Abstract
Background
The COVID-19 pandemic may worsen the mental health of people reporting subjective cognitive decline (SCD) and therefore their clinical prognosis. We aimed to investigate the association between the intensity of SCD and anxious/depressive symptoms during confinement and the underlying mechanisms.
Methods
Two hundred fifty cognitively unimpaired participants completed the Hospital Anxiety and Depression Scale (HADS) and SCD-Questionnaire (SCD-Q) and underwent amyloid-β positron emission tomography imaging with [18F] flutemetamol (N = 205) on average 2.4 (± 0.8) years before the COVID-19 confinement. During the confinement, participants completed the HADS, Perceived Stress Scale (PSS), Brief Resilience Scale (BRS), and an ad hoc questionnaire on worries (access to primary products, self-protection materials, economic situation) and lifestyle changes (sleep duration, sleep quality, eating habits). We investigated stress-related measurements, worries, and lifestyle changes in relation to SCD. We then conducted an analysis of covariance to investigate the association of SCD-Q with HADS scores during the confinement while controlling for pre-confinement anxiety/depression scores and demographics. Furthermore, we introduced amyloid-β positivity, PSS, and BRS in the models and performed mediation analyses to explore the mechanisms explaining the association between SCD and anxiety/depression.
Results
In the whole sample, the average SCD-Q score was 4.1 (± 4.4); 70 (28%) participants were classified as SCD, and 26 (12.7%) were amyloid-β-positive. During the confinement, participants reporting SCD showed higher PSS (p = 0.035) but not BRS scores (p = 0.65) than those that did not report SCD. No differences in worries or lifestyle changes were observed. Higher SCD-Q scores showed an association with greater anxiety/depression scores irrespective of pre-confinement anxiety/depression levels (p = 0.002). This association was not significant after introducing amyloid-β positivity and stress-related variables in the model (p = 0.069). Amyloid-β positivity and PSS were associated with greater HADS irrespective of pre-confinement anxiety/depression scores (p = 0.023; p < 0.001). The association of SCD-Q with HADS was mediated by PSS (p = 0.01).
Conclusions
Higher intensity of SCD, amyloid-β positivity, and stress perception showed independent associations with anxious/depressive symptoms during the COVID-19 confinement irrespective of pre-confinement anxiety/depression levels. The association of SCD intensity with anxiety/depression was mediated by stress perception, suggesting stress regulation as a potential intervention to reduce affective symptomatology in the SCD population in the face of stressors.
Supplementary Information
The online version contains supplementary material available at 10.1186/s13195-022-01068-7.
Keywords: Subjective cognitive decline, Stress, Anxiety, Depression, COVID-19 confinement, Alzheimer’s disease
Background
The COVID-19 pandemic and subsequent measures implemented to prevent the spread of the disease, such as home confinement, are having a global impact on the mental health of the general population [1, 2]. Several studies conducted during this period indicated that the mental health burden of the pandemic may be greater in clinically vulnerable populations [3–5] such as those with subjective cognitive decline (SCD) [6, 7].
During the preclinical stage, Alzheimer’s disease (AD) pathologies—amyloid beta (Aβ) and tau—slowly aggregate in the brain and interact with other risk factors including anxiety and depression [8–10], eventually leading to cognitive decline and dementia [11]. Before the onset of the cognitive impairment, some individuals may experience SCD defined as the perception of cognitive decline despite having objectively normal performance in neuropsychological testing [12, 13]. SCD is associated with a higher risk of developing AD [14, 15] as well as with the earliest AD-related pathophysiological processes such as Aβ accumulation [14, 16, 17].
Affective symptoms have been linked with an elevated risk for developing AD in cognitively unimpaired (CU) adults [8, 9, 18]. Moreover, growing evidence suggests associations between anxious/depressive symptoms and biomarkers of AD in CU older adults [19–21], such as with brain Aβ burden [22, 23]. These symptoms may also underlie SCD and/or interact with it [24]. Several studies reported that anxiety, depression, and worries are among the most common concurrent affective symptoms with SCD in older adults [24–26]. Moreover, the co-occurrence of SCD with these symptoms has been linked with an increased likelihood of cognitive decline [27–31].
Recent studies found associations between subjective memory complaints and stressful life events in CU older adults [32, 33]. COVID-19-related home confinement, as one of the main stressors related to the pandemic [33, 34], has been related to increased anxious/depressive symptoms as evidenced by numerous studies [1, 2, 34, 35]. Yet, many studies addressing this topic focused on the general population [1, 2] or individuals with dementia [4, 36]. Investigating the mental health implications of confinement in populations at elevated risk of cognitive decline can be instrumental to plan interventions and improve their clinical prognosis. In this regard, stress perception and stress resilience are especially interesting as they are shaped by individual factors or can be enhanced by training.
Herein, we focused on CU adults with a subgroup experiencing SCD, many of them being first-order descendants of sporadic AD patients and thus at a higher risk of developing cognitive decline and AD dementia [37, 38]. Our main aim was to investigate whether the intensity of SCD was associated with greater anxious/depressive symptoms during the COVID-19 confinement independent of pre-confinement anxiety/depression levels. Furthermore, we investigated the impact of brain Aβ status, stress perception, and stress resilience on these associations. Finally, we evaluated whether participants with or without SCD showed differences in worries and lifestyle changes during the confinement.
Methods
Participants and study design
Participants were selected from the longitudinal ALzheimer’s and FAmilies (ALFA+) study, nested to the ALFA parent cohort. ALFA parent cohort participants were recruited between 2013 and 2014 and included 2743 middle-aged (45–74 years) CU individuals, with the majority having a family history (FH) of sporadic AD [37]. The nested ALFA+ study included 451 cognitively intact participants (Clinical Dementia Rating = 0, Mini-Mental State Examination ≥ 27, semantic fluency ≥ 12) without any significant systematic illness, major psychiatric disorder, or unstable medical condition. ALFA+ participants underwent detailed cognitive testing, clinical interviews, lifestyle questionnaires, APOE genotyping, and blood and cerebrospinal fluid extractions, as well as magnetic resonance imaging and Aβ positron emission tomography (PET) data acquisitions [37]. These data were collected between 2016 and 2019 (average time lapse from ALFA+ HADS data acquisition to confinement data acquisition: 2.4 ± 0.8 years) from ALFA+ participants and referred to as “pre-confinement” measurements in the current study.
On May 8, 2020, during the COVID-19-related home confinement de-escalation periods in Spain [39], an invitation to participate in the current study was sent via an online link to 2582 participants of the ALFA parent cohort. Among these, 265 participants from the ALFA+ study responded positively to the invitation and completed an online assessment battery between May 8, 2020, and July 7, 2020, the period referred to as “confinement” in the present study. The assessment battery consisted of measurements of anxiety, depression, stress perception, stress resilience, and an ad hoc questionnaire developed to investigate specific worries and changes in lifestyle during the confinement. A total of 250 ALFA+ participants that completed the Hospital Anxiety and Depression Scale (HADS) in the assessment battery were included in the current study. As described below, participants had clinical data (SCD and HADS assessments) and Aβ PET data (N = 205) acquired during the pre-confinement period.
Clinical assessments
SCD intensity and status
During the pre-confinement period (in the context of the ALFA+ study), participants filled out the MyCog part of the Subjective Cognitive Decline-Questionnaire (SCD-Q) [40] before neuropsychological testing. The SCD-Q measures the presence or absence of a self-perceived deterioration in cognitive performance in the last two years of daily life. The MyCog part consists of 24 questions inquiring about the subjective experience of difficulties in performing memory, language, and executive tasks. Here, we investigated the intensity of SCD using MyCog scores as a continuous measurement of SCD symptoms, with higher scores indicating greater complaints in cognitive performance [26, 40]. The rationale behind this approach was based on the characteristic of our sample (middle-aged adults) and in consequence the relatively low percentage of participants classified as SCD with a binary classification (yes/no). Nevertheless, to allow comparison with previous and future research and test the between-group differences, we also classified participants as having SCD if the answer to the first question “Do you perceive memory or cognitive difficulties?” was affirmative and without SCD if the answer was negative [41]. Finally, we performed a sensitivity analysis with participants classified as SCD aged 60 and above.
