Abstract
The gut microbiota has become a significant factor associated with health and disease. Although many studies have reported the implications of changes in the gut microbiota on cardiovascular diseases, there are no reports on the relationship between heart rate variability (HRV) and the gut microbiota. Therefore, we investigated the association between gut microbiota abundance and HRV parameters in this cross-sectional study of the general Japanese population. This study included 950 participants of the Iwaki Health Promotion Project who underwent a medical examination in 2019 that included HRV and gut microbiota measurements. At the genus level, multivariate regression analysis showed that higher gut microbial diversity was associated with a higher standard deviation of RR intervals (SDNN). Moreover, a higher SDNN was associated with a higher relative count of Lachnospiraceae incertae sedis. L. incertae sedis abundance was associated with higher HRV parameters such as SDNN, coefficient of variation of RR intervals, low-frequency component power (LF)/high-frequency component power, and LF. In the general Japanese population, higher gut microbial diversity and L. incertae sedis abundance were associated with higher HRV parameters.
Keywords: general Japanese population, heart rate variability, gut microbiota, gut microbial diversity, Lachnospiraceae incertae sedis
1. Introduction
The gut microbiota has become a major factor associated with health and disease, and its influence on physiological, behavioral, and cognitive functions of the brain is well recognized. However, the exact mechanisms have not been completely understood [1]. The brain–gut interaction involves the gut microbiota and its metabolic, enteric nervous system, neuroendocrine system, neural-immune system, central nervous system, and autonomic nervous system [2]. Moreover, the abundance of gut microbiota has been suggested to influence homeostasis by changing the properties of autonomic nerve signaling and related neuronal networks [2,3].
The autonomic nervous system is part of the peripheral nervous system and regulates involuntary physiological processes such as sexual arousal, digestion, respiration, blood pressure, and heartbeat [4]. It is also integral in maintaining homeostasis and behavioral functions [5]. The autonomic nervous function is clinically evaluated via heart rate variability (HRV) analysis, an index of autonomic nervous activity related to cardiovascular function. HRV analysis is a non-invasive measurement technique used in various fields [6,7]. In the general population, lower HRV has been associated with cardiovascular disease (CVD) risk and is clinically important [8]. Prevention of CVD is very important as these diseases are among the leading causes of morbidity and mortality in developed countries.
Although many studies have reported that changes in gut microbiota composition are implicated in CVD [9,10,11,12], there are no reports on the relationship between HRV and gut microbiota abundance. Therefore, this study aimed to investigate the association between gut microbiota abundance and HRV parameters in the general Japanese population using data from a medical examination in the Iwaki Health Promotion Project.
2. Results
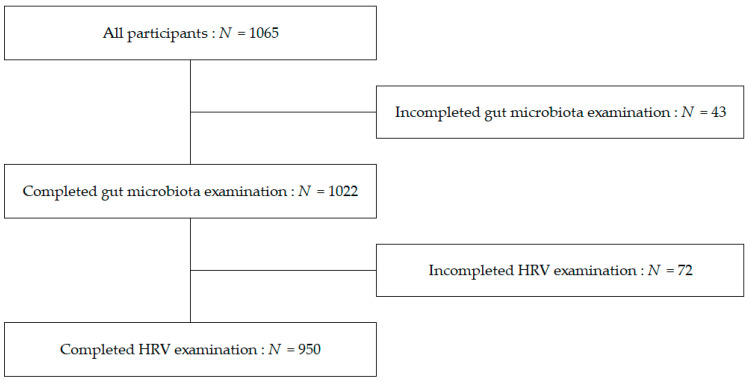
Figure 1 illustrates the number of examinations completed by study participants. Among the 1065 participants in the Iwaki Health Promotion Project medical examination, 43 did not have their gut microbiota examined; hence, 1022 completed the examination. An additional 72 participants did not complete HRV measurements because of an unstable heart rate (HR). Finally, 950 participants were included in the analysis.
Figure 1.
Number of examinations completed.
Table 1 shows the clinical characteristics of the study participants. The characteristics of the study cohort were as follows: sex distribution, 58.9% women and 41.1% men; mean age, 52.6 years; and mean body mass index (BMI), 23.1 kg/m2. In the HRV parameters, the mean intra-individual coefficient of variation of the RR interval (CVRR) and the inter-individual CVRR were 3.4% and 14.0%, respectively. Table 2 shows the relative proportions of the main gut microbial phyla among participants. The mean proportions were as follows: Actinobacteria, 11.9%; Bacteroidetes, 24.8%; Proteobacteria, 2.9%; and Firmicutes, 58.9%. For reference, the summary statistics of 52 gut microbial genera possessed by more than 50% of the participants are shown in Supplementary Materials Table S1.
Table 1.
Participant characteristics.
