Abstract
OBJECTIVE
To evaluate changes in the incidence, prevalence, severity, and onset age of visual impairment (VI) due to diabetic retinopathy (DR) and compare these trends in the screening and treatment of diabetes during 40 years based on Finnish national register data.
RESEARCH DESIGN AND METHODS
We included people with VI with nonproliferative DR (NPDR; n = 2,490, 73% women) or proliferative DR (PDR; n = 2,026, 53% women) as the main diagnosis for VI during 1980–2019 in the Finnish Register of Visual Impairment. The number of patients with treated diabetes during 1986–2019 was obtained from the Social Insurance Institution of Finland registers based on reimbursed medication data.
RESULTS
The annual incidence of reported VI due to DR has decreased since it peaked in the 1990s: regarding NPDR, it decreased from 102.3 to 5.5 per 100,000 patients with treated diabetes between the 1990s and 2010s; regarding PDR, the respective change was from 39.9 to 7.4. The incidence of patients with diabetes treated for DR increased during this period. Annual prevalence of reported VI and differences between sexes steadily decreased in the 2000s and 2010s. The severity of reported VI has decreased, and the age at the onset of reported VI increased during the 40 years.
CONCLUSIONS
Prevalence and incidence of VI due to DR have dramatically decreased and shifted to older age during the 40 years despite the increasing prevalence of diabetes. These positive trends highlight the successful development and effectiveness of screening and therapies for diabetes and DR.
Introduction
Diabetic retinopathy (DR) is the leading cause of visual impairment (VI) and blindness among people of working age worldwide, but increasingly also among elderly people (1–4). It is classified into nonproliferative DR (NPDR) and proliferative DR (PDR). Both conditions are associated with the presence of diabetes. In 2015, the globally estimated number of people with diabetes was 415 million, and by 2040, the projected figure is >600 million (5).
In Europe, the prevalence of DR among patients with diabetes is 25.7% (6). The prevalence of DR is 54.4% in people with type 1 diabetes and is 25.0% in people with type 2 diabetes (6). In 2010, the percentage of patients blinded by DR among blind people was 3.9% in North America and 4.2% in Western Europe, and the percentage of patients with VI due to DR among people with VI was 2.8% and 3.0%, respectively (7). In 2020, the estimated global prevalence of VI and blindness due to DR was 1.4% and 2.5%, respectively (8).
A significant number of patients with type 2 diabetes consider loss of vision the worst complication of the disease (9,10). In fact, even mild to moderate VI has been associated with a negative impact on quality of life and mental health due to fear of vulnerability and loss of independence, self-care, and mobility (11,12). As the number and life expectancy of people living with diabetes increases in the future, the number of people with DR and consequently VI is expected to rapidly rise (3,13,14). Hence, there is a significant need for evaluating the changes in VI due to DR over time for public health issues and response.
While there are studies that have investigated the changes in the incidence and prevalence of VI due to DR in past decades (13,15–21), these studies have usually been limited to small study samples, short follow-up periods, specific study regions, clinical trials, and/or specific VI or blindness classes. To our knowledge, comprehensive nationwide studies with long, 30- to 40-year follow-up periods have not been published before that would assess changes in VI due to DR and trends in the screening and treatment of the disease. A previous Finnish study investigated trends in VI due to DR during 1982–2010 based on Finnish national register data (4). In this study, we extended this investigation by providing a comprehensive overview of the temporal trends in VI due to DR during the past 40 years, a period in which many developments in the screening, diagnosis, and treatment of both diabetes and DR have occurred.
Research Design and Methods
The Finnish Register of Visual Impairment (The Finnish Federation of the Visually Impaired) is a national register regulated by the Act (556/89) and Decree (774/89) on National Personal Records kept under the Health Care System. Health care providers, specialists in ophthalmology, and the ophthalmological units of hospitals are, under the above-mentioned Act, responsible to submit information on people with permanent VI to the register without need for permission from the patients. Between 1980 and 2019, the register included data on 58,822 people with VI. Registered data include eye diagnoses, home region, date of birth and death, year of VI onset, and classification of VI. The time at the onset of VI is determined based on the notification data, and if it does not exist, the date of registration is used instead. VI is classified according to the Finnish definitions of VI using visual acuity (VA) and visual field (VF) from central fixation (22), which are based on the definitions of World Health Organization (23) with a modification of the nomenclature of the names of the VI classes: 1) mild VI (0.3 > VA ≥ 0.1), 2) moderate VI (0.1 > VA ≥ 0.05), 3) severe VI (0.05 > VA ≥ 0.02; 10° > VF ≥ 5°), 4) nearly total blindness (0.02 > VA – 1/∞; VF < 5°), and 5) total blindness (VA = 0; no sense of light). The classification of VI is updated if any further information is notified.
Our study population included people with VI who had NPDR or PDR as the main diagnosis for VI based on the data of the Finnish Register of Visual Impairment. The classification of NPDR and PDR is based on the notification of the reporting ophthalmologist usually familiar with the patient’s history of the eye disease.
