Abstract
Aims
The presence of pulmonary hypertension (PH) severely aggravates the clinical course of heart failure with preserved ejection fraction (HFpEF). To date, neither established heart failure therapies nor pulmonary vasodilators proved beneficial. This study investigated the efficacy of chronic treatment with the oral soluble guanylate cyclase stimulator riociguat in patients with PH-HFpEF.
Methods and Results
The phase IIb, randomized, double-blind, placebo-controlled, parallel-group, multicentre DYNAMIC trial assessed riociguat in PH-HFpEF. Patients were recruited at five hospitals across Austria and Germany. Key eligibility criteria were mean pulmonary artery pressure ≥25 mmHg, pulmonary arterial wedge pressure >15 mmHg, and left ventricular ejection fraction ≥50%. Patients were randomized to oral treatment with riociguat or placebo (1:1). Patients started at 0.5 mg three times daily (TID) and were up-titrated to 1.5 mg TID. The primary efficacy endpoint was change from baseline to week 26 in cardiac output (CO) at rest, measured by right heart catheterization. Primary efficacy analyses were performed on the full analysis set. Fifty-eight patients received riociguat and 56 patients placebo. After 26 weeks, CO increased by 0.37 ± 1.263 L/min in the riociguat group and decreased by −0.11 ± 0.921 L/min in the placebo group (least-squares mean difference: 0.54 L/min, 95% confidence interval 0.112, 0.971; P = 0.0142). Five patients dropped out due to riociguat-related adverse events but no riociguat-related serious adverse event or death occurred.
Conclusion
The vasodilator riociguat improved haemodynamics in PH-HFpEF. Riociguat was safe in most patients but led to more dropouts as compared to placebo and did not change clinical symptoms within the study period.
Keywords: Heart failure with preserved ejection fraction, Pulmonary hypertension, Riociguat, Soluble guanylate cyclase stimulation, Randomized controlled trial
Structured Graphical Abstract
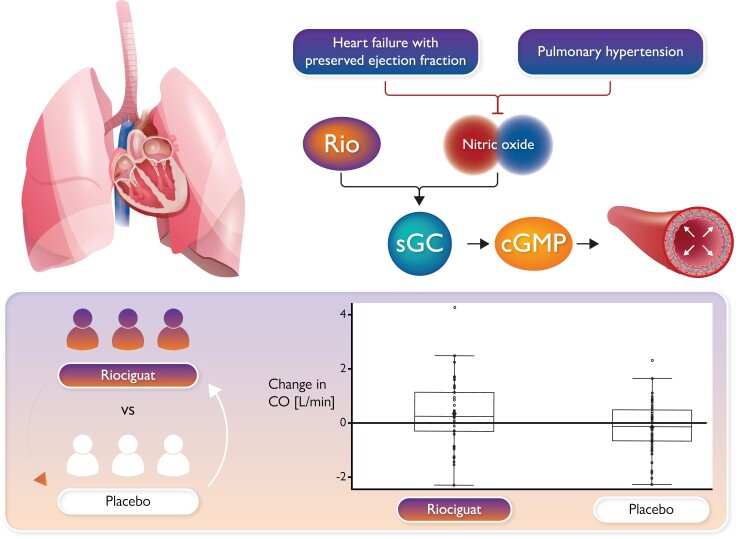
Structured Graphical Abstract.
A total of 114 consecutive patients with pulmonary hypertension associated with heart failure and preserved ejection fraction were randomized to receive riociguat (Rio) or placebo. Riociguat is an orally administered drug that stimulates soluble guanylate cyclase (sGC) directly, independent of nitric oxide, and also sensitizes sGC to endogenous nitric oxide. Stimulation of sGC results in an increase of the cyclic guanosine monophosphate (cGMP) concentration which leads to vasorelaxation. After 26 weeks, the primary efficacy endpoint—cardiac output (CO) at rest measured by right heart catheterization—improved significantly in response to riociguat, along with other haemodynamic parameters.
See the editorial comment for this article ‘Targeting pulmonary hypertension in patients with heart failure and preserved ejection fraction: rather static than DYNAMIC development?’, by Johann Bauersachs and Karen M. Olsson, https://doi.org/10.1093/eurheartj/ehac387.
Introduction
Heart failure (HF) with preserved ejection fraction (HFpEF) accounts for about 50% of all HF cases and is associated with poor quality of life (QoL), substantial healthcare resource utilization, and premature mortality.1 The presence of pulmonary hypertension (PH) contributes to disease progression and is associated with worse outcome in patients with HFpEF.2–4 To date, neither established HF therapies nor pulmonary vasodilators proved effective in the reduction of morbidity and mortality in PH-HFpEF patients.5 Accordingly, guidance is limited to the management of symptoms and comorbidities.6,7
Endothelial dysfunction with decreased nitric oxide (NO) bioavailability is typical for HF and PH-related diseases.8,9 The NO-soluble guanylate cyclase-cyclic guanosine monophosphate pathway mediates vasodilation, anti-inflammatory, antiproliferative, and antifibrotic effects and is involved in pathophysiological processes of PH, cardiac distensibility and relaxation, thereby representing a promising therapeutic target.10–12 Riociguat, an orally administered stimulator of soluble guanylate cyclase (sGC), both sensitizes sGC to endogenous NO and stimulates sGC directly.13,14 Riociguat is currently approved for precapillary forms of PH, i.e. inoperable chronic thromboembolic PH (CTEPH) and pulmonary arterial hypertension (PAH).15,16
The potential benefit of riociguat in postcapillary PH was previously examined in two phase II trials. The left ventricular systolic dysfunction associated with pulmonary hypertension riociguat trial (LEPHT) included 201 patients with PH resulting from HF with reduced ejection fraction (HFrEF) who were treated with riociguat or placebo for 16 weeks.17 The acute haemodynamic effects of riociguat in patients with pulmonary hypertension associated with diastolic heart failure trial (DILATE-1) included 39 patients with PH-HFpEF who were treated with single oral doses of riociguat or placebo.18 Based on the results of these trials, the present riociguat in pulmonary hypertension and heart failure with preserved ejection fraction haemoDYNAMIC trial (DYNAMIC) was designed to characterize haemodynamic and clinical effects, safety, and tolerability of chronic therapy with riociguat in PH-HFpEF.19
Methods
Study design
DYNAMIC was a 26-week multicentre, randomized, double-blind, placebo-controlled, parallel-group, clinical phase IIb trial. The design of DYNAMIC has been published previously.19 Briefly, patients were screened for eligibility during on-site visits at five hospitals across Austria (Vienna, Linz, Salzburg, and Graz) and Germany (Heidelberg). The study protocol was approved by the ethics committee of the Medial University of Vienna and local ethics committees (date of vote: 10 October 2014, reference number: 1570/2014; https://ekmeduniwien.at/core/catalog/2014/, EudraCT number: 2014-003055). The study was completed and all data were obtained following the principles of Good Clinical Practice. The trial was registered at www.clinicaltrials.gov (unique identifier: NCT02744339). The data shared will not be made universally available to any third party. Any requests should be directed towards the corresponding author.
Patients
Patients aged 18–80 years diagnosed with symptomatic PH-HFpEF, defined by (i) a left ventricular ejection fraction (LVEF) ≥50%, diagnosed by transthoracic echocardiography (TTE) or catheterization within 30 days before randomization, (ii) World Health Organization functional class (WHO-FC) II-IV, (iii) mean pulmonary artery pressure (PAPmean) ≥25 mmHg at rest, and (iv) pulmonary arterial wedge pressure (PAWP) >15 mmHg at rest, measured by right heart catheterization (RHC) within 12 weeks before randomization, were eligible, if concomitant medication which may affect haemodynamic parameters was not changed until study enrolment. Furthermore, clinical stability and unchanged medical treatment >30 days (>7 days for diuretics) were required prior to enrolment. Anaemia and thyrotoxicosis were contraindications for the performance of baseline right and left heart catheterization.19 Differential diagnoses such as cardiac amyloidosis, HFrEF, CTEPH, PAH, and significant pulmonary disease were ruled out according to accepted diagnostic procedures and guidelines.6,20 Patients with significant coronary artery disease (i.e. angina Canadian Cardiovascular Society ≥ III, need for percutaneous coronary intervention, coronary artery bypass graft ≤90 days) were excluded.19 All patients gave written informed consent in accordance with the Declaration of Helsinki.
