Abstract
Nucleic acid therapeutics have shown great potential for the treatment of numerous diseases, such as genetic disorders, cancer and infections. Moreover, they have been successfully used as vaccines during the COVID-19 pandemic. In order to unfold full therapeutical potential, these nano agents have to overcome several barriers. Therefore, directed transport to specific tissues and cell types remains a central challenge to receive carrier systems with enhanced efficiency and desired biodistribution profiles. Active targeting strategies include receptor-targeting, mediating cellular uptake based on ligand-receptor interactions, and chemical targeting, enabling cell-specific delivery as a consequence of chemically and structurally modified carriers. With a focus on synthetic delivery systems including polyplexes, lipid-based systems such as lipoplexes and lipid nanoparticles, and direct conjugates optimized for various types of nucleic acids (DNA, mRNA, siRNA, miRNA, oligonucleotides), we highlight recent achievements, exemplified by several nucleic acid drugs on the market, and discuss challenges for targeted delivery to different organs such as brain, eye, liver, lung, spleen and muscle in vivo.
Keywords: lipoplex, pDNA, polyplex, siRNA, targeting
Introduction
Over the last three decades, vectors for the delivery of therapeutic genetic material were extensively evaluated and optimized. Currently, more than 3000 clinical trials on gene therapy have been conducted or are still ongoing. With about 2/3 of clinical trials on cancer diseases, gene delivery to tumors represents the primary target for nucleic acid therapy. However, other indications, such as monogenetic diseases, infections, inflammatory diseases, neurological and ocular disorders are also interesting targets for gene therapy [1–3]. For gene transfer, viral vectors are still the most advanced delivery systems in clinical gene therapy studies, attributed to their excellent transduction efficacies [4, 5]. Part of their efficacy is that they are naturally built to introduce nucleic acids into foreign cells, presenting natural receptor targeting agents and peptide sequences on their surface that enable cellular entry.
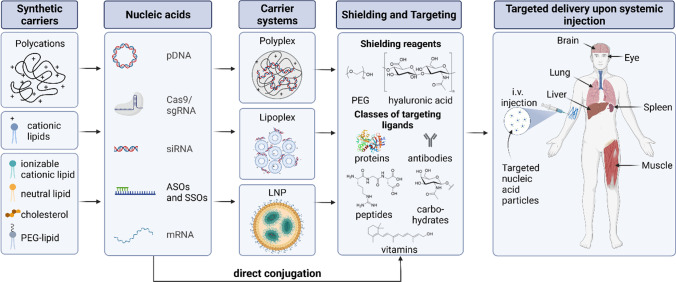
Synthetic delivery systems present a second class of transfer agents for nucleic acid therapeutics. They have the advantage that they can be designed to be non-immunogenic and have the potential to deliver a broad range of natural or synthetic and modified nucleic acids. Historically, transfections introduced functional genes by either using DNA transfer into the nucleus or RNA transfer into the cytosol [6–8]. In contrast to DNA, messenger RNA (mRNA) does not require nuclear entry since its site of action is located in the cytosol. Its great therapeutic potential [9, 10] was recently proven by the successful application of mRNA-based COVID-19 vaccines [11–13]. With the new millennium, RNA interference (RNAi) therapeutics were developed as another class of therapeutic nucleic acids, aiming for the down regulation of malignant gene expression by short interfering RNA (siRNA) [14] or micro-RNA (miRNA) [15]. In addition, now for more than 30 years, antisense oligonucleotides have been therapeutically applied by blocking or modulating splicing of complementary mRNA [16, 17]. Most recently, the CRISPR/Cas technology has entered therapeutic application as a promising tool for genome editing [18, 19], either via Cas9 mRNA/single guide (sg) RNA or as Cas9 protein/sgRNA ribonucleoprotein complex [20]. These synthetic carriers range from organic, polycationic carriers to lipid structures and inorganic particles and were extensively optimized in order to enhance transfection efficacy and become more and more like artificial viruses [21, 22]. First of all, these synthetic carriers are designed to compact the genetic material for protection against degradation and shielding against undesired interactions. This can be achieved by polycations such as poly-L-lysine (PLL) [23, 24], polyethylenimine (PEI) [25, 26], poly(amidoamines) (PAMAM) or dendrimers [27, 28] which interact with negatively charged nucleic acids and assemble in nano-sized particles, so-called “polyplexes”[29]. Lipid-based structures including “lipoplexes”, in which the genetic material is packaged by positively charged lipidic carriers, and “LNPs”, containing the nucleic acid inside multi-component lipid nanoparticles, have also proven great potential as nucleic acid delivery systems (Fig. 1) [29, 30].
Fig. 1.
Nonviral carriers for the delivery of different nucleic acids, including their main components, particle types as well as shielding and targeting agents for organ- or cell-specific delivery upon systemic injection. Created with BioRender.com
In order to show comparable transfection efficacies to viral vectors, especially for in vivo studies, synthetic delivery systems need to fulfill further demands. The particles should a) show no interaction with blood components or aggregation tendency in physiological environment, b) show prolonged blood circulation time to reach the target tissue, c) promote efficient cellular uptake and d) release the genetic material into the cytoplasm, so it can reach its site of action.
Within this delivery process, one major challenge remains the transport and transfer of nucleic acids to the desired cell type or tissue. This requirement can be approached by modifying synthetic carriers with functional domains giving targeted delivery systems, which was first successfully implemented 35 years ago by Wu et al. using GalNAc-presenting ligands on PLL-polyplexes for directed delivery to hepatocytes [24, 31].
In this review, we will give an overview about different active targeting strategies for synthetic delivery systems. We highlight recent advances in nucleic acid delivery to specific healthy tissues including the liver, lung, brain, immune cells, retina and muscle. Specific delivery to cell types can be mediated via defined ligand-receptor interactions (receptor-targeting) as well as modulating the physicochemical properties of the nucleic acid nanoparticles based on small structural variations of the synthetic carriers (chemical targeting). For tumor-specific targeting of nucleic acids we refer to other published work [32–34].
Strategies for specific delivery
Shielding
Synthetic carriers have proven to be potent transfer vehicles for nucleic acid delivery for in vitro studies thanks to various optimizations. However, during the delivery process from injection to gene expression, several obstacles and cellular bottlenecks must be overcome to unfold the full therapeutic potential of the nucleic acid. Besides efficient encapsulation of the genetic material to protect against degradation, it must be ensured that the carriers circulate in the blood until reaching the target tissue.
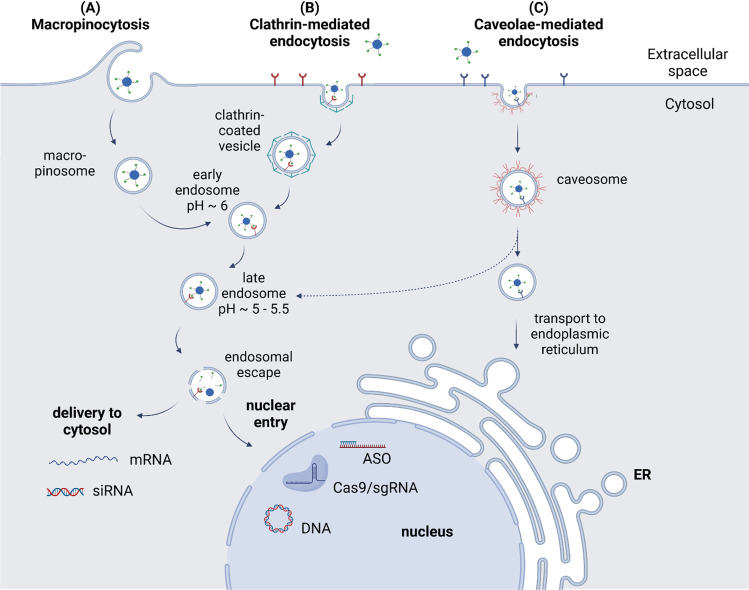
Cellular uptake of positively charged polyplexes is enabled by non-specific endocytosis, in particular macropinocytosis from the extracellular fluid [35] (see Fig. 2A). The uptake can be further improved, even though not specified, by incorporation of cell-penetrating peptides in the formulation, such as octaarginine motifs [36, 37]. In vivo applied nanocarriers, however, are confronted with blood components like plasma proteins that adsorb on particle surface and hence sustainably affect circulation, transport to tissues and cellular uptake [38–40]. For instance, the formation of a protein corona comprised of opsonins will mediate phagocytosis removing particles from the circulation.
Fig. 2.
Internalization pathways of nucleic acid carriers by receptor-mediated endocytosis. Created with BioRender.com
To this end, it is necessary to shield positively charged carriers against unspecific interactions with serum proteins that may result in phagocytosis of the particles. This can be achieved by incorporating shielding domains, for example polymers such as polyethylene glycol (PEG) [41], poly(N-(2- hydroxypropyl)methacrylamide) (pHPMA) [42], poly(2-oxazolines) [43] and polysarcosines [44] or polysaccharides such as hydroxyethyl starch [45] or hyaluronic acid (HA) [46–48] (Fig. 1). Shielding the carrier’s surface results in lowered surface charge and thus reduced interaction with serum proteins, which allows the particles to circumvent the reticuloendothelial system (RES) and increase circulation time in the blood [41]. However, poor targeting abilities resulting in off-target effects or low accumulation on the target site set limitations regarding the application of synthetic gene delivery systems in vivo. Consequently, to ensure tissue- or cell-selective delivery and to minimize accumulation in off-target sites, the particles can be surface-modified either by specific ligands that will interact with receptors on the targeted cell type for active targeting or by modification of their chemical composition leading to altered biodistribution for chemical targeting.
Active Targeting
Receptor Targeting
Decorating the carrier’s surface with ligands can yield specific cellular uptake based on receptor-mediated endocytosis. Here, the fact that specific tissues differently express certain receptors is used to enhance cell-specific uptake of nucleic acid carriers [49]. The types of ligands used for modification of delivery systems range from small chemical drug-derived compounds [50–53], peptides [54, 55] to large proteins [56, 57], antibodies [58, 59], carbohydrates [60–62] and vitamins [63, 64] (Fig. 1). Interaction with their specific receptors will lead to receptor-mediated endocytosis, a highly selective process of nanoparticle internalization.
In order to achieve receptor-mediated uptake, unspecific interactions of the carrier with blood-components have to be reduced by shielding. Cellular uptake is initiated by recognition of specific ligands by cell surface receptors. After cell binding, the receptor-ligand complex is internalized by the formation of vesicles from the cell membrane and delivered to the cytosol (Fig. 2B, C). The fate of nanocarrier transport through the cytosol is determined by the receptor type and the associated pathway of endocytosis, as summarized in Table I. For example, prominent receptors used for targeted gene delivery such as transferrin receptor (TfR) [65], asialoglycoprotein receptor (ASGPR) [66] or low-density lipoprotein receptor (LDLR) [67] undergo clathrin-mediated endocytosis into early endosomes, followed by endosome maturation and fusion with lysosomes [68]. Only small particles with a maximum size of 200 nm can be taken up by this route [69]. Other receptors, e.g., the folate receptor [70] or interleukin-2 receptor [71], are internalized via caveolae-mediated endocytosis [72]. This route tolerates uptake of particles up to 500 nm [69] and allows to bypass the fusion with lysosomes, leading to delivery to endoplasmic reticulum and facilitated nuclear transport [73]. Generally, the release of carriers from late endosomes is critical for efficient nucleic acid delivery to avoid either enzymatic degradation of the nucleic acid payload after fusion with lysosomes or exocytosis during receptor recycling. Conveniently, synthetic carriers have been designed to exhibit endosomolytic properties. For example, cationic carriers containing protonatable amines, e.g., PEI or histidine-containing constructs response to acidic pH in late endosomes promoting a proton-sponge effect, i.e. swelling and eventually disruption of the endosomes, leading to release of the nucleic acid to the cytosol [74, 75]. Lipid-based carriers are able to release their cargo by fusion with the negatively charged endosome membrane [76].
Table I.
Characteristics of Cellular Uptake via Clathrin- and Caveolae-Mediated Endocytosis
| Clathrin-mediated endocytosis | Caveolae-mediated endocytosis | |
|---|---|---|
| Vesicles for transport | Clathrin-coated vesicle | Caveosome |
| Examples for receptors | TfR, ASGPR, LDLR |
Folate receptor, VEGFR, Interleukin-2 |
| Tolerated nanoparticle size | Up to 200 nm | 200 to 500 nm |
| Intracellular fate of cargo |
Formation of endosomes, maturation to late endosomes, lysosomal degradation or endosomal escape |
Transport to endoplasmic reticulum, Golgi apparatus, facilitated delivery to nucleus |
Strategies to Incorporate Targeting Ligands into the Delivery System
Ligands for receptor-targeting as well as shielding domains can be integrated into the delivery system by both, pre-functionalization, and post-modification [77]. Multivalent ligand presentation on the carrier’s surface may promote receptor recognition and increases binding affinity [78, 79]. The density of ligands required for efficient targeting strongly depends on both the chosen carrier system as well as the type and avidity of ligand. For polyplexes, the ligand to polycation ratio may range from 2.5% to > 100% functionalization, depending on the type of ligand [80–82]. In LNPs, ligand-functionalized lipids may account for only 1–2 mol% per formulation, but still promote target-specific delivery [83, 84]. In direct conjugates such as trivalent GalNAc-siRNA, every nucleic acid is equipped with a targeting moiety.
Accessibility of the targeting ligand is also important for receptor binding [85].
Pre-functionalization has been evaluated for polymeric delivery systems which contain different domains for nucleic acid binding, shielding, and targeting [62, 86, 87]. An alternative functionalization strategy is based on the post-modification of pre-formed nanoparticles, mostly via covalent attachment of ligands to functional groups displayed on the surface. For example, copper-free [88, 89] or copper(I)-catalyzed [90] alkyne-azide click reactions were used for particle modification, as they are fast, selective and high-yielding. By means of this method, it can be ensured that the ligands are located on the surface of the particles. At the same time, the removal of excess ligands that may not have bound to the carrier may be required, as they could compete for the receptor and reduce cellular uptake.
Non-covalent binding of ligands to the particle surface can also be considered. This modification method was successfully realized for targeting of synthetic carriers with insulin [91] and hyaluronic acid [48].
Dual Targeting
Inspired by natural viruses, which have optimized cell association and cellular entry mechanisms by presenting several ligands on their surface, dual targeting represents an approach to further increase transfection efficacy. Here, cell entry properties of viral vectors are mimicked by using two (or more) ligands on a single carrier. For example, Nie et al. used dual-targeted PEGylated PEI-pDNA polyplexes, modified with the cell binding peptide B6 and the integrin targeting peptide RGD for increased transfection efficiency on DU145 and PC3 cells, showing increased transfection efficacy when both ligands were incorporated in the polyplex formulation. In addition, it was demonstrated how these ligands participate in both, cell association and internalization [92]. Additional studies of dual-targeted polyplexes with combinations of B6, GE11 (for EGFR targeting) and cyclic cRGDfk (for integrin targeting), respectively, revealed that the combination of B6 and GE11 was most promising for pDNA transfections to DU145 cells, which express all three receptors [93]. Dual-targeted LPEI-PEG polyplexes were also used for delivering the theranostic sodium iodide symporter gene to Huh7 cells, using a combination of GE11 and cMBP, which showed strong benefits compared to single-targeted polyplexes [94].
Cascade Targeting
Under certain circumstances, nucleic acid carriers have to overcome several barriers to reach their site of action, e.g., the blood–brain-barrier (BBB), followed by membranes of targeted cells. In order to generate cascade targeting delivery systems, nanoparticles can be designed to cross the BBB first and display targeting ligands selectively binding to receptors on specific cells behind the barrier. For example, Wang et al. developed such a gene delivery system by decorating the carrier with the I6P7 ligand, a heptapeptide derived from interleukin-6, which is able to promote both, BBB crossing and cell-specific delivery to interleukin-6 receptor presenting cells [95]. A cascade targeting concept was also used by Zhang et al. to deliver siRNA into neuronal cells after crossing the BBB for treatment of Alzheimer’s disease. In this study, BBB crossing was achieved via the ApoA-I ligand, which binds to the scavenger receptor B1, and selective uptake of the particles by neuronal PC12 cells could be demonstrated by incorporation of a peptide ligand, NL4 binding to tropomyosin receptor kinase A (TrkA). These findings were subsequently confirmed by in vivo studies resulting in downregulation of BACE1, an enzyme which is involved in pathogenesis of Alzheimer’s disease [96, 97].
Chemical Targeting
In addition to actively targeted delivery supported by ligand-receptor specific interaction, chemical properties of the carrier system can also generate cell- or tissue-specific delivery. Unmodified polycationic carriers such as poly-L-lysine or PEI naturally interact with negatively charged heparan sulfate proteoglycans of the plasma membrane which leads to particle uptake [98, 99]. Lipoplexes and liposomes containing cationic lipids were found to destabilize the phospholipid bilayer of cell membranes and are subsequently internalized via receptor-independent endocytosis [100]. Recently, it was observed that liposomes and LNPs typically accumulate in the liver in their classical composition due to non-covalent attachment of serum proteins, especially apolipoprotein E (ApoE) resulting in transport to hepatocytes and uptake via low-density lipoprotein receptor (LDLR) [101–104]. In fact, the composition of the carrier systems largely influences the interaction with blood proteins [105]. Therefore, together with active targeting for tissue- or cell-specific nucleic acid delivery by ligand-receptor specific interaction, chemical adjustment of the carrier system can be also utilized to generate organ-specific delivery. It could be observed that slight changes of the chemical or physicochemical properties of the formulation such as particle size and surface charge have a remarkable impact on the biodistribution and accumulation in certain tissues or cell types.
Chemical targeting has shown great potential for ligand-independent, but yet organ-specific delivery of nucleic acids. For example, Kowalski et al. observed organ-selective distribution of mRNA-LNPs with a library of amino-polyesters either in liver, spleen, or lung after i.v. injection [106]. Localization of structurally different LNPs after systemic administration was investigated by Dahlman et al. by developing a barcode tool in order to track biodistribution in vivo [107]. Furthermore, the group of Siegwart synthesized libraries of lipids for LNP formulations for delivery of mRNA as well as Cas9 with different properties by variation of charge, hydrophobicity and pKa, respectively, resulting in so-called “selective organ targeting” (SORT) lipids and studied the accumulation of these formulations in different tissues and cell types. For example, it was demonstrated that particles with higher amounts of positively charged dioleoyl-3-trimethylammonium propane (DOTAP) preferably accumulated in the lung, whereas the addition of negatively charged lipids led to selective delivery to the spleen [108, 109]. Recently, the mechanism behind SORT was studied, revealing both global and apparent pKa as well as the composition of the serum protein corona of the LNP formulation determining the selective delivery. The study revealed first that particles with a pKa around 6–7 accumulated in the lung, while LNPs with lower pKa from 2 to 6 were preferably delivered to the spleen and second that different serum proteins bind to LNPs dependent on the component composition, therefore representing pioneering results for understanding LNP delivery beyond hepatocytes for future fields of applications [110].
Liver as Target
The liver is a highly metabolic organ and source of numerous expressed genes and plasma proteins. Not surprisingly, this organ is also a main target for nucleic acid and gene therapy of a series of severe hereditary monogenetic diseases [111–113]. In addition, non-inherited hepatic diseases such as liver cirrhosis or hepatitis B and C or hepatocarcinoma are life-threatening [114]. Therefore, the liver presents a high-priority target for nucleic acid delivery. Targeting of hepatocytes can be approached either by active or indirect active targeting, dependent from the carrier system as summarized in Table II.
Table II.
