Abstract
Background
Mitochondrial long-chain fatty acid oxidation and carnitine metabolism defects are a group of inherited metabolic diseases. We performed a retrospective cohort study to report on the phenotypic and genotypic spectrum of mitochondrial long-chain fatty acid oxidation and carnitine metabolism defects as well as their treatment outcomes.
Methods
All patients with mitochondrial long-chain fatty acid oxidation and carnitine metabolism defects were included. We divided patients into two groups to compare outcomes of those treated symptomatically (SymX) and asymptomatically (AsymX). We reviewed patient charts for clinical features, biochemical investigations, molecular genetic investigations, cardiac assessments, neuroimaging, treatments, and outcomes.
Results
There were 38 patients including VLCAD (n = 5), LCHAD (n = 4), CACT (n = 3), MAD (n = 1), CPT-I (n = 13), CPT-II (n = 3) deficiencies and CTD (n = 9). Fourteen patients were diagnosed symptomatically (SymX), and 24 patients were diagnosed asymptomatically (AsymX). Twenty-eight variants in seven genes were identified in 36 patients (pathogenic/likely pathogenic n = 25; variant of unknown significance n = 3). Four of those variants were novel. All patients with LCHAD deficiency had the common variant (p.Glu474Gln) in HADHA and their phenotype was similar to the patients reported in the literature for this genotype. Only one patient with VLCAD deficiency had the common p.Val283Ala in ACADVL. The different genotypes in the SymX and AsymX groups for VLCAD deficiency presented with similar phenotypes. Eight patients were treated with carnitine supplementation [CTD (n = 6), CPT-II (n = 1), and MAD (n = 1) deficiencies]. Thirteen patients were treated with a long-chain fat restricted diet and MCT supplementation. A statistically significant association was found between rhabdomyolysis, and hypoglycemia in the SymX group compared to the AsymX group. A higher number of hospital admissions, longer duration of hospital admissions and higher CK levels were observed in the SymX group, even though the symptomatic group was only 37% of the study cohort.
Conclusion
Seven different mitochondrial long-chain fatty acid oxidation and carnitine metabolism defects were present in our study cohort. In our clinic, the prevalence of mitochondrial long-chain fatty acid oxidation and carnitine defects was 4.75%.
Supplementary Information
The online version contains supplementary material available at 10.1186/s13023-022-02512-5.
Keywords: Mitochondrial long-chain fatty acid oxidation, Carnitine metabolism defects, Medium chain triglycerides, Long-chain fat restricted diet, Newborn screening
Introduction
Mitochondrial long-chain fatty acid oxidation and carnitine metabolism defects are a group of inherited metabolic diseases. They are individually rare. The most commonly known mitochondrial long-chain fatty acid oxidation defects include long-chain 3-hydroxyacyl-CoA dehydrogenase (LCHAD) (encoded by HADHA; OMIM#600890) deficiency (OMIM#609016), very long-chain acyl-CoA dehydrogenase (VLCAD) (encoded by ACADVL; OMIM#609575) deficiency (OMIM#201475), carnitine transporter (encoded by SLC22A5; OMIM#603377) defect (CTD) (OMIM#212140), carnitine palmitoyltransferase I (CPT-I) (encoded by CPTIA; OMIM#600528) deficiency (OMIM#255120), carnitine palmitoyltransferase II (CPT-II) (encoded by CPT2; OMIM#600650) deficiency (OMIM#255110), carnitine acylcarnitine translocase (CACT) (encoded by SLC25A20; OMIM#613698) deficiency (OMIM#212138), and multiple acyl-CoA dehydrogenase (MAD) (encoded by ETFDH; OMIM#231675; ETFA; OMIM#608053 and ETFB; OMIM#130410) deficiency (OMIM#231680) [1–12].
The characteristic clinical presentations include acute hypoketotic hypoglycemia, encephalopathy, cardiomyopathy and myopathy [13, 14]. The symptoms can be precipitated by infections or prolonged fasting which results in hypoglycemia, elevated creatine kinase (CK), liver enzymes, lactate and ammonia levels. Plasma acylcarnitine profile and total and free carnitine levels can help identify specific enzyme or transporter deficiencies. The confirmation is by molecular genetic testing of candidate genes. Targeted next generation sequencing panel for myopathy and rhabdomyolysis typically includes these disorders [1, 15].
The mainstay of treatment is to prevent hypoglycemia and catabolism during intercurrent illness, vomiting or prolonged fasting which can result in death if untreated. Long-chain fat restricted diet, medium chain triglyceride (MCT), triheptanoin and carnitine supplementations are applied in mitochondrial long-chain fatty acid oxidation and carnitine metabolism defects [1, 15–21].
To report outcomes of mitochondrial long-chain fatty acid oxidation and carnitine metabolism defects, we performed a retrospective cohort study in the metabolic genetic clinic at our institution. We report on the phenotypic and genotypic spectrum of mitochondrial long-chain fatty acid oxidation and carnitine metabolism defects as well as their treatment outcomes. Furthermore, we compare the outcomes of patients identified by the manifestation of symptoms (SymX group) and asymptomatically by positive newborn screening or positive family history (AsymX group) in our study.
Methods
The Research Ethics Office, Health Research Ethics Board, University of Alberta (Study ID: Pro00108842) approved this retrospective cohort study. All patients with the mitochondrial long-chain fatty acid oxidation and carnitine metabolism defects were included. We divided patients into two groups to compare outcomes of those treated symptomatically and asymptomatically including Group 1) SymX group: diagnosed after the onset of symptoms; Group 2) AsymX group: diagnosed by positive family history or positive newborn screening (NBS).
We reviewed patient charts for clinical features, biochemical investigations, molecular genetic investigations, cardiac assessments, neuroimaging, treatments, and outcomes. We entered all information into an Excel database (Microsoft Corp., Redmond, WA, U.S.A.).
Molecular genetic investigations using patient and parent DNA samples were performed in clinical molecular genetic laboratories according to their methods. We applied American College of Medical Genetics and Genomics and the Association for Molecular Pathology (ACMG/AMP) variant classification guidelines for interpretation of variants [22]. We also searched all variants in the Genome Aggregation Database (gnomAD) (http://gnomad.broadinstitute.org/about) for their allele frequency in the general population [23].
We performed statistical analysis using R statistical software (version 4.0.2). Results are given as mean ± SD (range). Non-parametric Fisher’s exact and Wilcoxon rank-sum tests were chosen to compare outcomes between groups as indicated where appropriate. Results were considered statistically significant with a p-value of < 0.05.
Results
There were 38 patients (16 males, 22 females) from 27 unrelated families diagnosed with mitochondrial long-chain fatty acid oxidation and carnitine metabolism defects. Their current average age was 15.3 ± 16.9 standard deviation (SD) years (range 3 months-55 years). There were 15 adults (> 18 years of age) (average age of 36.5 ± 11.2 SD years and age range of 22–55 years) and 23 children (average age of 4.5 ± 3.1 SD years and range of 3 months-12 years). The mitochondrial long-chain fatty acid oxidation and carnitine metabolism defects diagnosed in our patient cohort were VLCAD (n = 5), LCHAD (n = 4), CACT (n = 3), MAD (n = 1), CPT-I (n = 13), CPT-II (n = 3) deficiencies and CTD (n = 9). The CTD, LCHAD and VLCAD deficiencies are included in the newborn screening programs in our Province of Alberta.
Fourteen patients were diagnosed symptomatically and were included in the SymX group. Their average age was 21.4 ± 18.3 SD years (range 7 months-55 years). Twenty-four patients were included in the AsymX group and were diagnosed either by positive NBS (n = 9) (average age of 5.8 ± 3.4 SD years and range of 3 months-12 years) or by positive family history (n = 15) (average age of 21 ± 19.1 SD years and range of 5 months-53 years). We summarized the type of mitochondrial long-chain fatty acid oxidation and carnitine metabolism defects, clinical features, biochemical features, genotypes, cardiac investigations, and treatments of patients in the SymX group in Table 1 and in the AsymX group in Table 2. Additionally, NBS results of the AsymX group are listed in Additional file1: Table S1.
Table 1.
