Abstract
Purpose
Previous studies have suggested that circulating extracellular vesicles (EVs) arise after high intensity exercise and urine could reflect the plasma proteome. Herein, we investigated the characteristic of urinary EVs from healthy young adult males who had completed a maximal effort exercise test.
Methods
Thirteen healthy men completed a 20 m shuttle run test (20 m SRT). Fresh urine samples were collected at first morning, right after, and 1 h rest after 20 m SRT. Also, blood lactate, heart rate, rating of perceived exertion, and blood pressure were measured before, right after, and 1 h rest after 20 m SRT. Urinary EVs were analyzed using Exoview instrument and microRNAs (miRNAs) sequencing on urinary EVs were performed.
Results
Urinary EVs increased significantly after exercise and returned to baseline value after 1 h of rest. miRNA sequencing on urinary EV revealed alterations in four miRNAs (1 up and 3 down) and nine miRNAs (2 up and 7 down) in pre- vs. post- and post- vs. post-1 h samples, respectively. Lastly, bioinformatic analysis of urinary EV miRNA suggests that predicted target genes could affect PI3K-Akt, mitogen-activated protein kinase, and insulin pathways by exercise.
Conclusions
Exercise to voluntary exhaustion increased the number of EVs in urine. Also, miRNAs in urinary EVs were altered after exercise. These findings could indicate the possibility of using the urinary EVs as a novel biomarker of acute exercise-induced fatigue.
Keywords: Urine, Extracellular vesicle, Fatigue, Exercise, miRNA
Introduction
Participating in regular exercise is one of the best ways to prevent chronic diseases and maintain longevity. Even a single bout of exercise could be beneficial for cardiac function (Boutcher et al. 2011; Pober et al. 2004). Exercise could change the metabolic phenotype of our system (Amar et al. 2021; Frampton et al. 2021; Kim et al. 2019), including increased insulin sensitivity, glucose uptake (Hayashi et al. 2005), oxidative capacity, secretion of various peptides, extracellular vesicles, and metabolites (McGee and Hargreaves 2020). However, excessive exercise or training without sufficient recovery could evoke short-term overreached or overtraining syndrome, resulting in constant fatigue and underperformance (Halson and Jeukendrup 2004; Urhausen and Kindermann 2002).
The common parameters for quantifying the intensity of training are heart rate (HR), rating of perceived exertion (RPE), and blood lactate (BLa) (Wallace et al. 2014). However, these markers have limitations. RPE, which is a subjective marker, sometimes overestimates the intensity (Scherr et al. 2013). BLa analysis has hygiene issues, and it is difficult to collect blood samples several times (Green et al. 2006). Similarly, although HR is a useful marker, self-monitoring and self-regulating the exercise intensity with HR is difficult (Ciolac et al. 2015). Blood pressure (BP), which increases progressively with exercise intensity, could elevate during rest due to anxiety (Miyai et al. 2002). Therefore, there is a need for an alternative marker to objectively detect individually perceived intensity or fatigue. We consider EVs to be a resource that complements, not replaces, these fatigue markers.
EVs are found in most bodily fluids, such as blood, urine, and saliva (Colombo et al. 2014). Among them, urine is easy to collect in relatively large volumes compared to other bodily fluids, and not only contains kidney-derived proteins, peptides, and metabolites, but also reveals the components present in plasma (Harpole et al. 2016). These small sized vesicles are secreted by the cells (Jeppesen et al. 2019). Thus, EVs contain integral components, such as proteins, mRNA, and miRNA of originating cells (Yellon and Davidson 2014). In this regard, they could act as representatives of the cellular origin and physiological state of the secreting cells (Zhang et al. 2019).
The kinetics of EVs in plasma shows a similar increase as BLa during incremental cycling (Frühbeis et al. 2015a). High intensity endurance exercise was reported to elevate EVs in circulation (Wilhelm et al. 2016, 2017). Resistance training after endurance exercise also facilitates multivesicular body (MVB) biogenesis, partially stimulating exosome biogenesis (Garner et al. 2020). Studies have suggested that an acute bout of exercise can stimulate the release of EVs from muscles and other tissues (Safdar et al. 2016; Whitham et al. 2018). In addition, endurance exercise can change the proteomics of urine (Kohler et al. 2015). Therefore, it is possible that exercise could affect the characteristics of urinary EVs and their components.
However, no study has elucidated the effects of exercise on urinary EVs. Therefore, this study attempted to define the characteristics and possible functions of urinary EVs after the maximal effort test with two hypotheses. First, the number of EVs will be altered after 20 m SRT. Second, the profiles of miRNAs in urinary EVs will be changed after 20 m SRT.
Materials and methods
Ethical approval
All experimental procedures were approved by the Institutional Review Board of Seoul National University in accordance with the standards of the Declaration of Helsinki of the World Medical Association (IRB No. 2009/003-028). All subjects were informed about the procedures and purpose of the study by oral and written forms, and they confirmed to take part in it.
Subjects
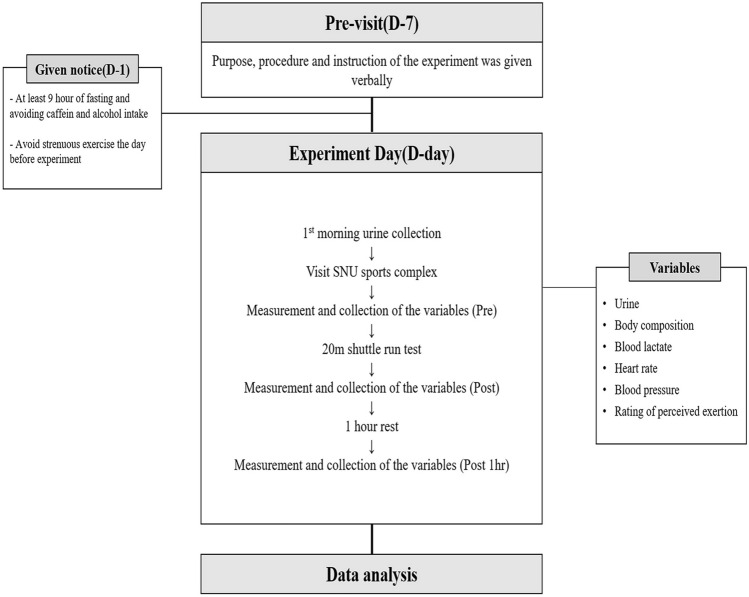
Thirteen healthy men aged 25.5 ± 2.0 years were recruited from Seoul National University in response to an advertisement. Men with a history of cardiovascular disease, musculoskeletal disease, or psychological disorder were excluded (more details about the inclusion and exclusion criteria are shown in Table 1). Subject characteristics are shown in Table 2. All participants were encouraged to fast for at least 9 h before the test day. They were also informed about the procedures of the study and the risks of 20 m SRT. Written consent was obtained from all participants before the experiment (Fig. 1).
Table 1.
Inclusion and exclusion criteria
| Inclusion criteria | Exclusion criteria |
|---|---|
|
Subject who(se) Agreed to participate in this study and sign a written consent from Aged 20 to 30 years BMI between 18.5 kg/m2 and 30.0 kg/m2 Normal BP less than 140 mmHg (systolic) and BP less than 90 mmHg (diastolic) Body temperature less than 37.5 °C when examined |
Subject who(se) BP is greater than 140 mmHg (systolic) or the relaxer BP is greater than 90 mmHg (diastolic) Had kidney disease BMI is less than or equal to 18.5 kg/m2 or greater than 30 kg/m2 Cannot walk or run Had hyperlipidemia, asthma, diabetes, bronchitis, anemia, thyroid disease, cardiovascular disease (hypertension, stroke, etc.), liver disease, musculoskeletal disorders, neurological disorders, or past history, or subscribed relate medications Had surgery within 6 months prior to his first visit Considered as inappropriate to participate in this study by a researcher |
Table 2.
Subject characteristics
| Anthropometric characteristics | |
|---|---|
| Variables (n = 13) | |
| Age (year) | 25.5 ± 2.0 |
| Height (cm) | 177.3 ± 5.6 |
| Weight (kg) | 72.0 ± 5.7 |
| Fat percentage (%) | 13.5 ± 4.6 |
| Fat free mass (kg) | 35.6 ± 3.2 |
| 20 m SRT result | |
| Repetition | 81.2 ± 13.9 |
Fig. 1.
Experimental procedure
Urine sample collection and preparation
Participants were asked to collect their first morning urine (Pre) into 50 mL sterile containers (SPL, Gyeonggi-do, Korea). Immediately after 20 m SRT (Post) and after 1 h (Post 1 h) of rest, urine samples were collected within 5 min. After collection, samples were filtered through a Minisart® 0.2 µm filter (Sartorius Stedim, Dourdan, France), and the filtered samples were frozen at − 80 °C for further analysis.
