Abstract
Purpose
ADAMTSL4-associated ectopia lentis is a rare autosomal recessive condition that is primarily associated with crystalline lens displacement. However, the prevalence of other ocular and systemic manifestations of this condition is poorly understood. In this study, we summarize the ocular and systemic phenotypic spectrum of this condition.
Methods
A cross-sectional case study series of four individuals with biallelic pathogenic or likely pathogenic ADAMTSL4 variants was performed alongside a literature review of individuals with ADAMTSL4-associated ectopia lentis on September 29, 2021. Ocular and systemic findings, complications, and genetic findings of all four individuals were collected and summarized.
Results
The phenotypic spectrum across 91 individuals sourced from literature and four individuals from this case study series was highly variable. The main ocular phenotypes included ectopia lentis (95/95, 100%), ectopia lentis et pupillae (18/95, 19%), iris transillumination (13/95, 14%), iridodonesis (12/95, 13%), persistent pupillary membrane (12/95, 13%), and early-onset cataract or lens opacities (12/95, 13%). Anterior segment features other than ectopia lentis appeared to be exclusively associated with biallelic loss of function variants (p<0.001). Pupillary block glaucoma had a prevalence of 1%. Post-lensectomy complications included retinal detachment (6/41, 15%), elevated intraocular pressure (4/41, 10%), and aphakic glaucoma (1/41, 2%). Most individuals were not reported to have had systemic features (69/95, 73%).
Conclusions
The clinical phenotype of ADAMTSL4-associated ectopia lentis was summarized and expanded. Clinicians should be aware of the varied ocular phenotype and the risks of retinal detachment, ocular hypertension, and glaucoma in the diagnosis and management of this condition.
Introduction
Autosomal recessive ectopia lentis associated with variants in the ADAMTSL4 gene (Gene ID: 54507; OMIM 610113) is a rare condition [1]. The phenotypic spectrum of ADAMTSL4-associated ectopia lentis was initially limited to isolated ectopia lentis or ectopia lentis et pupillae, a condition in which the direction of the corectopia is opposite that of the ectopia lentis [2]. More recent reports have expanded this spectrum to include congenital iris abnormalities, high myopia, raised intraocular pressure (IOP), and retinal detachment [3,4]. Systemic features are rarely documented, and major systemic involvement has not been suggested as a feature of the condition [1,3]. Differential diagnoses for ectopia lentis include conditions with systemic involvement such as Marfan syndrome (associated with variants of FBN1) and Weill-Marchesani syndrome (WMS; associated with variants of ADAMTS10, ADAMTS17, FBN1, and LTBP2) [5]. In this study, we describe a series of four previously unreported individuals from three pedigrees and summarize the phenotypic spectrum of ADAMTSL4-associated ectopia lentis.
Methods
Participants
The participants in the cross-sectional case series were part of the Australian and New Zealand Registry of Advanced Glaucoma [6]. This registry recruits individuals with anterior segment anomalies, including ectopia lentis, regardless of their glaucoma status [6]. Ophthalmic clinical details were obtained from the participants’ referring ophthalmologists. Genetic results and systemic features were obtained from the participants’ geneticists. Additional investigations, including electroretinography, Pentacam Scheimpflug imaging (Oculus, Wetzlar, Germany; for the measurement of the central corneal thickness [CCT], corneal curvature, and anterior chamber depth [ACD]), axial length measurement (Zeiss IOLMaster 500, Carl Zeiss Meditec, Jena, Germany), optical coherence tomography (OCT; CIRRUS SD-OCT, Carl Zeiss Meditec, Dublin, CA; for the measurement of the peripapillary retinal nerve fiber layer thickness), and the Humphrey visual field (HVF; Humphrey Field Analyzer; Carl Zeiss Meditec, Dublin, CA) were performed at the discretion of their referring ophthalmologist. The IOP was measured using rebound tonometry (iCare tonometer, Icare Finland Oy, Vantaa, Finland) or Goldmann applanation tonometry (GAT; Haag-Streit USA, Mason, OH). A high axial length was defined as ≥27 mm [7], and ocular hypertension was defined as IOP>21 mmHg. Ethics approval was obtained from the Southern Adelaide Clinical Human Research Ethics Committee, and this study adhered to the Tenets of the revised Declaration of Helsinki. All the participants provided their written informed consent.
Genetic testing
The genetic results were validated in a laboratory accredited by the National Association of Testing Authorities (SA Pathology, Adelaide, Australia or the Victorian Clinical Genetics Services, Melbourne, Australia). All the variants were reported using the GRCh37 reference genome, and all the ADAMTSL4 transcripts were annotated against the canonical DNA (NM_019032) and protein (NP_061905) transcripts. For the purposes of this study, loss of function (LoF) variants were defined as those that introduce a premature stop codon (nonsense), shift the transcriptional reading frame (frameshift), or alter the two essential splice-site nucleotides immediately upstream or downstream of a coding exon (splice donor or acceptor); and all missense variants were defined as non-LoF [8].
Literature review
PubMed, MEDLINE, Scopus, CINAHL, and PsycINFO were searched using the terms “ADAMTSL4,” “ADAMTS-Like protein 4” or “autosomal recessive ectopia lentis,” and “ectopia lentis” or “ectopia lentis et pupillae” or “lens subluxation.” No date restrictions were used. The search was last conducted on September 29, 2021 (Appendix 1).
Statistical analysis
All the calculations were performed using SPSS version 27.0 for Windows (IBM/SPSS Inc., Chicago, IL). The chi-square test with continuity correction or Fisher’s exact test was used for categorical variables as appropriate. A p value of <0.05 was considered statistically significant. Multiple testing adjustments were not used, as all the analyses were exploratory in nature.
Results
Clinical features
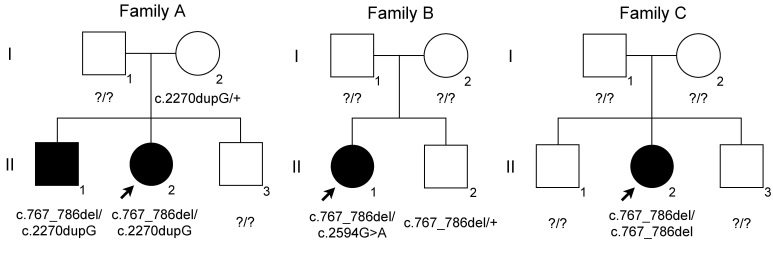
Four affected individuals from three unrelated pedigrees were included in this case study series (Figure 1). Their clinical characteristics are summarized in Table 1.
Figure 1.
Pedigrees of families A, B, and C. The filled symbols denote the affected individuals. The arrow denotes the proband.
