Abstract
Background:
Hospitalizations related to the consequences of opioid use are rising. National guidelines directing in-hospital opioid use disorder (OUD) management do not exist. OUD treatment guidelines intended for other treatment settings could inform in-hospital OUD management.
Purpose:
Evaluate the quality and content of existing guidelines for OUD treatment and management.
Data Sources:
OVID MEDLINE, PubMed, Ovid PsychINFO, EBSCOhost CINHAL, ERCI Guidelines Trust, websites of relevant societies and advocacy organizations, and selected international search engines.
Study Selection:
Guidelines published between January 2010 to June 2020 addressing OUD treatment, opioid withdrawal management, opioid overdose prevention, and care transitions among adults.
Data Extraction:
We assessed quality using the Appraisal of Guidelines for Research and Evaluation (AGREE) II instrument.
Data Synthesis:
Nineteen guidelines met selection criteria. Most recommendations were based on observational studies or expert consensus. Guidelines recommended use of non-stigmatizing language among patients with OUD; to assess patients with unhealthy opioid use for OUD using Diagnostic Statistical Manual of Diseases–5th Edition criteria; use of methadone or buprenorphine to treat OUD and opioid withdrawal; use of multimodal, non-opioid therapy, and when needed, short-acting opioid analgesics in addition to buprenorphine or methadone, for acute pain management; ensuring linkage to ongoing methadone or buprenorphine treatment; referring patients to psychosocial treatment; and ensuring access to naloxone for opioid overdose reversal.
Conclusions:
Included guidelines were informed by studies with various levels of rigor and quality. Future research should systematically study buprenorphine and methadone initiation and titration among people using fentanyl and people with pain, especially during hospitalization.
Keywords: opioid use disorder, hospitalization, buprenorphine, methadone, systematic review
Introduction
In 2019, an estimated 1.6 million Americans aged 12 years or older suffered from an opioid use disorder (OUD).1 In 2020, unintentional injuries, including drug-related overdose, increased by almost 30% from 2019, and were a leading cause of death in the United States.2,3 Hospitalizations related to the consequences of opioid use are also rising. On average, from 2014 to 2018, 1 in 9 hospitalized patients had a substance use disorder.4 In one statewide study of hospitalized patients with OUD, 7.8% of people died within one year, reflecting mortality similar to patients hospitalized with acute coronary syndrome (who have 5–9% one-year mortality).5
When taken regularly, medications for OUD (MOUD), including methadone and buprenorphine, are associated with an approximately 50% reduction in mortality.6,7 Despite their effectiveness, MOUD are vastly underutilized across the United States with access varying by geography,8,9 insurance status,10,11 treatment location,12–14 and race/ethnicity.10,15 Hospitalization offers an opportunity to provide treatment to hospitalized patients with OUD and hospitalists are uniquely positioned to provide this life-saving care.16
To our knowledge there are no existing national guidelines directing OUD treatment, opioid withdrawal management, overdose prevention, and care transitions among hospitalized adults. Given the overwhelming morbidity and mortality related to the opioid crisis, the Society of Hospital Medicine (SHM) sought to develop a consensus statement to assist inpatient clinicians caring for hospitalized patients with OUD. To inform the development of the consensus statement, SHM convened a working group to systematically review existing guidelines on OUD screening, assessment, and treatment, opioid withdrawal management, opioid overdose prevention, and care transitions. This article describes the methods and results of our systematic review of existing guidelines for OUD management. The consensus statement derived from these existing guidelines, applied to the hospital setting, appears in a companion article.
Methods
The reporting of this systematic review was guided by the standards of the Preferred Reporting Items for Systematic Review and Meta-Analysis (PRISMA) Statement.17 Steps in the systematic review process included: 1) conducting a search of relevant guidelines, 2) applying exclusion criteria, 3) assessing the quality of the guidelines, and 4) iteratively synthesizing guideline recommendations to identify issues potentially relevant to hospitalized patients with OUD. Details of the protocol for this systematic review were registered on PROSPERO accessible at: https://www.crd.york.ac.uk/prospero/display_record.php?RecordID=204849.
Data Sources, Process, and Guideline Screening
SLC worked with a librarian scientist to develop search teams and SLC conducted the search in July 2020. We limited the search to guidelines published in English between January 2010 to June 2020 since guidelines published before 2010 may contain outdated information.18 We used EndNote™19 to collect citations and remove duplicates. Information sources included Ovid MEDLINE, MEDLINE via PubMed, Ovid PsychINFO, EBSCOhost CINHAL, and ERCI Guidelines Trust. We identified additional records through relevant society websites, organizations, and international guideline search engines (see Figure 1; Appendix for search terms). SLC manually reviewed all article titles. If the title clearly suggested the article was not a guideline, the article was excluded. If the title did not clearly describe the content, then the abstract was reviewed. If the abstract did not clearly describe the content, then the full article text was reviewed.
Figure 1. PRISMA Flow Diagram.
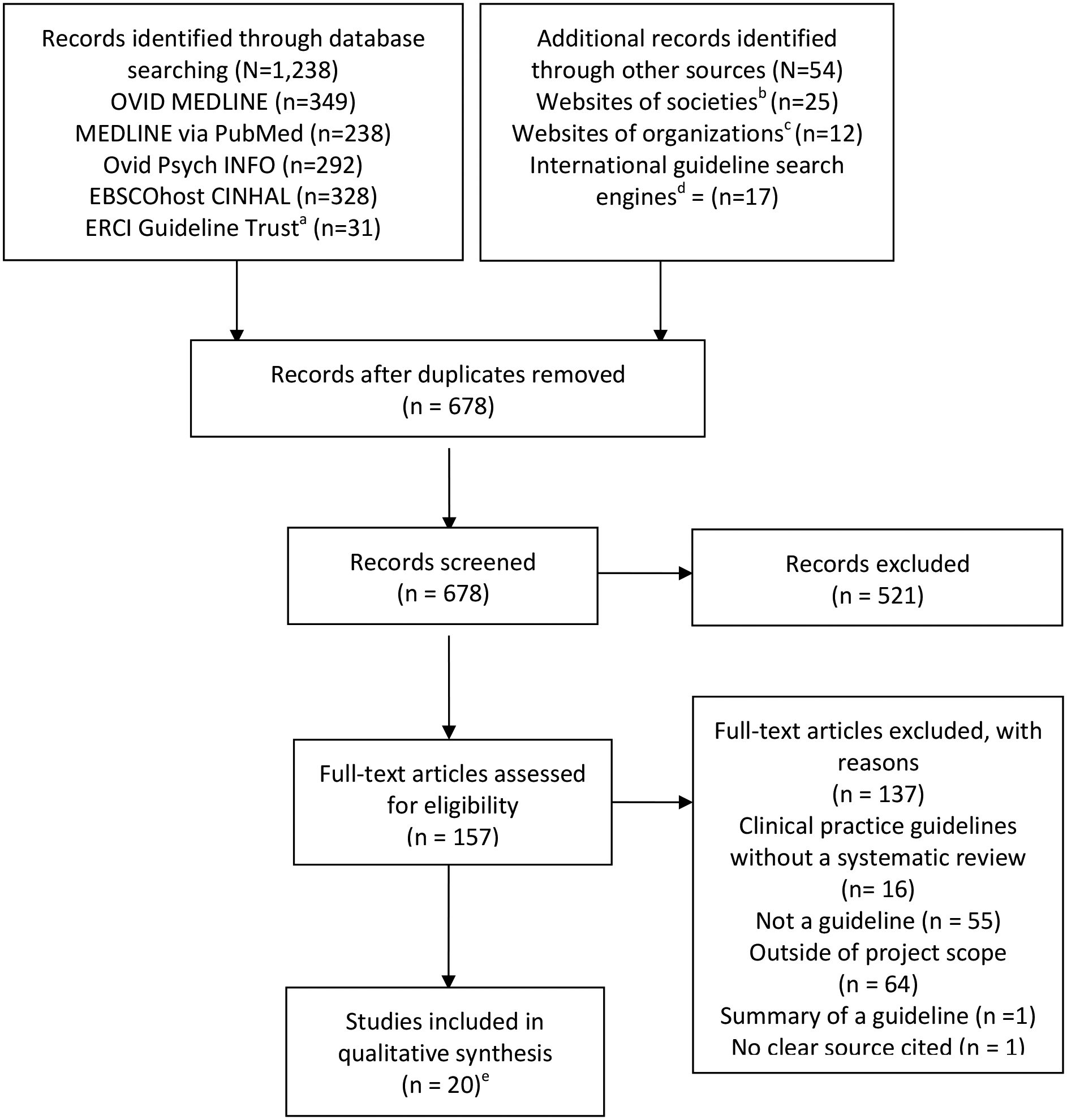
Summary of evidence search and selection.
