Abstract
In our previous studies, a novel antimutagenic compound, 2-ethoxy-3-(3,7,11,15-tetramethylhexadec-2-ethyl) naphthaquinone-1,4-dione (ethoxy-substituted phylloquinone; ESP) from spinach was characterized and mechanism contributing to its antimutagenicity was deduced. In the current study, anti-proliferative activity of ESP was assessed in lung cancer (A549) cells using MTT [3-(4,5-dimethylthiazole-2yl)-2,5-diphenyl tetrazolium bromide], clonogenic assays and cell cycle analysis. ESP treatment showed selective cytotoxicity against lung cancer cells and no cytotoxicity in normal lung (WI38) cells. Cell cycle analysis revealed that ESP treatment arrests A549 cell population in G2-M phase. In-silico analysis indicated positive drug-likeness features of ESP. Molecular docking showed H-bonding and hydrophobic interactions between ESP and B-DNA dodecamer residues at minor groove. SWATH-MS (Sequential Window Acquisition of All Theoretical Mass Spectra) based proteomic analysis indicated down-regulation of proteins involved in EGFR signaling, NEDDylation and other metabolic pathways and up-regulation of tumor suppressor (STAT1 and NDRG1) proteins. Treatment of spinach powder with gamma radiation (5–20 kGy) from cobalt (Co-60) enhanced the extractability of ESP up to 4.4-fold at the highest dose of 20 kGy. Scanning electron microscopy of spinach powder displayed decrease in smoothness and compactness with increase in radiation dose attributing to its enhanced extractability. Increase in the extractability of ESP with increasing radiation doses as measured by fluorescence intensity and dry weight basis was strongly correlated. Nonetheless, radiation treatment did not affect the functionality of ESP in terms of anti-proliferative and antimutagenic activities. Current findings thus highlight broad spectrum bioactivity of ESP from spinach, its underlying mechanism and applicability of radiation technology in enhancing extractability.
Supplementary Information
The online version contains supplementary material available at 10.1007/s13205-022-03264-6.
Keywords: Lung cancer cell, Molecular docking, SWATH-MS, Minor groove binding, Radiation technology
Introduction
Spinach (Spinacia oleracea) is an annual delicate herb native to south-western and central Asia being grown throughout the year at various locations. It is known to be a rich source of fibers, minerals (magnesium, manganese and iron) and vitamins such as phylloquinone (vitamin K1), carotene (β-carotene and lutein), α-tocopherol (vitamin E), and folate (vitamin B9) (Lester et al. 2013). The health benefits of spinach in terms of antioxidant, hepatoprotective, antiviral, anti-inflammatory and anticancer activities have been reported in earlier studies (Longnecker et al. 1997; Gupta and Singh 2006; Metha and Belemkar 2014). Recently, a novel antimutagenic compound was extracted from spinach leaf and characterized as 2-ethoxy-3-(3,7,11,15-tetramethylhexadec-2-ethyl) naphthaquinone-1,4-dione, in short named as ethoxy-substituted phylloquinone (ESP) (Fig. S1) (Kumar et al. 2016). The compound ESP was found to possess DNA minor grove binding ability and could inhibit cell division through modulation of regulatory metabolic steps leading to antimutagenicity and, therefore, a potential candidate molecule for exploring its anticancerous activity (Kumar et al. 2020).
Most of the anticancer drugs are cytotoxic to normal cells, making it essential to explore for newer potential compounds lacking such deleterious side effects to normal cells (Teerasripreecha et al. 2012). Diverse mechanisms have been earlier proposed to explain the anticancerous activity displayed by naphthoquinones which includes p53 regulation, repression/inhibition of telomerase activity, apoptosis induction through ER stress, regulation of tumor associated inflammation, MALT1 inhibition and ROS generation (Lim et al. 2015; Joerger and Fersht 2016; Corazzari et al. 2017; Pereyra et al. 2019). In the current study, anti-proliferative activity of ESP was assessed in lung cancer (A549) cell line and compared with its cytotoxicity to normal lung cell line (WI38). Subsequently, possible molecular mechanism was elucidated using SWATH-MS (Sequential Window Acquisition of All Theoretical Mass Spectra) proteomic analysis of control and ESP treated cells. Lung cancer has been reported to rank second with respect to new cases and first for deaths among the prevalent cancers (Sung et al. 2021). In the most cases, lung carcinoma epithelial cells, or tissues composed of epithelial cells get transformed to malignant cells (White and Ruparelia 2020).
Treatment with ionizing radiation like gamma rays from cobalt (Co-60) is an approved non-thermal and eco-friendly technology for achieving various objectives such as food preservation and enhanced extractability of bioactive compounds from agro-products and industrial residues (Madureira et al. 2020). However, yield of bioactive compound could depend upon its sensitivity to the radiation treatment as well as structure of source. In case of spinach powder, it is mainly constituted by pectin, cellulose and hemicelluloses (Voragen et al. 2009). Radiation technology was employed to study its effect on the extractability of ESP from spinach. Dehydrated spinach powder is comparatively better stable as compared to fresh green leaves due to low moisture content and often used as functional ingredient in several products such as soups (~ 12%), quiche (~ 12%), chapati premixes (~ 5%), hummus (~ 5%), tortillas (~ 5%), noodles (~ 4%), ultra-filtered soft chesses (~ 1%), and salad dressings (Khan et al. 2015; Lee et al. 2017; El-Sayed 2020).
Current study deals with anti-proliferative activity of ESP isolated from spinach powder using lung cancer cell line as a model system including cytotoxicity evaluation in normal lung cell line. Further, to gain mechanistic insights proteomic analysis using SWATH-MS has been performed. Besides, efficacy of gamma radiation processing has been evaluated for enhancing the extractability of ESP from spinach powder and its impact on functionality.
Materials and methods
Extraction of ethoxy-substituted phylloquinone (ESP) from spinach
Spinach (Spinacia oleracea var. semi-savoy) leaves were lyophilized (Alpha 2–4 Freeze Dryer, Martin Christ, Germany) to obtain its fine powder. ESP was extracted from the spinach powder to study its extractability as reported earlier (Kumar et al. 2016). In brief, 0.5 g of spinach powder was suspended in 15 and 32 ml of 2-propanol and hexane (3:2 v/v), respectively, and 32 ml of milliQ water. This was sonicated (45 s), vortexed (10 min), and centrifuged (5000×g; 5 min at 4 °C). The upper layer was collected which contained quinones and dried at ambient temperature under vacuum (Concentrator 5301, Eppendorf, Germany). The residue was dissolved in hexane (10 ml) and further purified by solid-phase extraction (SPE) using silica gel columns (Waters Corporation, Milford, USA). The SPE column was preconditioned sequentially with 8 ml hexane and diethyl ether in the ratio of 93:3 (v/v) and hexane (100%). The extract (2 ml) was applied on the column, washed (8 ml of 100% hexane) and eluted [hexane and diethyl ether (93:3 v/v; 8 ml)]. This was treated with charcoal at 4 °C for overnight and centrifuged twice (10000×g; 10 min at 4 °C). The supernatant was dried at ambient temperature (26 ± 2 °C).
Thin-layer chromatography (TLC) for purification and quantification of ESP
Sample was spotted on TLC silica gel 60 F254 plates of 0.25 mm thickness (Merck, Armstadt, Germany), developed using solvent system (petroleum ether: chloroform: acetone:: 5:1:0.106) and image captured under ultraviolet ray (UV; 366 nm) (Muttenz, CAMAG, Switzerland). The fluorescent band (ESP) was quantified using ImageJ software (NIH, U.S.A.). Further, preparative TLC was also carried out similarly on silica plates of 0.5 mm thickness using same solvent system. The ESP band was scraped, suspended in n-hexane and diethyl ether (93: 3), centrifuged at 15000×g for 40 min to remove the silica particles, dried under vacuum and weighed for quantification. To confirm the purity of ESP, high-performance liquid chromatography (HPLC) (Waters, U.S.A.) using a reverse-phase C-18 analytical column (250 × 4.6 mm, 10 μm pore size) and photodiode array (PDA) detector was performed (Kumar et al. 2016).
