Abstract
Stroke is a fatal cerebral vascular disease with a high mortality rate and substantial economic and social costs. ROS production and neuroinflammation have been implicated in both hemorrhagic and ischemic stroke and have the most critical effects on subsequent brain injury. SIRT1, a member of the sirtuin family, plays a crucial role in modulating a wide range of physiological processes, including apoptosis, DNA repair, inflammatory response, and oxidative stress. Targeting SIRT1 to reduce ROS and neuroinflammation might represent an emerging therapeutic target for stroke. Therefore, we conducted the present review to summarize the mechanisms of SIRT1-mediated oxidative stress and neuroinflammation in stroke. In addition, we provide a comprehensive introduction to the effect of compounds and natural drugs on SIRT1 signaling related to oxidative stress and neuroinflammation in stroke. We believe that our work will be helpful to further understand the critical role of the SIRT1 signaling pathway and will provide novel therapeutic potential for stroke treatment.
1. Introduction
Stroke is a type of fatal cerebral vascular disease with a high mortality rate [1], the second leading cause of death, and the leading cause of disability worldwide [2, 3]. As a result of stroke, a substantial economic and social burden is imposed [4]. Stroke can be classified as either an ischemic stroke or a hemorrhagic stroke [5, 6]. Furthermore, hemorrhagic stroke may be divided into intracerebral hemorrhages (ICHs) and subarachnoid hemorrhages (SAHs). In the event of hemorrhage and ischemia, overproduction of reactive oxygen species (ROS) and neuroinflammation have been linked to subsequent brain injury [7, 8, 9]. ROS overproduction is closely related to neuroinflammation, with each promoting and amplifying the other. After a stroke, mitochondria overproduce ROS, which then causes a wide array of destructive effects on cellular functions by damaging proteins, lipids, and nucleic acids [10, 11, 12, 13]. In addition, cell death pathways can be signaled by ROS as intracellular messengers [14]. Additionally, microglia and astrocytes are activated within hours of a stroke, releasing cytokines and chemokines in addition to leukocyte infiltration [15, 16, 17]. Reducing ROS and neuroinflammation has long been a focus of stroke treatment research. New therapies that reduce oxidative damage and inflammation after stroke could improve neurological recovery.
Silent mating type information regulation 2 homolog 1 (SIRT1), also known as sirtuin 1, is an NAD-dependent deacetylase located in the nucleus [18]. SIRT1 has been proven to play a critical role in modulating a wide range of physiological processes, including apoptosis, DNA repair, inflammatory response, metabolism, cancer, and stress [19, 20]. Previous studies have demonstrated that SIRT1 can protect against oxidative stress and inflammation in various research areas, including ischemic reperfusion-induced renal, brain, and heart injuries [21, 22, 23]. In recent years, SIRT1 has been recognized as particularly important in the pathogenesis of stroke [24, 25, 26]. SIRT1 modulates oxidative stress and inflammation through a variety of signaling pathways. Numerous studies have demonstrated that SIRT1 can protect neurons against oxidative and inflammatory damage induced by FOXOs, nuclear factor-κB (NF-κB), and p53 [25, 27, 28]. Reducing ROS and neuroinflammation by targeting SIRT1 may represent a promising therapeutic target for stroke.
During ischemic stroke, brain tissue is hypoxic due to vascular obstruction. The only FDA-approved treatment for ischemic stroke is tissue plasminogen activator (tPA) [29]. However, the limited therapeutic window of this drug is a major concern, as it can cause secondary damage that is ischemia–reperfusion injury [30, 31]. Despite significant progress in the clinical treatment of the disease, there has not been a medical or surgical therapy to improve outcomes for patients with hemorrhagic stroke. Surgical intervention in hemorrhagic strokes is still controversial. As a result, it is crucial to define the pathogenesis and targets for the prevention and treatment of stroke. In addition, we discuss the potential mechanisms and natural compounds used for stroke treatment by which SIRT1 protects against stroke through antioxidative and anti-inflammatory effects.
The following search terms were used: SIRT1, oxidation, neuroinflammation, and stroke. Once we found the target article, we continued our search in the similar articles section of PubMed. Then, we screened studies in English that investigated interventions for natural compounds in stroke. This review summarized the potential mechanisms and natural products used in stroke by which Sirt1 protects against stroke through antioxidative and anti-inflammatory effects.
2. Activation Mechanism
2.1. SIRT1-Mediated Antioxidation in Stroke
ROS are a class of oxygen-containing reactive chemicals that play essential roles in normal physiological processes, for instance, in controlling redox regulation of protein phosphorylation, ion channels, and transcription factors [32]. Typically, the primary intracellular source of ROS in mammalian cells is thought to be mitochondria [33, 34]. However, a high concentration of ROS can cause irreversible changes in protein, lipid, carbohydrate, and nucleic acid structures [35]. ROS has been proved to play a vital role in developing neuronal damage after a stroke [36, 37].
During ischemic injury, oxidative stress is one of the earliest outcomes, causing cascades of cellular and molecular processes that cause neurodegeneration and death of neurons [38]. Increased levels of ROS in cells, such as hydroxyl radicals, can result in oxidative stress and mitochondrial dysfunction, which can lead to cerebral ischemia and further aggravate the cerebral injury [39, 40].
SIRT1 is the most extensively studied sirtuin and is expressed in the brain at a higher level than other organs [41]. According to a previous study, mice overexpressing SIRT1 sustained less hippocampal damage following cerebral ischemia (bilateral common carotid artery occlusion) than mice lacking SIRT1, demonstrating that SIRT1 plays a prominent role in brain protection [42]. In addition, SIRT1 has been identified as playing an essential role in oxidative stress [41, 43]. SIRT1 is activated after the onset of stroke and can regulate multiple signaling pathways to affect oxidative stress, further regulating the pathological process of stroke. This section summarizes the critical signaling pathway proteins in the process of SIRT1-mediated antioxidation in stroke. The signaling pathway of SIRT1-mediated antioxidation in stroke is shown in Figure 1.
Figure 1.
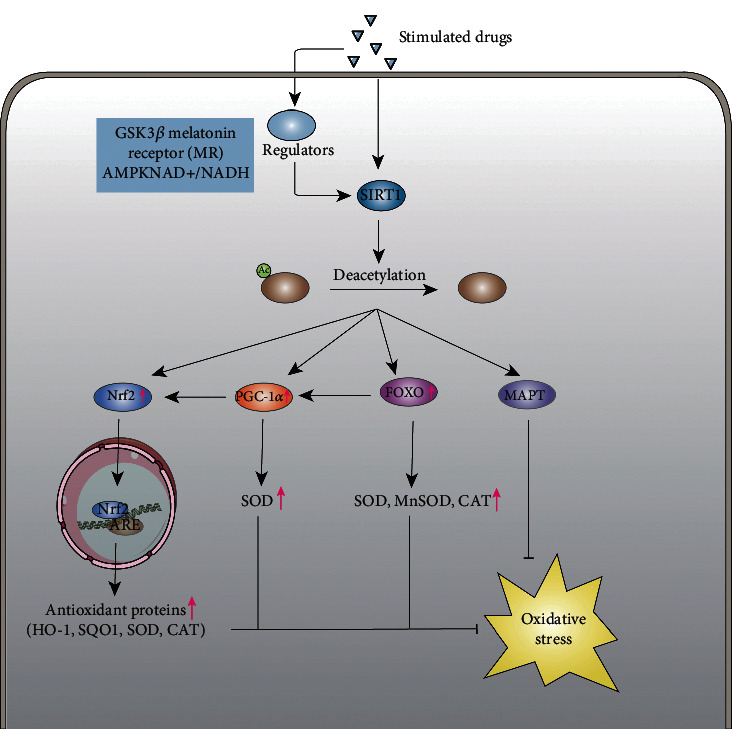
The signal pathway of SIRT1-mediated antioxidation in stroke. The picture shows the signal pathway of SIRT1-mediated antioxidation in stroke. In response to oxidative stress after stroke, SIRT1 mainly mediates four molecular modifications, including Nrf2, FOXO, PGC-1α, and MAPT. By deacetylating the major acetylation sites, the level of FOXO, PGC-1α, and Nrf2 is upregulated by SIRT1, while MAPT is downregulated. When the level of FOXO, PGC-1α, and Nrf2 increases, several kinds of antioxidant proteins also correspondingly increase, which further suppress oxidative stress. In addition, the accumulation of abnormal MAPT will be reduced through deacetylating MAPT, thus reducing the damaging effect on cells. Therefore, some drugs or upstream regulators can inhibit oxidative stress by acting on SIRT1, which further exerts neuroprotective effects.
2.1.1. PGC-1α
According to previous research, peroxisome proliferator-activated receptor gamma coactivator 1-α (PGC-1α) functions as a master regulator of mitochondrial function and biogenesis, such as oxidative phosphorylation (OXPHOS) and ROS detoxification, contributing to maintaining metabolic homeostasis [44]. The expression of PGC-1α is high in tissues with active oxidative metabolism, especially in the brain [45]. PGC1-α, mainly by upregulating oxidation defense, is critical in preventing cell death due to mitochondrial dysfunction [46]. To prevent oxidative stress-induced cell damage, the expression of PGC-1α is positively regulated by oxidative stress [47]. In addition to exerting a pivotal role in preventing oxidative damage, the upregulation of PGC-1α-antioxidant target genes also significantly decreased mitochondrial ROS levels and ensured mitochondrial integrity [48]. Synthesizing ATP in mitochondria inevitably leads to electrons leaking into oxygen, which eventually results in ROS formation [49]. By activating several transcription factors, including NRF, Tfam, ERRα, and PPARs, PGC-1α increases mitochondrial biogenesis and the function of genes [50]. Additionally, PGC-1α is involved in regulating mitochondrial antioxidant defenses. Manganese superoxide dismutase (MnSOD/SOD2), peroxiredoxin (Prx) 5, Prx3, thioredoxin reductase (TRXR) 2, UCP-2, thioredoxin (TRX) 2, and catalase are increased by PGC-1α and consequently prevent mitochondrial dysfunction in cells [46]. An increase in PGC-1α expression is related to preventing oxidative damage by cells when exposed to oxidative stress [47]. Mouse experiments confirmed that mice lacking PGC-1α are more susceptible to oxidative damage [51].
Regulation of PGC-1α occurs at the transcriptional and posttranslational levels. The significant transcription factors are FoxO1 and FoxO3, which regulate the expression of the PGC-1α gene in a tissue-dependent way [52]. More importantly, PGC-1α, a downstream effector of SIRT1, plays a neuroprotective role [53]. Numerous studies have shown that when SIRT1 is activated, it can deacetylate downstream PGC-1α, which exerts antioxidative and anti-inflammatory effects [53, 54]. As one of the targets of SIRT1, PGC-1α suppresses ROS production by inducing ROS detoxifying enzymes [55]. Thus, neurons are substantially protected against oxidative stress by increasing SIRT1-mediated PGC-1α levels [55]. The NAD+/NADH ratio measures the redox state of cells [56]. As NAD+/NADH levels rise, SIRT1 becomes activated, deacetylating PGC-1α [57].
An experimental study based on a cerebral ischemia model showed that SIRT1, FOXO1, PGC-1α, Bax, and Bcl-2 protein expressions were significantly reduced after oxygen-glucose deprivation/reperfusion (OGD/R) [58]. Calycosin-7-O-β-D-glucoside (CG), a representative drug, downregulated the expression of Bax, which further suggests that CG protects against ischemic stroke by activating the SIRT1/FOXO1/PGC-1α signaling pathway. In addition, many other drugs have been shown to exert neuroprotective effects by upregulating SIRT1-dependent PGC-1α expression to induce antioxidation in stroke. In previous studies, SIRT1/PGC-1α signaling was shown to enhance SOD levels, remove MDA from the cell, and decrease oxidative stress, leading to cell death resistance [59].
2.1.2. Forkhead Transcription Factors of the O Class (FOXO)
The mammalian FOXO have four members, including FOXO1, FOXO3, FOXO4, and FOXO6 [60]. Several processes in the body are controlled by FOXO, including the cell cycle, metabolism, stress resistance, and inflammation [61, 62]. Among them, FOXO3 has been demonstrated to play an essential role in the regulation of oxidative stress, which can upregulate the expression of several antioxidant proteins, including superoxide dismutase (SOD), manganese superoxide dismutase (MnSOD), and catalase (CAT) [63, 64, 65, 66]. FOXO3 can be phosphorylated and deacetylated to regulate its transcriptional activity [67].
