Abstract
Background
Older people and people with dementia experience a high prevalence of multiple health conditions. The terms ‘comorbidity’ and ‘multimorbidity’ are often used interchangeably to describe this, however there are key conceptual differences between these terms and their definitions. This has led to issues in the validity and comparability of research findings, potentially inappropriate intervention development and differences in quality of health care.
Objective
To review how the terms ‘comorbidity’ and ‘multimorbidity’ are defined within peer-reviewed dementia research and propose an operational framework.
Design
A scoping review of definitions within dementia research was carried out. Searches took place across five databases: Academic Search Premier, CINAHL Complete, MEDLINE, PsycARTICLES and PsycINFO. PRISMA-ScR guidelines were followed.
Results
Content analysis revealed five key themes, showing significant overlap and inconsistencies from both within, and between, the comorbidity and multimorbidity definitions; 1. Number of conditions; 2. Type of health conditions; 3. The co-occurrence of conditions; 4. The inclusion of an index disease (or not); 5. Use of medical language. The analysis also revealed gaps in how the underlying concepts of the definitions relate to people with dementia living with multiple health conditions.
Conclusion
This scoping review found that current definitions of comorbidity and multimorbidity are heterogeneous, reductionist and disease-focussed. Recommendations are made on the design of research studies including transparency and consistency of any terms and definitions used. A syndemic framework could be a useful tool for researchers, clinicians and policy makers to consider a more holistic picture of a person with dementia’s health and wellbeing.
Keywords: Multimorbidity, comorbidity, dementia, syndemic, scoping review
Introduction
There are numerous terms used in the literature to describe multiple health conditions, examples of which include ‘comorbidity’, ‘multimorbidity’, ‘patient complexity’ and ‘long-term conditions’ (see Table 1 for some examples of terms and associated definitions). However, there is a lack of consensus in the literature around how to define and conceptualise the various terminology used to describe people living with more than one health condition1–7 potentially leading to various interpretations or assumptions made by researchers and healthcare practitioners. For example, the term ‘comorbidity’ was defined originally by Feinstein in 1970 as ‘any distinct additional clinical entity that has existed or that may occur during the clinical course of a patient who has the index disease under study’ 8 (see Table 1) yet a review by Almirall and Fortin 7 in 2013 found at least 144 publications that used the term ‘comorbidity’ without referring to an index disease (an index disease refers to a primary condition of interest). Another example is the term ‘multimorbidity’. One definition describes this as ‘the co-existence of two or more chronic conditions, where one is not necessarily more central than the others’ 9 (see Table 1) however the Medical Subject Headings (MeSH) produced by the National Center for Biotechnology Information: U.S. National Library of Medicine 10 define multimorbidity as ‘the complex interactions of several co-existing diseases’. The latter definition does not state a minimum number of conditions nor place a position of whether one condition is more central than another.
Table 1.
Terms and definitions of multiple health conditions.
| Term | Definition a |
|---|---|
| Comorbidity | ‘any distinct additional clinical entity that has existed or that may occur during the clinical course of a patient who has the index disease under study’ 8 |
| Multimorbidity | ‘the co-existence of two or more chronic conditions, where one is not necessarily more central than the others’. 9 |
| Long-term conditions | ‘A long-term condition (LTC) is a condition that cannot, at present, be cured but is controlled by medication and/or other treatment or therapies’ 11 |
| Multiple chronic conditions | ‘Two or more chronic conditions that are expected to last at least 12 months and that confer a significant risk of death, decompensation, or functional decline’ 12 |
| Patient complexity | Schaink et al. 13 describe five elements of patient complexity: medical/physical health, mental health, demographics, social capital and health and social experiences. |
| Polymorbidity | ‘Although there is no universally accepted definition of poly-morbidity (also known as multimorbidity), some authors define it as being the co-occurrence of at least two chronic health conditions in the same person’. 14 |
| Multicomorbidity | No definition found. See research articles authored by Mamhidir et al. 15 or Wu et al. 16 as examples of the term being used. |
aNB various terms and definitions exist to describe multiple health conditions; the above are given here for illustration.
The lack of consensus around how to define these terms may cause conceptual and methodological differences in research. For example, researchers may differ in the number of conditions considered, the type of conditions considered relevant (e.g. physical, psychological, social health), or whether the research is centred around an index condition or whether all conditions are considered equally (see Table 1). This can create difficulties in making valid or reliable interpretations of findings across studies, and ultimately apply research into practice.
Dementia is a complex, neurodegenerative condition in which no two people may experience the same symptoms or progress in the same way. 17 Dementia is not a disease; it is a syndrome – an umbrella term for a cluster of symptoms which can be understood from a combination of biological, psychological and social perspectives. 18 There are over one hundred different types of dementia 19 that can affect different parts of the brain and result in both varying and overlapping symptoms from one type of dementia to another. 20 For example, a person diagnosed with Alzheimer’s Disease may experience difficulties with short term memory, judgement and planning, 21 whereas a person diagnosed with frontotemporal dementia may experience changes in personality, behaviour and movement problems. 22
In addition, the symptoms, behaviours or emotions that PwD experience may not always be caused by dementia, but can be due to other underlying or misdiagnosed conditions that may present with similar, overlapping symptoms as dementia, such as depression, diabetes or a urinary tract infection. 23 This is referred to as diagnostic overshadowing, where symptoms or behaviours are assumed to be caused by dementia. 24 It can be difficult for healthcare practitioners, carers and PwD themselves to identify what is thought to be caused by dementia, and what is the result of other underlying conditions. Furthermore, some behaviours experienced by PwD may not necessarily be caused by dementia or have a biological origin, but are due to unmet care needs.25,26
There are estimated to be between 850,000 to one million people living with dementia in the UK. 27 As of May 2022, dementia remained the leading cause of death in England. 28 There is a significant lack of support following a dementia diagnosis 29 as well as funding in comparison to other health conditions, such as cancer and coronary heart disease (CHD). 30 Yet dementia costs the health and social care system more than cancer, CHD and stroke combined. 30 PwD aged 65 or above are more likely to have a higher prevalence of multiple health conditions than those that do not have dementia. 31 However, PwD are less likely to receive primary, preventative health care 32 or receive treatment for other co-existing conditions, for example, older people living with dementia and cancer are less likely to be offered chemotherapy or diagnostic testing compared to those with cancer but without dementia. 33 Additionally, PwD often take multiple medications and are at risk of polypharmacy, 34 which has been linked to faster functional decline in PwD. 35 The National Institute of Health and Care Excellence (NICE) Multimorbidity guidelines state that the management of multimorbidity is more problematic in the presence of dementia and that careful consideration of any benefits or harms of interventions is required. 36 Overall, the above complexities of this syndrome warrant further investigation into how to conceptualise the ‘multimorbidity’ paradigm for people living with dementia.
In sum, there are many different terms and definitions used to describe people living with multiple health conditions. Indeed, this journal has renamed itself from the Journal of Comorbidity to the Journal of Multimorbidity and Comorbidity, in recognition of the distinction between comorbidity and multimorbidity as two separate concepts. 37 Furthering our understanding of these concepts will potentially help us to ‘improve research into the phenomenon of ill health in clinical care, epidemiology, and health services’, 4 improve intervention development 1 , as well as promote a shared language between healthcare practitioners and people living with multiple health conditions. 38 Given the complexity of dementia, it is important to review how to conceptualise the terms and definitions of living with ‘multiple health conditions’ in this group. Therefore, the overall aim of this review is to explore how ‘multiple health conditions’ are conceptualised in people living with dementia within published dementia research. The findings will be used to develop an operational framework that conceptualises the ‘multiple health conditions’ paradigm in PwD to guide future research, policy and clinical practice in dementia care.
The Population-Concept-Context framework (PCC) was used 39 to guide the focus of the review and to formulate the following research question: How is comorbidity and multimorbidity defined in relation to PwD within peer-reviewed dementia research and what are the key conceptual components that form these definitions?
