Abstract
Heart failure with preserved ejection fraction (HFpEF) encompasses nearly half of heart failure (HF) worldwide, and still remains a poor prognostic indicator. It commonly coexists in patients with vascular disease and needs to be recognized and managed appropriately to reduce morbidity and mortality. Due to the heterogeneity of HFpEF as a disease process, targeted pharmacotherapy to this date has not shown a survival benefit among this population. This article serves as a comprehensive historical review focusing on the management of HFpEF by reviewing past, present, and future randomized controlled trials that attempt to uncover a therapeutic value. With a paradigm shift in the pathophysiology of HFpEF as an inflammatory, neurohormonal, and interstitial process, a phenotypic approach has increased in popularity focusing on the treatment of HFpEF as a systemic disease. This article also addresses common comorbidities associated with HFpEF as well as current and ongoing clinical trials looking to further elucidate such links.
Keywords: HFpEF, diastolic dysfunction, preserved ejection fraction, heart failure, BNP, pathophysiology, treatment of HFpEF
Heart failure with preserved ejection fraction (HFpEF) is a clinical syndrome where the central pathogenetic abnormality is diastolic dysfunction resulting from impaired left ventricular (LV) relaxation and increase in chamber stiffness during diastole. Objectively, HFpEF is often characterized by the presence of heart failure (HF) symptoms with a left ventricular ejection fraction (LVEF) more than or equal to 50%. A clinical diagnosis of HF can be made based on the Framingham Diagnostic Criteria outlined in Table 1 . 1 Symptoms of congestion result in a sequelae of reduced LV compliance and elevated LV filling pressures that can be normal at rest and manifest only during periods of physical exertion. HFpEF is a disease process with a complex pathophysiology, and diagnostic criteria have evolved to reflect the heterogeneity of the condition. Approximately one-third of patients with HF are found to have preserved ejection systolic function. 2
Table 1. The major and minor Framingham diagnostic criteria for heart failure symptomatology.
| Major criteria | Acute pulmonary edema, cardiomegaly, hepatojugular reflex, neck vein distension, paroxysmal nocturnal dyspnea (PND)/orthopnea, pulmonary rales, S3 gallop, weight loss >4.5kg in 5 days w/ treatment |
| Minor criteria | Ankle edema, dyspnea on exertion, hepatomegaly, nocturnal cough, pleural effusion, tachycardia (hazard ratio [HR] >120) |
Two major criteria, or 1 major and 2 minor are required to diagnose heart failure (HF). 1
Several epidemiological studies have reported high morbidity and mortality rates, increased health care utilization costs, and poor quality of life (QOL) in patients with HFpEF. 3 4 5 6 Cardiovascular (CV) events are the major causes of death in this population; however, the rates of non-CV death are also much higher when compared to patients suffering from HF with reduced ejection fraction (HFrEF). 6 In contrast to HFrEF, the mechanism of HF in HFpEF is poorly understood and the diagnosis is routinely missed in community practice despite similar prevalence rates as HFrEF. This is largely due to difficulties with reliably assessing diastolic filling pressures clinically in patients presenting with symptoms of HF. At this time, we rely on a combination of clinical signs of high filling pressures (outlined above), elevated B-type natriuretic peptide (BNP) levels (although this could be unreliable in the presence of other comorbidities such as atrial fibrillation [AF] or obesity) and Doppler echocardiographic findings to make this diagnosis. 7 8 9 Over the past few years, several diagnostic clinical algorithms such as the H 2 FPEF score, European Society of Cardiology (ESC) algorithm for HFpEF, and the HFA-PEFF score have been proposed and validated in other study cohorts. 10 11 12 Most of the clinical trials that focused on pharmacological approaches to treatment of HFpEF have not shown survival benefit in patients with HFpEF. This persistent lack of evidence-driven guidelines for treatment can be explained partly by diagnostic uncertainties and heterogeneity in phenotypes among patients with HFpEF with an array of variability in driving comorbidities ( Fig. 1 ). In this review article, we aim to provide the readers with a comprehensive historical review of current literature on the treatment strategies, clinical trials both past ( Fig. 2 ) current ( Table 2 ), and future perspectives in the management of patients with HFpEF. We use a color coordinated scoring system to delineate positive, negative, and equivocal studies with specific criteria outlined in Table 3 .
Fig. 1.
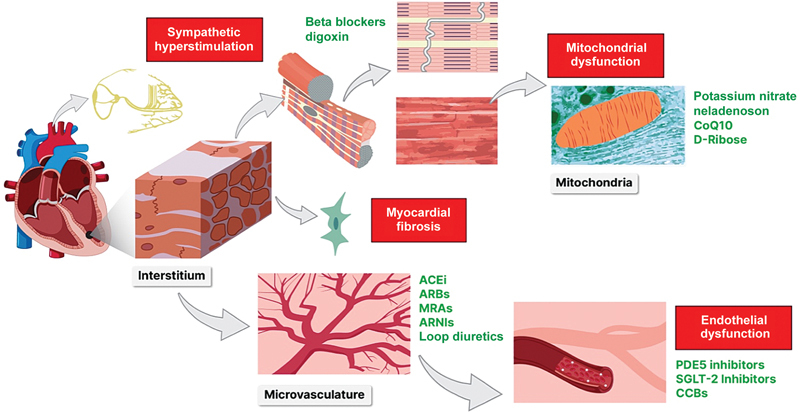
“Central Illustration”: This image illustrates the treatment rationale for drug therapies among heart failure with preserved ejection fraction (HFpEF) patients. Elevated left ventricular filling pressures (LVFP) are characteristic of diastolic dysfunction. Medications like diuretics, nitrates, phosphodiesterase 5 (PDE5) inhibitors, and Angiotensin Receptor Neprilysin Inhibitor (ARNIs) are thought to mitigate LVFPs and provide possible therapeutic benefit among HFpEF patients. Systemic inflammatory responses often driven by comorbidities leads to sympathetic hyperstimulation, myocardial fibrosis/interstitial remodeling, endothelial and mitochondrial dysfunction that can serve as major proposed pathophysiological targets. Renin-angiotensin-aldosterone system (RAAS) induced interstitial remodeling and myocardial fibrosis have both been implicated in structural changes seen within the myocardium of HFpEF patients. 75 76 77 78 79 80 81 Medications like Angiotensin-converting enzyme (ACE) inhibitors, Angiotensin receptor blockers (ARBs), mineralocorticoid receptor antagonists (MRA), ARNIs, and even loop diuretics may provide benefit to HFpEF patients by inhibiting the RAAS pathway from various points and deterring fibroblast proliferation. Endothelial dysfunction, microvascular inflammation, and arterial stiffening are also thought perpetuate imbalances in myocardial supply and demand, leading to chronic ischemia, increased angiogenesis, and fibrosis. Therapy thought to target this pathway include nitrates, PD5E inhibitors, Sodium-glucose co-transporter 2 (SGLT2) inhibitors, ARNI, and calcium channel blockers (CCB). 82 83 84 85 86 87 Sympathetic hyperstimulation is also seen among HFpEF patients. Drugs like beta blockers, and digoxin are thought to augment adrenergic activation in hopes to decreased rates of hospitalization and mortality within this population. 57 88 Mitochondrial dysfunction, decreased Adenosine Triphosphatase (ATP) production, and increased reactive oxygen species (ROS) has also been recently implicated in HFpEF perpetuating the inflammatory cascade. Targeted therapies like potassium nitrate, Coenzyme Q10 (CoQ10), Neladenoson, and D-Ribose are currently being researched to combat this phenomenon.
Fig. 2.
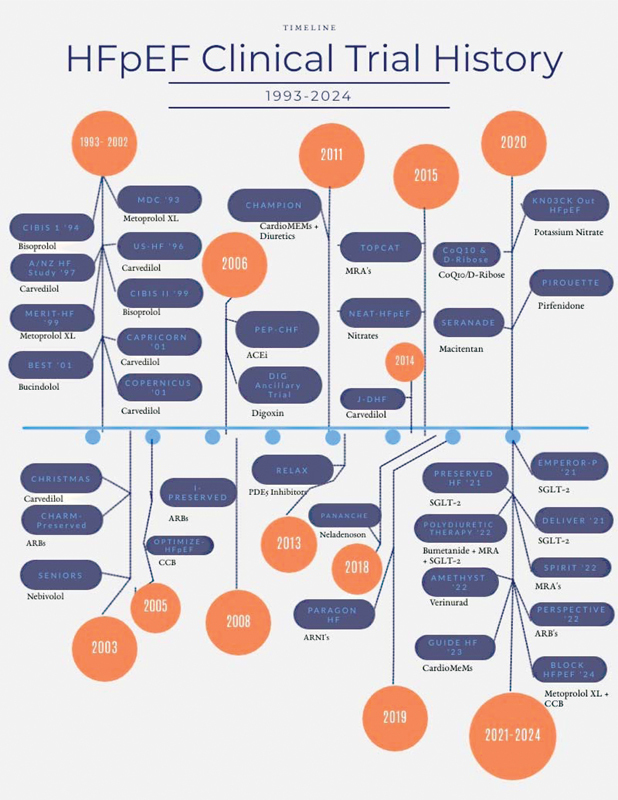
The timeline of key clinical trials and future trials that are currently being conducted among patients with HFpEF. Each trial is shown along with the medical therapy below. The clinical trial from 1993-2002 were RCTs looking at the utility of betablocker therapy among both HFpEF and HFrEF patients. The metanalysis by Cleland et al, extrapolated data from each of these trials to understand the role of beta blocker therapy among HFpEF patients. More details about this article are written within the body of the article. 29 Abbreviations: ACEi, angiotensin-converting enzyme inhibitor; CoQ10, CoEnzyme Q10; ARB, angiotensin receptor blocker; PDE5, phosphodiesterase 5 inhibitor; SGLT2, sodium glucose transporter 2 inhibitor.
