Abstract
Cancer is the deadliest disease in the world behind heart disease. Sadly, this remains true even as we suffer the ravages of the Covid-19 pandemic. Whilst current chemo- and radiotherapeutic treatment strategies have significantly improved the patient survival rate, disease reoccurrence continues to pose a deadly risk for all too many patients. Incomplete removal of tumour cells from the body increases the chances of metastasis and developing resistance against current treatments. Immunotherapy represents a therapeutic modality that has helped to overcome these limitations in recent decades. However, further progress is needed. So-called immunogenic cell death (ICD) is a recently discovered and unique mode of cell death that could trigger this necessary further progress. ICD involves stimulation of a tumour-specific immune response as a downstream effect. Facilitated by certain treatment modalities, cells undergoing ICD can trigger the IFN-γ mediated immune response involving cytotoxic T cells (CTLs) and γδ T cells that eradicate residual tumour cells. In recent years, there has been a significant increase in the number of small-molecules being tested as potential ICD inducers. A large number of these ICD inducers are metal-based complexes. In fact, anticancer metal drugs based on Pt, Ru, Ir, Cu, and Au are now known to give rise to an immune response against tumour cells as the result of ICD. Advances have also been made in terms of exploiting combinatorial and delivery strategies. In favourable cases, these approaches have been shown to increase the efficacy of otherwise ICD “silent” metal complexes. Taken in concert, rationally designed novel anticancer metal complexes that can act as ICD inducers show promise as potential new immunotherapies for neoplastic disease. This Tutorial Review will allow the readers to assess the progress in this fast-evolving field thus setting the stage for future advances.
Graphical Abstract
1. Introduction
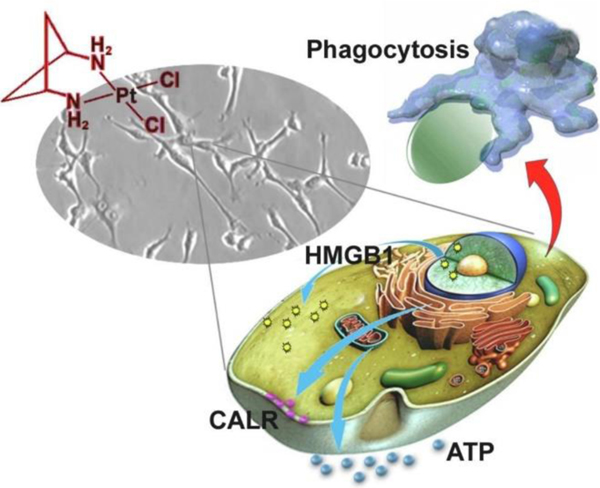
Every second, billions of cells in every healthy human undergo regulated cell death (RCD) in a “programmed” manner via the so-called apoptotic pathway. Such homeostatic cell death is usually considered to be “silent” to an immune response.1 This helps the body avoid activation of any unnecessary inflammatory signals and autoimmune reactions that might lead to catastrophic consequences. However, recent years have witnessed the discovery of another type of cell death that involves stimulation of immune response as its downstream affect. This immunostimulatory cell death modality, discovered and pioneered by the labs of Guido Kroemer and Laurence Zitvogel, is known as immunogenic cell death (ICD).2,3 Recently, the nomenclature committee on cell death (NCCD) has defined ICD in the following manner: “a form of RCD that is sufficient to activate an adaptive immune response in immunocompetent syngeneic hosts”.4 In the context of anticancer research, ICD can open a new avenue for treatment by exploiting immunomodulation to fight against tumour relapse and cancer metastasis. In principle, a therapeutic modality could militate the use of a patient’s dying cells as a vaccine that stimulates a cancer-specific immune response. To the extent this proves true, the patient would benefit from a rejuvenated immune system that would attack any residual cancer cells, resulting in inhibition of tumour growth or even complete eradication of the tumour.
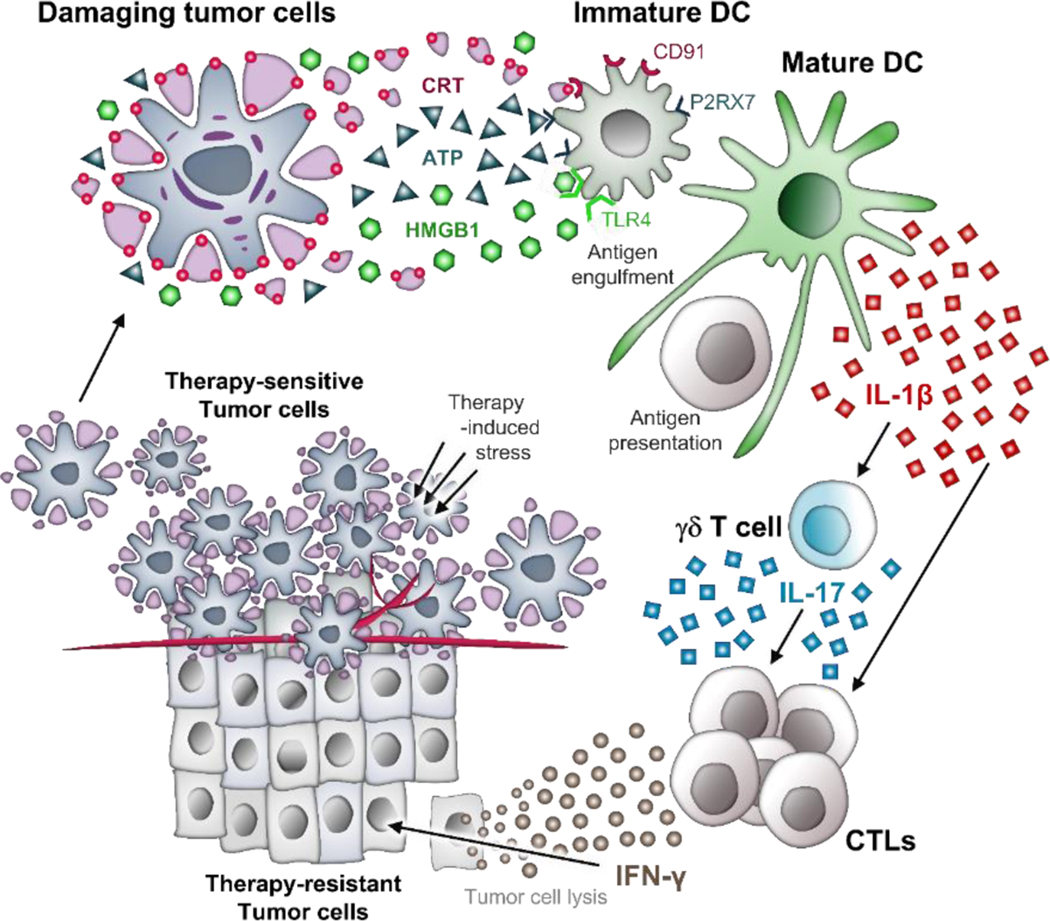
ICD can be characterized by its induction of three main damage-associated molecular patterns2,5,6 (DAMPs) which act as “biochemical hallmarks”: i) translocation of calreticulin (CRT) to the cell membrane of dying cells ii) active secretion of adenosine triphosphate (ATP) from dying cells, and iii) release of the high mobility group box 1 (HMGB1) protein by the damaging cells (Figure 1). Often, these biological events are associated with other molecular reactions, e.g., upregulation of endoplasmic reticulum (ER) chaperon proteins such as the heat-shock proteins 70 and 90 (HSP70 and HSP90) and/or enhanced expression of immunostimulatory cytokines, including interleukin 1β (IL-1β) and IL-17. The origin of these DAMP events and their effect on the immune response are discussed briefly below.
Figure 1:
Schematic representation of the immunogenic cell death mechanism.
1.a). Biochemical hallmarks of ICD
i). CRT translocation:
Calreticulin (CRT) is a Ca2+ binding chaperon protein present in the ER lumen. In healthy cells, besides acting as a Ca2+ buffer, CRT also plays a crucial role in the process of correct protein folding inside the ER.7 However, in the presence of ICD inducers, CRT translocation occurs from the ER lumen to the plasma membrane of damaging cells.8 A cascades of signalling events are then triggered by this CRT translocation. ER stress is believed to be a primary requirement for CRT exposure on the cell surface;8 however molecular mechanisms linking these two events have remained elusive.2 It is generally accepted that ER stress results in the phosphorylation of eukaryotic translation initiation factor eIF-2α by the protein kinase RNA like-endoplasmic reticulum kinase (PERK).9 This is followed by the caspase-8-mediated proteolysis of its target ER-sessile protein BAP31. Cleavage of BAP31 results in the activation of pro-apoptotic proteins BAX and BAK. This leads to perturbation of mitochondrial permeability and triggers release of cytochrome c into the cytosol. Altogether, these signalling events entail CRT translocation from the ER lumen to the plasma membrane via the Golgi apparatus. Two proteins are associated with this CRT transportation process: VAMP1 (vesicle associated membrane protein 1) and SNAP25 (synaptosomal-associated protein 25).10
CRT translocation dictates the immunogenicity of cancer cell death.11 However, CRT exposure by a chemotherapeutic can vary across the cell types. For example, cisplatin (CDDP) can exert CRT translocation in mouse ovarian cancer 2F8 cells, but not in mouse colorectal carcinoma CT26 cells.12 In the context of the immune response, CRT translocation acts as an “eat me” signal for dendritic cells (DCs).6 Following its release, CRT binds to its principal transmembrane receptor CD91 (also known as low density lipoprotein receptor-related protein LRP1) present on antigen-presenting cells (APCs). CRT-exposing cells are thus engulfed by CD91 positive APCs, (including dendritic cells, macrophages, and neutrophiles), a required event to initiate an immune response at a later stage (Figure 1).
ii). ATP release:
ATP represents the most abundant intracellular metabolite and is a small molecule that serves as the main source of energy for various cellular processes; however, it also plays a crucial role in cell signaling.2 In addition to the ATP release seen during neurotransmission, ATP release can also be triggered by physical or chemical-stress at the cellular level. While the precise mechanism whereby ICD inducers elicit ATP release is still unclear, pre-mortem autophagy has been established as a requirement for the release of substantial quantities of ATP from dying cells.13 The knockdown of either autophagy-related genes 5 or 7 (Atg5 and Atg7) has been reported as strongly inhibiting ICD-associated ATP secretion.14 ICD-mediated ATP release initiates through a complex mechanism involving the apparent relocalisation of vesicular ATP from lysosome to autolysosmes.10 Besides this, caspase 3 or caspase 8 mediated PANX1 (pannexin 1) activation, lysosomal-associated membrane protein 1 (LAMP 1)-dependent lysosomal exocytosis, and membrane blebbing also contribute to ATP release.10
Extracellular ATP release from dying cells acts as a “find me” signal for macrophages and dendritic cells where ATP behaves as the chemoattractant. Released ATP binds to its metabotropic receptor P2Y2 or ionotropic receptor P2RX7 on macrophages or dendritic cells (DCs) and induces their recruitment (Figure 1). In particular, binding the P2RX7 receptor results in K+ and Ca2+ efflux, which lead to activation of the NLRP3 inflammasome.15 Assembly of NLRP3 stimulates caspase-1 activation and subsequent secretion of the pro-inflammatory cytokines, interleukin 1β (IL-1β) and IL-18. This is crucial for immunogenic cell death because production of IL-1β initiates a cascade of immune response events that culminates in the intra-tumoural recruitment of γδ T cells and tumour antigen specific CD8+ αβ T lymphocytes.15
iii). HMGB1 release:
HMGB1 is a non-histone chromosomal binding protein that plays a multifunctional role, including regulation of DNA transcription, replication, and DNA repair in eukaryotic cells.16 In addition to its function in the nucleus, HMGB1 plays a role in cell motility, inflammatory machinery, and cancer cell invasion. In general, HMGB1 is secreted by DCs, natural killer cells (NKs), and monocytes as a response to cell injury or infection. At a late stage of ICD induction, nucleo-cytoplasmic HMGB1 is released from dying cells as a consequence of plasma membrane integrity loss.
Extracellular HMGB1 release from damaging cells acts as a “danger” signal in the ICD process. While the released HMGB1 can bind to many receptors, including the toll-like receptor 2 (TLR2) and the receptor for advanced glycation end products) (RAGE), in point of fact, it is the TLR4 that is thought to be the main receptor contributing to HMGB1-based effects on ICD. The binding of HMGB1 to TLR4 leads to activation of myeloid differentiation primary response gene 88 (MyD88).1 This activation is essential for the augmented expression of pro-IL 1β and optimal release of dead-cell antigens by DCs avoiding lysosomal degradation of the phagocytotic cargo. The importance of TLR4-HMGB1 engagement has been elucidated by the knockout of Tlr4 gene (but not Tlr2 or other Tlrs) which leads to deleterious immune responses in immunocompetent mice (Figure 1).17
CRT translocation, ATP release, and HMGB1 release fulfill the necessary requirements of ICD; however, they must occur in concert as part of a cumulative event, not separately.10 Together, these factors induce a cascade of processes that involve recruiting DCs to tumour cells, phagocytosis, antigen processing, DC maturation, and antigen presentation to cytotoxic T cells. This, in turn, results in an IFN-γ-mediated immune response involving γδ T cells and cytotoxic CD8+ T lymphocytes (CTL). These T cells then infiltrate the tumour microenvironment (TME) leading to inhibition of cancer cell growth and finally cancer cell death.6
In vitro assays to detect ICD-associated DAMPs can only identify anticancer agents to be explored as potential ICD inducers. The art-accepted gold standard for actually certifying a therapeutic modality as a bona fide ICD inducer requires vaccination assays with immunocompetent, syngeneic mice.4,10 Here, mouse cancer cells are incubated with a potential ICD inducer in vitro, followed by a thorough wash to remove any additional immunological adjuvant and exogenous chemical entities. Treated cancer cells are then resuspended in PBS, and inoculated subcutaneously (s.c.) into one flank of a sufficient number of immunocompetent syngeneic mice to provide an expected statistically meaningful result. Untreated cancer cells subjected to three freeze-thaw cycles to induce necrosis are typically inoculated into a separate cohort of mice as a negative control. One or two weeks later, the mice are challenged s.c. with living cancer cells of the same type as used originally, but into the opposite flank. Mice are then monitored over 40–60 days for the appearance or growth of tumours. Observing a significant number of mice that remain tumour-free at the end of the study or seeing a slowing in the growth of the challenge tumour relative to control are taken as an indication that the immunogenicity developed due to the putative ICD inducer is a real effect. To assess the durability of the triggered immune response, a second rechallenge model with the same type of cancer cells can be employed at a later date. Specificity of the immune response (if seen) can be tested by re-challenging any tumour-free mice obtained after the initial challenge with a different syngeneic cancer cell line. Other alternative approaches used to assess ICD in vivo include i) abscopal models (involving radiation therapy-induced antitumor immunity), ii) intracranial/extracranial models (useful for systemic chemotherapeutic agents that do not cross the blood-brain barrier). However, a detailed discussion of these latter models is beyond the scope of this review.
Depending on the treatment modality, ICD in cancer and infectious diseases can be subdivided in four groups: i) ICD caused by infectious pathogens, ii) ICD triggered by chemotherapeutics, iii) ICD elicited due to physical stress, iv) Necroptotic ICD.1 In this tutorial review we will mainly focus on ICD triggered by chemotherapeutics, in particular, metal-based complexes.
