Abstract
Coronavirus disease 2019 (COVID-19), caused by severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2), was first identified in Wuhan, China in December 2019. Since then, the disease has spread globally, leading to the ongoing pandemic. It can cause severe respiratory illness; however, many cases of pericarditis have also been reported. This systematic review aims to recognize the clinical features of pericarditis and myopericarditis in COVID-19 patients.
Google Scholar, Medline/PubMed, CINAHL, Cochrane Central, and Web of Science databases were searched for studies reporting “Coronavirus” or “COVID” and “Peri-myocarditis,” “heart,” or “retrospective.” Case reports and retrospective studies published from May 2020 to February 2021 were reviewed.
In total, 33 studies on pericarditis, myopericarditis, and pericardial infusion were included in this review. COVID-19 pericarditis affected adult patients at any age. The incidence is more common in males, with a male-to-female ratio of 2:1. Chest pain (60%), fever (51%), and shortness of breath (51%) were the most reported symptoms, followed by cough (39%), fatigue (15%), myalgia (12%), and diarrhea (12%). Laboratory tests revealed leukocytosis with neutrophil predominance, elevated D-dimer, erythrocyte rate, and C-reactive protein. Cardiac markers including troponin-1, troponin-T, and brain natriuretic peptide were elevated in most cases. Radiographic imaging of the chest were mostly normal, and only 31% of chest X-rays showed cardiomegaly and or bilateral infiltration. Electrocardiography (ECG) demonstrated normal sinus rhythm with around 59% ST elevation and rarely PR depression or T wave inversion, while the predominant echocardiographic feature was pericardial effusion. Management with colchicine was favored in most cases, followed by non-steroidal anti-inflammatory drugs (NSAIDs), and interventional therapy was only needed when patient developed cardiac tamponade. The majority of the reviewed studies reported either recovery or no continued clinical deterioration.
The prevalence of COVID-19-related cardiac diseases is high, and pericarditis is a known extrapulmonary manifestation. However, pericardial effusion and cardiac tamponade are less prevalent and may require urgent intervention to prevent mortality. Pericarditis should be considered in patients with chest pain, ST elevation on ECG, a normal coronary angiogram, and COVID-19. We emphasize the importance of clinical examination, ECG, and echocardiogram for decision-making, and NSAIDs, colchicine, and corticosteroids are considered to be safe in the treatment of pericarditis/myopericarditis associated with COVID-19.
Keywords: brain natriuretic peptide, chest x-ray, sars-cov-2, covid-19, myopericarditis, echocardiogram, pericarditis, electrocardiography (ecg), erythrocyte sedimentation rate, c-reactive protein
Introduction and background
The novel coronavirus disease 2019 (COVID-19) is a global pandemic caused by severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) [1]. The pandemic began in Wuhan, China in December 2019. While affecting dominantly the respiratory system, COVID-19 can also cause acute and chronic damage to the cardiovascular system. The cardiovascular manifestations of COVID-19 are diverse and include arrhythmia, acute coronary syndrome, left ventricular heart failure, myocarditis, and acute and subacute pericarditis with or without pericardial effusion [2-4].
Pericarditis refers to the inflammation of the pericardium, the thin fibrous sac surrounding the heart, that can present as an isolated disease or as a manifestation of a systemic disorder, diagnosed in approximately 0.1% of patients hospitalized for chest pain. Although acute pericarditis has many causes, it is most often idiopathic or is presumed to be viral in origin [5].
Acute effusive pericarditis is a rare manifestation of COVID-19, especially without concomitant pulmonary disease or myocardial injury; yet, very little research is available regarding pericarditis caused by SARS-CoV-2. It is important to maintain a high level of suspicion to ensure early diagnosis and treatment. Diagnosing these conditions can be challenging, and early appropriate treatment can improve the outcome. Therefore, we conducted a systematic review of COVID-19 patients with acute pericarditis to assess clinical characteristics, diagnostic testing, and current treatment therapy.
Acute pericarditis and myopericarditis share the same viral etiological agents, with myocardial involvement often found in the former [6]. Therefore, we have included myopericarditis in this study.
Review
Methodology
Protocol and Registration
The Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) checklist was followed for this systematic review [7]. The study protocol was not registered. The Joanna Briggs Institute critical appraisal tool for case reports was used in this systematic review [8].
Inclusion Criteria
Only articles reporting the association between COVID-19 and myopericarditis or pericarditis were included. Pericarditis is defined as an inflammatory process affecting the pericardial sac surrounding the heart [6]. The search terms that were used in our article screening were “COVID-19,” “Pericarditis,” “Pericardial effusion,” “Pericardial disease,” “Cardiac tamponade,” and “Myocarditis.”
Exclusion Criteria
Studies were excluded if: (1) they were not case reports, case series, or observational studies with a focus on myopericarditis or pericarditis; (2) they were reviews or editorials, (3) the patient tested negative for COVID-19; or (4) the study did not pass bias evaluation. The language of the article was not a limitation as all of the articles which were relevant in our literature search were written in the English language.
Information Sources and Search Strategies
We conducted a comprehensive literature search using Medline/PubMed, Google Scholar, CINAHL, Cochrane Central, and Web of Science databases up to and including February 28, 2021, using the terms “Coronavirus” or “COVID” and “pericarditis” or “myopericarditis.”
Study Selection
Articles were triaged based on whether titles or abstracts met the inclusion criteria. Full-text articles were then read, and those that did not satisfy the inclusion criteria were excluded. A summary of study characteristics is given in Table 1.
Table 1. Summary of the characteristics of included articles (n = 33).