Anxiety and depression
We used the 14-item HADS composed of the 7-item anxiety and the 7-item depression subscale to measure the symptoms of anxiety/depression. In each subscale, the items are scored from 0 to 3, generating a total score between 0 and 21. Higher scores indicate a greater level of anxiety or depression. The sum of 14 items generates a total anxiety/depression score [42].
Stress perception and stress resilience
Participants completed the 10-item Perceived Stress Scale (PSS) [43] as a measurement of self-reported stress perception during the confinement. Scores in the PSS range from 0 to 40 with higher ones reflecting a greater perception of stress. Furthermore, we evaluated the participants’ ability to resist or recover from stress using the 6-item Brief Resilience Scale (BRS) [44]. Higher scores in BRS indicate higher resilience to stress.
Worries and lifestyle changes during the COVID-19 confinement
We developed an ad hoc questionnaire that focused on the worries about access to primary products, self-protection materials, and economic situation, as well as changes in sleep duration, sleep quality, and eating habits during the confinement. The questions and the procedure to dichotomize the answers are provided in Additional file 1.
Aβ PET data acquisition and pre-processing
[18F] flutemetamol PET acquisitions were performed at the pre-confinement period in 205 participants in a Siemens Biograph mCT (Munich, Germany) following a cranial computed tomography scan for attenuation correction; 185 MBq (range 166.5–203.5 MBq) of [18F] flutemetamol was injected to the participants, and four frames of 5 min each were acquired 90 min post-injection. An OSEM3D algorithm with 8 iterations and 21 subsets was used for image reconstruction with point spread function and time-of-flight corrections into a matrix size of 1.02 × 1.02 × 2.03 mm.
All PET images were preprocessed using SPM12 (Statistical Parametric Mapping, Welcome Trust Centre for Neuroimaging, UK; http://www.fil.ion.ucl.ac.uk/spm). The averaged PET images were co-registered to the corresponding T1-weighted images. Then, T1-weighted images were segmented and normalized to Montreal Neurological Institute (MNI) space alongside the PET images. The standardized value uptake ratio (SUVR) was calculated in MNI space from the bilateral frontal and parietotemporal areas, and the whole cerebellum was used as a reference region. We then converted the SUVR values to the Centiloid (CL) [45] scale (see [46] for details). The cutoff value for CL was defined with a threshold of 12, and the scans were classified as “Aβ-negative” (< 12 CL) or “Aβ-positive” (≥ 12 CL) [46].
Statistical analyses
Descriptive statistics were generated with means and standard deviations for continuous variables and percentages and frequencies for categorical variables. We performed chi-square analyses to explore the differences in worries and lifestyle changes during the COVID-19 confinement between participants classified as SCD or without SCD.
We conducted analyses of covariance (ANCOVA) with HADS scores during the confinement as the dependent variable, adjusted by pre-confinement HADS scores. Firstly, we tested whether higher SCD-Q MyCog scores were associated with higher anxiety/depression scores during the confinement independent of the pre-confinement anxiety/depression levels. We adjusted the model by age, sex, years of education, and the inter-individual time variability between pre-confinement and confinement assessments. In the second step, we introduced Aβ positivity as well as stress perception (PSS) and stress resilience (BRS) in the model. Finally, we performed mediation analyses using bootstrap procedures (computed for N = 1000 bootstrapped samples) to investigate whether the tested association between SCD-Q and HADS scores was mediated by Aβ positivity or stress-related measurements.
Statistical analyses were conducted with IBM SPSS Statistics for Windows (Build 1.0.0.1447, Armonk, NY), and for the mediation analysis, the R Statistical Software (version 4.0.2) “mediation” package was used (R Foundation for Statistical Computing, Vienna, Austria). The results yielding a p value lower than 0.05 were considered as statistically significant.
Results
In the whole sample, participants’ age ranged from 52 to 72 years; 153 (61.2%) were women, and 248 (99.2%) were white Caucasians (0.8% Latinos). The average MyCog score was 4.1 (± 4.4) and 55 (22.3%) showed high-intensity SCD (MyCog scores ≥ 7) [47]. Seventy (28%) participants were classified as having SCD. A total of 197 participants (78.8%) and 53 of those classified as SCD (75.7%) had at least one parent diagnosed with sporadic AD. Twenty-six (12.7%) out of the 205 participants with available Aβ PET scans were Aβ-positive. Twenty-two (31.4%) of those classified as SCD sought medical help due to the perceived decline in cognitive abilities. During the confinement, 26 (10.4%) participants were diagnosed or under treatment for anxiety or depression. Table 1 displays the sociodemographic, clinical, and imaging data for the whole sample and participants classified as SCD vs. without SCD.
Table 1.
Demographic, imaging, and clinical characteristics of the study participants
| Overall | With SCD | Without SCD | |
|---|---|---|---|
| Variable | N = 250 | N = 70 | N = 180 |
| Age, years | 63.5 (4.8) | 63.9 (4.3) | 63.3 (4.9) |
| Female,n(%) | 153 (61.2) | 43 (61.4) | 110 (61.1) |
| Family history of sporadic AD,n(%)a | 197 (89.1) | 53 (75.7) | 144 (80) |
| Caucasian,n(%) | 248 (99.2) | 68 (97.1) | 180 (100) |
| Education, years | 13.3 (3.5) | 13.8 (3.4) | 13.1 (3.5) |
| Aβ-positive (≥ 12CL),n(%)b | 26 (12.7) | 7 (10) | 19 (10.6) |
| SCD-Q, MyCog scoresc | 4.1 (4.4) | 8.2 (4.5) | 2.5 (3.1) |
| Pre-confinement HADS total scores | 6.6 (4.8) | 8.3 (5.1) | 6 (4.5) |
| Confinement HADS total scores | 8.1 (5.9) | 9.4 (6.1) | 7.6 (5.7) |
| PSS scoresd | 16.1 (8.6) | 18 (9.1) | 15.4 (8.3) |
| BRS scorese | 3.2 (0.4) | 3.1 (0.3) | 3.2 (0.4) |
| Change in HADS from pre-confinement to confinement | 1.5 (5.8) | 1.1 (5.2) | 1.6 (5.9) |
| Currently diagnosed/under treatment for anxiety/depression,n(%)f | 26 (10.4) | 9 (12.9) | 17 (9.4) |
| Seeking medical help due to the self-perceived decline in cognition | 22 (8.8) | 22 (31.4) | – |
Data are presented as mean (standard deviation) unless otherwise indicated
Abbreviations: AD Alzheimer’s disease, SCD subjective cognitive decline, Aβ amyloid-beta, CL Centiloid, SCD-Q Subjective Cognitive Decline Questionnaire, HADS Hospital Anxiety and Depression Scale, PSS Perceived Stress Scale, BRS Brief Resilience Scale
aOverall N = 221
bOverall N = 205
cOverall N = 246
dOverall N = 238
eOverall N = 238
fOverall N = 248
Aβ burden (measured with continuous CL) did not show any correlation with PSS (r = 0.13; p = 0.078) or BRS (r = 0.044; p = 0.53) scores. We did not find a correlation between PSS and BRS scores during the confinement (r = 0.058; p = 0.37).
Participants having SCD showed significantly higher MyCog scores than those without SCD (t95.2=9.74; p < 0.001). Regarding anxiety/depression symptoms, we found a significant increase in HADS scores from pre-confinement to confinement (t249 = 4.04; p < 0.001) in the whole sample. Moreover, a lower frequency of going outside the home was associated with higher HADS scores during the confinement (F = 3.10; p = 0.017). This result suggests that the HADS evaluations are, at least partially, a reflection of the intensity of the COVID-19 confinement.
Participants with or without FH of sporadic AD did not present any difference in SCD-Q (t26.6 = − 1.51; p = 0.14) or HADS scores (pre-confinement: t31.2 = − 0.17; p = 0.87, during the confinement: t34.2 = 1.10; p = 0.29).