In addition, it has been shown that SIRT1 deacetylates histone and nonhistone proteins, including FOXO [68]. SIRT1 increases cellular resistance against oxidative stress by deacetylating and activating FOXO, which promotes the expression of antioxidant proteins such as SOD [69]. Deacetylation of FOXO3 by SIRT1 can prevent cell death induced by FOXO3 [69]. In response to stimulation, FOXO3a phosphorylates and binds to 14-3-3 proteins in the nucleus. This inhibits FOXO3a-dependent transcription [70]. Cells can resist oxidative stress and induce cycle arrest by deacetylating FOXO3 with SIRT1. Wu et al. found that the lncRNA SNHG12 inhibits the oxidative stress response of the ischemia/reperfusion (I/R) model by activating the SIRT1/FOXO3 pathway [71]. PGC-1α transcription is also regulated by FoxO transcription factors in different cell types. The FoxO3 signaling molecule interacts directly with PGC-1α in vascular endothelial cells and protects mitochondria from oxidative stress through a mechanism controlled by activation of SIRT1 [72]. When FoxO3 is inactivated and PGC-1α is downregulated, ROS detoxification is hampered [73]. FoxO3 can be inactivated by the PI3K/Akt signaling pathway to further decrease endothelial PGC-1α expression [69]. In the research of Duan et al., Akt/SIRT1/FOXO3a/PGC-1α pathways are shown to be regulated by saponin from Aralia taibaiensis (sAT), protecting brain cells from ischemia and reperfusion- (I/R-) induced mitochondrial dysfunction [74].
2.1.3. MAPT
Neurons of the central nervous system are the main site of microtubule-associated protein tau (MAPT) distribution, which plays an essential role in morphogenesis, axonal transport, and axonal extension by interfering with assemblage of microtubules and stabilization of the microtubule network [75, 76]. Posttranslationally modified MAPT facilitates tubulin binding, microtubule stability, and neuronal morphology [75]. Nevertheless, if MAPT is excessively phosphorylated, the microtubule binding capacity will be reduced. As microtubules are reduced in binding capacity, the neuronal cytoskeleton becomes less stable, resulting in neuronal death [77]. Research on MAPT is mainly conducted in neurodegenerative disease pathogenesis [78]. Aggregation and hyperphosphorylation of the MAPT proteins in the brain are pathological features that exist in a large group of neurodegenerative conditions, which are named tauopathies. In addition, oxidative stress (OS) is another crucial aspect of tauopathies' pathophysiology [78].
In recent years, MAPT has gained wide attention for its potential role in stroke. Researchers have found that brain ischemia/reperfusion (CIR) animal models have high abnormally hyperphosphorylated MAPT, which is closely linked to neurological deficits and neuronal apoptosis [79]. In the research of Fujii et al., an MCAO model showed abnormally high phosphorylation of MAPT 12 hours after CIR in Wistar rats [80]. Knockout of MAPT prevented brain damage in mice after MCAO-induced stroke [81]. Several studies have demonstrated that hyperacetylation of MAPT promotes the accumulation of phosphorylated MAPT, harming cells by accumulating abnormal MAPT [77, 82]. In recent years, SIRT1 has been shown to be closely associated with MAPT modification [83, 84]. Some specific lysine residues in MAPT can be deacetylated by SIRT1, which means that a pivotal role is played by SIRT1/MAPT pathway during stroke recovery [83]. Shi et al. proved that inhibition of Sirt1/MAPT signaling by astragaloside IV (AS-IV) protects rats from cerebral ischemia/reperfusion injury. By upregulating SIRT1 expression, AS-IV decreases acetylated MAPT (ac-MAPT) and phosphorylated MAPT (p-MAPT) levels, ultimately reducing infarction size and improving neurological function [85]. However, there is no direct evidence to prove that the Sirt1/MAPT pathway plays a role in stroke directly through antioxidant responses.
2.1.4. Nuclear Factor Erythroid 2-Related Factor 2 (Nrf2)
Nrf2, a basic leucine zipper protein (bZIP), is important for maintaining redox homeostasis in cells. Nrf2 is a pivotal regulator of endogenous antioxidant defenses [86], which can activate the transcription of its downstream antioxidant genes, including heme oxygenase-1 (HO-1), quinone oxidoreductase-1 (Nqo1), superoxide dismutase (SOD), catalase (CAT), and other phase II antioxidant enzymes [87, 88]. Typically, Kelch-like ECH-associated protein 1 (Keap1) retains Nrf2 in the cytoplasm. The Nrf2 protein translocates into the nucleus upon oxidative stress, binding to the antioxidant response element (ARE) and promoting antioxidant enzyme synthesis and phase II detoxification enzyme synthesis. According to recent research, neurons are protected against ischemic stroke injury by Nrf2. Knockout of the Nrf2 gene enormously increased the cerebral infarcted region and neurologic deficits in ischemia–reperfusion rats [89]. The Nrf2-target gene pathway is protective against ischemia/reperfusion injury in the brain. Increased Nrf2 activity can reduce the oxidative stress that occurs during stroke and alleviate brain injury.
Numerous regulators also control the activation of Nrf2, including SIRT1. A growing body of evidence indicates that SIRT1 plays a crucial role in regulating Nrf2. SIRT1 can deacetylate Nrf2 to increase downstream antioxidase expression. Mei et al. proved that a neuroprotective effect of diosmetin was observed both in vitro and in vivo against cerebral ischemia/reperfusion, which was mediated by inhibiting oxidative stress by regulating the SIRT1/Nrf2 signaling pathway [90]. Diosmetin induces the expression of SIRT1 and N-Nrf2, and T-Nrf2 promotes Nrf2 translocation to the nucleus and increases the expression of the downstream antioxidants NQO1 and HO-1.
2.2. SIRT1-Mediated Anti-inflammatory in Stroke
Inflammation plays a crucial role in stroke pathophysiology and is being targeted for stroke treatment. In human stroke cases, inflammatory cells infiltrate the brain within minutes to days after ischemic insult [91, 92], which further leads to the release of a large number of cytokines and chemokines [93]. Microglia are resident macrophages in the central nervous system. When they detect ischemia, they initiate a response immediately [94, 95, 96]. Microglial activation in the peri-infarct zone within 30 minutes to 1 hour after MCAO is accompanied by the appearance of CD11b, CD45, and Iba1 [97, 98]. At the same time, CD11b+ microglia within the infarct start fragmentation 12 hours after MCAO, and the number of microglia is reduced 24 hours later [98, 99]. There is persistent activation of microglial cells in the peri-infarct weeks after MCAO [100]. Apart from the above, infiltrating leukocytes, including polymorphonuclear leukocytes, monocytes/macrophages, and lymphocytes, also play crucial roles in ischemic stroke [101, 102, 103, 104]. Strokes caused by an ischemic stroke can induce the release of inflammatory cytokines, including IL-1β and IL-18 [105, 106]. An increase in inflammatory cytokines and neuroinflammation accompanies cerebral ischemia, aggravating neurodegeneration [107, 108]. Research suggests that interventions meant to reduce inflammation after cerebral ischemia can prevent brain damage from worsening.
SIRT1 is thought to play a critical role in brain protection. Several signaling pathways can be modulated by SIRT1 upon activation to affect neuroinflammation. SIRT1 plays a critical role in neuroprotection against brain ischemia through deacetylation and subsequent inhibition of p53 and nuclear factor-κB-induced inflammatory and apoptotic pathways [25]. Some drugs acting on SIRT1 have shown practical neuroprotective function due to their antioxidative stress and anti-inflammatory properties. In an experiment using the middle cerebral artery occlusion (MCAO) model, salvianolic acid B (SalB), which has been proven protective against ischemic stroke in previous studies, reduces brain injury induced by ischemic stroke by activating SIRT1 to reduce apoptosis and inflammation [109]. Notably, TNF-α and IL-1β levels in brain tissue decreased following treatment with SalB. Ischemic stroke is accompanied by the production and release of proinflammatory cytokines (mainly TNF-α and IL-1β), as previously demonstrated [110]. In addition, SMND-309, a novel derivative of SalB, has been proven to inhibit reperfusion injury in rat brains by targeting the JAK2/STAT3 [111]. Kou et al. found that magnolol, by modulating the expression of SIRT1, exerts anti-inflammatory effects by decreasing the expression of IL-1β and TNF-α in brain tissue to protect the brain against cerebral ischemic injury [112]. Additionally, magnolol causes a downregulation of Ac-FOXO1, induced by the SIRT1 expression, which means that magnolol can also exert a protective effect against ischemic stroke via resistance function against oxidative stress. The SIRT1-mediated anti-inflammatory signaling pathway in stroke is shown in Figure 2.
Figure 2.
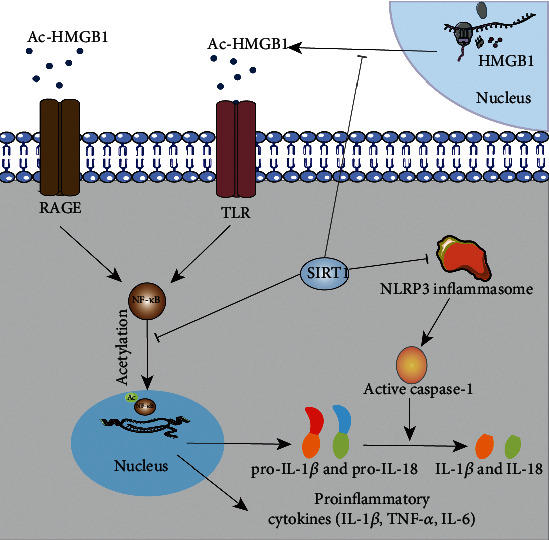
The signal pathway of SIRT1-mediated anti-inflammatory in stroke. The picture shows the signal pathway of SIRT1-mediated anti-inflammatory in stroke. SIRT1 can inhibit the production of proinflammatory factors through the deacetylation of HMGB1, NF-κB, and NLRP3 inflammasome, thus achieving an anti-neuroinflammatory effect. Through the deacetylation of HMGB1 and NF-κB, the synthesis of some proinflammatory cytokines such as TNF-α, IL-6, and IL-1β in the nucleus was inhibited. In addition, through the deacetylation of the NLRP3 inflammasome, the function of caspase-1 was restricted, and eventually, the pathway of conversion of pro-IL-1β and pro-IL-18 to IL-1β and IL-18 was blocked.
2.2.1. Nuclear Factor-κB (NF-κB)
The NF-κB protein complex is an inducible transcription factor family that consists of five members with correlated structures, including NF-κB1 (p50), NF-κB2 (p52), RelA (p65), RelB, and c-Rel [113]. NF-κB is widely considered a prototypical pathway that initiates inflammation, in which NF-κB regulates inflammation and immunity in various ways [114]. NF-κB is known to stimulate the expression of several proinflammatory genes, such as those that encode cytokines and chemokines, as well as those that regulate inflammasome activity [114, 115]. Typically, two signaling pathways, canonical and noncanonical (or alternative), are involved in activating NF-κB [116]. Among them, proinflammatory cytokines such as TNF-α and IL-1 activate the canonical pathway, activating RelA- or cRel-containing complexes [117]. The noncanonical pathway is triggered by several TNF family cytokines (excluding TNF-α), leading to RelB/p52 activation [118]. Both pathways are important for immune and inflammatory response regulation. When molecules such as TNF-α bind to TNF receptors of different types, NF-κB is activated, which further leads to subsequent activation processes [119]. To develop effective therapeutic strategies for inflammation-associated illnesses, a better understanding of the mechanisms that underlie NF-κB activation and proinflammatory function is crucial.
Previous work has demonstrated that neuroinflammation mediated by NF-κB contributes significantly to stroke-induced neurotoxicity [120, 121]. Several diseases are prevented by the anti-inflammatory action of SIRT1, which acts as a negative regulator of inflammation [122]. Notably, there has been extensive research on SIRT1 because it is abundantly present in brain tissue and plays a vital role in the central nervous system [123]. The interaction between SIRT1 and the RelA/p65 subunit of NF-κB plays a key role in this process: RelA/p65 is negatively affected by acetylation of lysine 310 by SIRT1, resulting in decreased transcriptional activity and expression of proinflammatory genes [124]. SIRT1 deacetylation of RelA/p65 promotes the p65/p50 complex's interaction with IκB-α. Through this association, the NF-B complex is transported back from the nucleus to the cytoplasm, thereby inactivating its activity. In a study by Deng et al., the activation of SIRT1 protected the brain after intracerebral hemorrhage by deacetylating NF-κB/p65 [120]. These results suggest that inhibition of the NF-κB inflammatory signaling pathway by activating SIRT1 can effectively protect brain tissue after stroke.