Body composition and blood pressure measurement
Weight, fat percentage, and fat-free mass of participants were measured using an Inbody 720 (Biospace, Korea). Unnecessary outwear and accessories were removed before measurements. Brachial BP was measured in duplicate using a digital sphygmomanometer (Omron™, Matsusaka, Japan). BP was assessed on the non-dominant arm (in this study all left) to measure the rest state (rested 3 min on chair) (Pre), immediately after 20 m SRT (Post), and after 1 h of rest (Post 1 h).
Measurement of rating of perceived exertion
The 15-point Borg rating scale of RPE (Borg 1970) which ranges from 6 to 20, was used to examine perceived exertion after 20 m SRT. Verbal instructions of the standard were given to the participants before measurements. RPE was obtained at the rest state before the test (Pre), immediately after 20 m SRT (Post), and after 1 h of rest (Post 1 h).
EV isolation
EV fractions from filtered urine samples were isolated by using miRCURY Exosome Cell/Urine/CSF Kit (Qiagen, Germany) following the manufacturer’s instructions. In short, 4 mL of precipitation buffer B was added to 10 mL of the urine sample. All samples were incubated at 4 °C overnight (16 h) and centrifuged at 3200 × g for 30 min at 20 °C. The obtained pellet was resuspended in 50 µL of resuspension buffer and frozen at − 80 °C.
Quantification of urine-derived EVs
The physical and biological properties of EVs in urine samples were characterized using ExoView R-100 and ExoView tetraspanin kit including anti-CD 81, anti-CD 63, and anti-CD 9 immobilized chips, labeling agents, washing solutions (solutions A and B), and blocking agents (Nanoview Bioscience, Boston, USA) (Gori et al. 2020).
First, 35 µL of the sample diluted 1:30 in distilled water was dropped onto the ExoView tetraspanin chip and incubated overnight at room temperature (r. t.). After the incubation process, the sample-loaded chip was washed thrice with 1 mL of solution A. Subsequently, the EVs on the chip were labeled using 250 µL of a mixture of anti-CD 81/AF 555, anti-CD 63/AF 647, and anti-CD 9/AF 488 and incubated for 1 h to analyze the colocalization of tetraspanin on the surface of the EV. In this case, the labeling antibody (Ab) was diluted in a mixture of solution A and blocking solution at 1:600. Finally, the chip was rinsed with 1 µL of solutions A and B and dried at r.t. The EV-captured chip was scanned using an ExoView R-100 (Nanoview Bioscience, Boston, USA).
BLa measurement
Using Accu-Chek Softclix plus lancing device and Softclix (Roche®, Germany), 5 µL of blood from the fingertip was aspirated into an enzyme-coated electrode test strip (Arkray, Japan). Concentration levels of BLa were checked with a Lactate Pro2 (Arkray, Japan) device before (Pre), immediately after (Post), and after 1 h of the 20 m SRT (Post 1 h).
All equipment was cleaned and operated according to the manufacturer’s instructions. Blood samples were analyzed within 30 s of collection.
20 m shuttle run test
The participants participated in continuous running between two cones placed 20 m apart in response to the recorded beeps. Before carrying out the test, participants made sure they could hear the beep clearly from each end of the line. After 5 min of warm-up, subjects started running back and forth in response to the beep sound and counts recorded in Korean, with an initial speed of 8 km/h. The speed progressively increased in accordance with the test grade. The total shuttle run number was checked when participants could not reach the end line in response to the beep more than twice.
The test was conducted between 09:00 and 11:00 AM in the Seoul National University Sports Complex.
miRNA sequencing and bioinformatic analysis
EV RNAs obtained from three urinary EV samples were used to generate sequencing libraries with the SMARTer smRNA-Seq Kit for Illumina (Takara Bio, Shiga, Japan) according to the manufacturer’s protocol. In short, input RNA was at first polyadenylated to provide a priming sequence for an oligo-(dT) primer. cDNA synthesis was primed by the 3′ smRNA dT Primer, which incorporates an adapter sequence at the 5′ end of each first-strand cDNA molecule. In the template-switching step, PrimeScript RT uses the SMART smRNA Oligo as a template to add a second adapter sequence to the 3′ end of each first-strand cDNA molecule. Full-length Illumina adapters were added during PCR amplification. The forward PCR primer binds to the sequence added by the SMART smRNA Oligo, while the reverse PCR primer binds to the sequence added by the 3′ smRNA dT primer. The resulting library cDNA molecules had included sequences required for clustering on an Illumina flow cell. The libraries were validated by assessing their size, purity, and concentration using an Agilent Bioanalyzer. The libraries were pooled in equimolar amounts and sequenced on an Illumina HiSeq 2500 instrument (Illumina, San Diego, CA, USA) to generate 51 base reads. Image decomposition and quality value calculations were performed using the modules of the Illumina pipeline.
We uploaded the miRNAs that changed significantly after the 20 m SRT into miRDB (Liu and Wang 2019; Wong and Wang 2015), and chose genes scored above 80. To integrate functional genomic annotations, the Kyoto Encyclopedia of Genes and Genomes (KEGG) pathway in the Database for Annotation, Visualization, and Integrated Discovery (DAVID) was used (Dennis et al. 2003; Huang et al. 2009).
Statistical analysis
Statistical analysis was performed using GraphPad Prism ver. 7 software (Graph Pad Software Inc., La Jolla, CA, USA), and all diagrams and data are presented as mean ± standard deviation. Statistical significance was set at p < 0.05. Since one participant could not collect urine sample, the Kruskal–Wallis test was used to compare the data within the group. Spearman’s rank coefficient of correlation and simple linear regression were used to evaluate the relationship between the data.
Results
Effect of the 20 m shuttle run test (SRT) on exercise-related fatigue markers
To verify whether the 20 m SRT could evoke fatigue to the participants, BLa, HR, RPE, and BP were measured before (Pre), immediately after (Post) the 20 m SRT, and after an hour of rest (Post 1 h) (Fig. 2). We observed significant changes in all fatigue markers after the 20 m SRT (Table 3, BLa, RPE, HR, and systolic blood pressure [SBP], p < 0.0001 and diastolic blood pressure [DBP], p < 0.01). All markers except for DBP (Table 3, p = 0.6435) were significantly elevated immediately after the 20 m SRT (Table 3, p < 0.0001). After 1 h of rest, BLa and RPE returned to normal state, HR was 7 ± 2 beat/min higher (p < 0.01), and SBP and DBP were 10 ± 3 mmHg (p < 0.05) and 6 ± 2 mmHg (p < 0.01) lower than the pre-exercise values, respectively (Table 3).
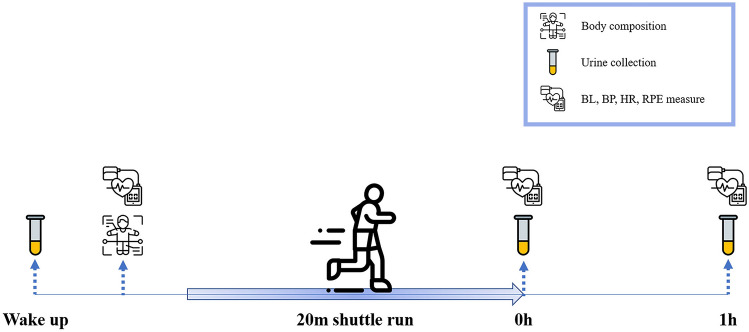
Fig. 2.
Workflow of the test day. Thirteen men participated in the 20 m SRT with their maximal efforts. Urine samples were collected and blood lactate (BLa), blood pressure (BP), heart rate (HR), and rating of perceived exertion (RPE) were measured before (Pre), after 0 h (Post), and after 1 h (Post 1 h) of the 20 m SRT
Table 3.