| Continuous Variables | Total (N = 950) | Men (N = 391) | Women (N = 559) | ||||
|---|---|---|---|---|---|---|---|
| (Unit) | Mean | (SD) | Mean | (SD) | Mean | (SD) | |
| Age | (years) | 52.6 | (14.77) | 52.4 | (14.58) | 52.7 | (14.90) |
| BMI | (kg/m2) | 23.1 | (3.63) | 24.1 | (3.51) | 22.4 | (3.54) |
| HbA1c | (%) | 5.7 | (0.62) | 5.7 | (0.71) | 5.7 | (0.54) |
| Glycoalbumin | (%) | 14.6 | (1.98) | 14.4 | (2.41) | 14.7 | (1.61) |
| Blood glucose | (mg/dL) | 96.3 | (16.52) | 99.8 | (18.29) | 93.9 | (14.70) |
| Triglyceride | (mg/dL) | 98.2 | (84.33) | 125.2 | (114.54) | 79.4 | (45.43) |
| Total cholesterol | (mg/dL) | 205.4 | (34.27) | 203.0 | (34.17) | 207.0 | (34.27) |
| HDL cholesterol | (mg/dL) | 65.0 | (16.66) | 58.2 | (15.00) | 69.8 | (16.08) |
| LDL cholesterol | (mg/dL) | 116.8 | (29.79) | 117.4 | (29.34) | 116.4 | (30.11) |
| ALT | (U/L) | 20.9 | (13.75) | 26.5 | (16.52) | 17.0 | (9.69) |
| AST | (U/L) | 21.8 | (7.86) | 23.8 | (8.37) | 20.5 | (7.18) |
| γ-GTP | (U/L) | 33.2 | (40.94) | 49.1 | (56.98) | 22.1 | (16.75) |
| Creatinine | (mg/dL) | 0.7 | (0.55) | 0.9 | (0.74) | 0.6 | (0.33) |
| Urea nitrogen | (mg/dL) | 14.5 | (4.50) | 15.4 | (4.52) | 14.0 | (4.40) |
| SBP | (mmHg) | 120.8 | (16.88) | 123.5 | (16.65) | 118.9 | (16.79) |
| DBP | (mmHg) | 77.1 | (11.20) | 79.5 | (11.48) | 75.3 | (10.67) |
| RR interval | (ms) | 866.9 | (121.76) | 885.9 | (127.19) | 853.7 | (116.09) |
| CVRR | (%) | 3.4 | (1.67) | 3.4 | (1.70) | 3.4 | (1.56) |
| SDNN | (ms) | 29.3 | (14.56) | 29.8 | (15.7) | 29.0 | (13.76) |
| LF | (ms2) | 343.5 | (522.26) | 421.4 | (625.19) | 289.0 | (428.32) |
| HF | (ms2) | 252.2 | (340.90) | 246.9 | (364.04) | 256.0 | (324.03) |
| LF/HF | (Ratio) | 2.7 | (3.99) | 3.3 | (4.73) | 2.2 | (3.32) |
| HR | (bpm) | 70.1 | (10.15) | 68.7 | (10.23) | 71.1 | (9.98) |
| Categorical variables | N | (%) | N | (%) | N | (%) | |
| Diabetes mellitus | No | 890 | (93.9%) | 361 | (92.3%) | 529 | (95.0%) |
| Yes | 58 | (6.1%) | 30 | (7.7%) | 28 | (5.0%) | |
| Hyperlipidemia | No | 778 | (82.2%) | 315 | (81.0%) | 463 | (83.1%) |
| Yes | 168 | (17.8%) | 74 | (19.0%) | 94 | (16.9%) | |
| High blood pressure | No | 711 | (74.9%) | 275 | (70.3%) | 436 | (78.1%) |
| Yes | 238 | (25.1%) | 116 | (29.7%) | 122 | (21.9%) | |
| Heart disease | No | 906 | (95.5%) | 366 | (93.6%) | 540 | (96.8%) |
| Yes | 43 | (4.5%) | 25 | (6.4%) | 18 | (3.2%) | |
| Gastric/Duodenal ulcer | No | 853 | (89.9%) | 342 | (87.5%) | 511 | (91.6%) |
| Yes | 96 | (10.1%) | 49 | (12.5%) | 47 | (8.4%) | |
| Antidiabetic medication use | No | 903 | (95.1%) | 367 | (93.9%) | 536 | (95.9%) |
| Yes | 47 | (4.9%) | 24 | (6.1%) | 23 | (4.1%) | |
| Antihyperlipidemic medication use | No | 846 | (89.1%) | 346 | (88.5%) | 500 | (89.4%) |
| Yes | 104 | (10.9%) | 45 | (11.5%) | 59 | (10.6%) | |
| Antihypertensive medication use | No | 728 | (76.6%) | 285 | (72.9%) | 443 | (79.2%) |
| Yes | 222 | (23.4%) | 106 | (27.1%) | 116 | (20.8%) | |
| Exercising (non-winter) | No | 732 | (77.4%) | 302 | (77.6%) | 430 | (77.2%) |
| Yes | 214 | (22.6%) | 87 | (22.4%) | 127 | (22.8%) | |
| Exercising (winter) | No | 731 | (77.7%) | 304 | (78.6%) | 427 | (77.1%) |
| Yes | 210 | (22.3%) | 83 | (21.4%) | 127 | (22.9%) | |
| Smoking | No | 596 | (63.3%) | 161 | (41.6%) | 435 | (78.4%) |
| Current | 161 | (17.1%) | 114 | (29.5%) | 47 | (8.5%) | |
| Previous | 185 | (19.6%) | 112 | (28.9%) | 73 | (13.2%) | |
| Alcohol consumption | No | 444 | (47.3%) | 111 | (28.6%) | 333 | (60.5%) |
| Current | 456 | (48.6%) | 266 | (68.6%) | 190 | (34.5%) | |
| Previous | 38 | (4.1%) | 11 | (2.8%) | 27 | (4.9%) | |
Abbreviations: γ-GTP, γ-glutamyl transferase; ALT, alanine transaminase; AST, aspartate transaminase; BMI, body mass index; bpm, beats per minute; CVRR, coefficient of variation of RR intervals; DBP, diastolic blood pressure; HbA1c, hemoglobin A1c; HDL, high-density lipoprotein; HF, high-frequency component power; HR, heart rate; LDL, low-density lipoprotein; LF, low-frequency component power; SBP, systolic blood pressure; SD, standard deviation; SDNN, standard deviation of RR intervals.
Table 2.
Relative count of gut microbial major phylum in participant.
| Continuous Variables | Total (N = 950) | Men (N = 391) | Women (N = 559) | ||||
|---|---|---|---|---|---|---|---|
| (Unit) | Mean | (SD) | Mean | (SD) | Mean | (SD) | |
| Actinobacteria | (%) | 11.9 | (9.02) | 11.8 | (9.31) | 12.0 | (8.82) |
| Bacteroidetes | (%) | 24.8 | (11.22) | 26.7 | (12.47) | 23.5 | (10.05) |
| Proteobacteria | (%) | 2.9 | (2.56) | 3.2 | (2.76) | 2.6 | (2.38) |
| Firmicutes | (%) | 58.9 | (12.27) | 56.2 | (13.04) | 60.8 | (11.33) |
Abbreviation: SD, standard deviation.
Table 3 and Table 4 show associations between HRV parameters and diversity (Simpson and Shannon indices) of the gut microbial genus. A higher Simpson index was associated with a higher standard deviation of RR intervals (SDNN) in multivariate regression analysis of Model 2 (β = 0.213; 95% confidence interval [CI], 0.012 to 0.413; p = 0.038). A higher Shannon index was associated with a higher SDNN in the multivariate regression analysis of Model 2 (β = 3.934; 95% CI, 0.444 to 7.424; p = 0.027).
Table 3.
