Abstract
Simple Summary
Reticulitermes flaviceps, as a main wood-boring pest, causes economically significant damage to wood materials. In this study, gas chromatography–mass spectrometry was used to detect and characterize the chemical constituents of lemongrass essential oil (Cymbopogon citratus (DC.) Stapf.), and we evaluated the associated vapor insecticidal effect on R. flaviceps worker adults. Lemongrass EO and its major constituent, citral, presented significant vapor toxicity against R. flaviceps, where the walking and gripping abilities of treated insects were reduced. Chitin content, thermal stability, and crystallinity were also reduced in the treated worker adults. The results of this study suggest that lemongrass EO can potentially be used to develop eco-friendly natural remedies for the management of R. flaviceps.
Abstract
Botanical pesticides are considered the most promising alternative to synthetic pesticides, considering their less negative impacts on the environment and human health. Here, we analyzed the components of lemongrass Cymbopogon citratus essential oil (EO) and evaluated its vapor activity against Reticulitermes flaviceps, in terms of the walking and gripping abilities of workers. In addition, the effects of lemongrass EO and its major component on the cuticular content and structure of chitin in termites were also observed. Our results indicate that cis-citral (36.51%) was the main constituent of lemongrass. In the vapor toxicity assay, the LC50 values of lemongrass EO and citral were 0.328 and 0.177 μL/L, respectively. When worker antennae were treated with lemongrass EO and citral, their walking and gripping capabilities were significantly inhibited. In addition, the cuticular content, thermal stability, and crystallinity of chitin in the termites were decreased after treatment with citral. Collectively, this study provides a basis for developing and utilizing lemongrass and citral as a new environment-friendly insecticide resource to control R. flaviceps.
Keywords: Reticulitermes flaviceps, Cymbopogon citratus, essential oil, citral, walking and gripping behavior, chitin structure
1. Introduction
Termites are important agricultural and forestry pests in tropical and sub-tropical regions, which damage crops, forests, garden trees, houses, and ancient buildings, thus causing an economic loss of more than USD 40 billion annually worldwide [1]. Reticulitermes flaviceps is widely distributed in China, and causes losses for the Chinese economy [2]. Synthetic pesticides are currently the most commonly used method for termite control, which often have a negative impact on the natural environment [3]. Due to the low toxicity, biodegradability, and environmentally friendly nature of essential oils (EOs), there is growing interest in their use as novel alternative synthetic insecticides [4].
The genus Cymbopogon (family Poaceae) comprises about 144 species, widely spread throughout the tropical and sub-tropical regions [5]. Plants in the genus Cymbopogon are good sources of EOs and are rich in bioactive compounds, including alcohols, aldehydes, and phenolic compounds. The chemical compositions of Cymbopogon spp. EOs, such as those from Cymbopogon citratus [6,7,8], Cymbopogon distans [9], Cymbopogon flexuosus [10], Cymbopogon martinii [11,12], Cymbopogon nardus [13,14,15], Cymbopogon nervatus [5,16], Cymbopogon schoenanthus [15,17,18], and Cymbopogon winterianus [19,20] were assessed in previous studies.
Cymbopogon spp. EOs present significant inter/intra-species differences in their chemical compositions. An EO of C. citratus from China is reported to be rich in citronellal (38.16%) [7] and geraniol (25.19%) [8], whereas cis-citral (Italy, 59.19%) [21], geranial (Brazil, 50.18%) [6] and neral (Benin, 24.6%) [22] were the major components in C. citratus oil in other countries. Similarly, citronellal (22.15–41.7%) [14,15,22,23] and citral (38.75%) [13] are reported as major components in C. nardus oil. Piperitone (59.2–71.5%) [15,17], α-eudesmol (17.89%) [18], and cis-p-Menth-2-en-1-ol (28.5%) [24] were the major components in C. schoenanthus oil. The major components of C. flexuosus oil were neral (30.4%) [10] and geranial (38.44%) [25]. A previous study reported that citronellal (24.0–55.4%) [19,26] was the main constituent in C. winterianus oil. Trans-p-mentha-2, 8-dien-1-ol (20.70%) [11] and geraniol (76.9%) [12] are reported as major constituents in C. martinii oil. Trans-p-mentha-1 (7), 8-dien-2-ol (32.6%) [16] and trans-p-mentha-2, 8-dien-1-ol (13.6%) [5] are reported as major components in C. nervatus oil.
According to previous reports, Cymbopogon EOs present good insecticidal activity against vector pests, such as Anopheles funestus [27], Aedes aegypti [28,29], Aedes albopictus [30], Cochliomyia hominivorax [31], Culex quinquefasciatus [32], Haemaphysalis longicornis [33], Musca domestica [34], Rhipicephalus microplus [35], and Sarcoptes scabiei [36]; storage pests, including Acanthoscelides obtectus [37], Callosobruchus maculatus [25,38,39], Dinoderus porcellus [22], Rhyzopertha dominica [40], Sitophilus granaries [41], Sitophilus oryzae [42,43,44], Sitophilus zeamais [45,46], Tenebrio molitor [47], Trogoderma granarium [48], Tribolium castaneum [49,50,51,52,53], and Ulomoides dermestoides [23,54]; and agricultural insect pests, such as Bemisia tabaci [55], Euprosterna elaeasa [56], Megalurothrips sjostedti [57], Phthorimaea operculella [58], Spodoptera exigua [59], Spodoptera frugiperda [60,61], Trichoplusia ni [62,63], and Tuta absoluta [64].
However, there exist almost no reports on the vapor toxicity efficacy of Cymbopogon spp. EOs against Reticulitermes flaviceps. Thus, the objectives of the present study were to: (1) evaluate the constituents of lemongrass EO; (2) evaluate the vapor activity of lemongrass EO and its major constituent against R. flaviceps in terms of their behavioral effect on the walking and gripping capability of workers; and (3) investigate the effects of citral on the chemical structure of chitin in R. flaviceps.
2. Materials and Methods
2.1. Insects
Two colonies of subterranean termite R. flaviceps were collected at Linglong Mountain in Lin’an, Hangzhou, and three colonies of R. flaviceps were collected from ZAFU campus, and reared with water and Mason’s pine (Pinus massoniana L.) in the laboratory. We selected healthy and active workers of uniform size for further experiments.
2.2. Lemongrass EO and the Constituents
Lemongrass EO was obtained from the Moellhausen Flagship Store (Shanghai, China) and its major constituent, citral (96%), was purchased from TCI Shanghai (Shanghai, China) and kept at 4 °C until further use.
2.3. GC-MS Analysis
The lemongrass EO was determined using a gas chromatograph (Agilent 6890A, Santa Clara, CA, USA), equipped with a mass spectrometer detector (Agilent 5975C, Santa Clara, CA, USA). The injector temperature was set at 250 °C, the oven temperature was programmed at 50–250 °C (10 °C/min), the He carrier gas flow was 1.0 mL/min (split ratio of 1:50), and a sample volume of 1.0 μL was injected. Compounds were identified using NIST11.LIB, through comparison of retention indices (RI) with respect to those reported in the Adams [65] library.
2.4. Vapor Toxicity
To conduct fumigations [3], filter paper strips were stuck to the lids of 1 L glass jars and 0.12–0.22 μL of lemongrass EO; its major component citral, or (acetone as a control) was added. Twenty healthy workers were put into a glass bottle, the bottle cap was quickly closed, and a moist filter paper was placed on the bottom of the bottle as food. The experiment was repeated three times with three colonies, and the glass jars were kept at 25 ± 1 °C and 80% RH. A portion of moistened filter paper was placed at the bottom of the bottle for water and food. After 24 h, the number of dead termites was observed and recorded.
2.5. Behavior Effect
2.5.1. Walking Behavior
R. flaviceps workers were anesthetized with carbon dioxide for 5 s, then 1 μL of the lemongrass EO or citral was applied to their antennae. The treated workers were transferred to petri dishes lined with moist filter paper and, after 2 h, we observed their walking ability. Workers who could walk continuously for more than 5 s were considered to have normal walking ability, and the number of workers with normal walking ability was recorded. Acetone treatment was used as control, and three replicates with three colonies of 20 workers were used for each dose.
2.5.2. Gripping Behavior
R. flaviceps workers were anesthetized with carbon dioxide for 5 s, then 1 μL of the lemongrass EO or citral was applied to their antennae. The treated workers were transferred to petri dishes lined with moist filter paper. After 2 h, the petri dish was covered with a piece of filter paper and gently inverted (with the mouth facing downward) for 5 s. Then, the petri dish was gently inverted back up again, and we immediately recorded the number of workers stuck to the paper. Acetone treatment was used as control, and three replicates with three colonies of 20 workers were used for each dose.
2.6. Effect of Chitin Structural Chemistry
2.6.1. Insect Treatment
Fifty healthy and active adult workers were placed in glass vials and treated with a sub-lethal concentration (LC20 = 0.16 μL/L) of citral for 6 h, as described in Section 2.4.
