Abstract
In the presence of trimethylamine N-oxide (TMAO), the TorS-TorR two-component regulatory system induces the torCAD operon, which encodes the TMAO respiratory system of Escherichia coli. The sensor protein TorS detects TMAO and transphosphorylates the response regulator TorR which, in turn, activates transcription of torCAD. The torR gene and the torCAD operon are divergently transcribed, and the short torR-torC intergenic region contains four direct repeats (the tor boxes) which proved to be TorR binding sites. The tor box 1-box 2 region covers the torR transcription start site and constitutes a TorR high-affinity binding site, whereas box 3 and box 4 correspond to low-affinity binding sites. By using torR-lacZ operon fusions in different genetic backgrounds, we showed that the torR gene is negatively autoregulated. Surprisingly, TorR autoregulation is TMAO independent and still occurs in a torS mutant. In addition, this negative regulation involves only the TorR high-affinity binding site. Together, these data suggest that phosphorylated as well as unphosphorylated TorR binds the box 1-box 2 region in vivo, thus preventing RNA polymerase from binding to the torR promoter whatever the growth conditions. By changing the spacing between box 2 and box 3, we demonstrated that the DNA motifs of the high- and low-affinity binding sites must be close to each other and located on the same side of the DNA helix to allow induction of the torCAD operon. Thus, prior TorR binding to the box 1-box 2 region seems to allow cooperative binding of phosphorylated TorR to box 3 and box 4.
Trimethylamine N-oxide (TMAO) is an organic compound widespread in nature, and very high levels of it accumulate in the tissues of fish, where it acts as a powerful osmoprotector (37). TMAO can also play the role of an alternative electron acceptor for bacterial anaerobic respiration (2, 3). In Escherichia coli, the genes encoding the TMAO reductase respiratory system are clustered in the torCAD operon (22). torC encodes a c-type cytochrome (TorC) anchored to the inner membrane, whereas torA encodes the periplasmic terminal enzyme (TorA). TorD, the product of the third gene of the torCAD operon, seems to be a TorA chaperone (28).
Expression of the torCAD operon is under the control of both anaerobiosis and TMAO or related compounds (35). The TMAO control is strict, as torCAD is rarely transcribed in the absence of TMAO (22). The anaerobic control is not as strong as that of TMAO, and expression of the tor operon decreases 5- to 10-fold under aerobic conditions (35). The tor anaerobic regulator, which is different from FNR or ArcA, is still unknown, and the TMAO control is mediated by the TorS-TorR two-component regulatory system (15, 35). We have shown that the sensor protein TorS seems to interact not only with TMAO but also with a periplasmic binding protein, TorT (16) and with the immature form of the TorC cytochrome (1). Whereas TorT is a positive regulator essential for tor operon induction, the apoform of TorC plays a negative autoregulatory role probably by inhibiting the TorS kinase activity. As TorS belongs to the family of the unorthodox sensors such as ArcB or BvgS (7, 24), the signal transduction from TorS to TorR involves a four-step phosphorelay (14). Once phosphorylated, TorR activates the tor operon transcription by binding to the torCAD promoter (34).
The short untranslated region between torR and torC contains four direct repeats of a decameric consensus motif (35) (see Fig. 2 and 3). These repeats have been called the tor boxes, and boxes 1, 2, and 4 correspond exactly to the same decameric sequence (CTGTTCATAT), whereas box 3 matches 7 of the 10 bases of the consensus (CCGTTCATCC). By using plasmid-born torC-lacZ transcriptional fusions, we have observed that a double substitution within one of the four tor boxes leads to a strong decrease in fusion activity (34). This clearly indicates that the tor boxes are important cis elements involved in tor operon expression. We have further shown that TorR binds specifically to the four tor boxes. However, we noticed from gel retardation assays and footprinting experiments that the box 1-box 2 region constitutes a high-affinity binding site for unphosphorylated TorR, whereas box 3 and box 4 are lower-affinity binding sites (33, 34). In our working model, we proposed that, first, a dimer of unphosphorylated TorR protein binds the box 1-box 2 region. Under inducing conditions, TorR is transphosphorylated and oligomerizes to form at least a tetramer. Then, two subunits of the TorR tetramer interact with box 3 and box 4 leading to the induction of tor operon expression. Alternatively, TorR∼P would first weakly bind as a dimer to box 3 and box 4, and this complex would then be stabilized by interaction with the TorR proteins previously bound to the box 1-box 2 site. In these models (34), we postulated that each TorR subunit binds one decameric direct repeat in a cooperative manner. This hypothesis is probably correct as TorR belongs to the OmpR family of response regulators (17, 21, 27, 35), and members of this family have been shown to bind cooperatively to multiple direct repeats (12, 29, 30, 38). Moreover, it has been reported that an OmpR binding site is comprised of two contiguous decameric repeats, more or less conserved, and that one OmpR molecule binds one DNA repeat (8, 9). The idea that emerges from several recent studies is that DNA binding of members of the OmpR family requires protein-protein interactions and occurs in a hierarchical manner so that high-affinity binding sites are filled first, and this binding facilitates interaction to weak binding sites by virtue of cooperativity (4, 10). Furthermore, phosphorylation of the response regulator seems to stimulate cooperative DNA binding (13).
