Abstract
Objective
The consequences of blood lipid abnormalities for cardiovascular disease risk in young adults is unclear. Optimal lipid levels may also vary depending on whether a statin drug is taken. It aimed to determine whether the optimal lipid levels in young adults differ depending on statin use.
Methods
Using a nationally representative database from the Korean National Health Insurance System, 6,350,400 participants aged 20–39 years who underwent a health examination between 2009–2012 were followed through to 2018. The primary outcome was incident myocardial infarction (MI). We assessed the associations between prespecified lipid levels and MI risk according to statin use.
Results
Among participants not taking statins, low-density lipoprotein cholesterol (LDL-C) levels ≥120 mg/dL were significantly associated with MI risk (hazard ratio [HR], 1.33; 95% confidence interval [CI], 1.27–1.40) compared with statin nonusers with LDL-C <80 mg/dL. Statin users with LDL-C categories <80, 80–100, 100–120, and ≥120 mg/dL all had significantly higher MI risk compared with statin nonusers with LDL-C <80 mg/dL; these HRs (95% CIs) were 1.66 (1.39–1.99), 1.68 (1.36–2.09), 1.63 (1.31–2.02), and 2.32 (2.07–2.60), respectively.
Conclusion
Young adults taking statins have an increased MI risk compared with statin nonusers, even when they have similar LDL-C levels. Specific lipid targets may need to differ depending on statin use.
Keywords: Statin, Cholesterol, Cardiovascular disease, Myocardial infarction
INTRODUCTION
Abnormal blood lipid levels are a major cause of coronary heart disease in middle-aged and older adults.1,2 A higher than expected proportion of young adults also have nonoptimal levels of low-density lipoprotein cholesterol (LDL-C ≥100 mg/dL), high-density lipoprotein cholesterol (HDL-C <60 mg/dL), or triglyceride (TG ≥150 mg/dL).1 Nonoptimal LDL-C and HDL-C levels during young adulthood are independently associated with coronary atherosclerosis 20 years later.1 However, there are insufficient guidelines for initiating statin therapy in adults younger than age 40 years.3,4,5,6 It is also uncertain whether long-term statin treatment leads to better outcomes, in contrast with postponing treatment until cardiovascular (CV) risk rises to a level at which treatment becomes warranted.3,4,5,6
Current guidelines suggest that an LDL-C goal of 100 mg/dL is warranted in patients with type 2 diabetes mellitus (DM), and 70 mg/dL for patients with atherosclerotic cardiovascular disease (CVD), regardless of whether either group are taking statins.5 We previously reported increased CVD risk in patients with type 2 DM and an LDL-C level ≥130 mg/dL who were not taking statins. We also found that CVD risk was significantly higher in those taking statins who had an LDL-C level ≥70 mg/dL.7 Thus, optimal cholesterol levels may differ depending on whether a statin drug is used. National Institute for Health and Care Excellence (NICE) guidelines aim to reduce baseline LDL-C by >40%, rather than to specific target lipid levels. When >40% reduction in baseline LDL-C within 24 months was defined as the optimal statin response, successful LDL-C reduction was not achieved within two years by over half of the general population patients who initiated statin therapy; these patients had significantly increased CVD risk.8
Thus, using a large, population-based cohort of young adults, we assessed the associations between prespecified lipid levels and myocardial infarction (MI) risk, according to statin use or nonuse. We also sought to determine whether the optimal lipid levels in young adults differ depending on statin use.
MATERIALS AND METHODS
1. Study participants
The Korean National Health Insurance Service (NHIS) 2009–2018 database was used for analyses. This single insurer provides comprehensive universal medical coverage for the entire Korean population (\xe2\x89\x8d50 million individuals). Available data include sociodemographic measures, medical expenses, diagnoses (based on the International Classification of Disease, Tenth Revision of Clinical Modification [ICD-10]), use of inpatient and outpatient services, pharmacy dispensing claims, and mortality.9,10 All enrolled participants (ages ≥20 years) are required to undergo annual or biannual health examinations. This health checkup includes a physical examination, routine blood tests, chest radiograph, and medical history self-report; all measures are included in the NHIS database. All data were anonymized.
The study flowchart is illustrated in Fig. 1. Adults aged 20–39 years who underwent routine NHIS health checkups from 2009–2012 were selected for analyses. Among these, subjects who had a history of MI (ICD-10 codes: I21, I22), stroke (ICD-10 codes: I63, I64) before 2009 were excluded (n=43,140). We also excluded 486,899 who had data missing for one or more variables. All participants with the occurrence of an endpoint prior to the one-year follow-up were also excluded from analyses. Finally, 6,350,400 participants were included in analyses. The study was approved by the Institutional Review Board of Yeouido St. Mary’s Hospital, The Catholic University of Korea (No. SC20ZISI0034).
Fig. 1. Flow chart of the study population.
MI, myocardial infarction.
