Abstract
Background
Anemia is common in patients with rheumatoid arthritis (RA). Higher hemoglobin (Hb) levels may be associated with better clinical outcomes and patient-reported outcomes (PROs). To assess this hypothesis, we conducted two post hoc analyses in three sarilumab phase III studies: TARGET, MOBILITY, and MONARCH.
Methods
Pooled data from combination therapy from placebo-controlled MOBILITY (sarilumab + methotrexate) and TARGET (sarilumab + conventional synthetic disease-modifying antirheumatic drugs [csDMARDs]) and monotherapy data from active-controlled MONARCH (sarilumab vs. adalimumab) studies were included. Associations between Hb levels and clinical measures and PROs were assessed over 24 weeks. The mean changes from baseline in clinical outcomes and PROs (to week 24) and radiographic outcomes (to week 52) were evaluated between low and normal Hb levels (based on the World Health Organization [WHO] criteria).
Results
From TARGET, MOBILITY, and MONARCH, 546, 1197, and 369 patients, respectively, were stratified according to Hb levels (low vs. normal). Over 24 weeks, higher Hb levels were found to be consistently associated with better clinical outcomes and PROs in combination therapy and monotherapy groups and were more pronounced among the patients treated with sarilumab than those treated with placebo and adalimumab. The mean change from baseline to week 24 in clinical efficacy measures and PROs was similar in patients with low vs. normal Hb at baseline. Differences between sarilumab and/or adalimumab, for all outcomes, were larger for low Hb subgroups. In MOBILITY, by week 52, the inhibition of progression of structural damage (assessed via Modified Total Sharp Score [mTSS]) was 84% (sarilumab 200 mg) and 68% (sarilumab 150 mg) vs. placebo in patients with low Hb and 97% (sarilumab 200 mg) and 68% (sarilumab 150 mg) vs. placebo in patients with normal Hb. Similar results were observed for other radiographic outcomes.
Conclusions
In these post hoc analyses, a consistent relationship was observed between higher Hb levels and better clinical outcomes and PROs in patients with RA. Irrespective of the baseline Hb levels, sarilumab treatment was associated with improvements in clinical measures and PROs over 24 weeks (improvements were more pronounced than those with adalimumab treatment) and mitigation of joint damage progression over 52 weeks.
Trial registration
ClinTrials.gov NCT01061736, NCT01709578, and NCT02332590
Supplementary Information
The online version contains supplementary material available at 10.1186/s13075-022-02891-x.
Keywords: Rheumatoid arthritis, Sarilumab, Patient-reported outcomes, Anemia, Hemoglobin, Radiographic outcomes, TARGET, MOBILITY, MONARCH
Introduction
Rheumatoid arthritis (RA) is a chronic, systemic autoimmune disease that, when untreated, progressively results in destruction and disability of the joints in 65 to 70% of patients and is associated with significant effects on social health and economic challenges. If left untreated, RA results in shortened life expectancy [1–4]. The global prevalence of RA in the adult population is approximately 0.3 to 1.0% [5].
Chronic inflammatory conditions may exhibit various hematologic abnormalities such as anemia, which develops in 15 to 47% [6–8] of patients with RA. The etiology of anemia in RA is complex and multifactorial [9]. Proinflammatory cytokines, including tumor necrosis factor (TNF)-alpha, interleukin (IL)-1, IL-6, and IL-10, play important roles in RA-associated anemia [10]. IL-6 may exert its effect by inducing hepcidin, which is a key regulator of body iron homeostasis [11]. The circulating serum hepcidin level is reported to be increased in RA patients with anemia of chronic disease [12, 13]. In such patients, anemia is associated with high disease activity [14], erosive arthritis [15], and an increased risk of mortality [7]. Hemoglobin (Hb) levels may also correlate with radiographic outcomes [7, 16].
The use of an anti-IL-6 receptor (IL-6R) monoclonal antibody in RA patients with anemia has been reported previously and has provided evidence for greater improvement in Hb levels compared to the control group [17, 18].
Two monoclonal antibodies that target IL-6 signaling, sarilumab and tocilizumab (TCZ), are approved for the treatment of RA. TCZ has been shown to improve anemia in patients with RA [17–19].
Sarilumab, an IL6-R antagonist, is approved for the treatment of adult patients with moderate-to-severe RA with an inadequate response or intolerance to one or more disease-modifying antirheumatic drugs (DMARDs) [20–22].
We hypothesized that, in RA patients, higher Hb levels are associated with better outcomes and that this association would be more pronounced among the patients treated with sarilumab. To evaluate this hypothesis, we conducted two post hoc analyses to assess the relationship between Hb and clinical outcomes and Hb and radiographic progression in three phase III studies of sarilumab, TARGET (NCT01709578), MOBILITY (NCT01061736), and MONARCH (NCT02332590).
Methods
Study design
Details of the phase III study designs have been described previously [21, 23, 24]. In brief, the MOBILITY study was conducted to investigate the efficacy and safety of sarilumab (vs. placebo) in combination with methotrexate (MTX) in patients with moderate-to-severe active RA and an inadequate response to methotrexate (MTX-IR) through 52 weeks. The TARGET study investigated the efficacy and safety of sarilumab (or placebo) in combination with background conventional synthetic disease-modifying antirheumatic drugs (csDMARDs) in patients with moderate-to-severe RA who were intolerant of, or who had an inadequate response to, tumor necrosis factor (TNF) inhibitors (TNFi-INT/IR) through 24 weeks. The MONARCH study compared the 24-week efficacy and safety of sarilumab monotherapy with adalimumab monotherapy in biologic DMARD-naïve patients with moderate-to-severe active RA who were intolerant of or had an inadequate response to MTX (MTX-INT/IR).
For the post hoc analysis that evaluated the relationship between Hb levels and clinical efficacy measures and PROs, the data sets included pooled data from combination therapy from TARGET and MOBILITY and monotherapy data from MONARCH through 24 weeks. The other post hoc analysis evaluated the proportion of RA patients with low Hb at baseline and whether such patients were at an increased risk of joint damage progression compared to those with normal baseline Hb levels. The baseline characteristics and radiographic outcomes in MOBILITY were analyzed by baseline Hb category (low vs. normal) [25].
The three protocols were approved by the appropriate ethics committees/institutional review boards, and each patient provided written informed consent before participating in the study. The studies were conducted in compliance with institutional review board regulations, the International Conference on Harmonization Guidelines for Good Clinical Practice, and the Declaration of Helsinki.
Assessments and endpoints
In all the three studies, Hb levels were defined as low (85–120 g/L [female patient]/85–130 g/L [male patient]) vs. normal (120–155 g/L [female patient]/130–175 g/L [male patient]). Hb levels were assessed at baseline to week 24 in all three studies and at baseline to week 52 in MOBILITY.