2.2.2. High Mobility Group Box 1 (HMGB1)
HMGB1 is a protein in eukaryotic cells that repairs DNA damage and maintains genomic stability [125]. HMGB1, as a typical DAMP, induces inflammatory responses in the innate immune system due to injury or stress [126]. Inflammasome activation releases HMGB1 from glia and neurons, which activates receptors for advanced glycation end products (RAGE) and Toll-like receptor 4 (TLR-4) on target cells. An inflammatory response mediated by HMGB1 plays a role in various conditions, such as ischemia [127]. HMGB1 contains several extracellular receptors, with RAGE and TLR4 having been extensively studied and demonstrated to be true fields [128]. In addition to initiating several cellular responses, including inflammation, HMGB1 also participates in the activation of inflammation by binding with RAGE and TLR4 [129]. In neurons and glial cells, HMGB1 is actively released following activation of the inflammasome. This stimulates the activation of two PRRs, namely, TLR4 and RAGE. In previous studies, SIRT1 was shown to play a vital role in activating HMGB1/NF-κB [130]. Various proinflammatory cytokines and chemokines are known to be induced by TLR4 activation, including TNF-α, IL-6, and IL-1β. Following SAH, proinflammatory cytokines can damage the surrounding neural cells and activate leukocytes, aggravating brain damage [131, 132, 133]. For HMGB1 to enter the extracellular environment and relocate to the cytoplasm, posttranslational modifications such as acetylation are critical. As HMGB1 relocates, hyperacetylation plays a critical role [134]. However, HMGB1 release is negatively regulated by SIRT1 [135]. HMGB1 is deacetylated by SIRT1 in vivo, thereby inhibiting the release of HMGB1 and the subsequent activation of inflammation [136].
2.2.3. NLRP3
The NLRP3 inflammasome is part of the NLR family, which consists of NLRP3, apoptosis-associated speck-like (ASC) adapter protein, and procaspase-1 [137]. NLRP3 inflammasomes are expressed in microglia, astrocytes, and neurons [138]. By regulating IL-1β and IL-18 secretion, they play a role in ischemic stroke [139]. Some studies have found that NLRP3 is upregulated in ischemic brains and is expressed in microglia and endothelial cells, indicating that these cells are major sources of NLRP3 [139]. Priming signals induced by the Toll-like receptor (TLR)/nuclear factor NF-κB pathway influence the transcription of NLRP3 [140]. The expression of pro-IL-1β and pro-IL-18 is increased by NF-kB stimuli, while the NLRP3 inflammasome activates caspase-1, which transforms inactive pro-IL-1β and pro-IL-18 into their active and secreted forms: IL-1β and IL-18. By initiating or amplifying downstream signaling pathways and driving proinflammatory responses, these cytokines cause cellular damage [141]. A previous study indicated that a lack of the NLRP3 inflammasome might enable rats to recover from cerebral injury after ischemic stroke by reducing infarcts and inflammatory responses [139]. Furthermore, neuronal cell death and behavioral deficits can be improved by inhibiting NLRP3 inflammasome activation in stroke models [139]. Importantly, several studies have confirmed that SIRT1 plays a key role in neuroprotection by inhibiting the activation of NLRP3 inflammasomes after ischemic stroke [142].
3. Therapeutic Effects of Natural Products Acting on SIRT1 in Stroke
Natural products are chemical compounds or substances produced by plants and microorganisms. From a particular perspective, natural products include everything produced by living things [143, 144]. There is high potential in developing new stroke treatments based on natural products [143, 145]. A natural product is typically seen as an environmentally sound, readily available resource with few adverse effects in the field [122] which makes them more clinically useful. In addition, SIRT1 plays an important role in stroke pathogenesis via its ability to modulate oxidative stress and inflammation [25]. This section is aimed at summarizing the natural products acting on SIRT1 for stroke treatment. The natural products working on SIRT1 in hemorrhagic and ischemic strokes are shown in Tables 1 and 2, respectively.
Table 1.
The natural products acting on SIRT1 in hemorrhagic stroke.
| Studied drugs | Mechanisms | Classes of action | Animal models | Reference |
|---|---|---|---|---|
| HLY78 | GSK3β/Sirt1/PGC-1α pathway | Antioxidation | ICH model (induced via autologous blood injection) | Jin et al. [148] |
| Resveratrol | NF-κB/p65 deacetylating | Anti-neuroinflammation | ICH model (intracranial injection of type IV collagenase) | Deng et al. [120] |
| Fucoxanthin (Fx) | Activating Sirt1 and further deacetylating FOXO and p53 | Antioxidation | SAH model (prechiasmatic cistern injection models) | Zhang et al. [150] |
| Salvianolic acid B (SalB) | Activating SIRT1 and Nrf2 signaling pathway | Antioxidation | SAH model (single blood injection) | Zhang et al. [153] |
| Melatonin (Mel) | Activating MR/Sirt1/NF-κB pathway | Anti-neuroinflammation and antioxidation | SAH model (endovascular perforation model) | Zhao et al. [158] |
| Berberine | Activating sirtuin 1 and suppressing HMGB1/NF-κB pathway | Anti-neuroinflammation | SAH model (prechiasmatic cistern injection models) | Zhang et al. [161] |
| Oleanolic acid | SIRT1-mediated HMGB1 deacetylation | Anti-neuroinflammation | SAH model (endovascular perforation model) | Han et al. [162] |
| Carnosic acid (CA) | Activating SIRT1/p66shc pathway | Antioxidation | SAH model (endovascular perforation model) | Teng et al. [164] |
| Wogonoside | Activating SIRT1 and further deregulating p53 | Anti-neuroinflammation | SAH model (endovascular perforation model) | Cheng et al. [166] |
Table 2.
The natural products acting on SIRT1 in hemorrhagic stroke.
| Studied drugs | Mechanisms | Classes of action | Animal models | Reference |
|---|---|---|---|---|
| Adiponectin (APN) | Activating SIRT1/PGC-1α | Antioxidation | Glutamate- (Glu-) induced excitotoxicity in mouse HT22 hippocampal cells | Yue et al. [53] |
| Salvianolic acid B (SalB) | Downregulation of Ac-FOXO1 | Anti-neuroinflammation and antioxidation | Middle cerebral artery occlusion (MCAO) model | Lv et al. [109] |
| Alpha-lipoic acid | Activating SIRT1/PGC-1α pathway | Antioxidation | MCAO | Fu et al. [186] |
| Astragaloside IV (AS-IV) | Activating Sirt1/Mapt pathway | Antioxidation | MCAO | Shi et al. [85] |
| Arctigenin (ARC) | NLRP3 inflammasome | Anti-neuroinflammation | MCAO | Zhang et al. [142] |
| Quercetin | Activating SIRT1/Nrf2 pathway | Antioxidation | MCAO | Yang et al. [177] |
| Momordica charantia polysaccharides (MCPs) | Activating SIRT1/β-catenin pathway | Antioxidation | MCAO | Hu et al. [179] |
| Icariin (ICA) | Activating SIRT1/PGC-1α pathway | Antioxidation | MCAO | Zhu et al. [180] |
| Kaempferol (KFL) | Activating SIRT1/P66shc pathway | Antioxidation | Oxygen and glucose deprivation (OGD) | Zhou and Li [182] |
| Calycosin-7-O-β-D-glucoside (CG) | Activating SIRT1/FOXO1/PGC-1α pathway | Antioxidation | OGD | Yan et al. [58] |
| Notoginseng leaf triterpenes (PNGL) | Activating SIRT1/2/3-Foxo3a-MnSOD/PGC-1α pathways | Antioxidation | OGD | Xie et al. [183] |
| Cycloastragenol (CAG) | Suppression of SIRT1/NF-κB activation | Anti-neuroinflammation | MCAO | Li et al. [185] |
| Piceatannol | Activating Sirt1/FoxO1 pathway | Antioxidation | MCAO | Wang et al. [168] |
3.1. Natural Products Acting on SIRT1 in Hemorrhagic Stroke
3.1.1. HLY78
As a novel lycorine derivative, HLY78 has shown antiapoptotic effects in tumors, ICH, and SAH [146, 147, 148]. After intranasal administration of HLY78 1 hour after ICH, it reduced oxidative stress and the extent of neuronal damage in the perihematomal region. Therefore, HLY78 may prove to be an effective drug for the treatment of ICH. Additionally, GSK3β/Sirt1/PGC-1α signaling was partially involved in the antiapoptotic and antioxidative effects [148].
3.1.2. Resveratrol
Natural resveratrol is found in the skin of numerous edible plants as a stilbene polyphenol. Strokes can be treated with resveratrol by its anti-inflammatory and antioxidant effects in the field [125], which means that the neuroprotective effects of resveratrol are associated with several signaling pathways. More importantly, resveratrol is also a SIRT1 activator. Through deacetylation of p65, SIRT1 inhibits NF-κB in ICH. In ICH patients, the activation of SIRT1 by resveratrol led to a neuroprotective effect and a reduction in Ac-p65, IL-1β, TNF-α, and apoptosis [120].
3.1.3. Fucoxanthin (Fx)
Fx is a kind of xanthophyll derivative extensively distributed in seaweeds. This compound has an allenic bond and a 5,6 monoepoxide structure, making it a potent antioxidant. The efficacy of FX in animal models of cerebral ischemia/reperfusion injury, Alzheimer's disease, and traumatic brain injury has been demonstrated [149]. By activating Sirt1 and further deacetylating FOXO and p53, Fx has been shown to decrease oxidative damage and brain injury after SAH, indicating that Fx may act as a promising therapeutic agent for SAH [150].
3.1.4. Salvianolic Acid B (SalB)
An extract of the traditional Chinese herb SalB has been shown to possess antioxidant and neuroprotective properties in vitro and in vivo [151, 152]. The central nervous system (CNS) can be directly affected by SalB because it can easily traverse the blood–brain barrier (BBB). When it is used in patients with SAH, it can reduce oxidative damage from SAH through SalB's ability to enhance SIRT1 activity and further promote Nrf2 signaling pathway activation [153].
3.1.5. Melatonin
Melatonin (Mel, N-acetyl-5-methoxytryptamine), mainly secreted by the pineal gland, has shown various benefits, such as antioxidation, anti-inflammation, and antiapoptosis [154, 155, 156]. Experimental SAH models have shown Mel to be protective, reducing mortality after a severe subarachnoid hemorrhage [157]. A subsequent experiment further confirmed that the MR/Sirt1/NF-κB pathway is activated by Mel and reduces apoptosis following SAH [158].
3.1.6. Berberine
Berberine is an isoquinoline alkaloid found in the Chinese herb Coptis chinensis that has anti-inflammatory and neuroprotective properties. The activation of HMGB1/NF-κB plays a crucial role in cerebral inflammation in SAH [159]. The interaction between HMGB1 and TLR4 triggers inflammatory responses after SAH insults. Following TLR4 activation, NF-κB is active, and proinflammatory cytokines and chemokines are released [160]. Inflammatory responses mediated by HMGB1/NF-κB activation can be inhibited by berberine supplementation by increasing the expression of SIRT1 [161].
3.1.7. Oleanolic Acid (OA)
OA (3b-hydroxy-olea-12-en-28-oic acid) is a triterpenoid compound that comes from various sources and can reduce inflammation and enhance antioxidant activity. Upon inflammasome activation, neurons and glia release HMGB1, which activates the RAGE receptor on target cells and the TLR4 receptor in the immune system. When OA is applied to patients with SAH, it can reduce acetylation of HMGB1 by activating SIRT1 to alleviate early brain injury after subarachnoid hemorrhage, which has been demonstrated to exert neuroprotective effects through its anti-inflammatory role [162].
3.1.8. Carnosic Acid (CA)
The CA is one of the most abundant phenolic compounds in rosemary and sage leaves, exhibiting antioxidant and antiapoptotic properties [163]. In a study, CA was applied to a vascular perforation model, which was used to mimic clinical SAH, and the desired therapeutic effect was obtained. There was a dramatic reduction in neuronal cell death and brain edema with CA treatment in ICH, as well as a diminished level of ROS [164]. The SIRT1/p66shc pathway may play a role in the protective effect of CA on SAH.
3.1.9. Wogonoside
The herb Scutellaria belongs to the Labiatae family (containing approximately 400 species) and is used for treating inflammation, allergies, and hepatitis, and as an antioxidant. A compound isolated from Scutellaria called wogonin is a radical scavenger, an anticancer agent, and an antioxidant [165]. When wogonoside is used in SAH treatment, it activates SIRT1 and decreases the level of p53 to prevent neuronal apoptosis [166], mainly through its anti-inflammatory properties.
