Abstract
Purpose
Trials aiming to lower homocysteine by B-vitamin supplementation have reported mixed results on slowing cognitive decline. We investigated if efficacy of B-vitamin supplementation is affected by baseline plasma omega-3 fatty acid levels.
Methods
This post-hoc analysis of the B-proof trial included 191 adults aged 65 years or older with baseline plasma total homocysteine ≥ 12 μmol/L, randomly assigned to 400 µg folic acid and 500 µg vitamin B12 or placebo daily for 2 years. Global and domain-specific cognitive functioning were assessed at baseline and after 2 years. The effect of B-vitamin supplementation was analyzed according to tertiles of baseline plasma omega-3 fatty acids concentrations combined, and eicosapentaenoic acid (EPA) and docosahexaenoic acid (DHA) individually using multiple linear regression analyses.
Results
The mean ± SD age of the participants was 71.6 ± 5.9 years and median [IQR] Mini-Mental State Examination was 29 [28–30]. The treatment effect of B-vitamins on global cognition was larger in participants in the high compared to the middle DHA tertile (difference in z-score, mean ± SE 0.22 ± 0.10, p = 0.03). There was no significant interaction between B-vitamin supplementation and combined omega-3 fatty acid (p = 0.49) and EPA (p = 0.99) tertiles. Similarly, the efficacy of B-vitamin treatment on domain-specific cognitive functioning did not link to omega-3 fatty acid, DHA, or EPA plasma levels.
Conclusion
This post-hoc analysis indicated that efficacy of B-vitamin supplementation in slowing cognitive decline relates to DHA status, with individuals with higher plasma DHA levels benefitting more from vitamin B12 and folic acid use. The results support earlier observations that positive effects of B-vitamins in cognitive ageing may be subgroup-specific.
Trial registration: Registered at clinicaltrials.gov (NCT00696514) on June 12, 2008.
Supplementary Information
The online version contains supplementary material available at 10.1007/s00394-022-02924-w.
Keywords: B-vitamins, Omega-3 fatty acids, Cognition, Older adults, Elderly, Healthy ageing
Introduction
Age-related cognitive decline leading to dementia poses a societal challenge with major medical, social and economic impact. In the absence of curative treatment for dementia, the focus is on prevention by management of risk factors [1]. Epidemiological studies show that individuals with elevated homocysteine levels are at greater risk of cognitive decline and dementia, identifying homocysteine as a possible modifiable risk factor [2].
Elevated homocysteine levels may reflect impaired B-vitamin status [3]. B-vitamin supplementation to lower homocysteine levels and thereby slowing down cognitive decline would seem a straightforward solution, yet proof of clinical benefits is lacking. While clinical trials show that B-vitamin treatment, usually existing of vitamin B12, B6 and/or folic acid, is effective in lowering homocysteine levels, its effect on slowing down cognitive decline remains inconclusive [4].
It has been hypothesized that the efficacy of B-vitamin supplementation in slowing cognitive decline is dependent on omega-3 fatty acid status, with B-vitamin supplementation being only effective in individuals with higher omega-3 fatty acid plasma levels. Indeed, results from several post-hoc analyses of B-vitamin trials underline this hypothesis [5, 6]. Surprisingly, opposite results have been demonstrated as well, with only individuals with lower omega-3 fatty acid status benefitting from B-vitamin supplementation [7]. This merits further research to disentangle the complex interaction between B-vitamins and omega-3 fatty acids in cognitive ageing.
Thus, the current study further investigates the interaction between B-vitamin supplementation and omega-3 fatty acids with respect to cognitive outcomes in healthy older adults without cognitive complaints. To this end, we investigated if the efficacy of B-vitamin supplementation was dependent on baseline omega-3 fatty acid plasma levels in cognitively healthy older adults in the B-proof trial (B-Vitamins for the Prevention of Osteoporotic Fractures). In the main study of the B-proof trial, no effects of B-vitamins on slowing cognitive decline were observed [8].
Methods
Study design and participants
The present study was conducted as a post-hoc analysis within the B-proof trial, a randomized, double-blind placebo-controlled trial investigating the effect of folic acid and vitamin B12 supplementation on fracture incidence. Cognitive functioning was measured as secondary outcome. Data was collected between October 2008 and March 2013 in three research centers in the Netherlands: Erasmus Medical Center (Rotterdam), VU University Medical Center (Amsterdam) and Wageningen University (Wageningen). This analysis is based on a subsample of the Wageningen participants for whom fatty acid data were available. The trial has been approved by the Medical Ethics committee from Wageningen University & Research and has been registered at clinicaltrials.gov (NCT00696514). All participants provided written informed consent.
Information on study design and participants has been described in detail previously [9]. In short, the intervention existed of daily administration of 400 µg folic acid and 500 µg vitamin B12 tablets versus placebo tablets for a period of 2 years. Both intervention and placebo tablets contained 15 µg vitamin D3. Participants received tablets every 6 months and they were requested to return any remaining tablets, as a measure of compliance. Participants were men and women aged 65 years and older, with elevated plasma homocysteine levels (12–50 µmol/L). Exclusion criteria were renal insufficiency (creatinine > 150 µmol/L), diagnosis of a malignancy in the past 5 years and current or recent (< 4 months) use of supplements with very high dose of folic acid (> 300 µg) or intramuscular injections with vitamin B12. Fatty acid data was available for 205 participants. Our analysis included data of 191 participants. Data from 13 participants were excluded due to missing follow-up (n = 3) or ApoE4 (n = 10) data, and data from 1 participant was excluded due to a follow-up MMSE score of 19, indicating possible dementia.