Targeted Nucleic Acid Delivery to Different Liver Cell Types
| Receptor | Delivery system | Ligand | Type of nucleic acid | Results | Reference | |
|---|---|---|---|---|---|---|
| Hepatocytes | ||||||
| ASGPR | PLL polyplex | ASOR | pDNA | First report on targeted, hepatocyte-specific gene delivery | [24, 31] | |
| PLL polyplex | Artificial tetra-antennary GalNAc ligand | pDNA | Conjugation of artificial ligand to the polyplexes results in comparable gene transfer efficiency as with the natural ligand asialofetuin, monovalent ligand does not improve gene expression | [60] | ||
| Polymer-nucleic acid conjugate | GalNAc | siRNA | in vitro and in vivo hepatocyte-specific delivery of siRNA | [120] | ||
| Direct conjugate | Tri-GalNAc | siRNA | FDA and EMA approval of several products: | |||
| Direct conjugate | Tri-GalNAc | siRNA | -Givosiran for treatment of acute intermittent porphyria | [123] | ||
| Direct conjugate | Tri-GalNAc | siRNA | -Lumarisan for treatment of primary hyperoxaluria type 1 | [124, 125] | ||
| Direct conjugate | Tri-GalNAc | siRNA | -Inclisiran for treatment of primary hypercholesterolaemia | [128] | ||
| LNP | Tri-GalNAc | siRNA | Exogenous ligand Tri-GalNAc mediates ASGPR-dependent uptake | [104] | ||
| Direct conjugate | Tri-GalNAc | ASO | Enhanced uptake, improved delivery and activity duration of clinically relevant ASOs to hepatocytes in vivo | [130] | ||
| Direct conjugate | Tri-GalNAc | ASO | Improved uptake and activity of targeted ASOs in human clinical trial | [133] | ||
| Direct conjugate | Tri-GalNAc | ASO | Safety, pharmacokinetic and pharmacodynamic study of GalNAc-ASO for treatment of β-thalassemia in monkeys | [134] | ||
| Direct conjugate | Tri-GalNAc | ASO | Enhanced uptake by hepatocytes, but not other liver cell types | [131] | ||
| Direct conjugate | Tri-GalNAc | ASO | Increased delivery of anti-miRNA-ASOs to hepatocyte in presence of ligand | [132] | ||
| Direct conjugate | Tri-GalNAc | Cas9 RNP | Disulfide linkage between Cas9 and GalNAc ligand led to receptor-dependent, selective uptake by hepatocytes and exhibited gene editing activity | [135] | ||
| LDLR | Direct conjugate | ApoB (endogenous) | siRNA | Delivery of siRNA to hepatocytes generated gene silencing of apoB protein expression | [136] | |
| Direct conjugate | AopB (endogenous) | ASO | Improved uptake of ASOs by hepatocytes after ligation of cholesterol | [131] | ||
| LNP | ApoE (endogenous) | siRNA | Uptake of LNPs is mediated by LDL-receptor determined by formation of ApoE-containing protein corona | [104] | ||
| LNP | ApoE (endogenous) | siRNA | Development of Patisiran for treatment of hereditary transthyretin amyloidosis, EMA and FDA approval in 2018 | [137] | ||
| LNP | ApoE |
Cas9-mRNA/ sgRNA |
Efficient TTR gene knockout in vivo in phase 1 clinical trial (57% after infusion of 0.1 mg/kg and 87% after 0.3 mg/kg); mild adverse effects | [19] | ||
| Hepatic stellate cells | ||||||
| RBP receptor | Liposome | Vitamin A | siRNA | Down-regulation of collagen synthesis after RBP receptor mediated uptake led to resolution of liver cirrhosis and fibrosis in rats after repeated treatments | [64, 141] | |
| PDGFR β | LNP | Cyclic peptide pPB | siRNA | Increased uptake of targeted SNALPs by HSCs; accumulation in liver after i.v. injection in mice, down regulation of gp46 mRNA expression, which is high in hepatic fibrosis | [83] | |
| Liver sinusoidal endothelial cells and Kupffer cells | ||||||
| n.a | LNP | None (chemical targeting) | Barcode DNA, siRNA, sgRNA, mRNA |
Alteration of cholesterol in LNP composition: Oxidized and esterified cholesterol mediated uptake by LSECs, cholesterol-oleate led to threefold enhanced gene editing activity in LSECs compared to hepatocytes |
[143, 144] | |
| n.a | LNP | None (chemical targeting) | Barcode DNA, mRNA | Exchange of DLin-MC3 by cKK-E12 leads partly to LNP uptake by LSECs and KCs | [142] | |
| n.a | LNP | None (chemical targeting) | Barcode DNA, mRNA | Adamantyl-phospholipids shifted distribution from hepatocytes to KCS and LSECs, but not to extrahepatic immune cells | [145] | |
| n.a | LNP | None (chemical targeting) | mRNA | Uptake by LSECs and KCs was achieved by replacing zwitterionic DSPC with anionic DSPG | [146] | |
Abbreviations: ASGPR, Asialoglycoprotein receptor; PLL, poly-L-lysine; ASOR, asialoorosomucoid; GalNAc, N-acetyl galactosamine; FDA, U.S. Food and Drug Administration; EMA, European medicines agency; LDLR, low-density lipoprotein receptor; ApoB, apolipoprotein B; ApoE, apolipoprotein E; RBP, retinol binding protein; PDGFR β, platelet-derived growth factor receptor β; pPB; SNALPs, stable nucleic acid lipid particle; HSC, hepatic stellate cell, LNP, lipid nanoparticle; LSECs, liver sinusoidal endothelial cells; DLin-MC3
Hepatocytes
Hepatocyte Targeting via Asialoglycoprotein Receptor
Delivering nucleic acids into hepatocytes is mostly achieved by receptor-mediated endocytosis via the ASGPR, which is found almost exclusively and abundantly on hepatocytes [115]. ASGPR binds highly selective to terminal, multi-antennary galactose and N-acetyl galactosamine (GalNAc) residues of glycoproteins with defined spatial geometry in presence of calcium(II)-ions [116–118].
The first targeted delivery of DNA by a non-viral delivery system was reported by Wu et al. by using asialoorosomucoid-modified (ASOR) polylysine polyplexes mediating delivery to the liver via ASGPR in vitro and in vivo after intravenous (i.v.) injection [24, 31]. Later, Plank et al. used an artificial tetra-antennary galactose ligand for gene transfer of pDNA-poly(lysine) polyplexes to hepatocytes in vitro [60]. Artificial ligands for ASGPR targeting were extensively optimized, as spatial distance of the carbohydrate residues, a well-balanced equilibrium of hydrophilicity and hydrophobicity of the linker largely impact the binding affinity towards the receptor [119].
Rozema et al. developed a dynamic polymer-nucleic acid conjugate, that fulfilled several tasks in one: The backbone itself provided endosomolytic properties, served as reaction site for the covalent attachment of siRNA and was grafted with GalNAc and PEG, giving a targeted and shielded vehicle. This formulation induced gene silencing activity in the liver after i.v. injection in mice [120].
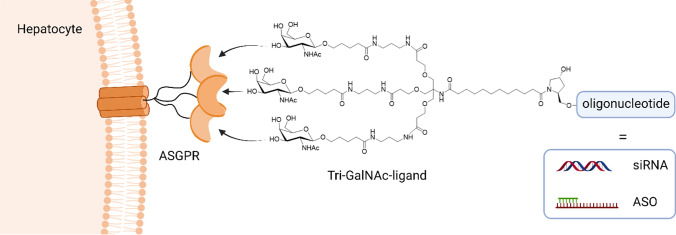
One milestone in both RNAi therapeutics and ASGPR mediated nucleic acid delivery is represented by the market release of Givosiran (Givlaari) in 2019 by the U.S. Food and Drug Administration (FDA) and European medicines agency (EMA). The nucleic acid, a chemically modified and stabilized siRNA, is directly conjugated to a tri-GalNAc ligand optimized towards ASGPR binding sites (Fig. 3). When administered subcutaneously, the direct conjugate enabled efficient gene silencing of aminolevulinic acid synthase 1 (ALAS1) aiming for reduced levels of ALA and PBG metabolites by RNAi for the treatment of acute hepatic porphyria [121–123]. Since then, two additional RNAi-therapeutics have been released to the market for treating rare, monogenetic hepatic diseases, using the same concept, i.e., the direct conjugation of trivalent GalNAc ligand to therapeutic siRNA. Lumasiran (Oxlumo) gained FDA approval in 2020 and targets the silencing of the gene encoding glycolate oxidase for the treatment of primary hyperoxaluria type 1 (PH1) [124, 125]. Inclisiran (Leqvio) also represents a direct siRNA-TriGalNAc conjugate targeting the inhibition of the translation of serum protease PCSK9 in order to regulate cholesterol blood level for the treatment of hypercholesteremia after subcutaneous injection. [126–128]. Thanks to the success of GalNAc-siRNA direct conjugates, several further therapeutics based on the same concept are currently in phase 2/3 clinical trials [129].
Fig.3.
Optimized trivalent GalNAc-ligand for hepatocyte delivery of direct conjugates with siRNA and ASOs, respectively, via ASGPR-mediated endocytosis. Created with BioRender.com
Additionally, the approach of direct conjugates has been further expanded to other cargos, e.g., antisense oligonucleotides [130–134] or Cas9 RNP complexes [135]. For example, trivalent GalNAc ligands were conjugated to antisense oligonucleotides, which enabled hepatocyte-specific delivery and enhanced the activity of clinically relevant human ASOs in mouse models [130], monkeys [134] and humans [133]. In another study, ASOs were conjugated with trivalent GalNAc and cholesterol, respectively, reporting enhanced uptake by hepatocytes [131]. Recently, Yamamoto et al. demonstrated that the conjugation of GalNAc to anti-miRNA ASOs led to highly increased potency [132]. A novel, trivalent GalNAc ligand which showed high affinity towards ASGPR (KD < 100 pM) mediated receptor-dependent, hepatocyte specific delivery and selective gene editing of CRISPR/Cas9 RNP complex [135].
Hepatocyte Targeting via LDL Receptor
In addition to active targeting of hepatocytes via ASGPR by GalNAc-modified formulations, uptake of lipid formulations can be also achieved by LDLR-mediated endocytosis. For example, cholesterol-siRNA direct conjugates for apolipoprotein B (ApoB) silencing have been found to exhibit gene silencing activity in hepatocytes in vivo [136]. LDLR-mediated uptake was observed due to interactions of cholesterol with serum proteins. The same approach was used for ASO direct conjugates by Watanabe et al. to enhance uptake and ASO activity in hepatocytes [131].
Furthermore, it was found that neutral liposomes interact mostly with apolipoprotein E (ApoE) in the blood, which directs the transport to hepatocytes by LDLR-mediated endocytosis [101–103]. Based on this observation, it was concluded that LNPs, multicomponent mixtures of cholesterol, an ionizable, cationic lipid, neutral helper lipids and a PEG-lipid for nucleic acid compaction, which appear almost neutral in serum, interact in a similar way with ApoE [104]. Thus, ApoE was identified as an endogenous ligand mediating the hepatocytic uptake of LNPs via LDLR [104].
In particular, Patisiran (Onpattro), the first siRNA product which was approved by FDA and EMA in 2018, is using LDLR interaction for the delivery of siRNA against transthyretin (TTR) mRNA to treat hereditary TTR mediated amyloidosis. By using a sheddable PEG-component, the formation of ApoE protein corona was observed, resulting in hepatocyte uptake via LDL receptor [137, 138]. In a recent phase 1 clinical study by Gilmore et al. the therapeutic effect of Cas9-mRNA/sgRNA targeting TTR, encapsulated by LNPs was evaluated. As a result of efficient TTR gene knockout, an average decrease of 87% of TTR protein levels was observed after one month in the patient group that received 0.3 mg/kg, accompanied with only mild side-effects. [19].
Targeting Liver Cell Types Beyond Hepatocytes
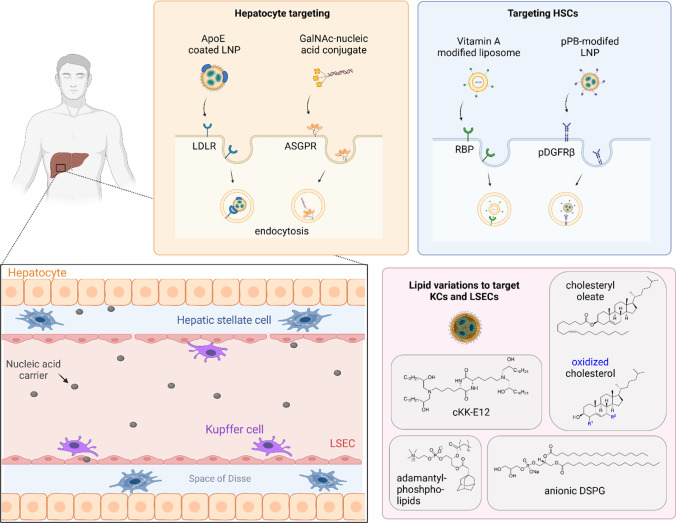
Approximately 80% of the liver is composed of hepatocytes. However, other cell types which are part of the hepatic reticuloendothelial system (RES), such as liver sinusoidal endothelial cells (LSECs), hepatic stellate cells (HSCs) and Kupffer cells (KCs) also represent interesting targets for nucleic acid therapeutics. Although KCs are very effective in removing and destroying nanosystems, they are much more difficult to be productively transfected with commonly used nucleic acid carriers. Therefore, several attempts were made to enable nucleic acid delivery to these cell types, including receptor-targeting strategies as well as the development of novel lipids for LNPs aiming for chemical targeting (Fig. 4).
Fig. 4.
Strategies to target different liver cell types: Hepatocytes (orange), hepatic stellate cells (blue), Kupffer cells (purple) and liver sinusoidal endothelial cells (light red). Created with BioRender.com
Hepatic Stellate Cells
While hepatic stellate cells make up about 5–8% of the cells in a healthy liver, the fibrotic liver consists of 15% HSCs. Nucleic acid delivery to activated HSCs is believed to reduce fibrosis by regulating fibrogenic cytokines [139, 140].
For example, Sato et al. accomplished HSC-targeted delivery of liposomes and LNPs by decorating the particle surface with vitamin A [64, 141]. As HSCs are a main storage for vitamin A, uptake of the liposomes was mediated by retinol binding receptor, which led to suppression of cirrhosis in a cirrhotic liver rat model by delivering therapeutic siRNA [64] and showed ability to promote regeneration of chronically injured liver [141]. Fibrotic HSCs were moreover targeted by modification of siRNA-LNPs with a cyclic peptide ligand (pPB) that interacts with the platelet-derived growth factor receptor β. It could be observed that pPB-targeted LNPs accumulated with high specificity in HSCs confirmed by biodistribution experiments after systemic injection [83].
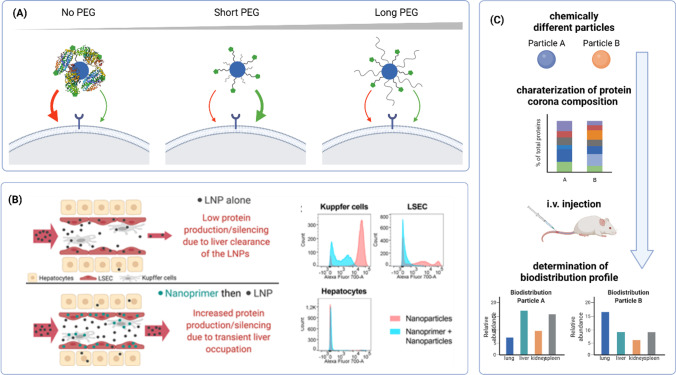
Studies conducted by the Dahlman group using the barcode technology described before for in vivo screening of lipid compositions aimed for delivery of chemically different LNP formulations beyond hepatocytes and the understanding of particle distribution in the liver microenvironment without the requirement for additional receptor-targeting ligands [142].
Liver Sinusoidal Endothelial Cells and Kupffer Cells
Targeted nucleic acid delivery to LSECs and KCs, which belong to the hepatic RES using chemical targeting has moved into the focus of attention.
Dahlman et al. used their barcode screening tool to tune the LNP biodistribution in mice based on alterations in the cholesterol component. As a consequence, the biodistribution shifted from hepatocytes to LSECs and KCs, respectively, using cholesterol-oleate or oxidized cholesterol [143, 144]. Additionally, the distribution of commercial LNP formulations with DLin-MC3 compared to the ionizable lipid cKK-E12 was assessed, revealing that both formulations were not only delivered to hepatocytes but partly to LSECs and KCs [142]. Furthermore, variations within the alkyl chains of the phospholipids giving “constrained” adamantyl-phospholipids which delivered the cargo specifically to KCs and LSECs, but not to immune cells outside the liver were evaluated [145].
Recently, Pattipeiluhu et al. developed LNPs for delivery of mRNA to hepatic RES resembling the Onpattro® formulation. By replacing the phospholipid within the LNP from the zwitterionic 1,2-distearoyl-sn-glycero-3-phosphocholine (DSPC) to the anionic 1,2-distearoyl-sn-glycero-3-phosphoglycerol (DSPG) a negative surface charge was created and specific uptake by LSECs under participation of Stabilin receptors in embryonic zebrafish and mice was achieved [146].
Immune Cells as Targets
Immune cells represent an interesting target for nucleic acid delivery as they play an important role in a wide range of diseases, including cancer, inflammatory or autoimmune diseases, etc. Besides the hepatic RES, immune cells are ubiquitous in the organism, especially in the spleen. Delivery of nucleic acid therapeutics to leukocytes, which include macrophages and dendritic cells as well as lymphocytes, offers the way to introduce genetic material with anti-inflammatory potential or to provoke T-cell modulation as a mean of immune stimulation [147, 148].
Macrophages
Macrophages, including the aforementioned Kupffer cells, express membrane lectins which recognize certain carbohydrate patterns, such as the mannose receptor CD206, that mediate endocytosis as a central function of immune response [149, 150]. This mechanism was used for targeted delivery of nucleic acids to macrophages. For example, DNA transfection to macrophages was accomplished by Erbacher et al. using mannosylated PLL polyplexes by interaction with the mannose receptor [151]. Mannosylation of PEG-PLL polyplexes caused an increase in transfection efficacy by about 8 times compared to untargeted polyplexes in a recent study by Lopukhov. Within the same study, the transfection efficacy of polyAsp(DET)-DNA polyplexes was boosted about 500 times when mannose residues were incorporated in the formulation [152]. Moreover, mannose-functionalized nanohydrogels have shown to efficiently deliver siRNA to CD206 + primary macrophages both in vitro and in vivo, which offers the opportunity for targeted gene regulation in immunosuppressive macrophages [153, 154]. In addition, Uehara et al. demonstrated efficient, ligand dependent gene silencing activity of a direct conjugate between siRNA and a tetravalent, chemically modified mannose in macrophages, which represents the first report of systemic delivery of siRNA-ligand conjugates to leukocytes [61].
Dendritic Cells
DCs, serving as antigen presenting cells (APCs), play a crucial role in antigen recognition of antigens and activation of immune response after uptake of foreign particles. Particles are internalized via phagocytosis or receptor-mediated endocytosis, dependent on their size and surface modifications. However, for immunotherapy it can be desirable to target DCs specifically (Table III).
Table III.
Active Targeting of Different Immune Cell Types
| Receptor | Ligand | Delivery System | Type of nucleic acid | Key results | Reference | |
|---|---|---|---|---|---|---|
| Macrophages | ||||||
| Mannose receptor | Mannose | PLL polyplexes | DNA | Transfection to monocyte-derived macrophages using Man-PLL polyplexes | [151] | |
| Mannose |
PLL polyplexes and pAsp(DET) polyplexes |
DNA | 8× increased transfection efficacy for Man-PLL polyplexes, 500× increased transfection efficacy for Man-pAsp(DET) polyplexes in murine bone marrow derived macrophages | [152] | ||
| CM Mannose | Direct conjugate | siRNA | Ligand-dependent gene silencing in monocyte-derived macrophages (in vitro) and in splenic and liver macrophages (in vivo) | [61] | ||
| Mannose | Cationic nano-hydrogel | siRNA | Receptor-dependent delivery of siRNA to M2 macrophages and efficient gene knockdown in primary cells and in mice | [153, 154] | ||
| Dendritic cells | ||||||
| Mannose receptor | Mannose | PEI polyplexes | pDNA | Increase of transfection efficacy by mannosylation of PEI polyplexes, uptake was reduced in presence of the inhibitor Mannose-BSA | [82] | |
| Mannose | PEGylated LNPs | mRNA | Variation of PEG-spacer length (PEG100, PEG1000 and PEG2000) was evaluated; LNPs with Man-PEG1000 showed highest transfection efficacy | [156] | ||
| CMM | Direct conjugate | siRNA | Ligand-dependent gene silencing activity in monocyte derived DCs | [61] | ||
| Mannan | LNP | Self-amplifying RNA | Enhanced immunization was observed for LNPs decorated with multivalent mannose residues | [157] | ||
| DEC205 | Anti-DEC205 scFv | LNP | siRNA | DEC205-dependency on uptake was demonstrated; targeted LNPs showed twofold increase in uptake compared to untargeted LNPs and LNPs with an isotype of scFv | [160] | |
| T-lymphocytes | ||||||
| CD3 T-cell receptor | Anti-CD3 antibodies | PLL polyplexes | pDNA | 1000-fold enhanced gene expression compared to unmodified PLL and Tf-PLL in T-cells; successful transfection to primary human lymphocytes | [58] | |
| CD4 | Anti-CD4 mAb | LNP | siRNA | Specific delivery to CD4+ cells ex vivo; gene silencing activity was observed in blood, bone marrow, spleen and lymph nodes | [59] | |
| Ly6c | Anti-Ly6-mAb | LNP | mmRNA | Targeted delivery to Ly6c positive cells in vitro; in vivo evaluation in IBD mouse model showed increased protein expression [20-fold in intestine, tenfold in spleen]; expression of anti-inflammatory IL-10 after delivery of IL-10 encoding mmRNA | [164, 165] | |
| n.a | None (chemical targeting) | LNP | Barcode siRNA, sgRNA |
Screening of 168 different LNP formulations in vivo with variations of head group, lipid alkyl chains, phospholipid and molar composition; adamantyl-DSPC delivered siRNA and sgRNA to T cells (and Kupffer cells) |
[166] | |
| n.a | None (chemical targeting) | LNP | siRNA | Variation of head group and alkyl chain; piperazine headgroup led to accumulation and gene silencing in the spleen | [167] | |
| Integrin β7 | Anti-β7-mAb | LNP | siRNA | CD45 mRNA silencing in CD4+ and CD8+ T cells in spleen and lymph nodes | [167] | |
Abbreviations: PLL, Poly-L-lysine; Man, Mannose; pAsp(DET) poly(N-[N-[2-aminoethyl]-2-aminoethyl] aspartamide); CM Mannose, chemically modified mannose; PEI, polyethylene imine; BSA, bovine serum albumin; DCs, dendritic cells; scFv, single chain antibody; CD, cluster of differentiation; Tf, Transferrin; mAb, monoclonal antibody; Ly6c, lymphocyte antigen 6 complex; mmRNA, modified messenger RNA; IBD, inflammatory bowel disease; mAb, monoclonal antibody
DNA delivery to DCs via mannose receptor was described by Diebold et al. by using mannose functionalized PEI polyplexes. Receptor-specific uptake was demonstrated in a competition assay with mannose albumin, which lowered gene expression of Man-polyplexes [82]. Gao et al. designed mannose ligands optimized towards the carbohydrate recognition domains of mannose receptor and the DC-specific intercellular adhesion molecule-3-grabbing non-integrin (DC-SIGN, CD209), respectively. Liposome uptake by DC2.4 cells and DC-SIGN expressing HEK293 cells was observed in a ligand-dependent manner. These particles were not yet used to deliver nucleic acids to DCs but provided deeper insight in the understanding of ligand design to improve receptor interaction [155]. The impact of PEG spacer lengths on particle size, stability and transfection efficacy was also examined using Man-PEG-cholesterol lipids in mRNA-LNP formulations. It could be shown that zetapotential and particle size remained unchanged by increasing PEG lengths, whereas PEG1000 showed highest transfection efficacy while maintaining serum stability [156]. Recently, the effect of mannan-coating of LNPs for the delivery of RNA vaccines was investigated by using mono- and multivalent mannose residues linked to cholesterol, resulting in an increased immunization arguing for the use of targeted, mannan-functionalized RNA vaccines [157].
Targeting of dendritic cells was also achieved via DEC205, another receptor from the mannose receptor family [158, 159]. Katakowski et al. formulated LNPs bearing a single-chain antibody to target DEC205 expressing murine DCs. Receptor-specific binding and uptake of the siRNA-LNPs were determined via flowcytometry, showing that uptake was twofold improved by targeted LNPs. DEC205-dependent internalization was further confirmed by reduced knock-down efficacy in DEC205 deficient mice [160].