COVID-19: coronavirus disease 2019; ED: emergency department; ICU: intensive care unit
| Reference, publication year | Study type | Patient profile (age in years, sex) | Symptoms | Diagnosis |
| Amoozgar et al., 2020 [9] | Case report | 56, Male | Non-radiating exertional chest pain with dyspnea for 1 week | Acute pericarditis |
| Asif et al., 2020 [10] | Case report | 70, Female | Chest pain, worsening dyspnea, myalgias for 3 days | Acute pericarditis |
| Blagojevic et al., 2020 [11] | Case report | 51, Male | Sudden but persistent chest pain for 1 day; the pain was sharp, worsened with deep breathing or a change in body position, and was alleviated while sitting | Acute pericarditis |
| Cairns et al., 2021 [12] | Case report | 58, Female | Fever, diarrhea, vomiting, poor oral intake | Acute Myopericarditis |
| Cizgici et al., 2020 [13] | Case report | 78, Male | Chest pain and shortness of breath | Acute myopericarditis |
| Faraj et al., 2021 [14] | Case report | 36, Male | Chest pain, worse when lying or on deep breathing | Acute pericarditis |
| Fox et al., 2020 [15] | Case report | 43, Male | Progressive orthopnea, conversational dyspnea, and chest pain (radiating to the neck and left shoulder) for 4 days; reported mild non-productive cough and subjective fever 2 weeks prior | Acute pericarditis |
| Fried et al., 2020 [16] | Case series | 64, Female | Persistent chest pressure for 2 days | Acute myopericarditis |
| García-Cruz et al., 2020 [17] | Case report | 64, Male | Chest pain, dry cough, fever, dyspnea | Acute pericarditis |
| Inciardi et al., 2020 [18] | Case report | 53, Female | Severe fatigue for 2 days, fever, and cough the week before | Acute myopericarditis |
| Karadeniz et al., 2020 [19] | Case report | 33, Male | Retrosternal chest pain for 5 days, worse with sitting forward, unresponsive to diclofenac, severe low back pain (for 1 week) | Acute pericarditis |
| Kazi et al., 2020 [20] | Case report | 73, Male | Dry cough, worsening fever, fatigue for 2 days before presenting to ED (6 days before transfer to ICU); dyspnea developed over next 4 days | Acute myopericarditis |
| Khalid et al., 2020 [21] | Case series | 34, Female | Chest heaviness, generalized weakness, subjective fever/chills, body aches for 3 days | Acute myopericarditis |
| Khatri et al., 2020 [22] | Case report | 50, Male | Fever, chills, generalized malaise, non-productive cough, dyspnea for 3-4 days, and an episode of near-syncope on the day of presentation | Acute myopericarditis |
| Kumar et al., 2020 [23] | Case report | 66, Male | Acute-onset severe pleuritic chest pain for 1 day (4 episodes lasting 10-15 minutes); pain worse lying down, relieved by leaning forward | Acute pericarditis |
| Legrand et al., 2020 [24] | Case report | 39, Male | Chest pain and dyspnea for 2 days | Acute myopericarditis |
| Li et al., 2020 [25] | Case report | 60, Male | fever, cough, worsening dyspnea, mild abdominal pain, diarrhea for 8 days | Acute myopericarditis |
| Marschall et al., 2020 [26] | Case report | 45, Male | Dyspnea with minimal exertion, orthopnea, bendopnea | Acute pericarditis |
| Naqvi et al., 2020 [27] | Case report | 55, Male | Chest pain for 24 hours | Acute pericarditis |
| Ortiz-Martínez et al., 2020 [28] | Case report | 25, Male | Myalgias, arthralgias, diarrhea, 2 days later: fever and nausea, began isolation, on 8th day: intense pleuritic centrothoracic chest pain, improved with sitting forward, worse with supine, dyspnea at rest | acute pericarditis |
| Özturan et al., 2020 [29] | Case report | 25, Male | Acute onset chest pain and shortness of breath, 4-day history of progressive fatigue and fever | Acute myopericarditis |
| Patel et al, 2021 [30] | Case report | 63, Male | Fever, dry cough, and malaise for 1 week; chest pain for 1 day | Acute pericarditis |
| Purohit et al., 2020 [31] | Case report | 82, Female | Productive cough, fever with chills, intermittent diarrhea for 5 days | Acute myopericarditis |
| Raymond et al., 2020 [32] | Case report | 7, Female | Cough, chest pain, orthopnea for 3 days | Acute pericarditis |
| Recalcati et al., 2020 [33] | Case report | 19, Female | Fever followed by chest pain, cutaneous rash | Acute myopericarditis |
| Sampaio et al., 2020 [34] | Case report | 45, Female | Dyspnea, fever, myalgia, postural hypotension progressing over 7 days | Acute myopericarditis |
| Sandino Pérez et al., 2020 [35] | Case report | 73, Male | Cough, dyspnea on moderate exertion, fever for 4 days | Acute pericarditis |
| Sauer et al., 2020 [36] | Case series | 60, Male | Asthenia for 1 week, acute anosmia, one month later prescribed amoxicillin-clavulanic acid for shivers and developed diffuse erythema 2 days later | Acute myopericarditis |
| Shah et al., 2020 [37] | Case series | 38, Male | Hospitalized for 3 weeks with COVID-19 pneumonia, presented to ED a few hours after discharge with pricking chest pain not related to breathing, exertion, or posture | Acute myopericarditis |
| Thrupthi et al., 2021 [38] | Case report | 68, Male | Chest tightness for 5 days, dry cough, mild fatigue, shortness of breath on exertion, chest pain | Acute pericarditis |
| Tiwary et al., 2020 [39] | Case report | 30, Male | Bilateral abdominal flank pain, shortness of breath, fatigue, tiredness, lightheadedness | Myopericarditis |
| Tung-Chen et al., 2020 [40] | Case report | 35, Female | Dry cough, anosmia, malaise, low-grade fever | Acute pericarditis |
| Walker et al., 2020 [41] | Case report | 30, Female | Fever, dry cough, exertional dyspnea for 3 days | Acute pericarditis |
Data Collection Process and Data Items
Data extracted from articles included the name of the first author, year of publication, country, and study design. Variables for which data were sought from all studies included patient age and sex and presenting complaints at the time of admission. Laboratory tests, diagnostic studies, management of pericarditis, and patient outcomes including complications were extrapolated from case studies.
Analysis of Results and Summary of Measures
Information was reviewed if it was reported by two or more articles. Subsequently, the data were tabulated, evaluated, and summarized.
Risk of Bias Across Studies
Potential bias across studies was analyzed within study characteristics. Two independent reviewers evaluated the methodological quality of the eligible studies. A third reviewer evaluated papers when there was no agreement. The Joanna Briggs Institute critical appraisal tool for case reports was used in this systematic review [8]. Bias was evaluated using a checklist of eight questions. Each question is specified in the Appendix concerning the risk of bias whereby an overall appraisal was made of each article: low risk of bias (included), high risk of bias (excluded), or uncertain risk of bias (more information is required). For this study, an answer of “yes” equal to or greater than 50% of the questions was considered to be low risk of bias. Similarly, an answer of “no” equal to or greater than 50% of questions was determined to be high risk of bias, whereas “unclear” answers were equal to or greater than a 50% response.