3.1.10. Piceatannol (Pic)
A hydroxylated analog of resveratrol called Pic has proven to be anti-inflammatory and potent antioxidant properties. A major mechanism through which Pic exerts its antioxidant effects is by upregulating antioxidant enzymes, such as SOD and CAT [167]. There was a dose-dependent increase in antioxidant activity and suppression of apoptosis with Pic, as well as upregulation of Sirt1/FoxO1 signaling in stroke [168]. It proved that Pic potentially exerts neuroprotective effects through upregulation of Sirt1/FoxO1 signaling in stroke.
3.2. Natural Products Acting on SIRT1 in Ischemic Stroke
3.2.1. Adiponectin (APN)
APN, which is entirely released by adipocytes, has been proven to act as a neuroprotectant in ischemia/reperfusion injuries [169, 170]. Past research suggested that there is an important role played by glutamate- (Glu-) induced excitotoxicity in stroke. When APN was used in HT22 neurons of Glu-induced injury, it upregulated the SIRT1, PGC-1α, and SOD. It downregulated the ROS levels, which suggested that the APN peptide activates PGC-1α signaling in HT22 neurons in response to Glu-induced injury [53].
3.2.2. Salvianolic Acid B
In addition to having a role in the SAH model, SalB is also protective against ischemic stroke. The expression of SIRT1 and Bcl-2 was upregulated by SalB in the MCAO model, while the expression of Ac-FOXO1 and Bax was downregulated. SalB treatment exerts cerebroprotective effects by reducing neuroinflammatory conditions and oxidative stress levels by activating SIRT1 signaling [109].
3.2.3. Alpha-Lipoic Acid (ALA)
The ALA is derived from octanoic acid, which acts as a cofactor in several mitochondrial dehydrogenases [171]. As an active free-radical scavenger, ALA exhibits powerful antioxidative properties [172]. After the systemic administration of ALA, SIRT1 and PGC-1α expressions were significantly increased, indicating the activation of SIRT1/PGC-1α might contribute to its beneficial effect.
3.2.4. Astragaloside IV (AS-IV)
For more than a thousand years, Astragalus membranaceus root and Mongolian Astragalus powder have been used to treat ischemic stroke [173], of which AS-IV is the predominant active component. The anti-inflammatory and antioxidative properties of AS-IV have been demonstrated in previous studies [174, 175]. In an earlier study, AS-IV contributed to the decrease in infarction area and the improvement of neurological functions. Additionally, the level of SIRT1 expression was upregulated, and ac-MAPT and p-MAPT declined [85]. Thus, the Sirt1/Mapt pathway is recognized to play a role in AS-IV's neuroprotective effect in rat models of CIR injury.
3.2.5. Arctigenin (ARC)
One of the most abundant and bioactive compounds in ARC has several pharmacological effects, including anti-inflammation, antiapoptosis, and antitumor effects [176]. It was demonstrated that ARC treatment could effectively suppress SIRT1-driven NLRP3 inflammasome activation by ischemic stroke in rat models [142]. In the light of this finding, ARC may play an important role in attenuating the damage to the brain caused by ischemic stroke.
3.2.6. Quercetin
A natural flavonoid, quercetin, is found in rutin and various fruits and vegetables. Quercetin is a potent ROS scavenger and has been shown to positively affect BBB function in acute ischemic strokes due to its ability to scavenge ROS. In research on the MCAO rat model, quercetin treatment was identified to suppress oxidative stress by activating Sirt1/Nrf2/HO-1 signaling pathways [177].
3.2.7. Momordica charantia Polysaccharides (MCPs)
MCPs, the main practical component extracted from Momordica charantia (MC), can protect against nerve damage after stroke by scavenging free radicals [178]. Under glutamate injury, MCPs activate SIRT1, improving the nuclear accumulation of β-catenin and contributing to NSC differentiation into neurons in the mimic IRI cell model [179]. Considering this study's findings, MCPs may protect patients recovering from ischemic stroke at late stages.
3.2.8. Icariin (ICA)
ICA is an active flavonoid with antioxidative and immunoregulatory effects. After MCAO, ICA has been shown to significantly attenuate brain damage, with the mechanism being an elevation of PGC-1α, which depends on SIRT1 [180]. It is, therefore, possible to develop ICA as a neuroprotectant for ischemia-related brain damage.
3.2.9. Kaempferol (KFL)
KFL is a natural product obtained from several natural sources that possesses antioxidative and immunoregulatory properties [181]. A cell model of ischemia/reperfusion injury showed that KFL effectively prevented OGD-induced cytotoxicity. The protection provided by KFL depends on the expression of SIRT1 and the inhibition of P66shc expression and acetylation [182].
3.2.10. CG
CG is a calycosin derivative compound derived from Astragali Radix (RA) and a representative component of isoflavones in RA. A significant improvement in cell viability and a reduction in neuronal apoptosis were observed with CG in the ischemia-reperfusion model in vitro. Additionally, CG treatment increased the expression of SIRT1, FOXO1, PGC-1α, and Bcl-2 while decreasing the expression of Bax [58]. This implies that GC exerts its neuroprotective role through activating SIRT1/FOXO1/PGC-1α signaling.
3.2.11. Notoginseng Leaf Triterpenes (PNGL)
PNGL are isolated from Panax notoginseng (Burk) and purified as a medicinal resource, functional, and typical food. Some studies have indicated that the neuronal apoptosis induced by ischemia is attenuated by PNGL, exerting potent neuroprotective effects. PNG exerts its protective effects via the NAMPT-NAD+ pathway and downstream SIRT1/2/3-Foxo3a-MnSOD/PGC-1α pathways [183].
3.2.12. Cycloastragenol (CAG)
CAG, an aglycone of astragaloside IV, was detected when looking for antiaging active ingredients in Astragalus membranaceus extracts [184]. It was found that a dose-dependent decrease in brain infarct volume was observed with CAG, as well as significant improvements in functional deficits and a decrease in neuronal loss in MCAO mice treated with CAG [185]. Its beneficial effect involves upregulating SIRT1, thereby inhibiting neuroinflammation.
4. Conclusion and Perspectives
Strokes are cerebral vascular diseases with a high mortality rate, deadly cerebral vascular disorders with a high mortality rate, and substantial economic and social costs. A stroke can be classified as either an ischemic stroke or a hemorrhagic stroke. Regardless of the type of stroke, however, oxidative stress and neuroinflammation have been linked to the subsequent brain injury and play vital roles. Accordingly, the treatment that reduces oxidative damage and inflammation after stroke to improve neurological recovery has long been a focus of stroke treatment research. SIRT1, as an NAD-dependent deacetylase in the nucleus, has been proven to play a critical role in oxidative stress and neuroinflammation in stroke. SIRT1 modulates oxidative stress and inflammation through a variety of signaling pathways. Therefore, a comprehensive understanding of the signaling pathway in the process of SIRT1-mediated antioxidation and anti-neuroinflammation in stroke is fundamental and can be used as the basis for therapies in stroke. At the same time, due to its numerous merits, such as readily available resources and few adverse effects, natural products have excellent application and practical clinical value in treating stroke. There are, however, some limitations to our literature review. Studies of natural compounds with possible neuroprotective effects in stroke are still in the exploratory stage, and clinical studies have yet to be conducted. This review discusses the potential mechanisms and natural products used in stroke by which Sirt1 protects against stroke through antioxidative and anti-inflammatory effects. It has considerable significance for future approaches to improving stroke treatment.
Acknowledgments
This work was supported by grants from the Zhejiang Provincial Natural Science Foundation of China (LY22H090020).
Contributor Information
Anke Zhang, Email: theanke@163.com.
Anwen Shao, Email: shaoanwen@zju.edu.cn.
Meiqing Lou, Email: meiqing_lou2020@163.com.
Data Availability
No data were used to support this study.
Conflicts of Interest
The authors declare that the research was conducted without any commercial or financial relationships construed as a potential conflict of interest.
Authors' Contributions
AZ, AS, and ML conceived, organized, and discussed the work. CF, HX, and LY contributed to manuscript writing and literature search. ZZ, XW, and YL revised the manuscript. All authors contributed to the review and approved the submitted version. Chaoyou Fang, Houshi Xu, and Ling Yuan contributed equally to this work.
References
- 1.Wang H., Naghavi M., Allen C., et al. Global, regional, and national life expectancy, all-cause mortality, and cause-specific mortality for 249 causes of death, 1980-2015: a systematic analysis for the Global Burden of Disease Study 2015. Lancet . 2016;388(10053):1459–1544. doi: 10.1016/S0140-6736(16)31012-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Donnan G. A., Fisher M., Macleod M., Davis S. M. Stroke. Lancet . 2008;371:1612–1623. doi: 10.1016/S0140-6736(08)60694-7. [DOI] [PubMed] [Google Scholar]
- 3.Sweid A., Hammoud B., Ramesh S., et al. Acute ischaemic stroke interventions: large vessel occlusion and beyond. Stroke and Vascular Neurology . 2020;5(1):80–85. doi: 10.1136/svn-2019-000262. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Johnson C. O., Nguyen M., Roth G. A., et al. Global, regional, and national burden of stroke, 1990-2016: a systematic analysis for the Global Burden of Disease Study 2016. Lancet Neurology . 2019;18(5):439–458. doi: 10.1016/S1474-4422(19)30034-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.World Healt Organization. Recommendations on stroke prevention, diagnosis, and therapy. Report of the WHO Task Force on Stroke and other Cerebrovascular Disorders. Stroke . 1989;20:1407–1431. doi: 10.1161/01.str.20.10.1407. [DOI] [PubMed] [Google Scholar]
- 6.Zhang A., Liu Y., Xu H., et al. CCL17 exerts neuroprotection through activation of CCR4/mTORC2 axis in microglia after subarachnoid haemorrhage in rats. Stroke and Vascular Neurology . 2022 doi: 10.1136/svn-2022-001659. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Sinha K., Das J., Pal P. B., Sil P. C. Oxidative stress: the mitochondria-dependent and mitochondria-independent pathways of apoptosis. Archives of Toxicology . 2013;87(7):1157–1180. doi: 10.1007/s00204-013-1034-4. [DOI] [PubMed] [Google Scholar]
- 8.Xiong X. Y., Wang J., Qian Z. M., Yang Q. W. Iron and intracerebral hemorrhage: from mechanism to translation. Translational Stroke Research . 2014;5(4):429–441. doi: 10.1007/s12975-013-0317-7. [DOI] [PubMed] [Google Scholar]
- 9.Aronowski J., Zhao X. Molecular pathophysiology of cerebral hemorrhage: secondary brain injury. Stroke . 2011;42(6):1781–1786. doi: 10.1161/STROKEAHA.110.596718. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Chan P. H. Reactive oxygen radicals in signaling and damage in the ischemic brain. Journal of Cerebral Blood Flow & Metabolism . 2001;21(1):2–14. doi: 10.1097/00004647-200101000-00002. [DOI] [PubMed] [Google Scholar]
- 11.Chan P. H. Oxygen radicals in focal cerebral ischemia. Brain Pathology . 1994;4(1):59–65. doi: 10.1111/j.1750-3639.1994.tb00811.x. [DOI] [PubMed] [Google Scholar]
- 12.Beal M. F. Mitochondrial dysfunction in neurodegenerative diseases and stroke: neuroprotective strategies. Journal of the Neurological Sciences . 2009;283(1-2):p. 240. doi: 10.1016/j.jns.2009.02.008. [DOI] [Google Scholar]
