Abstract
Background
There is considerable overlap of contributors to cardiovascular disease and the development of age-related macular degeneration (AMD). Compromised ocular microcirculation due to aging and vascular disease contribute to retinal dysfunction and vision loss. Decreased choroidal perfusion is evident in eyes with dry AMD and is thought to play a role in retinal pigment epithelial dysfunction, the rate of development of geographic atrophy, and the development of neovascularization. The aim of the study was to demonstrate that AMD is correlated with a compromised blood flow in the ocular pathway and show OA angioplasty as a potential treatment of late-stage AMD.
Methods
Based on the potential for the ophthalmic artery (OA) to be an anatomical target for the treatment of AMD as outlined above, five patients were found to be eligible for compassionate use treatment, presenting clinically significant late-stage AMD with profound vision loss in one or both eyes, and are included in this retrospective study.
Results
OA narrowing, or significant calcium burden at the ophthalmic segment of the internal carotid artery compromising the origin of the OA was confirmed in all cases. Subsequent OA cannulation was achieved in all patients with some difficulty. Subjective patient reports indicated that all patients perceived a benefit following the procedure; however, improved postoperative visual acuity did not confirm that perceived benefit for one of the patients.
Conclusions
Feasibility and safety of the OA angioplasty were demonstrated, and a benefit perceived in five patients with profound vision loss and a desire to achieve improved quality of life. A clinical trial with controlled schedule, imaging, and methodologies is needed to confirm these results.
Keywords: angioplasty, orbit, plaque, stenosis, atherosclerosis
Introduction
Age-related macular degeneration (AMD) is the leading cause of irreversible blindness among the elderly in the developed world.1 AMD is a gradually progressive disease that evolves through stages. The earliest sign of AMD is characterized by lipid- and protein-rich extracellular deposits (drusen) between the retinal pigment epithelium and Bruch’s membrane in the central macula, associated with pigmentary changes.2 AMD is a complex disease with multiple genetic and environmental risk factors. The role of genetic factors in the development of AMD is estimated to be between 46% and 71%.3 Susceptibility genes are particularly represented by immune modulation and complement pathway4; however, it is understood that oxidative stress, hypoxia, and compromised autophagy5 may contribute to complement activation,6 promoting disease progression. AMD is also influenced by environmental risk factors, including age, smoking, gender, diet, and cardiovascular disease.7 8
Currently, there are no available treatments for non-exudative AMD; however, studies have shown that dietary supplementation can play a protective role by potentially slowing the progression.9–13 In addition, although an unhealthy diet does not seem to contribute to AMD, those patients with AMD and an unhealthy diet may be more likely to progress to late-stage disease.14
There is considerable overlap in contributing factors to both cardiovascular disease and the development of AMD.8 15 16 Furthermore, compromised ocular microcirculation due to aging and vascular dysfunction contribute to retinal damage and vision loss.17 Decreased choroidal perfusion is evident in eyes with non-exudative AMD18 and is thought to play a role in retinal pigment epithelial dysfunction,19 the rate of geographic atrophy development,20 21 and the development of neovascularization.22
In an effort to evaluate whether or not the internal carotid artery (ICA) and/or the ophthalmic artery (OA) contribute to decreased retinal perfusion in eyes with AMD, a recent study was conducted at the Martinos Center for BioMedical Imaging (Boston, Massachusetts, USA) using 7 Tesla magnetic resonance imaging (7T MRI) and a custom built 32-channel head receive array coil with a birdcage transmit coil to non-invasively capture hemodynamic data of both arteries. Patients with late-stage AMD demonstrated a statistically significant decrease in both lumen diameter (p<0.01) and volumetric flow rates (p<0.01), and an increase in resistive index (p<0.01), which measures dynamic flow properties, in the OA of patients with AMD in comparison with age-matched controls without AMD. Further, they demonstrated as AMD disease progresses, there is a statistically significant (linear trend test p=0.002) declining trend of OA volumetric flow. No statistically significant differences were found in flow rates or resistive index in the ICA of the same AMD cohort as compared with age-matched controls without AMD.23 These findings indicate that the changes in the distal ICA, in or around the OA, might be contributing to decreased retinal perfusion in patients with AMD, and present a potential treatment target in a patient population without therapeutic options.