Results
Study Selection
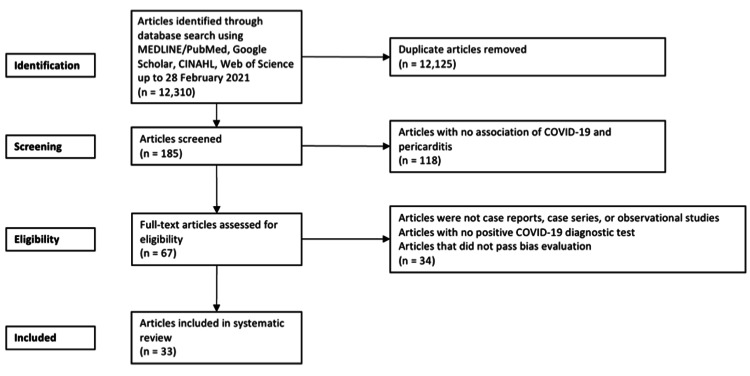
From the five databases, 12,310 articles were selected concerning COVID-19 and myocarditis. In total, 34 articles were selected once assessed for eligibility [9-42]. The study by Rauch et al. [42] was removed from this list as it did not meet the minimum criteria required when assessed for bias. A PRISMA flow diagram detailing the process of identification, inclusion, and exclusion of studies is shown in Figure 1.
Figure 1. Flow diagram of the literature search and selection criteria adapted from the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA).
Study Characteristics
In total, 29 articles selected for this study were case reports [9-15,17-20,22-35,38-41] while four were case series [16,21,36,37]. All articles were published in 2020 [9-11,13,15-29,31-37,39-41], except four, which were published in 2021 [12,14,30,38]. The majority of the studies were conducted in the United States [9,10,15,16,20-22,25,30-32,38-41], followed by countries from the European Union or United kingdom [11,12,18,23,24,26,33,35,36,40] and the Middle East or Asia [12,19,27,29,37]. Only three studies originated from Latin America [17,28,34] and one from northern Africa [14].
Risk of Bias Within Studies
In comparison to case reports, the majority of articles were rated to have a low risk of bias [9-41]. As mentioned previously, only one study was characterized as high risk of bias [42] and was removed from the systematic review.
Results of Individual Studies
The common theme of the studies was either the identification of only pericarditis [10,11,14,16,19,23,26,28,30,35,37,38,40], myopericarditis [13,18,20,21,24,25,29,33,34], or the association of cardiac tamponade or pericardial effusion and either pericarditis or myopericarditis [9,12,15,17,22,27,31,32,36,39,41] in COVID-19 patients.
Patient Profile
COVID-19 patients selected for the systematic review were 49.3 ± 18.5 years of age, with more males affected than females at a ratio of 2:1 [9-41]. The eldest was 82 years old [31], while the youngest was seven years old [32].
Presenting Complaints
The predominant symptom at admission was chest pain [9,11,13-17,19,21,23,24,27-30,32,33,37,38], followed by fever [12,15,17,18,20-22,25,28-31,33-35,40,41], shortness of breath [10,13,15,17,20,22,24-26,28,29,32,34,35,38,39,41], and cough [15,17,18,20,22,25,30-32,35,38,40,41]. The mean temperature recorded was 37.5 ± 1.1°C [9-12,14,16-21,22,30,32,33,35-38,41] while the mean blood pressure was 112.0 ± 20.3/71.7 ± 16.0 [3-12,14-21,23,26-30,34,37,38,41]. The average heart rate was also elevated at 103.9 ± 21.3 beats/minute [9-12,14-21,23-30,32,34,37,38,41]. The mean oxygen saturation was 96.2 ± 0.1% [9-12,14-21,23,25-30,33,34,36-38,41]. The distribution of presenting complaints and associated symptoms are listed in Table 2.
Table 2. Most common clinical manifestations on admission of patients with COVID-19 and pericarditis and myopericarditis as the proportion reported from all articles (n = 33).
| Symptom | % |
| Chest pain | 60.6 |
| Fever | 51.5 |
| Shortness of breath | 51.5 |
| Cough | 39.4 |
Medical History
Nearly half of the patients reported a medical history of hypertension [10-13,16,20,22,23,25,27,30,31,38,39], followed by diabetes mellitus [10,12,20,37,39] and hyperlipidemia [10,16,25,31].
Laboratory Tests
A summary of laboratory tests is presented in Table 3.
Table 3. Trends of laboratory values of COVID-19 patients with pericarditis and myopericarditis from all articles (n = 33).
SaO2: oxygen saturation (arterial blood); WBC: white blood cell; CRP: C-reactive protein; ESR: erythrocyte sedimentation rate; CK-MB: creatine kinase-myoglobin binding; BNP: brain natriuretic peptide; COVID-19: coronavirus disease 2019
| Trends | (Standard range) | |
| Vitals | ||
| Temperature, °C (n = 25) | Elevated | (<37.5) |
| Heart rate, beats/minute (n = 25) | Elevated | |
| Systolic blood pressure, mmHg (n = 22) | Normal | (90–120) |
| Diastolic blood pressure, mmHg (n = 21) | Normal | (60–80) |
| SaO2, % (n = 25) | Normal | (>94) |
| Inflammatory markers | ||
| WBC, cells/mm3 (n = 17) | Elevated | (4,500–11,000) |
| WBC predominance (n = 8) | Neutrophils | |
| CRP, mg/L (n = 21) | Elevated | (<8.0) |
| ESR, mm/hour (n = 5) | Elevated | (0–20) |
| D-dimer level, ng/mL (n = 15) | Elevated | (<250) |
| Cardiac markers | ||
| Troponin-I, ng/mL (n = 17) | Elevated | <0.04 |
| Troponin-T, ng/mL (n = 13) | Elevated | <0.01 |
| CK-MB, ng/mL (n = 3) | Elevated | <5.0 |
| BNP, pg/mL (n = 10) | Elevated | <125 |
All patients were confirmed COVID-19 positive [9-41]. The majority of studies reported leukocytosis [9,11,14,15,20,22,24-29,32-35,37,40] and neutrophil predominance [11,20,21,25,34,38-40], with a mean of 15,622 ± 2,654 cells/mm3. Increased C-reactive protein (CRP) was also reported by the majority of studies, with a mean of 15.0 ± 9.59 mg/dL [11,13-16,19-22,24-26,28-30,32-35,37-40]. Only six studies recorded erythrocyte sedimentation rate (ESR), with three reporting elevated values at a mean of 59.3 ± 25.7 mm/hour [11,21,22,30,32,38].
