Abstract
Background:
Research suggests that Gardnerella vaginalis (GV) is the keystone pathogen in bacterial vaginosis (BV). Knowledge gaps exist regarding the role of GV eradication in the development of BV. This study was designed to test the hypothesis that vaginal colonization with GV could be eradicated by treatment of women without BV with amoxicillin, a drug highly active against GV. If GV is necessary for the development of BV, then eradication of GV may prevent the development of BV.
Methods:
We conducted a randomized control trial of amoxicillin 500 mg twice daily versus placebo for 7 days in women ages 18–45 without vaginitis who screened positive for vaginal colonization with GV by qPCR. Test-of-cure visit for GV was conducted at day 21.
Results:
172 women met preliminary criteria and were screened for enrollment. Ninety-seven GV positive women were randomized to receive amoxicillin versus placebo. Eradication of GV occurred in 21% of women randomized to amoxicillin versus 16% on placebo (p=0.757). In the 4 weeks between screening and test-of-cure visit, 16/92 (17%) of participants developed Nugent scores >3 with 8/92 (9%) having BV. All of these were in participants in whom GV was not eradicated (p=0.035).
Conclusions:
The study failed to show a benefit of treatment with amoxicillin to eradicate GV. No participants in whom GV was eradicated had progression to abnormal vaginal flora during the study period.
Summary
Treatment with amoxicillin vs. placebo showed no significant difference in eradicating GV. Progression to abnormal vaginal flora did not occur in any woman with eradication of GV.
Keywords: Gardnerella vaginalis, amoxicillin, bacterial vaginosis, vaginal colonization, vaginal biofilm
Introduction
Gardnerella vaginalis (GV) is a putative pathogen for the syndrome of bacterial vaginosis (BV) (1), the most common vaginal infection characterized clinically by the presence of vaginal discharge and malodor (2). Recent in-vitro work has confirmed its pathogenic potential and has led to the hypothesis that GV is the keystone pathogen in BV (3, 4). GV is likely responsible for the biofilm characteristic of BV and works in concert with endogenous anaerobes to create the vaginal discharge and odor, symptoms of BV (4, 5).
BV is a syndrome significantly associated with sexual exposure to new and multiple partners (6). Public health interest in BV centers on its conferring increased risk for adverse outcomes including preterm birth and acquisition/transmission of sexually transmitted diseases (STDs) including human immunodeficiency virus (HIV) (7–9). Current treatments for BV are suboptimal in terms of cure, and recurrence rates are high (2). Although GV is highly associated with BV, it may also be found in women who do not meet the current definition of BV, leading some to question its pathogenicity (10). Furthermore, other co-pathogens including Atopobium, Prevotella, Bacteroides, Peptostreptococcus, Mobiluncus, Sneathia, Leptotrichia, Mycoplasma, and other BV-associated bacterium may also play a role in disease manifestation (11, 12).
There is no current recommendation to treat women who are colonized with GV. The current study was designed to test the hypothesis that vaginal colonization with GV could be eradicated by treatment of women without BV with amoxicillin, an amino penicillin, with high in vitro activity against GV and some activity against other bacteria in the vagina (12). If GV is necessary for the development of BV, then eradication of GV in women who do not meet the criteria for BV should abort the future development of BV.
Materials and Methods
Women ages 18–45 without evidence of vaginitis who were willing to use condoms during the study and were not menstruating at the time of the screening visit were invited to participate. Women were excluded from participation if they had an allergy to penicillin; used antibiotics in past 14 days; were HIV-positive or otherwise immunocompromised; or pregnant or nursing. The study was conducted at two sites – University of Alabama at Birmingham (UAB) student health clinic and Wake Forest University Health Sciences (WFUHS) at the Guilford County Department of Health and Human Services STD Clinic. The study was reviewed and approved by the UAB and WFUHS Institutional Review Boards.