Differences in SCD status in pre-confinement anxiety/depression measurements and confinement-related stress perception and stress resilience
Participants classified as having SCD had higher pre-confinement HADS scores (p < 0.001) than those without SCD. Regarding stress-related measurements, participants with SCD showed higher PSS scores (t236 = 2.12; p = 0.035) during the confinement compared to those without SCD. No differences were found in BRS scores between the two groups (t246 = − 0.45; p = 0.65).
In the whole sample, SCD-Q scores were positively correlated with pre-confinement HADS (r = 0.37; p < 0.001) and PSS (r = 0.43; p < 0.001) scores whereas they did not show any correlation with BRS scores (r = 0.044; p = 0.49).
Differences in SCD status in worries and lifestyle changes during the COVID-19 confinement
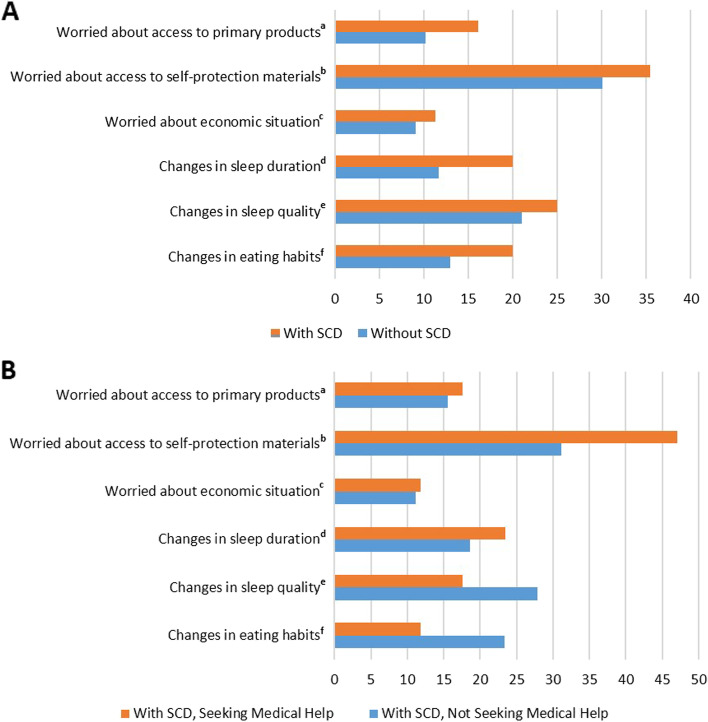
The results of the chi-square analyses investigating the differences between participants with and without SCD in confinement-related worries (access to primary products, self-protection materials, and economic situation) and lifestyle changes (sleep duration, sleep quality, and eating habits) are reported in Table 2. These analyses revealed that there were no significant differences between the two groups in any of the domains assessed (Fig. 1A).
Table 2.
Results from the chi-square analysis investigating the difference in confinement-related worries and lifestyle changes by SCD status
| With SCD (%) | Without SCD (%) | p value | |||
|---|---|---|---|---|---|
| Yes | No | Yes | No | ||
| Worries | |||||
| Access to primary productsa | 16.1 | 83.9 | 10.2 | 89.8 | 0.21 |
| Access to self-protection materialsb | 35.5 | 64.5 | 30.1 | 69.9 | 0.44 |
| Economic situationc | 11.3 | 88.7 | 9.1 | 90.9 | 0.62 |
| Lifestyle changes | |||||
| Changes in sleep durationd | 20 | 80 | 11.7 | 88.3 | 0.11 |
| Changes in sleep qualitye | 25 | 75 | 21 | 79 | 0.52 |
| Changes in eating habitsf | 20 | 80 | 13 | 87 | 0.19 |
Abbreviation: SCD subjective cognitive decline
aWith SCD N = 62, without SCD N = 167
bWith SCD N = 62, without SCD N = 166
cWith SCD N = 62, without SCD N = 165
dWith SCD N = 60, without SCD N = 162
eWith SCD N = 60, without SCD N = 162
fWith SCD N = 60, without SCD N = 162
Fig. 1.
Confinement-related worries and lifestyle changes in the whole sample and in the subsample with SCD. A In the whole sample, the percentage of participants showing specific worries and lifestyle changes during the confinement is displayed by SCD status. aWith SCD N = 62, without SCD N = 167. bWith SCD N = 62, without SCD N = 166. cWith SCD N = 62, without SCD N = 165. dWith SCD N = 60, without SCD N = 162. eWith SCD N = 60, without SCD N = 162. fWith SCD N = 60, without SCD N = 162. B In the subsample with SCD, the percentage of participants showing specific worries and lifestyle changes during the confinement is displayed for the participants who sought medical help and the participants who did not seek medical help. aWith SCD, seeking medical help N = 17; with SCD, not seeking medical help N = 45. bWith SCD, seeking medical help N = 17; with SCD, not seeking medical help N = 45. cWith SCD, seeking medical help N = 17; with SCD, not seeking medical help N = 45. dWith SCD, seeking medical help N = 17; with SCD, not seeking medical help N = 43. eWith SCD, seeking medical help N = 17; with SCD, not seeking medical help N = 43. fWith SCD, seeking medical help N = 17; with SCD, not seeking medical help N = 43. Abbreviations: SCD, subjective cognitive decline
Association between the intensity of SCD and anxiety/depression measurements
Higher SCD-Q MyCog scores were associated with greater HADS scores irrespective of the pre-confinement anxiety/depression levels. Furthermore, younger age, lower years of education, and having higher pre-confinement HADS scores showed independent associations with greater HADS scores during the confinement (Table 3).
Table 3.
Results from the model showing the association of SCD-Q with anxiety/depression scores
| Predictor | Confinement HADS | |
|---|---|---|
| B value (95% CI) | p value | |
| SCD-Q, MyCog scores | 0.25 (0.096 to 0.41) | 0.002 |
| Age, years | − 0.19 (− 0.33 to − 0.055) | 0.006 |
| Sex (women) | 1.25 (− 0.12 to 2.63) | 0.074 |
| Education, years | − 0.31 (− 0.50 to − 0.12) | 0.001 |
| Pre-confinement HADS | 0.42 (0.27 to 0.56) | < 0.001 |
| Time difference | 0.38 (− 0.26 to 1.03) | 0.240 |
The unstandardized B represents the variation in confinement HADS scores with 1-unit variation in a given predictor
Abbreviations: HADS Hospital Anxiety and Depression Scale, CI confidence interval, SCD-Q Subjective Cognitive Decline Questionnaire
Association between the intensity of SCD and anxiety/depression measurements: model including Aβ status and stress-related variables
Following the inclusion of Aβ positivity, PSS, and BRS in the model, SCD-Q MyCog scores did not show a significant association with HADS scores. Aβ positivity and PSS scores, instead, showed significant associations with higher HADS scores irrespective of the pre-confinement anxiety/depression levels. BRS scores were not associated with HADS scores. Additionally, sex (women) was associated with presenting greater HADS scores during the confinement (Table 4).
Table 4.