The spleen and more specifically DCs were chemically targeted by lipoplexes with an inversed lipid/RNA charge ratio giving negatively charged particles. After systemic administration, these particles successfully delivered mRNA encoding for antigens and promoted stimulation of APCs for cancer immunotherapy [161].
T-lymphocytes
T-lymphocytes also play a crucial role in cellular immune response [162]. Therapy of immune related disorders, e.g., inflammation or cancer, can be achieved by RNAi in T-lymphocytes. As gene delivery in these cells has appeared to be challenging [147, 163], the carrier systems have been decorated with ligands, mostly monoclonal antibodies, to improve nucleic acid delivery. For example, Buschle et al. achieved gene transfer to human T-lymphocytes by decorating Tf-PLL polyplexes with antibodies against the CD3 T cell receptor [58]. Ramishetti et al. aspired to deliver siRNA-LNPs to CD4 + T-lymphocytes by attachment of anti-CD4-monoclonal antibody, whereby the specific delivery to CD4 + lymphocytes could be confirmed ex vivo. Additionally, i.v. injection in mice showed gene silencing activity in spleen, lymph nodes, bone marrow and blood [59]. Veiga et al. evaluated LNPs loaded with modified mRNA for delivery to Ly6c + inflammatory leukocytes [164]. For this purpose, the targeting ligand was attached to the LNP via incorporation of lipoproteins interacting with antibodies [165]. Decoration with targeting mAbs towards inflammatory leukocytes led to strongly increased interleukin-10 expression in spleen and intestine [164].
Great advances regarding nucleic acid delivery to T-lymphocytes were made using the chemical targeting approach. Using the barcode screening technology, Lokugamage et al. studied the distribution of a library of 168 siRNA-LNP formulations with structural changes regarding the lipids in different cell types in vivo and showed that constrained LNPs preferably delivered the genetic material to splenic T lymphocytes instead of hepatocytes making these formulations interesting for immunotherapy [166].
A screening of 14 structurally different ionizable lipids by variation of linker backbone, head group and alkyl chains for delivery of siRNA-LNPs into leukocytes was reported by Ramishetti et al. The biodistribution after i.v. injection in mice showed accumulation in spleen for piperazine head group and in liver for tertiary amine head group [167]. In the same study, specificity was further improved by combination of both, chemical and active targeting. Decoration of the LNPs with anti-integrin β-mAbs resulted in CD45 mRNA knockdown in CD4 + and CD8 + lymphocytes in spleen and lymph nodes. Nevertheless, only a limited gene silencing ability was detected overall [167].
Lung as Target
Many severe, eventually lethal diseases are associated with the lung, for example cystic fibrosis, chronic obstructive pulmonary disease (COPD), asthma or pulmonary fibrosis amongst others. One advantage of nucleic acid delivery to the lung is certainly the accessibility of the lung via local and systemic administration routes. However, protective mechanisms and physiological barriers such as mucosal barrier or immune cells may impair the delivery of nucleic acids [168]. For selective, targeted nucleic acid transfer into the lung chemical as well as receptor-mediated targeting strategies have been evaluated with the key results summarized in Table IV.
Table IV.
Receptor-Mediated Non-Viral Nucleic Acid Delivery to the Lung
| Receptor | Ligand | Delivery System | Type of nucleic acid |
Key results | Reference |
|---|---|---|---|---|---|
| Insulin receptor | Insulin | PEI polyplex | pDNA | Selective delivery to alveolar epithelial cells | [91] |
| Lactoferrin receptor | Lactoferrin | PEI polyplex | pDNA | Selective delivery to bronchial epithelial cells | [81] |
| Integrin | RGD motif | Liposome | pDNA | High transfection efficacy in lung endothelial cells for targeted liposomes in vivo | [54] |
| TAT-RGD motif | Direct conjugate | pDNA | Enhanced uptake of targeted particles by pulmonary cells | [176] | |
| Cationic liposome | pDNA | Fivefold increased gene expression in A549 cells compared to lipofectamine | |||
| RGD motif | Lipoplex | pDNA | Significantly improved transfection efficacy for RGD-bearing polyplexes | [177] | |
|
Polymeric IgR |
Antisecretory component antibody | PLL polyplex | pDNA | Proof of concept for transfection efficacy to human tracheal epithelial cells, competition assay blocked uptake of targeted polyplexes | [178] |
| PECAM | Anti-PECAM antibody | PEI polyplex | pDNA | Enhanced gene transfer efficacy and reduced toxicity | [179] |
| Anti-PECAM antibody | LNP | mRNA | Enhanced protein expression in lung endothelial cells; reduced accumulation in hepatocytes | [84] | |
| Transferrin receptor | Transferrin | PEI polyplex | siRNA | Enhanced uptake of Tf-PEI polyplexes by pulmonary ATCs in vitro and in asthma mouse model after intratracheal application; improved endosomal escape by addition of melittin, 40% more effective than lipofectamine | [180, 181] |
| β2-adrenoceptor | Clenbuterol | PEI polyplex | pDNA | Enhanced gene expression in alveolar epithelial cells | [50] |
| Salbutamol | Chitosan polyplex | siRNA | Delivery to bronchial epithelial cells | [51] | |
| IP1 | Iloprost and Treprostinil | PEI polyplex | pDNA | Enhanced transfection efficacy | [52] |
| Lectins |
Galactose Glucose Lactose |
PLL polyplex | pDNA | Improved, sugar-type dependent gene expression in cystic fibrosis airway epithelial cells | [185-188] |
| Galactose | PEGylated PEI polyplex | pDNA | Increased transfection efficacy in vitro and in vivo | [189] |
Abbreviations: PEI, polyethylene imine; RGD, arginine-glycine-aspartic acid; TAT, transactivated transcription peptide; Ig, immunoglobulin receptor; PLL, poly-L-lysine; PECAM, platelet endothelial cell adhesion molecule; IP1, prostacyclin receptor; PEI, polyethylene imine
Due to their positive surface charge, many polyplex (PEI) as well as cationic liposome formulations automatically accumulate in the lung when injected systemically [169–172]. In addition, efforts were made to generate lung targeted LNPs, which typically accumulate in hepatocytes, by modification of lipid composition and the type of ionizable lipid. In the course of the development of “SORT”-LNPs, it was reported that increasing amounts of the positively charged component DOTAP shifted accumulation from hepatocytes to lung endothelial cells [108]. Chemical targeted synthetic carriers for specific lung delivery of mRNA and pDNA were developed by Kaczmarek et al. The hybrid polymer-lipid formulations used in their studies, consisting of poly(β-amino esters) (PBAEs) and PEGylated lipids, generated protein expression in the lung after i.v. injection in mice, but not in other organs [173–175].
Active targeting of lung epithelial cells resulting in receptor-mediated uptake of non-viral delivery systems was obtained by several classes of ligands, such as peptides, proteins, antibodies, carbohydrates and also small drugs. Elfinger et al. studied the selective delivery to different lung epithelial cell types. It was reported that pDNA/PEI polyplexes modified with lactoferrin delivered the nucleic acid selectively to bronchial epithelial cells via lactoferrin receptor, but not alveolar epithelial cells, whereas adsorption of insulin to pDNA/PEI polyplexes showed increased luciferase gene expression in alveolar epithelial cells, but not in bronchial epithelial cells [81, 91]. Integrins are also abundantly found on lung cells and attempts were made to achieve receptor-mediated uptake via caveolae-dependent pathway by incorporation of arginine-glycine-aspartic acid (RGD) motifs into the delivery system [54, 176, 177]. More specific targeting of the lung was achieved using antibodies as ligands. In early studies, Ferkol et al. observed targeted delivery of pDNA-polylysine complexes conjugated with Fab fragments of immunoglobulins directed against the polymeric immunoglobulin receptor (IgR) which is involved in the transport of immunoglobulins A and M from cell surface into lung epithelial cells. A competition assay with excess of Fab ligand blocked delivery gives further evidence for receptor-mediated uptake [178]. Additionally, lung-specific nucleic acid transfer was accomplished using antibodies directed against the platelet endothelial cell adhesion molecule (PECAM). For example, Li et al. were able to generate higher gene expression after i.v. injection of anti-PECAM-mAb decorated pDNA-PEI polyplexes in mice, furthermore observing reduced cytotoxicity when using ligand-modified carriers [179]. More recently, mRNA-LNPs, which are known to accumulate in the liver, were modified with monoclonal antibodies directed against PECAM-1. Intravenous injection in mice avoided accumulation in hepatocytes but resulted in enhanced protein expression in lung endothelial cells [84].
Transferrin (Tf) was used as targeting ligand to mediate the delivery of PEI-siRNA polyplexes to pulmonary activated T cells (ATCs). A study by Xie et al. has shown that transferrin modification led to enhanced cellular uptake and efficient, selective gene knockdown in vitro as well as in an asthma mouse model after intratracheal application [180]. Further optimization of the Tf-PEI polyplexes by blending with PEI bearing the endosomolytic peptide melittin improved endosomal escape capability of the cargo resulting in enhanced cellular uptake [181]. In fact, optimization of endosomal escape properties for delivery of siRNA polyplexes to the lung has been subject of further studies. For example, Pun et al. developed a virus-inspired polymer for endosomal release (VIPER) [182], which was applied for efficient pulmonary delivery of siRNA both in vitro and in vivo [183]. VIPER/siRNA polyplexes also showed antiviral effect by promoting suppression of viral replication of SARS-CoV-2 ex vivo in human lung tissues and in mouse models [184].
Furthermore, small chemical compounds, which have already been used effectively as drugs for asthma treatment were used as targeting ligands coupled to synthetic nucleic acid carriers for targeted nucleic acid delivery to the lung. For example, agonists for the β2-adrenoceptor were successfully used for targeted and improved delivery of nucleic acids to lung epithelial cells. Elfinger et al. demonstrated enhanced gene expression in alveolar epithelial cells in vitro as well as in vivo after inhalation of Clenbuterol-functionalized polyplexes [50]. Specific delivery of siRNA to bronchial epithelial cells could be improved by coupling of Salbutamol to the formulation, as shown by Luo et al. using guanidinylated chitosan carriers [51]. In addition, PEI-polyplexes modified with Iloprost and Treprostinil, prostacyclin derivatives targeting the prostacyclin receptor IP1, also exhibited enhanced transfection efficacy of pDNA polyplexes in lung epithelial cells as well in vivo in the lungs of mice after aerosol administration [52]. For these chemical ligands, their possible dual role as drugs was not explored.
Additionally, lectins have been studied for lung-specific uptake of non-viral delivery systems. Several studies by Kollen et al. showed that gene expression of pDNA/polylysine polyplexes could be increased through functionalization with β-galactose, α-glucose as well as lactose compared to other monosaccharides and the non-targeted formulation after transfection to cystic fibrosis cells [185–188]. Transfection of galactosylated polyplexes also resulted in improved gene expression compared to non-targeted polyplexes in A549 cells as well as in vivo experiments after intratracheal administration [189]. In both studies, lectins were hypothesized to play a role in the specific uptake of the particles, although the particular uptake route was not further addressed.
Brain as Target
Many neurodegenerative disorders such as Alzheimer’s disease, Huntington’s disease, Parkinson’s disease or amyotrophic lateral sclerosis (ALS) originate in the central nervous system (CNS). Treatment of these diseases via systemic administration routes remains challenging due to poor accessibility of the brain through the blood–brain barrier (BBB). Therapeutic nucleic acids compacted into synthetic carrier systems are not able to cross the BBB via diffusion [190]. However, nucleic acid delivery to the brain via systemic administration is highly desired, as topic routes like intracranial or intracerebroventricular injections as well as physical methods that enhance the permeability of the BBB are highly invasive. Thus, synthetic carriers must be decorated with ligands, which are recognized by receptors or carriers embedded in the BBB, becoming “trojan horses”, which are enabled to deliver nucleic acid to the brain through receptor-mediated transcytosis (RMT) or carrier-mediated transcytosis (CMT). Various synthetic carrier systems were modified with several ligands, ranging from proteins, peptides and aptamers to generate brain-targeted gene delivery (see Table V).
Table V.
Receptors and Ligands for Targeted Delivery of Nucleic Acids to the Brain
| Receptor | Ligand | Delivery system | Type of nucleic acid | Key findings | Reference |
|---|---|---|---|---|---|
| Transferrin receptor | Transferrin | Lipoplex | siRNA | Efficient gene silencing in primary murine cortical neuronal cells and in vivo without cytotoxicity | [193] |
| Transferrin | PEG-PAMAM dendrimer polyplex | pDNA | Enhanced gene expression in BCECs and in mice brain | [194] | |
| Transferrin | PPI polyplex | pDNA | Targeted polyplexes accumulated in mice brain | [195] | |
| Anti-TfR-mAb | Immunoliposome | pDNA | TfR-mAb promoted both, crossing of BBB and delivery to TfR-expressing glioma cells | [196] | |
| re-TfR-peptide | Lipo-oligo(amidoamine) polyplex | pDNA | Enhanced luciferase gene expression in N2a cells compared to non-targeted lipoplexes and scrambled peptide ligand | [55] | |
| Lactoferrin receptor | Lactoferrin | PEG-PAMAM polyplex | pDNA | 2.2-fold increased gene expression in vivo, selective gene transfer to the brain | [199] |
| Lactoferrin | PPI Polyplex | pDNA | 2.1-fold increased gene expression in vitro, significantly higher gene expression in vivo | [200] | |
| LRP1 | Angiopep-2 | PEG-PAMAM dendrimer polyplex | pDNA | Selective uptake of polyplexes by BCECs, accumulation of targeted polyplexes in brain, untargeted in spleen | [205] |
| Angiopep-2 | LNP | siRNA | In vitro study of cellular uptake and gene silencing efficacy in U87MG and b.End3 cells | [206] | |
| nAChR | RVG29 | Oligoarginine polyplex | siRNA | Enhanced gene expression in brain after i.v. injection, but not in other organs | [212] |
| RVG29 | PEG-PAMAM dendrimer polyplex | pDNA | Brain accumulation after systemic administration, GABA receptor involved in uptake | [213] | |
| RVG29 | PEI polyplex | miRNA | Reduced signal of reporter gene due to silencing activity, accumulation in brain | [267] | |
| RVG29 | Trimethylated chitosan polyplex | siRNA | Efficient gene silencing of BACE1, accumulation in brain | [214] | |
| RVG29 | Poly(mannitol-co-) PEI polyplex | siRNA | Efficient gene silencing of BACE1 | [215] | |
| RVG29 | Exosomes | siRNA | Gene knock-down of BACE1 | [216] | |
| RVG-9r | SNALP | siRNA | Efficient silencing of Machado Joseph disease involved proteins in vitro and in vivo | [217] | |
| Laminin receptor | EPRNEEK | Dendrigraft PLL polyplex | DNA | Improved uptake and gene expression by exogenous ligand compared to endogenous laminin ligand | [218] |
| Leptin receptor | Leptin-30 peptide | PEGylated PLL Polyplex | pDNA | Improved transfection efficacy in BV-2 cells; accumulation in brain after i.v. injection | [210] |
| VCAM1 | Anti-VCAM-antibody | LNP | mRNA | Specific mRNA delivery to inflammatory brain, but not to leukocytes, expression of anti-inflammatory protein | [219] |
| GLUT1 | Glucose | Polyplex | ASO | Accumulation in mice brain after i.v. injection depending on glucose-concentration | [223] |
Abbreviations: PAMAM, poly(amidoamine); BCECs, brain capillary endothelial cells; PPI, polypropylene imine; TfR-mAb, transferrin receptor monoclonal antibody; re-TfR, retro-enantio transferrin receptor, N2a, neuro2a cell line; LRP-1, low-density lipoprotein receptor related protein 1; GLUT1, glucose transporter 1; nAChR, nicotinic acetylcholine receptor; RVG29, rabies virus derived 29-mer peptide; GABA, gamma-aminobutyric acid; BACE1, beta-secretase 1; SNALP, stable nucleic acid lipid particle; PLL, poly-L-lysine; VCAM1, vascular cell adhesion molecule 1
A possible approach to facilitate nucleic acid transfer into the CNS is to use receptor-mediated transcytosis by transport proteins that enable the passage of essential nutrients, proteins or lipids across the BBB.
Receptor-Mediated Transcytosis
Receptors for Iron Transport Proteins
Transport of iron across the BBB is mediated by several iron transport protein receptors, including transferrin, lactoferrin or melanotransferrin. Above all, transferrin receptor was widely studied for targeted uptake of synthetic carriers to the CNS. As known from previous studies, the transferrin protein itself holds great potential to mediate receptor-dependent polyplex uptake by TfR-expressing cells and therefore, was explored for mediating transcytosis across the BBB [191, 192]. For example, Cardoso et al. prepared Tf-modified siRNA-lipoplexes that showed superior uptake by neuronal cells as well as significant gene silencing in both, in vitro and in vivo compared to non-targeted particles [193]. Moreover, dendrimer-polyplexes based on PAMAM or PPI were functionalized with human transferrin, resulting in successful pDNA delivery across the BBB. Biodistribution studies further confirmed accumulation of TfR-targeted dendrimers in the brain [194, 195]. TfR was also targeted by immunoliposomes bearing monoclonal antibodies (mAb) directed against rat TfR for RNAi therapy via delivery of plasmids encoding for short hairpin RNA (shRNA). It could be demonstrated that TfR-targeted immunoliposomes provided a dual targeting effect, as they promoted BBB crossing and subsequent uptake of glioma cells, which are also overexpressing TfR [196]. Recently, TfR-mediated delivery to neuronal cells was successfully accomplished by our group using a retro-enantio peptide sequence that showed high affinity towards the transferrin receptor. The “retro-enantio” approach provides stability against peptide degradation by inversion of the peptide order and usage of D-configurated amino acids while maintaining receptor binding affinity [197]. The retro-enantio ligand was conjugated to sequence defined lipo-oligo(amidoamines) (lipo-OAAs) for the delivery of both, siRNA and pDNA, to N2a cells [55].
Lactoferrin Receptor
Besides TfR, the lactoferrin receptor (LfR) is also involved in cellular iron uptake and expressed on the BBB [198]. By using LfR, increased gene expression of dendrimer polyplex formulations bearing lactoferrin (Lf) could be observed. Lf-modification of PAMAM dendrimer polyplexes resulted in 2.2-fold increase of luciferase gene expression in vivo compared to untargeted particles. In addition, selective delivery to the brain was reported after systemic administration [199]. Additionally, lactoferrin-PPI-dendrimers showed improved transfection in b.End.3 cells and significantly increased accumulation in mice brain [200].
Melanotransferrin
Furthermore, a short 12-amino acid peptide which has shown interaction with the iron transport protein melanotransferrin was able to mediate entry in the brain. Conjugation to siRNA enabled not only accumulation in the brain, but also in vivo gene silencing of NOX4, a gene that is upregulated during stroke [201, 202].
LRP1
Nucleic acid transfer across the BBB was furthermore achieved by low-density lipoprotein receptor related protein (LRP1) mediated transcytosis. Demeule et al. developed Kunitz domain derived peptides from aprotinin, named Angiopep, that showed the ability to overcome BBB via LRP1-mediated transport [203, 204]. Angiopep-2 was attached to DNA/PAMAM-dendrimers showing selective uptake by BCECs in vitro and a shift of the biodistribution from spleen (for untargeted carriers) to brain (for targeted polyplexes) in vivo [205]. The same ligand was later included in siRNA-LNP formulations by Bruun et al. for in vitro studies on uptake and gene silencing activity in human glioblastoma U87MG and murine brain endothelial bEnd.3 cell line. In addition to an increased gene knock-down, it could be observed that uptake could be improved about 2.4-fold by Angiopep-2 modification [206]. Moreover, a novel artificial ligand named L57 was found to enable BBB crossing in vivo by interaction with LRP1 [207]. Compared to Angiopep-7, L57 showed enhanced CNS uptake capability and low cytotoxicity [208].
Leptin Receptor
Moreover, the leptin receptor, which is responsible for recognition and transcytosis of the appetite regulating peptide leptin, was used for nucleic acid delivery to the brain [209]. It was shown that a leptin-derived 30-amino acid peptide attached to poly-L-lysine carrier was able to generate improved DNA transfection in BV-2 cells and accumulation in mice brain after i.v. injection [210].
Pathogen-Derived Peptide Ligands
In addition to ligands interacting with receptors which transport essential molecules across the BBB, another approach is to modify the carrier system with peptides deriving from viruses, bacteria or venoms that naturally show capabilities to enter the brain.
For example, the peptide RVG29 was studied for nucleic acid transfer across the BBB. The ligand derived from rabies virus glycoprotein (RVG), which naturally shows the ability to enter the brain as part of its pathology and targets mainly the nicotinic acetylcholine receptor (nAChR) [211]. RVG29 was included in polyplex and LNP formulations yielding nucleic acid transfer across the BBB. For example, Kumar et al. [212] reported efficient gene silencing using RVG-bearing siRNA oligo(arginine) formulations. A study by Liu et al., using RVG29-PEG-PAMAM/DNA polyplexes, revealed involvement of GABA receptor in uptake [213]. Efficient gene silencing of BACE1, a protein involved in Alzheimer’s disease, could be achieved by several groups using polyplex as well as exosome formulations that were functionalized with RVG29 [214-216]. Furthermore, a lipid-containing formulation bearing RVG-oligo(arginine) residues exhibited the ability to silence a mutant ataxin-3, involved in the pathology of Machado-Joseph disease, a hereditary ataxia disorder [217].
Furthermore, Liu et al. demonstrated the capability of a peptide sequence derived from meningitis-causing pathogen to mediate brain-specific delivery of surface-modified PLL/DNA-dendrimers via laminin receptor, resulting in enhanced cellular uptake by BCECs and U87MG cells compared to an endogenous laminin-targeting ligand [218].
VCAM1
A recent study by Marcos-Contreras et al. explored the specific delivery of antibody-modified mRNA-LNPs using an anti-vascular cell adhesion molecule 1 (anti-VCAM1) antibody to the inflammatory brain. It was found that the particles were delivered to brain endothelial cells but not to leukocytes. Furthermore, as a consequence of successful mRNA delivery, expression of anti-inflammatory thrombomodulin could be observed in a mouse model [219].
