Abstract
The Caulobacter crescentus CgtA protein is a member of the Obg-GTP1 subfamily of monomeric GTP-binding proteins. In vitro, CgtA specifically bound GTP and GDP but not GMP or ATP. CgtA bound GTP and GDP with moderate affinity at 30°C and displayed equilibrium binding constants of 1.2 and 0.5 μM, respectively, in the presence of Mg2+. In the absence of Mg2+, the affinity of CgtA for GTP and GDP was reduced 59- and 6-fold, respectively. N-Methyl-3′-O-anthranoyl (mant)–guanine nucleotide analogs were used to quantify GDP and GTP exchange. Spontaneous dissociation of both GDP and GTP in the presence of 5 to 12 mM Mg2+ was extremely rapid (kd = 1.4 and 1.5 s−1, respectively), 103- to 105-fold faster than that of the well-characterized eukaryotic Ras-like GTP-binding proteins. The dissociation rate constant of GDP increased sevenfold in the absence of Mg2+. Finally, there was a low inherent GTPase activity with a single-turnover rate constant of 5.0 × 10−4 s−1 corresponding to a half-life of hydrolysis of 23 min. These data clearly demonstrate that the guanine nucleotide binding and exchange properties of CgtA are different from those of the well-characterized Ras-like GTP-binding proteins. Furthermore, these data are consistent with a model whereby the nucleotide occupancy of CgtA is controlled by the intracellular levels of guanine nucleotides.
Small monomeric GTP-binding proteins have been identified in every organism examined thus far. These proteins are key players in a diverse array of essential cellular functions such as cell proliferation, signal transduction, protein synthesis, and protein targeting (3, 20, 54). The activity of GTP-binding proteins is controlled by the conformational state of the protein, being turned “on” when complexed with GTP and “off” when complexed with GDP. Conformational changes in the GTP-binding protein from the GDP- to the GTP-bound state are detected by downstream effector proteins. The specificity of the signaling cascade is due to the unique interactions between the GTP-binding protein and its effector proteins.
The balance between the amounts of GTP- and GDP-bound protein is a result of the affinity of the protein for guanine nucleotides and the rates of guanine nucleotide exchange and GTP hydrolysis. Typically, G proteins have very high affinities (in the nanomolar range) for nucleotides and low dissociation rates (on the order of hours) (3, 20, 54). Usually, the intrinsic hydrolysis rate of GTP is also low. In vivo, both dissociation and hydrolysis rates are controlled by three types of regulatory molecules. Guanine nucleotide exchange factors (GEFs) act as positive regulators that promote the release of guanine nucleotide (2, 16, 53). Since the intracellular concentration of GTP is usually high relative to that of GDP, the released nucleotide is almost always replaced with GTP, resulting in an active protein. GTPase-activating proteins (GAPs) act as negative regulators by stimulating the intrinsic GTPase activity of the protein to return it to the inactive form (11, 58). A third class of regulatory proteins, the guanine nucleotide dissociation inhibitors (GDIs), maintain the existing nucleotide state of some GTP-binding proteins such as Rho and Rab (27, 42).
Mg2+ plays a critical role in the control of guanine nucleotide exchange and GTP hydrolysis in the well-studied Ras-like GTP-binding proteins. Crystallographic studies of several GTP-binding proteins revealed a single Mg2+ ion in the guanine nucleotide binding pocket (9, 36, 46, 47, 55). When the concentration of Mg2+ is low, the protein exists in an open conformation and exchange of GDP for GTP is enhanced. In the presence of Mg2+, the protein-nucleotide complex exists in a closed conformation and exchange of the bound nucleotide occurs very slowly (6, 13, 19, 29). Thus, physiological levels of Mg2+ are sufficient to inhibit nucleotide exchange (6, 12, 18, 37), and it is thought that guanine nucleotide exchange in vivo is controlled by a GEF that overcomes the Mg2+ inhibition (19, 37, 38).
With the rapid expansion of bacterial genome sequence data, it is becoming clear that the bacterial Ras-like GTP-binding proteins are widespread and are likely to play critical cellular roles. Novel G-protein subfamilies such as Era (1, 5) and Obg (8, 15, 25, 33, 43, 45, 52, 57) are also present in archaea and eukaryotes. The bacterial Obg proteins are essential for cell viability (25, 34, 57) and appear to play critical roles in regulating DNA replication and/or cell differentiation (22, 34). It has been proposed that the guanine nucleotide state occupancy of the bacterial Obg-like proteins is directly controlled by the intracellular GTP pool (33, 34, 61). These proteins would be turned on (in the GTP-bound state) under growth conditions and off (in the GDP-bound state) under starvation conditions. Furthermore, it has been proposed that these proteins are involved in communicating changes in the GTP pool to pathways that are involved in cellular processes that occur under starvation conditions (33, 34). The most direct evidence comes from studies of the Obg protein in sporulating bacteria. For Bacillus subtilis, Obg protein was shown previously to be involved in communicating signals to the Spo0A sporulation pathway (60). The B. subtilis Obg protein binds to GDP with an affinity in the micromolar range and displays slow GTP hydrolysis (61). In Streptomyces spp., specific obg mutant alleles display dominant effects on sporulation (34). Overproduction of Streptomyces spp. Obg does not affect vegetative growth but does prevent the development of aerial mycelium (33). Furthermore, addition of decoyinine (a specific inhibitor of GMP synthetase) results in the restoration of aerial mycelium production in Streptomyces spp. strains overproducing Obg (34). These data suggest that the onset of differentiation is determined by the balance of Obg protein and GTP levels.
These studies clearly demonstrate a role of the Obg proteins in sporulation of B. subtilis and Streptomyces spp. However, Obg homologs are present in a diverse array of nonsporulating organisms and thus must play a different cellular role in these organisms. If the control of Obg-like proteins is mediated by intracellular GTP pools, then the guanine nucleotide binding and exchange and GTP hydrolysis parameters of these proteins should be consistent with this mode of regulation.
We are investigating the role of the Obg-like protein CgtA in Caulobacter crescentus, a nonsporulating bacterium. CgtA is essential for cell viability and is present throughout the C. crescentus cell cycle (25). Using fluorescent guanine nucleotide analogs (N-methyl-3′-O-anthranoyl–GDP [mant-GDP] and mant-GTP), we show here that the CgtA protein binds to mant-GDP and mant-GTP in an Mg2+ dose-dependent manner and that optimal complex formation occurs at physiological Mg2+ concentrations. CgtA displays moderate affinity for both GDP and GTP. Furthermore, CgtA, unlike the well-characterized Ras-like GTP-binding proteins, displays a high in vitro exchange rate constant for either nucleotide. Hydrolysis of GTP is relatively slow. Thus, the in vitro guanine nucleotide binding and exchange and GTP hydrolysis of CgtA support a model whereby the control of CgtA is directly mediated by changes in the intracellular guanine nucleotide pool without the benefit of GEFs or GAPs.
MATERIALS AND METHODS
Overexpression and purification of CgtA.
An NcoI site at the ATG translation initiation codon of the cgtA gene was generated by PCR amplification with the primers CGTA-NcoI (5′ GGACCCCATGGAATTCTTGGACCA 3′) and PROB1 (5′ GCGGCTCGAAAGCTTCTTCC 3′). The resulting 1.4-kb NcoI-to-HindIII fragment was cloned into the pET28a vector (Novagen) to create pJM625. In this vector, the ribosomal binding site was provided by the vector and expression of the transcriptional fusion was under the control of the T7 promoter, which, in turn, was expressed under the control of placUV. Transformation of plasmid pJM625 into the BL21DE3 strain of Escherichia coli and induction of mid-log-phase cells with 1 mM isopropyl-β-d-thiogalactopyranoside (IPTG) for 2 h at 37°C resulted in the accumulation of a prominent CgtA band as visualized by sodium dodecyl sulfate-polyacrylamide gel electrophoresis (SDS-PAGE).