FIG. 2.
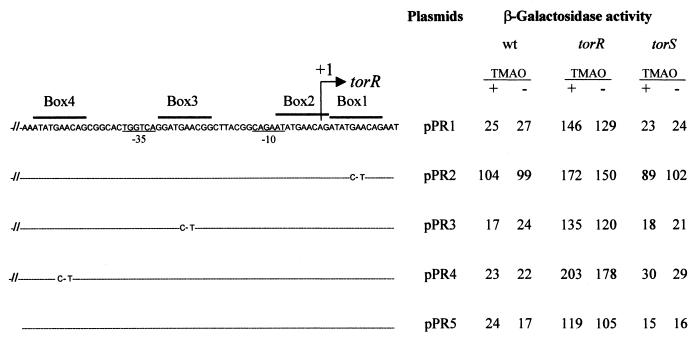
Activity of the torR promoter mutated or not mutated on tor boxes in different genetic backgrounds. (Left) The tor boxes are overlined, and the transcription start site of torR is indicated by a +1 arrow. Except for pPR5, the 5′ part of the cloned tor sequence is not shown (as indicated by −//−). The 5′ end of the cloned tor fragment corresponds to position −124, relative to the torR transcription start site for pPR1 to pPR4 and to position −53 for pPR5; the 3′ extremity of the cloned fragment for all pPR plasmids corresponds to position +15. For plasmids pPR2, pPR3, and pPR4, only the point mutations are indicated. (Right) The LCB506 (tor wild-type [wt]), LCB507 (torR), and LCB434 (torS) strains containing the pPR plasmids were grown anaerobically in the presence (+) or absence (−) of TMAO. β-Galactosidase activity of the plasmid-borne torR-lacZ fusions is expressed in Miller units.
FIG. 3.
Activity of the tor operon promoter with base pair deletions or insertions between box 2 and box 3. (Left) The tor boxes are overlined, the sequence insertions are in italics, and the transcription start site of torC is indicated by a +1 arrow. The 3′ part of the cloned tor sequence is not shown (as indicated by −//−). The 3′ end of the cloned tor fragment corresponds to position +275. (Right) The LCB506 (tor wild-type [wt]) and LCB507 (torR) strains containing the pPTor plasmids were grown anaerobically in the presence (+) or absence (−) of TMAO. β-Galactosidase activity of the plasmid-borne torC-lacZ fusions is expressed in Miller units. bp, the number of base pairs inserted (+) or deleted (−) between box 2 and box 3; ND, not determined.
As the TorR high-affinity binding site (box 1-box 2) covers the transcriptional start site of torR and overlaps its promoter −10 box (see Fig. 2), we decided to investigate whether or not the torR gene is autoregulated. We found that torR is negatively autoregulated and that full autoregulation requires an intact box 1-box 2 region. In contrast, the lower-affinity binding sites, box 3 and box 4, seem to play no role in this autogenous regulation. More surprisingly, torR autoregulation is unaffected in a torS mutant. Therefore, TorR seems to repress torR expression by binding the box 1-box 2 region even when it is unphosphorylated. Together, these results are consistent with our model in which the high-affinity binding site box 1-box 2 is bound by either the phosphorylated or the unphosphorylated form of TorR (34). In this model, the box 1-box 2 region plays the central role of a TorR oligomerization site, implying that the TorR binding site box 1-box 2 is properly positioned relative to box 3 and box 4 in order to allow the formation of a specific nucleoprotein complex able to activate tor operon transcription in inducing conditions. The effects produced by changing the spacing between box 2 and box 3 over the expression of a torC-lacZ fusion confirmed our proposal and showed that the motifs of the four boxes must be on the same side of the DNA helix. In addition, the activity of the tor operon promoter is at a maximum when the high- and low-affinity binding sites are close to each other.
MATERIALS AND METHODS
Strains, plasmids, growth conditions, and general methods.
