Abstract
Nanomaterials are currently widely exploited for their potential in the development of novel cancer therapies, and so far, mainly nanoparticles (NPs) consisting of liposomes and polymers have made their way into the clinic. However, major bottlenecks for the clinical translation of other types of NPs (i.e. inorganic) are the lack of knowledge concerning their long-term distribution in vivo and their potential toxicity. To counter this, various research groups have worked on soluble NPs, such as zinc oxide (ZnO), copper oxide (CuO), and silver (Ag), which tend to dissolve spontaneously into their ionic form, releasing toxic metal ions and leading to reactive oxygen species (ROS) generation when exposed to cellular environments. By fine-tuning the dissolution kinetics of these NPs, it is possible to control the level of ROS production and thus cytotoxicity to selectively destroy tumor tissue. Specifically, cancer cells tend to exhibit a higher basal level of oxidative stress compared to normal cells due to their higher metabolic rates, and therefore, by engineering NPs that generate sufficient ROS that barely exceed toxic thresholds in cancer cells, normal cells will only experience reversible transient damage. This review focuses on the use of these soluble inorganic NPs for selective cancer therapy and on the various in vitro and in vivo studies that have aimed to control the dissolution kinetics of these NPs, either through particle doping or surface modifications.
Nanomaterials are widely exploited for developing novel cancer therapies. Here, we discuss the potential of cancer-selective toxicity of metal oxide particle degradation.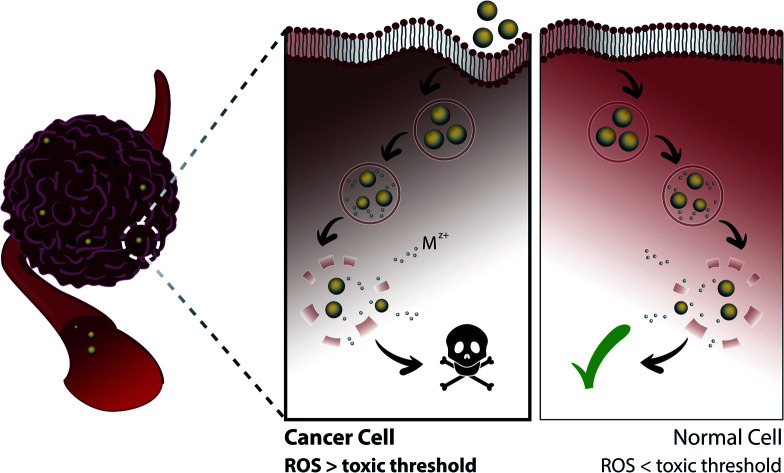
1. Introduction
Despite the long use of nanomaterials for biomedical applications, the field of nanomedicine only truly established itself as a separate field from nanotechnology in 2004 and involves the use of nanoparticles (NPs) that range from one to hundreds of nanometers for medical purposes.1 At this small scale, the surface area is significantly increased, altering the physical and chemical characteristics of the particles and giving rise to their various unique properties that render them suitable for a wide range of biomedical applications.2–4 For instance, NPs with varying compositions, sizes, and shapes can be used to enhance the solubility, biodistribution, and magnetic and optical properties of formulations, while their surfaces can be functionalized with various coatings and ligands to improve tissue selectivity and uptake into targeted cells.5,6 A wide range of different materials including liposomes, polymers, micelles, metal oxides (MOs) and other inorganic components can be employed for NP synthesis. These NPs can be subdivided in several ways based on their various characteristics. Two classifications that are often employed include chemical composition (organic or inorganic) and use (as delivery vehicle or direct physical/chemical therapy).
The focus of nanomedicine in the clinic so far has been in the treatment of cancer (i.e. Doxil©, Abraxane©, Onivyde MM-398©, DaunoXome©), anemia and Fe deficiency (i.e. Cosmofer©, Feraheme©), and infections (i.e. AmbiSome©), and in their use as contrast agents for non-invasive imaging (i.e. Ferumoxtran-10©, SonoVue©) and in vaccinations (i.e. Epaxal©, Inflexal©). Specifically, the anticancer regimens Doxil© and DaunoXome© consist of liposomes loaded with doxorubicin (DOX) and daunorubicin, respectively, and are approved for the treatment of HIV-associated Kaposi's sarcoma. Additional indications for Doxil© include ovarian cancer and multiple myeloma. Moreover, the approval of no less than three new nanomedicines, VYXEOS© (FDA-approved), Patisiran© (FDA-approved) and Hensify© (CE-Mark approved), in the last couple of years clearly shows that the field of nanomedicine is being actively investigated in by companies and academic and research institutions worldwide.7 VYXEOS© consists of liposomes loaded with daunorubicin and cytarabine and was approved for the treatment of acute myeloid leukemia cancer in 2017. Hensify©, on the other hand, is a hafnium oxide NP-based aqueous suspension that is used for enhanced radiotherapy treatment and was recently approved in 2019. Patisiran©, the first siRNA/RNAi therapeutic, is also encapsulated within liposomes and has received FDA-approval in 2018 for the treatment of rare amyloidogenic polyneuropathy. Currently, there are a few thousand ongoing studies that use NPs mainly focusing on cancer and gene therapy.7 These studies mostly involve the so-called ‘soft’ NPs, consisting of liposomes or polymers, which predominantly aim to obtain a more efficient delivery of traditional anticancer agents (Fig. 1).
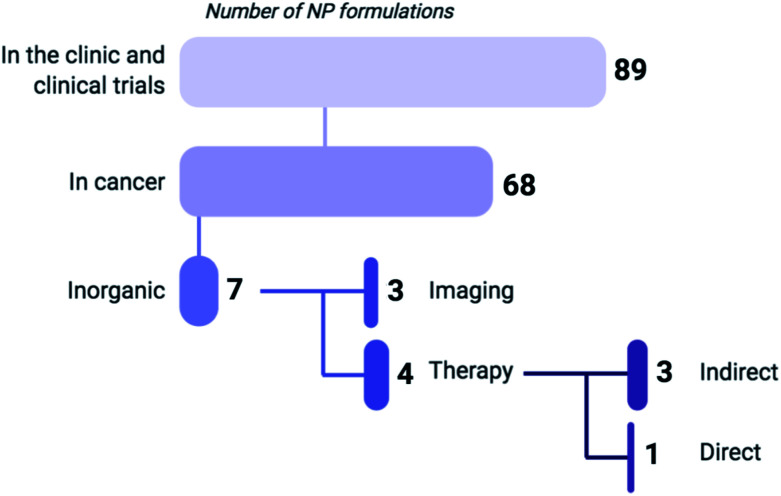
Fig. 1. Based on a recent review by Anselmo and Mitragotri (2019) listing 89 NP formulations that are currently approved for use in the clinic or undergoing clinical trials, 75% of these regimens are indicated for cancer treatment (including gene therapy) or diagnosis, of which 90% are composed of organic materials such as liposomes, polymers and micelles.7 Six of the inorganic NP formulations that are clinically approved are used for imaging, while only one (NU-0129) functions as a direct therapeutic. As of 2019, NU-0129, consisting of Au NPs surface-covered with nucleic acids, is being tested in phase I clinical trials for the treatment of glioblastoma. The indirect-acting NPs exert their anticancer activity through radiation or thermal ablation by an external source.
Harder NPs, often consisting of inorganic metals or MOs, have lower translational use, although a lot of progress has been made over the last years. For example, Fe oxide (IO) NPs are already clinically approved as hyperthermia agents for the treatment of multiforme glioblastoma (MagForce©) and are currently being tested on other types of tumors such as prostate cancer. Gold (Au) NPs are also being investigated in diverse cancer studies as agents for photothermal therapy (Aurolase©). In both cases, the NPs are used as indirect therapeutic agents, where external stimuli such as alternating magnetic fields and laser lights are applied upon administration of MagForce© and Aurolase©, respectively, to produce local heat, leading to cancer cell (CC) death. The lower translation of such NPs in the clinic has mainly been attributed to the lack of knowledge concerning their safety due to their potential to induce high levels of toxicity in biological systems compared to their bulk counterparts.8 This can be explained by the greater number of reactive sites on the NP surface associated with their higher surface-to-volume ratio as well as the high chemical reactivity that results from the structural modifications of their surface electronic properties.9,10 Concurrently, their nano size enables them to circumvent clearance by the reticuloendothelial system (RES), remain longer in blood circulation,11 passively traverse across the leaky tumor vasculature and penetrate deep into tumor tissue – a phenomenon known as the enhanced permeability and retention (EPR) effect.12,13 Put together, the larger exposure of the surface reactivity of NPs and their EPR effect lead to a higher level of particle interaction with their immediate surroundings, and therefore, greater potential for toxicity. However, this may not be the case for all types of materials, and even within a single type, the degree of toxicity is dependent upon multiple factors such as size, shape, coating and other surface modifications, as well as the nature of the exposed cell type.6,14–16
Therefore, there is a strong need to control NP-induced toxicity for effective tumor therapy. Alongside unaccounted-for toxicity, another factor that restricts the use of NPs in the clinic is the limited knowledge concerning their final destination, especially in the long-term. For example, Au NPs remain in the body for a long time and it is not yet entirely clear where these NPs will go after the therapy has been completed and what their long-term effects are, mainly in terms of the immune system, liver, spleen and kidneys.17–19 It has been suggested that such NPs remain intact within the tumor environment as a result of the thick extracellular matrix, poor lymphatic drainage, and eventual particle agglomeration. To counter this, various research groups have worked on soluble NPs, such as zinc oxide (ZnO), copper oxide (CuO), and silver (Ag). These NPs tend to dissolve spontaneously into their ionic form, releasing toxic metal ions (Mz+) when exposed to biotic/abiotic environments.20,21 In the context of the cell, the NPs are mainly taken up by endocytosis, during which their Mz+ release induces reactive oxygen species (ROS)-mediated toxicity in tumor tissues. This review will discuss the mechanisms by which these inorganic NPs degrade and exert their cytotoxicity, and how controlling the degree of Mz+ release from NPs can be harnessed to achieve selective tumor therapy. Although IONPs are the one of the most commonly used type of inorganic NPs for biomedical applications to date, their intracellular degradation has thus far not been linked to a cancer-selective toxicity (to our knowledge) particularly since the release of ferric ions is easily incorporated into the natural metabolism of the cell to be used in various biological functions and is thus tolerated at high levels.22 Therefore, this review will mainly focus on ZnO, CuO, and Ag NPs.
2. Inorganic NPs and their degradation
For all engineered NPs to be used for biomedical applications, their potential toxicity and removal from the body must be studied. In general, for organic materials, most commonly applied materials are biodegradable, resulting in the generation of biologically relevant components (e.g. polylactic-co-glycolic acid (PLGA) breaks down into lactic and glycolic acid, which are metabolites that can be processed in cellular Krebs cycle23). While the effect of these additional metabolites on cell growth and metabolism may not be negligible in all conditions, the toxicity thereof will be minimal. For inorganic NPs, this is typically a far more difficult story. Some NPs, such as Au NPs tend not to degrade and can persist very long inside the body.17 Other NPs can degrade, upon which they release Mz+. Any toxicity associated with this depends on multiple factors, including the chemical nature of the ions released and the rate of degradation. It is important to note that while some ions are physiologically relevant (e.g. Zn, Fe), their presence may affect other metabolites or disturb cellular homeostasis and hereby cause secondary toxicity. In the sections below, we aim to provide an overview of NP degradation (also referred to as “dissolution” throughout the text) with respect to how this can affect cellular homeostasis and how the process of NP degradation can be more controlled to selectively destroy CCs. Specifically, given that CCs exhibit a higher basal level of ROS compared to normal cells (NCs) due to their higher metabolic rates, NPs can be engineered to release Mz+ and generate ROS at finely-tuned levels that exceed toxic thresholds in CCs but only cause transient cellular damage in healthy tissue and allow gradual recovery.
2.1. Oxidative stress due to Mz+ release from inorganic NPs as main cause of toxicity
The mechanism by which metallic NPs exert their cytotoxic effects has been mainly attributed to the generation of ROS or ROS-induced damage (Fig. 2).4,24 These species can be classified as non-radicals (i.e. H2O2, HOCl, O3) or free radical-containing (i.e.1O2, O2˙−, HO˙, HO2˙).25,26 Under normal conditions, ROS are produced as intermediates by the cell's natural oxidative metabolism and serve as key mediators in processes such as cell survival and signaling, and production of inflammatory factors.27,28 Their intracellular levels are strongly maintained by antioxidants (i.e. glutathione GSH), and detoxifying enzymes, including superoxide dismutases (SODs), GSH peroxidase (GPx), and catalase (CAT).29 SOD serves as a catalyst for the conversion of O2− to O2 and H2O2, and CAT further reduces H2O2 to H2O. The cellular compartments that are mainly associated with ROS production are the mitochondria, endoplasmic reticula (ER), peroxisomes, and NADPH oxidase (NOX) complexes located on the cell membranes.29–31 At the early stages of the ROS production process, increased levels of cytoplasmic calcium (Ca2+) trigger the activation of the mitochondrial electron transport chain, leading to the production of ATP and therefore, ROS as by-products.32,33 Disruption in the redox balance that regulates ROS homeostasis leads to the oxidative damage of various biomolecules and consequently, interference with different cellular functions.34,35 Among the alterations that ROS generation exert is the peroxidation of lipids leading to cell membrane damage, oxidation of proteins which interferes with enzymatic function and membrane permeability, and nucleic acid oxidation that causes genotoxicity.4,36 ROS-induced oxidative stress has been delineated as a three-tier model.35,37 Tier 1 is characterized by a defensive response wherein detoxifying enzymes including HO1, NAD(P)H, SOD and CAT aim to restore the redox balance and minimize damage through the activation of the phase II genes by Nrf2.38,39 Tier 2 results from ROS-induced stress that surpasses the enzymes' defensive capacity, leading to the production of pro-inflammatory cytokines and chemokines and thus, activation of inflammatory cells such as macrophages and neutrophils.40,41 Among the pro-inflammatory responses is the activation of mitogen-activated protein kinase (MAPK) and nuclear factor κB (NFκB).42,43 Tier 3 involves perturbation of the mitochondrial membranes and their potential through the formation of transient pores and increased influx of intracellular calcium, resulting in morphological changes or cell death by apoptosis, autophagy or necrosis.35 When NPs are internalized by the cells, they are taken up into the endosomes where the acidic environment and cellular biomolecules stimulate metal ion release from the NPs causing an imbalance in the local metal ion homeostasis and producing a strong but gradual increase in ROS that reaches optimum/Tier 3 levels after 24–48 h. Specifically, transition metals located at the surface of NPs, such as Cu on CuO NPs, have been shown to react with H2O2 to produce HO˙ and oxidized Cu ions via Fenton-type reactions.44,45
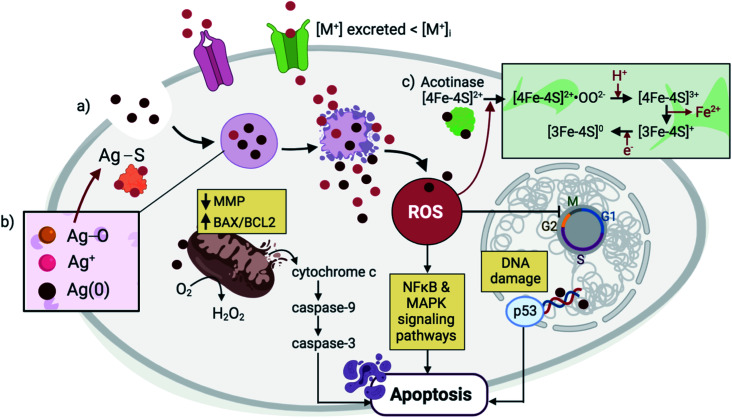
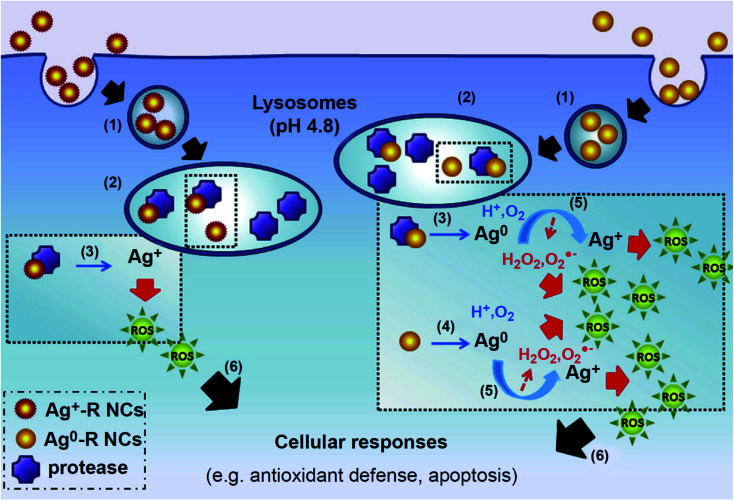
Fig. 2. NPs are endocytosed and are contained within lysosomal compartments, during which they are exposed to low pH environments that accelerate their dissolution. (a) The high concentration of local Mz+ release (pink) exceeds the metal transporters' capacity to excrete ions, leading to ROS generation and DNA damage. In response, p53 triggers cell cycle arrest with the aim of restoring oxidative balance. Further elevation in ROS levels causes a decrease in the MMP and an increase in the BAX/BCL2 ratio, triggering cytochrome c release and activation of a caspase signaling pathway that leads to apoptosis. Other signaling pathways such as NFκB and MAPK are also activated during NP-mediated apoptosis. (b) In the case of Ag NPs, three forms of Ag exist simultaneously in the lysosomes, including solid Ag(0), Ag+, and Ag2O.65 Subsequent interaction of Ag2O intermediates with cysteine residues results in Ag–S formation that cause disruptions in protein secondary structures. (c) Meanwhile, in the case of ZnO NPs, Mz+ release and ROS generation cause alterations in the Fe–S clusters that are found abundantly in several protein families including aconitase, leading to protein inactivation.42.
2.1.1. Mz+ released from NPs vs. their metal salt counterparts
It is generally agreed upon that the toxicity induced by these dissolving inorganic NPs is at least partially due to the release of Mz+.20 Several studies have been conducted throughout the years to confirm this hypothesis by comparing the toxicity induced by the NPs and their respective metal salts.46–50 The “Trojan Horse” mechanism has been used to describe NP-associated cytotoxicity, which is predominantly mediated by particle uptake into lysosomes, followed by a burst-like release of Cu2+, whereas the metal salts are not similarly internalized.50,51 However, others have argued that ZnO and CuO NPs exert the same toxic effects as their ionic counterparts.52–54 This strong discrepancy in the literature is a product of the different methodologies used in determining NP dissolution, as confirmed by Turney et al. (2012) revealing a fivefold difference in ZnO NP dissolution depending on whether the Zn species were separated by centrifugation or membrane filtration.55 As a means to settle this debate, Ivask et al. (2017) used synchrotron radiation-based X-ray absorption near edge structure (SR-XANES) spectroscopy which does not require particle and ion separation to identify elemental speciation, and the resulting spectra delineated similar speciation profiles between ZnO and CuO NPs and their metal salts in cells and cell culture media.56 Some have suggested that the observed toxicity within 24 h of NP exposure to cells is attributed to the NPs and their dissolution within the endosomes, while any subsequent effect is due to the Mz+ that have diffused into the cytoplasm.57,58 Concurrently, a bacterial proteomic study by Neal et al. (2012) has shown that in Cupriavidus necator bacteria, the proteins over-expressed upon ZnO NPs and Zn2+ exposure were associated with the synthesis of membrane proteins and cellular metabolism, respectively, suggesting varying mechanisms of toxicity induction of MO NPs and their salts.59 Similarly, although higher cytotoxicity was detected for CuO NPs than CuCl2, certain gene expression changes (i.e. for metallothioneins (MTs), which are key biomarkers in metal-induced toxicity60) were attributed to Cu2+ release.46 However, it is now clear that NPs exhibit differing exposure kinetics than their respective metal salts. When cells are exposed to metal salts, the Mz+ will typically diffuse throughout the entire cell almost uniformly. Conversely, once taken up by endosomes/lysosomes where the acidic environment speeds up their degradation (as will be explained more thoroughly in Section 2.2.1), NPs will locally induce high levels of Mz+ within the vicinity or inside the endosomal compartments, which is quite different than what is observed in the rest of the cell. These high Mz+ concentrations can locally exceed toxic thresholds while the overall cellular level may not be toxic. Combined with the oxidative stress linked to having the Mz+ in the acidic environments of the endosomal compartment, this can give rise to higher levels of toxicity than free Mz+ given in their salt form ref. 61.