a ERCI: Emergency Care Research Institute
b American Society of Addiction Medicine; California Society of Addiction Medicine; American College of Academic Addiction Medicine; American Academy of Addiction Psychiatry; American Academy of Family Physicians; American Academy of Pain Physicians; American College of Physicians; American Society of Hospital Medicine; American Society of Interventional Pain Physicians; American Cancer Society; American Society of Clinical Oncology; American Society of Clinical Oncology; American Psychiatric Association; Eastern Association for the Surgery of Trauma; National Pain Centre
c Substance Abuse and Mental Health Services Administration; United States Preventive Services Task Force; Centers for Disease Control and Prevention; Michigan Department of Health and Human Services; American Hospital Association; Magellan Healthcare; VA/DOD
d National Institute for Health and Care Excellence [NICE]; Canadian Medical Association Clinical Practice Guidelines Infobase; Australian Government National Health and Medical Research Council; Australian Clinical Practice Guidelines Portal Web; World Health Organization; New Zealand Ministry of Health Manatū Hauora
e Two studies included an executive summary of guidelines and were reviewed as a whole (ASAM 2015; CRISM); one guideline included a guideline and a separate publication outlining the systematic review methodology (USPSTF), also reviewed as a whole
Guideline Inclusion/Exclusion Criteria
We define guidelines as “statements that include recommendations intended to optimize patient care informed by a systematic review of evidence and an assessment of the benefits and harms of alternative care options”, consistent with the National Academies’ definition.20 To be eligible for inclusion, guidelines had to include recommendations on the assessment or management of OUD, opioid withdrawal, or overdose prevention, or the management of these conditions across care transitions. We included guidelines intended for emergency departments, hospitals, or other primary care or urgent care settings to which patients with OUD may present for medical care. We excluded guidelines intended for opioid treatment programs (OTP) which dispense methadone for OUD and guidelines derived entirely from another guideline. We excluded guidelines exclusively focused on other substances including alcohol, cannabis, and stimulant use, and guidelines specific to pregnant people, children, and adolescents with OUD.
Guideline Quality Assessment
We used the Appraisal of Guidelines for Research and Evaluation (AGREE) II instrument21–23 to evaluate the quality of each guideline selected for inclusion. The AGREE II instrument includes 23 statements spanning six domains: scope and purpose; stakeholder involvement; rigor of development; clarity of presentation; applicability; and editorial independence.21–23 Each guideline was rated by three appraisers (RB, MM, HE, ZW, MBW, EL, MVR, SH, TZ, MU, AP, KTL, MB, MTO, HS) who indicated the degree to which they agreed with each of the 23 statements using a scale from 1 (strongly disagree) to 7 (strongly agree) (Table 1) (see Supplementary Material, Table 1 for titles and affiliations of AGREE II reviewers). They also rated the overall quality of each guideline using the same scale while indicating whether or not they would recommend the guideline for use (Table 2). We report scaled domain scores as percentages and calculated an overall assessment score (Table 1).
Table 1.
Scaled Domain Scores Across Domains of the AGREE II Instrument, and Overall Assessment Scores
| Guidelinea | AGREE II Domainb | Overall Assessmentc | |||||
|---|---|---|---|---|---|---|---|
| Scope & Purpose | Stakeholder Involvement | Rigor of Development | Clarity of Presentation | Applicability | Editorial Independence | ||
| CCSMH | 100% | 78% | 85% | 93% | 74% | 97% | 6 |
| ACP | 54% | 39% | 41% | 54% | 53% | 61% | 4.67 |
| WFSBP | 70% | 54% | 65% | 76% | 50% | 56% | 5 |
| ASAM 2020 | 94% | 87% | 84% | 94% | 53% | 92% | 6 |
| CFPC | 91% | 72% | 83% | 91% | 62% | 86% | 5.67 |
| AUS | 89% | 78% | 57% | 78% | 74% | 64% | 4.67 |
| CRISM | 94% | 97% | 78% | 97% | 55% | 100% | 6.67 |
| APS/CPDD | 93% | 69% | 86% | 87% | 51% | 100% | 5.33 |
| ASAM 2011 | 63% | 63% | 40% | 81% | 68% | 36% | 4.33 |
| NSW | 93% | 70% | 15% | 87% | 57% | 60% | 4 |
| BC | 100% | 85% | 92% | 100% | 94% | 81% | 6.67 |
| ASERPQI | 100% | 43% | 66% | 91% | 69% | 83% | 6 |
| ASAM 2015 | 94% | 98% | 88% | 97% | 75% | 79% | 6.33 |
| PAIN | 100% | 96% | 95% | 96% | 38% | 97% | 5.67 |
| USPSTF | 94% | 54% | 74% | 91% | 61% | 97% | 5.8 |
| RAND | 63% | 37% | 47% | 70% | 54% | 22% | 2.3 |
| VA/DoD | 91% | 91% | 89% | 94% | 68% | 89% | 6.67 |
| NICE | 100% | 94% | 94% | 96% | 94% | 31% | 6.67 |
| UK | 91% | 96% | 44% | 57% | 61% | 44% | 3.67 |
| PEER | 96% | 80% | 73% | 98% | 24% | 92% | 5.33 |
CCSMH = Canadian Coalition of Seniors’ Mental Health;26 ACP = American College of Physicians;47 WFSBP = World Federation of Societies of Biological Psychiatry;39 ASAM 2020 = American Society of Addiction Medicine, 2020;37 CFPC = College of Family Physicians of Canada;43 AUS = Commonwealth of Australia;36 CRISM = Canadian Research Initiative Substance Misuse;33 APS/CPDD = American Pain Society, the College on Problems of Drug Dependence, and the Heart Rhythm Society;48 ASAM 2011 = American Society of Addiction Medicine, 2011;42 NSW = New South Wales;38 BC = British Columbia;25 ASERPQI = American Society for Enhanced Recovery and Perioperative Quality Initiative;46 ASAM 2015 = American Society of Addiction Medicine, 2015;31 PAIN = Perioperative Pain and Addiction Interdisciplinary Network;45 USPSTF = United States Preventive Services Task Force;18 RAND = Research and Development Corporation;41 VA/DoD = Department of Veterans Affairs, Department of Defense;40 NICE = National Institute for Healthcare Excellence;30 UK = United Kingdom;34 PEER = Patients, Experience, Evidence, Research Group27
Each individual item within a domain was rated on a Likert scale with a maximum of 7 points. The scores were averaged across three appraisers. The scaled domain score is calculated as follows: (obtained score [sum of the mean scores for the individual items within a domain] – minimum possible score) / (maximum possible score – minimum possible score).
Mean score on a scale of 1 – 7.
Table 2.
AGREE II Overall Recommendation for Use
| Yes | Yes, with modification | No | |
|---|---|---|---|
| CCSMH | 3 | 0 | 0 |
| ACP | 2 | 0 | 1 |
| WFSBP | 2 | 1 | 0 |
| ASAM 2020 | 2 | 1 | 0 |
| CFPC | 2 | 0 | 1 |
| AUS | 1 | 1 | 1 |
| CRISM | 3 | 0 | 0 |
| APS/CPDD | 2 | 1 | 0 |
| ASAM 2011 | 1 | 2 | 0 |
| NSW | 1 | 1 | 1 |
| BC | 2 | 1 | 0 |
| ASERPQI | 2 | 1 | 0 |
| ASAM 2015 | 3 | 0 | 0 |
| PAIN | 1 | 2 | 0 |
| USPSTF | 3 | 0 | 0 |
| RANDa | 0 | 0 | 3 |
| VA/DoD | 3 | 0 | 0 |
| NICE | 3 | 0 | 0 |
| UK | 0 | 1 | 2 |
| PEER | 1 | 2 | 0 |
Excluded following the AGREE II appraisal and recommendation process
Guideline Synthesis and Analysis
One author (SLC) formulated an a priori draft of key topics pertinent to the management of OUD, opioid withdrawal, and opioid overdose prevention in hospitalized adults, informed by the American Society of Addiction Medicine (ASAM) National Practice Guidelines.24,25 Members of our Working Group (RB, MM, HE, XW, MBW, EL, SH; see Supplementary Material, Table 1 for title and affiliations of Working Group members) met to review and discuss each drafted key topics, and to provide iterative feedback to refine and finalize the key topics. Using these key topics as a framework, we extracted recommendations from each guideline on the following topics: 1) non-stigmatizing medical communication and language for people who use opioids, 2) assessment of unhealthy opioid use and diagnosis of OUD, 3) MOUD for Diagnostic Statistical Manual of Diseases 5th Edition (DSM–5) confirmed diagnosis, 4) acute pain or management in the setting of OUD, and 5) care transitions.
Role of the Funding Source
SHM provided administrative and material support for the project but had no role in the design or execution of the scientific evaluation.