Method
A conceptual mapping scoping review was conducted, which is ‘a scoping study designed to establish how a particular term is used in what literature, by whom and for what purpose’. 40 Unlike a systematic review, which aims to synthesise findings from a narrow range of quality-assessed studies, a scoping review is a systematic approach to mapping the research evidence of a particular field and identify gaps in the literature. 41
The Arksey and O’Malley 41 scoping review framework was used, which consists of the following five iterative steps: 1: identifying the research question, 2: identifying relevant studies, 3: study selection, 4: charting the data and 5: collating, summarising and reporting the results. Recommendations made by Levac et al. 42 that extend and clarify steps one to five of the Arksey and O’Malley framework were also incorporated. Particular attention was paid to the recommendations made on ‘balancing feasibility with breadth and comprehensiveness; using an iterative team approach to select studies and extract data; incorporating a numerical summary and qualitative thematic analysis; identifying the implications of the study findings for policy, practice, or research and adopting consultation as a required component of scoping study methodology’. 42 Finally, the Preferred Reporting Items for Systematic reviews and Meta-Analyses extension for Scoping Reviews [PRISMA-ScR] Checklist 43 was used to ensure all of the relevant aspects required of a high-quality scoping review were completed (see Supplementary file 1).
Search strategy
The search strategy was developed with support from an Information Specialist. Five databases were searched from inception to 22nd March 2022, using EBSCOhost: Academic Search Premier, CINAHL Complete, MEDLINE, PsycARTICLES and PsycINFO. These search engines were chosen as they cover multi-disciplinary research articles from nursing, allied health, medicine and psychology disciplines. The following search terms were used: “comorbid*” OR “co-morbid*” OR “multimorbid*” OR “multi-morbid*” AND “dement*” OR “alzheimer*”.
An initial search was carried out using EBSCOhost within the five databases outlined above, for each of the terms given in Table 1. ‘Comorbidity’ and ‘multimorbidity’ produced the most results – comorbid* produced 644, 957 results, and multimorbid* produced 18, 377 results (as of 22nd March 2022). Therefore, in order to balance feasibility and breadth of the review as recommended by Levac et al. 42 the search terms were restricted to ‘comorbidity’ and ‘multimorbidity’ (the field code for these search terms was retained as ‘optional’ in order to retrieve papers from any indexed field) and the other terms described in Table 1 were not included in this review. The dementia search terms however were limited to ‘TI’ (Title), in order to reduce irrelevant papers. Additionally, the papers were limited to retrieve English language only papers at the start of the search, as translation facilities were not available. The papers identified through EBSCOhost were imported to EndNote bibliographic software 44 and duplicate papers were removed.
Inclusion and exclusion criteria
The title and abstracts of the retrieved papers were screened by two authors (RD and EC) according to the PCC framework. 39 Articles were included if they involved people living with dementia (Population); investigated or discussed comorbidity or multimorbidity in dementia (Concept) and were any type of study (e.g. case study, review) written in English (Context). After excluding papers according to the above criteria, the remaining papers were discussed with the team of authors. As there were thousands of papers still left to review following this first stage of screening, a further criterion was set to only include papers that were peer-reviewed journal articles, excluding books, theses, protocols and grey literature (Context). This made the review more feasible whilst still balancing breadth within the scoping review as recommended by Levac et al. 42 In addition, a quality assessment of included papers is not usually carried out in a scoping review41,42 however the inclusion of peer-reviewed papers only should theoretically be research that is published of a high quality. 45
The full text of the remaining articles was reviewed. Papers were included for final review if they contained a definition of comorbidity and/or multimorbidity and the research aims explicitly addressed comorbidity or multimorbidity within the research. The British library were contacted to obtain the full text of papers that were not available online; authors were contacted directly if they were not available from the British library. No restrictions were placed on the type of study or year of publication since it was important to gather a broad scope of the definitions used in the literature. Citation tracking and a hand search of the references of the final review papers were checked according to the above inclusion and exclusion criteria.
Data charting and analysis
A data charting form was developed to extract relevant data from the articles. 39 The charting form was used to extract data on two articles initially, then reviewed and updated iteratively following discussion with the team of authors. Data were extracted on the study characteristics (authors, year and location); the type of research design (e.g. case study, review); the aim of the research; the comorbidity and/or multimorbidity definition used; the reference given for the definition (if one was used); whether the reference given was in the field of dementia; the number and age of PwD (if applicable) and the number and type of health conditions researched in PwD (if reported). Inductive content analysis was used to analyse the definitions. This method was chosen as it is a systematic and flexible approach that allows the researcher to describe and quantify phenomena and extract meaning from textual data into content-related categories. 46 Content analysis was conducted following three main phases: preparation, organisation and reporting.
Phase 1: Preparation
The comorbidity and multimorbidity definitions were extracted from the papers and split into two separate tables to be analysed independently. The comorbidity definitions were analysed first; they were read several times to allow the author (RD) to make sense of the data and identify units of analysis. This consisted of words or phrases that had some meaning or relevance to the research question and reoccurred across the definitions. This process was repeated for the multimorbidity definitions.
Phase 2: Organisation
Open coding was carried out on the comorbidity definitions, and categories were developed where units of analysis reoccurred across the definitions. Numerical counts of the categories were made to enable comparisons between the data. The categories were then grouped into higher order headings. This process was repeated for the multimorbidity definitions.
Phase 3: Reporting
The final list of headings were developed by the author (RD) and then discussed with the team of authors (EC, EW and AH). The higher order headings were compared and contrasted across the comorbidity and multimorbidity definitions and described narratively.
Consultation
Arksey and O’Malley 41 and Levac et al. 42 recommend consulting with key stakeholders in order to glean insights beyond those found in the literature, thereby improving the methodological rigour of the scoping review process. The consultation therefore acts as a form of knowledge transfer, and provides the opportunity to share additional sources of information, perspectives, validation and meaning. The content analysis of the definitions were presented by the lead author (RD) to a special interest group in multimorbidity, who are also health and social care practitioners with many years’ experience of working with PwD and their supporters. A discussion of the findings took place and any additional insights or observations made from the consultation group members were documented by RD. The feedback from the consultation along with the content analysis of the definitions were used to co-create an operational framework that conceptualises the ‘multiple health conditions’ paradigm in PwD.
Ethics approval
Not applicable
Results
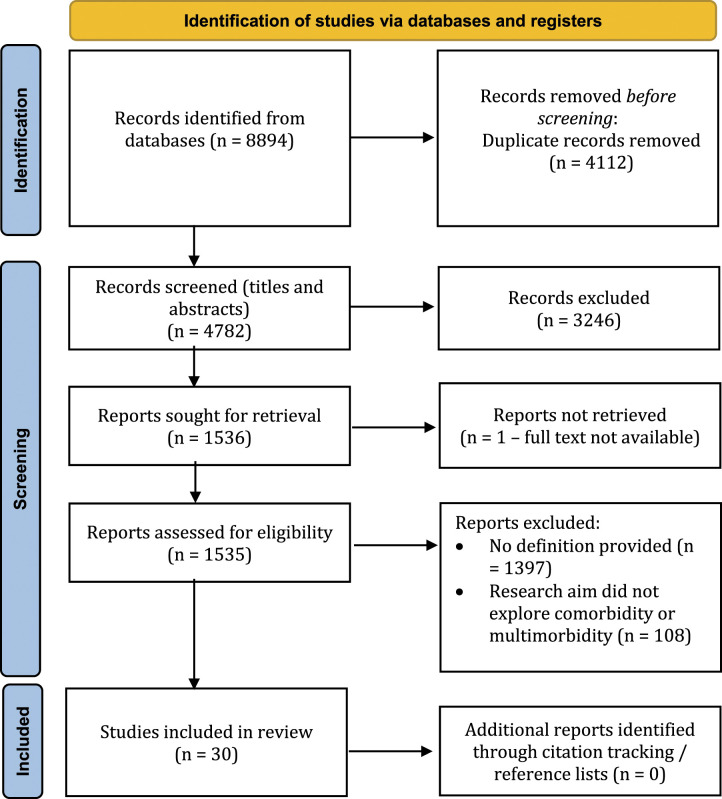
Figure 1 summarises the flow of the screening process. Initial searches found 8894 papers. Following the screening of titles, abstracts and full texts, 30 papers were included for final review that contained a definition of comorbidity or multimorbidity in relation to PwD.
Figure 1.
PRISMA Flow Diagram. 47
Study and participant characteristics
The majority of the papers were European (N = 24). Nearly two thirds of the study designs were observational (N = 18), ten of which were cohort studies, six were cross-sectional and two case control studies. Eight papers were reviews, two were case studies, one was a mixed methods study and one was an exploratory, qualitative study. The median (IQR) number of PwD recruited in the studies was 579.5 (6405.5) and the mean age (SD) was 80.4 years (± 4.4). Further details and references of the study and participant characteristics are available in supplementary files 2 and 3.