Table 1. Most recent clinical characteristics of the four individuals described in this case series.
| Individual | A-II-1 | A-II-2 | B-II-1 | C-II-2 |
|---|---|---|---|---|
|
Sex
|
M |
F |
F |
F |
|
Age at follow-up (years)
|
14 |
12 |
18 |
11 |
|
BCVA
(OD, OS)
|
20/20, 20/20 |
20/20, 20/32 |
20/40, LP |
CF, 20/63 |
|
SE (D)
(OD, OS)
|
+0.50, −0.50 |
−2.0, −3.75, |
+1.25 (aphakic), na |
−22.5, −3.25 |
|
CCT (µm)
(OD, OS)
|
594, 581 |
524, 527 |
632, 681 |
na |
|
IOP (mmHg; OD, OS)
|
28, 21 |
16, 13 |
17, 9 |
6, 11 |
|
CC (D)
(OD, OS)
|
42.6/44.1,
43.0/43.8 |
44.8/45.9,
44.1/46.2 |
na |
na |
|
AL (mm; OD, OS)
|
na |
na |
31.48, 31.61 |
27.30, 23.01 |
|
ACD (mm)
(OD, OS)
|
4.55, 4.90a |
4.26, 4.19a |
na |
na |
|
Ectopia lentis
|
OD |
OUb |
OU |
OD |
|
PPM
|
OU |
OU |
- |
- |
|
Iris TID
|
OU |
OU |
- |
- |
|
Spherophakia
|
OU |
OU |
- |
- |
|
Iris processes
|
OS |
- |
- |
- |
|
RD
|
- |
- |
OS |
- |
|
Lensectomy
|
- |
- |
OU |
- |
|
Glaucoma
|
- |
- |
ODc |
- |
| Systemic features | - | Learning difficulties, mild eczema, fructose and lactose intolerance | Motor delay, hypermobility, depression, anxiety, ASD | Learning difficulties, low vitamin D, ASD |
M: male; F: female; BCVA: best-corrected visual acuity; OD: right eye; OS; left eye; LP: light perception; CF: count fingers; SE: spherical equivalent; D: diopters; na: not available; CCT: central corneal thickness; IOP: intraocular pressure; CC: corneal curvature; AL: axial length; ACD: anterior chamber depth; OU: both eyes; PPM: persistent pupillary membrane; TID: transillumination defect; RD: retinal detachment; ASD: autism spectrum disorder. aOcular surgery was not performed on these individuals bIndividual A-II-2 had phacodonesis; a preceding sign of ectopia lentis cIndividual B-II-1 had aphakic glaucoma (i.e., glaucoma onset followed lensectomy)
Individual A-II-2
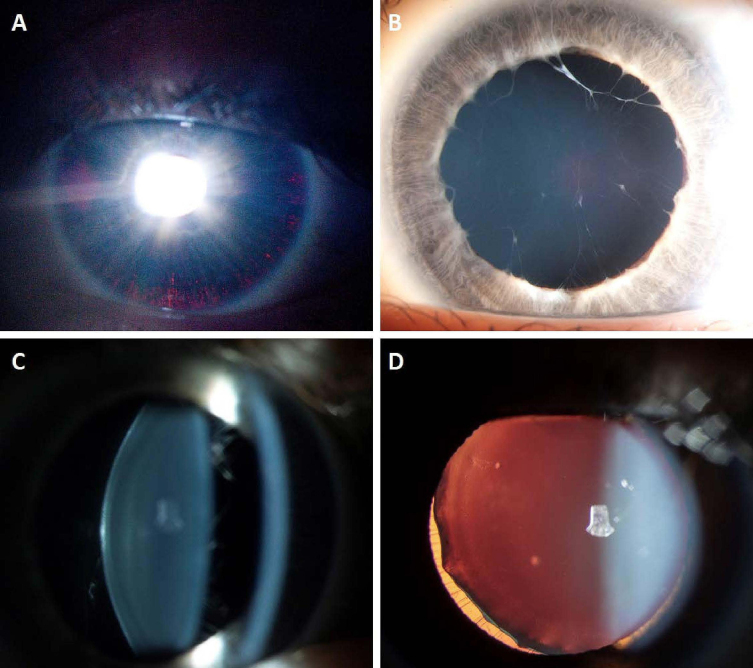
Individual A-II-2 experienced difficulty reading the notes on the classroom board from a distance at age 6 years. She had reported learning difficulties, mild eczema, and fructose and lactose intolerance. She had no significant family history of ocular disease or collagenopathies. In the clinical examination, her best-corrected visual acuity (BCVA) was 20/32 in the right eye (OD) and 20/50 in the left eye (OS) in an outdated mild myopic correction. The anterior segment examination revealed bilateral multiple iris transillumination defects (Figure 2A) and bilateral phacodonesis, a preceding sign of ectopia lentis. Extensive persistent pupillary membranes were found bilaterally adherent to the anterior lens surface (Figure 2B). The rebound tonometry IOP, corneal curvature, and CCT were within the normal range, and the ACDs were deep (Table 1). In the dilated examination, both lenses were clear but with posterior lentiglobus or spherophakia (Figure 2C). The mild myopic astigmatism was corrected with full-time spectacle wear, and the BCVA improved to 20/20 OD and 20/32 OS. The individual’s latest clinical review was performed at 12 years of age, and the findings were unchanged.
Figure 2.
Clinical photography. The slit lamp imaging shows A: iris transillumination defects, B: a persistent pupillary membrane, and C: posterior lentiglobus or spherophakia on the slit illumination observed in Individual A-II-2. D: Ectopia lentis seen in Individual A-II-1 OD.
Individual A-II-1
The brother of the proband was identified at age 13 years through family screening and was systemically well. The ophthalmic examination revealed bilateral mild myopic astigmatism with a BCVA of 20/20 in both eyes (OU). The anterior segment examination revealed iris thinning, mild iris stromal atrophy, and persistent pupillary membranes OU. Iris transillumination defects were later observed in high resolution slit-lamp imaging. The corneal curvature was normal, the ACDs were deep, and the CCTs were thick at 594 microns OD and 581 microns OS (Table 1). The gonioscopy revealed open angles and a deeply pigmented trabecular meshwork OD, and iris processes OS. Deposition of the pigment in other ocular structures (e.g., the corneal endothelium and the lens capsule) and backward bowing of the iris were not observed. The dilated examination clearly showed lens subluxation OD (Figure 2D) and posterior lentiglobus or spherophakia OU. Both fundi were unremarkable, and the optic nerve vertical cup-to-disc ratio (VCDR) was 0.3 OU. In the most recent review (at age 14 years), the individual was being monitored for ocular hypertension. Antiglaucoma medication has not yet been initiated.