We estimated the annual number of patients with treated diabetes based on reimbursements for diabetes medication (data available 1986–2019) acquired from the Social Insurance Institution of Finland registers. To assess the changes in different diagnoses and therapies related to DR, we received the annual number of patients diagnosed with type 1 or 2 diabetes and related eye complications (codes E10, E10.3, E11, and E11.3 of the ICD-10) in Finland during 1998–2019 from the Care Registers for Social Welfare and Health Care kept by the Finnish Institute for Health and Welfare. However, the data on type 2 diabetes are an underestimation because the patients receiving treatment in public or private primary health care only were not registered in the Care Registers for Social Welfare and Health Care during the whole study period. Therefore, data on type 2 diabetes presents those complicated cases referred to special health care. Based on this diagnosis data, we evaluated the annual number of patients with diabetes treated with endophotocoagulation of the retina, vitreoretinal surgery, or intravitreal injections (based on the operation procedure codes of Finnish Hospital League 3623–3628, 3631, 3633, 3724, and the Nordic Medico-Statistical Committee [NOMESCO] Classification for Surgical Procedures codes CKD40, CKD65, CKD91–95, and CKD05) in Finland during 1986–2019 from hospital data kept by the Finnish Institute for Health and Welfare. Patients with diabetes diagnosed with wet type of age-related macular degeneration (ICD-10 code H35.31) were excluded from intravitreal injections data. The annual population of Finland and the age-specific life expectancies of the general population were provided by Statistics Finland. At the end of 2019, the population of Finland was 5,525,292 (50.6% females).
This study was conducted in line with the tenets of the Helsinki Declaration. Because this was a register-based study, ethical committee approval was not required according to the Finnish legislation.
NPDR and PDR data were analyzed separately. Annual incidence and prevalence rates were calculated based on the number of patients with treated diabetes or all inhabitants at the end of each year, stratified or adjusted for age and sex. We also calculated average annual rates on decade basis. The expected number of years with VI was calculated by subtracting the mean age at the onset of reported VI from the mean age at death in each decade. Because the age data were left-skewed, the Mann-Whitney U test was used for between-group comparisons and the Kruskal-Wallis test for multiple comparisons, adjusted with the Dunn-Bonferroni correction. Statistical differences in annual incidence and prevalence rates were calculated using linear regression when appropriate. The χ2 test was used for decade-based rates. The Fisher exact test was used for comparing VI distribution. Two-tailed P values <0.05 were considered statistically significant. All statistical analyses were performed using R 4.1.1 software (R Core Team, Foundation for Statistical Computing, Vienna, Austria) with the DescTools 0.99.43 package (24).
Results
Between 1980 and 2019, the Finnish Register of Visual Impairment included 2,490 people (73.2% women) with reported VI due to NPDR and 2,026 people (52.9% women) due to PDR. Among registered women, NPDR was more common than PDR in the 1980s (452 of 697, 64.8%), 1990s (922 of 1,332, 69.2%), and 2000s (345 of 610, 56.6%), but in the 2010s, PDR was the leading cause of VI (151 of 254, 59.4%). Among registered men, PDR was the leading cause of VI in all decades (198 of 316, 62.7%; 283 of 561, 50.4%; 292 of 491, 59.5%; 182 of 255, 71.4%). The percentage of mild VI due to NPDR and PDR increased during the 40 years, whereas the percentage of more severe VI classes decreased (Supplementary Table 1). There were no significant differences in the distribution of VI classes between sexes in any decade.
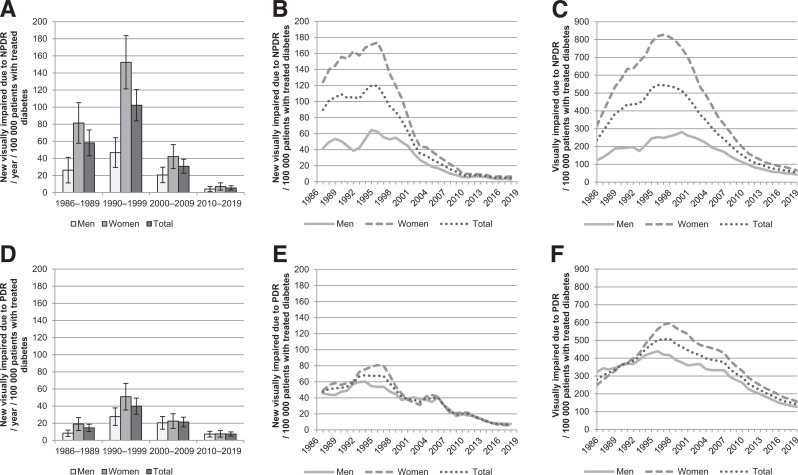
The incidence and prevalence of reported VI due to DR during the 40 years among patients with treated diabetes are shown in Fig. 1 and in total population in Supplementary Fig. 1. The incidence of reported VI due to both NPDR and PDR peaked in the 1990s in both sexes. The age- and sex-adjusted annual incidence of reported VI due to NPDR decreased from 102.3 (95% CI 84.0–120.6) per 100,000 patients with treated diabetes in the 1990s to 5.5 (95% CI 2.9–8.1) in the 2010s (P < 0.001). For PDR, the corresponding decrease was from 39.9 (95% CI 30.5–49.3) to 7.4 (95% CI 4.9–9.9; P < 0.001). In the total population (per 1,000,000 inhabitants), respective changes in incidence were from 22.8 (95% CI 18.7–26.8) to 3.0 (95% CI 1.6–4.5; P < 0.001) regarding NPDR and from 8.9 (95% CI 6.8–11.0) to 4.1 (95% CI 2.7–5.5; P < 0.001) regarding PDR. When incidences were observed for each year, the incidence of reported VI due to NPDR has gradually declined since 1996 and PDR since 2007 among patients with treated diabetes and in the total population. Women showed a higher incidence of reported VI due to both NPDR and PDR in the 1980s and 1990s (P < 0.001), but the difference between sexes has not been significant since.