Randomization and masking
A random list was created with unique 4-digit random numbers for medication kits stratified by dose-strength using a validated Statistical Analysis System (SAS) programme based on the in-built function RANUNI for generation of uniformly distributed variables. This list was uploaded into the electronic case report form (eCRF) system. Separately, random numbers for patients were generated stratified by centre following a 1:1 allocation ratio for treatment with riociguat or placebo. Upon request, the eCRF assigned the correct medication kit number to the respective patient automatically. Drug appearance and packaging of riociguat and placebo were identical and therefore indistinguishable. Information on treatment allocation was masked and remained blinded to all patients, investigators, study staff, and monitors until study completion and closure of the database.
In the event of a suspected unexpected serious adverse reaction (SUSAR) and relatedness to blinded treatment, the SUSAR with the patient’s actual treatment was reported to health authorities, ethics committees, and investigators.
Procedures
Details on the study phase have been published before.19 Briefly, after a pre-study phase of up to 4 weeks, the study phase consisted of an 8-week (up-)titration phase followed by 18 weeks of fixed-dose treatment. Patients started with a dose of 0.5 mg riociguat three times daily (TID) or placebo and were (up-)titrated to doses of 1.0 and 1.5 mg TID at three subsequent visits over the following 8 weeks in accordance with clinical condition and systolic blood pressure (SBP) (see Supplementary material online, Figure S1).
RHC was performed within 12 weeks before screening and at week 26. A pulmonary artery catheter was inserted via the femoral vein, zeroed to atmospheric pressure, and referenced at the midthoracic level. Cardiac output (CO) was assessed using the thermodilution method according to current guidelines20 using cold water injections until three measurements with a variability <10% were obtained; CO was calculated as a mean of these three measurements. All pressures including PAWP were recorded as averages of eight time-pressure integral derivations during several respiratory cycles.21
TTE was performed within 30 days before screening and at week 26. WHO-FC, N-terminal prohormone B-type natriuretic peptide (NT-proBNP), and 6-minute walking distance (6MWD) were assessed at baseline, after 8 and 26 weeks. The European Quality of Life five dimensions (EQ-5D) questionnaire22 and the heart failure-specific Minnesota Living with Heart Failure Questionnaire (MLHFQ)23 were assessed at baseline and after 26 weeks. Clinical assessments and laboratory testing were performed at screening, at baseline and at weeks 2, 4, 8, 16, and 26. After regular or premature discontinuation, a 30-day safety follow-up visit was performed. (Serious) treatment-emergent adverse events (TEAEs) were documented and followed up throughout the treatment period and until a 30-day safety follow-up visit.
Outcomes
The primary efficacy variable was the change in CO at rest from baseline to week 26 of treatment, measured by RHC. Secondary efficacy variables included the change from baseline to week 26 in pulmonary vascular resistance (PVR), systemic vascular resistance (SVR), transpulmonary pressure gradient (TPG), and PAWP, measured by RHC, as well as change in serum levels of NT-proBNP, and improvements by ≥1 WHO-FC. Exploratory analyses included changes from baseline to week 26 in 6MWD, EQ-5D, MLHFQ, and other cardiopulmonary variables derived from RHC and TTE.
Safety was assessed by reporting serious and non-serious TEAEs, changes in vital signs, 12-lead electrocardiogram, physical examination, and laboratory test results. All blood samples for safety (haematology, clinical chemistry, coagulation) were analysed by local laboratories of the participating hospitals.
Statistical analysis
For the determination of sample size, the significance level was set to 2.5% one-sided. Demanding 80% power to detect a mean difference of ≥0.6 L/min between riociguat and placebo with regard to the primary endpoint and assuming a standard deviation (SD) of the change in CO of 1.0 L/min, 45 evaluable patients were needed per treatment group. Allowing for a drop-out rate of 20%, 114 patients (57 per group) were required.
The safety analysis set (SAF) included all randomized patients who received at least one dose of study drug. Patients of the SAF belonged to the full analysis set (FAS), if they satisfied the major study entry criteria, had a valid measurement of the primary endpoint at baseline, and at least one valid post-baseline measure of efficacy at week 8 or 26. Patients of the FAS belonged to the per-protocol set (PPS), if they showed sufficient compliance (≥80% of study drug intake), had a valid post-treatment measurement of the primary endpoint at week 26, and were without any protocol violation that would have interfered with the interpretation of efficacy data. Primary efficacy analyses were performed on the FAS. Additionally, the primary and secondary endpoints were analysed on the PPS. All endpoints were analysed in a descriptive way using standard statistical methods.
An extremely high CO value of 17.6 L/min was reported at week 26 for one patient in the riociguat group (normal range: 4.0–8.0 L/min). Since this value was judged as clinically inconceivable before unblinding, it was excluded from the analysis. A sensitivity analysis revealed that inclusion would overestimate the effect of riociguat. The primary efficacy variable was compared between treatment regimens using an analysis of covariance (ANCOVA) model with baseline value as covariate, centre, treatment regimen, and the interaction term of centre and treatment regimen as fixed effects. Vienna was included in the model as one centre, and the other centres were pooled to a second centre due to low sample sizes (see Supplementary material online, Table S1). If the F-test for the treatment-by-centre interaction was not significant at the 0.2 level, the interaction term was removed from the model. The primary comparison was a one-sided test at the 2.5% significance level for the difference in treatment effects between riociguat and placebo. Analogously, secondary haemodynamic endpoints were subjected to separate ANCOVAs as described above without adjustment for multiplicity. Results of other endpoints (i.e. cardiopulmonary variables, 6MWD, EQ-5D, and MLHFQ) were analysed descriptively. Changes concerning the WHO-FC were evaluated by dichotomizing into two outcomes ‘improvement by at least one class’ and ‘no improvement or worsening’. Additionally, subgroup analyses were descriptively performed for patients with combined pre- and postcapillary PH [cpcPH, i.e. PVR >3 Wood units (WU, mmHg·min·L−1) ± diastolic pulmonary gradient (DPG) ≥7 mmHg] vs. patients with isolated postcapillary PH (ipcPH).The analysis of safety data was descriptively performed on the SAF. No imputation of missing values was performed. Statistical analysis was carried out using the SAS® software package version 9.4 (SAS Institute Inc., Cary, NC, USA). Trial data for patient safety were reviewed by a data safety monitoring committee at regular intervals.
Results
Patients
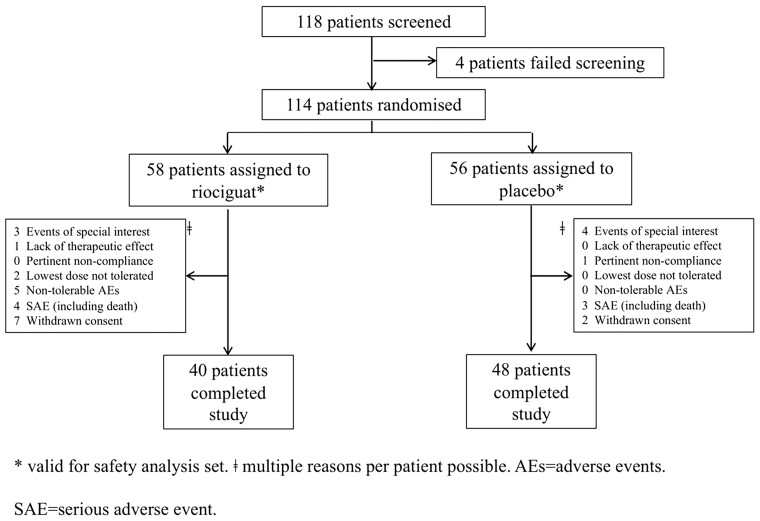
From 17 March 2016, until 29 February 2020, 118 patients were screened for eligibility and randomized at five centres across Austria and Germany. Thereof, 114 patients were randomized and received study treatment (Figure 1): 58 patients were assigned to riociguat and 56 patients to placebo. In total, 40 patients on riociguat and 48 patients on placebo completed the study. Eighteen patients in the riociguat (31.0%) and eight patients in the placebo group (14.3%) discontinued study procedures prematurely. Primary reasons for premature termination are specified in Supplementary material online, Table S2. Fifty patients on riociguat and 54 patients on placebo were included in the FAS. Of these, 36 patients on riociguat and 47 patients on placebo were included in the PPS. Of all patients included in the FAS, 40 patients on riociguat and 48 patients on placebo completed the study, i.e. they attended the visit at week 26. Of these, 38 riociguat-treated and 48 placebo-treated patients provided data for the primary endpoint. Major reasons for exclusion from PPS were non-availability of post baseline efficacy data and non-compliance.
Figure 1.
