Abstract
Chronic obstructive pulmonary disease (COPD) is a disabling condition that is characterised by poorly reversible airflow limitation and inflammation. Acute exacerbations of COPD are a common cause of hospitalisation and death among COPD patients. Several biochemical markers have been studied as outcome predictors in COPD; however, their measurement often requires significant time and resources. Relatively simple biomarkers of inflammation calculated from routine complete blood count tests, such as the neutrophil to lymphocyte ratio (NLR), might also predict COPD progression and outcomes. This review discusses the available evidence from studies investigating the associations between the NLR, COPD exacerbations and death in this patient group.
Short abstract
Blood neutrophil to lymphocyte ratio can predict stable COPD, its exacerbations and prognosis http://ow.ly/2aJ030hzUIn
Introduction
Chronic obstructive pulmonary disease (COPD) affects more than 200 million individuals and is currently the third leading cause of death worldwide (up from the sixth in 1990), with mortality rates projected to increase further [1–3]. COPD is a major contributor of healthcare costs due to frequent hospitalisation, loss of productivity and disability [4]. COPD is a heterogeneous collection of syndromes with complex pathophysiological mechanisms and different clinical phenotypes, which negatively affect accurate diagnosis, as well as the design and validation of effective studies and therapies [5–7].
By and large, COPD is characterised by a progressive destruction of the pulmonary tissue, often the result of an inflammatory response to external stimuli (i.e. long-term exposure to cigarette smoking, environmental pollution), which culminates in a non-fully reversible airflow limitation [8]. Typical clinical manifestations include chronic bronchitis, caused by large-airway inflammation and remodelling, and emphysema, a disease of the distal airways and the lung parenchyma, characterised by loss of alveolar respiratory surface. Shortness of breath and chronic productive cough can progress over time to chronic hypoxaemic and/or hypercapnic respiratory failure [9, 10]. Moreover, COPD is often characterised by extrapulmonary manifestations, such as systemic inflammation, cardiovascular comorbidities, cancer, cachexia and muscle dysfunction, osteoporosis, anaemia, depression and anxiety, which also contribute to disability and premature mortality [9, 11].
Acute exacerbations of COPD (AECOPD), defined as worsening of the patient's baseline dyspnoea, cough and/or sputum are an essential component of the natural history of the disease [12]. AECOPD are associated with increased risk of subsequent exacerbations, worsening of coexisting pathological conditions, poor performance status and physical activity, deterioration of respiratory function and, ultimately, death [13]. About half of AECOPD are triggered by bacterial and viral infections; however, non-infective factors such as environmental pollution can also contribute [14–17]. It is possible that bacterial colonisation itself, or repetitive and intermittent exacerbation caused by recurrent infections, may contribute to the chronic inflammatory state and progression of COPD [18]. Inflammation encompasses a complex network of interactions involving various immune-related cells, including neutrophils and lymphocytes, which can lead to persistent respiratory tissue injury and damage [19]. Moreover, it is likely that an excessive inflammatory response against bacteria contributes to chronic inflammation [18–21]. It has been reported that the absolute counts of key immune-related cell populations in the peripheral blood, and their ratios, can adequately reflect chronic inflammatory conditions [22–24].
In particular, the neutrophil to lymphocyte ratio (NLR) in peripheral blood is being increasingly studied as a systemic inflammatory marker, particularly considering its rapid, widely available, and relatively inexpensive assessment through routine blood count analysis. NLR has been shown to be an independent prognostic factor in various solid tumours, including lung, colorectal, pancreatic, breast, ovarian and gastric cancer [25–30]. Furthermore, it has been associated with disease severity, hospitalisation, malnutrition, recurrences and mortality in various chronic diseases, including cardiovascular and kidney diseases [31–33]. In recent years, the NLR has also been investigated as diagnostic and prognostic marker in COPD. Chronic inflammation in COPD causes the recruitment of both the main white blood cell populations, lymphocytes and neutrophils. The latter, once activated, release neutrophil elastase, cathepsin G, proteinase-3, matrix metalloproteinase (MMP)-8 and MMP-9, myeloperoxidase (MPO) and human neutrophil lipocalin, which participate actively in the pathophysiological mechanisms of emphysema and COPD. For example, neutrophil elastase is able to degrade insoluble elastin and MPO mediates the bactericidal effects of neutrophils. Furthermore, both neutrophil elastase and MPO favour tissue destruction in COPD [34, 35]. Therefore, NLR has been investigated as a putative marker of disease severity and prognosis. In this review, we discuss the results of published studies on the association between NLR, disease exacerbation and mortality in COPD.
NLR in stable COPD and exacerbations
The use of NLR as a marker in patients with stable COPD or during AECOPD has only been evaluated in the past few years (table 1). In 2014, Günay et al. [36] investigated the hospital records of 269 COPD patients with stable disease and acute exacerbations, as well as 50 sex- and age-matched healthy controls. There was a significant difference in NLR values between healthy controls (mean±sd 1.71±0.65), stable COPD (2.59±1.79) and AECOPD (4.28±4.12). However, there were no significant differences in relation to disease severity, both in stable and AECOPD patients. By contrast, there were significant positive correlations between NLR, C-reactive protein (CRP), and red cell distribution width (RDW), as well as a negative association between NLR and the mean platelet volume (MPV), both in stable COPD and AECOPD. Systemic inflammation reduces the survival of erythrocytes and platelets and deforms their membranes. High RDW and MPV have been associated with increased inflammatory activity in several diseases [57–60]. This might explain the correlations with NLR found by Günay et al. [36] in patients with stable COPD, and supports the potential applicability of simple blood count indexes in the clinical evaluation of the disease.
TABLE 1.