Figure 1.
Reported VI due to NPDR and PDR among patients with treated diabetes during 1980–2019. A: Average annual incidence of reported VI due to NPDR per decade adjusted for age and/or sex (with 95% CIs). B: Annual incidence of reported VI due to NPDR smoothed using a 3-year central moving average. C: Annual prevalence of reported VI due to NPDR. D: Average annual incidence of reported VI due to PDR per decade adjusted for age and/or sex (with 95% CIs). E: Annual incidence of reported visual impairment due to PDR smoothed using a 3-year central moving average. F: Annual prevalence of reported visual impairment due to PDR.
The prevalence of reported VI due to NPDR has steadily decreased since 1996 among patients with treated diabetes and since 2000 in the total population, and due to PDR since 1998 among patients with treated diabetes and since 2007 in the total population. Women showed higher prevalence during the 40 years (P < 0.001) regarding NPDR, although the difference became smaller in the 2010s. No significant difference was found between sexes regarding PDR.
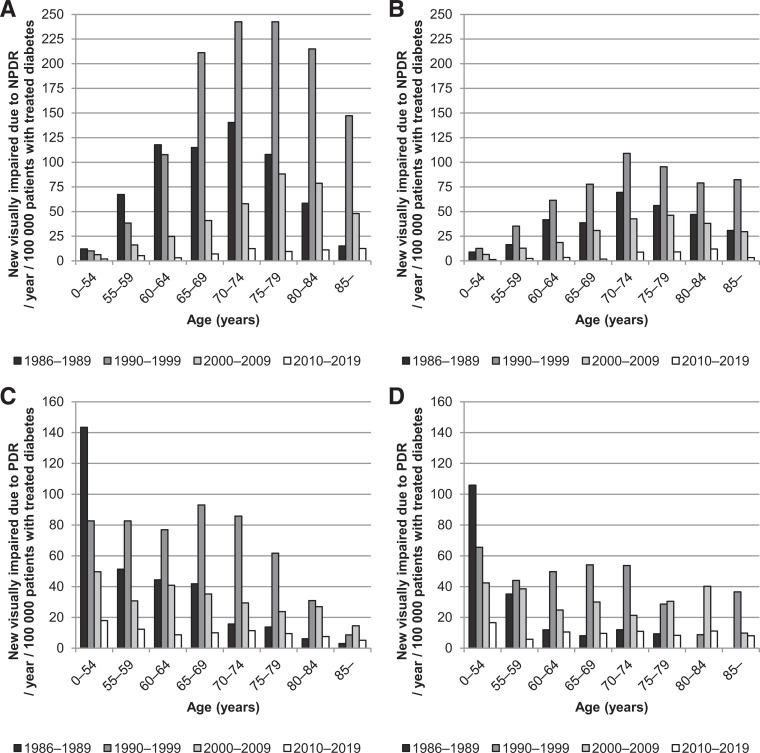
The incidence of reported VI due to DR by age among patients with treated diabetes is shown in Fig. 2 and in the total population in Supplementary Fig. 2. These figures show a shift to older age during the 40 years. Between the 1980s and the 2010s, the highest incidence of reported VI due to NPDR shifted from age-group 75–79 to ≥85 in women and from age group 70–74 to 80–84 in men compared with both patients with treated diabetes and the total population. Regarding PDR, the age shift was from 65–69 to 70–74 in women and 55–59 to 80–84 in men. This age shift is also demonstrated by a cumulative frequency of age at the onset of VI in Supplementary Fig. 3.
Figure 2.
Average annual incidence of reported VI due to NPDR and PDR per decade among patients with treated diabetes by age. A: NPDR, women. B: NPDR, men. C: PDR, women. D: PDR, men.