At the screening visit, a pelvic examination was performed and vaginal specimens were obtained to determine the presence or absence of vaginitis using wet prep microscopy, vaginal pH, whiff test, Nugent score, culture for trichomonas, and GV nucleic acid amplification test (NAAT) (13–15). The specimen for GV NAAT was collected with a swab placed into Aptima™ transport media and shipped to LabCorp (Molecular R&D 6500, 1447 York Court, Burlington, NC 27215) for quantitative GV testing. For this assay the limit of detection was 1000 copies/mL while the quantitative range was 3500–10,000,000 copies/mL(16). Participants were also administered a questionnaire regarding sexual history and vaginal hygiene behaviors.
Women who tested positive for GV by NAAT, negative for BV by Nugent (score of 0–6) and negative for trichomonas by culture were asked to return to the clinic 7–10 days after screening. At this visit they were randomized to treatment with amoxicillin 500 mg versus identical appearing placebo capsules by mouth twice daily for 7 days in a double-blinded fashion.
A test-of-cure visit was conducted at day 21 (allowable window: days 15 to 21) after randomization. At this visit subjects underwent a follow-up questionnaire, pill count to assess treatment compliance (receipt of at least 12 of the scheduled 14 doses), and a pelvic examination with sample collection.
Statistical Methods
Women were randomized 1:1 to receive amoxicillin or placebo using the online enrollment module of Advantage eClinicalSM (The Emmes Company, LLC, Rockville, MD). The study used a block randomization scheme prepared by the statisticians at the Statistical and Data Coordinating Center. The site pharmacists, participants, study personnel who performed study assessments, data entry personnel at the sites, and laboratory personnel were blinded to treatment assignment.
The study was powered to detect a decrease in GV colonization rate from 80% in the placebo arm to 44% among participants receiving amoxicillin at the two-sided 0.05 significance level with power of 0.90. It was anticipated that 40% of women without BV would have positive NAAT tests for GV (17) and assumed a 15% loss-to-follow-up rate between visits. Therefore, we required approximately 98 women randomized from 245 screened to obtain 82 evaluable participants. A chi-square test with continuity correction was used to compare GV infection rate in participants