Studies investigating the predictive roles of neutrophil to lymphocyte ratio (NLR) in chronic obstructive pulmonary disease (COPD)
| First author [ref.] | Patients enrolled | Study design | Main findings |
| NLR as predictor of exacerbations in stable COPD | |||
| Günay [36] | COPD: 178 stable, 91 exacerbations; 50 healthy controls | Retrospective | NLR significantly different between controls and COPD patients, but not between patients with stable COPD and with exacerbations |
| Furutate [37] | COPD: 141 stable, 49 exacerbations | Retrospective | NLR significantly higher in patients with exacerbations. Significant correlations with FEV1, BODE, BMI, 6MWT, mMRC, low-attenuation area and fat-free mass index |
| Lee [38] | 885 COPD, 233 exacerbations (1 year) | Retrospective | NLR significantly higher in patients with exacerbations. NLR predictor of exacerbations. Significant correlation with FEV1 |
| Taylan [39] | COPD: 100 stable with exacerbation; 80 healthy controls | Retrospective | NLR significantly higher in patients with exacerbations, NLR higher in stable COPD than in controls. NLR ≥3.29 predictor of exacerbation |
| Farah [40] | COPD: 13 stable, 72 exacerbations; 15 healthy controls | Retrospective | NLR significantly higher in patients with exacerbations. NLR ≥7.3 predictor of exacerbation |
| Kurtipek [41] | COPD: 94 male patients, 48 with exacerbations | Prospective | NLR significantly higher in exacerbations. NLR ≥3.3 predictor of exacerbation |
| Bilir [42] | COPD: 467 patients, 186 with exacerbations; 215 healthy controls | Retrospective | NLR significantly higher in COPD and in exacerbations. NLR ≥3.35 predictor of exacerbation |
| In [43] | COPD: 103 patients, 47 with exacerbations; 40 healthy controls | Retrospective | NLR significantly higher in COPD and in exacerbations. NLR ≥3.34 predictor of exacerbation |
| NLR as marker of exacerbation aetiology (infection) | |||
| Tanriverdi [44] | COPD: 77 patients with exacerbations | Prospective | Bacterial infection in 37.4% of the patients. NLR ≥11.5 significantly associated with bacterial infection |
| van de Geijn [45] | COPD: 17 stable and 45 exacerbations (22 bacterial and 23 non-bacterial) | Retrospective | NLR determined with different methods significantly higher in exacerbations than in stable, and in bacterial exacerbations than in non-bacterial ones |
| NLR in COPD patients with comorbidities | |||
| Vaguliene [46] | 139 lung cancer; COPD: 40 stable and 55 with lung cancer; 33 healthy controls | Prospective | NLR significantly higher in lung cancer patients. No significant difference in NLR between COPD and healthy patients |
| Yasar [47] | COPD: 140, 63 with metabolic syndrome; 50 healthy controls | Retrospective | NLR significantly higher in COPD patients than in controls, NLR significantly higher in those with metabolic syndrome. NLR ≥2.56 predictor of COPD |
| Arisou [48] | COPD: 40 stable; 40 healthy controls | Prospective | NLR was higher in COPD patients than in the control group. Significant positive correlation between tricuspid atrial conduction time and NLR |
| NLR as predictor of hospitalisation | |||
| Lee [49] | COPD: 61 stable, 59 exacerbations; 28 healthy controls | Prospective | NLR significantly higher in patients with exacerbations. Significant correlations with mMRC and 6MWT. NLR ≥2.8 predictor of hospitalisation |
| NLR as predictor of re-hospitalisation | |||
| Duman [50] | COPD: 1704 with exacerbations, 351 with >2% eosinophils | Retrospective | NLR significantly lower in the eosinophilic group. NLR predictor of re-admission and death |
| NLR as predictor of death | |||
| Rahimirad [51] | COPD: 315, 70 died in hospital | Retrospective | NLR significantly higher in patients who died in hospital. NLR ≥4 predictor of in-hospital death |
| Yao [52] | COPD: 303 with exacerbations, 33 died in hospital | Retrospective | NLR significantly higher in patients who died in hospital. NLR ≥6.24 predictor of in-hospital death |
| Xiong [53] | COPD: 368 stable, 96 died during follow-up; 298 healthy controls | Prospective | NLR significantly higher in COPD patients than in controls. NLR significantly higher in those who died during follow-up. NLR ≥3.3 predictor of death |
| Kumar [54] | COPD: 181 with exacerbation, 16 died within 90 days | Retrospective | NLR significantly higher in patients who died within 90 days from exacerbation |
| Saltürk [55] | COPD in ICU: 647, 62 with >2% eosinophils | Retrospective | NLR significantly lower in the eosinophilic group. NLR ≥16 predictor of death |
| NLR and oral corticosteroid therapy in COPD | |||
| Sørensen [56] | COPD: 386, 84 using glucocorticoids | Prospective | NLR significantly lower in patients without glucocorticoids, NLR predictor of death in patients not treated with glucocorticoids |
FEV1: forced expiratory volume in 1 s; BMI: body mass index; BODE: BMI, airflow obstruction, dyspnoea, exercise capacity; 6MWT: 6-min walking test; mMRC: modified Medical Research Council dyspnoea scale; ICU: intensive care unit.
In another study, Furutate et al. [37] enrolled 141 COPD patients to evaluate the association between the NLR and clinical parameters in stable disease, and to investigate potential changes during exacerbations in 49 patients. The NLR was significantly correlated with the forced expiratory volume in 1 s (FEV1) and the BODE (BMI, airflow obstruction, dyspnoea, exercise capacity) index, a composite marker of disease in COPD. There was also a significant correlation between the NLR and the percentage of low-attenuation area representing emphysematous changes, body mass index (BMI), fat-free mass index, nutritional status and COPD severity, 6-min walk test and the modified Medical Research Council dyspnoea scale (mMRC) score, but not with the fat-mass or the Charlson comorbidity index. The associations remained significant after adjusting for age, sex, Charlson co-morbidity index and smoking status, and suggest the potential role of the NLR to predict features of COPD severity. In this study, a significant association was also observed between CRP and NLR. In addition, during exacerbations, the NLR values were significantly elevated when compared to stable disease.