Results from the model including Aβ positivity and stress-related measurements
| Predictor | Confinement HADS | |
|---|---|---|
| B value (95% CI) | p value | |
| SCD-Q, MyCog scores | 0.12 (− 0.01 to 0.25) | 0.069 |
| Aβ positivity | 1.95 (0.28 to 3.63) | 0.023 |
| PSS scores | 0.49 (0.43 to 0.56) | < 0.001 |
| BRS scores | − 0.28 (− 1.56 to 1.01) | 0.670 |
| Age, years | − 0.11 (− 0.24 to 0.008) | 0.068 |
| Sex (women) | 1.11 (0.006 to 2.26) | 0.049 |
| Education, years | − 0.12 (− 0.28 to 0.033) | 0.120 |
| Pre-confinement HADS | 0.01 (0.11 to 0.13) | 0.870 |
| Time difference | 0.4 (− 0.17 to 0.98) | 0.170 |
The unstandardized B represents the variation in confinement HADS scores with 1-unit variation in a given predictor
Abbreviations: HADS Hospital Anxiety and Depression Scale, CI confidence interval, Aβ amyloid-beta, SCD-Q Subjective Cognitive Decline Questionnaire, PSS Perceived Stress Scale, BRS Brief Resilience Scale
Mediation analyses
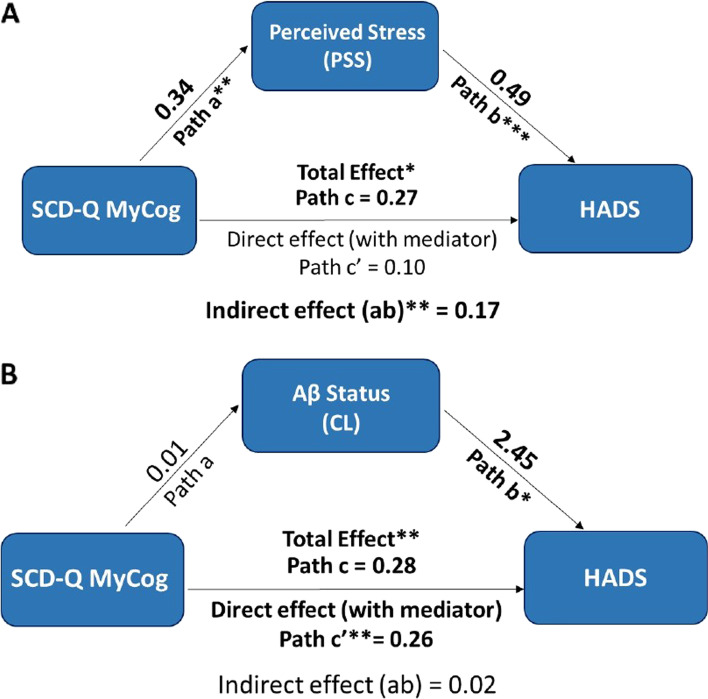
SCD-Q MyCog scores did not show a significant association with HADS scores after including Aβ status and stress-related variables in the model. Since both Aβ positivity and PSS were associated with HADS scores, we performed mediation analyses to investigate whether the effect of SCD-Q on anxiety/depression scores was mediated by Aβ positivity and/or stress perception. As illustrated in Fig. 2A, we found a significant partial mediation effect of stress perception on the association of SCD-Q MyCog scores with confinement HADS scores corresponding to 63% of the total association between the two variables (B = 0.17, 95% CI = 0.03 to 0.31, p = 0.01). Amyloid-β positivity, instead, did not show a mediating role between SCD-Q MyCog and HADS scores (B = 0.02, 95% CI = − 0.01 to 0.06, p = 0.45, respectively, see Fig. 2B).
Fig. 2.
Schematic illustration of the mediation analyses investigating the link between SCD and anxiety/depression scores. Regression coefficients for paths a, b, c, c′, and ab are reported in the figure. The total effect of SCD-Q MyCog on HADS (path c) is shown as the summation of the direct effect adjusted by the given mediator (c′) and the indirect effect (ab). The analyses were adjusted by age, sex, years of education, pre-confinement HADS scores, and time difference between the pre-confinement and confinement assessments. *p < 0.05, **p < 0.01, ***p < 0.001. A The results of the mediation analysis investigating the role of PSS as a mediator between SCD-Q MyCog scores and HADS scores. The indirect effect (path ab) was statistically significant (p = 0.01) corresponding to 63% of the total association between the two variables. B The results of the mediation analysis investigating the role of Aβ as a mediator between SCD-Q MyCog and HADS scores. The indirect effect (path ab) did not show a statistically significant effect (p = 0.45). Abbreviations: SCD-Q, Subjective Cognitive Decline Questionnaire; PSS, Perceived Stress Scale; HADS, Hospital Anxiety and Depression Scale; Aβ, amyloid-beta; CL, Centiloid
Sensitivity analyses
Models with SCD status
We repeated our main statistical analysis using the dichotomous SCD variable (participants classified as having SCD vs. without SCD) that is often used in the SCD field and in clinical practice [41]. The results showed that, unlike continuous SCD-Q scores, SCD status did not show an association with HADS scores during the confinement (95% CI = − 0.38 to 2.58, B = 1.10, p = 0.14).
SCD plus criteria-persistent SCD and age at onset of SCD after 60 years old
Given that the SCD-Q and SCD classification was performed based on pre-confinement evaluations, we repeated our main analysis with those participants that have a follow-up evaluation (N = 219) and were classified as SCD (N = 39). Persistent SCD (classified as SCD in both time points) was associated with greater HADS scores irrespective of pre-confinement HADS scores (95% CI = 0.07 to 3.73, B = 1.90, p = 0.042).
In addition, we repeated our models excluding those participants classified as SCD and aged below 60 (N = 27). The main results remained unchanged: the categorical SCD variable did not show a significant association with HADS scores (95% CI = − 0.15 to 3.66, B = 1.75, p = 0.071). Higher SCD-Q scores, instead, were associated with confinement HADS scores irrespective of pre-confinement anxiety/depression levels (95% CI = 0.13 to 0.52, B = 0.32, p = 0.001).
Differences in worries and lifestyle changes between participants with SCD that sought medical help vs. those that did not seek medical help
Within the group of participants classified as having SCD, we investigated whether those that sought medical help due to the perceived decline in cognition showed any differences in confinement-related worries and lifestyle changes than the ones that did not seek medical help (Fig. 1B). The rationale behind this sensitivity analysis is that seeking medical help is linked to worrying about the subjective decline in cognition [12, 47]. The two groups did not show any differences in worries about access to primary products (X2(1) = 0.04; p = 0.84), self-protection materials (X2(1) = 1.37; p = 0.24), or economic situation (X2(1) = 0.005; p = 0.94). We also did not observe any differences between the groups in changes in sleep duration (X2(1) = 0.18; p = 0.67), sleep quality (X2(1) = 0.68; p = 0.41), or eating habits (X2(1) = 1.005; p = 0.32) during the confinement.
Models adjusted by the change in sleep duration
We adjusted our main models by the change in sleep duration during the confinement. Following this adjustment, previously reported associations between SCD-Q and HADS scores did not change. Additionally, change in sleep duration showed a significant association with confinement HADS scores irrespective of pre-confinement anxiety/depression levels (95% CI = 1.49 to 5.35, B = 3.42, p < 0.001).
Discussion
In the current study, we investigated the association of the intensity of SCD with anxious/depressive symptoms during the COVID-19-related home confinement in a cohort of CU adults with an elevated risk of developing AD dementia. Our findings showed that higher levels of SCD, as measured by MyCog scores, were associated with presenting greater anxious/depressive symptoms during the COVID-19 confinement, irrespective of the pre-confinement anxiety/depression levels. Furthermore, Aβ positivity and higher stress perception showed independent associations with greater anxiety/depression scores. Mediation analyses revealed that the association of SCD intensity with higher anxiety/depression symptoms was mediated by perceived stress but not Aβ positivity. Lastly, we did not observe any differences in worries or lifestyle changes between the participants with or without SCD during the confinement.
During the COVID-19 pandemic, severe restrictions implemented to prevent the spread of the disease had negative effects on people’s mood, lifestyle, and mental health [33, 48]. In line with previous reports [49, 50], our sample showed increases in anxious/depressive symptoms between pre-confinement and confinement measurements. In addition, although the majority of the participants were not diagnosed with anxiety or depression during the confinement, the observed increase in anxiety/depression scores from pre-confinement (1.5 ± 5.8) was within the range of the minimum clinically significant difference suggested for the HADS [51, 52]. While participants having SCD had higher pre-confinement and confinement anxiety/depression scores than the ones without SCD, only the latter showed a minimum clinically significant difference from pre-confinement to confinement (mean change: 1.6 ± 5.9). Previous studies suggested an association of FH of dementia with greater cognitive complaints [53, 54] which could be mediated by depressive symptoms [55]. However, we did not observe any significant differences in HADS in those with and without FH of sporadic AD in our sample. Furthermore, having higher pre-confinement HADS scores independently contributed to explain higher HADS scores during the confinement. These results suggest that home confinement had a global influence on anxious/depressive symptomatology, including in individuals without cognitive complaints [2, 7, 36].