In the consideration of OA angioplasty or AMD, treatment of carotid artery disease by angioplasty and stenting in patients with ocular ischemic syndrome has been shown to mitigate vision loss,24 demonstrating the benefit of increased ocular perfusion in those patients. In at least one case of acute ischemic retinopathy, OA stenosis was diagnosed, and balloon angioplasty of the OA performed, effectively restoring vision.25 Hayreh and Zimmerman discovered that the OA can have a markedly stenosed lumen at its origin from the ICA with little or no stenosis found in the ICA itself,26 a situation that is echoed in the 7T MRI data discussed previously.
The premise that AMD is correlated with a compromised blood flow in the ocular pathway provided the foundation for OA angioplasty as a potential treatment of late-stage AMD.
Methods
Best corrected visual acuity (BCVA) was assessed at each visit, eccentric/extrafoveal fixation was allowed, and notations such as count fingers with distance measured was provided for patients with late-stage disease with more profound vision loss. In all patients, the eye with the worse BCVA was the treated eye. To provide aggregate analysis of the available data, all count fingers outcomes were converted to Snellen and all visual acuities to logarithm of the minimum angle of resolution (LogMAR) to determine means and SD.27
Regulatory oversight
Based on the potential for the OA to be an anatomical target for the treatment of AMD as outlined above, the treating interventional neuroradiologist sought and received approval from Argentina’s National Administration of Medicines, Foods, and Medical Technology (ANMAT) agency to treat patients under the Compassionate Use framework provided by the ANMAT Regulation 3315/2005.28 This approval provided limited permissions to use product(s) with current CE mark to perform femoral access neurovascular percutaneous transluminal angioplasty to the OA and its structures. The Compassionate Use procedure was approved to provide a potential treatment for those patients with advanced AMD in an environment where conventional, recognized therapy is ineffective or non-existent. Standard of care is driven only by the treating physician’s determination of appropriate evaluations or procedures at a given visit.
A retrospective study designed to evaluate the case series and first 6 months of postoperative follow-up was developed and reviewed by an independent review board, and approval to proceed was obtained. The retrospective study to evaluate this series of patients treated under the Compassionate Use approval was conducted in accordance with Argentine Data Protection Law No 25 326, the Declaration of Helsinki, and local regulations. All patients provided consent to allow their data to be used for analysis and publication purposes.
Patient population
Identification of a potential patient was provided by the treating ophthalmologist. A referral was then made to the interventional neuroradiologist for vascular evaluation and consideration for intervention. Patients were required to have late-stage AMD (history of wet or dry) as shown by the presence of geographic atrophy or stabilized history of choroidal neovascularization, visual acuity of 20/400 or worse, history of cardiovascular disease, and the likelihood of safe access and dilation of the OA anatomy to be eligible for treatment.
In all cases, the eye with the worse visual acuity was selected for treatment. Although the macula in these patients was severely compromised with reduced likelihood of a detectable improvement in vision, these patients were identified as viable candidates since an unsuccessful intervention was unlikely to affect their quality of life. Patient eligibility was confirmed jointly between therapeutic specialists prior to surgical intervention, and all patients provided written consent, indicating full understanding of the nature of the treatment and associated risks.
Interventional methods and materials
Review of systems, adjustments in concomitant and antiplatelet therapies, hydration, surgical preparation for femoral access, anesthesia, vasodilator use, analgesia, and ancillary devices were managed according to the interventionist’s standard of care.