All strains used in this study are derivatives of strain LCB506 (MC4100 pcnB). Strain LCB434 is a torS null mutant (14), and strain LCB507 was constructed by P1 transduction into strain LCB506 of a torR::mini-Tn10 allele (Cmr), obtained by random mini-Tn10 mutagenesis (1). The mini-Tn10 insertion site was determined by a rapid inverse PCR method as previously described (1), and it corresponds to position 583 relative to the torR transcription start site. Bacteria were grown on L broth medium (23) in the presence of TMAO (10 mM) where indicated. To maintain selection for plasmids or to select for transductant strains, we used antibiotics as follows: ampicillin, 50 μg · ml−1; chloramphenicol, 25 μg · ml−1; tetracycline, 25 μg · ml−1, and spectinomycin, 25 μg · ml−1. Anaerobic cultures were grown overnight without shaking at 37°C in full-cap tubes. DNA preparations were carried out with the high pure DNA isolation kit from Boehringer Mannheim. PCR amplifications and DNA restrictions were carried out using standard procedures according to the supplier's instructions. Electrotransformations were performed by the rapid method of Enderle and Farwell (5).
Primer extension analysis.
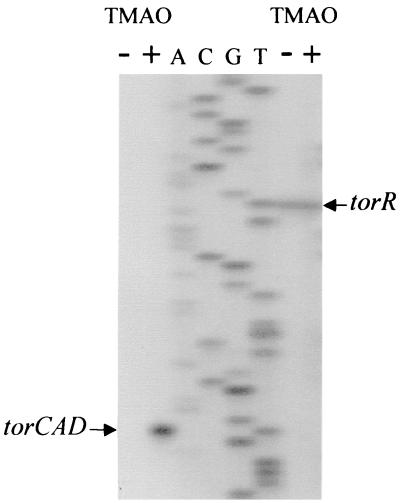
Strain MC4100 was grown anaerobically to late exponential phase with or without TMAO (10 mM). Total RNA was prepared by the hot-phenol method (23), the quality of the sample was checked electrophoretically, and quantification was done by spectroscopy. The two synthetic oligonucleotides complementary to sequences on the torR coding sequence (position 148 to 124 relative to the torR transcription start site) and the torC coding sequence (position 125 to 97 relative to the torCAD transcription start site) were end labeled with [γ-32P]ATP using T4 polynucleotide kinase (U. S. Biochemicals) and were coprecipitated with 20 μg of RNA. The same amount of primer and RNA was used in each experiment. The primer extension reaction was performed with Moloney murine leukemia virus reverse transcriptase as previously described (22, 35). A sequencing ladder was produced with a DNA template corresponding to the 5′ torC region and the oligonucleotide used for the primer extension of the torC mRNA.
Construction of plasmids.
The plasmid series pPR was created by PCR amplification of the torR promoter sequence from position −124 to position +15 for plasmids pPR1 to pPR4 and from position −53 to position +15 for pPR5, relative to the torR transcription start site, using chromosomal DNA as a template. We used mutagenic primers carrying two point mutations in box 1 (ATATGAACAG→ATATGCATAG), box 3 (GGATGAACGG→GGATGCATGG), and box 4 (ATATGAACAG→ATATGCATAG) for plasmids pPR2, pPR3, and pPR4, respectively. The PCR products, purified with Geneclean (BIO 101) and blunted using T4 DNA polymerase (Takara blunting kit), were then introduced into plasmid pGE593 (6) previously linearized with SmaI, thus placing the lacZ gene under the control of the torR promoter.
The plasmid series pPTor was created with a similar strategy by PCR amplification of the torCAD promoter sequence (from position −86 to position +276 relative to the torCAD transcription start site) and cloning into vector pGE593 (34). To create sequence insertions (pPTor28, -29, -32, and -33) or deletions (pPTor32 and -36) between box 2 and box 3, we performed PCR with insertion- or deletion-containing primers which start from the same 5′ extremity (see Fig. 3). Detailed information on the primer sequences is available from the authors on request.
All plasmids were checked by PCR with the upstream primer of the insert and a lacZ primer complementary to the lacZ sequence of pGE593 (1). The sequences of the PCR products were verified by direct sequencing with the lacZ primer.
β-Galactosidase assays.
β-Galactosidase activity was measured on whole cells by the method of Miller (23); the measures were repeated at least three times to confirm reproducibility, and the standard deviation was no more than 15%.
RESULTS AND DISCUSSION
Negative autoregulation of the torR gene.