2.6.2. Chitin Extraction
Chitin was extracted according to the procedure of Shah et al. [66], including three steps of demineralization, deproteinization, and decolorization. In brief, samples were first demineralized by treatment with 1 M HCl for 20 min at 100 °C. Then, the washed samples were deproteinized with 1 M NaOH solution for 24 h at 80 °C. Finally, the obtained chitin samples were decolorized through incubation in a mixture of chloroform, methanol, and water (1:2:4, v/v/v) for 20 min. The percentage of chitin weight was calculated based on the formula:
where W1 represents the weight of the raw sample and W2 represents the weight of the chitin.
2.6.3. Fourier Transform Infrared Spectroscopy (FTIR)
Fourier transform infrared spectroscopy (Prestige-21, SHIMADZU, Kyoto, Japan) was conducted to measure absorbance values between 250 and 4000 cm−1. Before measurement, a 1 mg sample of chitin was added to 100 mg of purified potassium bromide (KBr) powder. The effects of citral treatments on the molecular structure and composition of chitin were evaluated by observing the changes in the infrared band. Each experiment was repeated three times.
2.6.4. Thermogravimetric Analysis (TGA)
Citral-treated chitin samples were analyzed using a NETZSCH TG 209 F1 Libra thermal gravimetric analyzer (Selb, Germany). A 10 mg chitin sample was heated from 30 °C to 500 °C in nitrogen at a rate of 10 °C/min. The effect of citral on the thermal stability of the extracted chitin was analyzed by thermogravimetric (TG) and differential thermogravimetric (DTG) analyses.
2.6.5. X-ray Diffraction (XRD)
X-ray diffraction spectra were obtained using an X’Pert-Pro MPD X-ray diffractometer (Almelo, Holland). Data were collected on a copper target at a scan rate of 1°/min with a scan angle of 5–40°. The crystallinity index was calculated using the following equation:
where I110 is the maximum intensity at 2θ ≅ 20° and Iam is the intensity of amorphous diffraction at 2θ ≅ 16°.
2.6.6. Differential Scanning Calorimetry (DSC)
DSC analysis of 10 mg control and citral-treated chitin samples was conducted using a NETZSCH DSC 204F1 apparatus (Selb, Germany).
2.7. Statistical Analysis
Toxicity data were subjected to probit analysis in order to estimate the LC50 values of lemongrass EO and the major constituent. The mortality rate, chitin content, and walking and gripping ability data were subjected to one-way ANOVA and Tukey’s HSD test (p < 0.05).
3. Results
3.1. Chemical Composition of Lemongrass EO
The chemical compositions of the lemongrass s EO are shown in Table 1. Nine components comprising 96.84% of the total lemongrass EO composition were identified. cis-citral (36.51%), trans-citral (31.42%), and geraniol (8.78%) were identified as major components of the lemongrass oil.
Table 1.
Chemical composition of lemongrass EO.
| No. | Constituents | RI a | RI b | % |
|---|---|---|---|---|
| 1 | α-Pinene | 1.83 | ||
| 2 | β-Pinene | 935 | 932 | 0.64 |
| 3 | Limonene | 979 | 977 | 2.46 |
| 4 | 1,8-Cineole | 1029 | 1025 | 6.52 |
| 5 | Linalool | 1038 | 1036 | - |
| 6 | trans-Citral | 1097 | 1095 | 31.42 |
| 7 | Geraniol | 1240 | 1235 | 8.78 |
| 8 | Citronellyl formate | 1250 | 1249 | - |
| 9 | cis-Citral | 1277 | 1271 | 36.51 |
| 10 | Geranyl acetate | 1316 | 1312 | 4.85 |
| 11 | Neryl acetate | 1352 | 1350 | - |
| 12 | Caryophyllene | 1365 | 1359 | 3.83 |
| 1419 | 1417 | |||
| Total identified (%) | 96.84 |
a RI, linear retention indices on HP-5MS column, experimentally determined using homologue series of n-alkanes. b Retention indices (RI) values taken from Adams [65].
3.2. Vapor Activity of Lemongrass EO and the Major Constituent
The efficacy of lemongrass EO against R. flaviceps was significantly increased with increasing concentration (F = 32.73; df = 5, 12; p < 0.001; Table 2). The LC50 value of lemongrass EO against R. flaviceps showed high toxicity, with a value of 0.328 (Table 2). Additionally, citral also showed a significant variation in vapor toxicity at different concentrations (F = 184.85; df = 5, 12; p < 0.001; Table 2), with an LC50 of 0.177 (Table 2).
Table 2.
Mortality rate at 24 h, lethal concentration for 50% and 90% mortality of lemongrass EO and citral against R. flaviceps.
| Treatment | Conc. (μL/L) |
Mortality (%) ± SD at 24 h | LC50 a (LCL-UCL) | LC90 a (LCL-UCL) | Regression | χ2 b (d.f. = 4) | R2 c |
|---|---|---|---|---|---|---|---|
| Lemongrass EO | 0.14 0.16 |
0 e * 3.3 ± 2.9 e |
0.328 (0.222–0.391) | 0.595 (0.524–0.720) | y = 4.8167x − 0.655 | 3.685 n.s | 0.982 |
| 0.18 | 30.0 ± 5.0 d | ||||||
| 0.20 | 31.7 ± 5.0 d | ||||||
| 0.22 | 38.3 ± 7.6 cd | ||||||
| Citral | 0.14 0.16 |
6.7 ± 2.9 e 15.0 ± 5.0 e |
0.177 (0.171–0.185) | 0.214 (0.203–0.233) | y = 12.417x − 1.7383 | 32.464 n.s | 0.973 |
| 0.18 | 48.3 ± 2.9 c | ||||||
| 0.20 | 80.0 ± 10.0 b | ||||||
| 0.22 | 98.3 ± 2.9 a |
a LC50, LC90 = lethal concentration for 50% and 90% mortality with 95% confidence limit; LCL = lower confidence limit; UCL = upper confidence limit. b χ2 = chi-square value with α = 0.05. c R2 = regression coefficient. * The means in each row against R. flaviceps that are followed by different letters are significantly different (p < 0.05, by ANOVA and Tukey’s HSD Test). d.f. = degrees of freedom. n.s. = not significant (p > 0.05).
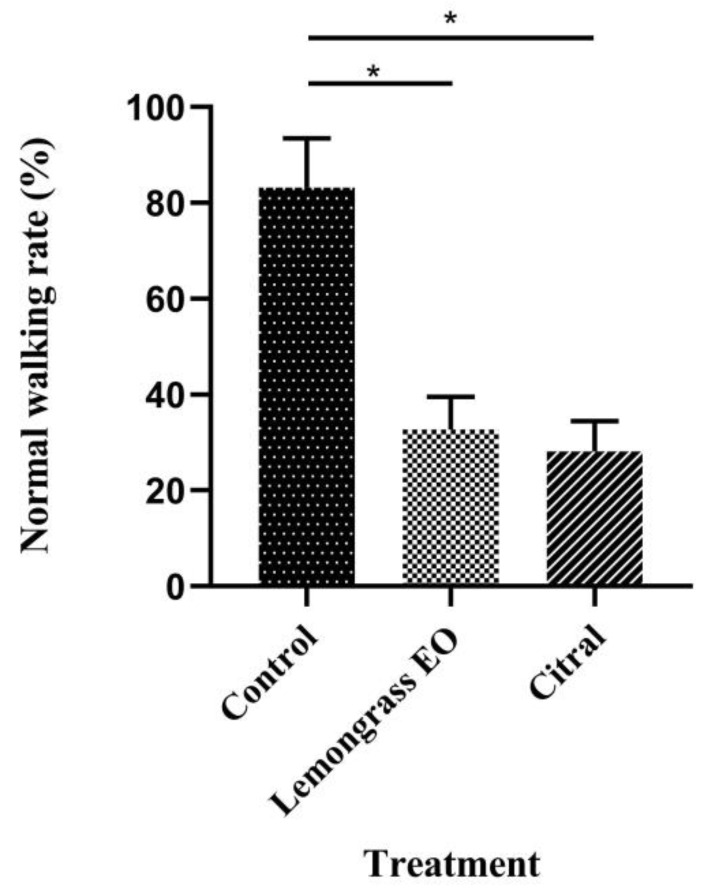
3.3. Effects of Lemongrass EO and the Major Constituent on Walking Behavior
Lemongrass EO and its major constituent significantly affected the walking ability of R. flaviceps workers, compared with controls (p < 0.01; Figure 1). In the control group, the workers walked quickly and lasted for a long time, while the walking rate of the workers in all treatment groups was significantly slowed down to where the workers could not walk continuously or even completely. In general, the walking ability of the citral-treated termites was lower than that of those treated with lemongrass EO.
Figure 1.
Effect of lemongrass EO and its major constituent on the walking behavior of R. flaviceps workers. Means (±SD) values with * show significant differences (p < 0.01), according to Tukey’s HSD test.