Cell cultures and their growth conditions
Human lung cancer cells (A549) and normal cells (WI38) were maintained in DMEM (Dulbecco’s modified Eagle’s medium; HiMedia Pvt. Ltd., India), whereas, human TK6 lymphoblast cells were maintained in RPMI-1640 (Roswell Park Memorial Institute Medium; HiMedia Pvt. Ltd.), supplemented with 10% fetal bovine serum (FBS), 100 units/ml penicillin and 100 μg/ml streptomycin (HiMedia Pvt. Ltd.). The cells were incubated in 5% CO2 humidified at 37 °C for growth.
MTT assay to evaluate cytotoxicity of ESP
Cytotoxic effect of ESP was assessed in human lung cancer cells (A549) using MTT [3-(4,5-dimethylthiazole-2yl)-2,5-diphenyl tetrazolium bromide] dye conversion assay (Kumar et al. 2020). In principle, MTT dye (yellow colored) is reduced to an insoluble formazan crystal (blue colored) in metabolically active cells by mitochondrial succinate dehydrogenase. In brief, 0.2 million A549 cells were cultured in medium (DMEM; HiMedia) supplemented with FBS (10%) (Sigma, U.S.A.), and antibiotics (200 U/mL penicillin and 50 μg/mL streptomycin) in the presence of ESP (1.56 to 75 µM) in 24 well plate for this assay. Dimethyl sulfoxide [DMSO (0.1%)] served as solvent control. Cells were incubated in a humidified chamber [CO2 atmosphere (5%) and air (95%)] at 37ºC for 24 h. Cells were washed with phosphate buffered saline (PBS, 10 mM, pH 7.2) and incubated for 4 h with MTT dye (0.5 mg/mL). The formazan crystals formed were dissolved in DMSO and the absorbance was measured at 550 nm using a microplate reader (Bio-Tek Instruments, Vt., U.S.A.). Similarly, cytotoxicity of ESP was analyzed in normal lung (WI38) cells.
Time-dependent microscopic examination of A549 cells upon ESP treatment
Cytotoxicity analysis of ESP was also carried out by time-dependent morphological observation. In brief, ~ 5 × 103 A549 cells were seeded in 96-well plate. Next day, after addition of ESP (6.25, 12.5, 25 and 50 µM) and solvent control [DMSO (0.1%)], time-dependent images of the cells were captured at the magnification of 10 × and resolution of 72 dpi using an IncuCyte S3 imaging system (Japan) equipped with CCD camera at every 2 h interval for 22 h. Cells were maintained at 37 °C in 5% CO2 atmosphere environment during the experiment.
Clonogenic assay for colony forming capacity of A549 cells upon ESP treatment
Clonogenic assay was conducted as per the method described earlier (Maurya 2017). Cells were grown to ~ 80% confluency, trypsinized and seeded (~ 2 × 103 cells) in each well of a 6-well plate. ESP (6.25, 12.5, 25 and 50 µM) and solvent control [DMSO (0.1%)] was added and allowed to grow for 12 days. Next, the cells were washed with PBS and incubated with 0.5% methylene blue solution (dissolved in 50% ethanol) for 30 min. Later, the methylene blue in the plates was washed off with tap water (running) and allowed to dry at ambient temperature. The images of the clonogenic plate were captured using Gel Doc system and the numbers of colonies formed in the plates were quantified by ColonyCountJ, an ImageJ add-on program (Maurya 2017).
Cell cycle analysis
For cell cycle analysis, ~ 0.25 × 103 A549 cells cultured in medium was treated with ESP (12.5 µM) and solvent control [DMSO (0.1%)] in a 24 well plate as discussed earlier (Kumar et al. 2012). The cells were allowed to grow in the CO2 incubator for 48 h. Later, cells were trypsinized and collected by centrifugation (900×g for 4 min), washed twice with PBS (10 mM) and finally suspended in 1 mL of PI staining solution [propidium iodide (PI; 50 μg/mL), RNase (40 μg/mL), sodium citrate (0.1%) and Triton X-100 (0.1%)]. A total of 20,000 cells were acquired in a flow cytometer (Partec, Germany) and analyzed for cell cycle using FlowJo 10.6.2 software.
In-silico analysis for structural features, drug-likeness properties and interaction with DNA
To determine structural and drug-likeness properties, pKCSM and SWISS-ADME servers were used, respectively. The molecular docking study of the compound was carried out using AutoDock4 tool and Ligplot for predicting the interaction of ESP with B-DNA dodecamer d(CGCGAATTCGCG)2 (PDB ID: 1BNA) (Laskowski et al. 2011).
SWATH-MS-based proteomic analysis for differential expression of proteins upon ESP treatment
In SWATH analysis, non‐labelled proteins are digested with trypsin enzyme and the resulting peptides are analyzed by liquid chromatography (LC) coupled to a tandem mass spectrometer (TMS) operating in the data‐independent acquisition (DIA) mode where all ionized peptides within a specified mass range are systematically fragmented. Around 2 million cells treated with ESP (12.5 µM) were washed with PBS and lysed in chilled RIPA buffer, centrifuged at 12000×g for 15 min at 4 °C and supernatant was collected. Protein precipitation was performed by mixing with 4 volumes of pre-chilled acetone and incubating at -20 °C for 1 h. The suspension was centrifuged at 15000×g at 4 °C. Protein pellet was suspended in 0.1 M Tris–HCl with 8 M urea (pH 8.5) and quantified using Bradford assay. For SWATH-MS analysis, reduction, alkylation and trypsin digestion, library generation and data acquisition, database searches and peak extraction have been conducted as per protocol reported earlier (Ghose et al. 2019; Singh and Sharma 2020). The detail of SWATH-MS protocol is given in the supplementary file.
Radiation processing of spinach powder for improving extractability of ESP
Spinach powder was radiation processed (5–20 kGy) at food irradiation facility located at the institute. Dosimetry of the irradiator was conducted using a standard ceric-cerous dosimeter (5 mM). The absorbed dose was determined by potentiometry with ± 1% uncertainty based upon potential difference vs. dose calibration data (Kumar et al. 2015). The dose uniformity ration (DUR; Dosemax/Dosemin) was measured as 1.2. ESP was extracted from irradiated and non-irradiated spinach powder, separately, subjected to TLC and comparative extractability/yield was analyzed as discussed above.
Scanning electron microscopy (SEM) of irradiated spinach powder
Samples were analyzed using scanning electron microscope (AIS2100, SERON Inc., Korea) to observe the microscopic changes in the spinach powder due to radiation treatment. For SEM, spinach powder was spread over the double-sided conducting carbon tape, loose powder was blown using air blower and then coated with gold (Au) (15 nm) using plasma based sputter coater. The SEM micrographs of the samples were acquired at similar magnification (2500×) and acceleration voltage (20 kV) for the ease of comparing the recorded micrographs. All the micrographs were saved at 96 dpi resolution.
Evaluation of functionality of ESP from irradiated spinach powder
ESP from irradiated spinach powder was evaluated for functionality in terms of antimutagenic and anti-proliferative activities. Antimutagenic assay is performed using the heterozygous thymidine kinase (tk+/−) gene present in TK6 human lymphoblast cell line. Upon mutagen exposure, the cells become thymidine kinase enzyme deficient (tk−/−) mutant and thus can survive in medium containing trifluorothymidine (TFT, a thymidine analogue), which is toxic for the TK-proficient cells. The assay was performed as per the method described earlier (Kumar et al. 2016). MTT assay for ESP extracted from irradiated spinach powder was also conducted against lung cancer (A549) and normal lung (WI38) cells as discussed above.