As with CuO and ZnO NPs, there have been several debates on the cytotoxicity induced by Ag NPs and their metal salts. In a study by Kim et al. (2009), the authors demonstrate comparable levels of cytotoxicity between Ag NPs and AgNO3, although the mechanism of toxicity differed for both.62 In cells treated with Ag NPs, no particular effect on the expression of MTs could be detected, which contrasts with the increased expression observed in AgNO3-exposed cells. When coupled with studies reporting that Ag NPs only partially dissolve in solution and thus release less free Ag+ compared with Ag salt, these findings suggest that the Ag NPs-induced toxicity is a product of oxidative stress rather than Mz+ release.63 Given that the mechanism of Ag+ release is shown to be via oxidative solution – during which surface metallic Ag reacts with protons and dissolved O2 to yield Ag2O, which is subsequently fully solubilized into Ag+ – the partial dissolution of Ag NPs has been attributed to the Ag+ readsorption onto the NP surface.64,65 In fact, Liu and Hurt (2010) demonstrate that three forms of Ag are found in colloidal suspensions of Ag NPs, including solid Ag(0), Ag+ that are available or have formed complexes with nutrients in the surrounding medium, and surface-adsorbed Ag+.65 The buildup of surface Ag+, in turn, blocks the O2 and proton access to metallic Ag, and this effect has been stated to be due to various NP surface coatings or particle agglomerations.65,66 However, others have reported the full dissolution of Ag NPs, and theoretical calculations suggest that Ag NPs are fully soluble in solutions with pH ranging from 4 to 12.65 At the same time, various studies have revealed that Ag NPs exhibit higher toxicity than Ag+.67,68 Specifically, a study by Eom and Choi (2010) indicates a time-dependent cytotoxic effect of Ag NPs,69 an observation that is similar to one made by Llop et al. (2014) on ZnO NPs.57 They show that, within 24 h of cellular incubation with Ag NPs and Ag+, the levels of oxidative stress were similar for both species. After 24 h, Ag NPs exhibit significantly higher toxicity compared to free Ag+, suggesting that they may serve as a long-term source of constant ion release. This data supports the notion that free Mz+ disperse themselves throughout the cells, while NPs tend to accumulate within endosomal compartments and release their Mz+ locally at high concentrations, exceeding toxic thresholds.
2.1.2. NP degradation disrupts the local Mz+ homeostasis
Under normal physiological conditions, the lowly abundant free intracellular Zn2+ are mainly protein-bound (i.e. with MTs) or contained within lysosomes and mitochondria, and their homeostasis is maintained by the Zn transporters (ZnTs) and Zrt/Irt-like proteins (ZIPs) that decrease or increase the level of cytosolic Zn, respectively, through extracellular release or sequestration into organelles and vesicles.70,71 In fact, ions of Zn and Cu that are located within the mitochondrial intermembrane space have been reported as catalysts in the conversion of superoxide anions into H2O2.72 Various ZnTs have been seen to co-localize with lysosomal proteins, and the increase in Zn2+ flux within lysosomes as a result of ZnT 2 overexpression highlights the importance of this organelle in maintaining cellular Zn levels.71 Upon ZnO NP dissolution within the cells, the excess burst-like release of Zn2+ is much higher than the rate of ion excretion by ZnTs, which disturbs the local Zn homeostasis, leading to ROS generation. The Zn2+ tend to also accumulate within the mitochondria, causing the organelle's damage and loss of its membrane potential (MMP) and ultimately, apoptosis.73 Among the various biological effects, Zn2+ have been shown to interfere with cytochrome bc1 and α-ketoglutarate dehydrogenase in complex III and I of mitochondria, respectively, significantly impeding the process of cellular respiration and leading to ATP depletion.74–76 Furthermore, it has been reported that Zn2+ levels and their buffering capacity are generally lower in CCs – which have lower thresholds of oxidative stress compared to their normal counterparts.77 As a result, the CCs tend to be more sensitive to elevating intracellular Zn2+ levels, providing a therapeutic window for ZnO NPs with finely-tuned dissolution kinetics to specifically target CCs.
Similarly, the homeostasis of Cu2+ is maintained by Cu importers such as CTR1 and ATPase that respectively increase or decrease the flux of Cu2+ within the cell.78,79 When the level of intracellular Cu2+ is increased beyond normal conditions, CTR1 is internalized while ATPase is transferred to the cell membrane to eliminate the excess ions. However, during cellular uptake and subsequent dissolution of CuO NPs, the release of Cu2+ is significantly higher and more rapid than the capacity of ATPase to excrete the excess ions, disrupting the redox balance and inducing ROS generation and oxidative stress. The cellular homeostasis of Ag is also tightly regulated by metal transport genes and proteins. In a study by Wang et al. (2015), exposure of cells to Ag NPs led to an increase in mRNA expression levels of genes responsible for the extracellular release or sequestration of Mz+ and metal-bound complexes into lysosomes, including ATP-binding cassette transporter subfamily C member C1 (ABCC1), divalent metal transporter 1 (DMT1) and Cu-transporting ATPase 1 (ATP7A).80 The presence of Ag NPs within the lysosomal compartments also led to the disruption of the organelle membrane, followed by a significant reduction in MMPs after 24 h.
2.1.3. NP-induced DNA damage, autophagy, and necrosis
Elevated levels of oxidative stress may result in DNA damage (characterized by the phosphorylation of histone γ-H2AX81), which in turn stimulates the tumor suppressor gene p53 to trigger cell cycle arrest as an attempt to reverse the damage or undergo self-apoptosis.82,83 Whether a cell proceeds with apoptosis is determined by the ratio of pro-apoptotic BAX and anti-apoptotic BCL2 proteins.84 Elevated BAX levels elicit the mitochondrial permeability transition and induce cytochrome c release from the mitochondrial outer membrane.85,86 Once this process is triggered, DNA is fragmented at the internucleosomal linker sites and activated caspase-3 leads to autocatalysis and triggers a caspase signaling cascade.87,88 Accordingly, exposure of different cell types to either ZnO, CuO or Ag NPs leads to overexpression of p53, bax and caspase-3 and downregulation of bcl2.89–91 Concurrently, the activities of SOD, CAT, and glutathione reductase (GSR) and GPx are significantly hindered as a result of NP treatment. While GSR recycles GSH disulfide to GSH and GPx converts lipid hydroperoxides to their hydroxyl form, they both contribute to the resistance towards oxidative stress. As such, due to the resulting NP-mediated antioxidant enzyme depletion, levels of intracellular ROS generation and lipid peroxidation become elevated, alongside intracellular Ca2+ flux, perturbation of mitochondrial membrane electron transfer and cell membrane leakage.92 In attempt to reduce the oxidative damage incurred by NP-mediated ROS generation, exposed cells often undergo autophagy in response to mitochondrial damage, a process whereby the components of a cell such as its organelles are degraded and recycled and that eventually leads to cell death if progressed without interference.93–95 Macroautophagy is the most commonly studied subtype of autophagy and involves the engulfment of the dysfunctional components by LC3-containing autophagosomes, followed by their delivery to the lysosomes, where local enzymes and the acidic pH initiate the degradation process. Among the accompanying cellular responses is the activation of the PI3K/AKT/mTOR signaling pathway. Another form of cell death that has been observed in MO NP-treated cells is necrosis, which is triggered secondarily in cases where apoptosis-associated caspase activation, cytochrome c release, and DNA fragmentation are hindered.94,96,97 Molecules that have shown to be involved the initiation and modulation of necrosis include receptor-interacting proteins 1 (RIP1) and 3 (RIP3), caspase inhibitors, and poly(ADP-ribose) polymerase (PARP).98 Although there is a general agreement regarding the cytotoxic effects of MO NPs, reported results still tend to vary among different studies due to inconsistencies in choices of assays, media, and other biological-related setups as well as differences in synthesis methods, which lead to varying structural defects and NP band structures.99 Specifically, the positions of the valence and conduction band edges of a NP determine its band gap energy, which, in turn, dictates the level of electrons in the conduction band or electropositive holes in the valence band under varying conditions. In the intracellular setting, oxygen molecules can react with the conduction band electrons and valence band holes to produce O2˙− (a precursor for the ROS ˙OH and 1O2) and ˙OH, respectively.100
2.2. Factors influencing NP dissolution
2.2.1. Effect of biological pH on NP dissolution
A major factor that impacts the solubility of these metal and MO NPs is the acidity in the surrounding medium.101 These NPs have been shown to be significantly pH sensitive, with their dissolution proceeding much faster in acidic environments.42,64 Specifically, ZnO NPs have shown to exhibit significantly high dissolution rates in low pH solutions (pH 1.5), reaching apparent equilibrium within 30 minutes, as compared with the equilibrium achieved after 60 minutes at neutral pH.102 In a separate study, ZnO NPs dissolved fully at pH 6.1 and around 80% at pH 7.6 after 5 days in aqueous media.101 Similar results have been confirmed for CuO NPs, in which ion measurements after 72 h of NP exposure in non-complexing buffer solutions indicate full particle dissolution at pH 5.5.103 Studies comparing Ag NP dissolution in neutral and acidic conditions have also revealed greater ion release at low pH.65,104,105 Peretyazhko et al. (2014) explain that the possible mechanism behind this observation is that while the intermediate Ag2O is covered with hydroxyl groups in water, its protonation under acidic conditions weakens the Ag–O bonds.64 Given that NPs are typically taken up into the cells via endocytosis, they experience varying levels of acidity, from pH 7.4 in the extracellular environment to pH 5.5 within endosomes and pH 4.5 in lysosomes,106 thereby releasing Mz+ following acid etching upon exposure to endosomal compartments. The greater dissolution of NPs within lysosomes is also due to the high ionic strength and multitudes of hydrolytic enzymes, including cathepsin L, that non-specifically degrade the protein corona that is tightly bound to the particle surface as well as any polymer coating.107–109 In fact, Kreyling et al. (2015) were able to reveal the clear separation between radiolabeled Au NPs and their radiolabeled polymer coat after being internalized within lysosomes.110 More importantly, this highly pH-dependent dissolution kinetics can be exploited for further selective tumor therapy as the local pH in the tumor microenvironment is lower than in healthy tissue, triggering Mz+ release preferentially in the vicinity of CCs.
2.2.2. Cellular proteins and amino acids affect NP dissolution
NP dissolution is also enhanced in nutrient-rich environments.111 In cells, covalent complexation between the Mz+ released from the MO NPs can occur with proteins or amino acids, causing crystal precipitation in the cytoplasmic fluid and ROS generation, which in turn can cause DNA damage and cell death following the Tier 3 biological pathways. In a recent work from our group, we show that Cu2+ released from CuO dissolution tend to complex with various amino acids and form light blue precipitates in glutamine solutions in the ratio 1 : 2, as confirmed using single-crystal X-ray diffraction (SC-XRD).79 These findings are in line with previously reported structural formations between Cu2+ and glutamine.112 In fact, several early works have described the crystal structure that results from Cu2+ complexation with various amino acids, such as asparagine,113 glutamic acid114 and aspartic acid.115 Records of electron paramagnetic resonance (EPR) spectra in growth medium also indicate that all the Cu2+ released immediately bind with the nutrients present and precipitate to form crystals.79 Additionally, Gilbert et al. (2012) used high-resolution X-ray spectromicroscopy and high elemental sensitivity X-ray microprobe analyses to reveal that the full intracellular ZnO NP dissolution generated Zn2+ that immediately formed complexes with molecular ligands.116 This NP–protein interaction is able to cause protein misfolding, fibrillation, thiol crosslinking and thus loss of function.117–119 Interestingly, in a study by Pokhrel et al. (2013), the authors developed a model explaining that Zn2+ released from ZnO NP dissolution interact with rhombic [2Fe–2S], cuboidal [3Fe–4S], and cubane [4Fe–4S] FeS clusters found in various intracellular proteins, causing changes in cellular responses as a result of the reduced enzymatic activity.42,120,121 Specifically, NP-induced ROS production is able to damage the highly oxidation-sensitive [4Fe–4S]2+ cluster found in the aconitase protein family,122 generating the intermediate [4Fe–4S]2+˙OO2−. This resulting species reacts with free protons and releases Fe2+ to produce [3Fe–4S]3+, which is then rendered inactive ([3Fe–4S]0) upon univalent reduction.123,124 Concurrently, Ag+ have been shown to complex with Cl−, Br− and thiol-containing biomolecules leading to further damage to cellular functionality.125–127 Using SR-XANES, Wang et al. (2015) revealed the chemical transformation of Ag NPs – as a result of surface oxidation by ROS and dissolved O2 – from Ag(0) to Ag2O, which then interacts with organic acid molecules to form AgS.80 Major sources of intracellular thiol include cysteine and methionine-containing peptides, proteins and antioxidants (i.e. MTs), which complex with Ag NPs to form more stable entities.63,128,129 In fact, the presence of Ag NPs caused a 5-fold and 10-fold increase in the mRNA expression of MTs after 12 h and 24 h, respectively, while circular dichroism spectra revealed alterations in the secondary structures of these antioxidants.63,80
2.2.3. A predictive model of NP toxicity based on its dissolution profile
There are various parameters that regulate NP dissolution, including ionic strength, dissolved O2 concentration and complexing ligands, surface coating, shape and size.130–133 For instance, NP size is typically inversely correlated with Ag NP dissolution, with the larger surface-to-volume ratio of smaller particles accounting for greater instability and increased solubility.9 In fact, one method of predicting the toxicity levels of MO NPs is examining specific characteristics including their oxidation number (Z), ionic potential (IP), surface reducibility (SR) and redox reactivity (RR). Based on the above listed physical–chemical parameters, a Bayesian classification system has been established by Simeone et al. (2019), in which they assign different categorical variables for each of the parameters mentioned above to determine the conditional probabilities of the cytotoxicity induced by MO NPs over a range of EC50 values.134 Increasing Z represents the strengthening of the electrostatic interactions within the NP structure and thus lowers the degree of NP dissolution, with oxides of Mz+ with Z ≤ 2, for instance, dissolving more than 10% of the structure while <1% is dissolved for cationic metals of Z > 3. NP surface charge is a key determinant of the level of NP interaction with biological media, particularly in the formation of the protein corona, and it has been repeatedly reported that positively charged NPs are preferentially taken up into cells compared with their negatively charged counterparts.132,133 Particle surface charge is mainly characterized by its acidity or IP, which is expressed as the ratio of Z to its ionic radius (r) and indicates the extent of hydrolysis. IPs of ≤3 and >5 lead to hydrated and deprotonated surface groups, respectively. SR delineates the type of NP surface defects and describes the MO as reducible or oxidizable according to the potential decrease or increase in Mz+ oxidation number, respectively.135 The final parameter RR categorizes NPs with redox potential <−0.8 V (against the standard hydrogen reduction potential at physiological pH) as “non-active”, while NPs with higher values were considered “active” and are thus capable of being reduced by GSH or oxidized by dissolved O2. According to this system, CuO NPs are classified as “highly toxic” due to their low Z (<2), low IP (≤3), surface reducibility, and “active” RR. ZnO NPs are similarly considered highly toxic due to their low Z and IP, although they are “non-reducible” and “non-active”. Both of their high toxicities are mainly attributed to the high likelihood of Mz+ bioavailability as result of their low Z. Within this classification system, the size factor is not incorporated into their assumptions. However, they explain that the chemical mode of interaction with the biological system is not affected, although the probability of this interaction can be increased due to particle size. Furthermore, the effect due to size is difficult to determine as particles can aggregate or dissolve upon interaction with the biological system, resulting in an effective size change. Long-term toxicities caused by aggregation are also not included in this model.
3. Controlling NP dissolution for selective tumor therapy
Selective tumor therapy can thus be achieved by engineering NPs that are responsive to the low pH surrounding tumor tissue and that generate sufficient levels of ROS to induce apoptotic responses in CCs with high metabolic rates, while the additional ROS experienced by normal peripheral cells with higher physiological pH and lower metabolic rates should not reach apoptotic thresholds and only cause temporary reversible damage (Fig. 3). Several methodologies have already been explored to tune the toxicity profiles of the NPs by controlling their distribution, dissolution and oxidative capacity. For instance, Le et al. (2016) conducted a multiparametric evaluation of ZnO NPs with varying physicochemical properties such as size (proportional to calcination temperature), aspect ratio (spheres, grains, rods, or needles), dopants with different concentrations (Fe, Mn, or Co), and coatings (polymethyl-methacrylate PMMA, silica, oleic acid OA, or serum protein) and assessed their effect on human umbilical vein endothelial cells (HUVECs) and hepatocellular liver carcinoma (HepG2) cells using three toxicity endpoints, including viability, membrane integrity and oxidative stress.136 The objective of this study was to determine the parameters that have the greatest impact on toxicity and subsequently develop computational models that would be able to predict NP behavior. The descriptors used for the doping elements were similar although not identical to those described above by Simeone et al. (2019), and include IP, conduction band energy (EC), and reduction potential (RP). With respect to cell viability, the parameters that had the greatest impact include NP concentration and size, extent of NP shielding from surrounding (solubility and coating type), and the resulting change in IP and EC associated with doping, particularly for Mn-doped oxides which had the largest cytotoxic effect, in contrast to serum-coated NPs that enhanced cell viability. Next, membrane damage was measured by the amount of lactate dehydrogenase (LDH) released, and was shown to be significantly affected by IP, EC, and RP, as well as NP shape, size, solubility and zeta potential. Specifically, in line with previously reported studies, positively-charged NPs exhibited higher LDH release compared to their negatively-charged counterparts, possibly due to the greater interaction of the cationic NPs with the abundant negative residues on cell membranes. Finally, antioxidant response elements (AREs) were used as markers for oxidative stress and were shown to be most affected by the dopant descriptors IP, EC and RP, particularly as a result of Mn- and Co-doping. However, in the context of selective cancer therapy using ZnO and CuO NPs, given that these particles in their bare form already exert the toxicity required for apoptosis in most cell lines as will be discussed in Section 4, doping them with elements such as Mn and Co that further induce cellular toxicity does not serve the purpose of reducing the impact on NCs. In contrast, doping these NPs with elements such as Fe reduces their dissolution kinetics and associated toxicity, which provides an opportunity to maintain the viability of normal tissue while only exceeding toxic thresholds in the highly metabolic CCs.
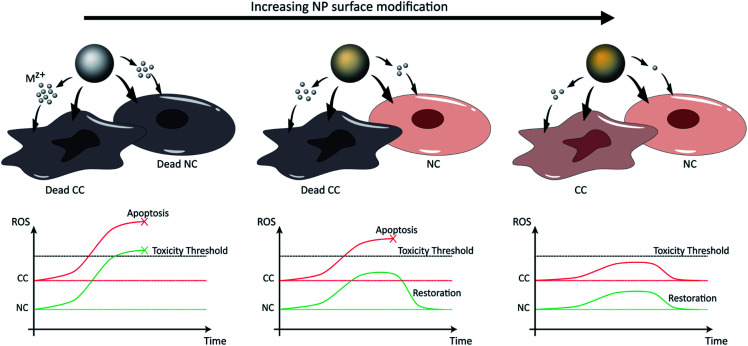
Fig. 3. NP dissolution kinetics and associated toxicity can be tuned by surface modification, including doping and coating, and are increased in the low pH tumor microenvironment. In the first scenario (left), pure NPs release high concentrations of ions that induce ROS generation at levels exceeding toxic thresholds for both CCs and NCs. In the second scenario (right), the excess surface modification of NPs causes a slow release of Mz+ at non-toxic amounts, preserving the viability of both cell types. Finally, in the third scenario (middle), the Mz+ release associated with specific extents of NP surface modification is sufficient to exceed toxic thresholds in CCs that possess high metabolism (higher basal levels of ROS), but low enough to exert only transient damage in NCs with low metabolism and to restore oxidative balance.
3.1. The effect of NP doping on dissolution properties
Doping the NPs with various elements leads to modifications in their electronic state density, magnetic moments, charge distribution, and lattice distortions, altering their reactivity, stability and various other properties. Given that kinetic factors hinder the incorporation of dopants into the NP structure, effective doping is only achieved when the growth rates of the host and dopant are balanced.137 In this way, the dopant properly substitutes the host core atoms rather than simply adsorb onto the NP surface. In an early work by Bilecka et al. (2011), ZnO NPs were doped with Fe, Mn, Ni, Co and V, and the greatest doping efficiency was found for Fe and Co due to their comparable reactivity and size of their divalent ions with Zn.138 Vanadium, however, had the least effective doping (<3% of dopant precursor content) as it has a larger ionic radius and is a much harder Lewis acid compared with Zn.