2. Measurements and definitions
Statin users were defined based on a history of statin use during the year of the index date. We did not include other classes of hyperlipidemic drugs such as ezetimibe, fibrate or omega-3 fatty acid. Covariates, based on index year data, included age, sex, socioeconomic status (income level), body mass index (BMI; kg/m2), current smoking status, alcohol consumption, physical activity (no, yes), and systolic/diastolic blood pressure (mmHg). Blood samples for the measurement of lipid profiles and glucose were drawn after an overnight fast. Lipid profiles, including the levels of total cholesterol (TC), HDL-C, and TG, were measured using an enzymatic method. LDL-C levels were calculated using the Friedewald formula11:
| LDL-C = TC − HDL-C − (TG/5). |
We excluded those with a TG level >400 mg/dL in LDL-C analysis. Laboratory test quality controls were conducted in accordance with the procedures of the Korean Association of Laboratory Quality Control.
3. Study outcomes and follow-up
The study endpoint was MI (defined as newly recorded ICD-10 code I21 or I22 during hospitalization) and only the first events of MI were included in this study. Participants without MI by follow-up (December 31, 2018) were considered to have completed the study on the date of their death or the end of follow-up, whichever occurred first.
4. Statistical analysis
Baseline characteristics are presented as the mean ± standard deviation (SD), median (interquartile range) or number (percentage). The primary outcome incidence rate was calculated by dividing the number of incident cases by the total follow-up duration (person years). The overall sample was divided into statin users or nonusers. Participants in each statin use group were then subdivided into categories based on each lipid component, and cutoffs were used for all analyses. The Cox proportional-hazards model was used to estimate hazard ratios (HR) and 95% confidence interval (CI) values for study outcomes according to the categories of lipid parameters: TC <160 mg/dL was the reference group, with 20 mg/dL intervals; TG <60 mg/dL was the reference group, with 30 mg/dL intervals; LDL-C <80 mg/dL was the reference group, with 20 mg/dL intervals; HDL-C <45 mg/dL was the reference group, with 10 mg/dL intervals. Multivariate adjusted proportional hazards models were used. Model 1 was adjusted for age and sex. Model 2 was additionally adjusted for BMI, income level, smoking, alcohol intake, physical activity, DM, hypertension and chronic kidney disease (defined as estimated glomerular filtration rate <60 mL/min/1.73 m2). Potential modifying effects of age, sex, and hypertension were evaluated by stratified analyses, and interactions evaluated by a likelihood ratio test. Subgroup analyses were also conducted based on age group (20–29 or 30–39 years), sex, and hypertension. Statistical analyses were performed using SAS version 9.4 (SAS Institute Inc., Cary, NC, USA), and p<0.05 was considered statistically significant.
RESULTS
1. Baseline characteristics
The sample included 74,876 (1.2%) statin users. Individuals taking statins were older (mean age 34.5 vs. 30.8 years), more likely to be men (83.6% vs. 59.1%), and had higher baseline TC and LDL-C levels. The prevalence of DM and hypertension was higher in statin users (Table 1).
Table 1. Baseline characteristics of the study population.
| Characteristics | Statin non-user | Statin user |
|---|---|---|
| No. of patients | 6,275,524 | 74,876 |
| Age (yr) | 30.8±5.0 | 34.5±3.9 |
| Sex (male) | 3,705,715 (59.1) | 62,556 (83.6) |
| BMI (kg/m2) | 22.96±3.59 | 26.23±3.93 |
| Systolic BP (mmHg) | 117.62±13.14 | 125.53±14.76 |
| Diastolic BP (mmHg) | 73.7±9.42 | 79.36±10.72 |
| Fasting glucose (mg/dL) | 90.67±15.94 | 107.73±43.7 |
| eGFR (mL/min/1.73 m2) | 96.1±49.6 | 92.7±54.3 |
| Baseline TC (mg/dL) | 184.3±35.8 | 219.3±52.9 |
| Baseline HDL-C (mg/dL) | 57.8±27.6 | 52.2±43.0 |
| Baseline TG (mg/dL) | 96.1 (96.0–96.1) | 186.1 (185.3–187.0) |
| Baseline LDL-C (mg/dL) | 102 (84–122) | 120 (87–158) |
| Current smoker | 2,177,566 (34.7) | 34,281 (45.8) |
| Alcohol drinking* | 549,285 (8.75) | 10,477 (14.0) |
| Regular exercise† | 802,702 (12.8) | 11,661 (15.6) |
| Income (lower 25%) | 1,357,336 (21.6) | 12,137 (16.2) |
| Diabetes mellitus | 107,711 (1.7) | 14,873 (19.9) |
| Hypertension | 438,053 (7.0) | 27,810 (37.1) |
Data are expressed as the means±standard deviation, median (25–75%), or number (%). The p-values for the trend were <0.001 for all variables because of the large size of the study population.
BMI, body mass index; BP, blood pressure; eGFR, estimated glomerular filtration rate; TC, total cholesterol; HDL-C, high-density lipoprotein cholesterol; TG, triglyceride; LDL-C, low-density lipoprotein cholesterol.
*Individuals who consumed ≥30 g of alcohol per day were defined as alcohol drinking.
†Regular exercise was defined as >30 minutes of moderate physical activity performed ≥5 times per week or >20 minutes of strenuous physical activity performed ≥3 times per week.