In line with our hypothesis, participants having higher levels of SCD showed higher anxious/depressive symptoms during the confinement irrespective of the pre-confinement anxiety/depression levels. These results are consistent with previous studies showing associations between SCD and affective symptoms [26, 56], specifically during the pandemic [3, 6, 7]. Previous studies performed during the pandemic focused on the differences in mental health outcomes between participants with SCD [7] and with mild cognitive impairment (MCI) or AD dementia [3, 6]. A strength of our study, compared to previous work, is that our cohort consisted of CU participants with a subgroup having SCD that allowed us to evaluate the differences in anxious/depressive symptoms between participants without and with SCD considering the intensity of the symptoms. When the categorical SCD variable was considered, having SCD did not show an association with anxiety/depression symptoms. When we performed the analysis using a longitudinal SCD classification (i.e., persistency of SCD over time), persistent SCD status was associated with HADS scores independent of pre-confinement HADS levels. The difference in the results depending on the approach used for SCD could reflect a limitation of using a single general question to determine the SCD status on a single time point. Considering that our main findings are consistent with the previous studies using SCD-Q MyCog scores as a continuous spectrum of SCD-related complaints [26, 40], these results suggest that the intensity of SCD could be a more sensitive approach to capture associations with anxiety/depression in middle-aged adults when only one time point assessment is available.
Our results showed that Aβ positivity measured 2.4 years before the confinement was associated with greater anxious/depressive symptoms during the confinement. These findings are consistent with the literature reporting associations between Aβ pathology and neuropsychiatric symptomatology in cognitively unimpaired older adults [19, 21–23], although the direction of causality between the two events is not clear. Previous studies suggested that anxious/depressive symptoms might facilitate or accelerate the expansion of Aβ and pathological progression of the disease [57, 58]. Conversely, specific neuropsychiatric symptoms, such as anxiety and depression, may represent an early manifestation of AD pathophysiology [23, 59]. Longitudinal studies are required to validate this hypothesis and unravel the potential causal relationship between the two events.
Higher levels of stress are often associated with negative emotions [60] and the development of affective symptoms, in particular during negative life events [61, 62]. The studies conducted during the pandemic suggested that confinement acts as the main stressor having an adverse impact on mental health [33, 35]. In our sample, we found that the association of higher levels of SCD with anxious/depressive symptoms was mediated by self-perceived stress during the confinement. This result is consistent with the reported associations between subjective cognitive complaints and stress perception in older adults [25, 33, 63]. The dysregulation in the hypothalamic-pituitary-adrenal axis may explain these associations such that stress-induced glucocorticoid secretion could have detrimental effects on memory performance [64, 65]. In agreement with these findings, our results suggest that people with higher levels of SCD are more vulnerable to stressors which may hasten the development of negative mental health outcomes. Overall, these findings support the previous research suggesting that interventions targeting stress alleviation can improve mental health outcomes in older adults with SCD [66].
Recent studies observed COVID-19 pandemic-related worries (e.g., related to getting the COVID-19 disease or socio-economic concerns) and lifestyle changes (e.g., changes in sleep or eating habits) in the general population [35, 48, 67] as well as in adults with SCD, MCI, or dementia [3, 6, 7]. We investigated whether the participants classified as SCD presented greater levels of worries and lifestyle changes during the confinement compared to those without SCD. Contrary to our hypothesis, the two groups did not show any differences in worries related to access to primary products, self-protection materials, or economic situation during the confinement. One possible explanation could be that the higher levels of worries observed in individuals with SCD [68] may be specific to cognitive decline-related concerns [12, 47] rather than reflecting a general trait of worrying in this population. However, our assessment did not include worries about the cognitive status during the confinement as in recent studies [3, 69]. Similarly, we did not observe significant differences in changes in sleep duration, sleep quality, or eating habits between the two groups. Previous studies found lower sleep duration [70] or sleep quality [71] in adults with SCD as compared to those without SCD using objective measurements. However, our questionnaire investigating the lifestyle changes was self-reported which could possibly explain the different results.
Lastly, we observed higher anxious/depressive symptoms during the COVID-19 confinement in younger participants and those with fewer years of education. Following the inclusion of Aβ positivity and stress-related measurements, sex (women) also showed an association with higher anxiety/depression scores. Overall, these results are in line with recent studies showing a higher prevalence of mental health problems in women, younger aged, and lower educated adults during the COVID-19 pandemic [2, 50, 72, 73]. Many studies attributed these findings to financial inequalities, poor economic status, and unemployment that have been shown as risk factors for developing anxiety and depression during the pandemic, particularly in these groups [2, 72]. Moreover, economic uncertainties and associated distress may exacerbate the negative consequences of the pandemic on the mental well-being of these populations in the long term [2, 74].
Limitations
Our study has several limitations. Firstly, our main results are reported on individuals recruited for a research study, and the majority have a FH of sporadic AD, linked previously with greater levels of cognitive complaints [53–55]. However, we did not observe any difference in SCD intensity between participants with or without FH of AD in our sample. Nevertheless, extrapolation to the general SCD population should not be done without further research. Secondly, among the ALFA+ participants that received the invitation to participate in the current study, only 59% of them responded. However, there were no significant differences in demographics or pre-confinement HADS scores between the responders and non-responders (data not shown). Furthermore, although the measurements of anxiety/depression were collected during the COVID-19-related home confinement, we cannot directly attribute the reported anxious/depressive symptoms in SCD to the confinement. Neuropsychiatric symptoms may also appear as part of the preclinical AD process [23, 59]. However, our HADS measurements were associated with the intensity of the confinement. In the same vein, the clinical measurements used in this study were self-reported, and objective measurements may provide different results. Yet, our results are supported by previous studies reporting higher anxiety, depression, or stress in similar cohorts during the confinement using subjective scales [3, 33]. Finally, our participants performed the SCD-Q before the pandemic, and SCD was not assessed during the COVID-19 confinement. Therefore, possible changes in the cognitive status of the participants from pre-confinement to confinement were not evaluated. However, with the available data collected after the confinement, we were able to repeat the main analysis only in the subsample that showed persistent SCD (N =39), and the results remained the same.
Conclusions
Our findings showed that adults at increased risk of AD and with higher levels of SCD are more vulnerable to adverse mental health outcomes during the COVID-19-related confinement. Furthermore, the mental health burden observed in adults with SCD may be mediated by stress perception. These results emphasize the importance of investigating the role of stress management in reducing the affective symptoms in the SCD population that could potentially lower the overall risk of cognitive impairment and dementia in the long term.
Supplementary Information
Additional file 1: Table S1. Selected questions from the online ad-hoc questionnaire on worries and lifestyle changes during the COVID-19 confinement.
Acknowledgements
This publication is part of the ALzheimer’s and FAmilies (ALFA) study. The authors would like to express their sincerest gratitude to ALFA project participants and relatives without whom this research would have not been possible. Collaborators of the ALFA study are Annabella Beteta, Raffaele Cacciaglia, Alba Cañas, Irene Cumplido, Ruth Dominguez, Maria Emilio, Carles Falcon, Laura Hernandez, Gema Huesa, Jordi Huguet, Paula Marne, Tania Menchón, Grégory Operto, Albina Polo, Blanca Rodríguez-Fernández, Sandra Pradas, Iman Sadeghi, Anna Soteras, Laura Stankeviciute, Marc Vilanova, and Natalia Vilor-Tejedor.
Abbreviations
- Aβ
Amyloid-beta
- AD
Alzheimer’s disease
- ALFA
ALzheimer’s and FAmilies
- BRS
Brief Resilience Scale
- CL
Centiloid
- FH
Family history
- HADS
Hospital Anxiety and Depression Scale
- PET
Positron emission tomography
- PSS
Perceived Stress Scale
- SCD
Subjective cognitive decline
- SCD-Q
Subjective Cognitive Decline Questionnaire
Authors’ contributions
Design/conceptualization of the study: EMAU, GSB, and MA. Acquisition, analysis, or interpretation of the data: MA, GSB, ABS, CPG, EP, MS, CD, SFJ, GS, JMDE, MSC, CM, KF, JLM, JDG, ORG, and EMAU. Drafting of the manuscript: MA, GSB, and EMAU. Revision of the manuscript and providing critical feedback: all authors. The authors read and approved the final manuscript.