A liter of cells was pelleted (6,000 × g, 5 min, 4°C), resuspended in 50 ml of TDGM (50 mM Tris-HCl [pH 8], 1 mM dithiothreitol, 10% glycerol, 5 mM MgCl2) supplemented with 1 mM phenylmethylsulfonyl fluoride and 10 μM GTP, and lysed by two passages through a French pressure cell. Approximately 50% of the CgtA protein was present in the supernatant (28,000 × g for 30 min, 4°C) of the cell extract. The supernatant was passed through 0.45- and 0.22-μm-pore-size filters and applied to a 20-ml (1.5 by 15 cm) Cibacron Blue 3GA agarose (Sigma) column (0.5 to 1 ml/min). The column was washed with 200 ml of TDGM and eluted with a 300-ml linear gradient of TDGM with 0 to 1 M KCl. The appropriate CgtA-containing fractions were pooled, diluted twofold with TDG (TDGM without MgCl2), and loaded on a 50-ml (1.5 by 30 cm) Toyopearl DEAE-650M (TosoHaas) column (1.5 to 5 ml/min). CgtA was eluted with a 200-ml linear gradient of TDG with 0 to 400 mM KCl. The relevant fractions were pooled, concentrated with a Centriprep-10 concentrator (Amicon), and applied to a 100-ml (1.5 by 70 cm) Sephadex G-75 (Pharmacia) gel filtration column (0.5 to 1 ml/min). CgtA was eluted with TDG containing 100 mM KCl.
The concentration of CgtA was first determined by the Bradford method (4) and then by UV absorption once the extinction coefficient was determined (7).
UV cross-linking.
Purified CgtA (10 μg/sample; 5 μM) was incubated with 10 μCi of [α-32P]GTP (3,000 Ci/mmol; NEN Life Science Products) in 50 μl of 1× binding buffer (50 mM Tris-HCl [pH 8.0], 50 mM KCl, 2 mM dithiothreitol, 5 μM ATP, 1 mM EDTA, and 10% [wt/vol] glycerol) supplemented with 5 mM Mg2+. In competition samples, 40 μM (each) nonradioactive nucleotide (ATP, GTP, GDP, or GMP) was added separately. The Mg2+ dependence of CgtA interaction with radiolabeled GTP was determined by incubating 3 μM CgtA with 0.5 μM [α-32P]GTP in binding buffer with or without 12 mM Mg2+. All samples were incubated on ice for 5 min, and the bound [α-32P]GTP was cross-linked to CgtA by UV treatment (254 nm, 1 J/cm2). Radiolabeled CgtA-GTP complexes were separated by SDS-PAGE. The gel was vacuum dried and exposed to X-ray film.
Synthesis of mant-GDP.
mant-GDP, mant-GTP, and mant-β-γ-methyleneguanosine-5′-triphosphate (mant–GMP-PCP) were synthesized by reaction of GDP, GTP, or GMP-PCP (Sigma) with N-methylisatoic anhydride (Acros) as described previously (23). To purify the mant-nucleotide, 3 ml of the synthesis reaction mixture was loaded on a 50-ml (1.5 by 30 cm) DEAE Sepharose Fast Flow (Pharmacia) column. The column was washed (2 ml/min) with 100 ml of 50 mM Tris-HCl buffer (pH 7.5), and the nucleotides were eluted with a 1,000-ml linear gradient of 50 mM Tris-HCl (pH 7.5) with 0 to 1 M KCl. The mant-nucleotide fractions were combined, diluted with 3 volumes of dH2O, and desalted by passage over a DEAE column (1.5 by 30 cm) preequilibrated with 0.2 M TBK (triethylamine hydrogen carbonate, pH 7.5). The column was washed with 100 ml of 0.2 M TBK, and the nucleotides were eluted (0.5 ml/min) with 100 ml of 2 M TBK. The relevant mant-nucleotide fractions were combined, freeze-dried, and resuspended in 1 ml of dH2O. The purity of the nucleotides was analyzed by thin-layer chromatography, and the absorption spectra (excitation at 240 to 400 nm) confirmed the identity of each mant-nucleotide (14). The concentration was determined by measuring the optical density at 252 nm (23). Aliquots were frozen at −80°C. The mant-GTP used in the initial phases of this study was a generous gift from Richard Neubig (39, 40).
Fluorescence measurements.
Fluorescence measurements were performed with a Shimadzu RF-5301PC spectrofluorometer equipped with a Hi-Tech SFA-20 stopped-flow apparatus. Unless otherwise indicated, all assays were performed at 30°C, and mant-nucleotide fluorescence was monitored with an excitation wavelength of 361 nm (slit width of 1.5 nm) and an emission wavelength of 446 nm (slit width of 20 nm). Emission and excitation profiles of the mant-nucleotides were generated from 0.3 μM free mant-GDP and mant-GTP in binding buffer. CgtA-guanine nucleotide complexes were generated by prebinding 10 μM CgtA with 0.3 μM mant-GDP or mant-GTP in binding buffer supplemented with 5 or 12 mM Mg2+, respectively, for 10 min at 30°C. As a negative control, denatured CgtA was generated by incubating CgtA for 10 min at 90°C in binding buffer with 0.7 mM SDS prior to the binding assay.
The Mg2+ dependence of CgtA interaction with mant-GTP and mant-GDP was determined by examining mant-GTP and mant-GDP fluorescence of 0.5 μM CgtA and 0.3 μM mant-nucleotides in 1× binding buffer (without EDTA) supplemented with 0 to 50 mM Mg2+. The data were corrected for the slight reduction in fluorescence due to dilution and the effect of Mg2+ quenching on the mant-nucleotide.
To monitor dissociation of CgtA–mant-nucleotide complexes, 1 μM mant-nucleotide was prebound and saturated with purified CgtA (approximately 3 μM). Dissociation of CgtA–mant-nucleotide complexes was initiated by rapidly mixing 150 μM GDP or GTP as a competitor, and the decrease of peak fluorescence (excitation slit width, 5 nm; emission slit width, 20 nm) was observed.
To monitor dissociation of GDP and GTP from CgtA, CgtA-guanine nucleotide complexes were generated by prebinding 1 μM CgtA with 0.3 μM GDP or GTP in binding buffer supplemented with 5 or 12 mM Mg2+, unless otherwise indicated. mant-GDP or mant-GTP (10 μM) was used as a competitor for approximately 0.3 μM prebound CgtA-guanine nucleotide complexes. In this case, we monitored the fluorescence resonance energy transfer (from tryptophan to the mant group) (21, 40) of CgtA binding to excess mant-GDP (excitation at 281 nm and emission at 446 nm). Data were collected at 20-ms intervals and curve fitted to a one-phase exponential decay equation. The initial rapid phase due to the association of unoccupied CgtA with mant-GDP was omitted due to the limitations of our recording device. The dissociation rate constant (kd) of each nucleotide was determined by averaging the kd values from a minimum of 10 trials.
The intrinsic GTPase activity of CgtA at 30°C was determined by monitoring the decrease in fluorescence of the CgtA–mant-GTP complex over time. mant-GTP or the nonhydrolyzable analog, mant–GMP-PCP (0.5 μM), was prebound with excess CgtA (10 μM) in 1× binding buffer supplemented with 12 mM MgCl2. The peak fluorescence was recorded over 3 h at 1-min intervals. The data were curve fitted to a first-order (single exponential decay) hydrolysis equation, and the rate constant (kh) and half-life (t1/2) of a single-turnover mant-GTP hydrolysis were determined by averaging the kh and t1/2 from four trials.
Equilibrium binding assays.
The affinity of CgtA for guanine nucleotides at 30°C was determined by an equilibrium centrifugal ultrafiltration assay (35). Each binding reaction mixture contained 200 μl of 1× binding buffer, 5 μM CgtA, and [8-3H]GDP (106 dpm/nmol; Amersham) or [α-32P]GTP (4 × 106 cpm/nmol) ranging from 0.1 to 8 μM. MgCl2 (5 or 12 mM) was added as indicated. Aliquots of 60 μl were withdrawn for scintillation counting, and the remaining reaction mixtures were transferred to Amicon Microcon-10 spin concentrators. After centrifugation at 16,000 × g for 8 min, 60-μl aliquots were withdrawn from the filtrate for scintillation counting and the free nucleotide concentration was calculated according to a standard curve. The concentration of bound nucleotide was calculated as the difference between total and free nucleotide concentrations. The equilibrium dissociation constants, Kd, were determined by curve fitting (Kaleidagraph 3.09; Synergy Software) the binding plots (bound versus total nucleotide) to a hyperbolic binding function. Assays were done in triplicate.