1.b). ICD inducers: introduction and classification
The initial years of anticancer drug discovery were largely focused on the development of cytotoxic compounds based on the notion that they would act as the equivalent of antibiotics vis à vis cancer cells. This hypothesis prompted academics and industrial drug developers alike to synthesize novel compounds with antiproliferative activity, irrespective of any effect on the host immune system.18 The famous review by Hanahan and Weinberg, published in 2000, defined “six hallmarks of cancers”; however, it did not mention any immunological effect on tumour cells.19 On the other hand, recent decades have borne witness to a paradigm shift in cancer care wherein the immune system plays a crucial role in controlling and eradicating tumours. It is now generally accepted that cancer cells can either hide from the host immune system (immunoevasion) or suppress the functionality of the host immune system to avoid immune-directed cell death (immunosubversion).20 This has also led to a reconsideration of the “hallmarks of cancer” where escape from immune surveillance is now included as a key attribute.21
Similar thinking has led to changes in how the development of novel anticancer drugs should be approached. The traditional understanding of chemotherapeutic treatment was that it would induce immunosuppression throughout the body.5 However, propelled by a knowledge of how the immune system can interact with anticancer drug-treated cells, it is becoming increasingly apparent that immune stimulation may be playing a major role in the efficacy of many frontline chemotherapeutic agents. In fact, recent studies have shown that certain anticancer drugs can stimulate tumour-specific immune responses and reinstate immunosurveillance.22 The underlying mechanisms include direct/indirect activation of immune effectors (NK cells, CTLs), cancer cell recognition (immune checkpoint inhibitors), and immune cell-mediated cell death. Stimulation of immunogenic cell death by anticancer agents is one specific example where chemotherapy induces rejuvenation of the immune system to recognize and eradicate cancer cells. Kroemer, et al. was the first to demonstrate that an ICD effect could be promoted by an anticancer molecule; these researchers showed that doxorubicin triggered the regression of established tumours in immunocompetent mice through the stimulation of ICD-mediated immune responses in vivo.23 Following this seminal paper, a number of chemotherapeutics were reported in the literature that promote ICD induction. A number of FDA-approved and experimental anticancer compounds are now known to elicit effects consistent with ICD in vitro; however, as noted above, validation of the effects in an in vivo vaccination model is considered necessary to claim a molecule as being a bona fide ICD inducer. Systems for which this has been done are still few in number.
ER stress and reactive oxygen species (ROS) generation have been confirmed as essential components that govern intracellular pathways of ICD.6 ICD has been found to be less effective in the presence of antioxidants, i.e., N-acetylcysteine.24 Conversely, cisplatin (CDDP) participates in redox metabolism but remained ineffective in inducing ICD, presumably due to its inability to trigger ER stress. It is known that CDDP can act as an ICD inducer in the presence of known ER stressors (i.e., tunicamycin and thapsigargin), findings that are discussed in detail in a later section.25 In 2013, Agostinis et al.26 first proposed a classification scheme for ICD inducers, dividing them into two classes, namely, Type I and Type II. The categorization is based on whether the ER is targeted directly or indirectly. Type I ICD do not target the ER as the primary site of action. Such agents trigger cell death through targets that are not directly linked mechanistically with the ER but induce an ICD-associated DAMP cascade through collateral ER stress effects. Thus, as a general rule, Type I inducers target different cellular compartments and include, for example, a) anthracyclines (e.g., doxorubicin, mitoxantrone) and oxaliplatin (OXP) that target DNA or DNA replication and repair proteins, b) shikonin and bortezomib that target cytosolic proteins, and c) cardiac glycosides, which target transmembrane proteins/channels. In contrast to Type I inducers, Type II ICD inducers involve the ER as their primary target organelle and typically promote both cancer cell death and ICD-mediated immunogenicity directly. For example, one of the few known Type II inducers, hypericin, predominantly localizes at the ER and upon irradiation trigger ROS-mediated ER stress therby triggering an ICD response. Presumably, an ICD-mediated immune response fostered by Type II inducers is more efficient. Such thinking is based on the simple fact that they typically elicit relatively higher levels of DAMPs and require simpler DAMP trafficking than Type I inducers.6,27
During the early years following the discovery of ICD, the majority of the systems were Type I inducers.28 However, recently an increasing number of small-molecule Type II ICD inducers have been reported. A large percentage of this latter set consists of metal-based chemotherapeutics. We review this progress in the ensuing subsections with a focus on compound class.
2. Metal based complexes as ICD inducers:
2.a). Platinum containing molecules triggering ICD:
The still-evolving story of platinum anticancer drugs begins with cisplatin, also known as Peyrone’s salt, which was first synthesized by Michele Peyrone back in 1844. However, it was only after more than 100 years that the antiproliferative activities of this salt were first reported by Barnett Rosenberg. Followed by the FDA approval of cisplatin (CDDP) as an anticancer drug in 1978, two other platinum(II) derivatives have received worldwide regulatory approval and many others have been studied as experimental drugs.29,30
2.a.i). FDA approved platinum drugs as ICD inducers:
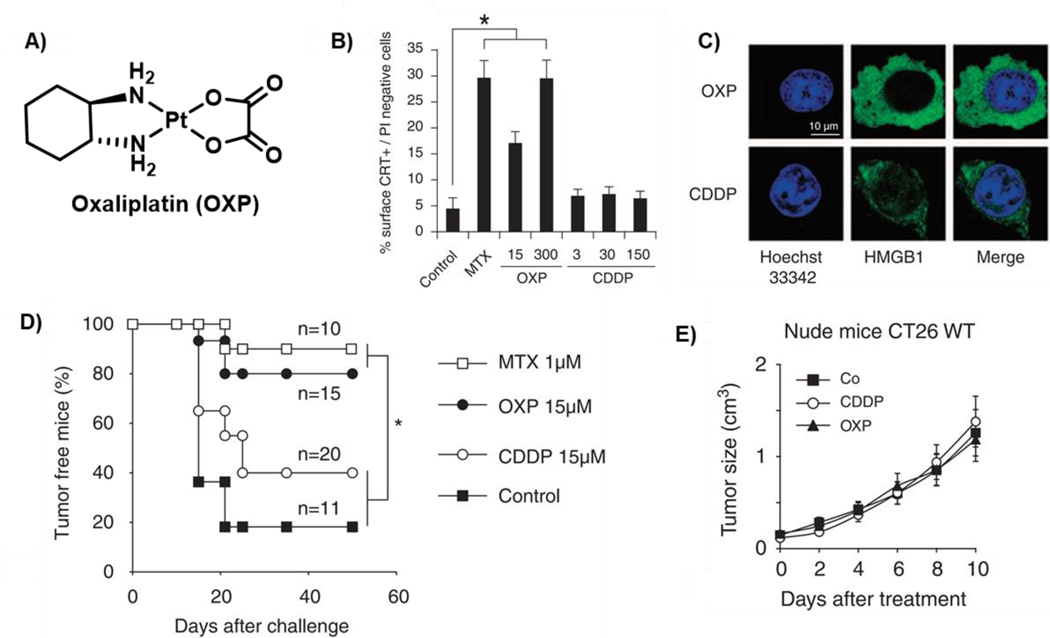
In spite of the effort devoted to exploring platinum drugs, their use as ICD inducers only dates back to 2010 when Kroemer et al.31 reported oxaliplatin (OXP) as the first metal-based chemotherapeutic that can elicit an ICD-mediated immune response in colon cancer cells (Figure 2a). Studies in the same paper served to highlight the contrasting behaviour of CDDP and OXP in terms of inducing ICD. As detailed by these researchers, treatment with OXP, but not CDDP, stimulated pre-apoptotic CRT exposure on the cell membrane in mouse colon cancer (CT26) cells (Figure 2b). However, HMGB1 release was observed in both OXP and CDDP treated cells (Figure 2c). Another study by Kroemer et al. revealed that CDDP is also unable to activate the PERK-dependent phosphorylation of eIF-2α, unlike OXP.25 These strikingly different phenotypic responses caused by CDDP, in comparison to OXP, resulted in its inability to trigger ICD. As a further validation of the ICD induction produced by OXP, in vivo studies involving a vaccination tumour model were performed. Here, immunocompetent BALB/c mice were injected (s.c.) with CT26 cells incubated with OXP (15 μM) for 4 h and then subsequently challenged with untreated CT26 cells on the opposite flank. OXP treatment was found to inhibit the tumour growth via an ICD mechanism (Figure 2d). Mitoxantrone (MTX) at 1 μM was used as a positive control in this experiment and found to inhibit distant tumour growth via an immunogenic effect. However, a similar outcome was not observed in the case of CDDP-treated mice even at higher doses. Interestingly, neither OXP nor CDDP was effective in retarding tumour growth against immunodeficient nu/nu mice (Figure 2e), thus indicating the underlying key role of immune system in ICD response. Additional studies involving knockout of Tlr4 (receptor for HMGB1) or RNA-mediated depletion of CRT levels in immunocompetent mice bearing CT26 xenografts led to a significant decrease in the anticancer immune response exhibited by OXP.31
Figure 2:
A) Chemical structure of FDA approved drug oxaliplatin (OXP) which can act as ICD inducer. B-C) CRT translocation (post 4 h incubation) and HMGB1 release (300 μM, post 24 h incubation) in CT26 cells upon treatment with OXP. D) In vivo demonstration of ICD-mediated challenge-tumour growth inhibition by OXP. E) Effect on tumour growth when OXP is administered to immunodeficient (nu/nu) mice. Reproduced with permission from ref. 31 (Oncogene, 2010, 29, 482–491). Copyright © Nature Publishing Group.
To test OXP as an ICD inducer in other tumour types, Li et al.32 studied its immunotherapeutic efficacy in Lewis lung carcinoma cells (LLC cells). Initial flow cytometry studies confirmed that OXP (20 μM) induced CRT exposure on the LLC cell surface 24 h post treatment. The release of ATP and HMGB1 was also observed 24 h after OXP treatment in LLC cells. Neither of these ICD hallmarks was observed upon treatment with equal concentrations of CDDP. To examine further the putative ICD-induction produced by OXP, in vivo vaccination studies were carried out. Here LLC cells treated with OXP (50 μM) were injected subcutaneously into C57BL/6J mice 7 days prior to challenge with fresh tumour cells. Taken together, these findings established that OXP was capable of promoting immunogenic lung cancer cell death in a LLC tumour model but that CDDP was unable to do so. It was found that OXP treatment increased the number of cytotoxic T cells (CD8+) and activated DC cells, both of which were believed to contribute to tumour regression during the in vivo vaccination studies. A follow-up paper revealed that co-administration of OXP with an α-PD-L1 immune checkpoint inhibitor gave rise to an even more pronounced immune-therapeutic effect, with more CD8+ T cells being elicited. The inferred synergy between OXP and the α-PD-L1 immune checkpoint inhibitor was ascribed to an increased expression of the immune checkpoint protein PD-L1 in the OXP treated animals, which served to reduce the initial cytotoxic therapeutic efficacy of OXP thus favouring ICD. This work highlights the promise associated with combining ICD with agents that promote an ICB (immune checkpoint blockade) effect.
Woodworth et al.33 reported that OXP can initiate ICD in glioma cells. These researchers found that OXP exposure caused phosphorylation of eIF-2α, which is a crucial key step in the early stages of ICD as noted above. Treatment of KR158-luc cells with 200 μM of OXP also led to an increase in CRT expression 24 h post incubation. On this basis, the authors concluded that OXP may act at critical junctures associated with glioblastoma pathobiology by triggering ER stress. ER stress leads to the disruption of STAT3 (a transcription factor that participates in cell signalling) activity, reduces expression of the DNA repair enzyme MGMT, and finally enhances immunogenicity through ICD. However, an in vivo demonstration of the presumed ICD effect was not included in this report.
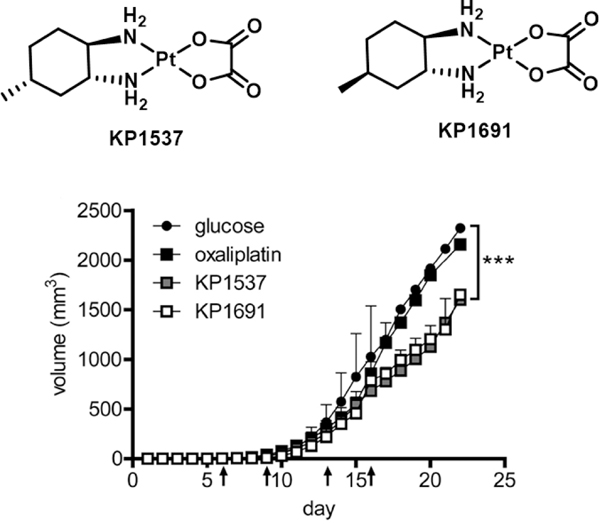
To investigate structure-activity relationships for OXP derivatives as putative ICD inducers, Berger et al.34 studied the effect of minor modifications in the drug motif. They found that structural modifications can have a profound impact on the immune response in different mouse models. In particular, these researchers identified two novel oxaliplatin derivatives, KP1537 and its stereoisomer KP1691 (Figure 3), which demonstrated significant tumour growth retardation in an immunodeficient SCID/BALB/c mouse model. In contrast, OXP treatment failed to achieve significant growth retardation, a finding in accord with the observations of Kroemer et al.31 The basis for this seemingly enigmatic difference between the effect of OXP and these derivatives is currently unknown.
Figure 3:
In vivo anticancer activity of OXP analogues KP1537 and KP1691 in immunodeficient SCID/BALB/c mice using a colon cancer xenograft model. Mice were treated for 2 weeks twice a week (days 6, 9, 13, and 16; arrows) with 9 mg/kg oxaliplatin, KP1537, and KP1691. Interestingly, OXP failed but OXP analogues were found to retard tumour growth significantly. Reprinted (adapted) with permission from ref. 34 (Mol. Pharmacol., 2012, 81, 719–728.). Copyright © 2009, Macmillan Publishers Limited.
In 2021, Brabec et al.35 carried out a structure-activity study involving derivatives of OXP. The main goal of this work was to compare and evaluate the plausible role of the cycloalkyl moiety in OXP derivatives in terms of inducing an ICD response. With this goal in mind, the authors screened a series of [Pt(cis-1,3-diaminocycloalkane)Cl2] and [Pt(cis-1,2-diaminocycloalkane)Cl2] complexes wherein the diaminocycloalkane group contains either cyclobutyl, cyclopentyl, or cyclohexyl moieties (denoted as DACB, DACP, and DACH respectively). The authors first confirmed that the cellular accumulation of platinum from these complexes was similar provided the cells were exposed to equitoxic concentrations (as determined via MTT assays). Thus, by using equitoxic concentrations of these platinum complexes, direct comparisons of their ICD inducing efficacy could be made in CT26 cells. It was found that DACB, the 1,3-diaminocyclobutyl derivative, was found to stimulate all 3 classic DAMP signals (CRT translocation, ATP release, and HMGB1 release) associated with ICD in CT-26 cells, and do so more effectively than OXP. This study also revealed that DACB was effective in promoting DC-mediated tumour cell phagocytosis, a key predictor of immunogenic response in vivo. On this basis, the authors concluded that DACB should be considered as a potential ICD inducer.