randomized to the treatment arm with those randomized to the placebo arm. The intention to treat (ITT) population was defined as the number of subjects who were randomized to the specified treatment group. For the ITT analyses, participants who had a missing GV NAAT result at the test of cure visit were classified as treatment failures. The per-protocol population (PP) included all randomized participants who met all inclusion/exclusion criteria, were evaluable, defined as having BV and GV statuses that could be determined at screening, and having a GV status that could be determined at the test of cure visit, adhered to the assigned study treatment regimen, returned to the study site for the test of cure visit within the specified window, and did not report interim vaginal sexual intercourse without a condom, pregnancy, or receipt of an oral or intravaginal antimicrobial (other than study drug). The safety population included all randomized participants who received at least one dose of study treatment.
Results
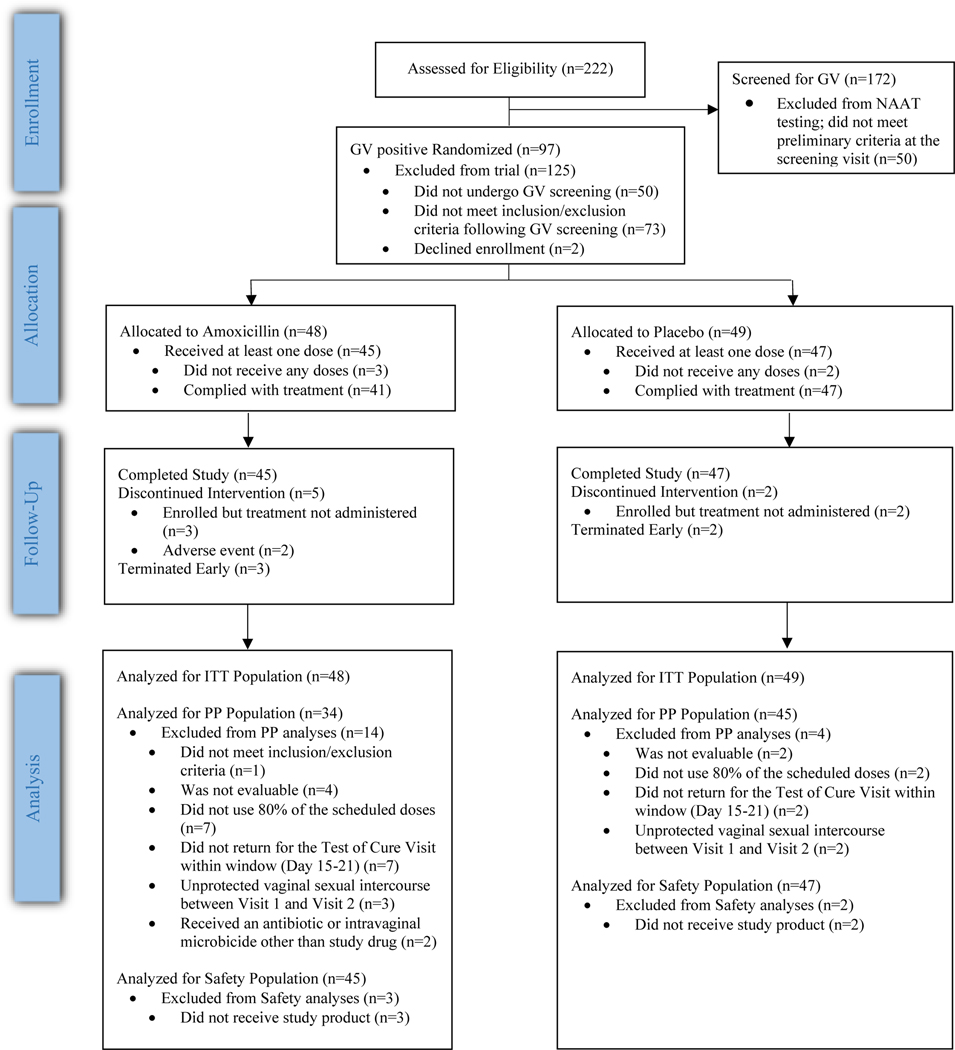
The CONSORT Flow Diagram is shown in Figure 1. Among 222 women screened for eligibility, 172 met the preliminary criteria (which included not having vaginitis) and were screened for GV. Among these women, GV was undetectable by NAAT in 57 (33%) and they were discontinued from the study. Ninety-seven women GV positive by NAAT were randomized into the treatment phase, with 81 (84%) seen at UAB and 16 (16%) seen at WFUHS. There were significant differences between the women enrolled at the WFUHS site versus UAB (Table 1). WFUHS participants were younger, more commonly reported a race(s) other than Caucasian or Hispanic ethnicity, and reported greater numbers of sexual partners in the 30 days prior to enrollment. There was no significant difference in episodes of BV in the last 6 months (9/48 versus 9/49, p=0.78) or recurrence of BV (more than one episode of BV) in the last year (5/48 versus 10/49, p=0.15) in the amoxicillin and placebo groups respectively. Overall, baseline characteristics were comparable for women in the amoxicillin and placebo groups.
Figure 1.
CONSORT Flow Diagram Evaluating the efficacy of eradicating Gardnerella vaginalis vaginal colonization with amoxicillin; a randomized, double-blind, phase 2 study
Abbreviations: GV, Gardnerella vaginalis; NAAT, nucleic acid amplification test; ITT, intention to treat; PP, per-protocol
Table 1.
