Abstract
Objective: To assess the prospective association between frailty and dietary diversity on mortality. Method: This prospective cohort study used the 2005–2008 Nutrition and Health Survey in Taiwan (N = 330; age ≥ 65 years) and this was linked to the Death Registry where we used the data that was recorded up to 31 January 2020. Dietary intake information was assessed using a 24-h dietary recall and food-frequency questionnaire, which were calculated a dietary diversity score (DDS; range, 0–6) and food consumption frequency. Assessment of frailty phenotypes was based on FRAIL scale which was proposed by the International Academy on Nutrition and Aging. Results: Frail older adults had a higher risk of all-cause mortality when they were compared to those with robust physiologies (hazard ratio [HR]: 3.73, 95% confidence interval [CI]: 2.13–6.52). Frailty and a lower DDS were associated with a higher risk of mortality (joint adjusted HR: 2.30, 95% CI: 1.11–4.75) which, compared with a robust physiology and higher DDS, were associated with a lower risk of mortality. Conclusions: Frailty and a lower DDS were associated with a higher mortality. Prefrailty and frailty with a higher DDS were associated with a lower risk of mortality when compared with those with prefrailty and frailty and a lower DDS. These results suggest that eating a wide variety of foods might reduce the risk of mortality in older adults with prefrailty and frailty.
Keywords: frailty, dietary diversity, mortality, older adults, Nutrition and Health Survey in Taiwan (NAHSIT)
1. Introduction
Frailty is a medical and complicated geriatric syndrome that is characterized by multisystem decline that is associated with decreased functional reserves and increased vulnerability with aging [1]. Most studies define physical frailty by unintentional weight loss, exhaustion, low physical activity, slowness, and low grip strength [2]. The possible causes of frailty include physiological, genetic, environmental, and nutritional factors [1]. The results from a systematic review and meta-analysis reported that the prevalence of frailty and prefrailty increases with age and it appears higher prevalence in upper middle-income countries, among community-dwelling older adults [3]. With the rapid growth of the older population, worldwide, frailty requires attention because those with this condition are at a higher risk of falling, developing a disability, hospitalization, and mortality, and they require more medical services [4].
Eating a healthy diet is an important non-pharmacological strategy for not only preventing and improving the variety of non-communicable diseases but also promoting independence, quality of life, and ultimately healthy aging [5,6]. Studies have explored dietary patterns [7,8] and dietary quality and how they might affect health or mortality [9]. A brief dietary measurement method that can predict general health or mortality would be useful for public health nutrition programs and in a community-dwelling setting. The dietary diversity score (DDS) is a dietary quality measurement method, which is a simple and rapid tool that requires no devices or complicated measurements [9,10]. Older adults with more dietary diversity usually have a better dietary nutrient and energy intake profile as well as a lower mortality [9].
Our previous study showed that older adults with frailty had a lower dietary diversity, and those with both had a higher risk of developing worsening cognitive function [11]. A Taiwanese cross-sectional study demonstrated that a dietary pattern with more fruit, nuts and seeds, tea, vegetables, whole grains, shellfish, milk/yogurt/cheese, and fish was associated with a reduced prevalence of frailty [12]. It is possible that the cause of frailty might not just be due to the inadequate intake of individual nutrients or foods. Older adults with a higher dietary diversity may have a reduced mortality that is related to cognitive impairment [13], poor appetite [14], and diabetes mellitus [15]. Moreover, the results from a prospective cohort study demonstrated that a poor diet quality may increase the incidence of frailty [16], subsequently leading to a higher risk of mortality [17].
It is unknown whether dietary diversity can reduce the mortality that is associated with frailty. Therefore, the aim of this study was to investigate the prospective associations of dietary diversity and frailty severity to all-cause mortality in older adults.