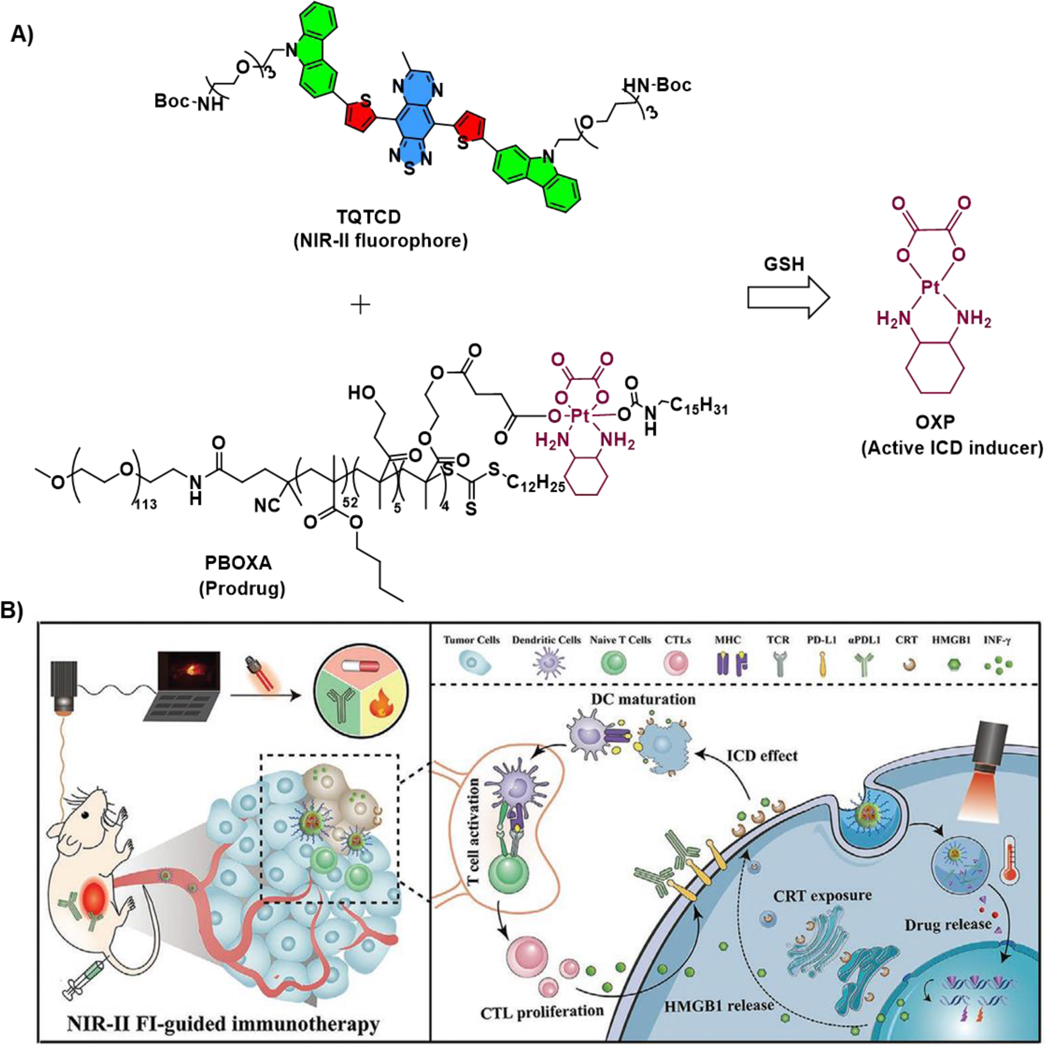
Yu et al. recently reported36 an OXP-based polymeric nanoparticle prodrug, compound 2.1 (denoted as PBOXA@TQTCD in the original work), which acts as an image-guided ICD inducer in a colorectal cancer model. This complex benefits from an NIR-II fluorophore, TQTCD, that displays a fluorescence emission maximum >1000 nm, and which facilitates a photothermal therapy (PTT) effect under 808 nm laser irradiation. This fluorophore also displays a large Stokes shift (>300 nm), which resulted into high signal-to-noise fluorescence imaging and makes it attractive for use in image-guided chemo-immunotherapeutic treatments. A copolymer named PBMA, comprised of butyl methacrylate (BMA) and hydroxyethyl methacrylate (HEMA), was used as the grafting material for an alkylated version of OXP. Glutathione (GSH) mediated reduction of these nanoparticles leads to OXP release, which in turn leads to ICD induction in CT26 cells. Immunogenicity was also elicited from the photothermal effect mediated by 2.1 under conditions of laser radiation. In vitro studies revealed that under laser irradiation compound 2.1 induces CRT expression on the cell membrane of CT26 cells, presumably reflecting the combined benefit of OXP-mediated chemotherapy and the PTT effect. Support for a strong cumulative effect on ICD induction came from the finding that the combined treatment led to higher HMGB1 efflux from cells compared to control PTT- or chemotherapy-alone cohorts. As an in vivo test of ICD induction, nanoparticles 2.1 were injected i.v. into CT26-bearing BALB/C mice when the tumour reached 50 mm3 in size. It was found that the group subject to laser irradiation after treatment with 2.1 displayed the greatest tumour growth inhibition. The role of ICD induction underlying the inhibitory effect was inferred from the higher levels of CRT expression on tumour cell membranes and CD8+ T cell infiltration as demonstrated by immunohistological studies. Finally, further the combination with an anti-PD-L1 antibody (αPDL1) led to an enhanced immune response. Taken in concert, this work establishes a novel approach to engineering ICD-inducing chemotherapeutics suitable for image-guided immunotherapeutic treatments.
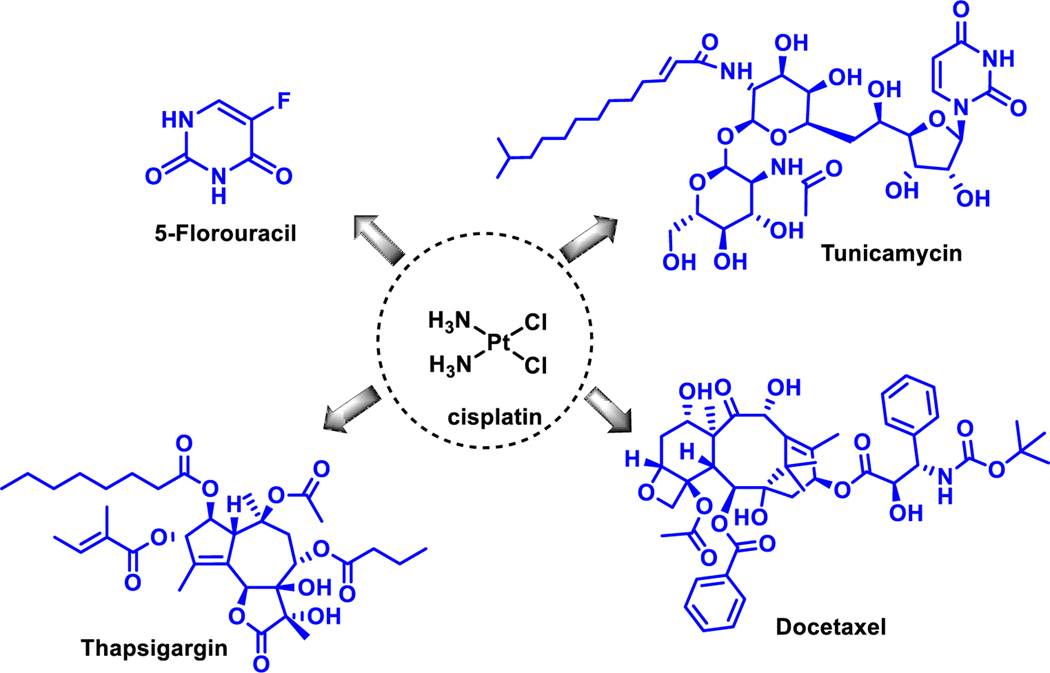
As noted above, CDDP fails to act as an ICD inducer in several tumour cell lines. This failure was primarily attributed to inefficient CRT translocation and an inability to promote ER stress in tumour cells. In an effort to elicit an immunotherapeutic effect using CDDP, Kroemer et al.25 co-administered CDDP with a known ER stressor thapsigargin (THAPS) (Figure 6); this combination was found to induce CRT exposure in several cell lines, including in CT26 and LLC cells. Further evidence of combination-induced immunogenicity came from in vivo studies carried out using immunocompetent C57BL/6 mice. Specifically, MCA205 cells treated overnight with CDDP (150 μM) or THAPS (5 μM), alone or in combination, were injected subcutaneously into the flank of C56BL/6 mice and subsequently rechallenged with untreated tumour cells injected on the opposite flank after 6 days. The CDDP-treated cells elicited no immune response. In contrast, the cells treated with both CDDP and THAPS elicited a strong immunogenic effect. In an effort to show the generality of this approach, a similar in vivo study was carried out using another ER stressor, tunicamycin (TUNICA) (Figure 6); again, immunogenicity was seen for CDDP when tested in conjunction with this stressor. Neither ER stressor (i.e., TUNICA or THAPS) alone was found to act as a CRT stimulating agent or to elicit an appreciable immune response. These studies underscore the major role of ER stress in ICD induction while highlighting a novel approach to triggering ICD by using a known cytotoxin (e.g., CDDP) in combination with an ER stressor.
Figure 6:
Representative drug molecules that act as adjuvants to convert the ICD-silent platinum drug cisplatin (CDDP) into an active ICD inducer.
In 2021, Ohira et al.37 reported that coadministration of 5-fluorouracil (5-FU)/CDDP can induce ICD in thoracic esophageal squamous cell carcinoma (ESCC) cells. This drug mixture constitutes a clinically approved neoadjuvant chemotherapy for stage II/III ESCC in Japan. However, the potential immunotherapeutic aspects of this therapy had not been previously explored. In a first set of in vitro studies, two ESCC cell lines were chosen, namely T.T and KYSE 30. Release of HMGB1 into the supernatant of both cell lines was seen post-treatment (48 h) with the co-administered drug mixture. However, CRT exposure and ATP release were not investigated in this study. The downstream effect of HMGB1 release was seen in terms of upregulation of DC maturation and enhanced expression of lysosome-associated membrane glycoprotein 3 (LAMP-3). Samples derived from human patients treated with this CDDP+5-FU cocktail (cf. Figure 6 for structure of 5-FU) also revealed a positive correlation between the number of LAMP-3 DCs and CD8+ T cells. Altogether, the authors concluded that this therapeutic modality can induce ICD in ESCC cells and that it thus may have a role to play in terms of modulating ESCC tumour progression in patients.
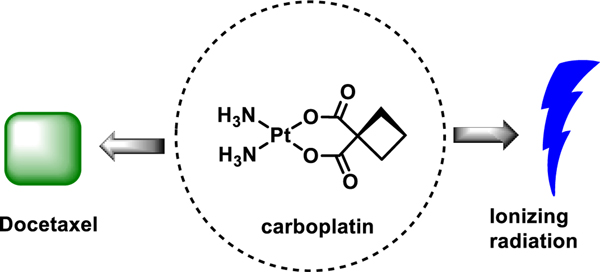
In a 2020 study, Jacobs et al.38 observed an ICD response when CDDP was used in combination with docetaxel (DOC) (cf. Figure 6 for structure) and tested against non-small cell lung cancer cells. The occurrence of DAMPs induced by the CDDP and DOC combination group was validated via in vitro assays. The combination group, consisting of CDDP and DOC, elicited higher levels of ICD markers and DC maturation, compared to the OXP control. (Note: to date DOC on its own has not been demonstrated as being particularly promising as an ICD inducer.) The same trend was seen when in vivo vaccination assays were carried out with immunocompetent C57BL/6J mice. In this study, mice were vaccinated twice with 3LL cells treated with the combined drug mixture or controls and subsequently challenged with fresh tumour cells. The authors observed that CDDP + DOC treatment inhibited tumour growth in five out of five mice, as compared to the OXP control wherein only 1 out of 5 mice remained tumour free in this particular model. Contrary to established thinking at the time, a vaccination with CDDP alone was found to exhibit an immunogenic effect in vivo in this tumour vaccination model (4 out of 5 mice remained tumour-free).38
In this same study,38 another frontline anticancer platinum drug, carboplatin, was also evaluated for its immunogenic effect. Unlike CDDP or OXP, carboplatin has not been extensively explored with regard its immunostimulatory potential.39 Carboplatin was found to express CRT translocation in a number of NSCLC cell lines (e.g., NCI-H1975, NCI-1650, and 3LL), but not in the commonly studied A549 human lung cancer cell line. It was found that ATP and HMGB1 release were not extensively triggered by carboplatin treatment. Nevertheless, carboplatin was able to inhibit tumour growth in an in vivo 3LL cell line vaccination model in 4 out of 5 mice. In contrast, OXP proved less efficient. Coadministration of DOC with carboplatin did not lead to an improvement in efficacy.
In 2014 Formenti et al.40 tested the use of ionizing radiation (IR) as a combination adjuvant to synergize the putative immunogenic effects of OXP and carboplatin. In this study, an in vitro assessment of ICD markers was performed wherein TSA breast cancer cells were subjected to OXP or carboplatin with a dose-dependent increase in IR (0–20 Gy). IR treatment (20 Gy) alone was found to enhance ATP release in TSA cells. The extent of ATP release was significantly increased in combination with OXP and IR, but a similar effect was not observed with carboplatin. Carboplatin was found to elicit CRT translocation alone, but a dose-dependent increase or synergy with IR was not observed. However, a fluorometric study monitoring HMGB1 release revealed a synergistic effect stemming from use of IR in combination with either OXP or carboplatin. The efficacy of the proposed immunogenic effect from carboplatin or carboplatin with IR was not, however, confirmed using an in vivo model.40 Thus, this work provides only a preliminary suggestion that clinically relevant doses of radiation alone, or in combination with chemotherapy might contribute towards triggering ICD.
The difference in the immunogenic potential of FDA-approved platinum drugs highlights the need for a thorough scrutiny of underlying mechanism of ICD induction and further studies of structure-activity relationships. In this context it is important to note that differences in dose (concentration), cell incubation time, and cell type can lead to apparent disparities in efficacy when conclusions are derived from comparisons across the literature, as opposed being the results of direct comparisons within the same set of in vitro or in vivo studies. Thus, caution is advised prior to reaching conclusions that are not experimentally derived under controlled conditions.39
2.a.ii). Novel Pt(II) complexes as ICD inducers:
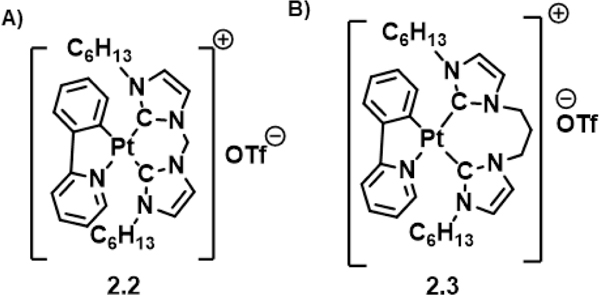
In 2015, Ang et al.28 reported a Pt(II) N-heterocyclic carbene complex, 2.2 (designated as Pt-NHC in the original report), which exhibited all three biological hallmarks of ICD in vitro in CT26 cells (Figure 8A). This was the first metal-chemotherapeutic to be reported, besides the FDA approved platinum drugs, which elicited an in vitro immunogenic response in cancer cells. This work revealed the engulfment of dying cancer cells via phagocytosis as a critical step for ICD. Interestingly, 2.2 was found to increase phagocytosis processes when CT26 cells were treated with 5 μM of 2.2, followed by co-incubation with J774 macrophages. In comparison, no other platinum drugs, including OXP at 50 μM, was found to be capable of promoting phagocytosis under similar conditions.