Characteristics of commonly used fatigue markers after 20 m SRT
| Pre | Post | Post 1 h | p | |
|---|---|---|---|---|
| BLa (mmol/L) | 1.4 ± 0.3 | 11.4 ± 4 | 1.9 ± 0.6 | < 0.0001 |
| RPE | 7 ± 1.5 | 17.5 ± 0.9 | 7.5 ± 1.2 | < 0.0001 |
| HR (beats/min) | 65 ± 12 | 143 ± 22 | 72 ± 13 | < 0.0001 |
| SBP (mmHg) | 120 ± 9 | 168 ± 25 | 110 ± 7 | < 0.0001 |
| DBP (mmHg) | 74 ± 8 | 76 ± 11 | 68 ± 6 | 0.1199 |
Characteristics of urinary extracellular vesicles (EVs) after the 20 m SRT
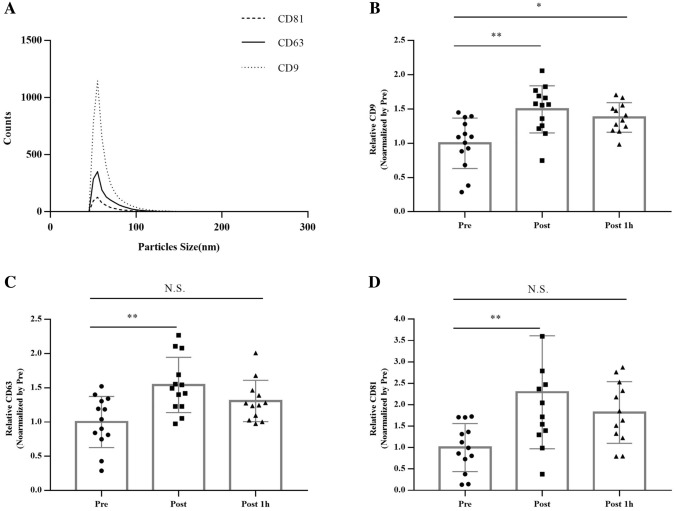
To validate the characteristics of urinary EVs, particle size analysis was performed on the ExoView R-100 instrument (Figs. 3a and 4). Particles detected on the instrument were mostly 50 nm in size. The 20 m SRT significantly altered urinary EV marker proteins (Fig. 3b–d, p < 0.01). In particular, when the value was normalized by Pre, CD9, CD63, and CD81 in urine showed 1.5 ± 0.3, 1.5 ± 0.4, and 2.3 ± 1.3 higher concentrations in post-exercise samples than in the first morning urine (Pre), respectively (Fig. 3b–d, p < 0.01). After an hour of rest (Post 1 h), only CD9 levels were significantly higher than those in pre-exercise samples (Fig. 3b, p < 0.05).
Fig. 3.
Characteristics of urinary EV marker proteins after the 20 m SRT. a The size distribution of all samples. (b–d) Relative concentrations of exosome marker proteins. Statistical analysis was performed with the aid of the ordinary one-way ANOVA with Tukey’s multiple comparisons test due to a missing sample (one participant could not collect the urine sample). The data represent means ± SEM. *p < 0.05, **p < 0.01, N.S. not significant
Fig. 4.

Representative images of captured urinary EV marker proteins (a–d). Captured EV marker proteins with anti-CD9 (a), anti-CD63 (b), anti-CD81 (c), and mouse IgG (d) on ExoView. Secondary labelling of exosomes captured on antibody capture spot: anti-CD 81/AF 555 (green), anti-CD 63/AF 647 (red) and anti-CD 9/AF 488 (blue)
Correlation between urinary EVs and exercise-related fatigue markers
After identifying the characteristics of urinary EVs after the 20 m SRT, we analyzed the relationship between EV marker proteins in urine and exercise-related fatigue markers. When the data were treated as delta values (Post–Pre, Post 1 h–Post), significant correlations were found between urinary EVs and fatigue markers (Except for DBP, only CD81 showed a significant relationship Table 4).
Table 4.
Correlation between urinary exosomal protein markers and BLa, RPE, HR, and BP
| Spearman r | |||||
|---|---|---|---|---|---|
| Δ RPE | Δ HR | Δ SBP | Δ DBP | Δ BLa | |
| Δ CD81 | 0.5342** | 0.3128 | 0.4548* | 0.2081 | 0.5061** |
| Δ CD63 | 0.6288*** | 0.5342** | 0.5442** | 0.3498 | 0.6854*** |
| Δ CD9 | 0.5213** | 0.4097* | 0.5175** | 0.1937 | 0.5308** |
*, **, *** indicates significant differences between urinary EV marker proteins and fatigue markers at p < 0.05, 0.01, 0.001, respectively
Urinary EV miRNA profiles after the 20 m SRT
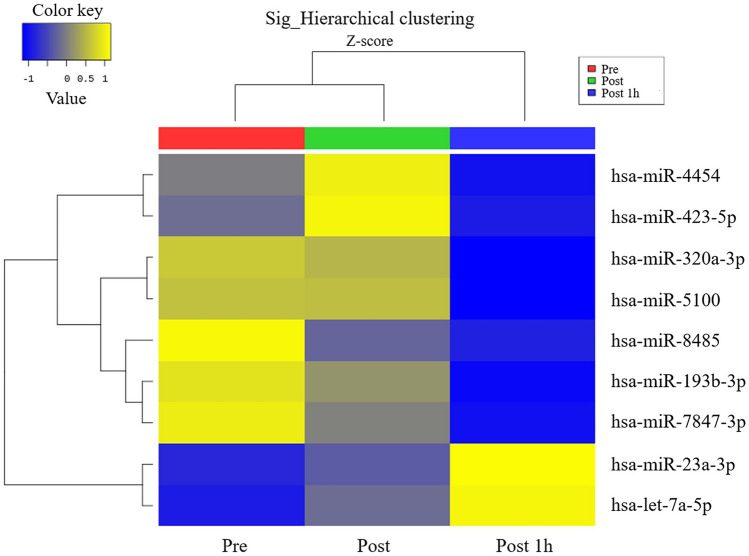
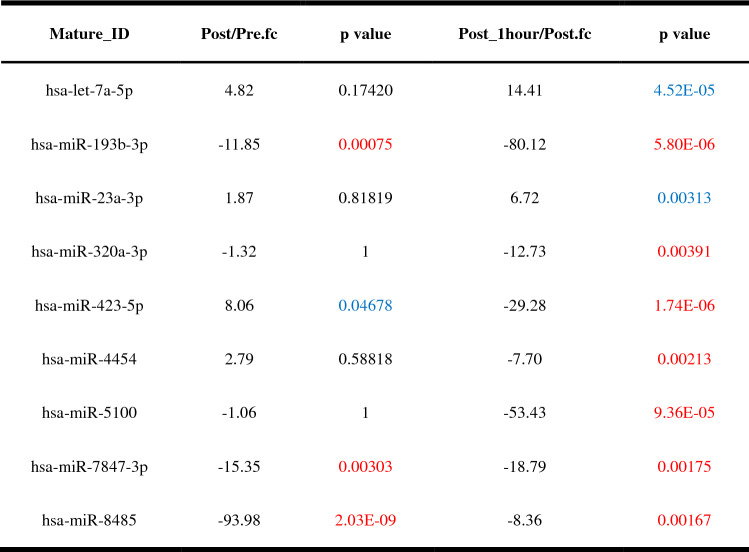
Next, we performed miRNA sequencing of urinary EVs to identify the effects of exercise on EV components. Sequencing results showed a total of 9 upregulated and downregulated miRNAs after 20 m SRT (Fig. 5, Table 5).
Fig. 5.
Effect of the 20 m SRT on urinary EV miRNA profiles. Microarray hierarchical clustering heat map image
Table 5.
The list of changed miRNAs in urinary exosomes after 20 m SRT
Significantly upregulated and downregulated miRNAs at p < 0.05 are shown in blue and red, respectively
Since we found a significant increase in urinary EVs immediately after the 20 m SRT, we analyzed genes and possible pathways related to four miRNAs (hsa-miR-193b-3p, hsa-miR- 423-5p, hsa-miR-7847-3p, and hsa-miR-8485) showing significant alterations. The target genes of each miRNA were predicted by miRDB (http://mirdb.org/) and were functionally classified using the web-based gene functional annotation tool DAVID (https://david.ncifcrf.gov/home.jsp). Target genes of hsa-miR-193b-3p are presented in Table 6 (the number of target genes of hsa-miR-8485 was 1055, which is above the score of 80 on miRDB, data not shown). We could not find exercise-related genes in hsa-miR-423-5p and hsa-miR-7847-3p (data not shown). Interestingly, target genes of both hsa-miR-193b-3p and hsa-miR-8485 showed insulin-related pathways and terms (Tables 7 and 8).
Table 6.