Two-thirds of studies showed elevated D-dimer levels [9,15,16,19,22,23,25,26,28,33-35,38,40,41], with two articles reporting values higher than 5,000 ng/mL [9,15]. Only 60.0% of studies reported either an increased troponin-I [9,11,15,16,19,21,22,24,27,29,31,32,34,36,38-40] or troponin-T [12-14,18,20,23,25,26,28,30,33,35,37]. The troponin-I values ranged from 0.544 ng/mL to 90.0 ng/mL, while mean troponin-T values ranged from 0.367 to 13.0 ng/mL. Nine out of 10 studies recorded elevated n-terminal brain natriuretic peptide (BNP), with a mean value of 4,770.1 ± 5,158.2 pg/mL [11,18,21,24,25,32,34,38,39,41]. Only three studies reported elevated creatine kinase-MB results, with a mean of 39.9 ± 13.3 ng/mL [22,25,29].
Diagnostic Studies
A summary of diagnostic studies is provided in Table 4.
Table 4. Common findings found on diagnostic tests of COVID-19 patients with pericarditis and myopericarditis as the proportion recorded from all articles (n = 33).
LVEF: left ventricular ejection fraction; CT: computed tomography; MRI: magnetic resonance imaging; COVID-19: coronavirus disease 2019
| Diagnostic study | % |
| Electrocardiography (n = 32) | |
| ST-segment elevation | 59 |
| PR depression | 28 |
| Tachycardia | 22 |
| T-wave inversion | 13 |
| Electric alternans | 3 |
| Echocardiography (n = 30) | |
| Pericardial effusion | 67 |
| Hypokinesis | 20 |
| Reduced LVEF | 20 |
| Ventricular wall thickening | 13 |
| Imaging (X-ray) (n = 16) | |
| Bilateral infiltrates | 31 |
| Cardiomegaly | 31 |
| Imaging (CT) (n = 18) | |
| Ground-glass opacities | 50 |
| Pericardial effusion | 39 |
| Pleural effusion | 33 |
| Imaging (MRI) (n = 4) | |
| Late gadolinium enhancements | 50 |
The predominant feature of electrocardiogram (ECG) recordings was sinus rhythm [10-11,15,16,20-22,32,33,36,40,41], with ST-segment elevation [10-11,13,15-18,20,22-27,30,33,35,37,38]. About one in four articles reported PR depression [10,11,15,20,23,26,29,30,37] while one in five presented with tachycardia [15,16,21,22,32,33,41]. Finally, inverted T waves were found in one in 10 studies [10,31,32,40] while only one study had the classic presentation of electric alternans [32].
Echocardiography showed pericardial infusion in nearly three out of four studies [9-12,14,15,17-19,21-22,24,25,27,28,30-32,34,35,39,41] while one in four demonstrated either hypokinesis [16,18,20,25,29,31] or reduced left ventricular ejection fraction (<50%) [16-18,21,25,29].
Less than one-third of the articles reported bilateral infiltrates on chest X-ray [10,17,26,37,38], with the same proportion highlighting cardiomegaly [9,10,14,15,32]. Likewise, computed tomography (CT) imaging of the chest revealed similar results, with 50% of images demonstrating ground-glass opacities in the lungs [9,13,19,24,28,34,39] while one in three reporting pleural effusion [9,14,19,24,34,39]. Pericardial effusion was reported in slightly more than one-third of articles [9,13,19,24,28,34,39]. Magnetic resonance imaging (MRI) was the least used modality, with only half demonstrating subepicardial delayed gadolinium enhancement [24,36].
Management of Myocarditis
The most common medical management was the use of colchicine by 45.1% of studies [14,15,19,21,23,24,26,28,30,32,35-38,40,41], followed by either aspirin or non-steroidal anti-inflammatory drugs (NSAIDs) in 41.9% of articles [11,15,19,26-30,32,33,37,38,41] and corticosteroids in 35.3% of patients [18,21,22,25,28,34,35,38]. Regarding surgical interventions, pericardiocentesis [10,12,15,21,22,27,31,32,34] or the creation of pericardial windows [10,17,39,41] were required in 13 case reports.
Outcomes
The outcomes of the patients were documented in the majority of case reports. Only two deaths were reported [20,22], with the majority being discharged from the hospital [9-19,21,23-41].
Risk of Bias Across Studies
Due to the nature of descriptive studies, the results being presented are liable to the investigator, procedure, and selection bias.
Limitation of the Study
Statistical analyses were not performed as there were no control/comparison groups in the included studies.
Discussion
Pathophysiology
SARS-CoV-2 is thought to lead to myocardial damage and inflammation through the following processes: (1) direct invasion, (2) ischemic injury, and (3) cytokine storm. SARS-CoV-2 typically invades the respiratory epithelium through direct invasion via binding to angiotensin-converting enzyme-2 (ACE2) receptors on the host cells. These receptors are predominantly found on the respiratory epithelium and are also expressed on cardiac cells, facilitating a pathway and allowing for direct invasion and damage to cardiomyocytes [43]. Hypoxemia, which can result from pulmonary compromise from SARS-CoV-2 infection, can also lead to impaired myocardial oxygen supply resulting in a supply and demand mismatch, which then leads to ischemic injury to cardiomyocytes [43]. Cytokine storm is a known physiologic mechanism associated with SARS-CoV-2 infection, leading to the widespread production of high levels of pro-inflammatory cytokines. Large-scale production of these pro-inflammatory cytokines has also been linked to myocardial damage through recorded elevations in troponin levels during these states [43]. Autopsy reports on patients who died of COVID-19 infection have also shown findings within cardiac tissue consistent with inflammatory cellular infiltrate and multi-nucleated giant cells. Additionally, findings of pericardial effusions have also been seen in autopsy examinations [43].
Clinical Presentation
The clinical manifestations of COVID-19 in order of prevalence are fever, cough, and fatigue [2]. A common clinical manifestation of pericarditis is typically sharp and pleuritic chest pain that radiates posteriorly to the bilateral trapezius ridges, which improves on sitting up or leaning forward [44]. The presence of fever, subacute course, large effusion, or tamponade are indicators of poor prognosis [45]. Auscultation of the left sternal border typically indicates a triphasic pericardial friction rub. Here, we identified that patients with COVID-19 pericarditis commonly presented with chest pain, in addition to fever and shortness of breath.
Laboratory Findings
It has been shown that thrombo-inflammatory biomarkers are associated with COVID-19 infection severity [46]. Our systematic review showed that COVID-19 patients with pericarditis exhibited leukocytosis with neutrophil predominance, elevated inflammatory markers (ESR and CRP), and elevated D-dimer levers as well. These case studies also exhibited elevated cardiac markers (troponin-I, troponin-T, CK-MB, and BNP) as well. Typically, 32-50% of cases of viral and idiopathic acute pericarditis as evidence of inflammatory myocardial damage and possible evidence of myopericarditis [47]. Patients with pericarditis and concomitant myocarditis have a potential for higher risk of complications, including left ventricular dysfunction and possible heart failure [48]. Similarly, our review showed that the deaths were from COVID-19 patients diagnosed with myopericarditis. Similarly, of the 10 studies to report COVID-19-related complications, eight of which were present in COVID-19 patients diagnosed with myopericarditis [20-22,24,25,34,36,39]. Furthermore, the only two deaths that were reported were from patients diagnosed with myopericarditis [20,22].