Statistical analysis
Data were analyzed and represented in mean and standard deviations (SD). One-way ANOVA followed by Fisher’s least significant difference (LSD) at p ≤ 0.05 were performed using BioStat 2009 Professional 5.8.0.0 software (AnalystSoft Inc., Vancouver, BC, Canada). Anti-proliferative such as clonogenic and cytotoxicity (MTT) assays as well as time-dependent microscopic studies were performed in three independent sets, where each set had five replicates. Cell cycle and SWATH-MS analysis were conducted twice in triplicate. Extractability analysis and yield determination was performed in three sets in triplicates for each radiation dose. Other experiments pertaining to SEM and bioactivity analysis (antimutagenicity and anti-proliferative) was conducted twice in triplicates.
Results and discussion
In our previous study, an antimutagenic naphthoquinone, ethoxy-substituted phylloquinone (ESP) was isolated and characterized from spinach (Fig. S1) and its mechanism was elucidated (Kumar et al. 2016, 2020). In the current study, anti-proliferative effect of ESP against lung cancer (A549) cells was evaluated and possible mechanism was deduced. Lung cancer is one of the most prevalent types of cancer. Further, spinach powder was subjected to gamma radiation treatment to evaluate its efficacy in enhancing the extractability of ESP.
ESP displayed strong selective anti-proliferative activity against lung cancer cells
The MTT assay showed that ESP treatment led to a concentration dependent decrease in the lung cancer (A549) cell viability which indicated its cytotoxic effects (Fig. 1A). The viability was not found to be significantly affected at ≤ 6.25 µM concentrations (Fig. 1A). At 12.5, 25 and 37.5 µM concentrations of ESP, viability was found to be reduced to 57, 21 and 17%, respectively, whereas, at further higher concentrations of 50 and 75 µM, viability was only 12 ± 3% (Fig. 1A). Interestingly, ESP was found to be non-toxic to the normal lung (WI38) cells (Fig. 1B) even at high concentrations up to 1 mM. In our earlier study too, ESP was not found to have any cytotoxicity to normal human lymphoblast TK6 cells up to 2.5 mM concentration (Kumar et al. 2020). Thus, this compound displayed selective cytotoxicity to lung cancer cell line. Such differential cytotoxicity has earlier been reported by other researchers too for dietary herbal acteoside bioactive (Cheimonidi et al. 2018).
Fig. 1.

Assessment of cytotoxicity of ESP in lung cancer cells (A549) (A) and normal lung cells (WI38) (B) using MTT assay. Different letters (a–e) on the top of the bars showed significant differences among means (p ≤ 0.05)
To study how early ESP started displaying its effect, a time-dependent microscopic study of A549 cells in the presence of different concentrations of ESP was performed. The untreated A549 cells are adherent and have epithelial-like morphology, whereas, ESP treated cells displayed changes from adherent to non-adherent with cell shrinkage and round morphology (Fig. 2A). Our observation reveals a concentration dependent cytotoxicity of ESP in A549 cells (Fig. 2A) and at higher concentration (50 µM), it starts showing its effect from 4 h onwards (Fig. 2A). Also, it is clear that at end of the analysis, i.e., 22 h there was no difference between control and vehicle control but there was a significant reduction in the cell number in ESP treated group (Fig. 2A).
Fig. 2.
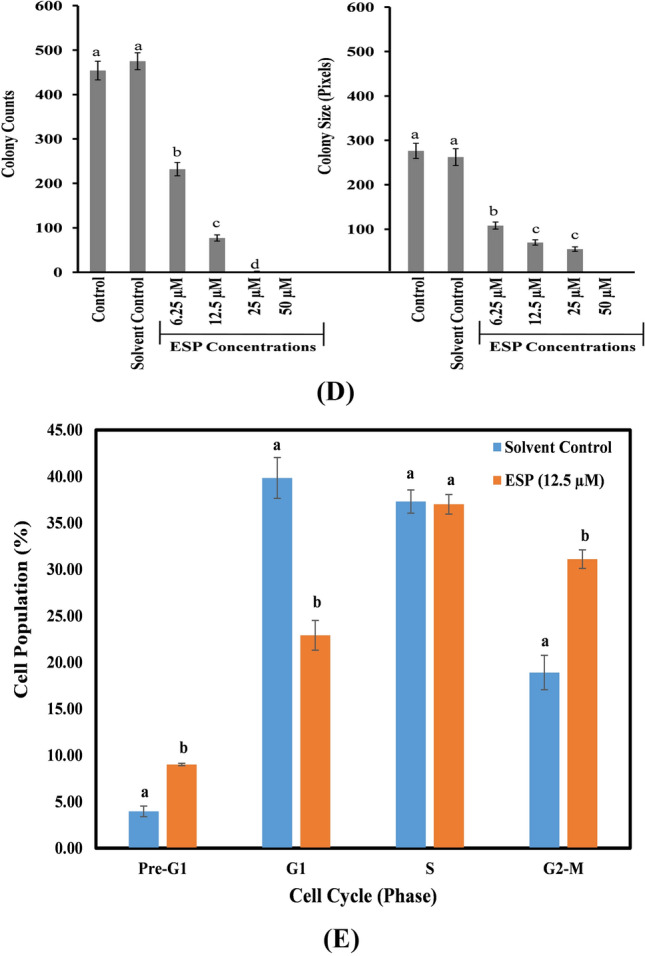
Time-dependent microscopic examination (A) and clonogenic assay of ESP against lung cancer (A549) cells (B) and normal lung (WI38) cells (C); Histogram showing effect of ESP on colony count and size of A549 cells (D), Histogram showing effect of ESP on A549 cells cell population (%) during cell cycle analysis by flow cytometry (E). Different letters (a–d) on the top of the bars showed significant differences among means (p ≤ 0.05)
Clonogenic assay is considered as gold standard assay for anti-proliferative study. This assay was performed to study the mitotic potential of the ESP treated A549 and WI38 cells. As shown in Fig. 2B, there was a concentration dependent inhibition of the macroscopic colony formation in A549 cells. The colony count in non-treated control cells was 465 ± 21 with size of 273 ± 17 pixels (Fig. 2B and D). At 6.25, 12.5, 25 and 50 µM concentrations of ESP, inhibition in colony formation was 55, 85, 99.5 and 100%, respectively, with significant reduction in colony size too (Fig. 2B and D). No significant inhibition in colony formation in normal lung WI38 cells was observed (Fig. 2C).
All these studies confirm that ESP has high anti-proliferative potential with preferential toxicity to the lung cancer cells. The findings of the time-dependent microscopy of A549 cells well corroborated with the MTT assay results. There was a strong correlation ('r' = 0.88) between MTT and clonogenic assays. However, the impact of ESP at same corresponding concentration during clonogenic assay was found to be more pronounced possibly due to prolonged treatment time of 12 days.
The cell cycle analysis revealed that ESP treatment decreases the G1 cell population and arrests the cell in the G2-M phase without any change on the S-phase of the cells cycle (Fig. 2E). There was also increase in the pre-G1 cell population because of ESP induced cell death (Fig. 2E). Naphthoquinone derivatives have been reported to exert anti-proliferative activity in cancer cell by cell cycle arrest at G2-M phase and induced cell death (Wang et al. 2019a, b).