- 13.Wan J., Ren H., Wang J. Iron toxicity, lipid peroxidation and ferroptosis after intracerebral haemorrhage. Stroke/Vascular Neurology . 2019;4(2):93–95. doi: 10.1136/svn-2018-000205. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Niizuma K., Yoshioka H., Chen H., et al. Mitochondrial and apoptotic neuronal death signaling pathways in cerebral ischemia. Biochimica et Biophysica Acta (BBA)-Molecular Basis of Disease . 2010;1802(1):92–99. doi: 10.1016/j.bbadis.2009.09.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Dugue R., Nath M., Dugue A., Barone F. C. Roles of Pro- and Anti-Inflammatory Cytokines in Traumatic Brain Injury and Acute Ischemic Stroke. Mechanical Neuroinflammation . 2017;211 [Google Scholar]
- 16.Jin R., Yang G., Li G. Inflammatory mechanisms in ischemic stroke: role of inflammatory cells. Journal of Leukocyte Biology . 2010;87(5):779–789. doi: 10.1189/jlb.1109766. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Murakami K., Koide M., Dumont T. M., Russell S. R., Tranmer B. I., Wellman G. C. Subarachnoid hemorrhage induces gliosis and increased expression of the pro-inflammatory cytokine high mobility group box 1 protein. Translational Stroke Research . 2011;2(1):72–79. doi: 10.1007/s12975-010-0052-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Kelly G. S. A review of the sirtuin system, its clinical implications, and the potential role of dietary activators like resveratrol: part 2. Alternative Medicine Review . 2010;15(4):313–328. [PubMed] [Google Scholar]
- 19.Zhang X. S., Lu Y., Li W., et al. Cerebroprotection by dioscin after experimental subarachnoid haemorrhage via inhibiting NLRP3 inflammasome through SIRT1-dependent pathway. British Journal of Pharmacology . 2021;178(18):3648–3666. doi: 10.1111/bph.15507. [DOI] [PubMed] [Google Scholar]
- 20.Zhang X. S., Wu Q., Wu L. Y., et al. Sirtuin 1 activation protects against early brain injury after experimental subarachnoid hemorrhage in rats. Cell Death & Disease . 2016;7(10, article e2416) doi: 10.1038/cddis.2016.292. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Mao H., Wang L., Xiong Y., Jiang G., Liu X. Fucoxanthin attenuates oxidative damage by activating the Sirt1/Nrf2/HO-1 signaling pathway to protect the kidney from ischemia-reperfusion injury. Oxidative Medicine and Cellular Longevity . 2022;2022:28. doi: 10.1155/2022/7444430.7444430 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Liu L., Cao Q., Gao W., et al. Melatonin ameliorates cerebral ischemia-reperfusion injury in diabetic mice by enhancing autophagy via the SIRT1-BMAL1 pathway. The FASEB Journal . 2021;35(12, article e22040) doi: 10.1096/fj.202002718RR. [DOI] [PubMed] [Google Scholar]
- 23.Wu B., Feng J. Y., Yu L. M., et al. Icariin protects cardiomyocytes against ischaemia/reperfusion injury by attenuating sirtuin 1-dependent mitochondrial oxidative damage. British Journal of Pharmacology . 2018;175(21):4137–4153. doi: 10.1111/bph.14457. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Ramadori G., Lee C. E., Bookout A. L., et al. Brain SIRT1: anatomical distribution and regulation by energy availability. Journal of Neuroscience . 2008;28(40):9989–9996. doi: 10.1523/JNEUROSCI.3257-08.2008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Hernández-Jiménez M., Hurtado O., Cuartero M. I., et al. Silent information regulator 1 protects the brain against cerebral ischemic damage. Stroke . 2013;44(8):2333–2337. doi: 10.1161/STROKEAHA.113.001715. [DOI] [PubMed] [Google Scholar]
- 26.Xia D. Y., Yuan J. L., Jiang X. C., et al. SIRT1 promotes M2 microglia polarization via reducing ROS-mediated NLRP3 inflammasome signaling after subarachnoid hemorrhage. Frontiers in Immunology . 2021;12, article 770744 doi: 10.3389/fimmu.2021.770744. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Kim E. J., Kho J. H., Kang M. R., Um S. J. Active regulator of SIRT1 cooperates with SIRT1 and facilitates suppression of p53 activity. Molecular Cell . 2007;28(2):277–290. doi: 10.1016/j.molcel.2007.08.030. [DOI] [PubMed] [Google Scholar]
- 28.Yang Y., Duan W., Li Y., et al. New role of silent information regulator 1 in cerebral ischemia. Neurobiology of Aging . 2013;34(12):2879–2888. doi: 10.1016/j.neurobiolaging.2013.06.008. [DOI] [PubMed] [Google Scholar]
- 29.National Institute of Neurological Disorders and Stroke rt-PA Stroke Study Group. Tissue plasminogen activator for acute ischemic stroke. New England Journal of Medicine . 1995;333(24):1581–1587. doi: 10.1056/NEJM199512143332401. [DOI] [PubMed] [Google Scholar]
- 30.Kaur J., Zhao Z., Klein G. M., Lo E. H., Buchan A. M. The neurotoxicity of tissue plasminogen activator? Journal of Cerebral Blood Flow & Metabolism . 2004;24(9):945–963. doi: 10.1097/01.WCB.0000137868.50767.E8. [DOI] [PubMed] [Google Scholar]
- 31.Wang C., Huang R., Li C., et al. Vepoloxamer enhances fibrinolysis of tPA (tissue-type plasminogen activator) on acute ischemic stroke. Stroke . 2019;50(12):3600–3608. doi: 10.1161/STROKEAHA.119.026049. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Brieger K., Schiavone S., Miller F. J., Jr., Krause K. H. Reactive oxygen species: from health to disease. Swiss Medical Weekly . 2012;142, article w13659 doi: 10.4414/smw.2012.13659. [DOI] [PubMed] [Google Scholar]
- 33.Andreyev A. Y., Kushnareva Y. E., Starkov A. A. Mitochondrial metabolism of reactive oxygen species. Biochemistry . 2005;70(2):200–214. doi: 10.1007/s10541-005-0102-7. [DOI] [PubMed] [Google Scholar]
- 34.Balaban R. S., Nemoto S., Finkel T. Mitochondria, oxidants, and aging. Cell . 2005;120(4):483–495. doi: 10.1016/j.cell.2005.02.001. [DOI] [PubMed] [Google Scholar]
- 35.Dröge W. Free radicals in the physiological control of cell function. Physiological Reviews . 2002;82:47–95. doi: 10.1152/physrev.00018.2001. [DOI] [PubMed] [Google Scholar]
- 36.Kim C. K., Kim T., Choi I. Y., et al. Ceria nanoparticles that can protect against ischemic stroke. Angewandte Chemie International Edition . 2012;51(44):11039–11043. doi: 10.1002/anie.201203780. [DOI] [PubMed] [Google Scholar]
- 37.Yang Y., Chen S., Zhang J. M. The updated role of oxidative stress in subarachnoid hemorrhage. Current Drug Delivery . 2017;14(6):832–842. doi: 10.2174/1567201813666161025115531. [DOI] [PubMed] [Google Scholar]
- 38.Candelario-Jalil E. Injury and repair mechanisms in ischemic stroke: considerations for the development of novel neurotherapeutics. Current Opinion in Investigational Drugs . 2009;10(7):644–654. [PubMed] [Google Scholar]
- 39.Mattiasson G., Shamloo M., Gido G., et al. Uncoupling protein-2 prevents neuronal death and diminishes brain dysfunction after stroke and brain trauma. Nature Medicine . 2003;9(8):1062–1068. doi: 10.1038/nm903. [DOI] [PubMed] [Google Scholar]
- 40.Slemmer J. E., Shacka J. J., Sweeney M. I., Weber J. T. Antioxidants and free radical scavengers for the treatment of stroke, traumatic brain injury and aging. Current Medicinal Chemistry . 2008;15(4):404–414. doi: 10.2174/092986708783497337. [DOI] [PubMed] [Google Scholar]
- 41.Michishita E., Park J. Y., Burneskis J. M., Barrett J. C., Horikawa I. Evolutionarily conserved and nonconserved cellular localizations and functions of human SIRT proteins. Molecular Biology of the Cell . 2005;16(10):4623–4635. doi: 10.1091/mbc.e05-01-0033. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Hattori Y., Okamoto Y., Nagatsuka K., et al. SIRT1 attenuates severe ischemic damage by preserving cerebral blood flow. Neuroreport . 2015;26(3):113–117. doi: 10.1097/WNR.0000000000000308. [DOI] [PubMed] [Google Scholar]
- 43.Singh V., Ubaid S. Role of silent information regulator 1 (SIRT1) in regulating oxidative stress and inflammation. Inflammation . 2020;43(5):1589–1598. doi: 10.1007/s10753-020-01242-9. [DOI] [PubMed] [Google Scholar]
- 44.Mastropasqua F., Girolimetti G., Shoshan M. PGC1α: friend or foe in cancer? Genes . 2018;9(1) doi: 10.3390/genes9010048. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Lin J., Handschin C., Spiegelman B. M. Metabolic control through the PGC-1 family of transcription coactivators. Cell Metabolism . 2005;1(6):361–370. doi: 10.1016/j.cmet.2005.05.004. [DOI] [PubMed] [Google Scholar]
- 46.Valle I., Alvarez-Barrientos A., Arza E., Lamas S., Monsalve M. PGC-1alpha regulates the mitochondrial antioxidant defense system in vascular endothelial cells. Cardiovascular Research . 2005;66(3):562–573. doi: 10.1016/j.cardiores.2005.01.026. [DOI] [PubMed] [Google Scholar]
- 47.Aquilano K., Baldelli S., Pagliei B., Cannata S. M., Rotilio G., Ciriolo M. R. p53 orchestrates the PGC-1α-mediated antioxidant response upon mild redox and metabolic imbalance. Antioxidants & Redox Signaling . 2013;18(4):386–399. doi: 10.1089/ars.2012.4615. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Baldelli S., Aquilano K., Ciriolo M. R. PGC-1α buffers ROS-mediated removal of mitochondria during myogenesis. Cell Death & Disease . 2014;5(11, article e1515) doi: 10.1038/cddis.2014.458. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Schilling T., Eder C. Amyloid-β-induced reactive oxygen species production and priming are differentially regulated by ion channels in microglia. Journal of Cellular Physiology . 2011;226(12):3295–3302. doi: 10.1002/jcp.22675. [DOI] [PubMed] [Google Scholar]
- 50.Bhatti J. S., Bhatti G. K., Reddy P. H. Mitochondrial dysfunction and oxidative stress in metabolic disorders - a step towards mitochondria based therapeutic strategies. Biochimica et Biophysica Acta (BBA)-Molecular Basis of Disease . 2017;1863(5):1066–1077. doi: 10.1016/j.bbadis.2016.11.010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Pérez S., Rius-Pérez S., Finamor I., et al. Obesity causes PGC-1α deficiency in the pancreas leading to marked IL-6 upregulation via NF-κB in acute pancreatitis. The Journal of Pathology . 2019;247(1):48–59. doi: 10.1002/path.5166. [DOI] [PubMed] [Google Scholar]
- 52.Fernandez-Marcos P. J., Auwerx J. Regulation of PGC-1α, a nodal regulator of mitochondrial biogenesis. The American Journal Of Clinical Nutrition . 2011;93(4):884s–8890. doi: 10.3945/ajcn.110.001917. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Yue L., Zhao L., Liu H., et al. Adiponectin protects against glutamate-induced excitotoxicity via activating SIRT1-dependent PGC-1α expression in HT22 hippocampal neurons. Oxidative Medicine and Cellular Longevity . 2016;2016:12. doi: 10.1155/2016/2957354.2957354 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Cantó C., Auwerx J. PGC-1alpha, SIRT1 and AMPK, an energy sensing network that controls energy expenditure. Current Opinion in Lipidology . 2009;20(2):98–105. doi: 10.1097/MOL.0b013e328328d0a4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.St-Pierre J., Drori S., Uldry M., et al. Suppression of reactive oxygen species and neurodegeneration by the PGC-1 transcriptional coactivators. Cell . 2006;127(2):397–408. doi: 10.1016/j.cell.2006.09.024. [DOI] [PubMed] [Google Scholar]
- 56.Berthiaume J. M., Kurdys J. G., Muntean D. M., Rosca M. G. Mitochondrial NAD(+)/NADH redox state and diabetic cardiomyopathy. Antioxidants & Redox Signaling . 2019;30(3):375–398. doi: 10.1089/ars.2017.7415. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Vinogradov A. D., Grivennikova V. G. Oxidation of NADH and ROS production by respiratory complex I. Biochimica et Biophysica Acta (BBA)-Bioenergetics . 2016;1857(7):863–871. doi: 10.1016/j.bbabio.2015.11.004. [DOI] [PubMed] [Google Scholar]
