Abstract
Motivation
Ligand–receptor (LR) network analysis allows the characterization of cellular crosstalk based on single cell RNA-seq data. However, current methods typically provide a list of inferred LR interactions and do not allow the researcher to focus on specific cell types, ligands or receptors. In addition, most of these methods cannot quantify changes in crosstalk between two biological phenotypes.
Results
CrossTalkeR is a framework for network analysis and visualization of LR interactions. CrossTalkeR identifies relevant ligands, receptors and cell types contributing to changes in cell communication when contrasting two biological phenotypes, i.e. disease versus homeostasis. A case study on scRNA-seq of human myeloproliferative neoplasms reinforces the strengths of CrossTalkeR for characterization of changes in cellular crosstalk in disease.
Availability and implementation
CrosstalkeR is an R package available at: Github: https://github.com/CostaLab/CrossTalkeR.
Supplementary information
Supplementary data are available at Bioinformatics online.
1 Introduction
Understanding cellular crosstalk is vital for uncovering molecular mechanisms associated to cell differentiation and disease progression. Single cell RNA sequencing (scRNA-seq) enables the characterization of tissue heterogeneity at an unprecedented level (Armingol et al., 2020; Luecken and Theis, 2019). However, information of cellular proximity and crosstalk is not directly captured by common single cell sequencing protocols. Computational methods, which search for pairs of cell types expressing compatible ligand–receptor (LR) pairs, have thus become a powerful approach for dissecting cellular crosstalk from scRNA-seq data (Armingol et al., 2020). Current state-of-art LR inference methods, such as CellphoneDB (CPDB) (Efremova et al., 2019), basically provide a ranked list of hundreds of LR and cell pairs for a given scRNA-seq dataset. As interpreting such ranked lists is challenging, this calls for tools to simplify the complexity of these results, i.e. ranking of cell types or genes within a pair of cell types. Also, most LR inference methods focus on the analysis of a single phenotype scRNA-seq experiment and are not able to characterize changes in cellular crosstalk between pairs of phenotypes, e.g. disease versus homeostasis.
To fill these gaps, we developed CrossTalkeR, which aggregates predictions from CPDB (Efremova et al., 2019) as cell and cell/gene networks. First it ranks cell types, cell–receptors and cell–ligand pairs regarding their importance in cellular crosstalk based on certain network topology measures. Second, CrossTalkeR enables researchers to perform a phenotype based comparative LR analysis. In comparison to competing methods, CrossTalkeR has two main unique features: (i) it is the most complete method in regard to using network topological measures in both cell–cell network and cell–gene network levels and (ii) the only one exploring network topological measures to contrast changes in cellular crosstalk between two conditions (Supplementary Section S5 and Supplementary Tables S1 and S2). CrossTalkeR is implemented as an R package. It presents the results either graphically or in tabular form summarized as a HTML report/PDF report and stores all results in an R container to foster data sharing and reproducibility of results.
2 Overview and implementation
The input to CrossTalkeR is a data-table, in which each line has five columns consisting of (i) a ligand, (ii) a receptor, (iii) a sender cell type that expresses the ligand, (iv) a receiver cell type that expresses the receptor and (v) a positive score associated with the weight of the interaction, e.g. as predicted by CPDB (Efremova et al., 2019). Specifically, for CPDB this positive score (weight) corresponds to a function of the mean expression levels of the ligand and the receptor within the respective cell types (Efremova et al., 2019). From this input data, CrossTalkeR builds network representations and estimates node statistics to highlight particular cells or cell/gene pairs. All the analyses can be performed either on data corresponding to a single phenotype, or data from two phenotypes can be compared.
2.1 Network construction
CrossTalkeR constructs two representations of the LR network: a cell–cell interaction (CCI) network and a cell–gene interaction (CGI) network. In the CCI network, the nodes are defined by each cell-type and the directed edges are weighted by characteristics of the interactions, e.g. number of LR pairs and sum of weights of LR pairs. In the CGI network, the nodes represent gene and cell pairs (ligand with sender cell type, or receptor with receiving cell type) and the edge weights are given by the mean LR expression levels. Edges are always directed from ligands to receptors. A detailed description can be find in Supplementary Section S1.