In-silico analysis indicated ESP toxicity, bioavailability and interaction with DNA minor groove
In-silico analysis of ESP indicated its high intestinal absorption (~ 95%) and bioavailability score (0.85) as well as no expected toxicity in liver cells or mutagenicity in AMES test (Fig. S1). In higher system (rat), expected tolerance for ESP was found to be at higher side (Fig. S1). Also, ESP displayed selective cytotoxicity to cancer cells compared to normal cells. These findings about ESP propelled further exploration of its intracellular targets. Previously, ESP was reported as minor groove binder through different approaches (Kumar et al. 2020). In-silico binding energy of ESP and B-DNA was determined as -7.82 kcal/mol which validated the minor groove binding ability of this compound (Fig. 3A). Further, ESP was found to form a hydrogen bond (2.91 Å distance) with its one of the quinone group at C4 and sixth deoxyadenosine (deoxyA) residue of dodecamer B-DNA (chain A) at N3-H group. Hydrophobic interactions of ESP with other B-DNA (chain A and B) residues were also predicted as shown in Fig. 3B. Minor groove binders may interact at the replication and transcription site of disassembled chromatin of cancer cells (Zihlif et al. 2010). These compounds have been reported to affect the expression profile by inhibiting DNA binding proteins such as transcription factors, DNA helicase, topoisomerase I and II, E2F1 and replication protein A possibly through direct steric interference leading to cytotoxic impact on cancer cells (Chen et al. 1993; Soderlind et al. 1999; Zhang et al. 2011).
Fig. 3.
Interaction of ESP at minor groove of B-DNA (A) and possible site of interactions at minor groove (B) using Chain A—deoxyC1GCGAATTCGCG12; Chain B—deoxyC13GCGAATTCGCG24 ;  H-bond;
H-bond;  Hydrophobic interactions
Hydrophobic interactions
Impact of ESP on SWATH-MS-based protein expression profile of lung cancer (A549) cells
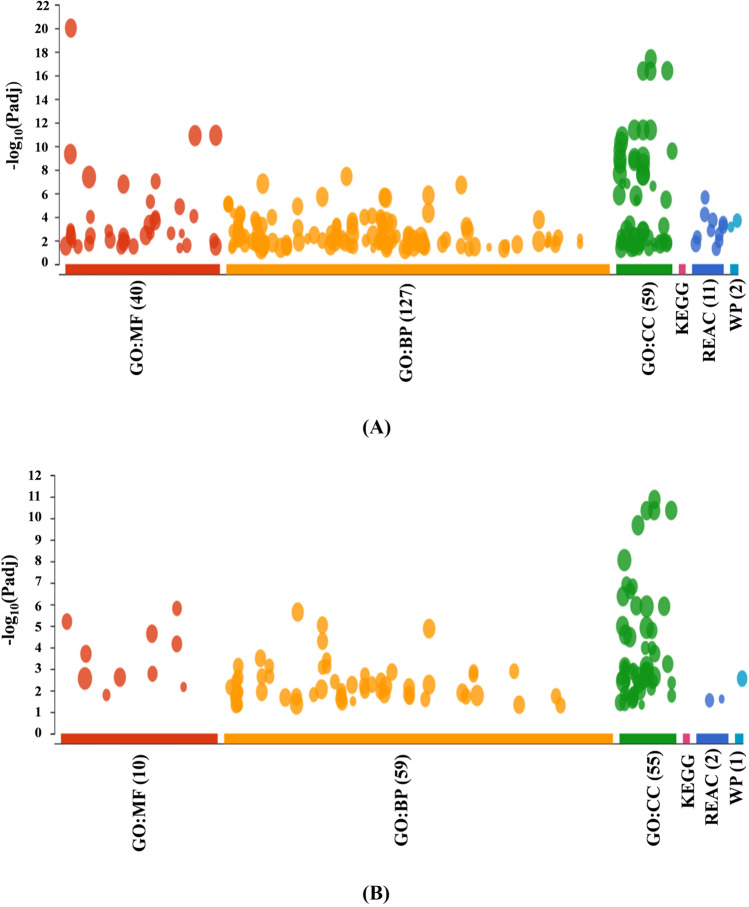
SWATH-MS has advantages of increased proteome depth due to combined impact of high reproducibility and sensitivity of targeted methods like selected reaction monitoring/multiple reaction monitoring (SRM/MRM) (Collins et al. 2017). A total of 1347 protein groups were identified in the spectral ion library at 1% FDR (false discovery rate) and 1141 proteins were identified and quantified in the SWATH-MS analysis. Around 783 proteins were down-regulated, whereas, 358 proteins were up-regulated in ESP treated as compared to untreated (control) A549 lung cancer cells. When ≥ twofold down-regulated proteins were subjected for functional enrichment analysis using gProfiler for the Gene Ontology (GO) and biological pathways, it was found that there was a total 40 GO-MF (Molecular Function), 127 GO-BP (Biological Process), 59 GO-CC (Cellular Component), and 1 KEGG, 11 Reac (Reactome), 2 WP (Wikipathways) biological pathways were affected (Fig. 4). Similarly, when ≥ twofold up-regulated proteins were subjected for the same analysis, there was only 10 GO-MF, 59 GO-BP, 55 GO-CC and 1 KEGG, 2 Reac and 1 WP biological pathways were affected (Fig. 4). Differentially expressed proteins were quite high in number. Thus, for understanding the mechanism of anticancerous activity of ESP, the proteins which showed down and up-regulated by ~ 20 and fivefold, respectively, and have been reported for their role in cancer were selected.
Fig. 4.
Functional enrichment analysis using gProfiler for the Gene Ontology (GO) and biological pathways using down-regulated (A) and up-regulated (B) proteins
Among the down-regulated protein, RalA (a Ras-related oncogenic protein), reported in EGFR signaling pathway was found to be 67-fold down-regulated (Table 1). Activated RalA (GTD bound) interacts with RalBP1 (Ral binding protein) and negatively regulates Rac-1 and Cdc4. Other effector proteins of RelA includes PLD (phospholipase D), Sec3 and Exo70 (components of exocyst complex), Filamin A (formation of filopodia) and ZONAB (ZO-1-associated nucleic acid binding protein). Thus, RalA affect cell proliferation, cell survival, tumor cell invasion and actin organization through these effector proteins and other signaling pathways (Moghadam et al. 2017). The NEDD8 protein (~ 23-fold down-regulated) is involved in NEDDylation pathway (Table 1) where ubiquitin ligase activity of the SCF (Skp-1, cullin and F-box protein) complex requires the conjugation of NEDD8 to cullins, i.e., scaffold proteins. Down-regulation of NEDD8 conjugation inhibits the ligase activity of SCF complexes required for cell cycle progression (Tanaka et al. 2012). Thus, in our study too, significant cell cycle arrest at G2-M phase was observed and has been discussed above. The expression of DDX21 protein, induced by EGF signaling in cancer cells is required for c-jun phosphorylation and AP-1 activity as well as rRNA processing and thus promotes tumorigenesis. In the current study, this protein was found to be down-regulated (21-fold) but has been reported to enhance the oncogenic activity of Ras when over-expressed (Table 1).
Table 1.