Results
We identified 678 unique records for screening from which we identified 20 guidelines meeting selection criteria (see Figure 1). Some guidelines were intended for the outpatient or community settings,26–31 other guidelines were intended across the spectrum of the inpatient and outpatient settings.25,32–40 Three guidelines were specific to buprenorphine for OUD treatment41–43 and two guidelines were specific to perioperative pain management.44–46 One guideline was a policy statement,47 one guideline was specific to methadone safety,48 and one guidelines was a U.S. Preventive Service Task Force (USPSTF) Screening for Unhealthy Drug Use recommendation49,50 No guidelines were specific to the hospital or emergency department settings.
Guideline Quality Assessment
See Table 1 for AGREE II scaled domain scores and for the ratings on each individual item within a domain. The range of scaled domain scores for each of the AGREE II domains were: scope and purpose 54% – 100%, stakeholder involvement 39% – 97%, rigor of development 41% – 95%, clarity of presentation 54% – 100%, applicability 38% – 94%, and editorial independence 31% – 100%. Overall guideline assessment scores ranged from 2.3 to 6.67 on a scale of 1 – 7. Table 2 lists recommendations for inclusion based upon AGREE II scores. Six guidelines were recommended for use without modification by all three appraisers,25,27,31,33,40,49 three guidelines were recommended for use with modification by two of three appraisers,28,42 and one guideline was not recommended for use by all three appraisers.41 Three guidelines had the highest score of 6.67 and included the Canadian Research Initiative in Substance Misuse (CRISM) National Guideline for the Clinical Management of OUD,33 the National Institute for Health and Care Excellence (NICE) Needle and Syringe Programmes,31 and the U.S. Department of Veterans Affairs/Department of Defense (VA/DoD) Clinical Practice Guideline for Management of Substance Use Disorders.40 Following completion of the AGREE II appraisals, 19 of the original 20 guidelines were included in our recommendation synthesis.
Table 3 includes the level of evidence reviewed for each guideline based on inclusion of evidence from randomized controlled trials or nonrandomized controlled trials; observational studies; expert consensus; or partially based on other guidelines. Each guideline included a systematic review of the literature, however the New South Wales Clinical Guidelines,38 the Australian Clinical Guidelines,36 and the United Kingdom Clinical Guidelines34 did not clearly link each recommendation to the underlying evidence base. When citations were present, we reviewed them to determine the type of data upon which the recommendations were based and included this information in Table 3. Due to the relative paucity of evidence from randomized controlled trials on many of the topics covered, the majority of the recommendations in Table 3 are based on observational studies, expert opinion and consensus, or other guidelines.
Table 3.
Selected Recommendations for Opioid Use Disorder Screening, Treatment, Overdose Prevention, and Care Transitions by Hospitalists
| Recommendation | Guideline | |||||
|---|---|---|---|---|---|---|
| BC1 | ASERPQI2 | ASAM 20153,4 | PAIN5,6 | USPSTF7,8 | VA/DoD9 | |
| Medical communication and language for people who use substances | ||||||
| Use non-stigmatizing and person-first language | ✓c,d | ✓c,d | ✓c,d | |||
| Assessment of unhealthy opioid use and diagnosis of opioid use disorder (OUD) | ||||||
| If unhealthy opioid use, assess for OUD; review PDMP | ✓c,d | ✓c,d | ✓a | ✓c,d | ||
| Use DSM-5 to diagnose OUD | ✓c,d | ✓c,d | ✓d | |||
| Offer HIV, hepatitis A, B, C, syphilis, pregnancy test, and urine drug analysis | ✓c,d | ✓c,d | ✓d | |||
| OUD medication for DSM – 5 confirmed diagnosis | ||||||
| Use shared decision making to medications for OUD (MOUD) including buprenorphine or methadone | ✓c,d | ✓c,d | ✓b.c,d | |||
| Offer MOUD to treat opioid withdrawal and OUD; use COWS10 to initiate buprenorphine | ✓c,d | ✓b.c,d | ✓a,d | |||
| Consider review of electrocardiogram | ✓c,d | ✓b,c,d | ✓c,d | |||
| Initiate methadone at 20 to 30 milligrams | ✓c,d | ✓c,d | ✓c,d | |||
| Use non-opioid adjunctive medications for withdrawal | ✓c,d | ✓a.b,c,d | ✓a,d | |||
| When applicable, offer intramuscular naltrexone | ✓a,b,c,d | ✓c,d | ✓a,b | |||
| Acute pain and/or perioperative pain management in setting of OUD | ||||||
| Assess and treat acute pain in the setting of OUD | ✓c,d | ✓c,d | ✓c | |||
| Continue MOUD in the perioperative period | ✓b,c | ✓b,c | ||||
| →Use short-acting opioids + adjunct analgesia in the perioperative period | ✓a,b,c | ✓c,d | ✓b,c | |||
| Care transition at hospital discharge | ||||||
| Obtain an X-Waiver | ✓e | ✓e | ||||
| Refer to a buprenorphine prescriber or OTP | ✓c,d | ✓a,c,d | ✓a,d | |||
| Refer to psychosocial and mental health treatment, mutual support groups, and harm reduction services | ✓c,d | ✓b,c,d | ✓a,b,d | |||
| Post-acute care facilities must continue MOUD | ||||||
| Prescribe/dispense naloxone at discharge | ✓c,d | ✓a,c,d | ||||
| Recommendation | Guideline | |||||
|---|---|---|---|---|---|---|
| NICE11,12 | UK13 | PEER14,15 | CCSMH16 | ACP17 | WFSBP18 | |
| Medical communication and language for people who use substances | ||||||
| Use non-stigmatizing and person-first language | ✓c,d | ✓c,d | ✓c | |||
| Assessment of unhealthy opioid use and diagnosis of OUD | ||||||
| If unhealthy opioid use, assess for OUD; review PDMP | ✓c,d | ✓a,d | ✓b,d | ✓a,b,c,d | ✓c | |
| Use DSM-5 to diagnose OUD | ||||||
| Offer HIV, hepatitis A, B, C, syphilis, pregnancy test, and urine drug analysis | ✓b,c,d | ✓c,d | ✓b,d | ✓c,d | ✓c | |
| OUD medication for DSM – 5 confirmed diagnosis | ||||||
| Use shared decision making to initiate MOUD, including buprenorphine or methadone | ✓c,d | ✓b,d | ✓b,c,d | ✓c | ✓a,b,c | |
| Offer MOUD as first line agents to treat opioid withdrawal and OUD; use COWS10 to initiate buprenorphine | ✓c,d | ✓c,d | ✓b,d | ✓b,c,d | ✓c | ✓c,d |
| Consider review of electrocardiogram | ✓c,d | ✓c | ||||
| Initiate methadone at 20 to 30 milligrams | ✓c,d | ✓b | ||||
| Use non-opioid adjunctive medications for withdrawal | ✓c,d | ✓c,d | ✓a.b,c | |||
| When applicable, offer intramuscular naltrexone | ✓c,d | ✓a,b,c,d | ✓a,b,c,d | ✓b.c,d | ✓a.b,c,d | |
| Acute pain and/or perioperative pain management in setting of OUD | ||||||
| Assess and treat acute pain in the setting of OUD | ✓c,d | |||||
| Continue MOUD in the perioperative period | ✓c,d | |||||
| →Use short-acting opioids + adjunct analgesia in the perioperative period | ✓c,d | ✓c,d | ||||
| Care transition at hospital discharge | ||||||
| Obtain an X-Waiver | ✓e | |||||
| Refer to a buprenorphine prescriber or OTP | ✓c,d | ✓a,c,d | ✓a | ✓c,d | ✓a,b | |
| Refer to psychosocial and mental health treatment, mutual support groups, and harm reduction services | ✓c,d | ✓b,c,d | ✓a,b,c | ✓b,c | ✓a,b,c | |
| Post-acute care facilities must continue MOUD | ||||||
| Prescribe/dispense naloxone at discharge | ✓c,d | ✓c,d | ✓b | ✓c | ✓b | |
| Recommendation | Guideline | ||||||
|---|---|---|---|---|---|---|---|
| ASAM 202019 | CFPC20 | AUS21 | CRISM22,23 | APS/CPDD24 | ASAM 201125 | NSW26 | |
| Medical communication and language for people who use substances | |||||||
| Use non-stigmatizing and person-first language | ✓c,d | ✓c,d | ✓c,d | ||||
| Assessment of unhealthy opioid use and diagnosis of OUD | |||||||
| If unhealthy opioid use, assess for OUD; review PDMP | ✓c,d | ✓c,d | ✓c,d | ✓c | ✓c | ✓c,d | |
| Use DSM-5 to diagnose OUD | ✓c,d | ✓c,d | ✓c,d | ✓c | ✓c | ✓c,d | |
| Offer HIV, hepatitis A, B, C, syphilis, pregnancy test, and urine drug analysis | ✓b,c,d | ✓c,d | ✓c,d | ✓b,c | ✓c | ✓c,d | ✓c,d |
| OUD medication for DSM – 5 confirmed diagnosis | |||||||
| Use shared decision making to initiate MOUD, including buprenorphine or methadone | ✓c,d | ✓b,c,d | ✓c,d | ✓b,c,d | ✓c | ✓c,d | |
| Offer MOUD to treat opioid withdrawal and OUD; use COWS10 to initiate buprenorphine | ✓a,b,c,d | ✓a,c,d | ✓c,d | ✓a,b | ✓c,d | ✓c,d | |
| Consider review of electrocardiogram | ✓c | ✓c,d | ✓c | ✓c,d | |||
| Initiate methadone at 20 to 30 milligrams | ✓c,d | ✓c,d | ✓b,c,d | ✓c | ✓c,d | ||