2. Materials and Methods
2.1. Participants’ Data Sources
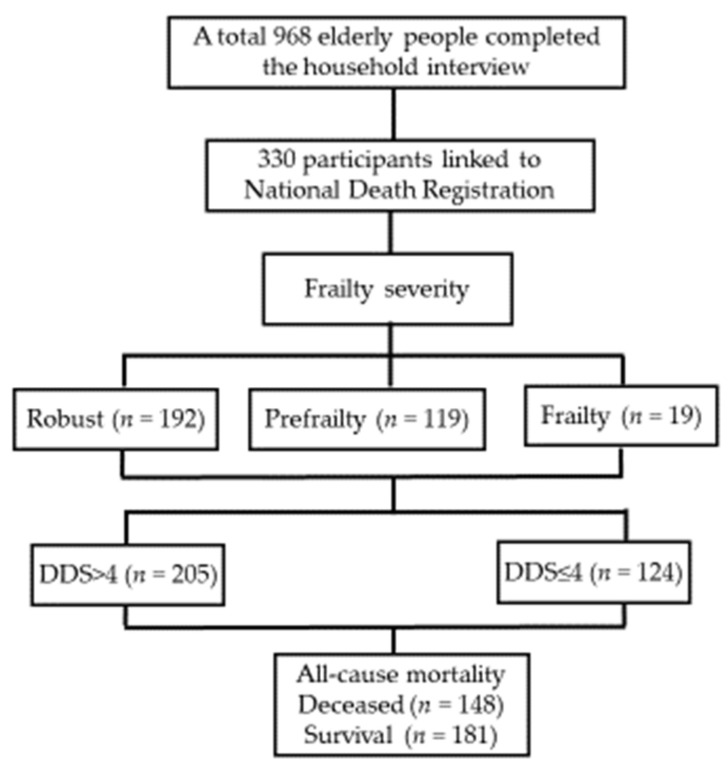
The data were obtained from the Nutrition and Health Survey in Taiwan (NAHSIT) 2005–2008; detailed information on the design and methods of this survey has been published elsewhere [18]. A total of 968 older adults (aged ≥ 65 years) completed the household interviews. There were 330 participants with a national identification (ID) number that could be used to determine their survival status (Figure 1); participants were followed up for a median of 11.7 years until 31 January 2020. All participants provided written informed consent, and this study was approved by the Research Ethics Committee of the National Health Research Institute (EC1080701-E; Miaoli, Taiwan).
Figure 1.
Flow chart for participant selection.
2.2. Frailty
Frailty and prefrailty were assessed using the FRAIL scale to assess frailty which was proposed by the International Academy on Nutrition and Aging [19]. It has been validated in previous studies [20]. The FRAIL scale includes five criteria: fatigue, resistance, ambulation, illnesses, and loss of weight [19]. Fatigue was measured by asking: “how much of the time during the past four weeks have participants felt tired?” The responses were measured from 1 (never) to 6 (all the time). Answers of 5 (most of the time) or 6 (all the time) were scored as 1; the other answers were scored as 0. Resistance was assessed by asking the participants how difficult it was to climb a flight of stairs. Responses of “Yes, limited a little or a lot” were scored as 1. Ambulation was assessed by asking the participants about the difficulty that they have walking a one block distance. Responses of “Yes, limited a lot” or “Yes, limited a little” were given a score of 1; the other responses were scored as 0. Illnesses were assessed by asking the participants if a medical professional had ever diagnosed them with any of the following illnesses: hypertension, cancer, diabetes, chronic lung disease, heart disease, asthma, arthritis, stroke, or kidney disease. A report of five or more illnesses received a score of 1. Weight loss was assessed by asking the participants if they had lost >5% of their weight in the past year. However, there was no equivalent variable that was available from the 2005–2008 NAHSIT database. Therefore, in this study, loss of weight was assessed by the participants who reported unintentional weight loss in the past month. Responses of “Yes” were given a score of 1. FRAIL scores ranged from 0 to 5, and these indicated frail (3–5), prefrail (1–2), and robust (0) conditions.
2.3. Dietary Intake Information
Dietary information was obtained by issuing the 24-h dietary recall and simplified food frequency questionnaire (FFQ). The 24-h dietary recall was used to estimate the nutrient intake, and the data were used to calculate the DDS [21]. The FFQ contains questions on the frequency of the consumption of 60 main food items per month/week/day during the month prior to taking the questionnaire. The validation of a similar simplified FFQ was previously reported. The Spearman rank correlation coefficients between the 24-h dietary recall and FFQ data ranged from 0.132 (whole grain) to 0.678 (dairy) for men, and −0.052 (whole grain) to 0.759 (dairy) for women [22].
Dietary quality was assessed using the DDS [10]. It was derived from the 24-h dietary recall of six food groups including dairy, soybeans/fish/eggs/meat, grain, fruit, vegetables, and oil/fat/nuts, in accordance with the Taiwanese Food Guides, and yielded a total score of 6 points. Achieving half of the recommended serving numbers per day of the six groups was required for a score of one [9]. The method that was used to estimate the serving numbers of the six food groups is provided in the Table S1. A DDS of >4 and ≤4 were considered high and low dietary diversity [9], respectively.
2.4. Mortality
The National Death Registry data were obtained from the Health and Welfare Data Science Center, Ministry of Health and Welfare. The 2005–2008 NAHSIT dataset of 330 participants with valid identification was linked to the National Death Registry to determine the participants’ survival status; they were followed up until 31 January 2020.
2.5. Statistical Analysis
All data analyses were performed using SAS statistical software (version 9.4; SAS Institute, Cary, NC, USA). Descriptive statistics are presented as the means ± standard deviations (SDs) and percentages for continuous and categorical variables, respectively. We evaluated the distribution of the differences within demographic and frailty severity by employing the one-way analysis of variance and the chi-square test or Fisher’s exact test. We calculated the follow-up time from the date of the interview to either the date of their death or 31 January 2020. The Cox proportional hazards regression model was used to evaluate the association between frailty severity and all-cause mortality, and the additional analyses were in accordance with the DDS. However, the sample size was limited for frailty; thus, to ensure that there was statistical power, we combined the categories of prefrailty and frailty into “frailty status” in the joint effect analysis.