Figure 8:
A) Pt(II) NHC complex 2.2 which can act as an ICD inducer in vitro. B) Modified Pt(II) NHC complex 2.3 which exhibits superior ICD stimulation in CT26 cells.
Complex 2.2 was also reported to behave as a Type II ICD inducer,28 which stands in contrast to the platinum drugs, e.g., OXP, which act as Type I ICD inducers. Evidence that 2.2 acts as a Type II inducer came from the finding that in MDA-MB-231 cells ROS generation was occurring in the ER, as shown using confocal microscopy. Of note, the authors found that OXP did not promote formation of ER ROS. Flow cytometry assay provided further support for the fact that ROS are generated at a relatively low dose of 5 μM in the CT26 and MDA-MB cell lines. DAMP tests confirmed that a dose as low as 5 μM was able to elicit all three classic ICD biomarkers. This work provides support for the notion that targeting oxidative ER stress could provide a rational approach to novel Type II inducers. On the other hand, no in vivo assessment of the putative ICD induction by 2.2 was carried out by the authors. Paradoxically, OXP was reported to not induce ICD in this work based on two parameters, namely, an inability to promote phagocytosis and generte ROS within the ER. However, the authors did not report on biomarker assays associated with ICD (i.e., CRT translocation, ATP release, and HMGB1 release) using OXP. This work thus underscores the need to consider in detail the experimental parameters used to support a conclusion for or against ICD induction in vitro and the importance of in vivo model studies in the context of ICD assessment.
Structural modifications of compound 2.2 were reported recently by the same group.41 By screening a library of molecules with subtle structural differences, compound 2.3 (designated as Platin-ER in the original work) was identified; it was found to stimulate an immunogenic response superior to that induced by complex, 2.2 (Figure 8B). For instance, relative to 2.2 an increase in ICD DAMPs was seen for 2.3 in CT26 cells. Compound 2.3 was also reported to activate heat-shock protein 90 (HSP90), another “eat me” signal, which was not activated by compound 2.2. Moreover, phagocytosis induction, a key step in the ICD pathway, was observed in CT26 cells when treated with 2.3. A concentration dependent increase in ER-localized intracellular ROS levels, ascribed to compound 2.3, was noted. The ROS production levels were found to correlate with the magnitude of phagocytosis and CRT translocation. However, in the presence of ROS scavengers, such as Trolox® or Tiron®, phagocytosis was completely eliminated. As suggested by the authors, this result highlights the importance of ROS in immunogenic cell death mechanisms. In vitro studies also served to underscore the crucial role of ER stress and eIF-2α phosphorylation in ICD. The relatively higher cytotoxicity, as well as the enhanced release of DAMP signals in CT26 cells induced by 2.3 as compared to 2.2, makes it an attractive system for future development.
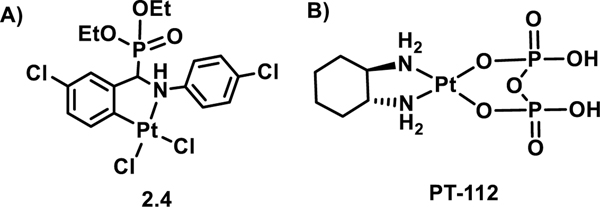
In 2019, Liang et al.42 reported a novel platinum(II) complex containing an aminophosphonate ligand, 2.4 (designated as Pt-1 in the original report), which can act as an ICD inducer (Figure 9A). An ICP-MS study with subcellular fractions from urinary bladder carcinoma (T-24) cells treated with compound 2.4 and harvested 24 h post incubation revealed the ER as the major subcellular organelle of localization for this complex. The accumulation of 2.4 in the ER was attributed to the presence of a phosphonate moiety in the ligan framework. Phosphonates are good ligands for Ca2+ ions, the levels of which are elevated in the ER. A time dependent increase in ROS along with ER stress generation in T-24 cells post-incubation with 2.4 prompted the authors to examine the putative ICD response from 2.4. They confirmed the activation of the three classic DAMP signals (CRT translocation, ATP release, and HMGB1 release) associated with ICD in T-24 cells upon treatment with 0.5 μM of compound 2.4 after incubation. Increased expression of the DC maturation markers CD83 and CD80 was also noted, along with an enhanced secretion of the proinflammatory cytokines IFN-γ and TNF-α, which are critical for eliciting an immune response. In vivo validation of the putative immunogenicity of compound 2.4 was carried out using a MB-49 mouse urothelial carcinoma vaccination model. Here, MB-49 cells treated with 2.4 (0.5 μM) or OXP (1.3 μM) for 48 h were injected (s.c.) into the left flanks of C57BL/6 mice and subjected to rechallenge after 7 days on the opposite flank. Cells treated with 2.4 were found to be slightly more effective in delaying tumour growth than OXP (30% vs 20% tumour free). In concert, these in vitro and in vivo observations led the authors to conclude that 2.4 can act as a novel scaffold for triggering ICD.
Figure 9:
A) Chemical structure of Pt(II) complex 2.4 that can stimulate ICD. B) Chemical structure of Pt(II) complex PT-112 that can act as ICD inducer and exhibit synergistic mechanism with immune checkpoint blockade therapy.
In 2020, Galluzzi et al.12 reported another platinum(II) complex comprising pyrophosphate ligands, PT-112, which behaves as a bona fide ICD inducer (Figure 9B). This complex was found to synergize with ICB therapy to induce a superior immunotherapeutic response. To characterize the release of DAMP signals, TSA cells were treated with PT-112 at a dose of 50 μg/ml for 24 h. It was found that PT-112 elicits all three ICD hallmarks (CRT, ATP, and HMGB1). Inspired by these positive results, the authors checked the efficacy of PT-112 in generating an ICD response in vivo. Here, immunocompetent BALB/c mice were injected with TSA cells treated with 150 μg/ml PT-112 and subsequently challenged after 7 days. Whilst TSA cells treated with the control mitoxantrone (a known ICD inducer) enabled only 40% tumour free survival 35 days post-challenge, PT-112 conferred 100% immunological protection. Furthermore, a second injection with fresh TSA cells 60 days post first challenge led to tumour growth in only 4 out of 9 mice, a finding consistent with the partial induction of a durable “immune memory effect”. PT-112 was also found to synergize with cytotoxic T-lymphocyte associated protein 4 (CTLA4) blockage therapy when tested in an abscopal model. Similar outcomes were also observed when PT-112 was co-administered with a programmed death-ligand 1 (PD-L1) blocker. The authors reported that PT-112 monotherapy was inefficient in extending the overall survival for BALB/c mice with CT26 tumours but was successful when used as a dual therapy (i.e., chemotherapy combined with ICB). The infiltration of cytotoxic T cells into tumour microenvironment induced by PT-112 was also found to be boosted by the use of PD-L1 blocker.12 On this basis, the authors concluded that PT-112 provides a novel approach to synergizing ICD and ICB and is a complex that shows promise for eradicating colorectal tumours with relatively high efficacy. In this context, PT-112 has also been assessed as monotherapy and in combination with the PD-L1 inhibitor avelumab in advanced metastatic castrate-resistant prostate cancer (mCRPC). In particular, mCRPC is considered to be immunologically “cold” and has a high rate of bone metastatic disease. A Phase 1 study revealed encouraging drug efficacy results for both therapeutic modalities tested (see clinical trial: NCT02266745 (monotherapy), NCT03409458 (with avelumab; ongoing)).43 PT-112 is now being evaluated in a Phase 2 study in patients with metastatic castration-resistant prostate cancer. The unique combination of ICD induction and osteotropism from PT-112 led to these positive results, as concluded by the researchers.
2.a. iii). Pt(IV) drugs as ICD inducers:
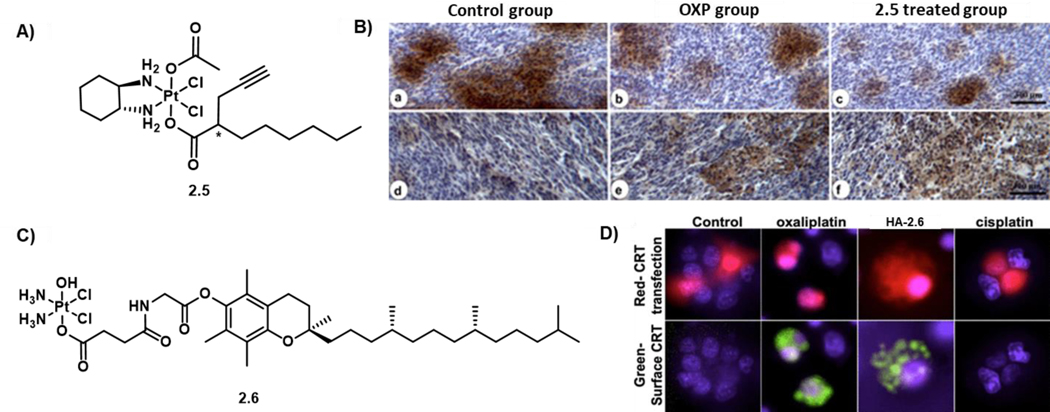
In an effort to create a next generation of platinum drugs capable of promoting immunogenic cell death, in 2019 Osella et al.44 reported a Pt(IV) prodrug, 2.5 (designated as compound 1 in the original report) (Figure 10A). Compound 2.5 was expected to be reduced to [PtCl2 (dach)] (dach = cyclohexane-1R,2R-diamine) in cellular environments while releasing the 2‑(2-propynyl)octanoate (POA) anion. The latter ligand can act as a histone deacetylase inhibitor, which is thought to also promote immunomodulatory effects in cancer cells. The authors did not report any in vitro studies evaluating DAMP signals in tumour cells treated with 2.5. However, a higher abundance of CD8+ T cells and CD68+ cells (macrophages) in tumour masses was seen in vivo studies carried out with BALB/c mice bearing CT26 colon cancer cells (Figure 10B). A greater effect compared to OXP as a control was also seen. This singular observation led them to conclude that 2.5 can potentially induce a stronger ICD response than OXP in colon cancer cells.44 As the authors concluded, Both metabolites, [PtCl2(dach)] and POA, likely contribute to this process.
Figure 10:
A) Axially derivatized Pt(IV) complex 2.5 which triggers tumour mass invasion by cytotoxic CD8+ T lymphocytes associated with ICD. B) Immunohistochemistry analysis showing a higher abundance of CD8+ T lymphocytes present in the tumour masses of mice treated with 2.5 (i.v. administration weekly, 13.6 mg kg−1). C) A carrier-based platinum(IV) agent 2.6 capable of eliciting immunogenic cell death. D) CRT translocation to the AT84 cell surface seen upon treatment with HA-2.6 (500 μM, 15h incubation). Reprinted (adapted) with permission from refs. 44 (J. Med. Chem., 2019, 62, 3395–3406) & 45 (Mol. Pharm., 2020, 17, 4334–4345), respectively. Copyright © 2019, 2020, American Chemical Society.
In 2020, Forrest et al.45 reported an axially derivatized Pt(IV)-tocopherol complex, 2.6, which was shown to act as a carrier-based ICD inducer (Figure 10C). A hyaluronic acid (HA)-tocopherol polymer-based delivery platform was exploited as a nanocarrier for the active Pt(IV) anticancer payload, giving rise to complex HA-2.6 (designated as HA-Pt4 in the original report). The authors hypothesized that the clinically relevant HA-based drug carrier approach would ensure efficient delivery of the drug into cancer cells, thus facilitating a superior ICD response. To probe the putative immunogenic properties of HA-2.6, the authors first investigated CRT translocation promoted by HA-2.6. Using confocal microscopy, 500 μM of compound HA-2.6 proved effective in promoting a CRT response 24 h post treatment in oral squamous cancer (AT84) cells that were transfected with a mouse CRT-HaloTag-KDEL fusion protein (Figure 10D). However, other DAMP signals including ATP release and HMGB1 release were not explored in this study. In vivo studies were carried out using a tumour efficacy model that involved both immunocompetent (C3H) and immunodeficient athymic nude mice. Better efficacy was seen in the C3H mice carrying the AT84 allograft as compared to the immunodeficient ones bearing MDA1986 xenografts upon subjecting both cohorts to 3 mg/kg intratumoural injections of HA-2.6. The improved efficacy seen in the immunocompetent group was attributed to an ICD effect induced by HA-2.6. Immunohistochemistry studies with tumour tissues confirmed a higher abundance of cytotoxic T cells than in the group treated with CDDP, a finding that correlates with the higher immune response produced by HA-2.6.45
2.a.iv). Photo-responsive platinum agents as ICD inducers:
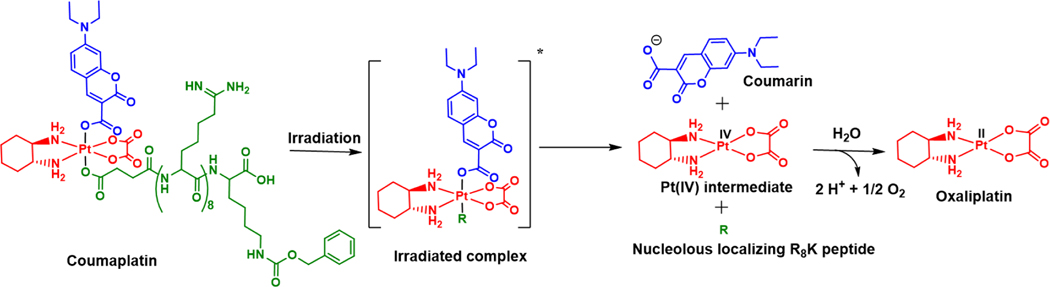
Stimuli-responsive anticancer drugs that can elicit an ICD response have not been extensively reported in the literature. This lack of precedence makes photoactivated platinum drugs particularly interesting. Rationally designed photocaged platinum complexes, which can be activated controllably “on site” with light, can help to avoid “off target” effects and elicit ICD mediated antitumour response. Appreciating this potential benefit, Zhu et al.46 reported a photocaged, nucleolus targeting platinum(IV) complex, coumaplatin (cf. for structure see Figure 11). Predicated on an OXP motif, coumaplatin features two axial ligands: coumarin as the photo-caging group and an R8K peptide for nucleolar localization. The authors demonstrated that coumaplatin provided for efficient nucleolus localization and displayed adequate photocytotoxicity. Coumaplatin was then tested in several cell lines. In the absence of light, coumaplatin (5 μM) did not trigger CRT translocation in cisplatin resistant lung cancer cells (A549cisR). However, when irradiated with blue light (450 nm, 8mW/cm2) for 1 h, cells treated with coumaplatin showed CRT translocation to the cell membrane. Photoactivated coumaplatin proved efficient in triggering other ICD biomarkers, namely ATP and HMGB1 release. Moreover, photoactivated coumaplatin-treated A549cisR cells were characterized by increased T cell proliferation, compared to OXP or cells treated with coumaplatin in the absence of photoactivation. The immune related positive outcomes from coumaplatin were attributed to the generation of OXP upon photoactivation. However, a stronger response was observed in terms of DAMP signals and greater T cell proliferation was seen for coumaplatin compared to OXP. On the other hand, an in vivo demonstration of the effectiveness was not included in the original paper46 or any follow-up study of which the authors of this review are aware.
Figure 11:
A photocaged, nucleolus-targeting Pt(IV) complex, coumaplatin, that can induce ICD. The proposed photoactivation mechanism for coumaplatin generates OXP, which contributes to triggering an immune response.
