Abstract
Resveratrol (RSV), a non-flavonoid stilbene polyphenol, possesses anti-carcinogenic activities against all the major stages of cancer. Zein nanoparticles (ZN NPs) have been utilized successfully in delivery of variant therapeuticals by virtue of their histocompatible nature. The goal of this work was to comparatively explore the antiproliferative, pro-apoptotic and oxidative stress potentials of RSV-ZN NPs versus RSV against human colorectal carcinoma HCT-116 cells. ZN-RSV NPs were developed and assayed for particle size analysis and RSV diffusion. The selected formula obtained 137.6 ± 8.3 nm as mean particle size, 29.4 ± 1.8 mV zeta potential, 92.3 ± 3.6% encapsulation efficiency. IC50 of the selected formula was significantly lower against HCT-116 cells versus Caco-2 cells. Also, significantly enhanced cellular uptake was generated from RSV-ZN NPs versus free RSV. Enhanced apoptosis was concluded due to increased percentage cells in G2-M and pre-G1 phases. The pro-apoptotic potential was explained by caspase-3 and cleaved caspase-3 increased mRNA expression in addition to NF-κB and miRNA125b decreased expression. Biochemically, ZN-RSV NPs induced oxidative stress as demonstrated by enhanced reactive oxygen species (ROS) generation and endothelial nitric oxide synthase (eNOS) isoenzyme increased levels. Conclusively, ZN-RSV NPs obtained cell cycle inhibition supported with augmented cytotoxicity, uptake and oxidative stress markers levels in HCT-116 tumor cells in comparison with free RSV. These results indicated intensified chemopreventive profile of RSV due to effective delivery utilizing ZN nano-dispersion against colorectal carcinoma HCT-116 cells.
Subject terms: Molecular biology, Molecular medicine, Cancer, Nanobiotechnology
Introduction
Resveratrol (trans-3,4′,5-trihydroxystilbene; RSV) is a natural occurring stilbene polyphenolic chemopreventive and anticancer drug. RSV can be naturally obtained daily via grapes, blueberries, raspberries, mulberries, and peanuts1. RSV has been sorted as a natural lipophilic phytoalexin for being synthesized by plants due to infection2, fungal attack3, ozone exposure4 and UV exposure5. RSV was first isolated by the Japanese scientist Mitchio Takaoka from Veratrum grandiflorum O. Loes. in 19396. RSV belongs to a vast group of physiologically active compounds that exhibit a wide variety of therapeutical effects including anti-oxidant, anti-inflammatory, anti-atherogenic, anti-fungal, cardioprotective, anti-atherosclerosis and anti-tumor effects7–9. RSV was first introduced in the early nineties10. The cytotoxic properties of RSV were first introduced in 1997 when this compound proved to possess activity against cancer stages of initiation via inducing phase II drug-metabolizing enzymes, promotion via hindering cyclooxygenase and hydroperoxidase functions and progression via inducing human promyelocytic leukemia cell differentiation11. RSV anti-cancer molecular mechanism is still not illustrated methodically12 in spite of over 20,000 research papers and around 130 human clinical trials interested about RSV6. RSV is also reported to suppress oxidative stress affecting nitric oxide generation that affects DNA damage, cell cycle, apoptosis, and proliferation13. Specifically, RSV enhanced apoptosis of HCT-116 cells via variant finite molecular mechanisms9,14–19.
Colorectal carcinoma (CRC) is a universal leading cause of death in men (10%) and women (9.2%) and ranks the third most common cancers and the fourth most common cause of cancer-related death14,20,21. Globally, CRC mortality will be increased by 56% between 2020 and 204021. In 2040, the estimated number of deaths due to the disease is 1.6 million deaths worldwide according to the international Agency for Research on Cancer (IARC)21. The elevated percent of CRC patients can be related to increased ageing population, non-healthy dietary habits and an increase in risks of smoking, obesity and low physical exercise21,22.
The obtained resistance to tumors due to conventional therapeuticals is a crucial incident in cancer treatment. Particularly, apoptosis resistance enables tumors cells evade innate surveillance mechanisms and disturbing intracellular signaling cascade leading to tumor promotion and metastasis. Distraction of apoptosis pathways may shift cancerous cells to be treatment resistant beside tumorigenesis promotion23,24. Induction of apoptosis by anti-carcinogenic molecules has been conformed with tumor response24. Treatment resistance was reported to be as a result of apoptosis potential failure especially when apoptosis is the main action of anticancer mechanism23. Apoptosis enhancement is a site of research interest to support cancer clinical management. Induction of apoptosis in tumor cells is a frontier strategy with limit concurrent damage to normal cells25.
Nanoparticulate delivery systems are characterized by eminent capability of enhancing drug solubilization, sustained circulation time, targeting at tumor via augmented permeability and retention effects26, enhanced overcoming of intratumoral heterogeneity in cancerous cells due to surface receptor expression27 and reduction of antitumor molecule toxicities and dose as a result of improving cytotoxicity and cellular uptake28. Zein nano-particulates (ZN NPs) are highly suggested nanocomposites because of their plant origin and appropriate method of fabrication. ZN is obtained from maize plant. According to United States Food and Drug Administration, ZN is Generally Recognized as Safe (GRAS)29, biocompatible and inexpensive excipient that used ordinarily in food industry30, pharmaceutical industry31, vaccine delivery32, tissues engineering33 and biomedical applications34,35. Also, zein has elevated thermal resistance and good oxygen barrier capability for the loading of thermosensitive compounds36. Pharmaceutical nanotechnology enhances antitumor therapeuticals profiles37,38. The aim of this study was to comparatively investigate the antiproliferative, pro-apoptotic and oxidative stress profiles of RSV-ZN NPs in comparison to free RSV in human colorectal carcinoma HCT-116 cells.
Results and discussion
RSV was studies extensively along last decade for obtaining cancer therapeutics promising outcomes in both cancer prevention and treatment. The anticancer molecular mechanism is still not elucidated. Effective formulation of RSV was frequently faced the difficulty of very low oral bioavailability due to reduced water solubility and low photostability. Just less than 1% bioavailability was reported due to severe metabolism in liver and intestines39. Pharmaceutical nanotechnological techniques offer successful approach to overcome this obstacle. Nanostructures of RSV are highly recommended to increase water solubility, facilitate biological membranes crossing, ameliorate absorption and stability and enhance pharmacokinetic parameters and bioavailability40. This will have significant and promising effect on therapeutical potential of RSV to ameliorate its cytotoxicity and pro-apoptotic activity against HCT-116 cells. The efficiency of nanostructures loaded with RSV will extended to overcome the suggested multiple drug resistance in various tumors and minimize toxicity via enhancing permeation and retention effect of nanostructures41.
Characterization of ZN-RSV NPs
RSV Nanocomposites were fabricated in three variant formulae; R1, R2 and R3. Table 1 displayed formulation characteristics for all developed formulae. The most reduced mean particle size was related to formula R1 which was 137.6 ± 8.3 nm. This obtained mean particle size of ZN-RSV NPs is suggested to enhance absorption and residence time inside biological systems34. As shown, particle size reduction was directly related to elevation of ZN content through the prepared formulae. The higher particle size of R3 formula could be generated due increased quantity of RSV on nanosphere surface, which could lead to maximized interfacial tension between particle surface and aqueous medium, so consequently particle diameter will be enlarged42. Also, R1 formula obtained the highest zeta potential magnitude (29.4 ± 1.8 mV) and the least polydispersity index magnitude (0.19 ± 0.01). Elevated zeta potential value reflects nanostructures dispersion stability as it will withstand aggregations because of charge of ZN surface35. R1 formula obtained 92.3% ± 3.6 encapsulation efficiency. The enhanced EE by increasing of in ZN amount in formula could be related to RSV partitioning to the hydrophobic matrix of ZN. The elevated encapsulation efficiency by elevation in ZN amount in formula might be due to integrated partitioning of RSV to ZN hydrophobic matrix which reflects a relation between RSV binding affinity and ZN polymerization degree43. The ability of nano-delivery systems to incorporate high percent of payloads was previously reported26,40,44. The method utilized for fabrication was adequate in obtaining high encapsulation efficiency values. This could be explained regarding to the natural unfolding of ZN molecules at alkaline pH which offers more reactive sites for maximized crosslinking and also remarkable reduction of void spaces inside ZN nanospheres35 which will positively contribute to ameliorated bioavailability, delivery and consequently the therapeutical response45,46.