Imaging
The typical ECG presentation of a COVID-19 patient with pericarditis is sinus rhythm with ST-segment elevation, while a minority of individuals exhibit PR-segment depression or tachycardia. ECG presentation of widespread ST-segment elevation and PR depression, although this finding is considered characteristic for pericarditis, es only found in slightly more than half of the patients with acute pericarditis, particularly among younger males [44]. Disease progression may evolve to include T waves [44].
Echocardiographic patterns of the brightness of the pericardial layers, which are associated with fibrinous pericarditis, and pericardial effusion have been described in pericarditis [44]. Approximately 60% of patients with acute pericarditis were found to have developed a pericardial effusion, although its absence does not exclude pericarditis. Our systematic review found that the predominant feature found in the majority of patients with pericarditis is pleural effusion, while a minority of patients have hypokinesis or depressed ejection fraction.
Chest radiograph findings in patients with acute pericarditis are usually normal except in the setting of very large pericardial effusion, which would convey the characteristic water bottle-shaped cardiac silhouette [44]. CT and cardiac MRI with gadolinium contrast are recommended as secondary imaging techniques after echocardiography and chest radiographs to detect and confirm pericardial inflammation, assess pericardial thickening or late gadolinium enhancement, and identify the presence of effusion or constrictive physiology [44,49]. Likewise, the case resorts in our systematic reported predominantly COVID-19 disease of the lungs with bilateral infiltrates or ground-glass opacities, with characteristic pericarditis features of pericardial effusion or cardiomegaly demonstrated in a minority of patients. In diagnostic CT scans, findings of pericardial effusions were also studied and reported to be present in a small number of patients as well, and the degree of pericardial effusion was also seen to correlate to the severity of infection. Evidence of myocarditis was also reported on CT scans, with increases in cardiac wall thickness, myocardial edema, and wall hypokinesia. In cardiac MRIs, these studies are limited; however, there have been reports of myocardial edema and significant findings of wall motion abnormalities in patients already clinically diagnosed with acute myocarditis. The use of positron emission tomography scan in detailing diagnostic findings of pericardial effusions and cardiomegaly only highlighted small studies which did not reveal any evidence of cardiac involvement [50].
Treatment
There was no prevailing strategy regarding the medical management to treat pericarditis in patients with COVID-19. We found that colchicine, aspirin, NSAIDs, and/or corticosteroids were used in the majority of studies. NSAIDs, most commonly ibuprofen (600 to 800 mg every six to eight hours), indomethacin (25 to 50 mg every eight hours), and aspirin (2 to 4 g daily in divided doses) have long been the mainstay of medical therapy for both acute and recurrent pericarditis whether or not it is of idiopathic or viral causes [41,51-53]. Corticosteroid therapy (0.2 to 0.5 mg of prednisone per kg of body weight daily) used to be the initial choice in treating pericarditis with pericardial effusions or recurrent episodes unresponsive to aspirin or NSAIDs; however, it has been shown to be associated with an increased risk in adverse effects and higher recurrence risk in non-randomized studies [44,52]. There is considerable evidence highlighting that the use of colchicine is efficacious and safe for the treatment and prevention of both acute and recurrent pericarditis, as well as for reducing rehospitalizations and symptom duration in the process [54-58]. In the cases that were reviewed in our study, the majority utilized colchicine (17/33), and 13 cases utilized NSAIDs either in the form of aspirin, ibuprofen, or indomethacin, all of which were usually used in conjunction with colchicine, with the exception of 5/13 of the cases. Among 10/39 of the cases, there was no use of NSAID or colchicine therapy. Additionally, one case reported the use of Anakinra [19] after noting poor results with NSAID and colchicine therapy. All patients received some form of standardized COVID-19 therapy as well, including intravenous corticosteroids. All of the doses reported in the treatment of myopericarditis and pericarditis were within the appropriate dosages discussed in the standard therapies above.
Conclusions
Our systematic review provides a comprehensive characterization of the clinical features among COVID-19 patients with pericarditis. Currently, as data are limited, more research is needed to improve our understanding of COVID-19 pericarditis.
Acknowledgments
Pramod Theetha Kariyanna, Ahmed Sabih, Bayu Sutarjono, Jeremy Lewis, and Rebecca Yu contributed equally to the work.
Appendices
Table 5. Risk of bias across studies.