The torR gene and the torCAD operon are divergently transcribed, and their transcriptional start sites have been previously determined by primer extension (22, 35) (for the position of the transcription start sites see Fig. 2 and 3). From these experiments, we deduced that the torR and the torCAD promoters are back-to-back and so close that no intervening DNA sequence is found between the two promoter −35 boxes. As torCAD is expressed only in the presence of TMAO under anaerobic conditions, we wondered whether the torR gene was also regulated by TMAO. To answer this question, we carried out the same primer extension experiments as previously described, but RNA was prepared from cells grown either in the presence or absence of TMAO. As shown in Fig. 1, the level of torR transcription was almost the same in the presence or absence of TMAO. In contrast and as expected, no transcription was detected for the torCAD operon in the absence of TMAO, whereas tor operon transcription was observed in the presence of inducer.
FIG. 1.
Primer extension analysis of torR and torCAD. The labeled primers were annealed to RNA from MC4100 cells grown anaerobically in the absence (− lanes) or presence (+ lanes) of 10 mM TMAO and extended with reverse transcriptase. Lanes A, C, G, and T are a sequencing ladder of the torC DNA region made with the same primer as that used in the primer extension reaction of torC.
To confirm that the torR promoter was constitutively expressed whatever the growth conditions, we constructed a hybrid plasmid in which the torR promoter region was fused to the lacZ coding sequence of the operon fusion vector pGE593. In this plasmid (pPR1), the tor DNA fragment carries the four tor boxes and extends from position −124 to position +15 relative to the torR transcription start site (+1). To avoid any artifactual effect on fusion activity due to the high copy number of the plasmid, we introduced pPR1 and the other plasmids used in this study into pcnB strains. Because of the pcnB mutation, the copy number of the plasmid remains very low in this type of strain (18). In a tor wild-type context, the torR-lacZ fusion from pPR1 was expressed at almost the same low level in the presence or absence of TMAO under anaerobic conditions (Fig. 2). This result is consistent with the primer extension analysis described above and confirms that TMAO does not affect torR expression. In addition, expression of the torR-lacZ fusion was not significantly modified under aerobic growth conditions (data not shown). Therefore, in contrast to several response regulator genes (25, 36), the torR gene seems to be expressed at the same low level whatever the growth conditions.
As the TorR response regulator mediates TMAO induction of the tor operon promoter, we thought at first that the torR gene was constitutively expressed regardless of TorR. To test this hypothesis, we introduced pPR1 into a torR strain. Surprisingly, β-galactosidase activity was five- to sixfold higher than that measured in a torR+ strain (Fig. 2). This result strongly suggests that the torR gene is negatively autoregulated, but an apparent paradox is that TorR autoregulation also takes place in the absence of TMAO (Fig. 1 and 2). An attractive possibility is that TorR can downregulate its own gene expression even when it is unphosphorylated. To clarify this last point, we decided to introduce pPR1 into a torS strain, since TorS transphosphorylates TorR in TMAO inducing conditions (14). As shown in Fig. 2, β-galactosidase activity measured in the torS strain is low whatever the growth conditions, and it is equivalent to that observed in a tor wild-type strain. TorS is thus not required for the TorR autoregulatory process. This result agrees with the fact that torR gene expression is unaffected by TMAO availability and supports the idea that TorR acts as a negative autoregulator in its phosphorylated, as well as unphosphorylated, form. Alternatively, a small amount of phosphorylated TorR might fulfil the autoregulatory function in the absence of TMAO, but this is unlikely because TorS is the only known sensor partner of TorR (1, 15).
TorR autoregulation requires only the high-affinity binding site.
As the four tor boxes are necessary for torCAD operon induction and constitute TorR DNA-binding sites (34), we supposed that the same cis-acting elements were implicated in TorR negative autoregulation as well. To test this hypothesis, we separately changed the tor boxes 1, 3, and 4 by a double substitution as previously described (34). Indeed, substitutions at conserved positions 6 and 8 of the decameric consensus sequence (AAC to CAT [Fig. 2]) in any one of the four boxes strongly decreased the activity of a torC-lacZ fusion under inducing conditions. In the present study, we decided not to mutate tor box 2 because the torR transcription start site is located within this box, and mutations close to this site might artificially affect the level of expression of the torR-lacZ fusion. Figure 2 shows that the double substitution within tor box 1 (pPR2) increased the torR-lacZ fusion activity in the tor wild-type strain about fourfold. In contrast, expression of the torR promoter remained nearly unchanged when either box 3 (pPR3) or box 4 (pPR4) was mutated (Fig. 2). These results clearly indicate that tor box 3 and box 4 are not required for torR autoregulation, whereas box 1 is essential for this process. Considering that the box 1-box 2 region constitutes a single TorR binding site (33, 34), our results strongly suggest that this high-affinity binding site alone is responsible for torR negative autoregulation.