2.2 Network topological measures
We use graph theoretic tools to rank nodes [cell types or cell–gene pairs of the CCI (and CGI) network]. We compute the following scores:
Influencer score. We compute the number of out-going edges, i.e. the number of signals sent by a cell.
Listener score. We compute the number of in-going edges per node. Nodes with high listener score represent nodes that receive many signals under the studied biological condition.
Moderator score. We compute the betweeness centrality of a node to measure the importance of a node to mediate communication between cells.
Importance score. We characterize the overall importance of nodes within the network by computing their PageRank score. Note that the PageRank can be interpreted probabilistically as the stationary probability distribution of a certain diffusion process on the network.
When comparing two different phenotypes, we obtain differential scores as follows. For the influencer, listener and moderator scores we simply compute the score difference for each node. For importance (pagerank), we compute log-odds of posteriors probabilities estimated via the pagerank scores. See Supplementary Sections S2 and S3 for details.
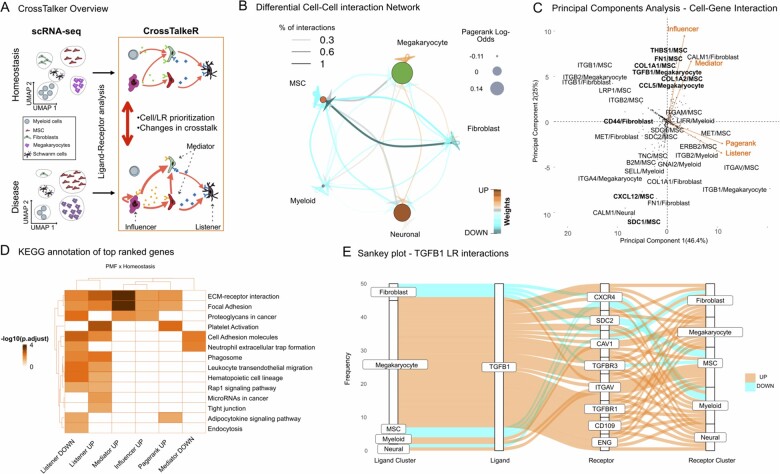
HTML/PDF report and graphical representations:CrossTalkeR creates graphical and tabular representations in the form of a HTML/PDF report. This includes representations of the CCI networks, where edge thickness, color and node sizes represent their characteristics (Fig. 1B). CrossTalker generates PCA plots on the node feature space to describe GCI nodes. It highlights nodes with deviating PCA scores, i.e. nodes outside the 95% confidence interval assuming PCA values are normally distributed (Fig. 1C). CrossTalkeR also provides a pathway characterization (Supplementary Section S4) of top ligand/receptors per node feature to support functional interpretation of results (Fig. 1D). Moreover, it is possible to list all interactions associated within a particular ligand or receptor via a Sankey plot (Fig. 1E). The report also includes tabular representation of rankings, which can be sorted by distinct criteria or searched for particular genes and PC analysis. All results can be exported to spreadsheet formats. The package also produces an R object, which can be used for generation of further graphs, i.e. generation of Sankey plots of ligand or receptors of interest or simplification of plots by filtering ligand, receptors or cells.
Fig. 1.