SWATH-MS analysis to understand differential expression of relevant proteins in lung cancer (A549) cells upon ESP treatment leading to anti-proliferative effect
| (A) Down-regulated proteins (≥ 20-fold) in A549 cell after ESP treatment | |||
|---|---|---|---|
| Proteins | Fold change | Function and role related to cancer development | References |
| RalA | 67 | Ras-related protein, RAL-A involve in EGFR signaling pathway; Activation of RalA signaling considered critical step in Ras-induced transformation and tumorigenesis | Lim et al. (2005) |
| NEDD8 | 23 | Involved in NEDDylation pathway and required for cell proliferation; Overexpressed in cancer cells | Guardavaccaro and Pagano (2004) |
| STMN 1 | 21 | Microtubule-destabilizing protein regulates the dynamics of microtubules; Regarded as an oncogene and overexpresses in cancer cells | Bao et al. ( 2017) |
| DDX21 | 21 | DDX21 (DExD-Box Helicase 21) promotes tumorigenesis; Induced by EGF signaling; Enhance the oncogenic activity of Ras | Zhang et al. (2014) |
| EIF1 | 22 | Eukaryotic translation initiation factor 1; Silencing of eIF1reduced proliferation and the clonogenicity of cancer (HCT116) cells | Golob-Schwarzl et al. (2017) |
| EIF3G | 33 | Eukaryotic translation initiation factor 3 subunit G is one of the subunits of EIF3, reported to be involved in the process of translation re-initiation; Positive relationship with chemoresistance and expression in breast and colon cancer cells | Yang et al. (2018) |
| ELOC | 38 | Elongin-C activates elongation by RNA polymerase II | Schoenfeld et al. (2000) |
| TFAM | 23 | Mitochondrial Transcription Factor A required for replication, transcription and maintenance of mitochondrial DNA; Down-regulation inhibits the tumorigenesis of non-small cell lung cancer by reducing cellular bioenergetics | Xie et al. (2016) |
| SRSF3 | 48 | Ser/Arg-rich splicing factor 3 are RNA-binding proteins known as constitutive and alternative splicing regulator; Upregulated in multiple types of human cancer and its silencing inhibit tumor cells | Zhou et al. (2020) |
| S100P | 59 | Interaction with or regulate several molecules related to actin cytoskeleton dynamics and extracellular matrix remodeling, cell progression and differentiation; Calcium-binding protein overexpresses in cancer cell and promote tumor progression | Cong et al. (2020) |
| CD151 | 24 | Cell surface glycoprotein involved in cell development, growth and motility; Overexpressed in cancer cells (non-small cell lung cancer) for cancer progression, neoangiogenesis and epithelial-mesenchymal transition (EMT), and tumor cell invasion | Kwon et al. (2013) |
| ATP5J | 21 | ATP synthase-coupling factor 6, mitochondrial; Overexpression reported in cancer cells confer drug (5-FU) resistance | Zhu et al. (2013) |
| G6PD | 24 | Glucose-6-phosphate dehydrogenase (G6PD) is involved in maintenance of redox homeostasis of cell; Down-regulation of G6PD in cancer cell retards growth and promote cell death | Yang et al. (2019) |
| CPLX2 | 31 | Encoded by the complexin/synaphin gene family function in synaptic vesicle exocytosis; Overexpressed in high grade lung neuroendocrine tumors (LNETs) | Komatsu et al. (2013) |
| GGCT | 91 | Gamma-glutamylcyclotransferase high level reported in cancer cell | Kageyama et al. (2015) |
| CYB5B | 91 | 21 kDa protein constitutively over-expressed in lymphoma cells; Negligible expression on the cell surface of non-neoplastic lymphocytes | Murphy et al. (2010) |
| (B) Upregulated proteins (≥ fivefold) in A549 cell after ESP treatment | |||
|---|---|---|---|
| STAT1 | 24 | STAT1 enhances inflammation and innate and adaptive immunity, triggering in most instances anti-proliferative and pro-apoptotic responses in tumor cells | Pensa et al. (2013) |
| NDRG1 | 5 | Protein NDRG1 inhibit F-actin polymerization and metastasis | Sun et al. (2013) |
| CALR | 7 | Cell surface calreticulin promotes phagocytic uptake of cancer cells by immune system | Fucikova et al. (2015) |
| PSME1 | 5 | Proteasome activator complex subunit 1 considered as prognostic markers for enhancing survival and prognostic accuracy of patients | Wang et al. (2019a, b) |
| HYOU1 | 5 | Hypoxia up-regulated protein 1 belongs to the Hsp70 family, induced by a variety of stimuli and diseases | Rao et al. (2021) |
| RCN1 | 7 | Reticulocalbin-1 | |
| CAPZB | 11 | F-actin-capping protein subunit beta | |
STMN 1 protein that regulates the dynamics of microtubules and regarded as oncogenic was also down-regulated (21-fold) (Table 1). Furthermore, eukaryotic translation initiation factor (1 and 3G), transcriptional (ELOC) protein, mitochondrial transcriptional factor A (TFAM) and splicing regulator (SRSF3) were down-regulated indicating anti-proliferative effect of ESP (Table 1). Interestingly, various marker proteins (S100P, CD151, ATP5J, G6PD, CPLX2, GGCT and CYB5B) reported to be over-expressed in cancer cells were significantly down-regulated due to treatment of ESP (Table 1).
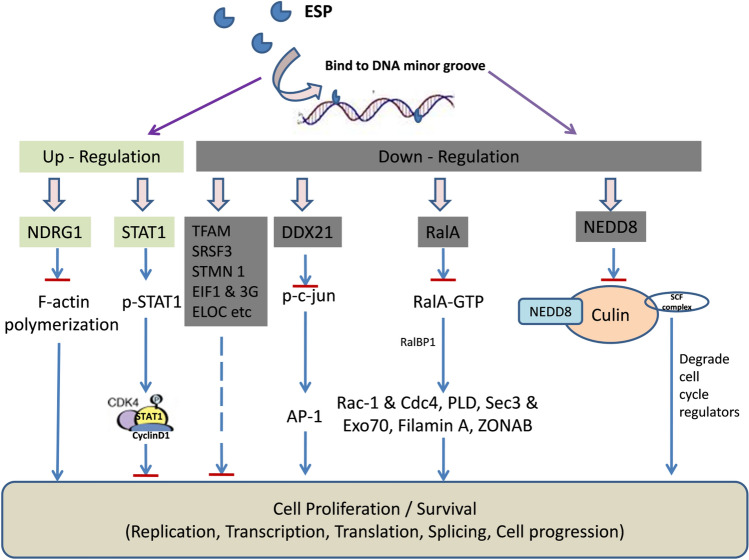
Among the up-regulated proteins, STAT1, considered as tumor suppresser, due to its inhibitory effects on cell progression, growth and angiogenesis was found to be 24-fold up-regulated (Table 1). STAT 1 in its phosphorylated form interacts with CDK4 and cyclin D1 and inhibits cell cycle progression. Besides, NDRG1, a metastasis suppressor protein that inhibits F-actin polymerization was also found to be up-regulated (fivefold) (Table 1). In our previous study, the same compound (ESP) was found to up-regulate dgcP genes in E. coli which stalls cell division by inhibiting initiation of septal invagination (Kumar et al. 2020). CALR protein which plays role in phagocytic immune response against cancer cells was also up-regulated (Table 1). Interestingly, only few marker proteins (HYOU1, PSME1, RCN1 and CAPZB) remained up-regulated (Table 1). Based upon SWATH-MS analysis, a model has been proposed to explain possible mechanism of anti-proliferative activity of ESP (Fig. 5).
Fig. 5.
Model explaining possible mechanism of anti-proliferative activity of ESP based on SWATH-MS proteomic analysis. Down-regulation (≥ 20-fold) of RalA, NEDD8, DDX21 and other proteins (TFAM, SRSF3, STMN1, EIF1 & 3G, ELOC, etc.) may inhibit the cell proliferation /survival by finally affecting the replication, transcription, translation, splicing and cell progression. Upregulated (≥ fivefold) STAT1 and NDRG1 possibly inhibited cell progression and F-actin polymerization required for cell division, respectively  Normal Pathway (in absence of ESP);
Normal Pathway (in absence of ESP);  Inhibition (in presence of ESP)
Inhibition (in presence of ESP)
Extractability of ESP significantly enhanced by radiation treatment of spinach powder
ESP found to have high antimutagenicity and anti-proliferative activity and, therefore, could be required in higher quantity during its potential applications. To address this concern, radiation technology was explored for its efficacy to enhance the extractability of ESP from spinach. Thus, spinach powder was radiation treated prior to its standard extraction process which included ultrasonication in solvent. The process for ESP extraction has been schematically depicted in Fig. 6A. The maximum approved radiation dose for food is up to 25 kGy as per the regulations issued by Food Safety and Standards Authority of India (FSSAI 2016). Thus, keeping dose uniformity ratio (DUR) into consideration, samples were subjected to maximum allowed radiation dose up to 20 kGy.
