To the Editor:
Hospitalizations to treat patients infected with COVID-19 have strained health systems worldwide. Monitored outpatient treatment of select low-risk patients with COVID-19 could help preserve hospital resources and reduce costs while aiming to maintain a high standard of care. However, criteria to identify patients appropriate for outpatient monitoring do not exist. Isolated exertional hypoxia may be an early predictor of moderate-to-severe COVID-19 and could be an indication for remote monitoring or inpatient admission; however, data to support its use are limited.1, 2, 3 Our institution implemented a standardized ED triage process that included exertional hypoxia and the COVID-19-associated hyperinflammation (cHIS) score.4 The cHIS scoring includes fever, degree of cytopenia, hyperferritinemia, D-dimer, markers of hepatic injury (lactate dehydrogenase or aspartate aminotransferase), and markers of cytokinemia (c-reactive protein, IL-6, or hypertriglyceridemia) and is associated with progression to mechanical ventilation and death. This triage process identified patients who were considered to be at an acceptable morbidity and mortality risk for discharge to home on low-flow home oxygen with remote SpO2 and vital sign monitoring. In this retrospective cohort analysis, we report the safety and effectiveness of the COVID-19 remote patient monitoring with home oxygen (cRPM-O2) program as a strategy to reduce hospitalization days and preserve resource utilization.
Methods
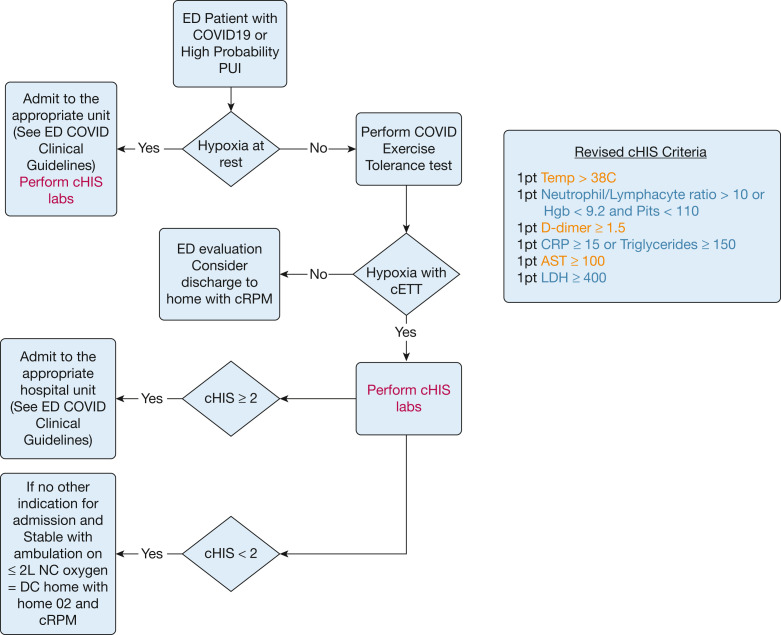
The cRPM-O2 algorithm was implemented across 23 of Intermountain Healthcare’s integrated hospitals from November 2020 to April 2021, during the COVID-19 alpha variant surge. Included patients had an ED encounter within 14 days after a confirmed COVID-19 positive test, received a COVID exercise tolerance test (cETT) to assess exertional hypoxia per protocol, and had a cHIS < 2 by laboratory results that were obtained during the index ED encounter. cETT consisted of either briskly walking for 1 min or sit-to-stand for 1 min. Any SpO2 result < 90% or severe dyspnea that prevented completion of the test was considered a positive cETT. The clinical criteria for cRPM-O2 inclusion and triage process are presented in Figure 1 . Patients who met the criteria for cRPM-O2 participation from the ED, but with an inpatient admission, constituted the control arm. The primary outcome was inpatient hospital days associated with the first admission within 14 days after the index ED encounter modeled by ordinary least squares regression (covariates are listed in Table 1 ). Secondary outcomes were ICU length of stay and combined ICU admission or all-cause death within 28 days. ICU length of stay was modeled in the same fashion as the primary outcome. Death was identified by state death database review to ascertain any death that occurs outside of our system. The composite binary outcome of ICU admission or death was modeled with the use of a logistic regression. Patient COVID-19 vaccination status was not available. The Intermountain Healthcare Institutional Review Board approved the analysis.
Figure 1.
COVID-19-Associated Hyperinflammation Score Assessment Flow Diagram. AST = aspartate aminotransferase; cETT = COVID exercise tolerance test; cHIS = COVID-19-associated hyperinflammation score; cRPM = COVID remote patient monitoring; Hgb = hemoglobin; LDH = lactate dehydrogenase.
Table 1.