The equilibrium binding constant of CgtA–mant-GDP was also determined by examining peak fluorescence intensities (excitation slit width, 1.5; emission slit width, 15 nm) of binding reaction mixtures (5 mM Mg2+) containing serial dilutions of mant-GDP (0.2 to 4 μM) with or without CgtA (4 μM concentration for nonsaturating; 50 μM concentration for saturating). Triplicate experiments were curve fitted and averaged.
RESULTS
CgtA is a GDP-GTP-binding protein.
In order to determine the in vitro guanine nucleotide-binding properties of CgtA, we overexpressed and purified the protein. We obtained approximately 140 mg of at least 95% pure CgtA per liter. Purified CgtA migrated at 40 kDa on SDS-PAGE gels, 2 kDa larger than the predicted value of 38 kDa. The purified protein was subjected to N-terminal sequencing and found to begin with Met, indicating that it was not N-terminally modified. Additional sequencing cycles confirmed that the purified protein was indeed CgtA. Electrospray mass spectrometry indicated that the mass of intact CgtA is 37,931 ± 16 Da. Due to the generation of the NcoI restriction site at the beginning of the cgtA gene, the purified protein contained a Lys-to-Glu mutation at the second amino acid. We have demonstrated that this change appears to be silent in vivo, as the mutant allele complements a cgtA-null allele (data not shown).
The association of CgtA with several nucleotides was determined by examining their ability to directly outcompete the binding of [α-32P]GTP (Fig. 1). CgtA binds [α-32P]GTP rapidly. This interaction is efficiently competed with GTP or GDP. However, little effect on the binding of [α-32P]GTP was observed in the presence of a >400-fold excess of GMP or ATP (Fig. 1). Thus, CgtA displays an inherent specificity for the di- and triphosphate forms of guanine nucleotides.
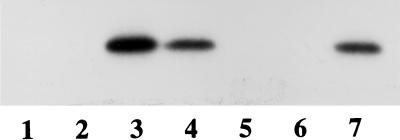
FIG. 1.
CgtA binds to GDP and GTP. Shown is an autoradiogram of CgtA-[α-32P]GTP complexes separated by SDS-PAGE. CgtA (5 μM) was incubated with 10 μCi of [α-32P]GTP in binding buffer supplemented with 5 mM Mg2+. Without UV cross-linking (lanes 1 and 2), no CgtA-[α-32P]GTP complexes are observed in either the absence (lane 1) or the presence (lane 2) of competing nucleotide (>400-fold ATP added). However, UV cross-linking (lanes 3 to 7) resulted in CgtA-[α-32P]GTP complexes detected without competing nucleotide (lane 3) or in the presence of >400-fold ATP and GMP (lanes 4 and 7, respectively). CgtA-[α-32P]GTP binding is inhibited by excess GTP and GDP (lanes 5 and 6, respectively).
mant-guanine nucleotide analogs are useful probes of G-protein activation and conformational state (19, 31, 32, 40, 41, 44). Changes in mant fluorescence reflect the hydrophobic environment of the nucleotide analog. We monitored the change in fluorescence of the guanine nucleotide analogs, mant-GTP and mant-GDP, as an indication of CgtA–mant-guanine nucleotide binding.
The emission (data not shown) and excitation profiles (Fig. 2) of free mant-GDP and mant-GTP were similar to profiles described previously (31, 32, 40). mant-GDP and mant-GTP nucleotides had an optimal excitation of 361 nm and an optimal emission at 446 nm. Figure 2 shows emissions at 446 nm at varying excitation wavelengths. Aside from the major excitation peak at 361 nm, an additional excitation peak at 256 nm due to the excitation of the nucleotide was observed. Preincubation of the mant-nucleotides with denatured CgtA resulted in an excitation profile similar to that of free mant-nucleotide, but with an additional excitation peak at 285 nm. Denatured CgtA did not enhance the mant-GTP or mant-GDP excitation at 361 nm, indicating that CgtA–mant-nucleotide complexes did not form.
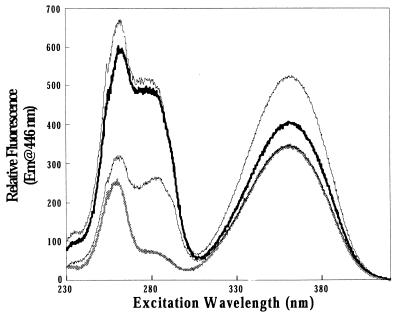
FIG. 2.
Excitation spectra of free and CgtA-complexed mant-nucleotides. CgtA (10 μM) was incubated with 0.3 μM mant-nucleotide as described in Materials and Methods. Shown are the relative excitation spectra of mant-GDP (thick, light line), CgtA–mant-GDP (thick, dark line), CgtA–mant-GTP (thin, light line), and denatured CgtA incubated with mant-GTP (thin, dark line). The spectrum of free mant-GDP overlays the free mant-GTP spectrum, and the denatured CgtA incubated with mant-GDP overlays the denatured CgtA incubated with mant-GTP (data not shown). Em, emission.
Binding of CgtA to mant-GTP and mant-GDP nucleotides led to a substantial increase in mant-nucleotide fluorescence, providing direct evidence that CgtA binds guanine nucleotides. An increase in fluorescence was observed in the CgtA–mant-GTP and CgtA–mant-GDP complexes upon excitation at 361 nm (optimum for mant), 256 nm (optimum for the nucleotide), and 285 nm (optimum for tryptophan). On binding to CgtA, mant-GTP showed a 1.5- to 1.6-fold enhancement of fluorescence intensity and mant-GDP showed a 1.2- to 1.3-fold enhancement when excited at 361 nm (Fig. 2). A significant increase in CgtA–mant-GTP and CgtA–mant-GDP fluorescence (excitation at 285 nm) was also observed due to the resonance energy transfer from tryptophan to the mant group. CgtA contains five tryptophan residues, none of which are in the proposed guanine nucleotide binding pocket. However, one or more of these tryptophan residues must be spatially located such that transfer of fluorescence energy can occur.
CgtA binds guanine nucleotides at physiological Mg2+ concentrations.
In the well-studied G proteins, millimolar Mg2+ concentrations inhibit exchange (6, 10, 12, 13, 19, 28, 29, 37) and binding (12, 13, 28, 49, 50, 56, 59) of guanine nucleotides. We determined the effects of Mg2+ on guanine nucleotide binding to CgtA by measuring the increase in mant-GTP and mant-GDP fluorescence upon binding to CgtA at different added Mg2+ concentrations (Fig. 3).
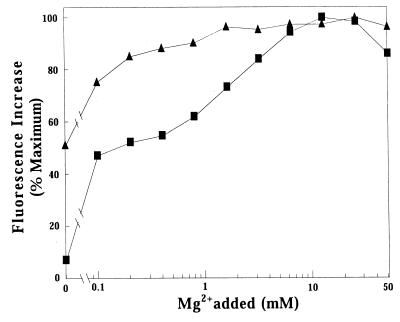
FIG. 3.
The effect of added Mg2+ on the binding of mant-GDP or mant-GTP to CgtA. Exchange reactions were performed at 30°C with 0.5 μM CgtA, 0.3 μM mant-nucleotide, and the indicated [Mg2+] added. Fluorescence of CgtA–mant-GTP (squares) and CgtA–mant-GDP (triangles) was measured. Shown is the profile of Mg2+ dependence as the percent maximal fluorescence versus [Mg2+] on a log scale. An additional experiment was carried out with the addition of 1 mM EDTA to the buffer without Mg2+. The values for relative fluorescence for mant-GTP and mant-GDP in buffer containing 1 mM EDTA were 8 and 51%, respectively.
The binding profiles obtained for mant-GDP versus mant-GTP at varying concentrations of Mg2+ were different. CgtA bound mant-GDP at 50% of maximal levels in the absence of Mg2+. The addition of 1 mM EDTA had no effect on the relative fluorescence, indicating that little Mg2+ was present in the solutions. Optimal binding occurred at 5 mM Mg2+, although CgtA-GDP complexes formed over a wide range of Mg2+ concentrations (Fig. 3, triangles). In contrast, Mg2+ had a strong dose-dependent effect on CgtA binding to mant-GTP (Fig. 3, squares). In the absence of supplemental Mg2+ (with or without EDTA added), less than 10% of maximal CgtA–mant-GTP complexes formed. Efficient CgtA–mant-GTP complex formation occurred between 5 and 15 mM Mg2+ and was inhibited at higher concentrations. Most significantly, CgtA–mant-GDP and CgtA–mant-GTP complexes formed most efficiently at physiologically relevant Mg2+ concentrations (in the millimolar range).
