Abstract
Clonal hematopoiesis of indeterminate potential (CHIP) is associated with a small risk of developing hematologic malignancies and a higher risk of cardiovascular diseases (CVD). We asked whether the reverse correlation exists and cardiometabolic risk factors have an impact on the progression of myelodysplastic syndrome (MDS) to acute myeloid leukemia (AML). We investigated the association between abnormal lipid profiles and inflammation in MDS, which shares many genetic mutations with CHIP, and the risk of developing acute leukemia. We examined the medical records of 11071 MDS patients. Among them, 5422 had at least one lipid profile or C-reactive protein (CRP) measurement. In univariate and multivariate analyses, elevated triglyceride and high-sensitive C-reactive protein (HS-CRP) were significantly associated with a diagnosis of acute leukemia in MDS patients. Next, we examined these associations in patients with available MDS prognostic scores (International Prognostic Scoring System, IPSS, or its revised version IPSS/R) (n = 2786 patients). We found that the statistical association between CRP and the progression of MDS to leukemia was independent of other variables in the scoring system. MDS patients with elevated CRP in both the high-risk and low-risk groups had a higher risk of progression to AML than those with a lower CRP. We speculate that inflammation might be a common denominator in developing hematologic malignancies and CVD in patients with clonal hematopoiesis.
Supplementary Information
The online version contains supplementary material available at 10.1186/s40164-022-00309-7.
Keywords: Myelodysplastic syndrome, Acute Myeloid Leukemia, Clonal hematopoiesis, Inflammation, Triglyceride
Keypoints
Elevated triglyceride and high-sensitive CRP are associated with a higher risk of acute leukemia in MDS.
Supplementary Information
The online version contains supplementary material available at 10.1186/s40164-022-00309-7.
To the editor:
MDS is a premalignant blood disorder, and about 30% of MDS patients develop AML. The presence of similar genetic mutations in abnormal hematopoietic clones in CHIP and MDS raises the possibility of a CHIP to MDS to AML continuum [1–6]. CHIP is also associated with a significant risk of CVD [7]. Preventive measures such as the treatment of hyperlipidemia, blood pressure, and diabetes are recommended in managing CHIP patients [8]. Whether cardiovascular risk factors impact the progression of CHIP to malignant blood disorders is unknown. MDS is also a clonal blood disorder but with a much higher conversion rate to AML than CHIP. We investigated the association between abnormal lipid profile and elevated inflammation biomarkers and the frequency of diagnosis of AML in a large MDS cohort.
Among 11071 MDS patients diagnosed and treated between 2003 and 2020 at MD Anderson Cancer Center, 5422 had at least one measurement of their lipid profile, CRP, or HS-CRP. Their average age was 56 (+ 12), and 59.4% were male. More information about the population and laboratory results are available in Additional file 1: Methods and Table S1. About 62.2% (n = 3375) of MDS patients were diagnosed with acute leukemia through follow-up. We compared laboratory values between MDS patients who developed AML and those who remained leukemia-free (n = 2047, 37.8%). Among the continuous variables, triglyceride and VLDL were higher, and HDL and LDL were lower in MDS patients who developed acute leukemia than in those who did not (Additional file 1: Table S2). In the univariate analysis, all the continuous lab variables, as well as the statin prescription, were significantly associated with the leukemia status (Table 1). In the multivariate analysis, only the triglyceride (OR 1.44; 95% CI 1.16–1.78) and HS-CRP (OR 1.13; 95% CI 1.06–1.21) remained significantly associated with a diagnosis of acute leukemia (Table 1). In the univariate analysis, treatment with statins was associated with an increased frequency of diagnosis of AML (OR 1.13, 95% CI 1.00–1.28, p = 0.044) (Table 1). However, one cannot study the effect of statins without considering the possibility of confounding effects of high LDL. In the multivariable analysis, when forced into the model, statins were associated with a modestly reduced risk of AML, with a borderline statistical significance (OR 0.8, CI 0.63–1.01, p = 0.061) (Table 1).
Table 1.