As a control, we also introduced the plasmids pPR2, pPR3, and pPR4 into the torR and torS strains (Fig. 2). As expected, the activity of the fusion was similar in both torS and tor wild-type strains for a given plasmid. This confirms that TorS plays no role in torR autoregulation and, consequently, expression of the torR promoter also increased in a torS strain when box 1 was mutated. In a torR context, activity originating from plasmids pPR2, pPR3, or pPR4 reached levels similar to that measured from pPR1, and this activity was always higher in the torR strain than in the other two strains. Together, these results highlight the negative action of TorR on torR gene expression. The fact that activity was higher in the torR strain even for the box 1-mutated torR promoter (pPR2) (Fig. 2) indicates that the effect of the mutations in box 1 is not as strong as that of torR inactivation. This is consistent with previous findings that showed that a double mutation in either box 1 or box 2 did not entirely prevent TorR binding to the box 1-box 2 region (33, 34). Thus, TorR might still slightly downregulate its own expression by binding loosely to the mutated box 1-box 2 region.
To confirm that repression of the torR promoter only requires TorR binding to the box 1-box 2 region, we cloned a small DNA fragment carrying just the torR promoter from position −53 to +15 relative to the torR transcription start site into pGE593. As expected, expression from this fusion (pPR5) (Fig. 2) remained almost unchanged in a wild-type or torS context, whereas it increased about sixfold in a torR strain. Together, these results clearly show that TorR negative autoregulation does not require auxiliary sites in addition to the box 1-box 2 region. As the box 1-box 2 TorR binding region overlaps the RNA polymerase binding region of the torR promoter, the interaction of TorR with this region might prevent RNA polymerase binding to the torR promoter by a simple steric hindrance mechanism. This repression mechanism is probable because most repressors act by limiting the access of the RNA polymerase to the promoter, and steric hindrance is one of the classical mechanisms used by repressors to achieve their function (26, 32).
TorR high- and low-affinity binding sites must be properly positioned to each other to allow tor operon induction.
It is striking that the DNA motifs of the tor boxes are found on the same side of the DNA helix (Fig. 2). This observation is consistent with our previous proposal in which TorR binds first as a dimer to the box 1-box 2 region in its unphosphorylated form and then interacts, under inducing conditions, with the weak binding sites, boxes 3 and 4, owing to cooperative interactions stimulated by phosphorylation (34). Formation of such an active nucleoprotein complex requires that the four TorR subunits are located close to each other on the same side of the DNA helix. A similar model has been recently proposed for regulation of ompF by OmpR (13), and activation of the pstS gene involves a DNA-PhoB complex that resembles the proposed DNA-TorR complex (19). However, an 11-bp intervening sequence is found between the high- and low-affinity binding sites in the case of the tor operon promoter. To examine the role of this intervening sequence over tor operon expression and to check that the TorR binding sites must be present on the same side of the DNA helix, we decided to change the distance between box 2 and box 3.
Figure 3 summarizes the effects of small insertions or deletions within the box 2-box 3 intervening region over a plasmid-borne torC-lacZ fusion. When the intervening region was changed so that the box 1-box 2 and box 3-box 4 DNA motifs were positioned on opposite sides of the DNA helix (pPTor28, pPTor32, and pPTor36), expression of the torC-lacZ fusion was very low whatever the growth conditions. In contrast, when the box 1-box 2 and box 3-box 4 DNA motifs were positioned on the same side of the DNA helix (pPTor29, pPTor33, and pPTor34), the fusion activity increased in the presence of TMAO. These results show that the DNA motifs of the high- and low-affinity binding sites must be positioned on the same face of the DNA helix to allow tor operon induction. Therefore, the TorR subunits activate tor operon expression by binding to the same side of the DNA helix.
Although a proper phasing between the tor boxes seems to be essential for tor operon expression, the distance between the high- and low-affinity binding sites appears to play an important role in the strength of the tor operon promoter. Indeed, insertion of one (pPTor29) or two (pPTor33) additional helical turns between box 2 and box 3 led to a strong decrease in torC-lacZ fusion activity under inducing conditions (Fig. 3). Furthermore, deletion of the box 2-box 3 intervening region (pPTor34) resulted in an even higher fusion activity than that of the wild-type promoter fusion. From this experiment, we conclude that the closer the high- and low-affinity binding sites are, the higher tor operon expression is. Finally, we introduced the same plasmids into a torR strain and, as expected, no activity above the background level was measured (Fig. 3).
