Abstract
A 10-kb DNA region of the cyanobacterium Anabaena variabilis ATCC 29413 containing the structural genes of the uptake hydrogenase (hupSL) was cloned and sequenced. In contrast to the hupL gene of Anabaena sp. strain PCC 7120, which is interrupted by a 10.5-kb DNA fragment in vegetative cells, there is no programmed rearrangement within the hupL gene during the heterocyst differentiation of A. variabilis. The hupSL genes were transcribed as a 2.7-kb operon and were induced only under nitrogen-fixing conditions, as shown by Northern blot experiments and reverse transcriptase PCR. Primer extension experiments with a fluorescence-labeled oligonucleotide primer confirmed these results and identified the 5′ start of the mRNA transcript 103 bp upstream of the ATG initiation codon. A consensus sequence in the promoter that is recognized by the fumarate nitrate reductase regulator (Fnr) could be detected. The hupSL operon in A. variabilis was interrupted by an interposon deletion (mutant strain AVM13). Under N2-fixing conditions, the mutant strain exhibited significantly increased rates in H2 accumulation and produced three times more hydrogen than the wild type. These results indicate that the uptake hydrogenase is catalytically active in the wild type and that the enzyme reoxidizes the H2 developed by the nitrogenase. The Nif phenotype of the mutant strain showed a slight decrease of acetylene reduction compared to that of the wild type.
The only microorganisms with an O2-producing photosynthesis that have a hydrogen metabolism are cyanobacteria and green algae (23, 25). In cyanobacteria, up to three enzymes can be involved in hydrogen metabolism: the nitrogenase which produces H2 during nitrogen fixation (24), the membrane-bound hydrogenase which reoxidizes the H2 (10), and the bidirectional hydrogenase catalyzing both oxidation of molecular hydrogen and reduction of protons (38). In cyanobacteria, the genetics of bidirectional hydrogenases are especially well characterized (3, 7). But, the H2 production in filamentous cyanobacteria during the reduction of nitrogen to NH3 is mainly catalyzed by the nitrogenase in the heterocysts. The Anabaena cells can oxidize the hydrogen with the uptake hydrogenase via the oxyhydrogen (Knallgas) reaction. It was suggested that the organism gets additional ATP while the Knallgas reaction can protect the O2-sensitive nitrogenase by removing the oxygen in the heterocysts (30).
Hydrogenases have been described for a large number of microorganisms and studied intensively in diverse phylogenetic groups of bacteria (22, 34, 54). The uptake hydrogenases are membrane-bound enzymes which consist of two subunits with [Fe-S] clusters as prosthetic groups. The large subunit, HupL, carries additionally a Ni atom in the active center. In most of the bacterial families, the hupSL genes are clustered in an operon in which hupS is located upstream of the hupL gene (20, 51, 53). Recently, some hydrogenase sequences from filamentous cyanobacteria were published (10, 31, 38). The uptake hydrogenase in the heterocyst-forming organism Anabaena sp. strain PCC 7120 is interrupted by a 10.5-kb element. Under nitrogen-fixing conditions, this fragment is excised by a site-specific recombinase that is encoded inside the right border of the hupL element (10). This rearrangement is not found in the hupSL genes of Nostoc sp. strain PCC 73102 (31).
In the present study, we isolated and characterized the hup gene region in Anabaena variabilis ATCC 29413. In contrast to the best characterized filamentous cyanobacterium Anabaena sp. strain PCC 7120, the closely related strain A. variabilis has some interesting features. Heterocyst differentiation in Anabaena sp. strain PCC 7120 is accompanied by developmentally regulated genome rearrangements that affect fdxN, nifD, and hupL gene expression (9, 10). A. variabilis does not contain the fdxN element (6), and in this study we show that the hupL gene is also not rearranged in A. variabilis.
In order to analyze the function of the uptake hydrogenase in nitrogen and hydrogen metabolism, we constructed a hupSL deletion mutant by the insertion of an interposon in the hupSL operon. Physiological studies to compare the mutant phenotype with that of the wild type were carried out. The transcriptional regulation of the hupSL genes was investigated by Northern analysis and reverse transcriptase PCR. Further transcriptional investigations were made by determining the 5′ end of the mRNA by the primer extension technique and analyzing the promoter region.
MATERIALS AND METHODS
Bacterial strains and growth conditions.
Bacterial strains and plasmids are listed in Table 1. Cells of the N2-fixing cyanobacterium A. variabilis ATCC 29413 were grown either in BG11, BG110 (12), or BG110 medium supplemented with 5 mM NH4Cl and 10 mM TES [N-tris (hydroxymethyl)methyl-2-aminoethane sulfonic acid] under continuous irradiance of 100 μmol m−2 s−1 and bubbled with air enriched with CO2 to 1% (vol/vol) at 30°C. To induce heterocyst formation, the cultures were pelleted, washed twice with BG110, and grown in BG110 for 24 h.
TABLE 1.
Bacterial strains and plasmids used in this studya
| Strain or plasmid | Relevant characteristic(s) | Reference(s) or source |
|---|---|---|
| Strains | ||
| E. coli | ||
| MC1061 | araD139 Δ(ara-leu)7696 Δ(lac)L74 galU galK hsdR2(rK− mK+) mcrB1 | 11 |
| HB101 | F−supE44 hsdS20 recA13 ara-14 proA2 lacY1 galK2 rpsL20 xyl-5 mtl-1 | 18 |
| A. variabilis | ||
| FD | Temperature-resistant wild type | 14 |
| AVM13 | A. variabilis hupSL::npt | This study |
| Plasmids | ||
| pUC18, pUC19 | Cloning vectors, Apr, mcs | 44, 55 |
| pBluescript SK(−) | Cloning vector, Apr, mcs | 40 |
| pSUP202 | Mobilizable plasmid, Apr, Cmr, Tcr | 41 |
| pRL443 | Conjugal plasmid, Apr, Tcr, Kms, derivative of RP4 | 19 |
| pRL623 | Helper plasmid, MobColK, M.AvaI, M.Eco4711, M.EcoT221, Apr, Cmr | 19 |
| pRL648 | Source of C.K3 cassette encoding Nmr | 18 |
| pBMB19 | Plasmid carrying the bom site in pUC18, Apr | 45 |
| pKS1 | 4.8-kb HindIII fragment carrying A. variabilis hupSL cloned in pBluescript | This study |
| pKS4I/II | Nmr gene replacing 2.1-kb XbaI fragment of pKS1 | This study |
| pKS13 | 0.7-kb XbaI-HindIII fragment, subclone of pKS1 with 3′ end of hupL | This study |
| pKS14 | 0.9-kb XbaI-HincII fragment, subclone of pKS1 with part of hupS | This study |
| pKS15 | 0.3-kb HincII-XbaI fragment, subclone of pKS1 with 5′ end of hupS | This study |
| pKS17 | 0.5-kb fragment containing part of hupL amplified by inverse PCR | This study |
| pKS18 | 4.8-kb XbaI fragment carrying A. variabilis hupL downstream region | This study |
| pAVM13 | pSUP202 containing a 3.7-kb HindIII fragment carrying hupSL::Nmr | This study |
| pIF1 | 0.8-kb HindIII-Asp700 fragment carrying part of hupL of Anabaena sp. strain PCC 7120 | This study |
| pTS1 | 0.6-kb Asp700-HindIII fragment carrying part of hupL of Anabaena sp. strain PCC 7120 | This study |
Ap, ampicillin; Cm, chloramphenicol; Km, kanamycin; Nm, neomycin; Tc, tetracycline.