Summary of Baseline Demographics, Sexual History, and Gardnerella vaginalis Screening by Enrollment Site – Intention to Treat Population
| Variable | Characteristic | Statistic | University of Alabama at Birmingham (N=81) | Wake Forest University Health Sciences (N=16) | All Subjects (N=97) |
|---|---|---|---|---|---|
| Age | Mean (Standard Deviation) | 30.7 (7.5) | 25.1 (6.1) | 29.8 (7.5) | |
| Median | 30.0 | 22.5 | 29.0 | ||
| Minimum, Maximum | 18, 45 | 20, 42 | 18, 45 | ||
| P-valued | 0.006 | -- | |||
| Ethnicity | Not Hispanic or Latino | n (%) | 78 (96) | 12 (75) | 90 (93) |
| Hispanic or Latino | n (%) | 3 (4) | 4 (25) | 7 (7) | |
| P-valuee | 0.013 | -- | |||
| Race | American Indian or Alaska Native | n (%) | 0 (0) | 1 (6) | 1 (1) |
| Asian | n (%) | 2 (2) | 0 (0) | 2 (2) | |
| Native Hawaiian or Other Pacific Islander | n (%) | 0 (0) | 0 (0) | 0 (0) | |
| Black or African American | n (%) | 53 (65) | 9 (56) | 62 (64) | |
| White | n (%) | 26 (32) | 2 (13) | 28 (29) | |
| Multi-Racial | n (%) | 0 (0) | 2 (13) | 2 (2) | |
| Unknown | n (%) | 0 (0) | 2 (13) | 2 (2) | |
| P-valuee | <0.001 | -- | |||
| Birth Control | Oral Contraceptives; Hormonal Implants; Contraceptive Patches; or NuvaRing | n (%) | 17 (21) | 11 (69) | 28 (29) |
| Hormonal Injections | n (%) | 1 (1) | 1 (6) | 2 (2) | |
| Intrauterine Device, Hormonal | n (%) | 10 (12) | 1 (6) | 11 (11) | |
| P-valuee | 0.149 | -- | |||
| Sexual History - General | Number of episodes of BV in prior 12 monthsa | n | 14 | 2 | 16 |
| Mean (Standard Deviation) | 2.3 (0.8) | 5.0 (2.8) | 2.6 (1.4) | ||
| Median | 2.0 | 5.0 | 2.0 | ||
| Minimum, Maximum | 1, 4 | 3, 7 | 1, 7 | ||
| P-valued | 0.063 | -- | |||
| Number of times the subject douched or used vaginal preparations or drying agents in the last 30 days | n | 18 | 7 | 25 | |
| Mean (Standard Deviation) | 5.1 (10.3) | 4.6 (11.2) | 5.0 (10.3) | ||
| Median | 0.0 | 0.0 | 0.0 | ||
| Minimum, Maximum | 0, 30 | 0, 30 | 0, 30 | ||
| P-valued | 0.828 | -- | |||
| Days since the subject last had sexual intercourse | n | 79 | 16 | 95 | |
| Mean (Standard Deviation) | 116.0 (272.5) | 28.9 (51.4) | 101.3 (251.2) | ||
| Median | 26.0 | 11.5 | 24.0 | ||
| Minimum, Maximum | 2, 1635 | 1, 210 | 1, 1635 | ||
| P-valued | 0.091 | -- | |||
| Days from the first day of the subject’s LMP to screening date | n | 81 | 16 | 97 | |
| Mean (Standard Deviation) | 50.3 (184.3) | 88.9 (220.9) | 56.6 (190.1) | ||
| Median | 14.0 | 20.0 | 14.0 | ||
| Minimum, Maximum | 3, 1458 | 7, 886 | 3, 1458 | ||
| P-valued | 0.042 | -- | |||
| Sexual History - In the last 30 days | Total number of partners | n | 81 | 16 | 97 |
| Mean (Standard Deviation) | 0.6 (0.6) | 0.9 (0.6) | 0.7 (0.6) | ||
| Median | 1.0 | 1.0 | 1.0 | ||
| Minimum, Maximum | 0, 2 | 0, 2 | 0, 2 | ||
| P-valued | 0.043 | -- | |||
| Number of new partners | n | 46 | 13 | 59 | |
| Mean (Standard Deviation) | 0.1 (0.3) | 0.2 (0.4) | 0.1 (0.3) | ||
| Median | 0.0 | 0.0 | 0.0 | ||
| Minimum, Maximum | 0, 1 | 0, 1 | 0, 1 | ||
| P-valued | 0.171 | -- | |||
| Sexual History - In the last 3 months | Total number of partners | n | 81 | 16 | 97 |
| Mean (Standard Deviation) | 1.0 (0.7) | 1.3 (0.7) | 1.1 (0.7) | ||
| Median | 1.0 | 1.0 | 1.0 | ||
| Minimum, Maximum | 0, 5 | 0, 3 | 0, 5 | ||
| P-valued | 0.132 | -- | |||
| Number of new partners | n | 68 | 15 | 83 | |
| Mean (Standard Deviation) | 0.2 (0.6) | 0.3 (0.6) | 0.2 (0.6) | ||