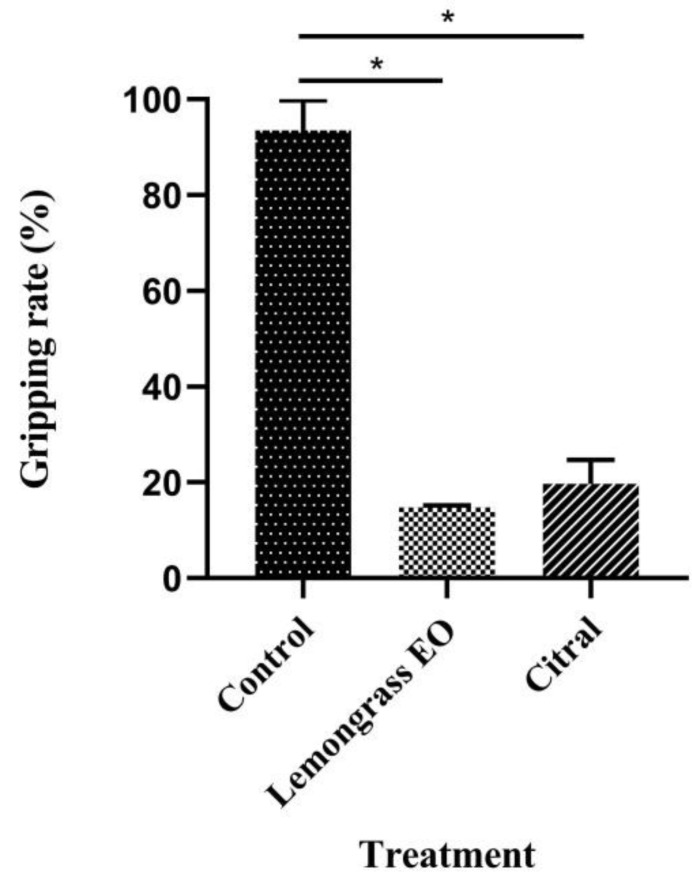
3.4. Effects of Lemongrass EO and Its Major Constituent on Gripping Behavior
The EO of lemongrass and its major constituent significantly affected the gripping ability of R. flaviceps workers, compared with controls (p < 0.01; Figure 2), where their gripping ability ranged from 11.84 to 22.02%.
Figure 2.
Effect of lemongrass EO and its major constituent on the gripping behavior of R. flaviceps workers. Mean (±SD) values with * show significant differences (p < 0.01), according to Tukey’s HSD test.
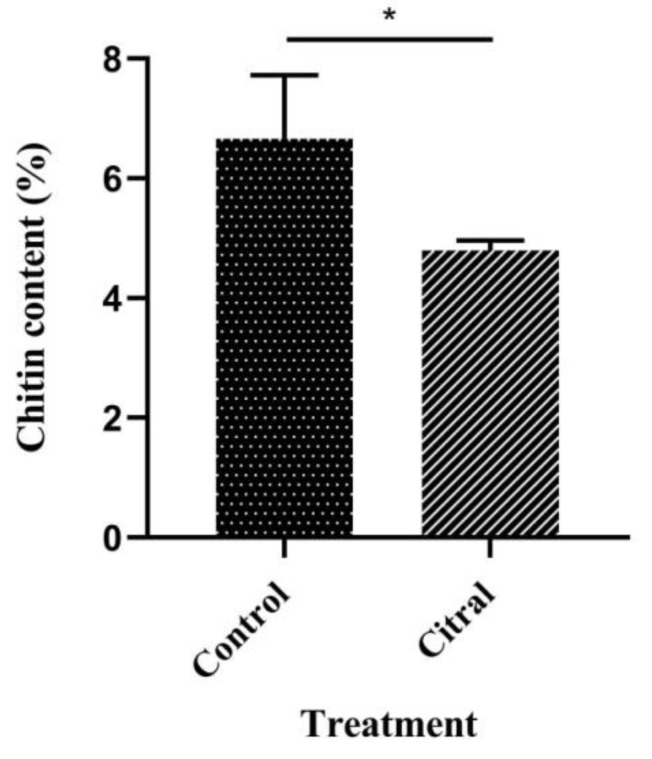
3.5. Chitin Content
In the present study, the chitin content in the control and citral-treated groups of R. flaviceps workers was investigated. As shown in Figure 3, citral (2.91%) led to significantly lower chitin content in R. flaviceps workers, compared with the control (6.67%; p < 0.01), indicating that citral decreased the chitin content.
Figure 3.
Citral decreases chitin content of R. flaviceps workers. Mean (±SD) values with * show significant difference (p < 0.01), according to Tukey’s HSD test.
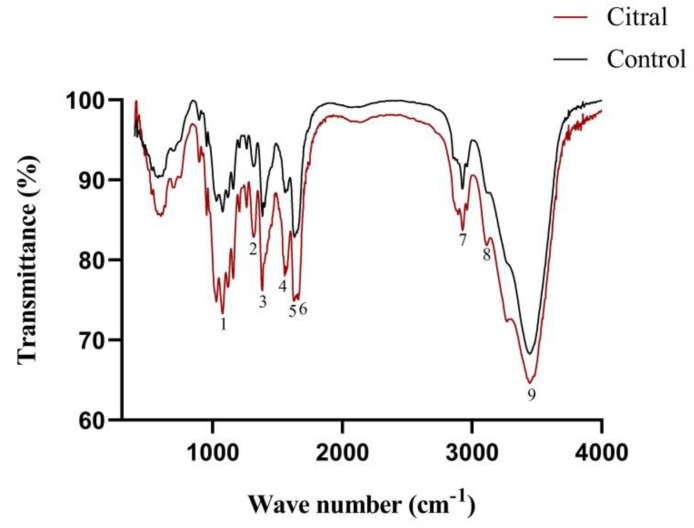
3.6. Fourier Transform Infrared Radiation
The FTIR spectra are presented in Figure 4 and Table 3. Significant changes in chitin chemical structure properties were observed in the citral-treated group. The chitin chemical structures of the control and citral-treated R. flaviceps workers presented three important amide bands at 1560, 1630, and 1656 cm−1, corresponding to N–H bending and C–N stretching (amide II), C=O secondary amide stretching (amide I) and C=O secondary amide stretching (amide I), respectively.
Figure 4.
FTIR spectra of α-chitin from R. flaviceps in the control and citral-treated groups.
Table 3.
Characteristics and variations in bands in the FTIR spectra of the chitin of R. flaviceps treated with citral and control insects.
| No. | Wave Number (cm−1) |
Functional Group and Vibration Modes |
Band Assignment |
Control | Citral |
|---|---|---|---|---|---|
| 1 | 1050 | C–O asym. stretch in phase ring |
- | 88.21 | 77.54 |
| 2 | 1315 | CH2 wagging | Amide III, components of proteins |
91.65 | 82.77 |
| 3 | 1385 | C–H bend, CH3 sym. Deformation |
- | 85.77 | 76.88 |
| 4 | 1560 | N–H bend, C–N stretch |
Amide II | 88.81 | 79.32 |
| 5 | 1630 | C=O secondary amide stretch |
Amide I | 82.92 | 75.05 |
| 6 | 1656 | C=O secondary amide stretch |
Amide I | 93.78 | 75.28 |
| 7 | 2932 | CH3 sym. stretch and CH2 asym. stretch |
Aliphatic compounds |
89.53 | 84.08 |
| 8 | 3111 | N–H secondary amine asym. stretch |
Amide II | 88.46 | 81.86 |
| 9 | 3440 | O–H hydroxyl stretching | - | 68.31 | 64.70 |
3.7. Thermogravimetric Analysis
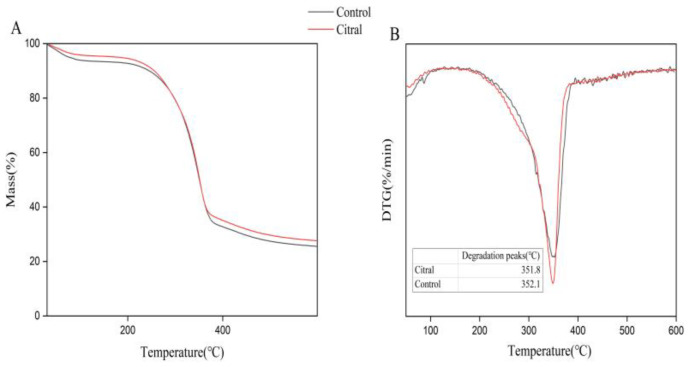
The thermal stability and degradability of chitin were analyzed by thermogravimetric analysis (TGA; Figure 5). As shown in Figure 5A, three degradation processes occurred: the first one occurred at 30–110 °C, with a weight loss of 5.5%. The second degradation process occurred at 280–370 °C; the degradation rate in this process was accelerated, and the weight loss was 67.8%. Finally, the third degradation process occurred at 403–500 °C, and the degradation tended to be stable in this process. Figure 5B shows the maximum degradation rates of chitin content by citral. The DTGmax of control chitin was at 352.1 °C, while the DTGmax of citral-treated chitin was at 351.8 °C, indicating that the thermal stability of chitin was affected by citral treatment.
Figure 5.
(A) TG curves of chitin in the control and citral-treated groups; (B) DTG profiles of the chitin of R. flaviceps in the control and citral-treated groups.
