Abstract
The coronavirus disease 2019 (COVID-19) pandemic has disproportionately affected patients with kidney disease, causing significant challenges in disease management, kidney research and trainee education. For patients, increased infection risk and disease severity, often complicated by acute kidney injury, have contributed to high mortality. Clinicians were faced with high clinical demands, resource shortages and novel ethical dilemmas in providing patient care. In this review, we address the impact of COVID-19 on the entire spectrum of kidney care, including acute kidney injury, chronic kidney disease, dialysis and transplantation, trainee education, disparities in health care, changes in health care policies, moral distress and the patient perspective. Based on current evidence, we provide a framework for the management and support of patients with kidney disease, infection mitigation strategies, resource allocation and support systems for the nephrology workforce.
Subject terms: Nephrology, Infectious diseases
In this Review, the authors summarize the challenges associated with the care of patients with kidney disease during the COVID-19 pandemic. They describe the major challenges and missed opportunities, global inequalities in health care, and offer a framework for future pandemic preparedness.
Key points
Acute kidney injury is common in severe coronavirus disease 2019 (COVID-19) and is associated with increased mortality.
Patients with chronic kidney disease are at a high risk of severe COVID-19 and severe outcomes, and should be prioritized for therapeutics, including vaccines.
Establishment of global collaborative registries is key to assessing the severity and risk factors of infection.
Interruptions in routine care have been common and highlighted the advantages of temporary implementation of telemedicine and home dialysis.
There have been gross inequities in access to COVID-19 testing, personal protective equipment, provision of dialysis services, COVID-19 vaccines and therapeutics rollout.
To prepare for future pandemics, it is important to stockpile emergency medical equipment, invest in resilient health care systems, have global cooperation in providing care, explore and advance remote care globally, address moral distress to improve the well-being of patients and care providers, build public trust in scientific recommendations and advocate for kidney patients to be included in clinical trials and global registries.
Introduction
The coronavirus disease 2019 (COVID-19) pandemic has impacted the kidney community on multiple levels. Acute kidney injury (AKI) is a common consequence of hospitalization with COVID-19 and portends high mortality1. Moreover, the rate of severe COVID-19 have been high in patients with chronic kidney disease (CKD), including dialysis patients and kidney transplant recipients1–3. Throughout the pandemic, a number of dilemmas have occurred, contributing to poor outcomes in patients with kidney disease. These include decisions surrounding the delivery of safe clinical care, triaging provision of kidney replacement therapy (KRT), the safety of donors and recipients for kidney transplantation, managing COVID-19 in patients with kidney disease, modulating immunosuppression in immune-mediated kidney disease and transplant recipients, and the best ways to protect patients and health care providers. Despite being at a high risk, however, patients with kidney disease were excluded from therapeutic clinical trials of COVID-19, largely because of a higher risk of mortality and concerns that CKD may alter the safety profile of drugs. As such, the lack of pre-specified subgroup analyses resulted in the application of novel therapies to this population with low evidence. Additional challenges in patient care faced by nephrologists have included nursing shortages and supply chain issues. Moreover, the pandemic has raised ethically challenging questions about the allocation of scant health care resources. Finally, there has been a significant impact on trainee education and on the emotional well-being of the nephrology workforce and patients4,5.
In this review, we discuss the challenges encountered in the care of patients across the spectrum of kidney disease during the COVID-19 pandemic. Strategies that were adopted to address these challenges, innovations in patient care delivery and advocacy efforts are described. We further discuss the global inequities in the provision of health care and highlight how the pandemic may exacerbate pre-existing disparities in kidney disease. Although some lessons learned by the kidney community during the pandemic have been heartening and stimulated innovation, others have had profoundly devastating effects. These experiences by the kidney community shape preparedness efforts for future pandemics. From the lessons learned thus far, we provide a framework for the management of patients with kidney disease for future pandemics.
Challenges and opportunities of COVID-19
AKI incidence throughout the pandemic
AKI is common in patients diagnosed with COVID-19 (ref.1). The mechanism of COVID-19 infection-induced AKI is multifactorial, involving both direct invasion of the virus and indirect mechanisms through acute tubular necrosis, dysregulation of the immune system, hypercoagulopathy and collapsing glomerulopathy6. In the early stages of the pandemic, the reported incidence of AKI ranged from 0.5 to 42%1,7,8, with a large percentage of patients requiring dialysis. Investigating how this incidence rate changed over time, one study found that in critically ill patients with COVID-19, although the incidence of AKI was 29.3%, the rates of AKI and KRT declined throughout the pandemic8. This reduced rate of AKI was likely attributable to multiple factors, including improved recognition and management, vaccine rollout and transmission of less virulent strains9. However, in low- and lower-middle-income countries, challenges were present in the management of COVID-19-associated AKI10. In a study of over 1,000 patients with COVID-19 in two teaching hospitals in South Africa, AKI occurred at a rate of 33.9%, with approximately a quarter of patients with AKI requiring ICU management and 8.6% receiving KRT11.
APOL1 and risk of COVID-19-related AKI
In April 2020, several case reports described collapsing glomerulopathy in patients with COVID-19. A subsequent study confirmed this finding, reporting that, out of 240 native kidney biopsies in patients with COVID-19, 62 (26%) had evidence of collapsing glomerulopathy12. Interestingly, a case series of six patients with collapsing glomerulopathy found that all six had variants in the APOL1 gene13. APOL1 genotyping studies have subsequently demonstrated that patients with high-risk APOL1 alleles had significantly higher odds of AKI and death than low-risk patients with 0 or 1 risk variants14. Furthermore, in African Americans who tested positive for COVID-19, APOL1 high-risk alleles were associated with greater odds of AKI and higher rates of persistent AKI and requirement for dialysis14.
Possible reasons for the higher risk of adverse kidney outcomes in individuals carrying APOL1 high-risk alleles include a heightened inflammatory response to COVID-19, leading to inflammatory kidney injury. Thus, the incidence and severity of AKI are likely increased in the presence of APOL1 variants, with an increased requirement for chronic dialysis.
COVID-19 and long-term kidney issues
Hospitalized patients with COVID-19 have a higher incidence of AKI overall, and an increased risk of higher stages of AKI when compared with patients with influenza15. The risk of developing CKD is high in patients with COVID-19, as demonstrated by an observational study from the UK showing that 16% of AKI survivors had progressed to CKD at 90 days16. Similarly, approximately 20% of critically ill patients with COVID-19 and AKI requiring KRT had no recovery at 3 months after discharge in a multicentre study from the USA17. A veterans administration study on long COVID reported a higher risk of CKD with COVID-19 (refs.18,19). Similarly, in a Chinese cohort study, a third of COVID-19-infected patients had reduced kidney function 6 months post-hospitalization18. Interestingly, patients developing AKI during their COVID-19 disease course had no increased risk of CKD at 12 months follow-up compared with non-COVID-19 AKI cases20, indicating that kidney disease progression is similar in patients with or without COVID-19. Further studies are needed to evaluate risk factors for progression to CKD following COVID-19-associated AKI.
Chronic kidney disease
The incidence of COVID-19 in patients with CKD is difficult to establish owing to under-reporting, under-diagnosis of CKD, and variations in access to SARS-CoV-2 testing. A large, nationally representative cohort from the UK demonstrated a CKD incidence and prevalence ranging from 0.5% to 37% and reported that CKD is associated with a high co-morbidity burden and high 1-year mortality21. CKD, including end-stage kidney disease (ESKD), is associated with an increased risk of COVID-19 and subsequent adverse outcomes including hospitalization, respiratory failure and mortality2,22–24, with risk proportional to kidney dysfunction22,24. There have been care delivery repercussions, including the postponement of kidney biopsies25, delays in arteriovenous fistula surgery and salvage, and disruption in the initiation of KRT26. There was a notable decline in incident ESKD in the USA early in the pandemic, because of both increased mortality in patients with advanced CKD and compromised access preparation for KRT27. Large-scale lockdowns resulted in interruption of routine health care, including access to dialysis, leading to preventable deaths, especially in those parts of the world where access to dialysis was already compromised, while highlighting the advantages of telemedicine in patients with CKD28–30.
There is a lack of guidelines related to the management of COVID-19 in patients with CKD. Dexamethasone was associated with both a reduced requirement for KRT and lower mortality in all patients with severe COVID-19, although fewer than 10% of patients in this trial had an estimated glomerular filtration rate of <30 ml/min/1.73 m2 (ref.31). Remdesivir, a direct-acting antiviral, remains off label in patients with an estimated glomerular filtration rate <30 ml/min/1.73 m2 (ref.32). Several monoclonal antibody therapies have been afforded emergency use authorization but their net benefit in patients with CKD remains untested. Tocilizumab, a recombinant anti-IL-6 receptor antibody was granted emergency use authorization in patients with moderate-to-severe COVID-19 (ref.33), and case reports demonstrate successful deployment in patients with ESKD. Unfortunately, phase III trials evaluating the benefits of vaccination against COVID-19 have placed little focus on specific subgroups such as patients with CKD34. Nonetheless, patients with CKD mounted a seroconversion rate that was comparable with the general population (Supplemental Table 1) and were prioritized for vaccination. It is noteworthy, however, that patients who did not receive a third dose of the vaccine had a significant decline in antibody titre after 3 to 6 months35.
Immune-mediated kidney disease
Managing patients with immune-mediated glomerular disease has presented unique challenges. At the start of the pandemic, the primary concerns for patients receiving immunosuppression included the assessment of the risk and severity of COVID-19, ways to mitigate infection risk, methods of clinical and laboratory surveillance, strategies to treat active disease and evaluation of the need for continued immunosuppressive therapy. After the vaccine rollout, questions were posed regarding the immunogenicity and safety of the vaccine in this patient subgroup, measurement of vaccine efficacy and need for modification of immunosuppressive therapy to improve vaccine response.
Although immunosuppressive therapy increases the risk of COVID-19 infection, there is currently no evidence to support deviation in the standard of care treatment of active kidney disease36. COVID-19 in patients with immune-mediated kidney disease conferred an increased risk of AKI and death37. Additionally, case reports of de novo immune-mediated kidney disease, including collapsing focal and segmental glomerulosclerosis, vasculitis and IgA nephropathy, have been described with COVID-19 infection. Among the immunosuppressive medications commonly used in this cohort, rituximab and prednisone dose ≥10 mg were associated with increased severity of COVID-19 (ref.38); information on other therapies such as cyclophosphamide has remained scarce.