Cognitive testing
Cognitive functioning was assessed at baseline and after 2 years of intervention with an extensive battery of cognitive tests administered by trained research assistants.
In the Rey Auditory Verbal Learning Test (RAVLT) [10], a list of 15 words was verbally presented to the participant at a rate of one word per two seconds. The participant was asked to recall the words in five trials immediately after presentation (subtest immediate), and after a 20-min delay (subtest delayed). Subsequently, the participant was asked to identify the 15 words in a list of 30 verbally presented words (subtest recognition). The number of correctly recalled words in each subtest was recorded.
In the Digit Span Task [11], the participant was verbally presented with digit sequences and asked to recall the sequence in either forward or backward order. Starting at a sequence length of three digits in the forward and two digits in the backward task, the length increased each two trials until an error was made or the maximum length of nine digits in the forward and eight in the backward task was reached. The maximum sequence length for the forward and backward version was recorded.
In the Trail Making Test (TMT) [12], participants were presented with a paper containing 25 circles. In two subtests, participants were asked to connect 25 circles containing numbers in chronological order (part A), and to alternate connecting circles containing numbers and letters in chronological and alphabetical order (part B). Time to complete each part was recorded.
The Stroop Colour-Word test [13] exists of three subtests, in which the participant was presented with colour words written in black ink (part I), coloured blocks (part II), or colour words written in an incongruent colour ink (part III). The participant is instructed to read aloud the words as fast as possible. The time needed to complete each part was documented.
In the Symbol Digit Modalities Test (SDMT) [14], symbols were paired with digits. The participant was presented with a sheet of symbols, and asked to match the symbols to the corresponding digit as fast as possible. The number of correctly matched pairs in 90 s was recorded.
In Letter Fluency [15], participants were given 60 s to name as many words as possible starting with the letter D, A and T (baseline) or K, O, and M (follow-up). The number of unique words was documented.
Parallel versions were used for RAVLT, TMT and Verbal Fluency to minimize learning effects. Individual cognitive test scores at baseline and follow-up were converted into Z-scores based on baseline mean and standard deviation, with higher scores indicating better cognitive functioning. The Z-scores for TMT and Stroop Colour-Word test were reversed as lower scores indicate better cognitive functioning. Individual Z-scores were clustered into composite scores for global and domain specific cognitive functioning:
Biochemical assays
Baseline omega-3 fatty acid concentrations were measured in the plasma phospholipid (PL) fractions from blood samples obtained after an overnight fast or a light breakfast. Samples had been collected by venipuncture using EDTA containing vacuum tubes. Plasma was obtained by centrifugation (10 min at 1200g) and stored at − 80 °C. Studies have shown that polyunsaturated fatty acids remain stable for up to 12 years under these conditions [16]. Total lipids were extracted from plasma with isopropanol/hexane (2:3, v/v) and separated into cholesteryl and PL fractions by solid phase extraction using silica columns. Subsequently, fatty acids in the PL fractions were transesterified using boron trifluoride in methanol yielding their methyl esters. Analysis was performed by gas chromatography with flame-ionization detection. Peaks were identified based on comparison of retention times to known standards. Fatty acid concentrations are presented in relative concentrations of total fatty acids. The relative concentration of plasma omega-3 fatty acids was derived by adding the proportions of eicosapentaenoic acid (EPA) and docosahexaenoic acid (DHA). A detailed description of the analytical procedure for fatty acids used in our lab has been published elsewhere [17].
Serum vitamin B12 and folate were determined using immune electrochemiluminescence assay (Elecsys, 2010, Roche). High-performance liquid chromatography was used to measure plasma total homocysteine [18]. DNA was isolated from buffy coats for genotyping. ApoE genotype was determined using TaqMan analysis.
Descriptive characteristics
Trained research assistants measured height with a stadiometer to the nearest 0.1 cm and weight with a calibrated scale to the nearest 0.5 kg. Body mass index (BMI) was calculated as weight (kg)/(height (m))2. Information on age, sex, education level (low, middle, high), smoking status (never, former, current) and physical activity [19] was obtained via questionnaires. MMSE (0–30 points) [20] was assessed by trained research assistants following a standardized protocol.
Statistical analysis
Data are expressed as n (%), mean (SD) or median (IQR) unless otherwise stated.
Baseline characteristics between intervention and omega-3 fatty acid groups were compared using independent sample t-test, ANOVA or Kruskal–Wallis test for continuous variables and chi-square for categorical variables. Multiple linear regression was performed to investigate if the efficacy of B-vitamin supplementation was dependent on baseline omega-3 fatty acid levels. We modelled the change in cognition Z-score between baseline and post-intervention for global cognition and domain-specific cognition as a function of intervention group (B-vitamins, placebo), baseline omega-3 fatty acid group (low, middle and high) and their interaction. To investigate if DHA and/or EPA status modified B-vitamin supplementation efficacy separately, additional models were run replacing baseline omega-3 fatty acid levels (by groups) by either DHA or EPA concentrations (by groups). To create the omega-3 fatty acid status groups, baseline omega-3 fatty acid, DHA and EPA concentrations were divided into tertiles. The analyses were adjusted for baseline cognitive Z-score, age, sex, education, ApoE4 status, baseline homocysteine level, physical activity and smoking status, all measured at baseline. Tukey correction for multiple comparisons was applied when examining treatment effects within the omega-3 fatty acid tertiles. p-values < 0.05 were considered statistically significant, for interaction terms the cut-off was set at p < 0.10. All analyses were performed using RStudio Version 1.1.463 [21].