Table 1.
Particle size, zeta potential, polydispersity index and encapsulation efficiency of ZN-RSV NPs formulae.
| RSV:ZN ratio | Mean Particle Size (nm) | Zeta Potential (mV) | Poly-dispersity index | Encapsulation efficiency (%) | |
|---|---|---|---|---|---|
| R1 | 1:5 | 137.6 ± 8.3 | 29.4 ± 1.8 | 0.19 ± 0.01 | 92.3 ± 3.6 |
| R2 | 1:1 | 158.8 ± 11.2 | 33.4 ± 1.2 | 0.33 ± 0.02 | 88.3 ± 2.4 |
| R3 | 5:1 | 246.4 ± 18.1 | 22.6 ± 1.8 | 0.61 ± 0.02 | 81.4 ± 4.9 |
ZN-RSV NPs formula diffusion is presented in Fig. 1. R1, R2 and R3 formulae displayed sustained RSV diffused profile, which obtained magnitudes of 94.1 ± 3.6%, 90.3 ± 4.5 and 84.4% ± 6.9 after 36 h, respectively. R1 formula was chosen for further investigations. Sustaining RSV diffusion could be attributed to ZN nature hydrophobic structure. ZN generates a delay in the water penetration which could attributed to the measured RSV sustained diffusion34. RSV diffusion exhibited biphasic sustained permeation profile. The diffusion displayed initial burst effect related to rapid release of trapped RSV to direction of ZN NPs surfaces. After initial burst, a reduction in diffusion rate was attained as RSV has to penetrate longer path in core matrix to completely release from nanospheres. Also, RSV diffusion rate was affected by water intake rate42,45,47. The prolonged release of RSV due to ZN NPs will support the pharmacokinetic potential due to short half-life and reported irregular profile in human after oral dose of RSV by48–50 that obtained two peak plasma concentration after 1 h and 6 h due to enteric recirculation.
Figure 1.
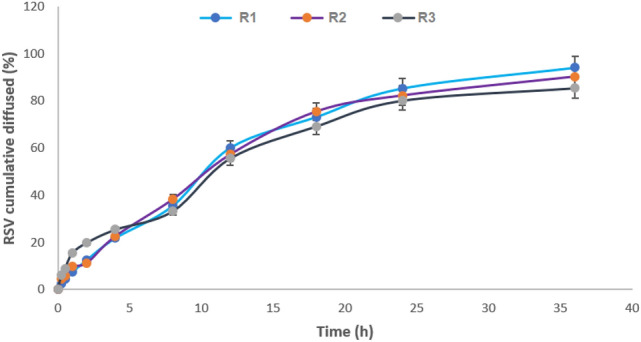
In vitro diffusion profile of ZN-RSV NPs formulations.
The particles morphology of R1 formula was examined by SEM imaging and showed compactly dispersed spheres with smooth surfaces (Fig. 2). The diameter of scanned spheres was consistent with particle size measured by laser diffraction technique. The noticeable compactness of nano-particulates may be related to freeze drying process51.
Figure 2.

SEM image of ZN-RSV NPs.
Serum stability
ZN-RSV NPs colloidal stability was estimated by monitoring the changes in sample mean particle size (Fig. 3). After incubation in FBS, ZN-RSV NPs displayed initial elevation in its mean particle size in the first 15 min and then decreased rapidly to starting value with non-significant difference. The developed RSV nanocomposites attained satisfied pattern in FBS which suggest similar profile in in in vivo studies due absence of persistent aggregation due to possible interacting with variant molecules inside the biological environment around nanocomposites.
Figure 3.
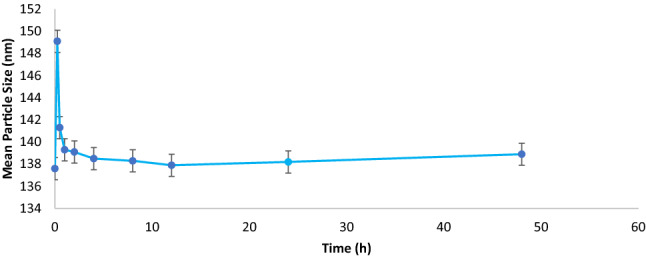
ZN-RSV NPs serum stability.
Cytotoxicity
Cytotoxicity of the different treatments using mitochondrial function (MTT reduction) results are exhibited in Fig. 4. It indicates distinguished augmentation of ZN-RSV NPs antiproliferative activity against HCT-116 cells. ZN-RSV NPs recorded enhanced activity of about (IC50 = 4.15 µg/ml) compared to free RSV (46 µg/ml). This premium cytotoxicity revealed the potential of ZN NPs due to noticeable reduced particle size of prepared nano-particulates that can interact with biomolecules within cells or on their surfaces, in addition to adequate encapsulation inside ZN NPs and complete diffusion of RSV that enhance potentiate cytotoxicity52,53. Also, it was very noticeable the cytotoxic effect of ZN alone in HCT-116 cell culture. ZN cytotoxicity may explain the obtained enhanced anti-proliferative action of ZN-RSV NPs. Cytotoxicity was enhanced in similar studies utilizing ZN nanoparticles as reported for dual delivery of exemestane and RSV54, Piceatannol55 and pterostilbene56. Similar studies reported highlighting cytotoxicity of ZN against several types of cancers cells44. Conclusively, ZN represented an optimistic biomaterial for fabrication of RSV nano-formulations and also cytotoxicity ameliorating.
Figure 4.
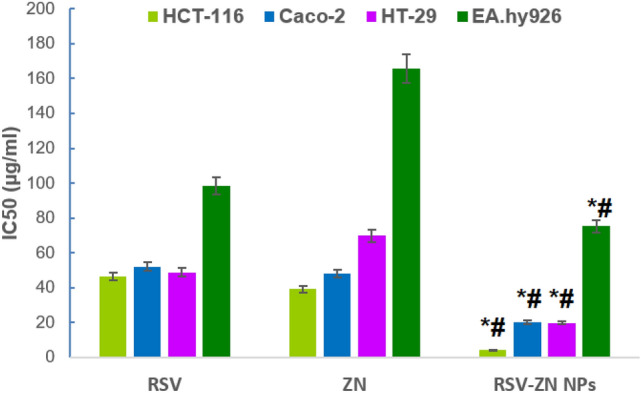
IC50 of RSV, ZN and ZN-RSV NPs in HCT-116, Caco-2, HT-29 and EA.hy926 cell line. *Significantly different in comparison with RSV (p < 0.05). #Significantly different in comparison with ZN (p < 0.05).
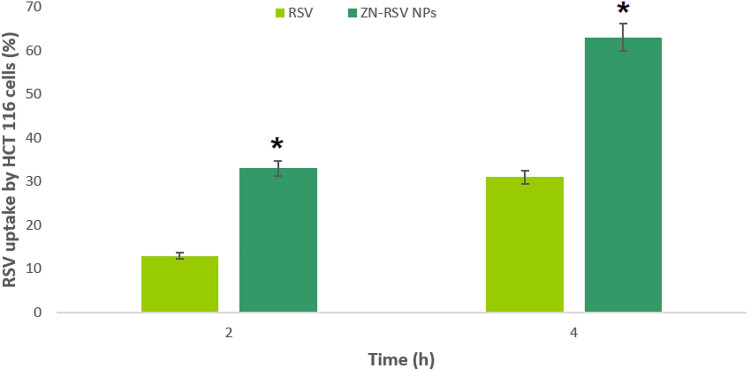
Cellular uptake
Figure 5 exhibited that free RSV cellular uptake obtained 19.1 ± 1.3% and 31.4 ± 2.7% at 2 and 4 h of incubation, respectively. Significantly elevated cellular uptakes were observed with ZN-RSV NPs incubations, which reached 41.6 ± 23.1% and 62.3 ± 5.2% at after 2 and 4 h of incubation, respectively. The results indicated ameliorated cellular uptake significantly by ZN-RSV NPs compared to free RSV. This also confirmed the ability of ZN NPs to enhance HCT-116 cells uptake of RSV that may ameliorate pro-apoptotic profile. The obtained results concluded significant ameliorated RSV cellular uptake due to the developed formula. This confirmed the ability of ZN NPs to enhance HCT-116 cells uptake of RSV which also confirm the efficacy of ZN improved Enhanced Permeability and Retention (EPR) cellular uptake in tumor microenvironment. The cellular uptake of polymeric nanoparticles which possess < 200 nm mean diameter is described by an endocytotic action57. Also, positively charged nanoparticles generated a higher rate of membrane internalization in comparison with negatively charged ones due to enhanced adsorptive-mediated transcytosis catalyzed by electrostatic interaction yielded between positively charged structures of ZN and negatively charged cells membranes58.
