Abstract
Common outdoor air pollutants present threats to fetal and neonatal health, placing neonatal-perinatal clinical specialists in an important role for harm reduction through patient counseling and advocacy. Climate change is intertwined with air pollution and influences air quality. There is increasing evidence demonstrating the unique vulnerability in the development of adverse health consequences from exposures during the preconception, prenatal, and early postnatal periods, as well as promising indications that policies aimed at addressing these toxicants have improved birth outcomes. Advocacy by neonatal-perinatal providers articulating the potential impact of pollutants on newborns and mothers is essential to promoting improvements in air quality and reducing exposures. The goal of this review is to update neonatal-perinatal clinical specialists on the key ambient air pollutants of concern, their sources and health effects, and to outline strategies for protecting patients and communities from documented adverse health consequences.
Subject terms: Epidemiology, Health policy
Introduction
Many neonatal-perinatal providers are unaware that climate change-related and air pollution exposures during infancy, pregnancy, and preconception (known critical windows of vulnerability) can lead to multiple adverse health outcomes including preterm birth, poor fetal growth, abnormal lung development, and impaired neurodevelopment [1, 2]. We therefore outline the need for neonatal-perinatal providers to understand the role of outdoor air quality, and how this can affect maternal and infant health and influence clinical practice. Our knowledge of the effects of tobacco smoke on pregnancy and infancy is instructive to understanding similar concerns with outdoor air pollution. This paper focuses on climate change, the intersection with outdoor air pollution, and mitigation of effects, which can also impact indoor air quality. We also review important policies aimed at reducing air pollution, and their influence on birth outcomes. Further, we delineate opportunities for neonatologists and perinatal care providers to address both air pollution and climate change on individual, community, and regional/national levels. The AAP Policy Statement and Technical Reports on climate change and air pollution are key reference documents that are important resources for clinicians [1–3].
Climate change
Attention on climate change has increased over the past four decades. In August 2021, the United Nations’ Intergovernmental Panel on Climate Change released a comprehensive report definitively identifying human activities as causation for the accelerated warming of the planet and subsequent extreme weather effects that are increasing in frequency, such as heat waves, wildfires, droughts and floods, hurricanes, and rising sea levels [4]. These human activities generate greenhouse gases, predominantly carbon dioxide (CO2), from the combustion of fossil fuels, and have contributed to altered levels of ozone and particulate matter (PM) [5–7]. Wildfires, which result from warmer, drier conditions, are more intense and are lasting longer, thus posing serious threats to human health, not just through immediate loss of life, but with the release of toxic emissions that have long-term effects [8]. Climate change is also contributing to the production of extreme heat which is intensified in large cities where urban heat islands are created from replacing trees and vegetation with buildings and pavement that absorb instead of reflect heat [9]. Air pollution is exacerbated by heat and climate change, and as a result, more focus needs to be paid to declining air quality and its adverse effects on human health worldwide. This is especially critical during pregnancy and preconception, vulnerable periods of time in which environmental exposures can have harmful effects on both the mother and fetus.
Key ambient air pollutants and their major sources
Air pollution is a mixture of gases and solid particles suspended in the air, and encompasses a variety of inhaled substances that have adverse effects on human health. Many of the components of outdoor air pollution are similar to those in tobacco smoke, such as carbon monoxide (CO), polycyclic aromatic hydrocarbons (PAHs), nitrogen dioxide (NO2), volatile organic compounds (VOCs), and PM [10]. As perinatal providers are already familiar with tobacco smoke and its resultant health effects on pregnancy and infancy, this established knowledge facilitates discussions on outdoor air pollution.
Management of air pollution is focused on understanding the contributors to these suspended mixtures [1, 6]. Hazardous air pollutants such as PAHs, metals, and solvents, are toxicants that are known or suspected to cause cancer or other serious health effects [1]. In the United States, the Environmental Protection Agency (EPA) regulates air quality under the Clean Air Act by implementing limits on 187 air toxicants and six outdoor criteria air pollutants, defined as PM, photochemical oxidants including ozone, CO, sulfur oxides, nitrogen oxides, and lead, through the National Emission Standards for Hazardous Air Pollutants and National Ambient Air Quality Standards, respectively [11–14]. The most common outdoor air pollutants are described in Table 1 along with their major sources and associated health effects [6, 7, 13, 15].
Table 1.
Major outdoor air pollutants, health effects, and mitigation measures for neonatal-perinatal providers.
| Pollutant | Description and Common Sources of Exposure | Mechanism of Action | Neonatal/Perinatal Effectsa [3–5, 19, 23, 24] | % of children in US exposed to levels above EPA NAAQS as of 2019b [86] | General Mitigation/Prevention Strategies for Outdoor Air Pollution |
|---|---|---|---|---|---|
| Ozone (O3) |
• Major component of urban smog • Greenhouse gas • In the presence of heat and sunlight, NO species and volatile organic compounds (VOCs) undergo a photochemical reaction to form ozone • VOCs derive from emissions from industrial facilities and motor vehicles, gasoline vapors, and chemical solvents |
• Reacts with, and damages, cell membranes in respiratory tract (nose to alveoli) and mucus membranes |
• Low birth weight • Risk of asthma • Decreased lung function |
46.2% |
Individual Action • Masking (primarily for PM) • Use of HEPA filters (primarily for PM) • Spending time indoors when AQI is unhealthy or unhealthy for sensitive groups Community Action • Plant trees and expand access to parks/green spaces • Promote community-wide shifts to non-fossil fuel based transportation and accessible mass transit • Maintain emissions standards as well as requirements for routine inspections and vehicle maintenance • Limit proximity of schools, child care facilities, and residences to traffic corridors • Anti-idling policies at schools • Electric school buses Clinical Practice • Address underlying conditions and co-morbidities • Mental health counseling related to climate change • Identify local resources • Display Climate Action posters in medical offices • Action plans for excessive heat • Educate patients about their neighborhood’s Air Quality Index (AQI) and toxicant exposures (www.AirNow.gov) • NY State Prescriptions for Prevention (www.nyscheck.org/rx) • Pediatric Environmental Health Specialty Units (www.pehsu.net) • Moms Clean Air Force (www.momscleanairforce.org) • The Medical Society Consortium on Climate and Health (www.medsocietiesforclimatehealth.org) • Climate for Health (www.climateforhealth.org) Advocacy • Encourage hospitals to promote sustainability and energy efficiency: Ex. National Academy of Medicine’s Action Collaborative on Decarbonizing the US Health Sector, joined by AAP in 2022 as a Network Organization (www.nam.edu/programs/climate-change-and-human-health/action-collaborative-on-decarbonizing-the-u-s-health-sector) and Health Care Climate Challenge (www.greenhospitals.net/about-challenge) • Encourage legislators to enact policy, such as stricter federal car emissions standards or expanding the Clean Air Act to include wildfire smoke • Emphasize the importance of considering the perinatal and neonatal life stages when implementing programs and policies related to ambient air contaminants |
| Particulate matter (PM) |
• Airborne mixture of solid particles and liquid droplets • Deposition pattern determined by particle size, with diameter >10 µm unable to pass nasal passages PM10: Diameter <10 µm • Thoracic region PM2.5 (fine): Diameter <2.5 µm • Small conducting airways and alveoli Ultrafine: Diameter <0.1 µm • Olfactory region of nose and can travel along olfactory nerve to brain • Can pass into epithelial tissues and enter systemic circulation • Fuel combustion from motor vehicles, power plants, and industrial operations • Combustion of organic material in fireplaces and wood stoves; wildfires • Dust from mechanical breakdown of solid matter (ex. rocks, soil) |
• Oxidative stress/ production of reactive oxygen species • Pro-inflammatory • Alters DNA methylation processes |
• Preterm birth • Low birth weight • Increased risk of autism spectrum disorder • Risk of asthma |
PM2.5 (24 hour): 20% PM2.5 (annual): 4.4% PM10 (24 hour): 10.2% |
|
| Nitrogen dioxide (NO2) |
• Nitrogen and oxygen combine in atmosphere during high temperature combustion • Often used as proxy for traffic emissions • High temperature fuel combustion, vehicles and coal • Road traffic, electricity generation, power plants • Enclosed spaces such as homes with poorly-vented gas appliances (ex. stoves, heaters, boilers) |
• Interacts with other chemicals to form secondary pollutants such as ozone and particulate matter |
• Impaired host defenses • Increased airway responsiveness and inflammation • Preterm birth • Low birth weight • Risk of asthma |
0.7% | |
| Sulfur dioxide (SO2) |
• Formed in atmosphere from oxidative reaction of sulfuric acid with ammonia in presence of moisture • Burning of coal, diesel, and sulfur-containing oil • Power plants, smelters, ships, pulp and paper mills that burn coal |
• Respiratory irritant |
• Preterm birth • Low birth weight |
2.3% |
Summary of major outdoor air pollutants, including common sources of exposure, mechanism of action, neonatal/perinatal health effects, percent of children in US exposed to levels above National Ambient Air Quality Standards (NAAQS), and clinician-specific mitigation/prevention strategies. In 2019, the percentage of children ages 0-17 years living in counties in which the level of air quality standards was exceeded by any standard was overall 50.5%, which has decreased from 76% in 1999. In addition to the criteria pollutants listed in the table, carbon monoxide (CO) and lead have had significant improvements over time, with levels at 0% and 0.3%, respectively, hallmarking the success of the Clean Air Act. Ultrafine PM is italicized as a pollutant as it is not specifically regulated by NAAQS. Of note, wildfire smoke contributes to air pollution but is not directly addressed in the Clean Air Act [78, 79].