For all covariates except for body mass index (BMI), less than 6% of the values were missing and all of them were imputed to the mode value (all covariates were categorical variables). The proportion of missing values was high for BMI (34.5%). To avoid the massive imputation for a non-negligible number of participants, the exclusion of those with missing data, or the introduction of a bias, we included a missing class into the models for this variable.
3. Results
The baseline characteristics of the participants are shown by their frailty severity as shown in Table 1. A total of 330 participants (180 men [54.5%] and 150 women [45.5%]) were included in this study, and the proportions of prefrailty and frailty were 36.1% and 5.8%, respectively. The distribution of frailty severity was similar to that of the original dataset (Table S2). Frail older adults often had a lower education level, poorer perceived health and sleep quality, and a higher number of drug treatments. Those with prefrailty consumed a significantly higher proportion of dietary supplements. It is noteworthy that when they were compared to those features of men, frail, older women had a higher BMI, waist circumference, and hip circumference (all p < 0.05; Tables S3 and S4).
Table 1.
Baseline characteristics by frailty severity among NAHSIT 2005–2008 older adults (n = 330).
| Frailty Severity | ||||
|---|---|---|---|---|
| Robust | Prefrailty | Frailty | P-Trend | |
| n, % | 192 (58.2) | 119 (36.1) | 19 (5.8) | |
| Gender, % | 0.763 | |||
| Men | 56.3 | 52.1 | 52.6 | |
| Women | 43.8 | 47.9 | 47.4 | |
| Age (years), % | 71.8 ± 5.47 | 73.5 ± 2.20 | 73.7 ± 5.44 | 0.008 |
| 65–69 | 42.7 | 30.3 | 36.8 | 0.234 |
| 70–74 | 25.0 | 26.9 | 15.8 | |
| 75–79 | 22.9 | 29.4 | 26.3 | |
| ≥80 | 9.38 | 13.5 | 21.1 | |
| Education, % | 0.048 | |||
| Illiterate | 19.8 | 26.9 | 36.8 | |
| Primary school | 60.9 | 45.4 | 42.1 | |
| High school and above | 19.3 | 27.7 | 21.1 | |
| Smoking, % | 35.4 | 36.2 | 36.8 | 0.985 |
| Drink alcohol, % | 42.1 | 37.1 | 36.8 | 0.659 |
| Enough money, % | 0.811 | |||
| More than enough | 8.47 | 10.3 | 5.88 | |
| Just enough | 56.6 | 50.0 | 52.9 | |
| difficult | 34.9 | 36.7 | 41.2 | |
| Perceived health status, % | <0.001 | |||
| Good | 34.2 | 24.4 | 0.00 | |
| Fair | 54.6 | 42.6 | 17.7 | |
| Poor | 11.2 | 33.0 | 82.4 | |
| Poor sleep quality, % | 7.3 | 17.2 | 26.3 | 0.006 |
| Number of diseases | 2.03 ± 1.23 | 3.86 ± 2.20 | 5.84 ± 1.68 | <0.001 |
| Number of medicines | 1.03 ± 1.03 | 2.06 ± 1.36 | 3.63 ± 1.95 | <0.001 |
| Cognitive impairment¶, % | 15.3 | 22.4 | 31.6 | 0.342 |
| Supplement use, % | 43.3 | 62.2 | 36.8 | 0.002 |
| BMI (kg/m2) | 24.4 ± 3.17 | 24.6 ± 3.52 | 26.7 ± 5.48 | 0.181 |
| Waist circumference (cm) | 85.3 ± 10.1 | 86.2 ± 10.2 | 91.5 ± 9.11 | 0.106 |
| Hip circumference (cm) | 92.6 ± 6.48 | 93.4 ± 6.68 | 96.8 ± 9.70 | 0.080 |
| DDS (score) | 4.74 ± 0.97 | 4.61 ± 1.10 | 4.32 ± 1.16 | 0.069 |
Abbreviations: NAHSIT, Nutrition and Health Survey in Taiwan; BMI, body mass index; DDS: dietary diversity score. Categorical variables are presented as a percentage, and continuous variables are presented as mean ± standard deviation (SD). General linear model regression and chi-square or Fisher’s exact tests were used for continuous and categorical variables, respectively. Cognitive impairment¶ was defined by a short, portable mental status questionnaire (SPMSQ) with ≥3 errors.
The DDS for robust, prefrail, and frail participants were 4.74 ± 0.97, 4.61 ± 1.10, and 4.32 ± 1.16, respectively, but it did not show a significant difference for frailty severity in this study (p = 0.069). In addition, the mean consumption frequencies of egg, dairy products, noodles products, breakfast cereals, vegetables, mushroom, fresh fruit, nuts and seeds, poultry/livestock blood and other parts, and coffee increase with the DDS score. Those with a lower DDS had consumed less dairy, vegetables and fruits (Table S5).