For deletion mutagenesis, a wild-type strain of A. variabilis with a spontaneous mutation (FD strain) which also grows at 40°C was used. The mutant AVM13 was grown in BG11 medium supplemented with 50 μg of neomycin per ml. The growth conditions, media, and antibiotic concentrations for Escherichia coli strains were described elsewhere (47).
Nucleic acid isolation.
Genomic DNA of the A. variabilis wild type and the mutant was isolated according to the method of Smoker and Barnum (42). Plasmid DNA was obtained by standard techniques (36). Total cellular RNA was isolated from 200-ml cultures grown in different media (BG11, BG110, and NH4+). The cells were disrupted with glass beads (450 microns), followed by organic extraction and ethanol precipitation. To remove contaminating DNA and proteins, a cesium-chloride gradient ultracentrifugation purification step (4) was done. The concentration of RNA was determined by measuring the absorbance at 260 nm.
Hybridization.
For Southern blot hybridization analysis, chromosomal DNA of A. variabilis and the exconjugants, as well as the plasmid DNA from the clones of the partial libraries, was isolated and cut with appropriate restriction enzymes. Following separation by electrophoresis in 1% agarose gels, the DNA was transferred to Hybond N nylon membranes (Amersham Pharmacia Biotech) by the capillary method described by Sambrook et al. (36). Prehybridization, overnight hybridization, and washing steps were carried out at 58 to 62°C. Labeling of probes and detection of hybridization signals were performed with the DIG Labeling and Detection kit (Roche Diagnostics, Boehringer Mannheim).
Northern blot experiments were carried out by separating total RNA of the different cultures in 1% denaturing formaldehyde agarose gels and transferring to Hybond N nylon. pKS14 was labeled with digoxigenin-UTP by in vitro transcription with the T7-RNA-polymerase (Roche Diagnostics, Boehringer Mannheim). RNA-RNA hybridization was performed overnight at 65°C in DIG Easy Hyb solution.
Screening of partial gene banks and sequencing.
Genomic DNA of A. variabilis was digested by different restriction endonucleases. After separation on a 1% agarose gel, HindIII and XbaI fragments between 4.5 and 5.0 kb were eluted out of the gel pieces. The extracted gel fragments were ligated to HindIII- and XbaI-linearized and -dephosphorylated pBluescript SK(−) vectors and transformed into competent E. coli MC1061 cells by the CaCl2 method (13; modified by reference 36). A 0.8-kb HindIII-Asp700 fragment (pIF1) and a 0.6-kb Asp700-HindIII fragment (pTS1), obtained from the plasmid pAM1311 (10), were digoxigenin-labeled and used for Southern blot experiments with the clones of the partial libraries.
The plasmids pKS1 and pKS18 were treated with exonuclease III and S1 nuclease of an Erase-a-Base kit (Promega). Following ligation and transformation, the deletion clones were analyzed by restriction. Sequencing was then performed by the dideoxy chain termination method (37).
PCR and primer extension analysis.
The 5′ end of the uptake hydrogenase mRNA was determined by a primer extension protocol (29) with fluorescence-labeled oligonucleotides and an automated DNA sequencer (2). The synthetic cy5-labeled oligonucleotide HupS7 (5′-Cy5-CGCATACTGTCGGTTCTTCGGC-3′) was a 22-mer complementary to the bases 85 to 64 downstream of the translation start codon ATG of the hupS gene.
Primer extension mixtures included 10 to 20 μg of RNA of the different preparations, 10 pmol of the HupS7 oligonucleotide, 50 U of RNasin Ribonuclease Inhibitor, 1 mM concentrations of deoxynucleoside triphosphates, and 5 U of avian myeloblastosis virus reverse transcriptase. The mixtures were incubated at 42°C for 1 h. The dideoxy sequencing reaction using a clone containing the 5′ region of the hupS gene (pKS15) and the same HupS7 primer was performed according to the instructions of the cy5-Auto Read Sequencing kit (Amersham Pharmacia Biotech). The primer extension product and the sequence ladder were loaded together onto a gel of the A.L.F. Express DNA Sequencer, and the transcription start point was determined by comparing their retention times.
Inverse PCR as described by Pang and Knecht (32) was used to determine the flanking sequences of the 3′ end of the hupL gene. Two primers which were complementary to bases 1230 to 1310 (HupL2 [5′-CGCTTGGCGATATAACTTGA-3′]) and identical to bases 1337 to 1362 (HupL3 [5′-GTCACTGGATAGATATCGAAGGTGGC-3′]) of the hupL gene were selected. These primers, facing outwards from the known DNA sequence, were used to carry out PCR with genomic DNA from A. variabilis.
Constructions of plasmids and conjugative plasmid transfer.
Different cargoplasmids were constructed and then transferred from E. coli HB101 to A. variabilis. A 2.15-kb XbaI fragment containing the hupS gene and part of the hupL gene was removed from the plasmid pKS1 by replacing it with a 1.1-kb neomycin resistance cassette from C.K3, yielding pKS4. C.K3 contains the npt gene from Tn5 with a promoter from the psbA gene of Amaranthus hybridus (18). pAVM13 was constructed by inserting the resulting 3.77-kb HindIII fragment from pKS4 in pSUP202 (41).
The resulting cargoplasmid pAVM13 was introduced from E. coli cells into the A. variabilis FD strain via triparental mating (using the conjugal plasmid pRL443 and the helper plasmid pRL623 for mobilization). The conjugative plasmid transfer was performed according to the method of Elhai et al. (19). Instead of the normal wild-type strain, a spontaneous mutant (FD strain) which grows at 40°C was used for triparental mating. Exconjugants were selected by plating on BG11 plates containing 50 μg of neomycin per ml. Nmr exconjugants were picked and grown in liquid medium, one added with neomycin and one added with ampicillin, to select the Amps exconjugants which have made the marker rescue via double crossover. One ampicillin-sensitive mutant chosen for further studies was designated AVM13. Insertion of the neomycin resistance cassette and complete segregation in AVM13 was shown by Southern hybridization.