Concluding remarks.
In the present study, we show that the TorR response regulator is not only an activator of the tor structural operon but also a repressor of its own gene expression (20, 32). The torR gene and the tor operon are divergently transcribed, and our data indicate that the box 1-box 2 TorR high-affinity binding site, located within the torR-torC intergenic region, is absolutely required for both repression of torR and activation of torCAD. The fact that binding of TorR to the weaker binding sites of box 3 and box 4, which are essential for tor operon expression, does not enhance torR repression strongly suggests that in contrast to many repressors (26, 32) TorR does not need to bind auxiliary binding sites to achieve full repression. In addition, TorR seems to bind the box 1-box 2 region in vivo in its unphosphorylated form, since torR negative autoregulation depends on neither the TorS sensor partner nor the presence of TMAO (Fig. 1 and 2). Accordingly, torR negative autoregulation is likely to maintain TorR concentration at a low level whatever the growth conditions.
Although the box 2-box 3 intervening sequence is essential for torR gene expression, as it is located within the torR promoter and carries part of the torR promoter −10 box, this DNA region proved to be dispensable not only for TMAO induction of the tor operon (Fig. 3) but also for the anaerobic control of the same operon (data not shown). However, tor operon induction requires strict spacing between the high- and low-affinity binding sites, so that the repeat sequences of the four tor boxes are oriented to the same face of the DNA helix. This further supports our previous model concerning the formation of an active nucleoprotein complex in which a TorR∼P tetramer binds the four tor boxes simultaneously and probably bends the tor regulatory region (34).
To extend our model, we propose now that RNA polymerase binds the torR promoter on the same side as TorR. Therefore, repression efficiency might depend mainly on the competition between TorR and the RNA polymerase for their overlapping binding sites. If this is true, then transcription of the torR gene should occur only in the absence of TorR binding, and as expression of the torCAD operon is strictly TorR-dependent, transcription initiations of torR and torCAD might be mutually exclusive. Additional experiments are required to better understand this complex regulatory process. It also remains to be answered how TorR activates transcription of the tor operon and whether or not the RNA polymerase that binds the tor operon promoter is positioned on the same side as TorR (11, 31).
ACKNOWLEDGMENTS
We thank C. Iobbi-Nivol, C. Jourlin-Castelli, and M. C. Pascal for helpful discussions. We are grateful to D. Cazeilles for technical assistance and to S. Wells for critical reading of the manuscript.
This work was supported by grants from the Centre National de la Recherche Scientifique, the Université de la Méditerranée, and the MENRT (Programme de Recherche Fondamentale en Microbiologie et Maladies Infectieuses et Parasitaires). M.A. was supported by grants from the MENRT and from the Fondation pour la Recherche Médicale (FRM).
REFERENCES
- 1.Ansaldi M, Bordi C, Lepelletier M, Méjean V. TorC apocytochrome negatively autoregulates the trimethylamine N-oxide (TMAO) reductase operon in Escherichia coli. Mol Microbiol. 1999;33:284–295. doi: 10.1046/j.1365-2958.1999.01468.x. [DOI] [PubMed] [Google Scholar]
- 2.Barrett E L, Kwan H S. Bacterial reduction of trimethylamine oxide. Annu Rev Microbiol. 1985;39:131–149. doi: 10.1146/annurev.mi.39.100185.001023. [DOI] [PubMed] [Google Scholar]
- 3.Dos Santos J P, Iobbi-Nivol C, Couillault C, Giordano G, Méjean V. Molecular analysis of the trimethylamine N-oxide (TMAO) reductase respiratory system from a Shewanella species. J Mol Biol. 1998;284:421–433. doi: 10.1006/jmbi.1998.2155. [DOI] [PubMed] [Google Scholar]