On the day of intervention, patients were admitted and administered 10 000 units of heparin and an additional 1000 units/h to achieve and maintain a target activated clotting time of 250–300 s. Percutaneous access was achieved in the femoral artery, the ICA accessed, and various 0.014" guidewires and microcatheters (0.0165 in) were employed in the event of challenging anatomy and to achieve OA cannulation. Local injection of a calcium channel blocker (nimodipine) into the OA via the microcatheter was used as needed to mitigate vasospasm pre- and postintervention. A rapid exchange neurovascular balloon catheter (pITA, phenox, Bochum) was advanced over the guidewire to the OA. The balloon was centered in the OA ostium with 5 mm protruding into the OA short limb, followed by up to two balloon inflations to 10 atm for 20 s each. Following dilation, all devices were removed, standard closure was performed, and patients were monitored and discharged according to standard practice, and returned to their referring ophthalmologist for follow-up examinations.
Diagnostic and Intraoperative imaging
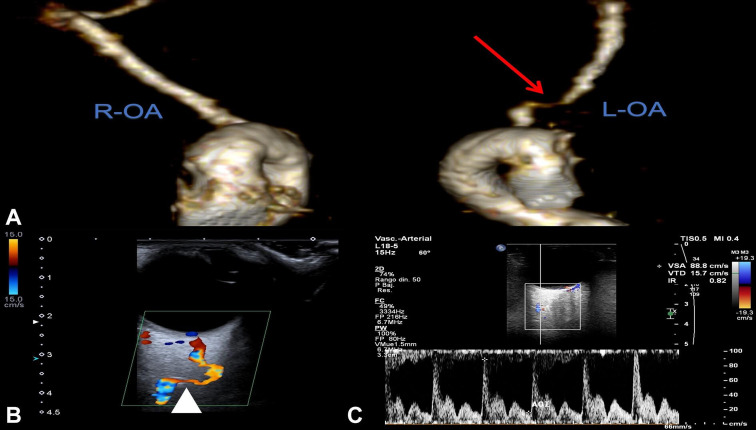
Patients underwent 3T MRI with angiography with the field of view centered on the OA origin using a 32-channel head coil from Philips (Eindhoven, Netherlands) and an EcoDoppler analysis prior to the interventional procedure in an attempt to identify compromised blood flow in the OA (figure 1). From the work of Hayreh and Zimmerman,26 it was expected that compromised flow in the OA would manifest as a discrete lesion in the OA ostium or short limb, a general luminal narrowing of the OA ostium or short limb, and/or vascular disease in the ICA extending over or around the OA ostium. However, a discrete lesion in the OA short limb was not readily discernable by MRI in all patients. Better definition of the OA was achieved by intracranial DSA. Conventional two-dimensional digital subtraction angiography combined with three-dimensional rotational angiography (Philips; Amsterdam, Netherlands) are the current standard intracranial imaging techniques used during percutaneous transluminal angioplasty procedures.
Figure 1.
Diagnostic workup for case 002. (A) 3T brain MR angiography 3D reconstruction depicting the origin of both ophthalmic arteries (OAs). The red arrow denotes stenosis in the long limb segment of the ophthalmic artery. EcoDoppler analysis, white (B) the arrow head depicts stenotic segment of the ophthalmic artery with (C) peak systolic velocities in the flow velocity waveform.
In cases 001 and 005, although narrowing of the OA was not severe, important amount of calcium burden was observed by VasoCT reconstruction at the ophthalmic segment of the ICA comprising its origin.