Clinical, biochemical and molecular genetic features of SymX patients are summarized in Table 1
| Patient#/study ID/sex/diagnosis/age of diagnosis/current age | Presenting symptom (age of onset)/other clinical features | Initial investigations AC1,2,3/TC1,2,3/FC1,2,3/UOA | Molecular genetic investigations | Outcome |
|---|---|---|---|---|
| 1/1/F/CPT-I deficiency/2yrs/26yrs | Seizure (2yrs)/hypoglycemia, GTCS | N/N/N/↑AA, SUA, 3OH-SEA/NP | HMZ c.1436C > T (p.Pro479Leu) in CPTIA | Hospital admissions (n = 5), normal echo (26yrs) |
| 2/3/F/CPT- I deficiency /neonatal/3yrs | Hypoglycemia (neonatal)/no |
N/N/N/↑2-oxoadipic acid, 4-HPPA, 4-HPLA/NP |
HMZ c.1436C > T (p.Pro479Leu) in CPTIA | Normal |
| 3/7/F/CPT- I deficiency /2mo/3yrs | Hepatosplenomegaly (2mo)/no | N/NP/62.01/N/NP | HMZ c.1436C > T (p.Pro479Leu) in CPTIA | Normal echo (3yrs) |
| 4/10/F/CPT- I deficiency/neonatal/3yrs | Hypoglycemia (neonatal)/no | NP | HMZ c.1436C > T (p.Pro479leu) in CPTIA | Hospital admissions (n = 1) |
| 5/49/F/CPT- I deficiency/5yrs/22yrs | RM (5yrs)/hypoglycemia | N/NP/18.5/NP/NP | HMZ c.1436C > T (p.Pro479Leu) in CPTIA | Hospital admissions (n = 2) |
| 6/6/M/CPT-II deficiency/35yrs/51yrs | Myopathy (18yrs)/ RM, ATN, fatigue, myalgia, myoglobinuria | N/NP/NP/↑lactic acid/NP | NP | Hospital admissions (n = 3) |
| 7/14/F/CPT-II deficiency/15yrs/24yrs | RM (15yrs)/myalgia | C18:1OH = 0.05/N/N/NP/NP | CMP HTZ c.298delG (p.Val100Leufs*30)/c.338C > T (p.Ser113Leu) in CPT2 | Hospital admissions (n = 12) |
| 8/15/F/CPT-II deficiency/35yrs/36yrs | RM (20yrs)/myoglobinuria, myopathy, ATN | C16 = 0.74, C18:1 = 0.85, C18:2 = 0.51/N/N/NP/NP | CMP HTZ c.341-2621_1121del/c.338C > T (p.Ser113Leu) in CPT2 | Hospital admissions (n = 2), normal echo (35yrs) |
| 9/37/M/LCHAD deficiency/47yrs/55yrs | RM (4yrs)/retinopathy, myopathy, peripheral neuropathy | C16:1OH = 0.08, C16OH = 0.18, C18:1OH = 0.16, C18OH = 0.16/N/N/NP/NP | HTZ c.1528G > C (p.Glu474Gln) in HADHA | Hospital admissions (n = 16), normal echo (53yrs) |
| 10/42/M/VLCAD deficiency/10mo/25yrs | Hypoglycemia (10mo)/DCMP, myalgia, RM, myopathy, hypoglycemia | C14:1 = 4.15, C14:2 = 0.46/78/57/N/NP |
CMP HTZ c.605 T > A (p.Leu202His) /c.1182 + 1G > A in ACADVL |
Hospital admissions (n = 3), normal echo (23yrs) |
| 11/52/F/VLCAD deficiency/neonatal/7mo | Hypoglycemia (neonatal)/hypotonia | C14:2 = 0.94, C14:1 = 9.03/119.9/32.6/↑AA, SUA, SEA/NP |
CMP HTZ c.1182 + 1G > A/ c.1406G > A (p.Arg469Gln) in ACADVL |
Hospital admissions (n = 4), normal echo (2wks) |
| 12/45/M/CACT deficiency/neonatal/3yrs | Hypoglycemia (neonatal)/no |
C16 = 1.36, C18:1 = 0.70, C16-DC = 0.15 /N/N/NP/NP |
CMP HTZ c.897dupC (p.Asn300Glnfs*24)/c.269 T > G (p.Phe90Cys) in SLC25A20 | Normal echo (3yrs) |
| 13/47/M/CACT deficiency/neonatal/deceased | Hypotonia, cardiac arrest, lactic acidemia, hypoglycemia (neonatal)/no | C16 = 13.66, C18:1 = 4.31/N/20.51/NP/NP |
CMP HTZ c.326 + 1delG (IVS3 + 1delG) /c.10C > T (p.Gln4X) in SLC25A20 |
Hospital admissions (n = 1), left ventricular dysfunction, MV, TV tricuspid regurgitation in echo (5 d) |
| 14/48/M/MAD deficiency/4yrs/26yrs | Lethargy (8mo)/seizure, hypoglycemia | Elevated C4-C14a/106/40/↑AA/NP |
CMP HTZ c.524G > A (p.Arg175His) /c.1001 T > C (p.Leu334Pro) in ETFDH |
Hospital admissions (n = 3), normal echo (24yrs) |
Reference Ranges: Free carnitine1 7.3–30.4; Free carnitine2 18.6–55.0; Free carnitine3 25.3–57.0; Total carnitine1 14.0–47.0; Total carnitine2 24.9–72.1; Total carnitine3 32.5–73.6; C161 0.04–0.41; C162 0.03–0.22; C163 0.03–0.19; C16-DC1 < 0.06; C16-DC2 < 0.03; C16-DC3 0.03–0.09; C16-OH1 < 0.05; C16-OH2 < 0.04; C16-OH3 < 0.02; C18:11 0.04–0.20; C18:12 0.03–0.23; C18:13 0.03–0.26; C18:21 < 0.10; C18:22 < 0.12; C18:23 < 0.13; C18-OH1 < 0.03; C18-OH2 < 0.03; C18-OH3 < 0.02; C18:1OH1 < 0.02; C18:1OH2 < 0.03; C18:1OH3 < 0.02; C16:1OH1 < 0.27; C16:1OH2 < 0.11; C16:1OH3 < 0.03; C14:11 < 0.26; C14:12 < 0.28; C14:13 < 0.11; C14:21 < 0.10; C14:22 < 0.13; C14:23 < 0.11
1 Indicates reference range for age group newborn-2 months
2 Indicates reference ranges for age group 2 months-18 years
3 Indicates reference ranges for age group 18 years and up
4-HPPA 4-OH-phenylpyruvic acid; 4-HPLA 4-hydroxyphenyllactic acid; AA adipic acid; AC acylcarnitines; ATN acute tubular necrosis; CACT carnitine acylcarnitine translocase; Car carnitine; CMP HTZ compound heterozygous; CPT-I carnitine palmitoyltransferase I; CPT-II carnitine palmitoyltransferase II; CTD carnitine transporter defect; DCMP dilated cardiomyopathy; echo echocardiography; FC free carnitine; F female; HMZ homozygous; LCHAD long-chain 3-hydroxyacyl-CoA dehydrogenase; MAD multiple acyl-CoA dehydrogenase; mo months; MV mitral valve; NB newborn; N normal; NP not performed; NR not reported; RM rhabdomyolysis; SEA sebacic acid; SUA suberic acid; TV tricuspid valve; UOA urine organic acids; yrs year(s); VLCAD very long-chain acyl-CoA dehydrogenase
aResult report did not provide values
Table 2.
Clinical, biochemical and molecular genetic features of AsymX patients are summarized in Table 2
| Patient #/study ID/sex/diagnosis/age of diagnosis/current age | Presenting symptom (age of onset)/other clinical features | Initial investigations AC1,2,3/TC1,2,3/FC1,2,3/UOA/TRR% | Molecular genetic investigations | Outcome |
|---|---|---|---|---|
| 1/2/M/CPT-I deficiency/neonatal/3yrs | No | N/NP/75.4/↑3-HIVA /NP | HMZ c.1436C > T (p.Pro479Leu) in CPTIA | Normal |
| 2/5/F/CPT-I deficiency/6yrs/26yrs | No | NP | HMZ c.1436C > T (p.Pro479Leu) in CPTIA | Normal |
| 3/8/M/CPT-I deficiency/45yrs/53yrs | No | NP | HMZ c.1436C > T (p.Pro479Leu) in CPTIA | Normal |
| 4/9/F/CPT-I deficiency/neonatal/6yrs | No | N/N/117.75/NP/NP | HMZ c.1436C > T (p.Pro479Leu) in CPTIA | Normal echo (4yrs) |
| 5/11/F/CPT-I deficiency/39yrs/48yrs | No | NP | HMZ c.1436C > T (p.Pro479Leu) in CPTIA | Normal |
| 6/12/F/CPT-I deficiency/neonatal/2yrs | No | N/66.9/51.3/N/NP | HMZ c.1436C > T (p.Pro479Leu) in CPTIA | atrial septal aneurysm, PFO, ASD, PDA (1mo) |
| 7/50/F/CPT-I deficiency/2yrs/3yrs | No | NP | HMZ c.1436C > T (p.Pro479Leu) in CPTIA | Normal |
| 8/51/M/CPT-I deficiency/neonatal/5yrs | No | NP | HMZ c.1436C > T (p.Pro479Leu) in CPTIA | Normal |
| 9/18/M/CTD deficiency/neonatal/7yrs | No | NP/NP/NP/NP/NP | HMZ c.769C > T (p.Arg257Trp) in SLC22A5 | Normal echo (5yrs) |
| 10/19/F/CTD deficiency/32yrs/37yrs | No | NP/2.2/1.7/N/90.88 | HMZ c.845G > A (p.Arg282Gln) in SLC22A5 | Normal |
| 11/24/M/CTD deficiency/neonatal/3yrs | No | N/6.4/4.2/N/NP | HMZ c.845G > A (p.Arg282Gln) in SLC22A5 | Hospital admissions (n = 2), normal echo (2yrs) |
| 12/25/M/CTD deficiency/neonatal/3mo | No | NP/6.1/4.8/N/NP | HMZ c.845G > A (p.Arg282Gln) in SLC22A5 | Normal |
| 13/22/F/CTD deficiency/38yrs/44yrs | No/ fatigue (retrospectively at the time of diagnosis) | NP/ < 2/ < 2/NP/NP | HMZ c.248G > T (p.Arg83Leu) in SLC22A5 | Normal echo (42yrs) |
| 14/23/F/CTD deficiency/28yrs/40yrs | No/ fatigue (retrospectively at the time of diagnosis) | NP/ < 2/ < 2/NP/NP | NP | Normal echo (30yrs) |
| 15/28/M/CTD deficiency/neonatal/6yrs | No | N/10.6/8.4/NP/NP | CMP HTZ c.1463G > A (p.Arg488His)/c.424G > T (p.Ala142Ser)/c.136C > T (p.Pro46Ser) in SLC22A5 | Normal echo (5yrs) |
| 16/32/F/CTD deficiency/26yrs/33yrs | No/ fatigue (retrospectively at the time of diagnosis) | NP/10.1/5.1/N/NP |
CMP HTZ c.364G > T (p.Asp122Tyr)/ c.505C > T (p.Arg169Trp) in SLC22A5 |
Normal echo (27yrs) |
| 17/36/M/CTD deficiency/neonatal/9yrs | No/ myalgia (8yrs) | NP/NP/NP/NP/NP |
CMP HTZ c.424G > T (p.Ala142Ser) /c.1463G > A (p.Arg488His) /c.1324GC_1325AT (p.Ala442Ile) in SLC22A5 |
Normal echo (6yrs) |
| 18/38/M/LCHAD deficiency/neonatal/6yrs | No/ RM (2yrs), retinopathy, myopathy (6 yrs) |
C16OH = 0.73, C18OH = 0.87, C18:1OH = 0.54/N/N/N/NP |
HMZ c.1528G > C (p.Glu474Gln) in HADHA | Hospital admissions (n = 6), normal echo (6yrs) |