aThere are many health effects that are beyond the scope of this paper, as those predominantly relate to adult health. Though this paper focuses on perinatal/neonatal health effects, this additional information can be found using the EPA’s Integrated Science Assessment (www.epa.gov/isa).
bNAAQS are established by the EPA under the Clean Air Act to regulate atmospheric concentrations of six criteria pollutants in outdoor air. Available at www.epa.gov/criteria-air-pollutants/naaqs-table.
Concentrations of air pollutants are dependent on wind, temperature, altitude, time of day, and seasonal weather changes. In addition, indoor air pollution can have a significant impact on health outcomes. Not only can outdoor air pollution permeate indoors depending on ventilation rate and insulation, but people who may conceive, pregnant persons, and neonates can have airborne exposure from secondhand smoke, pesticides, biological materials, and other household chemicals derived indoors [5, 6]. Exposure to air pollution also does not exclusively occur through inhalation, as pollutants can contaminate drinking water and soil, ultimately impacting the food supply [16, 17].
The interplay of climate change and air pollution
Climate change and air pollution exist in a vicious feedback loop where each exacerbates the other. The combustion of fossil fuels produces ambient air pollutants of health concern, as well as greenhouse gases that contribute to climate change and extreme weather events, such as longer and hotter summers, and more frequent and intense storms and wildfires [4, 7, 18]. In many areas of the world, massive and prolonged forest fires have increased PM and gaseous pollutant concentrations [18, 19]. Additionally, there is secondary air pollution from the precursors produced in combustion processes that then combine with other substances. For example, tropospheric (low-altitude) ozone, a major contributor to smog, is formed from oxygen and hydroxyl radicals produced via a photochemical reaction from nitrogen oxide species and VOCs [5, 7]. These derive from automobile exhaust on hot, sunny days which are increasingly common with climate change. In contrast to stratospheric (high-altitude) ozone, which naturally occurs in the upper atmosphere and forms a protective layer against UV rays, ground-level ozone prevents solar heat radiation from escaping [5].
Effects of ambient air pollution exposure during fetal development and early infancy
There are many parallels between outdoor air pollutants and the harmful chemicals found in tobacco smoke, and the data on the detrimental effects of tobacco smoke are illustrative for understanding the health impacts of air pollution [20]. It has been shown through CT scans that exposure to outdoor air pollution corresponds to the emphysematous lung changes as seen in tobacco smokers [21]. Both ambient air pollution and tobacco smoke are risk factors for pre-eclampsia, preterm births, poor fetal growth, immune system alterations, neurodevelopmental abnormalities, and childhood asthma [22–28]. In addition, multiple studies have suggested that maternal exposure to the toxic gases emitted from wildfire smoke, as well as PM2.5 as a major component, increases the risk of preterm birth and term low birth weight (LBW) [8, 29–32]. For these reasons, tobacco smoke, itself a recognized air contaminant, is often used as an example to educate clinicians regarding the ill effects of ambient air pollution, as clinicians are already familiar with its adverse outcomes.
Children and neonates are particularly susceptible to the effects of air pollution due to their increased respiratory and metabolic rates which lead to greater intake of air, food, and water per kilogram of body weight. Additionally, both the fetus and child have organ systems with critical windows of development for formation and functionality. Children may also have an inability to remove themselves from hazardous exposures and have decreased thermoregulatory capabilities in response to extreme heat [1, 5, 6, 28, 33]. In comparison to mature cells, germ and fetal cells replicate and differentiate faster, and have higher sensitivity to perturbations in cell signaling [24].
These vulnerabilities set the fetus and neonate up for higher risk of adverse birth outcomes. It has been reported that indoor air pollution contributes to 20% of LBW infants and 25% of stillbirths in low-middle income countries, where high-polluting cooking fuels are a major contributor [34]. A systematic review that analyzed over 32 million births in the United States reported an association of PM2.5, ozone, and heat exposure with statistically increased risks of preterm birth, term LBW, and stillbirth [35]. Trasande et al. have estimated that over 3% of all preterm births nationwide can be attributed to PM2.5 exposure, equating to 15,808 preterm births, and over $5 billion in medical care and lost economic productivity over a one year period [36]. Studies support the hypothesis that the adverse consequences on lung health of in utero and early postnatal exposure to air pollutants are higher in preterm and LBW infants when compared to those born at term [37, 38]. In 2020, Perera et al predicted that $66 million in public health and economic benefits could be attained if the improvements in air quality achieved in New York City during the COVID-19 lockdown were sustained over five years. This study also estimated that $55 to $183 million of costs related to childhood asthma (new cases, emergency department visits, and hospitalizations) could be saved with improved air quality [39].