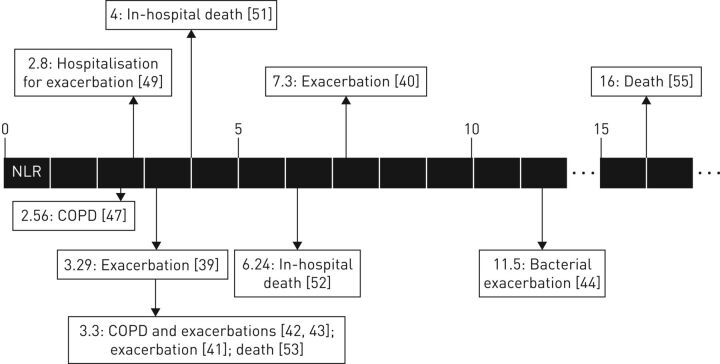
Lee et al. [49] prospectively measured NLR in 59 patients with COPD exacerbation, 61 patients with stable disease and 28 healthy controls. NLR values were significantly higher in patients with COPD exacerbation when compared to those with stable disease and healthy controls (12.4±10.6, 2.4±0.7 and 1.4±0.5, respectively). The NLR differences between patients with stable COPD and controls were significant using an unpaired t-test, but not using other statistical tests. Moreover, patients recovering from exacerbations had a significantly lower NLR value, compared with values during the exacerbations (4.5±4.6 versus 11.5±8.8). Regarding the correlations between NLR and clinical parameters, in 33 patients with stable COPD and eight with AECOPD, it was found that associations with BMI and FEV1 were not significant, as opposed to correlations with mMRC score and 6-min walk test. Furthermore, receiver operating curve (ROC) analysis showed that 2.8 was the optimal NLR cut-off value with maximal sensitivity (60%) and specificity (60.9%) for hospitalisation. In univariate logistic regression analysis a NLR ≥2.8 independently predicted hospitalisation, together with low BMI and FEV1 values. In multivariate analysis a NLR ≥2.8 remained an independent predictor of hospitalisation. This cut-off value, also assessed in other studies, is close to values observed in healthy subjects, which might limit its use in clinical practice (table 1, figure 1).
FIGURE 1.
Neutrophil to lymphocyte ratio (NLR) cut-off values in chronic obstructive pulmonary disease (COPD) identified in the studies reviewed.
Lee et al. [38] performed a wider study enrolling 885 patients from the Korean COPD Subtype Study cohort. The study investigated the relationship between the NLR and the severity of airflow limitation, measured with spirometry (primary outcome) and exacerbations during the first year of follow-up and the previous year according to NLR quartiles (Q1≤1.43, 1.43<Q2≤2.04, 2.04<Q3≤2.94, and 2.94<Q4). Both clinical symptoms and quality of life, assessed using the St George's Respiratory Questionnaire, mMRC and the COPD assessment test significantly deteriorated with increasing NLR quartiles. In addition, participants with higher NLR were more likely to have shorter 6-min walk test distance. As the NLR quartile increased, respiratory functional parameters (FEV1 (%), FEV1 (L), forced vital capacity (FVC; %), FVC (L), and FEV1/FVC) significantly deteriorated, and inhaled corticosteroids were more commonly prescribed in the higher NLR group. NLR values were inversely associated with the severity of airflow limitation determined by FEV1 % predicted and absolute values, also after adjusting for age, sex, BMI, pack-years of smoking and inhaled corticosteroid use. There were no significant differences in the prevalence of comorbidities (cardiovascular diseases, diabetes mellitus, gastro-oesophageal reflux disease and osteoporosis) among the NLR quartiles. This suggests that NLR alterations in COPD are mainly caused by the disease itself.
With regard to COPD exacerbations during the previous year, patients in the fourth quartile of NLR had the highest rate of exacerbations. In univariate logistic regression analysis, a one unit increase in NLR was significantly associated with exacerbation in the first year. In multivariate binary regression, using the first NLR quartile as reference, the fourth NLR quartile was an independent predictor of exacerbations during the first year of follow-up after adjusting for demographic, clinical and functional factors. Furthermore, the predictive ability of NLR during the first year of follow-up (area under the curve (AUC) 0.586, 95% CI 0.528–0.643) was higher than that of white blood cell (WBC) and neutrophil counts. This suggests that the combination of different blood cell populations, expressed by the NLR, might better reflect the inflammatory status in COPD.
In a retrospective study of 100 patients with AECOPD, Taylan et al. [39] confirmed that the NLR was the most sensitive indicator of exacerbation by comparing the ROC curves for NLR, CRP, WBC and erythrocyte sedimentation rate (ERS). In this study, 43 cases were classified as Global Initiative for Chronic Obstructive Lung Disease (GOLD) III and 57 as GOLD IV. Analyses were carried out 3 months after AECOPD and during stable disease; a control group of 80 healthy subjects was also studied. The mean NLR values of controls, stable COPD patients and AECOPD patients were 1.7±0.9, 3.1±2.5 and 7.1±5.4, respectively (p <0.001). Mean NLR values in AECOPD patients were significantly higher than those of patients with stable disease and controls, whereas the NLR values of patients with stable COPD were higher than controls. Pearson correlation analysis showed significant associations between the NLR and CRP, WBC and ERS. A NLR cut-off value of 3.29, one point higher than that proposed by Lee et al. [49], showed higher sensitivity (80.8%) and specificity (77.7%) in predicting exacerbations.
Similar results, albeit using a higher cut-off level, were reported in another prospective study by Farah et al. [40] in 13 patients with stable COPD, 72 patients with AECOPD and 15 healthy volunteers. In the AECOPD group the mean NLR values were, in agreement with previous studies, significantly higher than those of stable COPD patients and controls. Furthermore, there was a positive correlation between NLR and CRP. Using a NLR cut-off of 7.3, the sensitivity and specificity for AECOPD were 76.8% and 73.1%, respectively (AUC 0.793).