Baseline Characteristics of Patients and Outcomes
| Model Input and Patient Characteristicsa | ED Admission to Floor (n = 522) | COVID Remote Patient Monitoring With Home Oxygen (n = 233) | P value |
|---|---|---|---|
| Age, mean (SD), y | 61.5 (47.5-71.5) | 55.1 (43.6-64.9) | < .001 |
| Sex, No. (%) | .82 | ||
| Male | 251 (48.1) | 110 (47.2) | |
| Female | 271 (51.9) | 123 (52.8) | |
| Insurance, No. (%) | < .001 | ||
| Private or commercial | 253 (48.5) | 167 (71.7) | |
| Medicaid | 46 (8.8) | 11 (4.7) | |
| Medicare | 204 (39.1) | 45 (19.3) | |
| Other | 19 (3.6) | 10 (4.3) | |
| Collapsed race/ethnicity, No. (%) | .28 | ||
| White, not Hispanic | 370 (70.9) | 174 (74.7) | |
| Hispanic or other | 152 (29.1) | 59 (25.3) | |
| Charlson Comorbidity Index | 1.6 (2.7) | 0.8 (2.0) | < .001 |
| Lowest oxygen saturation during ED visit | 87.6 (4.4) | 87.3 (3.2) | .38 |
| Unadjusted patient outcomesb | |||
| Inpatient hospital days, No. (%) | 4.0 (3.2) | 1.5 (4.7) | < .001 |
| ICU length of stay, d (%) | 0.2 (1.8) | 0.5 (2.1) | .079 |
| ICU level care or death, No. (%) | 22 (4.2) | 15 (6.4) | .19 |
| 28-Day all-cause deaths, No. (%) | 5 (1.0) | 2 (0.9) | .90 |
| cRPM-O2 adjusted outcomesc | Coefficient/OR | 95% CI | |
| Hospital days (coefficient) | −2.27 | −3.01 to −1.53 | < .001 |
| < 70 Years old | −2.57 | −3.30 to −1.84 | < .001 |
| ≥ 70 Years old | −0.05 | −3.44 to 2.44 | .74 |
| ICU days (coefficient) | 0.33 | 0.00 to 0.65 | .048 |
| < 70 Years old | 0.18 | −0.16 to 0.53 | .30 |
| ≥ 70 Years old | 1.05 | 0.07 to 2.031 | .04 |
| ICU admission of death (OR) | 1.93 | 0.94 to 3.95 | .074 |
| < 70 Years old | 1.40 | 0.63 to 3.14 | .41 |
| ≥ 70 Years old | 5.44 | 0.93 to 31.75 | .06 |
Covariates adjusted for in our model for all 3 outcomes.
Unadjusted outcome measures.
Adjusted outcomes (ordinary least squares and logistic regression).
Patients who enrolled in cRPM-O2 received a Bluetooth-enabled pulse oximeter paired to their smartphone via an application downloaded and confirmed by the ED nurse or technician prior to ED discharge. Patients were asked to record SpO2/heart rate biometrics at least twice daily, or as often as they preferred, for 14 days. Submitted biometric data from enrolled patients were monitored 24 hours per day, 7 days per week. Patients without transmitted vital signs for > 18 hours or with SpO2 < 88% and/or pulse rate > 120 beats/min were contacted by a monitoring technician. If new hypoxemia was identified despite supplemental oxygen, the patient was instructed to return to the ED. Patients with concerns about their symptoms or the device could contact a monitoring technician via the application at any point, and concerns could be escalated to the monitoring center's critical care nurse or nurse practitioner. The application included built-in text and video visit functionality to facilitate remote patient assessment. The pulse oximeter device, application, and monitoring service were offered to patients without out-of-pocket charges.
Results
Of the 755 patients identified in the study cohort, 522 patients comprised the control group, and 233 patients were in the cRPM-O2 group. The cRPM-O2 group was younger and had fewer comorbidities (Table 1). After adjustment for age, sex, ethnicity/race identifiers, insurance provider, Charlson Comorbidity Index, and lowest recorded oxygen saturation during index ED visit, cRPM-O2 participation was associated with a reduction of 2.27 hospital days per patient (95% CI, 1.53 to 3.01; P < .001). Of the 233 patients who received cRPM-O2, 39 patients (16.7%) subsequently were admitted with a median of 4 days (interquartile range, 2 to 6) from ED visit to admission. cRPM-O2 participation was associated with a numeric increase in combined ICU admission and deaths (OR, 1.93; P = .07; 95% CI, 0.94 to 3.95); ICU days were significantly increased in cRPM-O2 participants (0.33 days; 95% CI, 0.00 to 0.65; P = .05). In the preplanned subgroup analysis that was limited to patients ≥ 70 years old, hospital length of stay did not differ with cRPM-O2 participation (−0.50 days; 95% CI, −3.44 to 2.44; P = .74) and participation in cRPM-O2 was associated with increased ICU length of stay (1.05 days; 95% CI, 0.07 to 2.0; P = .04) and a numerically increased odds of ICU admission or death (OR, 5.44; 95% CI, 0.93 to 31.75; P = .06). In this subgroup, there were four deaths in the control group and no deaths in the cRPM-O2 group. In contrast, cRPM-O2 for patients < 70 years of age was associated with hospital length of stay reduction of 2.56 days (95% CI, 1.84 to 3.30; P < .001), no change in ICU length of stay (+0.18 days; 95% CI, −0.16 to 0.53; P = .296), and no difference in the odds of combined ICU admission or death (OR, 1.40; 95% CI, 0.63 to 3.14; P = .410).