Endpoints were the mean change from baseline through 24 weeks in clinical assessments including clinical disease activity index (CDAI), C-reactive protein (CRP), Disease Activity Score 28-joint count-CRP (DAS28-CRP), physician-assessed 28 swollen and tender joint counts (SJC28 and TJC28), Physician Global Assessment (MDGA) and patient-reported assessments including the patient global assessment (PtGA), pain, and morning stiffness using visual analog scale (pain-VAS and morning stiffness-VAS using 0–100 mm VAS), and Functional Assessment of Chronic Illness Therapy-Fatigue (FACIT-F).
Radiographic outcomes assessed in this study were Modified Total Sharp Score (mTSS), joint space narrowing (JSN, sum of JSN scores from 42 joints), and erosion score (sum of erosion scores from 44 joints). Endpoints were the changes in mTSS, JSN, and erosion score from baseline to 24 weeks and 52 weeks.
Statistical methods
All analyses that were based on low-Hb and normal-Hb level patient groups were conducted post hoc. Bivariate linear regression analysis evaluated the relationships between Hb levels and outcomes at baseline and weeks 4, 8, 12, 20, and 24. In these regression models, the dependent variable was a clinical efficacy or PRO endpoint, and independent variables were treatment, Hb group, and interaction between treatment and Hb group (as indicated in Figs. 1 and 2). Categorical variables were summarized using the number and percentage (%) of patients. The mean changes from baseline in clinical efficacy measures and PROs were summarized as the mean (standard deviation [SD]) values. Forest plots were used to depict the mean difference between baseline and week 24 and week 52 for patients with low and normal baseline Hb by treatment arm and for each radiographic endpoint. Rank analysis of covariance (ANCOVA) models including prior biologic use, region, and baseline radiographic values as covariates were applied to assess the impact of sarilumab on the radiographic outcomes. A nominal P value of < 0.05 was considered significant in this analysis.
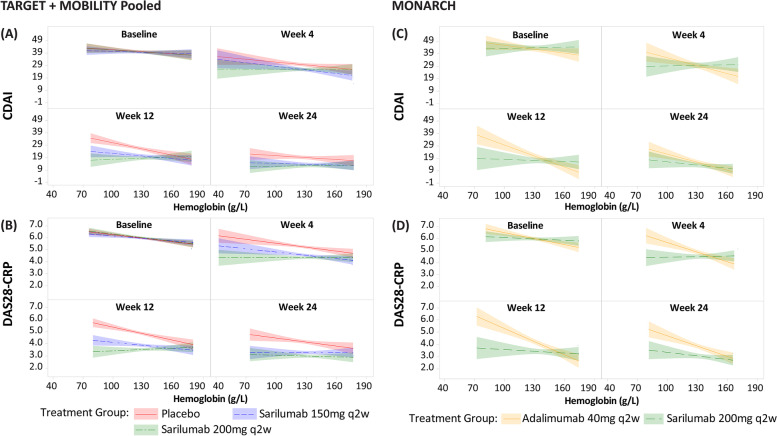
Fig. 1.
Relationship between Hb levels and clinical efficacy outcomes: CDAI and DAS-28 CRP over time. Slicefit plots of CDAI and DAS-28 CRP (Y-axis) against hemoglobin (g/L) (X-axis) by a visit from baseline to week 24. Patients with non-missing CRP (mg/L) and hemoglobin (g/L) values were considered. Each panel in the regression model shows the relationship between an outcome and Hb level at a given point in time, stratified by treatment. The lines, with shading, depict the treatment-specific regression coefficient (i.e., slope) and 95% CI associated with the linear regression in which Hb is the independent variable and the outcome is the dependent variable. CDAI, Clinical Disease Activity Index; CRP, C-reactive protein; DAS, Disease Activity Scores; Hb, hemoglobin; q2w, every 2 weeks
Fig. 2.
Relationship between Hb levels and clinical efficacy outcomes: PtGA and pain VAS over time. Slicefit plots of PtGA (MM) and pain VAS (MM) (Y-axis) against hemoglobin (g/L) (X-axis) by a visit from baseline to week 24. Each panel in the regression model shows the relationship between an outcome and Hb level at a given point in time, stratified by treatment. The lines, with shading, depict the treatment-specific regression coefficient (i.e., slope) and 95% CI associated with the linear regression in which Hb is the independent variable and the outcome is the dependent variable. Hb, hemoglobin; pain VAS, pain visual analog scale; PtGA, Patient Global Assessment; q2w, every 2 weeks
Results
Patients’ baseline characteristics and disposition
Across the three phase III studies, 186 patients in TARGET (N = 546), 414 patients in MOBILITY (N = 1197), and 92 patients in MONARCH (N = 369) had low Hb levels (Table 1). In all the evaluated study groups, low Hb was more common in female patients vs. male patients (TARGET [86.0% vs. 14.0%], MOBILITY [85.8% vs. 14.3%], and MONARCH [84.8% vs. 15.2%]). In MOBILITY, low Hb was more common in Asians (14% vs. 5%), young patients (mean age 49 vs. 51 years), and those with lower body weight (mean 69 vs. 77 kg); all P values < 0.01. In MOBILITY, there was a significant difference in CRP between low baseline Hb and normal baseline Hb groups (mean: 30.2 [SD: 28.5] vs. 17.3 [18.5] mg/L; P < 0.0001). Differences in CRP between low baseline Hb and normal baseline Hb groups were also observed in TARGET and MONARCH studies.
Table 1.