Table 2. Lists of current and future clinical trials, their corresponding .gov identifiers, expected completion, therapy studied, and number of participants in each trial.
| Current ongoing clinical trials for HFpEF | ||||
|---|---|---|---|---|
| Trial name | Drug being studied | Trial identifier | Expected completion | No. of participants |
| CoQ10 & D-Ribose a | CoQ10 | NCT03133793 | 2020 | 250 |
| SERENADE a | Macitentan | NCT03153111 | 2020 | 300 |
| KN03CK Out HFpEF a | Potassium nitrate | NCT02840799 | 2020 | 76 |
| PIROUETTE a | Pirfenidone | NCT02932566 | 2020 | 129 |
| SPIRRIT | Spironolactone | NCT02901184 | 2022 | 3,200 |
| DELIVER | SGLT2 | NCT03619213 | 2021 | 4,700 |
| Dapagliflozin + pioglitazone in HFpEF | Pioglitazone plus dapagliflozin | NCT03794518 | 2021 | 648 |
| Polydiuretic therapy for HFpEF | Bumetanide 0.5 mg + eplerenone 25 mg + dapagliflozin 5 mg | NCT04697485 | 2022 | 20 |
| PERSPECTIVE | Valsartan | NCT02468232 | 2022 | 520 |
| AMETHYST | Verinurad | NCT04327024 | 2022 | 435 |
| GUIDE-HF | CardioMEMS HF system | NCT03387813 | 2023 | 3,600 |
| BLOCK HFpEF | CCB/BB | NCT04434664 | 2024 | 50 |
Abbreviations: BB, beta-blocker; CCB, calcium channel blocker; HFpEF; heart failure with preserved ejection fraction; SGLT-2, sodium-glucose cotransporter 2.
denotes expected completion in 2020, but results have yet to be published.
Table 3. Our scoring system.
| Therapeutic target scoring system | ||
|---|---|---|
| Positive | Equivocal | Negative |
| Reduction in at least ONE of the primary end-points (all-cause mortality, HF hospitalizations, etc.) | Reduction in secondary end-points, or trends to improvement in primary end-points, more favorable results with subgroup analysis | Negative study on all fronts, no reduction in primary or secondary end-points, nor a trend towards improvement in primary end-points |
Abbreviation: HF, heart failure.
Each therapeutic target mentioned in this article is given 1 of the following 3 scores based on data from landmark randomized control trials (RCTs) within each category.
Current Management of Heart Failure
According to ACCF/AHA guidelines published in 2013 and the subsequent 2017 focused update for the management of HF, pharmacotherapy for HFpEF was given primarily class II and below recommendations. This sheds light on the lack of unanimity and inconclusive clinical evidence seen for therapies found to be efficacious among HFrEF patients. The only class 1 recommendations for patients with HFpEF focus on the management of systolic and diastolic blood pressure in concordance to published clinical guidelines, and the use of diuretics for symptomatic relief. 13 New 2021 guidelines from the ESC have made the distinction of HF with mildly reduced ejection fraction (HFmrEF; EF: 41–49%) creating more uncertainty when trying to find any true therapeutic options for patients with HFpEF. 14 This will become more evident throughout the article. Treatments that have shown to have a mortality benefit in patients with HFrEF have been equivocal in patients with diastolic dysfunction. In 2013, class 2a recommendations state that the use of beta-blocking agents, angiotensin converting enzyme inhibitor (ACEi), angiotensin-receptor blockers (ARBs), is reasonable in hypertensive patients who have HFpEF but this recommendation is primarily to stabilize systolic and diastolic blood pressure mentioned in the class 1 recommendation stated above. 13 In the subsequent 2017 focused update, Aldactone was given a class 2b recommendation for HFpEF patients with elevated BNP levels as well as recent HF hospitalization. 13 However, in the updated 2021 ESC guidelines, class II indications for the drugs mentioned above are only are given to patients with HFmrEF and not HFpEF, shedding light on the further ambiguity in therapeutic treatment options for patients with HFpEF. 14 In summary, no clear mortality benefit has been shown by medical management for patients with HFpEF, and the current recommendations are aimed to stabilize systolic/diastolic blood pressure, and diuretics for symptomatic patients.
Medical Therapy and Corresponding Key Randomized Controlled Trials
Beta-Blockers
Consensus: Equivocal
Evidence: Beta-Blockers for Heart Failure with Reduced, Mid-Range, and Preserved Ejection Fraction: An Individual Patient-Level Analysis of Double-Blind Randomized Trials; Effects of Carvedilol on Heart Failure with Preserved Ejection Fraction: The Japanese Diastolic Heart Failure Study (J-DHF)
This study was a patient level meta-analysis of 11 randomized controlled trials (RCTs) ( Table 4 ) across multiple countries to assess the efficacy of beta-blocker use among patients with HFpEF. The inclusion criteria for each RCT included in this analysis were placebo controlled trials with more than 600 patients, and a mean planned follow-up of more than 6 months with explicit documentation of mortality as a primary outcome. 15 The trials included are outlined in the table below. Patients from each trial included those with baseline LVEF, and electrocardiogram showing sinus rhythm or AF/AFlutter (all 3 subgroups analyzed separately). 16 Those with paced rhythm or heart block found in these trials were excluded.
Table 4. The meta-analysis by Cleland et al extrapolated data from each of these trials in green to understand the role of beta-blocker therapy among HFpEF patients.
| Randomized controlled trials included in meta-analysis | |
|---|---|
| Inclusion criteria: >600 patients, >6 months follow-up, mortality as a primary outcome | |
| 1. | The Australia/New Zealand Heart Failure Study (ANZ) 65 |
| 2. | The Beta-Blocker Evaluation Survival Trial (BEST) 15 |
| 3. | Carvedilol Post-Infarct Survival Control in LV Dysfunction Study (CAPRICORN) 66 |
| 4. | Carvedilol Hibernating Reversible Ischemia Trial: Marker of Success Study (CHRISTMAS) 67 |
| 5. | Cardiac Insufficiency Bisoprolol Study (CIBIS I) 68 |
| 6. | The Cardiac Insufficiency Bisoprolol Study II (CIBIS-II) 69 |
| 7. | Carvedilol Prospective Randomized Cumulative Survival Study (COPERNICUS) 70 |
| 8. | Metoprolol in Idiopathic Dilated Cardiomyopathy Study (MDC) 71 |
| 9. | Metoprolol CR/XL Randomized Intervention Trial in Congestive Heart Failure (MERIT-HF) 72 |
| 10. | Study of the Effects of Nebivolol Intervention on Outcomes and Rehospitalization in Seniors with Heart Failure (SENIORS) 73 |
| 11. | U.S. Carvedilol Heart Failure Program (US-HF) 74 |
Abbreviation: HFpEF, heart failure with preserved ejection fraction.
A total of 18,637 patients were studied (14,262 in sinus rhythm), with primary outcomes including all-cause mortality, and CV death over and 1.3 years average follow-up. 16 This study stratified their results into three subsections based on LVEF: less than 40%, 41 to 49%, more than or equal to 50%. In patients with LVEF less than 40%, beta-blocker therapy statistically significantly reduced the both the all-cause mortality and CV death especially in patients with an EF of 35 to 39% (hazard ratio [HR]: 0.67, 95% confidence interval [CI]: 0.50–0.90). 16 In patients with LVEF 40 to 49%, all-cause mortality was ostensibly but not statistically significant (HR: 0.59, 95% CI: 0.34–1.03), while death due to CV process was substantially reduced (HR: 0.48, 95% CI: 0.24–0.97). 16 On the other hand, patients with LVEF of more than or equal to 50%, which defines HFpEF, there was no reduction in both all-cause mortality and CV death among this subgroup (HR: 1.79, 95% CI: 0.78–4.10), (HR: 1.77, 95% CI: 0.61–5.14), respectively. 16 This meta-analysis shows a mortality benefit among those with reduced LV function, but shows no efficacy among patients with preserved EF defined by an LVEF greater than 50%.
A decade later the J-DHF, a multicenter, prospective, open, blind-end-point RCT was published studying the effects of carvedilol in HFpEF patients. 17 The primary composite outcome studied was CV death and unplanned HF hospitalizations, with a mean follow-up of 3.2 years. The primary end-point was seen among 29 patients in the treatment group, and 34 in the control (HR: 0.902, 95% CI: 0.546–1.488, p = 0.6854). 17 Interestingly, when stratifying based on dosage of carvedilol, patients taking more than 7.5mg/day when compared to control versus patients taking less than 7.5mg/day versus control showed a statistically significant decrease in compost outcomes (HR: 0.539, 95% CI: 0.303–0.959; p = 0.0356). 17 This study suggests that although carvedilol did not amount a survival benefit, this study suggests higher tolerable doses may be beneficial. Hopefully further studies like the BLOCK-HFpEF (NCT04434664) may yield more favorable results.