Direct Conjugates for BBB-Targeting
Some of the aforementioned ligands, such as Angiopep and RVG-29, as well as further peptide ligands were used for direct conjugation to phosphorodiamidate morpholino oligomers (PMOs), which function as splice-switching oligonucleotides. It was demonstrated that a truncated peptide-derivative of ApoE mediates PMO delivery in the CNS [220].
Carrier-Mediated Transcytosis
Transcytosis via GLUT1
An additional pathway to overcome BBB is through transcytosis by glucose transporter 1 (GLUT1). As transport carrier for glucose, GLUT1 is abundantly expressed on brain capillary endothelial cell (BCEC) membrane, ensuring adequate glucose supply of the brain [221]. Researchers exploited this transport mechanism by using glucose modified nanoparticles as “trojan horses” to induce gene transfer into the brain and other GLUT1-rich cells. For example, Kataoka et al. developed glucose-decorated polymeric carriers, which facilitated nanoparticle delivery to GLUT1-rich cancer cells under glycemic control [62, 222]. These block-copolymers were applied for the delivery of ASOs to the brain via intravenous injection into mice, providing efficient gene knockdown [223]. Another glucose-ligand capable of crossing the BBB is an opioid-derived glyco-heptapeptide (g7). Even though the transport mechanism of g7-functionalized particles into the brain is not fully understood, decoration of nanoparticles with g7 showed brain specific uptake after i.v. injection into rat and therefore presents a promising ligand for nucleic acid delivery to the CNS [224-227].
Ocular Targeting
Retina
Many genetic eye disorders, either inherited or environment-dependent, may lead to loss of vision eventually due to impaired functions of photoreceptors or retinal pigment endothelium (RPE). Therapeutic approaches mostly use classic gene therapy with DNA for gene replacement or gene-editing methods. Formulations are usually injected subretinal or intravitreal due to the blood ocular barrier and in order to reduce off-target effects or elimination by immune system. Furthermore, persistent high levels of gene expression after single injections are highly desired for retinal gene therapy. Sustainable gene expression was achieved by using PEG-PLL/DNA polyplexes, which were locally injected in mouse models [228-230]. Delivery of nucleic acid therapeutics to the retina resulting in long-term gene expression was also reported by the group of Zheng-Rong Lu [231]. Lu and co-workers have developed a multifunctional ionizable lipid, called “ECO”, which served as efficient gene carrier system in several applications [232-234]. A hybrid ECO/G4-dendrimer formulation was applied successfully as carrier system for GFP reporter gene to human ARPE-19 cells and in animal experiments [235]. Moreover, ECO served as carrier for the therapeutic ABCA4 plasmid, supported by a rhodopsin promoter, for the treatment of Stargardt disease. These formulations provided up to 8 months of gene expression and disease progression delay for 6 months in ABCA4 deficient mice [236]. ECO/DNA formulations were also functionalized with PEG3.4 kDa-all-trans-retinylamine to target the interphotoreceptor retinoid binding protein (IRBP), a key protein in the retinoid cycle [237]. In this study, high transfection efficacy of the reporter gene GFP could be demonstrated in ARPE-19 cells and mouse models of Leber’s congenital amaurosis (LCA) type 2 after subretinal injection with high expression levels up to 120 days [238]. Additionally, a chemically stable retinoid analogue ACU4429, linked to the carrier system via pH-sensitive hydrazone-PEG3.4 kDa-spacer, was used for IRBP-mediated delivery of ECO/DNA particles and gene expression of ABCA4 in ARPE-19 cells as well as in abca4-/- mice [239].
Furthermore, liposome-protamine complexes have shown potential to promote long term gene expression or gene knock-down, respectively [240-242]. More recently, LNP formulations encapsulating either mRNA [243] or siRNA [244] were screened to investigate cell-specific retinal nucleic acid transfer depending on surface charge and LNP composition. It was observed that mRNA-LNPs containing ionizable lipid are preferably internalized by RPE cells, suggesting ApoE-mediated uptake, whereas formulations with permanently cationic lipids showed only low transfection efficacies in the retina [243]. Another study using siRNA-LNPs demonstrated that LNPs with positive zetapotential around +35 mV distributed preferably in the vitreous and retina after local injection [244].
Cornea
In order to address corneal gene delivery, researchers used hyaluronic acid as a targeting ligand for the CD44-receptor, which is expressed by human corneal epithelial cells and is responsible for turnover of HA [245-247]. For instance, de la Fuente et al. developed HA-chitosan nanoparticles loaded with reporter DNA which successfully transfected human corneal epithelial (HCE) cells and conjunctiva cells via the CD44-receptor [248, 249]. Further investigations of the internalization pathway revealed that the particles were endocytosed by caveolae-mediated endocytosis [49, 250]. CD44-receptor mediated intracorneal uptake was also assessed by Hornof et al. using HA-coated PEI/DNA polyplexes demonstrating that nanoparticles coated with low-molecular weight HA generated well shielded, stable particles while maintaining efficient transfection activity [48].
Muscle as Target
The delivery of nucleic acids to skeletal and cardiac muscles allows the treatment of muscle-related disorders such as muscular dystrophy. Efforts were made to develop lipid-siRNA conjugates that enable muscle-targeted delivery upon systemic injection. Therefore, a library of lipid-ASO conjugates were screened regarding their ability to deliver functional ASOs to muscle cells after i.v. injection in mice. It was demonstrated that delivery to muscle cells was dependent on the length of the fatty acid, with C16 to C22 showing highest accumulation based on their affinity to albumin which mediated transport to muscle cells [251]. The palmitic-ASO conjugate was further evaluated revealing a slight increase in ASO activity, but relatively rapid clearance [252]. A following study evaluated ASO potency and association with human, rodent and monkey plasma proteins, showing a preferred binding of palmitate-ASOs to human and rodent albumin as well as histidine-rich glycoprotein possibly explaining enhanced ASO activity in the muscle. Additionally, an enhanced in vivo ASO potency was observed in rodents, but only a modest improvement in monkeys [253]. Moreover, different lipids were conjugated to chemically stabilized siRNA for a distribution study in mice. Most formulations accumulated in clearance organs such as liver or kidney, while docosanoic acid-siRNA conjugate (DCA) delivered siRNA partly to other tissues [254]. Compared to cholesterol-siRNA, delivery of DCA-conjugates to skeletal and cardiac muscles was enhanced about threefold and 2.5-fold, respectively. Using DCA conjugates, sustained silencing of myostatin mRNA in muscles was obtained leading to reduced myostatin protein levels and promotion of muscle growth after systemic injection [255].
Active targeting: In Vitro Versus In Vivo
It is noticeable that delivery systems using targeting ligands are rather rare on the medical market. Only a few products such as GalNAc direct siRNA conjugates and ApoE endogenously targeted Patisiran were approved by FDA. In fact, the majority of targeting ligands was evaluated in cell culture studies, demonstrating improved in vitro performance. However, most formulations have not taken the step to product development for in vivo applications.
Potential reasons for this observation must be considered and evaluated in order to find explanations for this translational bottleneck. There is a great discrepancy between the results obtained from in vitro studies and in vivo performance, making predictions for (pre-)clinical studies questionable when drawn from cell culture evaluation [38-40].
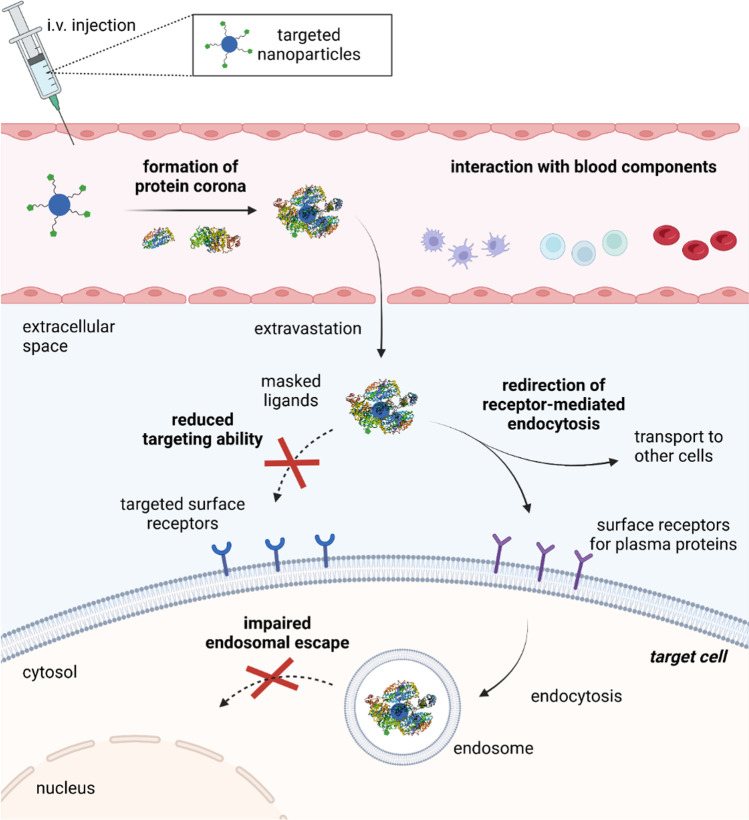
In vivo nucleic acid delivery appears to be affected by several barriers. In contrast to small molecule drugs, nucleic acids exhibit unfavored pharmacokinetic and pharmacodynamic profiles due to their high molecular weight and charge, excluding membrane diffusion as internalization route. The demand for endocytic internalization pathways, membrane barriers become more difficult to overcome and formulation properties have to be carefully tailored, resulting in complex, multi-component nanoparticle formulations. Furthermore, different types of nucleic acid therapeutics have different requirements on the formulation. Therefore, addressing new obstacles, i.e., cellular barriers such as nanoparticle uptake and endosomal escape represent major bottlenecks for clinical translation, as discussed in detail by several researchers [256-259]. Prior to cellular uptake and endosomal escape, interaction of carriers with blood components determine not only transport but eventually also efficiency. Thus, the behavior in plasma represents a critical obstacle for the delivery system. Depending on their physicochemical properties, including size, zeta potential and surface modification, certain proteins will adsorb on their surface to form a “protein corona”. This protein corona largely determines the characteristics of the particles in the organism, i.e., biodistribution, pharmacokinetics and immunogenicity. It was observed that the adsorption of plasma proteins leads to reduced accessibility or interaction between exogenously incorporated ligands and their targeted receptors (Fig. 5). For example, several studies reported that transferrin-coated nanoparticles showed reduced or lacking specificity towards TfR in presence of the protein corona as a result of ligand blockade [260-262]. The resulting “biological identity” was highly dependent on the composition of the protein corona. In particular, in vitro protein corona depleted targeting capability, whereas a protein corona resembling in vivo conditions caused only a reduction in specific receptor-mediated uptake [261].
Fig. 5.
Interaction of i.v. injected targeted nanoparticles with blood components and consequences for the delivery process. Formation of protein corona leads to reduced intended targeting ability due to masked ligands. Protein corona may lead to transport to other cells or uptake via receptors recognizing plasma proteins. Additionally, endosomal escape can be hampered by protein layer. Created with BioRender.com
Interestingly, two studies could prove that the in vivo protein corona even enhanced the overall uptake of the nanoparticles, hypothesizing redirection of cellular uptake by the protein layer and opening new paths for particle internalization [262, 263]. As a consequence, plasma protein adsorption could also lead to redirected transport to off-target cells such as the RES, which hampers selective nucleic acid delivery and efficacy. In addition, Tonigold et al. observed that antibodies covalently bound to nanoparticles lose their targeting ability almost completely in presence of serum. Particles with physically adsorbed antibodies, however, maintained their targeting ability, probably due to the fact that these antibodies were not completely exchanged or masked by serum proteins [85].
Traditionally, masking the surface properties causing plasma protein adsorption is a key strategy to overcome this issue and to maintain targeting ability. PEGylation has proven to reduce undesired interactions and consequently retain ligand-accessibility which is essential for targeted delivery in vivo [41]. One approach to further attenuate particle interaction with plasma proteins is PEG “backfilling” (Fig. 6A). Therefore, the surface of gold nanoparticles (Au-NPs), functionalized with targeting transferrin-PEG(5 kDa) was further modified at free reaction sites with PEG spacers of varying lengths, preventing the particles from protein corona formation. In order to enable receptor-recognition, PEG chains for shielding had to be shorter than the spacer between particle and targeting ligand [264]. Despite these difficulties, there are promising examples which have already demonstrated efficient, cell- or tissue-specific delivery of nucleic acids. Additionally, scientists have started to exploit the protein corona to tune particle distribution in vivo. It is commonly known that plasma proteins adsorption depends on particle surface characteristics. Hence, slight structural changes of the particle surface have shown to affect the protein corona composition. As a consequence of protein corona modification, transport to target cells by coating with certain plasma proteins which serve as endogenous ligands can be obtained in vivo. For example, DOTAP/DNA lipoplexes have shown to adsorb vitronectin which mediated receptor-dependent uptake by tumor cells expressing αVβ3 integrins [265]. Patisiran is another prominent example for targeted transport to hepatocytes, mediated by coating with endogenous ApoE [104, 137]. Manipulation of the interaction with receptors in vivo and therefore targeted delivery to other cell types could be achieved by an altered protein corona as a consequence of exchanging certain lipid components of the Patisiran formulation [146]. Additionally, Saunders et al. used “nanoprimers” administered shortly before injection of therapeutic LNPs that were taken up by cells of the hepatic RES (Fig. 6B). By inhibiting KCs and LSECs, LNPs could be preferentially delivered to hepatocytes, the desired target site [266].
Fig. 6.
Strategies to improve or maintain targeting ability in vivo. (A) PEG-Backfilling avoids formation of protein corona, which would mask targeting ligands. Short PEG chains are necessary to maintain accessibility of the ligands [264]. (B) Application of a nanoprimer is reducing off-target LNP uptake by Kupffer cells and LSECs and enhances delivery to hepatocytes. [266]. Reproduced with permission from reference with Copyright © 2020, American Chemical Society. (C) Adjustment of surface chemistry leads to modified protein corona composition and can be used for targeted delivery by altered biodistribution profile. Created with BioRender.com
Based on these observations, methods which enable fast screening of broad libraries of nucleic acid carriers in vivo were developed [106-109]. By means of that, evaluation and characterization of predominant plasma proteins in the corona and tuning the biodistribution profile based on facile structural variations becomes feasible, pathing the way for improved targeted, cell-specific nucleic acid delivery in vivo (see Fig. 6C) [110, 142-145].
Conclusion
Cell-specific delivery, especially for in vivo applications, remains a central challenge for the development of new nucleic acid therapeutics. Tremendous efforts were put into the optimization of existing delivery systems as well as in the development of new carriers due to numerous barriers that have to be overcome for efficient delivery and activity of the nucleic acid therapeutic. Receptor-mediated and chemical targeting strategies represent key approaches for targeted delivery and improved performance of synthetic carriers and have proven their ability for enhanced transfection efficacy in the desired cell type. Receptor-mediated nucleic acid delivery can be highly specific towards certain cell types (e.g., ASGPR) and even allows receptor-mediated transport across internal barriers (e.g., BBB). New, high-affinity ligands for specific delivery are discovered continuously and can be conjugated to synthetic carriers in numerous ways. Chemical targeting achieved by structural alterations of the particle components has also shown great potential for promoting cell-specific nucleic acid delivery. As a result, shifted biodistribution profiles were observed which enabled delivery to desired cells. In addition, new technologies as the barcoding method combined with high throughput processes could path the way for future applications, readily adjustable formulations and a deeper understanding of in vivo performance.
Funding
Open Access funding enabled and organized by Projekt DEAL. Funding of the work by the German Research Foundation (DFG) via the Collaborative Research Centre SFB 1032 sub-project B4 and by the UPGRADE (Unlocking Precision Gene Therapy) project from the European Union’s Horizon 2020 research and innovation programme under grant agreement No 825825.
Declarations
Conflict of Interest
The authors have no conflicts of interest to declare.
Footnotes
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
References
- 1.Gene Therapy Clinical Trials Worldwide. 09.05.2022. Available from: https://a873679.fmphost.com/fmi/webd/GTCT.
- 2.Mulligan RC. The basic science of gene therapy. Science. 1993;260(5110):926–932. doi: 10.1126/science.8493530. [DOI] [PubMed] [Google Scholar]
- 3.Friedmann T, Roblin R. Gene therapy for human genetic disease? Science. 1972;175(4025):949–955. doi: 10.1126/science.175.4025.949. [DOI] [PubMed] [Google Scholar]
- 4.Lundstrom K. Viral Vectors in Gene Therapy. Diseases. 2018;6(2). [DOI] [PMC free article] [PubMed]
- 5.Bulcha JT, Wang Y, Ma H, Tai PWL, Gao G. Viral vector platforms within the gene therapy landscape. Signal Transduct Target Ther. 2021;6(1):53. doi: 10.1038/s41392-021-00487-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Vaheri A, Pagano JS. Infectious poliovirus RNA: a sensitive method of assay 2. Virology. 1965;27(3):434–436. doi: 10.1016/0042-6822(65)90126-1. [DOI] [PubMed] [Google Scholar]
- 7.McCutchan JH, Pagano JS. Enchancement of the infectivity of simian virus 40 deoxyribonucleic acid with diethylaminoethyl-dextran. J Natl Cancer Inst. 1968;41(2):351–357. [PubMed] [Google Scholar]
- 8.Lopata MA, Cleveland DW, Sollner WB. High level transient expression of a chloramphenicol acetyl transferase gene by DEAE-dextran mediated DNA transfection coupled with a dimethyl sulfoxide or glycerol shock treatment. Nucleic Acids Res. 1984;12(14):5707–5717. doi: 10.1093/nar/12.14.5707. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Yamamoto A, Kormann M, Rosenecker J, Rudolph C. Current prospects for mRNA gene delivery. Eur J Pharm Biopharm. 2009;71(3):484–489. doi: 10.1016/j.ejpb.2008.09.016. [DOI] [PubMed] [Google Scholar]
- 10.Kowalski PS, Rudra A, Miao L, Anderson DG. Delivering the Messenger: Advances in Technologies for Therapeutic mRNA Delivery. Mol Ther. 2019;27(4):710–728. doi: 10.1016/j.ymthe.2019.02.012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Sahin U, Muik A, Derhovanessian E, Vogler I, Kranz LM, Vormehr M, Baum A, Pascal K, Quandt J, Maurus D, Brachtendorf S, Lorks V, Sikorski J, Hilker R, Becker D, Eller AK, Grutzner J, Boesler C, Rosenbaum C, Kuhnle MC, Luxemburger U, Kemmer-Bruck A, Langer D, Bexon M, Bolte S, Kariko K, Palanche T, Fischer B, Schultz A, Shi PY, Fontes-Garfias C, Perez JL, Swanson KA, Loschko J, Scully IL, Cutler M, Kalina W, Kyratsous CA, Cooper D, Dormitzer PR, Jansen KU, Tureci O. COVID-19 vaccine BNT162b1 elicits human antibody and TH1 T cell responses. Nature. 2020;586(7830):594–599. doi: 10.1038/s41586-020-2814-7. [DOI] [PubMed] [Google Scholar]
- 12.Baden LR, El Sahly HM, Essink B, Kotloff K, Frey S, Novak R, Diemert D, Spector SA, Rouphael N, Creech CB, McGettigan J, Khetan S, Segall N, Solis J, Brosz A, Fierro C, Schwartz H, Neuzil K, Corey L, Gilbert P, Janes H, Follmann D, Marovich M, Mascola J, Polakowski L, Ledgerwood J, Graham BS, Bennett H, Pajon R, Knightly C, Leav B, Deng W, Zhou H, Han S, Ivarsson M, Miller J, Zaks T, Group CS Efficacy and Safety of the mRNA-1273 SARS-CoV-2 Vaccine. N Engl J Med. 2021;384(5):403–416. doi: 10.1056/NEJMoa2035389. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Schoenmaker L, Witzigmann D, Kulkarni JA, Verbeke R, Kersten G, Jiskoot W, Crommelin DJA. mRNA-lipid nanoparticle COVID-19 vaccines: Structure and stability. Int J Pharm. 2021;601:120586. doi: 10.1016/j.ijpharm.2021.120586. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Elbashir SM, Harborth J, Lendeckel W, Yalcin A, Weber K, Tuschl T. Duplexes of 21-nucleotide RNAs mediate RNA interference in cultured mammalian cells. Nature. 2001;411(6836):494–498. doi: 10.1038/35078107. [DOI] [PubMed] [Google Scholar]
- 15.Zhang B, Farwell MA. microRNAs: a new emerging class of players for disease diagnostics and gene therapy. J Cell Mol Med. 2008;12(1):3–21. doi: 10.1111/j.1582-4934.2007.00196.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Bauman J, Jearawiriyapaisarn N, Kole R. Therapeutic potential of splice-switching oligonucleotides. Oligonucleotides. 2009;19(1):1–13. doi: 10.1089/oli.2008.0161. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Uhlmann EP, A. Antisense oligonucleotides: a new therapeutic principle. Chem Rev. 1990;90(4):543–584.