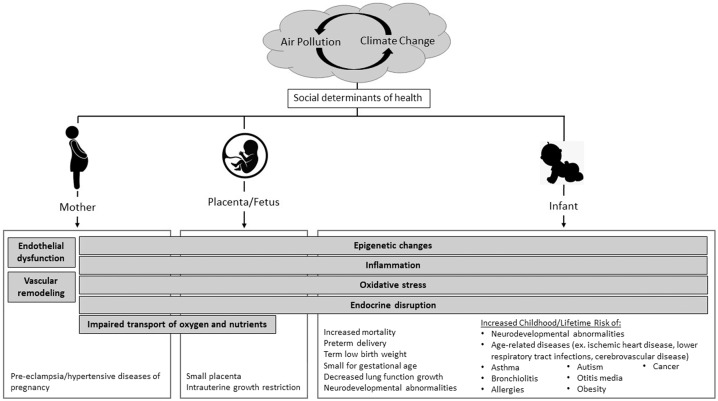
Research has suggested that air pollution exposure during pregnancy can affect the fetus directly via the placenta or indirectly through physiological changes in the mother, contributing to decreased fertility, pregnancy losses, pre-eclampsia and other hypertensive diseases of pregnancy, and gestational diabetes mellitus or insulin resistance [40–45]. This is supported by multiple epidemiological studies, including meta-analyses showing statistically increased risks for pre-eclampsia in association with exposure to PM2.5 and NO2 [46–48]. Studies have hypothesized that air pollutants can impact maternal health via multiple pathways, such as through endothelial dysfunction and vascular remodeling which can affect maternal-placental oxygen and nutrient transport to the fetus, endocrine disruption that mimics the effect of androgens and estrogens, alterations in DNA methylation processes, or promotion of oxidative stress and inflammatory processes [49]. Smoking causes reproducible DNA demethylation differences that can be used as markers for smoking status, including length of time and number of cigarettes [50]. In parallel to tobacco exposure, multiple studies have demonstrated DNA hypomethylation in adults and cord blood with exposure to PM and PAHs [51–53]. Animal and human epidemiological studies, as reviewed by Carré et al. in 2017, suggest these mechanisms can also negatively affect fertility in both men and women by causing defects during gametogenesis [54, 55]. This is an emerging field of study that holds promise for use in future perinatal research and providing biological plausibility for epidemiological studies. As our understanding of these biological processes expands, our ability to further delineate potential future implications of air pollution on pregnancy and infancy will be enhanced. Mechanisms and related health effects are shown in Fig. 1.
Fig. 1. Health impacts of air pollution across life stages.
Air pollution, which is interlinked with climate change, has health impacts that are important to neonatal-perinatal clinical specialists. The best-supported adverse health effects are listed here for individual life stages, while the gray bars indicate mechanisms of action, some of which are multi-temporal and span across multiple timeframes. The severity of health effects can be influenced by other social determinants of health, such as socioeconomic factors and race/ethnicity-related disparities.
Studies have suggested that many toxicants readily cross the placenta, and that ultrafine particles and nanoparticles are present on both the maternal and fetal sides [56, 57], with aggregates in the cytoplasm of placental trophoblastic cells [58]. These studies support the idea that the smallest components of PM can easily pass into the maternal respiratory tract, translocate into the systemic circulation, and ultimately impair placental growth as well as transport of oxygen and nutrients to the fetus [56, 58]. Gaseous pollutants, such as CO, can also affect oxygen delivery to the fetus after crossing the placental barrier, potentially causing tissue hypoxia that can reduce fetal growth [59]. Air pollution particles are able to generate free radicals or reactive oxygen/nitrogen species, inducing oxidative stress. Telomeres are particularly sensitive to oxidative stress and shorten prematurely. Research has shown an association between telomere shortening, and age-related diseases and mitochondrial dysfunction [60]. Although more studies are needed, air pollutants may have an epigenetic impact, with changes in histone acetylation, global DNA methylation, candidate gene methylation, and microRNA expression [61–63]. These epigenetic alterations can affect embryonic development of multiple organ systems, and have long-term, even multi-generational, health effects [64].
An example of how these mechanisms interact to exert health effects is the neuro-immuno-hormonal intersection. Recent research has shown that air pollution can decrease leukocytes, impair neonatal toll-like-receptor-mediated immune responses, alter cytokine levels such as IL-10 and IL-1β, and affect production of T lymphocytes, B lymphocytes, and natural killer cells in infants and cord blood [65–67]. Air pollution, specifically ultrafine PM (less than 0.1 µm in diameter), can enter the central nervous system via the olfactory nerve or through the blood-brain barrier, causing inflammation and neurodegeneration, alterations in the fine balance of excitatory and inhibitory factors in the brain, and impairment of cell proliferation, migration, differentiation, and ultimately, synaptogenesis during critical windows of development [68–70]. Higher pregnancy and infancy air pollution exposures have also been associated with altered volumes of brain structures, including the corpus callosum, hippocampus and cerebellum [71, 72]. Consistent with volume changes, air pollutants have been linked with reduced mental development index and IQ scores, psychosocial symptoms such as anxiety and depression, and delayed psychomotor development [73]. Additionally, autism spectrum disorders have been associated with prenatal air pollutant exposure [74–76]. These neurologic and neurodevelopmental effects can have lasting consequences into childhood and adulthood.
Policies, programs, and regulatory activities to reduce exposure to ambient air pollutants
The United States Clean Air Act of 1970, further amended in 1990, represents landmark legislation that is credited with a 70% reduction in mean levels of six criteria air pollutants as of 2020, and is believed to have extended the life expectancy of the average American by 1.4 years [18, 77]. However, while wildfires are becoming increasingly common, creating PM and contributing to poor air quality, the Clean Air Act exempts wildfire smoke, which is considered an “exceptional event” by the EPA [78, 79]. In August 2015, the Clean Power Plan was unveiled with the goal of reducing greenhouse gas emissions from coal-burning power plants by increasing the use of renewable energy; yet, its implementation stalled in 2017 [80, 81]. Between 2017 and 2020, over 100 environmental regulations were either eliminated or weakened, and in 2017, the United States officially withdrew from the Paris Climate Agreement, ultimately rejoining in 2021 [82, 83]. Most recently, in June 2022, the Supreme Court ruled that the Clean Air Act does not provide the EPA with the broad authority to regulate emissions from power plants, thus limiting the federal government’s ability to implement a progressive climate policy.
National regulations may appear to be slow in addressing air pollution, but stricter regulations have often been implemented at the community or state levels in advance of federal changes. One example is California, a state that has exceeded federal car emissions standards since the 1960’s, and where the successful implementation of air pollution-reduction policies has been associated with improved lung function in children [84, 85]. Thirteen additional states and Washington D.C. have adopted California’s more stringent Clean Cars legislation, and as of 2021, five states, led by Pennsylvania in 1971, have had environmental rights amended to their state constitutions [86, 87]. Most recently, New York’s Proposal 2, effective in January 2022, establishes that residents have a legal right to clean air and water, and a healthful environment [88].
Environmental justice
Perinatal providers are well aware of longstanding disparities in maternal and neonatal mortalities and morbidities. Underscoring this phenomenon is that the degree of exposure to air pollution is also disparately related to both socioeconomic status and race/ethnicity [35, 73, 89]. In 2019, it was reported that 50.5% of children in the United States reside in a county that exceeds the EPA standard for at least one criteria pollutant [90]. EPA data from 2017 demonstrated a lower percentage of white children living in counties with pollutant concentrations above the standards (76.4% for Hispanics and Asian/Pacific Islanders, and 64.1% for non-Hispanic Blacks, compared to 53.7% for non-Hispanic Whites) [90]. The effects of these exposures may be compounded by other disparities, such as inadequate nutrition or food insecurity, low-quality drinking water, substandard housing conditions, limited access to high-quality health care, and toxic stress [18, 73, 91]. In addition, stark racial disparities persist regarding pregnancy outcomes. In the United States, non-Hispanic Black and American Indian/Alaska Native women have the highest rates of maternal mortality and severe maternal morbidity compared to non-Hispanic White women [92–94]. These groups also have increased rates of infant mortality [95].