Calcium Channel Blockers
Consensus: Negative
Evidence: Calcium Channel Blockers and Outcomes in Older Patients with HFpEF: OMPTIMIZE-HF
This retrospective outcome-blinded, propensity-matched balanced cohort study looked at efficacy of calcium channel blocker (CCB) among 810 matched pairs or 1,620 HFpEF patients. The Organized Program to Initiate Lifesaving Treatment in Hospitalized Patients with Heart Failure (OPTIMIZE-HF) is a national registry of 48,612 HF across 259 hospitals and 48 different states. 18 Of these, 10,570 patients were more than or equal to 65 years, and had HFpEF more than or equal to 40%. Among the 10,570 patients, 7514 had no prior CCB use, with 815 (11%) newly discharged with CCB. The primary composite outcomes studied were all-cause mortality or HF hospitalizations, which occurred in 82 and 81% of patients taking or not taking CCBs, respectively, HR for CCBs: all-cause mortality 1.05 (0.94–1.18), and HF hospitalizations 1.05 (0.91–1.21). 18 Similar relationships were seen when patients were stratified by amlodipine versus nonamlodipine CCBs 1.03 (0.95–1.12) and 1.02 (0.94–1.11), respectively. 18 This study showed, that among older patients with HFpEF, new discharge prescription of CCB did not provide an all-cause mortality benefit nor reduce the frequency of HF hospitalizations. No prospective RCT with a large enough patient population has been studied, and it would be interesting to see if this would yield different results. In addition, this study did not find any major differences in composite outcomes among patients taking dihydropyridine versus nondihydropyridine CCBs. In 2001, a small study found increased exercise tolerance and improved LV diastolic function among 15 elderly patients with HFpEF taking verapamil for 3 months. 19 Given such a small sample size, larger RCTs looking specifically at non-dihydropyridine CCBs among HFpEF patients need to be done to confer such association. In addition, both trials studied primarily older HFpEF patients and included those with HFmrEF (EF≥ 40%). It would be interesting to see if there are any benefit of CCBs among HFpEF patients in varying age groups and higher EF cutoffs.
Angiotensin II Receptor Blocker
Consensus: Equivocal
Evidence: Effects of Candesartan in Patients with Chronic Heart Failure and Preserved Left-Ventricular Ejection Fraction: The CHARM-Preserved Trial and Irbesartan in Patients with Heart Failure and Preserved Ejection Fraction: I-PRESERVE
The CHARM-Preserved was a RCT enrolling 3,023 patients with New York Heart Association (NYHA) class II to IV and LVEF more than 40%, studying the mortality and rate of hospitalization among individuals ARBs. 20 The treatment arm consisted of 1,514 patients with a target dose of 32 mg of candesartan daily, with a mean follow-up of 36.6 months. 20 The primary outcomes studied were rate of CV mortality, and incidence of hospitalization due to CV event. Results were just shy of statistical significance in CV mortality and hospitalization between both groups 0.86 (95% CI: 0.74–1.0, p = 0.051). This can be partly explained by the notable decreases in first chronic heart failure (CHF) hospitalization among the candesartan treatment group (230 vs. 279, p = 0.017). 20 This trial showed some utility of ARBs in preventing CV hospitalization in patients with HF and LVEF more than 40% without a significant mortality benefit.
Similar to the CHARM-Preserved, the I-PRESERVE was a RCT that enrolled 4,128 individuals in NYHA class II to IV, LVEF more than 45% and mean EF of 59%, to study all-cause mortality and CV mortality among patients taking 300 mg of Irbesartan daily with a mean follow-up time of 49.5 months. 21 The primary outcomes studied included CV mortality from HF, myocardial infarction, unstable angina, arrhythmia, or stroke. 21 Secondary outcomes investigated included specific death or hospitalization from HF, and QOL. 21 This study found no significant difference in either all-cause mortality (HR: 1.00; 95% CI, 0.88 to 1.14; p = 0.98), or rates of CV-related hospitalization (HR: 0.95; 95% CI: 0.85–1.08; p = 0.44) and no difference in the secondary outcomes stated above. 21 Both trials studied the efficacy of ARB therapy among patients with relatively preserved EFs. Although the CHARM-Preserved showed a notable decreased in hospitalizations for HF, both studies failed to prove a mortality benefit among HFpEF patients on ARB therapy. In addition, the CHARM Preserved included those with EF more than 40%. We define HFpEF with an EF more than 50% which may confound partially favorable results noted in this study. Hopefully more promising results may be seen with valsartan, in the PERSPECTIVE trial NCT02468232, currently recruiting patients with an estimated date of completion in 2022.
ACE Inhibitors
Consensus: Positive
Evidence: Perindopril in Elderly People with Chronic Heart Failure (PEP-CHF) Trial
This study was a double-blind, multicenter, global, RCT that studied the effects of perindopril and placebo in patients with HFpEF. Eight-hundred fifty patients with an average age of 70 years, treated with diuretics for diastolic disfunction, and CV hospitalization within 6 months of enrollment were included. 22 Some specific exclusion criteria were hemodynamically significant valvular disease, cerebrovascular event within the last month, history of ACEi, ARB, or potassium (K) sparing diuretic intolerance within 1 week of enrollment, or systolic blood pressure (SBP) less than 100 mm Hg, serum creatinine (Cr) more than 200 µmol/L and K more than 5.4. 22 The primary end-points studied were all cause mortality or unplanned HF-related hospitalization. Secondary end-points included CV mortality, increase in diuretic treatment, length of hospitalization for HF symptoms, and change in NYHA class within a year from baseline. 22
The 850 patients included in this study were given either perindopril or a placebo with a minimum follow up for 1 year and median of 2.1 years. By 1 year, no statistically significant reduction in all-cause mortality was seen (HR: 0.692: 95% CI: 0.474–1.010; p = 0.055) but there was a statistically notable reduction in the hospitalization rate for HF (HR: 0.628: 95% CI: 0.408–0.966; p = 0.033), NYHA functional class ( p < 0.030), and 6-min corridor walk distance ( p = 0.011) among the treatment arm. 22 This study concluded that there is quite a bit of uncertainty around ACEi and long-term morbidity/mortality benefits, but possible utility in increasing exercise and functional capacity, while decreasing rates of hospitalizations among HFpEF patients.
Mineralocorticoid Receptor Antagonists
Consensus : Equivocal
Evidence: Treatment of Preserved Cardiac Function Heart Failure with an Aldosterone Antagonist Trial
Treatment of Preserved Cardiac Function Heart Failure with an Aldosterone Antagonist (TOPCAT) trial published in 2014 outlined the utility of aldosterone antagonist in the medical management of patients with HFpEF. This randomized, double-blind trial took 3,445 patients with symptomatic HF and LV functions more than or equal to 45% and assigned individuals to either spironolactone treatment and placebo group. 23 Major exclusion criteria included uncontrolled hypertension, K more than or equal to 5.0 mmol/L, Cr more than or equal to 2.5 mg/dL, or estimated glomerular filtration rate (GFR) less than 30 mL/min. 9 Patients were followed for 3.3 years with primary outcomes including cardiac arrest, hospitalization for HF, or death from CV disease. 23
These primary outcomes among participants in both arms of the study occurred at comparable rates with 18.6 and 20.4% among the treatment and placebo group, respectively, with a HR of 0.89; 95% CI: 0.77–1.04; p = 0.14. 24 Total number of deaths as well as hospitalizations followed similar rates. Despite the lack of significant primary outcomes between both groups, there was as statistically significant reduction in hospitalization for HF among the spironolactone treatment group (HR: 0.83; 95% CI: 0.69–0.99, p = 0.04). 23 In addition lower EF cutoffs were used with EF more than or equal to 45%, including those with HFmrEF, as opposed to only patients with EF more than 50%.
But there is more to the TOPCAT trial than meets the eye. One year after it was published, a post hoc analysis showed roughly a fourfold difference in composite event rates when stratified based on region. Regional differences between the Americas and Russia/Georgia showed larger differences in placebo groups geographically. 24 In the Americas primary composite outcome (Americas: 0.82 [95% CI: 0.69–0.98]; Russia/Georgia: 1.10 [95% CI: 0.79–1.51]; p = 0.12), CV death (Americas: 0.74 [95% CI: 0.57–0.97]; Russia/Georgia: 1.31 [95% CI: 0.91–1.90]; p = 0.012 for interaction), and HF hospitalizations (Americas: 0.82 [95% CI: 0.67–0.99]; Russia/Georgia: 0.76 [95% CI: 0.44–1.32]; p = 0.81 for interaction) were significantly reduced in the spironolactone treatment arm. 24 Irrespective of treatment arm, patients from Russia/Georgia were younger, had lower rates of AF, but had higher incidences of myocardial infarctions/HF hospitalizations. Similarly, patients enrolled from the Americas had higher incidences of hyperkalemia, and doubling of Cr when compared to patients from Russia/Georgia. 24
Despite the equivocal data from the initial trial, regional differences have complicated interpretations of the TOPCAT trial. Baseline differences show that within the trial itself diagnostic criteria were not standardized. However, although higher rates of hyperkalemia and elevated Cr were seen among the Americas, there were significant reduction in hospitalizations and CV death among this population. This perpetuated the update of Aldactone in the 2017 AHA guidelines as a class IIb recommendation among HFpEF patients whose GFR was more than 30 mL/min, Cr less than 2.5 mg/dL, and K less than 5 mEq/L to reduce hospitalizations. 13
Nitrates
Consensus: Negative
Evidence: Nitrate's Effect on Activity Tolerance in Heart Failure with Preserved Ejection Fraction (NEAT-HFpEF) Trial
This trial was a multicenter, placebo-controlled, double-blind, crossover study encompassing 110 patients with HFpEF randomly assigned a 6-week isosorbide mononitrate (ISMN) in a dose escalated fashion (30mg → 60mg → 120mg 1x daily) or placebo with cross over at 6 weeks. 25 Patients included in this study were more than or equal to 50 years old, LVEF more than or equal to 50%, and objective evidence of HF (criteria outlined in the study) within the last year prior to enrollment. Patients excluded from the trial were on the basis of SBP less than 110 or more than 180, and previous adverse reaction to long-term nitrate use or phosphodiesterase type 5 inhibitor therapy. 25
The primary outcome studied was daily activity level, calculated by the average daily accelerometer units achieved during the high does (120 mg) phase of the trial. 25 Secondary outcomes studied included daily hour of activity during the high dose phase, daily accelerometer units during all three dose regimens, 6-minute walk (meters), and N-terminal (NT)-proBNP levels. 25 Between the high dose ISMN 120 mg treatment arm and the placebo, there was statistically insignificant tendency toward lower daily activity (−381 accelerometer units; 95% CI, −780 to 17; p = 0.06), but a substantial decrease in the hours of daily activity among the ISMN arm (−0.30 hours; 95% CI, −0.55 to −0.05; p = 0.02). 25 All dose regimens within the treatment arm showed a lower and incremental decrease in activity level than its placebo counterpart (−439 accelerometer units; 95% CI, −792 to −86; p = 0.02). 25 There were no statistically significant difference between the treatment and placebo in the secondary outcomes outlined above. Overall, this study showed that nitrate therapy for HFpEF patients decreases to some extent their functional capacity and is generally not recommended as medical therapy. Hopefully further studies with potassium nitrate like the KNO3CK Out-HFpEF (NCT02840799) may yield more favorable results.