- 18.Doudna JA, Charpentier E. Genome editing. The new frontier of genome engineering with CRISPR-Cas9. Science. 2014;346(6213):1258096. [DOI] [PubMed]
- 19.Gillmore JD, Gane E, Taubel J, Kao J, Fontana M, Maitland ML, Seitzer J, O'Connell D, Walsh KR, Wood K, Phillips J, Xu Y, Amaral A, Boyd AP, Cehelsky JE, McKee MD, Schiermeier A, Harari O, Murphy A, Kyratsous CA, Zambrowicz B, Soltys R, Gutstein DE, Leonard J, Sepp-Lorenzino L, Lebwohl D. CRISPR-Cas9 In Vivo Gene Editing for Transthyretin Amyloidosis. N Engl J Med. 2021;385(6):493–502. doi: 10.1056/NEJMoa2107454. [DOI] [PubMed] [Google Scholar]
- 20.Lin Y, Wagner E, Lachelt U. Non-viral delivery of the CRISPR/Cas system: DNA versus RNA versus RNP. Biomater Sci. 2022;10(5):1166–1192. doi: 10.1039/d1bm01658j. [DOI] [PubMed] [Google Scholar]
- 21.Zuber G, Dauty E, Nothisen M, Belguise P, Behr JP. Towards synthetic viruses. AdvDrug DelivRev. 2001;52(3):245–253. doi: 10.1016/s0169-409x(01)00213-7. [DOI] [PubMed] [Google Scholar]
- 22.Wagner E. Strategies to improve DNA polyplexes for in vivo gene transfer: will "artificial viruses" be the answer? PharmRes. 2004;21(1):8–14. doi: 10.1023/b:pham.0000012146.04068.56. [DOI] [PubMed] [Google Scholar]
- 23.Wagner E, Ogris M, Zauner W. Polylysine-based transfection systems utilizing receptor-mediated delivery. Adv Drug Deliv Rev. 1998;30(1–3):97–113. doi: 10.1016/s0169-409x(97)00110-5. [DOI] [PubMed] [Google Scholar]
- 24.Wu GY, Wu CH. Receptor-mediated in vitro gene transformation by a soluble DNA carrier system. J Biol Chem. 1987;262(10):4429–4432. [PubMed] [Google Scholar]
- 25.Boussif O, Lezoualc'h F, Zanta MA, Mergny MD, Scherman D, Demeneix B, Behr JP. A versatile vector for gene and oligonucleotide transfer into cells in culture and in vivo: polyethylenimine. Proc Natl Acad Sci U S A. 1995;92(16):7297–7301. doi: 10.1073/pnas.92.16.7297. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Wightman L, Kircheis R, Rossler V, Carotta S, Ruzicka R, Kursa M, Wagner E. Different behavior of branched and linear polyethylenimine for gene delivery in vitro and in vivo. J Gene Med. 2001;3(4):362–372. doi: 10.1002/jgm.187. [DOI] [PubMed] [Google Scholar]
- 27.Dufes C, Uchegbu IF, Schatzlein AG. Dendrimers in gene delivery. Adv Drug Deliv Rev. 2005;57(15):2177–2202. doi: 10.1016/j.addr.2005.09.017. [DOI] [PubMed] [Google Scholar]
- 28.Zhou J, Wu J, Hafdi N, Behr JP, Erbacher P, Peng L. PAMAM dendrimers for efficient siRNA delivery and potent gene silencing. Chem Commun (Camb) 2006;22:2362–2364. doi: 10.1039/b601381c. [DOI] [PubMed] [Google Scholar]
- 29.Felgner PL, Barenholz Y, Behr JP, Cheng SH, Cullis P, Huang L, Jessee JA, Seymour L, Szoka F, Thierry AR, Wagner E, Wu G. Nomenclature for synthetic gene delivery systems. Hum Gene Ther. 1997;8(5):511–512. doi: 10.1089/hum.1997.8.5-511. [DOI] [PubMed] [Google Scholar]
- 30.Kulkarni JA, Witzigmann D, Chen S, Cullis PR, van der Meel R. Lipid Nanoparticle Technology for Clinical Translation of siRNA Therapeutics. Acc Chem Res. 2019;52(9):2435–2444. doi: 10.1021/acs.accounts.9b00368. [DOI] [PubMed] [Google Scholar]
- 31.Wu GY, Wu CH. Receptor-mediated gene delivery and expression in vivo. J Biol Chem. 1988;263(29):14621–14624. [PubMed] [Google Scholar]
- 32.Vetter VC, Wagner E. Targeting nucleic acid-based therapeutics to tumors: Challenges and strategies for polyplexes. J Control Release. 2022;346:110–135. doi: 10.1016/j.jconrel.2022.04.013. [DOI] [PubMed] [Google Scholar]
- 33.Vaughan HJ, Green JJ, Tzeng SY. Cancer-Targeting Nanoparticles for Combinatorial Nucleic Acid Delivery. Adv Mater. 2020;32(13):e1901081. doi: 10.1002/adma.201901081. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Xiao Y, Shi K, Qu Y, Chu B, Qian Z. Engineering Nanoparticles for Targeted Delivery of Nucleic Acid Therapeutics in Tumor. Mol Ther Methods Clin Dev. 2019;12:1–18. doi: 10.1016/j.omtm.2018.09.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Bareford LM, Swaan PW. Endocytic mechanisms for targeted drug delivery. Adv Drug Deliv Rev. 2007;59(8):748–758. doi: 10.1016/j.addr.2007.06.008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Nakamura Y, Kogure K, Futaki S, Harashima H. Octaarginine-modified multifunctional envelope-type nano device for siRNA. J Control Release. 2007;119(3):360–367. doi: 10.1016/j.jconrel.2007.03.010. [DOI] [PubMed] [Google Scholar]
- 37.Kawaguchi Y, Takeuchi T, Kuwata K, Chiba J, Hatanaka Y, Nakase I, Futaki S. Syndecan-4 Is a Receptor for Clathrin-Mediated Endocytosis of Arginine-Rich Cell-Penetrating Peptides. Bioconjug Chem. 2016;27(4):1119–1130. doi: 10.1021/acs.bioconjchem.6b00082. [DOI] [PubMed] [Google Scholar]
- 38.Bros M, Nuhn L, Simon J, Moll L, Mailander V, Landfester K, Grabbe S. The Protein Corona as a Confounding Variable of Nanoparticle-Mediated Targeted Vaccine Delivery. Front Immunol. 2018;9:1760. doi: 10.3389/fimmu.2018.01760. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Berger SB, M.; Bantz, C.; Maskos, M.; Wagner, E. Performance of nanoparticles for biomedical applications: The in vitro/in vivo discrepancy. Biophysics Reviews. 2022;3(1). [DOI] [PMC free article] [PubMed]
- 40.Francia V, Schiffelers RM, Cullis PR, Witzigmann D. The Biomolecular Corona of Lipid Nanoparticles for Gene Therapy. Bioconjug Chem. 2020;31(9):2046–2059. doi: 10.1021/acs.bioconjchem.0c00366. [DOI] [PubMed] [Google Scholar]
- 41.Ogris M, Brunner S, Schuller S, Kircheis R, Wagner E. PEGylated DNA/transferrin-PEI complexes: reduced interaction with blood components, extended circulation in blood and potential for systemic gene delivery. Gene Ther. 1999;6(4):595–605. doi: 10.1038/sj.gt.3300900. [DOI] [PubMed] [Google Scholar]
- 42.Johnson RN, Chu DS, Shi J, Schellinger JG, Carlson PM, Pun SH. HPMA-oligolysine copolymers for gene delivery: optimization of peptide length and polymer molecular weight. J Control Release. 2011;155(2):303–311. doi: 10.1016/j.jconrel.2011.07.009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Manzenrieder F, Luxenhofer R, Retzlaff M, Jordan R, Finn MG. Stabilization of virus-like particles with poly(2-oxazoline)s. Angew Chem Int Ed Engl. 2011;50(11):2601–2605. doi: 10.1002/anie.201006134. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Klein PM, Klinker K, Zhang W, Kern S, Kessel E, Wagner E, Barz M. Efficient Shielding of Polyplexes Using Heterotelechelic Polysarcosines. Polymers (Basel). 2018;10(6). [DOI] [PMC free article] [PubMed]
- 45.Noga M, Edinger D, Rodl W, Wagner E, Winter G, Besheer A. Controlled shielding and deshielding of gene delivery polyplexes using hydroxyethyl starch (HES) and alpha-amylase. J Control Release. 2012;159(1):92–103. doi: 10.1016/j.jconrel.2012.01.006. [DOI] [PubMed] [Google Scholar]
- 46.Sato T, Nakata M, Yang Z, Torizuka Y, Kishimoto S, Ishihara M. In vitro and in vivo gene delivery using chitosan/hyaluronic acid nanoparticles: Influences of molecular mass of hyaluronic acid and lyophilization on transfection efficiency. J Gene Med. 2017;19(8). [DOI] [PubMed]
- 47.Luo J, Schmaus J, Cui M, Horterer E, Wilk U, Hohn M, Dather M, Berger S, Benli-Hoppe T, Peng L, Wagner E. Hyaluronate siRNA nanoparticles with positive charge display rapid attachment to tumor endothelium and penetration into tumors. J Control Release. 2021;329:919–933. doi: 10.1016/j.jconrel.2020.10.022. [DOI] [PubMed] [Google Scholar]
- 48.Hornof M, de la Fuente M, Hallikainen M, Tammi RH, Urtti A. Low molecular weight hyaluronan shielding of DNA/PEI polyplexes facilitates CD44 receptor mediated uptake in human corneal epithelial cells. J Gene Med. 2008;10(1):70–80. doi: 10.1002/jgm.1125. [DOI] [PubMed] [Google Scholar]
- 49.Ogris M, Wagner E. To be targeted: is the magic bullet concept a viable option for synthetic nucleic acid therapeutics? Hum Gene Ther. 2011;22(7):799–807. doi: 10.1089/hum.2011.065. [DOI] [PubMed] [Google Scholar]
- 50.Elfinger M, Geiger J, Hasenpusch G, Uzgun S, Sieverling N, Aneja MK, Maucksch C, Rudolph C. Targeting of the beta(2)-adrenoceptor increases nonviral gene delivery to pulmonary epithelial cells in vitro and lungs in vivo. J Control Release. 2009;135(3):234–241. doi: 10.1016/j.jconrel.2009.01.012. [DOI] [PubMed] [Google Scholar]
- 51.Luo Y, Zhai X, Ma C, Sun P, Fu Z, Liu W, Xu J. An inhalable beta(2)-adrenoceptor ligand-directed guanidinylated chitosan carrier for targeted delivery of siRNA to lung. J Control Release. 2012;162(1):28–36. doi: 10.1016/j.jconrel.2012.06.005. [DOI] [PubMed] [Google Scholar]
- 52.Geiger J, Aneja MK, Hasenpusch G, Yuksekdag G, Kummerlowe G, Luy B, Romer T, Rothbauer U, Rudolph C. Targeting of the prostacyclin specific IP1 receptor in lungs with molecular conjugates comprising prostaglandin I2 analogues. Biomaterials. 2010;31(10):2903–2911. doi: 10.1016/j.biomaterials.2009.12.035. [DOI] [PubMed] [Google Scholar]
- 53.Lachelt U, Wittmann V, Muller K, Edinger D, Kos P, Hohn M, Wagner E. Synthetic polyglutamylation of dual-functional MTX ligands for enhanced combined cytotoxicity of poly(I:C) nanoplexes. Mol Pharm. 2014;11(8):2631–2639. doi: 10.1021/mp500017u. [DOI] [PubMed] [Google Scholar]
- 54.Jenkins RG, Herrick SE, Meng QH, Kinnon C, Laurent GJ, McAnulty RJ, Hart SL. An integrin-targeted non-viral vector for pulmonary gene therapy. Gene Ther. 2000;7(5):393–400. doi: 10.1038/sj.gt.3301095. [DOI] [PubMed] [Google Scholar]
- 55.Benli-Hoppe T, Gol Ozturk S, Ozturk O, Berger S, Wagner E, Yazdi M. Transferrin Receptor Targeted Polyplexes Completely Comprised of Sequence-Defined Components. Macromol Rapid Commun. 2021:e2100602. [DOI] [PubMed]
- 56.Wagner E, Zenke M, Cotten M, Beug H, Birnstiel ML. Transferrin-polycation conjugates as carriers for DNA uptake into cells. Proc Natl Acad Sci U S A. 1990;87(9):3410–3414. doi: 10.1073/pnas.87.9.3410. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Zenke M, Steinlein P, Wagner E, Cotten M, Beug H, Birnstiel ML. Receptor-mediated endocytosis of transferrin-polycation conjugates: an efficient way to introduce DNA into hematopoietic cells. Proc Natl Acad Sci U S A. 1990;87(10):3655–3659. doi: 10.1073/pnas.87.10.3655. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Buschle M, Cotten M, Kirlappos H, Mechtler K, Schaffner G, Zauner W, Birnstiel ML, Wagner E. Receptor-mediated gene transfer into human T lymphocytes via binding of DNA/CD3 antibody particles to the CD3 T cell receptor complex. Hum Gene Ther. 1995;6(6):753–761. doi: 10.1089/hum.1995.6.6-753. [DOI] [PubMed] [Google Scholar]
- 59.Ramishetti S, Kedmi R, Goldsmith M, Leonard F, Sprague AG, Godin B, Gozin M, Cullis PR, Dykxhoorn DM, Peer D. Systemic Gene Silencing in Primary T Lymphocytes Using Targeted Lipid Nanoparticles. ACS Nano. 2015;9(7):6706–6716. doi: 10.1021/acsnano.5b02796. [DOI] [PubMed] [Google Scholar]
- 60.Plank C, Zatloukal K, Cotten M, Mechtler K, Wagner E. Gene transfer into hepatocytes using asialoglycoprotein receptor mediated endocytosis of DNA complexed with an artificial tetra-antennary galactose ligand. Bioconjug Chem. 1992;3(6):533–539. doi: 10.1021/bc00018a012. [DOI] [PubMed] [Google Scholar]
- 61.Uehara K, Harumoto T, Makino A, Koda Y, Iwano J, Suzuki Y, Tanigawa M, Iwai H, Asano K, Kurihara K, Hamaguchi A, Kodaira H, Atsumi T, Yamada Y, Tomizuka K. Targeted delivery to macrophages and dendritic cells by chemically modified mannose ligand-conjugated siRNA. Nucleic Acids Res. 2022. [DOI] [PMC free article] [PubMed]
- 62.Anraku Y, Kuwahara H, Fukusato Y, Mizoguchi A, Ishii T, Nitta K, Matsumoto Y, Toh K, Miyata K, Uchida S, Nishina K, Osada K, Itaka K, Nishiyama N, Mizusawa H, Yamasoba T, Yokota T, Kataoka K. Glycaemic control boosts glucosylated nanocarrier crossing the BBB into the brain. Nat Commun. 2017;8(1):1001. doi: 10.1038/s41467-017-00952-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63.Dohmen C, Frohlich T, Lachelt U, Rohl I, Vornlocher HP, Hadwiger P, Wagner E. Defined Folate-PEG-siRNA Conjugates for Receptor-specific Gene Silencing. Mol Ther Nucleic Acids. 2012;1:e7. doi: 10.1038/mtna.2011.10. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64.Sato Y, Murase K, Kato J, Kobune M, Sato T, Kawano Y, Takimoto R, Takada K, Miyanishi K, Matsunaga T, Takayama T, Niitsu Y. Resolution of liver cirrhosis using vitamin A-coupled liposomes to deliver siRNA against a collagen-specific chaperone. Nat Biotechnol. 2008;26(4):431–442. doi: 10.1038/nbt1396. [DOI] [PubMed] [Google Scholar]
- 65.Hopkins CR, Miller K, Beardmore JM. Receptor-mediated endocytosis of transferrin and epidermal growth factor receptors: a comparison of constitutive and ligand-induced uptake. J Cell Sci Suppl. 1985;3:173–186. doi: 10.1242/jcs.1985.supplement_3.17. [DOI] [PubMed] [Google Scholar]
- 66.Wall DA, Hubbard AL. Receptor-mediated endocytosis of asialoglycoproteins by rat liver hepatocytes: biochemical characterization of the endosomal compartments. J Cell Biol. 1985;101(6):2104–2112. doi: 10.1083/jcb.101.6.2104. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 67.Goldstein JL, Anderson RG, Brown MS. Coated pits, coated vesicles, and receptor-mediated endocytosis. Nature. 1979;279(5715):679–685. doi: 10.1038/279679a0. [DOI] [PubMed] [Google Scholar]
- 68.McMahon HT, Boucrot E. Molecular mechanism and physiological functions of clathrin-mediated endocytosis. Nat Rev Mol Cell Biol. 2011;12(8):517–533. doi: 10.1038/nrm3151. [DOI] [PubMed] [Google Scholar]
- 69.Rejman J, Oberle V, Zuhorn IS, Hoekstra D. Size-dependent internalization of particles via the pathways of clathrin- and caveolae-mediated endocytosis. Biochem J. 2004;377(Pt 1):159–169. doi: 10.1042/BJ20031253. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 70.Dauty E, Remy JS, Zuber G, Behr JP. Intracellular delivery of nanometric DNA particles via the folate receptor. Bioconjug Chem. 2002;13(4):831–839. doi: 10.1021/bc0255182. [DOI] [PubMed] [Google Scholar]
- 71.Lamaze C, Dujeancourt A, Baba T, Lo CG, Benmerah A, Dautry-Varsat A. Interleukin 2 receptors and detergent-resistant membrane domains define a clathrin-independent endocytic pathway. Mol Cell. 2001;7(3):661–671. doi: 10.1016/s1097-2765(01)00212-x. [DOI] [PubMed] [Google Scholar]
- 72.Pelkmans L, Helenius A. Endocytosis via caveolae. Traffic. 2002;3(5):311–320. doi: 10.1034/j.1600-0854.2002.30501.x. [DOI] [PubMed] [Google Scholar]
- 73.Nichols BJ. A distinct class of endosome mediates clathrin-independent endocytosis to the Golgi complex. Nat Cell Biol. 2002;4(5):374–378. doi: 10.1038/ncb787. [DOI] [PubMed] [Google Scholar]
- 74.Akinc A, Thomas M, Klibanov AM, Langer R. Exploring polyethylenimine-mediated DNA transfection and the proton sponge hypothesis. J Gene Med. 2005;7(5):657–663. doi: 10.1002/jgm.696. [DOI] [PubMed] [Google Scholar]
- 75.Lachelt U, Kos P, Mickler FM, Herrmann A, Salcher EE, Rodl W, Badgujar N, Brauchle C, Wagner E. Fine-tuning of proton sponges by precise diaminoethanes and histidines in pDNA polyplexes. Nanomedicine. 2014;10(1):35–44. doi: 10.1016/j.nano.2013.07.008. [DOI] [PubMed] [Google Scholar]
- 76.Cullis PR, Hope MJ. Lipid Nanoparticle Systems for Enabling Gene Therapies. Mol Ther. 2017;25(7):1467–1475. doi: 10.1016/j.ymthe.2017.03.013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 77.Blessing T, Kursa M, Holzhauser R, Kircheis R, Wagner E. Different strategies for formation of pegylated EGF-conjugated PEI/DNA complexes for targeted gene delivery. Bioconjug Chem. 2001;12(4):529–537. doi: 10.1021/bc0001488. [DOI] [PubMed] [Google Scholar]
- 78.Gestwicki JE, Cairo CW, Strong LE, Oetjen KA, Kiessling LL. Influencing receptor-ligand binding mechanisms with multivalent ligand architecture. J Am Chem Soc. 2002;124(50):14922–14933. doi: 10.1021/ja027184x. [DOI] [PubMed] [Google Scholar]
- 79.Fasting C, Schalley CA, Weber M, Seitz O, Hecht S, Koksch B, Dernedde J, Graf C, Knapp EW, Haag R. Multivalency as a chemical organization and action principle. Angew Chem Int Ed Engl. 2012;51(42):10472–10498. doi: 10.1002/anie.201201114. [DOI] [PubMed] [Google Scholar]
- 80.Wagner E, Cotten M, Foisner R, Birnstiel ML. Transferrin-polycation-DNA complexes: the effect of polycations on the structure of the complex and DNA delivery to cells. Proc Natl Acad Sci U S A. 1991;88(10):4255–4259. doi: 10.1073/pnas.88.10.4255. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 81.Elfinger M, Maucksch C, Rudolph C. Characterization of lactoferrin as a targeting ligand for nonviral gene delivery to airway epithelial cells. Biomaterials. 2007;28(23):3448–3455. doi: 10.1016/j.biomaterials.2007.04.011. [DOI] [PubMed] [Google Scholar]
- 82.Diebold SS, Kursa M, Wagner E, Cotten M, Zenke M. Mannose polyethylenimine conjugates for targeted DNA delivery into dendritic cells. J Biol Chem. 1999;274(27):19087–19094. doi: 10.1074/jbc.274.27.19087. [DOI] [PubMed] [Google Scholar]
- 83.Jia Z, Gong Y, Pi Y, Liu X, Gao L, Kang L, Wang J, Yang F, Tang J, Lu W, Li Q, Zhang W, Yan Z, Yu L. pPB Peptide-Mediated siRNA-Loaded Stable Nucleic Acid Lipid Nanoparticles on Targeting Therapy of Hepatic Fibrosis. Mol Pharm. 2018;15(1):53–62. doi: 10.1021/acs.molpharmaceut.7b00709. [DOI] [PubMed] [Google Scholar]
- 84.Parhiz H, Shuvaev VV, Pardi N, Khoshnejad M, Kiseleva RY, Brenner JS, Uhler T, Tuyishime S, Mui BL, Tam YK, Madden TD, Hope MJ, Weissman D, Muzykantov VR. PECAM-1 directed re-targeting of exogenous mRNA providing two orders of magnitude enhancement of vascular delivery and expression in lungs independent of apolipoprotein E-mediated uptake. J Control Release. 2018;291:106–115. doi: 10.1016/j.jconrel.2018.10.015. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 85.Tonigold M, Simon J, Estupinan D, Kokkinopoulou M, Reinholz J, Kintzel U, Kaltbeitzel A, Renz P, Domogalla MP, Steinbrink K, Lieberwirth I, Crespy D, Landfester K, Mailander V. Pre-adsorption of antibodies enables targeting of nanocarriers despite a biomolecular corona. Nat Nanotechnol. 2018;13(9):862–869. doi: 10.1038/s41565-018-0171-6. [DOI] [PubMed] [Google Scholar]