| Median | 0.0 | 0.0 | 0.0 | ||
| Minimum, Maximum | 0, 4 | 0, 2 | 0, 4 | ||
| P-valued | 0.481 | -- | |||
| Baselineb GV qPCR - Subjects with measurablec numeric results | n | 37 | 7 | 44 | |
| Mean (Standard Deviation) | 1.9 × 106 (2.8 × 106) | 1.3 × 106 (1.2 × 106) | 1.8 × 106 (2.6 × 106) | ||
| Median | 7.4 × 105 | 1.5 × 106 | 8.5 × 105 | ||
| Minimum, Maximum | 1.3 × 103, 9.6 × 106 | 5.0 × 104, 2.8 × 106 | 1.3 × 103, 9.6 × 106 | ||
| P-valued | 0.633 | -- | |||
Abbreviations: BV, bacterial vaginosis; LMP, last menstrual period; GV, Gardnerella vaginalis; qPCR, quantitative polymerase chain reaction.
Notes:
Among subjects who reported recurrent BV in the past 12 months.
Screening Visit 0.
Statistics were calculated based on the valid numeric values from the GV qPCR. GV positive subjects with results outside the lower and upper limits of quantification were excluded (52 subjects).
P-value from Wilcoxon rank-sum two-sided test.
P-value from Fisher’s exact two-sided test
Efficacy of treatment in eradicating GV as detected by PCR are shown in Table 2. There was no significant difference in the proportions of GV eradication between treatment arms in the ITT population (10/48 versus 8/49, p=0.76) or the PP population (8/34 versus 8/45, p=0.73. It is notable that 14 participants were excluded from the amoxicillin group due to protocol deviations compared to 4 from the placebo group in the PP analysis (Figure 1). Of the 97 women in the ITT population, one participant in the amoxicillin group was GV negative at screening; a laboratory reporting error initially indicated the result as positive prior to randomization. This participant remained negative at the day 21 study visit (Visit 2). By definition, this subject was included in the ITT analysis population as having GV eradicated at Visit 2. In the placebo arm the spontaneous resolution rate of GV was 16% in the ITT population. Comparison of women in the placebo group who had spontaneous resolution of GV versus those who did not showed no significant differences in demographics or behaviors with the exception of a trend towards spontaneous resolution among those women who did not report a diagnosis of BV within the prior six months (88% versus 41%, data not shown).
Table 2.
Gardnerella vaginalis Eradication at Visit 2 by Treatment Group
| n/N | % | CI | Diff. in % CI | P-value | ||
|---|---|---|---|---|---|---|
|
| ||||||
| ITT | Amoxicillin | 10/48 | 21 | (12, 34) | 5 (−11, 20) | 0.757 |
| Placebo | 8/49 | 16 | (9, 29) | |||
|
| ||||||
| All | 18/97 | 19 | (12, 27) | |||
|
| ||||||
| PP | Amoxicillin | 8/34 | 24 | (12, 40) | 6 (−12, 24) | 0.729 |
| Placebo | 8/45 | 18 | (9, 31) | |||
|
| ||||||
| All | 16/79 | 20 | (13, 30) | |||
Abbreviations: ITT, intention to treat; PP, per-protocol
In the approximately 4 weeks between the screening and test-of-cure visit, 16/92 (17%) of participants with determinate GV results developed Nugent scores in the intermediate or BV categories with 8/92 (9%) having BV (Table 3). All of these were in participants in whom GV was not eradicated (p=0.035). The majority of participants in both the amoxicillin and placebo arms in whom GV was eradicated had GV detected, but beneath the lower limit required for quantification. However, a few were as high as 2.8 million copies to even greater than the upper limit of quantification.