Diuretics
Consensus: Positive
Evidence: Telemonitoring of Fluid Status in Heart Failure: CHAMPION Trial
The CHAMPION trial was a prospective, multicenter, single-blind clinical trial composed of 550 patients within NYHA class III regardless of LVEF, and history of HF exacerbation that required hospitalization in the last year. In the Champion trial the CardioMEMS HF System (Micro-Electro-Mechanical HF System, Abbott Medical, Inc., Abbott Park, IL) was implemented to provide pulmonary artery (PA) pressures to help dictate management of HF primarily in the form of diuresis. 26 Depending of the PA pressure, doses of diuretics were either uptitrated or reduced to ameliorate pulmonary vascular congestion before symptoms can manifest. The goal of the trial to was see if strict control of PA pressures would ultimately prevent or reduce the number of HF exacerbations.
In the CHAMPION trial, a subdivision of 119 patients who fit the initial inclusion criteria stated above also had HFpEF. Stratification of the data showed a 15% decrease in hospitalization among the treatment group (incidence rate ratio, 0.54; 95% CI: 0.38–0.70; p <0.0001). 27 28 These rates were observed to be statistically significantly lower than their HFrEF counterparts. This study showed that there is some utility in hemodynamically guided diuretic therapy in HFpEF patients as it decreases the frequency of decompensation and subsequent hospitalization in comparison of standard management. Similar to the TOPCAT trial for aldosterone antagonists, the CHAMPION trial shows the utility of diuretics therapy to decrease incidence of hospitalization without providing a direct mortality benefit. Hopefully the much larger GUIDE HF trial (NCT03387813) of 3600 have more promise with an estimated completion date 2023.
Sodium-Glucose Cotransporter 2 Inhibitors
Consensus: Positive
Evidence: Dapagliflozin in PRESERVED Ejection Fraction Heart Failure (PRESERVED-HF), Empagliflozin Outcome Trial in Patients with Chronic Heart Failure with Preserved Ejection Fraction (EMPEROR-Preserved), Dapagliflozin Evaluation to improve the LIVEs of Patients with Preserved Ejection Fraction Heart Failure (DELIVER)
Sodium-glucose cotransporter 2 (SGLT2) inhibitors (dapagliflozin and empagliflozin) are one of the latest medications that have shown a mortality benefit among HFrEF patients independent of the presence of comorbid diabetes, as seen in the DAPA-HF trials and EMPEROR-Reduced trials published in 2019 and 2020, respectively. 29 30 With these studies published, showing promising outcomes among HFrEF patients, 3 PRESERVED-HF, EMPEROR-Preserved, and DELIVER are currently being conducted to see if empagliflozin and dapagliflozin have similar benefits in HFpEF patients.
Initial data published from the EMPEROR-P trial on August 28,2021 shows a reduction in combined risk of CV death or hospitalization; however, benefits are driven primarily by reduction in HF hospitalizations and not mortality. 31 Results showed CV death or HF hospitalizations 13.8 versus 17.1% (HR: 0.79, 95% CI: 0.69–0.90, p < 0.001), CV death 7.3 versus 8.2% (HR: 0.91, 95% CI: 0.79–1.09) and HF hospitalizations: 8.6 versus 11.8% (HR: 0.71, 95% CI: 0.0–0.83). 31 Initial data for dapagliflozin have yet to be published; however, recent evidence shows promise. 32 The specific cardioprotective role of SGLT2s has not been clearly established but role in reducing preload, blood pressure, and afterload through natriuresis/osmotic diuresis may decrease afterload and arterial stiffness to promote subendocardial blood flow. 33
Phophodiesterase-5 inhibitors
Consensus: Negative
Evidence: Effect of Phosphodiesterase-5 Inhibition on Exercise Capacity and Clinical Status in Heart Failure with Preserved Ejection Fraction (RELAX) Trial
This study was another multicenter, double-blind, parallel-grouped, RCT of 216 stable patients with HFpEF defined by LVEF more than or equal to 50%, elevated NT-proBNP or elevated invasively measured filling pressures, and reduced exercise capacity. 34 Inclusion criteria consisted of NYHA II to IV, stable medical therapy for 30 days, and at least one of the following within the last year: hospitalization for CHF exacerbation, acute treatment for CHF with IV loop diuretics or hemofiltration, mean pulmonary capillary wedge pressure (PCWP) more than 15 mm Hg, left ventricular end-diastolic pressure more than 18 mm Hg, and NT-proBNP more than or equal to 400 pg/mL (or 200 w/ PCWP > 20 @ rest and >25 with exercise). 34
Patients were followed for 24 weeks with primary end-point being peak oxygen consumption (VO 2 ) after 24 weeks of therapy with sildenafil administered at 20 mg thrice daily (TID) for 12 weeks and 60 mg TID for the remaining 12 weeks. Secondary outcomes studied included 6-minute walk distance, clinical status score (higher number = better status), CV hospitalization, and changes in QOL for patients that were not hospitalized. 34 After 24 weeks, the sildenafil treatment arm showed no significant change in the peak VO 2 from baseline when compared to the placebo group −0.20 (95% CI: −0.70 to 1.00) versus −0.20 (95% CI: −1.70 to 1.11; p = 0.90). 34 This study also showed no clinically or statistically significant difference in clinical rank score, change in 6 minute walk distance, hospitalization secondary to CV process, or change in quality life among both groups. Based on this trial, the use of phosphodiesterase 5-inhibotors in HFpEF does not show any potential benefit and therefore generally not recommended.
Angiotensin Receptor-Neprilysin Inhibitor
Consensus: Equivocal
Evidence: Angiotensin–Neprilysin Inhibition in Heart Failure with Preserved Ejection Fraction PARAGON-HF
This has been the most recent study published on September 1, 2019 trying to shed light on efficacy of Angiotensin receptor-neprilysin inhibitor (ARNI) therapy among patients with HFpEF. This was a major randomized, double-blind, active-comparator trial consisting of 4,822 patients with NYHA class II to IV, EF more than or equal to 45%, elevated BNP, and echographic confirmation of structural heart disease, with an intention to treat analysis. 35 The treatment arm was given twice daily sacubitril–valsartan, with target dose for each half being 97 and 103 mg, respectively, and the control given twice daily valsartan with a target dose of 160mg. 35 These patients were followed for 35 months with total hospitalization for HF and CV death as primary outcomes studied. Predetermined secondary outcomes evaluated were changes in NYHA class, decline in renal function, and changes in Kansas City Cardiomyopathy Questionnaire (KCCQ) which the study describes as higher scores indicating fewer symptoms and physical limitations. 35
This study found similar incidences of CV death with 8.5% among the ARNI group and 8.9% with the ARB control group (HR: 0.95; 95% CI: 0.79–1.16). 35 Although not statistically significant, there was a general trend toward decreased number of total CV hospitalization among the sacubitril–valsartan group: 690 compared to the valsartan arm: 797 (rate ratio [RR]: 0.85; 95% CI: 0.72–1.00). 35 In terms of secondary outcomes, NYHA class improvement was seen in 15 versus 12.6% of patients, decline in renal function 1.4 versus 2.7% in the ARNI and ARB group, respectively, group (OR: 1.45; 95% CI: 1.13–1.86); HR: 0.50, 95% CI: 0.33–0.77). 35 There was no significant change in the KCCQ score between both arms of the study. In summary, this was a landmark study assessing the effects of ARNI therapy among HFpEF patients. This study proved no CV mortality or hospitalization benefit in patients taking daily sacubitril–valsartan versus valsartan alone. In addition, lower EF cutoffs were used with EF more than or equal to 45%, including those with HFmrEF, as opposed to only patients with EF more than 50%.
Digoxin
Consensus: Negative
Evidence: Effects of Digoxin on Morbidity and Mortality in Diastolic Heart Failure: The Ancillary Digitalis Investigation Group (DIG) Trial
The main DIG trial was a RCT looking at the effects of digoxin on the HF mortality and hospitalization in 6,800 patients with LVEF less than or equal to 45%. 36 The DIG ancillary trial was conducted in a parallel fashion composed of 988 patients with LVEF more than 45% assessing the same primary outcomes stated above. The mean follow-up was 37 months in which the treatment arm received a median daily dose of 0.25 mg of Lanoxin. 36 Between both groups, there was no difference between the primary end-points of the study. Although there was a general trend toward decreased HF hospitalization (HR: 0.79; 95% CI: 0.59–1.04; p = 0.094), both outcomes including HF mortality were not statically significant between the study groups (HR: 0.82; 95% CI: 0.63–1.07; p = 0.136). 36 Interestingly, use of digoxin showed a general increase in the rate of hospitalization among patients with unstable angina (HR: 1.37; 95% CI: 0.99–1.91; p = 0.061). 36 The DIG ancillary trial proved no mortality or reduction in hospitalization benefit among HFpEF patients on digoxin therapy. One criticism, as mentioned in previous trials above, lower EF cutoffs were used with EF more than or equal to 45% as opposed to only patients with EF more than 50%.