Comorbidity and multimorbidity research aims and definitions
Of the 30 papers reviewed, 23 papers aimed to explore or discuss comorbidity in PwD and only six papers had the specific research aim of exploring multimorbidity in PwD. One paper 48 uses both terms interchangeably within their research aim. Of the 23 papers that aimed to explore comorbidity, 15 papers provided a definition for comorbidity, five papers provided definitions for both comorbidity and multimorbidity, and three papers provided a definition for multimorbidity, even though the study aim was to research comorbidity.49–51 Of the six papers that aimed to explore multimorbidity in PwD, five papers provided a definition for multimorbidity, and one paper provided definitions for both multimorbidity and comorbidity (see Supplementary file 3 for references pertaining to detailed study characteristics).
37 definitions were found across the 30 papers. 22 papers provided a definition for comorbidity. 13 of the comorbidity definitions were referenced to sources such as Feinstein 8 and van den Akker et al., 5 and nine were a priori (a priori refers to the researcher’s own assumptions or definition). 15 papers provided a definition for multimorbidity, of which 11 were referenced to sources such as Boyd and Fortin 9 and four were a priori. None of the references used by the authors to cite either definitions were from the field of dementia. Tables 2 and 3 present the comorbidity and multimorbidity definitions used across the studies.
Table 2.
Comorbidity definitions.
| Paper reference | Definition | Definition a priori* or referenced |
|---|---|---|
| Aldridge and Harrison Dening 80 | ‘Comorbidity is the term used when a person has two or more chronic, long-term conditions such as schizophrenia and diabetes’ | NICE 81 † |
| Aldridge and Harrison Dening 48 | ‘Comorbidity, on the other hand, refers to situations where one or more conditions coexist with one being a primary or index condition. For example, an oncologist may consider the impact of comorbid conditions in the treatment and management of cancer’. | Valderas et al. 4 and Bunn et al. 53 |
| Aubert et al. 52 | ‘… yet older adults can develop an accumulation of comorbidities, which are defined as pathologies that influence the onset or evolution of an index pathology…’ | Extermann 82 |
| Beghi and Beghi 66 | ‘A comorbid condition is one that occurs in association with a given disease’. | a priori |
| Browne et al. 69 | ‘We defined comorbidity as the presence of one or more chronic health conditions, excluding dementia itself as the index condition, based on the list of long-term conditions in UK primary care published by Barnett et al…’ | Barnett et al. 31 |
| Bunn et al. 53 | ‘Comorbidity - the presence of more than one distinct condition in one individual. Definitions tend to consider that one condition assumes a central place’. | Feinstein 8 |
| Bunn et al. 54 | ‘… There are different conceptualisations of comorbidity but they are based on the core concept of more than one distinct condition in an individual…’ | Valderas et al. 4 |
| Clodomiro et al. 70 | ‘People affected by dementia often present with additional chronic medical conditions (comorbidity)’. | a priori |
| Damiani et al. 55 | ‘…we defined comorbid disease as the simultaneous occurrence of at least one medical condition in the same person in addition to dementia…’ | Van den Akker et al. 5 |
| Dasch et al. 56 | ‘… comorbidity — i.e., they suffer from several diseases simultaneously…’ | a priori |
| Formiga et al. 57 | ‘comorbidity (i.e. the presence of one or more disorders in addition to dementia)’ | a priori |
| Gerritsen et al. 58 | ‘Comorbidity, which is any clinical condition that occurs during the course of an index disease…’ | a priori |
| Griffith et al. 71 | ‘The terms “comorbidity” and “multimorbidity” both refer to the occurrence of multiple chronic conditions within the same individual; however “comorbidity” refers to the effects of additional conditions in reference to an index chronic condition (such as diabetes, dementia, and stroke)’. | Van den Akker et al. 5 |
| Haaksma et al. 59 | ‘Comorbidity is defined as any additional co-existing ailment in a patient with a particular index disease’. | Feinstein 8 |
| Haaksma et al. 60 | ‘…dementia patients are likely to suffer from concurrent diseases, ie, comorbidity’ | a priori |
| Heun et al. 61 | ‘A co-morbid disease was defined as any diagnosis of a somatic or mental disorder other than AD’ | a priori |
| Kaczynski et al. 62 | ‘comorbidity (one or more other diseases in addition to the underlying disease)’ | Valderas et al. 4 and Van den Akker et al. 5 |
| Kim et al. 63 | ‘Although the definition of comorbidity is simple—the “means two or more medical conditions or disease processes that are additional to an initial diagnosis and directly or in-directly affects health outcomes (Feinstein, 1970)” - the underlying construct is considerably more complex’. | Feinstein 8 |
| Martin-Garcia et al. 64 | ‘In this study, comorbidity is defined as the co-occurrence of pre-existing age-related health conditions or diseases in one person 83 in a way that symptoms, progress, and treatment interact, making health and social care of older adults a challenging issue’. | Yancik et al. 83 |
| Nelis et al. 65 | ‘Co-morbidity is the term given to the presence of conditions existing concurrently with a condition considered as the primary or index disease’ | Yancik et al. 83 |
| Poblador-Plou et al. 72 | ‘patients with dementia have on average 2 to 8 additional chronic diseases (comorbidities)’ | a priori |
| Volicer and Hurley 73 | ‘Comorbidity can be defined as “any distinct clinical entity that has existed or that may occur during the clinical course of a patient who has the index disease under study”. In a strict sense, comorbidity applies only to coexistent diseases, not symptoms’. | Feinstein 8 |
| ‘The two main types of comorbidity are (1) random comorbidity, in which Alzheimer’s disease coexists with another disease by chance alone, and (2) epidemiological comorbidity, in which there is a relationship between prevalence of Alzheimer’s disease and other diseases’. ‘Diagnostic comorbidity exists when two diseases have the same symptoms’. ‘Prognostic comorbidity exists when the presence of another disorder affects the prognosis of a patient’. ‘Incidental comorbidity is that which has no influence on either disease detection or course. Examples of incidental comorbidity are gout, or other chronic nonlife threatening conditions’. |
a priori |
*a priori indicates this is the researchers own definition.
†National Institute for Health and Care Excellence
Table 3.
Multimorbidity definitions.
| Paper reference | Definition | Definition given a priori* or referenced |
|---|---|---|
| Aldridge and Harrison Dening 80 | ‘multimorbidity there are more than two conditions’. | NICE 81 † |
| Aldridge and Harrison Dening 48 | ‘Multimorbidity refers to the co-occurrence of two or more physical and/or psychiatric conditions experienced by an individual, which may or may not directly interact with each other’ … ‘whereas multimorbidity captures the overall complexity of patients without focusing on any single disease’ | Barnett et al. 31 |
| Browne et al. 69 | ‘Multimorbidity—the presence of two or more chronic health conditions…’ | Barnett et al. 31 |
| Bunn et al. 53 | ‘Multimorbidity - the co-occurrence of multiple chronic or acute diseases and medical conditions in one person. One condition is not necessarily more central than the other. The term encompasses multiple, potentially interacting medical and psychiatric conditions’. | Boyd and Fortin 9 |
| Butler et al. 50 | ‘These conditions are also common in multimorbidity (defined as at least two comorbid conditions) in older adults’. | a priori |
| Cervellati et al. 68 | ‘the most demented patients are affected by multimorbidity, that is the co-occurrence of multiple diseases or medical conditions within a single person’. | Valderas et al. 4 |
| Cunningham et al. 76 | ‘Multi-morbidity has been variably defined and often used interchangeably, or conflated with, comorbidity (Nicholson et al., 2019). For our purposes, multi-morbidity refers to where an individual has two or more long-term health conditions and the consequences of this’. | NICE 81 † |
| Griffith et al. 71 | ‘Multimorbidity, i.e., the co-existence of 2 or more chronic conditions in the same person’ ‘The terms “comorbidity” and “multimorbidity” both refer to the occurrence of multiple chronic conditions within the same individual; …whereas the term “multimorbidity” indicates that no single condition holds priority over any of the co-occurring conditions’. | Boyd and Fortin 9 and Lefevre et al. 84 |
| Kaczynski et al. 62 | ‘Multimorbidity (the simultaneous existence of several diseases)’ | Van den Akker et al. 5 and Van den Bussche et al. 85 |
| Knight and Harrison Dening 51 | ‘Multimorbidity is where two or more medical conditions or disease processes that are additional to an initial diagnosis co-exist and where one is not necessarily more central than the others..’ | Boyd and Fortin 9 |
| Melis et al. 74 | ‘Multimorbidity was defined as any co-occurrence of two or more chronic conditions in the same individual’ | Van den Akker et al. 86 |
| Mondor et al. 75 | ‘The co-occurrence of multiple chronic conditions in an individual (multimorbidity)’ | a priori |
| Nelis et al. 65 | ‘Multimorbidity is the co-occurrence of two or more diseases or active health conditions that may or may not be linked by a causal relationship or with no consistent dominant index disorder’ | a priori |
| Quinones et al. 77 | ‘Co-existing, multiple chronic health conditions (multimorbidity)’ | a priori |
| Vogelgsang et al. 49 | ‘… . Although the definition of multimorbidity varies, it is commonly defined as the presence of two or more diseases or risk factors…’ | Willadsen et al. 6 |
*a priori indicates this is the researchers own definition.