Figure 5.
Uptake by RSV by HCT-116 cells at 2 and 4 h. *Significant difference (p < 0.05).
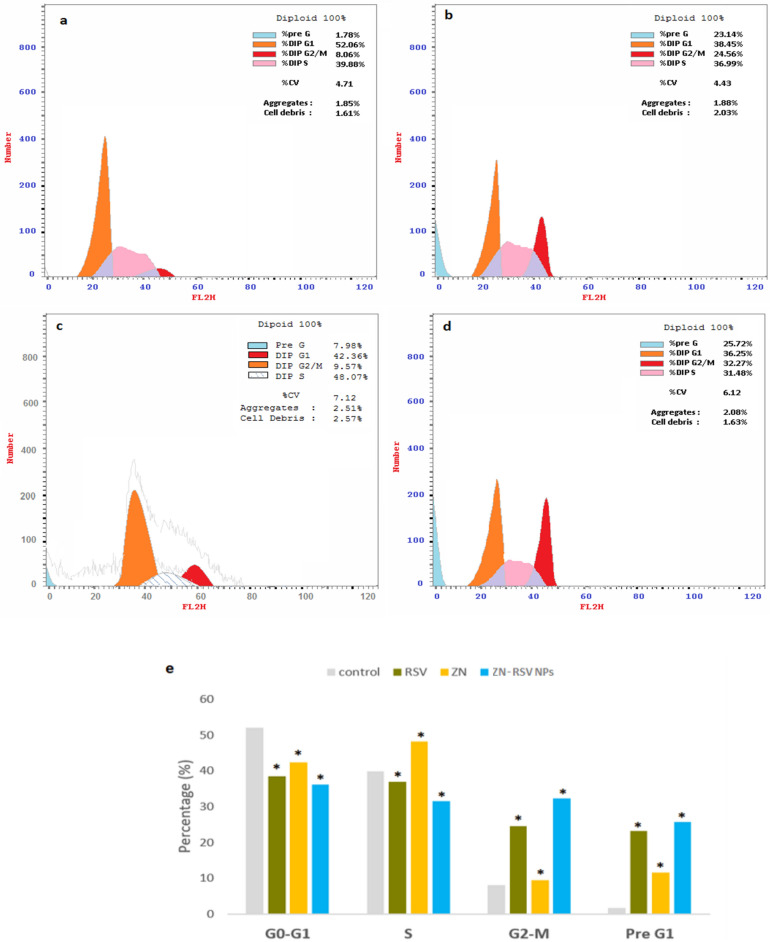
Cell cycle progression
Rapid growth properties were exhibited from control untreated HCT-116 cells. According to Fig. 6a, it obtained 52.06 ± 3.7% at the G0/G1 phase, 39.08 ± 2.9% at the S phase, 8.06 ± 0.4% at the G2-M phase, and 1.78 ± 0.6% at the pre-G1 phase. Other RSV, ZN and ZN-RSV NPs incubations displayed down the proliferation of HCT-116 cells, especially in the G0/G1 and S phases (Fig. 6b–d). Regarding to accumulation of cells in the pre-G phase, it records 1.78 ± 0.6%, 23.14 ± 1.1%, 7.98 ± 0.5 and 25.72 ± 1.4% of control HCT-116 cells, RSV, ZN, and ZN-RSV NPs groups, respectively. RSV antiproliferative potential is correlated with apoptosis induction capability and G1 phase cell cycle arrest in colon cancer cells59. Also, the concluded pro-apoptotic activity was in harmony with the obtained RSV enhanced induced apoptosis in HCT-116 cells three-dimensional culture leading to formation of a luminal cavity by suppressing PDE4 activity60. Figure 6e exhibited the obtained changes in cell cycle phases graphically.
Figure 6.
Impact of ZN-RSV NPs on cell cycle phases. (a) Control, (b) RSV, (c) ZN, (d) ZN-RSV NPs, (e) graphical presentation of each phase. *Significantly different in comparison with control group at p < 0.05.
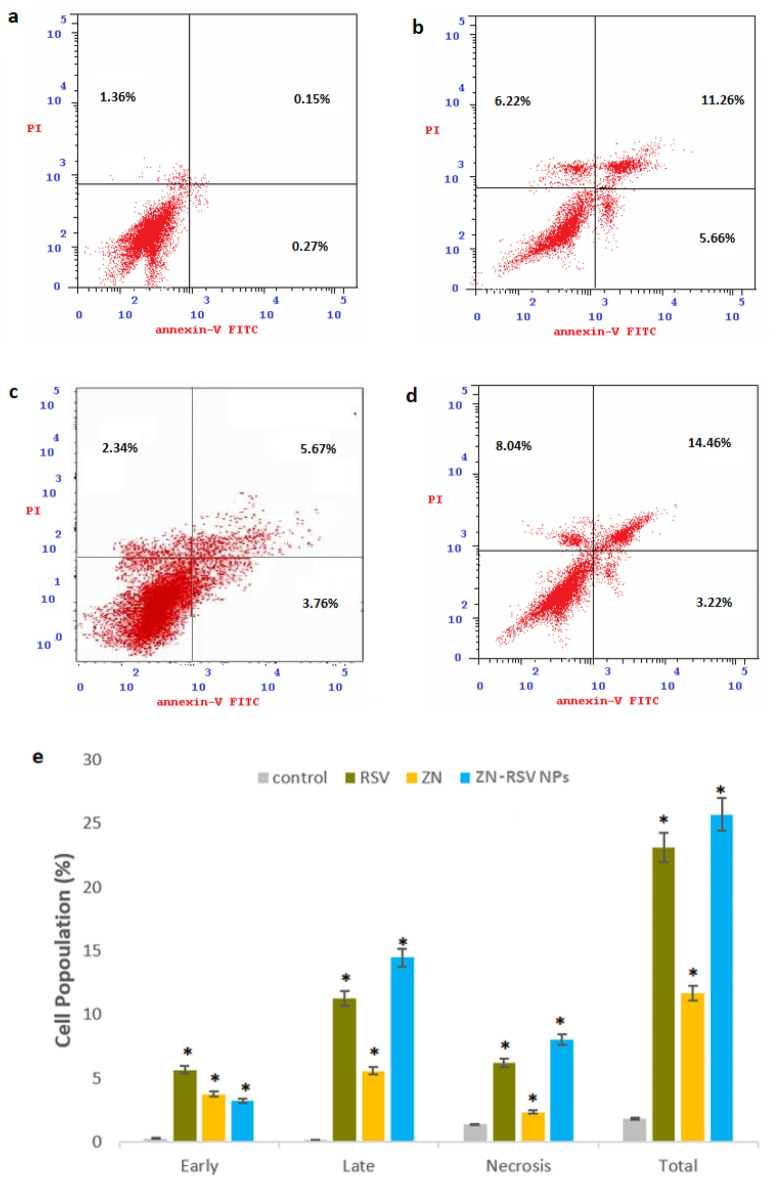
Annexin-V staining
In order to confirm the generated cell apoptotic death, the percentage of HCT-116 cells with positive annexin-V staining were estimated in the control, RSV, ZN and ZN-RSV NPs incubations (Fig. 7a–d). ZN-RSV NPs distinctly increased the early, late, and total cell death in comparison with other incubations. A graphical explanation for cell death different types was explained in Fig. 7e. ZN-RSV NPs obtained the most functional activity in enhancing pre-G phase, which confirms apoptotic cell death. This indicated early and late apoptotic death, as well as total cell death. The obtained results confirmed the potential of RSV to induce apoptosis in colon cancer cells61–64. Similar RSV enhanced pro-apoptotic was obtained in HT29 colon cancer cell65. Also, these results support the utilization of ZN Nano-dispersed delivery systems for enhancing apoptotic and cellular uptake potential of piceatannol55 and pterostilbene56.