Alternative Treatment Strategies
Cardiac Rehabilitation
Cardiac Rehabilitation (CR) has been shown to increase exercise capacity, increase vagal tone while reducing sympathetic stimulation known to be detrimental among HFpEF patients. 37 As discussed with beta-blockers and digoxin, sympathetic hyperstimulation reduces LV filling time while making the myocardium more prone to tachyarrhythmias. Suppression of adrenergic impulse through CR may provide a therapeutic benefit among HFpEF patients. 37
Consensus: Equivocal
Evidence: Exercise Training in Older Patients with Heart Failure and Preserved Ejection Fraction: A Randomized, Controlled, Single-Blind Trial and Several Other Meta-Analyses and RCTs
This study was a 16-week randomized, attention-controlled, single blinded study investigating the effects of exercise training on exercise intolerance and QOL among 53 elderly HFpEF patients. 38 The primary outcomes studied included peak exercise oxygen uptake ( p = 0.0002), peak power output ( p = 0.002), 6-minute walk distance ( p = 0.002), ventilatory anaerobic threshold ( p = 0.002), and QOL score ( p = 0.03/0.11). 38 This study found statistically significant outcomes in all primary end-points with the exception of QOL score. It is important to mention although no statistical significance was seen with the total QOL score ( p = 0.11), there were trends toward improvement with the physical QOL ( p = 0.03). 38 This study showed the utility of CR in improving functional capacity among elderly HFpEF patients. Another RCT conducted by Gary et al investigated similar end-points among women with diastolic dysfunction. 39 This study showed comparable outcomes in terms of functional capacity improvements, but marked improvement in QOL metrics among the women was included. 39 More recent meta-analyses and smaller cohort studies have mirrored outcomes to the ones mentioned above, some even looking at CR's effect on HFpEF versus HFrEF. 40 41 42 43 Although CR has shown to be overall less effective among HFpEF patients, comparable trends toward improvement in functional capacity are seen in both groups. 42 43 Despite morality being an end-point not investigated, exercise capacity and QOL metrics showed improvement among all studies mentioned. Currently, “Cardiac Rehab effects in HFpEF” (NCT04506606) is a RCT said to publish its data in the upcoming year looking specifically at CR and its effect on neurocirculatory deficits that may be contributing to exercise intolerance. It would be interesting to see if future longitudinal RCTs may shed light on the utility of CR in providing a mortality benefit among HFpEF patients, and not just increased functional capacity.
Atrial Fibrillation
Proper left atrial (LA) function becomes a crucial component to LV filling in HFpEF patients. LV stiffness reduces the amount of passive filling that occurs following isovolumic relaxation. 44 45 The stiffened LV becomes increasingly dependent on LA contraction to augment the stroke volume. 45 46 For this reason, HFpEF patients who develop AF lose this atrial kick that has been shown to decrease exercise capacity and increase mortality. 47 48 49 Apart from hemodynamic considerations, AF can perpetuate and worsen HF through rate dependent deterioration of cardiac function, fibrosis, and neurohormonal vasoconstriction. 50 Management of AF among HFpEF patients is an important therapeutic strategy. But the question becomes whether it should be through rate or rhythm control?
Consensus: Equivocal
Evidence: “A Comparison of Rate Control and Rhythm Control in Patients with Atrial Fibrillation”—(AFFIRM) Trial and The Atrial Fibrillation and Congestive Heart Failure (AF-CHF) trial
AF is more common among HFpEF when compared to its HFrEF counterpart. 51 The prevalence of AF within the HFpEF community is as high as two thirds. 49 With increased frequency of AF seen in patients with diastolic disfunction treatment is compulsory. The biggest question that arises, is should we manage AF in HFpEF patients through controlling the rate or rhythm? Managing the rate in hopes of mitigating chances of tachycardia mediated cardiomyopathy or controlling the rhythm in hopes to maintain sinus rhythm and preserve hemodynamic benefits of synchronized atrial contraction. Current recommendations suggest initial rate control, followed by rhythm control if symptoms persist, but no large RCTs comparing rate versus rhythm-controlled AF among HFpEF patients has been done. 52 53 That being said extensive study on AF in patients with and without HF has allowed extrapolation of possible therapeutic benefits between the two. The AFFIRM trial a landmark study published nearly two decades ago showed no mortality benefit between rate and rhythm control regardless of HF status. 54 A follow-up investigation of the AFFIRM study done a few year later showed that patients who spent longer in sinus rhythm did have a lower risk of death. 55 Another subgroup analysis of the AF-CHF trial showed similar results with time under sinus rhythm improved NYHA functional class among HF patients. 56 Left ventricular filling in HFpEF occurs primarily towards the end of diastole and relies more on atrial contraction than normal hearts. For this reason, HFpEF patients in sinus rhythm benefit from synchronized atrial contraction that might largely be why rhythm control may be more beneficial. Because no large study has been done on rate/rhythm control in AF-HFpEF patients, this continues to remain speculative.
HFpEF as a Systemic Inflammatory Process
Due to the heterogeneity of HFpEF as a disease process, clinical phenotyping has shed light on categorizing HFpEF based on common comorbidities/systemic diseases and providing targeted therapies for these patients. Comorbidities like coronary artery disease, hypertension, and obesity are thought to play an important role in catalyzing an inflammatory cascade, leading to microvascular endothelial dysfunction across many organ systems including the heart. A study published within the last decade identified a novel paradigm in which a systemic proinflammatory state is responsible for cardiomyocyte stiffness and interstitial fibrosis contributing to high diastolic pressures in the LV. 57 Below we discuss several comorbidities/systemic inflammatory conditions and their association with HFpEF prevalence.
Obesity-Related HFpEF
Consensus: Equivocal
Evidence: Effect of Caloric Restriction or Aerobic Exercise Training on Peak Oxygen Consumption and Quality of Life in Obese Older Patients with Heart Failure with Preserved Ejection Fraction: A Randomized Clinical Trial
Obesity is a growing epidemic and common among HFpEF patients. It has a detrimental impact on the CV system. Its deleterious effects are perpetuated by changes in volume status, inflammation, cardiac remodeling, and cardiac loading among others. 58 59 60 61 This study was a randomized, attention-controlled, two by two factorial study conducted over a 7-year period comprised of 100 older obese patients with chronic stable HFpEF. 62 Patients were divided into different treatment arms outlined in Table 4 . Specific inclusion and exclusion criteria are outlined within the study. Primary outcomes quantified were exercise capacity (measured as peak VO 2 mL/kg/min), and QOL (measured using the Minnesota Living with Heart Failure questionnaire). 62 Although the study spanned 7 years, the intervention period consisted of 20 weeks of diet and/or exercise. Exercise attendance and diet adherence were 84 and 99%, respectively. 62 Exercise capacity measured using peak VO2 increased significantly with both diet, 1.3 mL/kg body mass/min (95% CI: 0.8–1.8), p < 0.001 and exercise, 1.2 mL/kg body mass/min (95% CI: 0.7–1.7), p < 0.001, with an additive benefit among patients given both interventions, 2.5 mL/kg/min. 62 Despite improvements in patients functional capacity, similar QOL metrics were not seen in any intervention. This study highlights weight loss through diet and exercise may serve as a targeted therapeutic option for obesity-related HFpEF, and further reinforces the utility of phenotypic specific targeted treatment modalities.
Hypertension
Consensus: Equivocal
Evidence: Effects of Blood Pressure Lowering in Patients with Heart Failure with Preserved Ejection Fraction: A Systematic Review and Meta-Analysis
This meta-analysis included 10 RCTs involving 13,901 patients with HFpEF between January 1996 and July 2017. All studies looked at all-cause mortality, CV mortality, and HF hospitalization as primary end-points. Within these 10 RCTs, there was a statistically significant reduction in blood pressure among the treatment groups (134.7–130.2 mm Hg) when compared to the controls (134.4–133.3 mm Hg) as well as HF hospitalizations (RR: 0.89 [0.82–0.97], p = 0.006). 63 Interestingly, when studies were stratified based on renal function larger blood pressure changes were seen among the treatment groups (134–129.6 mm Hg) versus the control (134.4–132.8) with increased incidence of renal dysfunction (RR: 1.52 [1.31–1.76], p < 0.00001). 63 Despite significant decreases in SBP, none of the RCTs showed an individual nor as a composite benefit in all-cause mortality and CV mortality. 63 This study highlights that although targeting blood pressure alone does not provide a mortality benefit among HFpEF patients, achieving SBP near 130s is associated with a reduction in HF hospitalizations. But also keep in mind aggressive blood pressure management may increase the risk of renal dysfunction. 63 There is growing association between of diastolic dysfunction and hypertensive heart disease as the phenotypic model for the treatment of HFpEF gain traction. The PREFERS Hypertension study (NCT04190420) is a prospective cohort trial currently recruiting patients looking specifically at hypertension and its role in HFpEF disease progression using biomarkers and cardiac imaging variables.
Coronary Artery Disease—Revascularization Therapy
Consensus: Positive
Evidence: Implications of Coronary Artery Disease in Heart Failure with Preserved Ejection Fraction
This retrospective cohort study of 376 HFpEF patients, 255 (68%) having angiographically proven coronary artery disease (CAD). Of those 205 (80%) underwent revascularization (68% percutaneous coronary intervention; 37% surgical bypass). Repeat echocardiography was performed in 151 of the 255 patients with CAD with a median time of 1,219 days after catheterization. 64 CAD-HFpEF patients experienced statistically significant decline in EF and increased mortality when compared to non-CAD-HFpEF patients independent of other predictors (HR: 1.71, 95% CI: 1.03–2.98; p = 0.04). 64 There was also a twofold greater decline in EF in CAD-HFpEF patients who did not undergo revascularization. Complete revascularization was associated with less deterioration in EF and a mortality benefit when compared to incompletely revascularized patients independent of other predictors (HR: 0.56, 95% CI: 0.33–0.93; p = 0.03). 64 This study shows the presence of CAD among HFpEF patients serves as an independent negative prognostic indicator among HFpEF patients, and revascularization is associated with preservation of cardiac function, and improved outcomes. Prospective studies are required to establish the appropriate treatment of CAD in HFpEF patients.