Patients’ demographics and baseline disease characteristics
| TARGET (TNFi-INT/IR) | MOBILITY (MTX-IR) | MONARCH (MTX-INT/IR) | ||||
|---|---|---|---|---|---|---|
| Low Hb (N = 186) | Normal Hb (N = 360) | Low Hb (N = 414) | Normal Hb (N = 783) | Low Hb (N = 92) | Normal Hb (N = 277) | |
| Age, years | 52.1 (13.9) | 53.4 (11.5) | 49.2 (12.6) | 51.3 (11.0) | 50.0 (13.3) | 53.0 (11.9) |
| Female, n (%) | 160 (86.0) | 287 (79.7) | 355 (85.8) | 622 (79.4) | 78.0 (84.8) | 229 (82.7) |
| Male, n (%) | 26 (14.0) | 73 (20.3) | 59 (14.3) | 161 (20.6) | 14 (15.2) | 48 (17.3) |
| Weight, kg | 74.7 (20.9) | 80.0 (21.6) | 69.5 (16.9) | 76.9 (18.8) | 69.3 (19.8) | 73.0 (16.1) |
| BMI, kg/m2 | 28.7 (7.5) | 30.0 (7.0) | 26.7 (6.0) | 29.1 (6.4) | 26.2 (7.1) | 27.5 (5.7) |
| Race, n (%) | ||||||
| White | 118 (63.4) | 270 (75.0) | 336 (81.2) | 695 (88.8) | 82 (89.1) | 253 (91.3) |
| Black | 14 (7.5) | 6 (1.7) | 11 (2.7) | 17 (2.2) | 1 (1.1) | 3 (1.1) |
| Asian/Oriental | 3 (1.6) | 2 (0.6) | 58 (14.0) | 40 (5.1) | 5 (5.4) | 6 (2.2) |
| Others | 51 (27.4) | 82 (22.8) | 9 (2.2) | 31 (4.0) | 4 (4.4) | 15 (5.4) |
| Duration of RA since diagnosis, years | 12.2 (9.6) | 12.0 (9.3) | 8.6 (8.0) | 9.3 (7.8) | 6.3 (7.3) | 7.7 (8.2) |
| Anti-CCP [n (%)] positive | 147 (79.9) | 275 (77.3) | 350 (84.8) | 686 (88.0) | 73 (81.1) | 199 (74.3) |
| Rheumatoid factor [n (%)] positive | 147 (79.5) | 262 (73.4) | 344 (83.3) | 665 (85.5) | 63 (70.0) | 172 (64.4) |
| TJC28 (0–28) | 17.1 (7.4) | 15.8 (6.6) | 16.0 (6.7) | 15.6 (6.6) | 16.7 (6.4) | 16.7 (6.2) |
| SJC28 (0–28) | 14.5 (6.3) | 13.1 (5.9) | 12.2 (5.7) | 11.8 (5.4) | 13.3 (5.8) | 12.8 (5.5) |
| HAQ-DI (0–3) | 1.9 (0.6) | 1.7 (0.6) | 1.7 (0.7) | 1.6 (0.6) | 1.7 (0.5) | 1.6 (0.6) |
| CRP, mg/L* | 36.9 (30.5) | 21.6 (21.4) | 30.2 (28.5) | 17.3 (18.5) | 35.0 (35.0) | 16.0 (21.5) |
| CDAI | 46.0 (14.0) | 42.4 (12.4) | 41.2 (12.4) | 40.1 (12.2) | 43.6 (12.9) | 42.8 (11.8) |
| DAS28-CRP | 6.5 (1.0) | 6.1 (0.9) | 6.1 (0.9) | 5.9 (0.9) | 6.3 (0.9) | 5.9 (0.9) |
| Pain-VAS, mm | 75.9 (18.2) | 70.7 (18.8) | 66.2 (20.2) | 64.7 (21.3) | 71.5 (17.9) | 71.5 (19.1) |
| Morning stiffness VAS, 0–100 mm | 72.4 (22.0) | 67.7 (23.1) | Not Available | Not Available | 68.6 (20.5) | 69.7 (20.2) |
| FACIT-Fatigue | 22.8 (10.0) | 23.8 (10.8) | 25.3 (10.4) | 27.0 (10.1) | 23.0 (9.2) | 24.0 (9.8) |
All values are mean (SD), unless specified otherwise
N number of patients in each study group with low or normal Hb subgroup, BMI body mass index, CCP cyclic citrullinated peptide, CRP C-reactive protein, CDAI Clinical Disease Activity Index, DAS Disease Activity Scores, DAS28-CRP Disease Activity Score 28-joint count-C-reactive protein, FACIT-F Functional Assessment of Chronic Illness Therapy-Fatigue, HAQ-DI Stanford Health Assessment Questionnaire-Disability Index, Hb hemoglobin, MTX-INT/IR intolerant of, or an inadequate response to, MTX, MTX-IR An inadequate response to methotrexate, RA rheumatoid arthritis, SD standard deviation, SE standard error, SJC28 Swollen 28-Joint Count, TJC28 Tender 28-Joint Count, TNFi-INT/IR intolerant of, or an inadequate response to, tumor necrosis factor inhibitors, VAS visual analog scale
*Significant difference in CRP between low and normal baseline Hb groups in MOBILITY study (P < 0.0001)
Duration of RA and other factors were similar between patients with low and normal baseline Hb in all the evaluated study groups (TARGET, MOBILITY, and MONARCH). Prior RA medication use was also similar between the patients with low and normal baseline Hb in all study groups (Suppl Table 1).
TARGET MOBILITY Pooled and MONARCH: mean change in Hb levels over time
During the TEAE period (from baseline to 52 weeks), the mean changes in Hb (g/L) were as follows: placebo—mean change: − 0.6 (95% CI: − 2.15–0.87); sarilumab 150 mg—mean change: 9.1 (95% CI: 7.76–10.42); and sarilumab 200 mg—mean change: 10.0 (95% CI: 8.72–11.29) (Table 2).
Table 2.
Mean change in Hb levels over time by treatment arm (TARGET, MOBILITY, and MONARCH)
| Hb (g/L) | Baseline | Mean change from baseline to 24 weeks | Mean change from baseline to 52 weeks |
|---|---|---|---|
| TARGET | |||
| Placebo + DMARD | 126.6 | − 0.3 | – |
| Sarilumab 150 mg q2w + DMARD | 128.1 | + 8.0 | – |
| Sarilumab 200 mg q2w + DMARD | 125.7 | + 9.0 | – |
| MOBILITY | |||
| Placebo + MTX | 127.0 | − 0.3 | − 0.6 |
| Sarilumab 150 mg + MTX | 125.6 | + 7.9 | + 9.1 |
| Sarilumab 200 mg + MTX | 126.6 | + 8.2 | + 10.0 |
| MONARCH | |||
| Adalimumab 40 mg q2w | 129.5 | + 1.1 | – |
| Sarilumab 200 mg q2w | 130.1 | + 6.0 | – |
Mean changes in Hb levels were reported at baseline, week 24, and week 52 (MOBILITY-B only)
DMARD disease-modifying antirheumatic drugs, Hb hemoglobin, q2w every 2 weeks, SD standard deviation
TARGET+MOBILITY Pooled and MONARCH: relationship between Hb and multiple endpoints from baseline through week 24
From baseline to week 24, higher Hb levels were consistently associated with better outcomes, irrespective of the study groups, in TARGET+MOBILITY Pooled and MONARCH; however, we observed that this association was more pronounced among the patients treated with sarilumab than those treated with placebo and/or adalimumab. Moreover, a steady disconnect between Hb and clinical outcomes was observed in sarilumab-treated patients through week 24 and not observed in placebo or adalimumab (Fig. 1).
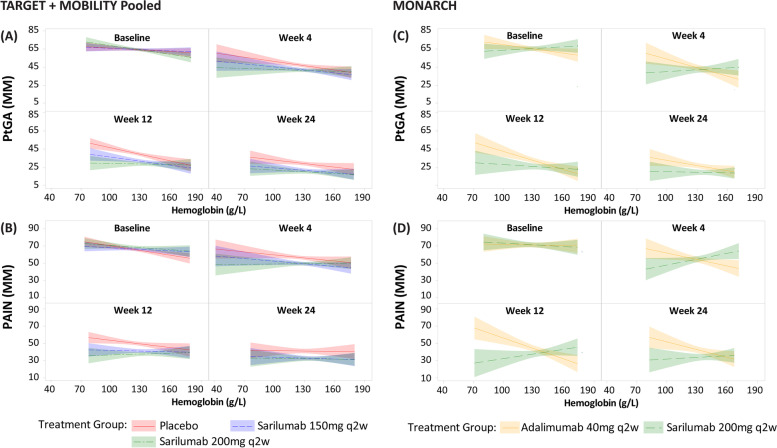
Disassociation between Hb level and endpoints was consistent between the two evaluated study groups (TARGET+MOBILITY Pooled) and MONARCH through week 24 across multiple additional clinical endpoints including PtGA and pain-VAS (Fig. 2), TJC28 and SJC28 (Suppl. Fig. 1), and FACIT-F and morning stiffness-VAS (Suppl. Fig. 2).