The list of target genes that regulated by has-miR-193b-3p (data from miRDB)
| Target rank | Target score | Gene symbol | Target rank | Target score | Gene symbol | Target rank | Target score | Gene symbol | Target rank | Target score | Gene symbol |
|---|---|---|---|---|---|---|---|---|---|---|---|
| 1 | 99 | MAPK10 | 23 | 89 | TAPT1 | 45 | 86 | ATF7IP2 | 67 | 83 | LRP4 |
| 2 | 98 | PIGA | 24 | 89 | NT5E | 46 | 86 | ST6GALNAC5 | 68 | 83 | ZNF618 |
| 3 | 97 | DCAF7 | 25 | 89 | PLAU | 47 | 85 | LAMC2 | 69 | 83 | NAV3 |
| 4 | 97 | RAPGEF6 | 26 | 89 | TAOK1 | 48 | 85 | KIT | 70 | 83 | ADGRE5 |
| 5 | 95 | ERBB4 | 27 | 88 | IGFBP5 | 49 | 85 | KRAS | 71 | 83 | RBMXL1 |
| 6 | 95 | ABI2 | 28 | 88 | CYREN | 50 | 85 | RGMA | 72 | 83 | OSMR |
| 7 | 94 | DMXL2 | 29 | 88 | CLTC | 51 | 85 | RUNX1T1 | 73 | 83 | ADARB1 |
| 8 | 94 | FLI1 | 30 | 88 | HNRNPUL2 | 52 | 85 | INO80D | 74 | 82 | CBX7 |
| 9 | 93 | PLAG1 | 31 | 88 | FAT4 | 53 | 85 | AREL1 | 75 | 82 | AAK1 |
| 10 | 93 | VN1R1 | 32 | 88 | CADM1 | 54 | 85 | LAMC1 | 76 | 82 | DNAAF4 |
| 11 | 93 | SOS2 | 33 | 88 | CNOT6 | 55 | 84 | HOXD13 | 77 | 81 | MSANTD2 |
| 12 | 92 | KCNJ2 | 34 | 87 | CCND1 | 56 | 84 | TGFBR3 | 78 | 81 | SRSF2 |
| 13 | 92 | IL17RD | 35 | 87 | EPHA5 | 57 | 84 | AIMP2 | 79 | 81 | ZBTB25 |
| 14 | 92 | FHDC1 | 36 | 87 | CLOCK | 58 | 84 | YWHAZ | 80 | 81 | AJUBA |
| 15 | 91 | LYRM2 | 37 | 87 | WDR82 | 59 | 84 | UBP1 | 81 | 81 | PLXNC1 |
| 16 | 91 | CARF | 38 | 87 | AP2M1 | 60 | 84 | P2RX5 | 82 | 80 | ABCB8 |
| 17 | 90 | ARHGEF12 | 39 | 87 | GPR20 | 61 | 83 | ZMAT3 | 83 | 80 | NF2 |
| 18 | 90 | MARF1 | 40 | 86 | TSC1 | 62 | 83 | CREBRF | 84 | 80 | USP53 |
| 19 | 90 | RRAS2 | 41 | 86 | CCDC28A | 63 | 83 | ETV1 | |||
| 20 | 90 | SLC10A6 | 42 | 86 | ETS1 | 64 | 83 | TIMM8B | |||
| 21 | 90 | SIAH1 | 43 | 86 | HEG1 | 65 | 83 | PLCH1 | |||
| 22 | 89 | SLC16A6 | 44 | 86 | UPRT | 66 | 83 | EARS2 |
Table 7.
Functional annotation results of target genes regulated by has-miR-193b-3p
| Category | Term | Genes | p value |
|---|---|---|---|
| KEGG_PATHWAY | hsa05221:Acute myeloid leukemia | CCND1, KIT, KRAS, SOS2, RUNX1T1 | 3.84E−04 |
| KEGG_PATHWAY | hsa05200:Pathways in cancer | MAPK10, ARHGEF12, CCND1, KIT, KRAS, LAMC2, LAMC1, ETS1, SOS2, RUNX1T1 | 5.87E−04 |
| KEGG_PATHWAY | hsa04151:PI3K-Akt signaling pathway | CCND1, KIT, TSC1, KRAS, LAMC2, LAMC1, OSMR, YWHAZ, SOS2 | 1.14E−03 |
| KEGG_PATHWAY | hsa04917:Prolactin signaling pathway | MAPK10, CCND1, KRAS, SOS2 | 9.68E−03 |
| KEGG_PATHWAY | hsa04014:Ras signaling pathway | MAPK10, KIT, RRAS2, KRAS, ETS1, SOS2 | 1.28E−02 |
| KEGG_PATHWAY | hsa04012:ErbB signaling pathway | MAPK10, ERBB4, KRAS, SOS2 | 1.68E−02 |
| KEGG_PATHWAY | hsa05231:Choline metabolism in cancer | MAPK10, TSC1, KRAS, SOS2 | 2.48E−02 |
| KEGG_PATHWAY | hsa04510:Focal adhesion | MAPK10, CCND1, LAMC2, LAMC1, SOS2 | 3.88E−02 |
| KEGG_PATHWAY | hsa04068:FoxO signaling pathway | MAPK10, CCND1, KRAS, SOS2 | 5.07E−02 |
| KEGG_PATHWAY | hsa04910:Insulin signaling pathway | MAPK10, TSC1, KRAS, SOS2 | 5.45E−02 |
| KEGG_PATHWAY | hsa04664:Fc epsilon RI signaling pathway | MAPK10, KRAS, SOS2 | 6.71E−02 |
| KEGG_PATHWAY | hsa04010:MAPK signaling pathway | MAPK10, TAOK1, RRAS2, KRAS, SOS2 | 7.20E-02 |
Table 8.
Functional annotation results of target genes regulated by has-miR-8485
| Category | Term | Genes | p value |
|---|---|---|---|
| KEGG_PATHWAY | hsa04151:PI3K-Akt signaling pathway | ATF2, GSK3B, FLT1, IRS1, YWHAB, ITGB3, PTEN, FASLG, PIK3R1, EFNA5, IGF1R, BCL2L11, FGF9, YWHAQ, PPP2R1A, CD19, ITGB6, JAK2, PCK1, PDGFRA, CHUK, INSR, F2R, PRKCA, PPP2R5C, VEGFA, FGF14, CDK6, PPP2R2C, GNB4, COL6A3, GRB2, SGK1, EIF4E2, MET, FGFR3, CREB5 | 5.33E−05 |
| KEGG_PATHWAY | hsa04010:MAPK signaling pathway | ATF2, SRF, CACNA1B, FASLG, NLK, STK3, RASGRP3, CRKL, ELK4, CACNG8, DUSP10, FGF9, GNA12, MAP3K2, PDGFRA, TGFB2, JUND, CHUK, PLA2G4A, PRKCA, MAPK14, DUSP6, CACNB2, PPM1B, FGF14, MAPKAPK2, GRB2, MAPT, LAMTOR3, FGFR3 | 5.49E−05 |
| KEGG_PATHWAY | hsa05200:Pathways in cancer | GSK3B, CTBP2, HHIP, PTEN, FASLG, PIK3R1, CBL, ETS1, FOXO1, GNAI1, IGF1R, RASGRP3, CRKL, EDNRA, FGF9, GNA12, E2F1, EP300, E2F3, SMAD2, STAT5A, PDGFRA, STAT5B, TGFB2, TCF7L1, CHUK, PTCH1, F2R, ARNT, PRKCA, RUNX1, VEGFA, BMP2, FGF14, CDK6, GNB4, CCDC6, GRB2, MET, FGFR3 | 7.90E−05 |
| KEGG_PATHWAY | hsa05161:Hepatitis B | STAT5A, ATF2, STAT5B, TGFB2, EGR3, CHUK, YWHAB, SRC, PTEN, FASLG, PRKCA, PIK3R1, DDB1, CDK6, YWHAQ, E2F1, EP300, GRB2, E2F3, CREB5 | 1.95E−04 |
| KEGG_PATHWAY | hsa04068:FoxO signaling pathway | SMAD2, TGFB2, USP7, CHUK, IRS1, INSR, PTEN, FASLG, CSNK1E, PIK3R1, NLK, MAPK14, FOXO1, IGF1R, BCL2L11, EP300, GRB2, SGK1, PCK1 | 2.09E−04 |
| KEGG_PATHWAY | hsa04360:Axon guidance | EPHA5, GSK3B, EPHA4, UNC5A, UNC5B, SEMA6D, SEMA4C, UNC5C, EFNA5, GNAI1, ABLIM1, DPYSL2, PAK6, PLXNC1, SRGAP3, PAK3, MET, EPHB3 | 3.26E−04 |
| KEGG_PATHWAY | hsa05220:Chronic myeloid leukemia | STAT5A, STAT5B, TGFB2, CTBP2, CHUK, PIK3R1, CBL, CRKL, RUNX1, CDK6, E2F1, GRB2, E2F3 | 3.29E−04 |
| KEGG_PATHWAY | hsa04810:Regulation of actin cytoskeleton | CYFIP2, PDGFRA, CYFIP1, ITGAM, GSN, SRC, ITGB3, F2R, RDX, PIK3R1, SSH2, FGD1, SSH1, CRKL, MYLK, FGF14, FGF9, GNA12, PIP4K2A, ARHGEF4, PAK6, ITGB6, PAK3, FGFR3 | 6.27E−04 |
Discussion
In the present study, urinary EVs showed higher levels after the 20 m SRT. Moreover, the change in urinary EVs was related to the commonly used fatigue markers. Another key finding of this study was that acute exercise altered urinary EV miRNAs including has-let-7a-5p, hsa-miR-193b-3p, hsa-miR-23a-3p, hsa-miR-320a-3p, hsa-miR-423-5p, hsa-miR-4454, hsa-miR-5100, hsa-miR-7847-3p, and hsa-miR-8485. It has been well documented that exercise induces the secretion of EVs in circulation (Safdar et al. 2016; Siqueira et al. 2021); however, alterations in urinary EVs after physical exercise have not been explored. To the best of our knowledge, the present study is the first to demonstrate that exercise induces changes the number of urinary EVs.