As a result, Fe-doping has been heavily explored for ZnO and CuO NPs, and has been shown to significantly reduce particle dissolution and cytotoxicity at levels proportional to the extent of doping,92 although the toxicity reduction mechanisms for both NP types differ. Within the CuO NP structure, Cu resides among two and four oxygen ligands in the apical and planar positions respectively, which result in Jahn–Teller distortions upon Fe-doping as well as CuFe2O4 spinal formation.139 The resulting increased structural stability and surface spinal formation hinders excess Cu2+ release. In Fe-doped ZnO NPs, the increased stability compared with pure ZnO is due to the stronger structural binding of Fe than Zn to O, while the Fe atoms simultaneously serve as kinetic constraints for Zn release.140,141 It has also been suggested that the additional cytoplasmic Fe2+ availability originating from the Fe-doped ZnO NPs is able to reduce the previously mentioned oxidized FeS clusters to reverse the protein inactivation process by regenerating the active [4Fe–4S]2+ cluster.42 These reports were further supported by an experiment conducted in E. coli that reveal that unstable [4Fe–4S]2+ clusters tend to form inactive [4Fe–4S]+, and subsequent exposure to Fe ions and dithiothreitol leads to protein reassembly with a 60% gain in protein activity within the span of a few minutes.142 Therefore, this mechanism of protein function salvaging may also contribute to the reduced cytotoxicity observed with Fe-doped NPs compared to their pure counterparts. Additionally, doping MOs results in the narrowing or expansion of the NP band structure and changes in the positioning of the conduction band edges, altering the levels of ROS generation.99
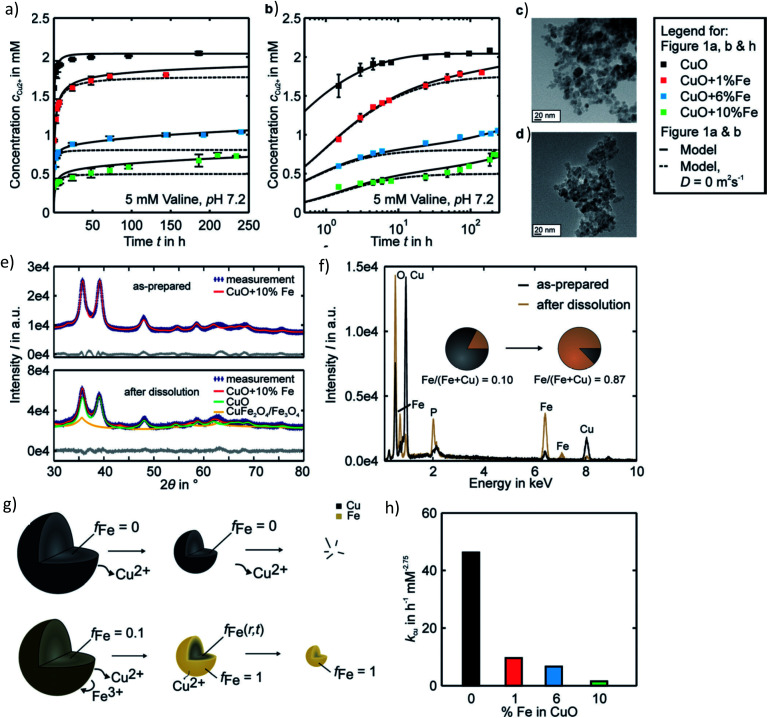
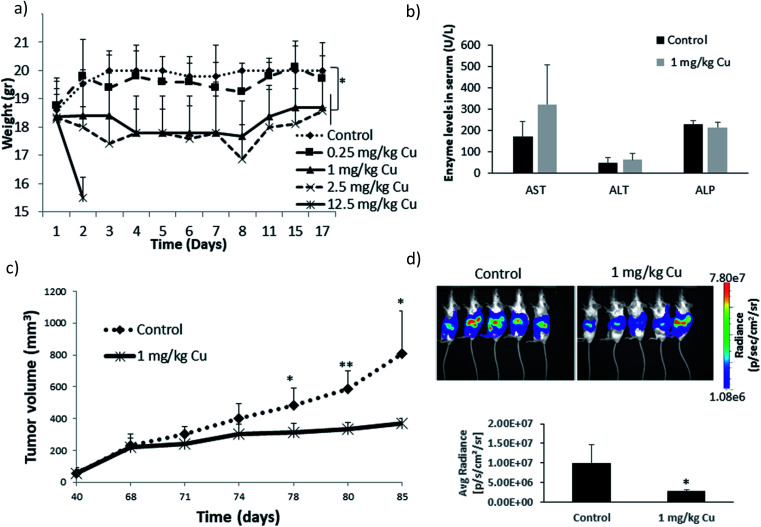
In a recent study from our group investigating the effects of varying Fe doping levels on CuO NP toxicity and cancer therapy, we found that the rapid release of Cu2+ from pure CuO causes cell death in both normal and tumor cells, whereas a too slow release leaves both cell types mostly unaffected.79 By adjusting the Fe-doping levels to fit the exact desired release kinetics of the Mz+, it was possible to selectively target CCs. The most beneficial response was achieved at Fe doping levels of 6%. Although the release rate of 6% Fe-doped CuO NPs was initially rapid and overlapped with that of pure CuO, it significantly diminished with time, leading to a slow long-term release (Fig. 4a and b). This decrease in dissolution rate is due to the strong Jahn–Teller distortion that adjusts the planar and apical Cu–O bond lengths. In fact, a 10% Fe-doping of CuO has been previously associated with a 6.4% plane elongation and 4.1% distortion compared to pure CuO.139 Our EPR spectra showed a Cu release of ∼65% from pure CuO and only 8% from 10% Fe-doped CuO in the first 10 minutes of NP exposure to cell culture medium. Using powder diffraction, Rietveld analysis and EDX spectroscopy, we suggested that dissolution causes a transformation from CuO → CuFe2O4 → Fe3O4 as well as the formation of Fe–O (Fig. 4e and f). Taking into account several assumptions, such as a dissolution rate that is proportional to the surface Cu concentration which itself is proportional to the active surface area for instance, we developed a two-step model to describe the dissolution kinetics of Fe-doped CuO (Fig. 4g). The first step involves the repeated Fe redistribution on the NP surface during Cu2+ release and thus a continuous increase in the Fe/Cu surface ratio until all the surface Cu is released, forming an Fe shell. This step is followed by the slow solid-state diffusion of the core Cu to the surface until all the Cu has been released. The remaining Fe particles are then degraded into their ionic form (i.e. via Fenton reactions, enzymes44), which are then incorporated into the natural Fe metabolism of the cell with the help of ferritin. In fact, this Fe integration has been evidenced through the radioactive labeling of magnetic FeO NPs and the subsequent localization of labeled Fe within the hemoglobin of newly-developed erythrocytes in vivo.22 Therefore, given the safety and success of Fe doping in controlling Mz+ release and reducing NP cytotoxicity, efforts have been made to test various core-dopant combinations for selective tumor therapy, such as Zn-doped CuO NPs, which have shown to have significant anticancer properties in vitro and in vivo. These findings will be discussed further in Sections 4 and 5.
Fig. 4. Pharmacokinetics of Fe-doped CuO NPs as described by Naatz et al.79 (a and b) Cu2+ release profiles showing a slow long-term release in the case of the higher doped NPs in the logarithmic time scale. (c and d) TEM images of 10% Fe-doped NPs before and after dissolution in the span of 4 weeks. (e and f) Powder diffraction patterns and EDX spectra depicting the change in particle composition before and after dissolution of the 10%-doped CuO NPs. (g) Proposed two-step model of Fe-doped CuO NP dissolution wherein all the surface available Cu is first released until an Fe shell forms, followed by the solid-state diffusion of core Cu to the surface until all the particle Cu is removed. (h) Decrease in the dissolution rate constants with increasing Fe doping. This image has been reproduced with permission from Naatz et al.,79 © Wiley-VCH, 2020.
3.2. The effect of NP coating on dissolution properties
Besides doping, surface modification of NPs using a wide variety of coatings have also been heavily investigated for their potential in controlling the dissolution kinetics of NPs. For instance, given that silica is highly stable in neutral and acidic environments, a silica coating has been developed around ZnO NPs with the aim of reducing NP degradation and toxicity.143 This strong hydrophilic coating turns the previously positively-charged surface of the ZnO NPs negative, and the absolute increase in the zeta potential enhances NP stability in water. Optimizing the amount of silica coating could thus result in ZnO NPs with specific degradation profiles. In two separate studies, other types of surface coatings were assessed on either CuO144 or ZnO145 NPs with the aim of completely reducing the surface reactivity and Mz+ release from the particles. Osmond-McLeod et al. (2013) demonstrate almost full reduction of ZnO NP toxicity as a result of surface coating them with dimethoxy-diphenylsilane/triethoxy-caprylylsilane crosspolymer (MAX) compared to pure ZnO.145 However, coating the NPs with triethoxycaprylylsilane (HP1) resulted in moderate cytotoxic response. Given that the aim of the study was to fully reduce NP dissolution as a safety measure and as a result, the HP1-coated NPs were not further investigated, these findings suggest the need to further test the HP1-coated NPs for the purposes of cancer therapy by tuning their dissolution kinetics. Similar conclusions can be derived from the study by Cai et al. (2017), who show that after coating with ethylenediamine tetra (methylene phosphonic acid) (EDTMP), the extent of CuO dissolution was less than 0.2% and almost no ROS generation and toxicity were observed in THP-1 and BEAS-2B cells.144 They also report that the coating does not disrupt the CuO crystal structure and morphology. Meanwhile, they tested the impact of coating NPs with citrate and PVP (polyvinylpyrrolidone), which had limited and moderate protective effects, respectively. Similarly to the case of HP1-coated ZnO NPs, optimizing NP dissolutions profiles may be achieved by investigating varying degrees of NP coating with PVP. A follow-up study by Líbalová et al. (2018) revealed that varying NP surface modifications (anionic sodium citrate CIT, sodium ascorbate ASC, neutral PVP, cationic polyethylenimine PEI) only moderately affected Cu2+ release and that the toxicity observed in a murine macrophage cell line was mainly determined by the NP coating agent and Cu bioavailability rather than the intracellular Cu burden, with the PEI-coated NPs being the most toxic.47
4. Cellular studies on cancer-specific NP toxicity in vitro
Although a few types of doping and coatings were introduced in the previous section, there exists a variety of other surface materials that have been experimented with in order to fine tune the dissolution kinetics of NPs. In this next part, we initially discuss the various studies that have tested the anticancer activity of pure ZnO, CuO, and Ag NPs in vitro, followed by the efforts that have been made over the years to further enhance CC selectivity using the assorted doping and coating materials and their cellular responses. Please note that a wide range of different cell types have been used in the various studies, which will not be described in detail as this would be outside of the scope of the current manuscript. For clarity purposes, we have listed all cell types described with their abbreviation and full name in Table 1. A summary of all the discussed studies is also provided in Tables 2–4.
Cell types referred to in the discussed studies and their abbreviations.
| Cell line | Type | Cell line | Type |
|---|---|---|---|
| A172 | Human glioblastoma | KCL22 | Human chronic myeloid leukemia |
| A549 | Human lung adenocarcinoma | KLN205 | Murine lung carcinoma |
| AsPC-1 | Human pancreatic tumor | L02 | Human hepatic |
| B16 | Murine melanoma | L5178Y-R | Murine lymphoma |
| BEAS-2B | Human lung epithelial | LS174T | Human colon cancer |
| BeL7402 | Human hepatocellular carcinoma | MCF-7 | Human breast cancer |
| BJ | Human normal fibroblasts | MCF-7/ADR | MCF-7 adriamycin-resistant (ADR) |
| BxPC-3 | Human pancreatic cancer | MDA-MB-231 | Human breast cancer |
| C6 | Rat glioma | MG-63 | Human osteosarcoma |
| CHO | Chinese hamster ovary epithelial | MIA Paca-2 | Human pancreatic cancer |
| COLO 205 | Human colon adenocarcinoma | MSC | Mesenchymal stem cells |
| CT26 | Murine colorectal carcinoma | N417 | Human small cell lung cancer |
| DAL | Canine hemangiosarcoma | NCI-H460 | Human non-small lung cancer |
| EAC | Murine Ehrlich Ascites carcinoma | NIH3T3 | Mouse embryonic fibroblasts |
| ESC | Embryonic stem cells | OUS-11 | Human lung normal |
| H1299 | Human non-small lung cancer | PANC1 | Human pancreatic cancer |
| H187 | Human small cell lung carcinoma | Panc28 | Human pancreatic adenocarcinoma |
| H82 | Human small cell lung carcinoma | PBMC | Peripheral blood mononuclear cells |
| HCC | Human hepatocellular carcinoma | PC3 | Human prostatic cancer |
| HCT116 | Human colorectal carcinoma | RAW264.7 | Murine macrophage |
| HeLa | Human Henrietta Lacks immortal cells | SHSY5Y | Human neuroblastoma |
| HepG2 | Human liver hepatocellular carcinoma | SK-MES-1 | Human lung cancer |
| HT-29 | Human colon cancer | SKBR3 | Human breast cancer |
| HT1080 | Human fibrosarcoma | SUM159 | Mesenchymal TNBC |
| hTERT-HPNE | Immortalized human pancreatic duct | TNBC | Triple-negative breast cancer |
| HUVEC | Human umbilical vein endothelial | U251 | Human glioblastoma |
| Jurkat | Human immortalized T lymphocytes | U87 | Human primary glioblastoma |
| K562 | Human myelogenous leukemia | WI38 | Human lung fibroblasts |
| K562/A02 | Human leukemic | WISH | Human amnion |
Summary of In vitro and in vivo studies testing the effects of pure and surface-modified ZnO NPsa.
| Surface mod. | Size (in nm) | Main finding | VT | VV | Model systems | Dose/concentration | Cellular/tumoral effects | CCS | Mechanism | Ref. |
|---|---|---|---|---|---|---|---|---|---|---|
| Pure | 21C, 131HD | ZnO NPs selectively induce apoptosis in CCs via ROS, mediated by p53, bax/bcl-2 and caspase pathways | ● | HepG2, A549, BEAS-2B, rat astrocytes & hepatocytes | 5–15 μg mL−1 | Dose-dependent ↓ cell viability | ● | ↑ p53 and bax and ↓ bcl2 | 89 | |
| Pure | 5C, 6C | Newly synthesized ZnO QDs with promising optical properties show cytotoxic effects in CC lines | ● | K562, K562/A02, HepG2 | 1.25–100 μg mL−1 | Dose-dependent ↓ cell viability | 146 | |||
| Pure | 35C | ZnO NPs restore oxidative balance, hepatocyte integrity and tumor markers to normal levels in HCC | ● | ● | VT: HepG2, PC3, A549; VV: HCC | VT: 0–1000 μmol L; VV: 10 μg kg−1 per week | Dose-dependent ↓ cell viability | Restoration of oxidant and antioxidant activity (MDA, GSH, GPx, GSR, SOD, CAT), hepatocyte integrity (ALT, AST, LDH) and tumor biomarkers (AFP, AFU) to normal levels in CCs | 147 | |
| Pure | 95HD | ZnO NPs exert toxicity in normal and CCs through ROS generation, whereas CeO2 and TiO2 NPs do not | ● | A549, NCI-H460, SK-MES-1, HeLa, Jurkat, AT II | 2–6 μg cm−2 | ↓ cell viability | Complete particle dissolution and ↑ ROS | 57 | ||
| Pure | 100HD | ZnO and Ag NPs evoke stress-induced autophagy in pulmonary and hepatic cells while TiO2 NPs do not | ● | A549, HepG2 | 1–500 μg mL−1 | Dose-dependent ↓ cell viability | Autophagy induction (↑ LC3B, atg4b, p62 and ↓ atg12, atg5) in a time-dependent manner, and apoptotic cell death via caspase-3 | 148 | ||
| Pure | 40C | Apoptosis and oxidative stress as relevant mechanisms of antitumor activity and genotoxicity of ZnO-NPs | ● | ESC | 50, 300 and 500 mg kg−1 per body weight | Oxidative stress (↑ MDA and ↓ CAT, GST) and DNA damage. Apoptosis (↑ Bax and p53, ↓ Bcl2). NAC restores oxidative balance in the liver and kidney without ↓ the antitumor efficacy of NPs | 184 | |||
| Pure | 20HD, 70HD | Genotoxic anticancer effects of ZnO NPs via ROS, leading to non-apoptotic cell death in an orthotopic mouse model of human small-cell lung cancer | ● | ● | VT: H82, H187, BEAS-2B, MCF-7, OUS-11, LS174T, N417; VV: N417 | VT: 0–20 μg mL−1; VV: 0.04–0.25 mg kg−1 | Dose-dependent ↓ cell viability. ↓ tumor density | ● | ↑ ROS and DNA leakage from nuclei. Phos. of CHK2 in N417 and LS174T cells. ↑ cleaved PARP in LS174T cells. Q-VD-OPh (inhibitor of caspase-3, 1, 8, 9, slightly ↑ viability by inhibiting apoptosis, but did not block ZnO toxicity) | 185 |
| Fe-doped (0–10%) | 20.2C–8.3C (0–10%) | Fe doping reduces ZnO toxicity in animals due to decreased NP dissolution rates and associated toxicological responses | ● | ● | VT: RAW264.7, BEAS-2B; VV: rat & mouse lung, zebrafish embryo | VT: 12.5 μg mL−1; VV: 0–50–150 μg mL−1 | ↓ toxicity with ↑ Fe doping | ↑ Fe doping leads to ↓ inhibitory effect of Zn2+ in zebrafish embryo hatching, ↓ PMN cell counts in the BAL fluid and IL-6 mRNA and ↑ heme oxygenase 1 in mouse lung, and ↓ BAL PMN cell counts, LDH, and albumin in rat lung | 163 | |
| Fe-doped (0–10%) | 11C, 5.5C (0, 10%) | 2% Fe-doped ZnO NPs are found to be optimal to cause selective CC death and reduce metastasis formation | ● | ● | VT: mMSC, Beas-2B, HeLa, KLN205; VV: KLN 205 | VT: 0–35 μg mL−1; VV: 125 μg per animal | ↓ cell viability. ↓ toxicity with ↑ Fe doping. ↓ tumor growth and metastasis (2, 10%) | ● | ↑ ROS, membrane and mitochondrial damage and autophagy. ↓ toxicological response with ↑ Fe doping due to resulting ↓ in cellular levels of Zn2+. VV: pure NPs led to weight loss and premature death of mice | 77 |
| La-doped (0–5%) | 33C, 29C (0, 5%) | Doping ZnO NPs with La increases NP photocatalytic activity and cytotoxicity | ● | MDA-MB-231, KCL22, HeLa | 6–500 μg mL−1 | Dose-dependent ↓ viability. ↑ toxicity with La doping | 164 | |||
| Sm-, Eu-, Gd-doped | >100C | Sm-doped ZnO NPs displays the most significant antitumor activity compared to other lanthanide-doped ZnO NPs | ● | ● | EAC | VT: 0–0.05 mol; VV: 150–350 mg kg−1 | Dose-dependent ↓ cell viability for Sm3+ : ZnO NPs | Compared to pure NPs, Sm-doping ↓ tumor size, ↓ PI3K, Akt and mTOR, ↓ CXCR4 and P450, ↓ Bcl2 : Bax ratio, ↓ liver function enzymes (AST, ALT) and induce G2 cell cycle arrest | 165 | |
| PEG or starch coated | 40–1200HD | Toxicity towards osteoblast CCs is dependent on NP size, aspect ratio and coating. PEG-capped NPs exhibit higher toxicity than starch-capped NPs | ● | MG-63 | 1 μM to 7 mM | Dose-dependent ↓ cell viability. ↓ toxicity with ↑ NP size | 167 |
Ccore size, HDhydrodynamic size, VT in vitro, VV in vivo, CCS cancer-cell specific.