| Use non-opioid adjunctive medications for withdrawal | ✓a,c,d | ✓c,d | ✓a | ✓b,c,d | ✓c,d | ||
| When applicable, offer intramuscular naltrexone | ✓a,b,c,d | ✓c,d | ✓a,b | ✓c,d | |||
| Acute pain and/or perioperative pain management in setting of OUD | |||||||
| Assess and treat acute pain in the setting of OUD | ✓c,d | ✓c | ✓c,d | ✓c,d | |||
| Continue MOUD in the perioperative period | ✓c,d | ✓b,c | ✓c,d | ✓c,d | |||
| →Use short-acting opioids + adjunct analgesia in the perioperative period | ✓b,c,d | ✓b,c | ✓c,d | ✓c,d | |||
| Care transition at hospital discharge | |||||||
| Obtain an X-Waiver | ✓e | ||||||
| Refer to a buprenorphine prescriber or OTP | ✓a,b,c,d | ✓a,b,c | ✓c,d | ✓a,b,c,d | ✓b,c,d | ✓c,d | |
| Refer to psychosocial and mental health treatment, mutual support groups, and harm reduction services | ✓a,b,c,d | ✓a,b | ✓c,d | ✓a,b,c | ✓a,c | ✓c,d | |
| Post-acute care facilities must continue MOUD | |||||||
| Prescribe/dispense naloxone at discharge | ✓b,c,d | ✓c,d | ✓b,c,d | ✓c,d | |||
Evidence from randomized, controlled trial/trials or nonrandomized controlled trial/trials
Evidence from observational study/studies
Evidence from expert consensus
Based on other guidelines
United States Law
Guideline Synthesis and Analysis
Table 3 contains a synthesis of the recommendations relative to our five pre-specified content areas. While the evidence supporting the recommendations is generally of lower quality, i.e., few randomized controlled trials, there were many areas of agreement across the guidelines.
Use of non-stigmatizing medical communication and language for people who use opioids:
Examples of non-stigmatizing language includes ‘person who uses drugs,’ ‘person who injects drugs,’ or ‘person with OUD’. Nine guidelines recommended that health care professionals approach people who use substances in a non-judgmental, non-stigmatizing way to address the patient’s needs and concerns regarding their substance use.25,26,31,33,34,36,38,40,47 These recommendations were based on expert consensus or other guidelines.
Assessment of unhealthy opioid use and diagnosis of OUD:
The term “unhealthy use” is defined by the USPSTF as “including conditions such as the full spectrum of unhealthy use, e.g., problem use or a use disorder, meeting any DSM criterion for a use disorder, heavy use, e.g., using a substance twice or more per day, or negative consequences or problems related to drug use.50 Fifteen guidelines recommended an assessment of OUD among people with unhealthy opioid use.25–28,34,36–40,42,43,47–49 When available, four guidelines recommended review of prescription drug monitoring program data (PDMP) for prescribed scheduled medication use.25,37,40,47 Seven guidelines recommended the use of the DSM–5 to diagnose OUD;26,32,33,36–38,40 two older guidelines recommended the use of the DSM-4th Edition to diagnose opioid dependence.42,43 Among patients with OUD, fifteen guidelines recommended testing for infections, pregnancy, or checking a urine drug analysis.26–28,31–34,36–40,42,43,48 These recommendations were based on observational studies, expert consensus, or other guidelines.
Use of medications for OUD (MOUD) for a DSM–5 confirmed diagnosis of OUD:
Fourteen guidelines recommended the use of shared decision making when initiating MOUD.25–28,33,34,36–40,43,47,48 These recommendations were based on observational studies, expert consensus, or other guidelines. Fifteen guidelines recommended the use of methadone or buprenorphine as first line agents to treat opioid withdrawal or OUD.25–28,31,33,34,36–40,42,43,47 These recommendations were based on randomized or nonrandomized controlled trials, observational studies, expert consensus, or other guidelines. Eleven guidelines recommended the use of a validated tool, e.g., the Clinical Opiate Withdrawal Scale, to measure and assess severity of opioid withdrawal prior to starting buprenorphine, to initiate buprenorphine for mild to moderate opioid withdrawal symptoms, and to titrate buprenorphine dosing based upon ongoing cravings or withdrawal symptoms.25,26,28,34,36–40,42,43 These recommendations were based on expert consensus or other guidelines. When initiating methadone for OUD treatment or prevention of opioid withdrawal, nine guidelines recommended review of an electrocardiogram when a patient has risk factors for QTc interval prolongation.25,26,34,36–40,48 These recommendations were based on expert consensus or other guidelines. When starting methadone, ten guidelines recommended an initial dose of 10 to 30 milligrams with further titration based on patient reported opioid withdrawal symptoms or opioid cravings.25,26,33,34,36–40,48 These recommendations were informed by observational studies, expert consensus, or other guidelines. Eleven guidelines recommended the use of non-opioid adjunctive medications for opioid withdrawal symptoms during initial dose titration of methadone or buprenorphine.25,26,28,33,34,36–40,42 These recommendations were based on randomized or nonrandomized controlled trials, observational studies, expert consensus, or other guidelines. If the patient prefers opioid antagonist treatment, twelve guidelines recommended the use of intramuscular naltrexone, and against the use of oral naltrexone, for OUD treatment.25–28,33,34,36–40,47 These recommendations were based on randomized or nonrandomized controlled trials, observational studies, expert consensus, or other guidelines.
Management of acute or perioperative pain in the setting of OUD:
Nine guidelines recommended that patients with OUD should be assessed and treated for acute pain.25–27,36–38,43,44,46 These same guidelines recommended the use of multimodal therapies to treat acute pain, starting with non-opioid analgesics including neuropathic agents, ketamine, lidocaine, non-steroidal anti-inflammatories, etc. With persistent acute pain, nine guidelines recommended escalating to short-acting full agonist opioids at higher doses than used with opioid naïve patients and at shorter intervals.25–27,36–38,43,44,46
Guideline recommendations for buprenorphine management in the perioperative period evolved over time. In 2015, the American Society of Addiction Medicine (ASAM) published practice guideline which recommended discontinuation of buprenorphine for OUD in the setting of severe acute pain, which included the perioperative period.25 An updated ASAM guideline published in 2020 stated that “discontinuation of buprenorphine (or methadone) before surgery is not required” and “increasing the dose or frequency of methadone or buprenorphine may be effective for managing pain.” Furthermore, “the use short acting full agonist opioids, at higher dose than those required in opioid naïve patients, can be effective to manage moderate to severe acute pain.”37 In total, six guidelines recommended continuation of buprenorphine or methadone in the perioperative period or with non-operative, acute pain.34,36–38,43,45 These recommendations were based on observational studies, expert consensus, or other guidelines.
Care transition at hospital discharge:
In the United States an “X-Waiver” is required to prescribe buprenorphine for OUD in the outpatient setting. “X-Waiver” refers to the Drug Addiction Treatment Act (DATA 2000) legislation that authorized the outpatient use of buprenorphine for the treatment of OUD. Four guidelines discussed this requirement,25,37,40,47 one of which advocated for the removal of barriers to provide buprenorphine (and methadone) to patients with OUD.47 Because the X-Waiver requirement is unique to the United States, international guidelines included in this systematic review did not address this topic.26–28,31,33,36,38,39,43,45 Fourteen guidelines recommended continuation of buprenorphine or methadone following treatment initiation compared to the use of these medications for medically managed opioid withdrawal (detoxification).25–28,33,34,36–40,42,43,47 These recommendations were based on randomized or nonrandomized controlled trials, observational studies, expert consensus, or other guidelines. No guideline specifically addressed best practices in medication continuation for OUD at hospital discharge because no guidelines included in the systematic reviewed addressed the care of hospitalized patients or patients in the emergency department.