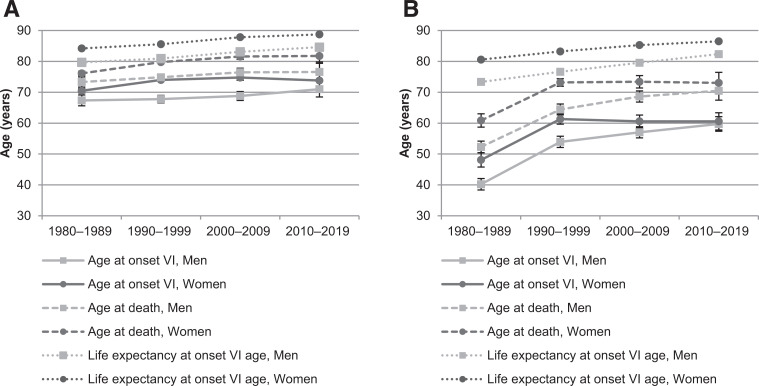
The age at the onset of reported VI and death by decade of onset VI is shown in Fig. 3 and more detailed in Supplementary Table 2. The mean age at the onset of reported VI due to NPDR and PDR increased significantly from the 1980s to the 2010s in both sexes. This increase was particularly noticeable in PDR, with almost a 20-year increase in men. The mean age at death also increased significantly during the 40 years in both sexes. Women had a significantly older mean age at the onset of reported VI in all decades except in the 2010s regarding both NPDR and PDR, as well as an older mean age at death in all decades, except in the 2010s regarding PDR.
Figure 3.
Age at the onset of reported VI due to NPDR (A) and PDR (B) and age at death with 95% CIs by decade of onset. For comparison, the life expectancy of the general population at the age of onset VI is shown.
There was no significant change in expected number of years with VI during the 40 years in neither NPDR nor PDR. However, the expected number of years with VI was significantly lower compared with the life expectancy of the general population at the age at the onset of reported VI in every decade (P < 0.001): regarding NPDR, the difference by each decade was 8.1, 5.8, 6.3, and 7.0 years in women and was 6.4, 6.1, 6.7, and 8.0 years in men, respectively; regarding PDR, the difference was 19.7, 10.0, 11.9, and 13.5 years in women and was 21.1, 12.2, 10.9, and 11.9 years in men.
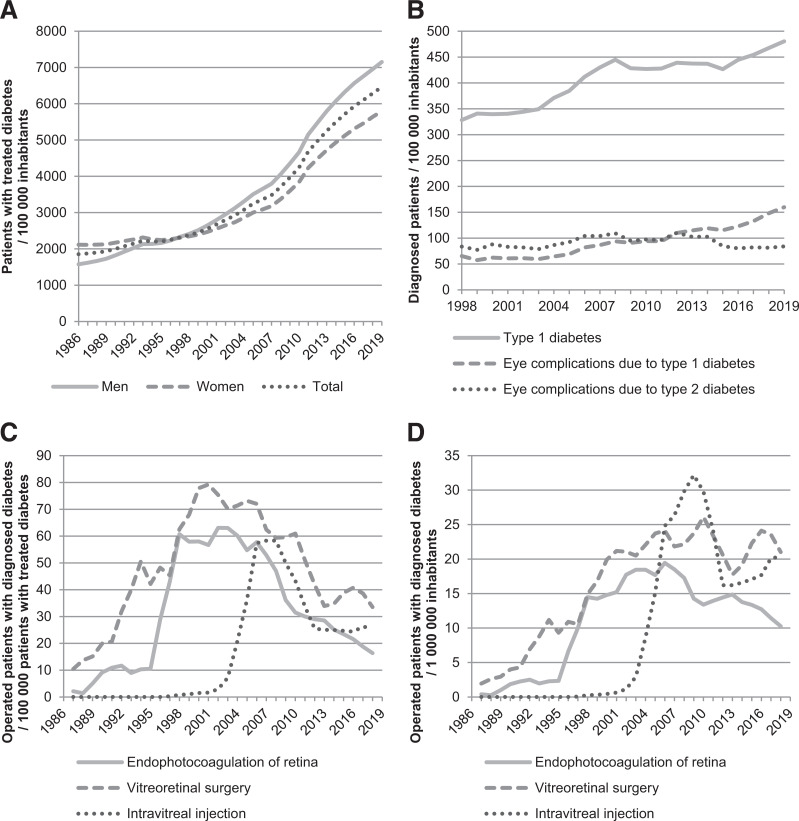
The annual prevalence of treated diabetes, incidence of diabetes and related eye complication diagnoses, and incidence of DR-related therapies are shown in Fig. 4. The annual prevalence of patients with treated diabetes per 100,000 inhabitants increased with each decade: 1,890, 2,225, 3,180, and 5,516. The incidence of people diagnosed with type 1 diabetes per 100,000 inhabitants increased from 328 in 1998 to 480 in 2019. The incidence of people diagnosed with eye complications due to type 1 diabetes per 100,000 inhabitants increased from 65 in 1998 to 160 in 2019, but regarding type 2 diabetes, the incidence was 84 in 1998 and has stayed relatively the same since. When different DR-related treatments were observed, the incidence of patients with diabetes treated with endophotocoagulation of the retina and vitreoretinal surgery increased gradually during the 1980s and 1990s, and intravitreal injections particularly in the late 1990s. During the 2000s and 2010s, these incidences stayed relatively the same or decreased among patients with treated diabetes and in the total population.
Figure 4.
Trends in diabetes diagnoses and treatment for DR. A: Annual prevalence of patients with treated diabetes in the total population during 1986–2019. B: Annual incidence of people diagnosed with diabetes and related eye complications in the total population during 1998–2019. C: Annual incidence of operated-on people with diagnosed diabetes among patients with treated diabetes during 1986–2019 smoothed using a 3-year central moving average. D: Annual incidence of operated-on people with diagnosed diabetes in total population during 1986–2019 smoothed using a 3 year central moving average.