Scatterplots from MONARCH were generated to evaluate the improvement in CDAI by Hb status (low-left of the WHO cutoff lines; normal-right of WHO cutoff lines) in patients treated with sarilumab (panel A) or adalimumab (panel B) over 24 weeks (Suppl. Fig. 3). Improvement in CDAI by Hb status was higher with sarilumab (68%; panel A) vs. adalimumab (61%; panel B).
By week 24, the proportion of patients with improvement in CDAI and Hb (“responders” defined as patients with a CDAI improvement from baseline of ≥ 50% and Hb as ≥ 120 g/L for women or Hb ≥ 130 g/L for men) was 71.2% (n = 111/156) in the sarilumab group and 60.8% (n = 90/148) in the adalimumab group (Suppl. Table 2).
For TARGET+MOBILITY Pooled data, the improvement in CDAI and Hb was more pronounced in patients receiving sarilumab 150 mg and 200 mg q2w vs. placebo (Suppl. Fig. 4). By week 24, the proportion of responders was 42.9% (n = 145/338) in the placebo group, 64.6% (n = 227/429) in the sarilumab 150 mg group, and 71.6% (n = 313/437) in the sarilumab 200 mg group (Suppl. Table 3).
TARGET+MOBILITY Pooled and MONARCH: mean change in PROs from baseline to week 24
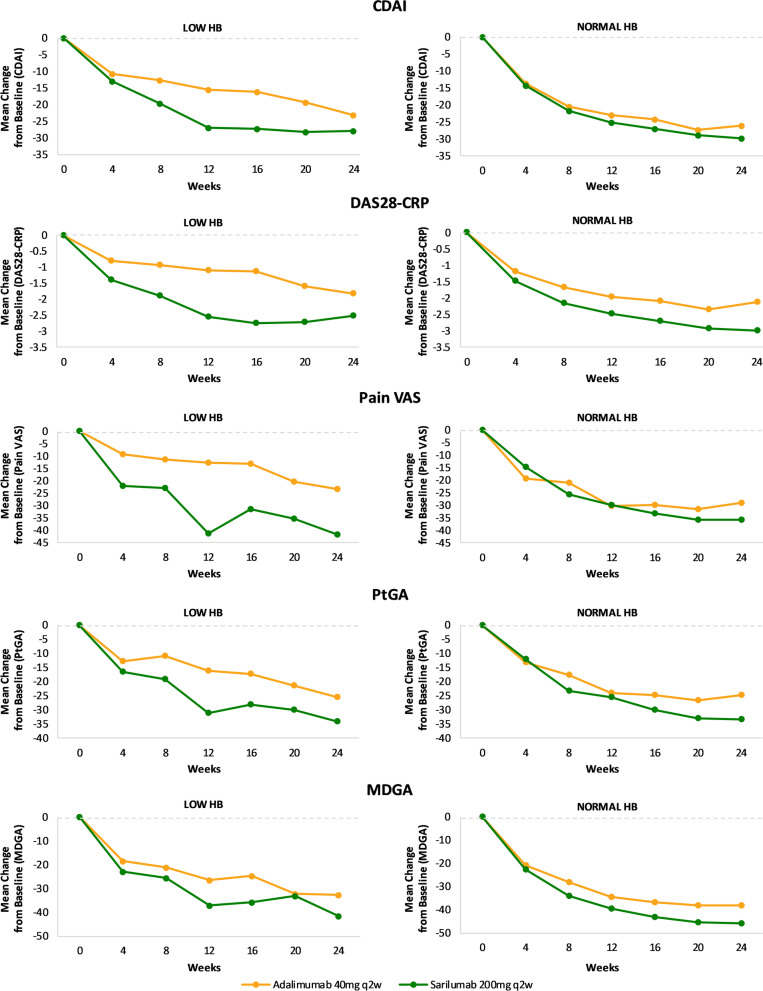
The mean change in CDAI, DAS-28-CRP, and MDGA at week 24 was similar in both low Hb and normal Hb subgroups. The results for the PROs (pain-VAS, PtGA) in MONARCH (Fig. 3) and TARGET+MOBILITY are shown in Suppl. Fig. 5. The effect size (difference between sarilumab and comparator adalimumab) was larger for the low Hb subgroup in MONARCH, but there were no obvious conclusions regarding the relationship between Hb and PROs in TARGET +MOBILITY (Suppl Fig. 5). Similarly, the effect size observed for TJC28, SJC28, FACIT-F, and morning stiffness-VAS in the low Hb subgroup were modest with the combination therapy group (Suppl. Fig. 6) compared to the monotherapy group (Suppl. Fig. 7).
Fig. 3.
Mean change in treatment outcomes over time (stratified by Hb level at baseline)—MONARCH. CDAI, Clinical Disease Activity Index; CRP, C-reactive protein; DAS28-CRP, Disease Activity Score-28 for Rheumatoid Arthritis with CRP; Hb, hemoglobin; PtGA, Patient Global Assessment; MDGA, Physician Global Assessment; q2w, every 2 weeks; VAS, visual analog scale
MOBILITY: mean change in radiographic outcomes (from baseline to week 52)
Modified Total Sharp Score
The mean (SD) change from baseline to weeks 24 and 52 in mTSS in the sarilumab 200 mg, sarilumab 150 mg, and placebo arms is shown in Fig. 4 (stratified by Hb level).
Fig. 4.
Mean change in mTSS from baseline at week 24 and week 52 (95% CI)—MOBILITY. All patients received weekly MTX, and sarilumab or placebo were administered q2w. At week 52, low Hb: P < 0.001 by rank ANCOVA for each sarilumab dose vs. placebo; normal Hb: rank ANCOVA P < 0.001 for 200 mg and P < 0.01 for 150 mg sarilumab dose vs. placebo. Hb, hemoglobin; mTSS, Modified Total Sharp Score; MTX, methotrexate; q2w, every 2 weeks; SD, standard deviation
By week 52, the inhibition of progression of structural damage was 84% (sarilumab 200 mg) and 68% (sarilumab 150 mg) vs. placebo in patients with low Hb and 97% (sarilumab 200 mg) and 68% (sarilumab 150 mg) vs. placebo in patients with normal Hb. Overall, the patients who received placebo + MTX had significantly more radiographic progression than the patients in the sarilumab 150 mg and 200 mg + MTX treatment groups, as assessed by mTSS through week 52.
Joint space narrowing
The mean (SD) change from baseline to weeks 24 and 52 in JSN in the sarilumab 200 mg, sarilumab 150 mg, and placebo arms is shown in Suppl. Fig. 8 (stratified by Hb level).