Funding
This research was supported by Alzheimer’s Association research grants (AARG 2019-AARG-644641, AARG 2019-AARG-644641-RAPID) to EMAU. EMAU holds a “Ramón y Cajal” fellowship (RYC2018-026053-I). EMAU holds a grant from the Ministry of Science and Innovation (PID2019-111514RA-I00). The research leading to these results has received funding from the “la Caixa” Foundation (LCF/PR/GN17/10300004), the Alzheimer’s Association, and an international anonymous charity foundation through the TriBEKa Imaging Platform project. Additional support has been received from the Universities and Research Secretariat, Ministry of Business and Knowledge of the Catalan Government under grant 2017-SGR-892.
MSC receives funding from the European Research Council (ERC) under the European Union’s Horizon 2020 research and innovation programme (Grant Agreement No. 948677) and the Instituto de Salud Carlos III (PI19/00155) and a fellowship from “la Caixa” Foundation (ID 100010434) and the European Union’s Horizon 2020 research and innovation programme under the Marie Skłodowska-Curie grant agreement no. 847648 (LCF/BQ/PR21/11840004).
Availability of data and materials
The data supporting the results of the current study are not publically available but may be available upon a reasonable request.
Declarations
Ethics approval and consent to participate
The ALFA+ study protocol and informed consent were approved by the Independent Ethics Committee “Parc de Salut Mar” (ALFA-FPM-0311) and registered at ClinicalTrials.gov (Identifier: NCT02485730). The COVID-19 protocol (CovidImpact_BBRC2020) was approved on March 16, 2020 (2020/9255). The informed consent for the COVID-19 study was obtained from all participants via an online form. All methods were carried out in accordance with the ethical standards as laid down in the 1964 Declaration of Helsinki and its later amendments or comparable ethical standards.
Consent for publication
NA.
Competing interests
MSC has served as a consultant and on advisory boards for Roche Diagnostics International Ltd. and has given lectures in symposia sponsored by Roche Diagnostics, S.L.U, and Roche Farma, S.A.
Footnotes
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Muge Akinci and Gonzalo Sánchez-Benavides contributed equally to this work.
Contributor Information
Eider M. Arenaza-Urquijo, Email: earenaza@barcelonabeta.org
for the ALFA Study:
Annabella Beteta, Raffaele Cacciaglia, Alba Cañas, Irene Cumplido, Ruth Dominguez, Maria Emilio, Carles Falcon, Laura Hernandez, Gema Huesa, Jordi Huguet, Paula Marne, Tania Menchón, Grégory Operto, Albina Polo, Blanca Rodríguez-Fernández, Sandra Pradas, Iman Sadeghi, Anna Soteras, Laura Stankeviciute, Marc Vilanova, and Natalia Vilor-Tejedor
References
- 1.Nochaiwong S, Ruengorn C, Thavorn K, et al. Global prevalence of mental health issues among the general population during the coronavirus disease-2019 pandemic: a systematic review and meta-analysis. Sci Rep. 2021;11(1):10173. doi: 10.1038/s41598-021-89700-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Xiong J, Lipsitz O, Nasri F, et al. Impact of COVID-19 pandemic on mental health in the general population: a systematic review. J Affect Disord. 2020;277:55–64. doi: 10.1016/j.jad.2020.08.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.van Maurik IS, Bakker ED, van den Buuse S, et al. Psychosocial effects of corona measures on patients with dementia, mild cognitive impairment and subjective cognitive decline. Front Psychiatry. 2020;11:585686. doi: 10.3389/fpsyt.2020.585686. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Suárez-González A, Rajagopalan J, Livingston G, Alladi S. The effect of COVID-19 isolation measures on the cognition and mental health of people living with dementia: a rapid systematic review of one year of quantitative evidence. EClinicalMedicine. 2021;39. 10.1016/j.eclinm.2021.101047. [DOI] [PMC free article] [PubMed]
- 5.Amerio A, Aguglia A, Odone A, et al. COVID-19 pandemic impact on mental health of vulnerable populations. Acta Biomed. 2020;91, 95(9-S):–96. 10.23750/abm.v91i9-S.10112. [DOI] [PMC free article] [PubMed]
- 6.Di Santo SG, Franchini F, Filiputti B, Martone A, Sannino S. The effects of COVID-19 and quarantine measures on the lifestyles and mental health of people over 60 at increased risk of dementia. Front Psychiatry. 2020;11:578628. doi: 10.3389/fpsyt.2020.578628. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Soldevila-Domenech N, Forcano L, Boronat A, et al. Effects of COVID-19 home confinement on mental health in individuals with increased risk of Alzheimer’s disease. J Alzheimers Dis. 2021;79(3):1015–1021. doi: 10.3233/JAD-201408. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Ownby RL, Crocco E, Acevedo A, John V, Loewenstein D. Depression and risk for Alzheimer disease: systematic review, meta-analysis, and metaregression analysis. Arch Gen Psychiatry. 2006;63(5):530–538. doi: 10.1001/archpsyc.63.5.530. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Gimson A, Schlosser M, Huntley JD, Marchant NL. Support for midlife anxiety diagnosis as an independent risk factor for dementia: a systematic review. BMJ Open. 2018;8(4):e019399. doi: 10.1136/bmjopen-2017-019399. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Gatchel JR, Rabin JS, Buckley RF, et al. Longitudinal association of depression symptoms with cognition and cortical amyloid among community-dwelling older adults. JAMA Netw Open. 2019;2(8):e198964. doi: 10.1001/jamanetworkopen.2019.8964. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Dubois B, Hampel H, Feldman HH, et al. Preclinical Alzheimer’s disease: definition, natural history, and diagnostic criteria. Alzheimers Dement. 2016;12(3):292–323. doi: 10.1016/j.jalz.2016.02.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Jessen F, Amariglio RE, van Boxtel M, et al. A conceptual framework for research on subjective cognitive decline in preclinical Alzheimer’s disease. Alzheimers Dement. 2014;10(6):844–852. doi: 10.1016/j.jalz.2014.01.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Molinuevo JL, Rabin LA, Amariglio R, et al. Implementation of subjective cognitive decline criteria in research studies. Alzheimers Dement. 2017;13(3):296–311. doi: 10.1016/j.jalz.2016.09.012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Jessen F. Subjective and objective cognitive decline at the pre-dementia stage of Alzheimer’s disease. Eur Arch Psychiatry Clin Neurosci. 2014;264(1):3–7. doi: 10.1007/s00406-014-0539-z. [DOI] [PubMed] [Google Scholar]