- 86.York AW, Zhang Y, Holley AC, Guo Y, Huang F, McCormick CL. Facile synthesis of multivalent folate-block copolymer conjugates via aqueous RAFT polymerization: targeted delivery of siRNA and subsequent gene suppression. Biomacromol. 2009;10(4):936–943. doi: 10.1021/bm8014768. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 87.Kakizawa Y, Kataoka K. Block copolymer micelles for delivery of gene and related compounds. Adv Drug Deliv Rev. 2002;54(2):203–222. doi: 10.1016/s0169-409x(02)00017-0. [DOI] [PubMed] [Google Scholar]
- 88.Klein PM, Wagner E. Click-Shielded and Targeted Lipopolyplexes. Methods Mol Biol. 2019;2036:141–164. doi: 10.1007/978-1-4939-9670-4_8. [DOI] [PubMed] [Google Scholar]
- 89.Klein PM, Kern S, Lee DJ, Schmaus J, Hohn M, Gorges J, Kazmaier U, Wagner E. Folate receptor-directed orthogonal click-functionalization of siRNA lipopolyplexes for tumor cell killing in vivo. Biomaterials. 2018;178:630–642. doi: 10.1016/j.biomaterials.2018.03.031. [DOI] [PubMed] [Google Scholar]
- 90.Glass EB, Masjedi S, Dudzinski SO, Wilson AJ, Duvall CL, Yull FE, Giorgio TD. Optimizing Mannose "Click" Conjugation to Polymeric Nanoparticles for Targeted siRNA Delivery to Human and Murine Macrophages. ACS Omega. 2019;4(16):16756–16767. doi: 10.1021/acsomega.9b01465. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 91.Elfinger M, Pfeifer C, Uezguen S, Golas MM, Sander B, Maucksch C, Stark H, Aneja MK, Rudolph C. Self-assembly of ternary insulin-polyethylenimine (PEI)-DNA nanoparticles for enhanced gene delivery and expression in alveolar epithelial cells. Biomacromol. 2009;10(10):2912–2920. doi: 10.1021/bm900707j. [DOI] [PubMed] [Google Scholar]
- 92.Nie Y, Schaffert D, Rodl W, Ogris M, Wagner E, Gunther M. Dual-targeted polyplexes: one step towards a synthetic virus for cancer gene therapy. J Control Release. 2011;152(1):127–134. doi: 10.1016/j.jconrel.2011.02.028. [DOI] [PubMed] [Google Scholar]
- 93.Kos P, Lachelt U, He D, Nie Y, Gu Z, Wagner E. Dual-targeted polyplexes based on sequence-defined peptide-PEG-oligoamino amides. J Pharm Sci. 2015;104(2):464–475. doi: 10.1002/jps.24194. [DOI] [PubMed] [Google Scholar]
- 94.Urnauer S, Schmohl KA, Tutter M, Schug C, Schwenk N, Morys S, Ziegler S, Bartenstein P, Clevert DA, Wagner E, Spitzweg C. Dual-targeted NIS polyplexes-a theranostic strategy toward tumors with heterogeneous receptor expression. Gene Ther. 2019;26(3–4):93–108. doi: 10.1038/s41434-019-0059-6. [DOI] [PubMed] [Google Scholar]
- 95.Wang S, Reinhard S, Li C, Qian M, Jiang H, Du Y, Lachelt U, Lu W, Wagner E, Huang R. Antitumoral Cascade-Targeting Ligand for IL-6 Receptor-Mediated Gene Delivery to Glioma. Mol Ther. 2017;25(7):1556–1566. doi: 10.1016/j.ymthe.2017.04.023. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 96.Zhang C, Gu Z, Shen L, Liu X, Lin H. A Dual Targeting Drug Delivery System for Penetrating Blood-Brain Barrier and Selectively Delivering siRNA to Neurons for Alzheimer's Disease Treatment. Curr Pharm Biotechnol. 2017;18(14):1124–1131. doi: 10.2174/1389201019666180226152542. [DOI] [PubMed] [Google Scholar]
- 97.Zhang C, Gu Z, Shen L, Liu X, Lin H. In vivo Evaluation and Alzheimer's Disease Treatment Outcome of siRNA Loaded Dual Targeting Drug Delivery System. Curr Pharm Biotechnol. 2019;20(1):56–62. doi: 10.2174/1389201020666190204141046. [DOI] [PubMed] [Google Scholar]
- 98.Mislick KA, Baldeschwieler JD. Evidence for the role of proteoglycans in cation-mediated gene transfer. Proc Natl Acad Sci U S A. 1996;93(22):12349–12354. doi: 10.1073/pnas.93.22.12349. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 99.Kopatz I, Remy JS, Behr JP. A model for non-viral gene delivery: through syndecan adhesion molecules and powered by actin. J Gene Med. 2004;6(7):769–776. doi: 10.1002/jgm.558. [DOI] [PubMed] [Google Scholar]
- 100.Hafez IM, Maurer N, Cullis PR. On the mechanism whereby cationic lipids promote intracellular delivery of polynucleic acids. Gene Ther. 2001;8(15):1188–1196. doi: 10.1038/sj.gt.3301506. [DOI] [PubMed] [Google Scholar]
- 101.Bisgaier CL, Siebenkas MV, Williams KJ. Effects of Apolipoproteins A-IV and A-I on the Uptake of Phospholipid Liposomes by Hepatocytes. J Biol Chem. 1989;264(2):862–866. [PubMed] [Google Scholar]
- 102.Cullis PR, Chonn A, Semple SC. Interactions of liposomes and lipid-based carrier systems with blood proteins: Relation to clearance behaviour in vivo. Adv Drug Deliv Rev. 1998;32(1–2):3–17. doi: 10.1016/s0169-409x(97)00128-2. [DOI] [PubMed] [Google Scholar]
- 103.Yan X, Kuipers F, Havekes LM, Havinga R, Dontje B, Poelstra K, Scherphof GL, Kamps JA. The role of apolipoprotein E in the elimination of liposomes from blood by hepatocytes in the mouse. Biochem Biophys Res Commun. 2005;328(1):57–62. doi: 10.1016/j.bbrc.2004.12.137. [DOI] [PubMed] [Google Scholar]
- 104.Akinc A, Querbes W, De S, Qin J, Frank-Kamenetsky M, Jayaprakash KN, Jayaraman M, Rajeev KG, Cantley WL, Dorkin JR, Butler JS, Qin L, Racie T, Sprague A, Fava E, Zeigerer A, Hope MJ, Zerial M, Sah DW, Fitzgerald K, Tracy MA, Manoharan M, Koteliansky V, Fougerolles A, Maier MA. Targeted delivery of RNAi therapeutics with endogenous and exogenous ligand-based mechanisms. Mol Ther. 2010;18(7):1357–1364. doi: 10.1038/mt.2010.85. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 105.Ritz S, Schottler S, Kotman N, Baier G, Musyanovych A, Kuharev J, Landfester K, Schild H, Jahn O, Tenzer S, Mailander V. Protein corona of nanoparticles: distinct proteins regulate the cellular uptake. Biomacromol. 2015;16(4):1311–1321. doi: 10.1021/acs.biomac.5b00108. [DOI] [PubMed] [Google Scholar]
- 106.Kowalski PS, Capasso Palmiero U, Huang Y, Rudra A, Langer R, Anderson DG. Ionizable Amino-Polyesters Synthesized via Ring Opening Polymerization of Tertiary Amino-Alcohols for Tissue Selective mRNA Delivery. Adv Mater. 2018:e1801151. [DOI] [PMC free article] [PubMed]
- 107.Dahlman JE, Kauffman KJ, Xing Y, Shaw TE, Mir FF, Dlott CC, Langer R, Anderson DG, Wang ET. Barcoded nanoparticles for high throughput in vivo discovery of targeted therapeutics. Proc Natl Acad Sci U S A. 2017;114(8):2060–2065. doi: 10.1073/pnas.1620874114. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 108.Cheng Q, Wei T, Farbiak L, Johnson LT, Dilliard SA, Siegwart DJ. Selective organ targeting (SORT) nanoparticles for tissue-specific mRNA delivery and CRISPR-Cas gene editing. Nat Nanotechnol. 2020;15(4):313–320. doi: 10.1038/s41565-020-0669-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 109.Liu S, Cheng Q, Wei T, Yu X, Johnson LT, Farbiak L, Siegwart DJ. Membrane-destabilizing ionizable phospholipids for organ-selective mRNA delivery and CRISPR-Cas gene editing. Nat Mater. 2021;20(5):701–710. doi: 10.1038/s41563-020-00886-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 110.Dilliard SA, Cheng Q, Siegwart DJ. On the mechanism of tissue-specific mRNA delivery by selective organ targeting nanoparticles. Proc Natl Acad Sci U S A. 2021;118(52). [DOI] [PMC free article] [PubMed]
- 111.Lee K, Jang B, Lee YR, Suh EY, Yoo JS, Lee MJ, Lee JY, Lee H. The cutting-edge technologies of siRNA delivery and their application in clinical trials. Arch Pharm Res. 2018;41(9):867–874. doi: 10.1007/s12272-018-1069-4. [DOI] [PubMed] [Google Scholar]
- 112.Witzigmann D, Kulkarni JA, Leung J, Chen S, Cullis PR, van der Meel R. Lipid nanoparticle technology for therapeutic gene regulation in the liver. Adv Drug Deliv Rev. 2020;159:344–363. doi: 10.1016/j.addr.2020.06.026. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 113.Nienhuis AW, Nathwani AC, Davidoff AM. Gene Therapy for Hemophilia. Hum Gene Ther. 2016;27(4):305–308. doi: 10.1089/hum.2016.018. [DOI] [PubMed] [Google Scholar]
- 114.Asrani SK, Devarbhavi H, Eaton J, Kamath PS. Burden of liver diseases in the world. J Hepatol. 2019;70(1):151–171. doi: 10.1016/j.jhep.2018.09.014. [DOI] [PubMed] [Google Scholar]
- 115.Ashwell G, Morell AG. The role of surface carbohydrates in the hepatic recognition and transport of circulating glycoproteins. Adv Enzymol Relat Areas Mol Biol. 1974;41:99–128. doi: 10.1002/9780470122860.ch3. [DOI] [PubMed] [Google Scholar]
- 116.Baenziger JU, Fiete D. Galactose and N-acetylgalactosamine-specific endocytosis of glycopeptides by isolated rat hepatocytes. Cell. 1980;22(2 Pt 2):611–620. doi: 10.1016/0092-8674(80)90371-2. [DOI] [PubMed] [Google Scholar]
- 117.Lee RT, Lee YC. Affinity enhancement by multivalent lectin-carbohydrate interaction. Glycoconj J. 2000;17(7–9):543–551. doi: 10.1023/a:1011070425430. [DOI] [PubMed] [Google Scholar]
- 118.Pricer WE, Ashwell G. The Binding of Desialylated Glycoproteins by Plasma Membranes of Rat Liver. J Biol Chem. 1971;246(15):4825–4833. [PubMed] [Google Scholar]
- 119.Huang X, Leroux JC, Castagner B. Well-Defined Multivalent Ligands for Hepatocytes Targeting via Asialoglycoprotein Receptor. Bioconjug Chem. 2017;28(2):283–295. doi: 10.1021/acs.bioconjchem.6b00651. [DOI] [PubMed] [Google Scholar]
- 120.Rozema DB, Lewis DL, Wakefield DH, Wong SC, Klein JJ, Roesch PL, Bertin SL, Reppen TW, Chu Q, Blokhin AV, Hagstrom JE, Wolff JA. Dynamic PolyConjugates for targeted in vivo delivery of siRNA to hepatocytes. Proc Natl Acad Sci U S A. 2007;104(32):12982–12987. doi: 10.1073/pnas.0703778104. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 121.Nair JK, Willoughby JL, Chan A, Charisse K, Alam MR, Wang Q, Hoekstra M, Kandasamy P, Kel'in AV, Milstein S, Taneja N, O'Shea J, Shaikh S, Zhang L, van der Sluis RJ, Jung ME, Akinc A, Hutabarat R, Kuchimanchi S, Fitzgerald K, Zimmermann T, van Berkel TJ, Maier MA, Rajeev KG, Manoharan M. Multivalent N-acetylgalactosamine-conjugated siRNA localizes in hepatocytes and elicits robust RNAi-mediated gene silencing. J Am Chem Soc. 2014;136(49):16958–16961. doi: 10.1021/ja505986a. [DOI] [PubMed] [Google Scholar]
- 122.Balwani M, Sardh E, Ventura P, Peiro PA, Rees DC, Stolzel U, Bissell DM, Bonkovsky HL, Windyga J, Anderson KE, Parker C, Silver SM, Keel SB, Wang JD, Stein PE, Harper P, Vassiliou D, Wang B, Phillips J, Ivanova A, Langendonk JG, Kauppinen R, Minder E, Horie Y, Penz C, Chen J, Liu S, Ko JJ, Sweetser MT, Garg P, Vaishnaw A, Kim JB, Simon AR, Gouya L, Investigators E. Phase 3 Trial of RNAi Therapeutic Givosiran for Acute Intermittent Porphyria. N Engl J Med. 2020;382(24):2289–2301. doi: 10.1056/NEJMoa1913147. [DOI] [PubMed] [Google Scholar]
- 123.Goma-Garces E, Perez-Gomez MV, Ortiz A. Givosiran for Acute Intermittent Porphyria. N Engl J Med. 2020;383(20):1989. doi: 10.1056/NEJMc2026458. [DOI] [PubMed] [Google Scholar]
- 124.Garrelfs SF, Frishberg Y, Hulton SA, Koren MJ, O'Riordan WD, Cochat P, Deschenes G, Shasha-Lavsky H, Saland JM, Van't Hoff WG, Fuster DG, Magen D, Moochhala SH, Schalk G, Simkova E, Groothoff JW, Sas DJ, Meliambro KA, Lu J, Sweetser MT, Garg PP, Vaishnaw AK, Gansner JM, McGregor TL, Lieske JC, Collaborators I-A. Lumasiran, an RNAi Therapeutic for Primary Hyperoxaluria Type 1. N Engl J Med. 2021;384(13):1216–1226. doi: 10.1056/NEJMoa2021712. [DOI] [PubMed] [Google Scholar]
- 125.Scott LJ, Keam SJ. Lumasiran: First Approval. Drugs. 2021;81(2):277–282. doi: 10.1007/s40265-020-01463-0. [DOI] [PubMed] [Google Scholar]
- 126.Frank-Kamenetsky M, Grefhorst A, Anderson NN, Racie TS, Bramlage B, Akinc A, Butler D, Charisse K, Dorkin R, Fan Y, Gamba-Vitalo C, Hadwiger P, Jayaraman M, John M, Jayaprakash KN, Maier M, Nechev L, Rajeev KG, Read T, Rohl I, Soutschek J, Tan P, Wong J, Wang G, Zimmermann T, de Fougerolles A, Vornlocher HP, Langer R, Anderson DG, Manoharan M, Koteliansky V, Horton JD, Fitzgerald K. Therapeutic RNAi targeting PCSK9 acutely lowers plasma cholesterol in rodents and LDL cholesterol in nonhuman primates. Proc Natl Acad Sci U S A. 2008;105(33):11915–11920. doi: 10.1073/pnas.0805434105. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 127.Fitzgerald K, White S, Borodovsky A, Bettencourt BR, Strahs A, Clausen V, Wijngaard P, Horton JD, Taubel J, Brooks A, Fernando C, Kauffman RS, Kallend D, Vaishnaw A, Simon A. A Highly Durable RNAi Therapeutic Inhibitor of PCSK9. N Engl J Med. 2017;376(1):41–51. doi: 10.1056/NEJMoa1609243. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 128.Lamb YN. Inclisiran: First Approval. Drugs. 2021;81(3):389–395. doi: 10.1007/s40265-021-01473-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 129.Zhang MM, Bahal R, Rasmussen TP, Manautou JE, Zhong XB. The growth of siRNA-based therapeutics: Updated clinical studies. Biochem Pharmacol. 2021;189:114432. doi: 10.1016/j.bcp.2021.114432. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 130.Prakash TP, Graham MJ, Yu J, Carty R, Low A, Chappell A, Schmidt K, Zhao C, Aghajan M, Murray HF, Riney S, Booten SL, Murray SF, Gaus H, Crosby J, Lima WF, Guo S, Monia BP, Swayze EE, Seth PP. Targeted delivery of antisense oligonucleotides to hepatocytes using triantennary N-acetyl galactosamine improves potency 10-fold in mice. Nucleic Acids Res. 2014;42(13):8796–8807. [DOI] [PMC free article] [PubMed]
- 131.Watanabe A, Nakajima M, Kasuya T, Onishi R, Kitade N, Mayumi K, Ikehara T, Kugimiya A. Comparative Characterization of Hepatic Distribution and mRNA Reduction of Antisense Oligonucleotides Conjugated with Triantennary N-Acetyl Galactosamine and Lipophilic Ligands Targeting Apolipoprotein B. J Pharmacol Exp Ther. 2016;357(2):320–330. doi: 10.1124/jpet.115.230300. [DOI] [PubMed] [Google Scholar]
- 132.Yamamoto T, Mukai Y, Wada F, Terada C, Kayaba Y, Oh K, Yamayoshi A, Obika S, Harada-Shiba M. Highly Potent GalNAc-Conjugated Tiny LNA Anti-miRNA-122 Antisense Oligonucleotides. Pharmaceutics. 2021;13(6). [DOI] [PMC free article] [PubMed]
- 133.Crooke ST, Baker BF, Xia S, Yu RZ, Viney NJ, Wang Y, Tsimikas S, Geary RS. Integrated Assessment of the Clinical Performance of GalNAc3-Conjugated 2'-O-Methoxyethyl Chimeric Antisense Oligonucleotides: I. Human Volunteer Experience Nucleic Acid Ther. 2019;29(1):16–32. doi: 10.1089/nat.2018.0753. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 134.Zanardi TA, Korbmacher B, Boone L, Engelhardt JA, Wang Y, Burel S, Prill B, Aghajan M, Guo S, Henry SP. Safety, Pharmacokinetic, and Pharmacodynamic Evaluation of a 2'-(2-Methoxyethyl)-D-ribose Antisense Oligonucleotide-Triantenarry N-Acetyl-galactosamine Conjugate that Targets the Human Transmembrane Protease Serine 6. J Pharmacol Exp Ther. 2021;377(1):51–63. doi: 10.1124/jpet.120.000222. [DOI] [PubMed] [Google Scholar]
- 135.Rouet R, Thuma BA, Roy MD, Lintner NG, Rubitski DM, Finley JE, Wisniewska HM, Mendonsa R, Hirsh A, de Onate L, Compte Barron J, McLellan TJ, Bellenger J, Feng X, Varghese A, Chrunyk BA, Borzilleri K, Hesp KD, Zhou K, Ma N, Tu M, Dullea R, McClure KF, Wilson RC, Liras S, Mascitti V, Doudna JA. Receptor-Mediated Delivery of CRISPR-Cas9 Endonuclease for Cell-Type-Specific Gene Editing. J Am Chem Soc. 2018;140(21):6596–6603. doi: 10.1021/jacs.8b01551. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 136.Soutschek J, Akinc A, Bramlage B, Charisse K, Constien R, Donoghue M, Elbashir S, Geick A, Hadwiger P, Harborth J, John M, Kesavan V, Lavine G, Pandey RK, Racie T, Rajeev KG, Rohl I, Toudjarska I, Wang G, Wuschko S, Bumcrot D, Koteliansky V, Limmer S, Manoharan M, Vornlocher HP. Therapeutic silencing of an endogenous gene by systemic administration of modified siRNAs. Nature. 2004;432(7014):173–178. doi: 10.1038/nature03121. [DOI] [PubMed] [Google Scholar]
- 137.Adams D, Gonzalez-Duarte A, O'Riordan WD, Yang CC, Ueda M, Kristen AV, Tournev I, Schmidt HH, Coelho T, Berk JL, Lin KP, Vita G, Attarian S, Plante-Bordeneuve V, Mezei MM, Campistol JM, Buades J, Brannagan TH, 3rd, Kim BJ, Oh J, Parman Y, Sekijima Y, Hawkins PN, Solomon SD, Polydefkis M, Dyck PJ, Gandhi PJ, Goyal S, Chen J, Strahs AL, Nochur SV, Sweetser MT, Garg PP, Vaishnaw AK, Gollob JA, Suhr OB. Patisiran, an RNAi Therapeutic, for Hereditary Transthyretin Amyloidosis. N Engl J Med. 2018;379(1):11–21. doi: 10.1056/NEJMoa1716153. [DOI] [PubMed] [Google Scholar]
- 138.Coelho T, Adams D, Silva A, Lozeron P, Hawkins PN, Mant T, Perez J, Chiesa J, Warrington S, Tranter E, Munisamy M, Falzone R, Harrop J, Cehelsky J, Bettencourt BR, Geissler M, Butler JS, Sehgal A, Meyers RE, Chen Q, Borland T, Hutabarat RM, Clausen VA, Alvarez R, Fitzgerald K, Gamba-Vitalo C, Nochur SV, Vaishnaw AK, Sah DW, Gollob JA, Suhr OB. Safety and efficacy of RNAi therapy for transthyretin amyloidosis. N Engl J Med. 2013;369(9):819–829. doi: 10.1056/NEJMoa1208760. [DOI] [PubMed] [Google Scholar]
- 139.Chen SL, Zheng MH, Shi KQ, Yang T, Chen YP. A new strategy for treatment of liver fibrosis: letting MicroRNAs do the job. BioDrugs. 2013;27(1):25–34. doi: 10.1007/s40259-012-0005-2. [DOI] [PubMed] [Google Scholar]
- 140.Omar R, Yang J, Liu H, Davies NM, Gong Y. Hepatic Stellate Cells in Liver Fibrosis and siRNA-Based Therapy. Rev Physiol Biochem Pharmacol. 2016;172:1–37. doi: 10.1007/112_2016_6. [DOI] [PubMed] [Google Scholar]
- 141.Sato Y, Yoneda A, Shimizu F, Nishimura M, Shimoyama R, Tashiro Y, Kurata W, Niitsu Y. Resolution of fibrosis by siRNA HSP47 in vitamin A-coupled liposomes induces regeneration of chronically injured livers. J Gastroenterol Hepatol. 2021;36(12):3418–3428. doi: 10.1111/jgh.15587. [DOI] [PubMed] [Google Scholar]