Trial profile. *Valid for safety analysis set. ‡Multiple reasons per patient possible. AEs, adverse events; SAE, serious adverse event.
Table 1 summarizes demographic characteristics. There were 46 (79.3%) female patients on riociguat compared to 37 (66.1%) on placebo. Measurements of mean LVEF at 60.6 ± 6.3%, PAPmean at 36.1 ± 9.86 mmHg, and PAWP at 20.8 ± 4.86 mmHg reflect the presence of PH-HFpEF. The mean treatment duration of patients who completed the study (FAS) was 186 ± 16.9 days for riociguat-treated and 186 ± 18.8 days for placebo-treated patients.
Table 1.
Patient demographics (safety analysis set)
| Riociguat N = 58 | Placebo N = 56 | Total N = 114 | |
|---|---|---|---|
| Sex | |||
| Female | 46 (79.3) | 37 (66.1) | 83 (72.8) |
| Male | 12 (20.7) | 19 (33.9) | 31 (27.2) |
| Age (years) | 70.6 ± 8.0 | 72.1 ± 8.5 | 71.4 ± 8.2 |
| Race | |||
| White | 57 (98.3) | 56 (100) | 113 (99.1) |
| Other | 1 (1.7) | 0 (0.0) | 1 (0.9) |
| BMI (kg/m2) | 32.1 ± 6.4 | 30.3 ± 6.4 | 31.2 ± 6.5 |
| LVEF (%)a | 61.0 ± 6.7 | 60.1 ± 6.0 | 60.6 ± 6.3 |
| PAPmean (mmHg)a | 36.3 ± 10.23 | 35.9 ± 9.60 | 36.1 ± 9.86 |
| PAWP (mmHg)a | 20.3 ± 4.59 | 21.2 ± 5.11 | 20.8 ± 4.86 |
| GFR (mL/min/1.73 m2) | 63.4 ± 21.9 | 61.7 ± 20.1 | 62.6 ± 20.9 |
| Comorbidities | |||
| Arterial hypertension | 38 (65.5) | 34 (60.7) | 72 (63.2) |
| Atrial fibrillation | 36 (62.1) | 37 (66.1) | 73 (64.0) |
| Chronic kidney diseaseb | 27 (46.6) | 27 (48.2) | 54 (47.4) |
| COPD | 5 (8.6) | 5 (8.9) | 10 (8.8) |
| Coronary artery disease | 7 (12.1) | 8 (14.3) | 15 (13.2) |
| Diabetes mellitus | 16 (27.6) | 16 (28.6) | 32 (28.1) |
| Obstructive sleep apnoea | 1 (1.7) | 2 (3.6) | 3 (2.6) |
| Valvular heart diseasec | 9 (15.5) | 4 (7.1) | 13 (11.4) |
| Drug and device therapy | |||
| Pacemaker | 8 (13.8) | 9 (16.1) | 17 (14.9) |
| ACEI or ARB | 42 (72.4) | 40 (71.4) | 82 (71.9) |
| β-blockers | 43 (74.1) | 42 (75.0) | 85 (74.6) |
| MRA | 40 (69.0) | 42 (75.0) | 82 (71.9) |
| Loop diuretics | 40 (69.0) | 45 (80.4) | 85 (74.6) |
| Thiazide like diuretics | 7 (12.1) | 4 (7.1) | 11 (9.6) |
| Antiplatelet drugs | 8 (13.8) | 13 (23.2) | 21 (18.4) |
| Vitamin K antagonists | 13 (22.4) | 11 (19.6) | 24 (21.1) |
| Direct oral anticoagulants | 34 (58.6) | 31 (55.4) | 65 (57.0) |
Data are numbers (%) or mean ± SD.
Data based on full analysis set.
Defined as GFR <60 mL/min/1.73 m2.
Defined as previous surgical or interventional repair or replacement.
ACEI, angiotensin-converting enzyme inhibitor; ARB, angiotensin II receptor blocker; BMI, body mass index; COPD, chronic obstructive pulmonary disease; GFR, glomerular filtration rate; LVEF, left ventricular ejection fraction; MRA, mineralocorticoid receptor antagonist; PAPmean, mean pulmonary arterial pressure; PAWP, pulmonary arterial wedge pressure; SD, standard deviation.
As the study drug dose was titrated based on clinical condition and SBP, 40 patients (69.0%) received 1.5 mg, 10 patients (17.2%) 1.0 mg, and 8 patients (13.8%) 0.5 mg riociguat TID after 26 weeks. Baseline values for haemodynamic parameters, NT-proBNP, WHO-FC, 6MWD, EQ-5D, and MHLFQ are shown in Table 2 and Supplementary material online, Table S4.
Table 2.
Baseline characteristics (full analysis set)
| Riociguat | Placebo | Total | |
|---|---|---|---|
| Haemodynamic parameters | |||
| CO (L/min) | 5.16 ± 1.131 | 5.05 ± 1.494 | 5.11 ± 1.327 |
| TPG (mmHg) | 16.0 ± 8.07 | 14.8 ± 7.69 | 15.4 ± 7.87 |
| PVR (dyn.s.cm−5) | 257.4 ± 139.1 | 245.7 ± 128.7 | 251.4 ± 133.3 |
| SVR (dyn.s.cm−5) | 1434 | 1422.5 ± 455.8 | 1428.2 ± 479.8 |
| .1 ± 508.3 | |||
| DPG (mmHg) | 2.6 ± 6.75 | 1.6 ± 6.86 | 2.1 ± 6.79 |
| RAPmean (mmHg) | 12.2 ± 5.52 | 12.8 ± 4.99 | 12.5 ± 5.23 |
| PAPmean (mmHg) | 36.3 ± 10.23 | 35.9 ± 9.60 | 36.1 ± 9.86 |
| PVRi (dyn.s.cm−5.m2) | 517.0 ± 297.6 | 465.3 ± 235.2 | 490.1 ± 266.8 |
| Cardiac index (L/min/m2) | 2.63 ± 0.497 | 2.63 ± 0.695 | 2.63 ± 0.605 |
| Stroke volume (mL) | 73.7 ± 23.5 | 72.8 ± 22.7 | 73.2 ± 23.0 |
| Haemoglobin (g/dL) | 12.4 ± 1.64 | 12.5 ± 1.91 | 12.5 ± 1.78 |
| Biomarker | |||
| NT-proBNP (pg/mL)a | 794.1 (264.8–1400) | 1065 (366.8–1722) | 859.0 (359.0–1593) |
| Clinical parameters | |||
| WHO functional class | |||
| II | 28 (56.0) | 26 (48.1) | 54 (51.9) |
| III | 22 (44.0) | 28 (51.9) | 50 (48.1) |
| 6MWD (m) | 308.83 ± 109.41 | 342.57 ± 116.14 | 326.21 ± 113.62 |
| EQ-5Db | |||
| Anxiety/depression | 33 (67.3) | 27 (55.1) | 60 (61.2) |
| Mobility | 16 (33.3) | 18 (36.0) | 34 (34.7) |
| Pain/discomfort | 14 (29.2) | 19 (38.8) | 33 (34.0) |
| Self-care | 43 (89.6) | 43 (87.8) | 86 (88.7) |
| Usual activities | 18 (36.7) | 23 (46.0) | 41 (41.4) |
| EQ-5D, VAS | 58.08 ± 18.278 | 58.17 ± 17.679 | 58.13 ± 17.888 |
| MLHF score | |||
| Total | 38.4 ± 19.50 | 39.4 ± 18.12 | 38.9 ± 18.73 |
| Physical dimension | 21.8 ± 9.89 | 21.0 ± 8.02 | 21.4 ± 8.95 |
| Emotional dimension | 7.7 ± 6.54 | 8.3 ± 6.23 | 8.0 ± 6.36 |
Data are n (%) or mean ± SD.
Data are median (IQR).
No problems’ in respective categories were reported. The EQ-5D provides a descriptive health profile composed of five dimensions (i.e. mobility, self-care, every-day activities, pain/discomfort and anxiety/depression) with three possible answers to each item (no/moderate/severe problems). In addition, patients rate their general health state on a visual analogue scale (range from 0–100, 100 signifying the best state). The MLHF assesses health-related QoL of patients with HF and is composed of two domains (i.e. physical and emotional dimensions). Scores are collected using 6-point Likert scales (lower scores indicating improvement) for 21 items and the sum of item responses is calculated to obtain total and dimension scores.