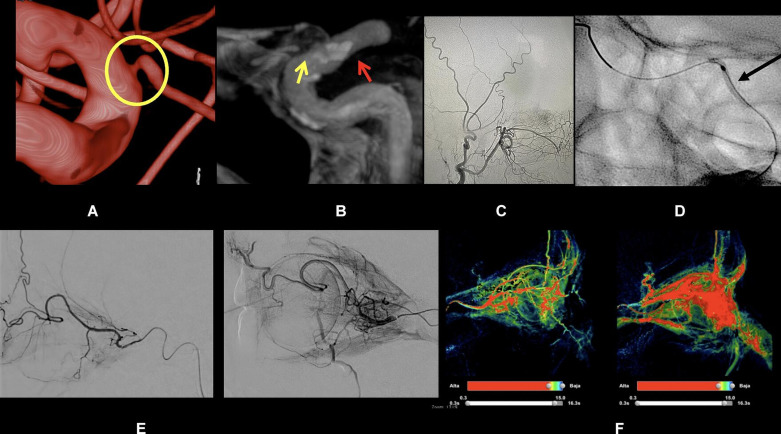
At the fluoroscopy suite setting, the image protocol for OA stenosis was based on angiographic DSA and the intraoperative cone-beam CT performed with a Philips BiPlane (Eindhoven, Netherlands) or monoplane angiographic flat detector 20/10 system. Intraoperative images were successfully captured for the precise OA origin and the relationship with an atheroma nearby or in the artery, and processed with VasoCT software from a Philips workstation29 (figure 2).
Figure 2.
Ophthalmic artery (OA) angioplasty for case 001. (A, B) 3D digital subtraction angiography reconstruction view of the internal carotid artery/OA ostium. Yellow circle highlighting the OA stenosis at its origin. (B) Cone-beam CT of the OA. The red arrow represents the plaque surrounding the OA origin and the yellow arrow represents the OA. (C) Balloon angioplasty at the OA short limb segment indicated by the black arrow. (D, E) 2D and 3D perfusion imaging preprocedure and postprocedure.
Results and discussion
Demographics and baseline characteristics
This cohort of five patients reflect a 60/40 split male to female, and an aged population, with a mean of 80.4 years and a range of 77 to 84. One of the patients (004) presented clinically significant late-stage AMD with profound vision loss in both eyes (OU) and the remaining patients in the left eye (OS) only. All patients presented with hypertension, two of them with a history of dyslipidemia (001 and 003), and one of those with a history of cardiac stenting and chronic obstructive pulmonary disease (001) and the other (003), Hashimoto’s disease.
Procedural results
The OA was successfully cannulated in all patients but with some difficulty due to the acute take-off angle of the OA, partial restriction of the OA ostium, and challenging device performance. Multiple guidewires, microcatheters, and tip shaping were necessary in most cases to accomplish OA cannulation. The balloon catheter was successfully advanced into the OA in all the patients. In patient 002, repeated attempts to advance the balloon catheter into the OA caused the guidewire to back out due to excessive stiffness of the balloon catheter and poor back-up support. When balloon catheter cannulation was successful, complete inflation of the balloon was confirmed by fluoroscopy.
Adverse events
The patients were managed postoperatively according to the standard of care and all patients had an uneventful postoperative period in this regard. All patients experienced minor adverse events associated with femoral access endovascular procedures, including localized inflammation or mild pain at the surgical site. The only intraoperative complication (patient 003) involved a 0.035” guidewire tracking into a false lumen of the right ICA causing a small dissection, which was treated by placement of a carotid artery stent. The dissection was resolved at the time of treatment without sequelae, and the remainder of the planned intervention was completed. The patient demonstrated a postoperative modified Rankin Scale score of 0 at 90 days. No other adverse events occurred and there were no neurological complications or deaths in this cohort of patients.
Ocular results
Visual acuity
Owing to the nature of treatment in a standard clinical setting and resulting variability of postoperative visit timelines between patients, data were grouped in as narrow a window as possible because a prospective visit schedule was not used. As shown in table 1, mean preoperative BCVA was 20/710 and improved to 20/383 (1.55 to 1.28 LogMAR) at 1 week postoperatively, representing a mean gain of nearly three lines, remaining stable through month 6. Patients 005 and 004 did not have data available for week 3 and month 6, respectively.
Table 1.