Determination of nitrogenase and hydrogenase activity.
In vivo nitrogenase activity was measured by acetylene reduction assay (17) using a Hewlett-Packard gas chromatograph Model 5890 Series II with a 6-ft Porapak N80/100 column for acetylene-ethylene separation and a flame ionization detector. The specific nitrogenase activity was expressed as follows: nanomoles of ethylene (C2H4)/micrograms of chlorophyll α × h. The developed H2 was quantified in a gas chromatograph (Hewlett-Packard Model 5890 Series II) equipped with a thermal conductivity detector and a molecular sieve column and expressed as follows: nanomoles of H2/nanograms of chlorophyll α × h. The hydrogen uptake was measured during incubation at 25°C by the decrease of a known amount of H2 (2 ml of 0.3% H2–99.7% argon) added to the probe.
Nucleotide sequence accession number.
The nucleotide sequence data reported in this study is available from the EMBL, GenBank, and DDBJ nucleotide sequence databases under accession no. Y13216.
RESULTS
Isolation, characterization, and sequence comparison of the hupSL region in A. variabilis ATCC 29413.
To identify the hup gene region in A. variabilis ATCC 29413, the hupSL genes were cloned by partial genomic library screening using different fragments within the hupL gene of Anabaena sp. strain PCC 7120 as probes in Southern hybridization experiments (see Materials and Methods). First, a HindIII (pKS1) clone with a 4,824-bp insert was isolated. This nucleotide sequence has two open reading frames (ORFs) that show over 90% homology to the hupSL genes of Anabaena sp. strain PCC 7120. As a result, no stop codon was found at the 3′ end of the hupL gene, indicating that hupL was only partially encompassed by the insert of pKS1.
A second partial gene bank was constructed to obtain the remaining part of hupL. After screening with a DNA probe (pTS1) containing the downstream region of the Anabaena sp. strain PCC 7120 hupL gene, one positive clone (pKS18) which contained the 120 bp of the 3′ end of the A. variabilis hupL gene was obtained. The missing 30 bp of the hupL gene were then identified by inverse PCR (32) using two inverted oligonucleotide primers which were designed from within the hupL sequence of the HindIII fragment. Both strands of the inserts, which contain the complete sequence of the hupSL gene region, were determined (data not shown). Together, the nucleotide sequence revealed 10 complete ORFs and one partial ORF (Fig. 1). Comparisons of their deduced amino acid and DNA sequences with known sequences in databases were performed (1, 21).
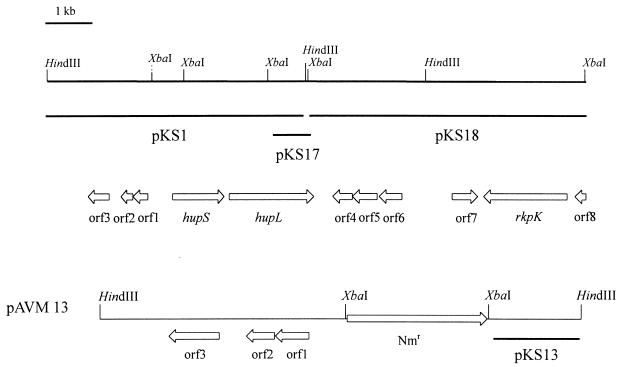
FIG. 1.
Physical and genetical maps of the 10-kb hupSL gene region of A. variabilis. The physical maps are for the enzymes HindIII and XbaI. The region is divided into three subclones, marked with horizontal lines. The organization of the genes is indicated by arrows under the restriction map; ORF8 has been only partially sequenced. hupSL code for the two subunits of the uptake hydrogenase; rkpK probably encodes the UDP-glucose dehydrogenase. The gene arrangement of the cargoplasmid pAVM13 with the integrated neomycin resistance cassette (Nmr) is shown. Note that pAVM13 is drawn on a different scale.
Two ORFs encode the small and large subunits of the uptake hydrogenase (hupSL). The proteins consist of 321 and 531 amino acid residues with calculated molecular masses of 35.1 and 61.9 kDa, respectively. Comparisons of the deduced amino acid sequences of hupS and hupL gene products with the HupS and HupL proteins of the filamentous cyanobacteria Anabaena sp. strain PCC 7120 and Nostoc sp. strain 73102 revealed very high similarities. Both subunits in A. variabilis showed more than 95% identity with the homologous region from Anabaena sp. strain PCC 7120. This is not surprising for these closely related organisms since 90% and higher similarities are also known for the nifHDK cluster (95% nucleotide identity) (6) and the ndh genes (93% nucleotide identity; T. Happe, personal communication). The intergenic region between hupS and hupL in A. variabilis consists of 76 bp and is only 60% homologous to the intergenic DNA sequence (54 bp) of Anabaena sp. strain PCC 7120. Unlike the hupL gene of Anabaena sp. strain PCC 7120, the hupSL operon in vegetative cells of A. variabilis is not interrupted by a 10.5-kb element. In Anabaena sp. strain PCC 7120, this DNA fragment is excised during heterocyst differentiation. Southern hybridizations with different DNA probes support the sequence data of a contiguous hupSL operon in A. variabilis.
The three ORFs upstream of the hupSL genes (Fig. 1) encode products that showed no similarity to any known protein. They probably do not belong to the hupSL operon because they are transcribed in the opposite direction. Also, no homologies were found for ORF4, ORF5, and ORF6. Interestingly, one (ORF7, 159 amino acids encoded) showed about 44% amino acid identity to a regulator protein of Enterococcus faecalis. One ORF (453 amino acids) 2 kb downstream of the hupL gene, showed homology to a UDP-glucose dehydrogenase of Synechocystis sp. strain PCC 6803 (46% identity) and Sinorhizobium meliloti (43% identity). In S. meliloti, the dehydrogenase catalyzes the reaction from UDP-glucose to UDP-glucuronic acid and is involved in the synthesis of capsular polysaccharides. The partial ORF at the 3′ end of the XbaI clone demonstrated homology to the C-terminal end of a UDP-glucose dehydratase from Synechocystis sp. strain 6803 (48% identity).
Transcriptional analysis.