†National Institute for Health and Care Excellence
Analysis of comorbidity and multimorbidity definitions
Content analysis of the extracted definitions revealed five key themes, showing significant overlap and inconsistencies from both within, and between, the comorbidity and multimorbidity definitions.
Type of health conditions
73% (N = 16) of comorbidity definitions48,52–66 and 53% (N = 8) of multimorbidity definitions48–51,62,65,67,68 did not provide a description of the specific types of conditions they intended to explore. 27% (N = 6) of comorbidity definitions67,69–73 and 40% (N = 6) of multimorbidity definitions69,71,74–77 described the presence of ‘chronic’ or ‘long-term’ conditions, as part of the concept. Only one multimorbidity definition considered both chronic and acute conditions within the concept. 53
Looking beyond the definitions, and only at studies that included participant data, 15 studies measured or identified specific conditions within a population of PwD49,50,52,54,56,57,60,62,64–66,68,69,71,75 and five studies recorded any condition in a person with dementia58,61,63,74,76. The majority of these conditions appeared to be chronic and varied greatly between studies (see supplementary files 2 and 3 for further information). Eight studies used the International Classification of Diseases, Ninth Revision Clinical Modification [ICD-9-CM] 78 and the International Classification of Diseases, Tenth Revision 79 [ICD-10],49,50,58,62,63,71,72,75 six studies used comorbidity indexes, such as the Charlson Comorbidity Index (CCI)52,57,60,64,65,68, three studies used electronic health records61,69,74 and one study used death certificates, 56 in order to identify and code conditions in PwD.
Co-occurrence of conditions
36% (N = 8) of comorbidity definitions described the simultaneous, ‘co-occurrence’ or ‘co-existence’ of conditions within an individual.48,55,56,59,60,64,65,73 Similarly, 67% (N = 10) of multimorbidity definitions described the ‘co-occurrence’ of conditions48,51,53,62,65,68,71,74,75,77 14% (N = 3) of comorbidity definitions described a temporal aspect to the definition, where the presence of one condition may influence the onset of another, or occur during the course of one condition52,58,73 and 13% (N = 2) of multimorbidity definitions referred to the idea that health conditions ‘may or may not be linked by a causal relationship’ 65 or ‘which may or may not directly interact with each other’. 48
Biomedical use of language
100% (N = 37) of the comorbidity and multimorbidity definitions used medical language, for example ‘disease’, ‘condition’, ‘health/medical condition’, ‘pathology’, ‘disorder’, ‘clinical condition’, ‘ailment’, ‘somatic’, ‘diagnosis’ and ‘symptoms’. However, only two comorbidity definitions referred to mental health within the concept; Aldridge and Harrison Dening 80 provided ‘schizophrenia’ as an example and Heun et al. 61 described it as a ‘mental disorder’. Similarly, only two multimorbidity definitions referred to mental health within their definitions, framed as ‘psychiatric conditions’.48,53
The number of conditions
There were inconsistencies within comorbidity definitions regarding how many conditions constituted comorbidity. 32% (N = 7) of comorbidity definitions implied that comorbidity is the presence of a condition in addition to an index disease (not necessarily dementia) within a person, indicating the co-existence of two conditions48,55,57,59,62,70,71. However, 27% (N = 6) of comorbidity definitions stated ‘two or more’ or ‘more than one’53,54,63,67,69,72 and 14% (N = 3) stated ‘several’, ‘multiple’ or ‘an accumulation’52,56,71. 27% (N = 6) of comorbidity definitions do not specify a number58,60,61,64–66. In contrast, 67% (N = 10) of the multimorbidity definitions stated ‘two or more’48–51,65,67,69,71,74,76 and 33% (N = 5) stated ‘multiple’ or ‘several’.53,62,68,75,77
The inclusion of an index disease
55% (N = 12) of comorbidity definitions described the inclusion of one condition as an ‘index disease’ that ‘assumes a central place’48,52,53,58,59,62,63,65,66,69,71,73. Although not explicitly stated within the definitions, seven papers identified dementia as the primary index disease elsewhere within the research48,52,58,59,62,65,73 however one paper explicitly excluded dementia as the primary index condition within the definition 69 and one paper chose to explore comorbidities across three index conditions; diabetes, dementia and stroke. 71 33% (N = 5) of multimorbidity definitions described multimorbidity as where ‘one condition is not necessarily more central than the other’ or that there is ‘no consistent dominant index disorder’ 48,51,53,65,71 67% (N = 10) multimorbidity definitions did not make a distinction as to whether one condition assumed a central place or whether conditions were considered equally.
Consultation
Four health and social care practitioners from the Multimorbidity Special Interest Group at the University of Hull, consisting of a Social Worker, Clinical Psychologist, Dementia Support Advisor and Pharmacist, took part in a consultation to discuss the issues outlined from the scoping review. As a research group the findings were reviewed and a discussion took place on other factors that may also be important to consider when conceptualising ‘multiple health conditions’ in PwD. The findings from the consultation were grouped into themes and sub-themes (see Table 4).
Table 4.
Findings from the consultation.
| Theme | Sub-themes / further descriptions |
|---|---|
| The perspectives and experiences of PwD, carers and families | The importance of person and relationship-centred care |
| The severity and type of dementia | Dementia is a heterogenous, complex condition where no two people are the same |
| Psychological distress/mental health | Psychological distress / mental health difficulties are highly prevalent in PwD and carers |
| Social health | The importance of reducing stigma |
| Social isolation and loneliness can have a negative impact on cognition and health | |
| Polypharmacy | Older people living with multiple health conditions often take multiple medications and are at risk of polypharmacy |
| Environmental factors | The covid-19 pandemic has had a devastating impact on PwD and their families |
Discussion
This scoping review found that the definitions used in the dementia literature are heterogeneous, reductionist and disease-focussed, with considerable overlap and inconsistencies from both within and between the different definitions. Crucially, there were many elements and concepts missing from these definitions that failed to capture aspects that are important to consider in the health and wellbeing of PwD.
Type of health conditions
Limited details were reported on the types of health conditions considered within the various definitions, and consequently within the overall study. There was a reliance on the use of classification systems such as the ICD-9-CM, 78 the ICD-10 79 and electronic health records. However, there are limitations to using these methods of measuring comorbidity or multimorbidity. For example, the ICD-10 has over 70,000 codes 79 and the more recent version, ICD-11, has over 120,000 codes; 18 how do researchers decide on which ones to use? None of the studies in this review provided a justification for how or why they chose these conditions, or why they chose to use certain methods to measure comorbidity or multimorbidity over others (e.g. the choice to use ICD-10 codes, the CCI or electronic health records). The lack of clarification reported in studies on the types of conditions included and the reasons behind their choices may have implications for research reproducibility and comparability; what one researcher considers a ‘condition’ another may not, for example ‘substance misuse’. 87
Acute, ‘short-term’ conditions, such as urinary tract infections or pneumonia are common in PwD, but can often go undetected, either because the person with dementia is unable to report the symptoms or because the healthcare professional or carer mistakes those symptoms as symptoms of dementia. 88 If left untreated, acute illnesses can lead to serious health issues, for example a urinary tract infection left untreated can lead to delirium (the symptoms of delirium can be very similar to those of dementia). 89 The onset of delirium can speed up the progression of dementia, and some people never fully return to their previous cognitive ability.90,91 Furthermore, the rate of mortality is high in PwD following acute illness; 92 indeed, the recent Coronavirus 2019 (COVID-19) pandemic has shown that PwD are at an increased risk from contracting the virus, including an increased risk of hospitalisation, psychological and cognitive decline and mortality.93–95 The results from this scoping review found that comorbidity and multimorbidity definitions mainly included ‘chronic’ or ‘long-term conditions’, which can be defined as ‘a condition that cannot, at present, be cured but is controlled by medication and/or other treatment or therapies’. 11 However, the detection and treatment of acute conditions in PwD is paramount to prevent further decline and mortality, and should be considered along with chronic conditions within the concept.