Analysis of the association between HRV parameters and the Simpson index (%).
| Characteristics | Univariate | Model 1 | Model 2 | ||||||
|---|---|---|---|---|---|---|---|---|---|
| β | 95% CI | p-Value | β | 95% CI | p-Value | β | 95% CI | p-Value | |
| SDNN (ms) | 0.113 | −0.093 to 0.320 | 0.282 | 0.174 | −0.020 to 0.369 | 0.078 | 0.213 | 0.012 to 0.413 | 0.038 |
| CVRR (%) | 0.011 | −0.012 to 0.034 | 0.368 | 0.017 | −0.004 to 0.038 | 0.116 | 0.020 | −0.002 to 0.042 | 0.070 |
| LF (ms2) | 0.743 | −6.673 to 8.159 | 0.844 | 3.963 | −3.174 to 11.100 | 0.276 | 4.598 | −2.823 to 12.018 | 0.224 |
| HF (ms2) | 0.564 | −4.277 to 5.405 | 0.819 | 1.024 | −3.738 to 5.786 | 0.673 | 2.305 | −2.589 to 7.199 | 0.356 |
| LF/HF | 0.013 | −0.043 to 0.070 | 0.641 | 0.034 | −0.022 to 0.090 | 0.236 | 0.034 | −0.024 to 0.092 | 0.254 |
| HR (bpm) | −0.022 | −0.166 to 0.122 | 0.762 | −0.033 | −0.175 to 0.109 | 0.648 | −0.051 | −0.195 to 0.093 | 0.484 |
Model 1: Adjusted for age, sex, and BMI. Model 2: Adjusted for age, sex, BMI, antidiabetic, antihyperlipidemic, antihypertensive, physical activity (non-winter and winter months), smoking, and alcohol consumption. Abbreviations: BMI, body mass index; CI, confidence interval; CVRR, coefficient of variation of RR intervals; HF, high-frequency component power; HR, heart rate; HRV, heart rate variability; LF, low-frequency component power; SDNN, standard deviation of RR intervals.
Table 4.
Analysis of the association between HRV parameters and the Shannon index.
| Characteristics | Univariate | Model 1 | Model 2 | ||||||
|---|---|---|---|---|---|---|---|---|---|
| β | 95% CI | p-Value | β | 95% CI | p-Value | β | 95% CI | p-Value | |
| SDNN (ms) | 0.681 | −2.931 to 4.293 | 0.711 | 3.542 | 0.142 to 6.942 | 0.041 | 3.934 | 0.444 to 7.424 | 0.027 |
| CVRR (%) | −0.056 | −0.458 to 0.346 | 0.786 | 0.302 | −0.064 to 0.667 | 0.106 | 0.346 | −0.030 to 0.721 | 0.071 |
| LF (ms2) | −38.512 | −168.009 to 90.984 | 0.560 | 57.731 | −67.340 to 182.803 | 0.365 | 63.993 | −65.218 to 193.205 | 0.331 |
| HF (ms2) | 3.120 | −81.242 to 87.663 | 0.942 | 38.975 | −44.438 to 122.388 | 0.359 | 50.440 | −34.732 to 135.612 | 0.245 |
| LF/HF | −0.014 | −1.003 to 0.975 | 0.978 | 0.342 | −0.647 to 1.331 | 0.498 | 0.346 | −0.671 to 1.363 | 0.504 |
| HR (bpm) | −2.232 | −4.745 to 0.280 | 0.082 | −1.608 | −4.088 to 0.872 | 0.204 | 0.676 | −4.132 to 0.876 | 0.202 |
Model 1: Adjusted for age, sex, and BMI. Model 2: Adjusted for age, sex, BMI, antidiabetic, antihyperlipidemic, antihypertensive, physical activity (non-winter and winter months), smoking, and alcohol consumption. Abbreviations: BMI, body mass index; CI, confidence interval; CVRR, coefficient of variation of RR intervals; HF, high-frequency component power; HR, heart rate; HRV, heart rate variability; LF, low-frequency component power; SDNN, standard deviation of RR intervals.
These results showed that a higher gut microbial diversity was associated with a higher SDNN after adjusting for age, sex, BMI, antidiabetic, antihyperlipidemic, antihypertensive, physical activity, smoking, and alcohol consumption. However, no significant associations with CVRR, low-frequency component power (LF), high-frequency component power (HF), LF/HF, and HR were confirmed.
Table 5 shows the associations between HRV parameters and Lachnospiraceae incertae sedis. In the univariate and multivariate regression analyses, L. incertae sedis was the only gut microbiota associated with SDNN (Tables S2–S4). Multivariate regression analysis (Model 2) revealed that a higher relative count of L. incertae sedis was associated with a higher SDNN (β = 1.449; 95% CI, 0.616 to 2.282; p = 0.001), CVRR (β = 0.135; 95% CI, 0.045 to 0.225; p = 0.003), LF (β = 59.687; 95% CI, 28.954 to 90.420; p < 0.001), and LF/HF (β = 0.367; 95% CI, 0.124 to 0.609; p = 0.003). In addition, HR was associated with a higher relative count of L. incertae sedis in the multivariate regression analysis of Model 1 only (β = −0.626; 95% CI, −1.212 to −0.040; p = 0.036).
Table 5.
Analysis of the association between HRV parameters and the relative count (%) of Lachnospiraceae incertae sedis.
| Characteristics | Univariate | Model 1 | Model 2 | ||||||
|---|---|---|---|---|---|---|---|---|---|
| β | 95% CI | p-Value | β | 95% CI | p-Value | β | 95% CI | p-Value | |
| SDNN (ms) | 1.024 | 0.169 to 1.878 | 0.019 | 1.390 | 0.589 to 2.191 | 0.001 | 1.449 | 0.616 to 2.282 | 0.001 |
| CVRR (%) | 0.087 | −0.008 to 0.183 | 0.072 | 0.127 | 0.040 to 0.213 | 0.004 | 0.135 | 0.045 to 0.225 | 0.003 |
| LF (ms2) | 41.007 | 10.402 to 71.611 | 0.009 | 57.949 | 28.590 to 87.309 | <0.001 | 59.687 | 28.954 to 90.420 | <0.001 |
| HF (ms2) | 7.118 | −12.926 to 27.163 | 0.486 | 10.824 | −8.899 to 30.548 | 0.282 | 11.525 | −8.885 to 31.935 | 0.268 |
| LF/HF | 0.271 | 0.037 to 0.505 | 0.023 | 0.357 | 0.124 to 0.590 | 0.003 | 0.367 | 0.124 to 0.609 | 0.003 |
| HR (bpm) | −0.586 | −1.182 to 0.010 | 0.054 | −0.626 | −1.212 to −0.040 | 0.036 | −0.576 | −1.175 to 0.023 | 0.060 |
Model 1: Adjusted for age, sex, and BMI. Model 2: Adjusted for age, sex, BMI, antidiabetic, antihyperlipidemic, antihypertensive, physical activity (non-winter and winter months), smoking, and alcohol consumption. Abbreviations: BMI, body mass index; CI, confidence interval; CVRR, coefficient of variation of RR intervals; HF, high-frequency component power; HR, heart rate; HRV, heart rate variability; LF, low-frequency component power; SDNN, standard deviation of RR intervals.