Fig. 6.

Schematic representation of extraction process with possible explanation involved in isolating ESP from spinach powder (A); Extractability of ESP from irradiated in the range of 5 to 20 kGy of spinach powder in terms of fluorescence intensity upon TLC (inset) and yield (weight; µg/g) (B); Scanning electron microscopy (SEM) of irradiated (5–20 kGy) spinach powder (C); Functionality [antimutagenicity (D) and anti-proliferative activity based on MTT assay (E)] of ESP from irradiated spinach powder. Different letters (a–d) on the top of the bars showed significant differences among means (p ≤ 0.05)
The yield of ESP was only ~ 391 µg/g from untreated powder (Fig. 6B). It was observed that radiation treatment enhanced the extraction of this bioactive compound from the spinach powder. Interestingly, there was a dose-dependent increase in the yield of the compound, i.e., 664 µg/g (1.7-fold of untreated) at 5 kGy to 1720 µg/g (4.4-fold of untreated) at 20 kGy (Fig. 6B). Fluorescence based relative fold change values corroborated with weight based quantification of ESP. Strong positive correlation (‘r’) of 0.996 was observed between both these quantification methods. Also, no change in relative front (Rf: 0.315) on TLC plate was found for ESP extracted from radiation treated spinach powder as compared to non-treated samples (Fig. 6B). Application of radiation exposure for enhancing the extractability of bioactive(s) has also been reported earlier (Taheri et al. 2014), wherein exposure of 20 kGy of gamma radiation to Curcuma alismatifolia leaves increased the yield of total phenolics and flavonoids up to twofold as compared to the non-irradiated ones.
Further, SEM technique was used to explore mechanism for the increased extractability of ESP from spinach powder after radiation exposure. SEM data show that there is dose-dependent reduction in smoothness and compactness in spinach powder (Fig. 6C). Radiation treatment at high dose could result in breakdown of polysaccharides such as pectin, cellulose and hemicelluloses present in the cell wall and other components (Roy et al. 2020). Energy from ultrasound generates cavities or bubbles in liquid which eventually becomes unstable and thus implodes creating waves could cause cell rupture (Sango et al. 2014). Ultrasonic cavitations are reported to be hindered due to energy attenuation in the direction of propagation (Wang et al. 2021). Besides, it has also been reported that fibril destruction due to treatment(s) promotes extraction of the compound(s) through the increase in surface area exposed to solvent action (Chemat et al. 2017; Freitas et al. 2020). This indicates that prior radiation treatment of spinach powder has led to softening of tissue which might have facilitated improved cavitations in the sample due to reduced ultrasonic energy attenuation and thus enhanced extraction of ESP in the preferable solvent.
Functionality of ESP from irradiated spinach remained unaltered
Functionality of ESP extracted from irradiated (20 kGy) spinach powder was conducted using both antimutagenicity and anti-proliferative activities. Study showed that the antimutagenicity of ESP from non-irradiated spinach powder was found to be ~ 60 and 80% at 1.1 (0.5 mg/mL) and 2.07 mM (1 mg/mL) with respect to positive (EMS treated) controls, respectively (Fig. 6D). Almost similar level of antimutagenicity, i.e., ~ 65 and 75% was displayed by ESP from irradiated spinach powder, respectively (Fig. 6D). It was also observed that ESP extracted from irradiated or non-irradiated spinach powder has similar cytotoxicity against A549 lung cancer cells, however, no cytotoxicity observed in normal lung (WI38) cells (Fig. 6E). This indicated that radiation treatment has enhanced the extraction of ESP without affecting its functionality.
Conclusion
Findings of the current study have demonstrated anti-proliferative effect of a vitamin K1 derivative (ethoxy-substituted phylloquinone) purified from spinach on lung cancer cell without negatively affecting normal lung cells. ESP treatment induces cell death and arrested lung cancer cell in the G2-M phase of cell cycle. The compound binds to the DNA minor groove and possibly down-regulates many key proteins involved in tumorigenesis such as EGFR signaling and NEDDylation as well as up-regulates certain tumor suppressors like STAT1 and NDRG1 during proteomic analysis. It was also observed that gamma radiation treatment enhanced the extractability of ESP and thus its content from spinach without affecting the functionality for potential applications.
Supplementary Information
Below is the link to the electronic supplementary material.
Acknowledgements
Authors acknowledge Dr. Dheeraj Jain (Chemistry Division, BARC) and Smt. Reema Devi Singh, Nuclear Agricultural and Biotechnology Division, BARC for their technical assistance. BARC is a Government of India R&D institute and so funding information is not applicable.
Author contributions
SK, SG and DKM: conceptualization, writing-original draft and editing; SK, JT, DKM and JN: investigation/analysis and validation; SK: data curation; SG: supervision.
Declarations
Conflict of interest
Authors declare no conflict of interest with respect to the study described in the manuscript.
References
- Bao P, Yokobori T, Altan B, Iijima M, Azuma Y, Onozato R, et al. High STMN1 expression is associated with cancer progression and chemo-resistance in lung squamous cell carcinoma. Ann Surg Oncol. 2017;24:4017–4024. doi: 10.1245/s10434-017-6083-0. [DOI] [PubMed] [Google Scholar]
- Cheimonidi C, Samara P, Polychronopoulos P, Tsakiri EN, Nikou T, Myrianthopoulos V, et al. Selective cytotoxicity of the herbal substance acteoside against tumor cells and its mechanistic insights. Redox Biol. 2018;16:169–178. doi: 10.1016/j.redox.2018.02.015. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chemat F, Rombaut N, Sicaire AG, Meullemiestre A, Fabiano-Tixier AS, Abert-Vian M. Ultrasound assisted extraction of food and natural products. mechanisms, techniques, combinations, protocols and applications. A Review Ultrason Sonochem. 2017;34:540–560. doi: 10.1016/j.ultsonch.2016.06.035. [DOI] [PubMed] [Google Scholar]
- Chen AY, Yu C, Gatto B, Liu LF. DNA minor groove-binding ligands: a different class of mammalian DNA topoisomerase I inhibitors. Proc Natl Acad Sci USA. 1993;90:8131–8135. doi: 10.1073/pnas.90.17.8131. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Collins BC, Hunter CL, Liu Y, Schilling B, Rosenberger G, Bader SL, et al. Multi-laboratory assessment of reproducibility, qualitative and quantitative performance of SWATH-mass spectrometry. Nat Commun. 2017;8:1–12. doi: 10.1038/s41467-016-0009-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cong Y, Cui Y, Wang S, Jiang L, Cao J, Zhu S, et al. Calcium-binding protein S100P promotes tumor progression but enhances chemosensitivity in breast cancer. Front Oncol. 2020;10:1–12. doi: 10.3389/fonc.2020.00001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Corazzari M, Gagliardi M, Fimia GM, Piacentini M. Endoplasmic reticulum stress, unfolded protein response, and cancer cell fate. Front Oncol. 2017;7:1–11. doi: 10.3389/fonc.2017.00078. [DOI] [PMC free article] [PubMed] [Google Scholar]
- El-Sayed SM. Use of spinach powder as functional ingredient in the manufacture of UF-soft cheese. Heliyon. 2020;6:1–6. doi: 10.1016/j.heliyon.2020.e03278. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Food Safety and Standards Authority of India (FSSAI) (2016) Draft food safety and standards (packaging and labelling) & (food products standards and food additives) (Amendment) regulations, 2015 related to inclusion of new atomic energy (radiation processing of food and allied products), Rules, 2012.