Results
Participant characteristics
Table 1 presents baseline characteristics of the study population. The mean age of the participants was 71.5 ± 5.8 years and 56% was male. The average BMI was 27.5 ± 4.2 kg/m2, with 76% being overweight (i.e. BMI ≥ 25 kg/m2). Total homocysteine levels were elevated with a median of 13.7 [IQR 12.9–15.8] µmol/L. The study population was cognitively healthy, as indicated by a median MMSE score of 29 [IQR 28–30] at baseline. Five participants (2.6%) had MMSE scores equal to or lower than 24, indicating cognitive impairment. Participants in the B-vitamin group were younger (p = 0.01) compared to participants in the placebo group. Furthermore, a larger proportion of participants in the middle omega-3 fatty acid tertile had never smoked compared to participants in the high omega-3 fatty acid tertile (p = 0.02) (Supplementary Table 1). Mean baseline cognitive scores did not differ between either intervention or omega-3 fatty acid status groups. Compliance to treatment was high with an average of 97%. There was no difference in compliance between treatment and/or omega-3 fatty acid groups.
Table 1.
Baseline characteristics per treatment group in the B-proof study
| Characteristic | Overall (n = 191) | B vitamin (n = 94) | Placebo (n = 97) | p-value |
|---|---|---|---|---|
| Age (years) | 71.5 ± 5.8 | 70.3 ± 5.1 | 72.7 ± 6.3 | > 0.01 |
| Sex n (%) | ||||
| Male | 107 (56%) | 52 (55%) | 55 (57%) | 0.96 |
| Female | 84 (44%) | 42 (45%) | 42 (43%) | |
| Level of education n (%) | 0.27 | |||
| Low | 76 (40%) | 41 (43%) | 35 (36%) | |
| Middle | 46 (24%) | 18 (19%) | 28 (29%) | |
| High | 69 (36%) | 35 (37%) | 34 (35%) | |
| BMI (kg/m2) | 27.5 ± 4.2 | 27.5 ± 4.4 | 27.5 ± 4.0 | 0.95 |
| Physical activity (kcal/d) | 561 (358–863) | 596 (386–879) | 525 (326–810) | 0.09 |
| Smoking behaviour n (%) | 0.54 | |||
| Current smoker | 11 (6%) | 7 (7%) | 4 (4%) | |
| Former smoker | 123 (64%) | 61 (65%) | 62 (64%) | |
| Never smoker | 57 (30%) | 26 (28%) | 31 (32%) | |
| ApoE4 carriers n (%) | 55 (29%) | 28 (30%) | 27 (28%) | 0.89 |
| Biochemical measures | ||||
| Total homocysteine (µmol/L) | 13.7 (12.9–15.8) | 13.7 (13.0–15.3) | 13.7 (12.9–16.4) | 0.65 |
| Folate (nmol/L) | 17.4 (14.1–23.5) | 16.9 (13.9–22.4) | 17.7 (14.3–24.7) | 0.12 |
| Vitamin B12 (pmol/L) | 256 (201–334) | 253 (203–308) | 275 (196–366) | 0.10 |
| MMA (μmol/L) | 0.22 (0.19–0.29) | 0.22 (0.19–0.29) | 0.22 (0.19–0.31) | 0.80 |
| holoTC (pmol/L) | 62 (46–80) | 63 (47–76) | 62 (46–82) | 0.73 |
| 25(OH)D (nmol/L) | 60 ± 23 | 61 ± 24 | 60 ± 22 | 0.84 |
| Omega-3 status (sum DHA and EPA, %)a | 5.7 ± 1.9 | 5.5 ± 1.8 | 5.9 ± 2.1 | 0.20 |
| DHA (%)a | 4.3 ± 1.2 | 4.2 ± 1.2 | 4.5 ± 1.3 | 0.11 |
| EPA (%)a | 1.4 ± 0.9 | 1.3 ± 0.8 | 1.4 ± 1.0 | 0.54 |
| MMSE score | 29 (28–30) | 29 (27–30) | 29 (28–30) | 0.85 |
| Global cognition Z-score | 0.00 ± 0.52 | 0.01 ± 0.54 | − 0.02 ± 0.51 | 0.69 |
| Episodic memory Z-score | 0.00 ± 0.70 | 0.03 ± 0.70 | − 0.02 ± 0.70 | 0.59 |
| Attention and working memory Z-score | 0.00 ± 0.86 | 0.01 ± 0.89 | − 0.00 ± 0.85 | 0.94 |
| Information processing speed Z-score | 0.00 ± 0.77 | − 0.02 ± 0.79 | 0.02 ± 0.76 | 0.73 |
| Executive functioning Z-score | 0.00 ± 0.69 | 0.04 ± 0.69 | − 0.05 ± 0.69 | 0.37 |
B-proof subjects with available fatty acid and cognition data at both time points
BMI: body mass index, MMA: methylmalonic acid, holoTC: holotranscobalamin, DHA: docosahexaenoic acid, EPA: eicosapentaenoic acid, MMSE: Mini Mental State Examination. Data are mean ± SD, median (IQR) or number (%)
aMeasured in phospholipid fractions
Comparing our subsample with the total Wageningen and B-proof study populations, our subsample was younger than the Wageningen (72.9 ± 5.7, p < 0.01) and B-proof (74.3 ± 6.6y, p < 0.01) study populations. Median Mini-Mental State Examination (MMSE) score was similar in our subsample and the total Wageningen study population (29 [28–30] for both, p = 0.46). The total B-proof study population showed lower MMSE scores (28 [27–29], p < 0.01).