Summary of in vitro and in vivo studies testing the effects of pure and surface-modified CuO NPsa.
| Surface mod. | Size (in nm) | Main finding | VT | VV | Model systems | Dose/concentration | Cellular/tumoral effects | CCS | Mechanism | Ref. |
|---|---|---|---|---|---|---|---|---|---|---|
| Pure | 7C, 127HD | CuO NPs inhibit pancreatic tumor growth primarily by targeting TICs via ROS and mitochondrial pathway | ● | ● | PANC1 | VT: 0–50 μg mL−1; VV: 0–12.5 mg kg−1 | Dose- and time-dependent ↓ cell viability. Tumor growth inhibition | ● | ↑ ROS, ↓ MMP, apoptosis of TICs (arrest in sub G1 phase) | 149 |
| Pure | 22C, 167HD | CuO NPs induce mitochondria-mediated apoptosis in human hepatocarcinoma cells | ● | HepG2 | 0–50 μg mL−1 | Dose-dependent ↓ cell viability | Oxidative stress (↑ MDA, ↓ GSH) ↑ ROS, DNA damage. Mitochondria-mediated apoptosis (↓ MMP, ↑ P53, ↑ BAX/BCL2 and caspase-3) | 150 | ||
| Pure | 20C | CuO NPs induce cytotoxicity via mitochondrial pathway | ● | K562, PBMC | 0–25 mg mL−1 | Dose-dependent ↓ cell viability | ● | ↑ ROS, mitochondria-mediated pathway, ↑ P53 and Bax/Bcl2 | 151 | |
| Pure | 30C, 235HD | Autophagy is the main mechanism of CuO NP-induced cell death, while apoptosis is only triggered secondarily | ● | MCF7 | 0–12 μg mL−1 | Dose- and time-dependent ↓ cell viability | Autophagy: ↑ MAP-LC3-II, Beclin1 and ATG5. 3 MA inhibits autophagy, and further Beclin1 KD leads to apoptosis (↑ PARP-cleavage, BAD dephosphorylation and caspase-3) | 95 | ||
| Pure | 12C | CuO NPs synthesized from Eucalyptus globulus induce apoptosis in breast carcinoma | ● | MCF7 | 0–100 μg mL−1 | Dose-dependent ↓ cell viability | Impaired MMP, ↑ ROS, ↑ p53, bax, caspase-3, and caspase-9, cell cycle arrest in G1, S and G2/M phases | 90 | ||
| Fe-doped (0–10%) | 11.8C–10.7C (0–10%) | 6% Fe-doped CuO NPs induce inhibition of tumor growth and complete tumor remission when combined with immunotherapy | ● | ● | VT: MSC, Beas-2B, HeLa, KLN205; VV: KLN205 | VT: 0–35 μg mL; VV: 0–225 μg kg−1 bw (6%) | ↓ toxicity with ↑ Fe doping. ↓ tumor growth (6%-doped) | ● | ↑ membrane damage, ROS, autophagy. ↓ toxicity with ↑ Fe doping. VV: ↑ local antitumor immune response (activation of CD8+ and NK cells). Complete tumor remission due to treatment with 6%-doped NPs and EPAC | 79 |
| Fe-doped (10%) | 235HD, 247HD (0, 10%) | Fe-doping of CuO NPs lowers their toxic potential on glioblastoma cells by slowing down Cu release | ● | C6 | 0–1000 μM | Dose- and time-dependent ↓ cell viability. ↓ toxicity with ↑ Fe doping | ↑ ROS and oxidative stress. Cu chelators can prevent Cu-induced toxicity | 168 | ||
| Zn-doped | 30C | Zn–CuO NPs exert selective antitumor activity (inhibition of glioblastoma growth) and reverse temozolomide resistance in glioblastoma by inhibiting AKT and ERK1/2 | ● | ● | VT: Panc28, HCT116, U87, C6, HELA, BeL7402, U251, A172, HUVEC, NIH3T3; VV: U87 | VT: 5.0–20.0 μg mL−1; VV: 0–100 mg kg−1 | Dose-dependent ↓ GBM cell proliferation. ↓ tumor growth, cell migration and invasion | ● | ↑ ROS, apoptosis (↑ procaspase-9, procaspase-3 and ↓ bcl-2/bax ratio), inhibition of AKT and ERK1/2 | 169 |
| Zn-doped | 30C | Zn–CuO NPs inhibit pancreatic cancer growth by inducing autophagy through AMPK/mTOR pathway | ● | ● | VT: AsPC1, MIA Paca2, HepG2, BxPC3, PANC1, HT29; VV: AsPC1 | VT: 0–160 μg mL−1; VV: 5 and 10 mg kg−1 | Dose-dependent ↓ cell viability. Inhibition of tumor growth | ↑ ROS. Autophagy induced via AMPK/mTOR pathway (↑ p-AMPK, p-ULK1, Beclin-1 and LC3-II/LC3-I ratio, ↓ mTOR phosphorylation) | 170 | |
| Zn-doped | 2–10C | Zn–CuO NPs inhibit tumor growth by NF-κB pathway. NAC restores the balance disrupted by autophagy and apoptosis | ● | ● | VT: HepG2, Panc28; VV: Panc28 | VT: 0–40 μg mL−1; VV: 5 and 10 mg kg−1 | Dose-dependent ↑ cell proliferation inhibitory rates | NF-κB signaling involved in ROS-induced apoptosis (↑ Bax and caspase 3, Bcl-2 ↓) and autophagy (↑ LC3B and LC3 B/A). DNA, ER & Golgi damage. All effects restored with NAC | 172 | |
| Zn-doped | 3C | Zn–CuO NPs inhibit human CC growth through ROS-mediated NFκB activations | ● | HepG2, Bel7402, A549, Panc28, HT1080, Hela, HUVEC, L02 | 0–60 μg mL−1 | Dose-dependent ↑ cell proliferation inhibitory rates | ● | ↑ ROS and NF-κB pathway activation (↑ p-IKKα/β and nucleus p-NF-κB p65, ↓ IKKα, IKKβ, IκBα and nucleus NF-κB p65 expression). Induction of G2/M cell cycle arrest | 171 | |
| Carbon (C)-coated | 10.4–19.4C (CuO–C/Cu) | C-coat decreases cytotoxicity of CuO NPs due to reduced solubility, and CuO NPs induce greater toxicity than Cu2+ | ● | CHO, HeLa | 30 ppm C/Cu, 34 ppm CuO | Dose-dependent ↓ cell viability. ↓ toxicity with C-coating | ● | 103 | ||
| Protein coating (DMSA) | 141HD (pure), 167HD (coated) | pCuO-NP-induce cell death in glioblastoma cells to a lesser extent than pure CuO NPs, due to reduced Cu ion release | ● | C6, primary astrocytes | 0–1000 μM | Dose- and time-dependent ↓ cell viability. ↓ toxicity with protein coat | ↓ LDH and ↓ MTT reduction capacity. Cu chelators and low temperature ↓ toxicity | 173 |
Ccore size, HDhydrodynamic size, VT in vitro, VV in vivo, CCS cancer-cell specific.
Summary of in vitro and in vivo studies testing the effects of pure and surface-modified Ag NPsa.
| Surface mod. | Size (in nm) | Main finding | VT | VV | Model systems | Dose/concentration | Cellular/tumoral effects | CCS | Mechanism | Ref. |
|---|---|---|---|---|---|---|---|---|---|---|
| Pure | 8–22C | Ag NPs synthesized from plant extracts induce anticancer activity in lung cancer models | ● | ● | H1299 | VT: 2–30 μg mL−1; VV: 10 μg g−1 | Dose-dependent ↓ cell viability. ↓ tumor size & growth | Morphological alterations, ↓ NF-κB transcriptional activity and Bcl2, ↑ caspase-3 and survivin | 152 | |
| Pure | 5–35C | Bio-synthesized Ag NPs exhibit anticancer, antioxidant and anti-angiogenic activity with no adverse effect on liver and kidney | ● | DAL | VT: 0–100 μg mL; VV: 50 μg mL−1 | Dose-dependent ↓ cell viability ex vivo | 187 | |||
| Pure | 9.4–25.9C | Ag NPs synthesized from Nostoc linckia extracts exert anticancer activity in breast cancer models | ● | ● | VT: MCF-7, WI38, WISH; VV: EAC | VT: 1.56–100 μg mL−1; VV: 5 mg kg−1 | Dose-dependent ↓ cell viability. ↓ cell volume, cell count and weight of tumors | ● | 153 | |
| Pure | 31.8C | Ag NPs synthesized from Acorus calamus show antitumor activity with complete NP elimination 89 days post-treatment | ● | ● | VT: Hep2, COLO 205, SH-SY5Y | 0–200 μg mL−1 | ↓ cell viability | ↑ ROS, ↓ MMP, ↑ caspases-8, 9 and 3, ↑ lamin and PARP, ↑ MDA, ↓ SOD, GPx and CAT | 154 | |
| Pure | 8–20C | Ag NPs synthesized from Agaricus bisporus cause apoptosis and anti-angiogenesis in combination with gamma radiation | ● | ● | VT: MCF-7; VV: EAC | VV: 0.1–10 μg kg−1 | Dose-dependent ↓ cell viability | ↑ caspase-3, ↑ nitric oxide and MDA, ↑ ROS, DNA damage | 155 | |
| Pure | 2.6C, 18C | Ag NPs induce size-dependent cell death in chemo-resistant pancreatic CCs via apoptosis, autophagy, necroptosis and mitotic catastrophe | ● | PANC-1, hTERT-HPNE | 0–5μg mL−1 (2.6 nm), 0–100 μg mL−1 (18 nm) | Dose-dependent ↓ cell viability. ↓ toxicity with ↑ NP size | ● | Apoptosis: ↑ bax, p53, ↓ bcl-2. Necroptosis: ↑ MLKL, RIP1, RIP3. Autophagy: ↑ LC3-II | 91 | |
| Citrate-coated | 5C | Ag NPs initiate antitumor effects and trigger the activation of a tumor cell-specific immune response | ● | ● | VT: HeLa, A549, KLN205; VV: KLN205, CT26 | VT: 5–50 μg mL−1; VV: 100 μg | Dose-dependent ↓ cell viability. Tumor inflammation and ↓ tumor size | ↑ ROS, mitochondrial damage, autophagy, immunomodulatory effects (↑ NFκB pathway and ↑ IL-1α) | 175 | |
| PVP-coated | 30–50 | Ag NPs induce toxicity to CCs via necrosis and increase survival of mice | ● | ● | L5178Y-R | VT: 9–579 nM; VV: 20 mg kg−1 | Dose-dependent ↓ cell viability | 97 | ||
| PVP-coated | 5–75C | Ag NPs show selective cytotoxicity against TNBC cells regardless of size, shape or coating | ● | ● | VT: TNBC, MDA-MB-231, SUM159; VV: TNBC | VT: 0–60 μg mL−1; VV: 6 mg kg−1 | ↓ proliferation. ↓ tumor growth rate, ↑ survival rate | ● | Impairment of cellular redox balance, ↑ ER stress, UPR activation, ↑ CHOP, DNA damage | 178 |
| EPS-coated | 11C | Ag NPs biogenerated by Klebsiella oxytoca DSM 29614 show anticancer activity mainly by induction of autophagy | ● | SKBR3 | 5 and 50 μg mL−1 | Dose- and time-dependent ↓ colony-forming ability | ↑ ROS. Autophagy: ↑ ATG5, ATG7, LC3-II and Beclin-1, ↓ AKT, p-AKT, p62 and HSP90 | 177 | ||
| Ag+-R, Ag0-R | <2C | Compared to Ag+-R NCs, Ag0-R nanoclusters (NCs) exhibit greater release of Ag species | ● | BJ | 62.5, 250 and 1000 mM | Toxicity by Ag0-R NCs > Ag+-R NCs | Activation of p53 | 176 | ||
| TAT-coated | 8C | Antitumor activity in both multidrug resistant and non-resistant CCs is greater with TAT functionalization | ● | ● | VT: HeLa, MCF-7(/ADR), B16; VV: B16 | VV: 1 nmol kg−1 | ↓ cell viability. ↓ tumor growth at lower doses than DOX | 191 |
Ccore size, HDhydrodynamic size, VT in vitro, VV in vivo, CCS cancer-cell specific.
4.1. CC-specific toxicity of pure NPs
4.1.1. ZnO NPs
Throughout the years, ZnO NPs have been tested for their toxicity in a variety of cell types and have clearly demonstrated selective anticancer properties in vitro. In an early study by Akhtar et al. (2012), the underlying mechanisms of ZnO NP cytotoxicity were investigated in three human CC lines and two primary rat cell lines (astrocytes and hepatocytes).89 The NPs were shown to selectively kill CCs without having an impact on normal tissue, and subsequent molecular data demonstrated that the toxicity mechanisms include the upregulation of mRNA and protein levels of p53 and bax, as well as the downregulation of bcl-2. Moreover, ZnO NPs induced the activation of caspase-3 and DNA fragmentation as a result of ROS generation and oxidative stress. In a similar study, two different ZnO NPs – green- and yellow-green-emitting – were shown to have selective cytotoxic impact on human CC lines including HepG2, leukemia K562 and K562/A02.146 Increasing the concentration of these NPs directly impacted their cellular uptake and thus, toxicity. Hassan et al. (2017) also reported enhanced toxicity associated with increasing ZnO NP concentration in three human CC lines (HepG2, prostate cancer PC3, and lung adenocarcinoma A549), although the absolute toxicity levels differed between cell types.147 Additionally, in a study comparing the effects of Al2O3, TiO2, CeO2−x and ZnO NPs on A549, NCI-H460 (hypotriploid cell line), SK-MES-1 (lung CC line), HeLa, and Jurkat (T-lymphocytes to study T-cell Leukemia), ZnO NPs exerted the highest toxicity after a 24 h exposure compared to the remaining NPs which exhibited minimal to no impact, and their subsequent coating with protein coronas significantly improved cell viability.57 Another comparative study assessing the viability of HepG2 and A549 cells in response to treatment with various NPs showed that the 50% lethality (LC50) was reached for both cell types treated with Ag and ZnO NPs as well as increased caspase-3 expression levels, whereas minimal toxicity was observed in TiO2 NP-treated cells.148
4.1.2. CuO NPs
As with ZnO NPs, CuO NPs have been heavily investigated as anti-cancer therapeutic agents in a wide variety of cell lines. In human pancreatic CC (PANC1) cultures enriched with tumor initiation cells (TICs), a considerable decrease in cell viability was found upon CuO NP treatment compared with standard PANC1 cultures.149 TICs are a subpopulation of CCs with stem cell properties, which are highly resistant to most of the current treatments and are therefore assumed responsible for tumor regrowth after therapy. Underlying mechanisms of their therapeutic resistance are linked to ROS scavenging, as lower ROS levels are exhibited in TICs compared to other cells in the tumor tissue. Interestingly, however, administration of CuO NPs was able to induce elevated levels of ROS and oxidative stress in TICs, leading to mitochondrial damage and apoptosis.
Similarly to the previously reported findings concerning the cytotoxic effects of ZnO NPs, CuO NPs were found to have a dose-dependent (DD) toxic effect on HepG2 (ref. 150) and K562 (leukemia)151 cells in two separate studies, and were associated with the upregulation of p53, increased ratio of BAX/BCL2, and decrease in MMP, suggesting that a mitochondria-mediated pathway is involved in CuO NP-induced apoptosis. Expression levels of caspase-3 were also measured in the HepG2 cell line and were found to be elevated. Multiple studies have also tested the effects of CuO NPs in human breast CCs. In the MCF7 cell line, CuO NPs induced dose- and time-dependent autophagy as a possible defence mechanism against CuO-induced toxicity.95 Apoptosis-related cell death was indicated by PARP cleavage, bcl2-associated death promoter (BAD) de-phosphorylation and increased caspase 3 cleavage. A more recent in vitro study on MCF7 cells by Ali et al. (2020) used CuO NPs synthesized from Eucalyptus globulus leaf extract (ELE) with anticancer and antifungal activity.90 They demonstrated a considerable reduction in cell survival via an impairment of MMP and elevation in intracellular ROS. Cell cycle analysis indicated disruption/arrest in different phases (G1, S and G2/M phases) upon CuO NP administration, leading to apoptosis. This data was further confirmed by the upregulated expression levels of p53, bax, caspase-3, and caspase-9 genes.
4.1.3. Ag NPs
Dose-dependent toxicity in a variety of cell lines has also been observed in the context of Ag NPs. For instance, studies testing the effects of Ag NPs on human lung cancer (H1299)152 and MCF-7 cells153 showed a DD decrease in viability. In the H1299 cells, induction of apoptosis, confirmed by the assessment of morphological characteristics such as cell shrinkage and nuclear condensation also displayed such a relationship.152 As seen with ZnO and CuO NPs, a decrease in bcl2 and NF-κB transcriptional activity was detected, alongside an increase in both caspase-3 and survivin levels. In the MCF-7 cells, a lower IC50 value was obtained as compared to two NC lines – human lung fibroblast cells (WI38) and human amnion cells (WISH) – further highlighting the selective antitumor properties of Ag.153
With the aim of circumventing the use of toxic and expensive chemicals, Nakkala et al. (2018) synthesized Ag NPs using a green pathway (rhizome extract of Acorus calamus), and these NPs significantly impacted the viabilities of different CC lines, including Hep2 (human epidermoid carcinoma), COLO 205 (human colon adenocarcinoma) and SH-SY5Y (neuroblastoma).154 Hep2 cells, which clearly showed lower IC50 values after 24 h, displayed characteristics of late apoptosis such as the rounding up of nuclei, cell shrinkage, nuclear condensation and fragmentation, and loss of MMP. Furthermore, an increase of ∼66% in ROS levels and 1.2-fold in MDA levels (marker of oxidative stress), as well as a decrease in SOD, GPx and CAT antioxidant activity were detected. It was thereby concluded that both intrinsic and extrinsic apoptotic pathways are involved in the cell death of Ag NP-treated cells. Other biologically-synthetized Ag NPs were also shown to induce a DD reduction in viability in MCF-7 cells.155 The selectivity of Ag towards CCs was further highlighted in a study by Zielinska et al. (2018), wherein 2.6 and 18 nm Ag NPs exerted higher cytotoxicity against PANC-1 CCs compared to non-tumor cells of the same tissue (hTERT-HPNE cells).91 Both Ag NPs induced, in a size- and concentration-dependent manner, a significant decrease in proliferation and cell death after 24 h, which appeared to be regulated by apoptosis, necroptosis, autophagy and/or mitotic catastrophe as a result of alterations in the protein levels associated with each mechanism. The role of apoptosis in Ag NPs-induced PANC-1 cell death was confirmed by significant increases in the levels of BAX and P53 and very low levels of BCL2. Evidence for necroptosis and autophagy was shown by a size- and concentration-dependent increase in MLKL, RIP1 and RIP3 protein levels and an increase in LC3-II, respectively. Finally, the role of mitotic catastrophe in Ag NPs-induced cell death was confirmed by the observed increase in cell size combined with multinucleation.
4.1.4. IONPs
Various IONP formulations have been clinically approved as MRI contrast agents, Fe replacement strategy or as mediator for magnetic hyperthermia in cancer therapy.156 While IONPs are therefore used in cancer therapy, and furthermore are known to degrade in a pH-dependent manner, this degradation has thus far not been linked to a cancer-selective toxicity, to our knowledge. Fe is essential to many biological functions.157 However, much like other MO NPs, the redox properties of IONPs which are intimately associated to its activity with proteins can potentially give rise to toxicity. The presence of ferric ions in the degradative environment of lysosomes can lead to the production of free oxide radicals due to the Fenton reaction, which can cause substantial toxicity when intracellular concentrations of IONPs are exceeded. This degradation followed by the Fenton reaction has been shown to be particularly affecting cells of neuronal lineage, as was first demonstrated by Pisanic and colleagues.158
IONPs have been shown to induce some interesting effects, linked to their degradation, which are unique among the other NPs. Firstly, when relatively low levels of IONPs are used, the released ferric ions can promote cell proliferation, as demonstrated for mesenchymal stem cells, as Fe is a vital component in cell cycle progression.159 Secondly, upon degradation of IONPs, it has been shown that they keep evolving with time. Initially, they undergo a rapid burst of degradation, after which ferric ions are transported to ferritin for long-term storage under its ferrihydrite form ref. 160 and 161. It has been shown that stem cells can however resynthesize novel biogenic IONPs with a magnetite structure.162
4.2. Effect of NP doping and surface modification on cellular toxicity in vitro
4.2.1. Metal-doped or coated ZnO NPs
Although the pure forms of the NPs have been shown to have intrinsic anticancer properties, this selectivity towards CCs can be further optimized by controlling the dissolution kinetics of the NPs. As previously mentioned, one method of fine-tuning Mz+ release is by doping these NPs with other metals. Specifically, Fe doping exhibits high efficiency in lowering cytotoxicity in a dopant concentration-dependent manner. An early study on Fe doping demonstrated that the toxicity profiles of 1, 2, 4, 6, 8 and 10% Fe-doped ZnO NPs in RAW264.7 (mouse macrophages) and BEAS-2B (human bronchial epithelial) cells showed a progressive decline in all tested toxicological responses with increasing levels of Fe doping due to reduced dissolution rates.163 Later in 2017, a similar conclusion was made by Manshian et al. when testing the same set of Fe-doped ZnO NPs on normal murine mesenchymal stem (mMSCs) and BEAS-2B cells, as well as on murine lung squamous carcinoma (KLN205) and human cervical cancer (HeLa) cells.77 They assessed the rate of dissolution of this NP series and found that NPs with no (0%) or low (1% and 2%) Fe-doping showed a significantly higher dissolution rate in CCs compared to NCs, possibly due to the higher metabolic rates and acidic environments associated with CCs. Using high-content imaging, they assessed cellular parameters such as viability, membrane damage, mitochondrial ROS generation and health, among others, and found a clear negative correlation between the degree of Fe-doping and the level of toxicity in a concentration-dependent manner. They further confirmed the higher sensitivity of CCs towards the 2%-doped ZnO NPs in co-culture experiments, during which the levels of oxidative stress were high enough to cause severe CC death, but not enough to affect the viability of NCs with lower metabolic rates. In contrast, pure ZnO NPs exhibited significant toxicity in both cancer and NCs, while 10%-doped NPs showed almost no effect in either of the cell types.