Conclusion
This clinical trial review on the efficacy of medical therapy for patients with HFpEF was designed to shed light as to why there is a lack of evidence-based recommendations and highlight important RCTs that have perpetuated class II and below guidelines for management. HFpEF encompasses nearly half of HF worldwide, and still remains a poor prognostic indicator. Due to the heterogeneity of HFpEF as a disease process, targeted pharmacotherapy has become increasingly difficult. Although there is data supporting beta-blockers, mineralocorticoid receptor antagonists, ACEi, revascularization, cardiac rehabilitation, and more recently SGLT2's as therapeutic targets to reduce hospitalizations, no therapeutic category has found a mortality benefit. We are continuing to learn more about HFpEF as a complex interplay of not only neurohormonal but also myocardial interstitial disturbances driven by comorbid and systemic inflammatory conditions. Future targeted therapies need to focus on treatment of HFpEF as a systemic disease process. This is a major criticism of current major landmark clinical trials discussed throughout this review. Recently published in the 2021 ESC HF guidelines, a new distinction of HFmrEF EF 41 to 49% has become increasingly evident. Many of the RCTs included in this study have included those with HFmrEF. This is a major criticism as it further confounds any therapeutic potential among HFpEF patients. Further research needs to be done with stricter EF cutoffs to find any therapeutic treatment strategies for HFpEF patients. This comprehensive review was also created to give readers a glimpse of the past, present, and future RCTs to address the million-dollar question of medical management of HFpEF. We hope readers develop an understanding and appreciation for the continued efforts to established pharmacotherapeutic links among this subgroup of patients.
Footnotes
Conflict of Interest None declared.
References
- 1.McKee P A, Castelli W P, McNamara P M, Kannel W B. The natural history of congestive heart failure: the Framingham study. N Engl J Med. 1971;285(26):1441–1446. doi: 10.1056/NEJM197112232852601. [DOI] [PubMed] [Google Scholar]
- 2.Butrous H, Pai R G. Heart failure with normal ejection fraction: current diagnostic and management strategies. Expert Rev Cardiovasc Ther. 2013;11(09):1179–1193. doi: 10.1586/14779072.2013.827468. [DOI] [PubMed] [Google Scholar]
- 3.Shah A M, Claggett B, Loehr L R. Heart failure stages among older adults in the community: the Atherosclerosis Risk in Communities Study. Circulation. 2017;135(03):224–240. doi: 10.1161/CIRCULATIONAHA.116.023361. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Bleumink G S, Knetsch A M, Sturkenboom M C. Quantifying the heart failure epidemic: prevalence, incidence rate, lifetime risk and prognosis of heart failure The Rotterdam Study. Eur Heart J. 2004;25(18):1614–1619. doi: 10.1016/j.ehj.2004.06.038. [DOI] [PubMed] [Google Scholar]
- 5.Framingham Heart Study . Lloyd-Jones D M, Larson M G, Leip E P. Lifetime risk for developing congestive heart failure: the Framingham Heart Study. Circulation. 2002;106(24):3068–3072. doi: 10.1161/01.cir.0000039105.49749.6f. [DOI] [PubMed] [Google Scholar]
- 6.Murphy T M, Waterhouse D F, James S. A comparison of HFrEF vs HFpEF's clinical workload and cost in the first year following hospitalization and enrollment in a disease management program. Int J Cardiol. 2017;232:330–335. doi: 10.1016/j.ijcard.2016.12.057. [DOI] [PubMed] [Google Scholar]
- 7.Shah P M, Pai R G. Diastolic heart failure. Curr Probl Cardiol. 1992;17(12):781–868. doi: 10.1016/0146-2806(92)90016-h. [DOI] [PubMed] [Google Scholar]
- 8.Pellikka P A, She L, Holly T A. Variability in ejection fraction measured by echocardiography, gated single-photon emission computed tomography, and cardiac magnetic resonance in patients with coronary artery disease and left ventricular dysfunction. JAMA Netw Open. 2018;1(04):e181456–e181456. doi: 10.1001/jamanetworkopen.2018.1456. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Pai R G, Buech G C. Newer Doppler measures of left ventricular diastolic function. Clin Cardiol. 1996;19(04):277–288. doi: 10.1002/clc.4960190402. [DOI] [PubMed] [Google Scholar]
- 10.Reddy Y NV, Carter R E, Obokata M, Redfield M M, Borlaug B A. A simple, evidence-based approach to help guide diagnosis of heart failure with preserved ejection fraction. Circulation. 2018;138(09):861–870. doi: 10.1161/CIRCULATIONAHA.118.034646. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Almeida P, Rodrigues J, Lourenço P, Maciel M J, Bettencourt P. Prognostic significance of applying the European Society of Cardiology consensus algorithm for heart failure with preserved systolic function diagnosis. Clin Cardiol. 2012;35(12):770–776. doi: 10.1002/clc.22044. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Barandiarán Aizpurua A, Sanders-van Wijk S, Brunner-La Rocca H P. Validation of the HFA-PEFF score for the diagnosis of heart failure with preserved ejection fraction. Eur J Heart Fail. 2020;22(03):413–421. doi: 10.1002/ejhf.1614. [DOI] [PubMed] [Google Scholar]
- 13.Yancy C W, Jessup M, Bozkurt B. 2017 ACC/AHA/HFSA Focused Update of the 2013 ACCF/AHA Guideline for the Management of Heart Failure: a report of the American College of Cardiology/American Heart Association Task Force on Clinical Practice Guidelines and the Heart Failure Society of America. J Am Coll Cardiol. 2017;70(06):776–803. doi: 10.1016/j.jacc.2017.04.025. [DOI] [PubMed] [Google Scholar]
- 14.ESC Scientific Document Group . McDonagh T A, Metra M, Adamo M. 2021 ESC Guidelines for the diagnosis and treatment of acute and chronic heart failure. Eur Heart J. 2021;42(36):3599–3726. doi: 10.1093/eurheartj/ehab368. [DOI] [PubMed] [Google Scholar]
- 15.Beta-Blocker Evaluation of Survival Trial Investigators . Eichhorn E J, Domanski M J, Krause-Steinrauf H, Bristow M R, Lavori P W. A trial of the beta-blocker bucindolol in patients with advanced chronic heart failure. N Engl J Med. 2001;344(22):1659–1667. doi: 10.1056/NEJM200105313442202. [DOI] [PubMed] [Google Scholar]
- 16.Beta-blockers in Heart Failure Collaborative Group . Cleland J GF, Bunting K V, Flather M D. Beta-blockers for heart failure with reduced, mid-range, and preserved ejection fraction: an individual patient-level analysis of double-blind randomized trials. Eur Heart J. 2018;39(01):26–35. doi: 10.1093/eurheartj/ehx564. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.J-DHF Investigators . Yamamoto K, Origasa H, Hori M. Effects of carvedilol on heart failure with preserved ejection fraction: the Japanese Diastolic Heart Failure Study (J-DHF) Eur J Heart Fail. 2013;15(01):110–118. doi: 10.1093/eurjhf/hfs141. [DOI] [PubMed] [Google Scholar]
- 18.Patel K, Fonarow G C, Ahmed M. Calcium channel blockers and outcomes in older patients with heart failure and preserved ejection fraction. Circ Heart Fail. 2014;7(06):945–952. doi: 10.1161/CIRCHEARTFAILURE.114.001301. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Hung M J, Cherng W J, Kuo L T, Wang C H. Effect of verapamil in elderly patients with left ventricular diastolic dysfunction as a cause of congestive heart failure. Int J Clin Pract. 2002;56(01):57–62. [PubMed] [Google Scholar]
- 20.CHARM Investigators and Committees Yusuf S, Pfeffer M A, Swedberg K.Effects of candesartan in patients with chronic heart failure and preserved left-ventricular ejection fraction: the CHARM-Preserved Trial Lancet 2003362(9386):777–781. [DOI] [PubMed] [Google Scholar]
- 21.I-PRESERVE Investigators . Massie B M, Carson P E, McMurray J J. Irbesartan in patients with heart failure and preserved ejection fraction. N Engl J Med. 2008;359(23):2456–2467. doi: 10.1056/NEJMoa0805450. [DOI] [PubMed] [Google Scholar]
- 22.PEP-CHF Investigators . Cleland J G, Tendera M, Adamus J, Freemantle N, Polonski L, Taylor J. The perindopril in elderly people with chronic heart failure (PEP-CHF) study. Eur Heart J. 2006;27(19):2338–2345. doi: 10.1093/eurheartj/ehl250. [DOI] [PubMed] [Google Scholar]
- 23.TOPCAT Investigators . Pitt B, Pfeffer M A, Assmann S F. Spironolactone for heart failure with preserved ejection fraction. N Engl J Med. 2014;370(15):1383–1392. doi: 10.1056/NEJMoa1313731. [DOI] [PubMed] [Google Scholar]
- 24.Pfeffer M A, Claggett B, Assmann S F. Regional variation in patients and outcomes in the Treatment of Preserved Cardiac Function Heart Failure With an Aldosterone Antagonist (TOPCAT) trial. Circulation. 2015;131(01):34–42. doi: 10.1161/CIRCULATIONAHA.114.013255. [DOI] [PubMed] [Google Scholar]