Cognitive performance
Global cognitive functioning
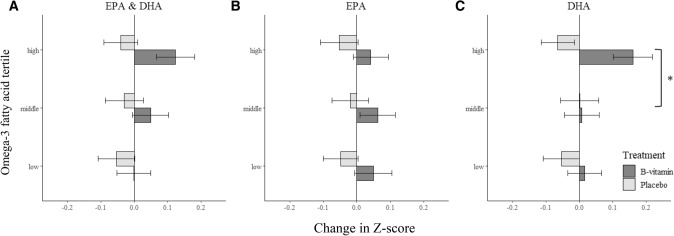
The treatment effects of B-vitamins versus placebo on global cognition were numerically positive (i.e. larger than 0) in all omega-3 fatty acid (EPA and DHA combined) tertiles, indicating that the group that received B-vitamins improved more over time compared to the placebo group, irrespective of the omega-3 fatty acid blood levels (Table 2; Fig. 1). Despite the larger treatment effect in participants in the high omega-3 fatty acid tertile (difference 0.16 ± 0.07, p = 0.25) compared to the middle and low tertiles (respectively, 0.08 ± 0.07, p = 0.75; 0.05 ± 0.07, p = 0.97), there was no significant overall interaction between B-vitamin supplementation and omega-3 fatty acid tertile (p = 0.60), meaning that there is no difference in treatment effect of B-vitamins between the low, middle and high omega-3 fatty acid tertiles.
Table 2.
Changes in global cognition Z-scores following B-vitamin versus placebo supplementation according to omega-3 fatty acid, EPA and DHA status tertile
| Treatment effecta | Overall interactionb | Tertiles pairwise comparisonc | |||||
|---|---|---|---|---|---|---|---|
| Crude | Adjusted | p-value | p-value | Low vs middle | Low vs high | Middle vs high | |
| Omega-3 fatty acid status | 0.60 | ||||||
| Low tertile | 0.10 ± 0.07 | 0.05 ± 0.07 | 0.97 |
diff = 0.04 ± 0.10 p = 0.69 |
diff = 0.10 ± 0.10 p = 0.32 |
diff = 0.06 ± 0.10 p = 0.54 |
|
| Middle tertile | 0.13 ± 0.07 | 0.08 ± 0.07 | 0.75 | ||||
| High tertile | 0.16 ± 0.07 | 0.16 ± 0.07 | 0.25 | ||||
| EPA status | 0.97 | ||||||
| Low tertile | 0.16 ± 0.07 | 0.10 ± 0.07 | 0.72 |
diff = 0.02 ± 0.10 p = 0.82 |
diff = 0.02 ± 0.10 p = 0.98 |
diff = 0.02 ± 0.10 p = 0.84 |
|
| Middle tertile | 0.09 ± 0.07 | 0.07 ± 0.07 | 0.88 | ||||
| High tertile | 0.12 ± 0.07 | 0.09 ± 0.07 | 0.75 | ||||
| DHA status | 0.06 | ||||||
| Low tertile | 0.12 ± 0.07 | 0.06 ± 0.07 | 0.95 |
diff = 0.05 ± 0.10 p = 0.59 |
diff = 0.18 ± 0.10 p = 0.07 |
diff = 0.23 ± 0.10 p = 0.02 |
|
| Middle tertile | 0.04 ± 0.07 | 0.01 ± 0.07 | 1.00 | ||||
| High tertile | 0.24 ± 0.07 | 0.24 ± 0.07 | 0.01 | ||||
Data available for n = 191 participants. Data is presented as mean β ± SEM
EPA: eicosapentaenoic acid; DHA: docosahexaenoic acid
aTreatment effect is the difference in change in Z-score over time between the B-vitamin and placebo treatment groups within an omega-3 fatty acid tertile as analyzed using linear multiple regression, equal to Δ Z-score B-vitamin − Δ Z-score placebo
Crude model: adjusted for baseline cognitive Z-score; Adjusted model: adjusted for baseline cognitive Z-score, age, sex, level of education, ApoE4 status, baseline homocysteine concentration, baseline body mass index, physical activity, smoking status
bThe overall interaction indicates similarity of treatment effects in the low, middle and high omega-3 fatty acid tertiles
cThe pairwise comparison tests for differences in treatment effects between omega-3 fatty acid tertiles
Fig. 1.