3.8. X-ray Diffraction
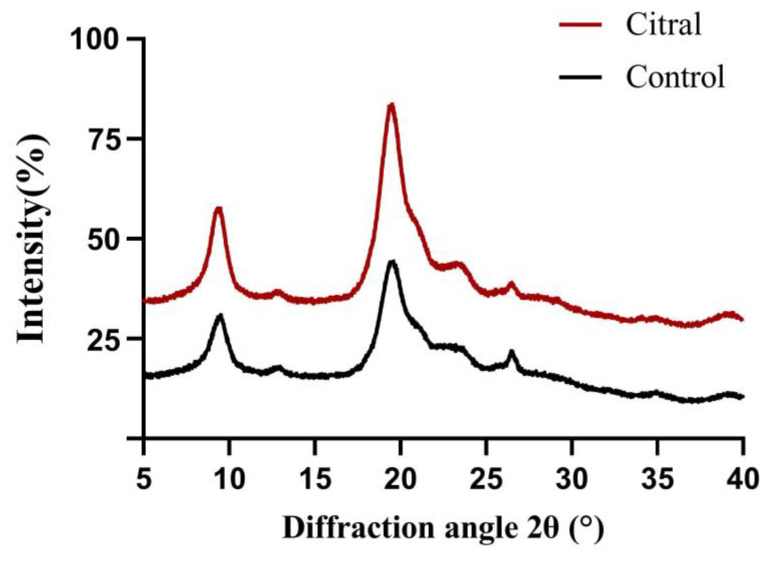
The α-crystalline structure of chitin samples was analyzed by X-ray diffraction. As shown in Figure 6, the crystal reflection peaks were located at 9 and 19°. The chitin crystallinity indices of the control and citral-treated samples were 60.8 and 41.2%, respectively. The results indicate that citral destroyed the chemical structure of chitin to varying degrees.
Figure 6.
Comparison of X-ray powder diffractograms of the chitin of R. flaviceps in the control and citral-treated groups.
3.9. Differential Scanning Calorimetry
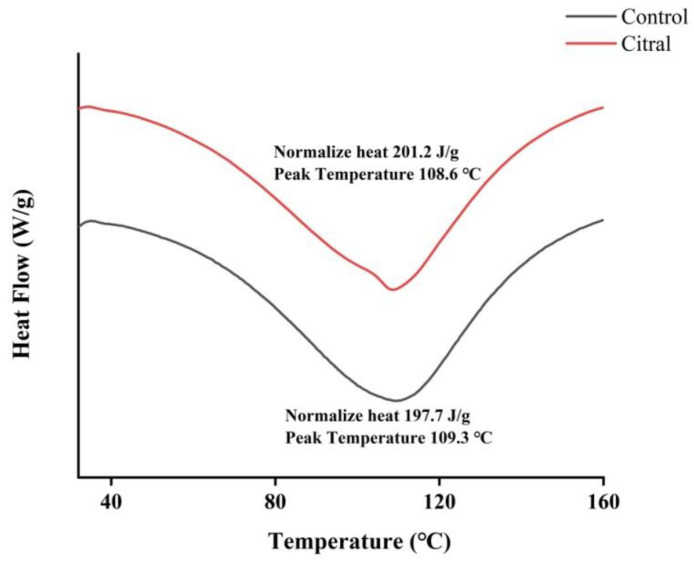
The thermal stability of the chitin samples was analyzed by differential scanning calorimetry. As shown in Figure 7, the heat value of the control group was 201.2 J/g, which was significantly higher than that of the treatment group. The maximum decomposition temperature was 109.3 °C in the control group and 108.6 °C in the citral-treated group. These results indicate that treatment with citral changed the thermal stability of chitin, which was consistent with the results of the previous two tests.
Figure 7.
DSC thermogram of the chitin of R. flaviceps in the control and citral-treated groups.
4. Discussion
The findings presented here reveal that the main components of lemongrass EO are cis-citral (36.51%), trans-citral (31.42%), and geraniol (8.78%); see Table 1. In general, citral (i.e., a mixture of cis- and trans-citral) was the major component of C. citratus EO, in agreement with the results of Boukhatem et al. [67], Pinto et al. [68], Feriotto et al. [21], Brugger et al. [69], Manh et al. [28], Soonwera and Sittichok [30], Aungtikun et al. [34], and Loko et al. [22], with slight differences in the relative content. However, previous studies also show that citronellal [7] and geraniol [8] are the major components of C. citratus EO, which is not in accordance with our results (Table S1). Genotypic variations, cultivation techniques, extraction method, and agricultural and environmental conditions can influence the chemical composition of plant EOs of the same species [30,34].
In this study, the results clearly demonstrate that the lemongrass EO had insecticidal efficacy against R. flaviceps (Table 2). This is in agreement with Xie et al. [70], who demonstrated the antitermitic activity of Syzgium aromaticum EO against R. chinensis (LC50 = 12.5 μg/g) after 7 d. Studies also reported the effectiveness of Eugenia caryophyllata EO against Coptotermes formosanus [71], R. speratus [72], and Odontotermes obesus [73]. Similarly, Pandey et al. [74] also reported the antitermitic activity of Sy. aromaticum EO on O. assamensis. Yang et al. [3] recently demonstrated that the LC50 value of spearmint EO against R. dabieshanensis was 0.194 μL/L.
There are no previous studies on the insecticidal activities of lemongrass EO against R. flaviceps; however, there are previous reports on the insecticidal potential of C. citratus EO. In the previous reports, C. citratus EO exhibited insecticidal activities against a variety of pest insects belonging to the orders Hemiptera [55], Coleoptera [75], Lepidoptera [59,60], and Diptera [28,30]. These bioassay results demonstrate that C. citratus EO has a significant insecticidal effect, which is worthy of further development in the future.
Our results demonstrate that the lemongrass EO and its major component, citral, had strong vapor activity in R. flaviceps. Similar results were obtained for citral (LC50 of 0.01 μL/L), in terms of its good termiticidal activity against R. chinensis [76]. Additionally, citral is shown to possess strong vapor toxicity against M. domestica (LC50 = 0.74 μL/L) [77]. Similarly, Lee et al. [78], Palacios et al. [79], and Kumar et al. [80] found that citral presents an insecticidal effect against M. domestica. The results of the above studies suggest that citral presents similar trends in toxicity for various insect species.
Interestingly, not only did lemongrass EO and its major component show excellent fumigation activity against R. flaviceps workers, but they also significantly affected the walking and gripping ability of workers when applied to their antennae. Similarly, Zhang et al. [81] reported that Citrus paradisi EO and its main compounds significantly suppressed the walking and gripping abilities of Solenopsis invicta workers. Fu et al. [82] also reported that camphor EO affects attacking, feeding, and climbing behaviors in S. invicta workers. Any reduced walking, gripping, and climbing abilities of social insects results in their inability to adapt to the environment. The above conclusions indicate that the vapor activity of C. citratus EO and citral may be related to their effects on the social behaviors of the insects.
We observed a significant decrease in chitin content in the citral-treated group. Further, through the FTIR analysis results, it was found that the chitin of R. flaviceps exists in the α-form (α-chitin). This is consistent with the previous reports of Zhang et al. [83] and Shah et al. [66], who showed that most insect chitin exists in the α-form. Crystalline α-chitin usually exhibits FTIR bands in the range of 1550, 1620, and 1650 cm−1 [66,84]. Based on this, it was found that citral treatment induced changes in the chemical structure of R. flaviceps chitin.
In the TGA analysis plot of the extracted chitin structures, mass loss was seen in two steps (Figure 5). In the first step, 8% to 10% weight loss was recorded due to water vaporization [66,85,86]; meanwhile, in the second step, the decomposition could be attributed to the decomposition of chitin saccharide structures [87]. In a previous study, the DTGmax decomposition temperature of α-chitin extracted from Sitotroga cerealella was 388 °C [66]. In addition, Wang et al. [88] found that the DTGmax decomposition temperature of α-chitin extracted from organisms such as crab, shrimp, krill, and crayfish ranged between 350 and 381 °C. Therefore, the DTGmax disintegration temperatures of α-chitin structures extracted from different organisms are different. According to Aranaz et al. [89], the decomposition temperature of chitin affects its usefulness. In the present study, the disintegration temperature of chitin extracted from the control and citral-treated groups was similar to that reported in other studies, varying between 385 and 389 °C [66]. TGA showed that treatment with citral slightly reduced the thermal stability of chitin, which may also cause changes in its chemical structure and reduce its crystallinity. These differences may be due to the N-acetylated polymer units of chitin being more stable in the control group than in the treated group. This is consistent with the results of the previous FTIR analysis. Zia et al. [90] reported that chitin has a highly ordered crystal structure. The XRD analysis results in this study were consistent with previous findings [66,91]. As shown in Figure 6, a broad signal centered at 2θ = 9° was presented, which was attributed to the GlcNAc sequence [92]. Furthermore, due to the GlcN sequence, the intensity of the broad signal was centered at 2θ = 19° [66].
In addition, it is known in the literature that most of the EOs and their major components can exert their toxic efficacy on insects notably through inhibition of P450 cytochromes (CYPs), GABA receptors, octopamine synapses, and the inhibition of acetylcholinesterase (AchE) [3]. Alves et al. [38] found that lemongrass EO and citral showed AChE inhibitory efficacy in Callosobruchus maculatu. Decreased AChE activity causes a direct change in insect behavior, such as flight, copulation, and oviposition, as well as changes in many other biological processes of insects [38]. Therefore, the exact toxicity mode of action and target of lemongrass EO and citral against the tested R. flaviceps in this study need to be revealed and confirmed by further experiments.