Individual B-II-1
Individual B-II-1 initially presented to an ophthalmologist at age 4 years with an uncorrected visual acuity of 20/400 OU. She was reported to have a motor delay, hypermobility, autism spectrum disorder, depression, and anxiety. There was no significant family history of ophthalmic or systemic disease. She had high myopia and lens subluxation OU, accompanied by small amplitude nystagmus and esotropia OS. Her electroretinography result was normal. With myopic correction, her BCVA improved to 20/60 OD and 20/120 OS. At age 10 years, her BCVA dropped to 20/400 OS due to worsening lens subluxation and consequent vitreous prolapse into the anterior chamber. She underwent lensectomy, pars plana vitrectomy (PPV), and prophylactic 360° retinopexy OU. As her axial lengths were high (Table 1), she was left aphakic OU. Her BCVA improved to 20/40 OD and 20/50 OS nine months post-operatively. Myopic degeneration was noted OU.
Five years post-surgery, she presented with a 2-week history of worsening vision OS. Her BCVA was ‘light-perception’ OS due to chronic total retinal detachment OS that was not deemed suitable for surgery. Her IOPs (in GAT) were 32 mmHg OD and 9 mmHg OS. Her CCTs were thick at 632 microns OD and 681 microns OS (Table 1), and the optic nerve head was tilted OD with peripapillary atrophy and a VCDR of 0.5. The average peripapillary retinal nerve fiber layer thickness in the OCT imaging was 79 µm. A corresponding nasal-step visual field defect was revealed in HVF 24–2 SITA Fast perimetry. Aphakic glaucoma was diagnosed, and topical glaucoma therapy was commenced. After 2 years of using latanoprost once daily, her treatment was escalated to latanoprost–timolol combination drops due to increasing IOP. In her last review (at age 18 years), her condition was stable and her IOP was controlled with latanoprost–timolol combination drops.
Individual C-II-2
Individual C-II-2 presented at the age of 11 years with chronic lens subluxation, high myopia, anisometropia, exotropia, and dense amblyopia OD. She had no family history of ophthalmic or systemic disease. She had been diagnosed with autism spectrum disorder, learning difficulties, and low vitamin D. Her BCVA was ‘count fingers’ OD and 20/63 OS. Her axial length OD was high but her IOP was normal (Table 1). The slit-lamp examination revealed superotemporal lens subluxation and a tilted optic disc OD. The contralateral lens and posterior segment were unremarkable.
Genetic results
Pathogenic variants in ADAMTSL4 were identified in all four individuals (Table 2). All variants have been previously described. The frameshift variant c.767_786del has been reported as a European founder variant [9].
Table 2. Genetic findings of the four individuals described in this case series.
| Individual | A-II-1 | A-II-2 | B-II-1 | C-II-2 |
|---|---|---|---|---|
|
cDNA
|
c.767_786del/
c.2270dupG |
c.767_786del/
c.2270dupG |
c.767_786del/
c.2594G>A |
c.767_786del/
c.767_786del |
|
Protein
|
p.Gln256Profs*38/
p.Gly758Trpfs*59 |
p.Gln256Profs*38/
p.Gly758Trpfs*59 |
p.Gln256Profs*38/
p.Arg865His |
p.Gln256Profs*38/
p.Gln256Profs*38 |
| Pathogenicitya | pathogenic/ likely pathogenic | pathogenic/ likely pathogenic | pathogenic/ likely pathogenic | pathogenic/ pathogenic |
aPathogenicity determined as per the 2015 American College of Medical Genetics and Genomics guidelines [41]
Literature review
A literature review identified 14 articles that described individuals with ADAMTSL4-associated lentis (Appendix 1) [3-5,9-19]. Across these studies, 91 individuals with homozygous or compound heterozygous variants in ADAMTSL4 were described and included. With the inclusion of the current series, there were 95 reported individuals with ADAMTSL4-associated ectopia lentis. Of these, 45% (39/87) were female. The age at diagnosis was recorded in 59% (56/95) of the individuals as a median of 3 years (interquartile range: 1.1–6 years). The age at last follow-up was recorded in 61% (58/95) of the individuals as a median of 11 years (interquartile range: 6–31 years).
To investigate potential genotype-phenotype correlations, all the variants reported were considered either LoF or non-LoF (Appendix 2). Consequently, 75% (71/95) of the individuals had biallelic LoF variants, and 25% (24/95) had at least one non-LoF allele (Appendix 3). The most common genetic variant was the European founder frameshift variant c.767_786del, which was identified in 58% (55/95) of the individuals [3,5,9-11,14-17]. This variant was reported in 73% (52/71) of the individuals with biallelic LoF variants [3,5,9-11,14-17] and in 13% (3/24) of the individuals with at least one non-LoF allele (Appendix 3) [11]. Among the individuals with the European founder frameshift variant, 60% (33/55) were reported to have European ancestry [5,9,11,15,17] and 2% (1/55), New Zealand Māori ancestry [11]. The ancestry of the remaining 38% (21/55) of the individuals [3,10,14,16,17] was not reported. The ancestries reported for the other variants are listed in Appendix 4. One additional individual reported in the literature was excluded from this study, as the variant (c.1delA/p.?Met1) could not be verified as pathogenic due to the existence of alternative start codon transcripts [5].
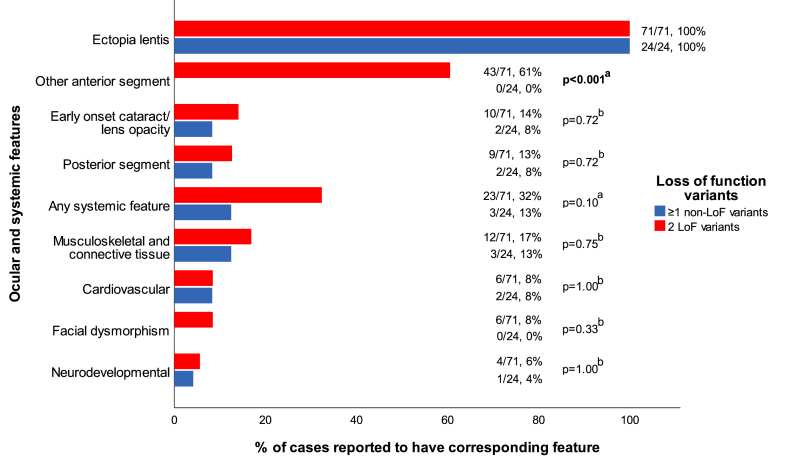
The ocular anterior segment findings of the 95 individuals with ADAMTSL4-associated ectopia lentis are summarized in Table 3. All the individuals reported ectopia lentis, with 19% (18/95) reporting ectopia lentis et pupillae. Iris transillumination defects were reported in 14% (13/95), iridodonesis in 13% (12/95), and persistent pupillary membrane in 13% (12/95) of the individuals. Early-onset cataract or lens opacity was noted in 13% (12/95) of the individuals. Of these 12 individuals, 75% (9/12) had cataract or lens opacity diagnosed at <40 years of age [3-5,13], and the remaining 25% (3/12) had an unknown age at onset but had undergone cataract surgery at ≤50 years of age [16]. Anterior segment features, excluding cataract and ectopia lentis, were exclusively reported in individuals with biallelic LoF variants, as shown in Figure 3 (p<0.001, chi-square test with continuity correction).