6MWD, 6-minute walking distance; CO, cardiac output; DPG, diastolic pressure gradient; EQ-5D, European Quality of Life five dimensions; IQR, interquartile range; MLHF, Minnesota Living with Heart Failure; NT-proBNP, N-terminal pro-brain natriuretic peptide; PAPmean, mean pulmonary arterial pressure; PVR, pulmonary vascular resistance; PVRi, pulmonary vascular resistance index; QoL, Quality of Life; RAPmean, mean right atrial pressure; SD, standard deviation; SVR, systemic vascular resistance; TPG, transpulmonary pressure gradient; VAS, visual analogue scale; WHO, World Health Organization.
Right heart haemodynamics
Results of the primary efficacy variable are shown in Table 3 and Figure 2. After 26 weeks, CO increased by 0.37 ± 1.263 L/min in the riociguat group and decreased by −0.11 ± 0.921 L/min in the placebo group. ANCOVA showed a significant mean difference (based on least-square [LS] means) between treatment groups of 0.54 L/min (95% confidence interval [CI]: 0.112, 0.971; P = 0.0142) and thus confirmed superiority of riociguat over placebo. Heart rate (HR) remained stable with mean changes from baseline of +1.9 ± 20.8 bpm in the riociguat and −0.9 ± 20.0 bpm in the placebo group. Analysis based on PPS showed a comparable result for CO change with a mean difference between treatments of 0.50 L/min (95% CI: 0.058, 0.937; P = 0.0271).
Table 3.
Change from baseline to week 26 in primary and secondary hemodynamic parameters (full analysis set)
| Riociguat | Placebo | LS means | LS means difference | p-value | |||
|---|---|---|---|---|---|---|---|
| N | Change | N | Change | Rio/Plac | Rio − Plac (95% CI) | ||
| Primary endpoint | |||||||
| CO (L/min) | 38 | 0.37 ± 1.263 | 48 | −0.11 ± 0.921 | 0.41/−0.13 | 0.54 (0.112; 0.971) | 0.0142 |
| Secondary endpoints | |||||||
| PAWP (mmHg) | 39 | −0.2 ± 6.74 | 48 | −0.4 ± 8.33 | −0.579/−0.663 | 0.084 (−3.239; 3.406) | 0.9601 |
| TPG (mmHg) | 39 | −2.5 ± 5.89 | 47 | 0.0 ± 7.10 | −3.225/2.444 | −5.669 (−9.251; −2.086) | 0.0023 |
| PVR (dyn.s.cm−5) | 38 | −38.1 ± 126.8 | 47 | 6.6 ± 137.7 | −63.584/45.292 | −108.88 (−186.89; −30.86) | 0.0068 |
| SVR (dyn.s.cm−5) | 33 | −91.1 ± 500.0 | 41 | −54.9 ± 392.1 | −103.61/−49.337 | −54.27 (−236.99; 128.44) | 0.5555 |
Data are mean ± SD.
CI, confidence interval; CO, cardiac output; LS, least squares; PAWP, pulmonary arterial wedge pressure; Plac, placebo; PVR, pulmonary vascular resistance; Rio, riociguat; SD, standard deviation; SVR, systemic vascular resistance; TPG, transpulmonary pressure gradient.
Figure 2.
Change in cardiac output after 26 weeks of treatment (full analysis set). Individual changes in cardiac output after 26 weeks of treatment were plotted for each treatment group (excluding the implausible value) and are presented by open circles. The box presents the interquartile range and the whiskers the minimum and maximum values. The median is presented by the line and the mean by the black diamond.
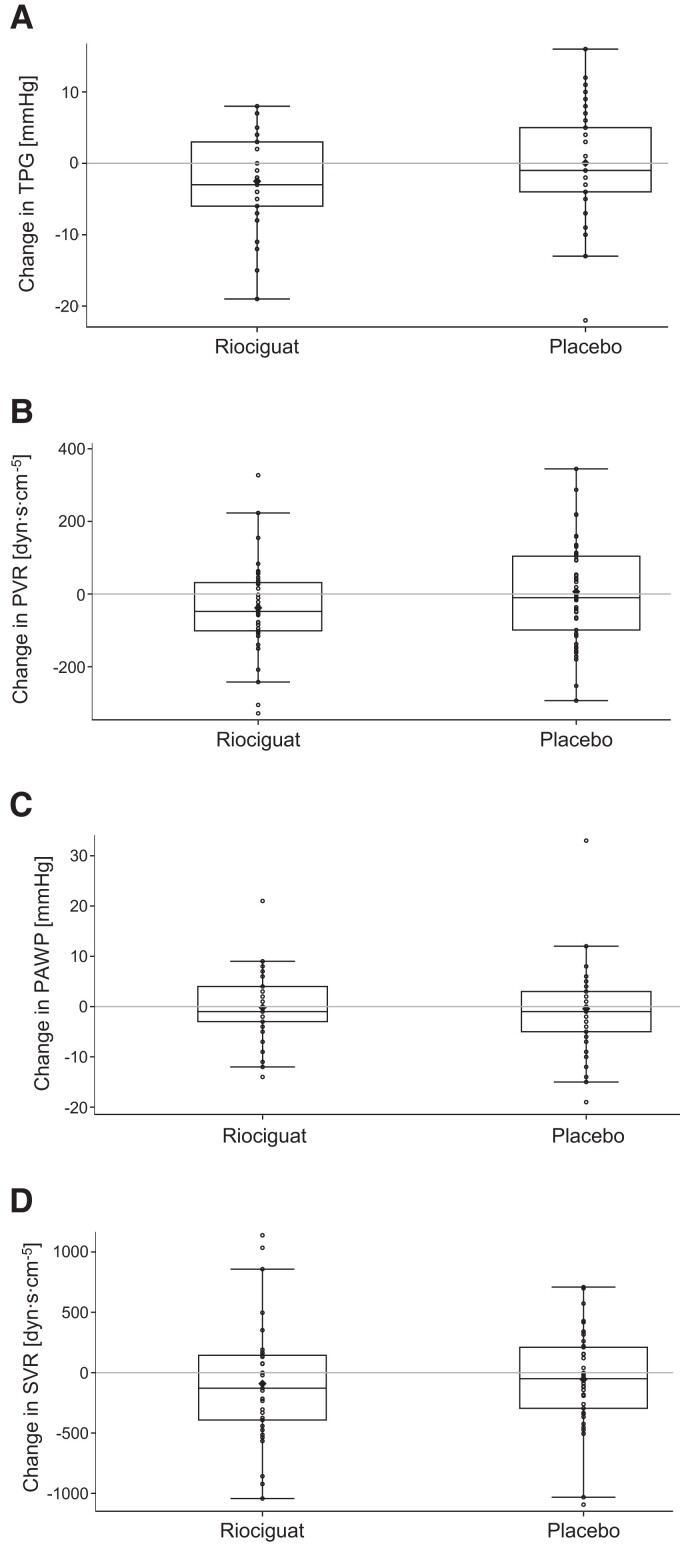
Results for secondary haemodynamic efficacy variables after 26 weeks of treatment are shown in Table 3 and Figure 3. Mean TPG was reduced by −2.5 ± 5.89 mmHg in the riociguat group and remained unchanged in the placebo group at 0.0 ± 7.10 mmHg (LS mean difference: −5.669 mmHg; 95% CI: −9.251, −2.086; P = 0.0023). Mean PVR decreased by −38.1 ± 126.8 dyn·s·cm−5 in the riociguat group and increased by 6.6 ± 137.7 dyn·s·cm−5 in the placebo group (LS mean difference: −108.88 dyn·s·cm−5; 95% CI: −186.89, −30.86; P = 0.0068). Analyses based on PPS showed comparable results with a mean difference between treatments of −6.221 mmHg (95% CI: −9.905, −2.537; P = 0.0012) for TPG, and −110.81 dyn·sec·cm−5 (95% CI: −189.41, −32.21; P = 0.0063) for PVR.
Figure 3.
Change in secondary haemodynamic parameters after 26 weeks of treatment (full analysis set). Individual changes in transpulmonary pressure gradient (A), pulmonary vascular resistance (B), pulmonary arterial wedge pressure (C), and systemic vascular resistance (D) after 26 weeks of treatment were plotted for each treatment group and are presented by open circles. The box presents the interquartile range and the whiskers the minimum and maximum values. The median is presented by the line and the mean by the black diamond.