Conclusions
Here we have shown that both the incidence and prevalence of VI due to DR have significantly decreased since the peak years in the 1990s despite the increased prevalence of diagnosed and treated diabetes. Even though DR has been previously associated with the working age population, a noticeable shift to older age has occurred during the 40 years. The severity of reported VI decreased during the same time period. Differences between sexes equalized in the 2000s and 2010s.
This study extended the previous report by Laatikainen et al. (4), who observed increased age at the time of VI notification, decreased severity of VI due to DR, and older age at death in people with VI during 1982–2010. These improvements mostly occurred in the 1990s.
Our study shows that both the incidence and prevalence of VI due to DR increased gradually in the 1980s and 1990s. This is likely explained by the increased prevalence of diabetes in Finland since the 1980s (14,25,26). Furthermore, the Social Insurance Institution of Finland register data show a threefold increase in the prevalence of patients with treated diabetes during the 40 years. However, the incidence and prevalence of VI due to DR started to decrease in the late 1990s.
The positive trends in the VI since the late 1990s are likely attributable to many factors. The treatment of diabetes was intensified in the late 1990s (27). In addition, Saramies et al. (14) reported that the proportion of undiagnosed diabetes in the Finnish population had decreased from 63 to 33% in 1996–2019. Furthermore, based on a population-based study of Finnish adults, the increase in the prevalence of hyperglycemia in previous decades had stagnated in the 2010s (28). The screening and treatment of DR have also improved since the 1990s, as indicated by the increased incidence of DR-related treatments among patients with treated diabetes in our data. The national screening program was intensified by the use of regular and standardized photographic methods. Intravitreal injections of steroids and, later in the 2000s, anti-vascular endothelial growth factor improved the prognosis of patients with diabetes (29,30). These, as well as timely laser therapy for DR and vitrectomy surgery for advanced DR, all contribute to the improved prevention of vision loss due to DR (2).
The changes in the incidence and prevalence of VI due to DR have considerably varied in previous studies, which have usually been limited to small study populations, short follow-up periods, or specific region of study. In Sweden, Bäcklund et al. (15) reported decreased incidence of blindness among patients with diabetes by 47% during 1981–1995 based on vision rehabilitation center data in Stockholm County. In Denmark, Hovind et al. (16) demonstrated in a clinic-based follow-up study consisting of 600 patients with type 1 diabetes during 1965–2000 that the VA was better in later cohorts than in earlier cohorts. In the U.S. in Wisconsin, Klein et al. (19) observed during a follow-up from 1980 to 2007 that the prevalence of VI was lower among people with type 1 diabetes diagnosed in more recent years. In Ireland, on the basis of a 10-year follow-up, the incidence of VI due to DR among diagnosed patients with diabetes registered in the National Council for the Blind of Ireland almost doubled between 2004 and 2013, whereas the incidence of blindness halved during the same period (20). In a systematic review of medical literature based on collected data from different countries between 1990 and 2010, Bourne et al. (7) reported that the estimated blindness due to DR remained unchanged in various high-income countries. In a meta-analysis of global scale, DR showed an increase in an estimated age-standardized prevalence between 1990 and 2020 worldwide, even though other vision-threatening eye diseases, such as age-related macular degeneration, decreased (8). Therefore, even though DR shows positive trends in Finland and other high-income countries, it continues to be a significant cause of VI worldwide.
The incidence of VI due to DR showed a shift to older age during the 40 years. This is further supported by the increased age at the onset of VI. This age shift is at least partly explained by the increasing prevalence of type 2 diabetes, which is more common among older people than type 1 diabetes in Finland, and the prevalence is likely to keep increasing due to the aging of the population and an increase of overweight and obesity in the population (26). As the treatment of diabetes and DR has improved and the life expectancy of patients with diabetes increases, VI is more likely to occur at later age among other age-associated vision-threatening diseases.
We reported that both the incidence and prevalence of VI due to DR were higher in women in the 1980s and 1990s, but these differences equalized in the 2000s and 2010s. This could be explained by the declining share of women among patients with diabetes that was observed during 1997–2007 in Finland (26). In global scale, the estimated prevalence of VI and blindness due to DR in 2020 was still higher in women (8).
The decline in the severity of VI due to DR during the past decades, as shown in this study, is likely associated with the declined rate of VI among patients with diabetes reported in previous studies. In Iceland, the proportion of legally blind patients with diabetes decreased from 2.4% in 1980 to 0.5% in 2005 (31,32). In the U.S. in Wisconsin, the estimated annual rate of any VI among patients with early-onset type 1 diabetes decreased from 1.2 in the 1980s to 0.3 in the early 2000s (33). They suggested that better glycemic and blood pressure control, as well as avoidance of smoking, likely contributed to these trends.