These results indicated that higher levels of HRV parameters, such as SDNN, CVRR, LF, and LF/HF, were associated with a higher relative count of L. incertae sedis after adjusting for age, sex, BMI, antidiabetic, antihyperlipidemic, antihypertensive, physical activity, smoking, and alcohol consumption.
3. Discussion
Among the participants in the Iwaki Health Promotion Project medical examination, a higher gut microbial diversity and L. incertae sedis abundance were associated with higher HRV parameters. In this study, the relative counts of the four major phyla in the gut (Firmicutes, Bacteroidetes, Actinobacteria, and Proteobacteria) were similar to those reported among participants in urban Kyoto City [13]. This study investigated the gut microbiota in the general Japanese population. A decreased gut microbial diversity has been linked to inflammatory bowel disease (IBD) [14], irritable bowel syndrome (IBS) [15], obesity [16], and a Western diet high in fat and sugar compared to a low-fat plant-based diet [17]. Understanding gut microbial diversity could facilitate the development of personalized nutritional and drug strategies [18].
L. incertae sedis abundance was associated with higher HRV parameters such as SDNN, CVRR, LF, and LF/HF, suggesting its association with overall cardiac autonomic and sympathetic activity. These findings are relevant because, to the best of our knowledge, no other study has reported an association between HRV and gut microbiota abundance among many participants. L. incertae sedis belongs to the Lachnospiraceae family phylogenetically and morphologically heterogeneous taxon belonging to the clostridial cluster XIVa of the phylum Firmicutes, which hydrolyzes starch and other sugars to produce butyrate and other short-chain fatty acids (SCFAs) [19]. The clostridial cluster XIVa, derived from human feces, may induce T regulatory cells and suppress inflammatory conditions such as IBD via SCFA butyrate production [20,21]. Previous studies have reported a higher prevalence of clostridial cluster XIVa in rural Kyotango City than in the urban city of Kyoto [13]. Kyotango City is a long-lived province with multiple centenarians, and emerging evidence suggests that the brain–gut interaction may influence the etiology of IBD. Clinical studies have shown that alterations in the brain–gut interaction are associated with autonomic nervous system dysfunction, affecting the central nervous and digestive systems [22]. Crohn’s disease (CD) and ulcerative colitis (UC) are classified as IBD as they have similar symptoms and lead to digestive disorders and inflammation in the digestive system [23]. Patients with UC and CD have lower time-domain parameters, such as SDNN, and HRV frequency domain parameters, such as LF and HF, than healthy participants [22]. The relationship between L. incertae sedis contained in butyrate-producing bacterium and some HRV parameters was confirmed in this study; hence, further studies to investigate the relationship between butyrate-producing bacteria and IBD via HRV are warranted.
Moreover, butyrate-producing bacteria reduce obesity by modulating G-protein coupled receptors (GPRs) 41 and 43 [24,25]. GPR-41 is most abundantly expressed in sympathetic ganglia in mice and humans, indicating its importance in these cells [24]. Obesity develops when energy intake exceeds energy expenditure; thus, increasing cellular energy expenditure may be an attractive approach [26]. Direct regulation of sympathetic activation via GPR-41 may serve as a key physiological mechanism regulating the body’s energy balance since modulating sympathetic nervous system (SNS) activity causes an increase or decrease in energy expenditure [24]. Incidentally, it has been reported that obesity lowers SDNN; however, no association between obesity and HRV parameters related to SNS was identified [27,28]. Whether targeting the SNS directly improves obesity or metabolism remains unknown but merits further attention [29]. In obesity, the composition of the intestinal flora is disrupted and is associated with cardiac and HRV dysfunction [30]. This study results highlight the importance of further investigating the anti-obesity and cardioprotective effects of prebiotics, probiotics, and synbiotics of butyrate-producing bacteria.
Changes in the gut microbiota composition and gut microbial metabolism have been implicated in the etiology of CVD. Gut-derived metabolites play important roles in maintaining healthy cardiac and vascular function [9,10,11,12]. CVD, as with IBD, is characterized by chronic inflammation and exhibits similar physiological mechanisms [31]. Furthermore, gastrointestinal disorders generally occurs such as IBS when the autonomic nervous system fails to regulate gastrointestinal motility [32]. Previous studies have suggested a relationship between IBS and the gut microbiota [15]. Clinical applications of the relationship between autonomic nerves and the gut microbiota may contribute to the prevention and treatment of CVD, IBD, and IBS.
In addition, the association between the gut and vaginal microbiota has recently attracted attention. The vaginal microbiota, as with the gut microbiota, have been reported to be associated with HRV and suggested to be related to an anti-inflammatory response due to autonomic nervous activity during parturition [33]. The relationship between the gut and vaginal microbiota is thought to be due to the vagina’s proximity to the anus, which allows the gut microbiota to reach the reproductive organs via the rectum and perineum [33]. Further studies on the relationship between the gut microbiota and the vaginal microbiota via autonomic nervous system are warranted.
This study had some limitations. First, the measurement of HRV was not optimal. Generally, HRV should be measured in the supine position for at least 24 h or 5 min [34]. However, in this study, measurements were taken in the sitting position for a relatively short time, 90 s [35]. Therefore, individuals with unstable HR and those who did not measure HRV were excluded from the analysis, which may have biased the results [35]. Furthermore, only limited HRV parameters were analyzed in this study; the association between HRV parameters not measured in VM302 and the gut microbiota could not be determined. In addition, methods that analyze HRV in the time or frequency domain may not be sufficient to characterize the complex dynamics of the heartbeat [36]. More reliable non-linear regression analyses are required to calculate HRV parameters. Second, although we adjusted for antidiabetic, antihyperlipidemic and antihypertensive use, the effects of specific classes of these medications that may be related to cardiac autonomic function and the gut microbiota could not be completely surveyed. Third, because of the cross-sectional design [34], we could not determine a causal relationship between HRV and gut microbiota abundance. Longitudinal data are needed to elucidate this relationship. Fourth, this study included participants who voluntarily participated in the Iwaki Health Promotion Health Examination [34]. Therefore, we may not be able to generalize the results of this study due to selection bias [34]; additional longitudinal studies with large sample sizes are needed to clarify the causal relationship between HRV and gut microbiota abundance.