Although numerous studies support the beneficial effect of exercise in preventing chronic diseases and early death (Penedo and Dahn 2005; Viña et al. 2016), excessive exercise could result in fatigue (Cordeiro et al. 2017) that reduces exercise capacity (De Becker et al. 2000). The commonly used markers of exercise-induced fatigue or internal intensity are BLa (Finsterer 2012), RPE (Falk Neto et al. 2020), HR (Berglund et al. 2019), and BP (Sharman and Lagerche 2015). In this study, we first examined whether the 20 m SRT could evoke fatigue in study participants. A previous study suggested that 20 m SRT predicts VO2max and induces peak BLa and HR as high as the incremental exercise test on a treadmill (Aandstad et al. 2011). Herein, significantly elevated levels of BLa, HR, RPE, and SBP in each individual after the 20 m SRT support the evidence that the 20 m SRT could elicit exhaustion (Ahmaidi et al. 1992). In accordance with a previous study, DBP did not show significant changes after the 20 m SRT (Miyai et al. 2002). However, variables that were measured at the post time point may not represent the peak values, since the participants took at least 30 s to reach to the measurement spot (Pescatello et al. 1991). Nevertheless, overall, we conclude that 20 m SRT could evoke exercise-induced fatigue to the participants.
EVs are naturally secreted from cells and are bound to lipid bilayers containing proteins, lipids, mRNA, non-coding RNA, and miRNA (Linxweiler and Junker 2020). After release, exosomes transfer bioactive molecules to recipient cells and are highlighted as key regulators in cell-to-cell communication (Wortzel et al. 2019). In recent studies, EVs have been presented as potentially novel biomarkers of diverse diseases and disorders (Mussack et al. 2019; Pitt et al. 2016; Shibata 2021; Yang et al. 2020).
Previous studies have suggested that the number of EV particles in circulation is elevated immediately after exercise bout both in humans (Frühbeis et al. 2015b; Whitham et al. 2018) and animals (Oliveira et al. 2018). One study demonstrated that the concentrations of CD9, CD63, and CD81 in plasma EVS were increased after exercise with exhaustion, but the particle counts did not change (Brahmer et al. 2019). The present data describing the characteristics of urinary EVs after a single bout of exercise until voluntary exhaustion suggests that EV particles were abundant in the urine after exercise. Here, we used the ExoView platform (Nanoview Bioscience, Boston, USA), which captures CD9, CD63, and CD81-positive EV particles on a single-vesicle basis, to assess the characteristics of urinary EVs (Bachurski et al. 2019). Since the platform identifies each particle that is colored with respective antibodies (CD9, CD63, and CD81), it could detect EVs in urine precisely (Bachurski et al. 2019). We had found all EV marker protein-positive particles were elevated in urine, which coincides with that of plasma EVs after cycling exercise until exhaustion (Brahmer et al. 2019; Whitham et al. 2018). However, since circulating EVs have been reported to be elevated, not only late but also early in high intensity exercise when participants may not feel fatigue, more time points of measuring urinary EVs is needed to elucidate the relationship between urinary EVs and fatigue.
Next, we determined if the quantitative changes in urinary EVs (CD9-, CD63-, and CD81-positive particles) correlated with RPE, HR, SBP, and BLa. Urinary EVs were found to be associated with commonly used exercise fatigue or intensity markers. In line with our findings, previous studies have reported that exosome secretion could be facilitated by various acute stressors (Beninson and Fleshner 2014) and that elevated plasma exosome concentration could be observed in patients with chronic fatigue syndrome (Castro-Marrero et al. 2018). CD81 can be upregulated by oxidative stress, enhancing monocyte adhesion to endothelium under flow (Rohlena et al. 2009). EVs could manage reciprocal communication between heart and other organs (Gabisonia et al. 2022) and take part in the development of various hypertensive disorder(Z. Z. Liu et al. 2021). Consequently, we suggest that urinary EVs might reflect the acute fatigue state.
Since exercise modifies the urinary proteome (Kohler et al. 2015) and EV miRNA profile in plasma (Yoon et al. 2021), and both urine and urinary EVs comprise the plasma proteome (Dear et al. 2013; Harpole et al. 2016), we hypothesized that exercise could change the urinary EV miRNA contents. After 20 m SRT and 1 h of rest, a total of 9 miRNAs in urinary EVs were found to be altered. Among them, five miRNAs (hsa-let-7a-5p. hsa-miR-193b-3p, hsa-miR-23a-3p, hsa-miR-320a-3p, and hsa-miR-423-5p) were highly abundant in human urinary EVs, supporting our data (Cheng et al. 2014). Another previous study showed that hsa-miR-5100 was found only in urine while hsa-let-7a-5p, hsa-miR-23a-3p, hsa-miR-320a-3p, hsa-miR-423-5p, hsa-miR-7847-3p, hsa-miR-4454, and hsa-miR-8485 were common in both urine and serum (Park et al. 2020). A pilot study conducted in 2021 revealed that hsa-miR-23a-3p was elevated in urinary as well as in plasma EVs (Kuji et al. 2021), which is similar to our finding that hsa-miR-23a-3p gradually increased with time. After intensive exercise, circulatory hsa-miR-23a-3p was upregulated in plasma EVs, but downregulated in the muscles (D’souza et al. 2018).
Next, using an online database for miRNA target prediction, we identified 84 genes and 1055 genes that were regulated by hsa-miR-193b-3p and hsa-miR-8485, respectively. Hsa-miR-423-3p targeted only 3 genes, while hsa-miR-7847-3p targeted 74 genes (data not shown). Then, using DAVID, which is a Web-based functional genomic annotation tool, we verified whether these genes could mediate exercise response. Interestingly, hsa-miR-193b-3p target genes might functionally modulate the PI3K-Akt, insulin, and mitogen-activated protein kinase (MAPK) pathways. A similar result was obtained for the hsa-miR-8485 target genes. Given the beneficial effects of exercise (Prior et al. 2004) and the possible function of EVs in cell-to-cell communication (Mathieu et al. 2019), it is possible that altered urinary EV miRNAs are involved in insulin sensitivity by direct interaction with organs responding to insulin (Kumar et al. 2021). Normally, exercise until exhaustion can induce a small hyperglycemic response and persists for up to 1 h, and this is accompanied by hyperinsulinemia in recovery (Marliss and Vranic 2002). In addition, a single bout of exercise is reported to increase insulin sensitivity for at least 16 h (Borghouts and Keizer 2000). In this study, hsa-miR-193b-3p and hsa-miR-8485 were downregulated 1 h after 20 m SRT.
Exercise stimulation increases the activity of MAPK, which is involved in the phosphorylation of substrates related to carbohydrate, fat metabolism, and hypertrophy (Kramer and Goodyear 2007). In accordance with this, secreted extracellular vesicles after exercise showed upregulated glycolytic proteins, suggesting that circulating EVs modulate crosstalk between tissues 60. In addition, a previous study has reported that exercise affects growth hormone and insulin-like growth factor-1 (IGF-1) and PI3K/AKT pathways (Moon et al. 2020), resulting in cardiac hypertrophy and protection (Weeks et al. 2017). Thus, EVs may be involved in the regulation of energy metabolism (de Mendonça et al. 2020; Göran Ronquist 2019).