- 58.Yan X., Yu A., Zheng H., Wang S., He Y., Wang L. Calycosin-7-O-β-D-glucoside attenuates OGD/R-induced damage by preventing oxidative stress and neuronal apoptosis via the SIRT1/FOXO1/PGC-1α pathway in HT22 cells. Neural Plasticity . 2019;2019:11. doi: 10.1155/2019/8798069.8798069 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59.Waldman M., Cohen K., Yadin D., et al. Regulation of diabetic cardiomyopathy by caloric restriction is mediated by intracellular signaling pathways involving ‘SIRT1 and PGC-1α’. Cardiovascular Diabetology . 2018;17(1):p. 111. doi: 10.1186/s12933-018-0754-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Wang Y., Zhou Y., Graves D. T. FOXO transcription factors: their clinical significance and regulation. BioMed Research International . 2014;2014:13. doi: 10.1155/2014/925350.925350 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.Dejean A. S., Hedrick S. M., Kerdiles Y. M. Highly specialized role of forkhead box O transcription factors in the immune system. Antioxidants & Redox Signaling . 2011;14(4):663–674. doi: 10.1089/ars.2010.3414. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62.Chung S., Ranjan R., Lee Y. G., et al. Distinct role of FoxO1 in M-CSF- and GM-CSF-differentiated macrophages contributes LPS-mediated IL-10: implication in hyperglycemia. Journal of Leukocyte Biology . 2015;97(2):327–339. doi: 10.1189/jlb.3A0514-251R. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63.Giannakou M. E., Partridge L. The interaction between FOXO and SIRT1: tipping the balance towards survival. Trends in Cell Biology . 2004;14(8):408–412. doi: 10.1016/j.tcb.2004.07.006. [DOI] [PubMed] [Google Scholar]
- 64.Sunters A., Fernández de Mattos S., Stahl M., et al. FoxO3a transcriptional regulation of Bim controls apoptosis in paclitaxel-treated breast cancer cell lines. Journal of Biological Chemistry . 2003;278(50):49795–49805. doi: 10.1074/jbc.M309523200. [DOI] [PubMed] [Google Scholar]
- 65.Kops G. J., Dansen T. B., Polderman P. E., et al. Forkhead transcription factor FOXO3a protects quiescent cells from oxidative stress. Nature . 2002;419(6904):316–321. doi: 10.1038/nature01036. [DOI] [PubMed] [Google Scholar]
- 66.Nemoto S., Finkel T. Redox regulation of forkhead proteins through a p66shc-dependent signaling pathway. Science . 2002;295(5564):2450–2452. doi: 10.1126/science.1069004. [DOI] [PubMed] [Google Scholar]
- 67.Accili D., Arden K. C. FoxOs at the crossroads of cellular metabolism, differentiation, and transformation. Cell . 2004;117(4):421–426. doi: 10.1016/S0092-8674(04)00452-0. [DOI] [PubMed] [Google Scholar]
- 68.Mangia A., Saponaro C., Vagheggini A., et al. Should tumor infiltrating lymphocytes, androgen receptor, and FOXA1 expression predict the clinical outcome in triple negative breast cancer patients? Cancers . 2019;11(9):p. 1393. doi: 10.3390/cancers11091393. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 69.Brunet A., Sweeney L. B., Sturgill J. F., et al. Stress-dependent regulation of FOXO transcription factors by the SIRT1 deacetylase. Science . 2004;303(5666):2011–2015. doi: 10.1126/science.1094637. [DOI] [PubMed] [Google Scholar]
- 70.Gilley J., Coffer P. J., Ham J. FOXO transcription factors directly activate bim gene expression and promote apoptosis in sympathetic neurons. The Journal of Cell Biology . 2003;162(4):613–622. doi: 10.1083/jcb.200303026. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 71.Wu Y., Huang Y., Cai J., Zhang D., Liu S., Pang B. LncRNA SNHG12 improves cerebral ischemic-reperfusion injury by activating SIRT1/FOXO3a pathway through I nhibition of autophagy and oxidative stress. Current Neurovascular Research . 2020;17(4):394–401. doi: 10.2174/1567202617666200727142019. [DOI] [PubMed] [Google Scholar]
- 72.Olmos Y., Valle I., Borniquel S., et al. Mutual dependence of Foxo3a and PGC-1alpha in the induction of oxidative stress genes. Journal of Biological Chemistry . 2009;284(21):14476–14484. doi: 10.1074/jbc.M807397200. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 73.Borniquel S., García-Quintáns N., Valle I., et al. Inactivation of Foxo3a and subsequent downregulation of PGC-1 alpha mediate nitric oxide-induced endothelial cell migration. Molecular and Cellular Biology . 2010;30(16):4035–4044. doi: 10.1128/MCB.00175-10. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 74.Duan J., Cui J., Zheng H., et al. Aralia taibaiensis protects against I/R-induced brain cell injury through the Akt/SIRT1/FOXO3a pathway. Oxidative Medicine and Cellular Longevity . 2019;2019:18. doi: 10.1155/2019/7609765.7609765 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 75.Avila J., Lucas J. J., Perez M., Hernandez F. Role of tau protein in both physiological and pathological conditions. Physiological Reviews . 2004;84:361–384. doi: 10.1152/physrev.00024.2003. [DOI] [PubMed] [Google Scholar]
- 76.Ballatore C., Lee V. M., Trojanowski J. Q. Tau-mediated neurodegeneration in Alzheimer’s disease and related disorders. Nature Reviews Neuroscience . 2007;8(9):663–672. doi: 10.1038/nrn2194. [DOI] [PubMed] [Google Scholar]
- 77.Cohen T. J., Guo J. L., Hurtado D. E., et al. The acetylation of tau inhibits its function and promotes pathological tau aggregation. Nature Communications . 2011;2(1):1–9. doi: 10.1038/ncomms1255. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 78.Alavi Naini S. M., Soussi-Yanicostas N. Tau hyperphosphorylation and oxidative stress, a critical vicious circle in neurodegenerative tauopathies? Oxidative Medicine and Cellular Longevity . 2015;2015:17. doi: 10.1155/2015/151979.151979 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 79.Basurto-Islas G., Gu J. H., Tung Y. C., Liu F., Iqbal K. Mechanism of tau hyperphosphorylation involving lysosomal enzyme asparagine endopeptidase in a mouse model of brain ischemia. Journal of Alzheimer's Disease . 2018;63(2):821–833. doi: 10.3233/JAD-170715. [DOI] [PubMed] [Google Scholar]
- 80.Fujii H., Takahashi T., Mukai T., et al. Modifications of tau protein after cerebral ischemia and reperfusion in rats are similar to those occurring in Alzheimer’s disease - hyperphosphorylation and cleavage of 4- and 3-repeat tau. Journal of Cerebral Blood Flow & Metabolism . 2017;37(7):2441–2457. doi: 10.1177/0271678X16668889. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 81.Bi M., Gladbach A., van Eersel J., et al. Tau exacerbates excitotoxic brain damage in an animal model of stroke. Nature Communications . 2017;8(1):1–15. doi: 10.1038/s41467-017-00618-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 82.Ferreon J. C., Jain A., Choi K. J., et al. Acetylation disfavors tau phase separation. International Journal of Molecular Sciences . 2018;19(5):p. 1360. doi: 10.3390/ijms19051360. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 83.Min S. W., Cho S. H., Zhou Y., et al. Acetylation of tau inhibits its degradation and contributes to tauopathy. Neuron . 2010;67(6):953–966. doi: 10.1016/j.neuron.2010.08.044. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 84.Min S. W., Sohn P. D., Li Y., et al. SIRT1 deacetylates tau and reduces pathogenic tau spread in a mouse model of tauopathy. Journal of Neuroscience . 2018;38(15):3680–3688. doi: 10.1523/JNEUROSCI.2369-17.2018. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 85.Shi Y. H., Zhang X. L., Ying P. J., et al. Neuroprotective effect of astragaloside IV on cerebral ischemia/reperfusion injury rats through Sirt1/Mapt pathway. Frontiers in Pharmacology . 2021;12, article 639898 doi: 10.3389/fphar.2021.639898. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 86.Liu Q., Jin Z., Xu Z., et al. Antioxidant effects of ginkgolides and bilobalide against cerebral ischemia injury by activating the Akt/Nrf2 pathway in vitro and in vivo. Cell Stress Chaperones . 2019;24(4):441–452. doi: 10.1007/s12192-019-00977-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 87.Jiang S., Deng C., Lv J., et al. Nrf2 weaves an elaborate network of neuroprotection against stroke. Molecular Neurobiology . 2017;54(2):1440–1455. doi: 10.1007/s12035-016-9707-7. [DOI] [PubMed] [Google Scholar]
- 88.Alfieri A., Srivastava S., Siow R. C., Modo M., Fraser P. A., Mann G. E. Targeting the Nrf2-Keap1 antioxidant defence pathway for neurovascular protection in stroke. The Journal of Physiology . 2011;589(17):4125–4136. doi: 10.1113/jphysiol.2011.210294. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 89.Shah Z. A., Li R. C., Thimmulappa R. K., et al. Role of reactive oxygen species in modulation of Nrf2 following ischemic reperfusion injury. Neuroscience . 2007;147(1):53–59. doi: 10.1016/j.neuroscience.2007.02.066. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 90.Mei Z., Du L., Liu X., et al. Diosmetin alleviated cerebral ischemia/reperfusion injury in vivo and in vitro by inhibiting oxidative stress via the SIRT1/Nrf2 signaling pathway. Food & Function . 2022;13(1):198–212. doi: 10.1039/D1FO02579A. [DOI] [PubMed] [Google Scholar]
- 91.Denes A., Thornton P., Rothwell N. J., Allan S. M. Inflammation and brain injury: acute cerebral ischaemia, peripheral and central inflammation. Brain, Behavior, and Immunity . 2010;24(5):708–723. doi: 10.1016/j.bbi.2009.09.010. [DOI] [PubMed] [Google Scholar]
- 92.Iadecola C., Alexander M. Cerebral ischemia and inflammation. Current Opinion in Neurology . 2001;14(1):89–94. doi: 10.1097/00019052-200102000-00014. [DOI] [PubMed] [Google Scholar]
- 93.Emsley H. C., Smith C. J., Gavin C. M., et al. An early and sustained peripheral inflammatory response in acute ischaemic stroke: relationships with infection and atherosclerosis. Journal of Neuroimmunology . 2003;139(1-2):93–101. doi: 10.1016/S0165-5728(03)00134-6. [DOI] [PubMed] [Google Scholar]
- 94.Lambertsen K. L., Finsen B., Clausen B. H. Post-stroke inflammation-target or tool for therapy? Acta Neuropathologica . 2019;137(5):693–714. doi: 10.1007/s00401-018-1930-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 95.Zhang S. Microglial activation after ischaemic stroke. Stroke and Vascular Neurology . 2019;4(2):71–74. doi: 10.1136/svn-2018-000196. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 96.Yan F., Cheng X., Zhao M., et al. Loss of Wip1 aggravates brain injury after ischaemia/reperfusion by overactivating microglia. Stroke and Vascular Neurology . 2021;6(3):344–351. doi: 10.1136/svn-2020-000490. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 97.Clausen B. H., Lambertsen K. L., Babcock A. A., Holm T. H., Dagnaes-Hansen F., Finsen B. Interleukin-1beta and tumor necrosis factor-alpha are expressed by different subsets of microglia and macrophages after ischemic stroke in mice. Journal of Neuroinflammation . 2008;5(1):1–18. doi: 10.1186/1742-2094-5-46. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 98.Ito D., Tanaka K., Suzuki S., Dembo T., Fukuuchi Y. Enhanced expression of Iba1, ionized calcium-binding adapter molecule 1, after transient focal cerebral ischemia in rat brain. Stroke . 2001;32(5):1208–1215. doi: 10.1161/01.STR.32.5.1208. [DOI] [PubMed] [Google Scholar]
- 99.Lambertsen K. L., Meldgaard M., Ladeby R., Finsen B. A quantitative study of microglial-macrophage synthesis of tumor necrosis factor during acute and late focal cerebral ischemia in mice. Journal of Cerebral Blood Flow & Metabolism . 2005;25(1):119–135. doi: 10.1038/sj.jcbfm.9600014. [DOI] [PubMed] [Google Scholar]
- 100.Perego C., Fumagalli S., De Simoni M. G. Temporal pattern of expression and colocalization of microglia/macrophage phenotype markers following brain ischemic injury in mice. Journal of Neuroinflammation . 2011;8(1):1–20. doi: 10.1186/1742-2094-8-174. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 101.Chu H. X., Arumugam T. V., Gelderblom M., Magnus T., Drummond G. R., Sobey C. G. Role of CCR2 in inflammatory conditions of the central nervous system. Journal of Cerebral Blood Flow & Metabolism . 2014;34(9):1425–1429. doi: 10.1038/jcbfm.2014.120. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 102.Selvaraj U. M., Stowe A. M. Long-term T cell responses in the brain after an ischemic stroke. Discovery Medicine . 2017;24(134):323–333. [PMC free article] [PubMed] [Google Scholar]