2. MI risk according to lipid profile in statin users and nonusers
During the median follow-up of 7.7 years (interquartile range 6.6–8.3 years), the number of cases of MI was 15,540 (0.24%). First, we compared MI risk after dividing participants according to statin use (Table 2), with reference groups defined as LDL-C <80 mg/dL within each group. Among participants not taking statins, LDL-C levels ≥120 mg/dL were significantly associated with MI risk (1.33 [1.27–1.40]). LDL-C ≥120 mg/dL was significantly associated with increased MI risk in statin users compared with statin users with LDL-C <80 mg/dL. Compared with LDL-C <80 mg/dL in each group, LDL-C categories 80–100 and 100–120 mg/dL were not associated with increased MI risk in either statin users or nonusers (Table 2).
Table 2. Hazard ratios and 95% confidence intervals for myocardial infarction according to lipid profiles in statin users and nonusers.
| Lipid profiles | Events | Incidence rate (per 1,000 person-years) | Model 1 | Model 2 | ||
|---|---|---|---|---|---|---|
| LDL-C (mg/dL) | ||||||
| Statin non-user | ||||||
| <80 | 2,428 | 0.27 | 1 (Ref.) | 1 (Ref.) | ||
| 80–100 | 3,169 | 0.26 | 0.93 (0.88–0.98) | 0.96 (0.91–1.02) | ||
| 100–120 | 3,350 | 0.29 | 0.95 (0.90–1.00) | 0.97 (0.92–1.03) | ||
| ≥120 | 5,938 | 0.46 | 1.35 (1.29–1.42) | 1.33 (1.27–1.40) | ||
| Statin user | ||||||
| <80 | 133 | 1.21 | 1 (Ref.) | 1 (Ref.) | ||
| 80–100 | 89 | 1.11 | 0.94 (0.72–1.22) | 1.01 (0.77–1.32) | ||
| 100–120 | 86 | 1.02 | 0.88 (0.67–1.15) | 0.98 (0.75–1.28) | ||
| ≥120 | 347 | 1.23 | 1.07 (0.88–1.31) | 1.39 (1.14–1.70) | ||
| TC (mg/dL) | ||||||
| Statin non-user | ||||||
| <160 | 2,444 | 0.23 | 1 (Ref.) | 1 (Ref.) | ||
| 160–180 | 2,833 | 0.25 | 1.03 (0.98–1.09) | 1.03 (0.97–1.08) | ||
| 180–200 | 3,077 | 0.29 | 1.09 (1.04–1.15) | 1.06 (1.01–1.12) | ||
| ≥200 | 6,531 | 0.48 | 1.63 (1.56–1.71) | 1.49 (1.42–1.56) | ||
| Statin user | ||||||
| <160 | 77 | 1.18 | 1 (Ref.) | 1 (Ref.) | ||
| 160–180 | 60 | 0.96 | 0.81 (0.58–1.14) | 0.88 (0.63–1.23) | ||
| 180–200 | 87 | 1.12 | 0.94 (0.69–1.28) | 1.07 (0.79–1.46) | ||
| ≥200 | 431 | 1.22 | 1.04 (0.82–1.33) | 1.32 (1.03–1.68) | ||
| TG (mg/dL) | ||||||
| Statin non-user | ||||||
| <60 | 1,918 | 0.19 | 1 (Ref.) | 1 (Ref.) | ||
| 60–90 | 2,884 | 0.23 | 1.05 (1.00–1.12) | 1.01 (0.96–1.07) | ||
| 90–140 | 3,740 | 0.32 | 1.25 (1.18–1.32) | 1.13 (1.07–1.20) | ||
| ≥140 | 6,343 | 0.54 | 1.85 (1.75–1.95) | 1.49 (1.40–1.57) | ||
| Statin user | ||||||
| <60 | 14 | 0.69 | 1 (Ref.) | 1 (Ref.) | ||
| 60–90 | 41 | 0.77 | 0.99 (0.54–1.82) | 0.94 (0.51–1.72) | ||
| 90–140 | 106 | 0.93 | 1.09 (0.62–1.90) | 0.94 (0.54–1.65) | ||
| ≥140 | 494 | 1.33 | 1.46 (0.86–2.48) | 1.15 (0.67–1.95) | ||
| HDL-C (mg/dL) | ||||||
| Statin non-user | ||||||
| <45 | 4,323 | 0.50 | 1 (Ref.) | 1 (Ref.) | ||
| 45–55 | 4,566 | 0.34 | 0.77 (0.74–0.81) | 0.82 (0.78–0.85) | ||
| 55–65 | 3,148 | 0.26 | 0.66 (0.63–0.69) | 0.72 (0.69–0.76) | ||
| ≥65 | 2,848 | 0.23 | 0.68 (0.64–0.71) | 0.76 (0.72–0.79) | ||
| Statin user | ||||||
| <45 | 305 | 1.55 | 1 (Ref.) | 1 (Ref.) | ||
| 45–55 | 178 | 1.01 | 0.67 (0.55–0.80) | 0.71 (0.59–0.86) | ||
| 55–65 | 102 | 0.98 | 0.67 (0.54–0.84) | 0.76 (0.61–0.95) | ||
| ≥65 | 70 | 0.87 | 0.67 (0.51–0.87) | 0.79 (0.61–1.03) | ||
Reference groups for both users and nonusers were LDL-C <80 mg/dL, TC <160 mg/dL, TG <60 mg/dL, and HDL-C <45 mg/dL. Model 1: adjusted for age and sex; Model 2: adjusted for age, sex, body mass index, income status, smoking, alcohol intake, physical activity, presence of diabetes mellitus, hypertension or chronic kidney disease.