Northern blot analysis was performed to examine the expression of the hupSL operon in N2-fixing and non-N2-fixing cultures. The Northern blot was hybridized with a digoxigenin-labeled probe (pKS14) from within the hupS gene. The transcript was only detected in RNA isolated from N2-fixing cultures (Fig. 2). Using an RNA marker, the size of the hupSL operon was determined to be 2.7 kb. Transcripts were completely missing in cells grown in medium supplemented with either ammonia or nitrate. The results of the Northern analysis were in good agreement with the results made by RT-PCR using total RNA from BG11 and BG110 cultures (data not shown). These results indicate that the A. variabilis hupSL operon is either nitrogen regulated or induced by hydrogen. In the cyanobacterium Nostoc sp. strain PCC 73102, the hupL transcript is also induced during heterocyst differentiation (5).
FIG. 2.
RNA gel blot analysis of the hupSL transcript. Hybridization of the hupS gene containing a 950-bp HincII fragment to total RNA (10 μg each) isolated from cells grown with NO3− (1), NH4+ (3) in the medium and under nitrogen-fixing conditions (2). The locations of the RNA size markers are shown.
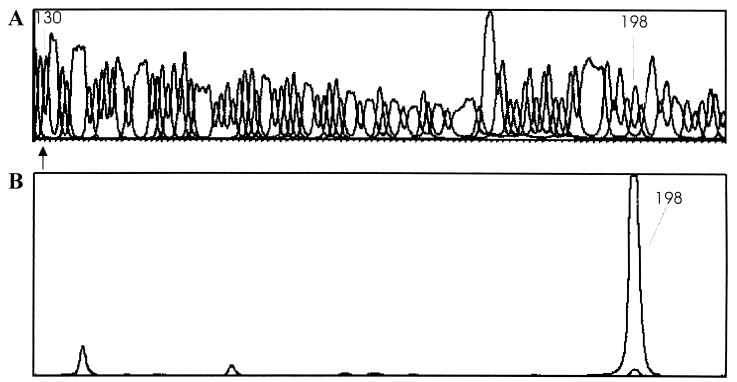
The 5′ end of the hupSL mRNA was determined by means of the primer extension technique with a fluorescence-labeled oligonucleotide primer and an automated DNA sequencer. Performing the extension reaction with RNA isolated under N2-limiting conditions, a major extension product was detected. By comparing the retention time of this product with the signal in the sequence chromatogram (Fig. 3), the first transcribed nucleotide, a thymidine, was located 103 nucleotides upstream of the translational start codon ATG of the hupS gene (Fig. 4). No primer extension product was found with RNA isolated from BG11 cultures (Fig. 3). This provides further evidence that the hupSL operon is expressed only under N2-fixing conditions. Fifteen base pairs upstream of the transcription initiation, a sequence that resembles a −10 consensus region (TAAACT) of the hup operon in E. coli (26) was found. Half of a sequence motif identical to the consensus Fnr-binding sequence 144 bp upstream of the transcription start site (Fig. 4) was also found. In addition, a fivefold direct repeat of the sequence TA/GACAAC upstream of the ATG start codon was obvious to identify.
FIG. 3.
Localization of the transcription start point of the hupSL genes. (A) Nucleotide sequence of the pKS1 clone containing the promoter region of the hupSL operon. Arrow indicates the start codon in the genome sequence. (B) Primer extension products obtained with RNA isolated from BG11 and BG110 cultures as the template and the oligonucleotide HupS7 as the primer. The retention times of the start codon and of the major extension product are shown. All signals result from the BG110 RNA except for the smaller peak at retention time 198.
FIG. 4.
Nucleotide sequence of the hupSL promoter region. The start codon of hupS is in bold; the N-terminal end of the amino acid sequence is written under the nucleotide sequence. Shine-Dalgarno homologies are underlined. The transcription start point is given by +1. The repetitive elements in the promoter sequence are boxed. The primer HupS7, used for primer extension, is also shown. The putative −10 consensus sequence and the Fnr-binding site are underlined and in bold.
Construction of a hupSL mutant and physiological characterization.
To investigate the biological properties of the hupSL genes, a mutant strain (AVM13) in which the complete hupS gene and the 5′ end of the hupL gene were deleted was constructed. A 2.15-kb XbaI fragment from within the plasmid pKS1 (4.8-kb HindIII fragment) was replaced by the selectable marker gene npt (Fig. 1).
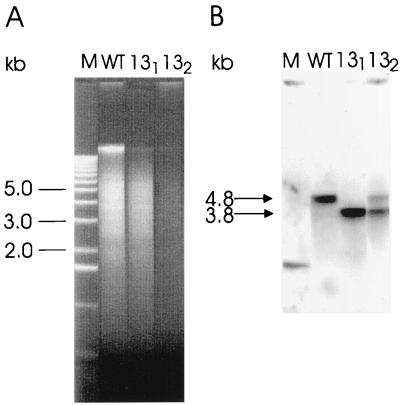
Total DNA from two homocygote recombinant clones (131 and 132) was prepared, and marker rescue and complete segregation was confirmed by Southern hybridization using a 715-bp fragment from within the hupL gene as a probe (pKS13) (Fig. 1 and 5). The probe hybridized with wild-type DNA at approximately 4.8 kb, corresponding to the original HindIII fragment. On the other hand, there is only one signal at 3.8 kb in the lane containing genomic DNA of the mutant 131. This result indicates that the genome of this clone is completely segregated while the genome of 132 is not because two signals at 4.8 and 3.8 kb occurred on the blot. The mutant 131 was designated AVM13 and was chosen for further investigations.
FIG. 5.
Southern analysis of the hupSL operon. (A) Total DNA (5 μg) from A. variabilis and the two mutant strains 131 and 132 was restricted with HindIII. (B) pKS13 containing part of the hupL gene was used as a probe. The arrows indicate the hybridization signals of the wild type (4.8 kb) and the mutants (3.8 kb).