In addition, PwD may also experience ‘borderline’ conditions and ‘episodic’ conditions. Borderline conditions are not clinically diagnosed, yet people are often told by healthcare professionals that they are ‘borderline diabetic’, or their blood pressure is ‘borderline’. 96 There is a lack of clinical diagnosis here, yet some kind of action or treatment is required for the person to improve and potentially recover (the opposite to the definition of ‘chronic’). Likewise, some people may experience ‘episodic’ conditions, such as certain types of cancers or depression, in which a course of treatment is required and the person may recover. For example, a person diagnosed with early-stages of prostate cancer may undergo a radical prostatectomy, and then be cancer-free. Or a person experiencing depression may receive counselling or treatment and their symptoms may improve (depression is very common in PwD 97 ). These are serious conditions which if left untreated, could result in further decline or death. Excluding them from concepts such as comorbidity and multimorbidity due to not fitting into the ‘chronic’ or ‘long-term’ caveat is a significant oversight in the context of dementia.
Co-occurrence of conditions
The idea of ‘simultaneous’ or ‘co-occurring’ conditions implies that people experience health conditions at the same time. However, some health conditions may act as a prodrome to the onset of another condition, some can occur later as a consequence of developing a condition and some may occur completely independently. For example, research evidence has demonstrated that depression in later life can act as a prodrome to developing dementia, it can occur as a consequence of dementia, or it can be a completely separate condition to dementia in its own right. 98 Studies have also found that other conditions increase the risk of dementia. For example, diabetes increases the risk of developing mild cognitive impairment (MCI) and progression from MCI to dementia. 99 Likewise, PwD appear to experience a higher prevalence of brain diseases, infections and eating disorders compared to age-matched control subjects 48,52,58,61,65,73
In addition, research has found that people’s symptoms, experiences or conditions can fluctuate from day to day. 96 For example, a person living with arthritis and diabetes may find that their arthritis is the most troublesome condition on one day, perhaps due to an increase in pain, and then on a different day find that managing their blood glucose levels are more problematic. Other days they may find they are managing both conditions well. This resonates with Leder’s 100 phenomenological work on the ‘dys’-appearing body, in which he distinguishes between bodily ‘dis’-appearance, where one is not aware of their body and its functions, and bodily ‘dys’-appearance, where one is very much aware of their body and perceives it as ‘ill’ or ‘bad’. Similarly, the Shifting Perspectives Model 101 posits that people’s perspectives of chronic illness ‘shift in the degree to which illness is in the foreground or background of their “world.”’. 101 Health status, therefore, is not a static concept that can be described as ‘simultaneous’ or ‘concurrent’ – it is a process that fluctuates, with peaks and troughs of improvements and decline. Definitions of comorbidity or multimorbidity should therefore avoid the use of describing conditions as ‘simultaneous’ or ‘co-occurring’, and include the potential for interaction or causation within the concept. See, for example, the two multimorbidity definitions that referred to the idea that health conditions ‘may or may not be linked by a causal relationship’ 65 or ‘which may or may not directly interact with each other’. 48
Biomedical use of language
The use of medical language implies the sole inclusion of pathological or physical health conditions, and arguably excludes other potential health aspects within the concept, such as psychological or social factors, which are important indicators in a person’s overall health and wellbeing. For example, the renowned biopsychosocial model of health, developed by George Engel 102 stated that interactions between biological (genetic make-up), psychological (mental health and behaviours), and social factors (e.g. culture and relationships) determine the cause, manifestation, and outcome of wellness and disease. However, this review found that the definitions used predominantly biomedical language, with a notable lack of psychological and social health inclusion.
The use of language here is relevant for two reasons. Firstly, the medical language used promotes the biomedical stance that a person’s overall health can be reduced to distinct disease entities that are biological in nature. However qualitative research on older adults living with multiple health conditions has shown that some people reject this notion of ‘illness’ and view their health in terms of how it affects their everyday life, rather than a list of diseases. 96 Research has found that mental health problems are highly prevalent in both PwD and carers,103,104 and how psychological distress experienced by carers can have a negative impact on the physical health outcomes of PwD. 105 Additionally, social isolation and loneliness has been found to negatively affect cognition and mental health in PwD, as observed from the social restrictions and lockdowns due to the COVID-19 pandemic. 95 The importance of promoting psychological and social health in PwD has been widely documented through research and policy,106–109 however the results from this scoping review show that these areas are neglected from current concepts of comorbidity or multimorbidity in dementia research.
Secondly, the use of language is important because low health literacy is linked to poorer health outcomes. Older people, black and ethnic minority groups, those without English as a first language, those with low job or education status and those in the poverty trap have low health literacy skills. 38 Additionally, PwD often experience cognitive issues such as the ability to recall and store information, sustain attention and make decisions. 18 It is important therefore that healthcare practitioners adapt their communication style and use language that is person-centred, easy to understand and follow, and holistically considers the patient’s perspectives of their health and wellbeing.
The number of conditions and the inclusion of an index disease
There was variation in the number of conditions that warrant ‘comorbidity’ or ‘multimorbidity’ (i.e. the co-existence of just two conditions, versus more than two, versus ‘multiple’ / ‘several’). There was also ambiguity and discrepancies from both between and within the various comorbidity and multimorbidity definitions regarding whether there is an index disease or not (and how researchers decide on which condition to explore as the primary index condition if they do). These inconsistencies make it difficult to replicate research findings or make cross comparisons across data sets. 1
As aforementioned, an index disease refers to a primary condition of interest. 8 However, the primary condition of interest may differ depending on the aims of the research, the type of healthcare practitioner, or whether they work in a specialist or generalist setting. For example, if a patient was diagnosed with dementia, diabetes and heart disease, a specialist such as an endocrinologist would consider diabetes as the primary index disease, with comorbid dementia and heart disease. In contrast, a generalist working in primary care would have a more holistic, person-centred approach, and would focus on the overall health of the individual, listening to their priorities and preferences for treatment. However, the majority of health care is designed around the singular index disease model, meaning people living with dementia and/or multiple health conditions often experience fragmented care. 110
The primary condition of interest may also differ according to the perspectives of the person experiencing multiple health conditions or the person’s family or carer. For example, a person living with dementia, arthritis and diabetes may consider their primary index condition to be arthritis, which is causing them pain and discomfort. In contrast, the carer may consider the diabetes as the primary condition, due to difficulties in administering medication or managing their diet. Indeed, PwD and carers have voiced that dementia is not always the biggest concern; ‘It was acknowledged that people are often living with multiple conditions, not just dementia. Other conditions can be more debilitating than dementia, and can affect end of life planning’. 111 It is important therefore that the perspectives and experiences of PwD, carers and families are considered in order to provide person and relationship-centred care.26,112,113
As previously discussed, people may experience fluctuations in relation to their symptom severity when living with multiple health conditions, and some health conditions can be mistaken for others (i.e. diagnostic overshadowing). Therefore, research conducted on multiple health conditions, underpinned by a definition describing the inclusion of a primary index condition that ‘assumes a central place’, may not be appropriate (however this would depend on the research aims).
There have been calls to action in order to standardise comorbidity and multimorbidity definitions going back as far as 1996 5 . Over twenty years later, Nguyen et al. (2019) state: ‘The need for a consistent operationalisation of multimorbidity is evident. It will enable more accurate estimations of disease burden and, consequently, more effective disease management and resources distribution’. 114 However, despite attempts to standardise definitions3,5,7 or operationalise a measurement of multimorbidity 115 the results from this scoping review demonstrate that there is still a lack of consensus around how to define these concepts, at least within peer-reviewed dementia research.