Although patients with immune-mediated kidney disease were prioritized for vaccines, understanding of the safety and immunogenicity of vaccines in this cohort was based only on observational studies. Studies in patients with rheumatic diseases on immunosuppressive therapy have consistently shown impaired humoral response with largely preserved cellular response following vaccination (Supplemental Table 1). Additionally, these studies have shown that prednisone (dose ≥10 mg), mycophenolate mofetil and rituximab were associated with impaired humoral response and that the response in rituximab-treated patients was improved when B cells were reconstituted or when time elapsed since the last rituximab administration was longer39. Temporary cessation of mycophenolate mofetil augmented humoral response following vaccination in patients with rheumatic disease40. In non-responders, administration of a booster vaccine dose was shown to increase the response rate. De novo or relapsing autoimmunity triggered by COVID-19 vaccines remains a concern, with isolated reports of de novo and relapsing minimal change disease, membranous nephropathy, IgA nephropathy and ANCA vasculitis having been described41. However, large cohorts with increased inflammatory disease incidences have not been described following COVID-19 vaccination.
In the absence of guidelines, caring during a pandemic requires personalizing immunosuppressive therapy to balance the risk of infection and disease control, and recognizing that treatment of active disease cannot be delayed. An important lesson learned is that the early establishment of global collaborative registries is critical to assessing the severity and risk factors of infection and guiding modulation of immunosuppressive therapy in this population.
Patients on chronic dialysis
Early data from Wuhan foreshadowed the major challenges that other regions would have to tackle in caring for patients on dialysis during the COVID-19 pandemic. Patients on dialysis were amongst those at highest risk of death, not just because of the propensity for serious illness but also because of missed treatments. These data generated a mandate for dialysis facilities to intensify infection prevention protocols to reassure patients and staff, to ultimately prevent missed sessions and loss of workforce. There was a strong recommendation for transition to home modalities42.
However, without additional resources to match the new guidance, there was a constant shortage of personal protective equipment (PPE)43 and SARS-CoV-2 tests. Data from universal screening of a single dialysis unit in the UK showed that patients experienced a spike in infection following a spike in infection of health care workers, implying potential transmission from health care worker to patient44. Nonetheless, widespread in-facility transmission was not reported. Lack of transportation, staff shortages and possibly personal fear or symptoms may have contributed to missed treatments in up to half of the surveyed facilities in Africa, Latin America, Middle East, South East Asia and South Asia43. Mortality among infected and hospitalized patients receiving dialysis exceeded 50% in most regions of the world45. The pivot to home modalities was not feasible in many environments that were already strained. Thus, despite early and sensible guidance on infection prevention and management — a majority of which still holds relevance in face of the changing nature of the pandemic — there was a stark loss of life among patients receiving dialysis46. With improvement in testing capacity and identification of patients with no or minimal symptoms, there has been improvement in crude mortality rates between the first and second wave of COVID-19 (ref.47).
The success of vaccines has been accompanied by challenges. Among countries that procured vaccine supply in late 2020 or early 2021, only European countries prioritized vaccination for patients receiving dialysis. In the USA, there was no specific prioritization until March 2021, at which point policy makers acknowledged that patients receiving dialysis were disproportionately from underserved populations and at a high risk of complications from COVID-19 illness. This led to a federal effort to offer vaccination in dialysis clinics, which improved vaccine access, especially for Hispanic and Black patients48. Despite this effort, however, some patients on dialysis expressed vaccine hesitancy, albeit to a lower extent than in the general population. Capitalizing on central record keeping, the US Centers for Disease Control and Prevention has created a real-time dashboard tracking vaccination in dialysis facilities, which indicates that more than 75% of patients on dialysis have had at least two doses of COVID-19 vaccine.
Several lines of evidence suggest that the immunogenicity of vaccination is lower among patients on dialysis than in the general population (Supplemental Table 1). Although the majority of patients ‘seroconverted’ post-vaccination, the strength of the early antibody response to vaccination was suboptimal. Furthermore, 20% lost a detectable antibody response within 6 months. Consistent with data from hepatitis B vaccination, low circulating antibody titres were associated with a 10-fold higher risk of infection among patients who had completed the initial vaccination series45. Data from a large not-for-profit dialysis centre indicated efficacy against hospitalization or death of 81%, only slightly lower than contemporaneous data for the general population, noting vaccine effectiveness of around 86–87%49. Vaccine immunogenicity by vaccine type has been studied closely, and concerns have been raised regarding the lack of immunogenicity of Ad26.COV2.S, although clinical effectiveness of this vaccine may be similar to the mRNA platform vaccines50. Furthermore, there was no difference in vaccine response by vaccine type among home dialysis versus in-centre patients51.
Boosters (third doses for persons with mRNA platform vaccines) have seen a lower uptake among patients on dialysis in the USA, with roughly 50% of patients reporting an additional dose to date. Again, there was a heterogenous policy implementation, with countries in Europe implementing earlier offers of third doses among patients on dialysis. Data from France and the USA on immunogenicity are encouraging, with strong responses reported even among older patient groups.
In this landscape, given the uncertainty around persistent immunogenicity to COVID-19 vaccines and substantial risk of ‘breakthrough’ infection — a majority of which will have some clinical consequences for patients receiving dialysis, and at the very least a need for isolation during in-centre dialysis — it will be critical to continue to develop future protocols for prevention, detection and early treatment of COVID-19.
Lessons for policymakers
Policymakers have important lessons to learn from the COVID-19 pandemic that are specific to the dialysis population28. Below, we outline key lessons learned from the USA.
First, policymakers should strongly consider extending waivers to exempt providers from pre-existing value-based purchasing programmes, including the Quality Incentive Program52 and the End-stage Renal Disease Treatment Choices model53. These programs have laudable goals but might pose a distraction for dialysis facilities that need to remain nimble during a public health emergency. To its credit, the Centers for Medicare and Medicaid Services rapidly implemented waivers early in the pandemic, allowing facilities to focus on the emergency54.
Second, the risk that in-centre dialysis poses for patients, particularly with airborne diseases, must be addressed. Early in the pandemic, providers rightly recognized that facilities could quickly become hubs for widespread infection55. One welcome addition was the broad expansion of telehealth benefits56. However, the beginning of the COVID-19 pandemic was also fraught because of strapped supply chains that exacerbated the already limited supply of PPE. The American Society of Nephrology (ASN), the European Renal Association and the International Society of Nephrology issued calls for governments around the world to prioritize PPE for dialysis personnel and increasing access to lifesaving dialysis. To prepare for the next pandemic, not only should policymakers stockpile more emergency medical equipment but they should also place a strong emphasis on distributing these supplies to providers of populations with complex medical issues, including dialysis facilities. A major concern is ongoing shortages in dialysis supplies, including dialysate.
Third, we must customize policies to address the specific needs of the dialysis population. For instance, per Centers for Medicare and Medicaid Services guidance, many hospitals deferred “non-essential” surgical procedures. However, these deferrals may have inadvertently harmed incident ESKD patients who needed dialysis access procedures27.
New therapeutics that can treat COVID-19 have shown incredible promise in reducing hospitalizations and mortality57,58. Dialysis facilities are unique in the health care industry because patients must return to the facility regularly. In the future, dialysis facilities should be considered a major site for the distribution of new therapeutics and vaccines.
Kidney transplant recipients
The pandemic has created significant challenges for kidney transplantation. Transplant candidates and recipients, especially in the early post-transplant period, experienced significant excess mortality related to COVID-19, with a disproportionate impact on racial minorities and socio-economically disadvantaged individuals59,60. Both the innate and adaptive immune systems seemed profoundly altered in transplant patients, with significantly lower levels of anti-spike antibodies up to 2 months following the onset of COVID-19 symptoms compared with patients on dialysis61. As such, prime concerns have been centred around continuing kidney transplant surgeries while minimizing the risk of infection and management of post-transplant immunosuppression.
Globally, living and deceased donor transplantation has been adversely impacted to various extents and at different periods of time. This is due, in large part, to efforts to conserve resources during a COVID-19 surge and concern for the risk of newly immunocompromised individuals given their increased risk and poor outcomes, particularly during periods of high community transmission3. The mortality rate was 20–30% in kidney transplant recipients during the first wave of the pandemic, with a reduction in mortality during the second wave47. Although there was a 16% global decrease in transplant activity, most notably during the first 3 months of the pandemic, there were substantial differences in transplant activity between countries62. Although living donation came to a nearly complete stop early on during the pandemic, it has resumed since then, but does not appear to have reached pre-pandemic levels63. Notably, previously decreased donor transplantation rates have continued to increase in the US allocation system, despite a dramatic increase in organ discards, reflecting increased selectivity of organs and patients64. There are notable differences in mortality rates of waitlisted individuals compared with transplant recipients, with the USA reporting higher mortality in waitlisted individuals, and Europe and the UK reporting higher mortality in transplant recipients. The decision to continue transplants during a pandemic needs to be individualized for each country and should take into consideration the mortality risk of waitlisted individuals as well as transplant recipients and infection risk in the immediate post-transplant period. In addition, the pandemic has also impacted transplant activity owing to concerns regarding donor-derived viral transmission. A systematic review of 69 transplants from 57 donors infected with SARS-CoV-2 demonstrated than non-lung transplantation was safe, with a low risk of transmission65.
Efforts to lower the risk of transmission along with overwhelmed health care systems created significant challenges in the care of patients. Health care systems pivoted quickly towards telehealth strategies and there was increased interest in the use of non-invasive biomarkers when kidney biopsies became a challenge66–68. Although the value of monitoring strategies for allograft health remains uncertain, there does not appear to have been a dramatic uptick in acute rejection episodes69.
The mainstay of treatment for COVID-19 in transplant recipients included reduction or cessation of antimetabolite therapy for 2 weeks or longer, in addition to standard adjuvant therapies used in the general population70. Although this approach also contributed to early concerns about adverse allograft consequences, recent data suggest that brief cessation of therapy was not associated with the development of donor-specific antibodies71. Additional concerns in the SARS-CoV-2-infected recipient include the abrupt increase in tacrolimus levels that has been observed at the time of presentation72. Immunosuppressed individuals also appear to have a prolonged high viral burden with persistent positive PCR results, which may have implications for when to allow patients with previous COVID-19 infection back in to the transplant clinic setting.
Although immunosuppressed patients and those with kidney disease were excluded from the initial vaccine trials, significant real-world experience has been gained in these groups. Studies of immunogenicity after vaccination revealed poor humoral responses to two doses of both mRNA and viral vector vaccines73,74. Older age, impaired allograft function, and use of triple maintenance immunosuppression, belatacept, steroids and anti-metabolites were associated with poor humoral response. Additionally, breakthrough infections were frequently observed in kidney transplant recipients, even before the omicron surge. An enhanced humoral response was observed after a third and fourth vaccine dose, use of heterologous vaccination and modulation of immunosuppression75–77.