4. Materials and Methods
4.1. Participants and Analysis
The Iwaki Health Promotion Project Health Examination has been conducted annually since 2005 to prevent lifestyle-related diseases, maintain and promote health, and increase longevity among residents of the Iwaki area of Hirosaki City [37]. The participants (n = 1065) in the 2019 health examination were men and women aged 20 years or older living in the Iwaki area of Hirosaki City, Aomori Prefecture [34]. This cross-sectional study included 950 participants who completed HRV and gut microbiota measurements. This study was approved by the Hirosaki University School of Medicine Ethics Review Committee (approval number: 2019-009) and was conducted in accordance with the principles of the Declaration of Helsinki. Written informed consent was obtained from all participants [37].
4.2. Clinical Features
All clinical examinations were performed in the morning in a fasting state [34]. BMI was assessed by physical examination, and systolic and diastolic blood pressures were measured. BMI was calculated from body weight and height (kg/m2). Blood pressure was measured with an automatic blood pressure meter Elemano 2 (Terumo Corporation, Tokyo, Japan) while the participant was seated and at rest. Blood tests were performed to determine glucose metabolic capacity by measuring hemoglobin A1c, glycoalbumin, and blood glucose; lipid metabolic capacity by measuring triglycerides, total cholesterol, high-density lipoprotein, and low-density lipoprotein; and liver function by measuring alanine transaminase, aspartate transaminase, γ-glutamyl transaminases were measured to evaluate liver function, and creatinine and urea nitrogen were measured to evaluate kidney function. Blood samples were collected from peripheral veins in the supine position, and blood tests were performed at LSI Medience Company (Tokyo, Japan) [34]. For analysis of lifestyle-related factors, investigations were collected for diabetes, dyslipidemia, hypertension, heart disease, gastric/duodenal ulcer, smoking, alcohol consumption, exercise (non-winter and winter), antidiabetic, antihyperlipidemic, and antihypertensive.
4.3. Measurement of HRV
HRV was measured in the morning while fasting [34]. Participants did not smoke or engage in strenuous exercise during the physical examination [34]. A Vital Monitor 302 (VM302) system (Hitachi Systems, Ltd., Tokyo, Japan) was used to ensure that the HR was stable, and measurements were taken for 90 s with the participant seated and eyes closed [34]. Data were analyzed using Memfmcc software (Fatigue Science Institute, Osaka, Japan) [34]. The VM302 can simultaneously perform electrocardiography (ECG) and photoelectric volumetric pulse waves from the fingertip and has been used in several clinical trials [38,39]. By monitoring HRV with ECG and photoplethysmography, 90 s of cardiac autonomic function data were collected at a sampling rate of 600 Hz [34]. The VM302′s built-in firmware uses a peak detection algorithm based on the Hill climbing method to detect the R-wave peak and transmits the obtained R-wave peak times to an external computer [34]. A time series of RR intervals was sequentially generated from the peak time of each R wave [34]. Noise in the digital signal was removed with a low-pass filter, but not the linear trend of the RR interval. The HR was calculated from the reciprocal of the RR interval for each heartbeat. In addition, instead of a steady-state test for RR interval variability, the error of the data for 30 s of the RR interval was set at 0.75-fold lower and 1.75-fold higher than the median value of the RR interval for 30 s, and the matched RR interval data were removed [34].
SDNN and CVRR, which express modulation of sympathetic and parasympathetic functions as time-domain parameters, were evaluated [34,40]. SDNN and CVRR were calculated as the SDNN and SDNN/mean value of the RR interval × 100, during the measurement. CVRR is SDNN normalized to the RR interval [41]. Variations in the RR interval are typical HRV parameters [42]. In addition, parameters in the frequency domain were evaluated; LF was calculated as power in the frequency range of 0.04 to 0.15 Hz, and HF as power in the frequency range of 0.15 to 0.4 Hz. The average values of LF, HF, and LF/HF obtained for each time series were representative of each measurement [34]. Frequency analysis of RR interval variation was performed using the maximum entropy method, which allows the estimation of power spectral density from short time series data and is suitable for studying HRV changes under different conditions over short periods [34].
4.4. Measurements of the Gut Microbiota
The detailed processing method for extracting deoxyribonucleic acid (DNA) from fecal samples is shown below [43]. Each participant promptly transferred 2–3 g of fresh feces into a storage container (TechnoSuruga Laboratory, Shizuoka, Japan) containing 3 mL of a guanidine thiocyanate stock solution (GTC buffer; 100 mM Tris-HCl [pH 9.0], 40 mM Tris-EDTA [pH 8.0], and 4 M guanidine thiocyanate). Fecal samples were stored at room temperature. Fecal sample suspensions (200 μL) were added to a tube with zirconium beads and 800 μL of GTC buffer and homogenized using a FastPrep 24 Instrument (MP Biomedicals, Santa Ana, CA, USA) at 5 m/s for 2 min. After cooling, the samples were centrifuged at 2400× g for 1 min. DNA was then purified from the bead-treated suspension using an automated DNA isolation system (GENE PREP STAR PI-480, KURABO, Osaka, Japan).
The detailed processing method for analyzing the gut microbiota using next-generation sequencing is mentioned here [44]. Sequences of the V3–V4 region of 16S recombinant DNA were used to identify the bacteria. Amplified fragments were purified using polymerase chain reaction (PCR) cleanup filter plates (Merck Millipore, Burlington, MA, USA) and quantified by real-time quantitative PCR. Bacterial sequences were detected and identified using the Illumina MiSeq system (Illumina, San Diego, CA, USA).
The detailed processing method for multiplexed paired-end reads is shown below [45]. Adaptor sequences and low-quality bases (threshold = 20) were trimmed at the 3′-end of the reads by Cutadapt (version: 1.13) [46]. N bases and reads containing less than 150 bases containing reads were discarded. Paired-end reads that exceeded the filter threshold were merged into a single read called a “merged read.” Merged reads < 370 or >470 bases were excluded by the fastq_mergepairs subcommand of VSEARCH (version: 2.4.3) [47]. Merged reads with multiple expected sequence errors were also excluded. After removing chimera reads detected by the uchime denovo subcommand of VSEARCH, the remaining merged reads were clustered with a sequence identity ≥ 97%. Taxonomy of the identified clusters was predicted by applying ribosomal database project Classifier (commit hash: 701e229dde7cbe53d4261301e23459d91615999d) based on representative reads. Results with confidence values less than 0.8 were treated as unclassified. The composition ratio of each genus of the gut microbiota was determined by dividing the number of read counts of each genus by the total number of read counts.