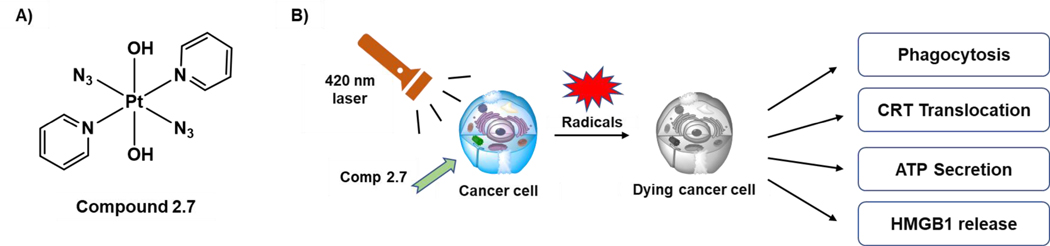
Separately, Brabec et al.47 reported a photoactivable platinum(IV) prodrug, 2.7 (designated as complex 1 in the original work) (Figure 12A). These authors reported that compound 2.7 remains unreactive in the dark but becomes potentially cytotoxic when photoactivated by UVA and visible light. It is thought that photoreduction of 2.7 occurs to generate a planar Pt(II) species and release azidyl and/or hydroxyl radicals. These photo-reduced species and daughter radicals serve to increase intracellular ROS levels, which may foster ICD in cancer cells. The ability of compound 2.7 to induce ICD DMAPs was tested. For the CRT translocation assay, human A2780 ovarian cancer cells were treated with increasing concentrations of 2.7 in the dark or when subject to photoirradiation (420 nm, 77 W m−2, 30 min). Under conditions of photoirradiation, but not in the dark, compound 2.7 was found to induce CRT exposure at a dose as low as 1 μM. Photoactivated 2.7 also proved efficient in inducing the release of HMGB1 and ATP and promoting tumour cell phagocytosis in a dose dependent manner in A2780 cells. However, no release was seen in the dark. On the basis of mechanistic studies, the authors concluded that autophagy is the main mode of cell death in A2780 cells treated with compound 2.7. This study thus provides support for the emerging impression that autophagy may be playing an essential role in promoting the immunogenic release of ATP from dying cells. Unfortunately, in vivo studies to support the putative benefit of this novel photoactivable approach to ICD induction were not included in the original report47 or, apparently, been the subject of follow-up efforts.
Figure 12:
A) Stimulation of ICD by a photoactivated Pt(IV) drug, compound 2.7. B) Schematic diagram of demonstrating biochemical hallmarks of ICD by compound 2.7 under photoirradiation.
2.b). Ruthenium containing molecules triggering ICD:
Ruthenium-based metal complexes have recently emerged as potential anticancer agents due to their unique mode of action.48 Currently, four ruthenium-based complexes are undergoing clinical trials as anticancer therapeutics. Nevertheless, until recently the potential of ruthenium compounds as ICD inducers had not been extensively explored.
2.b.i). Ruthenium(II) and ruthenium(III) complexes as ICD inducers:
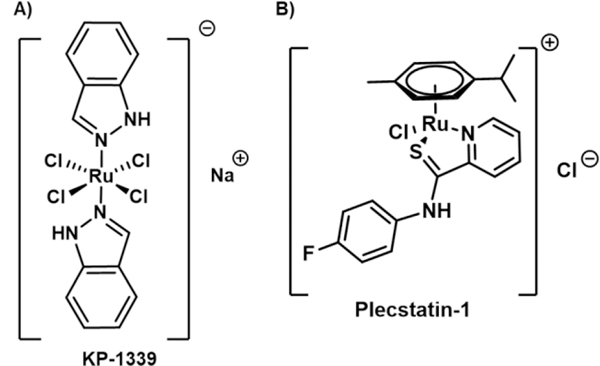
In 2019, Keppler et al.49 first showed that the ruthenium complex KP1339 (Figure 13A) can induce key markers associated with immunogenic cell death in vitro, using a spheroid model approach. The authors predicted KP1339 would stimulate an ICD response in cancer cells because of its ability to trigger ER stress. ER stress responses are presumably converted to ICD provoking biomarker release via activation of PERK pathway. To support their hypothesis, the authors showed PERK activation, followed by eIF-2α phosphorylation 24 h after incubating HCT116 human colon cancer cells with KP1339. These events are regarded as prerequisites for CRT translocation. CRT translocation was demonstrated using a HCT116 spheroid model. Flow cytometry and confocal microscopy studies revealed that treatment with 100 μM KP1339 leads to CRT exposure on the cell membrane of the spheroids. Using the same drug concentrations and incubation times, HMGB1 release was confirmed using an ELISA assay. Activation of autophagic cell death machinery and ATP release were also demonstrated in KP1339-treated cells. In an effort to classify this lead as a Type I or Type II ICD inducer, the production of ROS in cells following incubation with KP1339 was investigated using a fluorescent 2’,7’–dichlorofluorescein diacetate (DCFDA) assay; this served to confirm a dose dependent ROS level increase, leading to the inference that KP1339 is a Type II ICD inducer. However, it is to be noted that the in vivo efficacy of KP1339 would need to be established before it is appropriate to label it as a bona fide ICD inducer.49
Figure 13:
Ruthenium complexes A) KP-1339 and B) Plecstatin-1 which can trigger ICD as inferred from in vitro biomarker studies.
Keppler et al.50 also reported a Ru(II) arene complex, plecstatin-1, (Fig. 13B) which displayed ICD signature behaviour in colon cancer cells. The rationale behind choosing this molecule as a potential lead came from a prior study51 that showed treatment with plecstatin-1 in HCT116 colon carcinoma cells leads to phosphorylation of the eIF-2α protein. As noted above, eIF-2α phosphorylation is a key step for CRT translocation and hence ICD induction. To investigate in detail the ICD effect triggered by plecstatin-1, Keppler et al. used an in vitro spheroid model. Induction of oxidative stress in the form of a ca. 9-fold increase in ROS was observed in spheroids post-treatment with plecstatin-1 (200 μM). Presumably, this oxidative stress culminates in the phosphorylation of eIF-2α, which ultimately results in triggering ER stress and unfolded protein release (UPR). The downstream consequences of these presumed events were first evaluated by measuring the effect of plecstatin-1 on CRT exposure. Flow cytometry and confocal microscopy studies provided support for the contention that 200 μM of plecstatin-1 can induce CRT exposure on the cell membrane of HCT116 spheroids. This effect was less evident in HT29 colon cancer spheroids. HMGB1 and ATP release also followed the same trend where a stronger signal response was seen in the HCT116 spheroids as compared to the HT29 spheroids. Treatment with plecstatin-1 also led to an increased expression of the chaperon proteins HSP70 and HSP90, which play crucial roles in DC maturation and in enabling T cell representation. On the basis of these findings, it was concluded that plecstatin-1 can trigger ICD DAMP signals in vitro in colorectal cells.
2.b.ii). Photosensitizer-based ruthenium complexes as ICD inducers:
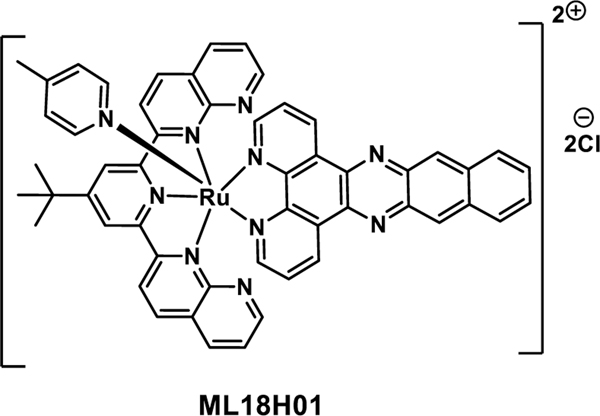
Many ruthenium-based metal complexes are highly efficient photosensitizers. They are well known for their “light in-ROS out” capabilities. Currently, a Ru(II) polypyridyl complex, TLD1433, is being evaluated in a phase II clinical trial against non-muscle invasive bladder cancer.52 However, the potential immunological ramification of Ru PDT-were not addressed until a 2020 report by Gujar et al.53 By means of screening studies, these researchers identified the lead complex ML18H01 (Figure 14) as being a particularly efficient singlet oxygen photosensitizer. They then evaluated the potential of this complex to trigger PDT-induced ICD in B16F10 melanoma cells. Melanoma cells have been reported to exhibit a positive outcome when treated with immunotherapeutic modalities, which led the authors to investigate a chemoimmunotherapeutic response with this drug. In fact, it was found that ML18H01-treated melanoma cells enhanced HSP90 and HSP1A gene expression and CRT translocation to the cell membrane. Increased levels of ATP release and HMGB1 were also reported upon ML18H01 treatment. Activation of inflammatory pathways, including upregulated expression of proinflammatory cytokines IL6, and CXCL10, were documented in this study. An in vivo demonstration of an ICD mediated immune response came from treating C57BL/6NCrl mice with ML18H01-treated melanoma cells and then challenging 7 days later with untreated B16F10 cells. It was found that this protocol conferred immunological protection as inferred from a delay in tumour growth or a reduction in tumour burden compared to the control group that was not treated with ML18H01. This study provides support for the notion that appropriately designed Ru PDT complexes could act as dual mode agents capable of eradicating tumours via PDT and protection against tumour relapse as the result of ICD.53
Figure 14:
Chemical structure of Ru(II) photodynamic therapy (PDT) complex ML18H01 which acts as an ICD inducer and a photosensitizer.
2.c). Other metal-based molecules triggering ICD
This subsection will summarize novel ICD inducers based on non-platinum or ruthenium metal complexes and their effectiveness in vitro or in vivo against different tumour models.
2.c.i). Iridium(III) complexes as ICD inducers:
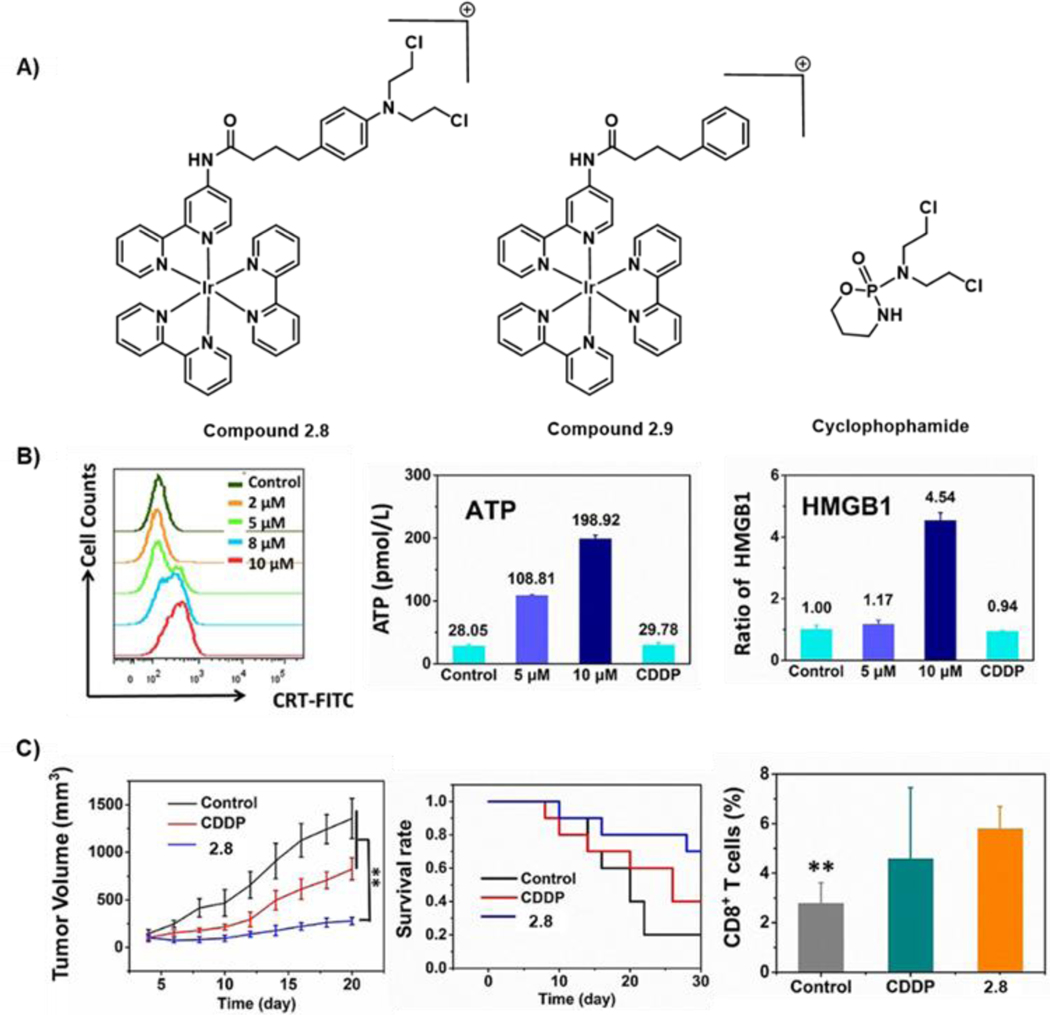
Iridium(III) complexes have been widely reported in the literature due to their strong anticancer activity. However, only very recently (2021), did Chao et al.54 report that an Ir(III) complex containing a bis(2-chloroethyl)-azane moiety, compound 2.8 (denoted as Ir1 in the original report, Figure 15A), acted as a potential ICD inducer. The pendant bis(2-chloroethyl)-azane moiety was chosen on the basis of its similarity with another ICD inducer, cyclophosphamide (cf. Figure 15A for structure). Compound 2.9 (denoted as Ir2 in the original report, cf. Figure 15A for structure), having the similar motif but lacking the bis(2-chloroethyl)-azane moiety, was tested a control molecule. The cancer being targeted in this study was non-small cell lung cancer, a choice rationalized by the fact that only a few complexes are known that elicit an ICD response in such tumours. Initial in vitro studies revealed that both compounds 2.8 and 2.9 not only localize selectively in ER of A549 cells, but also upregulate the ER stress-related C/EBP homologous protein (CHOP) and phosphorylated eIF-2α. Furthermore, a DCFDA assay revealed an increase in ROS levels in a dose dependent manner in A549 cells upon treatment with both compounds 2.8 and 2.9. These observations led the authors to investigate the DAMP cascade leading to ICD with both complexes. It was found that compound 2.8, but not compound 2.9, promoted autophagy-induced cell death. Likewise, CRT on the A549 cell surface was observed upon treatment with compound 2.8 (5 μM, 12 h), but not in the case of compound 2.9. The authors attributed this peculiar observation to very early-stage ROS generation in A549 cells (within 8 h) after treatment with compound 2.8. HMGB1 and ATP release were also detected in A549 cells treated with compound 2.8.
Figure 15:
A) Ir complex 2.8 that can act as an ICD inducer. A metal-free ICD inducer, cyclophosphamide, was conjugated to compound 2.9, an ICD-silent complex, to promote an immune response. B) ICD-mediated biomarker release by 2.8. C) In vivo results demonstrating tumour growth inhibition and showing the overall survival rate of mice inoculated with LLC cells. CD8+ T cells infiltration in the total cells isolated from C57BL/6 mice post treatment with PBS, CDDP (24 μM), or 2.8 (15 μM) are taken as evidence of an ICD-mediated immune response. Reprinted (adapted) with permission from ref. 54 (Angew. Chemie, 2021, 133, 4707–4715). Copyright © 2021, by Wiley-VCH GmbH.