| 19/39/F/LCHAD deficiency/neonatal/3yrs | No/RM, myopathy |
C16OH = 1.05, C18OH = 0.91, C18:1OH = 1.1/N/NP/NP/NP |
HMZc.1528G > C (p.Glu474Gln) in HADHA | Hospital admissions (n = 4), normal echo (3yrs) |
| 20/53/M/LCHAD deficiency/neonatal/7mo | Hypoglycemia (neonatal, but diagnosed prenatally) | C16:1OH = 0.33, C16OH = 0.78, C18:1OH = 0.45, C18OH = 0.40/N/N/N/NP | HMZ c.1528G > C (p.Glu474Gln) in HADHA | Hospital admissions (n = 2), normal echo (12yrs) |
| 21/40/F/VLCAD deficiency/neonatal/7yrs | No/ RM, myopathy (3 yrs) | C14:1 = 0.13, C14:2 = 0.06/N/N/NP/NP | CMP HTZ c.1375dupC (p. p.Arg459ProfsX4)/c.1700G > A (p.Arg567Gln) in ACADVL | Hospital admissions (n = 1), dilated ascending aorta in echo (6yrs) |
| 22/43/F/VLCAD deficiency/neonatal/12yrs | No/RM, myalgia (12 yrs) | N/N/N/↑AA, SUA, SEA/NP |
CMP HTZ c.848 T > C (p.Val283Ala)/ c.1375dupC (p.Arg459ProfsX4) in ACADVL |
Hospital admissions (n = 1) |
| 23/44/F/VLCAD deficiency/neonatal/9yrs | No/ myalgia (4yrs) | N/N/N/NP/NP |
CMP HTZ c.779C > T (p.Thr260Met/ c.1733 T > C (p.Met578Thr) in ACADVL |
Hospital admissions (n = 3), normal echo (7yrs) |
| 24/46/F/CACT deficiency/4yrs/8yrs | No |
C16 = 0.85, C18:1 = 0.83, C18:2 = 0.25/N/ 14.9/NP/NP |
CMP HTZ c.897dupC (p.Asn300Glnfs*24)/c.269 T > G (p.Phe90Cys) in SLC25A20 | PDA in echo (6mo) |
Reference Ranges: Free carnitine1 7.3–30.4; Free carnitine2 18.6–55.0; Free carnitine3 25.3–57.0; Total carnitine1 14.0–47.0; Total carnitine2 24.9–72.1; Total carnitine3 32.5–73.6; C161 0.04–0.41; C162 0.03–0.22; C163 0.03–0.19; C16-DC1 < 0.06; C16-DC2 < 0.03; C16-DC3 0.03–0.09; C16-OH1 < 0.05; C16-OH2 < 0.04; C16-OH3 < 0.02; C18:11 0.04–0.20; C18:12 0.03–0.23; C18:13 0.03–0.26; C18:21 < 0.10; C18:22 < 0.12; C18:23 < 0.13; C18-OH1 < 0.03; C18-OH2 < 0.03; C18-OH3 < 0.02; C18:1OH1 < 0.02; C18:1OH2 < 0.03; C18:1OH3 < 0.02; C16:1OH1 < 0.27; C16:1OH2 < 0.11; C16:1OH3 < 0.03; C14:11 < 0.26; C14:12 < 0.28; C14:13 < 0.11; C14:21 < 0.10; C14:22 < 0.13; C14:23 < 0.11
1 Indicates reference range for age group newborn-2 months
2 Indicates reference ranges for age group 2 months-6 years
3 Indicates reference ranges for age group 18 years and up
3-HIVA 3-OH-isovaleric acid; AA adipic acid; AC acylcarnitines; ASD atrial septal defect; CACT carnitine acylcarnitine translocase; Car carnitine; CPT-I carnitine palmitoyltransferase I; CPT-II carnitine palmitoyltransferase II; CMP HTZ compound heterozygous; CTD carnitine transporter defect; Echo echocardiography; FC free carnitine; F female; HMZ homozygous; LCHAD long-chain 3-hydroxyacyl-CoA dehydrogenase; MAD multiple acyl-CoA dehydrogenase; mo months; NB newborn; N normal; NP not performed; NR not reported; PDA patent ductus arteriosus; PFO patent foramen ovale; RM rhabdomyolysis; SEA sebacic acid; SUA suberic acid; TRR tubular reabsorption rate; UOA urine organic acids; yrs year(s); VLCAD very long-chain acyl-CoA dehydrogenase
Thirty-six patients had molecular genetic confirmation of their specific mitochondrial long-chain fatty acid oxidation and carnitine metabolism defects. All variants identified in our study cohort were re-classified using ACMG/AMP criteria (Additional file 1: Table S2). Unfortunately, two patients (CPT-II n = 1; CTD n = 1) did not have molecular genetic investigations due to the limited publicly available funding for molecular genetic investigations at the time of their diagnosis. In one patient with LCHAD deficiency, a single variant was identified. There were 28 different variants from 36 patients including 25 pathogenic or likely pathogenic variants and three variants of unknown significance. The most common variant type was missense variant (n = 20). Fourteen patients had compound heterozygous variants and 21 patients had homozygous variants (Tables 1 and 2). Four of the variants were not reported in the literature previously including CPT2 (n = 2), and SLC25A20 (n = 2).
Eight patients were treated with carnitine supplementation [CTD (n = 6), CPT-II (n = 1), and MAD (n = 1) deficiencies]. The average carnitine dose was 48.6 mg/kg/day (range 6.1–125 mg/kg/day). Thirteen patients were treated with a long-chain fat restricted diet and MCT supplementation (Table 3). Diet therapy recommendations were individualized based on the type of mitochondrial long-chain fatty acid oxidation and carnitine metabolism defects as well as clinical judgement regarding disease severity. The most severe patients were prescribed the greatest long chain fat restrictions and shortest fasting allowances. Recommendations for long chain fat restriction ranged from 5% of total energy for severe phenotypes to 35% of total energy for mild or moderate phenotypes. MCT was supplemented as an alternative energy source to meet total fat requirements, providing 0 to 35% of total energy. MCT supplementation was provided using either medical formulas (Lipistart) or module forms (MCT oil, MCT procal, Betaquick, or Liquigen). Age dependent maximum hours of fasting recommendations were applied. The longest allowed overnight fasting was up to 12 h in older children and adults. For illness management, carbohydrate was provided either through glucose polymers (such as Solcarb) or IV glucose, depending on the patient’s ability to tolerate feeding (and thus stay at home) versus require admission. MCT supplementation was also used in some cases as an alternative energy source to long-chain fat. Details of complex carbohydrate and/or MCT intakes prior to exercise as well as the compliance and moderation in exercise level are summarized in Additional file 1: Table S3.
Table 3.
All SymX and AsymX patients on the diet are summarized in Table 3
| Groups | Patient#/study ID/diagnosis | Long chain fat intake | MCT | Protein Intake (g/kg/d) |
||
|---|---|---|---|---|---|---|
| Recommended intake | Actual intake | Recommended intake | Actual intake | |||
| SymX group | 6/6/CPTII def | 20% | 30% | NA | NA | 1.09 |
| 7/14/CPTII def | 20% | < 20% | 10–15% | 1% | 1.17 | |
| 8/15/CPTII def | 35% | 44% | 10–20% | 10–20% | 1.33 | |
| 9/37/LCHAD def | 10% | 8% | 10–15% | 0% | NA | |
| 10/42/VLCAD def | 10–15% | 6% | 10–15% | 21% | 2.12 | |
| 11/52/VLCAD def | 5% | 5% | 35% | 35% | 2.94 | |
| 14/48/MAD def | 25–30% | 33% | NA | NA | 2.32 | |
| AsymX group | 24/38/LCHAD def | 10% | 7% | 15–25% | 23% | 2.57 |
| 25/39/LCHAD def | 10–15% | 10% | 20–25% | 22% | 2.13 | |
| 26/53/LCHAD def | 10% | 10% | 30% | 32% | 2.30 | |
| 27/40/VCLAD def | 15–25% | 26% | 5% | 0% | 0.97 | |
| 28/43/VLCAD def | 15–25% | 13% | 13% | 13% | 1.51 | |
| 29/44/VLCAD def | 10–20% | 10–12% | 15–20% | 15% | 2.15 | |
CACT carnitine acylcarnitine translocase; CPT-I carnitine palmitoyltransferase I; CPT-II carnitine palmitoyltransferase II; CTD carnitine transporter defect; def deficiency; LCHAD long-chain 3-hydroxyacyl-CoA dehydrogenase; MAD multiple acyl-CoA dehydrogenase; VLCAD very long-chain acyl-CoA dehydrogenase
Clinical and biochemical features and treatment outcomes of patients in the SymX group (n = 14)
There were 14 patients with CPT-I (n = 5), CPT-II (n = 3), LCHAD (n = 1), VLCAD (n = 2), CACT (n = 2) and MAD (n = 1) deficiencies. The average current age was 21.4 ± 18.3 SD years (range 7 months-55 years). The average age of symptom onset was 4.5 ± 7.4 SD years (range newborn-23 years). The average age of diagnosis was 10.6 ± 16.1 SD years (range newborn-47 years). Two patients with CPT-I deficiency presented at birth with hypoglycemia and two patients with CACT deficiency presented with symptoms prior to receiving NBS results. The average lack of time between the symptom onset and diagnosis was 6.1 ± 12.1 SD years (range newborn-43 years). The average duration of follow-up was 10.6 ± 8.8 years SD (range 7 months-24.2 years). The symptom frequency for each disorder is summarized in Table 4.