(A) CrossTalkeR scheme: given a pair of scRNA-seq, CrossTalkeR creates cell–cell networks (or cell–gene networks), where edge weights are proportional to the expression of LRs driving communication between the cells. Network topology measure is used to find nodes sending signals (influencers), nodes receiving signals (listener) or both (mediators). Moreover, random-walks estimates with pagerank indicate the importance of network nodes. These measures can be computed for each phenotype individually or in the comparative analyses. (B) Comparative CCI, where node size (edge thickness) represents importance of the cell (communication between cells). (C) PCA representation of nodes from the CGI based on comparative topological measures. We highlight cell–gene pairs with deviating PCA scores. Only directions associated to positive values are shown (negative values are in opposing order). (D) KEGG pathway enrichment provide further insights of genes associated to distinct topological measures (influencer, listener, mediator and node importance) with increase/decrease in disease versus control (up/down). (E) Sankey plot listing all predicted source, receptor and receiver interactions associated with a gene of interest, i.e. TGFB1
The software is provided as an open source package. This includes a tutorial to integrate the tool with LR analysis tools such as CPDB, as well as example code to reproduce the analysis of stromal cells in human myelofibrosis (Leimkühler et al., 2020).
3 Case study: cellular crosstalk in myelofibrosis
Here, we revisit the scRNAseq analysis comparing bone marrow stromal cells in myelofibrosis (MF) to those in homeostasis (Leimkühler et al., 2020). In the comparative CCI network graph (Fig. 1B) megakaryocytes (MK) show a high number of up-regulated autocrine and intercellular interactions. This results in a high pagerank log-odds for this cell population clearly highlighting MK as the most important signaling cells in this disease influencing a plethora of bone marrow cells such as neural cells and MSCs. We also observe a significant loss of interactions involving MSCs and fibroblasts compared to homeostasis. This is in line with the restricted myofibroblastic phenotype MSCs have been described to acquire. MSCs are usually strongly interconnected, moderating the quiescence and differentiation of hematopoietic cells, but loose this capacity as myelofibrosis progresses (Leimkühler et al., 2020; Schneider et al., 2017). PCA analysis of the CGI network (Fig. 1C) highlights matrix related proteins (FN1, THBS1, COL1A1), which are secreted by MSCs, and TGFB1, which is secreted by MK, to act as both influencers and mediators in myelofibrosis. These results are in accordance with known mechanisms associated to MF, e.g. the crosstalk between MK and MSCs via TGFb signaling; and the deposition on matrix via MSC cells (Gleitz et al., 2018). Exploration of TGFB1-mediated LR-interactions in the dataset as provided by CrossTalkeR also gives into the differential intercellular signaling present in the myelofibrotic bone marrow (Fig. 1E). The strong upregulation of TGFB1-TGFBR1 between MK and MSCs is in line with other publications, showing canonical TGFB1-signaling as most dominant pro-fibrotic element (Teodorescu et al., 2020). At the same time, differential expression of co-receptors CD109 and ENG show, how TGFB1-signals are modulated in MF (Bizet et al., 2011; Schoonderwoerd et al., 2020). Thus, CrossTalkeR manages to capture the multiplicity of signals expected to be present in biological networks at any given point in time. Similarly, a downregulation of the interaction between TGFB1 and TGFRB3, an endogeneous decoy receptor preventing downstream signaling, also indicates an additional way how TGFB1-signals are increased in MF (Andres et al., 1989). Similarly, CXCL12/MSC, a pivotal mediator of hematopoietic homing, is the node with the strongest decrease in the influencer score in disease. This is in line with the findings of the CCI and previously published data showing an extensive loss of hematopoiesis supporting capacities in myelofibrotic MSCs (Gleitz et al., 2018). KEGG pathway analysis (Fig. 1D) supports these interpretations with up-regulated mediators and influencers mostly being enriched for Extra Cellular Matrix (ECM) deposition, while down-regulated influenced nodes are associated with hematopoiesis and leukocyte migration. LR analysis with CrossTalkeR also revealed that genes with increased importance are enriched for the KEGG pathway ‘platelet activation’ (Fig. 1D). This recapitulates the finding that platelet related mediators such as CXCL4 contribute to the perpetual inflammatory environment in myelofibrosis (Gleitz et al., 2020). Taken together, these examples comprehensively illustrate how CrossTalkeR can characterize changes in the diverse cellular crosstalk between conditions from scRNAseq data.