Figure 7.
Impact of ZN-RSV NPs on annexin-V FITC positive staining HCT-116 cells. (a) Control, (b) RSV, (c) ZN, (d) ZN-RSV NPs, (e) graphical presentation of early and late apoptotic, necrotic and total cell death. *Significantly different in comparison with corresponding control group at p < 0.05.
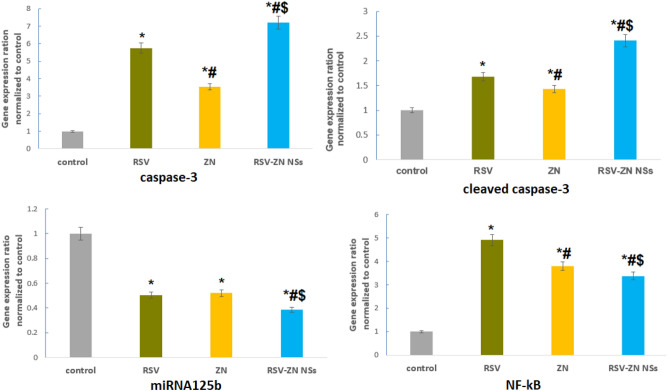
mRNA expression of apoptosis-related genes
Figure 8 displayed the results of mRNA expression of pro-apoptotic genes (CASP3 and cleaved caspase-3) and anti-apoptotic genes (miRNA125b and NF-κB) related to ZN-RSV NPs treated HCT-116. The cells exhibited statistically variant ratios. It was concluded that HCT-116 cells treated with ZN-RSV NPs obtained the highest values of expression related to caspase-3, cleaved caspase-3, miRNA125b and NF-κB genes.
Figure 8.
Impact of ZN-RSV NPs on caspase-3, cleaved caspase-3 miRNA125b and NF-κB in HCT-116 cells. *Significantly different in comparison with control (p < 0.05). #Significantly different in comparison with RSV (p < 0.05). $Significantly different in comparison with ZN (p < 0.05).
Activation of caspase-3 is so fundamental for apoptosis inducement and cancer targeting obtained by anti-tumor therapeuticals66; thus, it is considered a main indicator for sensitivity estimation of cancerous cells to therapeuticals. All treated groups (RSV, ZN and ZN-RSV NPs) exhibited folds change of 5.75, 3.54 and 7.21, respectively. Regarding to cleaved caspase-3 expression, all treated groups (RSV, ZN and ZN-RSV NPs) exhibited folds change of 1.68, 1.43 and 2.41, respectively. ZN-RSV NPs displayed elevated apoptotic effect in comparison with untreated positive control. ZN-RSV NPs obtained 1.25-fold elevation in caspase-3 level and 1.43-fold elevation in cleaved caspase-3 level in comparison with free RSV. Caspase-3 is considerably stimulated by the death protease, activating the specific cleavage of numerous major cell proteins, and it contributes mainly in programmed cell death67. A relation between RSV mechanism of action and caspase activation via blocking antiapoptotic proteins belonging to heat shock proteins was investigated68,69.
Regarding to miRNA125b expression, all treated groups (RSV, ZN and ZN-RSV NPs) displayed folds change of 0.503, 0.52 and 0.387, respectively. ZN-RSV NPs exhibited higher anti-apoptotic effect compared with untreated positive control. ZN-RSV NPs obtained 1.3-fold decrease in miRNA125b level compared with free RSV. These results confirmed the apoptotic role of RSV in cancer progression through modulation of microRNAs (miRNAs) which has not been studied70. Also, colon cancer progression was related to miRNAs that activating either as oncogenes or tumor-suppressor genes. In comparison to normal tissues, miRNAs have been dysregulated in colon cancer tissues51.
NF-κB pathway is the fundamental contributor to colon tumors where RSV inverts NF-κB activation, which is responsible for inducing inflammatory cytokines71. The results of NF-κB expression showed folds change of 3.51, 3.8 and 4.96, respectively. ZN-RSV NPs displayed elevated anti-apoptotic effect in comparison with untreated positive control. ZN-RSV NPs attained 1.41-fold decreasing in NF-κB level in comparison with free RSV. As investigated by72,73 NF-κB, as nuclear transcription factor involved in carcinogenesis, is implicated in cell proliferation, differentiation, apoptosis, tumorigenesis and cellular stress reactions. NF-κB activation might regulate several genes expression leading to cell survival. These results were in harmony with74 where RSV was concluded to suppress NF-κB by blocking TNF-induced activation of NF-κB in a dose- and time-dependent manner.
Reactive oxygen species assay (ROS)
As displayed in Fig. 9, ZN-RSV NPs obtained the largest green fluorescence intensity. ZN-RSV NPs obtained highest ROS activity (156.1 ± 9.4 Pg/106 million cells) which equal 1.27 upfolds of free RSV sample. This concluded that ZN-RSV NPs could induce oxidative stress in cancer cells by increasing ROS formation via oxidation of NADPH75. RSV demonstrated a protective potential regarding to ROS and nitric oxide synthesis61. ROS can generate subsequent toxic radicals like hydroxyl, hydrogen peroxide and superoxide which react to DNA and proteins on cellular level leading to DNA damage and lipid peroxidation69,76. The anti-peroxidative action of RSV was reported by77 and correlated to capability of phenolic groups to regain lipid hydroperoxyl, hydroxyl and superoxide anions. Also, apoptosis induced by RSV in NCI-H460 non-small cell lung cancer cells and human colon cancer cells was contributed to ROS levels regulations in that trigger downstream signaling pathways as antioxidant or pro-oxidant78,79.
Figure 9.
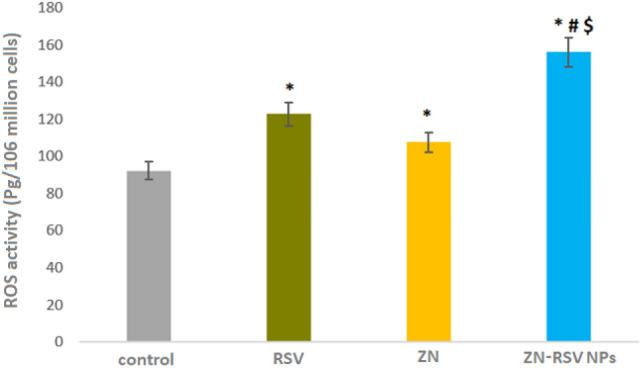
ROS activity in control, RSV and ZN-RSV NPs samples. *Significantly different in comparison with control group at p < 0.05. #Significantly different in comparison with RSV group at p < 0.05. $Significantly different in comparison with ZN group at p < 0.05.
eNOS activity assay
As reported in51 eNOS is expressed in many cell lines as colon cell line HCT-116. The response was explained as a response of tumor cells to the therapeutic agent. The results revealed increased levels of eNOS isoenzyme in ZN-RSV NPs sample in comparison with control and free RSV samples as exhibited in Fig. 10. There was increased significant production of eNOS in RSV and ZN-RSV NPs sample with obtained values of 10.72 and 12.58 ng/million cells, respectively. Zein base nano-particulates was a successful option for enhancing NO production induced by RSV in HCT-116 cells. This was in accordance with80 where suppression of tumorigenicity and abrogation of cancer metastasis is related directly to direct NO production, which confirm apoptosis potential. Also, it has been concluded that RSV enhanced endothelial NO release through long and short-term effects where rapid effect comprises phosphorylation of eNOS by AMP-activated kinase, or extracellular signal regulated kinase 1 and 2, and deacetylation of eNOS by sirtuin181.