A perpetuating cycle of environmental injustice is created in which those who are able to, will move away from less desirable areas, thus protecting their future generations from the hazardous effects of pollutants. People in communities of color or of lower socioeconomic status, who commonly have health conditions driven by social determinants of health, disproportionately reside in areas with greater pollutant exposure. More often located near major roadways, flood zones, or industrial plants, these communities will endure more toxicant exposures, adversely impacting infant and fetal health [96–99]. Globally, 91% of premature deaths related to air pollution occur in low-middle income countries [18]. Of concern is that emissions can migrate and affect the air quality of other countries, further emphasizing the idea of air quality being a global issue that requires an international approach.
Across the world, many countries have set their own air quality standards based on their determination of adverse health effects and acceptability of risk. National efforts to reduce pollution levels have improved health and birth outcomes, such as with strict regulations temporarily implemented by China prior to, and during, the 2008 Olympic and Paralympic Games. During this time, levels of most pollutants decreased by 20–60%, correlating with greater birth weights compared to unrestricted periods [100, 101]. Another example is the COVID-19 pandemic, which resulted in an unprecedented global shutdown. Studies have reported significantly improved air quality and visibility in many cities, based on satellite images and pollutant measurements, particularly PM2.5 and NO2 [102–104]. On average, there was a 23% and 34% improvement in PM2.5 and NO2 levels, respectively, in New York City [39], and a 20% reduction in daily global CO2 emissions during the 2020 COVID-19 lockdown [105]. It is possible that these decreases in pollutant exposure will translate to improvement in perinatal outcomes, such as preterm birth rates and term birth weights; however, preliminary data worldwide has demonstrated widely variable maternal and infant outcomes depending on resource availability, access to healthcare, and worsening socioeconomic inequality [106]. Nevertheless, these natural experiments suggest that although extreme lockdown measures are unsustainable long term, decreased emissions are achievable through reduced or altered human activities.
Role of the neonatal-perinatal specialist
One of the most effective ways of improving outdoor air pollution is to address pollutants that contribute to climate change. Healthcare providers can be powerful advocates for reducing reliance on fossil fuels and for improving air quality, both as constituents and as educators for local legislators, on how policy decisions can impact the health of families, particularly babies and children. While the greatest impact depends on policy changes at the local and federal level, neonatal-perinatal providers can encourage local corporations and healthcare systems to prioritize institutional accountability. The global healthcare system has a large environmental impact, with energy-intensive buildings, the manufacturing and use of complicated medical equipment, the production of medical waste, and the use of unsustainable materials and anesthetics that are themselves greenhouse gases [107]. On its own, the United States’ healthcare system would rank 13th as a country for greenhouse gas emissions [107, 108]. Few hospitals have instituted strategies to promote sustainability, citing expenses, regulatory compliance, operational hours, and infection control as main barriers. Neonatal-perinatal providers, who are primarily hospital-based, have a unique opportunity to educate administrators and advocate for change. In an effort to become more “green”, some hospitals have successfully implemented more efficient heating, ventilation, and air conditioning systems, used more renewable energy sources such as solar and wind energy, installed charging stations for electric cars, reprocessed medical equipment, and adopted sustainable food practices [109, 110]. The Health Care Climate Challenge (www.greenhospitals.net/about-challenge) was launched at the Paris Climate Conference in 2015 as an initiative for health care institutions worldwide to prioritize clean, renewable, healthy energy, and serve as advocates for improving patient and community health [111].
Clinicians are poised to connect with patients and parents to encourage them to make positive changes to address environmental health concerns. Pregnancy is a unique life stage that offers a window of opportunity in which positive life changes and healthy behavior can be significantly influenced by healthcare providers. It is also important that clinicians recognize the negative impact that air pollution, climate change, and heat stress can have on the mental health of parents. Antenatal and postnatal psychological distress can subsequently affect fetal and neonatal well-being.
Pregnant individuals who live in highly polluted areas, such as in wildfire-prone communities or traffic-dense urban environments, can benefit from using filtration devices [112, 113]. Holm et al. reviewed studies that reported a significant decrease in PM2.5 levels and attributable asthma morbidity, as well as higher standardized test scores, with the use of higher filtration efficiency filters [114]. A recent randomized controlled trial demonstrated that the use of high efficiency particulate air (HEPA) filter air cleaners in highly polluted settings in Mongolia was associated with significantly increased birth weights of term infants [115].
Neonatal-perinatal providers are in an ideal position to educate families about their exposures and the air quality index in their neighborhoods. Health promotion behaviors can be integrated into routine office visits [116], and there are several existing resources to facilitate provider discussions. For example, the New York State Children’s Environmental Health Centers created “Prescriptions for Prevention” (www.nyscheck.org/rx), one of which is to specifically educate families on outdoor air quality, local resources, and steps to reduce pollutant exposures [117]. The nationwide Pediatric Environmental Health Specialty Units network (www.pehsu.net) offers informational resources for clinicians on air pollution, in relation to reproductive and pediatric health [118]. In addition, the American Board of Pediatrics offers a Maintenance of Certification module to provide clinicians with additional education on climate, health, and equity. Additional resources for clinicians are provided in Table 1.
Conclusion
Air pollution and climate change contribute significantly to birth outcomes and health effects that can persist into childhood and adulthood, and a variety of mechanisms have been proposed, similar to those described for tobacco smoke exposure. The ever-changing climate and our increasing understanding of epigenetic and biological mechanisms mandates an expansive 21st century approach to reducing exposure to air pollution, such as broadening air quality policies to include exposure from wildfires. Neonatal-perinatal clinicians care for a uniquely vulnerable population, and through direct education and public health messaging, can have a significant influence on how patients and their communities understand climate change. Greater focus should be paid to clinically-relevant resources that allow physicians to better evaluate and treat the impacts of air pollution on health. For example, epigenetic screening may someday be of utility in gauging and educating patients on the insidious impacts of air pollution. Neonatal-perinatal providers can be role models for their patients not just on an individual level, but also as powerful advocates who can educate legislators on the impact to patients who may not yet have a vote or a voice to speak for themselves.
Author contributions
ML drafted the initial version, and revised several versions of the manuscript. CJK made critical revisions to the manuscript, and SIS provided significant contributions and revisions to the advocacy section. HLB mentored the primary author, and critically reviewed and revised the manuscript. All authors approved the final manuscript as submitted and agree to be accountable for all aspects of the work.
Competing interests
The authors declare no competing interests.
Footnotes
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
References
- 1.Brumberg HL, Karr CJ, Council On Environmental Health. Ambient air pollution: health hazards to children. Pediatrics. 2021;147:e2021051484. [DOI] [PubMed]
- 2.Council On Environmental H. Global climate change and children’s health. Pediatrics. 2015;136:992–7. doi: 10.1542/peds.2015-3232. [DOI] [PubMed] [Google Scholar]
- 3.Ahdoot S, Pacheco SE. Council On Environmental Health. Global climate change and children’s health. Pediatrics. 2015;136:e1468–84. doi: 10.1542/peds.2015-3233. [DOI] [PubMed] [Google Scholar]
- 4.IPCC. Climate change 2021: the physical science basis, Sixth Assessment Report. Cambridge University Press, Cambridge, United Kingdom and New York, NY, USA: IPCC; 2021. pp. 33–144.
- 5.Landrigan PJ, Etzel RA (editors). Textbook of children’s environmental health. New York, NY, USA: Oxford University Press; 2014.