PAWP and SVR were unaffected by treatment with riociguat. Mean changes of PAWP were −0.2 ± 6.74 mmHg in the riociguat group and −0.4 ± 8.33 mmHg in the placebo group (LS mean difference: 0.084 mmHg; 95% CI: −3.239, 3.406; P = 0.9601), and mean changes of SVR were −91.1 ± 500.0 dyn·sec·cm−5 in the riociguat group and −54.9 ± 392.1 dyn·s·cm−5 in the placebo group (LS mean difference: −54.27 dyn·s·cm−5; 95% CI: −236.99, 128.44; P = 0.5555). Results obtained from PPS-based analyses were similar (data not shown). Other cardiopulmonary, clinical and laboratory parameters after 26 weeks of treatment are shown in Table 4 and Supplementary material online, Table S5. Exploratory parameters derived from RHC and TTE were only slightly changed after 26 weeks of treatment. Exploratory subgroup analyses comparing patients with cpcPH (i.e. PVR >3 WU ± DPG ≥7 mmHg) vs. ipcPH revealed a more pronounced beneficial effect of riociguat on haemodynamic parameters in cpcPH (see Supplementary material online, Tables S7 and S8).
Table 4.
Change from baseline to week 26 in haemodynamic parameters, biomarker, and clinical parameters (full analysis set)
| Riociguat | Placebo | |||
|---|---|---|---|---|
| N | Change | N | Change | |
| Haemodynamic parameters | ||||
| RAPmean (mmHg) | 38 | −0.21 ± 6.502 | 48 | −0.98 ± 4.128 |
| PAPmean (mmHg) | 39 | −2.69 ± 7.726 | 48 | −0.38 ± 6.245 |
| PVRi (dyn.s.cm−5.m2) | 35 | −65.26 ± 266.7 | 46 | 7.14 ± 265.2 |
| Cardiac index (L/min/m2) | 35 | 0.14 ± 0.707 | 46 | −0.03 ± 0.471 |
| DPG (mmHg) | 39 | −1.8 ± 5.90 | 47 | −1.1 ± 7.33 |
| Stroke volume (mL) | 39 | 4.3 ± 32.8 | 45 | −3.1 ± 25.5 |
| Haemoglobin (g/dL) | 41 | −0.63 ± 1.316 | 48 | 0.13 ± 1.212 |
| Biomarker | ||||
| NT-proBNPa (pg/mL) | 36 | 60.35 (−184.20–238.00) | 43 | 76.00 (−169.50–533.00) |
| Clinical parameters | ||||
| 6MWD (m) | 37 | 21.26 ± 109.146 | 40 | 10.30 ± 55.016 |
| EQ-5D, VAS | 39 | 6.21 ± 19.600 | 39 | −0.17 ± 19.466 |
| MLHF score | ||||
| Total | 22 | −4.60 ± 15.120 | 22 | −9.50 ± 16.170 |
| Physical dimension | 38 | −3.10 ± 7.990 | 35 | −2.10 ± 6.540 |
| Emotional dimension | 38 | −0.10 ± 5.100 | 41 | −1.40 ± 7.440 |
Data are mean ± SD.
Data are median (IQR).
6-MWD, 6-minute walking distance; DPG, diastolic pressure gradient; EQ-5D = European Quality of Life five dimensions; IQR, interquartile range; MLHF, Minnesota Living with Heart Failure; NT-proBNP, N-terminal pro-brain natriuretic peptide; PAPmean, mean pulmonary arterial pressure; PVRi, pulmonary vascular resistance index; RAPmean, mean right atrial pressure; SD, standard deviation; SVR, systemic vascular resistance; TPG, transpulmonary pressure gradient; VAS, visual analogue scale.
Biomarker and clinical variables
Median serum levels of NT-proBNP slightly increased in both groups (riociguat: 60.35 pg/mL, IQR: −184.20, 238.00; placebo: 76.00 pg/mL, IQR: −169.50, 533.00). 6MWD improved by 21.26 ± 109.146 m in the riociguat group vs. 10.30 ± 55.016 m in the placebo group. With regard to EQ-5D, patients neither reported apparent changes with respect to health states on the visual analogue scale after 26 weeks of treatment (riociguat: 6.21 ± 19.60; placebo: −0.170 ± 19.47; Table 4), nor regarding the patient-reported health profiles (see Supplementary material online, Table S3). Similar, evaluation of the MLHFQ revealed no changes in QoL (Table 4). At week 26, 25.0% of patients on riociguat and 21.7% of patients on placebo reported symptomatic improvement by at least one WHO-FC (see Supplementary material online, Table S3). Mean changes in SBP differed slightly between groups (riociguat: −5.1 ± 28.80 mmHg; placebo: −2.3 ± 25.12 mmHg), whereas results were comparable for diastolic blood pressure (DBP) (riociguat: −2.2 ± 16.35 mmHg; placebo: −2.3 ± 13.74 mmHg).
Safety and tolerability
During the study period, one patient (1.7%) in the riociguat group died from cardiac arrest which was not judged as study drug-related. In the placebo group, two patients (3.6%) died during the study: one patient suffered cardiac arrest and for the second patient, the reason of death was not retraceable. Both patients who reportedly died due to cardiac arrest had no medical history of coronary artery disease.
Four patients (8.0%) in the riociguat group and five (9.3%) in the placebo group had an adverse event (AE) of special interest, indicating clinical worsening. Six patients (12.0%) in the riociguat group and four (7.4%) in the placebo group suffered either death from a cardiovascular cause or were hospitalized for a cardiovascular event. Study drug-related TEAEs were reported for 19 patients (32.8%) on riociguat and for 12 patients (21.4%) on placebo, of which one patient (1.8%) on placebo but none on riociguat had a serious TEAE (hypotension).
Most TEAEs were of mild or moderate intensity in both treatment groups. The most frequent TEAEs were peripheral oedema (riociguat: 29.3%; placebo: 30.4%), dyspnoea (riociguat: 25.9%; placebo: 21.4%), and hypotension (riociguat 22.4%; placebo: 12.5%). Nineteen (32.8%) study drug-related TEAEs occurred with riociguat and 12 (21.4%) with placebo. The most reported study drug-related TEAE for both treatments was hypotension (Table 5).
Table 5.
Incidence of the most frequent and most important treatment-emergent adverse events (safety analysis set)
| Event, N (%) | Riociguat (N = 58) | Placebo (N = 56) | Total (N = 114) | ||
|---|---|---|---|---|---|
| All | Drug-related | All | Drug-related | ||
| Any AE | 54 (93.1) | 19 (32.8) | 55 (98.2) | 12 (21.4) | 109 (95.6) |
| Most frequent AEs | |||||
| Oedema peripheral | 17 (29.3) | 1 (1.7) | 17 (30.4) | 1 (1.8) | 34 (29.8) |
| Dyspnoea | 15 (25.9) | 0 (0.0) | 12 (21.4) | 0 (0.0) | 27 (23.7) |
| Hypotension | 13 (22.4) | 12 (20.7) | 7 (12.5) | 5 (8.9) | 20 (17.5) |
| Cough | 11 (19.0) | 0 (0.0) | 7 (12.5) | 1 (1.8) | 18 (15.8) |
| Dizziness | 5 (8.6) | 1 (1.7) | 10 (17.9) | 1 (1.8) | 15 (13.2) |
| Fatigue | 7 (12.1) | 0 (0.0) | 7 (12.5) | 2 (3.6) | 14 (12.3) |
| Headache | 6 (10.3) | 1 (1.7) | 6 (10.7) | 0 (0.0) | 12 (10.5) |
| Cardiac failure | 3 (5.2) | 0 (0.0) | 8 (14.3) | 1 (1.8) | 11 (9.6) |
| Diarrhoea | 5 (8.6) | 0 (0.0) | 6 (10.7) | 1 (1.8) | 11 (9.6) |
| Gastroesophageal reflux disease | 7 (12.1) | 3 (5.2) | 3 (5.4) | 0 (0.0) | 10 (8.8) |
| Nasopharyngitis | 4 (6.9) | 0 (0.0) | 6 (10.7) | 0 (0.0) | 10 (8.8) |
| Hyperkalaemia | 5 (8.6) | 1 (1.7) | 5 (8.9) | 0 (0.0) | 10 (8.8) |
| Vertigo | 4 (6.9) | 1 (1.7) | 6 (10.7) | 2 (3.6) | 10 (8.8) |
| Most important AEs | |||||
| Syncope | 4 (6.9) | 2 (3.6) | 6 (5.3) | ||
| Death | 1 (1.7) | 2 (3.6) | 3 (2.6) | ||
| Cardiovascular | 1 (1.7) | 2 (3.6) | 3 (2.6) | ||
| Study drug-related | 0 (0.0) | 0 (0.0) | 0 (0.0) | ||
Most frequent AEs are sorted according to total frequency. Cut-off: N≥ 10 in total.