Visual acuity (n=5)
| BCVA | Preoperative | Window week 1 7±3 days |
Window week 3 21±10 days |
Window month 3 90±30 days |
Window month 6 180±30 days |
| n=5 | n=5 | n=4 | n=5 | n=4 | |
| LogMAR mean (SD) | 1.55 (0.26) | 1.28 (0.50) | 1.28 (0.39) | 1.30 (0.38) | 1.31 (0.44) |
| Mean Snellen | 20/710 | 20/383 | 20/372 | 20/400 | 20/406 |
| Snellen min, max | 400, 1334 | 150, 1334 | 200, 1334 | 150, 1334 | 150, 1334 |
BCVA, best corrected visual acuity; LogMAR, logarithm of the minimum angle of resolution.
Table 2 provides the visual acuity listing associated with each patient by visit. Patient 001 experienced a 10-line gain in the early postoperative period, that reduced after 3 weeks and returned to the 10-line gain through month 6. Patient 002 demonstrated no change in the early postoperative period; however, by month 3 the participant gained three lines of vision, which was maintained through month 6. Patient 003 demonstrated a gain of approximately 4 lines of vision early, which fluctuated in the postoperative period, but stabilized at a 3-line gain at the 6-month follow-up. Patient 004 gained three lines early, which was maintained over 3 months. Patient 005 demonstrated an early postoperative loss of 3 lines, which was maintained through months 3 and 6. It should be noted that patient 005 did not experience any procedure-related events, or ophthalmic signs that could explain the vision loss, and demonstrated fluctuating acuity that returned to preoperative levels during additional postoperative follow-up, approximately 9 months postoperatively.
Table 2.
Visual acuity by patient (n=5)
| Patient | Preoperative (Snellen) |
LogMAR | Window week 1 7 ±3 days (Snellen) |
LogMAR | Window week 3 21 ±10 days (Snellen) |
LogMAR | Window month 3 90 ±30 days (Snellen) |
LogMAR | Window month 6 180 ±30 days (Snellen) |
LogMAR |
| 001 | 20/1334 | 1.82 | 20/150 | 0.88 | 20/400 | 1.30 | 20/150 | 0.88 | 20/150 | 0.88 |
| 002 | 20/1334 | 1.82 | 20/1334 | 1.82 | 20/1334 | 1.82 | 20/667 | 1.52 | 20/667 | 1.52 |
| 003 | 20/400 | 1.30 | 20/150 | 0.88 | 20/200 | 1.00 | 20/400 | 1.30 | 20/200 | 1.00 |
| 004 | 20/400 | 1.30 | 20/200 | 1.00 | 20/200 | 1.00 | 20/200 | 1.00 | – | – |
| 005 | 20/667 | 1.52 | 20/1334 | 1.82 | – | – | 20/1334 | 1.82 | 20/1334 | 1.82 |
LogMAR, logarithm of the minimum angle of resolution.;
Although anecdotal, all patients expressed a perceived functional benefit in their day-to-day lives as a result of the treatment.
Discussion
As previously discussed, there is a single published case of OA angioplasty used to successfully restore vision in a patient presenting with OA stenosis and acute ischemic vision loss.25 However, this is the first known publication describing the use of OA angioplasty for the treatment of AMD. Our preliminary experience in a small cohort of patients suggests that OA angioplasty may be performed safely, and there is subjective evidence of efficacy.
In the fluoroscopy suite setting, the image protocol for identification of OA stenosis was based on angiographic DSA and the intraoperative cone-beam CT. Intraoperative images were successfully captured for the precise OA origin and the relationship with an atheroma nearby or in the artery and processed with VasoCT.29
Balloon cannulation and dilatation of the OA was successfully achieved in all cases, although with some degree of difficulty due to the limitation of having to use commercially available intravascular devices.
The challenges associated with the procedure itself, as well as the resulting procedural times, were associated with being restricted to the use of commercially available imaging and intravascular catheter-based devices. Although the cone-beam CT imaging was sufficient to visualize OA stenosis intraoperatively, the preference would be for a non-invasive technology that could be used outside the fluoroscopy suite. Regarding intravascular devices, although the compassionate use path allows for the use of CE marked products, there are currently no intravascular catheter-based devices available that are designed specifically to access the OA, which branches from the ICA at an acute angle. Access to stenosed OA can be difficult due to small lumen size, and a smaller profile, more flexible balloon designed specifically for this application might facilitate the procedure. Procedure times should be shortened, and specifically designed devices could assist with this.