Of the 294 genera detected (including unclassified), genera not possessed by more than 50% of the participants, which were too few to analyze accurately, were excluded from the analysis.
4.5. Statistical Analysis
Among the clinical characteristics of the participants, continuous variables are presented as the means and standard deviations, whereas categorical variables are presented as the frequency and percentages. The relative counts of gut microbial phylum and genera are presented as the means and standard deviations. To investigate the diversity (alpha-diversity) of microbial genus level associated with HRV, univariate and multivariate regression analyses were performed with various HRV parameters as the objective variables and the Simpson or Shannon index [48] as the explanatory variables. To investigate the relative counts of gut microbial genera associated with HRV, univariate and multivariate regression analyses were performed with the specific HRV parameter related to microbial diversity as the objective variables and the abundances of each gut microbial genus as the explanatory variables. Moreover, univariate and multivariate regression analyses were performed with various HRV parameters as the objective variables and abundances of the specific gut microbial genus detected above as the explanatory variables. In the multivariate analysis, Model 1 (adjusted for age, sex, and BMI) and Model 2 (adjusted for antidiabetic, antihyperlipidemic, antihypertensive, physical activity, smoking, and alcohol consumption in addition to age, sex, and BMI) were performed. The regression coefficient (β) was calculated using the standard least squares method. In these analyses, the significance level was set at p < 0.05 in two-tailed tests, and 95% CI for the regression coefficient (β) was calculated. We used JMP® Ver.16 software (SAS Institute, Inc., Cary, NC, USA) in these statistical analyses [34].
5. Conclusions
This study showed that a higher gut microbial diversity was associated with a higher SDNN. Moreover, L. incertae sedis abundance was associated with higher HRV parameters such as SDNN, CVRR, LF, and LF/HF. In the general Japanese population, higher gut microbial diversity and L. incertae sedis abundance were associated with higher HRV parameters. Clinical applications of autonomic nerves and the gut microbiota possibly contribute to the prevention and treatment of IBS, IBD, and CVD.
Acknowledgments
The authors would like to thank JST’s COI program (JPMJCE1302) and Iwaki Health Promotion Project.
Supplementary Materials
The following supporting information can be downloaded at: https://www.mdpi.com/article/10.3390/metabo12080730/s1, Table S1: The summary statistics of the relative count (%) of gut microbial genera. Table S2: Univariate analysis of the association between SDNN (ms) and the relative count (%) of gut microbial genera. Table S3: Multivariate analysis (Model 1) of the association between SDNN (ms) and the relative count (%) of gut microbial genera. Table S4: Multivariate analysis (Model 2) of the association between SDNN (ms) and the relative count (%) of gut microbial genera.
Author Contributions
Conceptualization, M.T.; methodology, M.T.; software, M.T.; validation, M.N.; formal analysis, M.T.; investigation, M.T.; resources, M.I.; data curation, M.T.; writing—original draft preparation, M.T.; writing—review and editing, M.T., M.N., T.M., M.I., T.H. and Y.T.; visualization, M.N.; supervision, T.M.; project administration, Y.T.; funding acquisition, T.M. All authors have read and agreed to the published version of the manuscript.
Institutional Review Board Statement
This study was conducted in accordance with the Declaration of Helsinki and approved by the Ethics Review Board of Hirosaki University School of Medicine (approval number: 2019-009).
Informed Consent Statement
Written informed consent has been obtained from the patients to publish this paper.
Data Availability Statement
The data cannot be shared publicly because of ethical concerns. Data are available from the Hirosaki University COI Program Institutional Data Access/Ethics Committee (contact via e-mail: coi@hirosaki-u.ac.jp) for researchers who meet the criteria for access to the data. Researchers need to have prior approval from the research ethics review boards of their respective affiliations.
Conflicts of Interest
The authors declare no conflict of interest. M.T. and M.N. were employed by FANCL Co., Ltd. (Tokyo, Japan). No patents, products under development, or products in the market have been declared. The funding organizations did not play any role in study design, data collection and analysis, decision to publish, or manuscript preparation.
Funding Statement
This research was funded by JST’s COI program Hirosaki University Base, grant number JPMJCE1302, and FANCL Co., Inc. (Kanagawa, Japan).
Footnotes
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations.
References
- 1.Fülling C., Dinan T.G., Cryan J.F. Gut Microbe to Brain Signaling: What Happens in Vagus. Neuron. 2019;101:998–1002. doi: 10.1016/j.neuron.2019.02.008. [DOI] [PubMed] [Google Scholar]
- 2.Wang H.-X., Wang Y.-P. Gut Microbiota-brain Axis. Chin. Med. J. 2016;129:2373–2380. doi: 10.4103/0366-6999.190667. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Bonaz B., Sinniger V., Pellissier S. Vagal tone: Effects on sensitivity, motility, and inflammation. Neurogastroenterol. Motil. 2016;28:455–462. doi: 10.1111/nmo.12817. [DOI] [PubMed] [Google Scholar]
- 4.Waxenbaum J.A., Reddy V., Varacallo M. StatPearls. StatPearls Publishing; Treasure Island, FL, USA: 2019. [(accessed on 29 July 2021)]. Anatomy, autonomic nervous system. Available online: https://www.ncbi.nlm.nih.gov/books/NBK539845/ [PubMed] [Google Scholar]
- 5.Coote J.H., Spyer K.M. Central control of autonomic function. Brain Neurosci. Adv. 2018;2:2398212818812012. doi: 10.1177/2398212818812012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Sztajzel J. Heart rate variability: A noninvasive electrocardiographic method to measure the autonomic nervous system. Swiss Med. Wkly. 2004;134:514–522. doi: 10.4414/smw.2004.10321. [DOI] [PubMed] [Google Scholar]