Table 4.
Frequency of clinical features of the SymX and AsymX patients are summarized in Table 4
| Features | Abnormal clinical, biochemical and organ involvement | SymX group (n = 14) |
AsymX group (NBS or asymptomatic with positive family history (n = 21) |
Statistical analysis (Fisher’s Exact Test) |
|---|---|---|---|---|
| Clinical features | Seizure |
14.3% (n = 2) |
0% | 0.1529 |
| Myalgia |
35.7% (n = 5) |
9.5% (n = 2) |
0.0897 | |
| Fatigue |
14.3% (n = 2) |
0% | 0.1529 | |
| Lethargy |
14.3% (n = 2) |
4.8% (n = 1) |
0.5508 | |
| Hepatomegaly |
14.3% (n = 2) |
0% | 0.1529 | |
| Hypotonia |
7.1% (n = 1) |
4.8% (n = 1) |
1 | |
| Headache | 0% | 0% | 1 | |
| Peripheral neuropathy |
7.1% (n = 1) |
0% | 0.4000 | |
| Retinopathy/maculopathy |
7.1% (n = 1) |
4.8% (n = 1) |
1 | |
| Investigations | Rhabdomyolysis |
50% (n = 7) |
9.5% (n = 2) |
0.0153* |
| Lactic acidemia |
14.3% (n = 2) |
0% | 0.1529 | |
| Myopathy |
14.3% (n = 2) |
0% | 0.1529 | |
| Cardiac arrhythmia |
7.1% (n = 1) |
0% | 0.4000 | |
| Dilated cardiomyopathy |
7.1% (n = 1) |
0% | 0.4000 | |
| Acute tubular necrosis (acute kidney insufficiency) |
14.3% (n = 2) |
0% | 0.1529 | |
| Hypoglycemia |
71.4% (n = 10) |
4.8% (n = 1) |
5.126 × 10–5* | |
| Myoglobinuria |
21.4% (n = 3) |
0% | 0.0556 | |
| Hospital admissionsa | CTD | No patients in this group with CTD |
0% (n = 0 out of 6) |
1 |
| CPT-I deficiency |
40% (n = 2 out of 5) |
0% (n = 0 out of 8) |
0.1282 | |
| CACT deficiency |
50% (n = 1 out of 2) |
100% (n = 1 out of 1) |
1 | |
| LCHAD deficiency |
100% (n = 1 out of 1) |
100% (n = 3 out of 3) |
1 | |
| VLCAD deficiency |
100% (n = 2 out of 2) |
100% (n = 3 out of 3) |
1 | |
| Treatmenta | Carnitine |
14.3% (n = 2 out of 14) |
28.6% (n = 6 out of 21) |
0.4307 |
| Long-chain fat restriction |
50% (n = 7 out of 14) |
28.6% (n = 6 out of 21) |
0.2882 |
CACT carnitine acylcarnitine translocase; CPT-I carnitine palmitoyltransferase I; CPT-II carnitine palmitoyltransferase II; CTD carnitine transporter defect; LCHAD long-chain 3-hydroxyacyl-CoA dehydrogenase; MAD multiple acyl-CoA dehydrogenase; VLCAD very long-chain acyl-CoA dehydrogenase
*Significant p-value (< 0.05)
aValues are reported as averages. Long-chain fat restriction and MCT supplementation indicate percentage of daily intake
The most common features were hypoglycemia (n = 10) and rhabdomyolysis (n = 7). Patients with CPT-I deficiency had hypoglycemia (n = 4), seizure (n = 1), rhabdomyolysis (n = 1) and hepatomegaly (n = 2). Patients with CPT-II deficiency had rhabdomyolysis (n = 3), myoglobinuria (n = 2), myalgia (n = 3), acute tubular necrosis (n = 2), hypoglycemia (n = 1) and fatigue (n = 1). One patient with LCHAD deficiency had retinopathy, rhabdomyolysis, fatigue, myalgia, myopathy, myoglobinuria, and peripheral neuropathy (symptoms of loss of deep tendon reflexes and pinprick and light touch sensation in lower extremities). Patients with VLCAD deficiency had hypoglycemia (n = 2), rhabdomyolysis (n = 2), cardiomyopathy (n = 1), myalgia (n = 1), myopathy (n = 1), lethargy (n = 1), hypotonia (n = 1), and lactic acidemia (n = 1). Patients with CACT deficiency had hypoglycemia (n = 2), hypotonia (n = 1), and lactic acidemia (n = 1). One patient with MAD deficiency had hypoglycemia, seizure, and lethargy. Interestingly one patient with VLCAD deficiency presented with neonatal hypoglycemia and was admitted to neonatal intensive care unit prior to NBS results becoming available.
Nine patients had alanine aminotransferase (ALT) (n = 9) and/or aspartate aminotransferase (AST) (n = 8) during regular follow-up appointments. Six patients (CPT-I n = 1; CPT-II n = 3; VLCAD n = 1 and CACT n = 1 deficiencies) had normal ALT levels. Six patients (CPT-I n = 2; CPT-II n = 2; VLCAD n = 1 and CACT n = 1 deficiencies) had normal AST levels.
Seven patients [(CPT-II (n = 3), LCHAD (n = 1), VLCAD (n = 2), MADD (n = 1) deficiencies] were treated with a long-chain fat restricted diet. The average age of diet initiation was 20.4 ± 18.2 SD years (range newborn-48 years). The average recommended long-chain fat intake was 19% ± 0.10 SD of total daily intake (range 5–35%), whereas the average of actual long-chain fat intake was 24% ± 0.1 SD of total daily intake (range 5–44%). The average recommended MCT intake was 16% ± 0.1 SD of daily total intake (range 0–35%), whereas the average actual MCT intake was 13% ± 0.1 SD of total daily intake (range 0–21%). We recommended pre-exercise complex carbohydrates in two patients (CPT-II n = 1; and MAD n = 1 deficiencies) to prevent rhabdomyolysis associated with exercise. Additionally, we recommended pre-exercise complex carbohydrates and MCT oil (in any form) in four patients (VLCAD n = 1; LCHAD n = 1; and CPT-II n = 2 deficiencies) to prevent rhabdomyolysis associated with exercise (Additional file 1: Table S3). Three patients were compliant with these recommendations including patients with CPT-II (n = 1), VLCAD (n = 1) and MAD (n = 1) deficiencies. Three patients were not compliant with our recommendations and for two of them with CPT-II deficiency we recommended limited level of exercise to prevent repeated rhabdomyolysis episodes. One non-compliant patient with LCHAD deficiency did not exercise. None of the patients received additional oil or cornstarch supplementation at the last assessment. One patient with CPT-II deficiency was non-compliant since the diagnosis (at age 9 years) due to very high exercise levels and failure to consume an appropriate amount of carbohydrates leading to several hospital admissions for the treatment of rhabdomyolysis. One patient with LCHAD deficiency had difficulty in being compliant with the long-chain fat restricted diet leading to multiple hospital admissions for rhabdomyolysis since the diagnosis. The adherence to the diet was improved with the use of nasogastric feeds, but this was not sustainable for long-term.
Adherence to diet recommendations was suboptimal in the SymX group. 50% (7 out of 14) of the patients were prescribed diet therapy including long chain fat restriction and, in some cases, MCT supplementation. 43% (3 out of 7) of the patients met their long chain fat goals within 1% of max recommendations or took less long chain fat than prescribed. Those that did not meet their long chain fat goals were not monitoring daily fat intake so had day-to-day variability in fat consumption. 71% (5 out of 7) of the patients were prescribed MCT supplements, where 60% (3 out of 5) of the patients met their goals. Two patients, who did not meet MCT goals, struggled with gastrointestinal side effects. One patient experienced significant abdominal discomfort and bloating. Another patient’s baseline irritable bowel syndrome like symptoms (abdominal discomfort and diarrhea) were exacerbated.