LDL-C, low-density lipoprotein cholesterol; TC, total cholesterol; TG, triglyceride; HDL-C, high-density lipoprotein cholesterol.
Compared with TC <160 mg/dL in each group, TC ≥200 mg/dL was associated with increased MI risk in both statin users and nonusers. Among participants not taking statins, TG categories 90–140 and ≥140 mg/dL significantly increased MI risk compared with statin nonusers with TG <60 mg/dL (Table 2). Among participants taking statins, TG ≥140 mg/dL was not associated with increased MI risk compared with statin users with TG <60 mg/dL.
Next, MI risk was compared with nonusers with LDL-C <80 mg/dL as a reference group (Table 3). MI risk was increased by 1.66 [1.39–1.99] in statin users with LDL-C <80 mg/dL compared with statin nonusers with LDL-C <80 mg/dL. Statin users with LDL-C categories 80–100 mg/dL, 100–120 mg/dL, and ≥120 mg/dL all had significantly associated MI risk (1.68 [1.36–2.09], 1.63 [1.31–2.02], and 2.32 [2.07–2.60], respectively) (Table 3).
Table 3. Hazard ratios and 95% confidence intervals for myocardial infarction according to lipid profile.
| Lipid profiles | Events | Incidence rate (per 1,000 person-years) | Model 1 | Model 2 | ||
|---|---|---|---|---|---|---|
| LDL-C (mg/dL) | ||||||
| Statin non-user | ||||||
| <80 | 2,428 | 0.27 | 1 (Ref.) | 1 (Ref.) | ||
| 80–100 | 3,169 | 0.26 | 0.93 (0.88–0.98) | 0.96 (0.91–1.02) | ||
| 100–120 | 3,350 | 0.29 | 0.95 (0.90–1.01) | 0.97 (0.92–1.03) | ||
| ≥120 | 5,938 | 0.46 | 1.35 (1.29–1.42) | 1.33 (1.27–1.40) | ||
| Statin user | ||||||
| <80 | 133 | 1.21 | 2.81 (2.36–3.35) | 1.66 (1.39–1.99) | ||
| 80–100 | 89 | 1.11 | 2.63 (2.13–3.26) | 1.68 (1.36–2.09) | ||
| 100–120 | 86 | 1.02 | 2.47 (1.99–3.06) | 1.63 (1.31–2.02) | ||
| ≥120 | 347 | 1.23 | 3.02 (2.70–3.38) | 2.32 (2.07–2.60) | ||
| p for trend | <0.0001 | <0.0001 | ||||
| TC (mg/dL) | ||||||
| Statin non-user | ||||||
| <160 | 2,444 | 0.23 | 1 (Ref.) | 1 (Ref.) | ||
| 160–180 | 2,833 | 0.25 | 1.03 (0.98–1.09) | 1.03 (0.97–1.08) | ||
| 180–200 | 3,077 | 0.29 | 1.09 (1.04–1.15) | 1.06 (1.01–1.12) | ||
| ≥200 | 6,531 | 0.48 | 1.63 (1.56–1.71) | 1.49 (1.42–1.56) | ||
| Statin user | ||||||
| <160 | 77 | 1.18 | 3.30 (2.63–4.14) | 1.85 (1.47–2.32) | ||
| 160–180 | 60 | 0.96 | 2.68 (2.07–3.46) | 1.62 (1.25–2.10) | ||
| 180–200 | 87 | 1.12 | 3.11 (2.51–3.85) | 1.98 (1.59–2.46) | ||
| ≥200 | 431 | 1.22 | 3.44 (3.10–3.81) | 2.43 (2.19–2.70) | ||
| p for trend | <0.0001 | <0.0001 | ||||
| TG (mg/dL) | ||||||