By week 52, the inhibition of progression of structural damage was 67.1% (sarilumab 200 mg) and 48.0% (sarilumab 150 mg) vs. placebo in patients with low Hb and 95.1% (sarilumab 200 mg) and 75.4% (sarilumab 150 mg) vs. placebo in patients with normal Hb.
Erosion score
The mean (SD) change from baseline to weeks 24 and 52 in erosion score in the sarilumab 200 mg, sarilumab 150 mg, and placebo arms is shown in Suppl. Fig. 9 (stratified by Hb level).
By week 52, the inhibition of progression of structural damage was 95.5% (sarilumab 200 mg) and 81.7% (sarilumab 150 mg) vs. placebo in patients with low Hb and 98.1% (sarilumab 200 mg) and 58.9% (sarilumab 150 mg) vs. placebo in patients with normal Hb.
Safety
The safety profile of sarilumab has been previously reported and was not part of this post hoc analysis. In a long-term safety analysis, the most frequent AEs associated with sarilumab were neutropenia, increased alanine aminotransferase, injection site erythema, upper respiratory infections, urinary tract infections, nasopharyngitis, and bronchitis [21, 23, 24].
Discussion
In these post hoc analyses from baseline to week 24, higher Hb levels were consistently associated with better disease outcomes (CDAI, DAS-28 CRP, pain-VAS, PtGA, and TJC28) in both study groups (pooled TARGET+MOBILITY, MONARCH). These results are in line with other published studies. Hashimoto et al. reported significant increases in Hb levels in anemic RA patients and better clinical outcomes (CDAI and CRP) after TCZ therapy compared with non-biologic therapies even after adjustment for baseline characteristics and baseline disease activity [17].
Strand et al. [26] reported that in sarilumab-treated patients, there was an improvement in PROs such as PtGA, pain-VAS, Stanford Health Assessment Questionnaire-Disability Index (HAQ-DI) score, and FACIT-F after 2 weeks of therapy. This improvement was numerically greater in the sarilumab 200 mg group vs. the sarilumab 150 mg q2w group and was sustained through week 52. Another exploratory analysis reported that at week 24, both sarilumab 150 mg and 200 mg q2w were superior to placebo in all clinical efficacy scores (American College of Rheumatology 20 (ACR20) response, DAS28-CRP remission rate, HAQ-DI score, and CDAI score) [27].
In these analyses, the association between improvement in Hb levels and clinical outcomes was more pronounced among patients treated with sarilumab than those treated with placebo and adalimumab. The findings are similar to those in the published literature [28, 29]. Song et al. [18] reported that in RA patients with anemia, treatment with both tocilizumab and TNF inhibitors (etanercept, infliximab, or adalimumab) resulted in significant improvements in anemia and disease activity, while the effect was more pronounced in patients treated with tocilizumab vs. those treated with TNF inhibitors. An analysis of the Rhumadata® clinical database and registry [30] confirmed that the improvement in anemia observed with IL-6 inhibitors was “numerically and statistically superior” to TNF inhibitors.
IL-6 induces hepcidin which plays a critical role in the development of anemia by causing alterations in iron homeostasis [31, 32]. A recent study addressed the laboratory and hematological parameters associated with RA and more specifically with anemia of chronic disease. The report confirms that patients with RA have a significant elevation of serum IL-6 levels including the patient group with anemia [33]. Our results suggest that the blockade of IL-6 receptor by sarilumab improves the hemoglobin level, possibly via decreasing hepcidin levels, and may contribute to the greater improvement in anemia observed with IL-6 inhibitors [34]. In the in vitro experiments conducted by Song et al., a decrease in serum IL-6 and hepcidin levels was observed in patients treated with TNF inhibitors suggesting that the TNF inhibitors may cause an indirect reduction in hepcidin levels by reducing IL-6 [18].
Although it can be postulated that IL-6 inhibition by sarilumab was effective in increasing Hb levels, independent of the baseline Hb levels, other possibilities cannot be ruled out; for example, IL-6Ri may counter at the target tissue level the inhibitory effects of elevated IL-6 level on erythropoiesis or the erythropoietin response as previously reported [34]. The findings did not address the molecular responses induced by sarilumab on erythropoiesis and therefore cannot fully address the causality between therapy and modification of Hb levels.
In these analyses of phase III randomized controlled trials, dissociation between Hb and clinical efficacy measures was observed by week 24 in sarilumab-treated patients but not in those treated with placebo or adalimumab. These results imply that the positive effect of treatment with sarilumab does not vary by whether a patient has low Hb at baseline.
Preventing joint damage progression and improvement in health-related quality of life are important therapeutic goals in RA [35]. The results showed that patients with low Hb at baseline were at a greater risk for joint damage progression over 52 weeks in the MOBILITY trial than those with normal Hb. Overall, Hb levels could have the potential to act as a surrogate biomarker for predicting the improvement of individual-tailored treatment and choice of drug therapies in RA patients. In the sarilumab + MTX groups, joint damage progression was mitigated compared with placebo + MTX. A similar magnitude of the effect was observed, irrespective of the Hb stratification. These results are consistent with other studies that have demonstrated improvements in long-term radiographic outcomes with early intensive treatment in patients with RA [36–39]. In an open-label extension study, 5-year follow-up data showed that sarilumab + MTX resulted in reductions in disease activity, inhibition of radiographic progression, and improvements in physical function [40].
This study has some limitations. Due to the lack of data on markers of iron status (serum iron, ferritin, and transferrin), hepcidin, or erythropoietin, we could not assess the precise mechanisms of anemia in the included study population. In this post hoc analysis, compensation for repeated analyses was not included in the methodology; as a result, nominal P values were generated without formal adjustments for multiple comparisons. Moreover, the limited sample size for patients with low Hb at baseline prevented pursuing stratification by characteristics linked to clinical outcomes or multivariate analysis with adjustment for baseline characteristics as previously reported by Hashimoto et al. [17]. Additional investigations would be required to potentially address these limitations.
Conclusions
In these post hoc analyses of the three phase III trials of sarilumab, improvements in clinical outcomes for the approved sarilumab 200 mg dose remained independent of changes in Hb level. Clinical, PROs, and laboratory improvements with sarilumab were greater than comparator groups in patients with low Hb levels. Sarilumab treatment was associated with improvement of the clinical outcomes over 24 weeks and mitigation of joint damage progression over 52 weeks, irrespective of baseline Hb levels. By week 24, more sarilumab-treated patients with low Hb levels showed improvement in clinical efficacy measures and PROs than adalimumab-treated patients.
Supplementary Information
Additional file 1: Supplementary Table 1. Concomitant Medications at Baseline. Supplementary Table 2. Proportion of Responders* With Improvement in CDAI and Hb at Respective Visit – MONARCH (Safety Population). Supplementary Table 3. Proportion of responders* With Improvement in CDAI and Hb at Respective Visit – TARGET + MOBILITY Pooled.