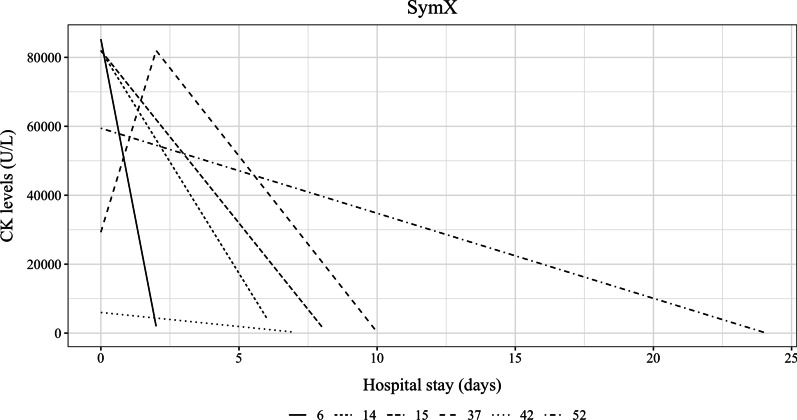
The patient with LCHAD deficiency had 16 acute intercurrent illnesses. One patient with VLCAD deficiency had four acute intercurrent illnesses and the other patient with VLCAD deficiency had 7 acute intercurrent illnesses. One patient with CACT deficiency reported one acute intercurrent illness treated at home. The number of episodes of elevated CK levels ranged from 0 to 19. The average peak CK during acute intercurrent illness or catabolism was 72,783.7 ± 60,708.7 SD U/L (range 246–197,836) (Additional file 1: Table S3). We had details for hospital admission and the CK levels for six patients as depicted in Fig. 1.
Fig. 1.
Hospital admissions of six patients with highest CK level at the time of the admission and duration of admission with the discharge CK levels are depicted in Fig. 1 for SymX group
Clinical and biochemical features and treatment outcomes of patients in the AsymX group (n = 24)
Twenty-four patients were asymptomatic at the time of diagnosis and were identified by positive NBS (n = 9) or by positive family history (n = 15) (Table 2). Patients with positive NBS included VLCAD (n = 2), LCHAD (n = 1), CPT-I (n = 1) deficiencies and CTD (n = 5). Patients with positive family history included CPT-I (n = 7), LCHAD (n = 2), VLCAD (n = 1), and CACT (n = 1) deficiencies and CTD (n = 4). The average current age was 15.3 ± 16.9 SD years (range 3 months-53 years). The average duration of follow-up was 5.6 ± 3.8 SD years (range 3 months-12 years) in 19 patients, as five asymptomatic patients with CPT-I deficiency were only seen once.
Interestingly, three patients with CTD (mothers of newborn screening positive babies) reported several symptoms retrospectively including fatigue (n = 3), muscle pain (n = 2), exercise intolerance (n = 1), and symptoms of presumed hypoglycemia (e.g., hungry, sweating) (n = 1).
Nine patients developed symptoms later including three patients with positive family history and six patients identified by positive NBS. The average lack of time between the diagnosis and symptom onset was 3.1 ± 2.8 SD years (range newborn-8 years). In three patients with positive family history, the symptom onset was during intercurrent illness including two patients with LCHAD deficiency and one patient with CACT deficiency. They presented with rhabdomyolysis, lethargy, hypotonia and hypoglycemia. The average age of symptom onset was 2 ± 3.5 years SD (range newborn-6 years). The average time between the diagnosis and symptom onset was 7.9 ± 13.8 months SD (range newborn-2 years). Six patients identified by positive NBS had subsequent symptom onset including retinopathy in one patient with LCHAD deficiency, rhabdomyolysis in two patients with LCHAD and myalgia in two patients with VLCAD deficiency (Table 2). The average age of symptom onset was 3.2 ± 2.7 SD years (range 1 month-8 years).
Twelve patients had alanine aminotransferase (ALT) (n = 12) and/or aspartate aminotransferase (AST) (n = 9) during regular follow-up appointments. Ten patients (VLCAD n = 3; LCHAD n = 1 and CACT n = 1 deficiencies, and CTD n = 5) had normal ALT levels. Nine patients (CTD n = 3; LCHAD n = 2; VLCAD n = 3 and CACT n = 1 deficiencies) had normal AST levels.
All patients with CPT-I deficiency (n = 8) were asymptomatic. One of the patients with CPT-I deficiency had atrial septal aneurysm with a residual patent foramen ovale and atrial septal defect, residual patent ductus arteriosus, and mild mitral incompetence in echocardiography at age one month. None of the patients with CTD, CACT, LCHAD and VLCAD deficiencies had cardiomyopathy.
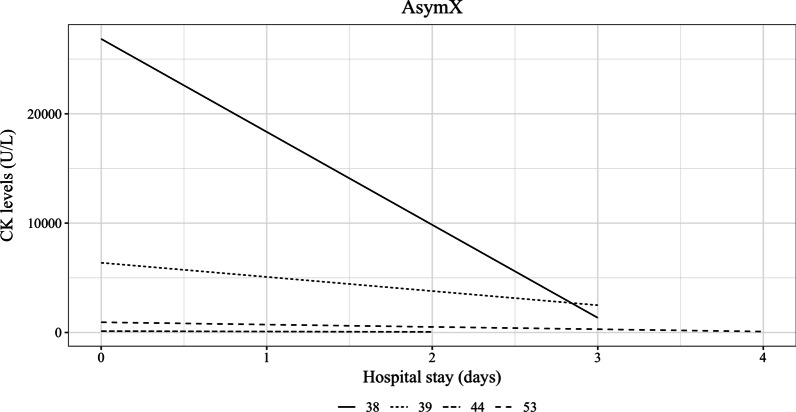
All patients received illness and emergency management. None of the patients with CPT-I deficiency required any treatments. Eight patients with CTD were treated with carnitine supplementation; one was not compliant with carnitine supplementation. All patients with LCHAD (n = 3) and VLCAD (n = 3) deficiencies received long-chain fat restricted diet. The average age of diet initiation was 2.2 ± 4.8 SD months (range newborn-13 months). The average recommended intake of long-chain fat was 18% of total energy intake ± 0.1 SD (range 10–25%). Whereas the average actual intake of long-chain fat was 13% ± 0.1 SD of total energy intake (range 7–26%). The average recommended intake of MCT was 20% of total energy intake ± 0.1 SD (range 13–35%). Whereas the average actual intake of MCT was 19% of total energy intake ± 0.1 SD (range 13–35%). Four patients received walnut oil supplementation (average 1.63 ml, range 0.5–4.0 ml), one patient received flax oil supplementation (0.5 ml), one patient received canola oil supplementation (30 ml), and two patients received cornstarch supplementation (average 0.8 mg/kg/day, range 0.6–0.9 mg/kg/day). The ASympX group demonstrated good adherence to dietary recommendations. 25% (6 out of 24) of the patients were prescribed diet therapy including long chain fat restriction and MCT supplementation. 100% of the patients met their long chain fat goals within 1% of max recommendations or took less long chain fat than prescribed. 83% (5 out of 6) of the patients met their MCT goals. The exception was one child who refused MCT supplementation due to poor palatability. We recommended pre-exercise complex carbohydrates in two patients with VLCAD deficiency to prevent rhabdomyolysis associated with the exercise. Additionally, we recommended pre-exercise complex carbohydrates and MCT oil (in any form) in two patients (VLCAD n = 1; and LCHAD n = 1 deficiencies) to prevent rhabdomyolysis associated with the exercise (Additional file 1: Table 3). Three patients were compliant with these recommendations including patients with VLCAD (n = 2) and LCHAD (n = 1) deficiencies and none of these patients had a reduced exercise level. Unfortunately, we are not certain about the compliance of one patient with VLCAD deficiency as the patient is no longer followed by our clinic due to moving out of province. The average number of acute intercurrent illnesses was 6.3 ± 3.3 SD (range 2–10) in patients with LCHAD deficiency. The average number of acute intercurrent illnesses was 4.7 ± 3.8 SD (range 2–9) in patients with VLCAD deficiency. The patient with CACT deficiency reported two acute intercurrent illnesses treated at home. The number of acute episodes of elevated creatine kinase (CK) levels ranged from 0 to 10. The average peak CK during acute intercurrent illness or catabolism was 22,842.5 ± 32,740.3 U/L SD (range 386–70,000) (Additional file 1: Table 3). Details for hospital admission and the CK levels were available for four patients and depicted in Fig. 2.
Fig. 2.
Hospital admissions of four patients with highest CK level at the time of the admission and duration of admission with the discharge CK levels are depicted in Fig. 2 for AsymX group
Comparison of patients in the SymX group and AsymX group
The symptom frequency for each group is summarized in Table 4. We excluded three patients who were diagnosed due to their positive family history but reported symptoms when they were asked for more information (Table 2). Statistically significant association was found for rhabdomyolysis, and hypoglycemia in the SymX group compared to AsymX group.
The total number of hospital admissions was 16 in one patient with LCHAD deficiency in the SymX group (n = 1), whereas the total number of hospital admissions was 12 in three LCHAD patients in the AsymX group (n = 3) (Individual admissions: 6, 4, 2) (p = 0.500, Wilcoxon rank-sum). The total number of hospital admissions was seven in two patients with VLCAD deficiency in the SymX group (n = 2) (individual admissions: 3, 4), whereas the total number of hospital admissions were five in three VLCAD patients in the AsymX group (n = 3) (individual admissions: 1, 1, 3) (p = 0.224, Wilcoxon rank-sum). The total number of hospital admissions was seven in five patients with CPT-I deficiency in the SymX group (n = 5) (individual admissions: 5, 0, 0, 1, 1), whereas none of the patients with CPT-I deficiency in the AsymX group (n = 8) (p = 0.0225, Wilcoxon rank-sum) had any hospital admissions. A higher number of hospital admissions, longer duration of hospital admissions and higher CK levels were observed in the SymX group, even though the SymX group was only 37% of the study cohort.