Kids, kidneys and COVID-19
Outcomes in children
Children and adolescents are a vulnerable group and are subject to special considerations in health care, research and public policy. Although the clinical impact of COVID-19 on paediatric patients has been less profound than in adults so far, successive waves of the pandemic have led to more children being directly impacted. At the start of the pandemic in the USA, there was an initial decrease in deceased and living donor kidney transplantation; however, rates of transplantation had returned to pre-pandemic levels by May 2020 (ref.78). Unlike in adults, children taking immunosuppression for kidney disease or kidney transplant and children on dialysis have not had worse outcomes from COVID-19 infection than the general paediatric population79–82. When sick enough to be admitted, however, 12–23% of hospitalized children with COVID-19 developed AKI83–85 and AKI is more common in patients with the multisystem inflammatory syndrome in children83. Consistent with other studies of AKI in children, AKI was associated with increased levels of care, length of hospital stay and worse outcomes83,85–87.
Gaps in science and child health policy
Compared with adults, many gaps in science remain for the paediatric population. Long-term COVID-19 outcomes are an important area of future study, and emphasis should be placed on developing safe and effective strategies to incorporate children into such studies. Research studying long COVID is lacking in children, despite evidence that it is at least as common for children as for adults88. Furthermore, although vaccination rates are much lower for children than for adults, we need to learn more about vaccination patterns and perceptions among children with kidney disease and their caregivers89. It will take years to understand the impact of educational disruptions that affected children with CKD, who already have lower cognition than the general population90. Moreover, paediatric research addressing the impact of pandemic disruptions on access to transplants, the early detection of kidney disease and the impact on family dynamics could aid in the development of more equitable and durable paediatric care delivery models and public policy. Table 1 outlines the challenges and missed opportunities faced by the kidney community in managing patients during the pandemic.
Table 1.
Challenges in managing patients with kidney disease during the COVID-19 pandemic
| Cohort type | Challenges | Solutions | Actions required |
|---|---|---|---|
| AKI |
Increased demand for bedside dialysis and CRRT Shortage of dialysis solutions and workforce |
Organization of multidisciplinary crisis team to include nephrologists, nurses and hospital administrators Taking inventory of all aspects of RRT Tracking daily need for RRT Modification of HD and CRRT protocols to meet increased demand Utility of acute PD Redeployment of faculty, trainees and nurses to meet needs |
Develop a framework for addressing system capacity, challenges in communication and allocating resources founded on ethical principles Educate patients about AKI and risks |
| CKD |
Interruption of new consultations and follow-up care Laboratory monitoring of CKD Limited pre-dialysis access care due to the pandemic Lack of data on therapeutics and vaccine response in the CKD population |
Adoption of telemedicine for all except urgent cases requiring in-person evaluation Restrict laboratory tests to those with rapid turnaround for clinical care and use non-hospital-based labs for blood draw Home urine dipstick monitoring Include vascular access surgeries and PD catheter placement among essential procedures |
Evaluate disparities in digital literacy and establish a protocol to include telemedicine navigators to facilitate telemedicine Advocacy for inclusion of CKD cohorts in clinical trials of therapeutics and vaccines |
| Patients with ESKD on maintenance dialysis |
Safe continuation of thrice weekly in-centre dialysis Training for home dialysis modalities and longitudinal care for home dialysis patients Delay in dialysis access placement Delays in transplant evaluation and placement on waiting list Lack of data on therapeutics and vaccine response in the ESKD population |
Protocol for symptom screening for infection and universal masking Cohorting infected patients in designated COVID units Wider adoption of home dialysis modalities Conversion from in-person to televisit for in-centre and home dialysis patients Inclusion of dialysis access as an essential procedure Reduction of dialysis sessions to twice a week Conversion to home dialysis |
Universal viral testing for symptomatic in-centre dialysis patients Stock-piling of emergency medical equipment and dialysis supplies Adoption of assisted peritoneal dialysis and home hemodialysis Development of algorithms to accelerate evaluation and placement of medically stable individuals on waiting list Inclusion of patients with ESKD in clinical trials of therapeutics and vaccines |
| Kidney transplant recipients |
Strategies to reduce risk of infection Continuation of evaluation for transplant candidacy Continuation of transplant surgeries Evaluating vaccine efficacy |
Adoption of telemedicine and remote monitoring Suspension of live donation and decrease in DDKT Vaccine prioritization, booster doses, vaccination of household contacts |
Leveraging health care technology to aid remote monitoring of vital signs and glucose Algorithm for individualized approach to continuing transplant surgery Strategies to enhance vaccine efficacy |
| Immune-mediated kidney disease |
Strategies to reduce risk of infection Identifying immunosuppressive classes associated with increased risk of infection Evaluating vaccine efficacy |
Adoption of telemedicine and remote monitoring Decrease in frequency of laboratory monitoring Delaying use of biologics in stable patients Vaccine prioritization, booster doses, vaccination of household contacts |
Leveraging health care technology to aid remote monitoring of disease activity and adoption of urine dipstick monitoring Comparing utility of non-invasive disease biomarkers and kidney biopsy for glomerular diseases Algorithms to personalize maintenance immunosuppressive therapy for relapsing diseases Strategies to enhance vaccine efficacy |
| Children living with kidney disease |
Interruption of follow-up care for CKD Continuation of transplant surgeries Lack of data on therapeutics and vaccine response in the CKD population Psychological impact of isolation and shielding Caregiver burden |
Adoption of telemedicine and remote monitoring Suspension of live donation |
Wider adoption of home dialysis Strategies to address caregiver burden Inclusion of children in clinical trials and prioritization of high-risk groups for vaccines Research into kidney health and cardiovascular consequences of the pandemic Address paediatric health equity through research and public policy Ensure resources to maintain critical services for children on dialysis Utilize patient-reported outcomes along with relevant health measures |
AKI, acute kidney injury; CKD, chronic kidney disease; CRRT, continuous renal replacement therapy; DDKT, deceased donor kidney transplantation; ESKD, end-stage kidney disease; HD, haemodialysis; PD, peritoneal dialysis; RRT, renal replacement therapy.
Psychosocial aspects of COVID-19
The COVID-19 pandemic has necessitated a change in almost every aspect of kidney care. Health care resources shifted to prevent, detect and manage waves of COVID-19, leading to dramatic alterations in routine kidney care in many countries. This section highlights some of the changes instituted, and their impact on patients, caregivers and health care providers.
As the potential severity of COVID-19 disease became clear, international efforts were made to identify people at risk. Many nations endorsed targeted public health measures to minimize both mortality and economic impact91,92. For example, in the UK, a targeted national policy of ‘shielding’ was implemented. Those considered most at-risk from COVID-19 were centrally identified using electronic records, and government letters were issued advising individuals to socially isolate themselves, restricting contact even within their household group, with the help of financial and logistical support. Many people with advanced kidney disease, kidney transplantation, and/or those requiring immunosuppressive treatment, were advised to shield93,94. Although these measures were broadly supported, the personal impact varied, with some feeling protected while others felt fearful and isolated94. In countries without such protective policies, public health messaging likely encouraged similar exposure-avoidant behaviours, particularly in at-risk groups95–97.
Additional strategies to minimize infection were implemented internationally, including reduced visitor access in hospitals98,99 and dialysis units100. End-of-life care provision was dramatically altered because of the restrictions, with limitations of social contact and rituals before and after death101. Even with technological innovations to provide human connection, the impact of reduced physical contact between patients, caregivers and clinicians was significant. Some patients experienced loneliness and depressive symptoms, and caregivers described heightened anxiety and an increased desire for information from health care professionals102. Many patient and provider groups described ethical compromise and psychological distress, as they felt unable to provide or receive care at pre-pandemic levels103,104. Furthermore, patient and provider groups have described adversity, fear, abandonment, hope and resilience105–108. A multinational mixed methods study of 251 kidney health care providers found that nearly one-third of respondents were at a high risk of burnout and mental health distress during the pandemic, with feelings of emotional exhaustion, depersonalization and a reduced sense of personal accomplishment109.
Moral distress in health care during the COVID-19 pandemic has been extensively described and may explain some of the negative psychological consequences described above104. Moral distress can occur when an individual perceives that they are unable to act according to their ethical values owing to external barriers110. If individuals perceive that their ethical duties are compromised in settings of severe resource constraints — where institutional, health policy or financial barriers limit access to optimal treatments that are clinically indicated — moral distress can occur111,112. Notably, it occurs in both patients and caregivers and has been described in relation to the intentional separation between loved ones during end-of-life care and hospital visitation restrictions103,108. The consequences of moral distress include experiences of anger, guilt, depersonalization and, for health care professionals, a desire to leave the workforce entirely113. If persistent, moral distress can result in moral injury, resulting in long-term social and psychological trauma114,115.
Children and young adults with kidney disease face unique and pervasive mental and behavioural health challenges, with higher rates of depression, anxiety and neurocognitive disorders than their peers90. One survey reported that children felt that they were missing out on work-related and educational opportunities, missing family and friends, and compared with their peers, they lived with more COVID-related restrictions90. Health-related quality of life and physical activity decreased significantly for both children and adolescents during the pandemic owing to school closures, social distancing and home confinement116. Although these strategies were employed to reduce virus transmission, their prolonged use requires assessment to mitigate the adverse psychological effects, especially in populations at a high risk of COVID-19. Furthermore, parents and caregivers of children with kidney disease experience significant psychosocial stressors that leave many families dysfunctional and disempowered. During the pandemic, this often-unseen care burden has been experienced disproportionately by families struggling with adverse social determinants of health and health disparities. Caregivers of children with kidney disease reported feelings of stress, anxiety, depression and insomnia during the pandemic, mirroring findings in parents of children with other chronic conditions117,118. Children with medical complexities have lost access to therapies, educational services and peer interactions, all while parents and caregivers have taken on additional responsibilities to navigate changes in employment and keeping their families healthy119.
A global health perspective
With its rapid spread across the globe, the COVID-19 pandemic revealed that most health systems were unprepared for, or at least underestimated, the challenge it would pose. Initially, the lack of readiness, combined with an almost lack of belief that such a pandemic could occur in the current day and age, resulted in acute shortages of many items needed for an effective response. A pertinent example is the scarcity of PPE early on in the pandemic, which sometimes resulted in hoarding by countries and by individuals. This “catastrophic breakdown in global cooperation”120 highlighted the need to develop global strategies to improve equity and access equipment, treatment and vaccines to treat COVID-19 (refs.120,121). Unfortunately, a lack of equity and empathy persists, with booster doses of vaccines being administered to most adults and children in some countries before adults at risk of poor outcomes even receive their first vaccine in others122. Hoarding, pricing, protection of intellectual property and dissemination of misinformation regarding vaccines have exacerbated inequities and contributed to deaths123. These persistent and pervasive inequities, which impact how individuals and nations have been (un)able to tackle the challenges posed by the pandemic have coined the term “political determinants of health”124.