AE, adverse event.
Discussion
DYNAMIC is the first study to assess haemodynamic and clinical effects of chronic treatment with the oral sGC stimulator riociguat in patients with PH-HFpEF previously unexposed to vasoactive treatment. The presented patient population was representative of an elderly, predominantly female HFpEF population with invasively confirmed PH, common comorbidities and concomitant medication. In this trial, riociguat improved CO at rest with a placebo-corrected change from baseline of 0.54 L/min (Structured Graphical Abstract). The treatment effect was consistent across all statistical analysis sets. Significant favourable haemodynamic effects of riociguat were also observed regarding decreased PVR and TPG, and stability of SVR and PAWP. The observed haemodynamic changes were not accompanied by significant improvements of NT-proBNP serum levels, WHO-FC, exercise capacity, or QoL. Overall, riociguat had a favourable safety profile with respect to patients who completed the study. Nevertheless, higher dropout rates demand careful monitoring in future trials.
Our results of the primary endpoint are in line with observations in the previous single-dose DILATE-1 study18 (baseline CO values: 4.2 to 5.2 L/min; change from baseline to 6 h in the 2 mg group: 0.9 [0.3; 1.4] L/min), showing that riociguat both promptly and continuously improves CO in patients with PH-HFpEF. In two pivotal trials conducted in patients with precapillary PH, CHEST-115 and PATENT-116, riociguat exerted directionally comparable effects on invasively measured haemodynamic parameters. However, improvements in CO (CHEST-1: 0.9 [0.6; 1.1] L/min; PATENT-1: 0.9 [0.7; 1.2] L/min) as well as PVR (CHEST-1: −246 [−303; −190] dyn.s.cm−5; PATENT-1: −226 [−281; −170] dyn.s.cm−5) were more pronounced in the aforementioned studies as compared to DYNAMIC. In fact, mean baseline values of CO were lower in the precapillary PH studies (CHEST-1: 4 ± 1 L/min; PATENT-1: 4 ± 1 L/min), while mean baseline PVR (CHEST-1: range from 779 to 791 dyn.s.cm−5; PATENT-1: range from 791 to 834 dyn.s.cm−5) was three times higher compared to DYNAMIC, which most likely explains the lower magnitude of effect in our study. As HR remained stable over the study course, the improvement in CO by riociguat can be attributed to an increased stroke volume following pulmonary vasodilation, but also other - less explored - mechanisms of action, such as beneficial remodelling effects on both ventricles.10–12 Furthermore, regarding the observation of unchanged TPG and PVR after single doses of riociguat in DILATE-1, an effect on the pulmonary vasculature beyond vasodilation in case of chronic administration seems plausible. It is important emphasizing that improved CO in our study was not accompanied by significant changes of PAWP, thereby matching the results of previous studies on riociguat in PH.15–18
SVR remained unchanged after 26 weeks of riociguat as compared to placebo and so were invasively measured systemic blood pressure (BP) values. This is in contrast to CHEST-115 and PATENT-116, where significant drops in mean systemic BP were encountered. This discrepancy could be explained by a type II error (for comparison, see non-invasive measurements in the Supplementary material online, Tables S9, S10, and S11), but also by the lower target dose of riociguat in the present study, which was chosen based on a comparable effect size and incidence of AEs after 2.5 mg and 1.5 mg riociguat TID in PATENT-116 and frequent comorbid kidney disease taking into account the partial renal elimination of riociguat.24 Beneficial haemodynamic changes were neither accompanied by reduced levels of NT-proBNP nor by improvements in WHO-FC, walking distance, or QoL. Similar observations of lacking clinical benefit were made in three larger trials, investigating the efficacy of the sGC stimulators vericiguat over 12 and 24 weeks (SOCRATES-PRESERVED, VITALITY-HFpEF)25,26 and praliciguat over 12 weeks (CAPACITY-HFpEF) in HFpEF.27 However, meaningful comparison between these studies and DYNAMIC is compromised by the non-uniformity of study populations, as the former were not restricted to the specific phenotype of PH-HFpEF. Ultimately, further studies are required to determine whether the effect of improved CO in response to riociguat will translate into a better clinical outcome in the specific phenotype of PH-HFpEF.
Preceding single-centre trials that investigated the efficacy of phosphodiesterase 5 inhibitors in PH-HFpEF yielded conflicting results. In a population of patients with higher PVR (indicative of predominant cpcPH), haemodynamics and right ventricular (RV) function improved significantly after 12 months.28 In contrast, no significant changes were observed after 12 weeks of treatment in a population with lower PVR and therefore a higher proportion of patients with ipcPH and milder forms of cpcPH.29 This discrepancy was addressed by Belyavskiy et al. who conducted a trial in patients with (echocardiographic diagnoses of) HFpEF and cpcPH. They found improved exercise capacity, pulmonary haemodynamic parameters, and RV function in response to sildenafil.30 Whether a subgroup of patients with cpcPH might benefit from treatment with sildenafil ultimately demands clarification in a multicentre randomized clinical trial with invasively confirmed PH. Respective subgroup analyses in the present study revealed a greater improvement in cpcPH regarding haemodynamic parameters in response to riociguat (see Supplementary material online, Tables S7 and S8). Since cpcPH displays pulmonary vascular changes typically seen in precapillary PH2,31 and riociguat has proven effective in this population,15,16 these exploratory results are not unexpected. However, the present study was not powered for subgroup analyses as patient numbers were too small and larger next generation trials should focus on cpcPH patients to clarify the therapeutic potential of riociguat in this subgroup.
In this population of HFpEF with invasively confirmed ipcPH/cpcPH, CO was chosen as primary endpoint. This choice was based on observations in precapillary PH where CO has proven a strong indicator of RV function, exercise capacity, and outcome at baseline and after initiation of therapy,20,32,33 although CO might not have been the ideal endpoint to measure efficacy in the present collective of PH-HFpEF patients, especially in the absence of clinical benefit, and further studies are warranted regarding other haemodynamic effects and clinical outcomes. In contrast to previously reported precapillary PH collectives, where CO correlated well with clinical outcome such as 6MWD, WHO-FC, and levels of NT-proBNP,15,16 we cannot report significant matching results in our study. Clearly, patients enrolled in DYNAMIC displayed a different subtype of PH with less severe haemodynamic changes as compared to precapillary PH collectives. Furthermore, patients received lower treatment doses.15,16 Moreover, DYNAMIC was not powered to detect such clinical changes. These factors need to be considered regarding the interpretation of results. In consideration of the previous neutral trials of other sGC stimulators in HFpEF,25–27 it is difficult to tell how the results of DYNAMIC, showing a haemodynamic effect, will change the landscape.
The majority of patients received vitamin K antagonists or direct oral anticoagulants (DOACs), mostly for atrial fibrillation (Table 1 and see Supplementary material online, Table S6). Drug-drug interactions have been reported for riociguat and DOACs which could lead to bioaccumulation and excessive bleeding.34 In this study, bleeding events necessitating blood transfusion occurred in two patients after riociguat due to gastrointestinal bleeding on day 41 and renal anaemia on day 25. Both patients recovered without sequalae and completed all study procedures. No bleeding events were observed after placebo.
Drop-out rates were higher for patients in the riociguat as compared to the placebo group. This difference occurred due to higher rates of initial intolerance, non-tolerable AEs, and withdrawing of consent (see Supplementary material online, Table S2). For 5 of the 18 riociguat-treated patients, the primary reason for premature termination was a study drug-related TEAE. This observation may indicate a safety signal which should be further monitored in PH-HFpEF patients.
The present study has some limitations that merit comment. A majority of patients were included in one of five study centres (82% in Vienna). Thirty-eight patients in the riociguat group and 48 patients in the placebo group were included in the analysis of the primary endpoint, which was slightly below but still sufficiently close to the required sample size of 45 per group. However, the difference between groups caused some loss of power due to imbalance. Inclusion criteria were not updated regarding the definition of PH as published in 2019,31 i.e. PAPmean threshold reduction from ≥25 mm to ≥20 mm. Accordingly, 53 patients (46.5%) were randomized based on previous standards. With regard to the invasive nature of RHC and inherent risk, RHC results obtained ≤12 weeks prior to study enrolment were deemed acceptable resulting in variable time between baseline RHC and enrolment and thereby additional variability. Although a change in concomitant medication which may affect haemodynamic parameters was not allowed between baseline RHC and enrolment, it cannot be excluded that other treatment with subtle haemodynamic changes might have affected the results of this study. Exploratory echocardiography did not include the assessment of pericardial effusion and tricuspid annular velocity. Parameters were not assessed by a central laboratory. ANCOVAs revealed significant treatment-by-centre interactions (P < 0.05) for TPG and PVR (involving Vienna and pooled other centres) presumably due to greater vulnerability to extreme values in the small-sized pooled centre. However, as treatment effects pointed in the same direction in each centre in favour of riociguat, the presented ANCOVA results are considered valid and can be reliably interpreted. No risk stratification was performed during the study period. Scheduled timeframes could not be maintained for few patients due to the coronavirus disease 2019 pandemic. Also, given the demographic situation of participating centres, the vast majority of patients were Caucasian which might limit generalizability of our findings.