| Statin non-user | ||||||
| <60 | 1,918 | 0.19 | 1 (Ref.) | 1 (Ref.) | ||
| 60–90 | 2,884 | 0.23 | 1.05 (1.00–1.12) | 1.01 (0.96–1.07) | ||
| 90–140 | 3,740 | 0.32 | 1.25 (1.18–1.32) | 1.13 (1.07–1.20) | ||
| ≥140 | 6,343 | 0.54 | 1.85 (1.75–1.95) | 1.49 (1.40–1.57) | ||
| Statin user | ||||||
| <60 | 14 | 0.69 | 2.75 (1.62–4.65) | 2.09 (1.23–3.53) | ||
| 60–90 | 41 | 0.77 | 2.72 (2.00–3.71) | 1.95 (1.43–2.67) | ||
| 90–140 | 106 | 0.93 | 2.99 (2.46–3.65) | 1.97 (1.61–2.40) | ||
| ≥140 | 494 | 1.33 | 4.01 (3.62–4.44) | 2.39 (2.15–2.66) | ||
| p for trend | <0.0001 | <0.0001 | ||||
| HDL-C (mg/dL) | ||||||
| Statin non-user | ||||||
| <45 | 4,323 | 0.50 | 1 (Ref.) | 1 (Ref.) | ||
| 45–55 | 4,566 | 0.34 | 0.77 (0.74–0.81) | 0.83 (0.78–0.85) | ||
| 55–65 | 3,148 | 0.26 | 0.66 (0.63–0.69) | 0.72 (0.69–0.76) | ||
| ≥65 | 2,848 | 0.23 | 0.68 (0.64–0.71) | 0.76 (0.72–0.79) | ||
| Statin user | ||||||
| <45 | 305 | 1.55 | 2.58 (2.30–2.90) | 1.76 (1.56–1.98) | ||
| 45–55 | 178 | 1.01 | 1.72 (1.48–2.00) | 1.26 (1.08–1.46) | ||
| 55–65 | 102 | 0.98 | 1.74 (1.43–2.12) | 1.34 (1.10–1.63) | ||
| ≥65 | 70 | 0.87 | 1.72 (1.36–2.18) | 1.39 (1.10–1.76) | ||
| p for trend | <0.0001 | <0.0001 | ||||
Statin nonusers with LDL-C <80 mg/dL, TC <160 mg/dL, TG <60 mg/dL, and HDL-C <45 mg/dL were used as references. Model 1: adjusted for age and sex; Model 2: adjusted for age, sex, body mass index, income status, smoking, alcohol intake, physical activity, presence of diabetes mellitus, hypertension or chronic kidney disease.
LDL-C, low-density lipoprotein cholesterol; TC, total cholesterol; TG, triglyceride; HDL-C, high-density lipoprotein cholesterol.
Results were similar for analyses performed using TC, TG, and HDL-C. MI risk increased by 1.85 (1.47–2.32) in statin users with TC <160 mg/dL compared with nonusers with TC <160 mg/dL. MI risk increased by 2.43 (2.19–2.70) in statin users with TC ≥200 mg/dL compared with nonusers with TC <160 mg/dL. MI risk increased by 1.76 (1.56–1.98) in statin users with HDL-C <45 mg/dL compared with statin nonusers with HDL-C <45 mg/dL.
3. Subgroup analyses
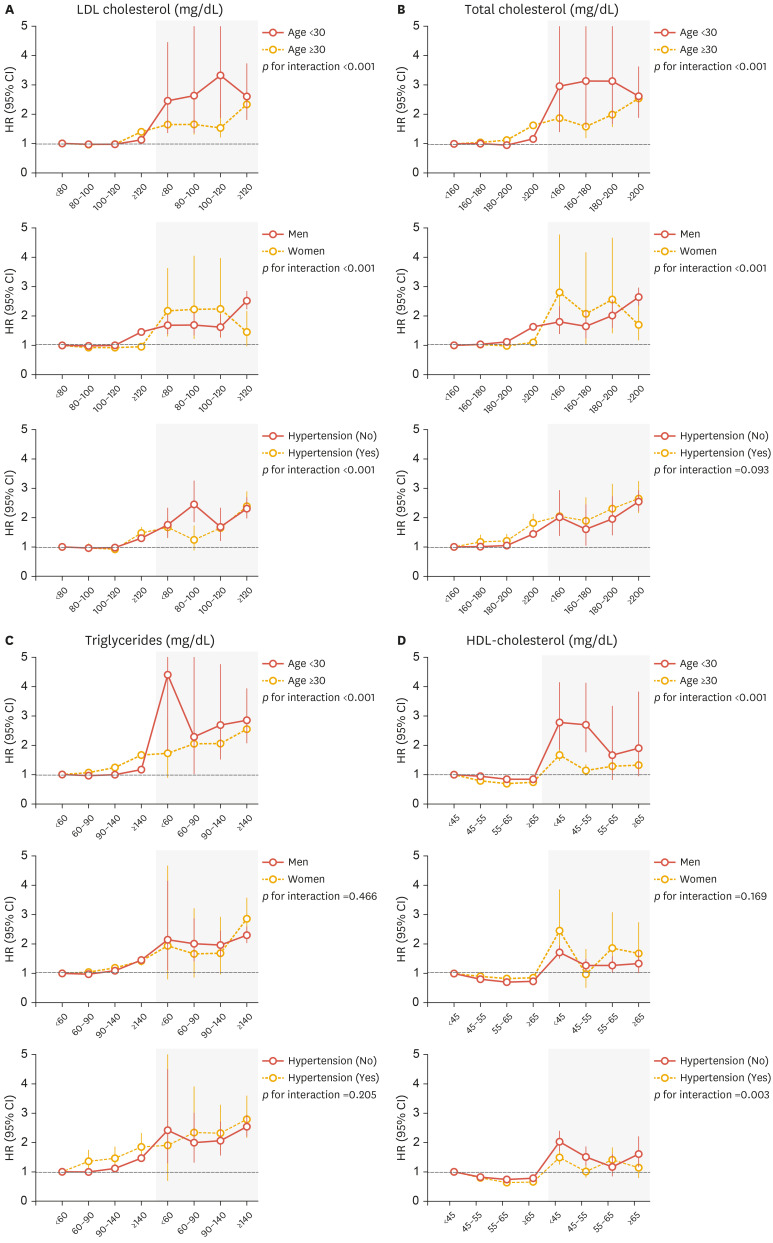
We performed stratified analyses by age, sex, and hypertension (Fig. 2). MI risk increased in statin users compared with nonusers in all subgroups regardless of lipid profile. Subgroup analyses revealed that the increased MI risk in statin users was more prominent in women, those <30 years, and those without hypertension (p for interaction <0.01).