Political and social determinants of health exist within as well as between countries, and the same populations who have experienced centuries of structural violence (such as African Americans and Indigenous populations) are those at the highest risk of serious illness from COVID-19 and most at risk of poor outcomes125–131. People living with chronic diseases, and especially kidney disease, are at the highest risk132. These facts have finally raised global awareness that we cannot continue to overlook non-communicable diseases, including when looking beyond the pandemic133,134. As the demand for hospital beds and health care services outstripped availability at various stages, triage guidance had to be urgently drawn up to allocate scarce intensive care unit beds, raising debates around which criteria would be morally acceptable135,136. Rationing of health care services became a reality, faced by many for whom the concept had been merely theoretical before then137–140.
The nephrology community was rapidly drawn into the eye of the storm. The capacity to provide dialysis became strained in some settings, leading to complex triage algorithms and in some cases deaths, even in high-income countries (HICs) because of a lack of access to dialysis140,141. People living with kidney disease are an at-risk population, tending to have lower socio-economic status, belong to minority populations and live with multiple co-morbidities142.
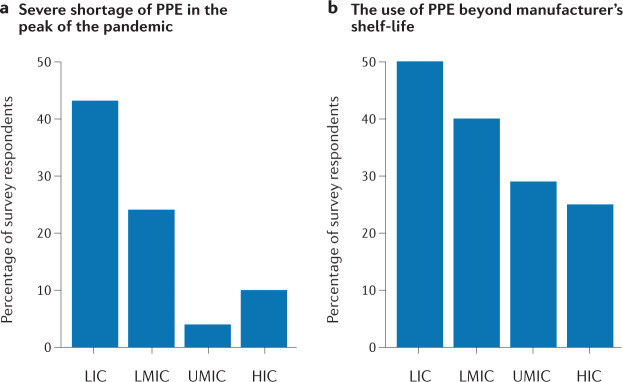
The pandemic has had a significant global impact on health care delivery in general, with consequences being particularly evident in low- and lower-middle-income countries (LLMICs)43. Notably, there were gross inequities in the provision of dialysis services143. Many guidelines were developed and disseminated for the management of patients with kidney disease, including those on dialysis, but most of these guidelines could not be adhered to because of the lack of resources in most LLMICs144. Surveys conducted by the International Society of Nephrology in partnership with the Dialysis Outcomes and Practice Patterns group were aimed at understanding how clinical practice was being impacted by the pandemic, and if and how people living with kidney disease were being prioritized across the globe43,145. Challenges affecting both staff and patients were common in LLMICs. The patient level impact reported by survey respondents included challenges in access to diagnostic testing, interruptions in haemodialysis delivery, restricted access to intensive care, mechanical ventilation and in-hospital haemodialysis, affecting patients in LLMICs more frequently than those in upper-middle-income countries and HICs. Staff in dialysis units in LLMICs had less access to COVID-19 testing, PPE (Fig. 1) and training in infection control, and suffered a greater psychological impact43.
Fig. 1. Shortage of personal protective equipment across countries by income status.
Data on country income derived from World Bank data. The graphs show the results of a global online survey of haemodialysis units, which was aimed at determining patterns and access to resources associated with haemodialysis care during the COVID-19 pandemic. As shown, LICs had the greatest shortage of personal protective equipment (PPE) at the peak of the pandemic (a) and the greatest use of PPE beyond manufacturer’s shelf life (b), the latter representing a need to continue using out-of-date PPE owing to a shortage of supply. HIC, high-income countries; LIC, low-income countries; LMIC, lower-middle-income countries; UMIC, upper-middle-income countries.
At the time of the survey, conducted during the first year of the pandemic, diagnostic tests for SARS-CoV-2 were unavailable or of limited availability, with longer turnaround times for test results in the majority of LLMICs43. Patients in LICs frequently had to pay out-of-pocket for diagnostic (PCR) testing. Owing to multiple factors including lockdowns, curfews and delays awaiting COVID-19 test results, patients in LLMICs missed dialysis with a greater frequency than pre-pandemic and these delays cost lives145.
A subsequent survey focused on access to vaccination for people living with advanced kidney disease. At least one COVID-19 vaccine was available in 97% of respondent countries. Over 90% of the respondent countries reported prioritization of health care workers within the first two phases of vaccine rollout, whereas patients living with stage 4/5 CKD, dialysis, or kidney transplants were prioritized within the first two phases in 51%, 71% and 62% of countries respectively. Overall, at least 50% of patients receiving in-centre haemodialysis, peritoneal dialysis or living with a kidney transplant were reported to have completed vaccination in around half of respondent countries, with the lowest rates reported in Africa and the highest rates in Western Europe. Vaccine hesitancy, vaccine shortages and difficulties in mass distribution of vaccines were common and reported more frequently in LLMICs than in HICs. Although the vaccination rate in the dialysis population may appear relatively high in lower income settings, indicating that the vulnerability of this group has been acknowledged globally, the global disparities echo the call by the World Health Organization for more equitable access to vaccines, having set a global target of 40% of the population of every country to have completed vaccination by the end of 2021 and 70% by mid-2022. Two major global efforts, COVAX and ACT-Accelerator, where richer countries should contribute to supplying and distributing vaccines to poorer countries, have been launched to facilitate global vaccination120,121,146. These schemes, however, have not yet translated into action in terms of global solidarity, although equity gaps may be beginning to narrow147,148.
Children are an inherently vulnerable population that modern society has a duty to protect. However, the unique social status of children places them at an equally unique risk of health inequities. Current research and pharmaceutical development processes are designed to protect children by studying drugs and diseases in adults first; however, the lack of interventions and immunizations that are available in a timely manner for children, including those with underlying chronic disease during the pandemic, has raised concern for age-based health inequity that should be re-evaluated. Protection of children is paramount; however, equipoise with timely availability of emerging therapies and robust safety information are critical to this endeavour for the ethical management of COVID-19 vaccines. At a national level, the prevalence of childhood poverty, specifically its relationship to health and its disproportionality across sociodemographic groups most at risk during the pandemic, highlights another important paediatric kidney health risk119. Evidence of social deprivation along racial, ethnic and class divisions has been shown to have adverse consequences in both children and adults with kidney disease, but the specific effect of the pandemic on this population is yet to be studied149,150.
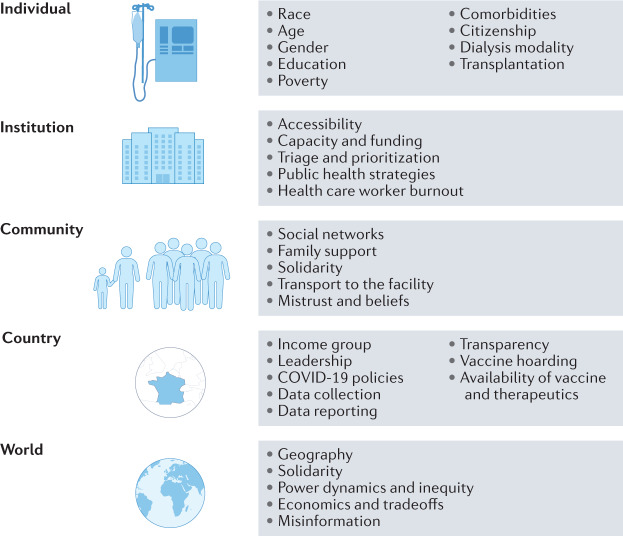
The almost miraculous rapidity with which the scientific community tackled the COVID-19 pandemic has been a simultaneous triumph and a failure. The rapid development of tests, vaccines and therapeutics has been life-saving for many, but has left many behind. The early, robust efforts to identify and publish potential management strategies led to great advances in clinical understanding and rapid knowledge sharing but also led to dissemination of pseudoscience and misinformation. This has severely impacted trust in health systems globally and has led to loss of life. The COVID-19 pandemic has revealed how necessary solidarity is at all levels, beginning with global governance and trickling down to the individual in the dialysis chair in a remote dialysis unit123,151,152 (Fig. 2). This requires attention being paid to justice and ethics at all levels. Global collaboration and cooperation are needed between countries, institutions, industry and academia. Collaborative focus on “building back better” is required such that health systems and societies emerge from the pandemic stronger, more resilient and fairer.
Fig. 2. Factors contributing to COVID-19-related health inequities.
A number of factors have resulted in COVID-19 health inequities, ranging from those that affect individuals (such as age and co-morbidities) to factors at the global level.
Trainee education
The surge of patients with COVID-19 also impacted nephrology training. Nephrology trainees have been at the front-line of care for COVID-19 patients, including in the intensive care units, dedicated COVID-19 wards and the new era of telemedicine. Although this comes with the possibility of reinvigorating interest in nephrology, it also comes with the threats of increased workload, stress and burnout for our trainees.
Activities our nephrology trainees have typically relied on for education and career advancement have undergone significant adjustments in this era. Major conferences such as the ASN Kidney Week and the European Renal Association meetings were held virtually with fewer opportunities for networking. Local conferences at one’s own institution were also shifted overnight to virtual formats, limiting in-person interaction with faculty, and requiring additional effort to stay engaged.
In the USA, adult and paediatric fellows and recent graduates of nephrology training were surveyed by the ASN Workforce and Training Committee in August–September 2020 using our Annual Fellows’ Survey instrument153. The impact of COVID-19 on fellows’ training experiences and wellbeing was measured, yielding 425 respondents (42% response rate). The majority of current fellows (84%) felt that their education was maintained during the pandemic. Fellows needed to adapt to this new landscape in real time, with up to 91% reporting adoption of telemedicine and 76% remote conferences. Although their education was maintained overall, 42% of fellows reported a negative impact of the pandemic on their overall quality of life, 33% reported a poorer work–life balance, and 15% scored as experiencing high distress measured using the Resident Well-Being Index. Similar findings were seen for trainees in the UK154, France and Belgium155.
The pandemic has offered opportunities to better understand, develop and train future nephrologists. Training programmes, such as those in Canada156, have seized this opportunity to develop personalized learning plans for postgraduate nephrology trainees. It remains to be seen whether resilience of nephrology fellows will be maintained through this pandemic, and what impact the pandemic will have on board certification, job prospects and recruitment of the next generation of nephrologists.
A patient’s voice on the pandemic
Telehealth offered a safe way to continue medical care and conferred multiple advantages, but limitations exist and must be acknowledged and addressed. A scoping review identified technical difficulties, digital illiteracy, lack of physical examination, privacy and confidentiality and loss of interpersonal interaction as the main barriers to telehealth157.