| Reference, publication year | Were patient’s demographic characteristics clearly described? | Was the patient’s history clearly described and presented as a timeline? | Was the current clinical condition of the patient on presentation clearly described? | Were diagnostic tests or assessment methods and results clearly described? | Was the intervention(s) or treatment procedure(s) clearly described? | Was the post-intervention clinical condition clearly described? | Were adverse events (harms) or unanticipated events identified and described? | Does the case report provide takeaway lessons? | Total score |
| Amoozgar et al., 2020 [9] | Some information missing | √ | √ | √ | √ | √ | √ | √ | 87.5% |
| Asif et al., 2020 [10] | √ | √ | √ | Some information missing | Some information missing | √ | √ | √ | 75.0% |
| Blagojevic et al., 2020 [11] | √ | √ | √ | √ | √ | √ | √ | √ | 100.0% |
| Cairns et al., 2021 [12] | √ | √ | √ | Some information missing | √ | √ | √ | √ | 87.5% |
| Cizgici et al., 2020 [13] | Some information missing | √ | Some information missing | √ | All information missing | All information missing | √ | √ | 50.0% |
| Faraj et al., 2021 [14] | Some information missing | √ | √ | √ | √ | √ | √ | √ | 87.5% |
| Fox et al., 2020 [15] | √ | √ | Some information missing | √ | √ | √ | √ | √ | 87.5% |
| Fried et al., 2020 [16] | Some information missing | √ | √ | All information missing | √ | √ | √ | √ | 75.0% |
| García-Cruz et al., 2020 [17] | √ | All information missing | √ | Some information missing | All information missing | All information missing | √ | √ | 50.0% |
| Inciardi et al., 2020 [18] | √ | √ | √ | √ | √ | √ | √ | √ | 100.0% |
| Karadeniz et al., 2020 [19] | Some information missing | √ | √ | √ | √ | √ | √ | √ | 87.5% |
| Kazi et al., 2020 [20] | √ | √ | √ | √ | Some information missing | √ | √ | √ | 87.5% |
| Khalid et al., 2020 [21] | Some information missing | √ | √ | √ | Some information missing | √ | √ | √ | 75.0% |
| Khatri et al., 2020 [22] | √ | √ | Some information missing | √ | Some information missing | √ | √ | √ | 75.0% |
| Kumar et al., 2020 [23] | Some information missing | √ | √ | √ | Some information missing | √ | √ | √ | 75.0% |
| Legrand et al., 2020 [24] | Some information missing | √ | Some information missing | √ | √ | √ | √ | √ | 75.0% |
| Li et al., 2020 [25] | Some information missing | √ | √ | √ | √ | √ | √ | √ | 87.5% |
| Marschall et al., 2020 [26] | Some information missing | √ | √ | √ | Some information missing | √ | √ | √ | 75.0% |
| Naqvi et al., 2020 [27] | √ | √ | √ | Some information missing | √ | All information missing | All information missing | √ | 62.5% |
| Ortiz-Martinez et al., 2020 [28] | Some information missing | √ | √ | √ | √ | √ | √ | √ | 87.5% |
| Özturan et al., 2020 [29] | Some information missing | √ | √ | √ | Some information missing | √ | √ | √ | 75.0% |
| Patel et al, 2021 [30] | Some information missing | √ | √ | √ | √ | √ | √ | √ | 87.5% |
| Purohit et al., 2020 [31] | Some information missing | √ | √ | √ | √ | √ | √ | √ | 87.5% |
| Raymond et al., 2020 [32] | Some information missing | √ | √ | √ | Some information missing | √ | √ | √ | 75.0% |
| Recalcati et al., 2020 [33] | Some information missing | √ | √ | √ | Some information missing | All information missing | √ | √ | 62.5% |
| Sampaio et al., 2020 [34] | Some information missing | √ | Some information missing | √ | √ | √ | √ | √ | 75.0% |
| Sandino Pérezet al., 2020 [35] | Some information missing | √ | √ | All information missing | √ | √ | √ | √ | 75.0% |
| Sauer et al., 2020 [36] | Some information missing | √ | Some information missing | Some information missing | √ | √ | √ | √ | 62.5% |
| Shah et al., 2020 [37] | Some information missing | √ | √ | √ | Some information missing | Some information missing | Some information missing | √ | 50.0% |
| Thrupthi et al., 2021 [38] | Some information missing | √ | √ | Some information missing | √ | √ | √ | √ | 75.0% |
| Tiwary et al., 2020 [39] | √ | √ | Some information missing | √ | Some information missing | √ | √ | √ | 75.0% |
| Tung-Chen et al., 2020 [40] | Some information missing | √ | Some information missing | √ | √ | √ | √ | √ | 75.0% |
| Walker et al., 2020 [41] | √ | √ | √ | √ | Some information missing | √ | √ | √ | 75.0% |
| Rauch et al., 2020 [42] | No | All information missing | All information missing | All information missing | All information missing | All information missing | √ | √ | 25.0% |
The content published in Cureus is the result of clinical experience and/or research by independent individuals or organizations. Cureus is not responsible for the scientific accuracy or reliability of data or conclusions published herein. All content published within Cureus is intended only for educational, research and reference purposes. Additionally, articles published within Cureus should not be deemed a suitable substitute for the advice of a qualified health care professional. Do not disregard or avoid professional medical advice due to content published within Cureus.
Footnotes
The authors have declared that no competing interests exist.
References
- 1.Report of the WHO-China Joint Mission on Coronavirus Disease 2019 (COVID-19) [ Jun; 2022 ];https://www.who.int/docs/default-source/coronaviruse/who-china-joint-mission-on-covid-19-final-report.pdf 2020
- 2.Clinical characteristics of coronavirus disease 2019 (COVID-19) in China: a systematic review and meta-analysis. Fu L, Wang B, Yuan T, et al. J Infect. 2020;80:656–665. doi: 10.1016/j.jinf.2020.03.041. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.COVID-19 and the cardiovascular system. Zheng YY, Ma YT, Zhang JY, Xie X. Nat Rev Cardiol. 2020;17:259–260. doi: 10.1038/s41569-020-0360-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Cardiovascular conundrums of COVID-19 pandemic. Otaal PS, Batta A, Makkar K, Vijayvergiya R. J Postgrad Med Educ Res. 2020;54:160–162. [Google Scholar]
- 5.2015 ESC Guidelines for the diagnosis and management of pericardial diseases: The Task Force for the Diagnosis and Management of Pericardial Diseases of the European Society of Cardiology (ESC) Endorsed by: The European Association for Cardio-Thoracic Surgery (EACTS) Adler Y, Charron P, Imazio M, et al. Eur Heart J. 2015;36:2921–2964. doi: 10.1093/eurheartj/ehv318. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Myopericarditis versus viral or idiopathic acute pericarditis. Imazio M, Cecchi E, Demichelis B, et al. Heart. 2008;94:498–501. doi: 10.1136/hrt.2006.104067. [DOI] [PubMed] [Google Scholar]