It is difficult to determine conclusively why inconsistencies between the terms and definitions exist, however it is possible that differences may occur due to variations in subjective views according to professional discipline (e.g. biomedical, psychological or social care practitioners), what researchers believe counts as a ‘health condition’ or due to the aims of the research. Additionally, although translations have been attempted across different countries for these terms and definitions 3 the application and accuracy of these terms and definitions may vary internationally. A singular term or definition therefore may not be appropriate to explain such a complex area. However, an operational framework could be a useful approach when a standard definition does not suffice to explain a phenomenon of interest. 116
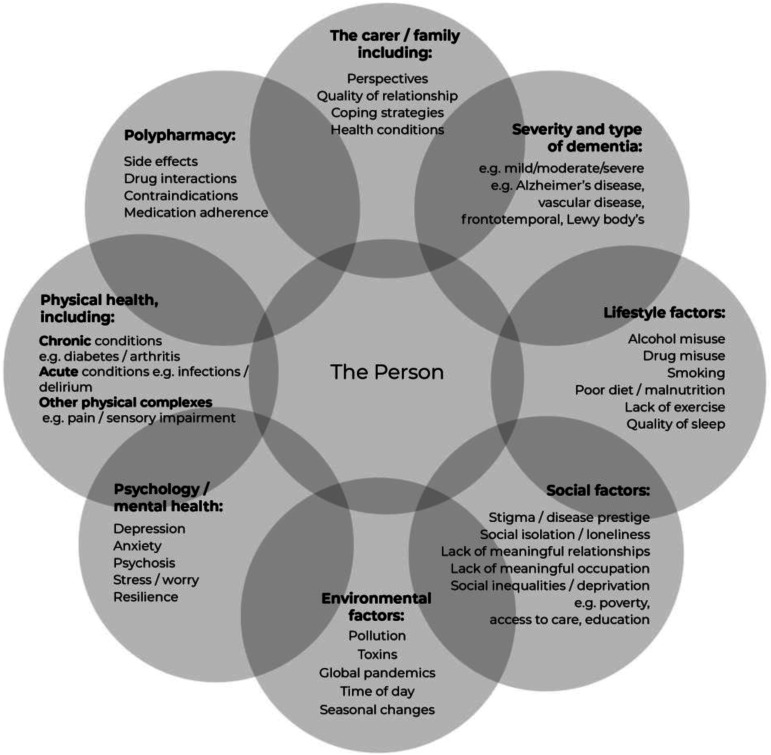
The findings from the consultation along with the gaps highlighted from the content analysis of the definitions, led to the development of a syndemic framework during an iterative process. A syndemic (synergistic-endemic) approach to health acknowledges that conditions do not just exist in parallel, and involves the interaction and consequences of biological, social, environmental, economic and lifestyle factors; 117 ‘Syndemics involve the adverse interaction between diseases and health conditions of all types (e.g. infections, chronic non-communicable diseases, mental health problems, behavioural conditions, toxic exposure, and malnutrition) and are most likely to emerge under conditions of health inequality caused by poverty, stigmatisation, stress, or structural violence’. 118 Dementia is increasingly being recognised as a syndemic disorder, given that PwD commonly live with several conditions or symptoms, the interaction of which can have greater consequences that singular conditions alone.119,120
Drawing from the results of this scoping review, in conjunction with syndemic theory, a syndemic framework is proposed in order to provide a more useful way of conceptualising ‘multiple health conditions’ in PwD, to be used by researchers, healthcare professionals and policy-makers (see Figure 2).
Figure 2.
Syndemic framework of health in dementia.
This syndemic framework does not serve to replace the comorbidity or multimorbidity definitions, rather to enhance our understanding and reflect on what is important when conducting research or providing care to a person living with dementia and multiple health conditions.
Strengths and limitations
In order to make the review feasible 42 only peer-reviewed journal articles were included, and books, theses, policies and grey literature were excluded. Therefore, some definitions may have been missed. Additionally, concepts of other terms used to describe people living with multiple health conditions, such as ‘long-term conditions’ or ‘patient complexity’, were not explored. This review focussed on the terms and definitions and did not review the methods or evaluation tools that may have been used by some studies, for example, CCI or ICD-10 codes. 79 As aforementioned, other potential reasons to further explain conceptual differences across definitions could be due to differences in research aims (e.g. risk factor assessment, outcome prediction, effectiveness of interventions), in the time-scales considered, in data availability and professional discipline. A full-scale analysis comparing these features with the authors’ definitions was beyond the scope of this review. However further analysis could be carried out in the future by extracting some of the data available from Supplementary file 3. Finally, the majority of papers found in the review were European (N = 24); we were only able to include English language papers as translation facilities were not available. This review had several strengths, however. Firstly, the review was conducted in a highly systematic approach, following comprehensive scoping review guidelines including Arksey and O'Malley, 41 Levac et al., 42 Joanna Briggs Institute [JBI] 39 and the PRISMA-ScR Checklist. 43 The screening process and review of included papers were discussed as a research team, ensuring high content validity. Additionally, the scoping review findings were discussed in consultation with dementia experts, in order to enhance our understanding further and create a useful framework that may help researchers, clinicians and policy-makers reflect beyond simplistic definitions.
Implications for research and practice
The key recommendations from this review are:
• To clearly state which health conditions are chosen to be researched and give clear reasons as to why they were included or excluded.
• To give a clear rationale as to why a particular method of measuring comorbidity or multimorbidity is chosen over another, for example if a researcher wishes to use the Charlson Comorbidity Index or ICD-11 codes. 18 (Lee et al., 2021 provide a list of multimorbidity instruments that can be used to measure against specific outcomes). 121
• To use the syndemic framework proposed from this review to reflect on the choices made when carrying out health-related research.
• To be consistent with any terms and definitions used, recognising the distinction between comorbidity and multimorbidity, as highlighted by Harrison et al. 37 but to also acknowledge the limitations posed (as highlighted from this review) when you use these terms and definitions.
Future research could explore PwD and carers’ perspectives on the language and definitions used to describe living with multiple health conditions as well as review and validate the syndemic framework. Further qualitative research is also required to explore the experiences of PwD and their carers living with other multiple health conditions.
Conclusion
This is the first scoping review to examine the definitions used to describe comorbidity and multimorbidity in dementia research. This scoping review found that current definitions of comorbidity and multimorbidity are heterogeneous, reductionist and disease-focussed, and fail to accurately reflect the health and wellbeing of people with dementia. A syndemic framework of health could be more usefully applied in health research, rather than reducing health into single disease entities. Although this research has concentrated on the field of dementia, the framework could be adapted to all health issues, and acts as a source of reflection for health and social care researchers.
Supplemental Material
Supplemental Material for Conceptualising comorbidity and multimorbidity in dementia: A scoping review and syndemic framework by Rosie Dunn, Eleanor Clayton, Emma Wolverson and Andrea Hilton in Journal of Multimorbidity and Comorbidity
Supplemental Material for Conceptualising comorbidity and multimorbidity in dementia: A scoping review and syndemic framework by Rosie Dunn, Eleanor Clayton, Emma Wolverson and Andrea Hilton in Journal of Multimorbidity and Comorbidity
Supplemental Material for Conceptualising comorbidity and multimorbidity in dementia: A scoping review and syndemic framework by Rosie Dunn, Eleanor Clayton, Emma Wolverson and Andrea Hilton in Journal of Multimorbidity and Comorbidity
Acknowledgements
We would like to thank Fiona Ware, Information Specialist, for her advice on the literature search and Rosie Connell, graphic designer, for her help in designing the syndemic framework illustration.
Author contributions: RD conceptualised the study, conducted the literature search, screening of articles, analysis, developed the syndemic framework and drafted the article. EC assisted in the literature search, screening of articles, reviewed the analysis and syndemic framework, critically edited the manuscript and approved the final version. EW and AH contributed to the design of the study, reviewed the analysis and syndemic framework, critically edited the manuscript and approved the final version.
Declarations of conflicting interests: The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding: The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental Material: Supplemental material for this article is available online.