- 25.NHLBI Heart Failure Clinical Research Network . Redfield M M, Anstrom K J, Levine J A. Isosorbide mononitrate in heart failure with preserved ejection fraction. N Engl J Med. 2015;373(24):2314–2324. doi: 10.1056/NEJMoa1510774. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Ayyadurai P, Alkhawam H, Saad M. An update on the CardioMEMS pulmonary artery pressure sensor. Ther Adv Cardiovasc Dis. 2019;13:1.753944719826826E15. doi: 10.1177/1753944719826826. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Krum H.Telemonitoring of fluid status in heart failure: CHAMPION Lancet 2011377(9766):616–618. [DOI] [PubMed] [Google Scholar]
- 28.Adamson P B, Abraham W T, Bourge R C. Wireless pulmonary artery pressure monitoring guides management to reduce decompensation in heart failure with preserved ejection fraction. Circ Heart Fail. 2014;7(06):935–944. doi: 10.1161/CIRCHEARTFAILURE.113.001229. [DOI] [PubMed] [Google Scholar]
- 29.DAPA-HF Trial Committees and Investigators . McMurray J JV, Solomon S D, Inzucchi S E. Dapagliflozin in patients with heart failure and reduced ejection fraction. N Engl J Med. 2019;381(21):1995–2008. doi: 10.1056/NEJMoa1911303. [DOI] [PubMed] [Google Scholar]
- 30.EMPEROR-Reduced Trial Investigators . Packer M, Anker S D, Butler J. Cardiovascular and renal outcomes with empagliflozin in heart failure. N Engl J Med. 2020;383(15):1413–1424. doi: 10.1056/NEJMoa2022190. [DOI] [PubMed] [Google Scholar]
- 31.EMPEROR-Preserved Trial Investigators . Anker S D, Butler J, Filippatos G. Empagliflozin in heart failure with a preserved ejection fraction. N Engl J Med. 2021;385(16):1451–1461. doi: 10.1056/NEJMoa2107038. [DOI] [PubMed] [Google Scholar]
- 32.Lim G B. Dapagliflozin improves exercise capacity in HFpEF. Nat Rev Cardiol. 2022;19(01):6–6. doi: 10.1038/s41569-021-00650-0. [DOI] [PubMed] [Google Scholar]
- 33.Lytvyn Y, Bjornstad P, Udell J A, Lovshin J A, Cherney D ZI. Sodium glucose cotransporter-2 inhibition in heart failure: potential mechanisms, clinical applications, and summary of clinical trials. Circulation. 2017;136(17):1643–1658. doi: 10.1161/CIRCULATIONAHA.117.030012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.RELAX Trial . Redfield M M, Chen H H, Borlaug B A. Effect of phosphodiesterase-5 inhibition on exercise capacity and clinical status in heart failure with preserved ejection fraction: a randomized clinical trial. JAMA. 2013;309(12):1268–1277. doi: 10.1001/jama.2013.2024. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.PARAGON-HF Investigators and Committees . Solomon S D, McMurray J JV, Anand I S. Angiotensin–neprilysin inhibition in heart failure with preserved ejection fraction. N Engl J Med. 2019;381(17):1609–1620. doi: 10.1056/NEJMoa1908655. [DOI] [PubMed] [Google Scholar]
- 36.Ahmed A, Rich M W, Fleg J L. Effects of digoxin on morbidity and mortality in diastolic heart failure: the ancillary digitalis investigation group trial. Circulation. 2006;114(05):397–403. doi: 10.1161/CIRCULATIONAHA.106.628347. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Roveda F, Middlekauff H R, Rondon M U. The effects of exercise training on sympathetic neural activation in advanced heart failure: a randomized controlled trial. J Am Coll Cardiol. 2003;42(05):854–860. doi: 10.1016/s0735-1097(03)00831-3. [DOI] [PubMed] [Google Scholar]
- 38.Kitzman D W, Brubaker P H, Morgan T M, Stewart K P, Little W C. Exercise training in older patients with heart failure and preserved ejection fraction: a randomized, controlled, single-blind trial. Circ Heart Fail. 2010;3(06):659–667. doi: 10.1161/CIRCHEARTFAILURE.110.958785. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Gary R A, Sueta C A, Dougherty M. Home-based exercise improves functional performance and quality of life in women with diastolic heart failure. Heart Lung. 2004;33(04):210–218. doi: 10.1016/j.hrtlng.2004.01.004. [DOI] [PubMed] [Google Scholar]
- 40.Smart N, Haluska B, Jeffriess L, Marwick T H. Exercise training in systolic and diastolic dysfunction: effects on cardiac function, functional capacity, and quality of life. Am Heart J. 2007;153(04):530–536. doi: 10.1016/j.ahj.2007.01.004. [DOI] [PubMed] [Google Scholar]
- 41.Pandey A, Parashar A, Kumbhani D. Exercise training in patients with heart failure and preserved ejection fraction: meta-analysis of randomized control trials. Circ Heart Fail. 2015;8(01):33–40. doi: 10.1161/CIRCHEARTFAILURE.114.001615. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Tanaka S, Sanuki Y, Ozumi K, Harada T, Tasaki H. Heart failure with preserved vs reduced ejection fraction following cardiac rehabilitation: impact of endothelial function. Heart Vessels. 2018;33(08):886–892. doi: 10.1007/s00380-018-1128-2. [DOI] [PubMed] [Google Scholar]
- 43.Iyer A M. Cardiac rehabilitation has important benefits for both HFpEF and HFrEF patients. J Card Fail. 2019;25(08):S143. [Google Scholar]
- 44.Varadarajan P, Pai R G. Prognosis of congestive heart failure in patients with normal versus reduced ejection fractions: results from a cohort of 2,258 hospitalized patients. J Card Fail. 2003;9(02):107–112. doi: 10.1054/jcaf.2003.13. [DOI] [PubMed] [Google Scholar]
- 45.Borlaug B A. The pathophysiology of heart failure with preserved ejection fraction. Nat Rev Cardiol. 2014;11(09):507–515. doi: 10.1038/nrcardio.2014.83. [DOI] [PubMed] [Google Scholar]
- 46.Ling L H, Kistler P M, Kalman J M, Schilling R J, Hunter R J. Comorbidity of atrial fibrillation and heart failure. Nat Rev Cardiol. 2016;13(03):131–147. doi: 10.1038/nrcardio.2015.191. [DOI] [PubMed] [Google Scholar]
- 47.Zakeri R, Borlaug B A, McNulty S E. Impact of atrial fibrillation on exercise capacity in heart failure with preserved ejection fraction: a RELAX trial ancillary study. Circ Heart Fail. 2014;7(01):123–130. doi: 10.1161/CIRCHEARTFAILURE.113.000568. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Melenovsky V, Hwang S J, Lin G, Redfield M M, Borlaug B A. Right heart dysfunction in heart failure with preserved ejection fraction. Eur Heart J. 2014;35(48):3452–3462. doi: 10.1093/eurheartj/ehu193. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Zakeri R, Chamberlain A M, Roger V L, Redfield M M. Temporal relationship and prognostic significance of atrial fibrillation in heart failure patients with preserved ejection fraction: a community-based study. Circulation. 2013;128(10):1085–1093. doi: 10.1161/CIRCULATIONAHA.113.001475. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.American College of Cardiology Foundation ; American Heart Association Task Force on Practice Guidelines . Yancy C W, Jessup M, Bozkurt B. 2013 ACCF/AHA guideline for the management of heart failure: a report of the American College of Cardiology Foundation/American Heart Association Task Force on Practice Guidelines. J Am Coll Cardiol. 2013;62(16):e147–e239. doi: 10.1016/j.jacc.2013.05.019. [DOI] [PubMed] [Google Scholar]
- 51.Sartipy U, Dahlström U, Fu M, Lund L H. Atrial fibrillation in heart failure with preserved, mid-range, and reduced ejection fraction. JACC Heart Fail. 2017;5(08):565–574. doi: 10.1016/j.jchf.2017.05.001. [DOI] [PubMed] [Google Scholar]
- 52.American College of Cardiology/American Heart Association Task Force on Practice Guidelines . January C T, Wann L S, Alpert J S. 2014 AHA/ACC/HRS guideline for the management of patients with atrial fibrillation: a report of the American College of Cardiology/American Heart Association Task Force on Practice Guidelines and the Heart Rhythm Society. J Am Coll Cardiol. 2014;64(21):e1–e76. doi: 10.1016/j.jacc.2014.03.022. [DOI] [PubMed] [Google Scholar]
- 53.Patel R B, Vaduganathan M, Shah S J, Butler J. Atrial fibrillation in heart failure with preserved ejection fraction: insights into mechanisms and therapeutics. Pharmacol Ther. 2017;176:32–39. doi: 10.1016/j.pharmthera.2016.10.019. [DOI] [PubMed] [Google Scholar]
- 54.Atrial Fibrillation Follow-up Investigation of Rhythm Management (AFFIRM) Investigators . Wyse D G, Waldo A L, DiMarco J P. A comparison of rate control and rhythm control in patients with atrial fibrillation. N Engl J Med. 2002;347(23):1825–1833. doi: 10.1056/NEJMoa021328. [DOI] [PubMed] [Google Scholar]
- 55.AFFIRM Investigators . Corley S D, Epstein A E, DiMarco J P. Relationships between sinus rhythm, treatment, and survival in the Atrial Fibrillation Follow-Up Investigation of Rhythm Management (AFFIRM) Study. Circulation. 2004;109(12):1509–1513. doi: 10.1161/01.CIR.0000121736.16643.11. [DOI] [PubMed] [Google Scholar]