Table 3. Prevalence of ocular anterior segment features in 95 individuals with ADAMTSL4-associated ectopia lentis.
| Ocular anterior segment features | Number of individuals in literature (n) | Individuals in this report (n) | Combined total (n) | Proportion of individuals (%) | References |
|---|---|---|---|---|---|
| Ectopia lentis |
91 |
4a |
95 |
100 |
[3–5,9–19] |
| Ectopia lentis et pupillae |
18 |
0 |
18 |
19 |
[3-5,10,15-17] |
| Iris TID |
11 |
2 |
13 |
14 |
[4,5,16] |
| Iridodonesis |
12 |
0 |
12 |
13 |
[4,5,9,19] |
| PPM |
10 |
2 |
12 |
13 |
[3-5,9,16] |
| Early onset cataract or
lens opacity |
12 |
0 |
12 |
13 |
[3-5,13,16] |
| Poor pupillary dilatation |
11 |
0 |
11 |
12 |
[4,16] |
| Spherophakia |
9 |
2 |
11 |
12 |
[3,4,9,16] |
| Corectopia |
4 |
0 |
4 |
4 |
[16] |
| Iris coloboma |
2 |
0 |
2 |
2 |
[5] |
| Lens coloboma |
2 |
0 |
2 |
2 |
[16] |
| Posterior synechiae |
1 |
0 |
1 |
1 |
[18] |
| Iris processes | 0 | 1 | 1 | 1 | - |
TID: transillumination defect; PPM: persistent pupillary membrane; aIndividual A-II-2 had phacodonesis; a sign which commonly precedes ectopia lentis.
Figure 3.
Prevalence of ocular and systemic features stratified by the predicted loss of the function allele count. Other anterior segment features included: ectopia lentis et pupillae, iris transillumination defect, iridodonesis, persistent pupillary membrane, poor pupillary dilatation, spherophakia, corectopia, iris coloboma, lens coloboma, posterior synechiae, and iris processes. The posterior segment features included retinal detachment, axial length ≥ 27.0 mm, and posterior staphyloma. The anterior segment features other than ectopia lentis appeared to have been exclusively associated with biallelic loss of function variants (p < 0.001). LoF: loss of function variant; non-LoF: non-loss of function variant. The bold values indicate statistical significance. aChi-square test with continuity correction, bFisher exact test.
Few reports described the presence of deep anterior chambers in unoperated eyes (median: 3.75 mm [range: 3.00–4.10 mm]) [16], increased mean CCTs of 566.1 µm (95% CI: 515.3–616.8 µm) [15], and median CCTs of 589 µm (range: 528–630 µm) [16]. The average corneal curvature might have been normal (i.e., >42.0 D) [11] or flattened (i.e., <42.0 D) [15]. The overall average values of either of these characteristics could not be calculated, as cohort medians or means were often reported rather than individual results.
The numbers of individuals who had disease complications either from surgical intervention (i.e., lensectomy) or without surgical intervention are summarized in Table 4. Lensectomies were performed in 43% (41/95) of the individuals [3,5,9,11,12,14-16,18,19]. Of these, 32% (13/41) of the individuals were left aphakic [11,14,18], 7% (3/41) individuals were pseudophakic [3,11,12], and 61% (25/41) had an unknown phakic status [3,5,9,12,15,16,19]. Lensectomy was complicated post-operatively by elevated IOP in 10% (4/41) of the individuals [5,9,16] and by retinal detachment in 15% (6/41) of the individuals [3,5,15,16,19]. Of those with retinal detachment, 33% (2/6) underwent a combined procedure that included PPV [15]. Aphakic glaucoma developed in one individual (B-II-1) 5 years post-lensectomy (1/41, 2%). High IOP was otherwise recorded in 7% (4/54) of the individuals without lensectomy [5,10,16], and pupillary block glaucoma was diagnosed in one individual at 21 years of age [13], representing a total prevalence of 1% (1/95).
Table 4. Prevalence of disease complications in 95 individuals with ADAMTSL4-associated ectopia lentis stratified by lensectomy status.
| Complication | No lensectomy (n=54) (n, %) | Lensectomy (n=41) (n, %) | Overall prevalence (n=95) (n, %) | References |
|---|---|---|---|---|
| Retinal detachment |
1 (2) |
6 (15)a |
7 (7) |
[3–5,15,16,19]b |
| High axial lengthc |
3 (6) |
3 (7) |
6 (6) |
[4,15]b |
| Posterior staphyloma |
1 (2) |
1 (2) |
2 (2) |
[15] |
| Ocular hypertension |
4 (7) |
4 (10) |
8 (8) |
[5,9,10,16]b |
| Glaucoma |
2 (4) |
1 (2) |
3 (3) |
[4,13]b |
| Pupillary block glaucoma |
1 (2) |
0 (0) |
1 (1) |
[13] |
| Aphakic glaucoma |
0 (0) |
1 (2) |
1 (1) |
b
|
| Unreported etiology | 1 (2) | 0 (0) | 1 (1) | [4] |
a33% (2/6) individuals had a concurrent pars plana vitrectomy (PPV) bIncludes individuals presented in this case series cHigh axial length defined as ≥27.0 mm
In 73% (69/95) of the individuals, systemic features were not reported (the specific systemic features are summarized in Appendix 5). There was no statistically significant difference between the prevalence of systemic features reported in individuals with biallelic LoF variants compared to individuals with at least one non-LoF variant (23/71, 32% versus 3/24, 13%, respectively, p = 0.10, chi-square test with continuity correction; Figure 3). Individual analysis of separate features (musculoskeletal and connective tissue, cardiovascular, facial dysmorphism, and development delay) did not show statistical significance between the two groups (Figure 3). Musculoskeletal and connective tissue features were otherwise the most reported features (15/95, 16%) [3,5,9,11,14,17]. Overall, there were no consistent phenotypes suggestive of an association with systemic disease. The presence of any feature was not dependent on whether an individual carried a European founder variant (Appendix 6).