Weaknesses presented by the current data include a small cohort of patients and subjective data, which should be interpreted with caution. Placebo effect is a concern and there were no prospective requirements on evaluation timing or methodology. It can be confirmed, however, that the methodologies used were uniformly applied and are consistent with standard of care. In addition, microperimetry and spectral domain optical coherence tomography were performed on most patients; however, the outcomes were inconclusive. With regard to microperimetry, it is hypothesized that central fixation necessary to accurately and reproducibly perform this test was not available in this population of patients with profound central vision loss. For spectral domain optical coherence tomography, in recent years there have been advances in the evaluation of deeper choroidal layers of the retina with the development of swept source optical coherence tomography. This technology may be better equipped to provide objective evidence of increased choroidal perfusion. In addition, new imaging technologies are in development, which may provide improved resolution and allow for better preprocedure and postprocedure characterization of the OA and surrounding anatomy. One such technology integrates fiber optic technologies into an intravascular catheter-based optical coherence tomography system. This type of intravascular imaging catheter may allow for the in vivo visualization of cerebral arteries and in situ stents with high image resolution.30
Wet AMD has limited treatment options that are invasive, costly, and at best will slow the disease progression. Dietary supplementation is the only treatment and monitoring disease progression is the only path for dry AMD. These limitations, recent work in the area of OA stenosis in eyes with AMD, and the experiences reported here indicate that treating the retina alone may not suffice.
Conclusion
The preliminary evidence provided by this small case series suggests that OA angioplasty can be performed safely, despite technical challenges associated with the use of currently available devices. Designing a specific tool set for access and treatment of the OA could contribute to shortening procedural times and reducing case complexity. Although preliminary efficacy is noted, a clinical trial with controlled schedule, imaging, and methodologies would be desirable in an effort to challenge and replicate the results reported here. Furthermore, the OA is a viable target to increase blood flow to the eye, restore retinal perfusion, and perhaps disrupt the disease process.
neurintsurg-2021-018222supp001.pdf (509.8KB, pdf)
Footnotes
Twitter: @eneri_neuro
Contributors: AAF and TZ composed the cohort; MC and PNL conceptualized the study; CB, NP, and PNL performed data preparation; JL, ES, and PNL supervised the clinical aspects of the study, IL performed data analyses and wrote the manuscript. All authors critically reviewed the manuscript and approved the final version. IL acts as the guarantor of the study.
Funding: The authors have not declared a specific grant for this research from any funding agency in the public, commercial or not-for-profit sectors.
Competing interests: PNL: consultant for OcuDyne, Phenox, Medtronic, grant or contract from Philips with institution; TZ: consultant for OcuDyne; MC: consultant for OcuDyne.
Provenance and peer review: Not commissioned; externally peer reviewed.
Supplemental material: This content has been supplied by the author(s). It has not been vetted by BMJ Publishing Group Limited (BMJ) and may not have been peer-reviewed. Any opinions or recommendations discussed are solely those of the author(s) and are not endorsed by BMJ. BMJ disclaims all liability and responsibility arising from any reliance placed on the content. Where the content includes any translated material, BMJ does not warrant the accuracy and reliability of the translations (including but not limited to local regulations, clinical guidelines, terminology, drug names and drug dosages), and is not responsible for any error and/or omissions arising from translation and adaptation or otherwise.
Data availability statement
Data are available upon reasonable request. Not applicable.
Ethics statements
Patient consent for publication
Not applicable.
Ethics approval
This study involves human participants and was approved by Comité de Ética de La Clínica La Sagrada Familia, Reference Number 47-6653/94-6. Participants gave informed consent to participate in the study before taking part.