There are some limitations to the present study. First, the small sample size and characteristics of the participants, as the effects of sex differences and aging on circulating EVs have been reported (Bertoldi et al. 2018; Jayachandran et al. 2015). Second, the measurement of HR, BP, and BLa was delayed by approximately 30 s since the participants had to come to the measuring spot. Third, we did not control the water consumption among the participants. Fourth, the present study proceeded without a control group. Further study is needed, including appropriate control (rest only or mild intensity exercise) with more measurement time points. Finally, for miRNA analysis, we pooled EV samples into one sample due to the small volume of total yield.
Despite these limitations, this study is the first to demonstrate urinary EVs and their contents after exercise. We found a significant increase in urinary EVs after 20 m SRT, and this increase subsided after 1 h of rest. The variation in urinary EVs showed a significant correlation with common exercise fatigue or internal intensity markers. In addition, we identified changes in urinary EV miRNAs in urine, their target genes, as well as possible biological functions that could be mediated by altered genes. These results confirmed that urinary EVs could be potential biomarkers of exercise-induced fatigue.
Conclusion
In conclusion, acute exercise until voluntary exhaustion changed the concentration of urinary EVs and miRNA profiles. In addition, downregulated miRNAs, hsa-miR-193b-3p and hsa-miR-8485, in urinary EVs were identified to mediate energy metabolism-related pathways. Therefore, this study suggests that urinary EVs are potential biomarkers of fatigue after a single exercise session in young adult males.
Abbreviations
- EV
Extracellular vesicle
- miRNA
MicroRNA
- BLa
Blood lactate
- HR
Heart rate
- RPE
Rating of perceived exertion
- SBP
Systolic blood pressure
- DBP
Diastolic blood pressure
- MVB
Multivesicular body
- SRT
Shuttle run test
Author contributions
SP and HYM conceived and designed research; SP performed experiments; SP analyzed data; SP and HYM interpreted results of experiments; SP wrote the manuscript, SP prepared figures and tables. SP and HYM reviewed the manuscript.
Funding
This work was supported by the National Research Foundation (NRF, Grant Number: 2020R1C1C1006414, 2022R1I1A4053049).
Data availability
The datasets that support the findings of this study are openly available in online repositories. The names of the repository/repositories and accession number(s) can be found at: https://www.ncbi.nlm.nih.gov/geo/query/acc.cgi?acc=GSE193186.
Declarations
Conflict of interest
The authors declare that they have no conflicts of interest.
Footnotes
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
References
- Aandstad A, Holme I, Berntsen S, Anderssen SA. Validity and reliability of the 20 meter shuttle run test in military personnel. Mil Med. 2011;176(5):513–518. doi: 10.7205/MILMED-D-10-00373. [DOI] [PubMed] [Google Scholar]
- Ahmaidi S, Collomp K, Caillaud C, Prefaut C. Maximal and functional aerobic capacity as assessed by two graduated field methods in comparison to laboratory exercise testing in moderately trained subjects. Int J Sports Med. 1992;13(3):243–248. doi: 10.1055/s-2007-1021261. [DOI] [PubMed] [Google Scholar]
- Amar D, Lindholm ME, Norrbom J, Wheeler MT, Rivas MA, Ashley EA. Time trajectories in the transcriptomic response to exercise - a meta-analysis. Nat Commun. 2021;12(1):1–12. doi: 10.1038/s41467-021-23579-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bachurski D, Schuldner M, Nguyen P-H, Malz A, Reiners KS, Grenzi PC, Babatz F, Schauss AC, Hansen HP, Hallek M, Pogge von Strandmann E. Extracellular vesicle measurements with nanoparticle tracking analysis: an accuracy and repeatability comparison between NanoSight NS300 and ZetaView. J Extracell Vesicles. 2019;8(1):1596016. doi: 10.1080/20013078.2019.1596016. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Beninson LA, Fleshner M. Exosomes: An emerging factor in stress-induced immunomodulation. Semin Immunol. 2014;26(5):394–401. doi: 10.1016/j.smim.2013.12.001. [DOI] [PubMed] [Google Scholar]
- Berglund IJ, Sørås SE, Relling BE, Lundgren KM, Kiel IA, Moholdt T. The relationship between maximum heart rate in a cardiorespiratory fitness test and in a maximum heart rate test. J Sci Med Sport. 2019;22(5):607–610. doi: 10.1016/j.jsams.2018.11.018. [DOI] [PubMed] [Google Scholar]
- Bertoldi K, Cechinel LR, Schallenberger B, Corssac GB, Davies S, Guerreiro ICK, Belló-Klein A, Araujo ASR, Siqueira IR. Circulating extracellular vesicles in the aging process: impact of aerobic exercise. Mol Cell Biochem. 2018;440(1–2):115–125. doi: 10.1007/s11010-017-3160-4. [DOI] [PubMed] [Google Scholar]
- Borg G. Perceived exertion as an indicator of somatic stress. Scand J Rehabil Med. 1970;2(2):92–98. [PubMed] [Google Scholar]
- Borghouts LB, Keizer HA. Exercise and insulin sensitivity: a review. Int J Sports Med. 2000;21(1):1–12. doi: 10.1055/s-2000-8847. [DOI] [PubMed] [Google Scholar]
- Boutcher YN, Hopp JP, Boutcher SH. Acute effect of a single bout of aerobic exercise on vascular and baroreflex function of young males with a family history of hypertension. J Hum Hypertens. 2011;25(5):311–319. doi: 10.1038/jhh.2010.62. [DOI] [PubMed] [Google Scholar]
- Brahmer A, Neuberger E, Esch-Heisser L, Haller N, Jorgensen MM, Baek R, Möbius W, Simon P, Krämer-Albers EM. Platelets, endothelial cells and leukocytes contribute to the exercise-triggered release of extracellular vesicles into the circulation. J Extracell Vesicles. 2019 doi: 10.1080/20013078.2019.1615820. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Castro-Marrero J, Serrano-Pertierra E, Oliveira-Rodríguez M, Zaragozá MC, Martínez-Martínez A, del Blanco-López MC, Alegre J. Circulating extracellular vesicles as potential biomarkers in chronic fatigue syndrome/myalgic encephalomyelitis: an exploratory pilot study. J Extracell Vesicles. 2018;7(1):1453730. doi: 10.1080/20013078.2018.1453730. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cheng L, Sun X, Scicluna BJ, Coleman BM, Hill AF. Characterization and deep sequencing analysis of exosomal and non-exosomal miRNA in human urine. Kidney Int. 2014;86(2):433–444. doi: 10.1038/ki.2013.502. [DOI] [PubMed] [Google Scholar]
- Ciolac EG, Mantuani SS, Neiva CM, Verardi CEL, Pessôa-Filho DM, Pimenta L. Rating of perceived exertion as a tool for prescribing and self regulating interval training: a pilot study. Biol Sport. 2015;32(2):103–108. doi: 10.5604/20831862.1134312. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Colombo M, Raposo G, Théry C. Biogenesis, secretion, and intercellular interactions of exosomes and other extracellular vesicles. Annu Rev Cell Dev Biol. 2014;30(1):255–289. doi: 10.1146/annurev-cellbio-101512-122326. [DOI] [PubMed] [Google Scholar]
- Cordeiro LMS, Rabelo PCR, Moraes MM, Teixeira-Coelho F, Coimbra CC, Wanner SP, Soares DD. Physical exercise-induced fatigue: the role of serotonergic and dopaminergic systems. Braz J Med Biol Res. 2017;50(12):1–13. doi: 10.1590/1414-431X20176432. [DOI] [PMC free article] [PubMed] [Google Scholar]
- De Becker P, Roeykens J, Reynders M, McGregor N, De Meirleir K. Exercise capacity in chronic fatigue syndrome. Arch Intern Med. 2000;160(21):3270–3277. doi: 10.1001/archinte.160.21.3270. [DOI] [PubMed] [Google Scholar]
- de Mendonça M, Rocha KC, de Sousa É, Pereira BMV, Oyama LM, Rodrigues AC. Aerobic exercise training regulates serum extracellular vesicle miRNAs linked to obesity to promote their beneficial effects in mice. Am J Physiol. 2020;319(3):E579–E591. doi: 10.1152/AJPENDO.00172.2020. [DOI] [PubMed] [Google Scholar]