Changes in Z-scores (mean β ± SE) in global cognition over the two year intervention period according to treatment group and A omega-3 fatty acid status, B eicosapentaenoic acid (EPA) status and C docosahexaenoic acid (DHA) status. *Significant (p < 0.05) difference in treatment effect between omega-3 fatty acid tertiles, as analyzed by linear multiple regression
Subsequently, we analysed the treatment effects related to EPA and DHA concentrations separately. The efficacy of B-vitamin supplementation related to plasma DHA levels. B-vitamin supplementation was more effective than placebo in maintaining global cognitive functioning in participants in the high DHA tertile (difference 0.24 ± 0.07, p = 0.01), while no difference in treatment effect was observed in participants in the middle or low DHA tertile (respectively, p = 1.00 and p = 0.95). The overall interaction between B-vitamin supplementation and DHA status was significant (p = 0.06). Participants in the high DHA tertile benefited significantly more from B-vitamin supplementation compared to participants in the middle DHA tertile (difference 0.23 ± 0.10, p = 0.02). Furthermore, there was a trend towards a difference in treatment effect between the high and low DHA tertile (0.18 ± 0.10 p = 0.07).
Corresponding analyses for potential interaction with EPA, i.e. comparing B-vitamin and placebo supplementation for EPA status, revealed that no treatment effect was observed in any of the EPA groups. In addition, there was no significant overall interaction between B-vitamin supplementation and EPA status (p = 0.97).
Domain-specific cognitive functioning
For none of the four cognitive domains separately; i.e. episodic memory, attention & working memory, information processing speed, and executive functioning, there was a difference in treatment effect in any of the combined omega-3 fatty acid groups (Supplementary Table 2). In addition, there was no significant overall interaction between B-vitamin supplementation and omega-3 fatty acid group. Similarly, when domain-specific performance was assessed in relation to tertiles of baseline concentrations of EPA and DHA individually, there were no significant treatment effects or interactions (Supplementary Tables 3, 4).
Discussion
This post-hoc analysis of the B-proof trial showed that the efficacy of B-vitamin supplementation on global cognition may be related to plasma DHA levels, but not to plasma total omega-3 fatty acid or EPA levels. Individuals with higher DHA plasma levels benefitted from B-vitamin supplementation, while individuals with lower DHA plasma levels did not. With respect to domain-specific cognitive performance, plasma omega-3 fatty acid combined, DHA or EPA levels separately did not modify the treatment effect of B-vitamins on episodic memory, attention & working memory, information processing speed nor executive functioning.
To date, the interaction between B-vitamins and omega-3 fatty acids in relation to cognitive decline have been investigated in three post-hoc analyses and one clinical trial, with mostly similar [5, 6, 22, 23] but also contrasting [7] findings. In line with our results, the VITACOG trial, in which older adults (> 70y) with MCI were supplemented with B-vitamins (folic acid, vitamin B6 and B12) versus placebo for 2 years, showed that omega-3 fatty acid status influenced B-vitamin treatment efficacy. Only individuals with higher plasma omega-3 fatty acid levels showed slower rates of cognitive decline [5] and brain atrophy [6] following B-vitamin supplementation. Similarly, a post-hoc analysis of the OmegAD randomized controlled trial showed that adequate levels of both omega-3 fatty acids and B-vitamins are needed [22]. In the OmegAD trial on the effect of 6 month daily supplementation with EPA and DHA versus placebo in AD patients, only subjects with lower homocysteine status benefited from omega-3 fatty acid supplementation. Further proof comes from a recent randomized controlled trial with a factorial design, in which older adults with MCI were supplemented with placebo, 0.8 mg folic acid, 0.8 mg DHA or a combination of the two daily for 6 months. Combined intervention of folic acid and DHA was more effective in improving cognition compared to supplementation with only folic acid or DHA, adding proof for the interaction from a factorial clinical trial [23].
Contrary to our current results and previous studies, the post-hoc analysis of the FACIT trial [7], performed by our group, showed that either sufficient availability of omega-3 fatty acids or B-vitamins may be needed. In this randomized controlled trial on the effect of 3-year daily supplementation with folic acid in cognitively healthy middle-aged adults (50–70 years) with elevated plasma homocysteine, folic acid supplementation was only beneficial in improving cognition in individuals with lower omega-3 fatty acid status, while individuals with higher omega-3 fatty acid status did not experience benefits.
The B-proof, VITACOG and OmegAD trials differed from the FACIT trial on various different aspects that could potentially explain the opposite findings. Importantly, B-proof, VITACOG and OmegAD participants were older, with an average age of over 70, versus an average age of 60 in the FACIT trial. In older individuals, needs for omega-3 fatty acids may be higher due to changes in dietary intake, bioavailability and increased membrane synthesis rates, as discussed previously [7]. Additionally, baseline omega-3 fatty acid status could be different between study populations, yet no direct comparison can be made due to differences in the fatty acid fractions analyzed, analytical methods and expressed measures. However, the omega-3 fatty acid distribution of our study population is similar to that of other study populations from European (non-Scandinavian) countries [24]. Vitamin B12 status also differed between study populations, as in the FACIT trial individuals with vitamin B12 deficiency were excluded. In our previous publication, we hypothesized that the contrasting findings of the FACIT trial could be attributed to differences in baseline homocysteine status and/or type of B-vitamin intervention. These factors now seem less probable, as homocysteine levels were both elevated in FACIT and B-proof trials and B-vitamin treatment included only folic acid in both the FACIT trial and in the clinical trial of Li and colleagues [7, 23]. We strongly encourage researchers with access to data on both B-vitamin and omega-3 fatty acid status to perform post-hoc analyses to be able to better define populations that may benefit from a combination of B-vitamins and omega-3 fatty acids. These results can be the basis for the design of future clinical trials with a factorial design (comparing B-vitamin supplementation only, omega-3 fatty acid supplementation only, combined supplementation versus placebo).