Discussion
We report 38 patients with seven different mitochondrial long-chain fatty acid oxidation and carnitine metabolism defects in our retrospective study from a single metabolic genetics center at our institution. The two most common defects were CPT-I deficiency (27%) and CTD (24%). Interestingly, 38% of the patients with CPT-I deficiency were identified symptomatically, whereas none of the patients with CTD were diagnosed symptomatically but identified due to positive NBS of CTD in their children. Asymptomatic diagnosis and application of treatments appears to decrease the number and duration of hospital admissions as well as peak CK levels. Our study reports the genetic landscape of mitochondrial long-chain fatty acid oxidation and carnitine metabolism defects in pediatric and adult patients from a single center.
The prevalence of maternal CTD was about 1 in 33,000 in a study from Taiwan [24]. In 91 families with abnormal NBS for CTD, 14 mothers were identified with two variants in SLC22A5. Eleven of those mothers were asymptomatic and their age ranged from 21 to 43 years at the time of the publication. Fatigability was reported in three of those mothers (21%) [25]. Interestingly, in our study 33% of the mothers reported symptoms at the time of their identification due to the positive NBS of CTD in their children. It is possible that more mothers with maternal CTD may have symptoms and their diagnosis may improve their quality of life and prevent deaths secondary to cardiomyopathy.
The prevalence of CPT-I deficiency in live newborns in three Canadian Territories were reported in 2010. The study genotyped all newborns born in Yukon, Northwest Territories and Nunavut for the p.Pro479Leu variant in CPT1A in 2006. The homozygosity rate was 0%, 3%, and 64% respectively in those Canadian Territories [26]. The prevalence of hypoglycemia in term newborns from the Kivalliq region of Nunavut was 22% in newborns with homozygous p.Pro479Leu variant in CPT1A [27]. Interestingly, 80% of the SymX patients with CPT-I deficiency had hypoglycemia in our patient cohort. None of the individuals with AsymX CPT-I deficiency reported any symptoms after the genotypic diagnosis. It is important to genotype symptomatic individuals, as asymptomatic individuals do not report any symptoms to the best of our knowledge. If there is a positive family history for symptomatic CPT-I deficiency, newborns in that family can be genotyped to prevent symptoms by application of illness management and emergency letters.
Trauma exposure and post-traumatic stress disorder like behavior in mice were studied using plasma and cerebellum metabolomics analysis. Investigators identified that medium and long chain acylcarnitines were elevated including C8, C12, C14, C16, C18:1 and C18. Additionally, free carnitine was reduced [28]. Interestingly, five adult patients had either suspected or diagnosed mental health condition in our study. Depression was reported in one patient with LCHAD, one patient with CPT-II deficiencies and one patient with CTD. Depression and anxiety were reported in one patient with CPT-I deficiency. Attention deficit disorder was reported in one patient with MAD deficiency. It is important to monitor abnormalities in acylcarnitine and carnitine levels in these patients with mitochondrial long-chain fatty acid oxidation and carnitine metabolism defects. This may increase our understanding of mental health conditions and help formulate better treatments.
Carnitine is a cofactor that transports long-chain fatty acids across the inner mitochondrial membrane for beta oxidation to provide energy to the skeletal and cardiac muscle. Energy demands of skeletal and cardiac muscle strongly require beta oxidation. Approximately 98% of the body’s carnitine is located within cardiac and skeletal muscle tissues. Orally taken carnitine is transported to skeletal and cardiac muscle via carnitine transporters. The half-life of a single dose (2 g) of oral l-carnitine is 60.3 ± 15 min and requires frequent oral intake to maintain carnitine levels. Oral l-carnitine intake of 2 g twice a day increases muscle carnitine by 21% of baseline. Carnitine supplementation increases plasma carnitine levels in individuals with CTD but does not normalize completely. High dose carnitine supplementation (up to 200–250 mg/kg/day) is usually required in individuals with CTD. It has been shown that blood and urine carnitine levels are increased on l-carnitine supplementation. However, there was no measurable increase in intracellular carnitine concentrations. Plasma carnitine levels on oral l-carnitine supplementation do not reflect the tissue carnitine levels. Especially during physical activity, energy demands of skeletal and cardiac muscle increase. It has been shown that during exercise there is an impaired beta oxidation capacity in individuals with CTD which is partially restored by carnitine supplementation [29, 30]. Unfortunately, high dose carnitine supplementation results in a fishy odor to the breath, sweat and urine. The frequent dosing and fishy body odor are major limiting factors for treatment compliance. There are unfortunately sudden deaths in childhood, adolescents, and adults with CTD. The sudden death is attributed to compliance problems, but it has not been proven to be correct. There are no extensive investigations to understand the mechanisms of sudden death in patients with CTD as well as how to prevent this devastating complication. It is timely to think about other treatments in addition to carnitine supplementation such as medium chain triglyceride supplementation or anaplerotic agent such as triheptanoin.
In nine patients with VLCAD deficiency, peripheral blood cells (including natural killer cells, B lymphocytes, T lymphocytes, dendritic cells, hematopoietic stem cells, and hematopoietic progenitor cells) were used to measure oxygen consumption rate and fatty acid oxidation rate. Patients with VLCAD deficiency showed impaired oxidation capacity and reduced total energy production. The supplementation of long-chain fatty acids resulted in the accumulation of long-chain fatty acid in CD8 + T cells, leading to impairment of mitochondrial functions, and decrease in the fatty acid metabolism in these cells [31]. It was previously reported that lymphocyte, monocyte, macrophage, and neutrophil functions were suppressed by long chain fats [32, 33]. The C8:0 supplementation did not result in an increase in oxygen consumption rate in VLCAD patient cells. The authors raised concerns of the efficacy of MCT supplementation in VLCAD deficiency [31]. Additionally, a VLCAD knock-out mouse model infected by influenza showed abnormalities in tissue specific acylcarnitine profiles compared to wild-type [34]. It seems that our patient cohort with mitochondrial long-chain fatty acid oxidation and some of the carnitine metabolism defects have had increased infection frequency resulting in metabolic decompensation and several hospital admissions. This is likely secondary to the energy deficiency and accumulation of long chain fatty acids affecting the immune system functions.
It is important to discuss the improved outcomes in patients who were diagnosed prenatally by positive family history or neonatally by NBS. Both early diagnostic processes are crucial to prevent morbidity and mortality in patients with mitochondrial fatty acid oxidation defects. There are several reports that NBS prevented disease related complications and improved outcomes in patients [4, 8, 12, 35–40]. A recent review article by Marsden et al. summarized outcomes of expanded NBS worldwide. There were several advantages including: 1) there was an increase in the incidence of fatty acid oxidation defects; 2) more patients were identified even if they were symptomatic prior to NBS results; 3) there were improvements in clinical outcomes; 4) there was a decrease in mortality rate; 4) there were less disease related complications due to early identification and treatment of patients with fatty acid oxidation defects identified by NBS such as cardiomyopathy and hypoglycemia; 5) there were improvements in the neurodevelopmental outcomes [41]. In our study, we compared several outcome measures between SymX group and AsymX group. Rhabdomyolysis and hypoglycemia were significantly lower in the AsymX group. None of the patients had myopathy, cardiac arrhythmia, cardiomyopathy, seizures or peripheral neuropathy in the AsymX group in our study.
To report genotype–phenotype correlations for mitochondrial long-chain fatty acid oxidation disorders, we reviewed the literature. Lim et al., [42] reported that the patients with LCHAD deficiency who had either homozygous or heterozygous p.Glu474Gln variant in HADHA presented with cardiomyopathy, hypoglycemia, retinopathy, rhabdomyolysis, and peripheral neuropathy [42]. The authors did not report their newborn screening status but compared this common genotype with other genotypes. In our study cohort, all patients with LCHAD deficiency had the common variant and their phenotype was similar to the patients reported in the literature for this genotype. It seems that despite identification by NBS or later in life symptomatically, they present with similar phenotypes. The p.Val283Ala in ACADVL was reported in about 10% of newborns identified by NBS by Miller et al. [38]. Interestingly, in our study, only one of the five patients with VLCAD deficiency was heterozygous for this variant who was identified by NBS. Our five patients with VLCAD deficiency were compound heterozygous for eight different variants (75% missense variants). The different genotypes in SymX and AsymX groups for VLCAD deficiency presented with similar phenotypes. The p.Ser113Leu variant in CPT2 is the common variant for CPT-II deficiency. This genotype is associated with the mild adult-onset phenotype, where most patients were homozygous for this variant [43, 44]. We had two patients with CPT-II deficiency who are heterozygous for this common variant and with a similar phenotype to the patients reported in the literature. Previous reports of large number of patients with CTD did not provide clear genotype–phenotype correlation [3, 7, 25]. We had eight patients with CTD who had nine different variants in our study cohort. We were not able to drove conclusions for genotype–phenotype correlations for those patients with CTD in our study. So far less than 30 patients with CACT deficiency were reported in the literature. There were two patients, 10 years and 16 months of age at the time of the publications who were compound heterozygous for the c.326 + 1delG variant in SLC25A20. Their phenotype was similar to our patient’s phenotype [5]. Our sibling pair with CACT deficiency was compound heterozygous for the known p.Asn300Glnfs*24 variant in SLC25A20. Interestingly, a patient with CACT deficiency who was homozygous for the same variant had a neonatal onset hypoglycemia but had a milder phenotype and was 9 years old at the time the publication [45]. It appears that this patient and our patients’ phenotypes are similar.