Risk of leukemia for all patients
| Univariate logistic regression | Multivariable logistic regression (based on backward model selection) |
Multivariable Model (based on a backward model selection with statin forced in the Model) | ||||
|---|---|---|---|---|---|---|
| OR (95% CI) | p-value | OR (95% CI) | p-value | OR (95% CI) | p-value | |
| HS-CRP* | 1.18 (1.11, 1.24) | < 0.001 | 1.13 (1.06,1.21) | < 0.001 | 1.14 (1.06,1.21) | < 0.001 |
| TRIG* | 1.38 (1.22, 1.56) | < 0.001 | 1.44 (1.16,1.78) | 0.001 | 1.44 (1.16,1.78) | 0.001 |
| CRP* | 1.28 (1.20, 1.37) | < 0.001 | ||||
| HDL | 0.99 (0.98, 0.99) | < 0.001 | ||||
| LDL | 1.00 (0.99, 1.00) | 0.001 | ||||
| VLDL | 1.01 (1.00, 1.01) | < 0.001 | ||||
| Statin, yes/no | 1.13 (1.00, 1.28) | 0.044 | 0.80 (0.63,1.01) | 0.061 | ||
*Log-transformed, OR > 1 means an increased risk
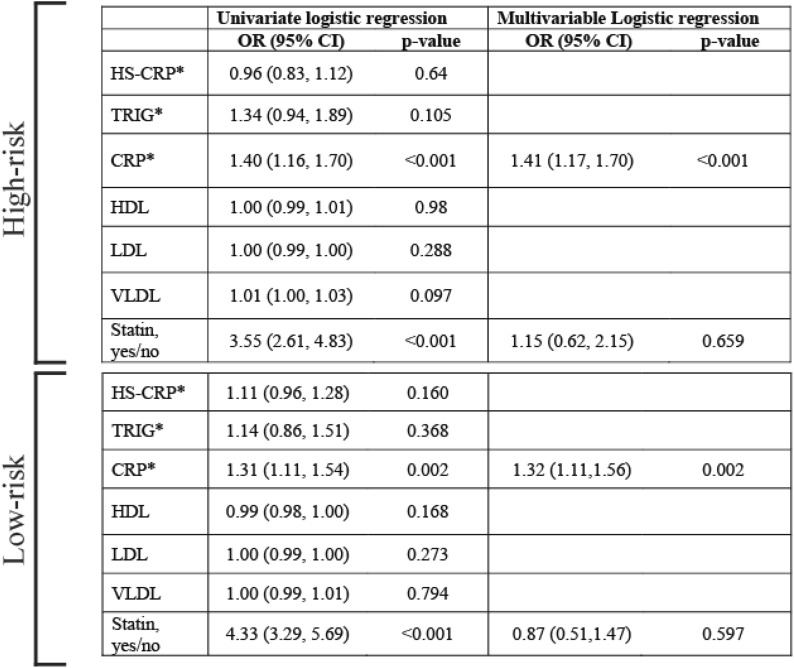
Using IPSS or IPSS/R classifications (whichever was available initially), MDS patients were categorized into two groups: patients with a high risk of conversion to leukemia (high and intermediate II according to IPSS and high and very high according to IPSS/R) and those with a low risk (Intermediate I and low according to IPSS and low and very low according to IPSS/R). In our cohort, 2786 patients had either IPSS or IPSS/R and lipid profile and CRP or HS-CRP. Among them, 1003 (36%) were in the high-risk group and 1783 (64%) in the low-risk group. The distribution of lipid profile and CRP and HS-CRP among these groups are shown in Additional file 1: Table S3. In the low-risk group, elevated CRP was more common in patients who developed leukemia than in those who did not (Additional file 1: Table S4). In the high-risk group, elevated CRP and triglyceride were more common in patients who developed leukemia (Additional file 1: Table S5). Elevated CRP was associated with an increased risk of conversion to AML in both high- and low-risk groups in univariate and multivariable logistic regression analyses (an estimated odd ratio of 1.3) (Table 2). Our results point to a predictive value for inflammatory biomarkers in MDS. In another study on a small number of low-risk MDS patients, CRP was found to be associated with a worse prognosis [9]. In univariate analysis, the use of statins was associated with an increase in the risk of AML but lost its significance in the multivariable analysis (Table 2). More information about the statistical methods is provided in the Additional file 1: Material.
Table 2.
Impact on the risk of leukemia in the high- and low-risk groups
Inflammation is important in the pathogenesis and progression of MDS [10, 11]. The pro-inflammatory effect of elevated triglycerides in atherosclerosis has been recently recognized [12] and might also be relevant to its impact on the bone marrow microenvironment. A limitation of our study is that we did not examine confounding factors such as age, gender, and chemotherapy protocols. Our results and speculations should be examined in other MDS cohorts.