5. Conclusions
In this study, we clearly demonstrated that lemongrass EO and citral show potential vapor toxicity against R. flaviceps. The higher vapor toxicity of the lemongrass EO can be ascribed to its major constituent, citral. In addition, the lemongrass EO and citral significantly inhibited the walking and gripping abilities of R. flaviceps workers. Analysis results indicate that citral significantly reduced the content of chitin and changed the chemical structure in R. flaviceps. As such, we expected that citral has great potential for development into a new type of termite control agent. Future research should include formulation development, in order to determine whether sustained-release formulations can be designed, which may be delivered by termite workers to the nest to achieve the vapor of other individuals in the nest, as well as study the effects of temperature on such a slow-release application.
Supplementary Materials
The following supporting information can be downloaded at: www.mdpi.com/article/10.3390/insects13090812/s1, Table S1: Major components of Cymbopogon citratus EO from different origins previously reported.
Author Contributions
Conceptualization, Y.X. and C.J.; methodology, C.J., B.L. and D.Z.; software, C.J.; validation, C.J., B.L. and Z.Z.; formal analysis, C.J.; investigation, H.H.; resources, H.H.; data curation, H.H.; writing—original draft preparation, Y.X. and C.J.; writing—review and editing, Y.X., B.L., Z.Z. and D.Z.; visualization, Z.Z. and D.Z.; supervision, Y.X. and D.Z.; project administration, Y.X.; funding acquisition, Y.X. All authors have read and agreed to the published version of the manuscript.
Institutional Review Board Statement
Not applicable.
Informed Consent Statement
Not applicable.
Data Availability Statement
The dataset utilized in this study is available upon request.
Conflicts of Interest
The authors declare no conflict of interest.
Funding Statement
This research received no external funding.
Footnotes
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations.
References
- 1.Ahmad F., Fouad H., Liang S.Y., Hu Y., Mo J.C. Termites and Chinese agricultural system: Applications and advances in integrated termite management and chemical control. Insect Sci. 2021;28:2–20. doi: 10.1111/1744-7917.12726. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Zhou J., Bai Y., Zhong H., Li G. Effect of nitenpyram on the control of Reticulitermes flaviceps. Int. J. Trop. Insect Sci. 2021;41:471–477. doi: 10.1007/s42690-020-00228-2. [DOI] [Google Scholar]
- 3.Yang X., Han H., Li B., Zhang D., Zhang Z., Xie Y. Fumigant toxicity and physiological effects of spearmint (Mentha spicata, Lamiaceae) essential oil and its major constituents against Reticulitermes dabieshanensis. Ind. Crop. Prod. 2021;171:113894. doi: 10.1016/j.indcrop.2021.113894. [DOI] [Google Scholar]
- 4.Benelli G., Pavela R., Petrelli R., Cappellacci L., Bartolucci F., Canale A., Maggi F. Origanum syriacum subsp. Syriacum: From an ingredient of Lebanese ‘manoushe’ to a source of effective and eco-friendly botanical insecticides. Ind. Crop. Prod. 2019;134:26–32. doi: 10.1016/j.indcrop.2019.03.055. [DOI] [Google Scholar]
- 5.Yagi S., Mohammed A.B.A., Tzanova T., Schohn H., Abdelgadir H., Stefanucci A., Mollica A., Zengin G. Chemical profile, antiproliferative, antioxidant, and enzyme inhibition activities and docking studies of Cymbopogon schoenanthus (L.) Spreng. and Cymbopogon nervatus (Hochst.) Chiov. from Sudan. J. Food Biochem. 2019;44:e13107. doi: 10.1111/jfbc.13107. [DOI] [PubMed] [Google Scholar]
- 6.Barbosa L.C.A., Pereira U.A., Martinazzo A.P., Maltha C.R.Á., Teixeira R.R., Melo E.D.C. Evaluation of the chemical composition of Brazilian commercial Cymbopogon citratus (DC) Stapf samples. Molecules. 2008;13:1864–1874. doi: 10.3390/molecules13081864. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Li C., Luo Y., Zhang W., Cai Q., Wu X., Tan Z., Chen R., Chen Z., Wang S., Zhang L. A comparative study on chemical compositions and biological activities of four essential oils: Cymbopogon citratus (DC.) Stapf, Cinnamomum cassia (L.) Presl, Salvia japonica Thunb. and Rosa rugosa Thunb. J. Ethnopharmacol. 2021;280:114472. doi: 10.1016/j.jep.2021.114472. [DOI] [PubMed] [Google Scholar]
- 8.Zhang X., Zhu H., Wang J., Li F., Wang J., Ma X., Li Y., Huang Y., Liu Z., Zhang L., et al. Anti-microbial activity of citronella (Cymbopogon citratus) essential oil separation by ultrasound assisted ohmic heating hydrodistillation. Ind. Crop. Prod. 2022;176:114299. doi: 10.1016/j.indcrop.2021.114299. [DOI] [Google Scholar]
- 9.Padalia R.C., Verma R.C., Chauhan A., Goswami P., Singh V.R., Verma S.K., Singh N., Kurmi A., Darokar M.P., Saikia D. p-Menthenols chemotype of Cymbopogon distans from India: Composition, antibacterial and antifungal activity of the essential oil against pathogens. J. Essent. Oil Res. 2018;30:40–46. doi: 10.1080/10412905.2017.1375035. [DOI] [Google Scholar]
- 10.Gao S., Liu G., Li J., Chen J., Li L., Li Z., Zhang X., Zhang S., Thorne R.F., Zhang S. Antimicrobial activity of Lemongrass essential oil (Cymbopogon flexuosus) and its active component citral against dual-species biofilms of Staphylococcus aureus and Candida species. Front. Cell. Infect. Microbiol. 2020;10:603858. doi: 10.3389/fcimb.2020.603858. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Verma R.S., Padalia R.C., Goswami P., Verma S.K., Chauhan A., Singh V.R., Darokar M.P. Chemical composition and antibacterial activity of p-menthane chemotype of Cymbopogon martini (Roxb.) W. Watson (Poaceae) from India. J. Essent. Oil Res. 2018;30:182–188. doi: 10.1080/10412905.2018.1429327. [DOI] [Google Scholar]
- 12.Nikolić M., Marković T., Marković D., Calhelha R.C., Fernandes Â., Ferreira I.C.F.R., Stojković D., Ćirić A., Glamočlija J., Soković M. Chemical composition and biological properties of Pelargonium graveolens, Leptospermum petersonii and Cymbopogon martini var. motia essential oils and of Rosa centifolia absolute. J. Serb. Chem. Soc. 2021;86:1291–1303. doi: 10.2298/JSC210729096N. [DOI] [Google Scholar]
- 13.Kandimalla R., Kalita S., Choudhury B., Dash S., Kalita K., Kotoky J. Chemical composition and anti-candidiasis mediated wound healing property of Cymbopogon nardus essential oil on chronic diabetic wounds. Front. Pharmacol. 2016;7:198. doi: 10.3389/fphar.2016.00198. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Kaur H., Bhardwaj U., Kaur R., Kaur H. Chemical composition and antifungal potential of citronella (Cymbopogon nardus) leaves essential oil and its major compounds. J. Essent. Oil Bear. Plants. 2021;24:571–581. doi: 10.1080/0972060X.2021.1942231. [DOI] [Google Scholar]
- 15.Sawadogo I., Paré A., Kaboré D., Montet D., Durand N., Bouajila J., Zida E.P., Sawadogo-Lingani H., Nikiéma P.A., Nebié R.H.C., et al. Antifungal and antiaflatoxinogenic effects of Cymbopogon citratus, Cymbopogon nardus, and Cymbopogon schoenanthus essential oils alone and in combination. J. Fungi. 2022;8:117. doi: 10.3390/jof8020117. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Omar E., Pavlović I., Drobac M., Radenković M., Branković S., Kovačević N. Chemical composition and spasmolytic activity of Cymbopogon nervatus (Hochst.) Chiov. (Poaceae) essential oil. Ind. Crop. Prod. 2016;91:249–254. doi: 10.1016/j.indcrop.2016.07.013. [DOI] [Google Scholar]
- 17.Pavlović I., Omar E., Drobac M., Radenković M., Branković S., Kovačević N. Chemical composition and spasmolytic activity of Cymbopogon schoenanthus (L.) Spreng. (Poaceae) essential oil from Sudan. Arch. Biol. Sci. 2017;69:409–415. doi: 10.2298/ABS160506113P. [DOI] [Google Scholar]