Three further studies, one prospective and two retrospective, were conducted in Turkey [41–43]. All these studies included patients with stable COPD and AECOPD, whereas healthy controls were only enrolled in the two retrospective studies by Bilir et al. [42] and In et al. [43]. These studies confirmed significantly higher NLR values in COPD patients when compared to healthy controls, whereas AECOPD patients had significantly higher NLR values when compared to patients with stable COPD. A positive correlation with CRP was observed in all studies. The NLR cut-off points predicting exacerbation in these studies were 3.3, 3.34 and 3.35, respectively [41–43].
NLR and infections in COPD exacerbations
Tanriverdi et al. [44] investigated the ability of NLR to detect infections which trigger AECOPD. The authors enrolled 77 patients with AECOPD and carried out cultures from tracheal aspirates or sputum on admission. Bacteria were isolated in 37.4% of patients and the procalcitonin (PCT), CRP and NLR values between them and the remaining cases were compared. NLR values were significantly higher in patients with bacterial exacerbation. The cut-off NLR value for predicting bacterial infections was 11.5 (sensitivity 61%, specificity was 58%, AUC 0.58); the AUC of PCT was significantly better in predicting bacterial exacerbation [44].
Van de Geijn et al. [45] studied 17 stable COPD, 22 bacterial and 23 non-bacterial AECOPD patients. Patients with AECOPD had significantly higher NLR values when compared to stable patients. Furthermore, AECOPD patients with bacterial infections had higher NLR values than those with non-bacterial infections. The authors tested several effective predictive models, most of them including NLR. Globally, the studies of Tanriverdi et al. [44] and van de Geijn et al. [45] demonstrate that infectious AECOPD are characterised by higher NLR values, probably due to a more intense inflammatory reaction involving the neutrophils rather than the lymphocytes.
NLR as a prognostic marker in COPD
The association between NLR and inflammation in COPD patients was investigated in other studies that focused on its ability to predict in-hospital and post-discharge mortality. Rahimirad et al. [51] retrospectively reviewed 315 hospital charts of COPD patients in two Iranian hospitals. NLR values were higher in patients who died in hospital than in those discharged alive. In addition, mortality rates were higher in patients with NLR ≥4 than those in patients with NLR <4. After adjusting for age, sex, anaemia and thrombocytopenia, a binary logistic regression showed that NLR independently predicted in-hospital mortality. ROC analysis indicated that a NLR with a cut-off value of 4 predicts in-hospital mortality with 87% sensitivity and 40% specificity (AUC 0.717).
In a recent retrospective study, Yao et al. [52] enrolled 303 patients with AECOPD. NLR values were significantly higher in patients who died in hospital than in those who survived. ROC analysis for using NLR to predict in-hospital mortality indicated an optimal cut-off of 6.24 (AUC 0.803), with 81.1% sensitivity and 69.2% specificity. In the entire cohort NLR values correlated positively with serum CRP levels, while the predictive capacity of NLR exceeded that of CRP and platelet to lymphocyte ratio (PLR) individually or in combination.
Xiong et al. [53] performed a prospective, multicentre, parallel-group, case–control study in 368 patients with moderate-to-very severe COPD and 296 controls, assessed every 3 months for 2 years. The baseline NLR values were significantly higher in COPD patients than in controls (2.98±1.89 versus 2.02±1.92). The mean NLR values of the 272 patients who survived during the follow-up were lower than those who died. Both univariate and multivariate analysis showed that NLR independently predicted death. ROC analysis (AUC 0.91) indicated that a NLR cut-off value of 3.3 predicted mortality with sensitivity of 85.8% and specificity of 89.7%. According to this cut-off point, COPD subjects were divided into a high NLR group and a low NLR group; exacerbations and mortality were significantly lower in the latter group. The same cut-off value was found to predict AECOPD in other studies, as previously discussed.
Kumar et al. [54] retrospectively studied 181 patients with acute COPD exacerbation. Death occurred in 16 (9%) patients within 90 days and in 32 (18%) patients within 12 months from discharge. The mean NLR values in patients who died were higher than those of survivors (13±10 versus 7±8). Nevertheless, in this study multivariable logistic regression analysis showed that NLR was not significantly associated with mortality at 90 days from discharge, after adjusting for age, haemoglobin, neutrophil count, PLR and urea.
Some studies assessed the prognostic role of NLR in relation to eosinophilic state. In the retrospective study of Saltürk et al. [55], 647 COPD subjects requiring intensive care unit (ICU) admission were divided into two groups according to their peripheral eosinophil count: 62 in the eosinophilic group (eosinophils >2%) and 585 in the non-eosinophilic group (eosinophils <2%). Blood NLR on ICU admission was significantly lower in the eosinophilic group (median (interquartile range) 4.6 (3.2–6.8)), compared to the non-eosinophilic group (13.0 (17.3–23.1)). Similar findings were observed on discharge and the difference in NLR between admission and discharge was significantly higher in the non-eosinophilic group. Logistic regression analysis, after adjusting for age, sex, BMI, peripheral eosinophilia, long-term oxygen or noninvasive mechanical ventilation applications use, APACHE (Acute Physiology and Chronic Health Evaluation) II score on admission, presence of arrhythmia, septic shock, resistant pathogen and CRP >50 mg·mL−1 showed that a NLR >16 independently predicted mortality. This value is consistently higher than other cut-offs reported in other studies and might reflect the severity of the patients evaluated (figure 1).
A similar retrospective observational cohort study was conducted by Duman et al. [50] in 1704 COPD patients hospitalised with exacerbations (351 patients with eosinophilic COPD as previously defined). Similarly, the NLR on admission was significantly higher in the non-eosinophilic group (6.0 (3.65–10.6)) in comparison to the eosinophilic group (3.73 (2.6–5.15)). NLR values in 504 readmitted patients within 6 months after discharge were higher (5.8 (3.5–10.4)) than those of patients not readmitted (4.9 (3.2–8.8)). The NLR was also higher in those who died in comparison to survivors, in accordance with previous studies. In addition, Cox regression analysis indicated that a NLR ≥7 was positively associated with 6-month mortality.