Discussion
This ED program to triage low-risk patients with COVID-19 with exertional hypoxia to treatment at home with remote monitoring and low-level supplemental oxygen was associated with fewer hospital days but with an increase in ICU days and trend toward an increase in the composite outcome of death or ICU for patients triaged to home with cRPM-O2, driven by events that occurred among patients ≥ 70 years old. In total, 83.3% of patients with cRPM-O2 were treated safely without hospitalization; however, only 67% in those patients aged ≥ 70 years. Inclusion in cRPM-O2 significantly increased ICU days, but by a clinically insignificant 0.33 days in our main analysis. When assessing only those patients admitted to the ICU in each group, we observed the adjusted ICU length of stay is not statistically different, which suggests that trends toward increased risk of ICU admission between groups accounts for some of the increased ICU length of stay finding. When assessing only patients who were admitted from the cRPM-O2, we found that patients who required hospitalization are more likely to be older and male and to have more comorbidities than those patients who remain at home. Inpatient interventions such as Remdesivir and steroids are considered confounders that may have influenced the differences in outcomes between groups in addition to expected confounders of a retrospective analysis. Additionally, this implementation from a single integrated health care system may not be generalizable externally.
ED triage of select patients with COVID-19 with isolated exertional hypoxia to home with low-flow supplemental oxygen plus remote monitoring is associated with fewer hospital days, and a net clinical benefit especially may be realized among patients < 70 years old. Appropriate risk assessment and treatment with cRPM-O2 may be a strategy to improve hospital capacity safely. Future study of subgroups is warranted to ensure the safety of this approach.
Financial/Nonfinancial Disclosures
The authors have reported to CHEST the following: S. C. W. reports service as co-chair of the American College of Chest Physicians Clinical Practice Guideline: Antithrombotic therapy for venous thromboembolic disease. H. L. C. reports that the Sean N. Parker Center for Allergy & Asthma at Stanford University sponsored a portion of his research time for COVID-19 specific research. I.D.P. reports grant support from the National Institutes of Health, Centers for Disease Control and Prevention, and Janssen Pharmaceuticals and support to his institution from Regeneron and Asahi Kasei Pharma. None declared (K. P., A. D., J. D., J. R. J., R. R., M. S., B. W., J. R. B.).
Acknowledgments
Author contributions: J. R. B. and H. L. C. are the guarantors of all content of this article. The authors participated in the following elements of the article: acquisition of data: H. L. C., I. D. P., J. D., J. R. J.; analysis of data: H. L. C., I. D. P.; interpretation of data: H. L. C., I. D. P., J. R. B.; drafting of manuscript: H. L. C., I. D. P., J. R. B., S. C. W.; critical revision of the manuscript: all authors; obtaining funding: H. L. C., I. D. P.
Other contributions: The authors thank David Hasleton, MD, and the Intermountain Healthcare Emergency Department Executive team, Intermountain Healthcare Emergency Medicine, and Mike Lyons, BS, and Lory Maddox, PhD, RN, of Intermountain Healthcare for assistance in data acquisition.
References
- 1.Bhasin A., Bregger M., Kluk M., Park P., Feinglass J., Barsuk J. Exertional hypoxia in patients without resting hypoxia is an early predictor of moderate to severe COVID-19. Intern Emerg. 2021;16:2097–2103. doi: 10.1007/s11739-021-02708-w. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Kalin A., Javid B., Knight M., Inada-Kim M., Greenhalgh T. Direct and indirect evidence of efficacy and safety of rapid exercise tests for exertional desaturation in Covid-19: a rapid systematic review. Syst Rev. 2021;10(1):77. doi: 10.1186/s13643-021-01620-w. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Hussain T., Saman H.T., Yousaf Z. Identification of exertional hypoxia and its implications in SARS-CoV-2 pneumonia. Am J Trop Med Hyg. 2020;103(4):1742–1743. doi: 10.4269/ajtmh.20-1012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Webb B.J., Peltan I.D., Jensen P., et al. Clinical criteria for COVID-19-associated hyperinflammatory syndrome: a cohort study. Lancet Rheumatol. 2020;2(12):e754–e763. doi: 10.1016/S2665-9913(20)30343-X. [DOI] [PMC free article] [PubMed] [Google Scholar]