- 142.Sago CD, Krupczak BR, Lokugamage MP, Gan Z, Dahlman JE. Cell Subtypes Within the Liver Microenvironment Differentially Interact with Lipid Nanoparticles. Cell Mol Bioeng. 2019;12(5):389–397. doi: 10.1007/s12195-019-00573-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 143.Paunovska K, Gil CJ, Lokugamage MP, Sago CD, Sato M, Lando GN, Gamboa Castro M, Bryksin AV, Dahlman JE. Analyzing 2000 in Vivo Drug Delivery Data Points Reveals Cholesterol Structure Impacts Nanoparticle Delivery. ACS Nano. 2018;12(8):8341–8349. doi: 10.1021/acsnano.8b03640. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 144.Paunovska K, Da Silva Sanchez AJ, Sago CD, Gan Z, Lokugamage MP, Islam FZ, Kalathoor S, Krupczak BR, Dahlman JE. Nanoparticles Containing Oxidized Cholesterol Deliver mRNA to the Liver Microenvironment at Clinically Relevant Doses. Adv Mater. 2019;31(14):e1807748. doi: 10.1002/adma.201807748. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 145.Gan Z, Lokugamage MP, Hatit MZC, Loughrey D, Paunovska K, Sato M, Cristian A, Dahlman JE. Nanoparticles containing constrained phospholipids deliver mRNA to liver immune cells in vivo without targeting ligands. Bioeng Transl Med. 2020;5(3):e10161. doi: 10.1002/btm2.10161. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 146.Pattipeiluhu R, Arias-Alpizar G, Basha G, Chan KYT, Bussmann J, Sharp TH, Moradi MA, Sommerdijk N, Harris EN, Cullis PR, Kros A, Witzigmann D, Campbell F. Anionic Lipid Nanoparticles Preferentially Deliver mRNA to the Hepatic Reticuloendothelial System. Adv Mater. 2022;34(16):e2201095. doi: 10.1002/adma.202201095. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 147.Freeley M, Long A. Advances in siRNA delivery to T-cells: potential clinical applications for inflammatory disease, cancer and infection. Biochem J. 2013;455(2):133–147. doi: 10.1042/BJ20130950. [DOI] [PubMed] [Google Scholar]
- 148.Granot-Matok Y, Kon E, Dammes N, Mechtinger G, Peer D. Therapeutic mRNA delivery to leukocytes. J Control Release. 2019;305:165–175. doi: 10.1016/j.jconrel.2019.05.032. [DOI] [PubMed] [Google Scholar]
- 149.Linehan SA, Martinez-Pomares L, Gordon S. Macrophage lectins in host defence. Microbes Infect. 2000;2(3):279–288. doi: 10.1016/s1286-4579(00)00300-2. [DOI] [PubMed] [Google Scholar]
- 150.Taylor ME, Bezouska K, Drickamer K. Contribution to ligand binding by multiple carbohydrate-recognition domains in the macrophage mannose receptor. J Biol Chem. 1992;267(3):1719–1726. [PubMed] [Google Scholar]
- 151.Erbacher P, Bousser MT, Raimond J, Monsigny M, Midoux P, Roche AC. Gene transfer by DNA/glycosylated polylysine complexes into human blood monocyte-derived macrophages. Hum Gene Ther. 1996;7(6):721–729. doi: 10.1089/hum.1996.7.6-721. [DOI] [PubMed] [Google Scholar]
- 152.Lopukhov AV, Yang Z, Haney MJ, Bronich TK, Sokolsky-Papkov M, Batrakova EV, Klyachko NL, Kabanov AV. Mannosylated Cationic Copolymers for Gene Delivery to Macrophages. Macromol Biosci. 2021;21(4):e2000371. doi: 10.1002/mabi.202000371. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 153.Leber N, Kaps L, Yang A, Aslam M, Giardino M, Klefenz A, Choteschovsky N, Rosigkeit S, Mostafa A, Nuhn L, Schuppan D, Zentel R. alpha-Mannosyl-Functionalized Cationic Nanohydrogel Particles for Targeted Gene Knockdown in Immunosuppressive Macrophages. Macromol Biosci. 2019;19(7):e1900162. doi: 10.1002/mabi.201900162. [DOI] [PubMed] [Google Scholar]
- 154.Kaps L, Leber N, Klefenz A, Choteschovsky N, Zentel R, Nuhn L, Schuppan D. In Vivo siRNA Delivery to Immunosuppressive Liver Macrophages by alpha-Mannosyl-Functionalized Cationic Nanohydrogel Particles. Cells. 2020;9(8). [DOI] [PMC free article] [PubMed]
- 155.Gao H, Goncalves C, Gallego T, Francois-Heude M, Malard V, Mateo V, Lemoine F, Cendret V, Djedaini-Pilard F, Moreau V, Pichon C, Midoux P. Comparative binding and uptake of liposomes decorated with mannose oligosaccharides by cells expressing the mannose receptor or DC-SIGN. Carbohydr Res. 2020;487:107877. doi: 10.1016/j.carres.2019.107877. [DOI] [PubMed] [Google Scholar]
- 156.Wang F, Xiao W, Elbahnasawy MA, Bao X, Zheng Q, Gong L, Zhou Y, Yang S, Fang A, Farag MMS, Wu J, Song X. Optimization of the Linker Length of Mannose-Cholesterol Conjugates for Enhanced mRNA Delivery to Dendritic Cells by Liposomes. Front Pharmacol. 2018;9:980. doi: 10.3389/fphar.2018.00980. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 157.Goswami R, O'Hagan DT, Adamo R, Baudner BC. Conjugation of Mannans to Enhance the Potency of Liposome Nanoparticles for the Delivery of RNA Vaccines. Pharmaceutics. 2021;13(2). [DOI] [PMC free article] [PubMed]
- 158.East L, Isacke CM. The mannose receptor family. Biochim Biophys Acta. 2002;1572(2–3):364–386. doi: 10.1016/s0304-4165(02)00319-7. [DOI] [PubMed] [Google Scholar]
- 159.Inaba K, Swiggard WJ, Inaba M, Meltzer J, Mirza A, Sasagawa T, Nussenzweig MC, Steinman RM. Tissue distribution of the DEC-205 protein that is detected by the monoclonal antibody NLDC-145. I. Expression on dendritic cells and other subsets of mouse leukocytes. Cell Immunol. 1995;163(1):148–156. [DOI] [PubMed]
- 160.Katakowski JA, Mukherjee G, Wilner SE, Maier KE, Harrison MT, DiLorenzo TP, Levy M, Palliser D. Delivery of siRNAs to Dendritic Cells Using DEC205-Targeted Lipid Nanoparticles to Inhibit Immune Responses. Mol Ther. 2016;24(1):146–155. doi: 10.1038/mt.2015.175. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 161.Kranz LM, Diken M, Haas H, Kreiter S, Loquai C, Reuter KC, Meng M, Fritz D, Vascotto F, Hefesha H, Grunwitz C, Vormehr M, Husemann Y, Selmi A, Kuhn AN, Buck J, Derhovanessian E, Rae R, Attig S, Diekmann J, Jabulowsky RA, Heesch S, Hassel J, Langguth P, Grabbe S, Huber C, Tureci O, Sahin U. Systemic RNA delivery to dendritic cells exploits antiviral defence for cancer immunotherapy. Nature. 2016;534(7607):396–401. doi: 10.1038/nature18300. [DOI] [PubMed] [Google Scholar]
- 162.Fabbri M, Smart C, Pardi R. T lymphocytes. Int J Biochem Cell Biol. 2003;35(7):1004–1008. doi: 10.1016/s1357-2725(03)00037-2. [DOI] [PubMed] [Google Scholar]
- 163.Peer D. Induction of therapeutic gene silencing in leukocyte-implicated diseases by targeted and stabilized nanoparticles: a mini-review. J Control Release. 2010;148(1):63–68. doi: 10.1016/j.jconrel.2010.06.029. [DOI] [PubMed] [Google Scholar]
- 164.Veiga N, Goldsmith M, Granot Y, Rosenblum D, Dammes N, Kedmi R, Ramishetti S, Peer D. Cell specific delivery of modified mRNA expressing therapeutic proteins to leukocytes. Nat Commun. 2018;9(1):4493. doi: 10.1038/s41467-018-06936-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 165.Kedmi R, Veiga N, Ramishetti S, Goldsmith M, Rosenblum D, Dammes N, Hazan-Halevy I, Nahary L, Leviatan-Ben-Arye S, Harlev M, Behlke M, Benhar I, Lieberman J, Peer D. A modular platform for targeted RNAi therapeutics. Nat Nanotechnol. 2018;13(3):214–219. doi: 10.1038/s41565-017-0043-5. [DOI] [PubMed] [Google Scholar]
- 166.Lokugamage MP, Sago CD, Gan Z, Krupczak BR, Dahlman JE. Constrained Nanoparticles Deliver siRNA and sgRNA to T Cells In Vivo without Targeting Ligands. Adv Mater. 2019;31(41):e1902251. doi: 10.1002/adma.201902251. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 167.Ramishetti S, Hazan-Halevy I, Palakuri R, Chatterjee S, Naidu Gonna S, Dammes N, Freilich I, Kolik Shmuel L, Danino D, Peer D. A Combinatorial Library of Lipid Nanoparticles for RNA Delivery to Leukocytes. Adv Mater. 2020;32(12):e1906128. doi: 10.1002/adma.201906128. [DOI] [PubMed] [Google Scholar]
- 168.Merkel OM, Zheng M, Debus H, Kissel T. Pulmonary gene delivery using polymeric nonviral vectors. Bioconjug Chem. 2012;23(1):3–20. doi: 10.1021/bc200296q. [DOI] [PubMed] [Google Scholar]
- 169.McLean JW, Fox EA, Baluk P, Bolton PB, Haskell A, Pearlman R, Thurston G, Umemoto EY, McDonald DM. Organ-specific endothelial cell uptake of cationic liposome-DNA complexes in mice. Am J Physiol. 1997;273(1 Pt 2):H387–404. doi: 10.1152/ajpheart.1997.273.1.H387. [DOI] [PubMed] [Google Scholar]
- 170.Zou SM, Erbacher P, Remy JS, Behr JP. Systemic linear polyethylenimine (L-PEI)-mediated gene delivery in the mouse. JGene Med. 2000;2(2):128–134. doi: 10.1002/(SICI)1521-2254(200003/04)2:2<128::AID-JGM95>3.0.CO;2-W. [DOI] [PubMed] [Google Scholar]
- 171.Goula D, Benoist C, Mantero S, Merlo G, Levi G, Demeneix BA. Polyethylenimine-based intravenous delivery of transgenes to mouse lung. Gene Ther. 1998;5(9):1291–1295. doi: 10.1038/sj.gt.3300717. [DOI] [PubMed] [Google Scholar]
- 172.Bragonzi A, Dina G, Villa A, Calori G, Biffi A, Bordignon C, Assael BM, Conese M. Biodistribution and transgene expression with nonviral cationic vector/DNA complexes in the lungs. Gene Ther. 2000;7(20):1753–1760. doi: 10.1038/sj.gt.3301282. [DOI] [PubMed] [Google Scholar]
- 173.Kaczmarek JC, Patel AK, Kauffman KJ, Fenton OS, Webber MJ, Heartlein MW, DeRosa F, Anderson DG. Polymer-Lipid Nanoparticles for Systemic Delivery of mRNA to the Lungs. Angew Chem Int Ed Engl. 2016;55(44):13808–13812. doi: 10.1002/anie.201608450. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 174.Kaczmarek JC, Kauffman KJ, Fenton OS, Sadtler K, Patel AK, Heartlein MW, DeRosa F, Anderson DG. Optimization of a Degradable Polymer-Lipid Nanoparticle for Potent Systemic Delivery of mRNA to the Lung Endothelium and Immune Cells. Nano Lett. 2018;18(10):6449–6454. doi: 10.1021/acs.nanolett.8b02917. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 175.Kaczmarek JC, Patel AK, Rhym LH, Palmiero UC, Bhat B, Heartlein MW, DeRosa F, Anderson DG. Systemic delivery of mRNA and DNA to the lung using polymer-lipid nanoparticles. Biomaterials. 2021;275:120966. doi: 10.1016/j.biomaterials.2021.120966. [DOI] [PubMed] [Google Scholar]
- 176.Renigunta A, Krasteva G, Konig P, Rose F, Klepetko W, Grimminger F, Seeger W, Hanze J. DNA transfer into human lung cells is improved with Tat-RGD peptide by caveoli-mediated endocytosis. Bioconjug Chem. 2006;17(2):327–334. doi: 10.1021/bc050263o. [DOI] [PubMed] [Google Scholar]
- 177.Scott ES, Wiseman JW, Evans MJ, Colledge WH. Enhanced gene delivery to human airway epithelial cells using an integrin-targeting lipoplex. J Gene Med. 2001;3(2):125–134. doi: 10.1002/jgm.172. [DOI] [PubMed] [Google Scholar]
- 178.Ferkol T, Kaetzel CS, Davis PB. Gene transfer into respiratory epithelial cells by targeting the polymeric immunoglobulin receptor. J Clin Invest. 1993;92(5):2394–2400. doi: 10.1172/JCI116845. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 179.Li S, Tan Y, Viroonchatapan E, Pitt BR, Huang L. Targeted gene delivery to pulmonary endothelium by anti-PECAM antibody. Am J Physiol Lung Cell Mol Physiol. 2000;278(3):L504–511. doi: 10.1152/ajplung.2000.278.3.L504. [DOI] [PubMed] [Google Scholar]
- 180.Xie Y, Kim NH, Nadithe V, Schalk D, Thakur A, Kilic A, Lum LG, Bassett DJP, Merkel OM. Targeted delivery of siRNA to activated T cells via transferrin-polyethylenimine (Tf-PEI) as a potential therapy of asthma. J Control Release. 2016;229:120–129. doi: 10.1016/j.jconrel.2016.03.029. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 181.Kandil R, Xie Y, Heermann R, Isert L, Jung K, Mehta A, Merkel OM. Coming in and Finding Out: Blending Receptor-Targeted Delivery and Efficient Endosomal Escape in a Novel Bio-Responsive siRNA Delivery System for Gene Knockdown in Pulmonary T Cells. Adv Ther (Weinh). 2019;2(7). [DOI] [PMC free article] [PubMed]
- 182.Cheng Y, Yumul RC, Pun SH. Virus-Inspired Polymer for Efficient In Vitro and In Vivo Gene Delivery. Angew Chem Int Ed Engl. 2016;55(39):12013–12017. doi: 10.1002/anie.201605958. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 183.Feldmann DP, Cheng Y, Kandil R, Xie Y, Mohammadi M, Harz H, Sharma A, Peeler DJ, Moszczynska A, Leonhardt H, Pun SH, Merkel OM. In vitro and in vivo delivery of siRNA via VIPER polymer system to lung cells. J Control Release. 2018;276:50–58. doi: 10.1016/j.jconrel.2018.02.017. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 184.Baldassi D, Ambike S, Feuerherd M, Cheng CC, Peeler DJ, Feldmann DP, Porras-Gonzalez DL, Wei X, Keller LA, Kneidinger N, Stoleriu MG, Popp A, Burgstaller G, Pun SH, Michler T, Merkel OM. Inhibition of SARS-CoV-2 replication in the lung with siRNA/VIPER polyplexes. J Control Release. 2022;345:661–674. doi: 10.1016/j.jconrel.2022.03.051. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 185.Kollen WJ, Midoux P, Erbacher P, Yip A, Roche AC, Monsigny M, Glick MC, Scanlin TF. Gluconoylated and glycosylated polylysines as vectors for gene transfer into cystic fibrosis airway epithelial cells. Hum Gene Ther. 1996;7(13):1577–1586. doi: 10.1089/hum.1996.7.13-1577. [DOI] [PubMed] [Google Scholar]
- 186.Kollen W, Erbacher P, Midoux P, Roche AC, Monsigny M, Glick MC, Scanlin TF. Glycosylated polylysines. Nonviral vectors for gene transfer into cystic fibrosis airway epithelial cells. Chest. 1997;111(6 Suppl):95S-96S. [DOI] [PubMed]
- 187.Kollen WJ, Mulberg AE, Wei X, Sugita M, Raghuram V, Wang J, Foskett JK, Glick MC, Scanlin TF. High-efficiency transfer of cystic fibrosis transmembrane conductance regulator cDNA into cystic fibrosis airway cells in culture using lactosylated polylysine as a vector. Hum Gene Ther. 1999;10(4):615–622. doi: 10.1089/10430349950018689. [DOI] [PubMed] [Google Scholar]
- 188.Kollen WJ, Schembri FM, Gerwig GJ, Vliegenthart JF, Glick MC, Scanlin TF. Enhanced efficiency of lactosylated poly-L-lysine-mediated gene transfer into cystic fibrosis airway epithelial cells. Am J Respir Cell Mol Biol. 1999;20(5):1081–1086. doi: 10.1165/ajrcmb.20.5.3417. [DOI] [PubMed] [Google Scholar]
- 189.Chen J, Gao X, Hu K, Pang Z, Cai J, Li J, Wu H, Jiang X. Galactose-poly(ethylene glycol)-polyethylenimine for improved lung gene transfer. Biochem Biophys Res Commun. 2008;375(3):378–383. doi: 10.1016/j.bbrc.2008.08.006. [DOI] [PubMed] [Google Scholar]
- 190.Pardridge WM. Blood-brain barrier delivery. Drug Discov Today. 2007;12(1–2):54–61. doi: 10.1016/j.drudis.2006.10.013. [DOI] [PubMed] [Google Scholar]
- 191.Descamps L, Dehouck MP, Torpier G, Cecchelli R. Receptor-mediated transcytosis of transferrin through blood-brain barrier endothelial cells. Am J Physiol. 1996;270(4 Pt 2):H1149–1158. doi: 10.1152/ajpheart.1996.270.4.H1149. [DOI] [PubMed] [Google Scholar]
- 192.Mills E, Dong XP, Wang F, Xu H. Mechanisms of brain iron transport: insight into neurodegeneration and CNS disorders. Future Med Chem. 2010;2(1):51–64. doi: 10.4155/fmc.09.140. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 193.Cardoso AL, Simoes S, de Almeida LP, Plesnila N, Pedroso de Lima MC, Wagner E, Culmsee C. Tf-lipoplexes for neuronal siRNA delivery: a promising system to mediate gene silencing in the CNS. J Control Release. 2008;132(2):113–123. [DOI] [PubMed]
- 194.Huang RQ, Qu YH, Ke WL, Zhu JH, Pei YY, Jiang C. Efficient gene delivery targeted to the brain using a transferrin-conjugated polyethyleneglycol-modified polyamidoamine dendrimer. FASEB J. 2007;21(4):1117–1125. doi: 10.1096/fj.06-7380com. [DOI] [PubMed] [Google Scholar]
- 195.Somani S, Blatchford DR, Millington O, Stevenson ML, Dufes C. Transferrin-bearing polypropylenimine dendrimer for targeted gene delivery to the brain. J Control Release. 2014;188:78–86. doi: 10.1016/j.jconrel.2014.06.006. [DOI] [PubMed] [Google Scholar]
- 196.Zhang Y, Boado RJ, Pardridge WM. In vivo knockdown of gene expression in brain cancer with intravenous RNAi in adult rats. J Gene Med. 2003;5(12):1039–1045. doi: 10.1002/jgm.449. [DOI] [PubMed] [Google Scholar]
- 197.Prades R, Oller-Salvia B, Schwarzmaier SM, Selva J, Moros M, Balbi M, Grazu V, de La Fuente JM, Egea G, Plesnila N, Teixido M, Giralt E. Applying the retro-enantio approach to obtain a peptide capable of overcoming the blood-brain barrier. Angew Chem Int Ed Engl. 2015;54(13):3967–3972. doi: 10.1002/anie.201411408. [DOI] [PubMed] [Google Scholar]
- 198.Huang RQ, Ke WL, Qu YH, Zhu JH, Pei YY, Jiang C. Characterization of lactoferrin receptor in brain endothelial capillary cells and mouse brain. J Biomed Sci. 2007;14(1):121–128. doi: 10.1007/s11373-006-9121-7. [DOI] [PubMed] [Google Scholar]
- 199.Huang R, Ke W, Liu Y, Jiang C, Pei Y. The use of lactoferrin as a ligand for targeting the polyamidoamine-based gene delivery system to the brain. Biomaterials. 2008;29(2):238–246. doi: 10.1016/j.biomaterials.2007.09.024. [DOI] [PubMed] [Google Scholar]
- 200.Somani S, Robb G, Pickard BS, Dufes C. Enhanced gene expression in the brain following intravenous administration of lactoferrin-bearing polypropylenimine dendriplex. J Control Release. 2015;217:235–242. doi: 10.1016/j.jconrel.2015.09.003. [DOI] [PubMed] [Google Scholar]
- 201.Singh CSB, Eyford BA, Abraham T, Munro L, Choi KB, Okon M, Vitalis TZ, Gabathuler R, Lu CJ, Pfeifer CG, Tian MM, Jefferies WA. Discovery of a Highly Conserved Peptide in the Iron Transporter Melanotransferrin that Traverses an Intact Blood Brain Barrier and Localizes in Neural Cells. Front Neurosci. 2021;15:596976. doi: 10.3389/fnins.2021.596976. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 202.Eyford BA, Singh CSB, Abraham T, Munro L, Choi KB, Hill T, Hildebrandt R, Welch I, Vitalis TZ, Gabathuler R, Gordon JA, Adomat H, Guns EST, Lu CJ, Pfeifer CG, Tian MM, Jefferies WA. A Nanomule Peptide Carrier Delivers siRNA Across the Intact Blood-Brain Barrier to Attenuate Ischemic Stroke. Front Mol Biosci. 2021;8:611367. doi: 10.3389/fmolb.2021.611367. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 203.Demeule M, Regina A, Che C, Poirier J, Nguyen T, Gabathuler R, Castaigne JP, Beliveau R. Identification and design of peptides as a new drug delivery system for the brain. J Pharmacol Exp Ther. 2008;324(3):1064–1072. doi: 10.1124/jpet.107.131318. [DOI] [PubMed] [Google Scholar]
- 204.Demeule M, Currie JC, Bertrand Y, Che C, Nguyen T, Regina A, Gabathuler R, Castaigne JP, Beliveau R. Involvement of the low-density lipoprotein receptor-related protein in the transcytosis of the brain delivery vector angiopep-2. J Neurochem. 2008;106(4):1534–1544. doi: 10.1111/j.1471-4159.2008.05492.x. [DOI] [PubMed] [Google Scholar]
- 205.Ke W, Shao K, Huang R, Han L, Liu Y, Li J, Kuang Y, Ye L, Lou J, Jiang C. Gene delivery targeted to the brain using an Angiopep-conjugated polyethyleneglycol-modified polyamidoamine dendrimer. Biomaterials. 2009;30(36):6976–6985. doi: 10.1016/j.biomaterials.2009.08.049. [DOI] [PubMed] [Google Scholar]