Figure 10.
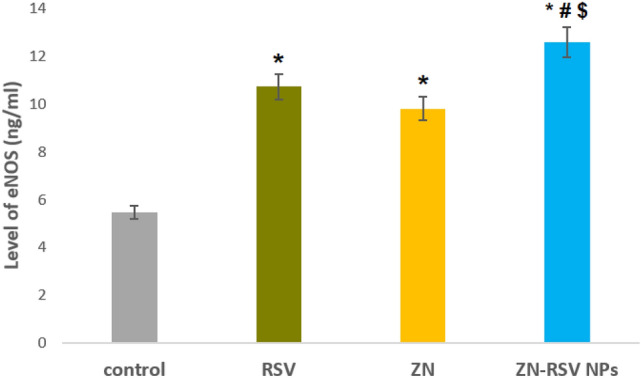
Levels of eNOS in control, RSV and ZN-RSV NPs samples. *Significantly different in comparison with control (p < 0.05). #Significantly different in comparison with RSV (p < 0.05).
Materials and methods
Materials
Zein (CAS number: 9010-66-6, ZN), RSV (CAS number: 501-36-0) and Absolute Ethanol were purchased from Sigma-Aldrich (St. Louis, MO, USA). All of other solvents and chemicals used were of analytical grade.
ZN-RSV NPs formulation
ZN-RSV NPs were prepared using nanoprecipitation method40,42,55. Three ZN-RSV NPs formulae (R1–R3) were prepared and varied according to the applied drug:polymer ratios. RSV and ZN were dissolved in 10 ml of 85% ethanol using of vortex (Velp scientifica, ZX3, Usmate, Italy) and ultrasonic probe (Vibra-Cell VCX750; Sonics and Materials Inc., Newtown, CT, USA). The ethanolic dispersion was flowed into deionized water and magnetically stirred at room temperature for 3 h at 2000 rpm to vaporize content of ethanol. The aqueous suspension was then centrifuged at 20,000×g speed then lyophilized using cryoprotectant of trehalose.
ZN-RSV NPs characterization
Particle size analysis
Laser diffraction technique was utilized for particle size analysis of ZN-RSV NPs formulae (R1–R3). Using disposable cuvette, one milliliter of the sample was diluted in deionized water. The average particle size was then determined.
Encapsulation efficiency (EE)
Ethanol solubilized samples were filtered through 0.22 µm filters, and analyzed for (EE) using reported high-performance liquid chromatography (HPLC) (Agilent 1200, Agilent Technologies, Santa Clara, CA, USA) equipped with a C18 column (5 μm, 4.6 mm × 250 mm), photodiode array detector (PDAD) at 306 nm (Waters, Milford, USA), adjusted column temperature at 35 °C, mixture of methanol and purified water (50:50 v/v) as mobile phase and 1 ml/min flow rate according to82. RSV EE was estimated according to the following equation:
| 1 |
In vitro RSV diffusion
RSV diffusion was carried out according to42 using automated Franz diffusion cell apparatus (MicroettePlus, Hanson Research, Chatsworth, CA, USA) of 7 ml chamber volume and 1.76 cm2 diffusion are using Phosphate-buffered saline (0.01 M, pH 7.0) at maintained temperature of 37 ± 0.5 °C. Synthetic nylon membrane of 0.45 μm pore size (Pall Corporation, Port Washington, NY, USA) was the utilized diffusion membrane. The study was proceeded for 36 h with selected intervals at 0.25, 0.5, 1, 2, 4, 8, 12, 18, 24 and 36 h. Aliquots were analyzed for RSV content utilizing validated HPLC method82.
Nanoparticles morphology
Scanning Electron Microscope (SEM) instrument (JEM 100-CX; JEOL, Tokyo, Japan) was used to investigate nanoparticles surface morphology of ZN-RSV NPs. Using the lyophilized powder, sample was coated under vacuum by gold after fixation.
Serum stability
ZN-RSV NPs Colloidal stability was examined in Fetal Bovine Serum (FBS, Gibco, Thermo Fisher Scientific, MA, USA)56. 1 ml of 70% FBS was added to 200 µl of sample suspension and then incubated at 37 °C for 48 h followed by magnetic stirring at 600 rpm. At selected time intervals of incubation, particle size analysis was carried out.
Cell lines and culture
Human colorectal carcinoma cell line (HCT-116), Human colorectal adenocarcinoma (Caco-2), Human colorectal adenocarcinoma (HT-29) and Human somatic cell hybrid (EA.hy926) cells were supplied from Nawah Scientific, Cairo, Egypt that were purchased from the American Type Culture Collection (ATCC; Manassas, VA, USA). HCT-116 were cultured in ATCC-formulated McCoy's 5a Medium Modified and Caco-2, HT-29 and EA.hy926 Cells were cultured in ATCC-formulated Eagle's Minimum Essential Medium. The mediums were supplemented with fetal bovine serum to a final concentration of 10% and 20%. Culture plates and flasks (SPL Life Sciences, Korea) were subjected to addition of Penicillin (100 U/ml) and streptomycin (100 mg/ml) previously. Cells were maintained inside carbon dioxide incubator at 37 °C in a humidified atmosphere containing 5% CO2 (Thermo Electron Corporation, Forma series II, 3141, USA) to keep the cells in a sub-confluent state.
Cytotoxicity assay
MTT assay was utilized to evaluate the antiproliferative activity using the kit of ABCAM, Cambridge, UK according to83, where HCT-116, Caco-2, HT-29 and EA.hy926 cells were introduced into 96-well plates (TPP, Switzerland) as 2 × 103 cells/well approximately. Wells were incubated with RSV, equivalent weight of ZN or ZN-RSV NPs using a range of concentrations at logarithmic manner at 37 °C in a carbon dioxide incubator for 48 h.
Cellular uptake analysis
In the presence of 5% CO2, HCT-116 cells were incubated overnight (1 × 105 cells/dish) at 37 °C for 2 and 4 h after treating with ZN-RSV NPs IC50 value, equivalent concentration of RSV or ZN. Lysis solution was added for 30 min at 37 °C after monolayers were washing. HPLC technique was utilized for analyzing of cell lysates aliquots.
Cell cycle progression analysis
This assay was carried out as described by55,56 using the same reagents and kits.
Annexin-V assay
This assay was carried out as described by55,56 using the same reagents and kits.
mRNA expression of apoptosis-related genes by quantitative real-time polymerase chain reaction (RT-PCR)
This assay was carried out as reported by55,56 using the same reagents and kits. Table 2 gives the primer sequences for caspase-3, miRNA125b, NF-κB and β-actin. Results were validated using the relative quantification (ΔΔCT) method. The genes expression was estimated in triplicates and runs mean was normalized with β-actin mean.
Table 2.
Primer sequences used for RT-PCR.
| Primer | Sequence | |
|---|---|---|
| Caspase-3 | Forward primer | 5′-GTGGAACTGACGATGATATGGC-3′ |
| Reverse primer | 5′-CGCAAAGTGACTGGATGAACC-3′ | |
| miRNA125b | Forward primer | 5′-CTTGCCAGAAACGTCAATGGA-3′ |
| Reverse primer | 5′-GTGCAACTACGTCATAGCCTG-3′ | |
| NF-κB | Forward primer | 5′-ATCCCATCTTTGACAATCGTGC-3′ |
| Reverse primer | 5′-CTGGTCCCGTGAAATACACCTC-3′ | |
| β-Actin | Forward primer | 5′-AAGATCCTGACCGAGCGTGG-3′ |
| Reverse primer | 5′-CAGCACTGTGTTGGCATAGAGG-3′ |
Reactive oxygen species assay (ROS)
IC50 concentrations of ZN-RSV NPs and equivalent concentrations of RSV and ZN were incubated in 96 well plates with adjusted cell density at 5 × 103 HCT-116 cells/well for period of 24 h. For staining, we used 10 μM 2,7-dichlorofluorescein diacetate (DCFDA) for 45 min. The used kit was ABCAM, Cambridge, UK.