In conjunction with MOUD, fifteen guidelines recommended that people with OUD should be linked to psychosocial treatment and mental health services. This includes cognitive-behavioral therapy and relapse prevention, contingency management, motivational enhancement/motivational interviewing and brief interventions.26–28,30,32–34,36–40,42,43,47 These recommendations were based observational studies, expert consensus, or other guidelines. Twelve guidelines recommended that people with OUD should be linked to psychosocial support, including resources for case management, housing, employment, and legal support.26,30,32–34,36–38,40,42,43,47 These recommendations were based on expert consensus or other guidelines. Ten guidelines recommended people with OUD should be linked to mutual support groups or peer recovery supports.26,32–34,36–38,40,42,47 They included randomized or nonrandomized controlled trials, observational studies, expert consensus, or other guidelines. Ten guidelines recommended that people with OUD should be referred to harm reduction services. Harm reduction services includes education on sterile syringe use and safer injection practices to reduce the risk of blood-borne (HIV, hepatitis C) and soft tissue infections, promoting access to take-home naloxone, and syringe distribution programs to reduce risk of blood-borne infection and fatal overdose among high-risk patients or patients with ongoing opioid use.26,30,32–34,36–39,47 These recommendations were based on observational studies, expert consensus, or other guidelines. Eleven guidelines specifically recommended that patients with OUD have access to naloxone for opioid overdose reversal.25–28,33,34,36–39,47 These recommendations were based on the safety profile of naloxone and its ability to reverse an opioid-related overdose. They included randomized or nonrandomized controlled trials, observational studies, expert consensus, or other guidelines.
Discussion
This systematic review identified nineteen guidelines which included recommendations on the management of OUD, opioid withdrawal, opioid overdose prevention, and care transitions. Guideline recommendations were primarily informed by observational studies and expert opinion. None of the included guidelines focused specifically on OUD-related care in the hospital setting, though many provided guidance for OUD-related care provision across the inpatient, outpatient, and community settings. While these factors may limit the recommendations’ applicability to the hospital setting, these guidelines represent the best guidance currently available to standardize and improve the management of OUD, opioid withdrawal, opioid overdose prevention, and care transitions for hospitalized patients with OUD.
Both methadone and buprenorphine are associated with a decreased risk of return to opioid use compared to no opioid agonist treatment.51 Lifesaving benefits of these medications include a maintenance of opioid tolerance and a reduction in opioid cravings leading to decreased use of opioid that are not prescribed, heroin, or illicitly manufactured fentanyl and its analogues.51 None of these guidelines discussed treatment outcomes by treatment location. However, to date, there is no evidence to suggest that medication effectiveness varies by administration location, e.g., hospital vs. outpatient vs. OTP. Most guidelines highlighted the risk of return to opioid use when methadone or buprenorphine are used to facilitate opioid detoxification alone, but not continued thereafter. Starting and continuing these medications in any setting is likely beneficial when people with OUD request this treatment. When a patient desires continuation of MOUD at care transitions, i.e., post-acute care facility or state or federal institutions, under the American with Disability Act, MOUD must be continued.52 Failure of such facilities to accept patients receiving MOUD, or to continue these medications, may result in legal action.53
Recommendations for buprenorphine initiation, dosing, and perioperative management strategies were primarily based on expert opinion, which carries a lower strength evidence. Decisions to continue or initiate buprenorphine in the hospital should be individualize, incorporating the types of opioids used, past patient experiences with pain management, and especially patient experiences with prior buprenorphine use. Due to widespread contamination of fentanyl and fentanyl analogs in the drug supply, and case reports describing instances of precipitated withdrawal with routine buprenorphine initiation among people using fentanyl,54 buprenorphine initiation and dosing practices are rapidly evolving. While most guidelines included in the systematic review recommended buprenorphine initiation with mild to moderate withdrawal symptoms, future guidelines may incorporate greater detail about when to use various buprenorphine initiation protocols based on the patient’s opioid use, i.e., regular use of fentanyl or methadone, or their desire to reach a therapeutic buprenorphine dose rapidly. This may include the use of a “low dose” method, in which the buprenorphine starting dose is very low and gradually increased to therapeutic levels over a period of days,55 or the use of a “high dose” method, in which buprenorphine dose titration is accelerated to achieve therapeutic buprenorphine levels in less than 3 to 4 hours, compared to the usual 48 to 72 hours.56
Most of the guidelines recommended that patients with OUD be referred to psychosocial treatment, mutual support groups, or harm reduction services, in addition to MOUD. High quality evidence supports the use of naloxone as harm reduction to reduce opioid-related overdose mortality.57,58 Observational data cited in many of these guidelines highlight public health benefits of integrating harm reduction strategies into clinical practice, e.g., reduction in HIV transmission. Data on the benefits of psychosocial treatment and mutual support groups, however, are mixed with some demonstrating reduction in substance use,59,60 and other with inconclusive benefit on substance use behaviors.61,62 Despite these limitations, psychosocial interventions, mental health treatment, and mutual support groups rarely report serious harms. Inclusion of these recommendations was fairly consistent across the guidelines.
Few guidelines explicitly addressed treatment disparities by race/ethnicity. One guideline noted that each treatment care plan should “take account of the patient’s ethnicity, gender, and sexuality”.36 Another guideline recommended that further research regarding treatment effectiveness should identify differences based on “socioeconomic status, age, gender, and ethnicity” but did not focus on addressing racism or structural issues underlying these disparities.30 In the United States, access to substance use disorder treatment for Black, Indigenous, and People of Color (BIPoC) is limited compared to treatment access for white people. National survey data from 2018 demonstrated that 88% of Black people and 90% of Latino people received no substance treatment due to “deep-seated inequities in health care for communities of color”.63 Another study of nationally representative survey data demonstrated that, from 2004 to 2015, Black people were 80% less likely to be prescribed buprenorphine than white people indicating buprenorphine treatment disparities on the basis of race/ethnicity and insurance ocverage.10 Black patients seeking substance treatment report experiencing discrimination by healthcare workers leading to delays in medical care.64 Similar treatment disparities were noted among patients who experienced a non-fatal opioid overdose where Black patients were half as likely to obtain a follow-up appointment or MOUD compared to white patients.65 Additional treatment disparities include the undertreatment of acute pain with opioid and non-opioid analgesia among Black people compared to white people66 and greater odds of opioid dose reduction for chronic opioid therapy among Black people versus white people. Emerging evidence supports that racial differences with access to MOUD noted in the outpatient setting are also present in the hospital setting. A recent study of hospitalized veterans with OUD reported that white patients were 11% more likely than Black patients to receive buprenorphine than methadone during hospitalization74. This matters because methadone for OUD requires near daily trips to the OTP whereas buprenorphine can be prescribed on a weekly or monthly basis, allowing greater flexibility to obtain MOUD following hospital discharge. Mechanisms to expand access to OUD treatment among BIPoC communities must address hospital- and community-level structural barriers to substance treatment access, systemic racism, and biased prescribing of MOUD. It must also include multi-sectoral, diverse community partnerships to develop community-guided prevention, treatment, and recovery strategies.67–69
An updated guideline search from July 2020 through April 2022 using the same search teams and sources revealed three additional guidelines which met our inclusion criteria.70–72 One guideline was an updated USPSTF recommendation,72 one guideline was an executive summary of a guideline included in this systematic review,71 and one guideline was intended for the care of adult patients presenting to an emergency department.70 Notably, these additional guideline recommendations align with the guideline recommendations included in this systematic review and do not offer any contrary recommendations.
Future research is needed to clarify and address treatment gaps identified during this systematic review. Frequency of infectious disease screening for people with OUD, or for people who inject drugs, was not addressed in these guidelines. Clarification on screening frequency is needed to ensure judicious use of healthcare resources. Rigorous and systematic research is needed to identify best practices for buprenorphine initiation and dosing among people who wish to transition off of methadone, for people who regularly use fentanyl or fentanyl analogues, or for people experiencing acute pain, especially during the perioperative period. Greater research is needed to develop best practice recommendations for co-administration of full agonist opioids, including hydromorphone or oxycodone, in addition to buprenorphine or methadone, when managing acute opioid withdrawal in the hospital setting. Importantly, significant OUD treatment disparities exist for BIPoC communities. Resources must be dedicated to address OUD treatment disparities to close discriminatory treatment gaps. Ongoing research will likely inform future iterations of practice recommendations for the management of OUD, opioid withdrawal, and opioid overdose prevention in hospitalized adults.