To confirm that the increase in fluorescence is due to an increase in the interaction between CgtA and the guanine nucleotide and not simply due to the conformational change in preexisting CgtA–mant-GTP complexes after Mg2+ binding, we assayed the binding of CgtA to radiolabeled GTP by UV cross-linking (Fig. 4). The resulting autoradiograph confirms that CgtA-GTP complexes were readily formed in the presence of 12 mM Mg2+ added (Fig. 4, lane 4) but were barely detectable without the addition of Mg2+ (Fig. 4, lane 3). Samples that were not UV cross-linked do not display any radioactivity (Fig. 4, lanes 1 and 2). Finally, we have also examined the binding of CgtA to radiolabeled GDP and observed ∼80% of control binding in the absence of Mg2+ (data not shown). Thus, optimal binding of modified and unmodified guanine nucleotides occurred at physiological Mg2+ concentrations, and GDP and GTP showed different Mg2+ dependence profiles.
FIG. 4.
CgtA-GTP complex formation requires Mg2+. Shown is an SDS-PAGE autoradiogram of CgtA-[α-32P]GTP. Each lane was loaded with 50 μl of 3 μM CgtA incubated with 0.5 μM [α-32P]GTP. Samples 1 and 3 had no Mg2+ added (and 1 mM EDTA), while samples 2 and 4 were supplemented with 12 mM MgCl2. Samples 3 and 4 were UV cross-linked prior to loading on the gel.
CgtA binds guanine nucleotides with moderate affinity.
To determine the intrinsic GDP and GTP equilibrium dissociation constants, we incubated CgtA with varying amounts of radiolabeled GDP or GTP and quantified the amount of CgtA-GDP or CgtA-GTP formed in the presence of 5 or 12 mM Mg2+ added by an equilibrium centrifugal ultrafiltration assay (35). These data result in a hyperbolic plot reflecting a single binding site with average apparent equilibrium dissociation constants, Kd, for CgtA-[8-3H]GDP of 0.56 ± 0.06 μM (Fig. 5B; Table 1) and 0.52 ± 0.03 μM (Table 1) in the presence of 5 and 12 mM Mg2+, respectively. The average apparent Kd values for CgtA-[α-32P]GTP were 1.27 ± 0.16 μM and 1.11 ± 0.13 μM in 5 and 12 mM Mg2+, respectively (Table 1). Under these conditions, CgtA displayed moderate affinity for both nucleotides with a twofold preference for GDP over GTP. The slightly stronger affinity of CgtA for [α-32P]GTP at 12 mM Mg2+ is consistent with the increase in fluorescence observed for CgtA–mant-GTP complexes at 12 mM Mg2+ added.
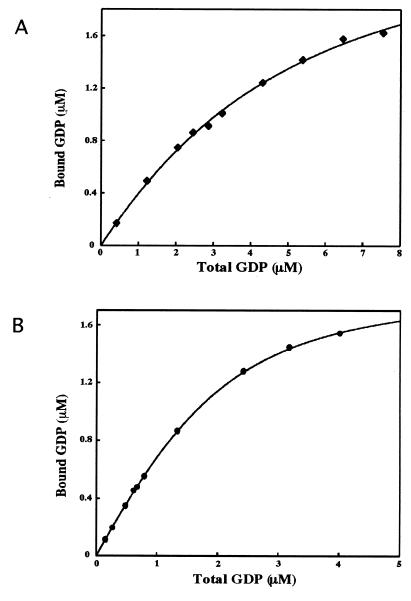
FIG. 5.
Equilibrium binding of CgtA to GDP. CgtA (5 μM) was incubated at 30°C with increasing concentrations of [8-3H]GDP in binding buffer containing 0 (A) or 5 (B) mM MgCl2. Aliquots were removed, and bound GDP was quantitated. Shown is the hyperbolic curve for GDP binding. The equilibrium binding constants, Kd, for GDP from triplicate experiments are 3.3 ± 0.2 and 0.56 ± 0.06 μM for 0 and 5 mM Mg2+, respectively.
TABLE 1.
Nucleotide binding to CgtAa
| Ligand | Mg2+ (mM) | Kd (μM) |
|---|---|---|
| GDP | 0 | 3.3 ± 0.2 |
| 5 | 0.56 ± 0.06 | |
| 12 | 0.52 ± 0.03 | |
| GTP | 0 | 65 ± 9 |
| 5 | 1.27 ± 0.16 | |
| 12 | 1.11 ± 0.13 |
Equilibrium binding constants, Kd, of CgtA-guanine nucleotides at different added [Mg2+] were determined as described in Materials and Methods.
In the absence of Mg2+, CgtA–mant-GTP-enhanced fluorescence is severely impaired and a reduction in the level of CgtA–mant-GDP-enhanced fluorescence is observed (Fig. 3). A parallel increase in the equilibrium binding constants for GTP and GDP was also observed in the absence of Mg2+ (Table 1). Without Mg2+, the affinity of CgtA-GDP was reduced 6-fold (compared to that for 5 mM Mg2+ added) whereas the affinity of CgtA-GTP was reduced 59-fold (compared to that for 12 mM Mg2+).
We also used the increase in mant-GDP fluorescence to determine the equilibrium binding constant of CgtA for mant-GDP. Peak fluorescence values of binding reaction mixtures containing varying concentrations of mant-GDP with or without CgtA were determined, and a Kd of 0.4 ± 0.1 μM at 5 mM Mg2+ was obtained. Therefore, under these conditions, CgtA binds mant-GDP with an affinity similar to that of unmodified GDP.
CgtA displays rapid guanine nucleotide dissociation kinetics.
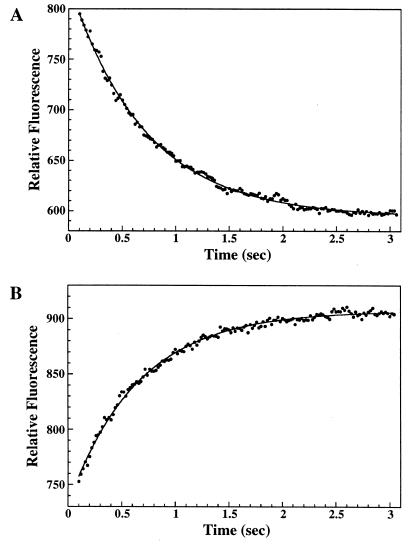
mant-guanine nucleotides are powerful tools for kinetic studies, since data can be collected continuously and in real time. Thus, we could determine the dissociation rate constant of the mant-nucleotides by monitoring the decrease in fluorescence that accompanied displacement of bound mant-nucleotide by a large excess of unlabeled nucleotide. CgtA was prebound to mant-GDP or mant-GTP until apparent saturation was achieved. Excess unlabeled nucleotide (GDP) was then added, and the rate constant of fluorescence decrease was measured (Table 2; Fig. 6A). In all cases, the dissociation followed a single exponential curve. The release of mant-GDP at 30°C occurred with an average rate constant of ∼1.4 s−1, and the release of mant-GTP occurred with an average rate constant of 1.3 to 1.4 s−1 (Table 2). The dissociation rate constants were essentially identical when GTP was utilized as the competing nucleotide (data not shown). We also obtained a rough measurement of the association rate constant (ka) for mant-GDP in the presence of 5 mM Mg2+ by using 0.05 μM CgtA with 0.2 to 1.5 μM mant-GDP. The observed ka value (>1 μM−1 s−1) was consistent with the calculated ka (3.6 μM−1 s−1).
TABLE 2.
Nucleotide exchange ratesa
| Nucleotide | Mg2+ (mM) | kd (s−1) |
|---|---|---|
| mant-GDP | 0 | 12.3 ± 0.7 |
| 5 | 1.44 ± 0.01 | |
| 12 | 1.43 ± 0.04 | |
| mant-GTP | 5 | 1.40 ± 0.02 |
| 12 | 1.28 ± 0.02 | |
| GDP | 0 | 11.2 ± 0.3 |
| 5 | 1.45 ± 0.01 | |
| 12 | 1.48 ± 0.06 | |
| GTP | 5 | 1.57 ± 0.03 |
| 12 | 1.54 ± 0.01 |
Dissociation rate constants, kd, of guanine nucleotides at different added [Mg2+] were determined as described in Materials and Methods by monitoring fluorescence with mant-nucleotides. Shown are the averages of >10 experiments.