Endothelial nitric oxide synthase (eNOS) activity assay
Enzyme-linked immunosorbent assay kit (RayBio® Human eNOS ELISA Kit, Norcross, GA, USA) was used to measure eNOS levels using 96-well plate. After adding of standards and samples into plates, biotinylated antihuman eNOS antibody is added where eNOS was present bounded to plate wells by immobilized antibody. HRP conjugated streptavidin is introduced to wells after washing. Second washing is carried out before the addition of TMB substrate solution. Then color was generated according to quantity of eNOS attached. Color was changed from blue to yellow after addition of stop solution. Finally, color intensity was measured at 450 nm.
Statistical analysis
Study data were displayed as means ± standard deviation (SD) of at least three independent runs. Comparative statistics between treatments was carried out by Student’s t test or one-way analysis of variance followed by Tukey’s test (criterion of significance was considered two-tailed p value less than 0.05). The utilized software was IBM SPSS software (Trial online version, SPSS Inc., Chicago, IL, USA).
Conclusions
The present study explored the ameliorating impact of nanoformulation on the molecular mechanism of RSV as anticancer molecule against Human CRC cells HCT-116. ZN-RSV NPs were fabricated with mean particle size of 137.6 ± 8.3 nm using the promising biomaterial of ZN. The developed ZN-RSV-NPs attained elevated EE%, 36 h sustained diffusion and colloidal stability. The enhanced cytotoxicity and cellular uptake, ameliorated pro-apoptotic potential supported by enhanced mRNA expression of caspase3, cleaved caspase-3, miRNA125b and NF-κB genes confirmed the enhanced anti-proliferative and pro-apoptotic potentials of ZN-RSV NPs. Biochemical examining exhibited increased generation of ROS and eNOS levels due to the ZN-RSV NPs formulation which displayed enhanced oxidant profile. The obtained results concluded therapeutical intensification of RSV antitumor potential against Human CRC cells HCT-116 due to the developed nano-delivery system.
Acknowledgements
The authors acknowledge financial support from the Researchers Supporting Project number (RSP-2022/111), King Saud University, Riyadh, Saudi Arabia.
Abbreviations
- CRC
Colorectal carcinoma
- EE
Encapsulation efficiency
- eNOS
Endothelial nitric oxide synthase
- HPLC
High-performance liquid chromatography
- NPs
Nanoparticles
- NF-κB
Nuclear factor kappa-light-chain-enhancer of activated B cells
- RSV
Resveratrol
- ROS
Reactive oxygen species
- RT-PCR
Real-time polymerase chain reaction
- SEM
Scanning electron microscope
- ZN
Zein
Author contributions
A.M.H., M.M.S., M.T.K., A.B.A. and D.F.T. designed and conducted the experiments. R.F.T., M.M.S., M.A.Z., H.I.K., A.B.A., E.M.O. and D.F.T. analyzed the results. All authors wrote and reviewed the manuscript.
Data availability
The datasets generated and analyzed during the current study are available from the corresponding author on reasonable request.
Competing interests
The authors declare no competing interests.
Footnotes
This article has been retracted. Please see the retraction notice for more detail: https://doi.org/10.1038/s41598-025-25537-9
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Change history
10/28/2025
This article has been retracted. Please see the Retraction Notice for more detail: 10.1038/s41598-025-25537-9
References
- 1.Jasiński, M., Jasińska, L. & Ogrodowczyk, M. Resveratrol in prostate diseases—A short review. Cent. Eur. J. Urol.66, 144 (2013). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Langcake, P. & Pryce, R. The production of resveratrol by Vitis vinifera and other members of the Vitaceae as a response to infection or injury. Physiol. Plant Pathol.9, 77–86 (1976). [Google Scholar]
- 3.Adrian, M., Jeandet, P., Veneau, J., Weston, L. A. & Bessis, R. Biological activity of resveratrol, a stilbenic compound from grapevines, against Botrytis cinerea, the causal agent for gray mold. J. Chem. Ecol.23, 1689–1702 (1997). [Google Scholar]
- 4.Schubert, R. et al. An ozone-responsive region of the grapevine resveratrol synthase promoter differs from the basal pathogen-responsive sequence. Plant Mol. Biol.34, 417–426 (1997). [DOI] [PubMed] [Google Scholar]
- 5.Douillet-Breuil, A.-C., Jeandet, P., Adrian, M. & Bessis, R. Changes in the phytoalexin content of various Vitis spp. in response to ultraviolet C elicitation. J. Agric. Food Chem.47, 4456–4461 (1999). [DOI] [PubMed] [Google Scholar]
- 6.Pezzuto, J. M. Resveratrol: Twenty years of growth, development and controversy. Biomol. Ther.27, 1 (2019). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Brotons-Canto, A., Gonzalez-Navarro, C. J., Gurrea, J., González-Ferrero, C. & Irache, J. M. Zein nanoparticles improve the oral bioavailability of resveratrol in humans. J. Drug Deliv. Sci. Technol.57, 101704 (2020). [Google Scholar]
- 8.Kundu, J. K. & Surh, Y.-J. Cancer chemopreventive and therapeutic potential of resveratrol: Mechanistic perspectives. Cancer Lett.269, 243–261 (2008). [DOI] [PubMed] [Google Scholar]
- 9.Liu, Z., Wu, X., Lv, J., Sun, H. & Zhou, F. Resveratrol induces p53 in colorectal cancer through SET7/9. Oncol. Lett.17, 3783–3789 (2019). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Richard, J. Coronary risk factors. The French paradox. Arch. Mal. Coeur Vaiss.80, 17–21 (1987). [PubMed] [Google Scholar]
- 11.Jang, M. et al. Cancer chemopreventive activity of resveratrol, a natural product derived from grapes. Science275, 218–220 (1997). [DOI] [PubMed] [Google Scholar]
- 12.Varoni, E. M., Lo Faro, A. F., Sharifi-Rad, J. & Iriti, M. Anticancer molecular mechanisms of resveratrol. Front. Nutr.3, 8 (2016). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Dembic, M. et al. Next generation sequencing of RNA reveals novel targets of resveratrol with possible implications for Canavan disease. Mol. Genet. Metab.126, 64–76 (2019). [DOI] [PubMed] [Google Scholar]
- 14.Amiri, F. et al. Synergistic anti-proliferative effect of resveratrol and etoposide on human hepatocellular and colon cancer cell lines. Eur. J. Pharmacol.718, 34–40 (2013). [DOI] [PubMed] [Google Scholar]
- 15.Honari, M., Shafabakhsh, R., Reiter, R. J., Mirzaei, H. & Asemi, Z. Resveratrol is a promising agent for colorectal cancer prevention and treatment: Focus on molecular mechanisms. Cancer Cell Int.19, 1–8 (2019). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Karimi Dermani, F. et al. Resveratrol inhibits proliferation, invasion, and epithelial–mesenchymal transition by increasing miR-200c expression in HCT-116 colorectal cancer cells. J. Cell. Biochem.118, 1547–1555 (2017). [DOI] [PubMed] [Google Scholar]
- 17.Liu, B. et al. Resveratrol inhibits proliferation in human colorectal carcinoma cells by inducing G1/S-phase cell cycle arrest and apoptosis through caspase/cyclin-CDK pathways. Mol. Med. Rep.10, 1697–1702 (2014). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Wang, Z. et al. Resveratrol induces AMPK-dependent MDR1 inhibition in colorectal cancer HCT116/L-OHP cells by preventing activation of NF-κB signaling and suppressing cAMP-responsive element transcriptional activity. Tumor Biol.36, 9499–9510 (2015). [DOI] [PubMed] [Google Scholar]
- 19.Zeng, Y.-H. et al. Resveratrol inactivates PI3K/Akt signaling through upregulating BMP7 in human colon cancer cells. Oncol. Rep.38, 456–464 (2017). [DOI] [PubMed] [Google Scholar]
- 20.Pan, M. H., Lai, C. S., Wu, J. C. & Ho, C. T. Molecular mechanisms for chemoprevention of colorectal cancer by natural dietary compounds. Mol. Nutr. Food Res.55, 32–45 (2011). [DOI] [PubMed] [Google Scholar]
- 21.The International Agency for Research on Cancer. Colorectal Cancer Awareness Month 2022 (2022).