Additional file 2: Supplementary Figure 1. Relationship Between Hb Levels and Clinical Efficacy Outcomes: TJC28 and SJC28 Over Time. Supplementary Figure 2. Relationship Between Hb Levels and Clinical Efficacy. Supplementary Figure 3. Scatterplot of CDAI Against Hb (g/L) by Visit and Baseline Hb Status– MONARCH. Supplementary Figure 4. Scatterplots of CDAI Against Hb (g/L) by Visit and Baseline Hb Status – TARGET + MOBILITY Pooled. Supplementary Figure 5. Mean Change in Treatment Outcomes: CDAI, DAS28-CRP, Pain-VAS, PtGA, MDGA – TARGET + MOBILITY Pooled. Supplementary Figure 6. Mean Change in Treatment Outcomes: TJC28, SJC28, FACIT-Fatigue, and Morning Stiffness – TARGET+MOBILITY Pooled Mean Change in Treatment Outcomes: CDAI, DAS28-CRP, Pain-VAS, PtGA, MDGA – TARGET + MOBILITY Pooled. Supplementary Figure 7. Mean Change in Treatment Outcomes: TJC28, SJC28, FACIT-Fatigue, and Morning Stiffness – MONARCH. Supplementary Figure 8. Mean change in JSN from Baseline at Week 24 and Week 52 (95% CI) – MOBILITY. Supplementary Figure 9. Mean change in Erosion score from Baseline at Week 24 and Week 52 (95% CI) – MOBILITY.
Acknowledgements
The authors acknowledge Nupur Chaubey and Keerthy Kanakamedala (ex-employees of Sanofi) for the medical writing and editorial assistance of the manuscript.
Abbreviations
- ACR
American College of Rheumatology
- ANCOVA
Analysis of covariance
- ART
Arthritis Research and Therapy
- BMI
Body mass index
- CCP
Cyclic citrullinated peptide
- CDAI
Clinical Disease Activity Index
- CI
Confidence interval
- CRP
C-reactive protein
- csDMARD
Conventional synthetic disease-modifying antirheumatic drug
- DAS
Disease Activity Scores
- DAS28-CRP
Disease Activity Score-28 for Rheumatoid Arthritis with CRP
- DMARD
Disease-modifying antirheumatic drug
- FACIT-F
Functional Assessment of Chronic Illness Therapy-Fatigue
- GABA
Gamma-aminobutyric acid
- HAQ-DI
Stanford Health Assessment Questionnaire-Disability Index
- Hb
Hemoglobin
- IL
Interleukin
- JSN
Joint Space Narrowing Score
- MDGA
Physician Global Assessment
- mTSS
Modified Total Sharp Score
- MTX
Methotrexate
- N
Total number of patients in each study group with low or normal Hb subgroup
- n
Number of patients in each subgroup
- NSAID
Nonsteroidal anti-inflammatory drug
- P value
Probability
- Pain-VAS
Pain as assessed by the visual analog scale
- PRO
Patient-reported outcome
- PtGA
Patient Global Assessment
- q2w
Every 2 weeks
- RA
Rheumatoid arthritis
- SD
Standard deviation
- SJC28
Swollen 28-Joint Count
- SSRI
Selective serotonin reuptake inhibitor
- TCZ
Tocilizumab
- TJC28
Tender 28-Joint Count
- TNF
Tumor necrosis factor
- TNFi
Tumor necrosis factor inhibitor
- VAS
Visual analog scale
- WHO
World Health Organization
Authors’ contributions
All authors read, critically revised, and approved the final version of the manuscript and are accountable for the accuracy and integrity of the data. In addition, A.RR., C.B., G.B., and S.F. contributed to the conception and design of the study, data analysis, and manuscript writing. D.F., V.B., and C. CS. contributed to the data analysis. A.P. contributed to the data acquisition and analysis and manuscript writing.
Funding
The study was sponsored by Sanofi (Cambridge, MA, USA) and Regeneron Pharmaceuticals, Inc. (Tarrytown, NY, USA).
Availability of data and materials
Qualified researchers may request access to patient-level data and related study documents including the clinical study report, study protocol with any amendments, blank case report form, statistical analysis plan, and data set specifications. Patient-level data will be anonymized, and study documents will be redacted to protect the privacy of our trial participants. Further details on Sanofi’s data sharing criteria, eligible studies, and process for requesting access can be found at https://www.vivli.org/.
Declarations
Ethics approval and consent to participate
The protocols used in the TARGET, MOBILITY, and MONARCH were approved by the appropriate ethics committees/institutional review boards, and each patient provided written informed consent before participating in the study. The studies were conducted in compliance with institutional review board regulations, the International Conference on Harmonization Guidelines for Good Clinical Practice, and the Declaration of Helsinki.
Competing interests
A. RR. has received honoraria for lectures and consultation from Sanofi, Abbvie, Lilly, Gilead, Roche, BMS, Boehringer, Novartis, Janssen, UCB, and Amgen.
D. F. has received grants and/or consulting fees from Actelion, Amgen, Bristol-Myers Squibb, Corbus, Galapagos, GlaxoSmithKline, Horizon, Kadmon, NIH, Novartis, Pfizer, Roche/Genentech, Talaris, and Sanofi Genzyme/Regeneron.
S. F. and A. P. are employees of Sanofi and may hold stock and/or stock options in the company.
V. B. has received consulting fees from Amgen; Bristol-Myers Squibb; Gilead; Pfizer; Regeneron Pharmaceuticals, Inc.; Sanofi Genzyme; and UCB, and her host institution has received research grants from Amgen, the Cedar Hill Foundation, and National Institutes of Health.
C. B. has received grants and/or consulting fees from AbbVie, Bristol-Myers Squibb, Janssen, Lilly, Pfizer, and Sanofi Genzyme/Regeneron.
C. CS. has received grants and/or consulting fees from AbbVie, Amgen, Bristol-Myers Squibb, Gilead, Octopharma, Pfizer, and Sanofi Genzyme/Regeneron.
G.B. has received grant/research support, consulting fees, and/or speaker fees/honoraria from AbbVie, Lilly, Merck Sharp & Dohme, Pfizer, Roche, Sanofi Genzyme, and UCB.
Footnotes
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
References
- 1.McInnes IB, Schett G. The pathogenesis of rheumatoid arthritis. N Engl J Med. 2011;365(23):2205–2219. doi: 10.1056/NEJMra1004965. [DOI] [PubMed] [Google Scholar]
- 2.Silman AJ. The changing face of rheumatoid arthritis: why the decline in incidence? Arthritis Rheum. 2002;46(3):579–581. doi: 10.1002/art.508. [DOI] [PubMed] [Google Scholar]
- 3.van Aken J, van Dongen H, le Cessie S, Allaart CF, Breedveld FC, Huizinga TW. Comparison of long term outcome of patients with rheumatoid arthritis presenting with undifferentiated arthritis or with rheumatoid arthritis: an observational cohort study. Ann Rheum Dis. 2006;65(1):20–25. doi: 10.1136/ard.2005.038471. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Wong JB, Ramey DR, Singh G. Long-term morbidity, mortality, and economics of rheumatoid arthritis. Arthritis Rheum. 2001;44(12):2746–2749. doi: 10.1002/1529-0131(200112)44:12<2746::AID-ART461>3.0.CO;2-Z. [DOI] [PubMed] [Google Scholar]
- 5.WHO. Chronic rheumatic conditions. Available from: https://www.who.int/chp/topics/rheumatic/en/. Accessed 11 Feb 2022.