- 18.Bellik F.Z., Benkaci-Ali F., Alsafra Z., Eppe G., Tata S., Sabaou N., Zidani R. Chemical composition, kinetic study and antimicrobial activity of essential oils from Cymbopogon schoenanthus L. Spreng extracted by conventional and microwave-assisted techniques using cryogenic grinding. Ind. Crop. Prod. 2019;139:111505. doi: 10.1016/j.indcrop.2019.111505. [DOI] [Google Scholar]
- 19.Verma R.S., Verma S.K., Tandon S., Padalia R.C., Darokar M.P. Chemical composition and antimicrobial activity of Java citronella (Cymbopogon winterianus Jowitt ex Bor) essential oil extracted by different methods. J. Essent. Oil Res. 2020;32:449–455. doi: 10.1080/10412905.2020.1787885. [DOI] [Google Scholar]
- 20.Kumar A., Jnanesha A.C., Chanotiya C.S., Srivastava S., Pant Y. Biplot investigation for essential oil yield and chemical compositions under the Deccan Plateau region of southern India in cultivars of Java citronella (Cymbopogon winterianus Jowitt) Ind. Crop. Prod. 2022;175:114249. doi: 10.1016/j.indcrop.2021.114249. [DOI] [Google Scholar]
- 21.Feriotto G., Marchetti N., Costa V., Beninati S., Tagliati F., Mischiati C. Chemical composition of essential oils from Thymus vulgaris, Cymbopogon citratus, and Rosmarinus officinalis, and their effects on the HIV-1 tat protein function. Chem. Biodivers. 2018;15:e1700436. doi: 10.1002/cbdv.201700436. [DOI] [PubMed] [Google Scholar]
- 22.Loko Y.L.E., Fagla S.M., Kassa P., Ahouansou C.A., Toffa J., Glinma B., Dougnon V., Koukoui O., Djogbenou S.L., Tamò M., et al. Bioactivity of essential oils of Cymbopogon citratus (DC) Stapf and Cymbopogon nardus (L.) W. Watson from Benin against Dinoderus porcellus Lesne (Coleoptera: Bostrichidae) infesting yam chips. Int. J. Trop. Insect Sci. 2021;41:511–524. doi: 10.1007/s42690-020-00235-3. [DOI] [Google Scholar]
- 23.Caballero-Gallardo K., Rodriguez-Niño D., Fuentes-Lopez K., Stashenko E., Olivero-Verbel J. Chemical composition and bioactivity of essential oils from Cymbopogon nardus L. and Rosmarinus officinalis L. against Ulomoides dermestoides (Fairmaire, 1893) (Coleoptera: Tenebrionidae) J. Essent. Oil Bear. Plants. 2021;24:547–560. doi: 10.1080/0972060X.2021.1936205. [DOI] [Google Scholar]
- 24.Malti C.E.W., Haci I.A.E., Hassani F., Paoli M., Gibernau M., Tomi F., Casanova J., Bekhechi C. Composition, chemical variability and biological activity of Cymbopogon schoenanthus essential oil from Central Algeria. Chem. Biodivers. 2020;17:e2000138. doi: 10.1002/cbdv.202000138. [DOI] [PubMed] [Google Scholar]
- 25.Barbosa D.R.S., Santos R.B.V., Santos F.M.P., Junior P.J.S., Neto F.M.O., Silva G.N., Dutra K.A., Navarro D.M.A.F. Evaluation of Cymbopogon flexuosus and Alpinia zerumbet essential oils as biopesticides against Callosobruchus maculate. J. Plant Dis. Prot. 2022;129:125–136. doi: 10.1007/s41348-021-00511-5. [DOI] [Google Scholar]
- 26.Piasecki B., Biernasiuk A., Skiba A., Skalicka-Woźniak K., Ludwiczuk A. Composition, Anti-MRSA activity and toxicity of essential oils from Cymbopogon Species. Molecules. 2021;26:7542. doi: 10.3390/molecules26247542. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Ntonga P.A., Baldovini N., Mouray E., Mambu L., Belong P., Grellier P. Activity of Ocimum basilicum, Ocimum canum, and Cymbopogon citratus essential oils against Plasmodium falciparum and mature-stage larvae of Anopheles funestus s.s. Parasite. 2014;21:33. doi: 10.1051/parasite/2014033. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Manh H.D., Hue D.T., Hieu N.T.T., Tuyen D.T.T., Tuyet O.T. The mosquito larvicidal activity of essential oils from Cymbopogon and Eucalyptus species in Vietnam. Insects. 2020;11:128. doi: 10.3390/insects11020128. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Castillo-Morales R.M., Serrano S.O., Villamizar A.L.R., Mendez-Sanchez S.C., Duque J.E. Impact of Cymbopogon flexuosus (Poaceae) essential oil and primary components on the eclosion and larval development of Aedes aegypti. Sci. Rep. 2021;11:24291. doi: 10.1038/s41598-021-03819-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Soonwera M., Sittichok S. Adulticidal activities of Cymbopogon citratus (Stapf.) and Eucalyptus globulus (Labill.) essential oils and of their synergistic combinations against Aedes aegypti (L.), Aedes albopictus (Skuse), and Musca domestica (L.) Environ. Sci. Pollut. Res. 2020;27:20201–20214. doi: 10.1007/s11356-020-08529-2. [DOI] [PubMed] [Google Scholar]
- 31.Bricarello P.A., Barros G.P., Seugling J., Podestá R., Velerinho M.B., Mazzarino L. Ovicidal, larvicidal and oviposition repelling action of a nanoemulsion of citronella essential oil (Cymbopogon winterianus) on Cochliomyia hominivorax (Diptera: Calliphoridae) J. Asia-Pac. Èntomol. 2021;24:724–730. doi: 10.1016/j.aspen.2021.06.006. [DOI] [Google Scholar]
- 32.Ilahi I., Yousafzai A.M., Hao T.U., Ali H., Rahim A., Sajad M.A., Khan A.N., Ahmad A., Ullah S., Zaman S., et al. Oviposition deterrence and adult emergence inhibition activities of Cymbopogon nardus against Culex quinquefasciatus with study on non-target organisms. Appl. Ecol. Environ. Res. 2019;17:4915–4931. doi: 10.15666/aeer/1702_49154931. [DOI] [Google Scholar]
- 33.Agwunobi D.O., Pei T., Wang K., Yu Z., Liu J. Effects of the essential oil from Cymbopogon citratus on mortality and morphology of the tick Haemaphysalis longicornis (Acari: Ixodidae) Exp. Appl. Acarol. 2020;81:37–50. doi: 10.1007/s10493-020-00485-3. [DOI] [PubMed] [Google Scholar]
- 34.Aungtikun J., Soonwera M., Sittichok S. Insecticidal synergy of essential oils from Cymbopogon citratus (Stapf.), Myristica fragrans (Houtt.), and Illicium verum Hook. f. and their major active constituents. Ind. Crop. Prod. 2021;164:113386. doi: 10.1016/j.indcrop.2021.113386. [DOI] [Google Scholar]
- 35.Silva L.C., Perinotto W.M.S., Sá A., Souza M.A.A., Bitencourt R.O.B., Sanavria A., Santos H.A., Marie-Magdeleine C., Angelo I.C. In vitro acaricidal activity of Cymbopogon citratus, Cymbopogon nardus and Mentha arvensis against Rhipicephalus microplus (Acari: Ixodidae) Exp. Parasitol. 2020;216:107937. doi: 10.1016/j.exppara.2020.107937. [DOI] [PubMed] [Google Scholar]
- 36.Li M., Liu B., Bernigaud C., Fischer K., Guillot J., Fang F. Lemongrass (Cymbopogon citratus) oil: A promising miticidal and ovicidal agent against Sarcoptes scabiei. PLoS Negl Trop Dis. 2020;14:e0008225. doi: 10.1371/journal.pntd.0008225. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Rodríguez-González Á., Álvarez-García S., González-López Ó., Silva F.D., Casquero P.A. Insecticidal properties of Ocimum basilicum and Cymbopogon winterianus against Acanthoscelides obtectus, insect pest of the common Bean (Phaseolus vulgaris, L.) Insects. 2019;10:151. doi: 10.3390/insects10050151. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Alves M.S., Campos I.M., Brito D.M.C., Cardoso C.M., Pontes E.G., Souza M.A.A. Efficacy of lemongrass essential oil and citral in controlling Callosobruchus maculatus (Coleoptera: Chrysomelidae) a post-harvest cowpea insect pest. Crop Prot. 2019;119:191–196. doi: 10.1016/j.cropro.2019.02.007. [DOI] [Google Scholar]
- 39.Aous W., Benchabane O., Outaleb T., Hazzit M., Mouhouche F., Yekkour A., Baaliouamer A. Essential oils of Cymbopogon schoenanthus (L.) Spreng from Algerian Sahara: Chemical variability, antioxidant, antimicrobial and insecticidal properties. J. Essent. Oil Res. 2019;31:562–572. doi: 10.1080/10412905.2019.1612790. [DOI] [Google Scholar]
- 40.Utono I.M., Coote C., Gibson G. Field study of the repellent activity of ‘Lem-ocimum’-treated double bags against the insect pests of stored sorghum, Tribolium castaneum and Rhyzopertha dominica, in northern Nigeria. J. Stored Prod. Res. 2014;59:222–230. doi: 10.1016/j.jspr.2014.03.005. [DOI] [Google Scholar]