- 22.Kuipers, E. J. et al. Colorectal cancer. Nat. Rev. Dis. Primers1, 15065. 10.1038/nrdp.2015.65 (2015). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Brown, J. M. & Attardi, L. D. The role of apoptosis in cancer development and treatment response. Nat. Rev. Cancer5, 231–237 (2005). [DOI] [PubMed] [Google Scholar]
- 24.Kim, R., Emi, M. & Tanabe, K. The role of apoptosis in cancer cell survival and therapeutic outcome. Cancer Biol. Ther.5, 1429–1442 (2006). [DOI] [PubMed] [Google Scholar]
- 25.Gerl, R. & Vaux, D. L. Apoptosis in the development and treatment of cancer. Carcinogenesis26, 263–270 (2005). [DOI] [PubMed] [Google Scholar]
- 26.Elzoghby, A., Freag, M., Mamdouh, H. & Elkhodairy, K. Zein-based nanocarriers as potential natural alternatives for drug and gene delivery: Focus on cancer therapy. Curr. Pharm. Des.23, 5261–5271 (2017). [DOI] [PubMed] [Google Scholar]
- 27.Houdaihed, L., Evans, J. C. & Allen, C. Dual-targeted delivery of nanoparticles encapsulating paclitaxel and everolimus: A novel strategy to overcome breast cancer receptor heterogeneity. Pharm. Res.37, 1–10 (2020). [DOI] [PubMed] [Google Scholar]
- 28.Houdaihed, L., Evans, J. & Allen, C. Delivery of paclitaxel and everolimus in dual-targeted polymeric nanoparticles to breast cancer cells. Ann. Oncol.29, iii21 (2018). [Google Scholar]
- 29.Luo, Y., Zhang, B., Whent, M., Yu, L. L. & Wang, Q. Preparation and characterization of zein/chitosan complex for encapsulation of α-tocopherol, and its in vitro controlled release study. Colloids Surf. B85, 145–152 (2011). [DOI] [PubMed] [Google Scholar]
- 30.Pérez-Masiá, R., López-Rubio, A. & Lagarón, J. M. Development of zein-based heat-management structures for smart food packaging. Food Hydrocolloids30, 182–191 (2013). [Google Scholar]
- 31.Berardi, A., Bisharat, L., AlKhatib, H. S. & Cespi, M. Zein as a pharmaceutical excipient in oral solid dosage forms: State of the art and future perspectives. AAPS PharmSciTech19, 2009–2022 (2018). [DOI] [PubMed] [Google Scholar]
- 32.Rosales-Mendoza, S., Sández-Robledo, C., Bañuelos-Hernández, B. & Angulo, C. Corn-based vaccines: Current status and prospects. Planta245, 875–888 (2017). [DOI] [PubMed] [Google Scholar]
- 33.Paliwal, R. & Palakurthi, S. Zein in controlled drug delivery and tissue engineering. J. Control. Release189, 108–122 (2014). [DOI] [PubMed] [Google Scholar]
- 34.Ahmed, O. A., Hosny, K. M., Al-Sawahli, M. M. & Fahmy, U. A. Optimization of caseinate-coated simvastatin-zein nanoparticles: Improved bioavailability and modified release characteristics. Drug Des. Dev. Ther.9, 655 (2015). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Lai, L. & Guo, H. Preparation of new 5-fluorouracil-loaded zein nanoparticles for liver targeting. Int. J. Pharm.404, 317–323 (2011). [DOI] [PubMed] [Google Scholar]
- 36.Anderson, T. J. & Lamsal, B. P. Zein extraction from corn, corn products, and coproducts and modifications for various applications: A review. Cereal Chem.88, 159–173 (2011). [Google Scholar]
- 37.Fernandes, R. S. et al. α-Tocopherol succinate loaded nano-structured lipid carriers improves antitumor activity of doxorubicin in breast cancer models in vivo. Biomed. Pharmacother.103, 1348–1354 (2018). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Oda, C. M. R. et al. Synthesis, characterization and radiolabeling of polymeric nano-micelles as a platform for tumor delivering. Biomed. Pharmacother.89, 268–275 (2017). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Summerlin, N. et al. Resveratrol nanoformulations: Challenges and opportunities. Int. J. Pharm.479, 282–290 (2015). [DOI] [PubMed] [Google Scholar]
- 40.Algandaby, M. M. et al. Curcumin-zein nanospheres improve liver targeting and antifibrotic activity of curcumin in carbon tetrachloride-induced mice liver fibrosis. J. Biomed. Nanotechnol.12, 1746–1757 (2016). [DOI] [PubMed] [Google Scholar]
- 41.Brigger, I., Dubernet, C. & Couvreur, P. Nanoparticles in cancer therapy and diagnosis. Adv. Drug Deliv. Rev.64, 24–36 (2012). [DOI] [PubMed] [Google Scholar]
- 42.Hashem, F. M., Al-Sawahli, M. M., Nasr, M. & Ahmed, O. A. Optimized zein nanospheres for improved oral bioavailability of atorvastatin. Int. J. Nanomed.10, 4059–4069. 10.2147/ijn.s83906 (2015). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Liversidge, G. G. & Cundy, K. C. Particle size reduction for improvement of oral bioavailability of hydrophobic drugs: I. Absolute oral bioavailability of nanocrystalline danazol in beagle dogs. Int. J. Pharm.125, 91–97 (1995). [Google Scholar]
- 44.Alhakamy, N. A. et al. Encapsulation of lovastatin in zein nanoparticles exhibits enhanced apoptotic activity in hepg2 cells. Int. J. Mol. Sci.20, 5788 (2019). [DOI] [PMC free article] [PubMed] [Google Scholar] [Retracted]
- 45.Lee, J. H. & Yeo, Y. Controlled drug release from pharmaceutical nanocarriers. Chem. Eng. Sci.125, 75–84 (2015). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Petros, R. A. & DeSimone, J. M. Strategies in the design of nanoparticles for therapeutic applications. Nat. Rev. Drug Discov.9, 615–627 (2010). [DOI] [PubMed] [Google Scholar]
- 47.Padua, G. W. & Wang, Q. Controlled self-organization of zein nanostructures for encapsulation of food ingredients. 143–156 (2009).