Our study had several limitations including: 1) It is a retrospective cohort study consisting of a chart review of patients who had been diagnosed with mitochondrial long-chain fatty acid oxidation and carnitine metabolism defects at our institution; 2) There were no single evaluation pathway for monitoring of these patients to assess outcomes in detail and systematically; 3) Some of the patients in the AsymX group had no clinical features during the last assessment. It may be still too early to see any symptoms; 4) Our data includes different treatment regimens, and different times for the initiation of treatment and does not allow us to compare the groups, genotypes and treatment outcomes statistically. Despite these limitations, the number of patients from a single metabolic genetics center and their outcomes are valuable and add additional knowledge regarding mitochondrial long-chain fatty acid oxidation and carnitine metabolism defects.
In conclusion, the prevalence of mitochondrial long-chain fatty acid oxidation and carnitine defects was 4.75% in our clinic. Asymptomatic diagnosis is important to improve quality of life and decrease acute complications.
Supplementary Information
Additional file 1. Table S1: Newborn screening results are summarized in Table S1. Table S2. All variants and their ACMG classification are summarized in Table S2. Table S3: Biochemical features, illness management, and number of lifetime hospital admissions are summarized in Table S3.
Acknowledgements
We would like to thank Genetic Counselors, Physicians, Metabolic Dieticians and Administrative Assistants for their excellent care for the management of our patients in the Metabolic Genetics Clinic. We would like to thank Khadine Wiltshire for the submission and receival of approvals from the Institutional Research Ethics Board for this study. We would like to thank Northern Alberta Clinical Trials + Research Center (NACTRC) for Operational Approval (O/A #57356) to conduct research at Alberta Health Services. We would like to thank Northern Alberta Clinical Trials and Research Centre (NACTRC) for their funding support for Summer Student Research Award to Anastasia Ambrose. This study was partially funded by Ultragenyx Pharmaceutical and we would like to thank the company for this funding support.
Author contributions
AA and MS: Reviewed charts, generated the database, drafted the manuscript, and conducted the work, read and approved the final manuscript. SB: Applied the statistical analysis, read and approved the final manuscript. AA and TA: Applied ACMG criteria for the variant classification, read and approved the final manuscript. S.G-J and A.C: Provided information for patients included into the study, read and approved the final manuscript. S.M-A: Planned, applied and received funding, designed the study, and drafted and revised the manuscript, read and approved the final manuscript. All authors read and approved the final manuscript.
Funding
This study is funded by Northern Alberta Clinical Trials and Research Centre (NACTRC) for Summer Student Research Award to Anastasia Ambrose. This study was partially funded by Ultragenyx Pharmaceutical.
Availability of data and materials
All data generated or analysed during this study are included in this published article and its supplementary information files.
Declarations
Ethics approval and consent to participate
The Research Ethics Office, Health Research Ethics Board, University of Alberta (Study ID: Pro00108842) approved this retrospective cohort study.
All procedures followed were in accordance with the ethical standards of the responsible committee on human experimentation (institutional and national) and with the Helsinki Declaration of 1975, as revised in 2000 (5).
Consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing interests.
Footnotes
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
References
- 1.Baker JJ, Burton BK. Diagnosis and clinical management of long-chain fatty-acid oxidation disorders: a review. touchREV Endocrinol. 2021;17:108–111. doi: 10.17925/EE.2021.17.2.108. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.El-Gharbawy A, Vockley J. Inborn errors of metabolism with myopathy: defects of fatty acid oxidation and the carnitine shuttle system. Pediatr Clin North Am. 2018;65:317–335. doi: 10.1016/j.pcl.2017.11.006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Longo N. Primary carnitine deficiency and newborn screening for disorders of the carnitine cycle. Ann Nutr Metab. 2016;68(Suppl 3):5–9. doi: 10.1159/000448321. [DOI] [PubMed] [Google Scholar]
- 4.Spiekerkoetter U. Mitochondrial fatty acid oxidation disorders: clinical presentation of long-chain fatty acid oxidation defects before and after newborn screening. J Inherit Metab Dis. 2010;33:527–532. doi: 10.1007/s10545-010-9090-x. [DOI] [PubMed] [Google Scholar]
- 5.Ryder B, Inbar-Feigenberg M, Glamuzina E, Halligan R, Vara R, Elliot A, Coman D, et al. New insights into carnitine-acylcarnitine translocase deficiency from 23 cases: management challenges and potential therapeutic approaches. J Inherit Metab Dis. 2021;44:903–915. doi: 10.1002/jimd.12371. [DOI] [PubMed] [Google Scholar]
- 6.Obaid A, Nashabat M, Alfadhel M, Alasmari A, Al Mutairi F, Alswaid A, Faqeih E, et al. Clinical, biochemical, and molecular features in 37 saudi patients with very long chain acyl CoA dehydrogenase deficiency. JIMD Rep. 2018;40:47–53. doi: 10.1007/8904_2017_58. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Lin Y, Lin B, Chen Y, Zheng Z, Fu Q, Lin W, Zhang W. Biochemical and genetic characteristics of patients with primary carnitine deficiency identified through newborn screening. Orphanet J Rare Dis. 2021;16:503. doi: 10.1186/s13023-021-02126-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Landau YE, Waisbren SE, Chan LM, Levy HL. Long-term outcome of expanded newborn screening at Boston children's hospital: benefits and challenges in defining true disease. J Inherit Metab Dis. 2017;40:209–218. doi: 10.1007/s10545-016-0004-4. [DOI] [PubMed] [Google Scholar]
- 9.Gallant NM, Leydiker K, Wilnai Y, Lee C, Lorey F, Feuchtbaum L, Tang H, Carter J, Enns GM, Packman S, Lin HJ, Wilcox WR, Cederbaum SD, Abdenur JE. Biochemical characteristics of newborns with carnitine transporter defect identified by newborn screening in California. Mol Genet Metab. 2017;122:76–84. doi: 10.1016/j.ymgme.2017.06.015. [DOI] [PubMed] [Google Scholar]
- 10.Ding M, Liu R, Qiubo L, Zhang Y, Kong Q. Neonatal-onset multiple acyl-CoA dehydrogenase deficiency (MADD) in the ETFDH gene: a case report and a literature review. Medicine. 2020;99:e21944. doi: 10.1097/MD.0000000000021944. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.den Boer ME, Wanders RJ, Morris AA, IJlst L, Heymans HS, Wijburg FA. Long-chain 3-hydroxyacyl-CoA dehydrogenase deficiency: clinical presentation and follow-up of 50 patients. Pediatrics. 2002;109:99–104. doi: 10.1542/peds.109.1.99. [DOI] [PubMed] [Google Scholar]
- 12.Bleeker JC, Kok IL, Ferdinandusse S, van der Pol WL, Cuppen I, Bosch AM, Langeveld M, et al. Impact of newborn screening for very-long-chain acyl-CoA dehydrogenase deficiency on genetic, enzymatic, and clinical outcomes. J Inherit Metab Dis. 2019;42:414–423. doi: 10.1002/jimd.12037. [DOI] [PubMed] [Google Scholar]
- 13.Immonen T, Ahola E, Toppila J, Lapatto R, Tyni T, Lauronen L. Peripheral neuropathy in patients with long-chain 3-hydroxyacyl-CoA dehydrogenase deficiency: a follow-up EMG study of 12 patients. Eur J Paediatr Neurol. 2016;20:38–44. doi: 10.1016/j.ejpn.2015.10.009. [DOI] [PubMed] [Google Scholar]
- 14.Bellusci M, Quijada-Fraile P, Barrio-Carreras D, Martin-Hernandez E, Garcia-Silva M, Merinero B, Perez B, et al. Carnitine palmitoyltransferase 1A deficiency: abnormal muscle biopsy findings in a child presenting with Reye's syndrome. J Inherit Metab Dis. 2017;40:751–752. doi: 10.1007/s10545-017-0041-7. [DOI] [PubMed] [Google Scholar]
- 15.Wanders RJA, Visser G, Ferdinandusse S, Vaz FM, Houtkooper RH. Mitochondrial fatty acid oxidation disorders: laboratory diagnosis, pathogenesis, and the complicated route to treatment. J Lipid Atheroscler. 2020;9:313–333. doi: 10.12997/jla.2020.9.3.313. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Spiekerkoetter U, Lindner M, Santer R, Grotzke M, Baumgartner MR, Boehles H, Das A, et al. Management and outcome in 75 individuals with long-chain fatty acid oxidation defects: results from a workshop. J Inherit Metab Dis. 2009;32:488–497. doi: 10.1007/s10545-009-1125-9. [DOI] [PubMed] [Google Scholar]
- 17.Spiekerkoetter U, Bastin J, Gillingham M, Morris A, Wijburg F, Wilcken B. Current issues regarding treatment of mitochondrial fatty acid oxidation disorders. J Inherit Metab Dis. 2010;33:555–561. doi: 10.1007/s10545-010-9188-1. [DOI] [PubMed] [Google Scholar]