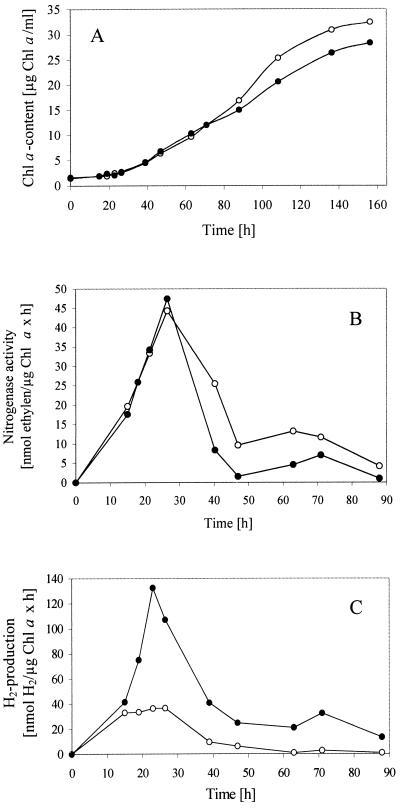
The physiological effects of the mutation in strain AVM13 were investigated by comparing the diazotrophic growth under aerobic conditions and the hydrogen metabolism (the in vivo nitrogenase and hydrogenase activity) of the mutant and the wild-type strains. Because the uptake hydrogenase is only active in heterocysts during nitrogen fixation, the deletion of the hup genes should have effects on a culture grown in medium without combined nitrogen. The A. variabilis wild type and mutant strain AVM13 were grown in media with combined nitrogen (BG11) or without combined N2 (BG110). Following the growth, the chlorophyll α content of the cultures was measured. In BG11, AVM13 cells have growth rates similar to that of the wild type (data not shown). During the first 80 h, the diazotrophic growth of both strains was nearly the same; however, after 80 h and until the end of the measurements, the mutant strain grew worse than did the wild type (Fig. 6A). Since the deletion of the hupSL genes in mutant AVM13 cells affected the growth under N2-fixing conditions, the effect of the mutation on the nitrogenase, the key enzyme of nitrogen fixation which is also involved in hydrogen metabolism, was examined. The maximum of the nitrogenase activity could be determined about 24 h after induction of the cells. Both cultures had nearly the same nitrogenase activity after 24 h, but in the following days, much higher values of acetylene reduction for the wild type were measured (Fig. 6B).
FIG. 6.
Bacterial growth (A), nitrogenase activity (B), and hydrogen production (C) of the A. variabilis wild type and AVM13 mutant strain under nitrogen-fixing conditions. After growing in BG11 medium containing combined nitrogen, the cells of the wild type and the mutant were washed with N-free medium BG110 and adjusted to the same cell density (measured by the chlorophyll α content). Open circles, A. variabilis wild type; closed circles, AVM13 (A. variabilis hupSL interposon mutant).
Concerning the H2 evolution, this paper demonstrates that the mutant AVM13 and the wild type produced equal rates of hydrogen in the first 16 h after induction with BG110 medium. At this time, the hupSL genes encoding the uptake hydrogenase were not expressed (data not shown). Contrastingly, after the expression of the hupSL operon, the H2 evolution of the mutant strain AVM13 increased dramatically in the next 20 h, while for the wild type no change in the rates of H2 evolution could be seen (Fig. 6C). Similar results were obtained for another mutant, in which the neomycin resistance cassette was cloned in the opposite direction to the hupSL operon. Under nitrogen-fixing conditions, the mutant developed very high rates of H2 (68 nmol H2/μg Chl α × h) because the produced hydrogen could not be oxidized. The wild-type cells, however, consumed even more hydrogen than the nitrogenase produced (−36 nanomoles of H2/micrograms of chlorophyll α × h). The activity of the bidirectional NAD+-hydrogenase assayed under non-N2-fixing conditions was very low for both the wild type and the mutant.
DISCUSSION
The similarity of the uptake hydrogenase from Anabaena sp. strain PCC 7120 and A. variabilis allowed for the isolation of a 4.8-kb HindIII fragment and of a 4.8-kb XbaI fragment of A. variabilis via hybridization of partial genomic libraries using the Anabaena sp. strain PCC 7120 hupL gene as a probe. The high degree of similarity between the hupSL operon of A. variabilis and other filamentous cyanobacteria (Anabaena sp. strain PCC 7120, 95%; Nostoc sp. strain PCC 73102, 90%) may reflect more stringent requirements for the conservation of amino acids in the uptake hydrogenase. The noncoding region between hupS and hupL, however, shows only slight similarities between A. variabilis and Anabaena sp. strain PCC 7120 and no similarities between A. variabilis and Nostoc sp. strain PCC 73102. This may hint not only at the physiological importance of the uptake hydrogenase but also at the phylogenetic divergence during evolution in filamentous cyanobacteria.
Protein sequence alignment of the HupS and HupL subunits was done with several Ni-Fe hydrogenases from different bacterial groups by using the ClustalW program (48; data not shown). The large subunit of Ni-Fe hydrogenases contains the Ni atom in the active site. As in the other organisms, one putative Ni-binding site (R-X-C-G-X-C) is located at the N-terminal site of the HupL protein in A. variabilis. The second conserved Ni-binding site (D-P-C-X-X-C) is found at the C-terminal end. The small subunit of A. variabilis contains 11 Cys residues. Nine of them correspond well to nine Cys residues of the known X-ray structure of the hydrogenase from Desulfovibrio gigas (52). HupS of D. gigas has two [4Fe-4S] clusters and one [3Fe-3S] cluster as prosthetic groups. The typical features of the small subunit of the dimeric Ni-Fe hydrogenase are the presence of a signal peptide at the N terminus and a motif located at the C terminus for anchoring the uptake hydrogenase to the membrane (51). Both features could not be found for the HupS protein of A. variabilis and of Nostoc sp. strain PCC 73102 (31). As Oxelfelt et al. pointed out, the role of the motifs is not yet clear and some examples of uptake hydrogenases which lack these features exist.
The cloning and sequencing of hydrogenase genes cluster led to the discovery of a number of accessory genes just up- and downstream of the two structural genes (51). In Alcaligenes eutrophus, up to 20 ORFs encoding proteins that are essential for the formation of the active hydrogenase have been characterized (20). Therefore, we sequenced 3 kb upstream and 4 kb downstream of the hupSL operon. Eight ORFs were discovered, but none of them showed similarities to other known accessory genes. Two kilobases downstream of the hupL gene, two ORFs which might encode for proteins of glucose metabolism could be detected. This means that the accessory genes of the uptake hydrogenase must be located elsewhere in the bacterial genome.
During late stages of heterocyst differentiation, three DNA rearrangements were found in Anabaena sp. strain PCC 7120 (10, 24). In vegetative cells, the genes nifD, fdxN, and hupL are interrupted by insertions of DNA elements. After excision by site-specific recombinases, the intact transcripts can be expressed in heterocysts. The nif1 region of A. variabilis, however, possesses only the 11-kb element in the nifD gene and no interruption in the fdxN gene. The data in this study revealed the presence of a contiguous hupSL operon in A. variabilis. These results are supported by the fact that the specific recombinase xisC of the hupL rearrangement of Anabaena sp. strain PCC 7120 could not be detected in Southern blot analysis with genomic DNA of A. variabilis (5). Beside the nif1 region, A. variabilis has an alternative Mo-dependent nitrogenase gene cluster (nif2) which is transcribed only under anaerobic conditions (46). Low-stringency Southern hybridization indicated that the hupL gene is a single-copy gene and that no similar ORFs for hupSL genes exist. Since the hupSL transcript is induced only under nitrogen-fixing conditions, A. variabilis has to regulate the uptake hydrogenase in a different way. In Northern analysis, we could show that the hupSL genes are probably transcribed as a dicistronic operon of a 2.7-kb size. In lithoautotrophic bacteria, most of the known genes for the uptake hydrogenase are clustered in a polycistronic operon, often with a third gene encoding HupC (51). The hupSL transcript could only be detected in cells grown 24 h on an N-free medium. The nifDHK genes of A. variabilis are induced earlier (12 h) during heterocyst differentiation (data not shown). This observation suggests that the hupSL genes are transcribed during the late stages of heterocyst differentiation when the nitrogenase is already active. It has been reported that the levels of uptake hydrogenase activity in Anabaena sp. strain PCC 7120 are up to five times higher in cultures grown under H2-N2-CO2 conditions (25), so the endogenous H2 produced by the nitrogenase may act as an inducer of the hydrogenase synthesis.