FIG. 6.
Dissociation of guanine nucleotides from CgtA. (A) Dissociation of mant-GDP. CgtA–mant-GDP (generated by prebinding 1 μM mant-GDP with 3 μM CgtA) in 5 mM Mg2+ was rapidly mixed with excess GDP (150 μM) in a stopped-flow fluorimeter, and the change in the relative fluorescence intensity over time was monitored, giving a kd of 1.44 ± 0.01 (average of 10 trials). (B) Dissociation of GDP. CgtA-GDP complexes (generated by prebinding 1 μM CgtA with 0.3 μM GDP) in 5 mM Mg2+ were rapidly mixed with excess mant-GDP (10 μM) in a stopped-flow fluorimeter, and the change in fluorescence intensity over time was monitored, resulting in a kd of 1.45 ± 0.01 (average of 10 trials). Representative profiles for the dissociation of guanine nucleotides are shown. The curve-fitted data are shown with a solid line. Relative fluorescence equals a + be−kdt).
The dissociation rate constants of unmodified nucleotides from CgtA-GDP and CgtA-GTP complexes were also determined. To reduce the background fluorescence of the mant-nucleotide, we monitored the fluorescence energy transfer from tryptophan to the mant group (21, 40). Prebound CgtA-nucleotide complexes were mixed with excess (>30-fold) mant-GDP, and the increase of fluorescence that accompanied the exchange of the unlabeled bound nucleotide for the mant-nucleotide was monitored (Fig. 6B). The exchange rate of either GDP or GTP for mant-GDP was ∼1.5 s−1 (Table 2). These data demonstrate that CgtA displays a rapid in vitro exchange of guanine nucleotides and that the exchange rate constants of GTP and GDP are essentially equivalent.
In the absence of Mg2+, Ras-like GTP-binding proteins typically display an enhanced guanine nucleotide exchange rate constant. We determined that the dissociation rate constants of mant-GDP and GDP were eightfold higher in the absence of Mg2+ than in its presence (Table 2). The dissociation rate constants of mant-GTP and GTP could not be determined by this assay due to weak binding of CgtA to the nucleotides (Table 1) and the resulting low increase in fluorescence signal under these conditions (Fig. 3).
GTP hydrolysis by CgtA.
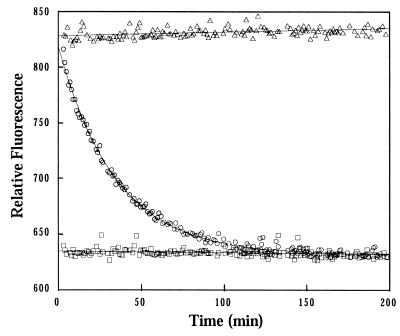
As has been shown previously for other GTP-binding proteins (31, 32, 39–41, 51), the CgtA–mant-GTP complexes displayed a peak fluorescence that is approximately 30% greater than that of CgtA–mant-GDP complexes. Thus, intrinsic GTPase activity of purified CgtA could be determined by measuring the reduction in fluorescence that accompanied the single-turnover conversion of bound mant-GTP to bound mant-GDP (Fig. 7). The decrease in fluorescence could be fitted to a single exponential curve with a first-order rate constant, kh, of 5.0 × 10−4 s−1 or a t1/2 of 23 ± 2 min. The rate constant for hydrolysis of mant-GTP by CgtA was well within the range observed under similar conditions for eukaryotic GTP-binding proteins (51).
FIG. 7.
Hydrolysis of mant-GTP by CgtA. The fluorescence intensity of CgtA–mant-GTP complexes (open circles), nonhydrolyzable CgtA–mant–GMP-PCP complexes (open triangles), and CgtA–mant-GDP (open squares) was monitored over time. The reduction in fluorescence that accompanies hydrolysis of mant-GTP in the CgtA–mant-GTP complexes fit a single exponential decay, and the single-turnover rate constant was 5.0 × 10−4 s−1.
DISCUSSION
We describe here the biochemical characterization of the C. crescentus CgtA protein, a member of the Obg family of GTP-binding proteins. Purified CgtA bound specifically to GTP and GDP with moderate affinity (in the micromolar range at 30°C) and bound with a twofold-higher affinity to GDP than to GTP. A similar equilibrium binding constant was obtained for the B. subtilis Obg protein for GDP (0.93 μM at 25°C [61]). Thus, it is possible that relatively weak binding to guanine nucleotides is a hallmark feature of this subfamily of proteins, in contrast to the high binding affinity (typically in the nanomolar range) reported for the vast majority of monomeric GTP-binding proteins.
Mg2+ is a critical cofactor for GDP and GTP binding by most, if not all, GTP-binding proteins (see references 20 and 54). For example, at millimolar levels of Mg2+, Ras exists in a closed conformation and the exchange rate of GDP for exogenous GTP is relatively low in vitro (t1/2 ∼ 60 min). The rate-limiting step has been shown to be the loss of bound nucleotide, and removal of Mg2+ allows free exchange of exogenous nucleotides. It is currently believed that the role of GEF proteins is to overcome the Mg2+ inhibition to allow more rapid exchange of the guanine nucleotide (38). In this study, we used radiolabeled and fluorescent GDP and GTP analogs to investigate the effect of Mg2+ on the interaction between these guanine nucleotides and CgtA. We demonstrated that purified CgtA binds mant-GTP in an Mg2+-dependent fashion, while its interaction with mant-GDP was relatively insensitive to a wide range of Mg2+ concentrations. Surprisingly, millimolar concentrations of Mg2+ did not inhibit binding of mant-GDP or mant-GTP to CgtA. In fact, optimal binding occurred at physiologically relevant Mg2+ concentrations. Thus, the Mg2+ requirements for CgtA-GDP and CgtA-GTP complex formation are unique.
The in vitro exchange of guanine nucleotides by CgtA was shown to be rapid at physiological levels of Mg2+. The dissociation rate constants for mant-GTP and mant-GDP at optimal [Mg2+] are 1.28 and 1.44 s−1, respectively. High exchange rate constants were observed regardless of whether GTP or GDP was used as the competing nucleotide. Similar dissociation rate constants were observed for unmodified GDP and GTP (Table 2), demonstrating that the mant-nucleotides behave as close analogs of GDP and GTP and can be used to accurately monitor CgtA-guanine nucleotide interactions. We observed an eightfold increase in the GDP dissociation rate constant in the absence of Mg2+ (Table 2). However, given the high dissociation rate constant in the presence of Mg2+, this difference may not be of biological significance. In contrast, the 59-fold decrease in affinity for GTP in the absence of Mg2+ could indicate that the magnesium ion plays a regulatory role in the control of CgtA-GTP complex formation.
Clearly, the guanine nucleotide binding and exchange and Mg2+ requirements of CgtA are different from those of the majority of Ras-like GTP-binding proteins. It is likely that some of the unique biochemical features of CgtA are due to nonconventional amino acids in the conserved guanine nucleotide binding pocket of the protein. For example, the amino acid residues G12 and Q61, critical for Ras function, are absent in CgtA. In addition, the G5 domain is not easily recognized (25). The amino acid at position 61 is part of the nucleotide binding site and is normally a glutamine residue; in CgtA, this amino acid residue is a leucine. In Ras, a Q61L mutation increases the GDP dissociation rate. The Rap2 protein has a threonine instead of glutamate at this position and displays an in vitro GDP dissociation rate constant fivefold higher than that of Ras (24). FtsY and Ffh2, two proteins that lack the invariant G12 and Q61 residues, also display a rapid exchange and micromolar affinities for guanine nucleotides (17, 30). However, in these proteins, other differences such as an insertion into the effector loop may also contribute to differences in nucleotide binding. An Mg2+ enhancement of guanine nucleotide binding has been observed for the eukaryotic Rad protein, although in this case, both GTP and GDP show a strong dose-dependent requirement for the cation (62). Rad also has nonconventional amino acids in the critical G1 and G3 binding domains (62).