During the median follow up of 11.7 years, 148 cases of death were recorded in the National Death Registry. Table 2 shows the associations between frailty severity and the risk of all-cause mortality. The crude model shows that frail, older people had a higher risk of mortality when it was compared with that of robust individuals (hazard ratio [HR]: 1.70, 95% confidence interval [CI]: 1.31–2.20). With adjustments for age and sex (Model 1), and additional adjustments for education level, smoking status, alcohol consumption, monetary status, sleep status, cognitive function, dietary supplement use, number of drugs that were being used (Model 2), DDS (Model 3), and perceived health status (Model 4), the HR was 3.31 (1.60–6.84).
Table 2.
Association between frailty severity and risk of all-cause mortality in 2005–2008 NAHSIT older adults (n = 330).
| Frailty Severity | ||||
|---|---|---|---|---|
| Robust | Prefrailty | Frailty | p-Value | |
| All-cause mortality | ||||
| Deceased/survival (n) | 75/117 | 58/61 | 15/4 | |
| Crude | 1.00 | 1.42 (1.01–2.00) | 3.73 (2.13–6.52) | <0.001 |
| Model 1 | 1.00 | 1.30 (0.92–1.85) | 3.80 (2.15–6.71) | <0.001 |
| Model 2 | 1.00 | 1.42 (0.98–2.07) | 5.09 (2.63–9.84) | <0.001 |
| Model 3 | 1.00 | 1.42 (0.98–2.07) | 5.10 (2.63–9.87) | <0.001 |
| Model 4 | 1.00 | 1.19 (0.80–1.78) | 3.31 (1.60–6.84) | 0.004 |
The Cox proportional hazards model was estimated for hazard ratios. Model 1: adjusted for age and sex. Model 2: Model 1 plus education level, smoking status, alcohol consumption, monetary status, sleep status, cognitive function, supplement use, and the number of drug treatments that were being used. Model 3: Model 2 plus DDS. Model 4: Model 3 plus BMI.
We merged the categories of prefrailty and frailty into ‘frailty status’ in the analysis. The combined effects of frailty status and DDS (≤4 and >4) on all-cause mortality are presented in Table 3. In Model 1, older adults with a frailty status and a lower DDS had increased risks of mortality (HR: 2.01, 95% CI: 1.26–3.19) than those without frailty and a diverse diet. Further adjustments for Model 2 and Model 3 covariates did not modify these findings. Sensitivity analyses (adjusted for Model 1 covariates), excluding participants who died in the first year of the follow up, provided similar results (HR: 2.25, 95% CI: 1.41–3.59; p = 0.007 for mortality risk).
Table 3.
Association between frailty status (combined prefrailty and frailty) stratified by DDS and all-cause mortality in 2005–2008 NAHSIT older adults (n = 329).
| Frailty Severity | |||||
|---|---|---|---|---|---|
| Robust | Prefrailty/Frailty | ||||
| DDS > 4 | DDS ≤ 4 | DDS > 4 | DDS ≤ 4 | p-Value | |
| All-cause mortality | |||||
| Deceased/survival | 44/79 | 31/37 | 39/43 | 34/22 | |
| Crude | 1.00 | 1.27 (0.80–2.02) | 1.50 (0.97–2.31) | 2.25 (1.44–3.53) | 0.004 |
| Model 1 | 1.00 | 1.16 (0.72–1.85) | 1.35 (0.88–2.09) | 2.01 (1.26–3.19) | 0.024 |
| Model 2 | 1.00 | 1.05 (0.64–1.74) | 1.56 (0.99–2.47) | 1.79 (1.07–3.02) | 0.062 |
| Model 3 | 1.00 | 1.08 (0.54–2.19) | 1.30 (0.72–3.74) | 2.30 (1.11–4.75) | 0.118 |
Cox proportional hazards model was estimated for hazard ratios. Model 1: adjusted for age and sex. Model 2: adjusted additionally for education level, smoking status, alcohol consumption, monetary status, sleep status, cognitive function, supplement use, and the number of drug treatments that were being used. Model 3: Model 2 plus BMI.
4. Discussion
In this prospective cohort study, we found that frailty which was based on the FRAIL scale definition can predict mortality among older Taiwanese people, and their survival may be improved by having dietary diversity. Older adults with a frailty status and a lower DDS (DDS ≤ 4) had a higher risk of mortality when it was compared with that of robust adults or those with a frail status and a higher DDS (DDS > 4).
The systematic review and meta-analysis showed that the FRAIL scale can effectively identify frailty severity and predict disability among community-dwelling middle-aged and older people [23]. However, mortality studies on frailty and its severity are still limited [24]. We found that the FRAIL scale can predict the risk of mortality, and these findings did not change after adjustments were made for several covariates. In addition, the prevalence of prefrailty and frailty was 35.8% and 3.9%, respectively, among NAHSIT 2005–2008 participants (Table S2). Our previous study used representative data from the 2014–2016 NAHSIT of 1115 participants (aged ≥ 65 years), and found that the prevalence of prefrailty and frailty was 37.3% and 6.2%, respectively [11]. These results demonstrate that the prevalence of prefrailty and frailty is on the rise in Taiwan, and thus, it needs special attention.