In cyanobacteria, promoter DNA elements that displayed a conserved element at −10 from the transcription initiation site could be found (15), which conforms to the E. coli −10 promoter consensus sequence (TATAAT). The controlling promoters of genes encoding chimeric hydrogenases of hydrogen-oxidizing bacteria have also been characterized (8, 35, 50). In most of the cases, sequence elements resembling a −24/−12 consensus sequence of ς54-dependent promoters are located just upstream of the transcription start (39). Neither a TATAAT motif nor a −24/−12 consensus sequence could be detected upstream of the hupSL operon of A. variabilis. Interestingly, the location of a fumarate nitrate reductase regulator (Fnr)-binding motif and a −10 consensus motif, which was found during the regulation of the hyp operon in E. coli (26), was confirmed. Comparing the putative Fnr-binding site of the upstream region of the hupSL operon with the promoter of the hypBCDE transcript, the same succession of bases could be demonstrated; only the distance of the motifs to the transcription site is different.
DNase footprinting experiments and in vitro transcription confirm the unusual localization of the Fnr-binding site in the napF control region in E. coli (16). As shown in Fig. 7, four bases of the napF promoter sequence are modified from the Fnr consensus sequence, but only one base is exchanged in the promoter region of hupSL as compared with the napF promoter. During anaerobic growth, the Fnr protein induces the expression of several operons in E. coli (43). In A. variabilis, the induction of the hupSL operon occurs within the heterocysts. Heterocysts are terminally differentiated cells whose interiors become anaerobic. This suggests a similar regulation of the hupSL operon compared with that of E. coli.
FIG. 7.
Nucleotide sequences of the consensus Fnr-binding site, the Fnr-binding site within the napF promoter of E. coli, and the promoter of the hupSL operon of A. variabilis. Three nucleotides out of 10 are different between the consensus sequence of the Fnr-binding site in E. coli, the napF (periplasmic nitrate reductase) promoter, and the hupSL promoter of A. variabilis.
The fivefold repeats (TA/GACAAC) downstream of the transcription site are another interesting structure which represent a new type of short tandemly repeated heptamers. Cyanobacterial genomes have a variety of such tandemly repeated sequences with unknown function, but most of them are specific to the heterocystous strains (24). It has been reported that these sequences might be a target of specific DNA-binding proteins for chromosome condensation (27). Further promoter analyses are needed to understand the transcription of the hupSL operon.
We also studied the induction of in vivo hydrogen uptake and nitrogenase activities under N-limiting conditions in the wild type and the hupSL mutant of A. variabilis. There are two hints that, in the light, the measured hydrogen originates from the nitrogenase activity. Firstly, hydrogen photoproduction of A. variabilis cultures did not occur under NO3−-saturated conditions in which the nitrogenase genes are not expressed. In BG11 medium, a slight H2 production was measured only in the dark, which indicates a low catalytic activity of the bidirectional hydrogenase. Secondly, the curve diagram of the H2 evolution corresponds well with the one of acetylene reduction. After 24 h of growth under nitrogen-fixing conditions, the maximal H2 production and nitrogenase activity could be observed. Similar effects have been described earlier (28, 49). Though a lot of biochemical and genetical studies have been done, the in vivo function of the uptake hydrogenase is poorly understood at present. In this study, the physiological data clearly show that the uptake hydrogenase reoxidizes the produced hydrogen at high rates in the wild type. Three to five times more hydrogen is produced by the hupSL mutant, dependent on the growth situations. It was shown that the electrons of the oxidized H2 are fed into the respiratory chain, proceeding to an oxyhydrogen reaction coupled to oxidative phosphorylation (33). The electron transport from H2 to O2 supports ATP synthesis and thereby supplies part of the energy required by the nitrogenase and also protects the nitrogenase by lowering intracellular O2 levels. The hupSL mutant fixed nitrogen after 35 h at lower rates than the wild type. However, the growth curves were almost the same, suggesting that the uptake hydrogenase is not absolutely essential under diazotrophic culture conditions. Obviously, the reduced rates of fixed nitrogen have only a slight effect on the growth of the mutant strain.
ACKNOWLEDGMENTS
We thank James W. Golden for the plasmids containing the hupL gene of Anabaena sp. strain PCC 7120 and C. Peter Wolk for the pRL plasmids. We also thank H. Geithmann for photographic work.
This work was supported by the Deutsche Forschungsgemeinschaft (Ha 2555/1-1).