- 48.Almeida, L. et al. Pharmacokinetic and safety profile of trans-resveratrol in a rising multiple-dose study in healthy volunteers. Mol. Nutr. Food Res.53, S7–S15 (2009). [DOI] [PubMed] [Google Scholar]
- 49.Walle, T., Hsieh, F., DeLegge, M. H., Oatis, J. E. & Walle, U. K. High absorption but very low bioavailability of oral resveratrol in humans. Drug Metab. Dispos.32, 1377–1382 (2004). [DOI] [PubMed] [Google Scholar]
- 50.Zu, Y. et al. Preparation and in vitro/in vivo evaluation of resveratrol-loaded carboxymethyl chitosan nanoparticles. Drug Deliv.23, 971–981 (2016). [DOI] [PubMed] [Google Scholar]
- 51.Adams, B. D., Guttilla, I. K. & White, B. A. Seminars in Reproductive Medicine 522–536 (Thieme Medical Publishers, 2008). [DOI] [PubMed] [Google Scholar]
- 52.Dhanapal, J. & Balaraman Ravindrran, M. Chitosan/poly (lactic acid)-coated piceatannol nanoparticles exert an in vitro apoptosis activity on liver, lung and breast cancer cell lines. Artif. Cells Nanomed. Biotechnol.46, 274–282 (2018). [DOI] [PubMed] [Google Scholar]
- 53.Oberdörster, G. et al. Principles for characterizing the potential human health effects from exposure to nanomaterials: Elements of a screening strategy. Part. Fibre Toxicol.2, 8 (2005). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Elzoghby, A. O., El-Lakany, S. A., Helmy, M. W., Abu-Serie, M. M. & Elgindy, N. A. Shell-crosslinked zein nanocapsules for oral codelivery of exemestane and resveratrol in breast cancer therapy. Nanomedicine12, 2785–2805 (2017). [DOI] [PubMed] [Google Scholar]
- 55.Algandaby, M. M. & Al-Sawahli, M. M. Augmentation of anti-proliferative, pro-apoptotic and oxidant profiles induced by piceatannol in human breast carcinoma MCF-7 cells using zein nanostructures. Biomed. Pharmacother.138, 111409 (2021). [DOI] [PubMed] [Google Scholar]
- 56.Kutbi, H. I., Kammoun, A. K. & El-Telbany, D. F. Amelioration of pterostilbene antiproliferative, proapoptotic, and oxidant potentials in human breast cancer MCF7 cells using zein nanocomposites. Int. J. Nanomed.16, 3059 (2021). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Conner, S. D. & Schmid, S. L. Regulated portals of entry into the cell. Nature422, 37–44 (2003). [DOI] [PubMed] [Google Scholar]
- 58.Kanwar, J. R., Samarasinghe, R. M., Sehgal, R. & Kanwar, R. K. Nano-lactoferrin in diagnostic, imaging and targeted delivery for cancer and infectious diseases. J. Cancer Sci. Ther.4, 31–42 (2012). [Google Scholar]
- 59.Li, D. et al. Resveratrol suppresses colon cancer growth by targeting the AKT/STAT3 signaling pathway. Int. J. Mol. Med.43, 630–640 (2019). [DOI] [PubMed] [Google Scholar]
- 60.Tsunoda, T. et al. Resveratrol induces luminal apoptosis of human colorectal cancer HCT116 cells in three-dimensional culture. Anticancer Res.34, 4551–4555 (2014). [PubMed] [Google Scholar]
- 61.Delmas, D. et al. Resveratrol-induced apoptosis is associated with Fas redistribution in the rafts and the formation of a death-inducing signaling complex in colon cancer cells. J. Biol. Chem.278, 41482–41490 (2003). [DOI] [PubMed] [Google Scholar]
- 62.Fouad, M., Agha, A., Merzabani, M. A. & Shouman, S. Resveratrol inhibits proliferation, angiogenesis and induces apoptosis in colon cancer cells: Calorie restriction is the force to the cytotoxicity. Hum. Exp. Toxicol.32, 1067–1080 (2013). [DOI] [PubMed] [Google Scholar]
- 63.Park, J.-W. et al. Resveratrol induces pro-apoptotic endoplasmic reticulum stress in human colon cancer cells. Oncol. Rep.18, 1269–1273 (2007). [PubMed] [Google Scholar]
- 64.Woo, K. J. et al. Elevated gadd153/chop expression during resveratrol-induced apoptosis in human colon cancer cells. Biochem. Pharmacol.73, 68–76 (2007). [DOI] [PubMed] [Google Scholar]
- 65.Feng, M., Zhong, L., Zhan, Z., Huang, Z. & Xiong, J. Enhanced antitumor efficacy of resveratrol-loaded nanocapsules in colon cancer cells: Physicochemical and biological characterization. Eur. Rev. Med. Pharmacol. Sci.21, 375–382 (2017). [PubMed] [Google Scholar]
- 66.Olsson, M. & Zhivotovsky, B. Caspases and cancer. Cell Death Differ.18, 1441–1449 (2011). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 67.Porter, A. G. & Jänicke, R. U. Emerging roles of caspase-3 in apoptosis. Cell Death Differ.6, 99–104 (1999). [DOI] [PubMed] [Google Scholar]
- 68.Santos, A. C. et al. Targeting cancer via resveratrol-loaded nanoparticles administration: Focusing on in vivo evidence. AAPS J.21, 1–16 (2019). [DOI] [PubMed] [Google Scholar]
- 69.Sengottuvelan, M., Deeptha, K. & Nalini, N. Resveratrol ameliorates DNA damage, prooxidant and antioxidant imbalance in 1, 2-dimethylhydrazine induced rat colon carcinogenesis. Chem. Biol. Interact.181, 193–201 (2009). [DOI] [PubMed] [Google Scholar]
- 70.Venkatadri, R., Muni, T., Iyer, A., Yakisich, J. & Azad, N. Role of apoptosis-related miRNAs in resveratrol-induced breast cancer cell death. Cell Death Dis.7, e2104 (2016). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 71.Arunachalam, G., Yao, H., Sundar, I. K., Caito, S. & Rahman, I. SIRT1 regulates oxidant-and cigarette smoke-induced eNOS acetylation in endothelial cells: Role of resveratrol. Biochem. Biophys. Res. Commun.393, 66–72 (2010). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 72.Lee, C. H., Jeon, Y. T., Kim, S. H. & Song, Y. S. NF-κB as a potential molecular target for cancer therapy. BioFactors29, 19–35 (2007). [DOI] [PubMed] [Google Scholar]
- 73.Yu, Y., Wan, Y. & Huang, C. The biological functions of NF-κB1. Curr. Cancer Drug Targets9, 566–571 (2009). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 74.Manna, S. K., Mukhopadhyay, A. & Aggarwal, B. B. Resveratrol suppresses TNF-induced activation of nuclear transcription factors NF-κB, activator protein-1, and apoptosis: Potential role of reactive oxygen intermediates and lipid peroxidation. J. Immunol.164, 6509–6519 (2000). [DOI] [PubMed] [Google Scholar]
- 75.Treml, J. et al. Antioxidant activity of selected stilbenoid derivatives in a cellular model system. Biomolecules9, 468 (2019). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 76.Heo, J. R., Kim, S. M., Hwang, K. A., Kang, J. H. & Choi, K. C. Resveratrol induced reactive oxygen species and endoplasmic reticulum stress-mediated apoptosis, and cell cycle arrest in the A375SM malignant melanoma cell line. Int. J. Mol. Med.42, 1427–1435 (2018). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 77.Murias, M. et al. Antioxidant, prooxidant and cytotoxic activity of hydroxylated resveratrol analogues: Structure–activity relationship. Biochem. Pharmacol.69, 903–912 (2005). [DOI] [PubMed] [Google Scholar]
- 78.Karthikeyan, S., Prasad, N. R., Ganamani, A. & Balamurugan, E. Anticancer activity of resveratrol-loaded gelatin nanoparticles on NCI-H460 non-small cell lung cancer cells. Biomed. Prev. Nutr.3, 64–73 (2013). [Google Scholar]
- 79.Miki, H. et al. Resveratrol induces apoptosis via ROS-triggered autophagy in human colon cancer cells. Int. J. Oncol.40, 1020–1028 (2012). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 80.Oktem, G. et al. Differential effects of doxorubicin and docetaxel on nitric oxide production and inducible nitric oxide synthase expression in MCF-7 human breast cancer cells. Oncol. Res. Featur. Preclin. Clin. Cancer Ther.14, 381–386 (2004). [DOI] [PubMed] [Google Scholar]
- 81.Ladurner, A. et al. Impact of trans-resveratrol-sulfates and-glucuronides on endothelial nitric oxide synthase activity, nitric oxide release and intracellular reactive oxygen species. Molecules19, 16724–16736 (2014). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 82.Cheng, H., Fang, Z., Bakry, A. M., Chen, Y. & Liang, L. Complexation of trans- and cis-resveratrol with bovine serum albumin, β-lactoglobulin or α-lactalbumin. Food Hydrocoll.81, 242–252 (2018). [Google Scholar]
- 83.Al-Gendy, A., Moharram, F. & Zarka, M. Chemical composition, antioxidant, cytotoxic and antimicrobial activities of Pimentaracemosa (Mill.) JW Moore flower essential oil. J. Pharmacogn. Phytochem.6, 312–319 (2017). [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
The datasets generated and analyzed during the current study are available from the corresponding author on reasonable request.