Discussion
This case study series reports four previously unreported individuals with ADAMTSL4-associated ectopia lentis and provides a detailed review of the ocular and systemic phenotype in 95 individuals reported to date [3-5,9-19]. ADAMTSL4 is one of seven ADAMTSL genes that maintain the function of the extracellular matrix. ADAMTSL4 proteins are expressed in the anterior and posterior segment structures of the eye, including in the cornea (epithelium, stroma, and endothelium), iris stroma, trabecular meshwork, ciliary body stroma, ciliary processes, lens epithelium, choroid, sclera, and optic nerve [20,21]. Whether ADAMTSL4 is expressed in the neuroretina remains unclear due to conflicting results of immunohistochemical and immunofluorescence studies [20,21]. It has been hypothesized that ADAMTSL4 is a fibrillin 1-binding protein that facilitates the assembly of microfibrils. Although ADAMTSL4 has not yet been isolated in the zonules, impairment of this function may result in impaired zonular formation and maintenance [20,21]. ADAMTSL4 has been isolated in the ciliary processes [21], which form the attachment site of the zonules [22]. Similarly, pathogenic ADAMTSL4 variants have been demonstrated in a murine model to result in unstable anchorage of zonule fibers to the lens capsule [23]. This may explain the prominent phenotype of ectopia lentis.
The ocular phenotype of ADAMTSL4-associated ectopia lentis is highly variable. Although all the reported individuals manifested ectopia lentis, other anterior-segment anomalies have been reported with inconsistent frequency. It is unclear whether this inconsistency is a consequence of different variants, reporting bias, or age at the surgical intervention (such that early-onset cataract or lens opacity may be more prevalent). Through an investigation of genotype-phenotype correlations, our data suggested a possible association between biallelic LoF variants and additional anterior-segment features. Although the physiologic function of ADAMTSL4 is not yet fully understood, the absence of other anterior-segment features seen in individuals with at least one non-LoF variant (i.e., the missense variant) may be due to a compensatory interactive role between FBN1 [20], ADAMTS10 [24], and ADAMTS17 [25] in microfibril biogenesis. Variants in these genes have also been implicated in ectopia lentis and, more particularly, in WMS (FBN1, ADAMTS10, ADAMTS17, and LTBP2) and in Marfan syndrome (FBN1) [26]. These findings, however, may be confounded by insufficient reporting or examination of previously described individuals and the low number of individuals with at least one non-LoF allele (25%). This observation may be further confounded by an overrepresentation of individuals with the European founder variant and their ancestry.
Iris transillumination, iridodonesis, and poor pupillary dilatation [27,28] are common features of Marfan syndrome, which further suggests a possible interaction between shared gene products (FBN1 and ADAMTSL4). In contrast, deep ACD [16] and increased CCT [15,16] are not features of Marfan syndrome, and thus, may represent important differentiating features between the two conditions [28]. However, these findings have been observed in only a few individuals, including two individuals each in this study, and should be interpreted with caution. The measurement of CCT and ACD in consecutive studies is consequently encouraged. Systemic features typically seen in Marfan syndrome (e.g., aortic root dilatation) may be differentiating features of the two conditions, but there is insufficient evidence of this [29]. Persistent pupillary membrane, which has been described in association with ADAMTSL4-associated ectopia lentis, is accepted to be a common congenital anomaly, observed in approximately 20% of the normal adult population [30]. Given its low reported prevalence in ADAMTSL4-associated ectopia lentis (13%), it is unclear whether this is a disease feature or a population or surgical artifact.
The low frequency of ocular hypertension (OHT; 7%) in individuals who did not undergo lensectomy, and low frequency of pupillary block glaucoma (1%) overall, represent additional differentiating features of other connective tissue disorders. Individuals with WMS report a glaucoma frequency of 80% [31]. Similarly, individuals with LTBP2-associated ectopia lentis and microspherophakia frequently report the onset of pupillary block glaucoma before approximately 10 years of age [32,33]. These differences may be due to the larger interactive role between LTBP2 and FBN1 in zonular stability and the consequent greater severity of lens displacement [34,35]. We hypothesize that the low rate of pupillary block glaucoma may be explained by the direction of the lens subluxation. Lens subluxation in ADAMTSL4 is most often within the pupillary plane rather than anterior [9,11]. Similarly, in Marfan syndrome, glaucoma prevalence may be low, as lens subluxation is frequently reported to be in the pupillary plane [28]. Nonetheless, tonometry (IOP measurement) and fundoscopy are recommended in routine clinical examinations to exclude OHT or glaucoma.
Posterior-segment abnormalities seen in ADAMTSL4-associated ectopia lentis are relatively low, in keeping with unclear protein expression in the neuroretina [20,21]. This may be confounded, however, by the relatively young median age at the last follow-up (11 years). Most retinal detachments occurred post-lensectomy (with or without PPV) and were equated with a prevalence of retinal detachment post-lensectomy of 15%. This is comparable to a study of individuals with Marfan syndrome, which reported retinal detachment post-lensectomy in seven of 39 individuals (i.e., an 18% prevalence) [28]. Retinal detachment is a well-documented risk of surgery for ectopia lentis [36] but may occur when the axial lengths increase in response to the retinal blur caused by ectopia lentis [4]. Subsequently, the prevalence of retinal detachment in ADAMTSL4-associated ectopia lentis may be explained by various factors, including surgical techniques and the presence of axial myopia, rather than as a result of its protein expression. ADAMTSL4 protein has further not been located within the vitreous humor [20,21].
Aphakic glaucoma is another well-known risk of lensectomy in children, and its risk increases with every year post-surgery [37]. This case study series reports a novel case of aphakic glaucoma in ADAMTSL4-associated ectopia lentis, equating to a prevalence of 2% at 5 years. This is comparable to Marfan syndrome, wherein aphakic glaucoma has been reported at least 1-year post-lensectomy in one of 43 individuals (i.e., 2% prevalence) [38]. A lensectomy for pediatric cataract after age 9 months carries a 1.8% risk of developing glaucoma 5 years post-surgery and increases to 4.1% at 10 years [37]. A lensectomy for congenital cataract between ages 0 and 2 years has an estimated risk of OHT of 9% [39], which is consistent with the rate of OHT following lensectomy that was observed in this study (10%). Subsequently, individuals who undergo lensectomy for ADAMTSL4-associated ectopia lentis appear unlikely to be at an additional risk of aphakic glaucoma, particularly as ADAMTSL4 proteins have not been isolated within the aqueous humor drainage structures [20,21]. Nonetheless, these individuals require long-term monitoring and management of their condition, as the risk of aphakic glaucoma may increase with age.