Other metals were also tested as potential dopants of ZnO NPs. For instance, in a study by Shakir et al. (2016), pure and different lanthanum-doped ZnO NPs (1%, 3% and 5% La/ZnO) were synthesized and their cytotoxicity was examined using various concentrations (6–500 μg mL−1) in different CC lines, including MDA-MB-231, KCL22 (chronic myeloid leukemia) and HeLa cells.164 Unlike Fe-doped NPs, La-doped ZnO NPs were shown to be more potent at inducing cytotoxicity than pure ZnO NPs under the same experimental conditions. These findings suggest that in cases where specific cell types are resistant to ZnO NPs, doping them with La may enhance the NP dissolution kinetics and potentially serve as a means of enhancing toxicity. Recently, another study was conducted evaluating the cytotoxic effects of ZnO NPs doped with various lanthanides, including samarium (Sm), europium (Eu) and gadolinium (Gd).165 They found that, in the Ehrlich ascites carcinoma (EAC) cell line, Sm3+ : ZnO displayed the most significant antitumor activity with 95% cell death, while both Eu3+ and Gd3+ caused only 10% and 5% tumor cell death, respectively. Pure ZnO NPs also showed weak antitumor activity, possibly due to the micro-sized particles utilized throughout the study. These findings are in line with the previously reported selective anticancer effects of nanocomplexes composed of Sm3+ and green tea polyphenols on the melanoma (B16F10) cell line.166
Finally, a study investigating the effects of varying NP surface modification was conducted using nano- and micro-particles of ZnO with sizes ranging from 40 nm to 1.2 μm.167 These particles were either bare or coated with polyethylene glycol (PEG) or starch and were tested at various concentrations (1 μM to 7 mM). In human osteoblast CCs (MG-63), PEG-capped particles exerted higher toxicity with decreasing size and increasing concentration. In contrast, starch-capped ZnO exerted the lowest toxicity compared with the other particles, highlighting the importance of surface capping on ZnO toxicity towards CCs.
4.2.2. Metal-doped or coated CuO NPs
As with ZnO NPs, Fe-doping has been heavily investigated on CuO NPs with the aim of controlling the rapid release of Cu2+ from the particle surface. In a study by our group that was previously discussed in Section 3.1 wherein a model of the dissolution kinetics of Fe-doped CuO was developed based on solid-state diffusion, a homologous series of engineered CuO NPs was introduced with different levels of Fe-doping (1–10%) and tested in normal (BEAS-2B and murine MSCs) and cancerous cells (HeLa and KLN205).79 In line with previous findings, there was a significant inverse correlation between Fe-doping and cell response in all tested cell types, including membrane damage, ROS production and cell death. The most significant anticancer activity was reported for the 6% Fe-doped CuO NPs at 12.5 μg mL−1, which left NCs statistically unaffected. As previously mentioned, the dissolution profiles of these doped NPs were characterized by a two-step process, an initial fast burst-like release of Cu followed by a slow long-term release, with the total Cu2+ release from the 10% Fe-doped CuO NPs being 8%, in contrast with the ∼65% observed in pure CuO NPs over a 10 minute time span. In a separate recent study comparing pure and 10% Fe-doped CuO NPs, results indicated similar intracellular Cu2+ accumulation and ROS generation for both NP types, although the doped NPs exhibited less redox activity and Cu2+ release as well as less severe loss in viability in C6 glioma cells.168 Interestingly, the toxicity induced by both types of NPs was completely averted upon the co-administration of Cu chelators as well as the neutralization of lysosomal pH by bafilomycin A1.
Another type of metal-doping of CuO NPs was performed using Zn with the aim of inducing selective anticancer activity in glioma cells both in vitro and in vivo (Fig. 5).169 Treatment with nZn-doped CuO NPs has shown to not only inhibit cell proliferation in a DD manner, but also trigger ROS generation and the intrinsic apoptotic pathway. Specifically, it was found that nZn–CuO NPs disturb cell growth by downregulating the gene expression of bcl-2 and inhibiting AKT and ERK1/2. In a similar study, the anticancer activity of these particles was tested on human pancreatic CCs (AsPC-1 and MIA Paca-2).170 Here, the authors highlight that the AMPK/mTOR pathway plays an important role in the inhibition of tumor growth induced by Zn–CuO NPs, as they increased both the protein levels and gene expression of p-AMPK, p-ULK1, Beclin-1 and the ratio of LC3-II/LC3-I, while NP exposure downregulated the phosphorylation of mTOR. The specificity of Zn–CuO NPs towards CCs was further tested in multiple cell lines.171 In a DD manner, Zn–CuO NPs exerted anti-proliferative effects on CCs, including HepG2, Bel7402 (human hepatoma), A549, Panc28 (human pancreatic), HT1080 (human cervical), and Hela. However, in NC lines such as HUVEC and L02 (human hepatocyte) cells, these anti-proliferative effects were significantly weaker compared to the impact on CC lines. It was further shown that Zn–CuO NPs affect HepG2 and Panc28 CCs through ROS generation, apoptosis and the induction of G2/M cell cycle arrest. Molecular data revealed an increase in the expression of p-IKKα/β and nucleus p-NF-κB p65 and a decrease of IKKα, IKKβ, IκBα and nucleus NF-κB p65 expression in both HepG2 and Panc28 CC lines. In a parallel study that also revealed the importance of the NF-κB pathway, further treatment with NAC restored all of the above effects on protein expression.172 Therefore, in addition to the activation of the AMPK/mTOR pathway, Zn–CuO NPs trigger the ROS-mediated NF-κB pathway during the selective inhibition of CC growth.
Fig. 5. Proposed mechanism of cellular responses induced by GSH-Ag NCs. GSH-Ag NCs were taken up by the cell via an endocytotic pathway (1) and send to the lysosome (2),where stable GSH-Agþ-R NCs were enzymatically degraded by the lysosomal protease (3) to form reactive Agþ. In contrast, GSH-Ag0-R NCs decomposition originated not only from enzymatic degradation of the clusters (3) but also caused by their instability (4) in lysosome environment. The result of this decomposition is the formation of Ag0, which then undergone oxidative dissolution (5), resulting in Ag+ ion formation with ROS as the byproducts. Ag+ ions being release in turn affects the cell's respiratory chain, leading to increase of ROS which then triggers cellular responses (6), such as activation of anti-oxidant defense and programmed cell death. Reproduced with permission from Setyawati et al.,176 © 2014, Elsevier Publishing.
Besides metal doping, CuO NPs have been synthesized with a variety of coatings with the aim of controlling NP dissolution. Studer et al. (2010) reported that coating CuO NPs with a stabilizing carbon layer not only reduced their toxicity in CCs (HeLa) but also altered the release and cellular internalization of Cu2+.103 Protein coats have also been applied to CuO NPs to tune their toxicity towards C6 glioma cells, including dimercaptosuccinic acid (DMSA) and bovine serum albumin (BSA),173 which are able to improve the colloidal stability of CuO NPs in physiological media.174 Within these cells, DMSA protein-coated NPs (pCuO-NPs) resulted in Cu accumulation and severe toxicity in a time-, concentration- and temperature-dependent manner. Exposure of the cells to pCuO-NPs for 30 minutes caused severe loss in enzyme activity, including cellular lactate dehydrogenase (LDH) activity and MTT reduction capacity, which are markers for membrane integrity and cell viability, respectively. This effect was only detected in C6 glioma cells when the Cu content exceeded 20 nmol mg−1. Once again, the addition of Cu chelators (TTM or BCS) led to lower intracellular Cu content and protected glioma cells against toxicity induced by pCuO-NPs.
4.2.3. Coated Ag NPs
Despite the lack of studies on metal-doped Ag NPs, various efforts have been made to control Ag NP dissolution through varying surface coatings. In a study by Manshian et al. (2017), the cytotoxicity of citrate-coated Ag NPs was investigated by exposing various CC lines, such as HeLa, A549, and KLN205 to different NP concentrations for 24 h up to 5 days.175 Using high-content imaging, they showed that these NPs induced toxicity through ROS generation, mitochondrial damage and autophagy, whereas the resulting immunomodulatory effects associated with NP treatment were characterized by an increase in the activity of the NFκB pathway and levels of the pro-inflammatory cytokine IL-1α in both HeLa and A549 cells under subcytotoxic conditions. PVP-coated Ag NPs were also tested for their toxicity on L5178Y-R murine lymphoma cells, which, at the maximum tested molar concentration of 57.9 × 10−8 M, were able to induce up to 78% cytotoxicity compared to the 100% toxicity caused by metal Ag at the same concentration.97 Both formulations were also tested on normal murine thymic lymphocytes showing that PVP-coated Ag NPs induced proliferation at lower concentrations whereas toxicity was observed at higher concentrations. Metal Ag did not reveal any toxic effects for the tested conditions. This study indicated that PVP-coated Ag NPs and metal Ag induced cell death via a necrotic rather than apoptotic pathway.
In a separate study, the toxicity of Ag nanoclusters (Ag NCs) with two different core surface speciation (Ag+-rich NCs (Ag+-R NCs) and Ag0-rich NCs (Ag0-R NCs)) of the same size (<2 nm core) was investigated on human neonatal foreskin fibroblast cells (BJ).176 Ag0-R NCs induced higher cellular toxicity in a dose- and time-dependent manner, as well as higher ROS generation compared to the Ag+-R NCs. As the Ag0-R bond is weaker than Ag+-R, making it more potent to release Ag in acidic environments, this difference in toxicity may be due to the faster release of Ag from Ag0-R NCs which then oxidizes immediately to Ag+ within the lysosomes. After 24 and 48 h of exposure to Ag0-R NCs, only 47% and 15% of the viable population remained, respectively, while Ag+-R NCs did not induce cell death in BJ cells. Activation of p53 was increased by 3.3-fold by Ag0-R NCs after 24 h, while only a 2-fold increase was observed for Ag+-R NCs (Fig. 5).
Ag NPs embedded in microbial exopolysaccharides (EPS) and biogenerated by Klebsiella oxytoca DSM 29614 under aerobic (Ag NPs-EPSaer) conditions were also investigated for their cytotoxic effects on SKBR3 cells.177 Significant inhibition of cellular proliferation was observed, and the colony-forming ability, which was used to assess cell viability, showed a dose- and time-dependent reduction after Ag NPs-EPSaer treatment. The selectivity of Ag NPs-EPSaer for SKBR3 cells was confirmed by calculating the selectivity index using DOX as a positive control and the non-tumoral mammary cell line HB2. Morphological assessment revealed apoptotic-like changes such as cell shrinkage, loss of contact between adjacent cells, membrane blebbing and nuclear chromatin condensation. Additionally, a concentration-dependent elevation of intracellular ROS was observed after 24 h of exposure. They found that autophagic cell death seemed to be the main mechanism in Ag NPs-EPSaer-induced cell death within the first 24 h of treatment, as supported by the evaluation in the expression levels of multiple autophagic markers. Specifically, the upregulation of ATG5, ATG7, LC3-II and Beclin-1 and downregulation of AKT, p-AKT, p62 and HSP90 were detected. It was also shown that DNA was not a primary target within the first 24 h of treatment but in the long-term, treatment with Ag NPs-EPSaer could increase and/or stabilise the interaction of Ag with DNA and thereby induce cell death. Together these results suggest that the cytotoxicity of Ag NPs-EPSaer consists of a direct effect caused by cellular uptake and induction of intracellular ROS as well as an indirect effect caused by the release of Ag+ – initially in the mitochondria and later on in the nuclei to interact with DNA – and is regulated by the induction of autophagy, followed by apoptosis.
Furthermore, Swanner et al. (2019) found that triple-negative breast cancer (TNBC) cells possess specific vulnerability to Ag NPs which can be exploited to avoid toxicity induction in normal tissue.178 They found that internalization of the NPs is essential to exert any TNBC-selective cytotoxic effects and that selective inhibition of TNBC cell growth takes place regardless of NP size, shape or coating (including PVP). However, they show that TNBC cells and their normal counterparts are comparably sensitive to Ag+, highlighting the importance of the NP itself in exerting a tumor selective response. Next, they followed the uptake and intracellular distribution of Ag NPs and reported that after 1 h of incubation with cells, Ag NPs were localized within the endosomes and exhibited clear degradation in MDA-MB-231 cells while remaining intact in non-malignant breast epithelial cells (MCF-10A). Interestingly, after 6 h, MCF-10A cells still displayed intact particles even upon endosomal fusion with lysosomes. The same observations were made when comparing the effects on iMEC and SUM159 cells, a non-malignant breast epithelial and TNBC cell line respectively. It was also shown that Ag NPs impaired the cellular redox balance in MDA-MB-231 cells and induced ER stress, UPR activation, and an increased expression of CHOP, a pro-apoptotic protein in TNBC cells. This effect was not observed in the MCF-10A cell line. Finally, MDA-MB-231 and non-neoplastic S1 mammary epithelial cells (S1 cells) were grown in 3D tumor organoid cultures, and in line with the previous findings, Ag NPs induced apoptosis and DNA damage only in MDA-MB-231 cells.
4.2.4. Coated IONPs
The degradation of IONPs has been clearly linked with the nature of the coating agent. Small molecules, such as citrate, have been shown to provide little protection against degradation, while polymeric coatings (dextran, or carboxydextran) were found to be slightly more stable.61,179 When a lipid bilayer was attached directly onto the surface of the IONPs, this resulted in far more stable formulations. Also, silica and metal-based coatings (e.g. gold) have been shown to provide excellent stability against degradation.180,181
5. Cancer-specific NP toxicity in vivo: an overview of preclinical results obtained for NP degradation
Despite the multitude of in vitro studies that have been conducted over the years exploiting the Mz+ release and ROS generation of ZnO, CuO and Ag NPs to selectively target CCs, only a few have made their way to the preclinical setting. Specifically, the lack of in vivo studies on pure ZnO and CuO NPs in the literature can be attributed to the high toxicity of these particles, which tend to cause major weight loss in animals and often premature death shortly after treatment. Thus, the termination of such experiments occurs due to the underlying ethical reasons associated with NP administration.77 In this next section, we discuss the few in vivo experiments associated with pure ZnO, CuO, and Ag NP, as well as the progress that has been made over the recent years in rendering such studies safer via doping and coating of these NPs. A summary of all the discussed studies is provided in Tables 2–4.
5.1. Effects of pure ZnO, CuO, Ag and IONPs in mouse tumor models
Among the few studies that have tested the effects of ZnO NPs in vivo, El-Shorbagy (2019) administered ZnO NPs alone or in combination with 100 mg kg−1 body weight N-acetyl cysteine (NAC) – a GSH precursor that has shown to lower intracellular ROS levels182,183 – to Ehrlich solid carcinoma (ESC)-bearing mice for a period of 7 days.184 During the treatment, three different doses of ZnO NPs including 50, 300 and 500 mg kg−1 were administered through oral gavage. It was shown that treatment with NPs inhibited the tumor growth by 31.5% and 46% for the 300 and 500 mg kg−1 doses, respectively. In line with respective in vitro studies, a significant increase in p53 and bax and a decrease in bcl2 expression levels were observed along with significant reductions of glutathione-S-transferase (GST) and CAT. Their results suggest that ZnO NPs have a significant effect on the inhibition of tumor growth through induction of oxidative stress, DNA damage and activation of the p53-mediated pathway, leading to apoptosis. Additionally, pre-treatment with NAC was shown to protect normal tissue including the liver and kidney without interfering with the antitumor activity of the ZnO NPs. In the latest study, ZnO NPs were used to treat lung cancer in mice and were found to exert anticancer activity when a minimum dose of 0.25 mg kg−1 was administered.185 However, contradicting results have been published by Hassan et al. (2017), wherein they observed an antioxidant effect of ZnO NPs, characterized by a decrease in rat serum α-fetoprotein (AFP) and α-l-fucosidase (AFU) levels and restoration of caspase-3 normal levels upon treatment of hepatocellular carcinoma (HCC)-bearing mice with ZnO NPs.147 It must be noted that AFP and AFU have been shown to be common biomarkers of HCC tumors.186
The cytotoxic effects of pure CuO NPs have been investigated in mice bearing pancreatic tumors (Fig. 6) and were shown to delay tumor growth and significantly increase the number of apoptotic TICs, which is in line with previously discussed in vitro studies.149
Fig. 6. In vivo studies of pure CuO NPs as reported by Benguigui et al.149 (a) Effect of different doses of NPs on the weight of mice with no tumors over time. (b) Changes in the biomarkers of liver damage (AST, ALT, ALP) as a result of NP treatment in mice with no tumors. (c and d) Effect of daily CuO NP treatment (total 7 days) on tumor size in mice bearing subcutaneous PANC-1 tumors (c) and on the bioluminescence signal in orthotopically-implanted PANC-1 (luciferase-tagged) tumors after 5 weeks (d). Reproduced with permission from Benguigui et al.149 © Nature Publishing Group, 2019.
With respect to Ag NPs, their effect on the progression and growth of human lung CCs (H1299) has been evaluated in immunodeficient SCID mice.152 The Ag NPs which were administered intraperitoneally were able to slow down the development of tumors and significantly reduce their size. Another study assessed the antitumor activity of pure Ag NPs in EAC-bearing mice and showed that a dose of 5 mg kg−1 could significantly inhibit the cell volume, cell count and weight of the tumors, as compared to the non-treated control group.153 A rapid increase in ascitic tumor volume and a decrease in body weight was observed in the control and treated mice, respectively. The body weight was correlated with the ascites fluid volume, which served as a direct nutritional source for tumor cells. These results are in agreement with a previous report on an Ehrlich ascites tumor model in mice, in which the antitumor activity of biologically synthetized Ag NPs was detected.155 In this study, increases in nitric oxide, MDA and ROS levels, and DNA damage were reported, and were associated with higher numbers of necrotic and apoptotic cells as shown on photomicrographs of H&E sections. Histopathological observations and cell cycle analysis also revealed an antiangiogenic effect of Ag NPs. In another similar study that employs biologically-synthesized particles, Ficus religiosa-derived Ag NPs were examined for their anticancer activity in a mouse model of Dalton's ascites lymphoma (DAL).187 As was the case in the previous study, NP-treated mice experienced loss of body weight (in line with reduced ascites fluid volume) as well as increased survival. These Ag NPs were shown to induce CC apoptosis via DNA damage and exert antioxidant (restoration of MDA, SOD and CAT levels) and antiangiogenic effects without affecting kidney and liver function.
For IONPs, no cancer-selective toxicity has been observed, but clinically approved dextran-coated IONPs have been shown to activate macrophages to a pro-inflammatory M1-type status. This then further resulted in a reduction in the growth of subcutaneous adenocarcinoma, due to a more aggressive immune microenvironment.188 This indirect effect could play an important role in macrophage-related cancer immunotherapies.
5.2. In vivo effects of doped and coated NPs
5.2.1. Metal-doped ZnO NPs
To enhance the anticancer selectivity and circumvent the major drawbacks associated with the in vivo administration of ZnO NPs, including rapid weight loss and animal death shortly after NP treatment, the promising results of doped ZnO NPs in the in vitro settings were followed by preclinical in vivo experiments using mouse models. In one of these studies, the effects of pure, 2% and 10% Fe-doped ZnO NPs were tested and compared in DBA/2 mice bearing firefly luciferase-expressing KLN205 tumors.77 When the tumor size reached a certain threshold (50 mm3), NPs were administered peritumorally, and after 24 h, the release of free Zn2+ was quantified by means of a fluorescent signal. In agreement with their in vitro results, the levels of Zn2+ had a negative correlation with the degree of Fe-doping. When testing the therapeutic efficacy of pure ZnO NPs, these particles showed signs of toxicity at levels that caused the premature deaths of several mice. However, for the 2% and 10% Fe-doped ZnO NPs there were no clear signs of toxicity on the general health of the mice, and the therapeutic effect only became significant after 10 days. For the 10%-doped treated animals, the tumor growth was delayed in comparison to the control group, while the 2% Fe-doped ZnO NPs exhibited more effective results, with near-steady tumor sizes lasting at least 5 weeks. However, under the employed conditions, no complete destruction of the tumor could be obtained, although the peritumoral administration of the Fe-doped ZnO NPs resulted in a clear decrease in the number of metastases. Therefore, the preclinical evaluation of the Fe-doped ZnO NPs indicated a localized increase in free Zn2+ within the CCs that not only obstructed tumor growth, but also reduced the formation of metastases.