A mechanistic explanation for the finding that B-vitamin supplementation was more effective in individuals with higher DHA status, may involve the interaction of B-vitamins with phospholipid metabolism [25]. Phosphatidylcholine (PC) plays a crucial role in the transport of omega-3 fatty acids, including DHA, to the brain. Interestingly, B-vitamins can influence the formation of PC [25]. In the one-carbon metabolism, the B-vitamins folic acid, B6 and B12 play an important role in regulating homocysteine levels. Inadequate B-vitamin status results in elevated levels of homocysteine and its precursor, S-adenosyl homocysteine (SAH) [26]. In turn, the accumulation of SAH slows down the enzyme phosphatidylethanolamine-N-methyltransferase, which converts phosphatidylethanolamine to PC [25]. In short, adequate B-vitamin status is needed to ensure sufficient PC production, and thus transport of omega-3 fatty acids to the brain. To support this possible mechanistic explanation, for further research it would be interesting to measure the proportion of omega-3 fatty acids bound to PC.
Here we demonstrated that DHA status, but not EPA or total omega-3 fatty acid status, modified efficacy of B-vitamin supplementation. An explanation may again involve the regulatory role of B-vitamins for omega-3 fatty acid transport to the brain. EPA and DHA have different mechanisms to promote brain health. While EPA is particularly known for its anti-inflammatory effects and is only present in the brain in limited amounts, DHA is the most abundant fatty acid in the brain. This omega-3 fatty acid increases membrane fluidity which is critical for synaptic vesicles and transmission of signals, demonstrating the importance of adequate DHA levels in the brain for proper functioning of the neuronal membrane [27]. Alternatively, the differences in study populations (cognitively healthy versus MCI) and treatment (dose, combination of B-vitamins versus folic acid) between our study and previous studies, may be responsible for the lack of interaction with EPA in the current study.
The current analyses were limited to the interaction between vitamin B12/folic acid and omega-3 fatty acids, yet there are indications that also other nutrients may be involved. Bowman and colleagues [28] demonstrated a possible role for vitamin D, by showing that adequate vitamin D status further enhances the protective effect of sufficient homocysteine and omega-3 fatty acid levels in cognitive ageing. Additionally, omega-3 fatty acids may interact with antioxidants: a post-hoc analysis of an antioxidant supplementation trial demonstrated that the association of omega-3 fatty acid intake with cognitive functioning was modulated by a multi-nutrient antioxidant supplement containing ascorbic acid, vitamin E, beta-carotene, selenium and zinc [29], illustrating the importance of a multi-nutrient approach in slowing down cognitive ageing. For the current study, although we did have dietary and blood nutrient assessment data available, unfortunately we were limited by our sample size to further look into the role of other nutrients in the interaction. Further research with larger sample size should consider incorporating vitamin D status and/or antioxidant intake and status.
A major limitation of the current post-hoc analysis is that we performed exploratory analyses not designed and adequately powered to investigate the modifying potential of omega-3 fatty acid status on B-vitamin supplementation efficacy. The small sample size may be responsible for the lack of findings for domain-specific cognitive functioning, and for the lack of significant differences between the low and high DHA tertiles. Additionally, omega-3 fatty acid status was only determined at baseline and in plasma phospholipids rather than red blood cells, which is a better proxy for long-term omega-3 fatty acid status. However, we assume that our measurements do represent longer-term status as dietary patterns (and thus omega-3 intake) in older adults are reasonably stable over time [30], and other factors that may influence variation (e.g. geographic and genetic reasons) also have remained stable. Though the 2-year duration of the trial is a fairly short period of time to recognize cognitive deteriorations in healthy older individuals, it can still be considered a strength as an intervention period of 2 years is quite long in comparison with other nutrition intervention studies to slow cognitive decline. Another strength of the study is the use of an extensive cognitive test battery with a focus on domain-specific tests, instead of general tests such as the MMSE or Telephone Interview for Cognitive Status.
In conclusion, this post-hoc analysis demonstrated that B-vitamin supplementation effectiveness in cognitive ageing is related to plasma DHA levels, with older adults with higher plasma DHA levels benefitting more from B-vitamin supplementation. The results support earlier observations that positive effects of B-vitamins in cognitive ageing may be subgroup-specific. Further research is needed to optimize defining subgroups that may be susceptible for B-vitamin supplementation, and subsequently to confirm this finding in a clinical trial with a factorial design.
Supplementary Information
Below is the link to the electronic supplementary material.
Acknowledgements
The authors gratefully acknowledge all study participants and the dedicated B-proof research team.
Abbreviations
- AD
Alzheimer’s disease
- BMI
Body mass index
- B-proof
B-vitamins for the prevention of osteoporotic fractures
- DHA
Docosahexaenoic acid
- EPA
Eicosapentaenoic acid
- MCI
Mild cognitive impairment
- MMSE
Mini mental state examination
- PC
Phosphatidylcholine
- RAVLT
Rey auditory verbal learning test
- SDMT
Symbol digit modalities test
- TMT
Trail making test
Author contributions
OvdR and LCPGMdG contributed to the study conception and design. APMvS analyzed the data and wrote the first draft of the manuscript. All authors contributed to the revision of the manuscript and read and approved the final manuscript.