- 103.Zou M., Feng Y., Xiu Y., et al. Pertussis toxin-induced inflammatory response exacerbates intracerebral haemorrhage and ischaemic stroke in mice. Stroke and Vascular Neurology . 2022;7(1):29–37. doi: 10.1136/svn-2021-000987. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 104.Li S., Liu K., Zhang R., et al. Lower lymphocyte to monocyte ratio is a potential predictor of poor outcome in patients with cerebral venous sinus thrombosis. Stroke and Vascular Neurology . 2019;4(3):148–153. doi: 10.1136/svn-2018-000180. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 105.Shan H., Bian Y., Shu Z., et al. Fluoxetine protects against IL-1β-induced neuronal apoptosis via downregulation of p53. Neuropharmacology . 2016;107:68–78. doi: 10.1016/j.neuropharm.2016.03.019. [DOI] [PubMed] [Google Scholar]
- 106.Zhang M. J., Zhou Y., Wang X., et al. Interleukin-18 gene promoter 607A polymorphism, but not 137C polymorphism, is a protective factor for ischemic stroke in the Chinese population: a meta-analysis. Meta Gene . 2016;9:165–172. doi: 10.1016/j.mgene.2016.06.006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 107.Zheng Z. V., Chen J., Lyu H., et al. Novel role of STAT3 in microglia-dependent neuroinflammation after experimental subarachnoid haemorrhage. Stroke and Vascular Neurology . 2022;7(1):62–70. doi: 10.1136/svn-2021-001028. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 108.Shen L., Bai Y., Han B., Yao H. Non-coding RNA and neuroinflammation: implications for the therapy of stroke. Stroke/Vascular Neurology . 2019;4(2):96–98. doi: 10.1136/svn-2018-000206. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 109.Lv H., Wang L., Shen J., et al. Salvianolic acid B attenuates apoptosis and inflammation via SIRT1 activation in experimental stroke rats. Brain Research Bulletin . 2015;115:30–36. doi: 10.1016/j.brainresbull.2015.05.002. [DOI] [PubMed] [Google Scholar]
- 110.Palencia G., Núñez-Medrano J. Á., Ortiz-Plata A., et al. Anti-apoptotic, anti-oxidant, and anti-inflammatory effects of thalidomide on cerebral ischemia/reperfusion injury in rats. Journal of the Neurological Sciences . 2015;351(1-2):78–87. doi: 10.1016/j.jns.2015.02.043. [DOI] [PubMed] [Google Scholar]
- 111.Zhu H., Zou L., Tian J., Du G., Gao Y. SMND-309, a novel derivative of salvianolic acid B, protects rat brains ischemia and reperfusion injury by targeting the JAK2/STAT3 pathway. European Journal of Pharmacology . 2013;714(1-3):23–31. doi: 10.1016/j.ejphar.2013.05.043. [DOI] [PubMed] [Google Scholar]
- 112.Kou D. Q., Jiang Y. L., Qin J. H., Huang Y. H. Magnolol attenuates the inflammation and apoptosis through the activation of SIRT1 in experimental stroke rats. Pharmacological Reports . 2017;69(4):642–647. doi: 10.1016/j.pharep.2016.12.012. [DOI] [PubMed] [Google Scholar]
- 113.Sun S. C., Chang J. H., Jin J. Regulation of nuclear factor-κB in autoimmunity. Trends in Immunology . 2013;34(6):282–289. doi: 10.1016/j.it.2013.01.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 114.Oeckinghaus A., Ghosh S. The NF-kappaB family of transcription factors and its regulation. Cold Spring Harbor perspectives in Biology . 2009;1(4, article a000034) doi: 10.1101/cshperspect.a000034. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 115.Lawrence T. The nuclear factor NF-kappaB pathway in inflammation. Cold Spring Harbor Perspectives in Biology . 2009;1(6, article a001651) doi: 10.1101/cshperspect.a001651. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 116.Vallabhapurapu S., Karin M. Regulation and function of NF-kappaB transcription factors in the immune system. Annual Review of Immunology . 2009;27:693–733. doi: 10.1146/annurev.immunol.021908.132641. [DOI] [PubMed] [Google Scholar]
- 117.Karin M., Ben-Neriah Y. Phosphorylation meets ubiquitination: the control of NF-[kappa]B activity. Annual Review of Immunology . 2000;18:621–663. doi: 10.1146/annurev.immunol.18.1.621. [DOI] [PubMed] [Google Scholar]
- 118.Bonizzi G., Karin M. The two NF-kappaB activation pathways and their role in innate and adaptive immunity. Trends Immunol . 2004;25(6):280–288. doi: 10.1016/j.it.2004.03.008. [DOI] [PubMed] [Google Scholar]
- 119.Albensi B. C. What is nuclear factor kappa B (NF-κB) doing in and to the mitochondrion? Frontiers in Cell and Developmental Biology . 2019;7:p. 154. doi: 10.3389/fcell.2019.00154. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 120.Deng H. J., Zhou C. H., Huang L. T., Wen L. B., Zhou M. L., Wang C. X. Activation of silent information regulator 1 exerts a neuroprotective effect after intracerebral hemorrhage by deacetylating NF-κB/p65. Journal of Neurochemistry . 2021;157(3):574–585. doi: 10.1111/jnc.15258. [DOI] [PubMed] [Google Scholar]
- 121.Wang Y. X., Yan A., Ma Z. H., et al. Nuclear factor-κB and apoptosis in patients with intracerebral hemorrhage. Journal of Clinical Neuroscience . 2011;18(10):1392–1395. doi: 10.1016/j.jocn.2010.11.039. [DOI] [PubMed] [Google Scholar]
- 122.Mendes K. L., Lelis D. F., Santos S. H. S. Nuclear sirtuins and inflammatory signaling pathways. Cytokine & Growth Factor Reviews . 2017;38:98–105. doi: 10.1016/j.cytogfr.2017.11.001. [DOI] [PubMed] [Google Scholar]
- 123.Qadir M. I., Anwar S. Sirtuins in brain aging and neurological disorders. Critical Reviews in Eukaryotic Gene Expression . 2017;27(4):321–329. doi: 10.1615/CritRevEukaryotGeneExpr.2017019532. [DOI] [PubMed] [Google Scholar]
- 124.Yeung F., Hoberg J. E., Ramsey C. S., et al. Modulation of NF-kappaB-dependent transcription and cell survival by the SIRT1 deacetylase. The EMBO Journal . 2004;23:2369–2380. doi: 10.1038/sj.emboj.7600244. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 125.Lange S. S., Vasquez K. M. HMGB1: the jack-of-all-trades protein is a master DNA repair mechanic. Molecular Carcinogenesis . 2009;48(7):571–580. doi: 10.1002/mc.20544. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 126.Yu Y., Tang D., Kang R. Oxidative stress-mediated HMGB1 biology. Frontiers in Physiology . 2015;6 doi: 10.3389/fphys.2015.0009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 127.Kim J. B., Sig Choi J., Yu Y. M., et al. HMGB1, a novel cytokine-like mediator linking acute neuronal death and delayed neuroinflammation in the postischemic brain. Journal of Neuroscience . 2006;26(24):6413–6421. doi: 10.1523/JNEUROSCI.3815-05.2006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 128.Andersson U., Yang H., Harris H. Extracellular HMGB1 as a therapeutic target in inflammatory diseases. Expert Opinion on Therapeutic Targets . 2018;22(3):263–277. doi: 10.1080/14728222.2018.1439924. [DOI] [PubMed] [Google Scholar]
- 129.Bianchi M. E., Manfredi A. A. High-mobility group box 1 (HMGB1) protein at the crossroads between innate and adaptive immunity. Immunological Reviews . 2007;220(1):35–46. doi: 10.1111/j.1600-065X.2007.00574.x. [DOI] [PubMed] [Google Scholar]
- 130.Chen F., Lu J., Chen F., et al. Recombinant neuroglobin ameliorates early brain injury after subarachnoid hemorrhage via inhibiting the activation of mitochondria apoptotic pathway. Neurochemistry International . 2018;112:219–226. doi: 10.1016/j.neuint.2017.07.012. [DOI] [PubMed] [Google Scholar]
- 131.Zhang X., Wu Q., Zhang Q., et al. Resveratrol attenuates early brain injury after experimental subarachnoid hemorrhage via inhibition of NLRP3 inflammasome activation. Frontiers in Neuroscience . 2017;11:p. 611. doi: 10.3389/fnins.2017.00611. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 132.Lu C., Gao S., Xu G. Geraniin inhibits TNF-α-induced impairments of osteogenesis through NF-κB and p38 MAPK signalling pathways in bone marrow stem cells. Stroke and Vascular Neurology . 2017;2:47–52. doi: 10.1136/svn-2016-000046. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 133.Li W., Zhang Y., Tian Z., et al. Statin treatment for unruptured intracranial aneurysms study: a study protocol for a double-blind, placebo-controlled trial. Stroke and Vascular Neurology . 2020;5(4):410–415. doi: 10.1136/svn-2020-000353. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 134.Lu B., Antoine D. J., Kwan K., et al. JAK/STAT1 signaling promotes HMGB1 hyperacetylation and nuclear translocation. Proceedings of the National Academy of Sciences of the United States of America . 2014;111(8):3068–3073. doi: 10.1073/pnas.1316925111. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 135.Hwang J. S., Lee W. J., Kang E. S., et al. Ligand-activated peroxisome proliferator-activated receptor-delta and -gamma inhibit lipopolysaccharide-primed release of high mobility group box 1 through upregulation of SIRT1. Cell Death & Disease . 2014;5, article e1432 doi: 10.1038/cddis.2014.406. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 136.Hwang J. S., Choi H. S., Ham S. A., et al. Deacetylation-mediated interaction of SIRT1-HMGB1 improves survival in a mouse model of endotoxemia. Scientific Reports . 2015;5(1):1–16. doi: 10.1038/srep15971. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 137.Wang L., Zha B., Shen Q., et al. Sevoflurane inhibits the Th2 response and NLRP3 expression in murine allergic airway inflammation. Journal of Immunology Research . 2018;2018:8. doi: 10.1155/2018/9021037.9021037 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 138.Cho M. H., Cho K., Kang H. J., et al. Autophagy in microglia degrades extracellular β-amyloid fibrils and regulates the NLRP3 inflammasome. Autophagy . 2014;10(10):1761–1775. doi: 10.4161/auto.29647. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 139.Yang F., Wang Z., Wei X., et al. NLRP3 deficiency ameliorates neurovascular damage in experimental ischemic stroke. Journal of Cerebral Blood Flow & Metabolism . 2014;34(4):660–667. doi: 10.1038/jcbfm.2013.242. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 140.Sutterwala F. S., Haasken S., Cassel S. L. Mechanism of NLRP3 inflammasome activation. Annals of the New York Academy of Sciences . 2014;1319(1):82–95. doi: 10.1111/nyas.12458. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 141.Schroder K., Tschopp J. The inflammasomes. Cell . 2010;140(6):821–832. doi: 10.1016/j.cell.2010.01.040. [DOI] [PubMed] [Google Scholar]
- 142.Zhang S., Jiang L., Che F., Lu Y., Xie Z., Wang H. Arctigenin attenuates ischemic stroke via SIRT1-dependent inhibition of NLRP3 inflammasome. Biochemical and Biophysical Research Communications . 2017;493(1):821–826. doi: 10.1016/j.bbrc.2017.08.062. [DOI] [PubMed] [Google Scholar]
- 143.Mutoh T., Mutoh T., Taki Y., Ishikawa T. Therapeutic potential of natural product-based oral nanomedicines for stroke prevention. Journal of Medicinal Food . 2016;19(6):521–527. doi: 10.1089/jmf.2015.3644. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 144.Romano J. D., Tatonetti N. P. Informatics and computational methods in natural product drug discovery: a review and perspectives. Frontiers in Genetics . 2019;10:p. 368. doi: 10.3389/fgene.2019.0036. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 145.Kakkar V., Muppu S. K., Chopra K., Kaur I. P. Curcumin loaded solid lipid nanoparticles: an efficient formulation approach for cerebral ischemic reperfusion injury in rats. European Journal of Pharmaceutics and Biopharmaceutics . 2013;85(3):339–345. doi: 10.1016/j.ejpb.2013.02.005. [DOI] [PubMed] [Google Scholar]