Even though the age at death has increased among DR patients during the 40 years, our data show that in the 2010s, the life expectancy among people with VI due to NPDR was still 7 years shorter and due to PDR 10 years shorter than in the general population. Similarly, Laatikainen et al. (4) reported that the standardized mortality ratios decreased among patients with VI due to DR between the 1980s and the 2000s, yet the mortality was still greater than in the general population. These adverse trends are likely attributable to the shorter life expectancy associated with diabetes, as patients with diabetes have an increased risk of life-threatening systemic vascular complications, such as stroke and heart failure (2,34).
All in all, these trends reflect the improvement and efficiency of the screening and treatment of DR during the past 40 years. Nevertheless, patients with DR are still at risk of VI and blindness. VA may not always improve above the mild vision loss level, and some patients with long-standing DR may end up becoming blind due to neuroretinal and pigment epithelial atrophy (35). Furthermore, the prognosis of treatment worsens the later the treatment begins during the course of DR (21). de Fine Olivarius et al. (36) reported that a significant vision loss can occur during 6 years after the diabetes diagnosis. Patients with diabetes also have an increased risk of other vision-threatening diseases, such as cataract and glaucoma (37). Hence, there is still a significant need to maintain and improve public awareness of vision-threatening complications of diabetes as well as systematic screening, early diagnosis, and prompt treatment of DR to reduce the magnitude of VI and blindness in patients with diabetes.
The strengths of our study include the large data set based on routinely collected health registers, thus ensuring that our results are generalizable to the population-level and comparable with those from studies in the other Western countries. The use of different registers made it possible to provide a comprehensive overview of changes in both DR and diabetes. In fact, the prevalence of diabetes in Finland is considered similar regardless of the data source (38). We had a unique opportunity to evaluate changes during a long, 40-year follow-up. The classification of VI is based on the Finnish national definitions and recommendations modified from the 1973 definitions by the World Health Organization that cover both decreased VA and VF constriction. These criteria remained the same during the entire 40-year period to ensure compatibility between decades, and the quality of the register data has been carefully followed. Therefore, the changes in the prevalence and incidence of VI caused by DR are likely to not reflect the changes in the notification methods.
Our study also has limitations. The reimbursement data for diabetes medicine do not cover patients with diabetes with diet treatment or people diagnosed while institutionalized. Hence, the prevalence of treated people with diabetes is not equivalent to the prevalence of diabetes, although we tried to improve the coverage of diabetes by providing diagnosis data from the Care Registers for Social Welfare and Health Care. Furthermore, in most cases, diabetes with diet treatment or without related medication is relatively mild and usually does not cause retinal complications (unpublished results from the Savitaipale study [14]). In 2017, the estimated number of patients with diabetes was 429,000 in the Finnish population aged ≥30 years, of which 48,000 (11.2%) were undiagnosed (28). Nevertheless, in most cases, the hidden diabetes form is also relatively mild and is not causing DR complications at the time of diagnosis. We could not cover the patients treated with laser treatments for DR as outpatients due to the development of the Care Registers for Social Welfare and Health Care during the first decades of the study. Also, the Current Care Recommendations in the Finnish Health care changed the practices of the doctors during the study period. As register data in general, the VI register data can have potential sources of biases, although not as remarkable as those in diabetes detection. These include difficulties in the estimation of the exact time point at which a person has become VI and when the disease itself has emerged, as well as the potential impact of other vision-threatening diseases. However, to minimize this bias, we analyzed only those patients whose main diagnosis causing VI was DR. The register may also lack information on specific populations, such as institutionalized people with dementia. Our data included predominantly people with a Finnish background; therefore, the results may not be directly applicable to other countries and ethnicities.
In conclusion, the incidence and prevalence of VI due to DR showed a gradual increase during the 1980s and 1990s but have since dramatically decreased despite the ever-increasing prevalence of diabetes. The severity of VI due to DR has decreased during the 40 years, and differences between sexes have equalized. Furthermore, the age at the onset of VI and age at death have increased in DR patients during the same period. These positive and encouraging trends underline the importance of efficient screening and timely treatment of diabetes and DR. In the future, more population-based studies with long follow-up periods in other countries could explore the situation in different regions of the world.
Article Information
Funding. This study was supported by Tampereen seudun Näkövammaisten tukisäätiö s.r, Tampere, Finland, and the Elsemay Björn Fund, Helsinki, Finland.
The funding sources did not influence the study design, data collection, analysis, interpretation, or writing of the publication. The decision of publishing the results was completely made by the authors.
Duality of Interest. No potential conflicts of interest relevant to this article were reported.
Author Contributions. P.K.M.P. conducted the statistical analysis. P.K.M.P., M.U.I.O., and M.G. investigated the data. P.K.M.P. and H.M.T.U wrote the manuscript. M.U.I.O. and M.G. reviewed the manuscript and contributed to discussion. P.K.M.P. is the guarantor of this work and, as such, had full access to all the data in the study and takes responsibility for the integrity of the data and the accuracy of the data analysis.
Footnotes
This article contains supplementary material online at https://doi.org/10.2337/figshare.20188460.
This article is featured in a podcast available at diabetesjournals.org/journals/pages/diabetes-core-update-podcasts.