References
- 1. Ratnapriya R, Chew EY. Age‐related macular degeneration—clinical review and genetics update. Clin Genet 2013;84:160–6. 10.1111/cge.12206 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2. Campagne M, LeCouter J, Yaspan B. Mechanisms of age-related macular degeneration and therapeutic opportunities. J Pathol 2014;232:151–64. 10.1002/path.4266 [DOI] [PubMed] [Google Scholar]
- 3. Warwick A, Lotery A. Genetics and genetic testing for age-related macular degeneration. Eye 2018;32:849–57. 10.1038/eye.2017.245 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4. Mousavi M, Armstrong RA. Genetic risk factors and age-related macular degeneration (AMD). J Optom 2013;6:176–84. 10.1016/j.optom.2013.07.002 [DOI] [PubMed] [Google Scholar]
- 5. Blasiak J, Petrovski G, Veréb Z, et al. Oxidative stress, hypoxia, and autophagy in the neovascular processes of age-related macular degeneration. Biomed Res Int 2014;2014:1–7. 10.1155/2014/768026 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6. Kaarniranta K, Salminen A, Eskelinen E-L, et al. Heat shock proteins as gatekeepers of proteolytic pathways—Implications for age-related macular degeneration (AMD). Ageing Res Rev 2009;8:128–39. 10.1016/j.arr.2009.01.001 [DOI] [PubMed] [Google Scholar]
- 7. Chakravarthy U, Wong TY, Fletcher A, et al. Clinical risk factors for age-related macular degeneration: a systematic review and meta-analysis. BMC Ophthalmol 2010;10:31. 10.1186/1471-2415-10-31 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8. Pennington KL, DeAngelis MM. Epidemiology of age-related macular degeneration (AMD): associations with cardiovascular disease phenotypes and lipid factors. Eye Vis (London) 2016;3:34. 10.1186/s40662-016-0063-5 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9. Age-Related Eye Disease Study Research Group . A randomized, placebo-controlled, clinical trial of high-dose supplementation with vitamins C and E, beta carotene, and zinc for age-related macular degeneration and vision loss. Arch Ophthalmol 2001;119:1417–36. 10.1001/archopht.119.10.1417 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10. Chew EY, Clemons TE, SanGiovanni JP, et al. Lutein + zeaxanthin and omega-3 fatty acids for age-related macular degeneration: the age-related eye disease study 2 (AREDS2) randomized clinical trial. JAMA 2013;309:2005–15. 10.1001/jama.2013.4997 [DOI] [PubMed] [Google Scholar]
- 11. Christen WG, Schaumberg DA, Glynn RJ, et al. Dietary ω-3 fatty acid and fish intake and incident age-related macular degeneration in women. Arch Ophthalmol 2011;129:921–9. 10.1001/archophthalmol.2011.34 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12. Millen AE, Voland R, Sondel SA, et al. Vitamin D status and early age-related macular degeneration in postmenopausal women. Arch Ophthalmol 2011;129:481–9. 10.1001/archophthalmol.2011.48 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13. Parekh N, Chappell RJ, Millen AE, et al. Association between vitamin D and age-related macular degeneration in the third National health and nutrition examination survey, 1988 through 1994. Arch Ophthalmol 2007;125:661–9. 10.1001/archopht.125.5.661 [DOI] [PubMed] [Google Scholar]
- 14. Dighe S, Zhao J, Steffen L, et al. Diet patterns and the incidence of age-related macular degeneration in the Atherosclerosis Tisk In Communities (ARIC) study. Br J Ophthalmol 2020;104:1–7. 10.1136/bjophthalmol-2019-314813 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15. van Leeuwen R, Ikram MK, Vingerling JR, et al. Blood pressure, atherosclerosis, and the incidence of age-related maculopathy: the Rotterdam study. Invest Ophthalmol Vis Sci 2003;44:3771–7. 10.1167/iovs.03-0121 [DOI] [PubMed] [Google Scholar]