COVID-19 patient communication impacted the entire spectrum of patients with kidney disease throughout the pandemic. Since the beginning of the pandemic, COVID-19 communication with kidney disease patient communities has remained unclear. From a patient perspective, the most impactful communications centred around the use of personal protective measures, risk and consequences of AKI, vaccine efficacy and updates and guidance on the use of COVID-19 therapeutics. In the absence of direction, navigation was dependent upon the initiative of the individual patient. Town halls were conducted by the American Society of Transplantation and webinars were facilitated by the ASN, but these were accessed only by patients who were part of professional patient advocacy groups.
Patient organizations such as the American Association of Kidney Patients, National Kidney Foundation and Kidney Care UK have attempted to address the gap in COVID-19 information. Although their efforts have been successful, they have reached only a small percentage of the CKD and ESKD patients. A process is needed in which all patients hear directly from their care team about the current guidance, and what actions they can take to avoid COVID-19 infection.
Future guidance and conclusions
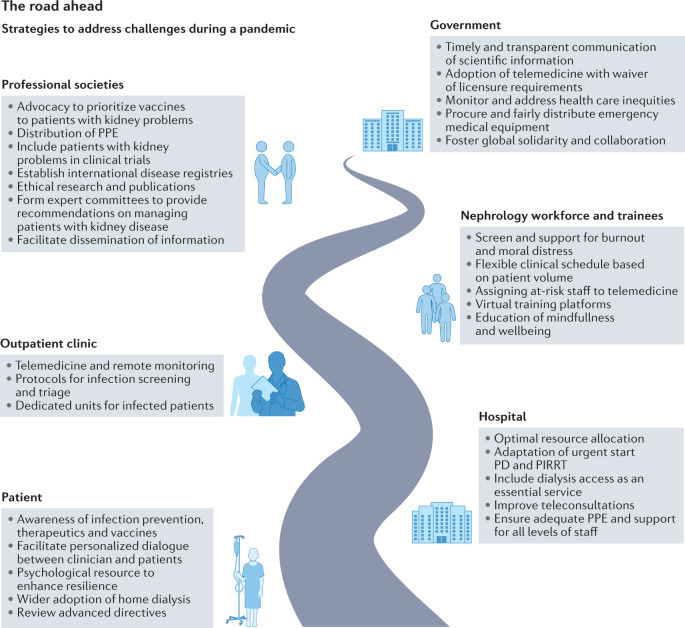
The COVID-19 pandemic has highlighted the challenges posed by surges in hospital occupancy, and has forced the digital transformation of many health care systems, underscored the need for authoritative and consistent communication from local, state and federal governments, and has identified striking global inequities in the care of patients with kidney disease. Additionally, the pandemic has highlighted the importance of proactively addressing psychosocial and ethical issues to ensure patient, caregiver and clinician wellbeing. The strategies to address the challenges faced by the kidney community during a pandemic are shown in Fig. 3. It is clear that these strategies will differ between low-, lower-middle-, upper-middle- and high-income countries, with a requirement for global collaboration and cooperation at all levels to ensure equity in health care delivery for patients with kidney disease.
Fig. 3. Strategies to address challenges during a pandemic.
PD, peritoneal dialysis; PIRRT, prolonged intermittent renal replacement therapy; PPE, personal protective equipment.
In summary, the COVID-19 pandemic has been one of the worst infectious disease crises in over a century and, given the increase in emerging diseases, pandemic threats may become a new normal. Our experience within the nephrology community has been extremely challenging. Hospitalization surges during the pandemic exposed fundamental weaknesses in all health care systems. Although this led to innovations in health care delivery and policy change, there remains an urgent need to invest in resilient health care systems and develop transparent and fair triage guidelines for scarce resources. The breakdown of global cooperation further exacerbated existing health care inequities. The pandemic illustrated that building public trust in scientific recommendations is critical to avoiding social and political divisions over following risk mitigation strategies. Moral distress has increased and must be recognized and addressed. One in 10 people is living with kidney disease. There is a need to include patients with kidney disease in clinical trials, especially during pandemics, to accelerate access to potential therapeutics, and to invest in the establishment of global collaborative disease registries to study risk factors and outcomes. Finally, we must be prepared to run a faster regulatory process during future public health emergencies. Strategic deregulation would accelerate the development and distribution of new therapeutics.
Supplementary information
Author contributions
The authors contributed equally to all aspects of the article.
Peer review
Peer review information
Nature Reviews Nephrology thanks Vivek Balkrishna Kute and the other, anonymous, reviewers for their contribution to the peer review of this work.
Competing interests
D.G. is a consultant to ChemoCentryx, Aurinia Inc and GSK, and received support from the Johns Hopkins Center for Innovative Medicine. A.K. is a consultant to Vifor, Otsuka, Delta4, Catalyst Biosciences and UriSalt, and received funding from Vifor and Otsuka. M.R. is funded by a Versus Arthritis Clinical Research Fellowship. S.A. has a grant from the National Institute of Diabetes and Digestive and Kidney Diseases (R01DK127138); Ascend Clinical Laboratory funded sample testing for S.A.’s work on SARS-CoV-2 seroepidemiology among patients on dialysis. E.L. received a consulting fee from Acumen, LLC, a federal contractor, and funding from National Institute of Diabetes and Digestive and Kidney Diseases (NIDDK) (K08 DK118213) and from the University Kidney Research Organization. V.T. is a consultant for AbbVie, Amgen, Baxter, Bayer, Boehringer Ingelheim, Calliditas, ChemoCentryx and Fresenius Medical Care, Omeros and Travere. V.T. is also a speaker for Bayer and Boehringer Ingelheim, and received travel expenses from AbbVie. D.C. was supported by grant 1 K24 HL148181 from the National Heart, Lung and Blood Institute, National Institutes of Health (NIH), and received research grant funding from Somatus Medical, Inc. and Baxter International, for unrelated work. N.C. received lecture fees Bristol Myers Squibb, and funding from The Danish Heart Foundation. V.J. received grant funding from GSK, Baxter Healthcare and Biocon, and honoraria from Bayer, AstraZeneca, Boehringer Ingelheim, NephroPlus and Zydus Cadilla, under the policy of all monies being paid to the organization. S.M. received grants from the NIH (DK126739, DK114893, DK130058, MD014161, DK116066) and the Kidney Transplant collaborative as well as personal fees from Kidney International Reports and HSAG, outside the submitted work. C.P. is a member of the advisory board of and owns equity in RenalytixAI, serves as a consultant for Genfit and Novartis, and is supported by NIH grants R01HL085757, UH3DK114866, U01DK106962 and R01DK093770. V.L. received funding from the Swiss Kidney Foundation. All other authors declare no competing interests.
Footnotes
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Related links
Dialysis COVID-19 Vaccination Data Dashboard: https://www.cdc.gov/nhsn/covid19/dial-vaccination-dashboard.html
These authors contributed equally: Duvuru Geetha, Andreas Kronbichler.
Change history
9/20/2022
A Correction to this paper has been published: 10.1038/s41581-022-00635-3
Supplementary information
The online version contains supplementary material available at 10.1038/s41581-022-00618-4.
References
- 1.Chan L, et al. AKI in hospitalized patients with COVID-19. J. Am. Soc. Nephrol. 2021;32:151–160. doi: 10.1681/ASN.2020050615. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Brogan M, Ross MJ. The impact of chronic kidney disease on outcomes of patients with COVID-19 admitted to the intensive care unit. Nephron. 2022;146:67–71. doi: 10.1159/000519530. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Pereira MR, et al. COVID-19 in solid organ transplant recipients: initial report from the US epicenter. Am. J. Transpl. 2020;20:1800–1808. doi: 10.1111/ajt.15941. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Singhala M, et al. Emergency production and collection of dialysate for CVVHD during the COVID-19 pandemic. Kidney Int. Rep. 2021;6:2200–2202. doi: 10.1016/j.ekir.2021.05.026. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Combe C, et al. At least 156 reasons to prioritize COVID-19 vaccination in patients receiving in-centre haemodialysis. Nephrol. Dial. Transpl. 2021;36:571–574. doi: 10.1093/ndt/gfab007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Hilton J, Boyer N, Nadim MK, Forni LG, Kellum JA. COVID-19 and acute kidney injury. Crit. Care Clin. 2022;38:473–489. doi: 10.1016/j.ccc.2022.01.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Hirsch JS, et al. Acute kidney injury in patients hospitalized with COVID-19. Kidney Int. 2020;98:209–218. doi: 10.1016/j.kint.2020.05.006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Charytan DM, et al. Decreasing incidence of acute kidney injury in patients with COVID-19 critical illness in New York City. Kidney Int. Rep. 2021;6:916–927. doi: 10.1016/j.ekir.2021.01.036. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Dellepiane S, et al. Acute kidney injury in patients hospitalized with COVID-19 in New York City: temporal trends from March 2020 to April 2021. Kidney Med. 2021;3:877–879. doi: 10.1016/j.xkme.2021.06.008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Tannor EK. Challenges in kidney care in a lower middle income country during the COVID-19 pandemic — the Ghanaian perspective. Kidney Int. Rep. 2021;6:2014–2016. doi: 10.1016/j.ekir.2021.06.019. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Diane NE, et al. Acute kidney injury during the COVID-19 pandemic–experience from two tertiary centres in South Africa. 2020. Wits J. Clin. Med. 2020;2:189–198. [Google Scholar]