References
- 1. Klein BE. Overview of epidemiologic studies of diabetic retinopathy. Ophthalmic Epidemiol 2007;14:179–183 [DOI] [PubMed] [Google Scholar]
- 2. Cheung N, Mitchell P, Wong TY. Diabetic retinopathy. Lancet 2010;376:124–136 [DOI] [PubMed] [Google Scholar]
- 3. Yau JW, Rogers SL, Kawasaki R, et al.; Meta-Analysis for Eye Disease (META-EYE) Study Group . Global prevalence and major risk factors of diabetic retinopathy. Diabetes Care 2012;35:556–564 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4. Laatikainen L, Ojamo M, Rudanko SL, et al. Improving visual prognosis of the diabetic patients during the past 30 years based on the data of the Finnish Register of Visual Impairment. Acta Ophthalmol 2016;94:226–231 [DOI] [PubMed] [Google Scholar]
- 5. Ogurtsova K, da Rocha Fernandes JD, Huang Y, et al. IDF Diabetes Atlas: global estimates for the prevalence of diabetes for 2015 and 2040. Diabetes Res Clin Pract 2017;128:40–50 [DOI] [PubMed] [Google Scholar]
- 6. Li JQ, Welchowski T, Schmid M, et al. Prevalence, incidence and future projection of diabetic eye disease in Europe: a systematic review and meta-analysis. Eur J Epidemiol 2020;35:11–23 [DOI] [PubMed] [Google Scholar]
- 7. Bourne RR, Jonas JB, Flaxman SR, et al. Vision Loss Expert Group of the Global Burden of Disease Study . Prevalence and causes of vision loss in high-income countries and in Eastern and Central Europe: 1990-2010. Br J Ophthalmol 2014;98:629–638 [DOI] [PubMed] [Google Scholar]
- 8. GBD 2019 Blindness and Vision Impairment Collaborators; Vision Loss Expert Group of the Global Burden of Disease Study . Causes of blindness and vision impairment in 2020 and trends over 30 years, and prevalence of avoidable blindness in relation to VISION 2020: the Right to Sight: an analysis for the Global Burden of Disease Study. Lancet Glob Health 2021;9:e144–e160 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9. Coyne KS, Margolis MK, Kennedy-Martin T, et al. The impact of diabetic retinopathy: perspectives from patient focus groups. Fam Pract 2004;21:447–453 [DOI] [PubMed] [Google Scholar]
- 10. Luckie R, Leese G, McAlpine R, et al.; DARTS/MEMO Collaboration . Fear of visual loss in patients with diabetes: results of the prevalence of diabetic eye disease in Tayside, Scotland (P-DETS) study. Diabet Med 2007;24:1086–1092 [DOI] [PubMed] [Google Scholar]
- 11. Taipale J, Mikhailova A, Ojamo M, et al. Low vision status and declining vision decrease health-related quality of life: results from a nationwide 11-year follow-up study. Qual Life Res 2019;28:3225–3236 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12. Purola PKM, Nättinen JE, Ojamo MUI, et al. Prevalence and 11-year incidence of common eye diseases and their relation to health-related quality of life, mental health, and visual impairment. Qual Life Res 2021;30:2311–2327 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13. Leasher JL, Bourne RR, Flaxman SR, et al.; Vision Loss Expert Group of the Global Burden of Disease Study . Global estimates on the number of people blind or visually impaired by diabetic retinopathy: a meta-analysis from 1990 to 2010. Diabetes Care 2016;39:1643–1649 [DOI] [PubMed] [Google Scholar]
- 14. Saramies J, Koiranen M, Auvinen J, et al. 22-year trends in dysglycemia and body mass index: a population-based cohort study in Savitaipale, Finland. Prim Care Diabetes 2021;15:977–984 [DOI] [PubMed] [Google Scholar]
- 15. Bäcklund LB, Algvere PV, Rosenqvist U. New blindness in diabetes reduced by more than one-third in Stockholm County. Diabet Med 1997;14:732–740 [DOI] [PubMed] [Google Scholar]
- 16. Hovind P, Tarnow L, Rossing K, et al. Decreasing incidence of severe diabetic microangiopathy in type 1 diabetes. Diabetes Care 2003;26:1258–1264 [DOI] [PubMed] [Google Scholar]
- 17. Younis N, Broadbent DM, Vora JP; Liverpool Diabetic Eye Study . Incidence of sight-threatening retinopathy in patients with type 2 diabetes in the Liverpool Diabetic Eye Study: a cohort study. Lancet 2003;361:195–200 [DOI] [PubMed] [Google Scholar]
- 18. Grauslund J, Green A, Sjølie AK. Blindness in a 25-year follow-up of a population-based cohort of Danish type 1 diabetic patients. Ophthalmology 2009;116:2170–2174 [DOI] [PubMed] [Google Scholar]
- 19. Klein R, Lee KE, Knudtson MD, Gangnon RE, Klein BE. Changes in visual impairment prevalence by period of diagnosis of diabetes: the Wisconsin Epidemiologic Study of Diabetic Retinopathy. Ophthalmology 2009;116:1937–1942 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20. Tracey ML, McHugh SM, Fitzgerald AP, Buckley CM, Canavan RJ, Kearney PM. Trends in blindness due to diabetic retinopathy among adults aged 18-69 years over a decade in Ireland. Diabetes Res Clin Pract 2016;121:1–8 [DOI] [PubMed] [Google Scholar]