At present, our findings support the hypothesis that ADAMTSL4-associated ectopia lentis is an isolated ocular condition [1], as its systemic features do not appear to fit a compelling phenotype. This could be due to the small number of individuals reported, or potentially, the lack of investigation or reporting of systemic features, or the presence of genetic variants in other genes that were not investigated. Learning difficulties and autism spectrum disorder are novel features reported in two individuals each in this series. However, these findings are unlikely to be related to ADAMTSL4-ectopia lentis, as they are among the top five most diagnosed conditions in general pediatric consultations across Australia [40]. The relative risks of musculoskeletal, connective tissue, and cardiovascular disorders appeared much lower than those of other syndromes associated with ectopia lentis, such as Marfan syndrome [29]. However, surveillance of systemic features is strongly recommended, as it remains unclear whether they are a consistent finding in ADAMTSL4-associated ectopia lentis. Further research is required in this area.
In conclusion, ADAMTSL4-associated ectopia lentis is most often an isolated ocular condition. Clinicians should be aware of its unique features and the complications of performing lensectomy. The ocular phenotype overlaps with that of other disorders, including Marfan syndrome, WMS, and LTBP2-associated ocular disease. However, the relatively normal systemic findings appear unique to the ADAMTSL4 spectrum, and may provide important differential diagnostic findings. Consistent and detailed reporting of ADAMTSL4-associated ectopia lentis is required to determine more accurate frequencies of its features and complications and to further elucidate the role of ADAMTSL4 proteins. These will guide the clinical and surgical management of the condition as well as genetic testing practices.
Acknowledgments
We thank Angela Chappell and Carly Emerson, at Flinders Medical Centre, Adelaide, Australia, for the clinical photographs used in this study (Figure 2). Funding support: This project was supported by the Australian National Health and Medical Research Council (NHMRC) Centres of Research Excellence Grant (APP1116360). OMS was supported by a Snow Fellowship, JEC was supported by an NHMRC Practitioner Fellowship and ES by an Early Career Fellowship from the Hospital Research Foundation. LSWK was funded by Flinders Health and Medical Research Institute and Flinders Foundation scholarships. The Centre for Eye Research Australia receives Operational Infrastructure Support from the Victorian Government. The sponsor or funding organization had no role in the design or conduct of this research. Prior presentation of manuscript data: Presented at the Royal Australian and New Zealand College of Ophthalmologists (RANZCO) 52nd Annual Scientific Congress 2022
Appendix 1. Flow diagram of the literature review of ADAMTSL4-associated ectopia lentis.
To access the data, click or select the words “Appendix 1.”
Appendix 2. The characteristics and frequency of the variants reported in 95 individuals with ADAMTSL4-associated ectopia lentis.
To access the data, click or select the words “Appendix 2.”
Appendix 3. The frequency of combinations of variants in 95 individuals with ADAMTSL4-associated ectopia lentis.
To access the data, click or select the words “Appendix 3.”
Appendix 4. Self-reported ancestry in 95 individuals with ADAMTSL4-associated ectopia lentis.
To access the data, click or select the words “Appendix 4.”
Appendix 5. Systemic features reported amongst 95 individuals with ADAMTSL4-associated ectopia lentis.
To access the data, click or select the words “Appendix 5.”
Appendix 6. A comparison of the presence of ocular or systemic features between individuals with at least one European founder variant (c.767_786del) and individuals who did not have the European founder variant.
To access the data, click or select the words “Appendix 6.”
References
- 1.Rødahl E, Mellgren AEC, Boonstra NE, Knappskog PM. ADAMTSL4-Related Eye Disorders. GeneReviews. 2012. [Google Scholar]
- 2.Cruysberg JR, Pinckers A. Ectopia lentis et pupillae syndrome in three generations. Br J Ophthalmol. 1995;79:135–8. doi: 10.1136/bjo.79.2.135. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Neuhann TM, Stegerer A, Riess A, Blair E, Martin T, Wieser S, Kläs R, Bouman A, Kuechler A, Rittinger O. ADAMTSL4-associated isolated ectopia lentis: Further patients, novel mutations and a detailed phenotype description. Am J Med Genet A. 2015;167A:2376–81. doi: 10.1002/ajmg.a.37157. [DOI] [PubMed] [Google Scholar]
- 4.Sharifi Y, Tjon-Fo-Sang MJ, Cruysberg JRM, Johannes RM, Matt-Kievit AJ. Ectopia lentis et pupillae in four generations caused by novel mutations in the ADAMTSL4 gene. Br J Ophthalmol. 2013;97:583–7. doi: 10.1136/bjophthalmol-2012-302367. [DOI] [PubMed] [Google Scholar]
- 5.Overwater E, Floor K, van Beek D, de Boer K, van Dijk T, Hilhorst-Hofstee Y, Hoogeboom AJM, van Kaam KJ, van de Kamp JM, Kempers M, Krapels IPC, Kroes HY, Loeys B, Salemink S, Stumpel CTRM, Verhoeven VJM, Wijnands-van den Berg E, Cobben JM, van Tintelen JP, Weiss MM, Houweling AC, Maugeri A. NGS panel analysis in 24 ectopia lentis patients; a clinically relevant test with a high diagnostic yield. Eur J Med Genet. 2017;60:465–73. doi: 10.1016/j.ejmg.2017.06.005. [DOI] [PubMed] [Google Scholar]