Table 3:
Nugent Scores at Visit 2 by Category, GV Eradication Status, and Treatment Group - Intention to Treat Population
| Nugent Score Category | Amoxicillin (N=48) | Placebo (N=49) | All Subjects (N=97) | |||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| GV Eradicated (N=10) | GV Not Eradicated (N=35) | Not Evaluablea (N=3) | GV Eradicated (N=8) | GV Not Eradicated (N=39) | Not Evaluablea (N=2) | GV Eradicated (N=18) | GV Not Eradicated (N=74) | Not Evaluablea (N=5) | ||||||||||
| n | % | n | % | n | % | n | % | n | % | n | % | n | % | n | % | n | % | |
| 0–3 (Normal) | 10 | 100 | 22 | 63 | - | - | 8 | 100 | 36 | 92 | - | - | 18 | 100 | 58 | 78 | - | - |
| 4–6 (Intermediate) | - | - | 7 | 20 | - | - | - | - | 1 | 3 | - | - | - | - | 8 | 11 | - | - |
| 7–10 (BV) | - | - | 6 | 17 | - | - | - | - | 2 | 5 | - | - | - | - | 8 | 11 | - | - |
Abbreviations: BV, bacterial vaginosis; GV, Gardnerella vaginalis
Notes:
Denominator for percentages is the number of subjects who were randomized to the specified treatment and specified GV eradication status.
Treatment group is the treatment to which the subject was randomized.
Subjects did not complete Visit 2; Nugent score was not determined.
Among participants receiving amoxicillin, eight (18%) experienced adverse events related to study product: two participants experienced gastrointestinal disorders (stomach pain and nausea) and two developed vulvovaginal candidiasis infections. Two additional, unrelated infections were reported: atypical pneumonia and BV (in the setting of interim sexual exposure reported without barrier protection). Three participants (6%) in the placebo group experienced adverse events: one had a gastrointestinal disorder (nausea) and one vulvovaginal candidiasis infection.
Discussion
The current study was designed as the first step in determining if eradication of GV colonization prevents the development of BV. Our study did not show a difference between the amoxicillin and placebo arms for eradication of GV. There could be several explanations for these results. GV infection is known to be associated with sexual activity (18). Unreported or misclassification of self-reported interim sexual activity by study subjects could lead to continued sexual transmission of GV from their sexual partner(s). Although we limited enrollment to women without evidence of vaginitis or vaginosis, the women enrolled at WFUHS were recruited from a STD Clinic and their exposures and behaviors reflected a population at higher risk for BV in general. Recent observational data suggest that sexual activity, specifically penile-vaginal sex can be associated with a GV dominant vaginal microbiota with multiple and diverse clades (19). Further, a detailed inventory of sexual exposures by anatomic site including non-coital sexual activities such toy-vaginal and digital-vaginal sex were not assessed and subsequently extravaginal reservoirs and non coital activities, which may be associated with BV (20–23) and contribute to GV colonization were not accounted for. Data support rapid and complete turnover of vaginal microbiota with exposure to antibiotics (24). Due to the limited frequency of specimen collection and short duration of follow up it is also possible that changes in vaginal flora were insufficiently captured. Additionally, our original sample size calculations may have been affected by the rate of spontaneous GV resolution in the placebo arm as well as the greater number of participants excluded in the PP evaluation in the amoxicillin versus placebo arm. Spontaneous resolution of BV has been reported to varied degrees in the literature (25–30) comparable studies to the present are lacking; more data are needed to understand the trend to GV resolution in our target population including the role of co pathogens.
This was a small study with a focused analysis on the contributions of GV as a keystone species. It is plausible that unstudied behavioral, microbiome, and metabolome factors may influence GV disposition and subsequent development of BV. Our study failed to show a benefit of treatment with amoxicillin to eradicate GV. Notably, participants in whom GV was eradicated did not progress to abnormal vaginal flora and only subjects in whom GV was not eradicated progressed to have Nugent scores in the intermediate (4–6) or BV (7–10) range. This lends evidence to the role GV plays in the development of BV and implies that if achieved, eradication of vaginal colonization of GV may contribute to the prevention of BV. Future studies may be warranted to pursue the hypotheses around which the current study was designed, including studies of improved strategies to eradicate vaginal colonization of GV and subsequent development of BV.