Compound 2.8 was unable to induce surface exposure of CRT in breast cancer cells (MDA-MB-231), a finding supporting the notion that 2.8 is unable to stimulate ICD in this particular cell line. Based on the results from the in vitro studies, the authors concluded that only compound 2.8, but not its analogue 2.9, can induce ICD. Therefore, in vivo challenge studies were carried out using only compound 2.8. Here, C57BL/6J female mice were injected (s.c.) with LLC (murine lung cancer) cells treated with compound 2.8 and challenged after 7 days with fresh untreated tumour cells. Compound 2.8 was found to inhibit the growth of the challenge tumour as compared to the control cohort. Treatment with compound 2.8 led to increase the levels of CD8+ and CD4+ T cells. This work served to demonstrate that Ir(III) complexes, such as 2.8, may have a role to play as ICD inducers, but careful attention needs to be paid to the specifics of the structure in question, as evidenced by the fact that an ostensible analogue of 2.8 (complex 2.9) failed to promote ICD. On the other hand, compound 2.9 was able to create ER stress and promote ROS production, attributes common to Type II ICD inducers. However, complicating the analysis was the finding that compound 2.8 was inefficient as an ICD inducer in breast cancer cells (MDA-MB-231). Further studies are needed to understand these systems and to elucidate the specific mechanisms leading to ICD in cancer cells.
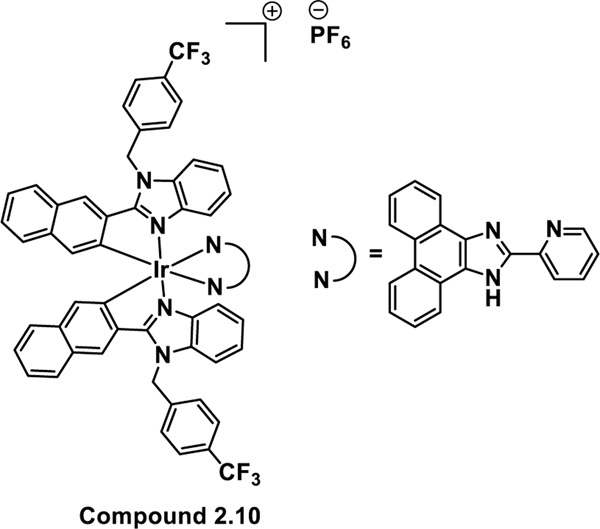
In 2021, Brabec et al.55 reported a photoactivated Ir(III) complex, compound 2.10 (Figure 16), that exhibits damage associated molecular patterns of ICD in melanoma cells. Compound 2.10 was shortlisted by these researchers as the preferred target compound from a series of Ir(III) complexes that differ in terms of subtle structural variations. The rationale behind this selection lies in the highest phototoxicity index, highest accumulation in cancer cells, and highest PDT-mediated ROS generation by compound 2.10 among the library of analogous complexes reported in this work. PDT agents have been previously documented to act as type II ICD inducers. Furthermore, compound 2.10 has been demonstrated to selectively localize in mitochondria and ER. Efforts were then made to prove the putative rationale that compound 2.10 can elicit ICD. First, dose dependent increase in calreticulin exposure was demonstrated when A375 human melanoma cells were treated with 2.10 and irradiated for 1 h (λmax = 420 nm, 77 ± 3 W m−2). Under analogous conditions, ATP release and HMGB1 secretion by the A375 cells were also detected. Interestingly, cells treated with 2.10 in the dark did not facilitate any significant level of biochemical hallmarks of ICD. Compound 2.10 was also found to provoke phagocytosis in CT26 cancer cells when irradiated and co-incubated with murine macrophages J774.A1. In contrast, cells treated with the drug in the dark do not promote phagocytosis. This work thus highlights the promise of using Ir(III) photosensitizers as potential ICD inducers in melanoma cells.
Figure 16:
Photoactivated Ir(III) complex 2.10 which induces biochemical hallmarks of ICD in human melanoma cells.
2.c.ii). Copper (I/II) complexes as ICD inducer:
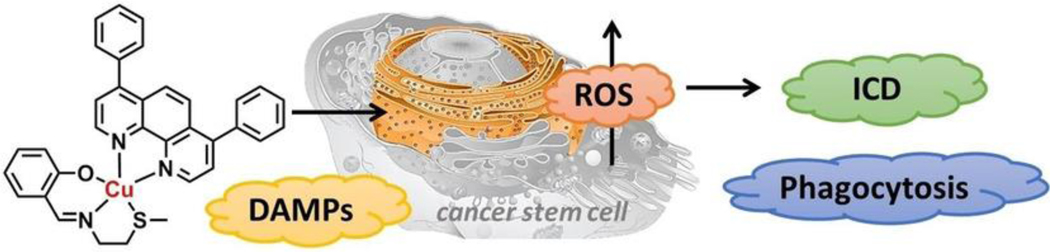
Many copper complexes act as redox-cycling agents that can increase efficiently the ROS levels inside cells. One consequence of this ROS generation is that certain copper complexes have been shown to be effective in inhibiting breast cancer stem cells (CSC). In light of these considerations, Suntharalingam et al.56 studied an ROS-generating copper complex that creates ER stress with the expectation that it could be used to facilitate an ICD response while treating CSCs.
After screening a library of copper complexes, these researchers identified the lead compound 2.11 (denoted as compound 4 in the original report, Figure 17) as being suitable system with which to probe the anticipated ICD effect. First, ROS generation and ER stress induction was confirmed in breast CSCs. ER stress was found to be a significant component of the cytotoxic mode of action of 2.11, along with ER stress related to the upregulation of the C/EBP homologous protein (CHOP) and phos-eIF-2α proteins. In vitro studies, involving treatment of CSC-enriched HMLER-shEcad breast cancer cells with 2.11, served to confirm CRT translocation to the cell membrane. ATP release was also confirmed in the same cell line. HMGB-1 release was indirectly established by the authors by measuring the total HMGB1 levels in HMLER-shEcad cells following treatment with 2.11 (0.3 and 0.6 μM for 48 h) and comparing them with the levels seen in untreated cells. The untreated cells showed higher levels of HMGB1 expression, a result consistent with intracellular HMGB-1 depletion being caused by compound 2.11. In aggregate, these results led the authors to conclude that compound 2.11 acts as Type II ICD inducer in vitro.56
Figure 17:
Copper based complex 2.11 which elicits ICD biomarkers in vitro within cancer stem cells. Reprinted (adapted) with permission from ref. 56 (ChemBioChem, 2020, 21, 3618–3624.). Copyright © 2020 by Wiley-VCH GmbH.
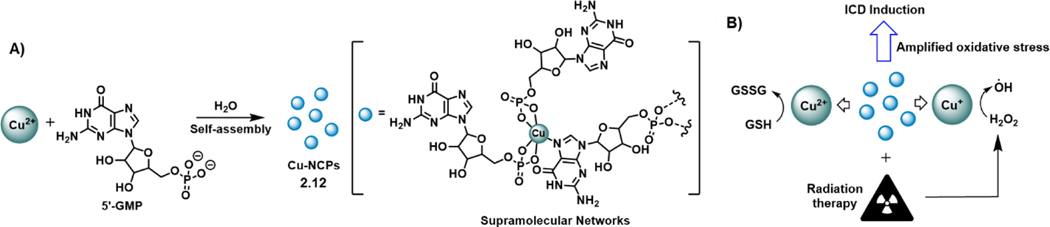
In 2021, Yuan et al.57 reported a copper-based nanoscale coordination polymer (Cu-NCP) 2.12 (Figure 18) that displayed synergy in terms of inducing ICD when used as a combination adjuvant with radiation therapy. Compound 2.12 was constructed via the self-assembly of CuSO4 and 5′-guanosine monophosphate in aqueous media at room temperature. High-resolution X-ray photoelectron spectroscopy (XPS) and electron spin resonance spectroscopy (ESR) studies revealed the presence of mixed valence copper ions (Cu(I) and Cu(II)) within the Cu-NCPs. The studies provided support for the design expectation that compound 2.12 would induce oxidative stress within the tumour cells via two different mechanism that operate contemporaneously: i) Cu(I) ion mediated ROS generation via Fenton-like reaction and ii) Cu(II) ion-facilitated inhibition of glutathione (GSH). The authors postulated that this dual mode of ROS generation would augment ICD induction when combined with a known ICD inducer, radiation therapy. It is noteworthy that radiation therapy (RT) can only trigger low levels of ICD response by itself, owing to inadequate ROS generation or unfavourable antioxidant mechanism inside cells. In support of their hypothesis, the authors first demonstrated an increase in CRT release in CT26 cells in the presence of Cu-NCPs and RT (6 Gy), compared with RT or the Cu-NCPs alone. A similar amplified effect was observed in terms of HMGB1 and ATP release in CT26 cells when treated with Cu-NCPs and RT as a combination cocktail. Further analysis using Western blots demonstrated upregulation of the CHOP protein, indicating the involvement of ER stress induction in triggering ICD. The in vivo effect from these DAMP events was reflected in an increased proportion of mature CD11c+ dendritic cells (DCs) in tumour-draining lymph nodes (TDLNs). Further evidence of the proposed immunological action of the Cu-NCPs in conjunction with RT was revealed in a CT26 bilateral abscopal tumour model. In this model, RT alone did not inhibit distant tumour growth, but the concurrent administration of RT and Cu-NCPs proved effective in reducing distant tumour growth. Enhanced intratumoural CD8+ and CD4+ T-cell infiltration in both primary and distant tumours also confirmed the promotion of systemic antitumour immunity by combined Cu-NCPs + RT treatment. Of particular note is that further combination with PDL1 blockade therapy amplified the immune response. Taken in concert, this work demonstrates the effect of Cu-NCPs in potentiating ICD induction and illustrates a novel way of augmenting the immunogenic profile of radiation therapy as an ICD inducer.
Figure 18:
A) Copper-based nanoscale coordination polymer (Cu-NCP) 2.12 which augments ICD when treated with radiation therapy (RT). B) Schematic diagram of Cu-NCPs producing •OH and eliminating GSH that potentiate immune response when treated with radiation therapy.
2.c.iii). Gold (I) complexes as ICD inducer:
Gold complexes are attracting increasing attention as potential anticancer agents. The cytotoxic effects of these drugs have been widely studied, leading to the consensus view that their main mode of action involves inhibition of thioredoxin reductase (TrxR), a key enzyme that serves to mitigate the effects of oxidative stress. However, their role in stimulating anticancer immunity and in particular, ICD, was not well investigated until recently.
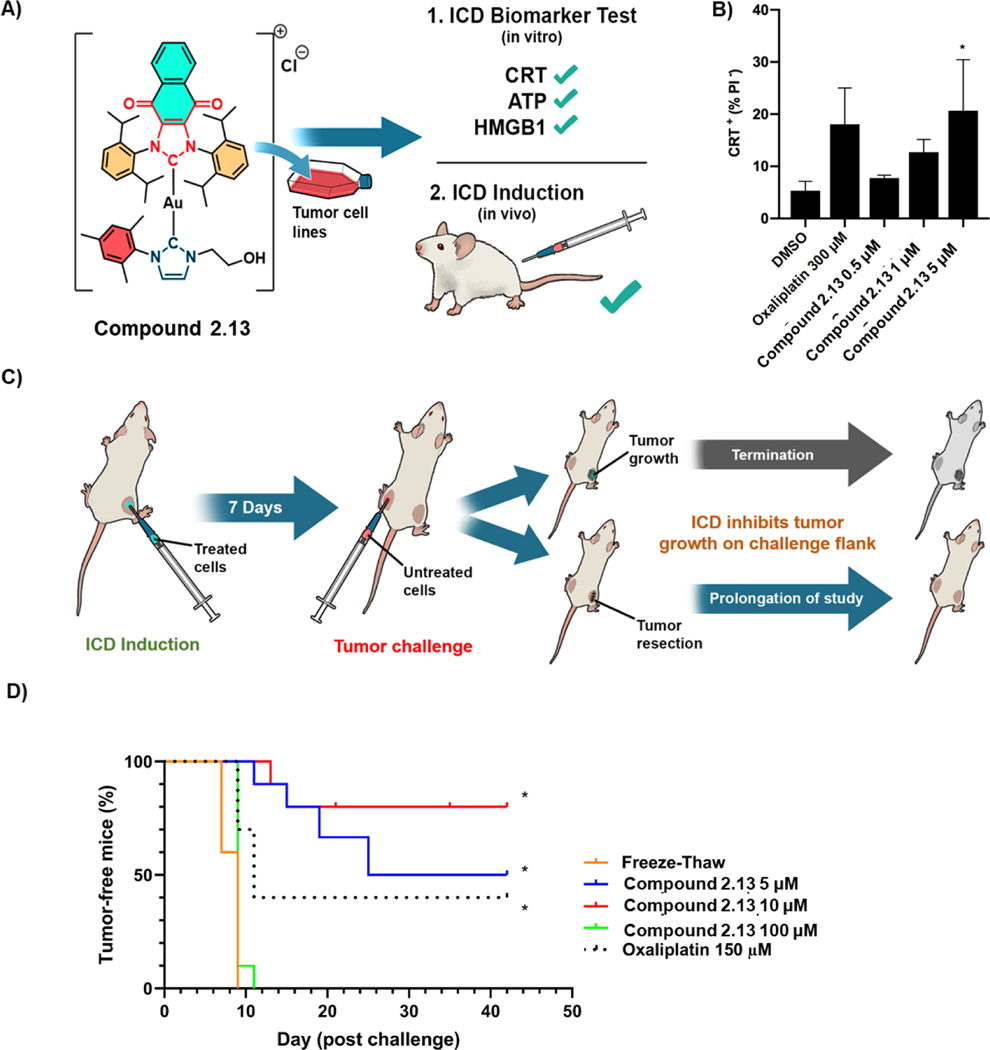
In 2021, Arambula et al.58 reported a rationally designed redox-active Au(I) bis N-heterocyclic carbene (NHC) compound 2.13 (Figure 19A) that can induce ICD both in vitro and in vivo. The authors postulated that dual targeting of the cancer antioxidant network and promotion of ER stress could provide a rational approach to Type II ICD induction. Redox-active Au(I) bis-NHCs have been associated with the upregulation of genes responsible for ER stress. Furthermore, naphthoquinone-appended Au(I) bis-NHCs can exhibit dual mode of action against the cancer antioxidant network as the result of combining TrxR2 inhibition (leading to a reduction in biological antioxidants) with increased oxidative stress (via redox cycling). In congruence with the synthetic design, compound 2.13 was first studied in the CT26 cell line to confirm that it can promote CRT translocation. Here, flow cytometry results revealed that 2.5 μM compound 2.13 can trigger cell membrane CRT translocation at a level similar level to that seen in cells treated with 300 μM of OXP post 24 h treatment (Figure 19B). Compound 2.13 also proved to be more effective than OXP in releasing ATP and HMGB1 in CT26 cells. Compound 2.13 was next assessed in vivo to determine whether it would engender an ICD effect as reflected in tumour growth inhibition in a challenge-type testing scenario (Figure 19C). With this objective in mind, a group of BALB/c mice were subcutaneously injected with CT26 cells pretreated with compound 2.13 at varying concentrations (5, 10, or 100 μM) of 2.13, as well as 150 μM of OXP for 4 h. Seven days later, the mice were challenged with naïve, live CT26 cells on the opposite flank. It was found that whilst mice cohorts treated with compound 2.13 (5 and 10 μM) or OXP experienced delayed or no tumour development on the challenge flank, 100 μM of compound 2.13 failed to do so (Figure 19D). These results provided support for the conclusion that compound 2.13 can act as an ICD inducer in vivo. However, it was also concluded that administrated concentrations should be carefully monitored since the inherent cytotoxicity of the compound can lead to direct tumour eradication, masking an in vivo ICD effect. This first study was not extended past the second week because tumour growth on the initial treatment flank and resulted in the premature end of the study. Therefore, in a follow up experiment, tumours growing on the treatment flank were resected when they reached a size of >200 mm3. The use of this revised protocol allowed the study time to be strategy helped to elongated and the tumour growth inhibition effect from ICD on the challenge flank to be more easily monitored. The data revealed that even after 42 days, compound 2.13 at 10 μM is more effective at delaying challenge tumour growth than OXP used at 150 μM (Figure 19D). Taken in concert, these findings justify the inclusion of these types of gold complexes in the lexicon of Type II ICD inducers list while highlighting the potential benefits of triggering ICD by perturbing intracellular redox homeostasis.