REFERENCES
- 1.Altschul S F, Miller W, Myers E W, Lipman D J. Basic local alignment search tool. J Mol Biol. 1997;215:403–410. doi: 10.1016/S0022-2836(05)80360-2. [DOI] [PubMed] [Google Scholar]
- 2.Ansorge W, Sproat B, Stegemann J, Schwager C, Zenke M. Automated DNA sequencing: ultrasensitive detection of fluorescent bands during electrophoresis. Nucleic Acids Res. 1987;15:4593–4602. doi: 10.1093/nar/15.11.4593. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Appel J, Schulz R. Sequence analysis of an operon of a nickel hydrogenase from the cyanobacterium Synechocystis sp. PCC 6803. Biochim Biophys Acta. 1996;1298:141–147. doi: 10.1016/s0167-4838(96)00176-8. [DOI] [PubMed] [Google Scholar]
- 4.Ausubel F M, Brent R, Kingston R E, Moore D D, Seidman J G, Smith J A, Struhl K, editors. Current protocols in molecular biology. New York, N.Y: Greene Publishing Associates and Wiley-Interscience; 1987. [Google Scholar]
- 5.Axelsson R, Oxelfelt F, Lindblad P. Transcriptional regulation of Nostoc uptake hydrogenase. FEMS Microbiol Lett. 1999;170:77–81. doi: 10.1111/j.1574-6968.1999.tb13357.x. [DOI] [PubMed] [Google Scholar]
- 6.Böhme H. Regulation of nitrogen fixation in heterocyst-forming cyanobacteria. Trends Plant Sci. 1998;9:346–351. [Google Scholar]
- 7.Boison G, Schmitz O, Mikheeva L, Shestakov S, Bothe H. Cloning, molecular analysis and insertional mutagenesis of the bidirectional hydrogenase genes from the cyanobacterium Anacystis nidulans. FEBS Lett. 1996;394:153–158. doi: 10.1016/0014-5793(96)00936-2. [DOI] [PubMed] [Google Scholar]
- 8.Brito B, Martinez M, Fernandez D, Rey L, Cabrera E, Palacios J M, Imperial J, Ruiz-Argueso T. Hydrogenase genes from Rhizobium leguminosarum bv. viciae are controlled by the nitrogen fixation regulatory protein nifA. Proc Natl Acad Sci USA. 1997;94:6019–6024. doi: 10.1073/pnas.94.12.6019. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Buikema W J, Haselkorn R. Molecular genetics of cyanobacterial development. Annu Rev Plant Physiol Plant Mol Biol. 1993;44:333–352. [Google Scholar]
- 10.Carrasco C D, Buettner J A, Golden J W. Programmed DNA rearrangement of the cyanobacterial hupL gene in heterocysts. Proc Natl Acad Sci USA. 1995;92:791–795. doi: 10.1073/pnas.92.3.791. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Casadaban M, Cohen S N. Analysis of gene control signals by DNA fusion and cloning in Escherichia coli. J Mol Biol. 1980;138:179–207. doi: 10.1016/0022-2836(80)90283-1. [DOI] [PubMed] [Google Scholar]
- 12.Castenholz R W. Culturing methods for cyanobacteria. Methods Enzymol. 1988;167:68–93. [Google Scholar]
- 13.Cohen S N, Chang A C, Hsu L. Nonchromosomal antibiotic resistance in bacteria: genetic transformation of Escherichia coli by R-factor DNA. Proc Natl Acad Sci USA. 1972;69:2110–2114. doi: 10.1073/pnas.69.8.2110. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Currier T C, Wolk C P. Characteristics of Anabaena variabilis influencing plaque formation by cyanophage N-1. J Bacteriol. 1979;171:4138–4145. doi: 10.1128/jb.139.1.88-92.1979. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Curtis S E, Martin J A. The transcription apparatus and the regulation of transcription initiation. In: Bryant D A, editor. The molecular biology of cyanobacteria. Dordrecht, The Netherlands: Kluwer Academic Publishers; 1994. pp. 769–823. [Google Scholar]
- 16.Darwin A J, Ziegelhoffer E C, Kiley P J, Stewart V. Fnr, NarP, and NarL regulation of Escherichia coli K-12 napF (periplasmic nitrate reductase) operon transcription in vitro. J Bacteriol. 1998;180:4192–4198. doi: 10.1128/jb.180.16.4192-4198.1998. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Dilworth M J. Acetylene reduction by nitrogen fixing preparations from Clostridium pasteurianum. Biochim Biophys Acta. 1966;127:285–294. doi: 10.1016/0304-4165(66)90383-7. [DOI] [PubMed] [Google Scholar]
- 18.Elhai J, Wolk C P. Conjugal transfer of DNA to cyanobacteria. Methods Enzymol. 1988;167:747–754. doi: 10.1016/0076-6879(88)67086-8. [DOI] [PubMed] [Google Scholar]
- 19.Elhai J, Vepritskiy A, Muro-Pastor A M, Flores E, Wolk C P. Reduction of conjugal transfer efficiency by three restriction activities of Anabaena sp. strain PCC 7120. J Bacteriol. 1997;179:1998–2005. doi: 10.1128/jb.179.6.1998-2005.1997. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Friedrich B, Schwartz E. Molecular biology of hydrogen utilization in aerobic chemolithotrophs. Annu Rev Microbiol. 1993;47:351–383. doi: 10.1146/annurev.mi.47.100193.002031. [DOI] [PubMed] [Google Scholar]
- 21.Gish W, States D J. Identification of protein coding regions by database similarity search. Nat Genet. 1993;3:166–172. doi: 10.1038/ng0393-266. [DOI] [PubMed] [Google Scholar]
- 22.Hahn D, Kueck U. Biochemical and molecular genetic basis of hydrogenases. Process Biochem. 1994;29:633–641. [Google Scholar]
- 23.Happe T, Naber J D. Isolation, characterization and N-terminal amino acid sequence of hydrogenase from the green alga Chlamydomonas reinhardtii. Eur J Biochem. 1993;214:475–481. doi: 10.1111/j.1432-1033.1993.tb17944.x. [DOI] [PubMed] [Google Scholar]
- 24.Haselkorn R, Buikema W J. Nitrogen fixation in cyanobacteria. In: Stacey G, Evans H J, Burris R H, editors. Biological nitrogen fixation. New York, N.Y: Chapman and Hall; 1992. pp. 166–190. [Google Scholar]
- 25.Houchins J P. The physiology and biochemistry of hydrogen metabolism in cyanobacteria. Biochim Biophys Acta. 1984;768:227–255. [Google Scholar]
- 26.Lutz S, Jacobi A, Schlensog V, Bohm R, Sawers G, Bock A. Molecular characterization of an operon (hyp) necessary for the activity of the three hydrogenase isoenzymes in Escherichia coli. Mol Microbiol. 1991;5:123–135. doi: 10.1111/j.1365-2958.1991.tb01833.x. [DOI] [PubMed] [Google Scholar]
- 27.Mazel D, Houmard J, Castets A M, Tandeau de Marsac N. Highly repetitive DNA sequences in cyanobacterial genomes. J Bacteriol. 1990;172:2755–2761. doi: 10.1128/jb.172.5.2755-2761.1990. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Mikheeva L E, Schmitz O, Shestakov S V, Bothe H. Mutants of the cyanobacterium Anabaena variabilis altered in hydrogenase activities. Z Naturforsch. 1995;50c:505–510. [Google Scholar]