Another recent work in 2020 was carried out in which Sm-doped ZnO NPs were tested in Swiss albino female mice after the LD50 was determined.165 The selected dose was 45 mg kg−1 (20% of LD50) given that it exhibited no mortality and was administered by intramuscular injection to the Ehrlich solid tumor-bearing mice. While the average tumor size in the control group increased over time, the treated mice experienced reduced tumor size and a downregulation in CXCR4 and cytochrome P450 expression, which are markers of different aspects of cancer.189,190
5.2.2. Metal-doped CuO NPs
Doped CuO NPs have also been investigated in vivo, and a recent study by our team demonstrates the effects of Fe-doping on CuO NPs in syngeneic subcutaneous mouse models (KLN205 and CT26).79 The percentage of Fe-doping was first optimized in several in vitro studies previously mentioned in Section 4.2.2, which consequently uncovered the 6% Fe-doped CuO NPs as the optimal formulation. While the effects of pure and 6% Fe-doped CuO NPs on tumor growth were comparable, the un-doped particles negatively affected the body weight, resulting in the premature death of some mice. Next, we tested the combination therapy of 6% Fe-doped CuO NPs and epacadostat (EPAC), a strong inhibitor of IDO1 immunosuppression, which showed increased therapeutic efficacy and complete tumor remission, compared to animals receiving a combined therapy of DOX and EPAC. Moreover, tumor cell rechallenge (via repeated vaccinations after complete remission) prevented tumor growth, and relapse was absent for up to 6 months. Additionally, no metastasis was observed throughout the experiment. While NPs alone resulted in the inhibition of cancer growth, a total remission was only achieved when combined with an immunosuppression inhibitor (Fig. 7). NP-induced sensitization of the immune system seemed to be regulated by NLRP3 inflammasome activation, combined with the immunogenic character of the CC-death mechanism. Evidence of a local anti-tumor immune response was shown by an increased influx of immune cells and activation of cytotoxic (CD8+) T cells and natural killer cells in CT26 tumors of BALB/c mice after combined treatment with 6% Fe-doped CuO NPs and EPAC. Together these results demonstrate that the gradual long-term release of Cu2+ due to NP-mediated degradation, combined with elevated ROS generation, exerts a vaccination-like effect for anti-cancer immune activation.
Fig. 7. Synergistic therapeutic effect of treatment combinations with 6% Fe-doped CuO NPs and EPAC in mice bearing firefly-expressing KLN205 (a–d) or CT26 (e–h) tumors, as assessed by Naatz et al.79 (a) Luminescence images of tumors after treatment over time. (b and d) Bioluminescence imaging (BLI) signals comparing combination treatments of EPAC with either NPs or DOX in normal (b) and DOX-resistant cells (d). (c) Survival curves of mice exposed to different treatments. (e–h) Fluorescence images of mice treated with a fluorescent pan-caspase probe (e) and neutrophil-specific peptide (g) and their respective signal quantifications. The DOX and NP-treated groups also received EPAC. This image has been reproduced with permission from Naatz et al.,79 © Wiley-VCH, 2020.
At the same time, the potential of Zn-doped CuO nanocomposites to selectively target CCs has been investigated in multiple studies in vivo. In a glioblastoma mouse model established by U87 cell transplantation, significant antitumor effects of Zn–CuO NPs have been demonstrated.169 Specifically, increased procaspase-9 and procaspase-3 levels and a decreased BCL2/BAX ratio indicated that NP-mediated cell death occurs through the mitochondrial apoptotic pathway. Histological studies confirmed a DD decrease in Ki-67, a marker for cell proliferation, as well as no toxicity in the main organs. Furthermore, this study demonstrated that temozolomide (TMZ)-resistance of cells can be reversed upon Zn–CuO NP exposure via inhibition of AKT/ERK1/2 activation. Similar observations on tumor growth inhibition have been made in mice bearing pancreatic cancer tumors (Panc28 (ref. 172) and AsPC-1 (ref. 170)) upon treatment with Zn–CuO NPs.
5.2.3. Coated Ag NPs
In an early study by Liu et al. (2012), cell-penetrating peptides (CPP-TAT) were functionalized on the surfaces of Ag NPs with the aim of enhancing the cellular uptake of the NPs.191 In contrast to the typical method of NP internalization via endocytosis, CPPs are able to directly transport NPs across cellular membranes and immediately gain cytosolic rather than endosomal access. This strategy was used for the treatment of multidrug-resistant (MDR) cancer and was compared with non-functionalized 8 nm Ag NPs. These TAT-Ag NPs did not only exhibit strong anticancer activity in vitro, but also significantly inhibited tumor growth in mice with malignant melanoma at a lower dose (1 nmol kg−1) than the effective dose of DOX (4.3 μmol kg−1). Although both groups of mice showed similar results with respect to tumor growth, DOX-treated mice were associated with significant weight loss, whereas mice treated with Ag NPs or AgNP-TAT gained weight throughout the therapy. PVP-coated Ag NPs were also evaluated for their antitumor activity in a L5178Y-R murine lymphoma cell mouse model and compared with metal Ag and vincristine, a common chemotherapeutic.97 Results showed that treatment with PVP-coated Ag NPs or metal Ag, at a dose of 20 mg kg−1 and given at the time of tumor inoculation via intramuscular injection, significantly increased the survival response compared to the following groups: (a) same treatments administered 7 days after tumor injection, (b) treatment with vincristine, and (c) the untreated L5178Y-R tumor-bearing mice. Survival rates at day 35 were reported as 60%, 70%, 25%, 55%, 50% and 20% for the various groups respectively. Furthermore, PVP-coated Ag NPs were tested on a TNBC mouse xenograft model and were shown to reduce the tumor growth rate compared the control group.178 All the NP-treated animals survived beyond the experimental period (100 days), in contrast to the one third survival rate experienced by the control group.
To investigate the contribution of the immune system to the effects of citrate-coated Ag NPs on the treatment of cancer, Manshian et al. (2017) administered Ag NPs to immune-deficient as well as immune-competent mice bearing KLN 205 CCs.175 In a first experiment, the pro-inflammatory effects of the Ag NPs were tested. The relative fluorescence intensity – originating from an inflammation-activatable probe given to the animals 5 days post saline or NP administration – exhibited a higher signal in the NP-treated immune-competent mice, indicating tumor inflammation and thus activation of the immune system. In a parallel experiment using the same animal groups, the therapeutic efficacy of the NPs was evaluated, during which a clear reduction in tumor size was shown upon NP treatment. Ten days post treatment, tumors regrew due to the dilution of cellular NP levels that results from continuous cell division. At the same time, for the treated immune-deficient mice, the tumor growth rate was nearly identical to that observed in the untreated group, highlighting the importance of the complementary role of the immune system during Ag NP cancer treatment.
6. Hypothetical mechanism summary
While all the NPs described above have redox potential and can induce ROS while undergoing degradation in the endo- or lysosomal environment, the true mechanism behind cancer-selective toxicity remains somewhat unclear, and the lack of effect for IONPs compared to other NPs also remains unanswered. From our perspective, and based on the literature review, we suggest a hypothetical mechanism for both activities.
First, the ability of CuO, ZnO or Ag NPs to induce cancer cell selective toxicity may be due to the fact that many cancer cells have a high metabolic rate and intrinsically have higher levels of (mitochondrial) ROS. Any additional ROS that is generated, such as that generated by the NPs, can therefore result in exaggerated toxic effects compared to non-cancerous counterparts. For every cell, cellular defense mechanisms against ROS will protect the cell against certain levels of ROS generation.192 However, when this threshold is exceeded, oxidative stress occurs, which can be lethal to the cells. It is therefore important to finely tune the kinetics of NP degradation and associated ROS generation to be able to induce sufficient ROS to exceed toxic limits in cancerous cells, while not inducing too many that they would also affect non-cancerous cells. Furthermore, the effects observed may not be generalized to every type of cancerous or normal cell. If the cancer cells have a high level of oxidative defense mechanisms, then they will not be easily targeted by this form of therapy. Cancer cells that contain mutations that limit their oxidative stress defenses would however be excellently suited to this type of therapy.
Secondly, the ability of some NPs to induce cancer-selective toxicity while others cannot (e.g. IONP), can simply be due to the chemical nature of the metal ion and how these are normally processed under typical physiological conditions. For Fe, any Fe to be taken up by cells will be bound to transferrin, and cellular uptake involves the transferrin molecule binding to the cell surface-located transferrin receptor. This triggers an internalization pathway, where the transferrin receptor/transferrin complex is shuttled inside early endosomes and at lower pH, Fe is released. The transferrin receptor can then be recycled back to the cell membrane, while Fe can be transferred to late endosomes or lysosomes. It can then bind to small molecules, such as citrate, which can shuttle it into the cytoplasm, where it can become part of the labile Fe pool.193 When cellular Fe levels increase, they can be stored long-term in Fe storage proteins (ferritin), the level of which depends on the amount of cytoplasmic Fe. For IONPs, this would essentially be a similar story, where internalization of IONPs results in endo- or lysosomal sequestration of the NPs followed by their degradation and transport of free Fe ions by means of citrate to become part of the labile Fe pool.160 Excessive amounts of Fe can be relatively well tolerated by increasing the level of ferritin storage complexes. Any toxicity would be linked to excessive amounts of Fe in the endo- or lysosomes, linked to high levels of Fenton reactions that exceed local toxicity thresholds.
For Ag, Zn or Cu, the physiological handling of these ions is far different. For Ag, which is extremely rare in our body, there are no specific transport pathways or predetermined routes of cellular processing. Ag ions will mainly be linked to calcium channels, and monovalent metal transporters.194 For Zn ions, a highly specific pathway is present. In short, for eukaryotic cells, Zn ions are transported from the extracellular space into the cell cytoplasm by the so-called Zrt-, Irt-like protein (ZIP, SLC39) while the cation diffusion facilitator (CDF, SLC30) work the opposite way and transport Zn from the cytoplasm to the extracellular space. Once inside the cytoplasm, Zn can be transported to the Golgi apparatus, or mitochondria, where it can be used in Zn-dependent proteins or matrices.195 For Cu, the process is similar. Here, Cu enters the cells mainly via the Cu transporter 1 (CTR1), and is immediately linked to its chaperones, such as Cu chaperone for SOD (CCS) or an enzyme involved in the synthesis of cytochrome c oxidase (Sco1).196 Cu can be then stored at low levels in MTs. For Ag, Zn or Cu, it is therefore important to note that transport of these ions from endo- and lysosomes towards the cytoplasm is very limited as it does not occur naturally. For metal-containing NPs, this thus poses an entirely different scenario, where the cell is not naturally equipped to deal with excess level of the Mz+ in its endosomal compartments. ZnO, CuO or Ag NP, all of which typically dissolve rather quickly, result in free Mz+ already present in cell medium due to premature degradation, and cellular uptake of excess Mz+ that can lead to cell death.197 Alternatively, leakage of Mz+ from endosomes can also lead to further oxidative stress and cytotoxicity, while the intraendosomal presence of these Mz+ in itself can generate additional ROS.
7. Conclusion and future outlook
Here, we have summarized recent advances and key mechanisms behind the degradation of degradable metal and MO NPs. We highlight the various types of doping strategies and surface coatings that have been used to control NP dissolution and achieve selective tumor targeting, as well as certain computational models that have been developed to predict the impact of various physicochemical alterations on cytotoxicity. Besides the composition of the surface modifications, the toxicity profiles of these NPs have been optimized by changing the size, aspect ratio, and concentration of dopants and surface coatings. We then discussed several recent in vitro and in vivo advances that utilized these variously modified NPs to highlight their great potential in selective cancer therapy.
Studies assessing the toxicity of pure ZnO, CuO and Ag NPs on a wide variety of cell lines showed a dose-dependent toxicity, as well as a time-dependent mechanism. In addition, cellular studies on Fe- and lanthanide-doped ZnO NPs showed the potential fine-tuning capacities of the doping approach to control their cytotoxicity. Similar results were found for carbon-coated, protein-coated and Fe-doped CuO NPs, and for citrate- and EPS-coated Ag NPs. At the same time, pure ZnO, CuO and Ag NPs have not been extensively investigated in vivo, due to their high cytotoxicity levels that cause premature death in animals shortly after treatment. Nevertheless, in vivo research on their coated and doped counterparts have gained tremendous interest over the recent years, although the NPs remain in the early phases of development. Specifically, doping of ZnO NPs with Fe and samarium, as well as coating Ag with citrate for instance, have provided a first glimpse of the potential use of these particles in cancer therapy. For CuO NPs, doping with Fe and Zn have exhibited promising results in vivo, especially in combination with epacadostat, which achieved complete tumor remission.79
Therefore, the latest advances on controlling NP dissolution provide a positive outlook in the direction of selective tumor targeting, indicating a promising future for the use of nanotechnology in cancer therapy. Still, several aspects require further investigation to improve the clinical translation of such NMs, including the inconsistencies in in vitro research concerning the setup, NP synthesis methods and evaluated parameters, making it hard to reproducibly come to the same results in follow-up studies. In particular, a GMP-certified synthesis method that can produce the levels of NPs required for clinical use is essential and more work is needed to set up such GMP-certified synthesis methods. Another aspect involves a further understanding of their underlying biological pathways. Several studies already suggest mechanisms and pathways involved in NP-induced cancer cell death, yet more understanding is needed to make firm conclusions. This in particular addresses the reason why some cancer cell types are more sensitive to this type of NP-mediated therapy than others, irrespective of the NP internalization levels. If any biomarkers could be found that can predict whether a particular tumor type would be sensitive to this of treatment or not would be a major breakthrough. Another point of attention would be the ability of the NPs to induce immunogenic cell death and hereby sensitize the tumors to immunotherapy. A broader spectrum of studies must be performed to understand whether this is true for all NPs or which NP-related parameters can enhance this. Additionally, also here the question of tumor-type specificity comes up, where the biological outcome of NP treatment will likely also depend on the nature of the tumor cells and its immune microenvironment. Lastly, while some promising preclinical studies have been performed, this must be repeated on a broader range of tumor types, and also include more relevant tumor models, such as patient-derived xenografts, and by combining different immunotherapy-NP combinations in view of the tumoral immune landscape.
Conflicts of interest
There are no conflicts to declare.
Supplementary Material
Biographies
Biography
Christy Maksoudian.

Christy Maksoudian obtained her BSc in Biomedical Sciences from New York University and MSc in Drug Discovery and Development from Imperial College London, during which she worked extensively on the use of cell-penetrating peptides and gold nanoparticles in cancer therapy, respectively. She is currently pursuing her PhD at KU Leuven, with her work mainly focusing on the optimization of nanoparticle delivery to solid tumors using a variety of biological methods, particularly cell-based therapy.
Biography
Neshat Saffarzadeh.

Neshat Saffarzadeh obtained her Masters' degree in Microbial Biotechnology from the University of Tehran in Iran in 2017 and is currently pursuing her PhD in Biomedical Science at KU Leuven under the supervision of Prof. Stefaan Soenen. Her research involves the use of various doped metal oxide nanoparticles for the treatment of cancer as well as bacterial and fungal infections.
Biography
Evelien Hesemans.

Evelien Hesemans obtained a Masters' degree in Pharmaceutical Sciences in 2017, with a specialization in Drug Development, followed by a Master in Management, both at KU Leuven. At the moment she is a PhD candidate in the NanoHealth and Optical Imaging lab supervised by Prof. Soenen. Her PhD project focuses on the development and validation of optical imaging tools for advanced preclinical cancer research. She strives to design an imaging platform to visualize small cell numbers including therapy-resistant cells, cells undergoing EMT and cancer stem cells.
Biography
Nora Dekoning.

Nora Dekoning graduated from the University of Antwerp with a Master's degree in Biochemical Engineering Technology in 2018. She joined the lab of Prof. Soenen at KU Leuven in 2019 as a PhD student in Biomedical Sciences in the cancer doctoral school. The main focus of her PhD is developing biomimetic nanoparticles for the optimization of innovative optical imaging techniques.
Biography
Kiana Buttiens.

Kiana Buttiens is a PhD candidate in the Doctoral School of Biomedical Sciences, within the domain of Cancer. After receiving her Masters' degree in Biochemistry and Biotechnology from KU Leuven in 2018, she joined Prof. Soenen's NanoHealth and Optical Imaging Lab. Her research focuses on the use of nanoparticles in cancer therapy, mainly exploring their effect on extracellular vesicle-mediated cellular communication.
Biography
Stefaan J. Soenen.

Stefaan J. Soenen heads the NanoHealth and Optical Imaging Group at KU Leuven, Belgium since January 2018. His main research interests lie in the use of nanomaterials for therapeutic and diagnostic purposes and on developing novel preclinical optical imaging methods. He has published over 75 manuscript with an h-index of 34 and received an ERC Starting Grant for his work on nanotherapeutics for cancer therapy.