- 21. Khan R, Chandra S, Rajalakshmi R, et al. Prevalence and incidence of visual impairment in patients with proliferative diabetic retinopathy in India. Sci Rep 2020;10:10513. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22. Ojamo M (Ed.). The Finnish Register of Visual Impairment – Annual Statistics 2019. Helsinki, Finnish Institute for Health and Welfare and Finnish Federation of the Visually Impaired, 2021. ISSN 1236-5114 [Google Scholar]
- 23. World Health Organization . The Prevention of Blindness: A Report of a WHO Study Group. Technical Report Series No 518 [meeting held in Geneva from 6 to 10 November 1972]. Geneva, World Health Organization, 1973. Accessed 7 November 2021. Available from https://apps.who.int/iris/handle/10665/38222
- 24. Signorell A, Aho K, Alfons A, et al. DescTools: Tools for Descriptive Statistics. R package version 0.99.43, 2021. Accessed 7 November 2021. Available from https://cran.r-project.org/package=DescTools
- 25. Harjutsalo V, Sjöberg L, Tuomilehto J. Time trends in the incidence of type 1 diabetes in Finnish children: a cohort study. Lancet 2008;371:1777–1782 [DOI] [PubMed] [Google Scholar]
- 26. Sund R, Koski S. FinDM II. On the register-based measurement of the prevalence and incidence of diabetes and its long-term complications. A technical report. Tampere, Finnish Diabetes Association, 2009 [Google Scholar]
- 27. Nathan DM, Genuth S, Lachin J, et al.; Diabetes Control and Complications Trial Research Group . The effect of intensive treatment of diabetes on the development and progression of long-term complications in insulin-dependent diabetes mellitus. N Engl J Med 1993;329:977–986 [DOI] [PubMed] [Google Scholar]
- 28. Koponen P, Borodulin K, Lundqvist A, Sääksjärvi K, Koskinen S. Health, functional capacity and welfare in Finland – FinHealth 2017 study. Helsinki, Finnish Institute for Health and Welfare, Report 4/2018. Accessed 7 November 2021. Available from https://urn.fi/URN:ISBN:978-952-343-105-8
- 29. Suomalaisen Lääkäriseuran Duodecimin; Suomen Silmälääkäriyhdistyksen; Diabetesliiton Lääkärineuvoston Asettama Työryhmä . Diabeettinen retinopatia. Duodecim 2006;122:1388–1401 [in Finnish] [PubMed] [Google Scholar]
- 30. Suomalaisen Lääkäriseuran Duodecimin, Diabetesliiton lääkärineuvoston ja Suomen sisätautilääkärien yhdistyksen asettama työryhmä . Diabeteksen Käypä hoito -suositus. Duodecim 2007;123:1490–1520 [in Finnish] [PubMed] [Google Scholar]
- 31. Stefánsson E, Bek T, Porta M, Larsen N, Kristinsson JK, Agardh E. Screening and prevention of diabetic blindness. Acta Ophthalmol Scand 2000;78:374–385 [DOI] [PubMed] [Google Scholar]
- 32. Zoega GM, Gunnarsdóttir T, Björnsdóttir S, Hreietharsson AB, Viggósson G, Stefánsson E. Screening compliance and visual outcome in diabetes. Acta Ophthalmol Scand 2005;83:687–690 [DOI] [PubMed] [Google Scholar]
- 33. Klein R, Lee KE, Gangnon RE, Klein BE. The 25-year incidence of visual impairment in type 1 diabetes mellitus: the Wisconsin Epidemiologic Study of Diabetic Retinopathy. Ophthalmology 2010;117:63–70 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34. Magliano DJ, Shaw JE, Shortreed SM, et al. Lifetime risk and projected population prevalence of diabetes. Diabetologia 2008;51:2179–2186 [DOI] [PubMed] [Google Scholar]
- 35. Hansson-Lundblad C, Holm K, Agardh CD, Agardh E. A small number of older type 2 diabetic patients end up visually impaired despite regular photographic screening and laser treatment for diabetic retinopathy. Acta Ophthalmol Scand 2002;80:310–315 [DOI] [PubMed] [Google Scholar]
- 36. de Fine Olivarius N, Siersma V, Almind GJ, Nielsen NV. Prevalence and progression of visual impairment in patients newly diagnosed with clinical type 2 diabetes: a 6-year follow up study. BMC Public Health 2011;11:80. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37. Chiang PP, Lamoureux EL, Zheng Y, et al. Frequency and risk factors of non-retinopathy ocular conditions in people with diabetes: the Singapore Malay Eye Study. Diabet Med 2013;30:e32–e40 [DOI] [PubMed] [Google Scholar]
- 38. Laatikainen T, Koponen P, Reinikainen J, et al. Monitoring, assessment and prediction of public health: what type of information can be obtained in Finland from care registers and what from population studies? Suom Lääkäril 2020;37:1853–1858 [Google Scholar]