- 6.American Academy of Pediatrics COEH. Pediatric environmental health. 4th ed. Itasca, IL: American Academy of Pediatrics; 2019.
- 7.Phalen RF, Phalen RN. Introduction to air pollution science: a public health perspective. Burlington, MA, USA: Jones & Bartlett Learning; 2013.
- 8.Amjad S, Chojecki D, Osornio-Vargas A, Ospina MB. Wildfire exposure during pregnancy and the risk of adverse birth outcomes: a systematic review. Environ Int. 2021;156:106644. doi: 10.1016/j.envint.2021.106644. [DOI] [PubMed] [Google Scholar]
- 9.O’Lenick CR, Wilhelmi OV, Michael R, Hayden MH, Baniassadi A, Wiedinmyer C, et al. Urban heat and air pollution: a framework for integrating population vulnerability and indoor exposure in health risk analyses. Sci Total Environ. 2019;660:715–23. doi: 10.1016/j.scitotenv.2019.01.002. [DOI] [PubMed] [Google Scholar]
- 10.American Lung Association. What’s In a Cigarette? 2020. https://www.lung.org/quit-smoking/smoking-facts/whats-in-a-cigarette.
- 11.United States Environmental Protection Agency. Hazardous air pollutants. https://www.epa.gov/haps. Accessed September 6, 2021.
- 12.United States Environmental Protection Agency. Criteria air pollutants. https://www.epa.gov/criteria-air-pollutants. Accessed September 6, 2021.
- 13.United States Environmental Protection Agency. Reviewing National Ambient Air Quality Standards (NAAQS): scientific and technical information. https://www.epa.gov/naaqs. Accessed September 6, 2021.
- 14.United States Environmental Protection Agency. National Emission Standards for Hazardous Air Pollutants (NESHAP). https://www.epa.gov/stationary-sources-air-pollution/national-emission-standards-hazardous-air-pollutants-neshap-9. Accessed December 8, 2021.
- 15.Registry AfTSaD. What are possible health effects from lead exposure? 2019. https://www.atsdr.cdc.gov/csem/leadtoxicity/physiological_effects.html.
- 16.United States Environmental Protection Agency. Hazardous air pollutants: sources and exposure. https://www.epa.gov/haps/hazardous-air-pollutants-sources-and-exposure. Accessed September 6, 2021.
- 17.Bantol KEA, Brumberg HL, Shah SI, Javier JR. Perspectives from the Society for Pediatric Research: contaminants of water and children’s health: Can we do better? Pediatr Res. 2020;88:535–43. doi: 10.1038/s41390-020-0985-4. [DOI] [PubMed] [Google Scholar]
- 18.WHO’s global air-quality guidelines. Lancet. 2006;368:1302. [DOI] [PubMed]
- 19.Haines A, Ebi K. The imperative for climate action to protect health. N Engl J Med. 2019;380:263–73. doi: 10.1056/NEJMra1807873. [DOI] [PubMed] [Google Scholar]
- 20.US Food and Drug Administration. Harmful and potentially harmful constituents in tobacco products and tobacco smoke; Established List. 2019. https://www.federalregister.gov/documents/2012/04/03/2012-7727/harmful-and-potentially-harmful-constituents-in-tobacco-products-and-tobacco-smoke-established-list.
- 21.Wang M, Aaron CP, Madrigano J, Hoffman EA, Angelini E, Yang J, et al. Association between long-term exposure to ambient air pollution and change in quantitatively assessed emphysema and lung function. JAMA. 2019;322:546–56. doi: 10.1001/jama.2019.10255. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Hackshaw A, Rodeck C, Boniface S. Maternal smoking in pregnancy and birth defects: a systematic review based on 173 687 malformed cases and 11.7 million controls. Hum Reprod Update. 2011;17:589–604. doi: 10.1093/humupd/dmr022. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Kahr MK, Suter MA, Ballas J, Ramphul R, Lubertino G, Hamilton WJ, et al. Preterm birth and its associations with residence and ambient vehicular traffic exposure. Am J Obstet Gynecol. 2016;215:111 e1–e10. doi: 10.1016/j.ajog.2016.01.171. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Korten I, Ramsey K, Latzin P. Air pollution during pregnancy and lung development in the child. Paediatr Respir Rev. 2017;21:38–46. doi: 10.1016/j.prrv.2016.08.008. [DOI] [PubMed] [Google Scholar]
- 25.Liu Y, Xu J, Chen D, Sun P, Ma X. The association between air pollution and preterm birth and low birth weight in Guangdong, China. BMC Public Health. 2019;19:1–10.. doi: 10.1186/s12889-018-6307-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Li C, Yang M, Zhu Z, Sun S, Zhang Q, Cao J, et al. Maternal exposure to air pollution and the risk of low birth weight: a meta-analysis of cohort studies. Environ Res. 2020;190:109970. doi: 10.1016/j.envres.2020.109970. [DOI] [PubMed] [Google Scholar]
- 27.Perera F, Ashrafi A, Kinney P, Mills D. Towards a fuller assessment of benefits to children’s health of reducing air pollution and mitigating climate change due to fossil fuel combustion. Environ Res. 2019;172:55–72. doi: 10.1016/j.envres.2018.12.016. [DOI] [PubMed] [Google Scholar]
- 28.Perera F, Nadeau K. Climate change, fossil-fuel pollution, and children’s health. N Engl J Med. 2022;386:2303–14.. doi: 10.1056/NEJMra2117706. [DOI] [PubMed] [Google Scholar]
- 29.Heft-Neal S, Driscoll A, Yang W, Shaw G, Burke M. Associations between wildfire smoke exposure during pregnancy and risk of preterm birth in California. Environ Res. 2022;203:111872. doi: 10.1016/j.envres.2021.111872. [DOI] [PubMed] [Google Scholar]
- 30.Holstius DM, Reid CE, Jesdale BM, Morello-Frosch R. Birth weight following pregnancy during the 2003 Southern California wildfires. Environ Health Perspect. 2012;120:1340–5. doi: 10.1289/ehp.1104515. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Requia WJ, Papatheodorou S, Koutrakis P, Mukherjee R, Roig HL. Increased preterm birth following maternal wildfire smoke exposure in Brazil. Int J Hyg Environ Health. 2022;240:113901. doi: 10.1016/j.ijheh.2021.113901. [DOI] [PubMed] [Google Scholar]
- 32.Abdo M, Ward I, O’Dell K, Ford B, Pierce JR, Fischer EV, et al. Impact of wildfire smoke on adverse pregnancy outcomes in Colorado, 2007-2015. Int J Environ Res Public Health. 2019;16:3720. [DOI] [PMC free article] [PubMed]
- 33.Smith CJ. Pediatric thermoregulation: considerations in the face of global climate change. Nutrients. 2019;11:2010. [DOI] [PMC free article] [PubMed]
- 34.Pope DP, Mishra V, Thompson L, Siddiqui AR, Rehfuess EA, Weber M, et al. Risk of low birth weight and stillbirth associated with indoor air pollution from solid fuel use in developing countries. Epidemiol Rev. 2010;32:70–81. doi: 10.1093/epirev/mxq005. [DOI] [PubMed] [Google Scholar]
- 35.Bekkar B, Pacheco S, Basu R, DeNicola N. Association of air pollution and heat exposure with preterm birth, low birth weight, and stillbirth in the US: a systematic review. JAMA Netw Open. 2020;3:e208243. doi: 10.1001/jamanetworkopen.2020.8243. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Trasande L, Malecha P, Attina TM. Particulate matter exposure and preterm birth: estimates of U.S. attributable burden and economic costs. Environ Health Perspect. 2016;124:1913–8. doi: 10.1289/ehp.1510810. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Lavigne E, Belair MA, Rodriguez Duque D, Do MT, Stieb DM, Hystad P, et al. Effect modification of perinatal exposure to air pollution and childhood asthma incidence. Eur Respir J. 2018;51:1701884. [DOI] [PMC free article] [PubMed]