Fig. 2. Subgroup analyses of associations between lipid components and myocardial infarction stratified by age, sex, and hypertension. Statin nonusers with LDL-C <80 mg/dL, TC <160 mg/dL, TG <60 mg/dL, and HDL-C <45 mg/dL were used as reference groups. Gray shaded areas represent statin users. (A) LDL-C, (B) TC, (C) TG, and (D) HDL-C.
Analyses adjusted for age, sex, body mass index, income status, smoking, alcohol intake, physical activity, presence of diabetes mellitus, hypertension or chronic kidney disease.
LDL-C, low-density lipoprotein cholesterol; TC, total cholesterol; TG, triglyceride; HDL-C, high-density lipoprotein cholesterol; HR, hazard ratio; CI, confidence interval.
DISCUSSION
We found that statin users had increased MI risk compared with nonusers with similar LDL-C levels. Among those with LDL-C <80 mg/dL, MI risk increased by 1.66 in statin users compared with nonusers. Statin users with LDL-C ≥120 mg/dL, probably at high risk, had a 3-fold higher risk of MI after adjusting for age and sex. This finding supports the necessity for intervention in young population with high CV risk. As shown herein, specific lipid targets may vary depending on whether statins are used.
Our participants were young adults aged 20–39 years. Even within this age range, CV risk associated with atherogenic lipid markers differed by age. The odds ratios for higher risk from elevated TC, LDL-C, and apolipoprotein-B are significantly higher among those who are younger.12 This suggests that CVD risk associated with elevated LDL-C is higher in younger individuals than it is in older individuals.12 This is consistent with the greater relative benefits of LDL-lowering therapy in younger compared with older individuals, and argues for initiating therapy in younger individuals with elevated lipids.13,14 The 2019 European Society of Cardiology/European Atherosclerosis Society dyslipidemia guidelines have raised concerns about young adults being treated inadequately for dyslipidemia, because a low absolute risk might conceal a high relative risk that may require intensive lifestyle changes.13,14 Even moderate LDL-C and TC elevations, if sustained, can lead to considerable 10-year CV risk, suggesting that important treatment opportunities are being lost.14,15,16
Currently, CVD risk is the primary guideline-base criterion for initiating lipid-lowering therapy.4,17 Because age is the principal determinant of risk (as calculated by guideline algorithms or risk engines), with the exception of those with markedly elevated LDL-C levels or DM, lipid-lowering interventions become common only after age 60 years.4,17 However, almost half of all CV events in men, and one third in women, occur before age 65 years.18 Forty- and 50-year-olds account, quantitatively, for an important fraction of the total number of events. Young adults who experience prolonged exposure to higher LDL-C levels have a significantly higher risk of coronary heart disease during their next decade.14,15,16 It was recently reported that cumulative LDL-C exposure during middle life is associated with risk of incident CVD.14,15,16 These cumulative findings suggest that maintaining optimal LDL-C level throughout middle age can minimize lifetime risk of atherosclerotic CVD.
Herein, about 1.2% of Koreans aged 20–39 years took statins. We found that statin users had an increased MI risk compared with nonusers, even at similar LDL-C or TC levels. These young adults who were taking statins were also more likely to have a greater number of CVD risk factors. In this study, statin users had a higher risk of MI compared with statin nonusers, even after adjusting for multiple confounding factors such as DM, hypertension, smoking, and chronic kidney disease. However, there is still the possibility of an increased risk of MI due to CV risk factors in statin users. Ultimately, even in the young population, the optimal LDL-C level should differ depending on the CV risk factors, and further studies related to this issue should be conducted. The risk of new-onset CVD is low before age 40 years, increases steadily from age 40–60 years, and jumps sharply thereafter. By age 60 years, asymptomatic intramural atherosclerotic disease is often advanced.3 Those taking statins at a relatively early age may also benefit from maintaining appropriate lipid levels during statin treatment, which may differ from those of young adults who do not use statins.
The risk of MI increased linearly from TG levels ≥90 mg/dL among statin nonusers. However, among statin users, elevated TG levels were not associated with an increased risk of MI. Previous epidemiological studies have shown that the effect of TG levels on CVD was more prominent in women, young adults (<50 vs. ≥ 50 years), and those without other CVD risk factors.19,20 The CVD risk related to TG levels has been reported to be relatively weak in the diabetes group but significant in subjects without DM.20 These results suggest that TG is an independent risk factor for overall CVD events in the clinically healthy population.