This study had several limitations. First, we only reviewed guidelines published in English, potentially missing research that has been conducted on OUD-related treatment globally. Second, while we used rigorous, pre-defined search criteria and registered our protocol on PROSPERO, it is possible that our search strategy missed relevant guidelines. Third, the results of the AGREE II appraisal should be viewed with caution, as different appraisers may interpret the items and scoring system differently. Despite this limitation, the AGREE II instrument provides an opportunity to evaluate the quality of guidelines systematically, specifically, and objectively across specialties and topics. Finally, guidelines do not specify how hospitals should deliver these recommendations. Various approaches to delivering hospital-based OUD treatment exist, and future work should explore what models best support effective delivery of evidence-based guidelines.73
In conclusion, we identified nineteen guidelines addressing OUD treatment and related conditions. These guidelines were developed from studies with various levels of rigor and quality which informed the strength of the final recommendations. Importantly, none of the guidelines were dedicated to the care of hospitalized patients with OUD. Despite the relative heterogeneity of the guidelines, e.g., international guidelines, guidelines from various specialties, guidelines directed to the care of older adults, or perioperative pain management, the recommendations related to our pre-specified topic areas were fairly consistent, allowing for the development of well-informed recommendations for the care of hospitalized adults with OUD.
Supplementary Material
Acknowledgements
Dr. Calcaterra had full access to all of the data in the study and takes full responsibility for the integrity of the data and the accuracy of the data analysis.
The authors would like to acknowledge and thank Chiara Mandel and Kevin Vuernick from SHM for their facilitation of this project and dedication to this purpose. The authors would also like to acknowledge Christi Piper, MLIS, AHIP for her work to build the search teams and statement for our systematic review.
Funding/Financial Support
The Society of Hospital Medicine (SHM) provided administrative assistance and material support but had no role in or influence on the scientific conduct of the study. Dr. Calcaterra is supported by a grant from the National Institutes of Health (NIH), National Institute on Drug Abuse (NIDA) (K08DA049905). Dr. Englander is supported by a grant from the NIH, NIDA (UG1DA015815). Dr. Herzig is supported by R01HS026215 from the Agency for Healthcare Research and Quality. The views and opinions expressed in this article are those of the authors and do not necessarily reflect those of the NIH.
Footnotes
Supplementary Material
Refer to Web Version on PubMed Central for supplementary material.
References
- 1.Administration SAaMHS. Key Substance Use and Mental Health Indicators in the United States: Results from the 2019 National Survey on Drug Use and Health. Rockville, MD: 2020. [Google Scholar]
- 2.Drug Overdose Deaths in the U.S. Top 100,000 Annually [press release]. Centers for Disease Control and Prevention; 2021. [Google Scholar]
- 3.Ahmad FB, Anderson RN. The Leading Causes of Death in the US for 2020. JAMA. 2021;325(18):1829–1830. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Suen LW, Makam AN, Snyder HR, et al. National Prevalence of Alcohol and Other Substance Use Disorders Among Emergency Department Visits and Hospitalizations: NHAMCS 2014–2018. Journal of General Internal Medicine. 2021. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.King C, Cook R, Korthuis PT, Morris CD, Englander H. Causes of Death in the 12 months After Hospital Discharge Among Patients With Opioid Use Disorder. J Addict Med. 2021. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Larochelle MR, Bernson D, Land T, et al. Medication for opioid use disorder after nonfatal opioid overdose and association with mortality: a cohort study. Annals of internal medicine. 2018;169(3):137–145. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Sordo L, Barrio G, Bravo MJ, et al. Mortality risk during and after opioid substitution treatment: systematic review and meta-analysis of cohort studies. bmj. 2017;357. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Andrilla CHA, Moore TE, Patterson DG, Larson EH. Geographic Distribution of Providers With a DEA Waiver to Prescribe Buprenorphine for the Treatment of Opioid Use Disorder: A 5-Year Update. J Rural Health. 2019;35(1):108–112. [DOI] [PubMed] [Google Scholar]
- 9.Joudrey PJ, Chadi N, Roy P, et al. Pharmacy-based methadone dispensing and drive time to methadone treatment in five states within the United States: A cross-sectional study. Drug and alcohol dependence. 2020;211:107968. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Lagisetty PA, Ross R, Bohnert A, Clay M, Maust DT. Buprenorphine Treatment Divide by Race/Ethnicity and Payment. JAMA Psychiatry. 2019;76(9):979–981. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Hartung DM, Johnston K, Geddes J, Leichtling G, Priest KC, Korthuis PT. Buprenorphine Coverage in the Medicare Part D Program for 2007 to 2018. JAMA. 2019;321(6):607–609. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Cabreros I, Griffin BA, Saloner B, Gordon AJ, Kerber R, Stein BD. Buprenorphine prescriber monthly patient caseloads: An examination of 6-year trajectories. Drug and Alcohol Dependence. 2021;228:109089. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Calcaterra SL, Binswanger IA, Edelman EJ, McNair BK, Wakeman SE, O’Connor PG. The impact of access to addiction specialist on attitudes, beliefs and hospital-based opioid use disorder related care: A survey of hospitalist physicians. Substance Abuse. 2020:1–9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Priest KC, Lovejoy TI, Englander H, Shull S, McCarty D. Opioid Agonist Therapy During Hospitalization Within the Veterans Health Administration: a Pragmatic Retrospective Cohort Analysis. J Gen Intern Med. 2020;35(8):2365–2374. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Goedel WC, Shapiro A, Cerdá M, Tsai JW, Hadland SE, Marshall BDL. Association of Racial/Ethnic Segregation With Treatment Capacity for Opioid Use Disorder in Counties in the United States. JAMA Network Open. 2020;3(4):e203711–e203711. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Englander H, Priest KC, Snyder H, Martin M, Calcaterra S, Gregg J. A Call to Action: Hospitalists’ Role in Addressing Substance Use Disorder. Journal of hospital medicine. 2019;14:E1–E4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.PRISMA. Transparent Reporting of Systematic Reviews and Meta-Analyses. http://www.prisma-statement.org/. Published 2021. Updated 2021. Accessed.
- 18.Shekelle PG, Ortiz E, Rhodes S, et al. Validity of the Agency for Healthcare Research and Quality Clinical Practice GuidelinesHow Quickly Do Guidelines Become Outdated? JAMA. 2001;286(12):1461–1467. [DOI] [PubMed] [Google Scholar]
- 19.EndNote. https://endnote.com/. Published 2021. Accessed December 11, 2021.
- 20.Graham R, Mancher M, Wolman D, Greenfield S, Steinberg E. Institute of Medicine. Clinical practice guidelines we can trust. Natl Acad Press. 2011. [PubMed] [Google Scholar]
- 21.Brouwers MC, Kho ME, Browman GP, et al. AGREE II: advancing guideline development, reporting and evaluation in health care. Cmaj. 2010;182(18):E839–E842. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Brouwers MC, Kho ME, Browman GP, et al. Development of the AGREE II, part 1: performance, usefulness and areas for improvement. Cmaj. 2010;182(10):1045–1052. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Brouwers MC, Kho ME, Browman GP, et al. Development of the AGREE II, part 2: assessment of validity of items and tools to support application. Cmaj. 2010;182(10):E472–E478. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.The ASAM National Practice Guideline for the Treatment of Opioid Use Disorder: 2020 Focused Update. J Addict Med. 2020;14(2S Suppl 1):1–91. [DOI] [PubMed] [Google Scholar]
- 25.Comer S, Cunningham C, Fishman MJ, Gordon A, Kampman K, Langlegen D, Nordstrom B, Oslin D, Woody G, Wright T, Wyatt S. The ASAM National Practice Guideline for the Use of Medications in the Treatment of Addiction Involving Opioid Use. 2015.