- Dear JW, Street JM, Bailey MA. Urinary exosomes: A reservoir for biomarker discovery and potential mediators of intrarenal signalling. Proteomics. 2013;13(10–11):1572–1580. doi: 10.1002/pmic.201200285. [DOI] [PubMed] [Google Scholar]
- Dennis G, Sherman BT, Hosack DA, Yang J, Gao W, Lane HC, Lempicki RA. DAVID: database for annotation, visualization, and integrated discovery. Genome Biol. 2003 doi: 10.1186/gb-2003-4-9-r60. [DOI] [PubMed] [Google Scholar]
- D’souza RF, Woodhead JST, Zeng N, Blenkiron C, Merry TL, Cameron-Smith D, Mitchell CJ. Circulatory exosomal miRNA following intense exercise is unrelated to muscle and plasma miRNA abundances. Am J Physiol. 2018;315(4):E723–E733. doi: 10.1152/ajpendo.00138.2018. [DOI] [PubMed] [Google Scholar]
- Falk Neto JH, Tibana RA, de Sousa NMF, Prestes J, Voltarelli FA, Kennedy MD. Session rating of perceived exertion is a superior method to monitor internal training loads of functional fitness training sessions performed at different intensities when compared to training impulse. Front Physiol. 2020 doi: 10.3389/fphys.2020.00919. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Finsterer J. Biomarkers of peripheral muscle fatigue during exercise. BMC Musculoskelet Disord. 2012;13(1):1. doi: 10.1186/1471-2474-13-218. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Frampton J, Cobbold B, Nozdrin M, Oo HTH, Wilson H, Murphy KG, Frost G, Chambers ES. The effect of a single bout of continuous aerobic exercise on glucose, insulin and glucagon concentrations compared to resting conditions in healthy adults: a systematic review meta-analysis and meta-regression. Sports Med. 2021;51(9):1949–1966. doi: 10.1007/s40279-021-01473-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Frühbeis C, Helmig S, Tug S, Simon P, Krämer-Albers E-M. Physical exercise induces rapid release of small extracellular vesicles into the circulation. J Extracell Vesicles. 2015;4(1):28239. doi: 10.3402/jev.v4.28239. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gabisonia K, Khan M, Recchia FA. Extracellular vesicle-mediated bidirectional communication between heart and other organs. Am J Physiol Heart Circ Physiol. 2022;322(5):H769–H784. doi: 10.1152/ajpheart.00659.2021. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Garner RT, Solfest JS, Nie Y, Kuang S, Stout J, Gavin TP. Multivesicular body and exosome pathway responses to acute exercise. Exp Physiol. 2020;105(3):511–521. doi: 10.1113/EP088017. [DOI] [PubMed] [Google Scholar]
- Göran Ronquist K. Extracellular vesicles and energy metabolism. Clin Chim Acta. 2019;488(2018):116–121. doi: 10.1016/j.cca.2018.10.044. [DOI] [PubMed] [Google Scholar]
- Gori A, Romanato A, Greta B, Strada A, Gagni P, Frigerio R, Brambilla D, Vago R, Galbiati S, Picciolini S, Bedoni M, Daaboul GG, Chiari M, Cretich M. Membrane-binding peptides for extracellular vesicles on-chip analysis. J Extracell Vesicles. 2020 doi: 10.1080/20013078.2020.1751428. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Green JM, McLester JR, Crews TR, Wickwire PJ, Pritchett RC, Lomax RG. RPE association with lactate and heart rate during high-intensity interval cycling. Med Sci Sports Exerc. 2006;38(1):167–172. doi: 10.1249/01.mss.0000180359.98241.a2. [DOI] [PubMed] [Google Scholar]
- Halson SL, Jeukendrup AE. Does overtraining exist? An analysis of overreaching and overtraining research. Sports Med. 2004;34(14):967–981. doi: 10.2165/00007256-200434140-00003. [DOI] [PubMed] [Google Scholar]
- Harpole M, Davis J, Espina V. Current state of the art for enhancing urine biomarker discovery. Expert Rev Proteomics. 2016;13(6):609–626. doi: 10.1080/14789450.2016.1190651. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hayashi Y, Nagasaka S, Takahashi N, Kusaka I, Ishibashi S, Numao S, Dong JL, Taki Y, Ogata H, Tokuyama K, Tanaka K. A single bout of exercise at higher intensity enhances glucose effectiveness in sedentary men. J Clin Endocrinol Metab. 2005;90(7):4035–4040. doi: 10.1210/jc.2004-2092. [DOI] [PubMed] [Google Scholar]
- Huang DW, Sherman BT, Lempicki RA. Systematic and integrative analysis of large gene lists using DAVID bioinformatics resources. Nat Protoc. 2009;4(1):44–57. doi: 10.1038/nprot.2008.211. [DOI] [PubMed] [Google Scholar]
- Jayachandran M, Lugo G, Heiling H, Miller VM, Rule AD, Lieske JC. Extracellular vesicles in urine of women with but not without kidney stones manifest patterns similar to men: A case control study. Biol Sex Differ. 2015 doi: 10.1186/s13293-015-0021-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jeppesen DK, Fenix AM, Franklin JL, Higginbotham JN, Zhang Q, Zimmerman LJ, Liebler DC, Ping J, Liu Q, Evans R, Fissell WH, Patton JG, Rome LH, Burnette DT, Coffey RJ. Reassessment of exosome composition. Cell. 2019;177(2):428–445.e18. doi: 10.1016/j.cell.2019.02.029. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kim J-S, Jeon T-B, Baek K-W, Yoo J-I, An S-H, Moon HY. Effects of 10-week treadmill exercise on the improvement of obesity-induced autophagy dysregulation in arterial endothelial cells of mice. Exercise Sci. 2019;28(3):263–269. doi: 10.15857/ksep.2019.28.3.263. [DOI] [Google Scholar]
- Kohler M, Schänzer W, Thevis M. Effects of exercise on the urinary proteome. Adv Exp Med Biol. 2015;845:121–131. doi: 10.1007/978-94-017-9523-4_12. [DOI] [PubMed] [Google Scholar]
- Kramer HF, Goodyear LJ. Exercise, MAPK, and NF-κB signaling in skeletal muscle. J Appl Physiol. 2007;103(1):388–395. doi: 10.1152/japplphysiol.00085.2007. [DOI] [PubMed] [Google Scholar]
- Kuji T, Sugasawa T, Fujita S, Ono S, Kawakami Y, Takekoshi K. A pilot study of miRNA expression profile as a liquid biopsy for full-marathon participants. Sports. 2021;9(10):134. doi: 10.3390/sports9100134. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kumar A, Sundaram K, Mu J, Dryden GW, Sriwastva MK, Lei C, Zhang L, Qiu X, Xu F, Yan J, Zhang X, Park JW, Merchant ML, Bohler HCL, Wang B, Zhang S, Qin C, Xu Z, Han X, Zhang HG. High-fat diet-induced upregulation of exosomal phosphatidylcholine contributes to insulin resistance. Nat Commun. 2021;12(1):1–21. doi: 10.1038/s41467-020-20500-w. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Linxweiler J, Junker K. Extracellular vesicles in urological malignancies: an update. Nat Rev Urol. 2020;17(1):11–27. doi: 10.1038/s41585-019-0261-8. [DOI] [PubMed] [Google Scholar]
- Liu W, Wang X. Prediction of functional microRNA targets by integrative modeling of microRNA binding and target expression data. Genome Biol. 2019;20(1):1–10. doi: 10.1186/s13059-019-1629-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Liu ZZ, Jose PA, Yang J, Zeng C. Importance of extracellular vesicles in hypertension. Exp Biol Med. 2021;246(3):342–353. doi: 10.1177/1535370220974600. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Marliss EB, Vranic M. Intense exercise has unique effects on both insulin release and its roles in glucoregulation: Implications for diabetes. Diabetes. 2002;51:271–283. doi: 10.2337/diabetes.51.2007.s271. [DOI] [PubMed] [Google Scholar]
- Mathieu M, Martin-Jaular L, Lavieu G, Théry C. Specificities of secretion and uptake of exosomes and other extracellular vesicles for cell-to-cell communication. Nat Cell Biol. 2019;21(1):9–17. doi: 10.1038/s41556-018-0250-9. [DOI] [PubMed] [Google Scholar]
- McGee SL, Hargreaves M. Exercise adaptations: molecular mechanisms and potential targets for therapeutic benefit. Nat Rev Endocrinol. 2020;16(9):495–505. doi: 10.1038/s41574-020-0377-1. [DOI] [PubMed] [Google Scholar]
- Miyai N, Arita M, Miyashita K, Morioka I, Shiraishi T, Nishio I. Blood pressure response to heart rate during exercise test and risk of future hypertension. Hypertension. 2002;39(3):761–766. doi: 10.1161/hy0302.105777. [DOI] [PubMed] [Google Scholar]