- 7.Vanderlei L.C., Pastre C.M., Hoshi R.A., Carvalho T., De Godoy M.F. Basic notions of heart rate variability and its clinical applicability. Rev. Bras. Cir. Cardiovasc. 2009;24:205–217. doi: 10.1590/S0102-76382009000200018. [DOI] [PubMed] [Google Scholar]
- 8.Hillebrand S., Gast K.B., de Mutsert R., Swenne C.A., Jukema J.W., Middeldorp S., Rosendaal F.R., Dekkers O.M. Heart rate variability and first cardiovascular event in populations without known cardiovascular disease: Meta-analysis and dose–response meta-regression. Europace. 2013;15:742–749. doi: 10.1093/europace/eus341. [DOI] [PubMed] [Google Scholar]
- 9.Tang W.H.W., Kitai T., Hazen S.L. Gut Microbiota in Cardiovascular Health and Disease. Circ. Res. 2017;120:1183–1196. doi: 10.1161/CIRCRESAHA.117.309715. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Wang Z., Zhao Y. Gut microbiota derived metabolites in cardiovascular health and disease. Protein Cell. 2018;9:416–431. doi: 10.1007/s13238-018-0549-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Witkowski M., Weeks T.L., Hazen S.L. Gut Microbiota and Cardiovascular Disease. Circ. Res. 2020;127:553–570. doi: 10.1161/CIRCRESAHA.120.316242. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Xu H., Wang X., Feng W., Liu Q., Zhou S., Liu Q., Cai L. The gut microbiota and its interactions with cardiovascular disease. Microb. Biotechnol. 2020;13:637–656. doi: 10.1111/1751-7915.13524. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Naito Y., Takagi T., Inoue R., Kashiwagi S., Mizushima K., Tsuchiya S., Itoh Y., Okuda K., Tsujimoto Y., Adachi A., et al. Gut microbiota differences in elderly subjects between rural city Kyotango and urban city Kyoto: An age-gender-matched study. J. Clin. Biochem. Nutr. 2019;65:125–131. doi: 10.3164/jcbn.19-26. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Dicksved J., Halfvarson J., Rosenquist M., Järnerot G., Tysk C., Apajalahti J., Engstrand L., Jansson J.K. Molecular analysis of the gut microbiota of identical twins with Crohn’s disease. ISME J. 2008;2:716–727. doi: 10.1038/ismej.2008.37. [DOI] [PubMed] [Google Scholar]
- 15.Carroll I.M., Ringel-Kulka T., Keku T.O., Chang Y.-H., Packey C.D., Sartor R.B., Ringel Y. Molecular analysis of the luminal- and mucosal-associated intestinal microbiota in diarrhea-predominant irritable bowel syndrome. Am. J. Physiol. Gastrointest. Liver Physiol. 2011;301:G799–G807. doi: 10.1152/ajpgi.00154.2011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Turnbaugh P.J., Bäckhed F., Fulton L., Gordon J.I. Diet-Induced Obesity Is Linked to Marked but Reversible Alterations in the Mouse Distal Gut Microbiome. Cell Host Microbe. 2008;3:213–223. doi: 10.1016/j.chom.2008.02.015. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Yatsunenko T., Rey F.E., Manary M.J., Trehan I., Dominguez-Bello M.G., Contreras M., Magris M., Hidalgo G., Baldassano R.N., Anokhin A.P., et al. Human gut microbiome viewed across age and geography. Nature. 2012;486:222–227. doi: 10.1038/nature11053. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Lozupone C.A., Stombaugh J.I., Gordon J.I., Jansson J.K., Knight R. Diversity, stability and resilience of the human gut microbiota. Nature. 2012;489:220–230. doi: 10.1038/nature11550. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Vacca M., Celano G., Calabrese F.M., Portincasa P., Gobbetti M., De Angelis M. The Controversial Role of Human Gut Lachnospiraceae. Microorganisms. 2020;8:573. doi: 10.3390/microorganisms8040573. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Atarashi K., Tanoue T., Oshima K., Suda W., Nagano Y., Nishikawa H., Fukuda S., Saito T., Narushima S., Hase K., et al. Treg induction by a rationally selected mixture of Clostridia strains from the human microbiota. Nature. 2013;500:232–236. doi: 10.1038/nature12331. [DOI] [PubMed] [Google Scholar]
- 21.Atarashi K., Tanoue T., Shima T., Imaoka A., Kuwahara T., Momose Y., Cheng G., Yamasaki S., Saito T., Ohba Y., et al. Induction of colonic regulatory T cells by indigenous clostridium species. Science. 2011;331:337–341. doi: 10.1126/science.1198469. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Kim K.-N., Yao Y., Ju S.-Y. Heart rate variability and inflammatory bowel disease in humans: A systematic review and meta-analysis. Medicine. 2020;99:e23430:1–e23430:10. doi: 10.1097/MD.0000000000023430. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Seyedian S.S., Nokhostin F., Malamir M.D. A review of the diagnosis, prevention, and treatment methods of inflammatory bowel disease. J. Med. Life. 2019;12:113–122. doi: 10.25122/jml-2018-0075. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Kimura I., Inoue D., Maeda T., Hara T., Ichimura A., Miyauchi S., Kobayashi M., Hirasawa A., Tsujimoto G. Short-chain fatty acids and ketones directly regulate sympathetic nervous system via G protein-coupled receptor 41 (GPR41) Proc. Natl. Acad. Sci. USA. 2011;108:8030–8035. doi: 10.1073/pnas.1016088108. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Kimura I., Ozawa K., Inoue D., Imamura T., Kimura K., Maeda T., Terasawa K., Kashihara D., Hirano K., Tani T., et al. The gut microbiota suppresses insulin-mediated fat accumulation via the short-chain fatty acid receptor GPR43. Nat. Commun. 2013;4:1829. doi: 10.1038/ncomms2852. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Tseng Y.-H., Cypess A.M., Kahn C.R. Cellular bioenergetics as a target for obesity therapy. Nat. Rev. Drug Discov. 2010;9:465–482. doi: 10.1038/nrd3138. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Almeida-Santos M.A., Barreto-Filho J.A., Oliveira J.L., Reis F.P., da Cunha Oliveira C.C., Sousa A.C.S. Aging, heart rate variability and patterns of autonomic regulation of the heart. Arch. Gerontol. Geriatr. 2016;63:1–8. doi: 10.1016/j.archger.2015.11.011. [DOI] [PubMed] [Google Scholar]