- 41.Moutassem D., Bellik Y., Sannef M.E.H. Toxicity and repellent activities of Thymus pallescens and Cymbopogon citratus essential oils against Sitophilus granaries. Plant Prot. Sci. 2021;57:297–309. doi: 10.17221/185/2020-PPS. [DOI] [Google Scholar]
- 42.Franz A.R., Knaak N., Fiuza L.M. Toxic effects of essential plant oils in adult Sitophilus oryzae (Linnaeus) (Coleoptera: Curculionidae) Rev. Bras. Èntomol. 2011;55:116–120. doi: 10.1590/S0085-56262011000100018. [DOI] [Google Scholar]
- 43.Devi M.A., Nameirakpam B., Devi T.B., Mayanglambam S., Singh K.D., Sougrakpam S.S., Shadia S., Tongbram M., Singh S.D., Sahoo D., et al. Chemical compositions and insecticidal efficacies of four aromatic essential oils on rice weevil Sitophilus oryzae L. Int. J. Trop. Insect Sci. 2020;40:549–559. doi: 10.1007/s42690-020-00102-1. [DOI] [Google Scholar]
- 44.Tawfeek M.E., Ali H.M., Akrami M., Salem M.Z.M. Potential insecticidal activity of four essential oils against the rice weevil, Sitophilus oryzae (L.) (Coleoptera: Curculionidae) BioResources. 2021;16:7767–7783. doi: 10.15376/biores.16.4.7767-7783. [DOI] [Google Scholar]
- 45.Hernández-Lambraño R., Pajaro-Castro N., Caballero-Gallardo K., Stashenko E., Olivero-Verbel J. Essential oils from plants of the genus Cymbopogon as natural insecticides to control stored product pests. J. Stored Prod. Res. 2015;62:81–83. doi: 10.1016/j.jspr.2015.04.004. [DOI] [Google Scholar]
- 46.Araújo A.M.N., Oliveira J.V., França S.M., Navarro D.N.A.F., Barbosa D.R.S., Dutra K.A. Toxicity and repellency of essential oils in the management of Sitophilus zeamais. Rev. Bras. Eng. Agríc. Ambient. 2019;23:372–377. doi: 10.1590/1807-1929/agriambi.v23n5p372-377. [DOI] [Google Scholar]
- 47.Wang X., Hao Q., Chen Y., Jiang S., Yang Q., Li Q. The effect of chemical composition and bioactivity of several essential oils on Tenebrio molitor (Coleoptera: Tenebrionidae) J. Insect Sci. 2015;15:116. doi: 10.1093/jisesa/iev093. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Feroz A. Efficacy and cytotoxic potential of deltamethrin, essential oils of Cymbopogon citratus and Cinnamonum camphora and their synergistic combinations against stored product pest, Trogoderma granarium (Everts) J. Stored Prod. Res. 2020;87:101614. doi: 10.1016/j.jspr.2020.101614. [DOI] [Google Scholar]
- 49.Olivero-Verbel J., Nerio L.S., Stashenko E.E. Bioactivity against Tribolium castaneum Herbst (Coleoptera: Tenebrionidae) of Cymbopogon citratus and Eucalyptus citriodora essential oils grown in Colombia. Pest Manag. Sci. 2010;66:664–668. doi: 10.1002/ps.1927. [DOI] [PubMed] [Google Scholar]
- 50.Caballero-Gallardo K., Olivero-Verbel J., Stashenko E.E. Repellency and toxicity of essential oils from Cymbopogon martinii, Cymbopogon flexuosus and Lippia origanoides cultivated in Colombia against Tribolium castaneum. J. Stored Prod. Res. 2012;50:62–65. doi: 10.1016/j.jspr.2012.05.002. [DOI] [Google Scholar]
- 51.Bossou A.D., Ahoussi E., Ruysbergh E., Adams A., Smagghe G., De Kimpe N., Avlessi F., Sohounhloue D.C.K., Mangelinckx S. Characterization of volatile compounds from three Cymbopogon species and Eucalyptus citriodora from Benin and their insecticidal activities against Tribolium castaneum. Ind. Crop. Prod. 2015;76:306–317. doi: 10.1016/j.indcrop.2015.06.031. [DOI] [Google Scholar]
- 52.Ahmad F., Iqbal N., Zaka S.M., Qureshi M.K., Saeed Q., Khan K.A., Ghramhe H.A., Ansari M.J., Jaleel W., Aasim M., et al. Comparative insecticidal activity of different plant materials from six common plant species against Tribolium castaneum (Herbst) (Coleoptera: Tenebrionidae) Saudi J. Biol. Sci. 2019;26:1804–1808. doi: 10.1016/j.sjbs.2018.02.018. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Devi M.A., Sahoo D., Singh T.B., Rajashekar Y. Toxicity, repellency and chemical composition of essential oils from Cymbopogon species against red flour beetle Tribolium castaneum Herbst (Coleoptera: Tenebrionidae) J. Consum. Prot. Food Saf. 2020;15:181–191. doi: 10.1007/s00003-019-01264-y. [DOI] [Google Scholar]
- 54.Plata-Rueda A., Martínez L.C., Rolim G.S., Coelho R.P., Santos M.H., Tavares W.S., Zanuncio J.C., Serrão J.E. Insecticidal and repellent activities of Cymbopogon citratus (Poaceae) essential oil and its terpenoids (citral and geranyl acetate) against Ulomoides dermestoides. Crop Prot. 2020;137:105299. doi: 10.1016/j.cropro.2020.105299. [DOI] [Google Scholar]
- 55.Kobenan K.C., Bini K.K.N., Kouakou M., Kouadio I.S., Zengin G., Ochou G.E.C., Boka N.R.K., Menozzi P., Ochou O.G., Dick A.E. Chemical composition and spectrum of insecticidal activity of the essential oils of Ocimum gratissimum L. and Cymbopogon citratus Stapf on the main insects of the cotton entomofauna in Côte d’Ivoire. Chem. Biodivers. 2021;18:e2100497. doi: 10.1002/cbdv.202100497. [DOI] [PubMed] [Google Scholar]
- 56.Hernández-Lambraño R., Caballero-Gallardo K., Olivero-Verbel J. Toxicity and antifeedant activity of essential oils from three aromatic plants grown in Colombia against Euprosterna elaeasa and Acharia fusca (Lepidoptera: Limacodidae) Asian Pac. J. Trop. Biomed. 2014;4:695–700. doi: 10.12980/APJTB.4.2014APJTB-2014-0178. [DOI] [Google Scholar]
- 57.Diabate S., Martin T., Murungi L.K., Fiaboe K.K.M., Subramanian S., Wesonga J., Deletre E. Repellent activity of Cymbopogon citratus and Tagetes minuta and their specific volatiles against Megalurothrips sjostedti. J. Appl. Entomol. 2019;143:855–866. doi: 10.1111/jen.12651. [DOI] [Google Scholar]
- 58.Jovanović J., Krnjajić S., Ćirković J., Radojković A., Popović T., Branković G., Branković Z. Effect of encapsulated lemongrass (Cymbopogon citratus L.) essential oil against potato tuber moth Phthorimaea operculella. Crop Prot. 2020;132:105109. doi: 10.1016/j.cropro.2020.105109. [DOI] [Google Scholar]
- 59.Murcia-Meseguer A., Alves T.J., Budia F., Ortiz A., Medina P. Insecticidal toxicity of thirteen commercial plant essential oils against Spodoptera exigua (Lepidoptera: Noctuidae) Phytoparasitica. 2018;46:233–245. doi: 10.1007/s12600-018-0655-9. [DOI] [Google Scholar]
- 60.Labinas M.A., Crocomo W.B. Effect of java grass (Cymbopogon winteranus) essential oil on fall armyworm Spodoptera frugiperda (J. E. Smith, 1979) (Lepidoptera, Noctuidae) Acta Sci. 2002;24:1401–1405. [Google Scholar]
- 61.Oliveira E.R., Alves D.S., Carvalho G.A., Oliveira B.M.R.G., Aazza S., Bertolucci S.K.V. Toxicity of Cymbopogon flexuosus essential oil and citral for Spodoptera frugiperda. Ciênc. Agrotec. 2018;42:408–419. doi: 10.1590/1413-70542018424013918. [DOI] [Google Scholar]
- 62.Jiang Z.L., Akhtar Y., Zhang X., Bradbury R., Isman M.B. Insecticidal and feeding deterrent activities of essential oils in the cabbage looper, Trichoplusia ni (Lepidoptera: Noctuidae) J. Appl. Entomol. 2012;136:191–202. doi: 10.1111/j.1439-0418.2010.01587.x. [DOI] [Google Scholar]
- 63.Tak J.H., Jovel E., Isman M.B. Synergistic interactions among the major constituents of lemongrass essential oil against larvae and an ovarian cell line of the cabbage looper, Trichoplusia ni. J. Pest Sci. 2017;90:735–744. doi: 10.1007/s10340-016-0827-7. [DOI] [Google Scholar]
- 64.Ngongang M.D.T., Eke P., Sameza M.L., Mback M.N.L.N., Djiéto-Lordon C., Boyom F.F. Chemical constituents of essential oils from Thymus vulgaris and Cymbopogon citratus and their insecticidal potential against the tomato borer, Tuta absoluta (Lepidoptera: Gelechiidae) Int. J. Trop. Insect Sci. 2022;42:31–43. doi: 10.1007/s42690-021-00514-7. [DOI] [Google Scholar]