- 38.Decrue F, Gorlanova O, Salem Y, Vienneau D, de Hoogh K, Gisler A, et al. Increased impact of air pollution on lung function in preterm versus term infants: the BILD study. Am J Respir Crit Care Med. 2022;205:99–107. doi: 10.1164/rccm.202102-0272OC. [DOI] [PubMed] [Google Scholar]
- 39.Perera F, Berberian A, Cooley D, Shenaut E, Olmstead H, Ross Z, et al. Potential health benefits of sustained air quality improvements in New York City: a simulation based on air pollution levels during the COVID-19 shutdown. Environ Res. 2021;193:110555. doi: 10.1016/j.envres.2020.110555. [DOI] [PubMed] [Google Scholar]
- 40.Bearblock E, Aiken CE, Burton GJ. Air pollution and pre-eclampsia; associations and potential mechanisms. Placenta. 2021;104:188–94.. doi: 10.1016/j.placenta.2020.12.009. [DOI] [PubMed] [Google Scholar]
- 41.Hong X, Liu C, Chen X, Song Y, Wang Q, Wang P, et al. Maternal exposure to airborne particulate matter causes postnatal immunological dysfunction in mice offspring. Toxicology. 2013;306:59–67. doi: 10.1016/j.tox.2013.02.004. [DOI] [PubMed] [Google Scholar]
- 42.Inoue K, Yan Q, Arah OA, Paul K, Walker DI, Jones DP, et al. Air pollution and adverse pregnancy and birth outcomes: mediation analysis using metabolomic profiles. Curr Environ Health Rep. 2020;7:231–42.. doi: 10.1007/s40572-020-00284-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Klepac P, Locatelli I, Korosec S, Kunzli N, Kukec A. Ambient air pollution and pregnancy outcomes: a comprehensive review and identification of environmental public health challenges. Environ Res. 2018;167:144–59.. doi: 10.1016/j.envres.2018.07.008. [DOI] [PubMed] [Google Scholar]
- 44.Koman PD, Hogan KA, Sampson N, Mandell R, Coombe CM, Tetteh MM, et al. Examining joint effects of air pollution exposure and social determinants of health in defining “at-risk” populations under the clean air act: susceptibility of pregnant women to hypertensive disorders of pregnancy. World Med Health Policy. 2018;10:7–54. doi: 10.1002/wmh3.257. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Sram RJ, Binkova B, Dejmek J, Bobak M. Ambient air pollution and pregnancy outcomes: a review of the literature. Environ Health Perspect. 2005;113:375–82. doi: 10.1289/ehp.6362. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Pedersen M, Stayner L, Slama R, Sorensen M, Figueras F, Nieuwenhuijsen MJ, et al. Ambient air pollution and pregnancy-induced hypertensive disorders: a systematic review and meta-analysis. Hypertension. 2014;64:494–500. doi: 10.1161/HYPERTENSIONAHA.114.03545. [DOI] [PubMed] [Google Scholar]
- 47.Cao L, Wang L, Wu L, Wang T, Cui X, Yu L, et al. Particulate matter and hypertensive disorders in pregnancy: systematic review and meta-analysis. Public Health. 2021;200:22–32. doi: 10.1016/j.puhe.2021.08.013. [DOI] [PubMed] [Google Scholar]
- 48.Gogna P, Villeneuve PJ, Borghese MM, King WD. An exposure-response meta-analysis of ambient PM2.5 during pregnancy and preeclampsia. Environ Res. 2022;210:112934. doi: 10.1016/j.envres.2022.112934. [DOI] [PubMed] [Google Scholar]
- 49.Saenen ND, Martens DS, Neven KY, Alfano R, Bove H, Janssen BG, et al. Air pollution-induced placental alterations: an interplay of oxidative stress, epigenetics, and the aging phenotype? Clin Epigenetics. 2019;11:124. doi: 10.1186/s13148-019-0688-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Philibert RA, Beach SR, Brody GH. Demethylation of the aryl hydrocarbon receptor repressor as a biomarker for nascent smokers. Epigenetics. 2012;7:1331–8. doi: 10.4161/epi.22520. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Bellavia A, Urch B, Speck M, Brook RD, Scott JA, Albetti B, et al. DNA hypomethylation, ambient particulate matter, and increased blood pressure: findings from controlled human exposure experiments. J Am Heart Assoc. 2013;2:e000212. doi: 10.1161/JAHA.113.000212. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Burris HH, Baccarelli AA. Air pollution and in utero programming of poor fetal growth. Epigenomics. 2017;9:213–6. doi: 10.2217/epi-2017-0008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Herbstman JB, Tang D, Zhu D, Qu L, Sjodin A, Li Z, et al. Prenatal exposure to polycyclic aromatic hydrocarbons, benzo[a]pyrene-DNA adducts, and genomic DNA methylation in cord blood. Environ Health Perspect. 2012;120:733–8. doi: 10.1289/ehp.1104056. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Carre J, Gatimel N, Moreau J, Parinaud J, Leandri R. Does air pollution play a role in infertility?: a systematic review. Environ Health. 2017;16:82. doi: 10.1186/s12940-017-0291-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Pires A, de Melo EN, Mauad T, Nascimento Saldiva PH, de Siqueira Bueno HM. Pre- and postnatal exposure to ambient levels of urban particulate matter (PM(2.5)) affects mice spermatogenesis. Inhal Toxicol. 2011;23:237–45. doi: 10.3109/08958378.2011.563508. [DOI] [PubMed] [Google Scholar]
- 56.Bove H, Bongaerts E, Slenders E, Bijnens EM, Saenen ND, Gyselaers W, et al. Ambient black carbon particles reach the fetal side of human placenta. Nat Commun. 2019;10:3866. doi: 10.1038/s41467-019-11654-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Bongaerts E, Nawrot TS, Van Pee T, Ameloot M, Bove H. Translocation of (ultra)fine particles and nanoparticles across the placenta; a systematic review on the evidence of in vitro, ex vivo, and in vivo studies. Part Fibre Toxicol. 2020;17:56. doi: 10.1186/s12989-020-00386-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Valentino SA, Tarrade A, Aioun J, Mourier E, Richard C, Dahirel M, et al. Maternal exposure to diluted diesel engine exhaust alters placental function and induces intergenerational effects in rabbits. Part Fibre Toxicol. 2016;13:39. doi: 10.1186/s12989-016-0151-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59.Salam MT, Millstein J, Li YF, Lurmann FW, Margolis HG, Gilliland FD. Birth outcomes and prenatal exposure to ozone, carbon monoxide, and particulate matter: results from the Children’s Health Study. Environ Health Perspect. 2005;113:1638–44. doi: 10.1289/ehp.8111. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Yang IA, Fong KM, Zimmerman PV, Holgate ST, Holloway JW. Genetic susceptibility to the respiratory effects of air pollution. Thorax. 2008;63:555–63.. doi: 10.1136/thx.2007.079426. [DOI] [PubMed] [Google Scholar]
- 61.Holloway JW, Savarimuthu Francis S, Fong KM, Yang IA. Genomics and the respiratory effects of air pollution exposure. Respirology. 2012;17:590–600. doi: 10.1111/j.1440-1843.2012.02164.x. [DOI] [PubMed] [Google Scholar]