This study was not without limitations. First, we lacked data on participants’ lipid profiles prior to initiating statin treatment. We also had no information on statin intensity and drug compliance or persistence. Second, a single lipid profile measurement was used; repeated assessments would more accurately classify participants due to the potential for day-to-day physiologic variability or laboratory error. Third, this was a retrospective study and, therefore, the association between lipid abnormalities and MI may not be causal, and optimal lipid levels as we presented may not be conclusive. To minimize the possible effects of reverse causality, we excluded those with previous MI or stroke and we set a lag period of 1 year and excluded 54,100 subjects to ensure the diagnosis of MI were newly made. Fourth, data on the study end points were obtained only from administrative data, which can be misleading. There is the possibility of over- or under-estimation due to low sensitivity of claim-based MI definition. However, the accuracy of NHIS claims data for the diagnosis of MI has been previously validated.21 Fifth, we did not consider other vascular outcomes such as ischemic stroke or peripheral artery disease. Further studies on lipid levels associated with an increased risk of ischemic stroke or peripheral artery disease should be conducted. Sixth, some patients with LDL-C≥120mg/dL in statin users may belong to the category of severe hypercholesterolemia. According to the study on familial hypercholesterolemia (FH) in Korea, patients with FH had a 2.18 times higher risk for CVD mortality.22 This could act as a confounder. However, the estimated prevalence of FH was low at 0.1% and of these, less than 0.6% had taken lipid lowering medications.22 Finally, because these results were derived from a single Asian country, they cannot necessarily be generalized to other regions or ethnicities. Nevertheless, the current study was based on nationwide claims data covering 97.0% of the Korean population, with a median follow-up duration of 7.7 years. This large-scale, long-term observational study included 6,350,400 adults in their 20s and 30s. In addition to claims data, we incorporated baseline clinical characteristics, including biochemical data from health checkups, allowing adjustments for potential confounding factors.
In conclusion, young adults who take statins have increased MI risk compared with statin nonusers, even at similar LDL-C levels. Specific lipid targets may differ depending on statin use among young Korean adults without CVD.
Footnotes
Funding: This study was supported by the big data analysis grant from the Korean Society of Lipid and Atherosclerosis. This work was supported in part by the National Research Foundation of Korea Grant funded by the Korean Government (NRF-2020R1F1A1049539). The funders of the study had no role in study design, data collection, data analysis, data interpretation, or writing of the report. The funding agency had no role in the design, collection, analysis, or interpretation of data; in the writing of the manuscript; or in the decision to submit the manuscript for publication.
Conflict of Interest: The authors have no conflicts of interest to declare. The funding source had no role in the collection of the data or in the decision to submit the manuscript for publication. No other potential conflicts of interest relevant to this article were reported.
- Conceptualization: Kim MK.
- Data curation: Han K.
- Formal analysis: Han K.
- Funding acquisition: Kim MK.
- Investigation: Jeong H, Kim MK.
- Methodology: Han K.
- Supervision: Yoo SJ.
- Writing – original draft: Jeong H.
- Writing - review & editing: Yoo SJ, Kim MK.
References
- 1.Pletcher MJ, Bibbins-Domingo K, Liu K, Sidney S, Lin F, Vittinghoff E, et al. Nonoptimal lipids commonly present in young adults and coronary calcium later in life: the CARDIA (Coronary Artery Risk Development in Young Adults) study. Ann Intern Med. 2010;153:137–146. doi: 10.1059/0003-4819-153-3-201008030-00004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Jeong IK, Kim SR. Efficacy and safety of Pitavastatin in a Real-wOrld setting: Observational study evaluating saFety in patient treated with pitavastatin in Korea (PROOF study) Endocrinol Metab (Seoul) 2020;35:882–891. doi: 10.3803/EnM.2020.723. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Domanski MJ, Fuster V, Diaz-Mitoma F, Grundy S, Lloyd-Jones D, Mamdani M, et al. Next steps in primary prevention of coronary heart disease: rationale for and design of the ECAD trial. J Am Coll Cardiol. 2015;66:1828–1836. doi: 10.1016/j.jacc.2015.08.857. [DOI] [PubMed] [Google Scholar]
- 4.Grundy SM, Stone NJ, Bailey AL, Beam C, Birtcher KK, Blumenthal RS, et al. 2018 AHA/ACC/AACVPR/AAPA/ABC/ACPM/ADA/AGS/APhA/ASPC/NLA/PCNA guideline on the management of blood cholesterol: executive summary: a report of the American College of Cardiology/American Heart Association Task Force on Clinical Practice Guidelines. Circulation. 2019;139:e1046–e1081. doi: 10.1161/CIR.0000000000000624. [DOI] [PubMed] [Google Scholar]