- 6.Souzeau E, Goldberg I, Healey PR, Mills RA, Landers J, Graham SL, Grigg JR, Usher B, Straga T, Crawford A, Casson RJ, Morgan WH, Ruddle JB, Coote MA, White A, Stewart J, Hewitt AW, Mackey DA, Burdon KP, Craig JE. Australian and New Zealand Registry of Advanced Glaucoma: methodology and recruitment. Clin Experiment Ophthalmol. 2012;40:569–75. doi: 10.1111/j.1442-9071.2011.02742.x. [DOI] [PubMed] [Google Scholar]
- 7.Jonas JB, Dong L, Da Chen S, Neumaier M, Findeisen P, Panda-Jonas S, Jonas RA. Intraocular epidermal growth factor concentration, axial length, and high axial myopia. Graefes Arch Clin Exp Ophthalmol. 2021;259:3229–34. doi: 10.1007/s00417-021-05200-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Karczewski KJ, Francioli LC, Tiao G, Cummings BB, Alföldi J, Wang Q, Collins RL, Laricchia KM, Ganna A, Birnbaum DP, Gauthier LD, Brand H, Solomonson M, Watts NA, Rhodes D, Singer-Berk M, England EM, Seaby EG, Kosmicki JA, Walters RK, Tashman K, Farjoun Y, Banks E, Poterba T, Wang A, Seed C, Whiffin N, Chong JX, Samocha KE, Pierce-Hoffman E, Zappala Z, O’Donnel-Luria AH, Minikel EV, Weisburd B, Lek M, Ware JS, Vittal C, Armean IM, Bergelson L, Cibulskis K, Connolly KM, Covarrubias M, Donnelly S, Ferriera S, Gabriel S, Gentry J, Gupta N, Jeandet T, Kaplan D, Llanwarne C, Munshi R, Novod S, Petrillo N, Roazen D, Ruano-Rubio V, Saltzman A, Schleicher M, Soto J, Tibbetts K, Tolonen C, Wade G, Talkowski ME, Genome Aggregation Database Consortium. Neale BM, Daly MJ, MacArthur DG. The mutational constraint spectrum quantified from variation in 141,456 humans. Nature. 2020;581:434–43. doi: 10.1038/s41586-020-2308-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Neuhann TM, Artelt J, Neuhann TF, Tinschert S, Rump A. A homozygous microdeletion within ADAMTSL4 in patients with isolated ectopia lentis: evidence of a founder mutation. Invest Ophthalmol Vis Sci. 2011;52:695–700. doi: 10.1167/iovs.10-5740. [DOI] [PubMed] [Google Scholar]
- 10.Safi M, Nejad SK, O’Hara M, Shankar SP. Ectopia lentis et pupillae caused by ADAMTSL4 pathogenic variants and an algorithm for work-up. J Pediatr Ophthalmol Strabismus. 2019;56:e45–8. doi: 10.3928/01913913-20190509-01. [DOI] [PubMed] [Google Scholar]
- 11.van Bysterveldt KA, Al Taie R, Ikink W, Oliver VF, Vincent AL. ADAMTSL4 assessment in ectopia lentis reveals a recurrent founder mutation in Polynesians. Ophthalmic Genet. 2017;38:537–43. doi: 10.1080/13816810.2017.1309552. [DOI] [PubMed] [Google Scholar]
- 12.Reinstein E, Smirin-Yosef P, Lagovsky I, Davidov B, Peretz Amit G, Nuemann D, Orr-Urtreger A, Ben-Shachar S, Basel-Vanagaite L. A founder mutation in ADAMTSL4 causes early-onset bilateral ectopia lentis among Jews of Bukharian origin. Mol Genet Metab. 2016;117:38–41. doi: 10.1016/j.ymgme.2015.11.011. [DOI] [PubMed] [Google Scholar]
- 13.Zhou XM, Wang Y, Zhao L, Yu HW, Fan N, Yan NH, Su Q, Liang YQ, Wang Y, Li LP, Cai SP, Jonas JB, Liu XY. Novel compound heterozygous mutations identified in ADAMTSL4 gene in a Chinese family with isolated ectopia lentis. Acta Ophthalmol. 2015;93:e91–2. doi: 10.1111/aos.12399. [DOI] [PubMed] [Google Scholar]
- 14.Chandra A, Aragon-Martin JA, Sharif S, Parulekar M, Child A, Arno G. Craniosynostosis with ectopia lentis and a homozygous 20-base deletion in ADAMTSL4. Ophthalmic Genet. 2013;34:78–82. doi: 10.3109/13816810.2012.710707. [DOI] [PubMed] [Google Scholar]
- 15.Chandra A, Aragon-Martin JA, Hughes K, Gati S, Reddy MA, Deshpande C, Cormack G, Child AH, Charteris DG, Arno G. A genotype-phenotype comparison of ADAMTSL4 and FBN1 in isolated ectopia lentis. Invest Ophthalmol Vis Sci. 2012;53:4889–96. doi: 10.1167/iovs.12-9874. [DOI] [PubMed] [Google Scholar]
- 16.Christensen AE, Fiskerstrand T, Knappskog PM, Boman H, Rødahl E. A novel ADAMTSL4 mutation in autosomal recessive ectopia lentis et pupillae. Invest Ophthalmol Vis Sci. 2010;51:6369–73. doi: 10.1167/iovs.10-5597. [DOI] [PubMed] [Google Scholar]
- 17.Aragon-Martin JA, Ahnood D, Charteris DG, Saggar A, Nischal KK, Comeglio P, Chandra A, Child AH, Arno G. Role of ADAMTSL4 mutations in FBN1 mutation-negative ectopia lentis patients. Hum Mutat. 2010;31:E1622–31. doi: 10.1002/humu.21305. [DOI] [PubMed] [Google Scholar]
- 18.Greene VB, Stoetzel C, Pelletier V, Perdomo-Trujillo Y, Liebermann L, Marion V, De Korvin H, Boileau C, Dufier JL, Dollfus H. Confirmation of ADAMTSL4 mutations for autosomal recessive isolated bilateral ectopia lentis. Ophthalmic Genet. 2010;31:47–51. doi: 10.3109/13816810903567604. [DOI] [PubMed] [Google Scholar]
- 19.Ahram D, Sato TS, Kohilan A, Tayeh M, Chen S, Leal S, Al-Salem M, El-Shanti H. A homozygous mutation in ADAMTSL4 causes autosomal-recessive isolated ectopia lentis. Am J Hum Genet. 2009;84:274–8. doi: 10.1016/j.ajhg.2009.01.007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Gabriel LAR, Wang LW, Bader H, Ho JC, Majors AK, Hollyfield JG, Traboulsi EI, Apte SS. ADAMTSL4, a secreted glycoprotein widely distributed in the eye, binds fibrillin-1 microfibrils and accelerates microfibril biogenesis. Invest Ophthalmol Vis Sci. 2012;53:461–9. doi: 10.1167/iovs.10-5955. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Chandra A, Jones M, Cottrill P, Eastlake K, Limb GA, Charteris DG. Gene expression and protein distribution of ADAMTSL-4 in human iris, choroid and retina. Br J Ophthalmol. 2013;97:1208–12. doi: 10.1136/bjophthalmol-2013-303353. [DOI] [PubMed] [Google Scholar]
- 22.Tamm ER, Lȕtjen-Drecoll E. Ciliary body. Microsc Res Tech. 1996;33:390–439. doi: 10.1002/(SICI)1097-0029(19960401)33:5<390::AID-JEMT2>3.0.CO;2-S. [DOI] [PubMed] [Google Scholar]