NLR in patients with COPD and other comorbidities
Three studies evaluated the role of NLR in patients with COPD and other comorbidities. Vaguliene et al. [46] compared NLR values between 139 patients with lung cancer, 55 with lung cancer and stable COPD, 40 with stable COPD and 33 healthy individuals. The NLR was significantly higher in lung cancer patients with COPD (2.92 (0.93–13.42)) or without COPD (3.08 (1.18–8.84)) when compared to non-cancer patients with COPD (2.35 (1.13–4.25)) or healthy individuals (1.86 (1.16–3.21)). However, the difference between COPD patients and healthy subjects was not statistically significant in this study.
Yasar et al. [47] recruited 140 patients with COPD, of whom 63 had metabolic syndrome, and 50 sex- and age-matched healthy controls. Among patients with COPD, the NLR was significantly higher in patients with metabolic syndrome than those without (3.40±0.93 versus 2.07±0.43). Arisou et al. [48] studied the relationship of NLR with atrial conduction time in a case–control study including 40 stable COPD patients and 40 controls. The authors evidenced that tricuspid atrial conduction time was significantly longer in COPD patients than in controls (26.4±11.4 ms versus 17.7±7.7 ms), and a significant positive correlation was identified between tricuspid atrial conduction time and NLR (r=0.38, p<0.001). These findings, as opposed to those of Lee et al. [38], which suggest that specific comorbidities can influence the kinetics of NLR.
NLR and steroid therapy in COPD
Glucocorticoid therapy is not the mainstay COPD treatment! It is the dual bronchodilation which is the pivotal treatment of COPD. Inhaled corticoids are only recommended when frequent exacerbations occur according to GOLD. Nevertheless, this is also questionable and likely that inhaled corticosteroids are essentially useful in eosinophilic COPD [61]. In most of the studies examined to date detailed data about the type, dose and regimen used are lacking. An evaluation of NLR temporal changes in relation to glucocorticoid therapies adopted was conducted by Sørensen et al. [56] in 386 patients with moderate-to-very severe stable COPD followed up for 10 years. The patients were divided into two groups according to systemic glucocorticoid use (the specific type of medications is not reported). Median NLR at baseline was lower in the group not treated with glucocorticoids (2.83 (2.03–4.57)) compared to that with glucocorticoid therapy (5.79 (4.17–9.66)). Furthermore, in this study the NLR was significantly associated with higher mortality in patients without systemic glucocorticoid treatment.
Future perspectives
In recent years, there has been an increasing interest in the potential clinical use of several inflammatory indexes obtained from routine complete blood count tests. In particular, the NLR has been evaluated in a range of pathological conditions, including chronic inflammatory and neoplastic diseases, as a diagnostic marker, an indicator of the intensity of the inflammatory background, and a predictor of the clinical evolution and prognosis of the disease. There are several features which make such indexes particularly attractive for clinical use, including their relatively simple derivation and low associated cost, and wide availability in laboratory practice and healthcare settings. This suggests the possible clinical utility and potential applications of NLR in COPD. In our review, the NLR correlated well with other markers of disease severity and predicted AECOPD and mortality, particularly in specific subgroups such as patients with an elevated eosinophilic state (table 2). Nevertheless, some methodological issues (most studies are retrospective) and the variability of the patient characteristics and comorbidities in different studies do not allow an immediate application of the NLR current clinical practice. Furthermore, data regarding traditional glucocorticoids and other COPD medications, such as long-acting β-agonists, long-acting muscarinic antagonists, phosphodiesterase-4 inhibitors and antibiotics [62, 63], are lacking in most of the studies evaluated. Despite the overall promising results in terms of predictive capacity, larger multicentre prospective studies with longer follow-up, in patients with various phases of the disease and different therapies, are required to better define the clinical role of the NLR in COPD. The latter also mandates the need for the identification and validation of specific NLR cut-off values for diagnosis, disease progression, response to therapy and prognosis.
TABLE 2.
Studies investigating relationships between neutrophil to lymphocyte ratio (NLR) and other biomarkers in chronic obstructive pulmonary disease (COPD)
| First author [ref.] | Main markers investigated | Main findings |
| Günay [36] | CRP, PDW, RDW, MPV | Increased levels of WBC, CRP, RDW and MPV in COPD. Significant positive correlation of NLR with CRP and RDW, and negative correlation with MPV in COPD |
| Furutate [37] | CRP | CRP significantly increased in COPD. Significant positive correlation between NLR and CRP |
| Taylan [39] | CRP, WBC, RDW, PDW, MPV, ESR | Significant correlations with CRP, WBC and ESR. High NLR values in AECOPD patients with low CRP, WBC or ESR levels |
| Farah [40] | CRP, WBC, RDW, MPV, CD64 | RDW, CRP and CD64 significantly elevated in AECOPD. Significant correlations of NLR with CRP and RDW |
| Kurtipek [41] | CRP, PLR | CRP and PLR are significantly higher in AECOPD. Significant positive correlation with CRP. CRP AUC was better than that of NLR, which was better than AUC for PLR |
| Bilir [42] | CRP | Significant positive correlation with CRP. CRP AUC better than AUC for NLR |