- Moon HY, Yoon KJ, Lee WS, Cho HS, Kim DY, Kim JS. Neural maturation enhanced by exercise-induced extracellular derivatives. Sci Rep. 2020;10(1):1–11. doi: 10.1038/s41598-020-60930-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mussack V, Wittmann G, Pfaffl MW. Comparing small urinary extracellular vesicle purification methods with a view to RNA sequencing: enabling robust and non-invasive biomarker research. Biomol Detect Quantif. 2019 doi: 10.1016/j.bdq.2019.100089. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Oliveira GP, Porto WF, Palu CC, Pereira LM, Petriz B, Almeida JA, Viana J, Filho NNA, Franco OL, Pereira RW. Effects of acute aerobic exercise on rats serum extracellular vesicles diameter, concentration and small RNAs content. Front Physiol. 2018;9(5):1–11. doi: 10.3389/fphys.2018.00532. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Park S, Lee K, Park IB, Kim NH, Cho S, Rhee WJ, Oh Y, Choi J, Nam S, Lee DH. The profiles of microRNAs from urinary extracellular vesicles (EVs) prepared by various isolation methods and their correlation with serum EV microRNAs. Diabetes Res Clin Pract. 2020;160:108010. doi: 10.1016/j.diabres.2020.108010. [DOI] [PubMed] [Google Scholar]
- Penedo FJ, Dahn JR. Exercise and well-being: A review of mental and physical health benefits associated with physical activity. Curr Opin Psychiatry. 2005;18(2):189–193. doi: 10.1097/00001504-200503000-00013. [DOI] [PubMed] [Google Scholar]
- Pescatello LS, Fargo AE, Leach CN, Scherzer HH. Short-term effect of dynamic exercise on arterial blood pressure. Circulation. 1991;83(5):1557–1561. doi: 10.1161/01.CIR.83.5.1557. [DOI] [PubMed] [Google Scholar]
- Pitt JM, Kroemer G, Zitvogel L. Extracellular vesicles: Masters of intercellular communication and potential clinical interventions. J Clin Investig. 2016;126(4):1139–1143. doi: 10.1172/JCI87316. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pober DM, Braun B, Freedson PS. Effects of a single bout of exercise on resting heart rate variability. Med Sci Sports Exerc. 2004;36(7):1140–1148. doi: 10.1249/01.MSS.0000132273.30827.9A. [DOI] [PubMed] [Google Scholar]
- Prior BM, Yang HT, Terjung RL, Hopkins N, Cable NT, Green DJ, Tian J, Fratz S, Hou Y, Lu Q, Görlach A, Hess J, Schreiber C, Black SM. What makes vessels grow with exercise training ? Skeletal and Cardiac Muscle Blood Flow What makes vessels grow with exercise training ? J Appl Physiol. 2004;97:1119–1128. doi: 10.1152/japplphysiol.00035.2004. [DOI] [PubMed] [Google Scholar]
- Rohlena J, Volger OL, van Buul JD, Hekking LHP, van Gils JM, Bonta PI, Fontijn RD, Post JA, Hordijk PL, Horrevoets AJG. Endothelial CD81 is a marker of early human atherosclerotic plaques and facilitates monocyte adhesion. Cardiovasc Res. 2009;81(1):187–196. doi: 10.1093/cvr/cvn256. [DOI] [PubMed] [Google Scholar]
- Safdar A, Saleem A, Tarnopolsky MA. The potential of endurance exercise-derived exosomes to treat metabolic diseases. Nat Rev Endocrinol. 2016;12(9):504–517. doi: 10.1038/nrendo.2016.76. [DOI] [PubMed] [Google Scholar]
- Scherr J, Wolfarth B, Christle JW, Pressler A, Wagenpfeil S, Halle M. Associations between Borg’s rating of perceived exertion and physiological measures of exercise intensity. Eur J Appl Physiol. 2013;113(1):147–155. doi: 10.1007/s00421-012-2421-x. [DOI] [PubMed] [Google Scholar]
- Sharman JE, Lagerche A. Exercise blood pressure: clinical relevance and correct measurement. J Hum Hypertens. 2015;29(6):351–358. doi: 10.1038/jhh.2014.84. [DOI] [PubMed] [Google Scholar]
- Shibata H. Exosomes and exosomal cargo in urinary extracellular vesicles: novel potential biomarkers for mineralocorticoid-receptor-associated hypertension. Hypertens Res. 2021 doi: 10.1038/s41440-021-00759-2. [DOI] [PubMed] [Google Scholar]
- Siqueira IR, Palazzo RP, Cechinel LR. Circulating extracellular vesicles delivering beneficial cargo as key players in exercise effects. Free Radical Biol Med. 2021;172(June):273–285. doi: 10.1016/j.freeradbiomed.2021.06.007. [DOI] [PubMed] [Google Scholar]
- Urhausen A, Kindermann W. Diagnosis of overtraining. Sports Med. 2002;32(2):95–102. doi: 10.2165/00007256-200232020-00002. [DOI] [PubMed] [Google Scholar]
- Viña J, Rodriguez-Mañas L, Salvador-Pascual A, Tarazona-Santabalbina FJ, Gomez-Cabrera MC. Exercise: the lifelong supplement for healthy ageing and slowing down the onset of frailty. J Physiol. 2016;594(8):1989–1999. doi: 10.1113/JP270536. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wallace LK, Slattery KM, Impellizzeri FM, Coutts AJ. Establishing the criterion validity and reliability of common methods for quantifying training load. J Strength Cond Res. 2014;28(8):2330–2337. doi: 10.1519/JSC.0000000000000416. [DOI] [PubMed] [Google Scholar]
- Weeks KL, Bernardo BC, Ooi JYY, Patterson NL, McMullen JR. The IGF1-PI3K-Akt signaling pathway in mediating exercise-induced cardiac hypertrophy and protection. In: Xiao J, editor. Exercise for cardiovascular disease prevention and treatment: from molecular to clinical, part 2. Singapore: Springer; 2017. pp. 187–210. [DOI] [PubMed] [Google Scholar]
- Whitham M, Parker BL, Friedrichsen M, Hingst JR, Hjorth M, Hughes WE, Egan CL, Cron L, Watt KI, Kuchel RP, Jayasooriah N, Estevez E, Petzold T, Suter CM, Gregorevic P, Kiens B, Richter EA, James DE, Wojtaszewski JFP, Febbraio MA. Extracellular vesicles provide a means for tissue crosstalk during exercise. Cell Metab. 2018;27(1):237–251.e4. doi: 10.1016/j.cmet.2017.12.001. [DOI] [PubMed] [Google Scholar]
- Wilhelm EN, González-Alonso J, Parris C, Rakobowchuk M. Exercise intensity modulates the appearance of circulating microvesicles with proangiogenic potential upon endothelial cells. Am J Physiol. 2016;311(5):H1297–H1310. doi: 10.1152/ajpheart.00516.2016. [DOI] [PubMed] [Google Scholar]
- Wilhelm EN, González-Alonso J, Chiesa ST, Trangmar SJ, Kalsi KK, Rakobowchuk M. Whole-body heat stress and exercise stimulate the appearance of platelet microvesicles in plasma with limited influence of vascular shear stress. Physiol Rep. 2017;5(21):e13496. doi: 10.14814/phy2.13496. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wong N, Wang X. miRDB: An online resource for microRNA target prediction and functional annotations. Nucleic Acids Res. 2015;43(D1):D146–D152. doi: 10.1093/nar/gku1104. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wortzel I, Dror S, Kenific CM, Lyden D. Exosome-mediated metastasis: communication from a distance. Dev Cell. 2019;49(3):347–360. doi: 10.1016/j.devcel.2019.04.011. [DOI] [PubMed] [Google Scholar]
- Yang E, Wang X, Gong Z, Yu M, Wu H, Zhang D. Exosome-mediated metabolic reprogramming: the emerging role in tumor microenvironment remodeling and its influence on cancer progression. Signal Transduct Target Ther. 2020;5(1):1–13. doi: 10.1038/s41392-020-00359-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yellon DM, Davidson SM. Exosomes nanoparticles involved in cardioprotection? Circ Res. 2014;114(2):325–332. doi: 10.1161/CIRCRESAHA.113.300636. [DOI] [PubMed] [Google Scholar]
- Yoon KJ, Park S, Kwak SH, Moon HY. Effects of voluntary running wheel exercise-induced extracellular vesicles on anxiety. Front Mol Neurosci. 2021 doi: 10.3389/fnmol.2021.665800. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhang Y, Liu Y, Liu H, Tang WH. Exosomes: biogenesis, biologic function and clinical potential. Cell Biosci. 2019;9(1):1–18. doi: 10.1186/s13578-019-0282-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
The datasets that support the findings of this study are openly available in online repositories. The names of the repository/repositories and accession number(s) can be found at: https://www.ncbi.nlm.nih.gov/geo/query/acc.cgi?acc=GSE193186.