- 56.AF-CHF Trial Investigators . Suman-Horduna I, Roy D, Frasure-Smith N. Quality of life and functional capacity in patients with atrial fibrillation and congestive heart failure. J Am Coll Cardiol. 2013;61(04):455–460. doi: 10.1016/j.jacc.2012.10.031. [DOI] [PubMed] [Google Scholar]
- 57.Georgakopoulos D, Little W C, Abraham W T, Weaver F A, Zile M R. Chronic baroreflex activation: a potential therapeutic approach to heart failure with preserved ejection fraction. J Card Fail. 2011;17(02):167–178. doi: 10.1016/j.cardfail.2010.09.004. [DOI] [PubMed] [Google Scholar]
- 58.Obokata M, Reddy Y NV, Pislaru S V, Melenovsky V, Borlaug B A. Evidence supporting the existence of a distinct obese phenotype of heart failure with preserved ejection fraction. Circulation. 2017;136(01):6–19. doi: 10.1161/CIRCULATIONAHA.116.026807. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59.Lauer M S, Anderson K M, Kannel W B, Levy D. The impact of obesity on left ventricular mass and geometry. The Framingham Heart Study. JAMA. 1991;266(02):231–236. [PubMed] [Google Scholar]
- 60.Abel E D, Litwin S E, Sweeney G. Cardiac remodeling in obesity. Physiol Rev. 2008;88(02):389–419. doi: 10.1152/physrev.00017.2007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.Lavie C J, Alpert M A, Arena R, Mehra M R, Milani R V, Ventura H O. Impact of obesity and the obesity paradox on prevalence and prognosis in heart failure. JACC Heart Fail. 2013;1(02):93–102. doi: 10.1016/j.jchf.2013.01.006. [DOI] [PubMed] [Google Scholar]
- 62.Kitzman D W, Brubaker P, Morgan T. Effect of caloric restriction or aerobic exercise training on peak oxygen consumption and quality of life in obese older patients with heart failure with preserved ejection fraction: a randomized clinical trial. JAMA. 2016;315(01):36–46. doi: 10.1001/jama.2015.17346. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63.Kawano H, Fujiwara A, Kai H. Effects of blood pressure lowering in patients with heart failure with preserved ejection fraction: a systematic review and meta-analysis. Hypertens Res. 2019;42(04):504–513. doi: 10.1038/s41440-019-0216-8. [DOI] [PubMed] [Google Scholar]
- 64.Hwang S-J, Melenovsky V, Borlaug B A.Implications of coronary artery disease in heart failure with preserved ejection fraction J Am Coll Cardiol 201463(25 Pt A, 25 Part A):2817–2827. [DOI] [PubMed] [Google Scholar]
- 65.Australia/New Zealand Heart Failure Research Collaborative Group Macmahon S, Sharpe N.Randomised, placebo-controlled trial of carvedilol in patients with congestive heart failure due to ischaemic heart disease Lancet 1997349(9049):375–380. [PubMed] [Google Scholar]
- 66.Dargie H J.Effect of carvedilol on outcome after myocardial infarction in patients with left-ventricular dysfunction: the CAPRICORN randomised trial Lancet 2001357(9266):1385–1390. [DOI] [PubMed] [Google Scholar]
- 67.Carvedilol hibernating reversible ischaemia trial: marker of success investigators Cleland J G, Pennell D J, Ray S G.Myocardial viability as a determinant of the ejection fraction response to carvedilol in patients with heart failure (CHRISTMAS trial): randomised controlled trial Lancet 2003362(9377):14–21. [DOI] [PubMed] [Google Scholar]
- 68.The Cardiac Insufficiency Bisoprolol Study (CIBIS). CIBIS Investigators and Committees . Committees C IBSCIA. A randomized trial of beta-blockade in heart failure. Circulation. 1994;90(04):1765–1773. doi: 10.1161/01.cir.90.4.1765. [DOI] [PubMed] [Google Scholar]
- 69.Poole-Wilson P A, Lechat P.The cardiac insufficiency Bisoprolol study II Lancet 1999353(9161):1360–1361. [DOI] [PubMed] [Google Scholar]
- 70.Carvedilol Prospective Randomized Cumulative Survival Study Group . Packer M, Coats A J, Fowler M B. Effect of carvedilol on survival in severe chronic heart failure. N Engl J Med. 2001;344(22):1651–1658. doi: 10.1056/NEJM200105313442201. [DOI] [PubMed] [Google Scholar]
- 71.Metoprolol in Dilated Cardiomyopathy (MDC) Trial Study Group Waagstein F, Bristow M R, Swedberg K.Beneficial effects of metoprolol in idiopathic dilated cardiomyopathy Lancet 1993342(8885):1441–1446. [DOI] [PubMed] [Google Scholar]
- 72.MERIT-HF Study Group Effect of metoprolol CR/XL in chronic heart failure: metoprolol CR/XL randomised intervention trial in-congestive heart failure (MERIT-HF) Lancet 1999353(9169):2001–2007. [PubMed] [Google Scholar]
- 73.SENIORS Investigators . Flather M D, Shibata M C, Coats A J. Randomized trial to determine the effect of nebivolol on mortality and cardiovascular hospital admission in elderly patients with heart failure (SENIORS) Eur Heart J. 2005;26(03):215–225. doi: 10.1093/eurheartj/ehi115. [DOI] [PubMed] [Google Scholar]
- 74.U.S. Carvedilol Heart Failure Study Group . Packer M, Bristow M R, Cohn J N. The effect of carvedilol on morbidity and mortality in patients with chronic heart failure. N Engl J Med. 1996;334(21):1349–1355. doi: 10.1056/NEJM199605233342101. [DOI] [PubMed] [Google Scholar]
- 75.Benetos A, Gautier S, Ricard S. Influence of angiotensin-converting enzyme and angiotensin II type 1 receptor gene polymorphisms on aortic stiffness in normotensive and hypertensive patients. Circulation. 1996;94(04):698–703. doi: 10.1161/01.cir.94.4.698. [DOI] [PubMed] [Google Scholar]
- 76.Jia G, Aroor A R, Hill M A, Sowers J R. Role of renin-angiotensin-aldosterone system activation in promoting cardiovascular fibrosis and stiffness. Hypertension. 2018;72(03):537–548. doi: 10.1161/HYPERTENSIONAHA.118.11065. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 77.van Empel V, Brunner-La Rocca H P. Inflammation in HFpEF: key or circumstantial? Int J Cardiol. 2015;189:259–263. doi: 10.1016/j.ijcard.2015.04.110. [DOI] [PubMed] [Google Scholar]
- 78.Rales Investigators . Zannad F, Alla F, Dousset B, Perez A, Pitt B. Limitation of excessive extracellular matrix turnover may contribute to survival benefit of spironolactone therapy in patients with congestive heart failure: insights from the randomized Aldactone evaluation study (RALES) Circulation. 2000;102(22):2700–2706. doi: 10.1161/01.cir.102.22.2700. [DOI] [PubMed] [Google Scholar]
- 79.Struthers A D. Aldosterone in heart failure: pathophysiology and treatment. Curr Heart Fail Rep. 2004;1(04):171–175. doi: 10.1007/s11897-004-0005-8. [DOI] [PubMed] [Google Scholar]
- 80.Gheorghiade M, De Luca L, Fonarow G C, Filippatos G, Metra M, Francis G S.Pathophysiologic targets in the early phase of acute heart failure syndromes Am J Cardiol 200596(6A):11G–17G. [DOI] [PubMed] [Google Scholar]
- 81.De Luca L, Fonarow G C, Adams K F., Jr Acute heart failure syndromes: clinical scenarios and pathophysiologic targets for therapy. Heart Fail Rev. 2007;12(02):97–104. doi: 10.1007/s10741-007-9011-8. [DOI] [PubMed] [Google Scholar]
- 82.Zakeri R, Levine J A, Koepp G A. Nitrate's effect on activity tolerance in heart failure with preserved ejection fraction trial: rationale and design. Circ Heart Fail. 2015;8(01):221–228. doi: 10.1161/CIRCHEARTFAILURE.114.001598. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 83.Battock D J, Levitt P W, Steele P P. Effects of isosorbide dinitrate and nitroglycerin on central circulatory dynamics in coronary artery disease. Am Heart J. 1976;92(04):455–458. doi: 10.1016/s0002-8703(76)80044-0. [DOI] [PubMed] [Google Scholar]
- 84.Elkayam U. Nitrates in the treatment of congestive heart failure. Am J Cardiol. 1996;77(13):41C–51C. doi: 10.1016/s0002-9149(96)00188-9. [DOI] [PubMed] [Google Scholar]
- 85.Takimoto E, Champion H C, Li M. Chronic inhibition of cyclic GMP phosphodiesterase 5A prevents and reverses cardiac hypertrophy. Nat Med. 2005;11(02):214–222. doi: 10.1038/nm1175. [DOI] [PubMed] [Google Scholar]
- 86.Solomon S D, Rizkala A R, Gong J. Angiotensin receptor neprilysin inhibition in heart failure with preserved ejection fraction: rationale and design of the PARAGON-HF trial. JACC Heart Fail. 2017;5(07):471–482. doi: 10.1016/j.jchf.2017.04.013. [DOI] [PubMed] [Google Scholar]
- 87.Vardeny O, Miller R, Solomon S D. Combined neprilysin and renin-angiotensin system inhibition for the treatment of heart failure. JACC Heart Fail. 2014;2(06):663–670. doi: 10.1016/j.jchf.2014.09.001. [DOI] [PubMed] [Google Scholar]
- 88.Bavishi C, Chatterjee S, Ather S, Patel D, Messerli F H. Beta-blockers in heart failure with preserved ejection fraction: a meta-analysis. Heart Fail Rev. 2015;20(02):193–201. doi: 10.1007/s10741-014-9453-8. [DOI] [PubMed] [Google Scholar]