| In [43] | CRP, ESR | Significant positive correlation with CRP and ESR |
| Tanriverdi [44] | CRP, PCT | PCT higher in infections. Significant but weak positive correlation with CRP. AUC value of PCT significantly better in predicting bacterial infection compared with the CRP or the NLR |
| van de Geijn [45] | CRP, PCT and others | CRP and PCT higher in AECOPD, especially bacterial ones. Addition of PCT to CRP increased specificity for bacterial AECOPD compared to CRP alone |
| Yasar [47] | CRP, RDW, MPV, PDW | CRP, RDW and PDW were higher while MPV was lower in COPD. CRP, RDW and PDW were higher and MPV was lower in patients who had metabolic syndrome |
| Lee [49] | CRP | CRP was higher in exacerbations than in healthy controls |
| Duman [50] | CRP, platelet/MPV | CRP was significantly higher in the non-eosinophilic group. The platelet/MPV ratio was not different between the two groups. Survival negatively influenced by elevated CRP in the non-eosinophilic group |
| Rahimirad [51] | dNLR, MLR, PLR | dNLR higher in dead patients than in those discharged alive. MLR and PLR not significant in multivariate analysis |
| Yao [52] | CRP, PLR | NLR levels correlated with serum CRP levels in AECOPD. CRP and PLR levels higher in dead patients than in survivors. CRP AUC higher than that of PLR in predicting death |
| Xiong [53] | ESR, EBR, CRP, RDW | EBR higher in COPD than in controls, and in survivors than in dead patients. EBR and CRP are predictors of mortality, not ESR or RDW |
| Kumar [54] | PLR, urea | PLR significantly associated to 90-days mortality, AUC poor |
| Saltürk [55] | CRP, platelet/MPV | CRP >50 mg·mL−1 risk factor for mortality |
| Sørensen [56] | Calprotectin, YKL-40 | Correlation with calprotectin. In the group not treated with systemic glucocorticoids, plasma calprotectin was significantly associated with mortality. In the group treated with systemic glucocorticoids, higher plasma YKL-40 was significantly associated with mortality |
CRP: C-reactive protein; PDW: platelet distribution width; RDW: red cell distribution width; MPV: mean platelet volume; WBC: white blood cell; ESR: erythrocyte sedimentation rate; PLR: platelet to lymphocyte ratio; PCT: procalcitonin; dNLR: derived neutrophil to lymphocyte ratio; MLR: monocyte to lymphocyte ratio; EBR: eosinophil to basophil ratio; AECOPD: acute exacerbation of COPD; AUC: area under the curve.
Conclusions
Blood NLR is a simple, inexpensive, widely available index which has been intensively evaluated in recent years in several clinical applications and in various diseases, including COPD. The studies reviewed showed that the NLR is a valuable predictor of AECOPD and mortality. Furthermore, it correlates well, and in some studies is even more accurate, than other traditional inflammatory indexes (WBC counts alone) or more complex and expensive markers (CRP and calprotectin). Additionally, several NLR cut-off values have been identified in several clinical situations (stable COPD, exacerbations and ICU patients), and with different prediction purposes (COPD itself, exacerbation, infection, and in hospital and general mortality). The routine use of the NLR in clinical practice should be further assessed in large, well-designed, prospective studies.
Footnotes
Conflict of interest: None declared.
Provenance: Submitted article, peer reviewed.
References
- 1.Lozano R, Naghavi M, Foreman K, et al. Global and regional mortality from 235 causes of death for 20 age groups in 1990 and 2010: a systematic analysis for the Global Burden of Disease Study 2010. Lancet 2012; 380: 2095–2128. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Mannino DM, Buist AS. Global burden of COPD: risk factors, prevalence, and future trends. Lancet 2007; 370: 765–773. [DOI] [PubMed] [Google Scholar]
- 3.Burney PG, Patel J, Newson R, et al. Global and regional trends in COPD mortality, 1990–2010. Eur Respir J 2015; 45: 1239–1247. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Rycroft CE, Heyes A, Lanza L, et al. Epidemiology of chronic obstructive pulmonary disease: a literature review. Int J Chron Obstruct Pulmon Dis 2012; 7: 457–494. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Agusti A, Calverley PM, Celli B, et al. Characterisation of COPD heterogeneity in the ECLIPSE cohort. Respir Res 2010; 11: 122. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Vestbo J, Agusti A, Wouters EF, et al. Should we view chronic obstructive pulmonary disease differently after ECLIPSE? A clinical perspective from the study team. Am J Respir Crit Care Med 2014; 189: 1022–1030. [DOI] [PubMed] [Google Scholar]
- 7.Han MK, Agusti A, Calverley PM, et al. Chronic obstructive pulmonary disease phenotypes: the future of COPD. Am J Respir Crit Care Med 2010; 182: 598–604. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.MacNee W. Pathogenesis of chronic obstructive pulmonary disease. Proc Am Thorac Soc 2005; 2: 258–268. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Tuder RM, Petrache I. Pathogenesis of chronic obstructive pulmonary disease. J Clin Invest 2012; 122: 2749–2755. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Turato G, Zuin R, Saetta M. Pathogenesis and pathology of COPD. Respiration 2001; 68: 117–128. [DOI] [PubMed] [Google Scholar]
- 11.Wouters EFM. Local and systemic inflammation in chronic obstructive pulmonary disease. Proc Am Thorac Soc 2005; 2: 26–33. [DOI] [PubMed] [Google Scholar]
- 12.Vestbo J, Hurd SS, Agusti AG, et al. Global strategy for the diagnosis, management and prevention of chronic obstructive pulmonary disease, GOLD executive summary. Am J Respir Crit Care Med 2013; 187: 347–365. [DOI] [PubMed] [Google Scholar]