- 12.May RM, et al. A multi-center retrospective cohort study defines the spectrum of kidney pathology in Coronavirus 2019 Disease (COVID-19) Kidney Int. 2021;100:1303–1315. doi: 10.1016/j.kint.2021.07.015. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Wu H, et al. AKI and collapsing glomerulopathy associated with COVID-19 and APOL 1 high-risk genotype. J. Am. Soc. Nephrol. 2020;31:1688. doi: 10.1681/ASN.2020050558. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Hung AM, et al. APOL1 risk variants, acute kidney injury, and death in participants with African ancestry hospitalized with COVID-19 from the million veteran program. JAMA Intern. Med. 2022 doi: 10.1001/jamainternmed.2021.8538. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Strohbehn IA, et al. Acute kidney injury incidence, recovery, and long-term kidney outcomes among hospitalized patients with COVID-19 and influenza. Kidney Int. Rep. 2021;6:2565–2574. doi: 10.1016/j.ekir.2021.07.008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Lumlertgul N, et al. Acute kidney injury prevalence, progression and long-term outcomes in critically ill patients with COVID-19: a cohort study. Ann. Intensive Care. 2021;11:123. doi: 10.1186/s13613-021-00914-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Gupta S, et al. AKI treated with renal replacement therapy in critically ill patients with COVID-19. J. Am. Soc. Nephrol. 2021;32:161–176. doi: 10.1681/ASN.2020060897. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Yende S, Parikh CR. Long COVID and kidney disease. Nat. Rev. Nephrol. 2021;17:792–793. doi: 10.1038/s41581-021-00487-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Bowe B, Xie Y, Xu E, Al-Aly Z. Kidney outcomes in long COVID. J. Am. Soc. Nephrol. 2021;32:2851–2862. doi: 10.1681/ASN.2021060734. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Sun S, et al. Short- and long-term recovery after moderate/severe AKI in patients with and without COVID-19. Kidney360. 2022;3:242–257. doi: 10.34067/KID.0005342021. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Dashtban A, et al. A retrospective cohort study measured predicting and validating the impact of the COVID-19 pandemic in individuals with chronic kidney disease. Kidney Int. 2022 doi: 10.1016/j.kint.2022.05.015. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Uribarri A, et al. Impact of renal function on admission in COVID-19 patients: an analysis of the international HOPE COVID-19 (Health Outcome Predictive Evaluation for COVID 19) registry. J. Nephrol. 2020;33:737–745. doi: 10.1007/s40620-020-00790-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Portoles J, et al. Chronic kidney disease and acute kidney injury in the COVID-19 Spanish outbreak. Nephrol. Dial. Transpl. 2020;35:1353–1361. doi: 10.1093/ndt/gfaa189. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Carlson N, et al. Increased vulnerability to COVID-19 in chronic kidney disease. J. Intern. Med. 2021;290:166–178. doi: 10.1111/joim.13239. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Hakroush S, Tampe D, Korsten P, Tampe B. Impact of the COVID-19 pandemic on kidney diseases requiring renal biopsy: a single center observational study. Front. Physiol. 2021;12:649336. doi: 10.3389/fphys.2021.649336. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Seet C, et al. The management of dialysis access thrombosis during the COVID-19 pandemic. J. Vasc. Access. 2021 doi: 10.1177/11297298211045578. [DOI] [PubMed] [Google Scholar]
- 27.Wetmore JB, et al. Changes in treatment of patients with incident ESKD during the novel coronavirus disease 2019 pandemic. J. Am. Soc. Nephrol. 2021;32:2948–2957. doi: 10.1681/ASN.2021040579. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Truong T, Dittmar M, Ghaffari A, Lin E. Policy and pandemic: the changing practice of nephrology during the coronavirus disease-2019 outbreak. Adv. Chronic Kidney Dis. 2020;27:390–396. doi: 10.1053/j.ackd.2020.06.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Chen G, et al. When the COVID-19 pandemic changed the follow-up landscape of chronic kidney disease: a survey of real-world nephrology practice. Ren. Fail. 2020;42:733–739. doi: 10.1080/0886022X.2020.1798783. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Ramachandran R, Jha V. Adding insult to injury: kidney replacement therapy during COVID-19 in India. Kidney Int. 2020;98:238–239. doi: 10.1016/j.kint.2020.04.019. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Group RC, et al. Dexamethasone in hospitalized patients with COVID-19. N. Engl. J. Med. 2021;384:693–704. doi: 10.1056/NEJMoa2021436. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Adamsick ML, et al. Remdesivir in patients with acute or chronic kidney disease and COVID-19. J. Am. Soc. Nephrol. 2020;31:1384–1386. doi: 10.1681/ASN.2020050589. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Salvarani C, et al. Effect of tocilizumab vs standard care on clinical worsening in patients hospitalized with COVID-19 pneumonia: a randomized clinical trial. JAMA Intern. Med. 2021;181:24–31. doi: 10.1001/jamainternmed.2020.6615. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Hou YC, Lu KC, Kuo KL. The efficacy of COVID-19 vaccines in chronic kidney disease and kidney transplantation patients: a narrative review. Vaccines. 2021 doi: 10.3390/vaccines9080885. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Quiroga B, et al. Humoral response to third dose of SARS-CoV-2 vaccines in the CKD spectrum. Clin. J. Am. Soc. Nephrol. 2022;17:872–876. doi: 10.2215/CJN.01770222. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Salas A, et al. ANCA vasculitis induction management during the COVID-19 pandemic. Kidney Int. Rep. 2021;6:2903–2907. doi: 10.1016/j.ekir.2021.08.009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Waldman M, et al. Results from the IRoc-GN international registry of patients with COVID-19 and glomerular disease suggest close monitoring. Kidney Int. 2021;99:227–237. doi: 10.1016/j.kint.2020.10.032. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Strangfeld A, et al. Factors associated with COVID-19-related death in people with rheumatic diseases: results from the COVID-19 global rheumatology alliance physician-reported registry. Ann. Rheum. Dis. 2021;80:930–942. doi: 10.1136/annrheumdis-2020-219498. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Prendecki M, Willicombe M, McAdoo SP. COVID-19 vaccination in patients with immunity-mediated kidney disease. Nat. Rev. Nephrol. 2021;17:790–791. doi: 10.1038/s41581-021-00502-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Connolly CM, et al. Temporary hold of mycophenolate augments humoral response to SARS-CoV-2 vaccination in patients with rheumatic and musculoskeletal diseases: a case series. Ann. Rheum. Dis. 2022;81:293–295. doi: 10.1136/annrheumdis-2021-221252. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Kronbichler A, Jung SY, Kim MS, Shin JI. Distinct glomerular disease association after vaccination with BNT162b2 and mRNA-1273: a VigiBase analysis. Kidney Int. 2022;101:415–416. doi: 10.1016/j.kint.2021.11.013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Oliver MJ, Crabtree JH. Prioritizing peritoneal catheter placement during the COVID-19 pandemic: a perspective of the American Society of Nephrology COVID-19 Home Dialysis Subcommittee. Clin. J. Am. Soc. Nephrol. 2021;16:1281–1283. doi: 10.2215/CJN.19141220. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Aylward R, et al. The global impact of the COVID-19 pandemic on in-center hemodialysis services: an ISN-dialysis outcomes practice patterns study survey. Kidney Int. Rep. 2022;7:397–409. doi: 10.1016/j.ekir.2021.12.011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Corbett RW, et al. Epidemiology of COVID-19 in an urban dialysis center. J. Am. Soc. Nephrol. 2020;31:1815–1823. doi: 10.1681/ASN.2020040534. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Alfano G, et al. The frail world of haemodialysis patients in the COVID-19 pandemic era: a systematic scoping review. J. Nephrol. 2021;34:1387–1403. doi: 10.1007/s40620-021-01136-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Ziemba R, et al. Excess death estimates in patients with end-stage renal disease — United States, February–August 2020. MMWR Morb. Mortal. Wkly. Rep. 2021;70:825–829. doi: 10.15585/mmwr.mm7022e2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Vart P, et al. COVID-19 pandemic waves and mortality among patients on kidney replacement therapy. Kidney Int. Rep. 2022 doi: 10.1016/j.ekir.2022.06.007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Patel PR, et al. Assessment of provision of COVID-19 vaccination in dialysis clinics and patient vaccination coverage. JAMA Intern. Med. 2022;182:676–678. doi: 10.1001/jamainternmed.2022.0627. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Anand Shuchi, et al. Antibody response to COVID-19 vaccination in patients receiving dialysis. J. Am. Soc. Nephrol. 2021;32:2435–2438. doi: 10.1681/ASN.2021050611. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Brunelli SM, et al. Comparative effectiveness of mRNA-based BNT162b2 vaccine versus adenovirus vector-based Ad26.COV2.S vaccine for the prevention of COVID-19 among dialysis patients. J. Am. Soc. Nephrol. 2022 doi: 10.1681/ASN.2021101395. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Garcia P, et al. COVID-19 vaccine type and humoral immune response in patients receiving dialysis. J. Am. Soc. Nephrol. 2022;33:33–37. doi: 10.1681/ASN.2021070936. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.CMS. Medicare Program; end-stage renal disease prospective payment system, payment for renal dialysis services furnished to individuals with acute kidney injury, and end-stage renal disease quality incentive program. Fed. Register. 2020;85:42132–42208. [PubMed] [Google Scholar]
- 53.CMS, HHS. Medicare Program; specialty care models to improve quality of care and reduce expenditures. Fed. Register. 2020;85:61114–61381. [Google Scholar]
- 54.CMS. COVID-19 Emergency Declaration Blanket Waivers for Health Care Providers. https://www.cms.gov/files/document/summary-covid-19-emergency-declaration-waivers.pdf (2021).