- 4.Eder S, Liu W, Hulett F M. Mutational analysis of the phoD promoter in Bacillus subtilis: implications for PhoP binding and promoter activation of Pho regulon promoters. J Bacteriol. 1999;181:2017–2025. doi: 10.1128/jb.181.7.2017-2025.1999. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Enderle P J, Farwell M A. Electroporation of freshly plated Escherichia coli and Pseudomonas aeruginosa cells. BioTechniques. 1998;25:954–958. doi: 10.2144/98256bm05. [DOI] [PubMed] [Google Scholar]
- 6.Eraso J M, Weinstock G M. Anaerobic control of colicin E1 production. J Bacteriol. 1992;174:5101–5109. doi: 10.1128/jb.174.15.5101-5109.1992. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Goudreau P N, Stock A M. Signal tranduction in bacteria: molecular mechanisms of stimulus-response coupling. Curr Opin Microbiol. 1998;2:160–169. doi: 10.1016/s1369-5274(98)80006-4. [DOI] [PubMed] [Google Scholar]
- 8.Harlocker S L, Bergstrom L, Inouye M. Tandem binding of six OmpR proteins to the ompF upstream regulatory sequence of Escherichia coli. J Biol Chem. 1995;270:26849–26856. doi: 10.1074/jbc.270.45.26849. [DOI] [PubMed] [Google Scholar]
- 9.Harrison-McMonagle P, Denissova N, Martinez-Hackert E, Ebright R H, Stock A M. Orientation of OmpR monomers within an OmpR:DNA complex determined by DNA affinity cleaving. J Mol Biol. 1999;285:555–566. doi: 10.1006/jmbi.1998.2375. [DOI] [PubMed] [Google Scholar]
- 10.Head C G, Tardy A, Kenney L J. Relative binding affinities of OmpR and OmpR-phosphate at the ompF and ompC regulatory sites. J Mol Biol. 1998;281:857–870. doi: 10.1006/jmbi.1998.1985. [DOI] [PubMed] [Google Scholar]
- 11.Hochschild A, Dove S L. Protein-protein contacts that activate and repress prokaryotic transcription. Cell. 1998;92:597–600. doi: 10.1016/s0092-8674(00)81126-5. [DOI] [PubMed] [Google Scholar]
- 12.Huang K J, Igo M M. Identification of the bases in the ompF regulatory region, which interact with the transcription factor OmpR. J Mol Biol. 1996;262:615–628. doi: 10.1006/jmbi.1996.0540. [DOI] [PubMed] [Google Scholar]
- 13.Huang K J, Lan C Y, Igo M M. Phosphorylation stimulates the cooperative DNA-binding properties of the transcription factor OmpR. Proc Natl Acad Sci USA. 1997;94:2828–2832. doi: 10.1073/pnas.94.7.2828. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Jourlin C, Ansaldi M, Méjean V. Transphosphorylation of the TorR response regulator requires the three phosphorylation sites of the TorS unorthodox sensor in Escherichia coli. J Mol Biol. 1997;267:770–777. doi: 10.1006/jmbi.1997.0919. [DOI] [PubMed] [Google Scholar]
- 15.Jourlin C, Bengrine A, Chippaux M, Méjean V. An unorthodox sensor protein (TorS) mediates the induction of the tor structural genes in response to trimethylamine N-oxide in Escherichia coli. Mol Microbiol. 1996;20:1297–1306. doi: 10.1111/j.1365-2958.1996.tb02648.x. [DOI] [PubMed] [Google Scholar]
- 16.Jourlin C, Simon G, Pommier J, Chippaux M, Méjean V. The periplasmic TorT protein is required for trimethylamine N-oxide reductase gene induction in Escherichia coli. J Bacteriol. 1996;178:1219–1223. doi: 10.1128/jb.178.4.1219-1223.1996. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Kondo H, Nakagawa A, Nishihira J, Nishimura Y, Mizuno T, Tanaka I. Escherichia coli positive regulator OmpR has a large loop structure at the putative RNA polymerase interaction site. Nat Struct Biol. 1997;4:28–31. doi: 10.1038/nsb0197-28. [DOI] [PubMed] [Google Scholar]
- 18.Lopilato J, Bortner S, Beckwith J. Mutations in a new chromosomal gene of Escherichia coli K-12, pcnB, reduce plasmid copy number of pBR322 and its derivatives. Mol Gen Genet. 1986;205:285–290. doi: 10.1007/BF00430440. [DOI] [PubMed] [Google Scholar]
- 19.Makino K, Amemura M, Kawamoto T, Kimura S, Shinagawa H, Nakata A, Suzuki M. DNA binding of PhoB and its interaction with RNA polymerase. J Mol Biol. 1996;259:15–26. doi: 10.1006/jmbi.1996.0298. [DOI] [PubMed] [Google Scholar]
- 20.Maloy S, Stewart V. Autogenous regulation of gene expression. J Bacteriol. 1993;175:307–316. doi: 10.1128/jb.175.2.307-316.1993. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Martinez-Hackert E, Stock A M. Structural relationships in the OmpR family of winged-helix transcription factors. J Mol Biol. 1997;269:301–312. doi: 10.1006/jmbi.1997.1065. [DOI] [PubMed] [Google Scholar]