Figure 19:
A) Au(I) complex 2.13 that promotes immunogenic cell death in a CT26 colon cancer model. B) In vitro CRT translocation study in CT26 cells treated with compound 2.13. C) Illustration of in vivo experiments carried out with 2.13. D) Tumour growth inhibition at the challenge tumour site in mice treated with 2.13 at different doses or OXP. Reprinted (adapted) with permission from ref. 58 (J. Am. Chem. Soc., 2020, 142, 20536–20541). Copyright © 2020, American Chemical Society.
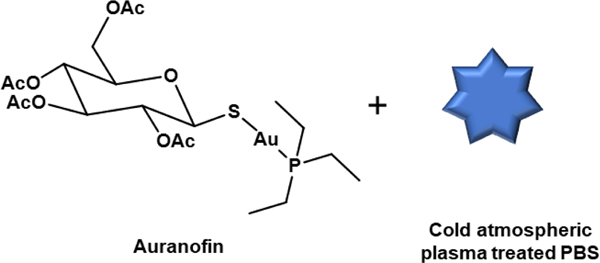
In 2021, Deben et al.59 reported induction of ICD in glioblastoma cells via perturbation of ROS homeostasis through sequential combinatorial treatment of auranofin (AF, Figure 20) and cold atmospheric plasma treated phosphate buffered saline (pPBS). Cold atmospheric plasma treated PBS was used as a unique treatment method for increasing oxidative stress levels in cancer cells via the exogenous delivery of ROS and reactive nitrogen species (RNS). The endogenous ROS level accentuation was achieved by the inhibition of TrxR1 with AF. The synergistic effect of oxidative stress from the combination of AF and pPBS resulted in ICD induction in U-87 cells (human primary glioblastoma cell) and LN-229 cells (human glioblastoma cells). Surface translocation of CRT, followed by extracellular ATP and HMGB1 release, were significantly elevated only in combination cohort of AF and pPBS treated cell lines compared to the untreated one. Next, the studies revealed a significant increase in the mature CD86+/MHCII+ DC population after coculture with treated T98G cell lines (human glioblastoma multiforme tumour cells). Maturation of DC can initiate an effective antitumour response, as discussed in earlier sections. However, a mild increase or no increase in DC maturation was observed in the LN-229 cells or U-87 cells, respectively, even in the presence of combined treatment. Additional experiments suggested that AF can significantly inhibit the phagocytosis of immature DCs as monotherapy or in combination with pPBS. The inhibition of phagocytotic activity of DCs by AF can hamper the immunostimulatory effects of ICD induction from AF and pPBS cotreatment. Therefore, as concluded by the authors, use of AF as an adjuvant to stimulate antitumour immune response should be carefully investigated in future studies.
Figure 20:
Au(I) complex auranofin (AF) that can promote immunogenic cell death in glioblastoma cells in conjugation with cold atmospheric plasma treated PBS (pPBS).
3. Conclusions and outlook:
Anti-cancer immunotherapy using immune checkpoint inhibitors has brought a paradigm shift in cancer treatment. This revolution in care is providing an incentive to develop small molecules that promote an immune response. In this context, metal-based ICD inducers may have an important role to play and could propel further the use of metal-drugs in treating cancerous disease. Although progress along the latter lines is being made, it remains a sad truism that only a limited number of platinum(II) complexes have been approved as cancer chemotherapeutics. Gratifyingly, in various guises, these agents are showing promise in the ICD-based treatment of colorectal cancer, gastric cancer, and pancreatic cancer.60 Of note is that the immunogenic profile of oxaliplatin can be further augmented by using it in conjunction with immunotherapeutic protocols currently employed in the clinic. One of the major limitations accounting for the paucity of ICD inducers reported in the literature is the requirement of using immunocompetent mice in in vivo model studies. This stands in contrast with the traditional anticancer drug screening approaches that generally rely on human tumor xenograft models and immunocompromised mice. Therefore, the gold-standard method (i.e., vaccination model) required to make a robust claim of ICD induction constitutes a requirement that is beyond the current testing capacity of many laboratories. Moreover, the need for a fully syngeneic setting in assessing immunogenicity also restricts the library of readily available cell lines and de facto has limited the number of established rodent models to C57BL/6 or BALB/c mice.4,61
Moving forward, determining the correct dose regimen and administration schedule to achieve a maximal immunological benefit will be a major challenge for clinical studies with this and other metal-based ICD-inducing agents. This is highly significant, as in certain cases, chemotherapeutics designed for inducing ICD negatively impact the antitumour immunity when administered through multiple treatment cycles. Therefore, strategies for effective immunogenic chemotherapy that employ metronomic schedules that provide for maximal patient benefit and tolerability need to be developed.62 Compounding this challenge is the fact that a direct correlation between MTD (maximum tolerable dose) and the ICD effective dose for a certain chemotherapeutics cannot yet be predicted. Recent studies have led to the conclusion that dose concentrations can also dictate the extent of immunogenicity seen in cancer cells treated with certain chemotherapeutics, e.g., CDDP.63 Thus, further pre-clinical studies are warranted to determine the dosage parameters that trigger anticancer immunity without appreciable deleterious side-effect. Little is known so far about how clinically approved anticancer agents interact with the host immune system. A few FDA approved anticancer drugs that act as bona fide ICD inducers have been developed preclinically using immunodeficient models of disease. However, human cancer cells cannot be directly assessed for stimulating an immune response in a murine microenvironment due to the inherent immunological incompatibilities across species.4 To this end, humanized mice models that utilize immunodeficient mice reconstituted with a human immune system are attractive. However, further effort dedicated to generating additional rodent models for the study of putative ICD inducers is warranted. Beyond the initial research stages, several well-designed longitudinal immuno-monitoring protocols are currently being implemented in several clinical trials involving oncological patients treated with FDA-approved chemotherapeutics that show promise as ICD inducers.60 The results from these trials will likely play a pivotal role in assessing the actual diagnostic value of preclinical ICD studies as prognosticators of clinical success. As such, they may or may not provide a added incentive for moving ew putative ICD agents into early stage clinical trials.
Subject to the above caveats r.e. clinical translation, in our view there remains a cogent need to discover new ICD inducers. In the context of metal based systems one can hypothesize that the development of drug conjugates that combine two classes of ICD inducers in the form of a hetero-bimetallic complex might lead to a superior anticancer immune response. The rationale behind this putative strategy lies in the pre-clinical success encountered in combining metal-based ICD inducers with other immunogenic treatment therapies. Separately, the library of metal-based complexes tested for ICD induction should be expanded with the caveat that new candidates are evaluated for in vivo efficacy. In this context, we suggest that further studies of gold, iron, arsenic, osmium, and rhenium complexes are warranted since complexes of these elements have already been shown to upregulate ER stress within cancer cells, a key progenitor of ICD.64 As an adjunct strategy, perturbing mitochondrial calcium homeostasis can lead to ICD stimulation.65 Therefore, progress with rationally designed novel calcium transporters is likely to make a significant contribution to this effort. On a similar note, intervening iron homeostasis pathway can also facilitate or augment immunogenic cell death.66 Recent studies with a rhenium complex conjugated to an iron chelator deferasirox67 showed upregulation of CRT translocation in MDA-MB-231 triple negative breast cancer cells. This is indicative of ICD induction in vitro; however, further experiments with similar complexes need to be carried out to confirm the rationale. Creating new systems that specifically target tumour environments is also important. Nanoparticle-based strategies for stimulating and/or potentiating type II ICD is particularly a burgeoning field of research.68–72
In conclusion, metal compounds inducing immunogenic cell death is a rapidly blooming field of anticancer research which has already demonstrated huge promises. To achieve its full potential, more proactive involvement of the pharmaceutical companies in translating these exciting metallodrugs to the next step of drug development for clinical studies is needed. It is hoped that the current summary of the state-of-the-art for metal-based ICD inducers will set the stage for future advances in this field, ushering in the next generation of immunotherapeutic drugs.
Figure 4:
Schematic showing how the OXP derivative DACB triggers ICD-associated DAMPs and induces phagocytosis in CT26 cells. Reprinted with permission from ref. 35 (J. Inorg. Biochem., 2022, 226, 111628.). Copyright © 2021, Elsevier Inc.
Figure 5:
A) Preparation of OXP-based nanoparticles 2.1 that integrate the polymeric prodrug PBOXA and a D–S–A–S–D-type TQTCD fluorophore. Compound 2.1 can facilitate image-guided ICD induction via release of OXP combined with a photothermal effect. B) Schematic diagram of image guided immunotherapy with nanoparticle 2.1. Reprinted (adapted) with permission from ref. 36 (Small, 2021, 17, 1–11). Copyright © 2021, Wiley-VCH GmbH.
Figure 7:
Treatment modalities used with FDA approved platinum drug carboplatin to induce ICD.
Key Learning points:
The significance of immunological cell death and key biological events associated with it.
Use of ICD-inducers as a cancer immunotherapeutic modality, either alone or in conjunction with immune checkpoint inhibitors.
Underlying principles for the design of metal-based ICD inducers.
Reinstating an immune response from ICD “silent” metal complexes and potentiating ICD mediated immune response.
Pre-clinical and clinical progress of ICD-inducers and future perspectives.
Acknowledgements.
The work in Austin was supported initially by NIH grant R15 CA232765 to J.F.A. Subsequent support was provided by the Robert A. Welch Foundation (F-0018 to J.L.S.). The work in Seoul was supported by the National Research Foundation of Korea (CRI Project Nos. 2018R1A3B1052702 and 2019M3E5D1A01068998, J.S.K.).
Abbreviations:
- 5-FU
5-Fluorouracil
- AF
Auranofin
- APCs
Antigen presenting cells
- ATP
Adenosine triphosphate
- BAK
Bcl-2 homologous antagonist/killer
- BAX
Bcl-2-associated X
- CDDP
Cis-diammine-dichloroplatinum(II)/Cisplatin
- CRT
Calreticulin
- CSC
Cancer stem cells
- CTLA4
Cytotoxic T-lymphocyte associated protein 4
- CTLs
Cytotoxic T-lymphocytes
- DAMPs
Damage associated molecular patterns
- DC
Dendritic cells
- DCFDA
Dichlorofluorescein diacetate
- DOC
Docetaxel
- eIF-2α
Eukaryotic initiation factor-2α
- ER
Endoplasmic reticulum
- HMGB1
High mobility group box protein 1
- HSP
Heat shock protein
- i.v.
Intravenous
- ICB
Immune checkpoint blockade
- ICD
Immunogenic cell death
- IFN-γ
Interferon- γ
- IL-1β
Interleukin-1β
- IR
Ionizing radiation
- MTD
Maximum tolerable dose
- NCP
Nanoscale coordination polymer
- NHC
N-heterocyclic carbene
- OXP
Oxaliplatin
- PD-L1
Programmed death-ligand 1
- PDT
Photodynamic therapy
- PERK
Protein kinase RNA like-endoplasmic reticulum kinase
- pPBS
Cold atmospheric plasma treated PBS
- RCD
Regulated cell death
- ROS
Reactive oxygen species
- RT
Radiation therapy
- s.c.