- 55.Rombola G, Brunini F. COVID-19 and dialysis: why we should be worried. J. Nephrol. 2020;33:401–403. doi: 10.1007/s40620-020-00737-w. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Lew SQ, Sikka N. Operationalizing telehealth for home dialysis patients in the United States. Am. J. Kidney Dis. 2019;74:95–100. doi: 10.1053/j.ajkd.2019.01.023. [DOI] [PubMed] [Google Scholar]
- 57.Mahase E. COVID-19: Pfizer’s paxlovid is 89% effective in patients at risk of serious illness, company reports. BMJ. 2021;375:n2713. doi: 10.1136/bmj.n2713. [DOI] [PubMed] [Google Scholar]
- 58.Wen W, et al. Efficacy and safety of three new oral antiviral treatment (molnupiravir, fluvoxamine and paxlovid) for COVID-19: a meta-analysis. Ann. Med. 2022;54:516–523. doi: 10.1080/07853890.2022.2034936. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59.Schold JD, et al. COVID-19 mortality among kidney transplant candidates is strongly associated with social determinants of health. Am. J. Transpl. 2021;21:2563–2572. doi: 10.1111/ajt.16578. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Mohan S, King KL, Husain SA, Schold JD. COVID-19-associated mortality among kidney transplant recipients and candidates in the United States. Clin. J. Am. Soc. Nephrol. 2021;16:1695–1703. doi: 10.2215/CJN.02690221. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.Alberici F, et al. SARS-CoV-2 infection in dialysis and kidney transplant patients: immunological and serological response. J. Nephrol. 2022 doi: 10.1007/s40620-021-01214-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62.Aubert O, et al. COVID-19 pandemic and worldwide organ transplantation: a population-based study. Lancet Public Health. 2021;6:e709–e719. doi: 10.1016/S2468-2667(21)00200-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63.Lentine KL, et al. Survey of US living kidney donation and transplantation practices in the COVID-19 era. Kidney Int. Rep. 2020;5:1894–1905. doi: 10.1016/j.ekir.2020.08.017. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64.Li MT, King KL, Husain SA, Schold JD, Mohan S. Deceased donor kidneys utilization and discard rates during COVID-19 pandemic in the United States. Kidney Int. Rep. 2021;6:2463–2467. doi: 10.1016/j.ekir.2021.06.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 65.Martinez-Reviejo R, et al. Solid organ transplantation from donors with recent or current SARS-CoV-2 infection: a systematic review. Anaesth. Crit. Care Pain. Med. 2022 doi: 10.1016/j.accpm.2022.101098. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 66.Chang JH, et al. Home care delivery and remote patient monitoring of kidney transplant recipients during COVID-19 pandemic. Prog. Transpl. 2021;31:381–384. doi: 10.1177/15269248211046005. [DOI] [PubMed] [Google Scholar]
- 67.Biancone L, et al. Telemedicine monitoring in the follow-up of kidney transplant recipients: consensus indications from an Italian panel of surgeons and nephrologists after the COVID-19 experience. J. Nephrol. 2022 doi: 10.1007/s40620-021-01193-w. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 68.Potter SR, Hinojosa R, Miles CD, O’Brien D, Ross DJ. Remote monitoring using donor-derived, cell-free DNA after kidney transplantation during the coronavirus disease 2019 pandemic. Kidney Res. Clin. Pract. 2020;39:495–500. doi: 10.23876/j.krcp.20.107. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 69.Masset C, et al. Occurrence of de novo donor specific antibodies after COVID-19 in kidney transplant recipients is low despite immunosuppression modulation. Kidney Int. Rep. 2022 doi: 10.1016/j.ekir.2022.01.1072. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 70.Abu Jawdeh BG. COVID-19 in kidney transplantation: outcomes, immunosuppression management, and operational challenges. Adv. Chronic Kidney Dis. 2020;27:383–389. doi: 10.1053/j.ackd.2020.07.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 71.Meena Priti, Crew RJohn. Understanding the risks of immunosuppression reduction for active COVID-19 infection. Kidney Int. Rep. 2022;7:937–938. doi: 10.1016/j.ekir.2022.03.005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 72.Salerno DM, et al. SARS-CoV-2 infection increases tacrolimus concentrations in solid-organ transplant recipients. Clin. Transpl. 2021;35:e14193. doi: 10.1111/ctr.14193. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 73.Watcharananan SP, et al. Comparison of the immunogenicity of ChAdOx1 nCoV-19 vaccine against the wild-type and delta variants in kidney transplant recipients and healthy volunteers. Am. J. Transpl. 2022 doi: 10.1111/ajt.16966. [DOI] [PubMed] [Google Scholar]
- 74.Boyarsky BJ, et al. Antibody response to 2-dose SARS-CoV-2 mRNA vaccine series in solid organ transplant recipients. JAMA. 2021;325:2204–2206. doi: 10.1001/jama.2021.7489. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 75.Masset C, et al. A third injection of the BNT162b2 mRNA COVID-19 vaccine in kidney transplant recipients improves the humoral immune response. Kidney Int. 2021;100:1132–1135. doi: 10.1016/j.kint.2021.08.017. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 76.Masset C, et al. Observations on improving COVID-19 vaccination responses in kidney transplant recipients: heterologous vaccination and immunosuppression modulation. Kidney Int. 2022;101:642–645. doi: 10.1016/j.kint.2021.11.024. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 77.Cristelli MP, Nakamura MR, Viana LA, Tedesco-Silva H, Medina-Pestana J. The fourth dose of coronavac vaccine results in a small increase of seroconversion and antibody values among kidney transplant recipients. Transplantation. 2022 doi: 10.1097/TP.0000000000004219. [DOI] [PubMed] [Google Scholar]
- 78.Charnaya O, et al. Effects of COVID-19 pandemic on pediatric kidney transplant in the United States. Pediatr. Nephrol. 2021;36:143–151. doi: 10.1007/s00467-020-04764-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 79.Marlais M, et al. COVID-19 in children treated with immunosuppressive medication for kidney diseases. Arch. Dis. Child. 2020 doi: 10.1136/archdischild-2020-320616. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 80.Varnell CD, et al. COVID-19 in pediatric kidney transplantation: the improving renal outcomes collaborative. Am. J. Transpl. 2021;21:2740–2748. doi: 10.1111/ajt.16501. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 81.Canpolat N, et al. COVID-19 in pediatric patients undergoing chronic dialysis and kidney transplantation. Eur. J. Pediatr. 2022;181:117–123. doi: 10.1007/s00431-021-04191-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 82.Mastrangelo A, et al. Impact of COVID-19 pandemic in children with CKD or immunosuppression. Clin. J. Am. Soc. Nephrol. 2021;16:449–451. doi: 10.2215/CJN.13120820. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 83.Basalely A, et al. Acute kidney injury in pediatric patients hospitalized with acute COVID-19 and multisystem inflammatory syndrome in children associated with COVID-19. Kidney Int. 2021;100:138–145. doi: 10.1016/j.kint.2021.02.026. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 84.Kari JA, et al. Acute kidney injury in children with COVID-19: a retrospective study. BMC Nephrol. 2021;22:202. doi: 10.1186/s12882-021-02389-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 85.Chopra S, et al. Acute kidney injury in hospitalized children with COVID19. J. Trop. Pediatr. 2021 doi: 10.1093/tropej/fmab037. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 86.Wang X, et al. Be aware of acute kidney injury in critically ill children with COVID-19. Pediatr. Nephrol. 2021;36:163–169. doi: 10.1007/s00467-020-04715-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 87.Kaddourah A, Basu RK, Bagshaw SM, Goldstein SL, AWARE Investigators. Epidemiology of acute kidney injury in critically ill children and young adults. N. Engl. J. Med. 2017;376:11–20. doi: 10.1056/NEJMoa1611391. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 88.No authors listed. Long COVID and kids: more research is urgently needed. Nature10.1038/d41586-022-00334-w (2022). [DOI] [PubMed]
- 89.Yilmaz M, Sahin MK. Parents’ willingness and attitudes concerning the COVID-19 vaccine: a cross-sectional study. Int. J. Clin. Pract. 2021;75:e14364. doi: 10.1111/ijcp.14364. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 90.Tse Y, et al. COVID-19: experiences of lockdown and support needs in children and young adults with kidney conditions. Pediatr. Nephrol. 2021;36:2797–2810. doi: 10.1007/s00467-021-05041-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 91.Acemoglu, D., Chernozhukov, V., Werning, I. & Whinston, M. D. A Multi-Risk SIR Model with Optimally Targeted Lockdown. Vol. 2020 (National Bureau of Economic Research Cambridge, 2020).
- 92.Neufeld Z, Khataee H, Czirok A. Targeted adaptive isolation strategy for COVID-19 pandemic. Infect. Dis. Model. 2020;5:357–361. doi: 10.1016/j.idm.2020.04.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 93.Rutter M, et al. Estimation of the burden of shielding among a cross-section of patients attending rheumatology clinics with SLE — data from the BSR audit of systemic lupus erythematosus. Rheumatology. 2020;60:1474–1479. doi: 10.1093/rheumatology/keaa620. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 94.Sloan M, et al. COVID-19 and shielding: experiences of UK patients with lupus and related diseases. Rheumatol. Adv. Pract. 2021 doi: 10.1093/rap/rkab003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 95.Qian M, et al. Anxiety levels, precautionary behaviours and public perceptions during the early phase of the COVID-19 outbreak in China: a population-based cross-sectional survey. BMJ Open. 2020;10:e040910. doi: 10.1136/bmjopen-2020-040910. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 96.Yıldırım M, Geçer E, Akgül Ö. The impacts of vulnerability, perceived risk, and fear on preventive behaviours against COVID-19. Psychol. Health Med. 2021;26:35–43. doi: 10.1080/13548506.2020.1776891. [DOI] [PubMed] [Google Scholar]
- 97.Bavel JJV, et al. Using social and behavioural science to support COVID-19 pandemic response. Nat. Hum. Behav. 2020;4:460–471. doi: 10.1038/s41562-020-0884-z. [DOI] [PubMed] [Google Scholar]
- 98.Lanphier, E., Mosley, L. & Antommaria, A. H. M. Assessing visitor policy exemption requests during the COVID-19 pandemic. Pediatrics148, e2021051254 (2021). [DOI] [PubMed]
- 99.Downar J, Kekewich M. Improving family access to dying patients during the COVID-19 pandemic. Lancet Respir. Med. 2021;9:335–337. doi: 10.1016/S2213-2600(21)00025-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 100.Nogueira GM, Oliveira MS, Moura AF, Cruz CMS, Moura-Neto JA. COVID-19 in dialysis units: a comprehensive review. World J. Virol. 2021;10:264. doi: 10.5501/wjv.v10.i5.264. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 101.Janssen DJ. Palliative care in COVID-19. Curr. Opin. Support. Palliat. Care. 2021;15:199. doi: 10.1097/SPC.0000000000000567. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 102.Hugelius K, Harada N, Marutani M. Consequences of visiting restrictions during the COVID‐19 pandemic: an integrative review. Int. J. Nurs. Stud. 2021;121:104000. doi: 10.1016/j.ijnurstu.2021.104000. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 103.Vittone, S. & Sotomayor, C. R. in HEC forum. Vol. 33, 415–423 (Springer, 2021). [DOI] [PMC free article] [PubMed]
- 104.Ducharlet K, et al. Moral distress and moral injury in nephrology during the COVID-19 pandemic. Semin. Nephrol. 2021;41:253–261. doi: 10.1016/j.semnephrol.2021.05.006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 105.Sloan M, et al. Will ‘the feeling of abandonment’ remain? Persisting impacts of the COVID-19 pandemic on rheumatology patients and clinicians. Rheumatology. 2022 doi: 10.1093/rheumatology/keab937. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 106.Shaw SC. Hopelessness, helplessness and resilience: the importance of safeguarding our trainees’ mental wellbeing during the COVID-19 pandemic. Nurse Educ. Pract. 2020;44:102780. doi: 10.1016/j.nepr.2020.102780. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 107.Ripp J, Peccoralo L, Charney D. Attending to the emotional well-being of the health care workforce in a New York City health system during the COVID-19 pandemic. Acad. Med. 2020;95:1136–1139. doi: 10.1097/ACM.0000000000003414. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 108.Morley, G., Sese, D., Rajendram, P. & Horsburgh, C. C. Addressing caregiver moral distress during the COVID-19 pandemic. Cleve. Clin. J. Med. 10.3949/ccjm.87a.ccc047 (2020). [DOI] [PubMed]
- 109.Mc Keaveney C, et al. Experiences of renal healthcare practitioners during the COVID-19 pandemic: a multi-methods approach. BMC Nephrol. 2021;22:1–10. doi: 10.1186/s12882-021-02500-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 110.Jameton, A. Nursing Practice: The Ethical Issues (Prentice-Hall, 1984).