Important strengths of this study were the meticulous characterization of patients by TTE and RHC directly before randomization, the LVEF cut-off at ≥50%,6 the exclusion of differential diagnoses such as cardiac amyloidosis and hypertrophic cardiomyopathy, and the long-term study drug treatment. The present study was powered to detect a mean difference of at least 0.6 L/min between riociguat and placebo with regard to the primary endpoint assuming an SD of 1.0 L/min. Actually, the estimated treatment mean difference of 0.54 L/min was close to the anticipated treatment effect of 0.6 L/min associated with an error variability (ANCOVA) of 0.994 L/min. Moreover, FAS and PPS based analyses provided highly consistent results. Therefore, the study was adequately powered and presented reliable and credible results.
In conclusion, riociguat improved CO, PVR, and further pulmonary haemodynamics, and was safe in most patients with PH-HFpEF, but led to more dropouts and did not change clinical symptoms within the study period. Whether clinically meaningful endpoints of morbidity and mortality may be influenced by treatment with riociguat merits further investigation in adequately designed trials.
Supplementary data
Supplementary data is available at European Heart Journal online.
Supplementary Material
Acknowledgements
We wish to thank the study investigators, site staff, including Kristen Kopp, patients, and their families who participated in this study. We thank Bayer AG for funding of this study by an unrestricted grant. We would also like to acknowledge the contributions of the members of the data safety monitoring committee: Prof. Dr. Wilhelm Haverkamp, Prof. Dr. Walter Lehmacher, and Prof. Dr. Horst Olschewski. We thank Dr. Janine Dreesen, Mr. Jochen Weinert, and Dr. Maren Carstensen from M.A.R.C.O. GmbH & Co for data management, monitoring, medical writing, statistical and editorial support.
Contributor Information
Theresa Marie Dachs, Division of Cardiology, Department of Internal Medicine II, Medical University of Vienna, Waehringer Guertel 18-20, 1090 Vienna, Austria.
Franz Duca, Division of Cardiology, Department of Internal Medicine II, Medical University of Vienna, Waehringer Guertel 18-20, 1090 Vienna, Austria.
René Rettl, Division of Cardiology, Department of Internal Medicine II, Medical University of Vienna, Waehringer Guertel 18-20, 1090 Vienna, Austria.
Christina Binder-Rodriguez, Division of Cardiology, Department of Internal Medicine II, Medical University of Vienna, Waehringer Guertel 18-20, 1090 Vienna, Austria.
Daniel Dalos, Division of Cardiology, Department of Internal Medicine II, Medical University of Vienna, Waehringer Guertel 18-20, 1090 Vienna, Austria.
Luciana Camuz Ligios, Division of Cardiology, Department of Internal Medicine II, Medical University of Vienna, Waehringer Guertel 18-20, 1090 Vienna, Austria.
Andreas Kammerlander, Division of Cardiology, Department of Internal Medicine II, Medical University of Vienna, Waehringer Guertel 18-20, 1090 Vienna, Austria.
Ekkehard Grünig, Centre for Pulmonary Hypertension, Thoraxklinik at Heidelberg University Hospital, Translational Lung Research Centre Heidelberg (TLRC), German Centre for Lung Research (DZL), 69126 Heidelberg, Germany.
Ingrid Pretsch, Division of Cardiology and Intensive Care, Department of Internal Medicine II, Paracelsus Medical University of Salzburg, Muellner Hauptstraße 48, 5020 Salzburg, Austria.
Regina Steringer-Mascherbauer, Division of Cardiology, Angiology and Intensive Care, Department of Internal Medicine II, Public Hospital Elisabethinen Linz, Fadingerstraße 1, 4020 Linz, Austria.
Klemens Ablasser, Division of Cardiology, Medical University of Graz, Auenbruggerplatz 15, 8036 Graz, Austria.
Manfred Wargenau, M.A.R.C.O. GmbH & Co. KG, Institute for Clinical Research and Statistics, Schirmerstraße 71, 40211 Duesseldorf, Germany.
Julia Mascherbauer, Division of Cardiology, Department of Internal Medicine II, Medical University of Vienna, Waehringer Guertel 18-20, 1090 Vienna, Austria; Division of Cardiology, Department of Internal Medicine III, University Hospital of St. Poelten, Dunant-Platz 1, 3100 St. Poelten, Austria.
Irene M Lang, Division of Cardiology, Department of Internal Medicine II, Medical University of Vienna, Waehringer Guertel 18-20, 1090 Vienna, Austria.
Christian Hengstenberg, Division of Cardiology, Department of Internal Medicine II, Medical University of Vienna, Waehringer Guertel 18-20, 1090 Vienna, Austria.
Roza Badr-Eslam, Division of Cardiology, Department of Internal Medicine II, Medical University of Vienna, Waehringer Guertel 18-20, 1090 Vienna, Austria.
Johannes Kastner, Division of Cardiology, Department of Internal Medicine II, Medical University of Vienna, Waehringer Guertel 18-20, 1090 Vienna, Austria.
Diana Bonderman, Division of Cardiology, Department of Internal Medicine II, Medical University of Vienna, Waehringer Guertel 18-20, 1090 Vienna, Austria; Division of Cardiology, Department of Internal Medicine V, Favoriten Clinic, Kundratstraße 3, 1100 Vienna, Austria.
Funding
This work was supported by an unrestricted grant from Bayer, who also provided the study medication. The funder had no role in study design, data collection, data analysis, data interpretation, or writing of the report. The sponsor of this investigator-initiated trial was the Medical University of Vienna (Diana Bonderman, MD). The sponsor was responsible for initiation, management, and financing of the study. Clinical project management, monitoring, data management, statistics, and medical writing assistance were provided by M.A.R.C.O. GmbH & Co. KG (Duesseldorf, Germany). The first and the corresponding author drafted and corrected the manuscript. All authors had access to trial data, reviewed the manuscript, and approved the final draft for submission for publication. All authors guarantee the completeness and accuracy of the data presented.
Data Availability
The data shared will not be made universally available to any third party. Any requests should be directed towards the corresponding author.