References
- Lancet, 2003, 362, 673. [PubMed] [Google Scholar]
- Baetke S. C. Lammers T. Kiessling F. Br. J. Radiol. 2015;88:20150207. doi: 10.1259/bjr.20150207. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wagner V. Dullaart A. Bock A.-K. Zweck A. Nat. Biotechnol. 2006;24:1211–1217. doi: 10.1038/nbt1006-1211. [DOI] [PubMed] [Google Scholar]
- Nel A. Xia T. Mädler L. Li N. Science. 2006;311:622–627. doi: 10.1126/science.1114397. [DOI] [PubMed] [Google Scholar]
- Euliss L. E. DuPont J. A. Gratton S. DeSimone J. Chem. Soc. Rev. 2006;35:1095–1104. doi: 10.1039/B600913C. [DOI] [PubMed] [Google Scholar]
- Chauhan V. P. Jain R. K. Nat. Mater. 2013;12:958. doi: 10.1038/nmat3792. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Anselmo A. C. Mitragotri S. Bioeng. Transl. Med. 2019;4:e10143. doi: 10.1002/btm2.10143. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Buzea C. Pacheco I. I. Robbie K. Biointerphases. 2007;2:MR17–MR71. doi: 10.1116/1.2815690. [DOI] [PubMed] [Google Scholar]
- Sioutas C. Delfino R. J. Singh M. Environ. Health Perspect. 2005;113:947–955. doi: 10.1289/ehp.7939. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Oberdörster G. Maynard A. Donaldson K. Castranova V. Fitzpatrick J. Ausman K. Carter J. Karn B. Kreyling W. Lai D. Part. Fibre Toxicol. 2005;2:8. doi: 10.1186/1743-8977-2-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wei Y. Quan L. Zhou C. Zhan Q. Nanomedicine. 2018;13:1495–1512. doi: 10.2217/nnm-2018-0040. [DOI] [PubMed] [Google Scholar]
- Golombek S. K. May J.-N. Theek B. Appold L. Drude N. Kiessling F. Lammers T. Adv. Drug Delivery Rev. 2018;130:17–38. doi: 10.1016/j.addr.2018.07.007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Maeda H. Bharate G. Daruwalla J. Eur. J. Pharm. Biopharm. 2009;71:409–419. doi: 10.1016/j.ejpb.2008.11.010. [DOI] [PubMed] [Google Scholar]
- Karlsson H. L. Gustafsson J. Cronholm P. Möller L. Toxicol. Lett. 2009;188:112–118. doi: 10.1016/j.toxlet.2009.03.014. [DOI] [PubMed] [Google Scholar]
- Maksoudian C. Soenen S. J. Susumu K. Oh E. Medintz I. L. Manshian B. B. Bioconjugate Chem. 2020;31:1077–1087. doi: 10.1021/acs.bioconjchem.0c00078. [DOI] [PubMed] [Google Scholar]
- Akhtar M. J. Ahamed M. Alhadlaq H. A. Clin. Chim. Acta. 2018;487:186–196. doi: 10.1016/j.cca.2018.10.004. [DOI] [PubMed] [Google Scholar]
- Fraga S. Brandão A. Soares M. E. Morais T. Duarte J. A. Pereira L. Soares L. Neves C. Pereira E. de Lourdes Bastos M. Nanomedicine. 2014;10:1757–1766. doi: 10.1016/j.nano.2014.06.005. [DOI] [PubMed] [Google Scholar]
- Wang J.-Y. Chen J. Yang J. Wang H. Shen X. Sun Y.-M. Guo M. Zhang X.-D. Int. J. Nanomed. 2016;11:3475. doi: 10.2147/IJN.S106073. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Poon W. Zhang Y.-N. Ouyang B. Kingston B. R. Wu J. L. Wilhelm S. Chan W. C. ACS Nano. 2019;13:5785–5798. doi: 10.1021/acsnano.9b01383. [DOI] [PubMed] [Google Scholar]
- Sabella S. Carney R. P. Brunetti V. Malvindi M. A. Al-Juffali N. Vecchio G. Janes S. M. Bakr O. M. Cingolani R. Stellacci F. Nanoscale. 2014;6:7052–7061. doi: 10.1039/C4NR01234H. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Midander K. Cronholm P. Karlsson H. L. Elihn K. Möller L. Leygraf C. Wallinder I. O. Small. 2009;5:389–399. doi: 10.1002/smll.200801220. [DOI] [PubMed] [Google Scholar]
- Weissleder R. a. Stark D. D. Engelstad B. L. Bacon B. R. Compton C. C. White D. L. Jacobs P. Lewis J. Am. J. Roentgenol. 1989;152:167–173. doi: 10.2214/ajr.152.1.167. [DOI] [PubMed] [Google Scholar]
- Danhier F. Ansorena E. Silva J. M. Coco R. Le Breton A. Préat V. J. Controlled Release. 2012;161:505–522. doi: 10.1016/j.jconrel.2012.01.043. [DOI] [PubMed] [Google Scholar]
- Abdal Dayem A. Hossain M. K. Lee S. B. Kim K. Saha S. K. Yang G.-M. Choi H. Y. Cho S.-G. Int. J. Mol. Sci. 2017;18:120. doi: 10.3390/ijms18010120. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Augusto O. and Miyamoto S., Principles of free radical biomedicine, 2011, vol. 1, pp. 19–42 [Google Scholar]
- Halliwell B. Plant Physiol. 2006;141:312–322. doi: 10.1104/pp.106.077073. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mueller C. F. Laude K. McNally J. S. Harrison D. G. Arterioscler., Thromb., Vasc. Biol. 2005;25:274–278. doi: 10.1161/01.ATV.0000149143.04821.eb. [DOI] [PubMed] [Google Scholar]
- Akhtar M. J. Ahamed M. Alhadlaq H. A. Alshamsan A. Biochim. Biophys. Acta, Gen. Subj. 2017;1861:802–813. doi: 10.1016/j.bbagen.2017.01.018. [DOI] [PubMed] [Google Scholar]
- Li Y. R., Free Radical Biomedicine: Principles, Clinical Correlations, and Methodologies, Bentham Science Publishers, 2012 [Google Scholar]
- Thannickal V. J. Fanburg B. L. Am. J. Physiol.: Lung Cell. Mol. Physiol. 2000;279:L1005–L1028. doi: 10.1152/ajplung.2000.279.6.L1005. [DOI] [PubMed] [Google Scholar]
- Akhtar M. J. Ahamed M. Alhadlaq H. A. Clin. Chim. Acta. 2017;469:53–62. doi: 10.1016/j.cca.2017.03.020. [DOI] [PubMed] [Google Scholar]
- Finkel T. J. Biol. Chem. 2012;287:4434–4440. doi: 10.1074/jbc.R111.271999. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Brookes P. S. Yoon Y. Robotham J. L. Anders M. Sheu S.-S. Am. J. Physiol.: Cell Physiol. 2004;287:C817–C833. doi: 10.1152/ajpcell.00139.2004. [DOI] [PubMed] [Google Scholar]
- Tapeinos C. Pandit A. Adv. Mater. 2016;28:5553–5585. doi: 10.1002/adma.201505376. [DOI] [PubMed] [Google Scholar]
- Xia T. Kovochich M. Liong M. Madler L. Gilbert B. Shi H. Yeh J. I. Zink J. I. Nel A. E. ACS Nano. 2008;2:2121–2134. doi: 10.1021/nn800511k. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ray P. D. Huang B.-W. Tsuji Y. Cell. Signalling. 2012;24:981–990. doi: 10.1016/j.cellsig.2012.01.008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Xia T. Kovochich M. Brant J. Hotze M. Sempf J. Oberley T. Sioutas C. Yeh J. I. Wiesner M. R. Nel A. E. Nano Lett. 2006;6:1794–1807. doi: 10.1021/nl061025k. [DOI] [PubMed] [Google Scholar]
- Xiao G. G. Wang M. Li N. Loo J. A. Nel A. E. J. Biol. Chem. 2003;278:50781–50790. doi: 10.1074/jbc.M306423200. [DOI] [PubMed] [Google Scholar]
- Talalay P. Dinkova-Kostova A. T. Holtzclaw W. D. Adv. Enzyme Regul. 2003;43:121–134. doi: 10.1016/S0065-2571(02)00038-9. [DOI] [PubMed] [Google Scholar]
- Kennedy I. M., Wilson D. and Barakat A. I., Research report, Health Effects Institute, 2009, pp. 3–32 [PubMed] [Google Scholar]
- Zhang Z. Berg A. Levanon H. Fessenden R. W. Meisel D. J. Am. Chem. Soc. 2003;125:7959–7963. doi: 10.1021/ja034830z. [DOI] [PubMed] [Google Scholar]
- Pokhrel S. Nel A. E. Mädler L. Acc. Chem. Res. 2013;46:632–641. doi: 10.1021/ar300032q. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lee H.-M. Shin D.-M. Song H.-M. Yuk J.-M. Lee Z.-W. Lee S.-H. Hwang S. M. Kim J.-M. Lee C.-S. Jo E.-K. Toxicol. Appl. Pharmacol. 2009;238:160–169. doi: 10.1016/j.taap.2009.05.010. [DOI] [PubMed] [Google Scholar]
- Fenton H. J. Chem. Soc., Trans. 1894;65:899–910. doi: 10.1039/CT8946500899. [DOI] [Google Scholar]
- Wei Y. Guo M. Angew. Chem., Int. Ed. 2007;46:4722–4725. doi: 10.1002/anie.200604859. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Strauch B. M. Niemand R. K. Winkelbeiner N. L. Hartwig A. Part. Fibre Toxicol. 2017;14:28. doi: 10.1186/s12989-017-0209-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Líbalová H. Costa P. M. Olsson M. Farcal L. Ortelli S. Blosi M. Topinka J. Costa A. L. Fadeel B. Chemosphere. 2018;196:482–493. doi: 10.1016/j.chemosphere.2017.12.182. [DOI] [PubMed] [Google Scholar]
- Franklin N. M. Rogers N. J. Apte S. C. Batley G. E. Gadd G. E. Casey P. S. Environ. Sci. Technol. 2007;41:8484–8490. doi: 10.1021/es071445r. [DOI] [PubMed] [Google Scholar]
- Beer C. Foldbjerg R. Hayashi Y. Sutherland D. S. Autrup H. Toxicol. Lett. 2012;208:286–292. doi: 10.1016/j.toxlet.2011.11.002. [DOI] [PubMed] [Google Scholar]
- Cronholm P. Karlsson H. L. Hedberg J. Lowe T. A. Winnberg L. Elihn K. Wallinder I. O. Möller L. Small. 2013;9:970–982. doi: 10.1002/smll.201201069. [DOI] [PubMed] [Google Scholar]
- Gliga A. R. Skoglund S. Wallinder I. O. Fadeel B. Karlsson H. L. Part. Fibre Toxicol. 2014;11:11. doi: 10.1186/1743-8977-11-11. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Heinlaan M. Ivask A. Blinova I. Dubourguier H.-C. Kahru A. Chemosphere. 2008;71:1308–1316. doi: 10.1016/j.chemosphere.2007.11.047. [DOI] [PubMed] [Google Scholar]
- Franklin N. M. Rogers N. J. Apte S. C. Batley G. E. Gadd G. E. Casey P. S. Environ. Sci. Technol. 2007;41:8484–8490. doi: 10.1021/es071445r. [DOI] [PubMed] [Google Scholar]
- Wong S. W. Leung P. T. Djurišić A. Leung K. M. Anal. Bioanal. Chem. 2010;396:609–618. doi: 10.1007/s00216-009-3249-z. [DOI] [PubMed] [Google Scholar]
- Turney T. W. Duriska M. B. Jayaratne V. Elbaz A. O'Keefe S. J. Hastings A. S. Piva T. J. Wright P. F. Feltis B. N. Chem. Res. Toxicol. 2012;25:2057–2066. doi: 10.1021/tx300241q. [DOI] [PubMed] [Google Scholar]
- Ivask A. Scheckel K. G. Kapruwan P. Stone V. Yin H. Voelcker N. H. Lombi E. Nanotoxicology. 2017;11:150–156. doi: 10.1080/17435390.2017.1282049. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Llop J. Estrela-Lopis I. Ziolo R. F. González A. Fleddermann J. Dorn M. Vallejo V. G. Simon-Vazquez R. Donath E. Mao Z. Part. Part. Syst. Charact. 2014;31:24–35. doi: 10.1002/ppsc.201300323. [DOI] [Google Scholar]
- Misra S. K. Nuseibeh S. Dybowska A. Berhanu D. Tetley T. D. Valsami-Jones E. Nanotoxicology. 2014;8:422–432. doi: 10.3109/17435390.2013.796017. [DOI] [PubMed] [Google Scholar]
- Neal A. L. Kabengi N. Grider A. Bertsch P. M. Nanotoxicology. 2012;6:371–380. doi: 10.3109/17435390.2011.579633. [DOI] [PubMed] [Google Scholar]
- Asharani P. Hande M. P. Valiyaveettil S. BMC Cell Biol. 2009;10:65. doi: 10.1186/1471-2121-10-65. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Soenen S. J. Himmelreich U. Nuytten N. Pisanic T. R. Ferrari A. De Cuyper M. Small. 2010;6:2136–2145. doi: 10.1002/smll.201000763. [DOI] [PubMed] [Google Scholar]
- Kim S. Choi J. E. Choi J. Chung K.-H. Park K. Yi J. Ryu D.-Y. Toxicol. In Vitro. 2009;23:1076–1084. doi: 10.1016/j.tiv.2009.06.001. [DOI] [PubMed] [Google Scholar]
- Luther E. M. Schmidt M. M. Diendorf J. Epple M. Dringen R. Neurochem. Res. 2012;37:1639–1648. doi: 10.1007/s11064-012-0767-4. [DOI] [PubMed] [Google Scholar]
- Peretyazhko T. S. Zhang Q. Colvin V. L. Environ. Sci. Technol. 2014;48:11954–11961. doi: 10.1021/es5023202. [DOI] [PubMed] [Google Scholar]
- Liu J. Hurt R. H. Environ. Sci. Technol. 2010;44:2169–2175. doi: 10.1021/es9035557. [DOI] [PubMed] [Google Scholar]
- Ma R. Levard C. Marinakos S. M. Cheng Y. Liu J. Michel F. M. Brown Jr G. E. Lowry G. V. Environ. Sci. Technol. 2012;46:752–759. doi: 10.1021/es201686j. [DOI] [PubMed] [Google Scholar]
- Kawata K. Osawa M. Okabe S. Environ. Sci. Technol. 2009;43:6046–6051. doi: 10.1021/es900754q. [DOI] [PubMed] [Google Scholar]
- Fabrega J. Fawcett S. R. Renshaw J. C. Lead J. R. Environ. Sci. Technol. 2009;43:7285–7290. doi: 10.1021/es803259g. [DOI] [PubMed] [Google Scholar]
- Eom H.-J. Choi J. Environ. Sci. Technol. 2010;44:8337–8342. doi: 10.1021/es1020668. [DOI] [PubMed] [Google Scholar]
- Frazzini V. Rockabrand E. Mocchegiani E. Sensi S. Biogerontology. 2006;7:307–314. doi: 10.1007/s10522-006-9045-7. [DOI] [PubMed] [Google Scholar]
- Falcón-Pérez J. M. Dell'Angelica E. C. Exp. Cell Res. 2007;313:1473–1483. doi: 10.1016/j.yexcr.2007.02.006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Okado-Matsumoto A. Fridovich I. J. Biol. Chem. 2001;276:38388–38393. doi: 10.1074/jbc.M105395200. [DOI] [PubMed] [Google Scholar]
- Gazaryan I. G. Krasinskaya I. P. Kristal B. S. Brown A. M. J. Biol. Chem. 2007;282:24373–24380. doi: 10.1074/jbc.M611376200. [DOI] [PubMed] [Google Scholar]
- Brown A. M. Kristal B. S. Effron M. S. Shestopalov A. I. Ullucci P. A. Sheu K.-F. R. Blass J. P. Cooper A. J. J. Biol. Chem. 2000;275:13441–13447. doi: 10.1074/jbc.275.18.13441. [DOI] [PubMed] [Google Scholar]
- Sensi S. L. Yin H. Z. Carriedo S. G. Rao S. S. Weiss J. H. Proc. Natl. Acad. Sci. U. S. A. 1999;96:2414–2419. doi: 10.1073/pnas.96.5.2414. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dineley K. E. Votyakova T. V. Reynolds I. J. J. Neurochem. 2003;85:563–570. doi: 10.1046/j.1471-4159.2003.01678.x. [DOI] [PubMed] [Google Scholar]
- Manshian B. B. Pokhrel S. Himmelreich U. Tämm K. Sikk L. Fernández A. Rallo R. Tamm T. Mädler L. Soenen S. J. Adv. Healthcare Mater. 2017;6:1601379. doi: 10.1002/adhm.201601379. [DOI] [PubMed] [Google Scholar]
- Denoyer D. Masaldan S. La Fontaine S. Cater M. A. Metallomics. 2015;7:1459–1476. doi: 10.1039/C5MT00149H. [DOI] [PubMed] [Google Scholar]
- Naatz H. Manshian B. B. Rios Luci C. Tsikourkitoudi V. Deligiannakis Y. Birkenstock J. Pokhrel S. Madler L. Soenen S. J. Angew. Chem., Int. Ed. 2020;59:1828–1836. doi: 10.1002/anie.201912312. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wang L. Zhang T. Li P. Huang W. Tang J. Wang P. Liu J. Yuan Q. Bai R. Li B. ACS Nano. 2015;9:6532–6547. doi: 10.1021/acsnano.5b02483. [DOI] [PubMed] [Google Scholar]
- Toduka Y. Toyooka T. Ibuki Y. Environ. Sci. Technol. 2012;46:7629–7636. doi: 10.1021/es300433x. [DOI] [PubMed] [Google Scholar]
- Xie H. Mason M. M. Wise J. P. Rev. Environ. Health. 2011;26:251–268. doi: 10.1515/reveh.2011.033. [DOI] [PubMed] [Google Scholar]
- Farnebo M. Bykov V. J. Wiman K. G. Biochem. Biophys. Res. Commun. 2010;396:85–89. doi: 10.1016/j.bbrc.2010.02.152. [DOI] [PubMed] [Google Scholar]
- Antonsson B. Cell Tissue Res. 2001;306:347–361. doi: 10.1007/s00441-001-0472-0. [DOI] [PubMed] [Google Scholar]
- Brown J. M. Wouters B. G. Cancer Res. 1999;59:1391–1399. [PubMed] [Google Scholar]
- Jiang D. Sullivan P. G. Sensi S. L. Steward O. Weiss J. H. J. Biol. Chem. 2001;276:47524–47529. doi: 10.1074/jbc.M108834200. [DOI] [PubMed] [Google Scholar]
- Compton M. M. Cancer Metastasis Rev. 1992;11:105–119. doi: 10.1007/BF00048058. [DOI] [PubMed] [Google Scholar]
- Sánchez-Pérez Y. Chirino Y. I. Osornio-Vargas Á. R. Morales-Bárcenas R. Gutiérrez-Ruíz C. Vázquez-López I. García-Cuellar C. M. Cancer Lett. 2009;278:192–200. doi: 10.1016/j.canlet.2009.01.010. [DOI] [PubMed] [Google Scholar]
- Akhtar M. J. Ahamed M. Kumar S. Khan M. M. Ahmad J. Alrokayan S. A. Int. J. Nanomed. 2012;7:845. doi: 10.2147/IJN.S29129. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ali K. Saquib Q. Ahmed B. Siddiqui M. A. Ahmad J. Al-Shaeri M. Al-Khedhairy A. A. Musarrat J. Process Biochem. 2020;91:387–397. doi: 10.1016/j.procbio.2020.01.008. [DOI] [Google Scholar]
- Zielinska E. Zauszkiewicz-Pawlak A. Wojcik M. Inkielewicz-Stepniak I. Oncotarget. 2018;9:4675. doi: 10.18632/oncotarget.22563. [DOI] [PMC free article] [PubMed] [Google Scholar]
- George S. Pokhrel S. Xia T. Gilbert B. Ji Z. Schowalter M. Rosenauer A. Damoiseaux R. Bradley K. A. Mädler L. ACS Nano. 2010;4:15–29. doi: 10.1021/nn901503q. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mizushima N. Levine B. Cuervo A. M. Klionsky D. J. Nature. 2008;451:1069–1075. doi: 10.1038/nature06639. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Akhtar M. J. Alhadlaq H. A. Kumar S. Alrokayan S. A. Ahamed M. Arch. Toxicol. 2015;89:1895–1907. doi: 10.1007/s00204-015-1570-1. [DOI] [PubMed] [Google Scholar]
- Laha D. Pramanik A. Maity J. Mukherjee A. Pramanik P. Laskar A. Karmakar P. Biochim. Biophys. Acta, Gen. Subj. 2014;1840:1–9. doi: 10.1016/j.bbagen.2013.08.011. [DOI] [PubMed] [Google Scholar]
- Vandenabeele P. Berghe T. V. Festjens N. Science's STKE. 2006;2006:pe44. doi: 10.1126/stke.3582006pe44. [DOI] [PubMed] [Google Scholar]
- Lara-González J. H. Gomez-Flores R. Tamez-Guerra P. Monreal-Cuevas E. Tamez-Guerra R. Rodríguez-Padilla C. J. Adv. Med. Med. Res. 2013:1308–1316. doi: 10.9734/BJMMR/2013/3108. [DOI] [Google Scholar]
- Vandenabeele P. Galluzzi L. Berghe T. V. Kroemer G. Nat. Rev. Mol. Cell Biol. 2010;11:700–714. doi: 10.1038/nrm2970. [DOI] [PubMed] [Google Scholar]
- Saleh N. B. Milliron D. J. Aich N. Katz L. E. Liljestrand H. M. Kirisits M. J. Sci. Total Environ. 2016;568:926–932. doi: 10.1016/j.scitotenv.2016.06.145. [DOI] [PubMed] [Google Scholar]
- Burello E. Worth A. P. Nanotoxicology. 2011;5:228–235. doi: 10.3109/17435390.2010.502980. [DOI] [PubMed] [Google Scholar]