Funding
B-PROOF was supported and funded by The Netherlands Organization for Health Research and Development (ZonMw, grant 6130.0031), the Hague; support was also received from an unrestricted grant from NZO (Dutch Dairy Association), Zoetermeer; MCO Health, Almere; NCHA (Netherlands Consortium Healthy Ageing) Leiden/Rotterdam; Ministry of Economic Affairs, Agriculture and Innovation (project KB-15-004-003), the Hague; Wageningen University, Wageningen; VU University Medical Center, Amsterdam; and Erasmus Medical Center, Rotterdam. The fatty acid analysis was supported by the Nutrition and Healthcare Alliance, Ede.
Data availability
Data and code are available upon reasonable request in consultation with the study team.
Declarations
Conflict of interest
The authors have no relevant financial or non-financial interest to disclose.
Consent to participate and for publication
All participants gave written informed consent.
Ethics approval
This study was performed in line with the principles of the Declaration of Helsinki. Approval was granted by the Medical Ethics committee of Wageningen University and Research (ABR20783.081.07).
References
- 1.World Health Organization . Dementia: a public health priority. Geneva: World Health Organization; 2012. [Google Scholar]
- 2.Smith AD, Refsum H, Bottiglieri T, Fenech M, Hooshmand B, McCaddon A, Miller JW, Rosenberg IH, Obeid R. Homocysteine and dementia: an international consensus statement. J Alzheimers Dis. 2018;62(2):561–570. doi: 10.3233/JAD-171042. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Smith AD, Refsum H. Homocysteine, B vitamins, and cognitive impairment. Annu Rev Nutr. 2016 doi: 10.1146/annurev-nutr-071715-050947. [DOI] [PubMed] [Google Scholar]
- 4.Clarke R, Bennett D, Parish S, Lewington S, Skeaff M, Eussen SJ, Lewerin C, Stott DJ, Armitage J, Hankey GJ. Effects of homocysteine lowering with B vitamins on cognitive aging: meta-analysis of 11 trials with cognitive data on 22,000 individuals. Am J Clin Nutr. 2014;100(2):657–666. doi: 10.3945/ajcn.113.076349. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Oulhaj A, Jernerén F, Refsum H, Smith AD, de Jager CA. Omega-3 fatty acid status enhances the prevention of cognitive decline by B vitamins in mild cognitive impairment. J Alzheimers Dis. 2016;50(2):547–557. doi: 10.3233/JAD-150777. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Jernerén F, Elshorbagy AK, Oulhaj A, Smith SM, Refsum H, Smith AD. Brain atrophy in cognitively impaired elderly: the importance of long-chain ω-3 fatty acids and B vitamin status in a randomized controlled trial. Am J Clin Nutr. 2015;102(1):215–221. doi: 10.3945/ajcn.114.103283. [DOI] [PubMed] [Google Scholar]
- 7.van Soest AP, van de Rest O, Witkamp RF, de Groot LC. Positive effects of folic acid supplementation on cognitive aging are dependent on ω-3 fatty acid status: a post hoc analysis of the FACIT trial. Am J Clin Nutr. 2021;113(4):801–809. doi: 10.1093/ajcn/nqaa373. [DOI] [PubMed] [Google Scholar]
- 8.van der Zwaluw NL, Dhonukshe-Rutten RA, van Wijngaarden JP, Brouwer-Brolsma EM, van de Rest O, In't Veld PH, Enneman AW, van Dijk SC, Ham AC, Swart KM. Results of 2-year vitamin B treatment on cognitive performance: secondary data from an RCT. Neurology. 2014;83(23):2158–2166. doi: 10.1212/WNL.0000000000001050. [DOI] [PubMed] [Google Scholar]
- 9.Van Wijngaarden JP, Dhonukshe-Rutten RAM, Van Schoor NM, Van Der Velde N, Swart KMA, Enneman AW, Van Dijk SC, Brouwer-Brolsma EM, Zillikens MC, Van Meurs JBJ, Brug J, Uitterlinden AG, Lips P, De Groot LCPGM. Rationale and design of the B-PROOF study, a randomized controlled trial on the effect of supplemental intake of vitamin B 12and folic acid on fracture incidence. BMC Geriatr. 2011 doi: 10.1186/1471-2318-11-80. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Schmidt M. Rey auditory verbal learning test: a handbook. Los Angeles: Western Psychological Services; 1996. [Google Scholar]
- 11.Wechsler D (1981) WAIS-R manual: Wechsler adult intelligence scale-revised. Psychological Corporation
- 12.Reitan RM. Validity of the Trail Making Test as an indicator of organic brain damage. Percept Mot Skills. 1958;8(3):271–276. doi: 10.2466/pms.1958.8.3.271. [DOI] [Google Scholar]
- 13.Stroop JR. Studies of interference in serial verbal reactions. J Exp Psychol. 1935;18(6):643. doi: 10.1037/h0054651. [DOI] [Google Scholar]
- 14.Smith A. Symbol digit modalities test. Los Angeles: Western Psychological Services; 1982. [Google Scholar]
- 15.Lezak MD, Howieson DB, Loring DW, Fischer JS. Neuropsychological assessment. Oxford: Oxford University Press; 2004. [Google Scholar]