- 28.Oliveira C., Silveira E.A., Rosa L., Santos A., Rodrigues A.P., Mendonça C., Silva L., Gentil P., Rebelo A.C. Risk Factors Associated with Cardiac Autonomic Modulation in Obese Individuals. J. Obes. 2020;2020:7185249. doi: 10.1155/2020/7185249. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Lambert G.W., Schlaich M.P., Eikelis N., Lambert E.A. Sympathetic activity in obesity: A brief review of methods and supportive data. Ann. N. Y. Acad. Sci. 2019;1454:56–67. doi: 10.1111/nyas.14140. [DOI] [PubMed] [Google Scholar]
- 30.Tunapong W., Apaijai N., Yasom S., Tanajak P., Wanchai K., Chunchai T., Kerdphoo S., Eaimworawuthikul S., Thiennimitr P., Pongchaidecha A., et al. Chronic treatment with prebiotics, probiotics and synbiotics attenuated cardiac dysfunction by improving cardiac mitochondrial dysfunction in male obese insulin-resistant rats. Eur. J. Nutr. 2018;57:2091–2104. doi: 10.1007/s00394-017-1482-3. [DOI] [PubMed] [Google Scholar]
- 31.Łykowska-Szuber L., Rychter A.M., Dudek M., Ratajczak A.E., Szymczak-Tomczak A., Zawada A., Eder P., Lesiak M., Dobrowolska A., Krela-Kaźmierczak I. What Links an Increased Cardiovascular Risk and Inflammatory Bowel Disease? A Narrative Review. Nutrients. 2021;13:2661. doi: 10.3390/nu13082661. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Casado-Bedmar M., Keita Å.V. Potential neuro-immune therapeutic targets in irritable bowel syndrome. Ther. Adv. Gastroenterol. 2020;13:1756284820910630. doi: 10.1177/1756284820910630. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Reyes-Lagos J.J., Echeverría-Arjonilla J.C., Peña-Castillo M.Á., Montiel-Castro A.J., Pacheco-López G. Physiological, immunological and evolutionary perspectives of labor as an inflammatory process. Adv. Neuroimmune Biol. 2014;5:75–89. doi: 10.3233/NIB-140085. [DOI] [Google Scholar]
- 34.Tsubokawa M., Nishimura M., Tamada Y., Nakaji S. Factors Associated with Reduced Heart Rate Variability in the General Japanese Population: The Iwaki Cross-Sectional Research Study. Healthcare. 2022;10:793. doi: 10.3390/healthcare10050793. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Task Force of the European Society of Cardiology and the North American Society of Pacing and Electrophysiology Heart rate variability: Standards of measurement, physiological interpretation and clinical use. Circulation. 1996;93:1043–1065. doi: 10.1161/01.CIR.93.5.1043. [DOI] [PubMed] [Google Scholar]
- 36.De Godoy M.F. Nonlinear analysis of heart rate variability: A comprehensive review. J. Cardiol. Ther. 2016;3:528–533. doi: 10.17554/j.issn.2309-6861.2016.03.101-4. [DOI] [Google Scholar]
- 37.Nakaji S., Ihara K., Sawada K., Parodi S., Umeda T., Takahashi I., Murashita K., Kurauchi S., Tokuda I. Social innovation for life expectancy extension utilizing a platform-centered system used in the Iwaki health promotion project: A protocol paper. SAGE Open Med. 2021;9:20503121211002606. doi: 10.1177/20503121211002606. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Kume S., Nishimura Y., Mizuno K., Sakimoto N., Hori H., Tamura Y., Yamato M., Mitsuhashi R., Akiba K., Koizumi J.-I., et al. Music Improves Subjective Feelings Leading to Cardiac Autonomic Nervous Modulation: A Pilot Study. Front. Neurosci. 2017;11:108. doi: 10.3389/fnins.2017.00108. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Mizuno K., Sasaki A.T., Ebisu K., Tajima K., Kajimoto O., Nojima J., Kuratsune H., Hori H., Watanabe Y. Hydrogen-rich water for improvements of mood, anxiety, and autonomic nerve function in daily life. Med. Gas. Res. 2017;7:247–255. doi: 10.4103/2045-9912.222448. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Pop-Busui R. Cardiac Autonomic Neuropathy in Diabetes: A clinical perspective. Diabetes Care. 2010;33:434–441. doi: 10.2337/dc09-1294. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Silva L.E.V., Salgado H.C., Fazan R., Jr. Mean Heart Rate Level Does Not Affect All Heart Rate Variability Indices. Hypertension. 2017;69:e21–e22. doi: 10.1161/HYPERTENSIONAHA.117.09060. [DOI] [PubMed] [Google Scholar]
- 42.Aeschbacher S., Bossard M., Ruperti Repilado F.J., Good N., Schoen T., Zimny M., Probst-Hensch N.M., Schmidt-Trucksäss A., Risch M., Risch L., et al. Healthy lifestyle and heart rate variability in young adults. Eur. J. Prev. Cardiol. 2016;23:1037–1044. doi: 10.1177/2047487315623708. [DOI] [PubMed] [Google Scholar]
- 43.Takahashi S., Tomita J., Nishioka K., Hisada T., Nishijima M. Development of a Prokaryotic Universal Primer for Simultaneous Analysis of Bacteria and Archaea Using Next-Generation Sequencing. PLoS ONE. 2014;9:e105592. doi: 10.1371/journal.pone.0105592. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Nomura A., Matsubara A., Goto S., Takahata J., Sawada K., Ihara K., Nakaji S. Relationship between gut microbiota composition and sensitization to inhaled allergens. Allergol. Int. 2020;69:437–442. doi: 10.1016/j.alit.2019.12.010. [DOI] [PubMed] [Google Scholar]
- 45.Ozato N., Saito S., Yamaguchi T., Katashima M., Tokuda I., Sawada K., Katsuragi Y., Kakuta M., Imoto S., Ihara K., et al. Blautia genus associated with visceral fat accumulation in adults 20–76 years of age. NPJ Biofilms Microbiomes. 2019;5:28. doi: 10.1038/s41522-019-0101-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Martin M. Cutadapt Removes Adapter Sequences from High-Throughput Sequencing Reads. EMBnet J. 2011;17:10–12. doi: 10.14806/ej.17.1.200. [DOI] [Google Scholar]
- 47.Rognes T., Flouri T., Nichols B., Quince C., Mahé F. VSEARCH: A versatile open source tool for metagenomics. PeerJ. 2016;4:e2584. doi: 10.7717/peerj.2584. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Kim B.R., Shin J., Guevarra R.B., Lee J.H., Kim D.W., Seol K.H., Lee J.H., Kim H.B., Isaacson R.E. Deciphering Diversity Indices for a Better Understanding of Microbial Communities. J. Microbiol. Biotechnol. 2017;27:2089–2093. doi: 10.4014/jmb.1709.09027. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Data Availability Statement
The data cannot be shared publicly because of ethical concerns. Data are available from the Hirosaki University COI Program Institutional Data Access/Ethics Committee (contact via e-mail: coi@hirosaki-u.ac.jp) for researchers who meet the criteria for access to the data. Researchers need to have prior approval from the research ethics review boards of their respective affiliations.