Supplementary Information
Additional file 1: Table S1. Characteristics of the laboratory tests of the patients. Table S2. Lab test characteristics stratified by the leukemia status. Table S3. Distribution of Lab values in risk subgroups. Table S4. Lab test characteristics stratified by the leukemia status for the low-risk group (n=1783). Table S5. Lab test characteristics stratified by the leukemia status for the high-risk group (n=1003)
Author contributions
WQ analyzed data and interpreted the results. EY collected data and analyzed data. CF analyzed data. SL analyzed data. JJ analyzed data. LN Collected data. CMRH analyzed data, interpreted the results, and participated in writing the manuscript. GB interpreted the results. OG analyzed data, interpreted the results, and participated in writing the manuscript. VA-K designed the study, interpreted the results, and wrote the manuscript. All authors read and approved the final manuscript.
Funding
This work was supported by the National Institutes of Health/National Cancer Institute grants CA231141 to V. A-K and OG and CA177909 to V. A-K.
Availability of data and materials
Data generated or analyzed during this study are included in this published article [and its supplementary information files]. Additional raw data are available from the corresponding author on reasonable request and upon approval of the Institutional Review Boards.
Declarations
Ethics approval and consent to participate
All of the studies conducted in this manuscript were approved by the Institutional Review Board of the University of Texas M. D. Anderson Cancer Center (IRB protocol # 2021-0591. Lipid profile and inflammation biomarkers in myelodysplastic syndrome).
Consent for publication
Not Applicable. Our manuscript does not contain data from any individual person.
Competing interests
The authors disclose no potential conflicts of interest.
Footnotes
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
References
- 1.Genovese G, Kähler AK, Handsaker RE, et al. Clonal hematopoiesis and blood-cancer risk inferred from blood DNA sequence. N Engl J Med. 2014;371(26):2477–2487. doi: 10.1056/NEJMoa1409405. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Walter MJ, Shen D, Ding L, et al. Clonal architecture of secondary acute myeloid leukemia. N Engl J Med. 2012;366(12):1090–1098. doi: 10.1056/NEJMoa1106968. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Welch JS, Ley TJ, Link DC, et al. The origin and evolution of mutations in acute myeloid leukemia. Cell. 2012;150(2):264–278. doi: 10.1016/j.cell.2012.06.023. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Xie M, Lu C, Wang J, et al. Age-related mutations associated with clonal hematopoietic expansion and malignancies. Nat Med. 2014;20(12):1472–1478. doi: 10.1038/nm.3733. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Young AL, Challen GA, Birmann BM, Druley TE. Clonal haematopoiesis harbouring AML-associated mutations is ubiquitous in healthy adults. Nat Commun. 2016;7:12484. doi: 10.1038/ncomms12484. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Young AL, Tong RS, Birmann BM, Druley TE. Clonal hematopoiesis and risk of acute myeloid leukemia. Haematologica. 2019;104(12):2410–2417. doi: 10.3324/haematol.2018.215269. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Jaiswal S, Fontanillas P, Flannick J, et al. Age-related clonal hematopoiesis associated with adverse outcomes. N Engl J Med. 2014;371(26):2488–2498. doi: 10.1056/NEJMoa1408617. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Jaiswal S, Natarajan P, Silver AJ, et al. Clonal hematopoiesis and risk of atherosclerotic cardiovascular disease. N Engl J Med. 2017;377(2):111–121. doi: 10.1056/NEJMoa1701719. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Baba Y, Saito B, Shimada S, et al. Increased serum C-reactive protein is an adverse prognostic factor in low-risk myelodysplastic syndromes. Int J Hematol. 2021;114(4):441–448. doi: 10.1007/s12185-021-03187-7. [DOI] [PubMed] [Google Scholar]
- 10.Sallman DA, List A. The central role of inflammatory signaling in the pathogenesis of myelodysplastic syndromes. Blood. 2019;133(10):1039–1048. doi: 10.1182/blood-2018-10-844654. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Gañán-Gómez I, Wei Y, Starczynowski DT, et al. Deregulation of innate immune and inflammatory signaling in myelodysplastic syndromes. Leukemia. 2015;29(7):1458–1469. doi: 10.1038/leu.2015.69. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Libby P. Fat fuels the flame: triglyceride-rich lipoproteins and arterial inflammation. Circ Res. 2007;100(3):299–301. doi: 10.1161/01.RES.0000259393.89870.58. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Additional file 1: Table S1. Characteristics of the laboratory tests of the patients. Table S2. Lab test characteristics stratified by the leukemia status. Table S3. Distribution of Lab values in risk subgroups. Table S4. Lab test characteristics stratified by the leukemia status for the low-risk group (n=1783). Table S5. Lab test characteristics stratified by the leukemia status for the high-risk group (n=1003)
Data Availability Statement
Data generated or analyzed during this study are included in this published article [and its supplementary information files]. Additional raw data are available from the corresponding author on reasonable request and upon approval of the Institutional Review Boards.