- 26.Use BCCoS. A Guideline for the Clinical Management of Opioid Use Disorder. In. British Columbia: British Columbia Ministry of Health; 2017. [Google Scholar]
- 27.Rieb LM, Samaan Z, Furlan AD, et al. Canadian Guidelines on Opioid Use Disorder Among Older Adults. Can Geriatr J. 2020;23(1):123–134. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Korownyk C, Perry D, Ton J, et al. Managing opioid use disorder in primary care: PEER simplified guideline. Canadian Family Physician. 2019;65(5):321–330. [PMC free article] [PubMed] [Google Scholar]
- 29.Korownyk C, Perry D, Ton J, et al. Opioid use disorder in primary care: PEER umbrella systematic review of systematic reviews. Can Fam Physician. 2019;65(5):e194–e206. [PMC free article] [PubMed] [Google Scholar]
- 30.Excellence NIfHaC. Needle and syringe programmes (PH52). United Kingdom: 2014. [Google Scholar]
- 31.Excellence NIfHaC. Needle and Syringe Programmes (PH52). United Kingdom: 2019. [Google Scholar]
- 32.Kampman K, Jarvis M. American Society of Addiction Medicine (ASAM) National Practice Guideline for the Use of Medications in the Treatment of Addiction Involving Opioid Use. J Addict Med. 2015;9(5):358–367. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Wood EAK, Fairbairn N, Hering R, Lappalainen L, Matieschyn Z, Tupper K, Sharifi N, Fischer B, Selby P, Cirone S, Handford C, Kahan M. Ordean A, Pouteau K, Spithoff SM, Sproule BA, Wild TC, Poulin G, Athaide M, Badesha K, Butt PR, Evans LD, Fisher M, Isacc MR, Lim R, Martin RA, Rose M, Trew M, Bruneau J, Goyer ME, Barnes P, Boudreault P, Cox J, Dumont J, Dupont S, Hickox S, Hollett B, Hooley P, Martell D, Miller L, Pelletier MC, Webster D. CRISM National Guideline for the Clinical Management of Opioid Use Disorder. Canadian Research Initiative in Substance Misuse;2018. [Google Scholar]
- 34.Group CGoDMaDUIEW. Drug misuse and dependence: UK guidelines on clinical management. London: 2017. [Google Scholar]
- 35.Bruneau J, Ahamad K, Goyer M, et al. Management of opioid use disorders: a national clinical practice guideline. Cmaj. 2018;190(9):E247–e257. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Gowing LAR, Dunlop A, Farrell M, Lintzeris N. National Guidelines for Medication-Assisted Treatment of Opioid Dependence. Australia: 2014. [Google Scholar]
- 37.Cunningham CEM, Fishman M, Gordon AF, Jones HE, Kampman KM, Langleben D, Meyer M, Springer S, Woody G, Wright TE, Wyatt S. The ASAM National Practice Guideline for the Treatment of Opioid Use Disorder 2020 Focused Update. 11400 Rockville Pike, Suite 200, Rockville, MD 20852: 2020. [Google Scholar]
- 38.Health NMo. New South Wales Clinical Guidelines: Treatment of Opioid Dependence. New South Wales: 2018. [Google Scholar]
- 39.Soyka M, Kranzler HR, van den Brink W, Krystal J, Möller HJ, Kasper S. The World Federation of Societies of Biological Psychiatry (WFSBP) guidelines for the biological treatment of substance use and related disorders. Part 2: Opioid dependence. World J Biol Psychiatry. 2011;12(3):160–187. [DOI] [PubMed] [Google Scholar]
- 40.Affairs DoVs. VA/DoD Clinical Practice Guideline for Management of Substance Use Disorders. Washington, DC: U.S. Department of Defense;2015. [Google Scholar]
- 41.Farmer CM, Lindsay D, Williams J, et al. Practice Guidance for Buprenorphine for the Treatment of Opioid Use Disorders: Results of an Expert Panel Process. Subst Abus. 2015;36(2):209–216. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Kraus ML, Alford DP, Kotz MM, et al. Statement of the American Society Of Addiction Medicine Consensus Panel on the use of buprenorphine in office-based treatment of opioid addiction. J Addict Med. 2011;5(4):254–263. [DOI] [PubMed] [Google Scholar]
- 43.Handford CKM, Srivastava A, Cirone S, Sanghera S, Palda V, Lester MD, Janecek E, Franklyn M, Cord M, Selby P, Ordean A. Buprenorphine/Naloxone for Opioid Dependence: Clinical Practice Guideline. 2012.
- 44.Goel A, Azargive S, Weissman JS, et al. Perioperative Pain and Addiction Interdisciplinary Network (PAIN): protocol of a practice advisory for the perioperative management of buprenorphine using a modified Delphi process. BMJ Open. 2019;9(5):e027374. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Goel A, Azargive S, Weissman JS, et al. Perioperative Pain and Addiction Interdisciplinary Network (PAIN) clinical practice advisory for perioperative management of buprenorphine: results of a modified Delphi process. Br J Anaesth. 2019;123(2):e333–e342. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Edwards DA, Hedrick TL, Jayaram J, et al. American Society for Enhanced Recovery and Perioperative Quality Initiative Joint Consensus Statement on Perioperative Management of Patients on Preoperative Opioid Therapy. Anesthesia & Analgesia. 2019;129(2):553–566. [DOI] [PubMed] [Google Scholar]
- 47.Crowley R, Kirschner N, Dunn AS, Bornstein SS. Health and Public Policy to Facilitate Effective Prevention and Treatment of Substance Use Disorders Involving Illicit and Prescription Drugs: An American College of Physicians Position Paper. Ann Intern Med. 2017;166(10):733–736. [DOI] [PubMed] [Google Scholar]
- 48.Chou R, Cruciani RA, Fiellin DA, et al. Methadone safety: a clinical practice guideline from the American Pain Society and College on Problems of Drug Dependence, in collaboration with the Heart Rhythm Society. J Pain. 2014;15(4):321–337. [DOI] [PubMed] [Google Scholar]
- 49.Force UPST. Screening for Unhealthy Drug Use: US Preventive Services Task Force Recommendation Statement. JAMA. 2020;323(22):2301–2309. [DOI] [PubMed] [Google Scholar]
- 50.Patnode CD, Perdue LA, Rushkin M, et al. Screening for Unhealthy Drug Use: Updated Evidence Report and Systematic Review for the US Preventive Services Task Force. JAMA. 2020;323(22):2310–2328. [DOI] [PubMed] [Google Scholar]
- 51.Mattick RP, Breen C, Kimber J, Davoli M. Buprenorphine maintenance versus placebo or methadone maintenance for opioid dependence. Cochrane database of systematic reviews. 2014(2). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Division USDoJCR. The Americans with Disabilities Act and the Opioid Crisis: Combating Discrimination Against People in Treatment or Recovery April 5 2022.
- 53.Department of Justice USAsODoM. U.S. Attorney’s Office Settles Disability Discrimination Allegations with Operator of Skilled Nursing Facilities. September 19 2019.
- 54.Varshneya NB, Thakrar AP, Hobelmann JG, Dunn KE, Huhn AS. Evidence of Buprenorphine-precipitated Withdrawal in Persons Who Use Fentanyl. J Addict Med. 9000. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Cohen SM, Weimer MB, Levander XA, Peckham AM, Tetrault JM, Morford KL. Low Dose Initiation of Buprenorphine: A Narrative Review and Practical Approach. J Addict Med. 2021. [DOI] [PubMed] [Google Scholar]
- 56.Herring AA, Vosooghi AA, Luftig J, et al. High-Dose Buprenorphine Induction in the Emergency Department for Treatment of Opioid Use Disorder. JAMA Netw Open. 2021;4(7):e2117128. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Centers for Disease C, Prevention. Community-based opioid overdose prevention programs providing naloxone - United States, 2010. MMWR Morb Mortal Wkly Rep. 2012;61(6):101–105. [PMC free article] [PubMed] [Google Scholar]
- 58.Kerr D, Kelly AM, Dietze P, Jolley D, Barger B. Randomized controlled trial comparing the effectiveness and safety of intranasal and intramuscular naloxone for the treatment of suspected heroin overdose. Addiction. 2009;104(12):2067–2074. [DOI] [PubMed] [Google Scholar]
- 59.Mayet S, Farrell M, Ferri M, Amato L, Davoli M. Psychosocial treatment for opiate abuse and dependence. Cochrane database of systematic reviews. 2004(4). [DOI] [PubMed] [Google Scholar]
- 60.Dugosh K, Abraham A, Seymour B, McLoyd K, Chalk M, Festinger D. A Systematic Review on the Use of Psychosocial Interventions in Conjunction With Medications for the Treatment of Opioid Addiction. J Addict Med. 2016;10(2):93–103. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.Rice D, Corace K, Wolfe D, et al. Evaluating comparative effectiveness of psychosocial interventions adjunctive to opioid agonist therapy for opioid use disorder: A systematic review with network meta-analyses. PloS one. 2020;15(12):e0244401. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62.Gregory VL Jr, Ellis RJB. Cognitive-behavioral therapy and buprenorphine for opioid use disorder: A systematic review and meta-analysis of randomized controlled trials. The American Journal of Drug and Alcohol Abuse. 2020;46(5):520–530. [DOI] [PubMed] [Google Scholar]
- 63.(SAMHSA) SAaMHSA. Double jeopardy: COVID-19 and behavioral health disparities for Black and Latino communities in the US: 2020. 2020.