- Odzak N. Kistler D. Behra R. Sigg L. Environ. Pollut. 2014;191:132–138. doi: 10.1016/j.envpol.2014.04.010. [DOI] [PubMed] [Google Scholar]
- Avramescu M.-L. Rasmussen P. Chénier M. Gardner H. Environ. Sci. Pollut. Res. 2017;24:1553–1564. doi: 10.1007/s11356-016-7932-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Studer A. M. Limbach L. K. Van Duc L. Krumeich F. Athanassiou E. K. Gerber L. C. Moch H. Stark W. J. Toxicol. Lett. 2010;197:169–174. doi: 10.1016/j.toxlet.2010.05.012. [DOI] [PubMed] [Google Scholar]
- Zhang W. Yao Y. Sullivan N. Chen Y. Environ. Sci. Technol. 2011;45:4422–4428. doi: 10.1021/es104205a. [DOI] [PubMed] [Google Scholar]
- Elzey S. Grassian V. H. J. Nanopart. Res. 2010;12:1945–1958. doi: 10.1007/s11051-009-9783-y. [DOI] [Google Scholar]
- Cooper G. M. Ganem D. Nat. Med. 1997;3:1042. doi: 10.1038/nm0997-1042. [DOI] [Google Scholar]
- Müller S. Dennemärker J. Reinheckel T. Biochim. Biophys. Acta, Proteins Proteomics. 2012;1824:34–43. doi: 10.1016/j.bbapap.2011.07.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sée V. Free P. Cesbron Y. Nativo P. Shaheen U. Rigden D. J. Spiller D. G. Fernig D. G. White M. R. Prior I. A. ACS Nano. 2009;3:2461–2468. doi: 10.1021/nn9006994. [DOI] [PubMed] [Google Scholar]
- Jiang X. Miclăuş T. Wang L. Foldbjerg R. Sutherland D. S. Autrup H. Chen C. Beer C. Nanotoxicology. 2015;9:181–189. doi: 10.3109/17435390.2014.907457. [DOI] [PubMed] [Google Scholar]
- Kreyling W. G. Abdelmonem A. M. Ali Z. Alves F. Geiser M. Haberl N. Hartmann R. Hirn S. De Aberasturi D. J. Kantner K. Nat. Nanotechnol. 2015;10:619. doi: 10.1038/nnano.2015.111. [DOI] [PubMed] [Google Scholar]
- Moreau J. W. Weber P. K. Martin M. C. Gilbert B. Hutcheon I. D. Banfield J. F. Science. 2007;316:1600–1603. doi: 10.1126/science.1141064. [DOI] [PubMed] [Google Scholar]
- Schveigkardt J. M. Rizzi A. C. Piro O. E. Castellano E. E. Santana R. C. d. Calvo R. Brondino C. D. Eur. J. Inorg. Chem. 2002;2002:2913–2919. doi: 10.1002/1099-0682(200211)2002:11<2913::AID-EJIC2913>3.0.CO;2-P. [DOI] [Google Scholar]
- Stephens F. Vagg R. Williams P. Acta Crystallogr., Sect. B: Struct. Crystallogr. Cryst. Chem. 1975;31:841–845. doi: 10.1107/S0567740875003901. [DOI] [Google Scholar]
- Antolini L. Inorg. Chem. 1985;24:3621–3626. doi: 10.1021/ic00216a029. [DOI] [Google Scholar]
- Calvo R. Steren C. Piro O. Rojo T. Zuniga F. Castellano E. E. Inorg. Chem. 1993;32:6016–6022. doi: 10.1021/ic00078a019. [DOI] [Google Scholar]
- Gilbert B. Fakra S. C. Xia T. Pokhrel S. Mädler L. Nel A. E. ACS Nano. 2012;6:4921–4930. doi: 10.1021/nn300425a. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chen M. von Mikecz A. Exp. Cell Res. 2005;305:51–62. doi: 10.1016/j.yexcr.2004.12.021. [DOI] [PubMed] [Google Scholar]
- Linse S. Cabaleiro-Lago C. Xue W. F. Lynch I. Lindman S. Thulin E. Radford S. E. Dawson K. A. Proc. Natl. Acad. Sci. U. S. A. 2007;104:8691–8696. doi: 10.1073/pnas.0701250104. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Vertegel A. A. Siegel R. W. Dordick J. S. Langmuir. 2004;20:6800–6807. doi: 10.1021/la0497200. [DOI] [PubMed] [Google Scholar]
- Mansy S. S. Cowan J. Acc. Chem. Res. 2004;37:719–725. doi: 10.1021/ar0301781. [DOI] [PubMed] [Google Scholar]
- Holm R. H. Acc. Chem. Res. 1977;10:427–434. doi: 10.1021/ar50120a001. [DOI] [Google Scholar]
- Flint D. H. Tuminello J. Emptage M. J. Biol. Chem. 1993;268:22369–22376. [PubMed] [Google Scholar]
- Leal S. S. Gomes C. M. Biol. Chem. 2005;386:1295–1300. doi: 10.1515/BC.2005.147. [DOI] [PubMed] [Google Scholar]
- Gardner P. R. Raineri I. Epstein L. B. White C. W. J. Biol. Chem. 1995;270:13399–13405. doi: 10.1074/jbc.270.22.13399. [DOI] [PubMed] [Google Scholar]
- Levard C. Hotze E. M. Colman B. P. Dale A. L. Truong L. Yang X. Bone A. J. Brown Jr G. E. Tanguay R. L. Di Giulio R. T. Environ. Sci. Technol. 2013;47:13440–13448. doi: 10.1021/es403527n. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Levard C. m. Mitra S. Yang T. Jew A. D. Badireddy A. R. Lowry G. V. Brown Jr G. E. Environ. Sci. Technol. 2013;47:5738–5745. doi: 10.1021/es400396f. [DOI] [PubMed] [Google Scholar]
- Feng Q. L. Wu J. Chen G. Cui F. Kim T. Kim J. J. Biomed. Mater. Res. 2000;52:662–668. doi: 10.1002/1097-4636(20001215)52:4<662::AID-JBM10>3.0.CO;2-3. [DOI] [PubMed] [Google Scholar]
- Lowry G. V. Gregory K. B. Apte S. C. Lead J. R. Environ. Sci. Technol. 2012;46:6893–6899. doi: 10.1021/es300839e. [DOI] [PubMed] [Google Scholar]
- Zook J. M. Long S. E. Cleveland D. Geronimo C. L. A. MacCuspie R. I. Anal. Bioanal. Chem. 2011;401:1993. doi: 10.1007/s00216-011-5266-y. [DOI] [PubMed] [Google Scholar]
- Puzyn T. Rasulev B. Gajewicz A. Hu X. Dasari T. P. Michalkova A. Hwang H.-M. Toropov A. Leszczynska D. Leszczynski J. Nat. Nanotechnol. 2011;6:175. doi: 10.1038/nnano.2011.10. [DOI] [PubMed] [Google Scholar]
- Kaweeteerawat C. Ivask A. Liu R. Zhang H. Chang C. H. Low-Kam C. Fischer H. Ji Z. Pokhrel S. Cohen Y. Environ. Sci. Technol. 2015;49:1105–1112. doi: 10.1021/es504259s. [DOI] [PubMed] [Google Scholar]
- Mu Q. Jiang G. Chen L. Zhou H. Fourches D. Tropsha A. Yan B. Chem. Rev. 2014;114:7740–7781. doi: 10.1021/cr400295a. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Verma A. Stellacci F. Small. 2010;6:12–21. doi: 10.1002/smll.200901158. [DOI] [PubMed] [Google Scholar]
- Simeone F. C. Costa A. L. Environ. Sci.: Nano. 2019;6:3102–3112. doi: 10.1039/C9EN00785G. [DOI] [Google Scholar]
- Ganduglia-Pirovano M. V. Hofmann A. Sauer J. Surf. Sci. Rep. 2007;62:219–270. doi: 10.1016/j.surfrep.2007.03.002. [DOI] [Google Scholar]
- Le T. C. Yin H. Chen R. Chen Y. Zhao L. Casey P. S. Chen C. Winkler D. A. Small. 2016;12:3568–3577. doi: 10.1002/smll.201600597. [DOI] [PubMed] [Google Scholar]
- Buonsanti R. Milliron D. J. Chem. Mater. 2013;25:1305–1317. doi: 10.1021/cm304104m. [DOI] [Google Scholar]
- Bilecka I. Luo L. Djerdj I. Rossell M. D. Jagodic M. Jaglicic Z. Masubuchi Y. Kikkawa S. Niederberger M. J. Phys. Chem. C. 2011;115:1484–1495. doi: 10.1021/jp108050w. [DOI] [Google Scholar]
- Naatz H. Lin S. Li R. Jiang W. Ji Z. Chang C. H. Köser J. Thöming J. Xia T. Nel A. E. ACS Nano. 2017;11:501–515. doi: 10.1021/acsnano.6b06495. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wang R. Sleight A. W. Subramanian M. J. Solid State Chem. 1996;125:224–227. doi: 10.1006/jssc.1996.0289. [DOI] [Google Scholar]
- Xiao J. Kuc A. Pokhrel S. Schowalter M. Parlapalli S. Rosenauer A. Frauenheim T. Mädler L. Pettersson L. G. Heine T. Small. 2011;7:2879–2886. doi: 10.1002/smll.201100963. [DOI] [PubMed] [Google Scholar]
- Jang S. Imlay J. A. J. Biol. Chem. 2007;282:929–937. doi: 10.1074/jbc.M607646200. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chia S. L. Leong D. T. Heliyon. 2016;2:e00177. doi: 10.1016/j.heliyon.2016.e00177. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cai X. Lee A. Ji Z. Huang C. Chang C. H. Wang X. Liao Y. P. Xia T. Li R. Part. Fibre Toxicol. 2017;14:13. doi: 10.1186/s12989-017-0193-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Osmond-McLeod M. J. Osmond R. I. Oytam Y. McCall M. J. Feltis B. Mackay-Sim A. Wood S. A. Cook A. L. Part. Fibre Toxicol. 2013;10:54. doi: 10.1186/1743-8977-10-54. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhang Y. Wang H. Jiang H. Wang X. Nanoscale. 2012;4:3530–3535. doi: 10.1039/C2NR30127J. [DOI] [PubMed] [Google Scholar]
- Hassan H. F. Mansour A. M. Abo-Youssef A. M. Elsadek B. E. Messiha B. A. Clin. Exp. Pharmacol. Physiol. 2017;44:235–243. doi: 10.1111/1440-1681.12681. [DOI] [PubMed] [Google Scholar]
- Kermanizadeh A. Jantzen K. Ward M. B. Durhuus J. A. Juel Rasmussen L. Loft S. Moller P. Nanotoxicology. 2017;11:184–200. doi: 10.1080/17435390.2017.1279359. [DOI] [PubMed] [Google Scholar]
- Benguigui M. Weitz I. S. Timaner M. Kan T. Shechter D. Perlman O. Sivan S. Raviv Z. Azhari H. Shaked Y. Sci. Rep. 2019;9:1–10. doi: 10.1038/s41598-018-37186-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Siddiqui M. A. Alhadlaq H. A. Ahmad J. Al-Khedhairy A. A. Musarrat J. Ahamed M. PLoS One. 2013;8:e69534. doi: 10.1371/journal.pone.0069534. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Shafagh M. Rahmani F. Delirezh N. Iran. J. Basic Med. Sci. 2015;18:993. [PMC free article] [PubMed] [Google Scholar]
- He Y. Du Z. Ma S. Liu Y. Li D. Huang H. Jiang S. Cheng S. Wu W. Zhang K. Int. J. Nanomed. 2016;11:1879. doi: 10.2147/IJN.S103695. [DOI] [PMC free article] [PubMed] [Google Scholar]
- El-Naggar N. E.-A. Hussein M. H. El-Sawah A. A. Sci. Rep. 2017;7:1–20. doi: 10.1038/s41598-016-0028-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nakkala J. R. Mata R. Raja K. Chandra V. K. Sadras S. R. Mater. Sci. Eng., C. 2018;91:372–381. doi: 10.1016/j.msec.2018.05.048. [DOI] [PubMed] [Google Scholar]
- El-Sonbaty S. Cancer Nanotechnol. 2013;4:73. doi: 10.1007/s12645-013-0038-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dadfar S. M. Roemhild K. Drude N. I. von Stillfried S. Knüchel R. Kiessling F. Lammers T. Adv. Drug Delivery Rev. 2019;138:302–325. doi: 10.1016/j.addr.2019.01.005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Aisen P. Enns C. Wessling-Resnick M. Int. J. Biochem. Cell Biol. 2001;33:940–959. doi: 10.1016/S1357-2725(01)00063-2. [DOI] [PubMed] [Google Scholar]
- Pisanic II T. R. Blackwell J. D. Shubayev V. I. Fiñones R. R. Jin S. Biomaterials. 2007;28:2572–2581. doi: 10.1016/j.biomaterials.2007.01.043. [DOI] [PubMed] [Google Scholar]
- Huang D.-M. Hsiao J.-K. Chen Y.-C. Chien L.-Y. Yao M. Chen Y.-K. Ko B.-S. Hsu S.-C. Tai L.-A. Cheng H.-Y. Biomaterials. 2009;30:3645–3651. doi: 10.1016/j.biomaterials.2009.03.032. [DOI] [PubMed] [Google Scholar]
- Soenen S. J. Himmelreich U. Nuytten N. De Cuyper M. Biomaterials. 2011;32:195–205. doi: 10.1016/j.biomaterials.2010.08.075. [DOI] [PubMed] [Google Scholar]
- Arami H. Khandhar A. Liggitt D. Krishnan K. M. Chem. Soc. Rev. 2015;44:8576–8607. doi: 10.1039/C5CS00541H. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Van de Walle A. Sangnier A. P. Abou-Hassan A. Curcio A. Hemadi M. Menguy N. Lalatonne Y. Luciani N. Wilhelm C. Proc. Natl. Acad. Sci. U. S. A. 2019;116:4044–4053. doi: 10.1073/pnas.1816792116. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Xia T. Zhao Y. Sager T. George S. Pokhrel S. Li N. Schoenfeld D. Meng H. Lin S. Wang X. ACS Nano. 2011;5:1223–1235. doi: 10.1021/nn1028482. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Shakir M. Faraz M. Sherwani M. A. Al-Resayes S. I. J. Lumin. 2016;176:159–167. doi: 10.1016/j.jlumin.2016.03.027. [DOI] [Google Scholar]
- Nabeel A. I. Tumor Biol. 2020;42 doi: 10.1177/1010428320909999. [DOI] [PubMed] [Google Scholar]
- Li K. Xiao G. Richardson J. J. Tardy B. L. Ejima H. Huang W. Guo J. Liao X. Shi B. Adv. Sci. 2019;6:1801688. doi: 10.1002/advs.201801688. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nair S. Sasidharan A. Rani V. D. Menon D. Nair S. Manzoor K. Raina S. J. Mater. Sci.: Mater. Med. 2009;20:235. doi: 10.1007/s10856-008-3548-5. [DOI] [PubMed] [Google Scholar]
- Joshi A. Naatz H. Faber K. Pokhrel S. Dringen R. Neurochem. Res. 2020:1–16. doi: 10.1007/s11064-020-02954-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wu N. Zhang C. Wang C. Song L. Yao W. Gedanken A. Lin X. Shi D. Nanomedicine. 2018;13:1303–1318. doi: 10.2217/nnm-2017-0359. [DOI] [PubMed] [Google Scholar]
- Li X. Xu H. Li C. Qiao G. Farooqi A. A. Gedanken A. Liu X. Lin X. Front. Pharmacol. 2019;10:319. doi: 10.3389/fphar.2019.00319. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yuan R. Xu H. Liu X. Tian Y. Li C. Chen X. Su S. Perelshtein I. Gedanken A. Lin X. ACS Appl. Mater. Interfaces. 2016;8:31806–31812. doi: 10.1021/acsami.6b09542. [DOI] [PubMed] [Google Scholar]
- Xu H. Yuan R. Liu X. Li X. Qiao G. Li C. Gedanken A. Lin X. Nanomedicine. 2019;14:131–149. doi: 10.2217/nnm-2018-0366. [DOI] [PubMed] [Google Scholar]
- Joshi A. Rastedt W. Faber K. Schultz A. G. Bulcke F. Dringen R. Neurochem. Res. 2016;41:3004–3019. doi: 10.1007/s11064-016-2020-z. [DOI] [PubMed] [Google Scholar]
- Ostermeyer A.-K. Kostigen Mumuper C. Semprini L. Radniecki T. Environ. Sci. Technol. 2013;47:14403–14410. doi: 10.1021/es4033106. [DOI] [PubMed] [Google Scholar]
- Manshian B. B. Jimenez J. Himmelreich U. Soenen S. J. Adv. Healthcare Mater. 2017;6:1601099. doi: 10.1002/adhm.201601099. [DOI] [PubMed] [Google Scholar]
- Setyawati M. I. Yuan X. Xie J. Leong D. T. Biomaterials. 2014;35:6707–6715. doi: 10.1016/j.biomaterials.2014.05.007. [DOI] [PubMed] [Google Scholar]
- Buttacavoli M. Albanese N. N. Di Cara G. Alduina R. Faleri C. Gallo M. Pizzolanti G. Gallo G. Feo S. Baldi F. Oncotarget. 2018;9:9685. doi: 10.18632/oncotarget.23859. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Swanner J. Fahrenholtz C. D. Tenvooren I. Bernish B. W. Sears J. J. Hooker A. Furdui C. M. Alli E. Li W. Donati G. L. Cook K. L. Vidi P. A. Singh R. FASEB BioAdv. 2019;1:639–660. doi: 10.1096/fba.2019-00021. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Shaterabadi Z. Nabiyouni G. Soleymani M. Mater. Sci. Eng., C. 2017;75:947–956. doi: 10.1016/j.msec.2017.02.143. [DOI] [PubMed] [Google Scholar]
- Malvindi M. A. De Matteis V. Galeone A. Brunetti V. Anyfantis G. C. Athanassiou A. Cingolani R. Pompa P. P. PLoS One. 2014;9:e85835. doi: 10.1371/journal.pone.0085835. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Javed Y. Lartigue L. Hugounenq P. Vuong Q. L. Gossuin Y. Bazzi R. Wilhelm C. Ricolleau C. Gazeau F. Alloyeau D. Small. 2014;10:3325–3337. doi: 10.1002/smll.201400281. [DOI] [PubMed] [Google Scholar]
- Spagnuolo G. D'Antò V. Cosentino C. Schmalz G. Schweikl H. Rengo S. Biomaterials. 2006;27:1803–1809. doi: 10.1016/j.biomaterials.2005.10.022. [DOI] [PubMed] [Google Scholar]
- Fukami G. Hashimoto K. Koike K. Okamura N. Shimizu E. Iyo M. Brain Res. 2004;1016:90–95. doi: 10.1016/j.brainres.2004.04.072. [DOI] [PubMed] [Google Scholar]
- El-Shorbagy H. M. Eissa S. M. Sabet S. El-Ghor A. A. Int. J. Nanomed. 2019;14:3911. doi: 10.2147/IJN.S204757. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tanino R. Amano Y. Tong X. Sun R. Tsubata Y. Harada M. Fujita Y. Isobe T. Mol. Cancer Ther. 2020;19:502–512. doi: 10.1158/1535-7163.MCT-19-0018. [DOI] [PubMed] [Google Scholar]
- Waidely E. Al-Yuobi A.-R. O. Bashammakh A. El-Shahawi M. S. Leblanc R. M. Analyst. 2016;141:36–44. doi: 10.1039/C5AN01884F. [DOI] [PubMed] [Google Scholar]
- Antony J. J. Sithika M. A. A. Joseph T. A. Suriyakalaa U. Sankarganesh A. Siva D. Kalaiselvi S. Achiraman S. Colloids Surf., B. 2013;108:185–190. doi: 10.1016/j.colsurfb.2013.02.041. [DOI] [PubMed] [Google Scholar]
- Zanganeh S. Hutter G. Spitler R. Lenkov O. Mahmoudi M. Shaw A. Pajarinen J. S. Nejadnik H. Goodman S. Moseley M. Nat. Nanotechnol. 2016;11:986–994. doi: 10.1038/nnano.2016.168. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rodriguez-Antona C. Ingelman-Sundberg M. Oncogene. 2006;25:1679–1691. doi: 10.1038/sj.onc.1209377. [DOI] [PubMed] [Google Scholar]
- Nobutani K. Shimono Y. Mizutani K. Ueda Y. Suzuki T. Kitayama M. Minami A. Momose K. Miyawaki K. Akashi K. PLoS One. 2015;10:e0130032. doi: 10.1371/journal.pone.0130032. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Liu J. Zhao Y. Guo Q. Wang Z. Wang H. Yang Y. Huang Y. Biomaterials. 2012;33:6155–6161. doi: 10.1016/j.biomaterials.2012.05.035. [DOI] [PubMed] [Google Scholar]
- Manke A. Wang L. Rojanasakul Y. BioMed Res. Int. 2013;2013:e942916. doi: 10.1155/2013/942916. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kurz T. Terman A. Gustafsson B. Brunk U. T. Histochem. Cell Biol. 2008;129:389–406. doi: 10.1007/s00418-008-0394-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Klíma P. Laňková M. Vandenbussche F. Van Der Straeten D. Petrášek J. Plant Cell Rep. 2018;37:809–818. doi: 10.1007/s00299-018-2269-6. [DOI] [PubMed] [Google Scholar]
- Eide D. J. Biochim. Biophys. Acta, Mol. Cell Res. 2006;1763:711–722. doi: 10.1016/j.bbamcr.2006.03.005. [DOI] [PubMed] [Google Scholar]
- Kaplan J. H. Maryon E. B. Biophys. J. 2016;110:7–13. doi: 10.1016/j.bpj.2015.11.025. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Liu S. Liu Y. Pan B. He Y. Li B. Zhou D. Xiao Y. Qiu H. Vijver M. G. Peijnenburg W. J. Chemosphere. 2020;245:125612. doi: 10.1016/j.chemosphere.2019.125612. [DOI] [PubMed] [Google Scholar]