A wide range of single-turnover hydrolysis rate constants have also been observed for the Ras-like GTP-binding proteins. Q61 has also been proposed to play an essential role in the hydrolysis of GTP, by activating a water molecule for the nucleophilic attack on the γ-phosphate (36, 46). Although CgtA has a leucine at the Q61 position, it displays a hydrolysis rate constant equivalent to that of Ras (t1/2 = 23 and 30 min, respectively [12]). Thus, the differences in the requirements for Mg2+, binding affinities, and exchanges of nucleotides between CgtA and Ras-like GTP-binding proteins may reflect differences in the guanine nucleotide binding pockets of these proteins.
The guanine nucleotide occupancy of GTP-binding proteins is determined by the affinity of the protein for guanine nucleotides and the rates of guanine nucleotide exchange and GTP hydrolysis. CgtA displays a rapid exchange of either GDP or GTP at physiological levels of Mg2+ and a relatively low hydrolysis rate. Thus, in vitro, the guanine nucleotide occupancy of the protein may be dictated by the guanine nucleotide concentration. The in vitro activities described here underscore that, in vivo, CgtA activity may be controlled in a manner distinct from that of the well-characterized Ras-like GTP-binding proteins. We envision three possible scenarios to explain the unique in vitro binding and exchange parameters described here.
First, it is possible that CgtA is controlled by GEF proteins and GAPs in vivo but that the GEF activity is associated with CgtA itself. CgtA may be a bimodal protein containing both a Ras-like GTP-binding domain and a guanine nucleotide exchange domain (GEF activity) at its N terminus. The Fts Y protein of E. coli has previously been proposed to be such a GEF-GTPase protein (30). Within the Obg-GTP1 subfamily, there are two distinct classes of proteins, (i) an N-terminally extended form that possesses an ∼150-amino-acid glycine-rich N terminus preceding the Ras-like domain and (ii) a C-terminally extended form that lacks the N-terminal extension but has additional residues at the C terminus. To date, all known bacterial Obg-like proteins, including CgtA, possess the N-terminal extension and all archaeal forms have the C-terminal extension, while both forms of Obg-like proteins can be identified in eukaryotes.
The unique C terminus found in archaeal and eukaryotic Obg-like proteins may play a role in protein-protein interactions. For example, the mouse protein DRG interacts specifically with the helix-loop-helix domain of the transcription factor TAL1 via two amphipathic helices at the C terminus of DRG (26). Similar amphipathic helices are readily identified by BLAST searches in all members of the Obg-GTP1 family that possess the C-terminal extension.
The role of the N-terminal extension is unknown. It is possible that this motif acts as an exchange factor; however, this seems unlikely. Although this domain plays a critical role in the function of B. subtilis Obg protein, a nonfunctional Obg protein harboring amino acid changes in the N terminus bound GTP with the same affinity as did the wild-type protein (61). An alteration in the rate of dissociation would usually result in an increase in the observed affinity. Moreover, the N-terminal sequence does not resemble the amino acid sequence of known GEF proteins.
A second possibility is that, in vivo, the exchange of guanine nucleotides by CgtA is suppressed by a GDI. GDIs maintain the existing nucleotide state in proteins such as Rho and Rab. Although a CgtA-specific GDI could modify or dictate the in vivo exchange properties of CgtA, to date no GDI proteins have been identified in bacteria, either by function or by similarity searches.
The third explanation for the unique guanine nucleotide-binding characteristics of CgtA is that the exchange of guanine nucleotides by CgtA is controlled directly by the intracellular pools of guanine nucleotides and the relative affinity of CgtA for GTP and GDP, without the benefit of GEFs or GAPs. In the intracellular milieu during exponential growth, the GTP concentration greatly exceeds that of GDP, and CgtA, with an equilibrium binding constant in the micromolar range and a high guanine nucleotide exchange rate, would be predicted to be predominantly GTP associated. However, upon entry into stationary growth, the GTP levels drop, and eventually, CgtA-GDP complexes predominate. This model also predicts that the relatively slow hydrolysis of GTP would not play a major role in controlling the guanine nucleotide occupancy state of CgtA, since exchange occurs extremely rapidly. Given a generation time of 90 min in rich medium for C. crescentus, it is unlikely that the intrinsic hydrolysis rate of CgtA (t1/2 ∼ 23 min) is of biological relevance unless control mechanisms to enhance the hydrolysis rate (such as GAPs) exist in vivo.
Several lines of evidence support a model whereby CgtA is controlled by the intracellular GTP-GDP pools. (i) The in vitro exchange of guanine nucleotides occurs at optimum physiological Mg2+ concentrations. Without Mg2+, there is an eightfold increase in the exchange rate constant of GDP. However, even with millimolar levels of Mg2+, the dissociation rate constant of CgtA is 80-fold higher than that seen for the Ras protein (12, 13, 48). Therefore, in the absence of a mechanism to reduce the exchange rate of guanine nucleotides (model 1), the occupancy of CgtA should reflect the guanine nucleotide pool. (ii) The GEF-GAP-regulated GTP-binding proteins typically have very high affinities for nucleotides (nanomolar range) and low dissociation rate constants (on the order of hours). In addition, GTP exchange is much slower than GDP exchange (3). CgtA has moderate to weak affinity for both nucleotides, and the dissociation rate constants of GDP and GTP are comparable. These features argue that occupancy of CgtA could be controlled by the GTP-GDP pool. (iii) Proteins similar in function or sequence to GEFs or GAPs have not been identified in bacteria. (iv) In Streptomyces spp., overproduction of Obg delays development of aerial mycelium (33) under normal conditions and suppresses early development induced by the addition of decoyinine (a specific inhibitor of GMP synthetase) (33, 34). These data strongly argue that differentiation in Streptomyces spp. is determined by the balance of Obg protein and GTP levels. Specifically, it has been proposed that the Streptomyces spp. Obg protein is in the GTP-bound state during exponential growth and in the GDP-bound state after cells enter into stationary phase and the GTP pools decrease (34).
This study demonstrates that the mechanism of regulation of CgtA is different from that of the well-characterized Ras-like GTP-binding proteins. The data are consistent with a model whereby the guanine nucleotide state of CgtA is controlled by the intracellular GTP-GDP pool. If this is true, the timing of the shift from CgtA-GTP complexes to CgtA-GDP complexes would not be limited by the rate of guanine nucleotide exchange but by the ratio of GTP to GDP in the cell and the relative affinity of CgtA for the guanine nucleotides. Clearly, the challenge ahead is to determine the functional consequences of the CgtA-GTP-to-CgtA-GDP shift during C. crescentus growth.
ACKNOWLEDGMENTS
We are grateful to Rick Neubig for his generous gift of mant-GTP and to Phil Andrews, Charlie Yocum, Jesse Hay, and members of the Maddock laboratory for critical reading of the manuscript. Protein structure analysis was performed at the University of Michigan Protein and Carbohydrate Structure Facility.
This work was supported by grant GM-55133 from the National Institutes of Health and grant MCB9723749 from the National Science Foundation. J.R.M. is partially supported by JFRA-626 from the American Cancer Society.