References
- 1. Dunlay SM, Roger VL, Redfield MM. Epidemiology of heart failure with preserved ejection fraction. Nat Rev Cardiol 2017;14:591–602. [DOI] [PubMed] [Google Scholar]
- 2. Guazzi M, Naeije R. Pulmonary hypertension in heart failure: pathophysiology, pathobiology, and emerging clinical perspectives. J Am Coll Cardiol 2017;69:1718–1734. [DOI] [PubMed] [Google Scholar]
- 3. Lam CS, Roger VL, Rodeheffer RJ, Borlaug BA, Enders FT, Redfield MM. Pulmonary hypertension in heart failure with preserved ejection fraction: a community-based study. J Am Coll Cardiol 2009;53:1119–1126. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4. Hoeper MM, Carolyn SP, Lam CSP, Vachiery JL, Bauersachs J, Gerges C, et al. Pulmonary hypertension in heart failure with preserved ejection fraction: a plea for proper phenotyping and further research. Eur Heart J 2017;38:2869–873. [DOI] [PubMed] [Google Scholar]
- 5. Borlaug BA. Evaluation and management of heart failure with preserved ejection fraction. Nat Rev Cardiol 2020;17:559–573. [DOI] [PubMed] [Google Scholar]
- 6. Ponikowski P, Voors AA, Anker SD, Bueno H, Cleland JGF, Coats AJS, et al. 2016 ESC Guidelines for the diagnosis and treatment of acute and chronic heart failure: the task force for the diagnosis and treatment of acute and chronic heart failure of the European Society of Cardiology (ESC). developed with the special contribution of the Heart Failure Association (HFA) of the ESC. Eur Heart J 2016;37:2129–2200. [DOI] [PubMed] [Google Scholar]
- 7. Yancy CW, Jessup M, Bozkurt B, Butler J, Jr CD, Drazne MH, et al. 2013 ACCF/AHA guideline for the management of heart failure: a report of the American College of Cardiology Foundation/American Heart Association task force on practice guidelines. J Am Coll Cardiol 2013;62:e147–239. [DOI] [PubMed] [Google Scholar]
- 8. Lam CS, Brutsaert DL. Endothelial dysfunction: a pathophysiologic factor in heart failure with preserved ejection fraction. J Am Coll Cardiol 2012;60:1787–1789. [DOI] [PubMed] [Google Scholar]
- 9. Giaid A, Saleh D. Reduced expression of endothelial nitric oxide synthase in the lungs of patients with pulmonary hypertension. N Engl J Med 1995;333:214–221. [DOI] [PubMed] [Google Scholar]
- 10. Stasch JP, Pacher P, Evgenov OV. Soluble guanylate cyclase as an emerging therapeutic target in cardiopulmonary disease. Circulation 2011;123:2263–2273. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11. van Heerebeek L, Hamdani N, Falcão-Pires I, Leite-Moreira AF, Begieneman MPV, Bronzwaer JGF, et al. Low myocardial protein kinase G activity in heart failure with preserved ejection fraction. Circulation 2012;126:830–839. [DOI] [PubMed] [Google Scholar]
- 12. Boerrigter G, Costello-Boerrigter LC, Cataliotti A, Lapp H, Stasch JP, Burnett JC Jr. Targeting heme-oxidized soluble guanylate cyclase in experimental heart failure. Hypertension 2007;49:1128–1133. [DOI] [PubMed] [Google Scholar]
- 13. Schermuly RT, Janssen W, Weissmann N, Stasch JP, Grimminger F, Ghofrani HA. Riociguat for the treatment of pulmonary hypertension. Expert Opin Investig Drugs 2011;20:567–576. [DOI] [PubMed] [Google Scholar]
- 14. Grimminger F, Weimann G, Frey R, Voswinckel R, Thamm M, Bölkow D, et al. First acute haemodynamic study of soluble guanylate cyclase stimulator riociguat in pulmonary hypertension. Eur Respir J 2009;33:785–792. [DOI] [PubMed] [Google Scholar]
- 15. Ghofrani HA, D’Armini AM, Grimminger F, Hoeper MM, Pavel Jansa P, Kim NH, et al. Riociguat for the treatment of chronic thromboembolic pulmonary hypertension. N Engl J Med 2013;369:319–329. [DOI] [PubMed] [Google Scholar]
- 16. Ghofrani HA, Galiè N, Grimminger F, Grünig E, Humbert M, Jing ZC, et al. Riociguat for the treatment of pulmonary arterial hypertension. N Engl J Med 2013;369:330–340. [DOI] [PubMed] [Google Scholar]
- 17. Bonderman D, Ghio S, Felix SB, Ghofrani HA, Michelakis E, Mitrovic V, et al. Riociguat for patients with pulmonary hypertension caused by systolic left ventricular dysfunction: a phase IIb double-blind, randomized, placebo-controlled, dose-ranging hemodynamic study. Circulation 2013;128:502–11. [DOI] [PubMed] [Google Scholar]
- 18. Bonderman D, Pretsch I, Steringer-Mascherbauer R, Jansa P, Rosenkranz S, Tufaro C, et al. Acute hemodynamic effects of riociguat in patients with pulmonary hypertension associated with diastolic heart failure (DILATE-1): a randomized, double-blind, placebo-controlled, single-dose study. Chest 2014;146:1274–1285. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19. Mascherbauer J, Grünig E, Halank M, Hohenforst-Schmidt W, Kammerlander AA, Pretsch I, et al. Evaluation of the pharmacoDYNAMIC effects of riociguat in subjects with pulmonary hypertension and heart failure with preserved ejection fraction: study protocol for a randomized controlled trial. Wien Klin Wochenschr 2016;128:882–889. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20. Galiè N, Humbert M, Vachiery JL, Gibbs S, Lang I, Torbicki A, et al. 2015 ESC/ERS Guidelines for the diagnosis and treatment of pulmonary hypertension: the joint task force for the diagnosis and treatment of pulmonary hypertension of the European Society of Cardiology (ESC) and the European Respiratory Society (ERS): endorsed by: Association for European Paediatric and Congenital Cardiology (AEPC), International Society for Heart and Lung Transplantation (ISHLT). Eur Heart J 2016;37:67–119. [DOI] [PubMed] [Google Scholar]
- 21. Kovacs G, Avian A, Pienn M, Naeije R, Olschewski H. Reading pulmonary vascular pressure tracings. How to handle the problems of zero leveling and respiratory swings. Am J Respir Crit Care Med 2014;190:1198–1199. [DOI] [PubMed] [Google Scholar]
- 22. EuroQol Group . EuroQol - a new facility for the measurement of health-related quality of life. Health Polic 1990;16:199–208. [DOI] [PubMed] [Google Scholar]
- 23. Rector TS, Kubo SH, Cohn JN. Patients’ self-assessment of their congestive heart failure. Part 2: content, reliability and validity of a new measure, The Minnesota Living with Heart Failure Questionnaire. Heart Failure 1987;3:198–209. [Google Scholar]
- 24. Frey R, Becker C, Saleh S, Unger S, van der Mey D, Mück W. Clinical pharmacokinetic and pharmacodynamic profile of riociguat. Clin Pharmacokinet 2018;57:647–661. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25. Pieske B, Maggioni AP, Lam CSP, Pieske-Kraigher E, Filippatos G, Butler J, et al. Vericiguat in patients with worsening chronic heart failure and preserved ejection fraction: results of the SOluble guanylate Cyclase stimulatoR in heArT failurE patientS with PRESERVED EF (SOCRATES-PRESERVED) study. Eur Heart J 2017;38:1119–1127. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26. Armstrong PW, Lam CSP, Anstrom KJ, Ezekowitz J, Hernandez AF, O’Connor CM, et al. Effect of vericiguat vs placebo on quality of life in patients with heart failure and preserved ejection fraction: the VITALITY-HFpEF randomized clinical trial. JAMA 2020;324:1512–1521. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27. Udelson JE, Lewis GD, Shah SJ, Zile MR, Redfield MM, Jr BJ, et al. Effect of praliciguat on peak rate of oxygen consumption in patients with heart failure with preserved ejection fraction the CAPACITY HFpEF randomized clinical trial. JAMA 2020;324:1522–1531. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28. Guazzi M, Vicenzi M, Arena R, Guazzi MD. Pulmonary hypertension in heart failure with preserved ejection fraction: a target of phosphodiesterase-5 inhibition in a 1-year study. Circulation 2011;124:164–74. [DOI] [PubMed] [Google Scholar]
- 29. Hoendermis ES, Liu LCY, Hummel YM, van der Meer P, de Boer RA, Berger RMF, et al. Effects of sildenafil on invasive haemodynamics and exercise capacity in heart failure patients with preserved ejection fraction and pulmonary hypertension: a randomized controlled trial. Eur Heart J 2015;36:2565–73. [DOI] [PubMed] [Google Scholar]
- 30. Belyavskiy E, Ovchinnikov A, Potekhina A, Ageev F, Edelmann F. Phosphodiesterase 5 inhibitor sildenafil in patients with heart failure with preserved ejection fraction and combined pre- And postcapillary pulmonary hypertension: a randomized open-label pilot study. BMC Cardiovasc Disord 2020;20:408. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31. Simonneau G, Montani D, Celermajer DS, Denton CP, Gatzoulis MA, Krowka M, et al. Haemodynamic definitions and updated clinical classification of pulmonary hypertension. Eur Respir J 2019;53:1801913. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32. Swiston JR, Johnson SR, Granton JT. Factors that prognosticate mortality in idiopathic pulmonary arterial hypertension: a systematic review of the literature. Respir Med 2010;104:1588–607. [DOI] [PubMed] [Google Scholar]
- 33. Miura Y, Fukumoto Y, Sugimura K, Oikawa M, Nakano M, Tatebe S, et al. Identification of new prognostic factors of pulmonary hypertension. Circ J 2010;74:1965–71. [DOI] [PubMed] [Google Scholar]
- 34. Margelidon-Cozzolino V, Delavenne X, Catella-Chatron J, De Magalhaes E, Bezzeghoud S, Humbert M, et al. Indications and potential pitfalls of anticoagulants in pulmonary hypertension: Would DOACs become a better option than VKAs? Blood Rev 2019;37:100579. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Data Availability Statement
The data shared will not be made universally available to any third party. Any requests should be directed towards the corresponding author.