- 65.Adams R.P. Identification of Essential Oil Components by Gas Chromatography/Mass Spectrometry. 4th ed. Allured Publishing Corporation; Carol Stream, IL, USA: 2007. [Google Scholar]
- 66.Shah S., Ma M., Ali A., Kaya M., Li X.G., Wu G., Yang F.L. Effects of diallyl trisulfide, an active substance from garlic essential oil, on structural chemistry of chitin in Sitotroga cerealella (Lepidoptera: Gelechiidae) Pestic. Biochem. Physiol. 2021;172:104765. doi: 10.1016/j.pestbp.2020.104765. [DOI] [PubMed] [Google Scholar]
- 67.Boukhatem M.N., Ferhat M.A., Kameli A., Saidi F., Kebir H.T. Lemon grass (Cymbopogon citratus) essential oil as a potent anti-inflammatory and antifungal drugs. Libyan J. Med. 2014;9:25431. doi: 10.3402/ljm.v9.25431. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 68.Pinto Z.T., Sánchez F.F., dos Santos A.R., Amaral A.C., Ferreira J.L., Escalona-Arranz J.C., Queiroz M.M. Chemical composition and insecticidal activity of Cymbopogon citratus essential oil from Cuba and Brazil against housefly. Rev. Bras. Parasitol. Vet. 2015;24:36–44. doi: 10.1590/S1984-29612015006. [DOI] [PubMed] [Google Scholar]
- 69.Brugger B.P., Martinez L.C., Plata-Rueda A., Castrol B.M.C., Soares M.A., Wilcken C.F., Carvalho A.G., Serao J.E., Zanuncio J.C. Bioactivity of the Cymbopogon citratus (Poaceae) essential oil and its terpenoid constituents on the predatory bug, Podisus nigrispinus (Heteroptera: Pentatomidae) Sci. Rep. 2019;9:8358. doi: 10.1038/s41598-019-44709-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 70.Xie Y., Yang Z., Cao D., Rong F., Ding H., Zhang D. Antitermitic and antifungal activities of eugenol and its congeners from the flower buds of Syzgium aromaticum (Clove) Ind. Crop. Prod. 2015;77:780–786. doi: 10.1016/j.indcrop.2015.09.044. [DOI] [Google Scholar]
- 71.Zhu B.C., Henderson G., Chen F., Fei H., Laine R.A. Evaluation of vetiver oil and seven insect-active essential oils against the Formosan subterranean termite. J. Chem. Ecol. 2001;27:1617–1625. doi: 10.1023/A:1010410325174. [DOI] [PubMed] [Google Scholar]
- 72.Park I.K., Shin S.C. Fumigant activity of plant essential oils and components from garlic (Allium sativum) and clove bud (Eugenia caryophyllata) oils against the Japanese termite (Reticulitermes speratus Kolbe) J. Agric. Food Chem. 2005;53:4388–4392. doi: 10.1021/jf050393r. [DOI] [PubMed] [Google Scholar]
- 73.Gupta A., Sharma S., Naik S.N. Biopesticidal value of selected essential oils against pathogenic fungus, termites, and nematodes. Int. Biodeterior. Biodegrad. 2011;65:703–707. doi: 10.1016/j.ibiod.2010.11.018. [DOI] [Google Scholar]
- 74.Pandey A., Chattopadhyay P., Banerjee S., Pakshirajan K., Singh L. Antitermitic activity of plant essential oils and their major constituents against termite Odontotermes assamensis Holmgren (Isoptera: Termitidae) of North East India. Int. Biodeterior. Biodegrad. 2012;75:63–67. doi: 10.1016/j.ibiod.2012.09.004. [DOI] [Google Scholar]
- 75.Kabera J., Gasogo A., Uwamariya A., Ugirinshuti V., Nyetera P. Insecticidal effects of essential oils of Pelargonium raveolens and Cymbopogon citratus on Sitophilus zeamais (Motsch) Afr. J. Food Sci. 2011;5:366–375. doi: 10.5897/AJFS.9000170. [DOI] [Google Scholar]
- 76.Xie Y., Wang K., Huang Q., Lei C. Evaluation toxicity of monoterpenes to subterranean termite, Reticulitermes chinensis Snyder. Ind. Crop. Prod. 2014;53:163–166. doi: 10.1016/j.indcrop.2013.12.021. [DOI] [Google Scholar]
- 77.Zhang Z., Xie Y., Wang Y., Lin Z., Wang L., Li G. Toxicities of monoterpenes against housefly, Musca domestica L. (Diptera: Muscidae) Environ. Sci. Pollut. Res. 2017;24:24708–24713. doi: 10.1007/s11356-017-0219-4. [DOI] [PubMed] [Google Scholar]
- 78.Lee S., Peterson C.J., Coats J.R. Fumigation toxicity of monoterpenoids to several stored product insects. J. Stored Prod. Res. 2003;39:77–85. doi: 10.1016/S0022-474X(02)00020-6. [DOI] [Google Scholar]
- 79.Palacios S.M., Bertoni A., Rossi Y., Santander R. Insecticidal activity of essential oils from native medicinal plants of Central Argentina against the housefly, Musca domestica (L.) Parasitol. Res. 2009;106:207–212. doi: 10.1007/s00436-009-1651-2. [DOI] [PubMed] [Google Scholar]
- 80.Kumar P., Mishra S., Malik A., Satya S. Biocontrol potential of essential oil monoterpenes against Musca domestica (Diptera Muscidae) Ecotoxicol. Environ. Saf. 2014;100:1–6. doi: 10.1016/j.ecoenv.2013.11.013. [DOI] [PubMed] [Google Scholar]
- 81.Zhang N., Liao Y., Xie L., Zhang Z., Hu W. Using essential oils from Citrus paradisi as a fumigant for Solenopsis invicta workers and evaluating the oils’ effect on worker behavior. Environ. Sci. Pollut. Res. 2021;28:59665–59672. doi: 10.1007/s11356-021-14910-6. [DOI] [PubMed] [Google Scholar]
- 82.Fu J.T., Tang L., Li W.S., Wang K., Cheng D.M., Zhang Z.X. Fumigant toxicity and repellence activity of camphor essential oil from Cinnamonum camphora Siebold against Solenopsis invicta workers (Hymenoptera: Formicidae) J. Insect Sci. 2015;15:129. doi: 10.1093/jisesa/iev112. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 83.Zhang M., Haga A., Sekiguchi H., Hirano S. Structure of insect chitin isolated from beetle larva cuticle and silkworm (Bombyx mori) pupa exuvia. Int. J. Biol. Macromol. 2000;27:99–105. doi: 10.1016/S0141-8130(99)00123-3. [DOI] [PubMed] [Google Scholar]
- 84.Waśko A., Bulak P., Polak-Berecka M., Nowak K., Polakowski C., Bieganowski A. The first report of the physicochemical structure of chitin isolated from Hermetia illucens. Int. J. Biol. Macromol. 2016;92:316–320. doi: 10.1016/j.ijbiomac.2016.07.038. [DOI] [PubMed] [Google Scholar]
- 85.Kaya M., Bitim B., Mujtaba M., Koyuncu T. Surface morphology of chitin highly related with the isolated body part of butterfly (Argynnis pandora) Int. J. Biol. Macromol. 2015;81:443–449. doi: 10.1016/j.ijbiomac.2015.08.021. [DOI] [PubMed] [Google Scholar]
- 86.Kaya M., Mujtaba M., Ehrlich H., Salaberria A.M., Baran T., Amemiya C.T., Galli R., Akyuz L., Sargin I., Labidi J. On chemistry of γ-chitin. Carbohydr. Polym. 2017;176:177–186. doi: 10.1016/j.carbpol.2017.08.076. [DOI] [PubMed] [Google Scholar]
- 87.Paulino A.T., Simionato J.I., Garcia J.C., Nozaki J. Characterization of chitosan and chitin produced from silkworm crysalides. Carbohydr. Polym. 2006;64:98–103. doi: 10.1016/j.carbpol.2005.10.032. [DOI] [Google Scholar]
- 88.Wang Y., Chang Y., Yu L., Zhang C., Xu X., Xue Y., Li Z., Xue C. Crystalline structure and thermal property characterization of chitin from Antarctic krill (Euphausia superba) Carbohydr. Polym. 2013;92:90–97. doi: 10.1016/j.carbpol.2012.09.084. [DOI] [PubMed] [Google Scholar]
- 89.Aranaz I., Mengíbar M., Harris R., Pãnos I., Miralles B., Acosta N., Galed G., Heras Á. Functional characterization of chitin and chitosan. Curr. Chem. Biol. 2009;3:203–230. [Google Scholar]
- 90.Zia K.M., Bhatti I.A., Barikani M., Zuber M., Sheikh M.A. XRD studies of chitin-based polyurethane elastomers. Int. J. Biol. Macromol. 2008;43:136–141. doi: 10.1016/j.ijbiomac.2008.04.009. [DOI] [PubMed] [Google Scholar]
- 91.Wada M., Saito Y. Lateral thermal expansion of chitin crystals. J. Poly. Sci. Part B Poly. Phys. 2001;39:168–174. doi: 10.1002/1099-0488(20010101)39:1<168::AID-POLB150>3.0.CO;2-0. [DOI] [Google Scholar]
- 92.Jayakumar R., Selvamurugan N., Nair S.V., Tokura S., Tamura H. Preparative methods of phosphorylated chitin and chitosan—An overview. Int. J. Biol. Macromol. 2008;43:221–225. doi: 10.1016/j.ijbiomac.2008.07.004. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Data Availability Statement
The dataset utilized in this study is available upon request.