REFERENCES
- 1.Ahnn J, March P E, Takiff H E, Inouye M. A GTP-binding protein of Escherichia coli has homology to yeast RAS proteins. Proc Natl Acad Sci USA. 1986;83:8849–8853. doi: 10.1073/pnas.83.23.8849. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Boguski M S, McCormick F. Proteins regulating Ras and its relatives. Nature. 1993;366:643–654. doi: 10.1038/366643a0. [DOI] [PubMed] [Google Scholar]
- 3.Bourne H R, Sanders D A, McCormick F. The GTPase superfamily: conserved structure and molecular mechanism. Nature. 1991;349:117–127. doi: 10.1038/349117a0. [DOI] [PubMed] [Google Scholar]
- 4.Bradford M M. A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye-binding. Anal Biochem. 1976;72:248–254. doi: 10.1016/0003-2697(76)90527-3. [DOI] [PubMed] [Google Scholar]
- 5.Britton R A, Powell B S, Dasgupta S, Sun Q, Margolin W, Lupski J R, Court D L. Cell cycle arrest in Era GTPase mutants: a potential growth rate-regulated checkpoint in Escherichia coli. Mol Microbiol. 1998;27:739–750. doi: 10.1046/j.1365-2958.1998.00719.x. [DOI] [PubMed] [Google Scholar]
- 6.Burstein E S, Macara I G. Interactions of the ras-like protein p25rab3A with Mg2+ and guanine nucleotides. Biochem J. 1992;282:387–392. doi: 10.1042/bj2820387. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Coligan J E, Dunn B M, Ploegh H L, Speicher D W, Wingfield P T, editors. Current protocols in protein science. New York, N.Y: John Wiley and Sons, Inc.; 1997. [Google Scholar]
- 8.Devitt M L, Maas K J, Stafstrom J P. Characterization of DRGs, developmentally regulated GTP-binding proteins, from pea and Arabidopsis. Plant Mol Biol. 1999;39:75–82. doi: 10.1023/a:1006178710443. [DOI] [PubMed] [Google Scholar]
- 9.DeVos A M, Tong L, Milburn M V, Matias P M, Jancarik J, Noguchi S, Nishimura S, Miura K, Ohtsuka E, Kim S-H. Three-dimensional structure of an oncogene protein: catalytic domain of human c-H-ras p21. Science. 1988;239:888–893. doi: 10.1126/science.2448879. [DOI] [PubMed] [Google Scholar]
- 10.Frech M, John J, Pizon V, Chardin P, Tavitian A, Clark R, McCormick F, Wittinghofer A. Inhibition of GTPase activating protein stimulation of Ras-p21 GTPase by the Krev-1 gene product. Science. 1990;249:169–171. doi: 10.1126/science.2164710. [DOI] [PubMed] [Google Scholar]
- 11.Gibbs J B, Schaber M D, Allard W J, Sigal I S, Scholnick E M. Purification of ras GTPase activating protein from bovine brain. Proc Natl Acad Sci USA. 1988;85:5026–5030. doi: 10.1073/pnas.85.14.5026. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Hall A, Self A J. The effect of Mg2+ on the guanine nucleotide exchange rate of p21N-ras. J Biol Chem. 1986;261:10963–10965. [PubMed] [Google Scholar]
- 13.Hara J, Tamaoki T, Nakano H. Guanine nucleotide binding properties of purified v-Ki-ras p21 protein produced in Escherichia coli. Oncogene Res. 1988;2:325–333. [PubMed] [Google Scholar]
- 14.Hiratsuka T. New ribose-modified fluorescent analogs of adenine and guanine nucleotides available as substrates for various enzymes. Biochim Biophys Acta. 1983;742:496–508. doi: 10.1016/0167-4838(83)90267-4. [DOI] [PubMed] [Google Scholar]
- 15.Hudson J D, Young P G. Sequence of the Schizosaccharomyces pombe gtp1 gene and identification of a novel family of putative GTP-binding proteins. Gene. 1993;125:191–193. doi: 10.1016/0378-1119(93)90327-y. [DOI] [PubMed] [Google Scholar]
- 16.Jacquet E, Vanoni M, Ferrari C, Alberghina L, Martegani E, Parmeggiani A. A mouse CDC25-like product enhances the formation of the active GTP complex of human ras p21 and Saccharomyces cerevisiae RAS2 proteins. J Biol Chem. 1992;267:24181–24183. [PubMed] [Google Scholar]
- 17.Jagath J R, Rodnina M V, Lentzen G, Wintermeyer W. Interaction of guanine nucleotides with the signal recognition particle from Escherichia coli. Biochemistry. 1998;57:15408–15415. doi: 10.1021/bi981523a. [DOI] [PubMed] [Google Scholar]
- 18.John J, Rensland H, Schlichting I, Vetter I, Borasio G D, Goody R S, Wittinghofer A. Kinetic and structural analysis of the Mg(2+)-binding site of the guanine nucleotide-binding protein p21H-ras. J Biol Chem. 1993;268:923–929. [PubMed] [Google Scholar]
- 19.John J, Sohment R, Feuerstein J, Linke R, Wittinghofer A, Goody R S. Kinetics of interaction of nucleotides with nucleotide-free H-ras p21. Biochemistry. 1990;29:6058–6065. doi: 10.1021/bi00477a025. [DOI] [PubMed] [Google Scholar]
- 20.Kjeldgaard M, Nyborg J, Clark B F C. The GTP-binding motif: variations on a theme. FASEB J. 1996;10:1347–1368. [PubMed] [Google Scholar]
- 21.Klebe C, Bischoff F R, Ponsting H, Wittinghofer A. Interaction of the nuclear GTP-binding protein Ran with its regulatory proteins RCC1 and RanGAP1. Biochemistry. 1995;34:639–647. doi: 10.1021/bi00002a031. [DOI] [PubMed] [Google Scholar]
- 22.Kok J, Trach K A, Hoch J A. Effect on Bacillus subtilis of a conditional lethal mutation in the essential GTP-binding protein Obg. J Bacteriol. 1994;176:7155–7160. doi: 10.1128/jb.176.23.7155-7160.1994. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Lenzen C, Cool R H, Wittinghofer A. Analysis of intrinsic and CDC25-stimulated guanine nucleotide exchange of p21ras-nucleotide complexes by fluorescence measurements. Methods Enzymol. 1995;255:95–109. doi: 10.1016/s0076-6879(95)55012-7. [DOI] [PubMed] [Google Scholar]
- 24.Lerosey I, Chardin P, Gunzburg J, Tavitian A. The product of the rap2 gene, member of the ras superfamily. J Biol Chem. 1991;266:4315–4321. [PubMed] [Google Scholar]
- 25.Maddock J, Bhatt A, Koch M, Skidmore J. Identification of an essential Caulobacter crescentus gene encoding a member of the Obg family of GTP-binding proteins. J Bacteriol. 1997;179:6426–6431. doi: 10.1128/jb.179.20.6426-6431.1997. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Mahajan M A, Park S T, Sun X-H. Association of a novel GTP binding protein, DRG, with TAL oncogenic proteins. Oncogene. 1996;12:2343–2350. [PubMed] [Google Scholar]
- 27.Masuda T, Tanaka K, Nonaka H, Yamochi W, Maeda A, Takai Y. Molecular cloning and characterization of yeast rho GDP dissociation inhibitor. J Biol Chem. 1994;269:19713–19718. [PubMed] [Google Scholar]
- 28.Menard L, Tomhave E, Casey P J, Uhing R J, Snyderman R, Didsbury J R. Rac1, a low-molecular-mass GTP-binding protein with high intrinsic GTPase activity and distinct biochemical properties. Eur J Biochem. 1992;206:537–546. doi: 10.1111/j.1432-1033.1992.tb16957.x. [DOI] [PubMed] [Google Scholar]
- 29.Mistou M-Y, Cool R H, Parmeggiani A. Effects of ions on the intrinsic activities of c-H-ras protein p21. A comparison with elongation factor Tu. Eur J Biochem. 1992;204:179–185. doi: 10.1111/j.1432-1033.1992.tb16621.x. [DOI] [PubMed] [Google Scholar]
- 30.Moser C, Mol O, Goody R S, Sinning I. The signal recognition particle receptor of Escherichia coli (FtsY) has a nucleotide exchange factor built into the GTPase domain. Proc Natl Acad Sci USA. 1997;94:11339–11344. doi: 10.1073/pnas.94.21.11339. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Neal S E, Eccleston J F, Webb M R. Hydrolysis of GTP by p21NRAS, the NRAS protooncogene product, is accompanied by a conformational change in the wild-type protein: use of a single fluorescent probe at the catalytic site. Proc Natl Acad Sci USA. 1990;87:3562–3565. doi: 10.1073/pnas.87.9.3562. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Nomanbhoy T K, Leonard D A, Manor D, Cerione R A. Investigation of the GTP-binding/GTPase cycle of the Cdc42Hs using extrinsic reporter group fluorescence. Biochemistry. 1996;35:4602–4608. doi: 10.1021/bi951743d. [DOI] [PubMed] [Google Scholar]