We conducted the analysis by sex and found that frail, older women, but not men, had a higher BMI. These findings were consistent with a French longitudinal study of 1593 non-institutionalized older people that were aged ≥65 years. The results demonstrated that older women were approximately 2-fold more likely to be frail when they were obese, but there was no such correlation in men [25]. Moreover, a higher BMI was inversely associated with skeletal muscle mass, muscle strength, and bone mass, which may have been due to adipose tissue involvement in the complex bone-muscle interaction [26]. Therefore, older adults should maintain both a stable weight (BMI 24–26.9 kg/m2) and also skeletal muscle mass to prevent frailty and subsequent disability and mortality [27,28].
In a Japanese prospective cohort study of 666 community-dwelling older adults, a “sugar and fat” dietary pattern had a positive association with frailty [29]. The consumption of a high sugar or fat diet has been shown to increase mitochondrial dysfunction and inflammation [30]. It may increase proteolysis and reduce skeletal muscle protein synthesis, and further reduce muscle function and strength [31]. Frailty may be the result of tissue damage that is caused by a poor antioxidant ability [32]. It has been suggested that frail, older adults tend to have a poor appetite or anorexia [33]. These diets were characterized by reducing the intake of certain food groups (e.g., protein-rich food and fruit and vegetables), and these adults often have a lower DDS, which typically infers the consumption of higher amounts of sugar and fat (e.g., refined foods and fatty meats) [14,33,34]. These aging-related overall dietary quality changes may increase the risk of frailty [16,35] and mortality [14,36].
The consumption of a single food or supplement alone cannot improve frailty [37]. Studies have been shown that dietary diversity is inversely associated with frailty [11,38]. Also, dietary interventions have been shown to improve frailty severity, which when they are combined with resistance training, lead to a great improvement in frailty severity [39]. Antioxidant polysaccharides from food sources such as vegetables, fruits, cereals, beans, mushrooms, tea, milk products, and shellfish are associated with antioxidant function [40]. Moreover, in this study, older adults with a higher DDS demonstrated a higher consumption frequency of vegetables, fruit, cereals, dairy products, and mushrooms (Table S5). Nevertheless, we did not find an association between dietary diversity and frailty severity in this cross-sectional study, but the DDS showed that there is a long-term protective effect. This study had some limitations. First, frailty and dietary data were collected at a baseline, which cannot assess the effects of the changes that occur or determine the causality of frailty. Second, we used one 24-h dietary recall test to measure the DDS, which may not completely capture the long-term DDS. Therefore, the composite responses from the FFQ were used to support the validity of the DDS from one 24-h dietary recall test in this study of the older adults. We have provided the distribution of food intake frequency as determined by the DDS in the Table S5. Moreover, these estimates, even though there is less variation in them, still illustrate our point, and the effect that they have may have been underestimated. Third, only 330 people with a valid identification could be linked to mortality. The information about mortality could be biased. The strength of this study is that it was a cohort study, with comprehensive socio-demographic characteristics as well as dietary and nutritional information.
5. Conclusions
In this prospective study, older adults with frailty had an increased risk of mortality. Higher dietary diversity might reduce the risk of mortality in older adults with prefrailty and frailty. Dietary diversity has health benefits. Health promotion interventions should emphasize the importance of eating a variety of foods, especially for older adults who tend to have reduced food intakes, and especially for those with a lower DDS who consume less dairy, vegetables and fruits. Further studies are needed to examine this association and investigate whether combined dietary and exercise interventions can reduce frailty and mortality in older adults.
Acknowledgments
Data analyzed in this paper (article) were collected by the research project “Nutrition and Health Survey in Taiwan 2005–2008” sponsored by the Department of Health in Taiwan. This research project was carried out by the Center for Survey Research of the Research Center for Humanities and Social Sciences of Academia Sinica, directed by Wen-Harn Pan. The Office of Nutrition Survey, the Institute of Biomedical Sciences, Academia Sinica is responsible for data distribution. The assistance provided by the institutes and efforts made by all contributed to the survey are greatly appreciated. The views expressed herein are solely those of the authors. Funding sources had no role in the design, methods, subject recruitment, data collection, analysis, or preparation of this manuscript.
Supplementary Materials
The following supporting information can be downloaded at: https://www.mdpi.com/article/10.3390/nu14183825/s1, Table S1: The definition of a serving for six food groups; Table S2: Baseline characteristics by frailty severity among NAHSIT 2005–2008 older adults (n = 968); Table S3: Baseline characteristics by frailty severity among NAHSIT 2005–2008 older men (n = 482); Table S4: Baseline characteristics by frailty severity among NAHSIT 2005–2008 older women (n = 486); Table S5: Distributions of food intake frequency by DDS among NAHSIT 2005–2008 older adults (n = 329).