- Freitas PAV, González-Martínez C, Chiralt A. Application of ultrasound pre-treatment for enhancing extraction of bioactive compounds from rice straw. Foods. 2020;9:1–15. doi: 10.3390/foods9111657. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Fucikova J, Becht E, Iribarren K, Goc J, Remark R, Damotte D, et al. Calreticulin expression in human non-small cell lung cancers correlates with increased accumulation of antitumor immune cells and favorable prognosis. Cancer Res. 2016;76:1746–1756. doi: 10.1158/0008-5472.CAN-15-1142. [DOI] [PubMed] [Google Scholar]
- Ghose S, Varshney S, Adlakha K, Bhat A, Naushin S, Seth S, et al. Quantitative proteomics study reveals differential proteomic signature in dilated, restrictive, and hypertrophic cardiomyopathies. J Proteins Proteomics. 2019;100:33–44. doi: 10.1007/s42485-019-00004-y. [DOI] [Google Scholar]
- Golob-Schwarzl N, Schweiger C, Koller C, Krassnig S, Gogg-Kamerer M, Gantenbein N, et al. Separation of low and high grade colon and rectum carcinoma by eukaryotic translation initiation factors 1, 5 and 6. Oncotarget. 2017;8:101224–101243. doi: 10.18632/oncotarget.20642. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Guardavaccaro D, Pagano M. Oncogenic aberrations of cullin-dependent ubiquitin ligases. Oncogene. 2004;23:2037–2049. doi: 10.1038/sj.onc.1207413. [DOI] [PubMed] [Google Scholar]
- Gupta RS, Singh D. Amelioration of CCl4-induced hepatosuppression by Spinacia oleracea L. leaves in wistar albino rats. Pharmacologyonline. 2006;3:267–278. [Google Scholar]
- Joerger AC, Fersht AR. The p53 pathway: origins, inactivation in cancer, and emerging therapeutic approaches. Annu Rev Biochem. 2016;85:375–404. doi: 10.1146/annurev-biochem-060815-014710. [DOI] [PubMed] [Google Scholar]
- Kageyama S, Hanada E, Ii H, Tomita K, Yoshiki T, Kawauchi A. Gamma-glutamylcyclotransferase: a novel target molecule for cancer diagnosis and treatment. Biomed Res Int. 2015;2015:1–5. doi: 10.1155/2015/345219. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Khan MA, Mahesh C, Semwal AD, Sharma GK. Effect of spinach powder on physico-chemical, rheological, nutritional and sensory characteristics of chapati premixes. J Food Sci Technol. 2015;52:2359–2365. doi: 10.1007/s13197-013-1198-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Komatsu H, Kakehashi A, Nishiyama N, Izumi N, Mizuguchi S, Yamano S, et al. Complexin-2 (CPLX2) as a potential prognostic biomarker in human lung high grade neuroendocrine tumors. Cancer Biomark. 2013;13:171–180. doi: 10.3233/CBM-130336. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kumar S, Chatterjee S, Tripathi J, Gautam S. Purification and characterization of the principal antimutagenic bioactive as ethoxy-substituted phylloquinone from spinach (Spinacea oleracea L.) based on evaluation in models including human lymphoblast TK+/− cells. J Agric Food Chem. 2016;64:8773–8782. doi: 10.1021/acs.jafc.6b03915. [DOI] [PubMed] [Google Scholar]
- Kumar S, Gautam S, Sharma A. Hurdle technology including chlorination, blanching, packaging and irradiation to ensure safety and extend shelf life of shelled sweet corn kernels. J Food Process Preserv. 2015;39:2340–2347. doi: 10.1111/jfpp.12481. [DOI] [Google Scholar]
- Kumar S, Kunwar A, Gautam S, Sharma A. Inactivation of A. ochraceus spores and detoxification of ochratoxin A in coffee beans by gamma irradiation. J Food Sci. 2012;77:44–51. doi: 10.1111/j.1750-3841.2011.02572.x. [DOI] [PubMed] [Google Scholar]
- Kumar S, Tripathi J, Srivastava AK, Gautam S. Molecular mechanism of antimutagenicity by an ethoxy-substituted phylloquinone (vitamin K1 derivative) from spinach (Spinacea oleracea L.) Chem Biol Interact. 2020;330:1–12. doi: 10.1016/j.cbi.2020.109216. [DOI] [PubMed] [Google Scholar]
- Kwon MJ, Seo J, Kim YJ, Kwon MJ, Choi JY, Kim TE, et al. Prognostic significance of CD151 overexpression in non-small cell lung cancer. Lung Cancer. 2013;81:109–116. doi: 10.1016/j.lungcan.2013.03.014. [DOI] [PubMed] [Google Scholar]
- Laskowski RA, Swindells MB. LigPlot+: multiple ligand-protein interaction diagrams for drug discovery. J Chem Inf Model. 2011;51:2778–2786. doi: 10.1021/ci200227u. [DOI] [PubMed] [Google Scholar]
- Lee MY, Kim HA, Kang MH. Comparison of lymphocyte DNA damage levels and total antioxidant capacity in Korean and American diet. Nutr Res Pract. 2017;11:33–42. doi: 10.4162/nrp.2017.11.1.33. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lester GE, Makus DJ, Hodges DM, Jifon JL. Summer (subarctic) versus winter (subtropic) production affects spinach (Spinacia oleracea L.) leaf bionutrients: vitamins (c, e, folate, k1, provitamin a), lutein, phenolics, and antioxidants. J Agric Food Chem. 2013;61:7019–7027. doi: 10.1021/jf401461z. [DOI] [PubMed] [Google Scholar]
- Lim KH, Baines AT, Fiordalisi JJ, Shipitsin M, Feig LA, Cox AD, et al. Activation of RalA is critical for Ras-induced tumorigenesis of human cells. Cancer Cell. 2005;7:533–545. doi: 10.1016/j.ccr.2005.04.030. [DOI] [PubMed] [Google Scholar]
- Lim SM, Jeong Y, Lee S, Im H, Tae HS, Kim BG, et al. Identification of β-lapachone analogs as novel MALT1 inhibitors to treat an aggressive subtype of diffuse large B-cell lymphoma. J Med Chem. 2015;58:8491–8502. doi: 10.1021/acs.jmedchem.5b01415. [DOI] [PubMed] [Google Scholar]
- Longnecker MP, Newcomb PA, Mittendorf R, Greenberg R, Willet W. Intake of carrots, spinach, and supplements containing vitamin A in relation to risk of breast cancer. Cancer Epidemiol Biomark Prev. 1997;6:887–892. [PubMed] [Google Scholar]
- Madureira J, Barros L, Verde SC, Margaça FMA, Santos-Buelga C, Ferreira ICFR. Ionizing radiation technologies to increase the extraction of bioactive compounds from agro-industrial residues: a review. J Agric Food Chem. 2020;68:11054–11067. doi: 10.1021/acs.jafc.0c04984. [DOI] [PubMed] [Google Scholar]
- Maurya DK. ColonyCountJ: A user-friendly Image J Add-on Program for quantification of different colony parameters in clonogenic assay. J Clin Toxicol. 2017;358:1–4. [Google Scholar]
- Metha D, Belemkar S. Pharmacological activity of Spinacia oleracea Linn. Asian J Pharm Res Dev. 2014;2:83–93. [Google Scholar]
- Moghadam AR, Patrad E, Tafsiri E, Peng W, Fangman B, Pluard TJ, et al. Ral signaling pathway in health and cancer. Cancer Med. 2017;6:2998–3013. doi: 10.1002/cam4.1105. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Murphy D, Parker J, Zhou M, Fadlelmola FM, Steidl C, Karsan A, et al. Constitutively overexpressed 21 kDa protein in Hodgkin lymphoma and aggressive non-Hodgkin lymphomas identified as cytochrome B5b (CYB5B) Mol Cancer. 2010;9:1–19. doi: 10.1186/1476-4598-9-14. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pensa S, Regis G, Boselli D, Novelli F, Poli V. (2013) STAT1 and STAT3 in tumorigenesis: two sides of the same coin? In: Madame Curie bioscience database. Landes Biosci Austin (TX).