- 7.Preferred reporting items for systematic review and meta-analysis protocols (PRISMA-P) 2015 statement. Moher D, Shamseer L, Clarke M, et al. Syst Rev. 2015;4:1. doi: 10.1186/2046-4053-4-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Conducting systematic reviews of association (etiology): the Joanna Briggs Institute's approach. Moola S, Munn Z, Sears K, et al. Int J Evid Based Healthc. 2015;13:163–169. doi: 10.1097/XEB.0000000000000064. [DOI] [PubMed] [Google Scholar]
- 9.Symptomatic pericardial effusion in the setting of asymptomatic COVID-19 infection: a case report. Amoozgar B, Kaushal V, Mubashar U, Sen S, Yousaf S, Yotsuya M. Medicine (Baltimore) 2020;99:0. doi: 10.1097/MD.0000000000022093. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Acute pericarditis and cardiac tamponade in a patient with COVID-19: a therapeutic challenge. Asif T, Kassab K, Iskander F, Alyousef T. Eur J Case Rep Intern Med. 2020;7:1701. doi: 10.12890/2020_001701. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Acute pericarditis and severe acute respiratory syndrome coronavirus 2: case report. Blagojevic NR, Bosnjakovic D, Vukomanovic V, Arsenovic S, Lazic JS, Tadic M. Int J Infect Dis. 2020;101:180–182. doi: 10.1016/j.ijid.2020.09.1440. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.COVID-19 myopericarditis with cardiac tamponade in the absence of respiratory symptoms: a case report. Cairns L, Abed El Khaleq Y, Storrar W, Scheuermann-Freestone M. J Med Case Rep. 2021;15:31. doi: 10.1186/s13256-020-02618-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.COVID-19 myopericarditis: it should be kept in mind in today's conditions. Cizgici AY, Zencirkiran Agus H, Yildiz M. Am J Emerg Med. 2020;38:1547–1546. doi: 10.1016/j.ajem.2020.04.080. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Acute pericarditis revealing COVID-19 infection: case report. Faraj R, Belkhayat C, Bouchlarhem A, El Aidouni G, Bkiyar H, Housni B. Ann Med Surg (Lond) 2021;62:225–227. doi: 10.1016/j.amsu.2021.01.053. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Acute effusive pericarditis: a late complication of COVID-19. Fox K, Prokup JA, Butson K, Jordan K. Cureus. 2020;12:0. doi: 10.7759/cureus.9074. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.The variety of cardiovascular presentations of COVID-19. Fried JA, Ramasubbu K, Bhatt R, et al. Circulation. 2020;141:1930–1936. doi: 10.1161/CIRCULATIONAHA.120.047164. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Cardiac tamponade in a patient with myocardial infarction and COVID-19: electron microscopy. García-Cruz E, Manzur-Sandoval D, Lazcano-Díaz EA, Soria-Castro E, Jiménez-Becerra S. JACC Case Rep. 2020;2:2021–2023. doi: 10.1016/j.jaccas.2020.07.042. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Cardiac involvement in a patient with coronavirus disease 2019 (COVID-19) Inciardi RM, Lupi L, Zaccone G, et al. JAMA Cardiol. 2020;5:819–824. doi: 10.1001/jamacardio.2020.1096. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Anakinra for the treatment of COVID-19-associated pericarditis: a case report. Karadeniz H, Yamak BA, Özger HS, Sezenöz B, Tufan A, Emmi G. Cardiovasc Drugs Ther. 2020;34:883–885. doi: 10.1007/s10557-020-07044-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Case 18-2020: a 73-year-old man with hypoxemic respiratory failure and cardiac dysfunction. Kazi DS, Martin LM, Litmanovich D, Pinto DS, Clerkin KJ, Zimetbaum PJ, Dudzinski DM. N Engl J Med. 2020;382:2354–2364. doi: 10.1056/NEJMcpc2002417. [DOI] [PubMed] [Google Scholar]
- 21.COVID-19 (SARS-CoV-2) and the heart - an ominous association. Khalid N, Chen Y, Case BC, et al. Cardiovasc Revasc Med. 2020;21:946–949. doi: 10.1016/j.carrev.2020.05.009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Coronavirus disease 2019 (Covid-19) presenting as purulent fulminant myopericarditis and cardiac tamponade: a case report and literature review. Khatri A, Wallach F. Heart Lung. 2020;49:858–863. doi: 10.1016/j.hrtlng.2020.06.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Acute pericarditis as a primary presentation of COVID-19. Kumar R, Kumar J, Daly C, Edroos SA. BMJ Case Rep. 2020;13:237617. doi: 10.1136/bcr-2020-237617. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Myopericarditis, rhabdomyolysis, and acute hepatic injury: sole expression of a SARS-CoV-2 infection. Legrand F, Chong-Nguyen C, Ghanem N. Circ Cardiovasc Imaging. 2020;13:0. doi: 10.1161/CIRCIMAGING.120.010907. [DOI] [PubMed] [Google Scholar]
- 25.Management of COVID-19 myopericarditis with reversal of cardiac dysfunction after blunting of cytokine storm: a case report. Li A, Garcia-Bengochea Y, Stechel R, Azari BM. Eur Heart J Case Rep. 2020;4:1–6. doi: 10.1093/ehjcr/ytaa224. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Acute pericarditis secondary to COVID-19. Marschall A, Concepción Suárez R, Dejuan Bitriá C, Fernández Pascual MC. https://pubmed.ncbi.nlm.nih.gov/32395943/ Emergencias. 2020;32:221–222. [PubMed] [Google Scholar]
- 27.Acute pericarditis and pericardial effusion in a hypertensive COVID-19 patient. Naqvi SG, Naseeb U, Fatima K, Riffat S, Memon AG. Cureus. 2020;12:0. doi: 10.7759/cureus.10705. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Pericarditis in a young internal medicine resident with COVID-19 in Colombia. Ortiz-Martínez Y, Cabeza-Ruiz LD, Vásquez-Lozano SH, Villamil-Gómez WE, Rodriguez-Morales AJ. Travel Med Infect Dis. 2020;37:101863. doi: 10.1016/j.tmaid.2020.101863. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Myopericarditis caused by severe acute respiratory syndrome coronavirus 2. Özturan İU, Köse B, Özkan B, Köse A. Clin Exp Emerg Med. 2020;7:326–329. doi: 10.15441/ceem.20.109. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Acute pericarditis in a patient with severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) infection: a case report and review of the literature on SARS-CoV-2 cardiological manifestations. Patel VD, Patel KH, Lakhani DA, Desai R, Mehta D, Mody P, Pruthi S. AME Case Rep. 2021;5:6. doi: 10.21037/acr-20-90. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Acute myopericarditis with pericardial effusion and cardiac tamponade in a patient with COVID-19. Purohit R, Kanwal A, Pandit A, et al. Am J Case Rep. 2020;21:0. doi: 10.12659/AJCR.925554. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Pediatric COVID-19 and pericarditis presenting with acute pericardial tamponade. Raymond TT, Das A, Manzuri S, Ehrett S, Guleserian K, Brenes J. World J Pediatr Congenit Heart Surg. 2020;11:802–804. doi: 10.1177/2150135120949455. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Colchicin treatment of COVID-19 presenting with cutaneous rash and myopericarditis. Recalcati S, Piconi S, Franzetti M, Barbagallo T, Prestinari F, Fantini F. Dermatol Ther. 2020;33:0. doi: 10.1111/dth.13891. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Rescue venoarterial extracorporeal membrane oxygenation after cardiac arrest in COVID-19 myopericarditis: a case report. Sampaio PP, Ferreira RM, de Albuquerque FN, et al. Cardiovasc Revasc Med. 2021;28S:57–60. doi: 10.1016/j.carrev.2020.09.038. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Pericarditis secundaria a infección por COVID-19 en un paciente trasplantado renal. Sandino Pérez J, Aubert Girbal L, Caravaca-Fontán F, Polanco N, Sevillano Prieto Á, Andrés A. Nefrologia. 2020;41:349–352. doi: 10.1016/j.nefro.2020.07.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Pericardial effusion in patients with COVID-19: case series. Sauer F, Dagrenat C, Couppie P, Jochum G, Leddet P. Eur Heart J Case Rep. 2020;4:1–7. doi: 10.1093/ehjcr/ytaa287. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Myocarditis and pericarditis in patients with COVID-19. Shah JZ, Kumar SA, Patel AA. Heart Views. 2020;21:209–214. doi: 10.4103/HEARTVIEWS.HEARTVIEWS_154_20. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.A rare case of acute pericarditis due to SARS-CoV-2 managed with aspirin and colchicine. Thrupthi K, Ganti A, Acherjee T, Mehmood MA, Vakde T. Cureus. 2021;13:0. doi: 10.7759/cureus.12534. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.A rare case of COVID-19 myocarditis with cardiac tamponade in a young diabetic adult with renal failure. Tiwary T, Baiswar S, Jinnur P. Cureus. 2020;12:0. doi: 10.7759/cureus.11632. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Acute pericarditis due to COVID-19 infection: an underdiagnosed disease? Tung-Chen Y. Med Clin (Engl Ed) 2020;155:44–45. doi: 10.1016/j.medcle.2020.06.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Pericardial effusion and cardiac tamponade requiring pericardial window in an otherwise healthy 30-year-old patient with COVID-19: a case report. Walker C, Peyko V, Farrell C, Awad-Spirtos J, Adamo M, Scrocco J. J Med Case Rep. 2020;14:158. doi: 10.1186/s13256-020-02467-w. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Right ventricular myopericarditis in COVID-19: a call for regular echocardiography. Rauch S, Regli IB, Clara A, Seraglio PM, Bock M, Poschenrieder F, Resch M. Minerva Anestesiol. 2020;86:1253–1254. doi: 10.23736/S0375-9393.20.14756-4. [DOI] [PubMed] [Google Scholar]
- 43.Pathophysiological characteristics and therapeutic approaches for pulmonary injury and cardiovascular complications of coronavirus disease 2019. Geng YJ, Wei ZY, Qian HY, Huang J, Lodato R, Castriotta RJ. Cardiovasc Pathol. 2020;47:107228. doi: 10.1016/j.carpath.2020.107228. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Evaluation and treatment of pericarditis: a systematic review. Imazio M, Gaita F, LeWinter M. JAMA. 2015;314:1498–1506. doi: 10.1001/jama.2015.12763. [DOI] [PubMed] [Google Scholar]
- 45.Indicators of poor prognosis of acute pericarditis. Imazio M, Cecchi E, Demichelis B, et al. Circulation. 2007;115:2739–2744. doi: 10.1161/CIRCULATIONAHA.106.662114. [DOI] [PubMed] [Google Scholar]
- 46.Thromboinflammatory biomarkers in COVID-19: systematic review and meta-analysis of 17,052 patients. Chaudhary R, Garg J, Houghton DE, et al. Mayo Clin Proc Innov Qual Outcomes. 2021;5:388–402. doi: 10.1016/j.mayocpiqo.2021.01.009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Corticosteroids for recurrent pericarditis: high versus low doses: a nonrandomized observation. Imazio M, Brucato A, Cumetti D, et al. Circulation. 2008;118:667–671. doi: 10.1161/CIRCULATIONAHA.107.761064. [DOI] [PubMed] [Google Scholar]
- 48.Prognosis of myopericarditis as determined from previously published reports. Imazio M, Brucato A, Spodick DH, Adler Y. J Cardiovasc Med (Hagerstown) 2014;15:835–839. doi: 10.2459/JCM.0000000000000082. [DOI] [PubMed] [Google Scholar]
- 49.Systematic review of non-invasive cardiovascular imaging in the diagnosis of constrictive pericarditis. Ardhanari S, Yarlagadda B, Parikh V, Dellsperger KC, Chockalingam A, Balla S, Kumar S. Indian Heart J. 2017;69:57–67. doi: 10.1016/j.ihj.2016.06.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Quantifying and reporting cardiac findings in imaging of COVID-19 patients. Mishra AK, Lal A, Sahu KK, Kranis M, Sargent J. Monaldi Arch Chest Dis. 2020;90 doi: 10.4081/monaldi.2020.1394. [DOI] [PubMed] [Google Scholar]
- 51.Clinical practice. Acute pericarditis. LeWinter MM. N Engl J Med. 2014;371:2410–2416. doi: 10.1056/NEJMcp1404070. [DOI] [PubMed] [Google Scholar]
- 52.Current and emerging strategies for the treatment of acute pericarditis: a systematic review. Sheth S, Wang DD, Kasapis C. J Inflamm Res. 2010;3:135–142. doi: 10.2147/JIR.S10268. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.The effective treatment of postpericardiotomy syndrome after cardiac operations. A randomized placebo-controlled trial. Horneffer PJ, Miller RH, Pearson TA, Rykiel MF, Reitz BA, Gardner TJ. J Thorac Cardiovasc Surg. 1990;100:292–296. [PubMed] [Google Scholar]
- 54.The use of colchicine in cardiovascular diseases: a systematic review. Siak J, Flint N, Shmueli HG, Siegel RJ, Rader F. Am J Med. 2021;134:735–744. doi: 10.1016/j.amjmed.2021.01.019. [DOI] [PubMed] [Google Scholar]
- 55.Meta-analysis for the value of colchicine for the therapy of pericarditis and of postpericardiotomy syndrome. Lutschinger LL, Rigopoulos AG, Schlattmann P, Matiakis M, Sedding D, Schulze PC, Noutsias M. BMC Cardiovasc Disord. 2019;19:207. doi: 10.1186/s12872-019-1190-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Colchicine in cardiac disease: a systematic review and meta-analysis of randomized controlled trials. Verma S, Eikelboom JW, Nidorf SM, Al-Omran M, Gupta N, Teoh H, Friedrich JO. BMC Cardiovasc Disord. 2015;15:96. doi: 10.1186/s12872-015-0068-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.A randomized trial of colchicine for acute pericarditis. Imazio M, Brucato A, Cemin R, et al. N Engl J Med. 2013;369:1522–1528. doi: 10.1056/NEJMoa1208536. [DOI] [PubMed] [Google Scholar]
- 58.Colchicine in addition to conventional therapy for acute pericarditis: results of the COlchicine for acute PEricarditis (COPE) trial. Imazio M, Bobbio M, Cecchi E, et al. Circulation. 2005;112:2012–2016. doi: 10.1161/CIRCULATIONAHA.105.542738. [DOI] [PubMed] [Google Scholar]