- 146.Luo X., Li L., Xu W., Cheng Y., Xie Z. HLY78 attenuates neuronal apoptosis via the LRP6/GSK3β/β-catenin signaling pathway after subarachnoid hemorrhage in rats. Neuroscience Bulletin . 2020;36(10):1171–1181. doi: 10.1007/s12264-020-00532-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 147.Guo X., Zhang L., Fan Y., et al. Oxysterol-binding protein-related protein 8 inhibits gastric cancer growth through induction of ER stress, inhibition of Wnt signaling, and activation of apoptosis. Oncology Research . 2017;25(5):799–808. doi: 10.3727/096504016X14783691306605. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 148.Jin P., Qi D., Cui Y., Lenahan C., Deng S., Tao X. Activation of LRP6 with HLY78 attenuates oxidative stress and neuronal apoptosis via GSK3β/Sirt1/PGC-1α pathway after ICH. Oxidative Medicine and Cellular Longevity . 2022;2022:18. doi: 10.1155/2022/7542468.7542468 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 149.Hu L., Chen W., Tian F., Yuan C., Wang H., Yue H. Neuroprotective role of fucoxanthin against cerebral ischemic/reperfusion injury through activation of Nrf2/HO-1 signaling. Biomedicine & Pharmacotherapy . 2018;106:1484–1489. doi: 10.1016/j.biopha.2018.07.088. [DOI] [PubMed] [Google Scholar]
- 150.Zhang X. S., Lu Y., Tao T., et al. Fucoxanthin mitigates subarachnoid hemorrhage-induced oxidative damage via sirtuin 1-dependent pathway. Molecular Neurobiology . 2020;57(12):5286–5298. doi: 10.1007/s12035-020-02095-x. [DOI] [PubMed] [Google Scholar]
- 151.Zhang J., Xie X., Tang M., et al. Salvianolic acid B promotes microglial M2-polarization and rescues neurogenesis in stress-exposed mice. Brain, Behavior, and Immunity . 2017;66:111–124. doi: 10.1016/j.bbi.2017.07.012. [DOI] [PubMed] [Google Scholar]
- 152.Kim D. H., Park S. J., Kim J. M., et al. Cognitive dysfunctions induced by a cholinergic blockade and Aβ 25-35 peptide are attenuated by salvianolic acid B. Neuropharmacology . 2011;61(8):1432–1440. doi: 10.1016/j.neuropharm.2011.08.038. [DOI] [PubMed] [Google Scholar]
- 153.Zhang X., Wu Q., Lu Y., et al. Cerebroprotection by salvianolic acid B after experimental subarachnoid hemorrhage occurs via Nrf2- and SIRT1-dependent pathways. Free Radical Biology and Medicine . 2018;124:504–516. doi: 10.1016/j.freeradbiomed.2018.06.035. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 154.Li J. G., Lin J. J., Wang Z. L., et al. Melatonin attenuates inflammation of acute pulpitis subjected to dental pulp injury. American Journal of Translational Research . 2015;7(1):66–78. [PMC free article] [PubMed] [Google Scholar]
- 155.Amin A. H., El-Missiry M. A., Othman A. I. Melatonin ameliorates metabolic risk factors, modulates apoptotic proteins, and protects the rat heart against diabetes-induced apoptosis. European Journal of Pharmacology . 2015;747:166–173. doi: 10.1016/j.ejphar.2014.12.002. [DOI] [PubMed] [Google Scholar]
- 156.Galano A., Tan D. X., Reiter R. J. Melatonin as a natural ally against oxidative stress: a physicochemical examination. Journal of Pineal Research . 2011;51(1):1–16. doi: 10.1111/j.1600-079X.2011.00916.x. [DOI] [PubMed] [Google Scholar]
- 157.Ayer R. E., Sugawara T., Chen W., Tong W., Zhang J. H. Melatonin decreases mortality following severe subarachnoid hemorrhage. Journal of Pineal Research . 2008;44(2):197–204. doi: 10.1111/j.1600-079X.2007.00508.x. [DOI] [PubMed] [Google Scholar]
- 158.Zhao L., Liu H., Yue L., et al. Melatonin attenuates early brain injury via the melatonin receptor/Sirt1/NF-κB signaling pathway following subarachnoid hemorrhage in mice. Molecular Neurobiology . 2017;54(3):1612–1621. doi: 10.1007/s12035-016-9776-7. [DOI] [PubMed] [Google Scholar]
- 159.Sun Q., Wu W., Hu Y. C., et al. Early release of high-mobility group box 1 (HMGB1) from neurons in experimental subarachnoid hemorrhage in vivo and in vitro. Journal of Neuroinflammation . 2014;11(1):1–17. doi: 10.1186/1742-2094-11-106. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 160.Okada T., Kawakita F., Nishikawa H., Nakano F., Liu L., Suzuki H. Selective Toll-like receptor 4 antagonists prevent acute blood-brain barrier disruption after subarachnoid hemorrhage in mice. Molecular Neurobiology . 2019;56(2):976–985. doi: 10.1007/s12035-018-1145-2. [DOI] [PubMed] [Google Scholar]
- 161.Zhang X. H., Peng L., Zhang J., et al. Berberine ameliorates subarachnoid hemorrhage injury via induction of sirtuin 1 and inhibiting HMGB1/Nf-κB pathway. Frontiers in Pharmacology . 2020;11:p. 1073. doi: 10.3389/fphar.2020.01073. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 162.Han Y., Tong Z., Wang C., Li X., Liang G. Oleanolic acid exerts neuroprotective effects in subarachnoid hemorrhage rats through SIRT1-mediated HMGB1 deacetylation. European Journal of Pharmacology . 2021;893, article 173811 doi: 10.1016/j.ejphar.2020.173811. [DOI] [PubMed] [Google Scholar]
- 163.Jordán M. J., Lax V., Rota M. C., Lorán S., Sotomayor J. A. Relevance of carnosic acid, carnosol, and rosmarinic acid concentrations in the in vitro antioxidant and antimicrobial activities of Rosmarinus officinalis (L.) methanolic extracts. Journal of Agricultural and Food Chemistry . 2012;60(38):9603–9608. doi: 10.1021/jf302881t. [DOI] [PubMed] [Google Scholar]
- 164.Teng L., Fan L., Peng Y., et al. Carnosic acid mitigates early brain injury after subarachnoid hemorrhage: possible involvement of the SIRT1/p66shc signaling pathway. Frontiers in Neuroscience . 2019;13 doi: 10.3389/fnins.2019.00026. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 165.Ikemoto S., Sugimura K., Yoshida N., et al. Antitumor effects of Scutellariae radix and its components baicalein, baicalin, and wogonin on bladder cancer cell lines. Urology . 2000;55(6):951–955. doi: 10.1016/S0090-4295(00)00467-2. [DOI] [PubMed] [Google Scholar]
- 166.Cheng Y., Li S., Liu Y., Li J., Chen Y., Zhao H. Treatment of brain edema by wogonoside is associated with inhibition of neuronal apoptosis and SIRT1 activation in rats. Medical Science Monitor . 2020;26, article e921250 doi: 10.12659/MSM.921250. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 167.Wen J., Lin H., Zhao M., et al. Piceatannol attenuates D-GalN/LPS-induced hepatoxicity in mice: involvement of ER stress, inflammation and oxidative stress. International Immunopharmacology . 2018;64:131–139. doi: 10.1016/j.intimp.2018.08.037. [DOI] [PubMed] [Google Scholar]
- 168.Wang K. J., Zhang W. Q., Liu J. J., Cui Y., Cui J. Z. Piceatannol protects against cerebral ischemia/reperfusion-induced apoptosis and oxidative stress via the Sirt1/FoxO1 signaling pathway. Molecular Medicine Reports . 2020;22(6):5399–5411. doi: 10.3892/mmr.2020.11618. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 169.Nishimura M., Izumiya Y., Higuchi A., et al. Adiponectin prevents cerebral ischemic injury through endothelial nitric oxide synthase dependent mechanisms. Circulation . 2008;117(2):216–223. doi: 10.1161/CIRCULATIONAHA.107.725044. [DOI] [PubMed] [Google Scholar]
- 170.Scherer P. E., Williams S., Fogliano M., Baldini G., Lodish H. F. A novel serum protein similar to C1q, produced exclusively in adipocytes. Journal of Biological chemistry . 1995;270(45):26746–26749. doi: 10.1074/jbc.270.45.26746. [DOI] [PubMed] [Google Scholar]
- 171.Martínez-Klimova E., Aparicio-Trejo O. E., Tapia E., Pedraza-Chaverri J. Unilateral ureteral obstruction as a model to investigate fibrosis-attenuating treatments. Biomolecules . 2019;9(4) doi: 10.3390/biom9040141. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 172.Panigrahi M., Sadguna Y., Shivakumar B. R., et al. alpha-Lipoic acid protects against reperfusion injury following cerebral ischemia in rats. Brain Research . 1996;717(1-2):184–188. doi: 10.1016/0006-8993(96)00009-1. [DOI] [PubMed] [Google Scholar]
- 173.Gong A. G. W., Duan R., Wang H. Y., et al. Evaluation of the pharmaceutical properties and value of Astragali Radix. Medicines . 2018;5(2) doi: 10.3390/medicines5020046. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 174.Chen J. K., Guo M. K., Bai X. H., et al. Astragaloside IV ameliorates intermittent hypoxia-induced inflammatory dysfunction by suppressing MAPK/NF-κB signalling pathways in Beas-2B cells. Sleep Breath . 2020;24(3):1237–1245. doi: 10.1007/s11325-019-01947-8. [DOI] [PubMed] [Google Scholar]
- 175.Wang Y., Jiang F., Cheng H., et al. Astragaloside IV protects against oxidative stress in calf small intestine epithelial cells via NFE2L2-antioxidant response element signaling. International Journal of Molecular Sciences . 2019;20(24):p. 6131. doi: 10.3390/ijms20246131. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 176.Kang H. S., Lee J. Y., Kim C. J. Anti-inflammatory activity of arctigenin from Forsythiae Fructus. Journal of Ethnopharmacology . 2008;116(2):305–312. doi: 10.1016/j.jep.2007.11.030. [DOI] [PubMed] [Google Scholar]
- 177.Yang R., Shen Y. J., Chen M., et al. Quercetin attenuates ischemia reperfusion injury by protecting the blood-brain barrier through Sirt1 in MCAO rats. Journal of Asian Natural Products Research . 2022;24(3):278–289. doi: 10.1080/10286020.2021.1949302. [DOI] [PubMed] [Google Scholar]
- 178.Gong J., Sun F., Li Y., et al. Momordica charantia polysaccharides could protect against cerebral ischemia/reperfusion injury through inhibiting oxidative stress mediated c-Jun N-terminal kinase 3 signaling pathway. Neuropharmacology . 2015;91:123–134. doi: 10.1016/j.neuropharm.2014.11.020. [DOI] [PubMed] [Google Scholar]
- 179.Hu Z., Li F., Zhou X., et al. Momordica charantia polysaccharides modulate the differentiation of neural stem cells via SIRT1/Β-catenin axis in cerebral ischemia/reperfusion. Stem Cell Research & Therapy . 2020;11(1):1–14. doi: 10.1186/s13287-020-02000-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 180.Zhu H. R., Wang Z. Y., Zhu X. L., Wu X. X., Li E. G., Xu Y. Icariin protects against brain injury by enhancing SIRT1-dependent PGC-1alpha expression in experimental stroke. Neuropharmacology . 2010;59(1-2):70–76. doi: 10.1016/j.neuropharm.2010.03.017. [DOI] [PubMed] [Google Scholar]
- 181.Yang Y. L., Cheng X., Li W. H., Liu M., Wang Y. H., Du G. H. Kaempferol attenuates LPS-induced striatum injury in mice involving anti-neuroinflammation, maintaining BBB integrity, and down-regulating the HMGB1/TLR4 pathway. International Journal of Molecular Sciences . 2019;20(3):p. 491. doi: 10.3390/ijms20030491. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 182.Zhou Y. P., Li G. C. Kaempferol protects cell damage in in vitro ischemia reperfusion model in rat neuronal PC12 cells. BioMed Research International . 2020;2020:10. doi: 10.1155/2020/2461079.2461079 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 183.Xie W., Zhu T., Zhou P., et al. Notoginseng leaf triterpenes ameliorates OGD/R-induced neuronal injury via SIRT1/2/3-Foxo3a-MnSOD/PGC-1α signaling pathways mediated by the NAMPT-NAD pathway. Oxidative Medicine and Cellular Longevity . 2020;2020:15. doi: 10.1155/2020/7308386.7308386 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 184.Ip F. C., Ng Y. P., An H. J., et al. Cycloastragenol is a potent telomerase activator in neuronal cells: implications for depression management. Neurosignals . 2014;22(1):52–63. doi: 10.1159/000365290. [DOI] [PubMed] [Google Scholar]
- 185.Li M., Li S. C., Dou B. K., et al. Cycloastragenol upregulates SIRT1 expression, attenuates apoptosis and suppresses neuroinflammation after brain ischemia. Acta Pharmaceutica Sinica B . 2020;41(8):1025–1032. doi: 10.1038/s41401-020-0386-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 186.Fu B., Zhang J., Zhang X., et al. Alpha-lipoic acid upregulates SIRT1-dependent PGC-1α expression and protects mouse brain against focal ischemia. Neuroscience . 2014;281:251–257. doi: 10.1016/j.neuroscience.2014.09.058. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
No data were used to support this study.