- 111.Flood D, et al. Challenges in the provision of kidney care at the largest public nephrology center in Guatemala: a qualitative study with health professionals. BMC Nephrol. 2020;21:1–10. doi: 10.1186/s12882-020-01732-w. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 112.Cervantes L, et al. Clinicians’ perspectives on providing emergency-only hemodialysis to undocumented immigrants: a qualitative study. Ann. Intern. Med. 2018 doi: 10.7326/M18-0400. [DOI] [PubMed] [Google Scholar]
- 113.Lamiani G, Borghi L, Argentero P. When healthcare professionals cannot do the right thing: a systematic review of moral distress and its correlates. J. Health Psychol. 2017;22:51–67. doi: 10.1177/1359105315595120. [DOI] [PubMed] [Google Scholar]
- 114.Myran DT, et al. Physician health care visits for mental health and substance use during the COVID-19 pandemic in Ontario, Canada. JAMA Netw. Open. 2022;5:e2143160. doi: 10.1001/jamanetworkopen.2021.43160. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 115.Litz BT, et al. Moral injury and moral repair in war veterans: a preliminary model and intervention strategy. Clin. Psychol. Rev. 2009;29:695–706. doi: 10.1016/j.cpr.2009.07.003. [DOI] [PubMed] [Google Scholar]
- 116.Nobari H, et al. Effect of COVID-19 on health-related quality of life in adolescents and children: a systematic review. Int. J. Environ. Res. Public Health. 2021 doi: 10.3390/ijerph18094563. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 117.Sharma R, et al. Distress, anxiety, and its correlates among caregivers of children with kidney diseases during COVID-19 pandemic lockdown. Arch. Pediatr. 2022 doi: 10.1016/j.arcped.2022.01.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 118.Alessi J, de Oliveira GB, Feiden G, Schaan BD, Telo GH. Caring for caregivers: the impact of the COVID-19 pandemic on those responsible for children and adolescents with type 1 diabetes. Sci. Rep. 2021;11:6812. doi: 10.1038/s41598-021-85874-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 119.Smitherman LC, Golden WC, Walton JR. Health disparities and their effects on children and their caregivers during the coronavirus disease 2019 pandemic. Pediatr. Clin. North. Am. 2021;68:1133–1145. doi: 10.1016/j.pcl.2021.05.013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 120.Moon S, et al. Governing the access to COVID-19 tools accelerator: towards greater participation, transparency, and accountability. Lancet. 2022;399:487–494. doi: 10.1016/S0140-6736(21)02344-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 121.Herzog LM, Norheim OF, Emanuel EJ, McCoy MS. Covax must go beyond proportional allocation of covid vaccines to ensure fair and equitable access. BMJ. 2021;372:m4853. doi: 10.1136/bmj.m4853. [DOI] [PubMed] [Google Scholar]
- 122.McIntyre PB, et al. COVID-19 vaccine strategies must focus on severe disease and global equity. Lancet. 2022;399:406–410. doi: 10.1016/S0140-6736(21)02835-X. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 123.Binagwaho A, Mathewos K, Davis S. Time for the ethical management of COVID-19 vaccines. Lancet Glob. Health. 2021;9:e1169–e1171. doi: 10.1016/S2214-109X(21)00180-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 124.Patel MS, Phillips CB. COVID-19 and the moral imagination. Lancet. 2021;397:648–650. doi: 10.1016/S0140-6736(21)00151-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 125.Ismail SJ, Tunis MC, Zhao L, Quach C. Navigating inequities: a roadmap out of the pandemic. BMJ Glob. Health. 2021 doi: 10.1136/bmjgh-2020-004087. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 126.Bignall ONR, 2nd, Crews DC. Stony the road we trod: towards racial justice in kidney care. Nat. Rev. Nephrol. 2021;17:79–80. doi: 10.1038/s41581-020-00389-w. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 127.Shadmi E, et al. Health equity and COVID-19: global perspectives. Int. J. Equity Health. 2020;19:104. doi: 10.1186/s12939-020-01218-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 128.Aldridge RW, et al. Black, Asian and minority ethnic groups in England are at increased risk of death from COVID-19: indirect standardisation of NHS mortality data. Wellcome Open Res. 2020;5:88. doi: 10.12688/wellcomeopenres.15922.2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 129.Baqui P, Bica I, Marra V, Ercole A, van der Schaar M. Ethnic and regional variations in hospital mortality from COVID-19 in Brazil: a cross-sectional observational study. Lancet Glob. Health. 2020;8:e1018–e1026. doi: 10.1016/S2214-109X(20)30285-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 130.Laster Pirtle WN. Racial capitalism: a fundamental cause of novel coronavirus (COVID-19) pandemic inequities in the United States. Health Educ. Behav. 2020;47:504–508. doi: 10.1177/1090198120922942. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 131.Yaya S, Yeboah H, Charles CH, Otu A, Labonte R. Ethnic and racial disparities in COVID-19-related deaths: counting the trees, hiding the forest. BMJ Glob. Health. 2020 doi: 10.1136/bmjgh-2020-002913. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 132.Council E-E, Group EW. Chronic kidney disease is a key risk factor for severe COVID-19: a call to action by the ERA-EDTA. Nephrol. Dial. Transpl. 2021;36:87–94. doi: 10.1093/ndt/gfaa314. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 133.WHO NCD Department. COVID-19 AND NCDS, https://www.google.com/url?sa=t&rct=j&q=&esrc=s&source=web&cd=&ved=2ahUKEwiMjPyu7ovrAhVt-yoKHU2MBi0QFjAIegQICBAB&url=https%3A%2F%2Fwww.who.int%2Fdocs%2Fdefault-source%2Fncds%2Fncd-covid-19%2Ffor-web---rapid-assessment---29-may-2020-(cleared).pdf%3Fsfvrsn%3D6296324c_8%26download%3Dtrue&usg=AOvVaw2fh7BVQyYEc4eozvMEaZxT (2020).
- 134.Luyckx VA. Equity is key to build back better after COVID-19: prioritize noncommunicable diseases and kidney health. Kidney360. 2021;2:747–750. doi: 10.34067/KID.0006932020. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 135.Vergano M, et al. SIAARTI recommendations for the allocation of intensive care treatments in exceptional, resource-limited circumstances. Minerva Anestesiol. 2020;86:469–472. doi: 10.23736/S0375-9393.20.14619-4. [DOI] [PubMed] [Google Scholar]
- 136.Jobges S, Vinay R, Luyckx VA, Biller-Andorno N. Recommendations on COVID-19 triage: international comparison and ethical analysis. Bioethics. 2020;34:948–959. doi: 10.1111/bioe.12805. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 137.Moosa MR, Luyckx VA. The realities of rationing in health care. Nat. Rev. Nephrol. 2021;17:435–436. doi: 10.1038/s41581-021-00404-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 138.Robert R, et al. Ethical dilemmas due to the COVID-19 pandemic. Ann. Intensive Care. 2020;10:84. doi: 10.1186/s13613-020-00702-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 139.White DB, Lo B. A framework for rationing ventilators and critical care beds during the COVID-19 pandemic. JAMA. 2020 doi: 10.1001/jama.2020.5046. [DOI] [PubMed] [Google Scholar]
- 140.Butler CR, Wong SPY, Wightman AG, O’Hare AM. US clinicians’ experiences and perspectives on resource limitation and patient care during the COVID-19 pandemic. JAMA Netw. Open. 2020;3:e2027315. doi: 10.1001/jamanetworkopen.2020.27315. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 141.Stevens JS, et al. Dashboards to facilitate nephrology disaster planning in the COVID-19 era. Kidney Int. Rep. 2020;5:1298–1302. doi: 10.1016/j.ekir.2020.06.033. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 142.Norton JM, et al. Social determinants of racial disparities in CKD. J. Am. Soc. Nephrol. 2016;27:2576–2595. doi: 10.1681/ASN.2016010027. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 143.Adapa S, et al. COVID-19 and renal failure: challenges in the delivery of renal replacement therapy. J. Clin. Med. Res. 2020;12:276. doi: 10.14740/jocmr4160. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 144.Hopman J, Allegranzi B, Mehtar S. Managing COVID-19 in low-and middle-income countries. JAMA. 2020;323:1549–1550. doi: 10.1001/jama.2020.4169. [DOI] [PubMed] [Google Scholar]
- 145.Tannor EK, et al. The COVID-19 pandemic identifies significant global inequities in hemodialysis care in low and lower middle-income countries — an ISN/DOPPS survey. Kidney Int. Rep. 2022 doi: 10.1016/j.ekir.2022.02.027. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 146.Storeng KT, de Bengy Puyvallee A, Stein F. COVAX and the rise of the ‘super public private partnership’ for global health. Glob. Public Health. 2021 doi: 10.1080/17441692.2021.1987502. [DOI] [PubMed] [Google Scholar]
- 147.Storeng KT, Stein F, de Bengy Puyvallee A. COVAX and the many meanings of sharing. BMJ Glob. Health. 2021 doi: 10.1136/bmjgh-2021-007763. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 148.Binagwaho A, Mathewos K, Davis S. Equitable and effective distribution of the COVID-19 vaccines — a scientific and moral obligation. Int. J. Health Policy Manag. 2021 doi: 10.34172/ijhpm.2021.49. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 149.Plumb LA, et al. Associations between deprivation, geographic location, and access to pediatric kidney care in the United Kingdom. Clin. J. Am. Soc. Nephrol. 2021;16:194–203. doi: 10.2215/CJN.11020720. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 150.Goldberg AM, Bignall ONR., 2nd Mind the gap: acknowledging deprivation is key to narrowing kidney health disparities in both children and adults. Clin. J. Am. Soc. Nephrol. 2021;16:185–187. doi: 10.2215/CJN.19321220. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 151.Alfano G, et al. Ethical challenges in managing unvaccinated patients receiving chronic in-centre haemodialysis. Clin. Kidney J. 2021 doi: 10.1093/ckj/sfab276. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 152.Taylor AL, et al. Solidarity in the wake of COVID-19: reimagining the International Health Regulations. Lancet. 2020;396:82–83. doi: 10.1016/S0140-6736(20)31417-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 153.Pivert KA, et al. Impact of the COVID-19 pandemic on nephrology fellow training and well-being in the United States: a National Survey. J. Am. Soc. Nephrol. 2021;32:1236–1248. doi: 10.1681/ASN.2020111636. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 154.Selvaskandan H, et al. Burnout and long COVID among the UK nephrology workforce: results from a national survey investigating the impact of COVID-19 on working lives. Clin. Kidney J. 2022;15:517–526. doi: 10.1093/ckj/sfab264. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 155.Maisons V, et al. Impact of the COVID-19 pandemic on training and well-being of nephrology residents in France and Belgium. Kidney Int. Rep. 2022;7:653–655. doi: 10.1016/j.ekir.2021.12.035. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 156.Cunningham A, Hung W, Levin A, Jamal A. Adapting nephrology training curriculum in the era of COVID-19. Can. J. Kidney Health Dis. 2021;8:2054358120988446. doi: 10.1177/2054358120988446. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 157.Ewart C, Baharani J, Wilkie M, Thomas N. Patient perspectives and experiences of remote consultations in people receiving kidney care: a scoping review. J. Ren. Care. 2022 doi: 10.1111/jorc.12419. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.