- 206.Bruun J, Larsen TB, Jolck RI, Eliasen R, Holm R, Gjetting T, Andresen TL. Investigation of enzyme-sensitive lipid nanoparticles for delivery of siRNA to blood-brain barrier and glioma cells. Int J Nanomedicine. 2015;10:5995–6008. doi: 10.2147/IJN.S87334. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 207.Sakamoto K, Shinohara T, Adachi Y, Asami T, Ohtaki T. A novel LRP1-binding peptide L57 that crosses the blood brain barrier. Biochem Biophys Rep. 2017;12:135–139. doi: 10.1016/j.bbrep.2017.07.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 208.Rodrigues JP, Prajapati N, DeCoster MA, Poh S, Murray TA. Efficient LRP1-Mediated Uptake and Low Cytotoxicity of Peptide L57 In Vitro Shows Its Promise as CNS Drug Delivery Vector. J Pharm Sci. 2021;110(2):824–832. doi: 10.1016/j.xphs.2020.09.019. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 209.Golden PL, Maccagnan TJ, Pardridge WM. Human blood-brain barrier leptin receptor. Binding and endocytosis in isolated human brain microvessels. J Clin Invest. 1997;99(1):14–18. [DOI] [PMC free article] [PubMed]
- 210.Liu Y, Li J, Shao K, Huang R, Ye L, Lou J, Jiang C. A leptin derived 30-amino-acid peptide modified pegylated poly-L-lysine dendrigraft for brain targeted gene delivery. Biomaterials. 2010;31(19):5246–5257. doi: 10.1016/j.biomaterials.2010.03.011. [DOI] [PubMed] [Google Scholar]
- 211.Huey R, Hawthorne S, McCarron P. The potential use of rabies virus glycoprotein-derived peptides to facilitate drug delivery into the central nervous system: a mini review. J Drug Target. 2017;25(5):379–385. doi: 10.1080/1061186X.2016.1223676. [DOI] [PubMed] [Google Scholar]
- 212.Kumar P, Wu H, McBride JL, Jung KE, Kim MH, Davidson BL, Lee SK, Shankar P, Manjunath N. Transvascular delivery of small interfering RNA to the central nervous system. Nature. 2007;448(7149):39–43. doi: 10.1038/nature05901. [DOI] [PubMed] [Google Scholar]
- 213.Liu Y, Huang R, Han L, Ke W, Shao K, Ye L, Lou J, Jiang C. Brain-targeting gene delivery and cellular internalization mechanisms for modified rabies virus glycoprotein RVG29 nanoparticles. Biomaterials. 2009;30(25):4195–4202. doi: 10.1016/j.biomaterials.2009.02.051. [DOI] [PubMed] [Google Scholar]
- 214.Gao Y, Wang ZY, Zhang J, Zhang Y, Huo H, Wang T, Jiang T, Wang S. RVG-peptide-linked trimethylated chitosan for delivery of siRNA to the brain. Biomacromol. 2014;15(3):1010–1018. doi: 10.1021/bm401906p. [DOI] [PubMed] [Google Scholar]
- 215.Park TE, Singh B, Li H, Lee JY, Kang SK, Choi YJ, Cho CS. Enhanced BBB permeability of osmotically active poly(mannitol-co-PEI) modified with rabies virus glycoprotein via selective stimulation of caveolar endocytosis for RNAi therapeutics in Alzheimer's disease. Biomaterials. 2015;38:61–71. doi: 10.1016/j.biomaterials.2014.10.068. [DOI] [PubMed] [Google Scholar]
- 216.Alvarez-Erviti L, Seow Y, Yin H, Betts C, Lakhal S, Wood MJ. Delivery of siRNA to the mouse brain by systemic injection of targeted exosomes. Nat Biotechnol. 2011;29(4):341–345. doi: 10.1038/nbt.1807. [DOI] [PubMed] [Google Scholar]
- 217.Conceicao M, Mendonca L, Nobrega C, Gomes C, Costa P, Hirai H, Moreira JN, Lima MC, Manjunath N, Pereira de Almeida L. Intravenous administration of brain-targeted stable nucleic acid lipid particles alleviates Machado-Joseph disease neurological phenotype. Biomaterials. 2016;82:124–137. [DOI] [PubMed]
- 218.Liu Y, He X, Kuang Y, An S, Wang C, Guo Y, Ma H, Lou J, Jiang C. A bacteria deriving peptide modified dendrigraft poly-l-lysines (DGL) self-assembling nanoplatform for targeted gene delivery. Mol Pharm. 2014;11(10):3330–3341. doi: 10.1021/mp500084s. [DOI] [PubMed] [Google Scholar]
- 219.Marcos-Contreras OA, Greineder CF, Kiseleva RY, Parhiz H, Walsh LR, Zuluaga-Ramirez V, Myerson JW, Hood ED, Villa CH, Tombacz I, Pardi N, Seliga A, Mui BL, Tam YK, Glassman PM, Shuvaev VV, Nong J, Brenner JS, Khoshnejad M, Madden T, Weissmann D, Persidsky Y, Muzykantov VR. Selective targeting of nanomedicine to inflamed cerebral vasculature to enhance the blood-brain barrier. Proc Natl Acad Sci U S A. 2020;117(7):3405–3414. doi: 10.1073/pnas.1912012117. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 220.Shabanpoor F, Hammond SM, Abendroth F, Hazell G, Wood MJA, Gait MJ. Identification of a Peptide for Systemic Brain Delivery of a Morpholino Oligonucleotide in Mouse Models of Spinal Muscular Atrophy. Nucleic Acid Ther. 2017;27(3):130–143. doi: 10.1089/nat.2016.0652. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 221.Maher F, Vannucci SJ, Simpson IA. Glucose transporter proteins in brain. FASEB J. 1994;8(13):1003–1011. doi: 10.1096/fasebj.8.13.7926364. [DOI] [PubMed] [Google Scholar]
- 222.Suzuki K, Miura Y, Mochida Y, Miyazaki T, Toh K, Anraku Y, Melo V, Liu X, Ishii T, Nagano O, Saya H, Cabral H, Kataoka K. Glucose transporter 1-mediated vascular translocation of nanomedicines enhances accumulation and efficacy in solid tumors. J Control Release. 2019;301:28–41. doi: 10.1016/j.jconrel.2019.02.021. [DOI] [PubMed] [Google Scholar]
- 223.Min HS, Kim HJ, Naito M, Ogura S, Toh K, Hayashi K, Kim BS, Fukushima S, Anraku Y, Miyata K, Kataoka K. Systemic Brain Delivery of Antisense Oligonucleotides across the Blood-Brain Barrier with a Glucose-Coated Polymeric Nanocarrier. Angew Chem Int Ed Engl. 2020;59(21):8173–8180. doi: 10.1002/anie.201914751. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 224.Costantino L, Gandolfi F, Tosi G, Rivasi F, Vandelli MA, Forni F. Peptide-derivatized biodegradable nanoparticles able to cross the blood-brain barrier. J Control Release. 2005;108(1):84–96. doi: 10.1016/j.jconrel.2005.07.013. [DOI] [PubMed] [Google Scholar]
- 225.Tosi G, Fano RA, Bondioli L, Badiali L, Benassi R, Rivasi F, Ruozi B, Forni F, Vandelli MA. Investigation on mechanisms of glycopeptide nanoparticles for drug delivery across the blood-brain barrier. Nanomedicine (Lond) 2011;6(3):423–436. doi: 10.2217/nnm.11.11. [DOI] [PubMed] [Google Scholar]
- 226.Tosi G, Vergoni AV, Ruozi B, Bondioli L, Badiali L, Rivasi F, Costantino L, Forni F, Vandelli MA. Sialic acid and glycopeptides conjugated PLGA nanoparticles for central nervous system targeting: In vivo pharmacological evidence and biodistribution. J Control Release. 2010;145(1):49–57. doi: 10.1016/j.jconrel.2010.03.008. [DOI] [PubMed] [Google Scholar]
- 227.Rigon L, Salvalaio M, Pederzoli F, Legnini E, Duskey JT, D'Avanzo F, De Filippis C, Ruozi B, Marin O, Vandelli MA, Ottonelli I, Scarpa M, Tosi G, Tomanin R. Targeting Brain Disease in MPSII: Preclinical Evaluation of IDS-Loaded PLGA Nanoparticles. Int J Mol Sci. 2019;20(8). [DOI] [PMC free article] [PubMed]
- 228.Koirala A, Makkia RS, Conley SM, Cooper MJ, Naash MI. S/MAR-containing DNA nanoparticles promote persistent RPE gene expression and improvement in RPE65-associated LCA. Hum Mol Genet. 2013;22(8):1632–1642. doi: 10.1093/hmg/ddt013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 229.Koirala A, Conley SM, Makkia R, Liu Z, Cooper MJ, Sparrow JR, Naash MI. Persistence of non-viral vector mediated RPE65 expression: case for viability as a gene transfer therapy for RPE-based diseases. J Control Release. 2013;172(3):745–752. doi: 10.1016/j.jconrel.2013.08.299. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 230.Han Z, Koirala A, Makkia R, Cooper MJ, Naash MI. Direct gene transfer with compacted DNA nanoparticles in retinal pigment epithelial cells: expression, repeat delivery and lack of toxicity. Nanomedicine (Lond) 2012;7(4):521–539. doi: 10.2217/nnm.11.158. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 231.Sun D, Schur RM, Lu ZR. A novel nonviral gene delivery system for treating Leber's congenital amaurosis. Ther Deliv. 2017;8(10):823–826. doi: 10.4155/tde-2017-0072. [DOI] [PubMed] [Google Scholar]
- 232.Malamas AS, Gujrati M, Kummitha CM, Xu R, Lu ZR. Design and evaluation of new pH-sensitive amphiphilic cationic lipids for siRNA delivery. J Control Release. 2013;171(3):296–307. doi: 10.1016/j.jconrel.2013.06.019. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 233.Wang XL, Ramusovic S, Nguyen T, Lu ZR. Novel polymerizable surfactants with pH-sensitive amphiphilicity and cell membrane disruption for efficient siRNA delivery. Bioconjug Chem. 2007;18(6):2169–2177. doi: 10.1021/bc700285q. [DOI] [PubMed] [Google Scholar]
- 234.Gujrati M, Malamas A, Shin T, Jin E, Sun Y, Lu ZR. Multifunctional cationic lipid-based nanoparticles facilitate endosomal escape and reduction-triggered cytosolic siRNA release. Mol Pharm. 2014;11(8):2734–2744. doi: 10.1021/mp400787s. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 235.Sun D, Maeno H, Gujrati M, Schur R, Maeda A, Maeda T, Palczewski K, Lu ZR. Self-Assembly of a Multifunctional Lipid With Core-Shell Dendrimer DNA Nanoparticles Enhanced Efficient Gene Delivery at Low Charge Ratios into RPE Cells. Macromol Biosci. 2015;15(12):1663–1672. doi: 10.1002/mabi.201500192. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 236.Sun D, Schur RM, Sears AE, Gao SQ, Vaidya A, Sun W, Maeda A, Kern T, Palczewski K, Lu ZR. Non-viral Gene Therapy for Stargardt Disease with ECO/pRHO-ABCA4 Self-Assembled Nanoparticles. Mol Ther. 2020;28(1):293–303. doi: 10.1016/j.ymthe.2019.09.010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 237.Gonzalez-Fernandez F, Ghosh D. Focus on Molecules: interphotoreceptor retinoid-binding protein (IRBP) Exp Eye Res. 2008;86(2):169–170. doi: 10.1016/j.exer.2006.09.003. [DOI] [PubMed] [Google Scholar]
- 238.Sun D, Sahu B, Gao S, Schur RM, Vaidya AM, Maeda A, Palczewski K, Lu ZR. Targeted Multifunctional Lipid ECO Plasmid DNA Nanoparticles as Efficient Non-viral Gene Therapy for Leber's Congenital Amaurosis. Mol Ther Nucleic Acids. 2017;7:42–52. doi: 10.1016/j.omtn.2017.02.005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 239.Sun D, Schur RM, Sears AE, Gao SQ, Sun W, Naderi A, Kern T, Palczewski K, Lu ZR. Stable Retinoid Analogue Targeted Dual pH-Sensitive Smart Lipid ECO/pDNA Nanoparticles for Specific Gene Delivery in the Retinal Pigment Epithelium. ACS Appl Bio Mater. 2020;3(5):3078–3086. doi: 10.1021/acsabm.0c00130. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 240.Rajala A, Wang Y, Zhu Y, Ranjo-Bishop M, Ma JX, Mao C, Rajala RV. Nanoparticle-assisted targeted delivery of eye-specific genes to eyes significantly improves the vision of blind mice in vivo. Nano Lett. 2014;14(9):5257–5263. doi: 10.1021/nl502275s. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 241.Takahashi Y, Chen Q, Rajala RVS, Ma JX. MicroRNA-184 modulates canonical Wnt signaling through the regulation of frizzled-7 expression in the retina with ischemia-induced neovascularization. FEBS Lett. 2015;589(10):1143–1149. doi: 10.1016/j.febslet.2015.03.010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 242.Liu HA, Liu YL, Ma ZZ, Wang JC, Zhang Q. A lipid nanoparticle system improves siRNA efficacy in RPE cells and a laser-induced murine CNV model. Invest Ophthalmol Vis Sci. 2011;52(7):4789–4794. doi: 10.1167/iovs.10-5891. [DOI] [PubMed] [Google Scholar]
- 243.Patel S, Ryals RC, Weller KK, Pennesi ME, Sahay G. Lipid nanoparticles for delivery of messenger RNA to the back of the eye. J Control Release. 2019;303:91–100. doi: 10.1016/j.jconrel.2019.04.015. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 244.Huang X, Chau Y. Investigating impacts of surface charge on intraocular distribution of intravitreal lipid nanoparticles. Exp Eye Res. 2019;186:107711. doi: 10.1016/j.exer.2019.107711. [DOI] [PubMed] [Google Scholar]
- 245.Zhu SN, Nolle B, Duncker G. Expression of adhesion molecule CD44 on human corneas. Br J Ophthalmol. 1997;81(1):80–84. doi: 10.1136/bjo.81.1.80. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 246.Lerner LE, Schwartz DM, Hwang DG, Howes EL, Stern R. Hyaluronan and CD44 in the human cornea and limbal conjunctiva. Exp Eye Res. 1998;67(4):481–484. doi: 10.1006/exer.1998.0567. [DOI] [PubMed] [Google Scholar]
- 247.Culty M, Nguyen HA, Underhill CB. The hyaluronan receptor (CD44) participates in the uptake and degradation of hyaluronan. J Cell Biol. 1992;116(4):1055–1062. doi: 10.1083/jcb.116.4.1055. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 248.de la Fuente M, Seijo B, Alonso MJ. Bioadhesive hyaluronan-chitosan nanoparticles can transport genes across the ocular mucosa and transfect ocular tissue. Gene Ther. 2008;15(9):668–676. doi: 10.1038/gt.2008.16. [DOI] [PubMed] [Google Scholar]
- 249.de la Fuente M, Seijo B, Alonso MJ. Novel hyaluronic acid-chitosan nanoparticles for ocular gene therapy. Invest Ophthalmol Vis Sci. 2008;49(5):2016–2024. doi: 10.1167/iovs.07-1077. [DOI] [PubMed] [Google Scholar]
- 250.Contreras-Ruiz L, de la Fuente M, Parraga JE, Lopez-Garcia A, Fernandez I, Seijo B, Sanchez A, Calonge M, Diebold Y. Intracellular trafficking of hyaluronic acid-chitosan oligomer-based nanoparticles in cultured human ocular surface cells. Mol Vis. 2011;17:279–290. [PMC free article] [PubMed] [Google Scholar]
- 251.Prakash TP, Mullick AE, Lee RG, Yu J, Yeh ST, Low A, Chappell AE, Ostergaard ME, Murray S, Gaus HJ, Swayze EE, Seth PP. Fatty acid conjugation enhances potency of antisense oligonucleotides in muscle. Nucleic Acids Res. 2019;47(12):6029–6044. [DOI] [PMC free article] [PubMed]
- 252.Chappell AE, Gaus HJ, Berdeja A, Gupta R, Jo M, Prakash TP, Oestergaard M, Swayze EE, Seth PP. Mechanisms of palmitic acid-conjugated antisense oligonucleotide distribution in mice. Nucleic Acids Res. 2020;48(8):4382–4395. [DOI] [PMC free article] [PubMed]
- 253.Ostergaard ME, Jackson M, Low A, A EC, R GL, Peralta RQ, Yu J, Kinberger GA, Dan A, Carty R, Tanowitz M, Anderson P, Kim TW, Fradkin L, Mullick AE, Murray S, Rigo F, Prakash TP, Bennett CF, Swayze EE, Gaus HJ, Seth PP. Conjugation of hydrophobic moieties enhances potency of antisense oligonucleotides in the muscle of rodents and non-human primates. Nucleic Acids Res. 2019;47(12):6045–6058. [DOI] [PMC free article] [PubMed]
- 254.Biscans A, Coles A, Haraszti R, Echeverria D, Hassler M, Osborn M, Khvorova A. Diverse lipid conjugates for functional extra-hepatic siRNA delivery in vivo. Nucleic Acids Res. 2019;47(3):1082–1096. doi: 10.1093/nar/gky1239. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 255.Biscans A, Caiazzi J, McHugh N, Hariharan V, Muhuri M, Khvorova A. Docosanoic acid conjugation to siRNA enables functional and safe delivery to skeletal and cardiac muscles. Mol Ther. 2021;29(4):1382–1394. doi: 10.1016/j.ymthe.2020.12.023. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 256.Hendricks WP, Yang J, Sur S, Zhou S. Formulating the magic bullet: barriers to clinical translation of nanoparticle cancer gene therapy. Nanomedicine (Lond) 2014;9(8):1121–1124. doi: 10.2217/nnm.14.63. [DOI] [PubMed] [Google Scholar]
- 257.Dowdy SF. Overcoming cellular barriers for RNA therapeutics. Nat Biotechnol. 2017;35(3):222–229. doi: 10.1038/nbt.3802. [DOI] [PubMed] [Google Scholar]
- 258.Kulkarni JA, Witzigmann D, Thomson SB, Chen S, Leavitt BR, Cullis PR, van der Meel R. The current landscape of nucleic acid therapeutics. Nat Nanotechnol. 2021;16(6):630–643. doi: 10.1038/s41565-021-00898-0. [DOI] [PubMed] [Google Scholar]
- 259.Tieu TW, Y.; Cifuentes-Rius, A.; Voelcker, N.H. Overcoming Barriers: Clinical Translation of siRNA Nanomedicines. Advanced Therapeutics. 2021;4(9).
- 260.Salvati A, Pitek AS, Monopoli MP, Prapainop K, Bombelli FB, Hristov DR, Kelly PM, Aberg C, Mahon E, Dawson KA. Transferrin-functionalized nanoparticles lose their targeting capabilities when a biomolecule corona adsorbs on the surface. Nat Nanotechnol. 2013;8(2):137–143. doi: 10.1038/nnano.2012.237. [DOI] [PubMed] [Google Scholar]
- 261.Zhang H, Wu T, Yu W, Ruan S, He Q, Gao H. Ligand Size and Conformation Affect the Behavior of Nanoparticles Coated with in Vitro and in Vivo Protein Corona. ACS Appl Mater Interfaces. 2018;10(10):9094–9103. doi: 10.1021/acsami.7b16096. [DOI] [PubMed] [Google Scholar]
- 262.Xiao W, Wang Y, Zhang H, Liu Y, Xie R, He X, Zhou Y, Liang L, Gao H. The protein corona hampers the transcytosis of transferrin-modified nanoparticles through blood-brain barrier and attenuates their targeting ability to brain tumor. Biomaterials. 2021;274:120888. doi: 10.1016/j.biomaterials.2021.120888. [DOI] [PubMed] [Google Scholar]
- 263.Wang Y, Zhang H, Xiao W, Liu Y, Zhou Y, He X, Xia X, Gong T, Wang L, Gao H. Unmasking CSF protein corona: Effect on targeting capacity of nanoparticles. J Control Release. 2021;333:352–361. doi: 10.1016/j.jconrel.2021.04.001. [DOI] [PubMed] [Google Scholar]
- 264.Dai Q, Walkey C, Chan WC. Polyethylene glycol backfilling mitigates the negative impact of the protein corona on nanoparticle cell targeting. Angew Chem Int Ed Engl. 2014;53(20):5093–5096. doi: 10.1002/anie.201309464. [DOI] [PubMed] [Google Scholar]
- 265.Caracciolo G, Cardarelli F, Pozzi D, Salomone F, Maccari G, Bardi G, Capriotti AL, Cavaliere C, Papi M, Lagana A. Selective targeting capability acquired with a protein corona adsorbed on the surface of 1,2-dioleoyl-3-trimethylammonium propane/DNA nanoparticles. ACS Appl Mater Interfaces. 2013;5(24):13171–13179. doi: 10.1021/am404171h. [DOI] [PubMed] [Google Scholar]
- 266.Saunders NRM, Paolini MS, Fenton OS, Poul L, Devalliere J, Mpambani F, Darmon A, Bergere M, Jibault O, Germain M, Langer R. A Nanoprimer To Improve the Systemic Delivery of siRNA and mRNA. Nano Lett. 2020;20(6):4264–4269. doi: 10.1021/acs.nanolett.0c00752. [DOI] [PubMed] [Google Scholar]
- 267.Hwang DW, Son S, Jang J, Youn H, Lee S, Lee D, Lee YS, Jeong JM, Kim WJ, Lee DS. A brain-targeted rabies virus glycoprotein-disulfide linked PEI nanocarrier for delivery of neurogenic microRNA. Biomaterials. 2011;32(21):4968–4975. doi: 10.1016/j.biomaterials.2011.03.047. [DOI] [PubMed] [Google Scholar]