- 13.Vogelmeier C, Hederer B, Glaab T, et al. Tiotropium versus salmeterol for the prevention of exacerbations of COPD. N Engl J Med 2011; 364: 1093–1103. [DOI] [PubMed] [Google Scholar]
- 14.Seemungal T, Harper-Owen R, Bhowmik A, et al. Respiratory viruses, symptoms, and inflammatory markers in acute exacerbations and stable chronic obstructive pulmonary disease. Am J Respir Crit Care Med 2001; 164: 1618–1623. [DOI] [PubMed] [Google Scholar]
- 15.Wedzicha JA. Role of viruses in exacerbations of chronic obstructive pulmonary disease. Proc Am Thorac Soc 2004; 1: 115–120. [DOI] [PubMed] [Google Scholar]
- 16.Papi A, Luppi F, Franco F, et al. Pathophysiology of exacerbations of chronic obstructive pulmonary disease. Proc Am Thorac Soc 2006; 3: 245–251. [DOI] [PubMed] [Google Scholar]
- 17.Peacock JL, Anderson HR, Bremner SA, et al. Outdoor air pollution and respiratory health in patients with COPD. Thorax 2011; 66: 591–596. [DOI] [PubMed] [Google Scholar]
- 18.Sethi S, Murphy TF. Infection in the pathogenesis and course of chronic obstructive pulmonary disease. N Engl J Med 2008; 359: 2355–2365. [DOI] [PubMed] [Google Scholar]
- 19.Nathan C. Points of control in inflammation. Nature 2002; 420: 846–852. [DOI] [PubMed] [Google Scholar]
- 20.Ishii T, Kida K. Predictors of chronic obstructive pulmonary disease exacerbations. Curr Opin Pulm Med 2014; 20: 138–145. [DOI] [PubMed] [Google Scholar]
- 21.Mantovani A, Cassatella MA, Costantini C, et al. Neutrophils in the activation and regulation of innate and adaptive immunity. Nat Rev Immunol 2011; 11: 519–553. [DOI] [PubMed] [Google Scholar]
- 22.Borregaard N. Neutrophils: from marrow to microbes. Immunity 2010; 33: 657–670. [DOI] [PubMed] [Google Scholar]
- 23.Nathan C. Neutrophils and immunity: challenges and opportunities. Nat Rev Immunol 2006; 6: 173–182. [DOI] [PubMed] [Google Scholar]
- 24.Brusselle GG, Joos GF, Bracke KR. New insights into the immunology of chronic obstructive pulmonary disease. Lancet 2011; 378: 1015–1026. [DOI] [PubMed] [Google Scholar]
- 25.Williams KA, Labidi-Galy SI, Terry KL, et al. Prognostic significance and predictors of the neutrophil-to-lymphocyte ratio in ovarian cancer. Gynecol Oncol 2014; 132: 542–550. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Roxburgh CS, McMillan DC. Role of systemic inflammatory response in predicting survival in patients with primary operable cancer. Future Oncol 2010; 6: 149–163. [DOI] [PubMed] [Google Scholar]
- 27.Kacan T, Babacan NA, Seker M, et al. Could the neutrophil to lymphocyte ratio be a poor prognostic factor for non small cell lung cancers? Asian Pac J Cancer Prev 2014; 15: 2089–2094. [DOI] [PubMed] [Google Scholar]
- 28.Paliogiannis P, Scognamillo F, Bellomo M, et al. Neutrophil to lymphocyte ratio as a predictor of thyroid papillary carcinoma. Act Med Mediterr 2015; 31: 371–375. [Google Scholar]
- 29.Azab B, Bhatt VR, Phookan J, et al. Usefulness of the neutrophil-to-lymphocyte ratio in predicting short- and long-term mortality in breast cancer patients. Ann Surg Oncol 2012; 19: 217–224. [DOI] [PubMed] [Google Scholar]
- 30.Stotz M, Gerger A, Eisner F, et al. Increased neutrophil-lymphocyte ratio is a poor prognostic factor in patients with primary operable and inoperable pancreatic cancer. Br J Cancer 2013; 109: 416–421. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Zahorec R. Ratio of neutrophil to lymphocyte counts: rapid and simple parameter of systemic inflammation and stress in critically ill. Bratisl Lek Listy 2001; 102: 5–14. [PubMed] [Google Scholar]
- 32.Duffy BK, Gurm HS, Rajagopal V, et al. Usefulness of an elevated neutrophil to lymphocyte ratio in predicting long-term mortality after percutaneous coronary intervention. Am J Cardiol 2006; 97: 993–996. [DOI] [PubMed] [Google Scholar]
- 33.Arbel Y, Finkelstein A, Halkin A, et al. Neutrophil/lymphocyte ratio is related to the severity of coronary artery disease and clinical outcome in patients undergoing angiography. Atherosclerosis 2012; 225: 456–460. [DOI] [PubMed] [Google Scholar]
- 34.Andelid K, Bake B, Rak S, et al. Myeloperoxidase as a marker of increasing systemic inflammation in smokers without severe airway symptoms. Respir Med 2007; 101: 888–895. [DOI] [PubMed] [Google Scholar]
- 35.Houghton AM, Rzymkiewicz DM, Ji H, et al. Neutrophil elastase mediated degradation of IRS-1 accelerates lung tumor growth. Nat Med 2010; 16: 219–223. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Günay E, Sarınç Ulaşlı S, Akar O, et al. Neutrophil-to-lymphocyte ratio in chronic obstructive pulmonary disease: a retrospective study. Inflammation 2014; 37: 374–380. [DOI] [PubMed] [Google Scholar]
- 37.Furutate R, Ishii T, Motegi T, et al. The neutrophil to lymphocyte ratio is related to disease severity and exacerbation in patients with chronic obstructive pulmonary disease. Intern Med 2016; 55: 223–229. [DOI] [PubMed] [Google Scholar]
- 38.Lee H, Um SJ, Kim YS, et al. Association of the neutrophil-to-lymphocyte ratio with lung function and exacerbations in patients with chronic obstructive pulmonary disease. PLoS One 2016; 11: e0156511. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Taylan M, Demir M, Kaya H, et al. Alterations of the neutrophil-lymphocyte ratio during the period of stable and acute exacerbation of chronic obstructive pulmonary disease patients. Clin Respir J 2017; 11: 311–317. [DOI] [PubMed] [Google Scholar]
- 40.Farah R, Ibrahim R, Nassar M, et al. The neutrophil/lymphocyte ratio is a better addition to C-reactive protein than CD64 index as a marker for infection in COPD. Panminerva Med 2017; 59: 203–209. [DOI] [PubMed] [Google Scholar]