4 Conclusion and future work
CrossTalkeR is a comprehensive framework for network analysis of LR interactions, and enables comparisons between LR interactions of two biological phenotypes.CrossTalkeR provides both graphical report, tabular data and R objects, enabling both interpretable and reproducible analysis. Future work includes the extension of network-based method analysis as well as the analysis of more complex experimental designs as time series or the comparison of several biological conditions.
Funding
This project was funded by the clinical research unit CRU344 supported by the German Research Foundation (DFG) and the E: MED Consortia Fibromap funded by the German Ministry of Education and Science (BMBF).
Conflict of Interest: none declared.
Data availability statement
The data underlying this article are available in Zenodo: https://zenodo.org/record/4740646.
Supplementary Material
Contributor Information
James S Nagai, Institute for Computational Genomics, Joint Research Center for Computational Biomedicine, RWTH Aachen University Medical School, Aachen 52062, Germany.
Nils B Leimkühler, Department of Hematology and Stem Cell Transplantation, University Hospital Essen, Essen 45147, Germany; Department of Hematology, Erasmus Medical Center, Rotterdam 3015 GD, The Netherlands.
Michael T Schaub, Department of Computer Science, RWTH Aachen University, Aachen 52062, Germany.
Rebekka K Schneider, Department of Hematology, Erasmus Medical Center, Rotterdam 3015 GD, The Netherlands; Department of Cell Biology, Institute for Biomedical Engineering, Faculty of Medicine, RWTH Aachen University, Aachen 52062, Germany; Oncode Institute, Erasmus Medical Center, Rotterdam 3015 GD, The Netherlands.
Ivan G Costa, Institute for Computational Genomics, Joint Research Center for Computational Biomedicine, RWTH Aachen University Medical School, Aachen 52062, Germany.
References
- Andres J.L. et al. (1989) Membrane-anchored and soluble forms of betaglycan, a polymorphic proteoglycan that binds transforming growth factor-beta. J. Cell Biol., 109, 3137–3145. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Armingol E. et al. (2020) Deciphering cell–cell interactions and communication from gene expression. Nat. Rev. Genet., 21, 71–88. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bizet A.A. et al. (2011) The tgf-β co-receptor, cd109, promotes internalization and degradation of tgf-β receptors. Biochim. Biophys. Acta (BBA) Mol. Cell Res., 1813, 742–753. [DOI] [PubMed] [Google Scholar]
- Efremova M. et al. (2019) CellPhoneDB v2.0: Inferring cell–cell communication from combined expression of multi-subunit receptor-ligand complexes. Nature Protocols, 15, 1484–1506. [DOI] [PubMed] [Google Scholar]
- Gleitz H.F. et al. (2018) Understanding deregulated cellular and molecular dynamics in the hematopoietic stem cell niche to develop novel therapeutics for bone marrow fibrosis. J. Pathol., 245, 138–146. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gleitz H.F. et al. (2020) Increased cxcl4 expression in hematopoietic cells links inflammation and progression of bone marrow fibrosis in MPN. Blood J. Am. Soc. Hematol., 136, 2051–2064. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Leimkühler N.B. et al. (2020) Heterogeneous bone-marrow stromal progenitors drive myelofibrosis via a druggable alarmin axis. Cell Stem Cell, 28, 637–652.e8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Luecken M.D., Theis F.J. (2019) Current best practices in single-cell RNA-seq analysis: a tutorial. Mol. Syst. Biol., 15, e8746. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Schneider R.K. et al. (2017) Gli1+ mesenchymal stromal cells are a key driver of bone marrow fibrosis and an important cellular therapeutic target. Cell Stem Cell, 20, 785–800. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Schoonderwoerd M.J. et al. (2020) Endoglin: beyond the endothelium. Biomolecules, 10, 289. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Teodorescu P. et al. (2020) Transforming growth factor β-mediated micromechanics modulates disease progression in primary myelofibrosis. J. Cell. Mol. Med., 24, 11100–11110. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Data Availability Statement
The data underlying this article are available in Zenodo: https://zenodo.org/record/4740646.