- 15.Studart A, Nitrini R. Subjective cognitive decline: the first clinical manifestation of Alzheimer’s disease? Dement Neuropsychol. 2016;10(3):170–177. doi: 10.1590/S1980-5764-2016DN1003002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Perrotin A, Mormino EC, Madison CM, Hayenga AO, Jagust WJ. Subjective cognition and amyloid deposition imaging: a Pittsburgh Compound B positron emission tomography study in normal elderly individuals. Arch Neurol. 2012;69(2):223–229. doi: 10.1001/archneurol.2011.666. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Snitz BE, Lopez OL, McDade E, et al. Amyloid-β imaging in older adults presenting to a memory clinic with subjective cognitive decline: a pilot study. J Alzheimers Dis. 2015;48(Suppl 1):S151–S159. doi: 10.3233/JAD-150113. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Jang JY, Ho JK, Blanken AE, Dutt S, Nation DA. Affective neuropsychiatric symptoms as early signs of dementia risk in older adults. J Alzheimers Dis. 2020;77(3):1195–1207. doi: 10.3233/JAD-200190. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Lavretsky H, Siddarth P, Kepe V, et al. Depression and anxiety symptoms are associated with cerebral FDDNP-PET binding in middle-aged and older nondemented adults. Am J Geriatr Psychiatry. 2009;17(6):493–502. doi: 10.1097/jgp.0b013e3181953b82. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Babulal GM, Ghoshal N, Head D, et al. Mood changes in cognitively normal older adults are linked to Alzheimer disease biomarker levels. Am J Geriatr Psychiatry. 2016;24(11):1095–1104. doi: 10.1016/j.jagp.2016.04.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Sannemann L, Schild AK, Altenstein S, et al. Neuropsychiatric symptoms in at-risk groups for AD dementia and their association with worry and AD biomarkers—results from the DELCODE study. Alzheimers Res Ther. 2020;12(1):131. doi: 10.1186/s13195-020-00701-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Krell-Roesch J, Lowe VJ, Neureiter J, et al. Depressive and anxiety symptoms and cortical amyloid deposition among cognitively normal elderly persons: the Mayo Clinic Study of Aging. Int Psychogeriatr. 2018;30(2):245–251. doi: 10.1017/S1041610217002368. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Donovan NJ, Locascio JJ, Marshall GA, et al. Longitudinal association of amyloid beta and anxious-depressive symptoms in conitively normal older adults. Am J Psychiatry. 2018;175(6):530–537. doi: 10.1176/appi.ajp.2017.17040442. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Hill NL, Mogle J, Wion R, et al. Subjective cognitive impairment and affective symptoms: a systematic review. Gerontologist. 2016;56(6):e109–e127. doi: 10.1093/geront/gnw091. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Jenkins A, Tree J, Tales A. Distinct profile differences in subjective cognitive decline in the general public are associated with metacognition, negative affective symptoms, neuroticism, stress, and poor quality of life. J Alzheimers Dis. 2021;80(3):1231–1242. doi: 10.3233/JAD-200882. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Pavisic IM, Lu K, Keuss SE, et al. Subjective cognitive complaints at age 70: associations with amyloid and mental health. J Neurol Neurosurg Psychiatry. 2021;24. 10.1136/jnnp-2020-325620. [DOI] [PMC free article] [PubMed]
- 27.Pietrzak RH, Maruff P, Woodward M, et al. Mild worry symptoms predict decline in learning and memory in healthy older adults. Am J Geriatr Psychiatry. 2012;20(3):266–275. doi: 10.1097/JGP.0b013e3182107e24. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Liew TM. Subjective cognitive decline, anxiety symptoms, and the risk of mild cognitive impairment and dementia. Alzheimers Dement. 2020;16(S6):e036611. doi: 10.1002/alz.036611. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Desai R, Whitfield T, Said G, et al. Affective symptoms and risk of progression to mild cognitive impairment or dementia in subjective cognitive decline: a systematic review and meta-analysis. Ageing Res Rev. 2021;71:101419. doi: 10.1016/j.arr.2021.101419. [DOI] [PubMed] [Google Scholar]
- 30.Hill NL, Bhargava S, Bratlee-Whitaker E, Turner JR, Brown MJ, Mogle J. Longitudinal relationships between subjective cognitive decline and objective memory: depressive symptoms mediate between-person associations. J Alzheimers Dis. 2021;20. 10.3233/JAD-210230. [DOI] [PMC free article] [PubMed]
- 31.Wang SM, do Han K, Kim NY, et al. Late-life depression, subjective cognitive decline, and their additive risk in incidence of dementia: a nationwide longitudinal study. PLoS One. 2021;16(7):e0254639. doi: 10.1371/journal.pone.0254639. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Jacob L, Haro JM, Koyanagi A. Physical multimorbidity and subjective cognitive complaints among adults in the United Kingdom: a cross-sectional community-based study. Sci Rep. 2019;9(1):12417. doi: 10.1038/s41598-019-48894-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Podlesek A, Komidar L, Kavcic V. The relationship between perceived stress and subjective cognitive decline during the COVID-19 epidemic. Front Psychol. 2021;12:647971. doi: 10.3389/fpsyg.2021.647971. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Xin M, Luo S, She R, et al. Negative cognitive and psychological correlates of mandatory quarantine during the initial COVID-19 outbreak in China. Am Psychol. 2020;75(5):607–617. doi: 10.1037/amp0000692. [DOI] [PubMed] [Google Scholar]
- 35.Stanton R, To QG, Khalesi S, et al. Depression, anxiety and stress during COVID-19: associations with changes in physical activity, sleep, tobacco and alcohol use in Australian adults. Int J Environ Res Public Health. 2020;17(11):4065. doi: 10.3390/ijerph17114065. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Manca R, De Marco M, Venneri A. The impact of COVID-19 infection and enforced prolonged social isolation on neuropsychiatric symptoms in older adults with and without dementia: a review. Front Psychiatry. 2020;11:585540. doi: 10.3389/fpsyt.2020.585540. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Molinuevo JL, Gramunt N, Gispert JD, et al. The ALFA project: a research platform to identify early pathophysiological features of Alzheimer’s disease. Alzheimers Dement (N Y) 2016;2(2):82–92. doi: 10.1016/j.trci.2016.02.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Arenaza-Urquijo EM, Salvadó G, Operto G, et al. Association of years to parent’s sporadic onset and risk factors with neural integrity and Alzheimer biomarkers. Neurology. 2020;95(15):e2065–e2074. doi: 10.1212/WNL.0000000000010527. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Ministerio de Sanidad. Orden SND/399/2020, de 9 de Mayo, Para La Flexibilización de Determinadas Restricciones de Ámbito Nacional, Establecidas Tras La Declaración Del Estado de Alarma En Aplicación de La Fase 1 Del Plan Para La Transición Hacia Una Nueva Normalidad. Vol BOE-A-2020-4911.; 2020:31998-32026. Accessed April 28, 2022. https://www.boe.es/eli/es/o/2020/05/09/snd399.