- 64.Hall OT, Jordan A, Teater J, et al. Experiences of racial discrimination in the medical setting and associations with medical mistrust and expectations of care among black patients seeking addiction treatment. Journal of Substance Abuse Treatment. 2022;133:108551. [DOI] [PubMed] [Google Scholar]
- 65.Kilaru AS, Xiong A, Lowenstein M, et al. Incidence of Treatment for Opioid Use Disorder Following Nonfatal Overdose in Commercially Insured Patients. JAMA Network Open. 2020;3(5):e205852–e205852. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 66.Meghani SH, Byun E, Gallagher RM. Time to Take Stock: A Meta-Analysis and Systematic Review of Analgesic Treatment Disparities for Pain in the United States. Pain Medicine. 2012;13(2):150–174. [DOI] [PubMed] [Google Scholar]
- 67.(SAMHSA) SAaMHSA. The Opioid Crisis and the Black/African American Population: An Urgent Issue. Office of Behavioral Health Equity;2020. [Google Scholar]
- 68.Humphreys K, Shover CL, Andrews CM, et al. Responding to the opioid crisis in North America and beyond: recommendations of the Stanford-Lancet Commission. Lancet. 2022;399(10324):555–604. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 69.Joudrey PJ, Edelman EJ, Wang EA. Drive times to opioid treatment programs in urban and rural counties in 5 US states. Jama. 2019;322(13):1310–1312. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 70.Hatten BW, Cantrill SV, Dubin JS, et al. Clinical Policy: Critical Issues Related to Opioids in Adult Patients Presenting to the Emergency Department. Ann Emerg Med. 2020;76(3):e13–e39. [DOI] [PubMed] [Google Scholar]
- 71.Crotty K, Freedman KI, Kampman KM. Executive Summary of the Focused Update of the ASAM National Practice Guideline for the Treatment of Opioid Use Disorder. J Addict Med. 2020;14(2):99–112. [DOI] [PubMed] [Google Scholar]
- 72.Force UPST. Primary Care–Based Interventions to Prevent Illicit Drug Use in Children, Adolescents, and Young Adults: US Preventive Services Task Force Recommendation Statement. JAMA. 2020;323(20):2060–2066. [DOI] [PubMed] [Google Scholar]
- 73.Englander H, Jones A, Krawczyk N, et al. A Taxonomy of Hospital-Based Addiction Care Models: a Scoping Review and Key Informant Interviews. Journal of General Internal Medicine. 2022. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 74.Priest KC, King CA, Englander H, Lovejoy TI, & McCarty D. (2022). Differences in the delivery of medications for opioid use disorder during hospitalization by racial categories: A retrospective cohort analysis. Substance Abuse, 43(1), 1251–1259. [DOI] [PMC free article] [PubMed] [Google Scholar]
Table 3 References
- 1.Use BCCoS. A Guideline for the Clinical Management of Opioid Use Disorder. In. British Columbia: British Columbia Ministry of Health; 2017. [Google Scholar]
- 2.Edwards DA, Hedrick TL, Jayaram J, et al. American Society for Enhanced Recovery and Perioperative Quality Initiative Joint Consensus Statement on Perioperative Management of Patients on Preoperative Opioid Therapy. Anesthesia & Analgesia. 2019;129(2):553–566. [DOI] [PubMed] [Google Scholar]
- 3.Kampman K, Jarvis M. American Society of Addiction Medicine (ASAM) National Practice Guideline for the Use of Medications in the Treatment of Addiction Involving Opioid Use. J Addict Med. 2015;9(5):358–367. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Comer S, Cunningham C, Fishman MJ, Gordon A, Kampman K, Langlegen D, Nordstrom B, Oslin D, Woody G, Wright T, Wyatt S. The ASAM National Practice Guideline for the Use of Medications in the Treatment of Addiction Involving Opioid Use. 2015.
- 5.Goel A, Azargive S, Weissman JS, et al. Perioperative Pain and Addiction Interdisciplinary Network (PAIN) clinical practice advisory for perioperative management of buprenorphine: results of a modified Delphi process. Br J Anaesth. 2019;123(2):e333–e342. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Goel A, Azargive S, Weissman JS, et al. Perioperative Pain and Addiction Interdisciplinary Network (PAIN): protocol of a practice advisory for the perioperative management of buprenorphine using a modified Delphi process. BMJ Open. 2019;9(5):e027374. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Force UPST. Screening for Unhealthy Drug Use: US Preventive Services Task Force Recommendation Statement. JAMA. 2020;323(22):2301–2309. [DOI] [PubMed] [Google Scholar]
- 8.Patnode CD, Perdue LA, Rushkin M, et al. Screening for Unhealthy Drug Use: Updated Evidence Report and Systematic Review for the US Preventive Services Task Force. JAMA. 2020;323(22):2310–2328. [DOI] [PubMed] [Google Scholar]
- 9.Affairs DoVs. VA/DoD clinical practice guideline for management of sbustance use disorders. Washington, DC: U.S. Department of Defense;2015. [Google Scholar]
- 10.Wesson DR, Ling W. The Clinical Opiate Withdrawal Scale (COWS). J Psychoactive Drugs. 2003;35(2):253–259. [DOI] [PubMed] [Google Scholar]
- 11.Excellence NIfHaC. Needle and syringe programmes (PH52). United Kingdom: 2014. [Google Scholar]
- 12.Excellence NIfHaC. Needle and Syringe Programmes (PH52). United Kingdom: 2019. [Google Scholar]
- 13.Group CGoDMaDUIEW. Drug misuse and dependence: UK guidelines on clinical management. London: 2017. [Google Scholar]
- 14.Korownyk C, Perry D, Ton J, et al. Managing opioid use disorder in primary care: PEER simplified guideline. Canadian Family Physician. 2019;65(5):321–330. [PMC free article] [PubMed] [Google Scholar]
- 15.Korownyk C, Perry D, Ton J, et al. Opioid use disorder in primary care: PEER umbrella systematic review of systematic reviews. Can Fam Physician. 2019;65(5):e194–e206. [PMC free article] [PubMed] [Google Scholar]
- 16.Rieb LM, Samaan Z, Furlan AD, et al. Canadian Guidelines on Opioid Use Disorder Among Older Adults. Can Geriatr J. 2020;23(1):123–134. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Crowley R, Kirschner N, Dunn AS, Bornstein SS. Health and Public Policy to Facilitate Effective Prevention and Treatment of Substance Use Disorders Involving Illicit and Prescription Drugs: An American College of Physicians Position Paper. Ann Intern Med. 2017;166(10):733–736. [DOI] [PubMed] [Google Scholar]
- 18.Soyka M, Kranzler HR, van den Brink W, Krystal J, Möller HJ, Kasper S. The World Federation of Societies of Biological Psychiatry (WFSBP) guidelines for the biological treatment of substance use and related disorders. Part 2: Opioid dependence. World J Biol Psychiatry. 2011;12(3):160–187. [DOI] [PubMed] [Google Scholar]
- 19.Cunningham CEM, Fishman M, Gordon AF, Jones HE, Kampman KM, Langleben D, Meyer M, Springer S, Woody G, Wright TE, Wyatt S. The ASAM National Practice Guideline for the Treatment of Opioid Use Disorder 2020 Focused Update. 11400 Rockville Pike, Suite 200, Rockville, MD 20852: 2020. [Google Scholar]
- 20.Handford CKM, Srivastava A, Cirone S, Sanghera S, Palda V, Lester MD, Janecek E, Franklyn M, Cord M, Selby P, Ordean A. Buprenorphine/Naloxone for Opioid Dependence: Clinical Practice Guideline. 2012.
- 21.Gowing LAR, Dunlop A, Farrell M, Lintzeris N. National Guidelines for Medication-Assisted Treatment of Opioid Dependence. Australia: 2014. [Google Scholar]
- 22.Bruneau J, Ahamad K, Goyer M, et al. Management of opioid use disorders: a national clinical practice guideline. Cmaj. 2018;190(9):E247–e257. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Wood EAK, Fairbairn N, Hering R, Lappalainen L, Matieschyn Z, Tupper K, Sharifi N, Fischer B, Selby P, Cirone S, Handford C, Kahan M. Ordean A, Pouteau K, Spithoff SM, Sproule BA, Wild TC, Poulin G, Athaide M, Badesha K, Butt PR, Evans LD, Fisher M, Isacc MR, Lim R, Martin RA, Rose M, Trew M, Bruneau J, Goyer ME, Barnes P, Boudreault P, Cox J, Dumont J, Dupont S, Hickox S, Hollett B, Hooley P, Martell D, Miller L, Pelletier MC, Webster D. CRISM National Guideline for the Clinical Management of Opioid Use Disorder. Canadian Research Initiative in Substance Misuse;2018. [Google Scholar]
- 24.Chou R, Cruciani RA, Fiellin DA, et al. Methadone safety: a clinical practice guideline from the American Pain Society and College on Problems of Drug Dependence, in collaboration with the Heart Rhythm Society. J Pain. 2014;15(4):321–337. [DOI] [PubMed] [Google Scholar]
- 25.Kraus ML, Alford DP, Kotz MM, et al. Statement of the American Society Of Addiction Medicine Consensus Panel on the use of buprenorphine in office-based treatment of opioid addiction. J Addict Med. 2011;5(4):254–263. [DOI] [PubMed] [Google Scholar]
- 26.Health NMo. New South Wales (NSW) Clinical Guidelines: Treatmetnt of Opioid Dependence - 2018. New South Wales: 2018. [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.