Acknowledgements
The authors would like to thank LabCorp for donating the quantitative PCR testing for GV, Amanda Pherson, PMP, MB (ASCP) for performing the testing, and all of the study participants.
Funding: The work was supported by the National Institute of Allergy and Infectious Diseases, part of the National Institutes of Health. Contract numbers [HHSN2722013000121; HHSN272000011].
Footnotes
Conflict of Interest: None
References
- 1.Schwebke JR, Muzny CA, Josey WE. Role of Gardnerella vaginalis in the pathogenesis of bacterial vaginosis: a conceptual model. J Infect Dis. 2014;210(3):338–43. [DOI] [PubMed] [Google Scholar]
- 2.Workowski KA, Bolan GA, Centers for Disease C, Prevention. Sexually transmitted diseases treatment guidelines, 2015. MMWR Recomm Rep. 2015;64(RR-03):1–137. [PMC free article] [PubMed] [Google Scholar]
- 3.Patterson JL, Stull-Lane A, Girerd PH, Jefferson KK. Analysis of adherence, biofilm formation and cytotoxicity suggests a greater virulence potential of Gardnerella vaginalis relative to other bacterial-vaginosis-associated anaerobes. Microbiology. 2010;156(Pt 2):392–9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Machado A, Cerca N. Influence of Biofilm Formation by Gardnerella vaginalis and Other Anaerobes on Bacterial Vaginosis. J Infect Dis. 2015;212(12):1856–61. [DOI] [PubMed] [Google Scholar]
- 5.Swidsinski A, Doerffel Y, Loening-Baucke V, et al. Gardnerella biofilm involves females and males and is transmitted sexually. Gynecol Obstet Invest. 2010;70(4):256–63. [DOI] [PubMed] [Google Scholar]
- 6.Fethers KA, Fairley CK, Hocking JS, Gurrin LC, Bradshaw CS. Sexual risk factors and bacterial vaginosis: a systematic review and meta-analysis. Clin Infect Dis. 2008;47(11):1426–35. [DOI] [PubMed] [Google Scholar]
- 7.Hillier S. The vaginal microbiol ecosystem and resistance to HIV. AIDS. 1998;14(1):17–21. [PubMed] [Google Scholar]
- 8.Hillier SL, Nugent RP, Eschenbach DA, et al. Association between bacterial vaginosis and preterm delivery of a low-birth-weight infant. The Vaginal Infections and Prematurity Study Group. N Engl J Med. 1995;333(26):1737–42. [DOI] [PubMed] [Google Scholar]
- 9.Martin HL, Richardson BA, Nyange PM, et al. Vaginal lactobacilli, microbial flora, and risk of human immunodeficiency virus type 1 and sexually transmitted disease acquisition. J Infect Dis. 1999;180(6):1863–8. [DOI] [PubMed] [Google Scholar]
- 10.Hickey R, Forney L. Reply to “Role of Gardnerella vaginalis in the pathogenesis of bacterial vaginosis-a conceptual model. J Infect Dis. 2014; 210(10): 1682–3. [DOI] [PubMed] [Google Scholar]
- 11.Onderdonk AB, Delaney ML, Fichorova RN. The Human Microbiome during Bacterial Vaginosis. Clinical microbiology reviews. 2016;29(2):223–38. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Catlin BW. Gardnerella vaginalis: characteristics, clinical considerations, and controversies. Clin Microbiol Rev. 1992;5(3):213–37. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Amsel R, Totten PA, Spiegel CA, Chen KCS, Eschenbach D, Holmes KK. Non-specific vaginitis: diagnostic and microbial and epidemiological associations. Am J Med. 1983;74:14–22. [DOI] [PubMed] [Google Scholar]