- 40.Rami L, Mollica MA, García-Sanchez C, et al. The Subjective Cognitive Decline Questionnaire (SCD-Q): a validation study. J Alzheimers Dis. 2014;41(2):453–466. doi: 10.3233/JAD-132027. [DOI] [PubMed] [Google Scholar]
- 41.Sánchez-Benavides G, Suárez-Calvet M, Milà-Alomà M, et al. Amyloid-β positive individuals with subjective cognitive decline present increased CSF neurofilament light levels that relate to lower hippocampal volume. Neurobiol Aging. 2021;104:24–31. doi: 10.1016/j.neurobiolaging.2021.02.026. [DOI] [PubMed] [Google Scholar]
- 42.Zigmond AS, Snaith RP. The Hospital Anxiety and Depression Scale. Acta Psychiatr Scand. 1983;67(6):361–370. doi: 10.1111/j.1600-0447.1983.tb09716.x. [DOI] [PubMed] [Google Scholar]
- 43.Cohen S, Kamarck T, Mermelstein R. A global measure of perceived stress. J Health Soc Behav. 1983;24(4):385–396. doi: 10.2307/2136404. [DOI] [PubMed] [Google Scholar]
- 44.Smith BW, Dalen J, Wiggins K, Tooley E, Christopher P, Bernard J. The brief resilience scale: assessing the ability to bounce back. Int J Behav Med. 2008;15(3):194–200. doi: 10.1080/10705500802222972. [DOI] [PubMed] [Google Scholar]
- 45.Klunk WE, Koeppe RA, Price JC, et al. The Centiloid Project: standardizing quantitative amyloid plaque estimation by PET. Alzheimers Dement. 2015;11(1):1–15.e4. doi: 10.1016/j.jalz.2014.07.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Salvadó G, Molinuevo JL, Brugulat-Serrat A, et al. Centiloid cut-off values for optimal agreement between PET and CSF core AD biomarkers. Alzheimers Res Ther. 2019;11(1):27. doi: 10.1186/s13195-019-0478-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Jessen F, Amariglio RE, Buckley RF, et al. The characterisation of subjective cognitive decline. Lancet Neurol. 2020;19(3):271–278. doi: 10.1016/S1474-4422(19)30368-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Ammar A, Trabelsi K, Brach M, et al. Effects of home confinement on mental health and lifestyle behaviours during the COVID-19 outbreak: insights from the ECLB-COVID19 multicentre study. Biol Sport. 2021;38(1):9–21. doi: 10.5114/biolsport.2020.96857. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Brooks SK, Webster RK, Smith LE, et al. The psychological impact of quarantine and how to reduce it: rapid review of the evidence. Lancet. 2020;395(10227):912–920. doi: 10.1016/S0140-6736(20)30460-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.O’Connor RC, Wetherall K, Cleare S, et al. Mental health and well-being during the COVID-19 pandemic: longitudinal analyses of adults in the UK COVID-19 Mental Health & Wellbeing study. Br J Psychiatry. 2020;21:1–8. doi: 10.1192/bjp.2020.212. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Puhan MA, Frey M, Büchi S, Schünemann HJ. The minimal important difference of the hospital anxiety and depression scale in patients with chronic obstructive pulmonary disease. Health Qual Life Outcomes. 2008;6:46. doi: 10.1186/1477-7525-6-46. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Lemay KR, Tulloch HE, Pipe AL, Reed JL. Establishing the minimal clinically important difference for the Hospital Anxiety and Depression Scale in patients with cardiovascular disease. J Cardiopulm Rehabil Prev. 2019;39(6):E6–E11. doi: 10.1097/HCR.0000000000000379. [DOI] [PubMed] [Google Scholar]
- 53.Vlachos GS, Cosentino S, Kosmidis MH, et al. Prevalence and determinants of subjective cognitive decline in a representative Greek elderly population. Int J Geriatric Psychiatry. 2019;34(6):846–854. doi: 10.1002/gps.5073. [DOI] [PubMed] [Google Scholar]
- 54.Wolfsgruber S, Kleineidam L, Weyrauch AS, et al. Relevance of subjective cognitive decline in older adults with a first-degree family history of Alzheimer’s disease. J Alzheimers Dis. 2022;11. 10.3233/JAD-215416. [DOI] [PubMed]
- 55.Rue AL, Small G, McPherson S, Komo S, Matsuyama SS, Jarvik LF. Subjective memory loss in age-associated memory impairment: family history and neuropsychological correlates. Aging Neuropsychol Cognit. 1996;3(2):132–140. doi: 10.1080/13825589608256618. [DOI] [Google Scholar]
- 56.Sabatini S, Woods RT, Ukoumunne OC, Ballard C, Collins R, Clare L. Associations of subjective cognitive and memory decline with depression, anxiety, and two-year change in objectively-assessed global cognition and memory. Neuropsychol Dev Cogn B Aging Neuropsychol Cogn. 2021;11:1–27. doi: 10.1080/13825585.2021.1923634. [DOI] [PubMed] [Google Scholar]
- 57.Hanseeuw BJ, Jonas V, Jackson J, et al. Association of anxiety with subcortical amyloidosis in cognitively normal older adults. Mol Psychiatry. 2020;25(10):2599–2607. doi: 10.1038/s41380-018-0214-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Peters ME, Lyketsos CG. Beyond memory: a focus on the other neuropsychiatric symptoms of dementia. Am J Geriatr Psychiatry. 2015;23(2):115–118. doi: 10.1016/j.jagp.2014.11.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59.Ng KP, Chiew H, Rosa-Neto P, Kandiah N, Ismail Z, Gauthier S. Associations of AT(N) biomarkers with neuropsychiatric symptoms in preclinical Alzheimer’s disease and cognitively unimpaired individuals. Transl Neurodegener. 2021;10(1):11. doi: 10.1186/s40035-021-00236-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Scott SB, Sliwinski MJ, Blanchard-Fields F. Age differences in emotional responses to daily stress: the role of timing, severity, and global perceived stress. Psychol Aging. 2013;28(4):1076–1087. doi: 10.1037/a0034000. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.Schneiderman N, Ironson G, Siegel SD. Stress and health: psychological, behavioral, and biological determinants. Annu Rev Clin Psychol. 2005;1:607–628. doi: 10.1146/annurev.clinpsy.1.102803.144141. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62.Lindert J, Lee LO, Weisskopf MG, McKee M, Sehner S, Spiro A. Threats to belonging—stressful life events and mental health symptoms in aging men—a longitudinal cohort study. Front Psychiatry. 2020;11 Accessed May 2, 2022. https://www.frontiersin.org/article/10.3389/fpsyt.2020.575979. [DOI] [PMC free article] [PubMed]
- 63.Steinberg SI, Negash S, Sammel MD, et al. Subjective memory complaints, cognitive performance, and psychological factors in healthy older adults. Am J Alzheimers Dis Other Dement. 2013;28(8):776–783. doi: 10.1177/1533317513504817. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64.Potter GG, Hartman M, Ward T. Perceived stress and everyday memory complaints among older adult women. Anxiety Stress Coping. 2009;22(4):475–481. doi: 10.1080/10615800802449610. [DOI] [PubMed] [Google Scholar]
- 65.Canet G, Hernandez C, Zussy C, Chevallier N, Desrumaux C, Givalois L. Is AD a stress-related disorder? Focus on the HPA axis and its promising therapeutic targets. Front Aging Neurosci. 2019;11:269. doi: 10.3389/fnagi.2019.00269. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 66.Marchant NL, Barnhofer T, Klimecki OM, et al. The SCD-Well randomized controlled trial: effects of a mindfulness-based intervention versus health education on mental health in patients with subjective cognitive decline (SCD) Alzheimers Dement (N Y) 2018;4:737–745. doi: 10.1016/j.trci.2018.10.010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 67.Reppas-Rindlisbacher C, Finlay JM, Mahar AL, et al. Worries, attitudes, and mental health of older adults during the COVID-19 pandemic: Canadian and U.S. perspectives. J Am Geriatr Soc. 2021;69(5):1147–1154. doi: 10.1111/jgs.17105. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 68.Schwarz C, Lange C, Benson GS, et al. Severity of subjective cognitive complaints and worries in older adults are associated with cerebral amyloid-β load. Front Aging Neurosci. 2021;13:675583. doi: 10.3389/fnagi.2021.675583. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 69.Bakker ED, van Maurik IS, Mank A, et al. Psychosocial effects of COVID-19 measures on (pre-)dementia patients during second lockdown. J Alzheimers Dis. 2022;86(2):931–939. doi: 10.3233/JAD-215342. [DOI] [PubMed] [Google Scholar]
- 70.Bubbico G, Di Iorio A, Lauriola M, et al. Subjective cognitive decline and nighttime sleep alterations, a longitudinal analysis. Front Aging Neurosci. 2019;11:142. doi: 10.3389/fnagi.2019.00142. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 71.Lauriola M, Esposito R, Pizzi SD, et al. Sleep changes without medial temporal lobe or brain cortical changes in community-dwelling individuals with subjective cognitive decline. Alzheimers Dement. 2017;13(7):783–791. doi: 10.1016/j.jalz.2016.11.006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 72.Varma P, Junge M, Meaklim H, Jackson ML. Younger people are more vulnerable to stress, anxiety and depression during COVID-19 pandemic: a global cross-sectional survey. Prog Neuro-Psychopharmacol Biol Psychiatry. 2021;109:110236. doi: 10.1016/j.pnpbp.2020.110236. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 73.Benke C, Autenrieth LK, Asselmann E, Pané-Farré CA. Lockdown, quarantine measures, and social distancing: associations with depression, anxiety and distress at the beginning of the COVID-19 pandemic among adults from Germany. Psychiatry Res. 2020;293:113462. doi: 10.1016/j.psychres.2020.113462. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 74.Ng KH, Agius M, Zaman R. The global economic crisis: effects on mental health and what can be done. J R Soc Med. 2013;106(6):211–214. doi: 10.1177/0141076813481770. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Additional file 1: Table S1. Selected questions from the online ad-hoc questionnaire on worries and lifestyle changes during the COVID-19 confinement.
Data Availability Statement
The data supporting the results of the current study are not publically available but may be available upon a reasonable request.