Author Contributions
W.-C.H.: Study conception and design, data analysis and interpretation, and manuscript drafting and revision. Y.-C.H., M.-S.L., J.-Y.D. and W.-H.P.: Study conception and design, and manuscript revision. H.-Y.C.: Research coordination; study conception and design; data acquisition, analysis, and interpretation; and manuscript drafting and revision. All authors have read and agreed to the published version of the manuscript.
Institutional Review Board Statement
This study was approved by the Research Ethics Committee of the National Health Research Institute (EC1080701-E; Miaoli, Taiwan) on 30 July 2019.
Informed Consent Statement
Informed consent was obtained from all subjects involved in the study.
Data Availability Statement
Restrictions apply to the availability of these data. Data was obtained from Wen-Harn Pan, and are available Wen-Harn Pan with the permission of https://www.ibms.sinica.edu.tw/wen-harn-pan/ (accessed on 31 July 2022).
Conflicts of Interest
The authors declare no conflict of interest.
Funding Statement
Funding for this study was provided by grants from the Health Promotion Administration, Ministry of Health and Welfare in Taiwan (DOH94-fs-6-4), and a grant from Academia Sinica. These funding sources had no role in the design, methods, subject recruitment, data collection, analysis, or preparation of this manuscript.
Footnotes
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations.
References
- 1.Clegg A., Young J., Iliffe S., Rikkert M.O., Rockwood K. Frailty in elderly people. Lancet. 2013;381:752–762. doi: 10.1016/S0140-6736(12)62167-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Fried L.P., Tangen C.M., Walston J., Newman A.B., Hirsch C., Gottdiener J., Seeman T., Tracy R., Kop W.J., Burke G., et al. Frailty in older adults: Evidence for a phenotype. J. Gerontol. A Biol. Sci. Med. Sci. 2001;56:M146–M156. doi: 10.1093/gerona/56.3.M146. [DOI] [PubMed] [Google Scholar]
- 3.Siriwardhana D.D., Hardoon S., Rait G., Weerasinghe M.C., Walters K.R. Prevalence of frailty and prefrailty among community-dwelling older adults in low-income and middle-income countries: A systematic review and meta-analysis. BMJ Open. 2018;8:e018195. doi: 10.1136/bmjopen-2017-018195. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Morley J.E., Vellas B., van Kan G.A., Anker S.D., Bauer J.M., Bernabei R., Cesari M., Chumlea W.C., Doehner W., Evans J., et al. Frailty consensus: A call to action. J. Am. Med. Dir. Assoc. 2013;14:392–397. doi: 10.1016/j.jamda.2013.03.022. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.World Health Organization Global Action Plan for the Prevention and Control of NCDs 2013–2020. [(accessed on 31 July 2022)]. Available online: http://www.who.int/nmh/events/ncd_action_plan/en/
- 6.Gopinath B., Russell J., Flood V.M., Burlutsky G., Mitchell P. Adherence to dietary guidelines positively affects quality of life and functional status of older adults. J. Acad. Nutr. Diet. 2014;114:220–229. doi: 10.1016/j.jand.2013.09.001. [DOI] [PubMed] [Google Scholar]
- 7.Stewart R.A.H. Primary Prevention of Cardiovascular Disease with a Mediterranean Diet Supplemented with Extra-Virgin Olive Oil or Nuts. N. Engl. J. Med. 2018;379:1388. doi: 10.1056/NEJMc1809971. [DOI] [PubMed] [Google Scholar]
- 8.Chuang S.Y., Lo Y.L., Wu S.Y., Wang P.N., Pan W.H. Dietary Patterns and Foods Associated with Cognitive Function in Taiwanese Older Adults: The Cross-sectional and Longitudinal Studies. J. Am. Med. Dir. Assoc. 2019;20:544–550.e544. doi: 10.1016/j.jamda.2018.10.017. [DOI] [PubMed] [Google Scholar]
- 9.Lee M.S., Huang Y.C., Su H.H., Lee M.Z., Wahlqvist M.L. A simple food quality index predicts mortality in elderly Taiwanese. J. Nutr. Health Aging. 2011;15:815–821. doi: 10.1007/s12603-011-0081-x. [DOI] [PubMed] [Google Scholar]
- 10.Kant A.K., Schatzkin A., Harris T.B., Ziegler R.G., Block G. Dietary diversity and subsequent mortality in the First National Health and Nutrition Examination Survey Epidemiologic Follow-up Study. Am. J. Clin. Nutr. 1993;57:434–440. doi: 10.1093/ajcn/57.3.434. [DOI] [PubMed] [Google Scholar]