- 16.Zeleniuch-Jacquotte A, Chajès V, Van Kappel A, Riboli E, Toniolo P. Reliability of fatty acid composition in human serum phospholipids. Eur J Clin Nutr. 2000;54(5):367–372. doi: 10.1038/sj.ejcn.1600964. [DOI] [PubMed] [Google Scholar]
- 17.Pertiwi K, Kok DE, Wanders AJ, de Goede J, Zock PL, Geleijnse JM. Circulating n-3 fatty acids and linoleic acid as indicators of dietary fatty acid intake in post-myocardial infarction patients. Nutr Metab Cardiovasc Dis. 2019;29(4):343–350. doi: 10.1016/j.numecd.2018.12.010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Ubbink JB, Hayward Vermaak WJ, Bissbort S. Rapid high-performance liquid chromatographic assay for total homocysteine levels in human serum. J Chromatogr B Biomed Sci Appl. 1991;565(1–2):441–446. doi: 10.1016/0378-4347(91)80407-4. [DOI] [PubMed] [Google Scholar]
- 19.Stel VS, Smit JH, Pluijm SM, Visser M, Deeg DJ, Lips P. Comparison of the LASA Physical Activity Questionnaire with a 7-day diary and pedometer. J Clin Epidemiol. 2004;57(3):252–258. doi: 10.1016/j.jclinepi.2003.07.008. [DOI] [PubMed] [Google Scholar]
- 20.Folstein MF, Folstein SE, McHugh PR. "Mini-mental state". A practical method for grading the cognitive state of patients for the clinician. J Psychiatr Res. 1975;12(3):189–198. doi: 10.1016/0022-3956(75)90026-6. [DOI] [PubMed] [Google Scholar]
- 21.R Core Team (2021) R: A language and environment for statistical computing. R Foundation for Statistical Computing. https://www.R-project.org/
- 22.Jernerén F, Cederholm T, Refsum H, Smith AD, Turner C, Palmblad J, Eriksdotter M, Hjorth E, Faxen-Irving G, Wahlund L-O. Homocysteine status modifies the treatment effect of omega-3 fatty acids on cognition in a randomized clinical trial in mild to moderate Alzheimer’s disease: The OmegAD Study. J Alzheimers Dis. 2019;69(1):189–197. doi: 10.3233/JAD-181148. [DOI] [PubMed] [Google Scholar]
- 23.Li M, Li W, Gao Y, Chen Y, Bai D, Weng J, Du Y, Ma F, Wang X, Liu H, Huang G. Effect of folic acid combined with docosahexaenoic acid intervention on mild cognitive impairment in elderly: a randomized double-blind, placebo-controlled trial. Eur J Nutr. 2021;60(4):1795–1808. doi: 10.1007/s00394-020-02373-3. [DOI] [PubMed] [Google Scholar]
- 24.Stark KD, Van Elswyk ME, Higgins MR, Weatherford CA, Salem N. Global survey of the omega-3 fatty acids, docosahexaenoic acid and eicosapentaenoic acid in the blood stream of healthy adults. Prog Lipid Res. 2016;63:132–152. doi: 10.1016/j.plipres.2016.05.001. [DOI] [PubMed] [Google Scholar]
- 25.Selley ML. A metabolic link between S-adenosylhomocysteine and polyunsaturated fatty acid metabolism in Alzheimer's disease. Neurobiol Aging. 2007;28(12):1834–1839. doi: 10.1016/j.neurobiolaging.2006.08.003. [DOI] [PubMed] [Google Scholar]
- 26.Kumar A, Palfrey HA, Pathak R, Kadowitz PJ, Gettys TW, Murthy SN. The metabolism and significance of homocysteine in nutrition and health. Nutr Metab. 2017 doi: 10.1186/s12986-017-0233-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Dyall SC. Long-chain omega-3 fatty acids and the brain: a review of the independent and shared effects of EPA, DPA and DHA. Front Aging Neurosci. 2015;7:52. doi: 10.3389/fnagi.2015.00052. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Bowman GL, Dodge HH, Guyonnet S, Zhou N, Donohue J, Bichsel A, Schmitt J, Hooper C, Bartfai T, Andrieu S, Vellas B, Carrié I, Brigitte L, Faisant C, Lala F, Delrieu J, Villars H, Combrouze E, Badufle C, Zueras A, Cantet C, Morin C, Van Kan GA, Dupuy C, Rolland Y, Caillaud C, Ousset PJ. A blood-based nutritional risk index explains cognitive enhancement and decline in the multidomain Alzheimer prevention trial. Alzheimer's Dementia Transl Res Clin Interv. 2019;5:953–963. doi: 10.1016/j.trci.2019.11.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Assmann KE, Adjibade M, Hercberg S, Galan P, Kesse-Guyot E. Unsaturated fatty acid intakes during midlife are positively associated with later cognitive function in older adults with modulating effects of antioxidant supplementation. J Nutr. 2018;148(12):1938–1945. doi: 10.1093/jn/nxy206. [DOI] [PubMed] [Google Scholar]
- 30.Jankovic N, Steppel MT, Kampman E, De Groot LC, Boshuizen HC, Soedamah-Muthu SS, Kromhout D, Feskens EJ. Stability of dietary patterns assessed with reduced rank regression. The Zutphen Elderly Study. Nutr J. 2014;13(1):1–9. doi: 10.1186/1475-2891-13-30. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Data Availability Statement
Data and code are available upon reasonable request in consultation with the study team.