- 18.Vockley J. Long-chain fatty acid oxidation disorders and current management strategies. Am J Manag Care. 2020;26(Suppl 7):147–154. doi: 10.37765/ajmc.2020.88480. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Vockley J, Enns GM, Ramirez AN, Bedrosian CL, Reineking B, Lu X, Ray K, et al. Response to triheptanoin therapy in critically ill patients with LC-FAOD: report of patients treated through an expanded access program. Mol Genet Metab. 2022;136:152–162. doi: 10.1016/j.ymgme.2022.04.001. [DOI] [PubMed] [Google Scholar]
- 20.Vockley J, Burton B, Berry G, Longo N, Phillips J, Sanchez-Valle A, Chapman K, et al. Effects of triheptanoin (UX007) in patients with long-chain fatty acid oxidation disorders: results from an open-label, long-term extension study. J Inherit Metab Dis. 2021;44:253–263. doi: 10.1002/jimd.12313. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Sklirou E, Alodaib AN, Dobrowolski SF, Mohsen AA, Vockley J. Physiological perspectives on the use of triheptanoin as anaplerotic therapy for long chain fatty acid oxidation disorders. Front Genet. 2021;11:598760. doi: 10.3389/fgene.2020.598760. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Richards S, Aziz N, Bale S, Bick D, Das S, Gastier-Foster J, Grody WW, et al. Standards and guidelines for the interpretation of sequence variants: a joint consensus recommendation of the American College of Medical Genetics and Genomics and the Association for Molecular Pathology. Genet Med. 2015;17:405–424. doi: 10.1038/gim.2015.30. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Lek M, Karczewski KJ, Minikel EV, Samocha KE, Banks E, Fennell T, O'Donnell-Luria AH, et al. Analysis of protein-coding genetic variation in 60,706 humans. Nature. 2016;536:285–291. doi: 10.1038/nature19057. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Lee NC, Tang NL, Chien YH, Chen CA, Lin SJ, Chiu PC, Huang AC, et al. Diagnoses of newborns and mothers with carnitine uptake defects through newborn screening. Mol Genet Metab. 2010;100:46–50. doi: 10.1016/j.ymgme.2009.12.015. [DOI] [PubMed] [Google Scholar]
- 25.Li FY, El-Hattab AW, Bawle EV, Boles RG, Schmitt ES, Scaglia F, Wong LJ. Molecular spectrum of SLC22A5 (OCTN2) gene mutations detected in 143 subjects evaluated for systemic carnitine deficiency. Hum Mutat. 2010;31:E1632–E1651. doi: 10.1002/humu.21311. [DOI] [PubMed] [Google Scholar]
- 26.Collins SA, Sinclair G, McIntosh S, Bamforth F, Thompson R, Sobol I, Osborne G, et al. Carnitine palmitoyltransferase 1A (CPT1A) P479L prevalence in live newborns in Yukon, Northwest Territories, and Nunavut. Mol Genet Metab. 2010;101:200–204. doi: 10.1016/j.ymgme.2010.07.013. [DOI] [PubMed] [Google Scholar]
- 27.Collins SA, Hildes-Ripstein GE, Thompson JR, Edmunds S, Miners A, Rockman-Greenberg C, Arbour L. Neonatal hypoglycemia and the CPT1A P479L variant in term newborns: a retrospective cohort study of Inuit newborns from Kivalliq Nunavut. Paediatr Child Health. 2020;26:218–227. doi: 10.1093/pch/pxaa039. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Preston G, Emmerzaal T, Kirdar F, Schrader L, Henckens M, Morava E, Kozicz T. Cerebellar mitochondrial dysfunction and concomitant multi-system fatty acid oxidation defects are sufficient to discriminate PTSD-like and resilient male mice. Brain Behav Immun Health. 2020;6:100104. doi: 10.1016/j.bbih.2020.100104. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Flanagan JL, Simmons PA, Vehige J, Willcox MD, Garrett Q. Role of carnitine in disease. Nutr Metab. 2010;7:30. doi: 10.1186/1743-7075-7-30. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Madsen KL, Preisler N, Rasmussen J, Hedermann G, Olesen JH, Lund AM, Vissing J. L-carnitine improves skeletal muscle fat oxidation in primary carnitine deficiency. J Clin Endocrinol Metab. 2018;103:4580–4588. doi: 10.1210/jc.2018-00953. [DOI] [PubMed] [Google Scholar]
- 31.Stenlid R, Olsson D, Cen J, Manell H, Haglind C, Chowdhury AI, Bergsten P, et al. Altered mitochondrial metabolism in peripheral blood cells from patients with inborn errors of β-oxidation. Clin Transl Sci. 2022;15:182–194. doi: 10.1111/cts.13133. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Buenestado A, Cortijo J, Sanz MJ, Naim-Abu-Nabah Y, Martinez-Losa M, Mata M, Issekutz AC, Martí-Bonmatí E, et al. Olive oil-based lipid emulsion's neutral effects on neutrophil functions and leukocyte-endothelial cell interactions. J Parenter Enteral Nutr. 2006;30:286–296. doi: 10.1177/0148607106030004286. [DOI] [PubMed] [Google Scholar]
- 33.Yaqoob P. Lipids and the immune response. Curr Opin Clin Nutr Metab Care. 1998;1:153–161. doi: 10.1097/00075197-199803000-00003. [DOI] [PubMed] [Google Scholar]
- 34.Tarasenko TN, Cusmano-Ozog K, McGuire PJ. Tissue acylcarnitine status in a mouse model of mitochondrial β-oxidation deficiency during metabolic decompensation due to influenza virus infection. Mol Genet Metab. 2018;125:144–152. doi: 10.1016/j.ymgme.2018.06.012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Maguolo A, Rodella G, Dianin A, Nurti R, Monge I, Rigotti E, Cantalupo G, et al. Diagnosis, genetic characterization and clinical follow up of mitochondrial fatty acid oxidation disorders in the new era of expanded newborn screening: a single centre experience. Mol Genet Metab Rep. 2020;24:100632. doi: 10.1016/j.ymgmr.2020.100632. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Rücklová K, Hrubá E, Pavlíková M, Hanák P, Farolfi M, Chrastina P, Vlášková H, et al. Impact of newborn screening and early dietary management on clinical outcome of patients with long chain 3-hydroxyacyl-coa dehydrogenase deficiency and medium chain Acyl-CoA dehydrogenase deficiency-a retrospective nationwide study. Nutrients. 2021;13:2925. doi: 10.3390/nu13092925. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Pena LD, van Calcar SC, Hansen J, Edick MJ, Walsh Vockley C, Leslie N, Cameron C, et al. Outcomes and genotype-phenotype correlations in 52 individuals with VLCAD deficiency diagnosed by NBS and enrolled in the IBEM-IS database. Mol Genet Metab. 2016;118:272–281. doi: 10.1016/j.ymgme.2016.05.007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Miller MJ, Burrage LC, Gibson JB, Strenk ME, Lose EJ, Bick DP, Elsea SH, et al. Recurrent ACADVL molecular findings in individuals with a positive newborn screen for very long chain Acyl-CoA dehydrogenase (VLCAD) deficiency in the United States. Mol Genet Metab. 2015;116:139–145. doi: 10.1016/j.ymgme.2015.08.011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Evans M, Andresen BS, Nation J, Boneh A. VLCAD deficiency: follow-up and outcome of patients diagnosed through newborn screening in Victoria. Mol Genet Metab. 2016;118:282–287. doi: 10.1016/j.ymgme.2016.05.012. [DOI] [PubMed] [Google Scholar]
- 40.Merinero B, Alcaide P, Martín-Hernández E, Morais A, García-Silva MT, Quijada-Fraile P, Pedrón-Giner C, et al. Four years’ experience in the diagnosis of very long–chain acyl-CoA dehydrogenase deficiency in infants detected in three Spanish newborn screening centers. JIMD Rep. 2018;39:63–74. doi: 10.1007/8904_2017_40. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Marsden D, Bedrosian CL, Vockley J. Impact of newborn screening on the reported incidence and clinical outcomes associated with medium- and long-chain fatty acid oxidation disorders. Genet Med. 2021;23:816–829. doi: 10.1038/s41436-020-01070-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Lim CC, Vockley J, Ujah O, Kirby RS, Edick MJ, Berry SA, Arnold GL. Outcomes and genotype correlations in patients with mitochondrial trifunctional protein or isolated long chain 3-hydroxyacyl-CoA dehydrogenase deficiency enrolled in the IBEM-IS database. Mol Genet Metab Rep. 2022;32:100884. doi: 10.1016/j.ymgmr.2022.100884. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Handig I, Dams E, Taroni F, Van Laere S, de Barsy T, Willems PJ. Inheritance of the S113L mutation within an inbred family with carnitine palmitoyltransferase enzyme deficiency. Hum Genet. 1996;97:291–293. doi: 10.1007/BF02185756. [DOI] [PubMed] [Google Scholar]
- 44.Di Mauro S, Papadimitriou A. Camitine palmitoyltransferase deficiency. In: Engel AG, Banker BQ, editors. Myology. 1. New York: McGraw-Hill; 1986. pp. 1697–1708. [Google Scholar]
- 45.Huizing M, Iacobazzi V, Ijlst L, Savelkoul P, Ruitenbeek W, van den Heuvel L, Indiveri C, Smeitink J, Trijbels F, Wanders R, Palmieri F. Cloning of the human carnitine-acylcarnitine carrier cDNA and identification of the molecular defect in a patient. Am J Hum Genet. 1997;61(6):1239–1245. doi: 10.1086/301628. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Additional file 1. Table S1: Newborn screening results are summarized in Table S1. Table S2. All variants and their ACMG classification are summarized in Table S2. Table S3: Biochemical features, illness management, and number of lifetime hospital admissions are summarized in Table S3.
Data Availability Statement
All data generated or analysed during this study are included in this published article and its supplementary information files.