- 11.Huang W.C., Huang Y.C., Lee M.S., Chang H.Y., Doong J.Y. Frailty Severity and Cognitive Impairment Associated with Dietary Diversity in Older Adults in Taiwan. Nutrients. 2021;13:418. doi: 10.3390/nu13020418. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Lo Y.L., Hsieh Y.T., Hsu L.L., Chuang S.Y., Chang H.Y., Hsu C.C., Chen C.Y., Pan W.H. Dietary Pattern Associated with Frailty: Results from Nutrition and Health Survey in Taiwan. J. Am. Geriatr. Soc. 2017;65:2009–2015. doi: 10.1111/jgs.14972. [DOI] [PubMed] [Google Scholar]
- 13.Chen R.C., Chang Y.H., Lee M.S., Wahlqvist M.L. Dietary quality may enhance survival related to cognitive impairment in Taiwanese elderly. Food Nutr. Res. 2011;55:7387. doi: 10.3402/fnr.v55i0.7387. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Huang Y.C., Wahlqvist M.L., Lee M.S. Appetite predicts mortality in free-living older adults in association with dietary diversity. A NAHSIT cohort study. Appetite. 2014;83:89–96. doi: 10.1016/j.appet.2014.08.017. [DOI] [PubMed] [Google Scholar]
- 15.Wahlqvist M.L., Xiu L., Lee M.S., Chen R.C., Chen K.J., Li D. Dietary diversity no longer offsets the mortality risk of hyperhomocysteinaemia in older adults with diabetes: A prospective cohort study. Asia Pac. J. Clin. Nutr. 2016;25:414–423. doi: 10.6133/apjcn.112015.06. [DOI] [PubMed] [Google Scholar]
- 16.Hengeveld L.M., Wijnhoven H.A.H., Olthof M.R., Brouwer I.A., Simonsick E.M., Kritchevsky S.B., Houston D.K., Newman A.B., Visser M. Prospective Associations of Diet Quality with Incident Frailty in Older Adults: The Health, Aging, and Body Composition Study. J. Am. Geriatr. Soc. 2019;67:1835–1842. doi: 10.1111/jgs.16011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Berry S.D., Ngo L., Samelson E.J., Kiel D.P. Competing risk of death: An important consideration in studies of older adults. J. Am. Geriatr. Soc. 2010;58:783–787. doi: 10.1111/j.1532-5415.2010.02767.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Tu S.H., Chen C., Hsieh Y.T., Chang H.Y., Yeh C.J., Lin Y.C., Pan W.H. Design and sample characteristics of the 2005-2008 Nutrition and Health Survey in Taiwan. Asia Pac. J. Clin. Nutr. 2011;20:225–237. [PubMed] [Google Scholar]
- 19.Abellan van Kan G., Rolland Y.M., Morley J.E., Vellas B. Frailty: Toward a clinical definition. J. Am. Med. Dir. Assoc. 2008;9:71–72. doi: 10.1016/j.jamda.2007.11.005. [DOI] [PubMed] [Google Scholar]
- 20.Woo J., Leung J., Morley J.E. Comparison of frailty indicators based on clinical phenotype and the multiple deficit approach in predicting mortality and physical limitation. J. Am. Geriatr. Soc. 2012;60:1478–1486. doi: 10.1111/j.1532-5415.2012.04074.x. [DOI] [PubMed] [Google Scholar]
- 21.Chen J.Y., Wu S.J., Tzeng M.S., Kao M.D. Nutrition and health survey in Taiwan (NAHSIT) 1993-1996: Dietary nutrient intakes assessed by 24-h recall. Nutr. Sci. J. 1999;24:11–39. [Google Scholar]
- 22.Huang Y.C., Lee M.S., Pan W.H., Wahlqvist M.L. Validation of a simplified food frequency questionnaire as used in the Nutrition and Health Survey in Taiwan (NAHSIT) for the elderly. Asia Pac. J. Clin. Nutr. 2011;20:134–140. [PubMed] [Google Scholar]
- 23.Kojima G. Quick and Simple FRAIL Scale Predicts Incident Activities of Daily Living (ADL) and Instrumental ADL (IADL) Disabilities: A Systematic Review and Meta-analysis. J. Am. Med. Dir. Assoc. 2018;19:1063–1068. doi: 10.1016/j.jamda.2018.07.019. [DOI] [PubMed] [Google Scholar]
- 24.Kojima G. Frailty Defined by FRAIL Scale as a Predictor of Mortality: A Systematic Review and Meta-analysis. J. Am. Med. Dir. Assoc. 2018;19:480–483. doi: 10.1016/j.jamda.2018.04.006. [DOI] [PubMed] [Google Scholar]
- 25.Monteil D., Walrand S., Vannier-Nitenberg C., Van Oost B., Bonnefoy M. The Relationship between Frailty, Obesity and Social Deprivation in Non-Institutionalized Elderly People. J. Nutr. Health Aging. 2020;24:821–826. doi: 10.1007/s12603-020-1465-6. [DOI] [PubMed] [Google Scholar]