- 16. Vingerling JR, Dielemans I, Bots ML, et al. Age-related macular degeneration is associated with atherosclerosis. Am J Epidemiol 1995;142:404–9. 10.1093/oxfordjournals.aje.a117648 [DOI] [PubMed] [Google Scholar]
- 17. Friedman E. A hemodynamic model of the pathogenesis of age-related macular degeneration. Am J Ophthalmol 1997;124:677–82. 10.1016/S0002-9394(14)70906-7 [DOI] [PubMed] [Google Scholar]
- 18. Friedman E, Krupsky S, Lane AM, et al. Ocular blood flow velocity in age-related macular degeneration. Ophthalmology 1995;102:640–6. 10.1016/S0161-6420(95)30974-8 [DOI] [PubMed] [Google Scholar]
- 19. Feigl B. Age-related maculopathy – linking aetiology and pathophysiological changes to the ischaemia hypothesis. Prog Retin Eye Res 2009;28:63–86. 10.1016/j.preteyeres.2008.11.004 [DOI] [PubMed] [Google Scholar]
- 20. Coleman DJ, Silverman RH, Rondeau MJ, et al. Age-related macular degeneration: choroidal ischaemia? Br J Ophthalmol 2013;97:1020–3. 10.1136/bjophthalmol-2013-303143 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21. Coleman DJ, Lee W, Chang S, et al. Treatment of macular degeneration with sildenafil: results of a two-year trial. Ophthalmologica 2018;240:45–54. 10.1159/000486105 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22. Metelitsina TI, Grunwald JE, DuPont JC, et al. Foveolar choroidal circulation and choroidal neovascularization in age-related macular degeneration. Invest Ophthalmol Vis Sci 2008;49:358–63. 10.1167/iovs.07-0526 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23. Hibert ML, Chen YI, Ohringer N, et al. Altered blood flow in the ophthalmic and internal carotid arteries in patients with age-related macular degeneration measured using noncontrast Mr angiography at 7T. AJNR Am J Neuroradiol 2021;42:1653–60. 10.3174/ajnr.A7187 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24. Ishii M, Hayashi M, Yagi F, et al. Relationship between the direction of ophthalmic artery blood flow and ocular microcirculation before and after carotid artery stenting. J Ophthalmol 2016;2016:1–6. 10.1155/2016/2530914 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25. Hwang GJ, Woo SJ, Hwang J-M, et al. Reversal of ischemic retinopathy following balloon angioplasty of a stenotic ophthalmic artery. J Neuroophthalmol 2010;30:228–30. 10.1097/WNO.0b013e3181dc2078 [DOI] [PubMed] [Google Scholar]
- 26. Hayreh SS, Zimmerman MB. Ocular arterial occlusive disorders and carotid artery disease. Ophthalmology Retina 2017;1:12–18. 10.1016/j.oret.2016.08.003 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27. Holladay JT. Proper method for calculating average visual acuity. J Refract Surg 1997;13:388–91. 10.3928/1081-597X-19970701-16 [DOI] [PubMed] [Google Scholar]
- 28. ANMAT . Provision 3315/2005-ANMAT. In: Registry of the administration national medicines, food, and medical technology, 2005. [Google Scholar]
- 29. Aadland TD, Thielen KR, Kaufmann TJ, et al. 3D C-arm conebeam CT angiography as an adjunct in the precise anatomic characterization of spinal dural arteriovenous fistulas. AJNR Am J Neuroradiol 2010;31:476–80. 10.3174/ajnr.A1840 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30. Gounis MJ, Ughi GJ, Marosfoi M, et al. Intravascular optical coherence tomography for neurointerventional surgery. Stroke 2018:STROKEAHA118022315. 10.1161/STROKEAHA.118.022315 [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
neurintsurg-2021-018222supp001.pdf (509.8KB, pdf)
Data Availability Statement
Data are available upon reasonable request. Not applicable.