- 6.Chen YF, Xu SQ, Xu YC, Li WJ, Chen KM, Cai J, et al. Inflammatory anemia may be an indicator for predicting disease activity and structural damage in Chinese patients with rheumatoid arthritis. Clin Rheumatol. 2020;39(6):1737–1745. doi: 10.1007/s10067-019-04873-y. [DOI] [PubMed] [Google Scholar]
- 7.Möller B, Scherer A, Förger F, Villiger PM, Finckh A. Anaemia may add information to standardised disease activity assessment to predict radiographic damage in rheumatoid arthritis: a prospective cohort study. Ann Rheum Dis. 2014;73(4):691–696. doi: 10.1136/annrheumdis-2012-202709. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Nikiphorou E, de Lusignan S, Mallen C, Khavandi K, Roberts J, Buckley CD, et al. Haematological abnormalities in new-onset rheumatoid arthritis and risk of common infections: a population-based study. Rheumatology (Oxford, England) 2020;59(5):997–1005. doi: 10.1093/rheumatology/kez344. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Papadaki HA, Kritikos HD, Valatas V, Boumpas DT, Eliopoulos GD. Anemia of chronic disease in rheumatoid arthritis is associated with increased apoptosis of bone marrow erythroid cells: improvement following anti–tumor necrosis factor-α antibody therapy. Blood. 2002;100(2):474–482. doi: 10.1182/blood-2002-01-0136. [DOI] [PubMed] [Google Scholar]
- 10.Means RT, Jr, Krantz SB. Progress in understanding the pathogenesis of the anemia of chronic disease. Blood. 1992;80(7):1639–1647. doi: 10.1182/blood.V80.7.1639.1639. [DOI] [PubMed] [Google Scholar]
- 11.Hashizume M, Mihara M. The roles of interleukin-6 in the pathogenesis of rheumatoid arthritis. Arthritis. 2011;2011:765624. doi: 10.1155/2011/765624. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Demirag MD, Haznedaroglu S, Sancak B, Konca C, Gulbahar O, Ozturk MA, et al. Circulating hepcidin in the crossroads of anemia and inflammation associated with rheumatoid arthritis. Intern Med (Tokyo, Japan) 2009;48(6):421–426. doi: 10.2169/internalmedicine.48.1578. [DOI] [PubMed] [Google Scholar]
- 13.Wrighting DM, Andrews NC. Interleukin-6 induces hepcidin expression through STAT3. Blood. 2006;108(9):3204–3209. doi: 10.1182/blood-2006-06-027631. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Ganna S. Prevalência de anemia na artrite reumatoide. Rev Bras Reumatol. 2014;54(4):257–259. doi: 10.1016/j.rbr.2014.03.023. [DOI] [PubMed] [Google Scholar]
- 15.Yuan S, Chen D, Xiao Y, Lao M, Qiu Q, Liang L, et al. Factors associated with erosive arthritis in rheumatoid arthritis and other connective tissue diseases: a retrospective study from a southern Chinese population. J Clin Rheumatol. 2016;22(1). [DOI] [PubMed]
- 16.van der Heijde DM, van’t Hof MA, van Riel PL, van Leeuwen MA, van Rijswijk MH, van de Putte LB. Validity of single variables and composite indices for measuring disease activity in rheumatoid arthritis. Ann Rheum Dis. 1992;51(2):177–181. doi: 10.1136/ard.51.2.177. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Hashimoto M, Fujii T, Hamaguchi M, Furu M, Ito H, Terao C, et al. Increase of hemoglobin levels by anti-IL-6 receptor antibody (tocilizumab) in rheumatoid arthritis. PLoS One. 2014;9(5):e98202. doi: 10.1371/journal.pone.0098202. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Song SN, Iwahashi M, Tomosugi N, Uno K, Yamana J, Yamana S, et al. Comparative evaluation of the effects of treatment with tocilizumab and TNF-α inhibitors on serum hepcidin, anemia response and disease activity in rheumatoid arthritis patients. Arthritis Res Ther. 2013;15(5):R141. doi: 10.1186/ar4323. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Isaacs JD, Harari O, Kobold U, Lee JS, Bernasconi C. Effect of tocilizumab on haematological markers implicates interleukin-6 signalling in the anaemia of rheumatoid arthritis. Arthritis Res Ther. 2013;15(6):R204. doi: 10.1186/ar4397. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Boyce EG, Rogan EL, Vyas D, Prasad N, Mai Y. Sarilumab: review of a second IL-6 receptor antagonist indicated for the treatment of rheumatoid arthritis. Ann Pharmacother. 2018;52(8):780–791. doi: 10.1177/1060028018761599. [DOI] [PubMed] [Google Scholar]
- 21.Genovese MC, Fleischmann R, Kivitz AJ, Rell-Bakalarska M, Martincova R, Fiore S, et al. Sarilumab plus methotrexate in patients with active rheumatoid arthritis and inadequate response to methotrexate: results of a phase III study. Arthritis Rheumatol (Hoboken, NJ) 2015;67(6):1424–1437. doi: 10.1002/art.39093. [DOI] [PubMed] [Google Scholar]
- 22.Huizinga TW, Fleischmann RM, Jasson M, Radin AR, van Adelsberg J, Fiore S, et al. Sarilumab, a fully human monoclonal antibody against IL-6Rα in patients with rheumatoid arthritis and an inadequate response to methotrexate: efficacy and safety results from the randomised SARIL-RA-MOBILITY Part A trial. Ann Rheum Dis. 2014;73(9):1626–1634. doi: 10.1136/annrheumdis-2013-204405. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Burmester GR, Lin Y, Patel R, van Adelsberg J, Mangan EK, Graham NMH, et al. Efficacy and safety of sarilumab monotherapy versus adalimumab monotherapy for the treatment of patients with active rheumatoid arthritis (MONARCH): a randomised, double-blind, parallel-group phase III trial. Ann Rheum Dis. 2017;76(5):840. doi: 10.1136/annrheumdis-2016-210310. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Fleischmann R, van Adelsberg J, Lin Y, Castelar-Pinheiro GD, Brzezicki J, Hrycaj P, et al. Sarilumab and nonbiologic disease-modifying antirheumatic drugs in patients with active rheumatoid arthritis and inadequate response or intolerance to tumor necrosis factor inhibitors. Arthritis Rheumatol (Hoboken, NJ) 2017;69(2):277–290. doi: 10.1002/art.39944. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.WHO. Haemoglobin concentrations for the diagnosis of anaemia and assessment of severity. Vitamin and Mineral Nutrition Information System. Geneva: World Health Organization; 2011. (WHO/NMH/NHD/MNM/11.1). http://www.who.int/vmnis/indicators/haemoglobin.pdf. Accessed 11 Feb 2022.