ORCID iD
Rosie Dunn https://orcid.org/0000-0001-8196-9339
References
- 1.Johnston MC, Crilly M, Black C, et al. Defining and measuring multimorbidity: a systematic review of systematic reviews. European Journal of Public Health 2019; 29: 182–189. DOI: 10.1093/eurpub/cky098 [DOI] [PubMed] [Google Scholar]
- 2.Lee YAJ, Xie Y, Lee PSS, et al. Comparing the prevalence of multimorbidity using different operational definitions in primary care in Singapore based on a cross-sectional study using retrospective, large administrative data. BMJ open 2020; 10: e039440. 10.1136/bmjopen-2020-039440 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Le Reste JY, Nabbe P, Rivet C, et al. The European general practice research network presents the translations of its comprehensive definition of multimorbidity in family medicine in ten European languages. PloS one 2015; 10: e0115796. DOI: 10.1371/journal.pone.0115796. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Valderas JM, Starfield B, Sibbald B, et al. Defining comorbidity: implications for understanding health and health services. The Annals of Family Medicine 2009; 7: 357–363. DOI: 10.1370/afm.983. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.van den Akker M, Buntinx F, Knottnerus JA. Comorbidity or multimorbidity: what's in a name? A review of literature. The European journal of general practice 1996; 2: 65–70. 10.3109/13814789609162146 [DOI] [Google Scholar]
- 6.Willadsen TG, Bebe A, Køster-Rasmussen R, et al. The role of diseases, risk factors and symptoms in the definition of multimorbidity–a systematic review. Scandinavian journal of primary health care 2016; 34: 112-121. DOI: 10.3109/02813432.2016.1153242. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Almirall J, Fortin M. The coexistence of terms to describe the presence of multiple concurrent diseases. Journal of comorbidity 2013; 3: 4–9. DOI: 10.15256/joc.2013.3.22. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Feinstein AR. The pre-therapeutic classification of co-morbidity in chronic disease. Journal of chronic diseases 1970; 23: 455–468. DOI: 10.1016/0021-9681(70)90054-8. [DOI] [PubMed] [Google Scholar]
- 9.Boyd CM, Fortin M. Future of multimorbidity research: how should understanding of multimorbidity inform health system design? Public health reviews 2010; 32: 451–474. 10.1007/BF03391611 [DOI] [Google Scholar]
- 10.National Center for Biotechnology Information: U.S. National Library of Medicine . Multimorbidity: MeSH Unique ID D000076322. www.ncbi.nlm.nih.gov/mesh/?term=multimorbidity (2018, accessed 11/01/2022).
- 11.Department of Health [DoH] . Long Term Conditions Compendium of Information: Third Edition. 2012. [Google Scholar]
- 12.Dattalo M, DuGoff E, Ronk K, et al. Apples and Oranges: Four Definitions of Multiple Chronic Conditions and their Relationship to 30-Day Hospital Readmission. Journal of the American Geriatrics Society 2017; 65: 712–720. Article. DOI: 10.1111/jgs.14539. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Schaink AK, Kuluski K, Lyons RF, et al. A scoping review and thematic classification of patient complexity: offering a unifying framework. Journal of comorbidity 2012; 2: 1–9. DOI: 10.15256/joc.2012.2.15. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Gomes F, Schuetz P, Bounoure L, et al. ESPEN guidelines on nutritional support for polymorbid internal medicine patients. Clinical nutrition 2018; 37: 336–353. DOI: 10.1016/j.clnu.2017.06.025. [DOI] [PubMed] [Google Scholar]
- 15.Mamhidir A-G, Wimo A, Kihlgren A. Fewer referrals to Swedish emergency departments among nursing home patients with dementia, comprehensive cognitive decline and multicomorbidity. The journal of nutrition, health & aging 2012; 16: 891–897. DOI: 10.1007/s12603-012-0069-1. [DOI] [PubMed] [Google Scholar]
- 16.Wu L-T, Zhu H, Ghitza UE. Multicomorbidity of chronic diseases and substance use disorders and their association with hospitalization: Results from electronic health records data. Drug and alcohol dependence 2018; 192: 316–323. DOI: 10.1016/j.drugalcdep.2018.08.013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Alzheimer's Society . From diagnosis to end of life: The lived experiences of dementia care and support. 2020. [Google Scholar]
- 18.World Health Organisation [WHO] . International Statistical Classification of Diseases and Related Health Problems (ICD-11), Version 11. 2019. [Google Scholar]
- 19.Alzheimer's Disease International . Types of dementia, https://www.alzint.org/about/dementia-facts-figures/types-of-dementia/ (2021, accessed 28.08.21). [Google Scholar]
- 20.National Institute for Health and Care Excellence (NICE) . Dementia: assessment, management and support for people living with dementia and their carers. NICE guideline (NG97). 2018. [PubMed] [Google Scholar]
- 21.Alzheimer's Society . What is Alzheimer's Disease? Factsheet 401LP. 2017. [Google Scholar]
- 22.Alzheimer's Society . What is frontotemporal dementia (FTD)? Factsheet 404LP. 2020. [Google Scholar]
- 23.Scrutton J, Brancati CU. Dementia and comorbidities: Ensuring parity of care. . London, UK: ILC, 2016. [Google Scholar]
- 24.Clarke C, Mantle R. Using risk management to promote person-centred dementia care. Nursing Standard 2016; 30. 41, 46, DOI: 10.7748/ns.30.28.41.s47. [DOI] [PubMed] [Google Scholar]
- 25.Kitwood T. Dementia Reconsidered: The person comes first (Rethinking Ageing). Buckingham. Open University Press., 1997. [Google Scholar]
- 26.Kitwood T. Dementia reconsidered, revisited: the person still comes first. Second ed.: London: : Open University Press; 2019, 2019. [Google Scholar]
- 27.Wittenberg R, Hu B, Barraza-Araiza L, et al. Projections of older people with dementia and costs of dementia care in the United Kingdom, 2019–2040. 2019. Care Policy and Evaluation Centre, London School of Economics and Political Science. [Google Scholar]
- 28.Office for National Statistics [ONS]. Monthly mortality analysis, England and Wales: May 2022. 2022. [Google Scholar]
- 29.Alzheimer's Society . Left to Cope Alone - The unmet support needs after a dementia diagnosis. 2022. [Google Scholar]
- 30.Luengo-Fernandez R, Leal J, Gray A. UK research spend in 2008 and 2012: comparing stroke, cancer, coronary heart disease and dementia. BMJ Open 2015; 5: e006648-e006648. DOI: 10.1136/bmjopen-2014-006648. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Barnett K, Mercer SW, Norbury M, et al. Epidemiology of multimorbidity and implications for health care, research, and medical education: a cross-sectional study. The Lancet 2012; 380: 37–43. DOI: 10.1016/S0140-6736(12)60240-2. [DOI] [PubMed] [Google Scholar]
- 32.Cooper C, Lodwick R, Walters K, et al. Inequalities in receipt of mental and physical healthcare in people with dementia in the UK. Age and ageing 2017; 46: 393–400. DOI: 10.1093/ageing/afw208 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Huang H-K, Hsieh J-G, Hsieh C-J, et al. Do cancer patients with dementia receive less aggressive treatment in end-of-life care? A nationwide population-based cohort study. Oncotarget 2017; 8: 63596–63604. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Gao L, Maidment I, Matthews FE, et al. Medication usage change in older people (65+) in England over 20 years: findings from CFAS I and CFAS II. Age and ageing 2018; 47: 220–225. DOI: 10.1093/ageing/afx158. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Borda MG, Castellanos-Perilla N, Tovar-Rios DA, et al. Polypharmacy is associated with functional decline in Alzheimer's disease and Lewy body dementia. Archives of gerontology and geriatrics 2021; 96: 104459. DOI: 10.1016/j.archger.2021.104459. [DOI] [PubMed] [Google Scholar]
- 36.National Institute for Health and Care Excellence (NICE) . Multimorbidity: clinical assessment and management. Full NICE guideline NG56. 2016. [Google Scholar]
- 37.Harrison C, Fortin M, van den Akker M, et al. Comorbidity versus multimorbidity: Why it matters. Journal of Comorbidity 2021: 1-3. DOI: 10.1177/2633556521993993. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Rowlands G, Protheroe J, Price H, et al. Health Literacy: Report from an RCGP-led health literacy workshop. 2014. [Google Scholar]