- 14.Nugent RP, Krohn MA, Hillier SL. Reliability of diagnosing bacterial vaginosis is improved by a standardized method of Gram stain interpretation. J Clin Microbiol. 1991;29(2):297–301. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Borchardt K, Smith R. An evaluation fo an InPouchTM TV culture method for diagnosing Trichomonas vaginalis infection. Genitourin Med. 1991;67:149–52. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Cartwright CP, Lembke BD, Ramachandran K, et al. Development and validation of a semiquantitative, multitarget PCR assay for diagnosis of bacterial vaginosis. J Clin Microbiol. 2012;50(7):2321–9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Schwebke JR, Flynn MS, Rivers CA. Prevalence of Gardnerella vaginalis among women with lactobacillus-predominant vaginal flora. Sex Transm Infect. 2014;90(1):61–3. [DOI] [PubMed] [Google Scholar]
- 18.Eren AM, Zozaya M, Taylor CM, Dowd SE, Martin DH, Ferris MJ. Exploring the diversity of Gardnerella vaginalis in the genitourinary tract microbiota of monogamous couples through subtle nucleotide variation. PLoS One. 2011;6(10):e26732. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Vodstrcil LA, Twin J, Garland SM, et al. The influence of sexual activity on the vaginal microbiota and Gardnerella vaginalis clade diversity in young women. PloS one. 2017;12(2):e0171856-e. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Marrazzo JM, Fiedler TL, Srinivasan S, et al. Extravaginal reservoirs of vaginal bacteria as risk factors for incident bacterial vaginosis. J Infect Dis. 2012;205(10):1580–8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Fredricks DN, Plantinga A, Srinivasan S, et al. Vaginal and Extra-Vaginal Bacterial Colonization and Risk for Incident Bacterial Vaginosis in a Population of Women Who Have Sex With Men. The Journal of Infectious Diseases. 2020. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Mitchell C, Manhart LE, Thomas KK, Agnew K, Marrazzo JM. Effect of sexual activity on vaginal colonization with hydrogen peroxide-producing lactobacilli and Gardnerella vaginalis. Sex Transm Dis. 2011;38(12):1137–44. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Brotman RM, Ravel J, Cone RA, Zenilman JM. Rapid fluctuation of the vaginal microbiota measured by Gram stain analysis. Sexually Transmitted Infections. 2010;86(4):297. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Mayer BT, Srinivasan S, Fiedler TL, Marrazzo JM, Fredricks DN, Schiffer JT. Rapid and Profound Shifts in the Vaginal Microbiota Following Antibiotic Treatment for Bacterial Vaginosis. J Infect Dis. 2015;212(5):793–802. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Hay PE, Morgan DJ, Ison CA, et al. A longitudinal study of bacterial vaginosis during pregnancy. Br J Obstet Gynaecol. 1994;101(12):1048–53. [DOI] [PubMed] [Google Scholar]
- 26.McDonald HM, O’Loughlin JA, Vigneswaran R, Jolley PT, McDonald PJ. Bacterial vaginosis in pregnancy and efficacy of short-course oral metronidazole treatment: a randomized controlled trial. Obstet Gynecol. 1994;84(3):343–8. [PubMed] [Google Scholar]
- 27.Schwebke JR. Asymptomatic bacterial vaginosis: response to therapy. Am J Obstet Gynecol. 2000;183(6):1434–9. [DOI] [PubMed] [Google Scholar]
- 28.Ugwumadu A, Reid F, Hay P, Manyonda I. Natural history of bacterial vaginosis and intermediate flora in pregnancy and effect of oral clindamycin. Obstet Gynecol. 2004;104(1):114–9. [DOI] [PubMed] [Google Scholar]
- 29.Klebanoff MA, Hauth JC, MacPherson CA, et al. Time course of the regression of asymptomatic bacterial vaginosis in pregnancy with and without treatment. Am J Obstet Gynecol. 2004;190(2):363–70. [DOI] [PubMed] [Google Scholar]
- 30.Hendler I, Andrews WW, Carey CJ, et al. The relationship between resolution of asymptomatic bacterial vaginosis and spontaneous preterm birth in fetal fibronectin-positive women. Am J Obstet Gynecol. 2007;197(5):488.e1-5. [DOI] [PubMed] [Google Scholar]