- 26.Strand V, Kosinski M, Chen CI, Joseph G, Rendas-Baum R, Graham NM, et al. Sarilumab plus methotrexate improves patient-reported outcomes in patients with active rheumatoid arthritis and inadequate responses to methotrexate: results of a phase III trial. Arthritis Res Ther. 2016;18(1):198. doi: 10.1186/s13075-016-1096-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Fleischmann R, Genovese MC, van Adelsberg J, Mangan E, Iglesias-Rodriguez M, Dukovic D, Huizinga T. Pooled Safety and Efficacy of Sarilumab in Rheumatoid Arthritis Patients 65 Years of Age and Older [abstract]. Arthritis Rheumatol. 2016;68(suppl 10).
- 28.Genovese MC, Fleischmann R, Furst D, Janssen N, Carter J, Dasgupta B, et al. Efficacy and safety of olokizumab in patients with rheumatoid arthritis with an inadequate response to TNF inhibitor therapy: outcomes of a randomised phase IIb study. Ann Rheum Dis. 2014;73(9):1607–1615. doi: 10.1136/annrheumdis-2013-204760. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Tanaka Y, Martin Mola E. IL-6 targeting compared to TNF targeting in rheumatoid arthritis: studies of olokizumab, sarilumab and sirukumab. Ann Rheum Dis. 2014;73(9):1595. doi: 10.1136/annrheumdis-2013-205002. [DOI] [PubMed] [Google Scholar]
- 30.Choquette D, Bessette L, Sauvageau LC, Ferdinand I, Haraoui P, Massicotte F, et al. The impact of il-6 and tnf inhibitors on hemoglobin levels: an analysis from rhumadata® clinical database and registry. Ann Rheum Dis. 2019;78(Suppl 2):705.
- 31.Choy E. Understanding the dynamics: pathways involved in the pathogenesis of rheumatoid arthritis. Rheumatology (Oxford, England) 2012;51(Suppl 5):v3–11. doi: 10.1093/rheumatology/kes113. [DOI] [PubMed] [Google Scholar]
- 32.Dayer JM, Choy E. Therapeutic targets in rheumatoid arthritis: the interleukin-6 receptor. Rheumatology (Oxford, England) 2010;49(1):15–24. doi: 10.1093/rheumatology/kep329. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Ali ET, Jabbar AS, Mohammed AN. A comparative study of interleukin 6, inflammatory markers, ferritin, and hematological profile in rheumatoid arthritis patients with anemia of chronic disease and iron deficiency anemia. Anemia. 2019;2019:3457347. doi: 10.1155/2019/3457347. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Nikolaisen C, Figenschau Y, Nossent JC. Anemia in early rheumatoid arthritis is associated with interleukin 6-mediated bone marrow suppression, but has no effect on disease course or mortality. J Rheumatol. 2008;35(3):380. [PubMed] [Google Scholar]
- 35.Scott DL, Wolfe F, Huizinga TW. Rheumatoid arthritis. Lancet (London, England) 2010;376(9746):1094–1108. doi: 10.1016/S0140-6736(10)60826-4. [DOI] [PubMed] [Google Scholar]
- 36.Lard LR, Visser H, Speyer I, vander Horst-Bruinsma IE, Zwinderman AH, Breedveld FC, et al. Early versus delayed treatment in patients with recent-onset rheumatoid arthritis: comparison of two cohorts who received different treatment strategies. Am J Med. 2001;111(6):446–451. doi: 10.1016/S0002-9343(01)00872-5. [DOI] [PubMed] [Google Scholar]
- 37.Nell VP, Machold KP, Eberl G, Stamm TA, Uffmann M, Smolen JS. Benefit of very early referral and very early therapy with disease-modifying anti-rheumatic drugs in patients with early rheumatoid arthritis. Rheumatology (Oxford, England) 2004;43(7):906–914. doi: 10.1093/rheumatology/keh199. [DOI] [PubMed] [Google Scholar]
- 38.Quinn MA, Emery P. Window of opportunity in early rheumatoid arthritis: possibility of altering the disease process with early intervention. Clin Exp Rheumatol. 2003;21(5 Suppl 31):S154–S157. [PubMed] [Google Scholar]
- 39.Steunebrink LMM, Versteeg LGA, Vonkeman HE, Ten Klooster PM, Hoekstra M, van de Laar M. Radiographic progression in early rheumatoid arthritis patients following initial combination versus step-up treat-to-target therapy in daily clinical practice: results from the DREAM registry. BMC Rheumatol. 2018;2:1. doi: 10.1186/s41927-018-0009-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Genovese MC, van der Heijde D, Lin Y, St John G, Wang S, van Hoogstraten H, et al. Long-term safety and efficacy of sarilumab plus methotrexate on disease activity, physical function and radiographic progression: 5 years of sarilumab plus methotrexate treatment. RMD Open. 2019;5(2):e000887. doi: 10.1136/rmdopen-2018-000887. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Additional file 1: Supplementary Table 1. Concomitant Medications at Baseline. Supplementary Table 2. Proportion of Responders* With Improvement in CDAI and Hb at Respective Visit – MONARCH (Safety Population). Supplementary Table 3. Proportion of responders* With Improvement in CDAI and Hb at Respective Visit – TARGET + MOBILITY Pooled.
Additional file 2: Supplementary Figure 1. Relationship Between Hb Levels and Clinical Efficacy Outcomes: TJC28 and SJC28 Over Time. Supplementary Figure 2. Relationship Between Hb Levels and Clinical Efficacy. Supplementary Figure 3. Scatterplot of CDAI Against Hb (g/L) by Visit and Baseline Hb Status– MONARCH. Supplementary Figure 4. Scatterplots of CDAI Against Hb (g/L) by Visit and Baseline Hb Status – TARGET + MOBILITY Pooled. Supplementary Figure 5. Mean Change in Treatment Outcomes: CDAI, DAS28-CRP, Pain-VAS, PtGA, MDGA – TARGET + MOBILITY Pooled. Supplementary Figure 6. Mean Change in Treatment Outcomes: TJC28, SJC28, FACIT-Fatigue, and Morning Stiffness – TARGET+MOBILITY Pooled Mean Change in Treatment Outcomes: CDAI, DAS28-CRP, Pain-VAS, PtGA, MDGA – TARGET + MOBILITY Pooled. Supplementary Figure 7. Mean Change in Treatment Outcomes: TJC28, SJC28, FACIT-Fatigue, and Morning Stiffness – MONARCH. Supplementary Figure 8. Mean change in JSN from Baseline at Week 24 and Week 52 (95% CI) – MOBILITY. Supplementary Figure 9. Mean change in Erosion score from Baseline at Week 24 and Week 52 (95% CI) – MOBILITY.
Data Availability Statement
Qualified researchers may request access to patient-level data and related study documents including the clinical study report, study protocol with any amendments, blank case report form, statistical analysis plan, and data set specifications. Patient-level data will be anonymized, and study documents will be redacted to protect the privacy of our trial participants. Further details on Sanofi’s data sharing criteria, eligible studies, and process for requesting access can be found at https://www.vivli.org/.