- 23.Collin GB, Hubmacher D, Charette JR, Hicks WL, Stone L, Yu Minzhong Y, Naggert JK, Krebs MP, Peachey NS, Apte SS, Nishina PM. Disruption of murine ADAMTSL4 results in zonular fiber detachment from the lens and in retinal pigment epithelium dedifferentiation. Hum Mol Genet. 2015;24:6958–74. doi: 10.1093/hmg/ddv399. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Kutz WE, Wang LW, Bader HL, Majors AK, Iwata K, Traboulsi EI, Sakai LY, Keene DR, Apte SS. ADAMTS10 protein interacts with fibrillin-1 and promotes its deposition in extracellular matrix of cultured fibroblasts. J Biol Chem. 2011;286:17156–67. doi: 10.1074/jbc.M111.231571. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Farias FHG, Johnson GS, Taylor JF, Giuliano E, Katz ML, Sanders DN, Schnabel RD, McKay SD, Khan S, Gharahkhani P, O’Leary CA, Pettitt L, Forman OP, Boursnell M, McLaughlin B, Ahonen S, Lohi H, Hernandez-Merino E, Gould DJ, Sargan DR, Mellersch C. An ADAMTS17 splice donor site mutation in dogs with primary lens luxation. Invest Ophthalmol Vis Sci. 2010;51:4716–21. doi: 10.1167/iovs.09-5142. [DOI] [PubMed] [Google Scholar]
- 26.Hubmacher D, Apte SS. ADAMTS proteins as modulators of microfibril formation and function. Matrix Biol. 2015;47:34–43. doi: 10.1016/j.matbio.2015.05.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Shah SS, Kurup SP, Ralay Ranaivo H, Mets-Halgrimson RB, Mets MB. Pupillary manifestations of Marfan syndrome: from the Marfan eye consortium of Chicago. Ophthalmic Genet. 2018;39:297–9. doi: 10.1080/13816810.2018.1424207. [DOI] [PubMed] [Google Scholar]
- 28.Gehle P, Goergen B, Pilger D, Ruokonen P, Robinson PN, Salchow DJ. Biometric and structural ocular manifestations of Marfan syndrome. PLoS One. 2017;12:e0183370. doi: 10.1371/journal.pone.0183370. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Loeys BL, Dietz HC, Braverman AC, Callewaert BL, De Backer J, Devereux RB, Hilhorst-Hofstee Y, Jondeau G, Faivre L, Milewicz DM, Pyeritz RE, Sponseller PD, Wordsworth P, De Paepe AM. The revised Ghent nosology for the Marfan syndrome. J Med Genet. 2010;47:476–85. doi: 10.1136/jmg.2009.072785. [DOI] [PubMed] [Google Scholar]
- 30.Kleiman NJ, Worgul BV. Chapter 15: Lens. In: Tasman W, Jaeger EA, eds. Duane’s foundations of clinical ophthalmology volume 1: Ocular anatomy, embryology and teratology. Philadelphia: Lippincott Williams & Wilkins 2007. [Google Scholar]
- 31.Faivre L, Dollfus H, Lyonnet S, Alembik Y, Mégarbané A, Samples J, Gorlin RJ, Alswaid A, Feingold J, Le Merrer M, Munnich A, Cormier-Daire V. Clinical homogeneity and genetic heterogeneity in Weill-Marchesani syndrome. Am J Med Genet A. 2003;123A:204–7. doi: 10.1002/ajmg.a.20289. [DOI] [PubMed] [Google Scholar]
- 32.Désir J, Sznajer Y, Depasse F, Roulez F, Schrooyen M, Meire F, Abramowicz M. LTBP2 null mutations in an autosomal recessive ocular syndrome with megalocornea, spherophakia, and secondary glaucoma. Eur J Hum Genet. 2010;18:761–7. doi: 10.1038/ejhg.2010.11. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Micheal S, Siddiqui SN, Zafar SN, Iqbal A, Khan MI, den Hollander AI. Identification of novel variants in LTBP2 and PXDN using whole-exome sequencing in developmental and congenital glaucoma. PLoS One. 2016;11:e0159259. doi: 10.1371/journal.pone.0159259. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Inoue T, Ohbayashi T, Fujikawa Y, Yoshida H, Akama TO, Noda K, Horiguchi M, Kameyama K, Hata Y, Takahashi K, Kusumoto K, Nakamura T. Latent TGF-β binding protein-2 is essential for the development of ciliary zonule microfibrils. Hum Mol Genet. 2014;23:5672–82. doi: 10.1093/hmg/ddu283. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Siggs OM, Souzeau E, Craig JE. Loss of ciliary zonule protein hydroxylation and lens stability as a predicted consequence of biallelic ASPH variation. Ophthalmic Genet. 2019;40:12–6. doi: 10.1080/13816810.2018.1561904. [DOI] [PubMed] [Google Scholar]
- 36.Miraldi Utz V, Coussa RG, Traboulsi EI. Surgical management of lens subluxation in Marfan syndrome. J AAPOS. 2014;18:140–6. doi: 10.1016/j.jaapos.2013.12.007. [DOI] [PubMed] [Google Scholar]
- 37.Haargaard B, Ritz C, Oudin A, Wohlfahrt J, Thygesen J, Olsen T, Melbye M. Risk of glaucoma after pediatric cataract surgery. Invest Ophthalmol Vis Sci. 2008;49:1791–6. doi: 10.1167/iovs.07-1156. [DOI] [PubMed] [Google Scholar]
- 38.Sen P, Attiku Y, Bhende P, Rishi E, Ratra D, Sreelakshmi K. Outcome of sutured scleral fixated intraocular lens in Marfan syndrome in pediatric eyes. Int Ophthalmol. 2020;40:1531–8. doi: 10.1007/s10792-020-01322-7. [DOI] [PubMed] [Google Scholar]
- 39.Eibenberger K, Kiss B, Schmidt-Erfurth U, Stifter E. Clinical characteristics and treatment of secondary glaucoma, glaucoma suspects and ocular hypertension after congenital cataract surgery. Eur J Ophthalmol. 2021;31:3309–17. doi: 10.1177/1120672121991356. [DOI] [PubMed] [Google Scholar]
- 40.Hiscock H, Roberts G, Efron D, Sewell JR, Bryson HE, Price AMH, Obserklaid F, South M, Wake MA. Children Attending Paediatricians Study: a national prospective audit of outpatient practice from the Australian Paediatric Research Network. Med J Aust. 2011;194:392–7. doi: 10.5694/j.1326-5377.2011.tb03028.x. [DOI] [PubMed] [Google Scholar]
- 41.Richards S, Aziz N, Bale S, Bick D, Das S, Gastier-Foster J, Grody WW, Hegde M, Lyon E, Spector E, Voelkerding K, Rehm HL, ACMG Laboratory Quality Assurance Committee Standards and guidelines for the interpretation of sequence variants: a joint consensus recommendation of the American College of Medical Genetics and Genomics and the Association for Molecular Pathology. Genet Med. 2015;17:405–24. doi: 10.1038/gim.2015.30. [DOI] [PMC free article] [PubMed] [Google Scholar]