- 22.Méjean V, Iobbi-Nivol C, Lepelletier M, Giordano G, Chippaux M, Pascal M C. TMAO anaerobic respiration in Escherichia coli: involvement of the tor operon. Mol Microbiol. 1994;11:1169–1179. doi: 10.1111/j.1365-2958.1994.tb00393.x. [DOI] [PubMed] [Google Scholar]
- 23.Miller J. Experiments in molecular genetics. Cold Spring Harbor, N.Y: Cold Spring Harbor Laboratory; 1972. [Google Scholar]
- 24.Mizuno T. Compilation of all genes encoding two-component phosphotransfer signal transducers in the genome of Escherichia coli. DNA Res. 1997;4:161–168. doi: 10.1093/dnares/4.2.161. [DOI] [PubMed] [Google Scholar]
- 25.Mouncey N J, Kaplan S. Cascade regulation of dimethyl sulfoxide reductase (dor) gene expression in the facultative phototroph Rhodobacter sphaeroides 2.4.1T. J Bacteriol. 1998;180:2924–2930. doi: 10.1128/jb.180.11.2924-2930.1998. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Müller-Hill B. Some repressors of bacterial transcription. Curr Opin Microbiol. 1998;1:145–151. doi: 10.1016/s1369-5274(98)80004-0. [DOI] [PubMed] [Google Scholar]
- 27.Parkinson J S, Kofoid E C. Communication modules in bacterial signaling proteins. Annu Rev Genet. 1992;26:71–112. doi: 10.1146/annurev.ge.26.120192.000443. [DOI] [PubMed] [Google Scholar]
- 28.Pommier J, Méjean V, Giordano G, Iobbi-Nivol C. TorD, a cytoplasmic chaperone that interacts with the unfolded trimethylamine N-oxide reductase enzyme (TorA) in Escherichia coli. J Biol Chem. 1998;273:16615–16620. doi: 10.1074/jbc.273.26.16615. [DOI] [PubMed] [Google Scholar]
- 29.Pratt L A, Silhavy T J. Porin regulon of Escherichia coli. In: Hoch J A, Silhavy T J, editors. Two-component signal transduction. Washington, D.C.: ASM Press; 1995. pp. 105–127. [Google Scholar]
- 30.Rampersaud A, Harlocker S L, Inouye M. The OmpR protein of Escherichia coli binds to sites in the ompF promoter region in a hierarchical manner determined by its degree of phosphorylation. J Biol Chem. 1994;269:12559–12566. [PubMed] [Google Scholar]
- 31.Rhodius V A, Busby S J. Positive activation of gene expression. Curr Opin Microbiol. 1998;1:152–159. doi: 10.1016/s1369-5274(98)80005-2. [DOI] [PubMed] [Google Scholar]
- 32.Rojo F. Repression of transcription initiation in bacteria. J Bacteriol. 1999;181:2987–2991. doi: 10.1128/jb.181.10.2987-2991.1999. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Simon G. Régulation de l'expression de la triméthylamine N-oxyde réductase chez Escherichia coli: étude du régulateur de réponse, TorR, et de son interaction avec la région promotrice de l'opéron torCAD. Ph.D. thesis. Marseille, France: Université de la Méditerranée; 1996. [Google Scholar]
- 34.Simon G, Jourlin C, Ansaldi M, Pascal M C, Chippaux M, Méjean V. Binding of the TorR regulator to cis-acting direct repeats activates tor operon expression. Mol Microbiol. 1995;17:971–980. doi: 10.1111/j.1365-2958.1995.mmi_17050971.x. [DOI] [PubMed] [Google Scholar]
- 35.Simon G, Méjean V, Jourlin C, Chippaux M, Pascal M C. The torR gene of Escherichia coli encodes a response regulator protein involved in the expression of the trimethylamine N-oxide reductase. J Bacteriol. 1994;176:5601–5606. doi: 10.1128/jb.176.18.5601-5606.1994. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Soncini F C, Vescovi E G, Groisman E A. Transcriptional autoregulation of the Salmonella typhimurium phoPQ operon. J Bacteriol. 1995;177:4364–4371. doi: 10.1128/jb.177.15.4364-4371.1995. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Wang A, Bolen D W. A naturally occurring protective system in urea-rich cells: mechanism of osmolyte protection of proteins against urea denaturation. Biochemistry. 1997;36:9101–9108. doi: 10.1021/bi970247h. [DOI] [PubMed] [Google Scholar]
- 38.Wietzorrek A, Bibb M. A novel family of proteins that regulates antibiotic production in streptomycetes appears to contain an OmpR-like DNA-binding fold. Mol Microbiol. 1997;25:1181–1184. doi: 10.1046/j.1365-2958.1997.5421903.x. [DOI] [PubMed] [Google Scholar]