- Pereyra CE, Dantas RF, Ferreira SB, Gomes LP, Silva-Jr FP. The diverse mechanisms and anticancer potential of naphthoquinones. Cancer Cell Int. 2019;19:1–20. doi: 10.1186/s12935-019-0925-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rao S, Oyang L, Liang J, Yi P, Han Y, Luo X, et al. Biological function of HYOU1 in tumors and other diseases. OncoTargets Ther. 2021;14:1727–1735. doi: 10.2147/OTT.S297332. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Roy R, Rahman MS, Raynie DE. Recent advances of greener pretreatment technologies of lignocelluloses. Curr Res Green Sustain Chem. 2020;3:1–17. doi: 10.1016/j.crgsc.2020.100035. [DOI] [Google Scholar]
- Sango DM, Abela D, McElhatton A, Valdramidis VP. Assisted ultrasound applications for the production of safe foods. J Appl Microbiol. 2014;116:1067–1083. doi: 10.1111/jam.12468. [DOI] [PubMed] [Google Scholar]
- Schoenfeld AR, Davidowitz EJ, Burk RD. Elongin BC complex prevents degradation of von Hippel-Lindau tumor suppressor gene products. Proc Natl Acad Sci USA. 2000;97:8507–8512. doi: 10.1073/pnas.97.15.8507. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Singh AN, Sharma N. Quantitative SWATH-based proteomic profiling for identification of mechanism-driven diagnostic biomarkers conferring in the progression of metastatic prostate cancer. Front Oncol. 2020;10:1–12. doi: 10.3389/fonc.2020.00001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Soderlind KJ, Gorodetsky B, Singh AK, Bachur NR, Miller GG, Lown JW. Bis-benzimidazole anticancer agents: targeting human tumour helicases. Anticancer Drug Des. 1999;14:19–36. [PubMed] [Google Scholar]
- Sun J, Zhang D, Bae DH, Sahni S, Jansson P, Zheng Y, et al. Metastasis suppressor, NDRG1, mediates its activity through signaling pathways and molecular motors. Carcinogenesis. 2013;34:1943–1954. doi: 10.1093/carcin/bgt163. [DOI] [PubMed] [Google Scholar]
- Sung H, Ferlay J, Siegel RL, Laversanne M, Soerjomataram I, Jemal A, et al. Global cancer statistics 2020 GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J Clin. 2021;71:209–249. doi: 10.3322/caac.21660. [DOI] [PubMed] [Google Scholar]
- Taheri S, Abdullah TL, Karimi E, Oskoueian E, Ebrahimi M. Antioxidant capacities and total phenolic contents enhancement with acute gamma irradiation in Curcuma alismatifolia (Zingiberaceae) leaves. Int J Mol Sci. 2014;15:13077–13090. doi: 10.3390/ijms150713077. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tanaka T, Nakatani T, Kamitani T. Inhibition of NEDD8-conjugation pathway by novel molecules: potential approaches to anticancer therapy. Mol Oncol. 2012;6:267–275. doi: 10.1016/j.molonc.2012.01.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Teerasripreecha D, Phuwapraisirisan P, Puthong S, Kimura K, Okuyama M, Mori H, et al. In vitro antiproliferative/cytotoxic activity on cancer cell lines of a cardanol and a cardol enriched from Thai Apis mellifera propolis. BMC Complement Altern Med. 2012;12:1–17. doi: 10.1186/1472-6882-12-27. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Voragen AGJ, Coenen GJ, Verhoef RP, Schols HA. Pectin, a versatile polysaccharide present in plant cell walls. Struct Chem. 2009;20:263–275. doi: 10.1007/s11224-009-9442-z. [DOI] [Google Scholar]
- Wang H, Luo YH, Shen GN, Piao XJ, Xu WT, Zhang Y, et al. Two novel 1,4-naphthoquinone derivatives induce human gastric cancer cell apoptosis and cell cycle arrest by regulating reactive oxygen species-mediated MAPK/Akt/STAT3 signaling pathways. Mol Med Rep. 2019;20:2571–2582. doi: 10.3892/mmr.2019.10500. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wang Q, Pan F, Li S, Huang R, Wang X, Wang S, et al. The prognostic value of the proteasome activator subunit gene family in skin cutaneous melanoma. J Cancer. 2019;10:2205–2219. doi: 10.7150/jca.30612. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wang X, Ning Z, Lv M, Sun C. The investigation of ultrasonic energy attenuation in aqueous solutions. J Phys Soc Japan. 2021;90:1–7. doi: 10.7566/JPSJ.90.104401. [DOI] [Google Scholar]
- White V, Ruparelia P. Kumar & Clark's clinical medicine. 10. Amsterdam: Elsevier; 2020. p. 975. [Google Scholar]
- Xie D, Wu X, Lan L, Shangguan F, Lin X, Chen F, et al. Downregulation of TFAM inhibits the tumorigenesis of non-small cell lung cancer by activating ROS-mediated JNK/p38MAPK signalling and reducing cellular bioenergetics. Oncotarget. 2016;7:11609–11624. doi: 10.18632/oncotarget.7018. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yang C, Liu X, Li C, Li S, Du W, Yang D. Eukaryotic translation initiation factor 3 subunit G (EI F3G) resensitized HC T116/5-Fu to 5-fluorouracil (5-Fu) via inhibition of MRP and MDR1. Onco Targets Ther. 2018;11:5315–5324. doi: 10.2147/OTT.S170854. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yang HC, Wu YH, Yen WC, Liu HY, Hwang TL, Stern A, et al. The redox role of G6PD in cell growth, cell death, and cancer. Cells. 2019;8:1–29. doi: 10.3390/cells8091055. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhang Y, Baysac KC, Yee LF, Saporita AJ, Weber JD. Elevated DDX21 regulates c-Jun activity and rRNA processing in human breast cancers. Breast Cancer Res. 2014;16:1–18. doi: 10.1186/s13058-014-0449-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhang X, Zhang SC, Sun D, Hu J, Wali A, Pass H, et al. New insight into the molecular mechanisms of the biological effects of DNA minor groove binders. PLoS ONE. 2011;6:1–12. doi: 10.1371/annotation/4056d03c-20ed-4eca-9568-3e9400e2312e. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhou Z, Gong Q, Lin Z, Wang Y, Li M, Wang L, et al. Emerging roles of SRSF3 as a therapeutic target for cancer. Front Oncol. 2020;10:1–17. doi: 10.3389/fonc.2020.00001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhu H, Chen L, Zhou W, Huang Z, Hu J, Dai S, et al. Over-expression of the ATP5J gene correlates with cell migration and 5-fluorouracil sensitivity in colorectal cancer. PLoS ONE. 2013;8:1–11. doi: 10.1371/journal.pone.0076846. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zihlif M, Catchpoole DR, Stewart BW, Wakelin LP. Effects of DNA minor groove binding agents on global gene expression. Cancer Genomics Proteomics. 2010;7:323–330. [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.