- 26.Choi K.M. Sarcopenia and sarcopenic obesity. Korean J. Intern. Med. 2016;31:1054–1060. doi: 10.3904/kjim.2016.193. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Chuang S.Y., Chang H.Y., Lee M.S., Chia-Yu Chen R., Pan W.H. Skeletal muscle mass and risk of death in an elderly population. Nutr. Metab. Cardiovasc. Dis. 2014;24:784–791. doi: 10.1016/j.numecd.2013.11.010. [DOI] [PubMed] [Google Scholar]
- 28.Ishii H., Tsutsumimoto K., Doi T., Nakakubo S., Kim M., Kurita S., Shimada H. Effects of comorbid physical frailty and low muscle mass on incident disability in community-dwelling older adults: A 24-month follow-up longitudinal study. Maturitas. 2020;139:57–63. doi: 10.1016/j.maturitas.2020.04.018. [DOI] [PubMed] [Google Scholar]
- 29.Huang C.H., Martins B.A., Okada K., Matsushita E., Uno C., Satake S., Kuzuya M. A 3-year prospective cohort study of dietary patterns and frailty risk among community-dwelling older adults. Clin. Nutr. 2021;40:229–236. doi: 10.1016/j.clnu.2020.05.013. [DOI] [PubMed] [Google Scholar]
- 30.Valencia A.P., Nagaraj N., Osman D.H., Rabinovitch P.S., Marcinek D.J. Are fat and sugar just as detrimental in old age? GeroScience. 2021;43:1615–1625. doi: 10.1007/s11357-021-00390-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Liguori I., Russo G., Curcio F., Bulli G., Aran L., Della-Morte D., Gargiulo G., Testa G., Cacciatore F., Bonaduce D., et al. Oxidative stress, aging, and diseases. Clin. Interv. Aging. 2018;13:757–772. doi: 10.2147/CIA.S158513. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Soysal P., Stubbs B., Lucato P., Luchini C., Solmi M., Peluso R., Sergi G., Isik A.T., Manzato E., Maggi S., et al. Inflammation and frailty in the elderly: A systematic review and meta-analysis. Ageing Res. Rev. 2016;31:1–8. doi: 10.1016/j.arr.2016.08.006. [DOI] [PubMed] [Google Scholar]
- 33.Donini L.M., Poggiogalle E., Piredda M., Pinto A., Barbagallo M., Cucinotta D., Sergi G. Anorexia and eating patterns in the elderly. PLoS ONE. 2013;8:e63539. doi: 10.1371/journal.pone.0063539. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Vitolins M.Z., Quandt S.A., Bell R.A., Arcury T.A., Case L.D. Quality of diets consumed by older rural adults. J. Rural Health. 2002;18:49–56. doi: 10.1111/j.1748-0361.2002.tb00876.x. [DOI] [PubMed] [Google Scholar]
- 35.Tsutsumimoto K., Doi T., Makizako H., Hotta R., Nakakubo S., Makino K., Suzuki T., Shimada H. Aging-related anorexia and its association with disability and frailty. J. Cachexia Sarcopenia Muscle. 2018;9:834–843. doi: 10.1002/jcsm.12330. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Lee M.S., Huang Y.C., Wahlqvist M.L. Chewing ability in conjunction with food intake and energy status in later life affects survival in Taiwanese with the metabolic syndrome. J. Am. Geriatr. Soc. 2010;58:1072–1080. doi: 10.1111/j.1532-5415.2010.02870.x. [DOI] [PubMed] [Google Scholar]
- 37.Dominguez L.J., Barbagallo M. The relevance of nutrition for the concept of cognitive frailty. Curr. Opin. Clin. Nutr. Metab. Care. 2017;20:61–68. doi: 10.1097/MCO.0000000000000337. [DOI] [PubMed] [Google Scholar]
- 38.Motokawa K., Watanabe Y., Edahiro A., Shirobe M., Murakami M., Kera T., Kawai H., Obuchi S., Fujiwara Y., Ihara K., et al. Frailty Severity and Dietary Variety in Japanese Older Persons: A Cross-Sectional Study. J. Nutr. Health Aging. 2018;22:451–456. doi: 10.1007/s12603-018-1000-1. [DOI] [PubMed] [Google Scholar]
- 39.Hsieh T.J., Su S.C., Chen C.W., Kang Y.W., Hu M.H., Hsu L.L., Wu S.Y., Chen L., Chang H.Y., Chuang S.Y., et al. Individualized home-based exercise and nutrition interventions improve frailty in older adults: A randomized controlled trial. Int. J. Behav. Nutr. Phys. Act. 2019;16:119. doi: 10.1186/s12966-019-0855-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Li H., Ding F., Xiao L., Shi R., Wang H., Han W., Huang Z. Food-Derived Antioxidant Polysaccharides and Their Pharmacological Potential in Neurodegenerative Diseases. Nutrients. 2017;9:778. doi: 10.3390/nu9070778. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Data Availability Statement
Restrictions apply to the availability of these data. Data was obtained from Wen-Harn Pan, and are available Wen-Harn Pan with the permission of https://www.ibms.sinica.edu.tw/wen-harn-pan/ (accessed on 31 July 2022).