- 62.Isaevska E, Moccia C, Asta F, Cibella F, Gagliardi L, Ronfani L, et al. Exposure to ambient air pollution in the first 1000 days of life and alterations in the DNA methylome and telomere length in children: a systematic review. Environ Res. 2021;193:110504. doi: 10.1016/j.envres.2020.110504. [DOI] [PubMed] [Google Scholar]
- 63.Martens DS, Cox B, Janssen BG, Clemente DBP, Gasparrini A, Vanpoucke C, et al. Prenatal air pollution and newborns’ predisposition to accelerated biological aging. JAMA Pediatr. 2017;171:1160–7. doi: 10.1001/jamapediatrics.2017.3024. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64.Mitchell C, Schneper LM, Notterman DA. DNA methylation, early life environment, and health outcomes. Pediatr Res. 2016;79:212–9. doi: 10.1038/pr.2015.193. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 65.Hertz-Picciotto I, Dostal M, Dejmek J, Selevan SG, Wegienka G, Gomez-Caminero A, et al. Air pollution and distributions of lymphocyte immunophenotypes in cord and maternal blood at delivery. Epidemiology. 2002;13:172–83.. doi: 10.1097/00001648-200203000-00012. [DOI] [PubMed] [Google Scholar]
- 66.Hertz-Picciotto I, Herr CE, Yap PS, Dostal M, Shumway RH, Ashwood P, et al. Air pollution and lymphocyte phenotype proportions in cord blood. Environ Health Perspect. 2005;113:1391–8. doi: 10.1289/ehp.7610. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 67.Latzin P, Frey U, Armann J, Kieninger E, Fuchs O, Roosli M, et al. Exposure to moderate air pollution during late pregnancy and cord blood cytokine secretion in healthy neonates. PLoS One. 2011;6:e23130. doi: 10.1371/journal.pone.0023130. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 68.Brockmeyer S, D’Angiulli A. How air pollution alters brain development: the role of neuroinflammation. Transl Neurosci. 2016;7:24–30. doi: 10.1515/tnsci-2016-0005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 69.Costa LG, Cole TB, Dao K, Chang YC, Garrick JM. Developmental impact of air pollution on brain function. Neurochem Int. 2019;131:104580. doi: 10.1016/j.neuint.2019.104580. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 70.Zheng X, Wang X, Wang T, Zhang H, Wu H, Zhang C, et al. Gestational exposure to particulate matter 2.5 (PM2.5) leads to spatial memory dysfunction and neurodevelopmental impairment in hippocampus of mice offspring. Front Neurosci. 2018;12:1000. doi: 10.3389/fnins.2018.01000. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 71.Lubczynska MJ, Muetzel RL, El Marroun H, Hoek G, Kooter IM, Thomson EM, et al. Air pollution exposure during pregnancy and childhood and brain morphology in preadolescents. Environ Res. 2021;198:110446. doi: 10.1016/j.envres.2020.110446. [DOI] [PubMed] [Google Scholar]
- 72.Calderon-Garciduenas L, Engle R, Mora-Tiscareno A, Styner M, Gomez-Garza G, Zhu H, et al. Exposure to severe urban air pollution influences cognitive outcomes, brain volume and systemic inflammation in clinically healthy children. Brain Cogn. 2011;77:345–55.. doi: 10.1016/j.bandc.2011.09.006. [DOI] [PubMed] [Google Scholar]
- 73.Perera FP. Multiple threats to child health from fossil fuel combustion: impacts of air pollution and climate change. Environ Health Perspect. 2017;125:141–8. doi: 10.1289/EHP299. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 74.Becerra TA, Wilhelm M, Olsen J, Cockburn M, Ritz B. Ambient air pollution and autism in Los Angeles county, California. Environ Health Perspect. 2013;121:380–6. doi: 10.1289/ehp.1205827. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 75.Raz R, Roberts AL, Lyall K, Hart JE, Just AC, Laden F, et al. Autism spectrum disorder and particulate matter air pollution before, during, and after pregnancy: a nested case-control analysis within the Nurses’ Health Study II Cohort. Environ Health Perspect. 2015;123:264–70.. doi: 10.1289/ehp.1408133. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 76.Volk HE, Lurmann F, Penfold B, Hertz-Picciotto I, McConnell R. Traffic-related air pollution, particulate matter, and autism. JAMA Psychiatry. 2013;70:71–7. doi: 10.1001/jamapsychiatry.2013.266. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 77.United States Environmental Protection Agency. Overview of the clean air act and air pollution. https://www.epa.gov/clean-air-act-overview.
- 78.United States Environmental Protection Agency. Treatment of data influenced by exceptional events. Research Triangle Park, NC USA: United States Environmental Protection Agency; 2016.
- 79.Williams E. Reimagining exceptional events: regulating wildfires through the clean air act. Seattle, WA, USA: University of Washington Law Review; 2021. p. 765–810.
- 80.United States Environmental Protection Agency. Fact sheet: overview of the clean power plan. Cutting Carbon Pollution from Power Plants. https://archive.epa.gov/epa/cleanpowerplan/fact-sheet-overview-clean-power-plan.html.
- 81.United States Environmental Protection Agency. Electric utility generating units: repealing the clean power plan. https://www.epa.gov/stationary-sources-air-pollution/electric-utility-generating-units-repealing-clean-power-plan.
- 82.Popovich N, Albeck-Ripka L, Pierre-Louis K. The Trump administration rolled back more than 100 environmental rules. Here’s the Full List. New York City, NY, USA: The New York Times; 2021. Retrieved from: https://www.nytimes.com/interactive/2020/climate/trump-environment-rollbacks-list.html.
- 83.US Department of State. The United States officially rejoins the Paris agreement. https://www.state.gov/the-united-states-officially-rejoins-the-paris-agreement/.
- 84.United States Environmental Protection Agency. Vehicle emissions california waivers and authorizations. https://www.epa.gov/state-and-local-transportation/vehicle-emissions-california-waivers-and-authorizations.
- 85.Gauderman WJ, Urman R, Avol E, Berhane K, McConnell R, Rappaport E, et al. Association of improved air quality with lung development in children. N. Engl J Med. 2015;372:905–13. doi: 10.1056/NEJMoa1414123. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 86.American Council of Energy Efficient-Economy. Tailpipe emission standards. https://database.aceee.org/state/tailpipe-emission-standards.
- 87.Williams K. Fundamental environmental rights: state constitutions as a vehicle of change. Jurist - Student Commentary. 2021. https://www.jurist.org/commentary/2021/11/kirsten-williams-environmental-rights-amendments.
- 88.New York state board of elections. 2021 statewide ballot proposals. 2021. https://www.elections.ny.gov/2021BallotProposals.html.
- 89.Ragavan MI, Marcil LE, Garg A. Climate change as a social determinant of health. Pediatrics. 2020;145:e20193169. [DOI] [PMC free article] [PubMed]
- 90.United States Environmental Protection Agency. America’s children and the environment. Washington, DC, USA: United States Environmental Protection Agency; 2019.