- 33.Okamoto S, Itoh M, Ochi K. Molecular cloning and characterization of the obg gene of Streptomyces griseus in relation to the onset of morphological differentiation. J Bacteriol. 1997;179:170–179. doi: 10.1128/jb.179.1.170-179.1997. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Okamoto S, Ochi K. An essential GTP-binding protein functions as a regulator of differentiation in Streptomyces coelicolor. Mol Microbiol. 1998;30:107–119. doi: 10.1046/j.1365-2958.1998.01042.x. [DOI] [PubMed] [Google Scholar]
- 35.Ormø M, Sjøberg B-M. An ultrafiltration assay for nucleotide binding to ribonucleotide reductase. Anal Biochem. 1990;189:138–141. doi: 10.1016/0003-2697(90)90059-i. [DOI] [PubMed] [Google Scholar]
- 36.Pai E F, Krengel U, Petsko G A, Goody R S, Kabsch W, Wittinghofer A. Structure of the guanine-nucleotide-binding domain of the Ha-ras oncogene product p21 in the triphosphate conformation. EMBO J. 1990;9:2351–2359. doi: 10.1002/j.1460-2075.1990.tb07409.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Pan J Y, Sanford J C, Wessling-Resnick M. Influence of Mg2+ on the structure and function of Rab5. J Biol Chem. 1996;271:1322–1328. doi: 10.1074/jbc.271.3.1322. [DOI] [PubMed] [Google Scholar]
- 38.Pan J Y, Wessling-Resnick M. GEF-mediated GDP/GTP exchange by monomeric GTPases: a regulatory role for Mg2+? Bioessays. 1998;20:516–521. doi: 10.1002/(SICI)1521-1878(199806)20:6<516::AID-BIES11>3.0.CO;2-3. [DOI] [PubMed] [Google Scholar]
- 39.Remmers A E, Neubig R R. Partial G protein activation by fluorescent guanine nucleotide analogs. Evidence for a triphosphate-bound but inactive state. J Biol Chem. 1996;271:4791–4797. doi: 10.1074/jbc.271.9.4791. [DOI] [PubMed] [Google Scholar]
- 40.Remmers A E, Posner R, Neubig R R. Fluorescent guanine nucleotide analogs and G protein activation. J Biol Chem. 1994;269:13771–13778. [PubMed] [Google Scholar]
- 41.Rensland H, Lautwein A, Wittinghofer A, Goody R S. Is there a rate-limiting step before GTP cleavage by H-ras p21? Biochemistry. 1991;30:11181–11185. doi: 10.1021/bi00110a023. [DOI] [PubMed] [Google Scholar]
- 42.Sasaki T, Kato M, Nishiyama T, Takai Y. The nucleotide exchange rates of Rho and Rac small GTP-binding proteins are enhanced to different extents by their regulatory protein Smg GDS. Biochem Biophys Res Commun. 1993;194:1188–1193. doi: 10.1006/bbrc.1993.1948. [DOI] [PubMed] [Google Scholar]
- 43.Sazuka T, Tomooka Y, Ikawa Y, Noda M, Kumar S. DRG: a novel developmentally regulated GTP-binding protein. Biochem Biophys Res Commun. 1992;189:363–370. doi: 10.1016/0006-291x(92)91567-a. [DOI] [PubMed] [Google Scholar]
- 44.Scheidig A J, Franken S M, Corrie J E T, Reid G P, Wittinghofer A, Pai E F, Goody R S. X-ray crystal structure analysis of the catalytic domain of the oncogene p21H-ras complexed with caged GTP and Mant dGppNHp. J Mol Biol. 1995;253:132–150. doi: 10.1006/jmbi.1995.0541. [DOI] [PubMed] [Google Scholar]
- 45.Schenker T, Lach C, Kessler B, Calderara S, Trueb B. A novel GTP-binding protein which is selectively repressed in SV40 transformed fibroblasts. J Biol Chem. 1994;269:25447–25453. [PubMed] [Google Scholar]
- 46.Schlichting I, Almo S C, Rapp G, Wilson K, Petratos K, Lentfer A. Time-resolved x-ray crystallographic study of the conformational change in Ha-Ras p21 protein on GTP hydrolysis. Nature. 1990;345:309–315. doi: 10.1038/345309a0. [DOI] [PubMed] [Google Scholar]
- 47.Schweins T, Scheffzek K, Abheuer R, Wittinghofer A. The role of the metal ion in the p21ras catalyzed GTP-hydrolysis: Mn2+versus Mg2+ J Mol Biol. 1997;266:847–856. doi: 10.1006/jmbi.1996.0814. [DOI] [PubMed] [Google Scholar]
- 48.Self A J, Hall A. Measurement of intrinsic nucleotide exchange and GTP hydrolysis rates. Methods Enzymol. 1995;256:67–76. doi: 10.1016/0076-6879(95)56010-6. [DOI] [PubMed] [Google Scholar]
- 49.Seo H S, Choi C H, Kim H Y, Jeong J Y, Lee S Y, Cho M J, Bahk J D. Guanine-nucleotide binding and hydrolyzing kinetics of ORrab2, a rice small GTP-binding protein expressed in Escherichia coli. Eur J Biochem. 1997;249:293–300. doi: 10.1111/j.1432-1033.1997.00293.x. [DOI] [PubMed] [Google Scholar]
- 50.Shoji I, Kikuchi A, Kuroda S, Takai Y. Kinetic analysis of the binding of guanine nucleotide to bovine brain smg p25A. Biochem Biophys Res Commun. 1989;162:273–281. doi: 10.1016/0006-291x(89)91992-x. [DOI] [PubMed] [Google Scholar]
- 51.Simon I, Zerial M, Goody R S. Kinetics of interaction of Rab5 and Rab7 with nucleotides and magnesium ions. J Biol Chem. 1996;271:20470–20478. doi: 10.1074/jbc.271.34.20470. [DOI] [PubMed] [Google Scholar]
- 52.Sommer K A, Peterson G, Bautz E K. The gene upstream of DmRP128 codes for a novel GTP-binding protein of Drosophila melanogaster. Mol Gen Genet. 1994;242:391–398. doi: 10.1007/BF00281788. [DOI] [PubMed] [Google Scholar]
- 53.Sownward J. The ras superfamily of small GTP-binding proteins. Trends Biochem Sci. 1990;15:469–472. doi: 10.1016/0968-0004(90)90300-z. [DOI] [PubMed] [Google Scholar]
- 54.Sprang S R. G protein mechanisms: insights from structural analysis. Annu Rev Biochem. 1997;66:639–678. doi: 10.1146/annurev.biochem.66.1.639. [DOI] [PubMed] [Google Scholar]
- 55.Tong L, deVos A M, Milburn M V, Kim S-H. Crystal structures at 2.2 Å resolution of the catalytic domains of normal ras protein and an oncogenic mutant complexed with GDP. J Mol Biol. 1991;217:503–516. doi: 10.1016/0022-2836(91)90753-s. [DOI] [PubMed] [Google Scholar]
- 56.Touchot N, Zahraoui A, Vielh E, Tavitian A. Biochemical properties of the YPT-related rab1B protein. Comparison with rab1A. FEBS Lett. 1989;256:79–84. doi: 10.1016/0014-5793(89)81722-3. [DOI] [PubMed] [Google Scholar]
- 57.Trach K, Hoch J A. The Bacillus subtilis spoOB stage O sporulation operon encodes an essential GTP-binding protein. J Bacteriol. 1989;171:1362–1371. doi: 10.1128/jb.171.3.1362-1371.1989. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Trahey M, McCormick F. A cytoplasmic protein stimulates normal N-ras p21 GTPase, but does not affect oncogenic mutants. Science. 1987;238:542–545. doi: 10.1126/science.2821624. [DOI] [PubMed] [Google Scholar]
- 59.Vedia L M, Ohmstede C-A, Lapetina E G. Properties of the exchange rate of guanine nucleotides to the novel RAP-2B protein. Biochem Biophys Res Commun. 1990;171:319–324. doi: 10.1016/0006-291x(90)91395-9. [DOI] [PubMed] [Google Scholar]
- 60.Vidwans S J, Ireton K, Grossman A D. Possible role for the essential GTP-binding protein Obg in regulating the initiation of sporulation in Bacillus subtilis. J Bacteriol. 1995;177:3308–3311. doi: 10.1128/jb.177.11.3308-3311.1995. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.Welsh K M, Trach K A, Folger C, Hoch J A. Biochemical characterization of the essential GTP-binding protein Obg of Bacillus subtilis. J Bacteriol. 1994;176:7161–7168. doi: 10.1128/jb.176.23.7161-7168.1994. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62.Zhu J, Reynet C, Caldwell J S, Kahn C R. Characterization of Rad, a new member of Ras/GTPase superfamily, and its regulation by a unique GTPase-activating protein (GAP)-like activity. J Biol Chem. 1995;270:4805–4812. doi: 10.1074/jbc.270.9.4805. [DOI] [PubMed] [Google Scholar]