Subcutaneous
- THAPS
Thapsigargin
- TrxR
Thioredoxin reductase
- TUNICA
Tunicamycin
- UPR
Unfolded protein release
- VAMP1
Vesicle Associated Membrane Protein 1
4. References:
- 1.Galluzzi L, Buqué A, Kepp O, Zitvogel L and Kroemer G, Nat. Rev. Immunol, 2017, 17, 97–111. [DOI] [PubMed] [Google Scholar]
- 2.Kroemer G, Galluzzi L, Kepp O and Zitvogel L, Annu. Rev. Immunol, 2013, 31, 51–72. [DOI] [PubMed] [Google Scholar]
- 3.Garg AD, Dudek-Peric AM, Romano E and Agostinis P, Int. J. Dev. Biol, 2015, 59, 131–140. [DOI] [PubMed] [Google Scholar]
- 4.Galluzzi L, Vitale I, Warren S, Adjemian S, Agostinis P, Martinez AB, Chan TA, Coukos G, Demaria S, Deutsch E, Draganov D, Edelson RL, Formenti SC, Fucikova J, Gabriele L, Gaipl US, Gameiro SR, Garg AD, Golden E, Han J, Harrington KJ, Hemminki A, Hodge JW, Hossain DMS, Illidge T, Karin M, Kaufman HL, Kepp O, Kroemer G, Lasarte JJ, Loi S, Lotze MT, Manic G, Merghoub T, Melcher AA, Mossman KL, Prosper F, Rekdal Ø, Rescigno M, Riganti C, Sistigu A, Smyth MJ, Spisek R, Stagg J, Strauss BE, Tang D, Tatsuno K, Van Gool SW, Vandenabeele P, Yamazaki T, Zamarin D, Zitvogel L, Cesano A and Marincola FM, J. Immunother. Cancer, 2020, 8, 1–22. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Terenzi A, Pirker C, Keppler BK and Berger W, J. Inorg. Biochem, 2016, 165, 71–79. [DOI] [PubMed] [Google Scholar]
- 6.Krysko DV, Garg AD, Kaczmarek A, Krysko O, Agostinis P and Vandenabeele P, Nat. Rev. Cancer, 2012, 12, 860–875. [DOI] [PubMed] [Google Scholar]
- 7.Fucikova J, Spisek R, Kroemer G and Galluzzi L, Cell Res., 2021, 31, 5–16. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Panaretakis T, Kepp O, Brockmeier U, Tesniere A, Bjorklund AC, Chapman DC, Durchschlag M, Joza N, Pierron G, Van Endert P, Yuan J, Zitvogel L, Madeo F, Williams DB and Kroemer G, EMBO J., 2009, 28, 578–590. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Kepp O, Menger L, Vacchelli E, Locher C, Adjemian S, Yamazaki T, Martins I, Sukkurwala AQ, Michaud M, Senovilla L, Galluzzi L, Kroemer G and Zitvogel L, Cytokine Growth Factor Rev., 2013, 24, 311–318. [DOI] [PubMed] [Google Scholar]
- 10.Kepp O, Senovilla L, Vitale I, Vacchelli E, Adjemian S, Agostinis P, Apetoh L, Aranda F, Barnaba V, Bloy N, Bracci L, Breckpot K, Brough D, Buqué A, Castro MG, Cirone M, Colombo MI, Cremer I, Demaria S, Dini L, Eliopoulos AG, Faggioni A, Formenti SC, Fučíková J, Gabriele L, Gaipl US, Galon J, Garg A, Ghiringhelli F, Giese NA, Guo ZS, Hemminki A, Herrmann M, Hodge JW, Holdenrieder S, Honeychurch J, Hu H-M, Huang X, Illidge TM, Kono K, Korbelik M, Krysko DV, Loi S, Lowenstein PR, Lugli E, Ma Y, Madeo F, Manfredi AA, Martins I, Mavilio D, Menger L, Merendino N, Michaud M, Mignot G, Mossman KL, Multhoff G, Oehler R, Palombo F, Panaretakis T, Pol J, Proietti E, Ricci J-E, Riganti C, Rovere-Querini P, Rubartelli A, Sistigu A, Smyth MJ, Sonnemann J, Spisek R, Stagg J, Sukkurwala AQ, Tartour E, Thorburn A, Thorne SH, Vandenabeele P, Velotti F, Workenhe ST, Yang H, Zong W-X, Zitvogel L, Kroemer G and Galluzzi L, Oncoimmunology, 2014, 3, e955691. [Google Scholar]
- 11.Obeid M, Tesniere A, Ghiringhelli F, Fimia GM, Apetoh L, Perfettini JL, Castedo M, Mignot G, Panaretakis T, Casares N, Métivier D, Larochette N, Van Endert P, Ciccosanti F, Piacentini M, Zitvogel L and Kroemer G, Nat. Med, 2007, 13, 54–61. [DOI] [PubMed] [Google Scholar]
- 12.Yamazaki T, Buqué A, Ames TD and Galluzzi L, Oncoimmunology, 2020, 9, 1721810. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Martins I, Wang Y, Michaud M, Ma Y, Sukkurwala AQ, Shen S, Kepp O, Métivier D, Galluzzi L, Perfettini JL, Zitvogel L and Kroemer G, Cell Death Differ., 2014, 21, 79–91. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Michaud M, Martins I, Sukkurwala AQ, Adjemian S, Ma Y, Pellegatti P, Shen S, Kepp O, Scoazec M, Mignot G, Rello-Varona S, Tailler M, Menger L, Vacchelli E, Galluzzi L, Ghiringhelli F, di Virgilio F, Zitvogel L and Kroemer G, Science (80-. )., 2011, 334, 1573–1577. [DOI] [PubMed] [Google Scholar]
- 15.Ghiringhelli F, Apetoh L, Tesniere A, Aymeric L, Ma Y, Ortiz C, Vermaelen K, Panaretakis T, Mignot G, Ullrich E, Perfettini JL, Schlemmer F, Tasdemir E, Uhl M, Génin P, Civas A, Ryffel B, Kanellopoulos J, Tschopp J, André F, Lidereau R, McLaughlin NM, Haynes NM, Smyth MJ, Kroemer G and Zitvogel L, Nat. Med, 2009, 15, 1170–1178. [DOI] [PubMed] [Google Scholar]
- 16.Luo Y, Chihara Y, Fujimoto K, Sasahira T, Kuwada M, Fujiwara R, Fujii K, Ohmori H and Kuniyasu H, Eur. J. Cancer, 2013, 49, 741–751. [DOI] [PubMed] [Google Scholar]
- 17.Apetoh L, Ghiringhelli F, Tesniere A, Obeid M, Ortiz C, Criollo A, Mignot G, Maiuri MC, Ullrich E, Saulnier P, Yang H, Amigorena S, Ryffel B, Barrat FJ, Saftig P, Levi F, Lidereau R, Nogues C, Mira JP, Chompret A, Joulin V, Clavel-Chapelon F, Bourhis J, André F, Delaloge S, Tursz T, Kroemer G and Zitvogel L, Nat. Med, 2007, 13, 1050–1059. [DOI] [PubMed] [Google Scholar]
- 18.Zitvogel L, Galluzzi L, Smyth MJ and Kroemer G, Immunity, 2013, 39, 74–88. [DOI] [PubMed] [Google Scholar]
- 19.Hanahan D and Weinberg RA, Cell, 2000, 100, 57–70. [DOI] [PubMed] [Google Scholar]
- 20.Zitvogel L, Tesniere A and Kroemer G, Nat. Rev. Immunol, 2006, 6, 715–727. [DOI] [PubMed] [Google Scholar]
- 21.Hanahan D and Weinberg RA, Cell, 2011, 144, 646–674. [DOI] [PubMed] [Google Scholar]
- 22.Zitvogel L, Apetoh L, Ghiringhelli F and Kroemer G, Nat. Rev. Immunol, 2008, 8, 59–73. [DOI] [PubMed] [Google Scholar]
- 23.Casares N, Pequignot MO, Tesniere A, Ghiringhelli F, Roux S, Chaput N, Schmitt E, Hamai A, Hervas-Stubbs S, Obeid M, Coutant F, Métivier D, Pichard E, Aucouturier P, Pierron G, Garrido C, Zitvogel L and Kroemer G, J. Exp. Med, 2005, 202, 1691–1701. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Menger L, Vacchelli E, Adjemian S, Martins I, Ma Y, Shen S, Yamazaki T, Sukkurwala AQ, Michaud M, Mignot G, Schlemmer F, Sulpice E, Locher C, Gidrol X, Ghiringhelli F, Modjtahedi N, Galluzzi L, André F, Zitvogel L, Kepp O and Kroemer G, Sci. Transl. Med, 2012, 4, 1–10. [DOI] [PubMed] [Google Scholar]
- 25.Martins I, Kepp O, Schlemmer F, Adjemian S, Tailler M, Shen S, Michaud M, Menger L, Gdoura A, Tajeddine N, Tesniere A, Zitvogel L and Kroemer G, Oncogene, 2011, 30, 1147–1158. [DOI] [PubMed] [Google Scholar]
- 26.Dudek AM, Garg AD, Krysko DV, De Ruysscher D and Agostinis P, Cytokine Growth Factor Rev., 2013, 24, 319–333. [DOI] [PubMed] [Google Scholar]
- 27.Wernitznig D, Kiakos K, Del Favero G, Harrer N, MacHat H, Osswald A, Jakupec MA, Wernitznig A, Sommergruber W and Keppler BK, Metallomics, 2019, 11, 1044–1048. [DOI] [PubMed] [Google Scholar]
- 28.Wong DYQ, Ong WWF and Ang WH, Angew. Chemie Int. Ed, 2015, 54, 6483–6487. [DOI] [PubMed] [Google Scholar]
- 29.Johnstone TC, Suntharalingam K and Lippard SJ, Chem. Rev, 2016, 116, 3436–3486. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Lebwohl D and Canetta R, Eur. J. Cancer, 1998, 34, 1522–1534. [DOI] [PubMed] [Google Scholar]
- 31.Tesniere A, Schlemmer F, Boige V, Kepp O, Martins I, Ghiringhelli F, Aymeric L, Michaud M, Apetoh L, Barault L, Mendiboure J, Pignon J-P, Jooste V, van Endert P, Ducreux M, Zitvogel L, Piard F and Kroemer G, Oncogene, 2010, 29, 482–491. [DOI] [PubMed] [Google Scholar]
- 32.Sun F, Cui L, Li T, Chen S, Song J and Li D, J. Recept. Signal Transduct, 2019, 39, 208–214. [DOI] [PubMed] [Google Scholar]
- 33.Roberts NB, Alqazzaz A, Hwang JR, Qi X, Keegan AD, Kim AJ, Winkles JA and Woodworth GF, J. Neurooncol, 2018, 140, 497–507. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Jungwirth U, Xanthos DN, Gojo J, Bytzek AK, Körner W, Heffeter P, Abramkin SA, Jakupec MA, Hartinger CG, Windberger U, Galanski M, Keppler BK and Berger W, Mol. Pharmacol, 2012, 81, 719–728. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Novohradsky V, Markova L, Kostrhunova H, Kasparkova J, Hoeschele J and Brabec V, J. Inorg. Biochem, 2022, 226, 111628. [DOI] [PubMed] [Google Scholar]
- 36.Zhu Q, Sun F, Li T, Zhou M, Ye J, Ji A, Wang H, Ding C, Chen H, Xu Z and Yu H, Small, 2021, 17, 1–11. [DOI] [PubMed] [Google Scholar]
- 37.Nishimura J, Deguchi S, Tanaka H, Yamakoshi Y, Yoshii M, Tamura T, Toyokawa T, Lee S, Muguruma K and Ohira M, In Vivo (Brooklyn)., 2021, 35, 743–752. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Flieswasser T, Van Loenhout J, Freire Boullosa L, Van den Eynde A, De Waele J, Van Audenaerde J, Lardon F, Smits E, Pauwels P and Jacobs J, Cells, 2020, 9, 1474. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Rébé C, Demontoux L, Pilot T and Ghiringhelli F, Biomolecules, 2020, 10, 1–22. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Golden EB, Frances D, Pellicciotta I, Demaria S, Helen Barcellos-Hoff M and Formenti SC, Oncoimmunology, 2014, 3, e28518. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Tham MJR, Babak MV and Ang WH, Angew. Chemie Int. Ed, 2020, 59, 19070–19078. [DOI] [PubMed] [Google Scholar]
- 42.Huang K-B, Wang F-Y, Feng H-W, Luo H, Long Y, Zou T, Chan ASC, Liu R, Zou H, Chen Z-F, Liu Y-C, Liu Y-N and Liang H, Chem. Commun, 2019, 55, 13066–13069. [DOI] [PubMed] [Google Scholar]
- 43.Bryce AH, Dronca RS, Costello BA, Infante JR, Ames TD, Jimeno J and Karp DD, J. Clin. Oncol, 2020, 38, 83. [Google Scholar]
- 44.Sabbatini M, Zanellato I, Ravera M, Gabano E, Perin E, Rangone B and Osella D, J. Med. Chem, 2019, 62, 3395–3406. [DOI] [PubMed] [Google Scholar]
- 45.Groer C, Zhang T, Lu R, Cai S, Mull D, Huang A, Forrest M, Berkland C, Aires D and Forrest ML, Mol. Pharm, 2020, 17, 4334–4345. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Deng Z, Wang N, Liu Y, Xu Z, Wang Z, Lau TC and Zhu G, J. Am. Chem. Soc, 2020, 142, 7803–7812. [DOI] [PubMed] [Google Scholar]
- 47.Novohradsky V, Pracharova J, Kasparkova J, Imberti C, Bridgewater HE, Sadler PJ and Brabec V, Inorg. Chem. Front, 2020, 7, 4150–4159. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Kenny RG and Marmion CJ, Chem. Rev, 2019, 119, 1058–1137. [DOI] [PubMed] [Google Scholar]
- 49.Wernitznig D, Kiakos K, Del Favero G, Harrer N, MacHat H, Osswald A, Jakupec MA, Wernitznig A, Sommergruber W and Keppler BK, Metallomics, 2019, 11, 1044–1048. [DOI] [PubMed] [Google Scholar]
- 50.Wernitznig D, Meier-Menches SM, Cseh K, Theiner S, Wenisch D, Schweikert A, Jakupec MA, Koellensperger G, Wernitznig A, Sommergruber W and Keppler BK, Metallomics, 2020, 12, 2121–2133. [DOI] [PubMed] [Google Scholar]
- 51.Meier-Menches SM, Zappe K, Bileck A, Kreutz D, Tahir A, Cichna-Markl M and Gerner C, Metallomics, 2019, 11, 118–127. [DOI] [PubMed] [Google Scholar]
- 52.Monro S, Colón KL, Yin H, Roque J, Konda P, Gujar S, Thummel RP, Lilge L, Cameron CG and McFarland SA, Chem. Rev, 2019, 119, 797–828. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Konda P, Lifshits LM, Roque JA, Cole HD, Cameron CG, McFarland SA and Gujar S, Oncoimmunology, 2021, 10, 1863626. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Wang L, Guan R, Xie L, Liao X, Xiong K, Rees TW, Chen Y, Ji L and Chao H, Angew. Chemie, 2021, 133, 4707–4715. [DOI] [PubMed] [Google Scholar]
- 55.Vigueras G, Markova L, Novohradsky V, Marco A, Cutillas N, Kostrhunova H, Kasparkova J, Ruiz J and Brabec V, Inorg. Chem. Front, 2021, 8, 4696–4711. [Google Scholar]
- 56.Kaur P, Johnson A, Northcote-Smith J, Lu C and Suntharalingam K, ChemBioChem, 2020, 21, 3618–3624. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Wang Y, Ding Y, Yao D, Dong H, Ji C, Wu J, Hu Y and Yuan A, Small, 2021, 17, 2006231. [DOI] [PubMed] [Google Scholar]
- 58.Sen S, Hufnagel S, Maier EY, Aguilar I, Selvakumar J, DeVore JE, Lynch VM, Arumugam K, Cui Z, Sessler JL and Arambula JF, J. Am. Chem. Soc, 2020, 142, 20536–20541. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59.Van Loenhout J, Freire Boullosa L, Quatannens D, De Waele J, Merlin C, Lambrechts H, Lau HW, Hermans C, Lin A, Lardon F, Peeters M, Bogaerts A, Smits E and Deben C, Cells, 2021, 10, 2936. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Vanmeerbeek I, Sprooten J, De Ruysscher D, Tejpar S, Vandenberghe P, Fucikova J, Spisek R, Zitvogel L, Kroemer G, Galluzzi L and Garg AD, Oncoimmunology, 2020, 9, 1703449. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.Garg AD, More S, Rufo N, Mece O, Sassano ML, Agostinis P, Zitvogel L, Kroemer G and Galluzzi L, Oncoimmunology, 2017, 6, e1386829. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62.Wu J and Waxman DJ, Cancer Lett., 2018, 419, 210–221. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63.Sakakibara K, Sato T, Kufe DW, VonHoff DD and Kawabe T, Oncotarget, 2017, 8, 78277–78288. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64.King AP and Wilson JJ, Chem. Soc. Rev, 2020, 49, 8113–8136. [DOI] [PubMed] [Google Scholar]
- 65.Zheng P, Ding B, Jiang Z, Xu W, Li G, Ding J and Chen X, Nano Lett., 2021, 21, 2088–2093. [DOI] [PubMed] [Google Scholar]
- 66.Zhang C, Liu H, Gong M, Yang M, Yang Z, Xie Y and Cai L, Mater. Des, 2021, 210, 110087. [Google Scholar]
- 67.Pan Z, Tan C, Rao L, Zhang H, Zheng Y, Hao L, Ji L and Mao Z, Angew. Chemie, 2020, 132, 18914–18921. [DOI] [PubMed] [Google Scholar]
- 68.Duan X, Chan C, Han W, Guo N, Weichselbaum RR and Lin W, Nat. Commun, 2019, 10, 1899. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 69.Ding B, Zheng P, Jiang F, Zhao Y, Wang M, Chang M, Ma P and Lin J, Angew. Chemie - Int. Ed, 2020, 59, 16381–16384. [DOI] [PubMed] [Google Scholar]
- 70.Zhou F, Feng B, Yu H, Wang D, Wang T, Ma Y, Wang S and Li Y, Adv. Mater, 2019, 31, 1805888. [DOI] [PubMed] [Google Scholar]
- 71.Feng B, Hou B, Xu Z, Saeed M, Yu H and Li Y, Adv. Mater, 2019, 31, 1902960. [DOI] [PubMed] [Google Scholar]
- 72.Gao J, qi Wang W, Pei Q, Lord MS and jun Yu H, Acta Pharmacol. Sin, 2020, 41, 986–994. [DOI] [PMC free article] [PubMed] [Google Scholar]