- 91.Blakstad MM, Smith ER. Climate change worsens global inequity in maternal nutrition. Lancet Planet Health. 2020;4:e547–e8.. doi: 10.1016/S2542-5196(20)30246-1. [DOI] [PubMed] [Google Scholar]
- 92.Holdt Somer SJ, Sinkey RG, Bryant AS. Epidemiology of racial/ethnic disparities in severe maternal morbidity and mortality. Semin Perinatol. 2017;41:258–65.. doi: 10.1053/j.semperi.2017.04.001. [DOI] [PubMed] [Google Scholar]
- 93.MacDorman MF, Thoma M, Declcerq E, Howell EA. Racial and ethnic disparities in maternal mortality in the United States using enhanced vital records, 2016-2017. Am J Public Health. 2021;111:1673–81.. doi: 10.2105/AJPH.2021.306375. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 94.Centers for Disease Control and Prevention. Racial and Ethnic Disparities Continue in Pregnancy-Related Deaths. Press release September 5, 2019. https://www.cdc.gov/media/releases/2019/p0905-racial-ethnic-disparities-pregnancy-deaths.html.
- 95.Ely DM, Driscoll AK. Infant mortality in the United States, 2018: data from the period linked birth/infant death file. Atlanta, GA, USA: Division of Reproductive Health, National Center for Chronic Disease Prevention and Health Promotion (NCCDPHP), Centers for Disease Control and Prevention; 2021. [PubMed]
- 96.Gutschow B, Gray B, Ragavan MI, Sheffield PE, Philipsborn RP, Jee SH. The intersection of pediatrics, climate change, and structural racism: Ensuring health equity through climate justice. Curr Probl Pediatr Adolesc Health Care. 2021;51:101028. doi: 10.1016/j.cppeds.2021.101028. [DOI] [PubMed] [Google Scholar]
- 97.Gochfeld M, Burger J. Disproportionate exposures in environmental justice and other populations: the importance of outliers. Am J Public Health. 2011;101:S53–63. doi: 10.2105/AJPH.2011.300121. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 98.Morello-Frosch R, Zuk M, Jerrett M, Shamasunder B, Kyle AD. Understanding the cumulative impacts of inequalities in environmental health: implications for policy. Health Aff. 2011;30:879–87. doi: 10.1377/hlthaff.2011.0153. [DOI] [PubMed] [Google Scholar]
- 99.Lamichhane DK, Lee SY, Ahn K, Kim KW, Shin YH, Suh DI, et al. Quantile regression analysis of the socioeconomic inequalities in air pollution and birth weight. Environ Int. 2020;142:105875. doi: 10.1016/j.envint.2020.105875. [DOI] [PubMed] [Google Scholar]
- 100.Huang C, Nichols C, Liu Y, Zhang Y, Liu X, Gao S, et al. Ambient air pollution and adverse birth outcomes: a natural experiment study. Popul Health Metr. 2015;13:17. doi: 10.1186/s12963-015-0050-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 101.Rich DQ, Kipen HM, Huang W, Wang G, Wang Y, Zhu P, et al. Association between changes in air pollution levels during the Beijing Olympics and biomarkers of inflammation and thrombosis in healthy young adults. JAMA. 2012;307:2068–78. doi: 10.1001/jama.2012.3488. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 102.Ching J, Kajino M. Rethinking air quality and climate change after COVID-19. Int J Environ Res Public Health. 2020;17:5167. [DOI] [PMC free article] [PubMed]
- 103.Sannigrahi S, Kumar P, Molter A, Zhang Q, Basu B, Basu AS, et al. Examining the status of improved air quality in world cities due to COVID-19 led temporary reduction in anthropogenic emissions. Environ Res. 2021;196:110927. doi: 10.1016/j.envres.2021.110927. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 104.Venter ZS, Aunan K, Chowdhury S, Lelieveld J. COVID-19 lockdowns cause global air pollution declines. Proc Natl Acad Sci USA. 2020;117:18984–90.. doi: 10.1073/pnas.2006853117. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 105.Le Quéré C, Jackson RB, Jones MW, Smith AJP, Abernethy S, Andrew RM, et al. Temporary reduction in daily global CO2 emissions during the COVID-19 forced confinement. Nat Clim Change. 2020;10:647–53.. doi: 10.1038/s41558-020-0797-x. [DOI] [Google Scholar]
- 106.Stock SJ, Zoega H, Brockway M, Mulholland RH, Miller JE, Been JV, et al. The international Perinatal Outcomes in the Pandemic (iPOP) study: protocol. Wellcome Open Res. 2021;6:21. doi: 10.12688/wellcomeopenres.16507.1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 107.Eckelman MJ, Sherman J. Environmental impacts of the U.S. health care system and effects on public health. PLoS One. 2016;11:e0157014. doi: 10.1371/journal.pone.0157014. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 108.Dzau VJ, Levine R, Barrett G, Witty A. Decarbonizing the U.S. health sector - a call to action. N Engl J Med. 2021;385:2117–9. doi: 10.1056/NEJMp2115675. [DOI] [PubMed] [Google Scholar]
- 109.Jablow MM. Teaching hospitals go green. AAMC News; 2021. https://www.aamc.org/news-insights/teaching-hospitals-go-green.
- 110.Harm HCW. Global road map for health care decarbonization. 2022. https://healthcareclimateaction.org/roadmap.
- 111.Harm GGaHH-HCW. Health care climate challenge. 2015. https://www.greenhospitals.net/about-challenge/.
- 112.Maestas MM, Brook RD, Ziemba RA, Li F, Crane RC, Klaver ZM, et al. Reduction of personal PM2.5 exposure via indoor air filtration systems in Detroit: an intervention study. J Expo Sci Environ Epidemiol. 2019;29:484–90. doi: 10.1038/s41370-018-0085-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 113.Mott JA, Meyer P, Mannino D, Redd SC, Smith EM, Gotway-Crawford C, et al. Wildland forest fire smoke: health effects and intervention evaluation, Hoopa, California, 1999. West J Med. 2002;176:157–62. doi: 10.1136/ewjm.176.3.157. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 114.Holm SM, Miller MD, Balmes JR. Health effects of wildfire smoke in children and public health tools: a narrative review. J Expo Sci Environ Epidemiol. 2021;31:1–20. doi: 10.1038/s41370-020-00267-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 115.Barn P, Gombojav E, Ochir C, Laagan B, Beejin B, Naidan G, et al. The effect of portable HEPA filter air cleaners on indoor PM2.5 concentrations and second hand tobacco smoke exposure among pregnant women in Ulaanbaatar, Mongolia: the UGAAR randomized controlled trial. Sci Total Environ. 2018;615:1379–89. doi: 10.1016/j.scitotenv.2017.09.291. [DOI] [PubMed] [Google Scholar]
- 116.Philipsborn RP, Cowenhoven J, Bole A, Balk SJ, Bernstein A. A pediatrician’s guide to climate change-informed primary care. Curr Probl Pediatr Adolesc Health Care. 2021;51:101027. doi: 10.1016/j.cppeds.2021.101027. [DOI] [PubMed] [Google Scholar]
- 117.Unit NYSCsEHCaPEHS. New York State prescriptions for prevention New York, NY: NYS Children’s Environmental Health Centers - Department of Environmental Medicine and Public Health Icahn School of Medicine at Mount Sinai; 2021. https://nyscheck.org/rx/.
- 118.Woolf AD, Sibrizzi C, Kirkland K. Pediatric environmental health specialty units: an analysis of operations. Acad Pediatr. 2016;16:25–33. doi: 10.1016/j.acap.2015.05.006. [DOI] [PubMed] [Google Scholar]