- 29.Myöhänen S, Wahlfors J. Automated fluorescent primer extension. BioTechniques. 1993;14:16–17. [PubMed] [Google Scholar]
- 30.Oxelfelt F, Tamagnini P, Salema R, Lindblad P. Hydrogen uptake in Nostoc sp. strain PCC 73102: effects of nickel, hydrogen, carbon, and nitrogen. Plant Physiol Biochem. 1995;33:617–623. [Google Scholar]
- 31.Oxelfelt F, Tamagnini P, Lindblad P. Hydrogen uptake in Nostoc sp. strain PCC 73102. Cloning and characterization of a hupSL homologue. Arch Microbiol. 1998;169:267–274. doi: 10.1007/s002030050571. [DOI] [PubMed] [Google Scholar]
- 32.Pang K M, Knecht D A. Partial inverse PCR: a technique for cloning flanking sequences. BioTechniques. 1997;22:1046–1048. doi: 10.2144/97226bm07. [DOI] [PubMed] [Google Scholar]
- 33.Peterson R B, Burris R H. Hydrogen metabolism in isolated heterocysts of Anabaena 7120. Arch Microbiol. 1978;116:125–132. [Google Scholar]
- 34.Przybyla A E, Robbins J, Menon N, Peck H D., Jr Structure-function relationships among nickel-containing hydrogenases. FEMS Microbiol Rev. 1992;88:109–136. doi: 10.1111/j.1574-6968.1992.tb04960.x. [DOI] [PubMed] [Google Scholar]
- 35.Richaud P, Colbeau A, Toussaint B, Vignais P M. Identification and sequence analysis of the hupR1 gene, which encodes a response regulator of the NtrC family required for hydrogenase expression in Rhodobacter capsulatus. J Bacteriol. 1991;173:5928–5932. doi: 10.1128/jb.173.18.5928-5932.1991. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Sambrook J, Fritsch E F, Maniatis T. Molecular cloning: a laboratory manual. 2nd ed. Cold Spring Harbor, N.Y: Cold Spring Harbor Laboratory Press; 1989. [Google Scholar]
- 37.Sanger F, Nicklen S, Coulson A R. DNA sequencing with chain-terminating inhibitors. Proc Natl Acad Sci USA. 1977;74:5463–5467. doi: 10.1073/pnas.74.12.5463. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Schmitz O, Boison G, Hilscher R, Hundeshagen B, Zimmer W, Lottspeich F, Bothe H. Molecular biological analysis of a bidirectional hydrogenase from cyanobacteria. Eur J Biochem. 1995;233:266–276. doi: 10.1111/j.1432-1033.1995.266_1.x. [DOI] [PubMed] [Google Scholar]
- 39.Schwartz E, Gerischer U, Friedrich B. Transcriptional regulation of Alcaligenes eutrophus hydrogenase genes. J Bacteriol. 1998;180:3197–3204. doi: 10.1128/jb.180.12.3197-3204.1998. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Short J M, Fernandez J M, Sorge J A, Huse W D. λ-ZAP: a bacteriophage λ expression vector with in vivo excision properties. Nucleic Acids Res. 1988;16:7538. doi: 10.1093/nar/16.15.7583. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Simon L, Priefer U, Pühler A. A broad host range mobilization system for in vivo genetic engineering: transposon mutagenesis in gram negative bacteria. Bio/Technology. 1983;1:784–791. [Google Scholar]
- 42.Smoker J A, Barnum S R. Rapid small scale DNA isolation from filamentous cyanobacteria. FEMS Microbiol Lett. 1988;56:119–122. [Google Scholar]
- 43.Spiro S, Guest J R. FNR and its role in oxygen-regulated gene expression in Escherichia coli. FEMS Microbiol Rev. 1990;6:399–428. doi: 10.1111/j.1574-6968.1990.tb04109.x. [DOI] [PubMed] [Google Scholar]
- 44.Stanier R, Cohen-Bazire G. Plasmide genetic. Annu Rev Microbiol. 1977;31:225–274. doi: 10.1146/annurev.mi.31.100177.001301. [DOI] [PubMed] [Google Scholar]
- 45.Stricker O, Masepohl B, Klipp W, Böhme H. Identification and characterization of the nifV-nifZ-nifT gene region from the filamentous cyanobacterium Anabaena sp. strain PCC 7120. J Bacteriol. 1997;179:2930–2937. doi: 10.1128/jb.179.9.2930-2937.1997. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Thiel T, Lyons E, Erker J C, Ernst A. A second nitrogenase in vegetative cells of a heterocyst-forming cyanobacterium. Proc Natl Acad Sci USA. 1995;92:9358–9362. doi: 10.1073/pnas.92.20.9358. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Thiel T. Isolation and characterization of the vnfEN genes of the cyanobacterium Anabaena variabilis. J Bacteriol. 1996;178:4493–4499. doi: 10.1128/jb.178.15.4493-4499.1996. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Thompson J D, Higgins D G, Gibson T J. CLUSTAL W: improving the sensitivity of progressive multiple sequence alignment through sequence weighting, position-specific gap penalties and weight matrix choice. Nucleic Acids Res. 1994;22:4673–4680. doi: 10.1093/nar/22.22.4673. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Troshina O Y, Serebryakova L T, Lindblad P. Induction of H2-uptake and nitrogenase activities in the cyanobacterium Anabaena variabilis ATCC 29413: effects of hydrogen and organic substrate. Curr Microbiol. 1996;33:11–15. doi: 10.1007/s002849900066. [DOI] [PubMed] [Google Scholar]
- 50.Van Soom C, de Wilde P, Vanderleyden J. HoxA is a transcriptional regulator for expression of the hup structural genes in free-living Bradyrhizobium japonicum. Mol Microbiol. 1997;23:967–977. doi: 10.1046/j.1365-2958.1997.2781648.x. [DOI] [PubMed] [Google Scholar]
- 51.Vignais P M, Toussaint B. Molecular biology of membrane-bound H2 uptake hydrogenases. Arch Microbiol. 1994;161:1–10. doi: 10.1007/BF00248887. [DOI] [PubMed] [Google Scholar]
- 52.Volbeda A, Charon M-H, Piras C, Hatchikian E C, Frey M, Fontecilla-Camps J-C. Crystal structure of the nickel-iron hydrogenase from Desulfovibrio gigas. Nature. 1995;373:580–585. doi: 10.1038/373580a0. [DOI] [PubMed] [Google Scholar]
- 53.Voordouw G. Evolution of hydrogenase genes. In: Cammack R, editor. Advances in inorganic chemistry, vol. 38, Iron-sulfur proteins. San Diego, Calif: Academic Press Inc.; 1992. pp. 397–422. [Google Scholar]
- 54.Wu L F, Mandrand M A. Microbial hydrogenases: primary structure, classification, signatures and phylogeny. FEMS Microbiol Rev. 1993;104:243–269. doi: 10.1111/j.1574-6968.1993.tb05870.x. [DOI] [PubMed] [Google Scholar]
- 55.Yanisch-Perron C, Vieira J, Messing J. Improved M13 phage cloning vectors and host strains: nucleotide sequences of the M13mp18 and pUC19 vectors. Gene. 1985;33:103–119. doi: 10.1016/0378-1119(85)90120-9. [DOI] [PubMed] [Google Scholar]