- 5.Hur KY, Moon MK, Park JS, Kim SK, Lee SH, Yun JS, et al. 2021 Clinical Practice Guidelines for Diabetes Mellitus of the Korean Diabetes Association. Diabetes Metab J. 2021;45:461–481. doi: 10.4093/dmj.2021.0156. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Rhee EJ, Kim MK, Lee WY. Best achievements in clinical medicine in diabetes and dyslipidemia in 2020. Endocrinol Metab (Seoul) 2021;36:41–50. doi: 10.3803/EnM.2021.106. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Kim MK, Han K, Joung HN, Baek KH, Song KH, Kwon HS. Cholesterol levels and development of cardiovascular disease in Koreans with type 2 diabetes mellitus and without pre-existing cardiovascular disease. Cardiovasc Diabetol. 2019;18:139. doi: 10.1186/s12933-019-0943-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Akyea RK, Kai J, Qureshi N, Iyen B, Weng SF. Sub-optimal cholesterol response to initiation of statins and future risk of cardiovascular disease. Heart. 2019;105:975–981. doi: 10.1136/heartjnl-2018-314253. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Lee SH, Han K, Kim HS, Cho JH, Yoon KH, Kim MK. Predicting the development of myocardial infarction in middle-aged adults with type 2 diabetes: a risk model generated from a nationwide population-based cohort study in Korea. Endocrinol Metab (Seoul) 2020;35:636–646. doi: 10.3803/EnM.2020.704. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Lee SH, Han K, Kwon HS, Kim MK. Frequency of exposure to impaired fasting glucose and risk of mortality and cardiovascular outcomes. Endocrinol Metab (Seoul) 2021;36:1007–1015. doi: 10.3803/EnM.2021.1218. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Friedewald WT, Levy RI, Fredrickson DS. Estimation of the concentration of low-density lipoprotein cholesterol in plasma, without use of the preparative ultracentrifuge. Clin Chem. 1972;18:499–502. [PubMed] [Google Scholar]
- 12.Sniderman AD, Islam S, McQueen M, Pencina M, Furberg CD, Thanassoulis G, et al. Age and cardiovascular risk attributable to apolipoprotein B, low-density lipoprotein cholesterol or non-high-density lipoprotein cholesterol. J Am Heart Assoc. 2016;5:e003665. doi: 10.1161/JAHA.116.003665. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Mach F, Baigent C, Catapano AL, Koskinas KC, Casula M, Badimon L, et al. 2019 ESC/EAS Guidelines for the management of dyslipidaemias: lipid modification to reduce cardiovascular risk. Eur Heart J. 2020;41:111–188. doi: 10.1093/eurheartj/ehz455. [DOI] [PubMed] [Google Scholar]
- 14.Park JB, Kim DH, Lee H, Lee HJ, Hwang IC, Yoon YE, et al. Effect of moderately but persistently elevated lipid levels on risks of stroke and myocardial infarction in young Korean adults. J Am Heart Assoc. 2021;10:e020050. doi: 10.1161/JAHA.120.020050. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Navar-Boggan AM, Peterson ED, D’Agostino RB, Sr, Neely B, Sniderman AD, Pencina MJ. Hyperlipidemia in early adulthood increases long-term risk of coronary heart disease. Circulation. 2015;131:451–458. doi: 10.1161/CIRCULATIONAHA.114.012477. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Zhang Y, Pletcher MJ, Vittinghoff E, Clemons AM, Jacobs DR, Jr, Allen NB, et al. Association between cumulative low-density lipoprotein cholesterol exposure during young adulthood and middle age and risk of cardiovascular events. JAMA Cardiol. 2021;6:1406–1413. doi: 10.1001/jamacardio.2021.3508. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Blankstein R, Singh A. Cholesterol guidelines: a missed opportunity for young adults? J Am Coll Cardiol. 2020;76:665–668. doi: 10.1016/j.jacc.2020.06.055. [DOI] [PubMed] [Google Scholar]
- 18.Sniderman AD, Thanassoulis G, Williams K, Pencina M. Risk of premature cardiovascular disease vs the number of premature cardiovascular events. JAMA Cardiol. 2016;1:492–494. doi: 10.1001/jamacardio.2016.0991. [DOI] [PubMed] [Google Scholar]
- 19.Liu J, Zeng FF, Liu ZM, Zhang CX, Ling WH, Chen YM. Effects of blood triglycerides on cardiovascular and all-cause mortality: a systematic review and meta-analysis of 61 prospective studies. Lipids Health Dis. 2013;12:159. doi: 10.1186/1476-511X-12-159. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Kim EH, Lee JB, Kim SH, Jo MW, Hwang JY, Bae SJ, et al. Serum triglyceride levels and cardiovascular disease events in Koreans. Cardiology. 2015;131:228–235. doi: 10.1159/000380941. [DOI] [PubMed] [Google Scholar]
- 21.Kimm H, Yun JE, Lee SH, Jang Y, Jee SH. Validity of the diagnosis of acute myocardial infarction in korean national medical health insurance claims data: the Korean Heart Study (1) Korean Circ J. 2012;42:10–15. doi: 10.4070/kcj.2012.42.1.10. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Jung KJ, Koh H, Choi Y, Lee SJ, Ji E, Jee SH. Familial hypercholesterolemia and atherosclerotic cardiovascular mortality among Korean adults with low levels of serum cholesterol. Atherosclerosis. 2018;278:103–109. doi: 10.1016/j.atherosclerosis.2018.09.012. [DOI] [PubMed] [Google Scholar]