- 41.Kurtipek E, Bekci TT, Kesli R, et al. The role of neutrophil-lymphocyte ratio and platelet-lymphocyte ratio in exacerbation of chronic obstructive pulmonary disease. J Pak Med Assoc 2015; 65: 1283–1287. [PubMed] [Google Scholar]
- 42.Bilir B, Altintas N, Aydin M, et al. The predictive pole of neutrophil to lymphocyte ratio in chronic obstructive pulmonary disease. Eur J Gen Med 2016; 13: 105–110. [Google Scholar]
- 43.In E, Kuluozturk M, Oner O, et al. The importance of neutrophil-to-lymphocyte ratio in chronic obstructive pulmonary disease. Turk Thorac J 2016; 17: 41–46. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Tanrıverdi H, Ornek T, Erboy F, et al. Comparison of diagnostic values of procalcitonin, C-reactive protein and blood neutrophil/lymphocyte ratio levels in predicting bacterial infection in hospitalized patients with acute exacerbations of COPD. Wien Klin Wochenschr 2015; 127: 756–763. [DOI] [PubMed] [Google Scholar]
- 45.van de Geijn GM, Van Denker S, Meuleman-van Waning V, et al. Evaluation of new laboratory tests to discriminate bacterial from nonbacterial chronic obstructive pulmonary disease exacerbations. Int J Lab Hematol 2016; 38: 616–628. [DOI] [PubMed] [Google Scholar]
- 46.Vaguliene N, Zemaitis M, Lavinskiene S, et al. Local and systemic neutrophilic inflammation in patients with lung cancer and chronic obstructive pulmonary disease. BMC Immunol 2013; 14: 36. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Yasar Z, Buyuksirin M, Ucsular FD, et al. Is an elevated neutrophil-to-lymphocyte ratio a predictor of metabolic syndrome in patients with chronic obstructive pulmonary disease? Eur Rev Med Pharmacol Sci 2015; 19: 956–962. [PubMed] [Google Scholar]
- 48.Arisou A, Memic K, Diken OE, et al. Evaluation of atrial conduction features in stable chronic obstructive pulmonary disease patients and its relationship with neutrophil to lymphocyte ratio. Acta Med Mediterr 2015; 31: 343–349. [Google Scholar]
- 49.Lee SJ, Lee HR, Lee TW, et al. Usefulness of neutrophil to lymphocyte ratio in patients with chronic obstructive pulmonary disease: a prospective observational study. Korean J Intern Med 2016; 31: 891–898. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Duman D, Aksoy E, Agca MC, et al. The utility of inflammatory markers to predict readmissions and mortality in COPD cases with or without eosinophilia. Int J Chron Obstruct Pulmon Dis 2015; 10: 2469–2478. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Rahimirad S, Ghaffary MR, Rahimirad MH, et al. Association between admission neutrophil to lymphocyte ratio and outcomes in patients with acute exacerbation of chronic obstructive pulmonary disease. Tuberk Toraks 2017; 65: 25–31. [PubMed] [Google Scholar]
- 52.Yao C, Liu X, Tang Z. Prognostic role of neutrophil-lymphocyte ratio and platelet-lymphocyte ratio for hospital mortality in patients with AECOPD. Int J Chron Obstruct Pulmon Dis 2017; 12: 2285–2290. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Xiong W, Xu M, Zhao Y, et al. Can we predict the prognosis of COPD with a routine blood test? Int J Chron Obstruct Pulmon Dis 2017; 12: 615–625. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Kumar P, Law S, Sriram KB. Evaluation of platelet lymphocyte ratio and 90-day mortality in patients with acute exacerbation of chronic obstructive pulmonary disease. J Thorac Dis 2017; 9: 1509–1516. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Saltürk C, Karakurt Z, Adiguzel N, et al. Does eosinophilic COPD exacerbation have a better patient outcome than non-eosinophilic in the intensive care unit? Int J Chron Obstruct Pulmon Dis 2015; 10: 1837–1846. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Sørensen AK, Holmgaard DB, Mygind LH, et al. Neutrophil-to-lymphocyte ratio, calprotectin and YKL-40 in patients with chronic obstructive pulmonary disease: correlations and 5-year mortality – a cohort study. J Inflamm (Lond) 2015; 12: 20. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Xanthopoulos A, Giamouzis G, Melidonis A, et al. Red blood cell distribution width as a prognostic marker in patients with heart failure and diabetes mellitus. Cardiovasc Diabetol 2017; 16: 81. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Hu ZD, Sun Y, Guo J, et al. Red blood cell distribution width and neutrophil/lymphocyte ratio are positively correlated with disease activity in primary Sjögren's syndrome. Clin Biochem 2014; 47: 287–290. [DOI] [PubMed] [Google Scholar]
- 59.Taşoğlu Ö, Şahin A, Karataş G, et al. Blood mean platelet volume and platelet lymphocyte ratio as new predictors of hip osteoarthritis severity. Medicine (Baltimore) 2017; 96: e6073. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Tanrikulu CS, Tanrikulu Y, Sabuncuoglu MZ, et al. Mean platelet volume and red cell distribution width as a diagnostic marker in acute appendicitis. Iran Red Crescent Med J 2014; 16: e10211. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.Ernst P, Saad N, Suissa S. Inhaled corticosteroids in COPD: the clinical evidence. Eur Respir J 2015; 45: 525–537. [DOI] [PubMed] [Google Scholar]
- 62.Vestbo J, Anderson J, Brook RD, et al. The study to understand mortality and morbidity in COPD (SUMMIT) study protocol. Eur Respir J 2013; 41: 1017–1022. [DOI] [PubMed] [Google Scholar]
- 63.Vestbo J, Agusti A, Wouters EF, et al. Evaluation of COPD longitudinally to identify predictive surrogate endpoints study investigators. Should we view chronic obstructive pulmonary disease differently after ECLIPSE? A clinical perspective from the study team. Am J Respir Crit Care Med 2014; 189: 1022–1030. [DOI] [PubMed] [Google Scholar]