- 39.Joanna Briggs Institute [JBI] . The Joanna Briggs Institute reviewers’ manual 2015: Methodology for JBI scoping reviews. 2015. [Google Scholar]
- 40.Anderson S, Allen P, Peckham S, et al. Asking the right questions: scoping studies in the commissioning of research on the organisation and delivery of health services. Health research policy and systems 2008; 6: 1–12. DOI: 10.1186/1478-4505-6-7 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Arksey H, O'Malley L. Scoping studies: towards a methodological framework. International journal of social research methodology 2005; 8: 19-32. DOI: 10.1080/1364557032000119616. [DOI] [Google Scholar]
- 42.Levac D, Colquhoun H, O'Brien KK. Scoping studies: advancing the methodology. Implementation science 2010; 5: 1–9. DOI: 10.1186/1748-5908-5-69 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Tricco AC, Lillie E, Zarin W, et al. PRISMA extension for scoping reviews (PRISMA-ScR): checklist and explanation. Annals of internal medicine 2018; 169: 467–473. DOI: 10.7326/M18-0850. [DOI] [PubMed] [Google Scholar]
- 44.The EndNote Team . EndNote. Philadelphia, PA: Clarivate, 2013. [Google Scholar]
- 45.Kelly J, Sadeghieh T, Adeli K. Peer review in scientific publications: benefits, critiques, & a survival guide. Ejifcc 2014; 25: 227. 243, DOI: PMC4975196. [PMC free article] [PubMed] [Google Scholar]
- 46.Elo S, Kyngäs H. The qualitative content analysis process. Journal of advanced nursing 2008; 62: 107–115. DOI: 10.1111/j.1365-2648.2007.04569.x. [DOI] [PubMed] [Google Scholar]
- 47.Page MJ, McKenzie JE, Bossuyt PM, et al. The PRISMA 2020 statement: an updated guideline for reporting systematic reviews. International Journal of Surgery 2021; 88: 105906. 10.1136/bmj.n71 [DOI] [PubMed] [Google Scholar]
- 48.Aldridge Z, Harrison Dening K. Managing comorbid conditions and dementia. Journal of Community Nursing 2021; 35: 54–59. [Google Scholar]
- 49.Vogelgsang J, Wolff‐Menzler C, Kis B, et al. Cardiovascular and metabolic comorbidities in patients with Alzheimer's disease and vascular dementia compared to a psychiatric control cohort. Psychogeriatrics 2018; 18: 393–401. DOI: 10.1111/psyg.12338. [DOI] [PubMed] [Google Scholar]
- 50.Butler LM, Houghton R, Abraham A, et al. Comorbidity Trajectories Associated With Alzheimer's Disease: A Matched Case-Control Study in a United States Claims Database. Frontiers in neuroscience 2021; 15: 749305. DOI: 10.3389/fnins.2021.749305. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Knight C, Harrison Dening K. Management of long-term conditions and dementia: The role of the Admiral Nurse. British Journal of Community Nursing 2017; 22: 295–302. DOI: 10.12968/bjcn.2017.22.6.295. [DOI] [PubMed] [Google Scholar]
- 52.Aubert L, Pichierri S, Hommet C, et al. Association between comorbidity burden and rapid cognitive decline in individuals with mild to moderate Alzheimer's disease. Journal of the American Geriatrics Society 2015; 63: 543–547. 10.1111/jgs.13314 [DOI] [PubMed] [Google Scholar]
- 53.Bunn F, Goodman C, Burn A-M. Multimorbidity and frailty in people with dementia. Nursing Standard 2015. 30, 45, 50, DOI: 10.7748/ns.30.1.45.e9816. [DOI] [PubMed] [Google Scholar]
- 54.Bunn F, Burn A-M, Goodman C, et al. Comorbidity and dementia: a mixed method study on improving healthcare for people with dementia (CoDem). Health services and delivery research; 2016. DOI: 10.3310/hsdr04080. [DOI] [PubMed] [Google Scholar]
- 55.Damiani G, Silvestrini G, Trozzi L, et al. Quality of dementia clinical guidelines and relevance to the care of older people with comorbidity: evidence from the literature. Clinical interventions in aging 2014; 9: 1399–1407, DOI: 10.2147/CIA.S65046. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Dasch B, Bausewein C, Feddersen B. Place of death in patients with dementia and the association with comorbidities: a retrospective population-based observational study in Germany. BMC palliative care 2018; 17: 1–12. DOI: 10.1186/s12904-018-0334-0 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Formiga F, Fort I, Robles M, et al. Comorbidity and clinical features in elderly patients with dementia: differences according to dementia severity. JNHA-The Journal of Nutrition, Health and Aging 2009; 13: 423–427. DOI: 10.1007/s12603-009-0078-x. [DOI] [PubMed] [Google Scholar]
- 58.Gerritsen AA, Bakker C, Verhey FR, et al. Prevalence of comorbidity in patients with young-onset Alzheimer disease compared with late-onset: a comparative cohort study. Journal of the American Medical Directors Association 2016; 17: 318–323. DOI: 10.1016/j.jamda.2015.11.011. [DOI] [PubMed] [Google Scholar]
- 59.Haaksma ML, Vilela LR, Marengoni A, et al. Comorbidity and progression of late onset Alzheimer’s disease: A systematic review. PloS one 2017; 12: e0177044. DOI: 10.1371/journal.pone.0177044. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Haaksma ML, Rizzuto D, Ramakers IH, et al. The impact of frailty and comorbidity on institutionalization and mortality in persons with dementia: a prospective cohort study. Journal of the American Medical Directors Association 2019; 20: 165–170. e162. DOI: 10.1016/j.jamda.2018.06.020. [DOI] [PubMed] [Google Scholar]
- 61.Heun R, Schoepf D, Potluri R, et al. Alzheimer's disease and co-morbidity: increased prevalence and possible risk factors of excess mortality in a naturalistic 7-year follow-up. European Psychiatry 2013; 28: 40–48. DOI: 10.1016/j.eurpsy.2011.06.001. [DOI] [PubMed] [Google Scholar]
- 62.Kaczynski A, Michalowsky B, Eichler T, et al. Comorbidity in dementia diseases and associated health care resources utilization and cost. Journal of Alzheimer's Disease 2019; 68: 635–646. 10.3233/JAD-180896 [DOI] [PubMed] [Google Scholar]
- 63.Kim J-H, Yoo K-B, Lee Y. Development and validation of the Korea Dementia Comorbidity Index (KDCI): A nationwide population-based cohort study from 2002 to 2013. Archives of gerontology and geriatrics 2017; 72: 195–200. DOI: 10.1016/j.archger.2017.06.001. [DOI] [PubMed] [Google Scholar]
- 64.Martín-García S, Rodríguez-Blázquez C, Martínez-López I, et al. Comorbidity, health status, and quality of life in institutionalized older people with and without dementia. International psychogeriatrics 2013; 25: 1077–1084. DOI: 10.1017/S1041610213000458. [DOI] [PubMed] [Google Scholar]
- 65.Nelis SM, Wu Y-T, Matthews FE, et al. The impact of co-morbidity on the quality of life of people with dementia: findings from the IDEAL study. Age and Ageing 2019; 48: 361–367. DOI: 10.1093/ageing/afy155. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 66.Beghi E, Beghi M. Epilepsy, antiepileptic drugs and dementia. Current opinion in neurology 2020; 33: 191–197. DOI: 10.1097/WCO.0000000000000802. [DOI] [PubMed] [Google Scholar]
- 67.Aldridge Z, Dening KH. Dementia and the management of comorbidity. Practice Nurse 2019; 49: 22–26. DOI: https://search.ebscohost.com/login.aspx?direct=true&db=ccm&AN=139135146&site=ehost-live [Google Scholar]
- 68.Cervellati C, Romani A, Bosi C, et al. Serum levels of hydroperoxides and multimorbidity among older patients with mild cognitive impairment or late-onset Alzheimer’s disease. Aging clinical and experimental research 2015; 27: 799–804. DOI: 10.1007/s40520-015-0352-1 [DOI] [PubMed] [Google Scholar]
- 69.Browne J, Edwards DA, Rhodes KM, et al. Association of comorbidity and health service usage among patients with dementia in the UK: a population-based study. BMJ open 2017; 7: e012546. DOI: 10.1136/bmjopen-2016-012546. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 70.Clodomiro A, Gareri P, Puccio G, et al. Somatic comorbidities and Alzheimer’s disease treatment. Neurological Sciences 2013; 34: 1581–1589. DOI: 10.1007/s10072-013-1290-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 71.Griffith LE, Gruneir A, Fisher K, et al. Insights on multimorbidity and associated health service use and costs from three population-based studies of older adults in Ontario with diabetes, dementia and stroke. BMC health services research 2019; 19: 1–11. DOI: 10.1186/s12913-019-4149-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 72.Poblador-Plou B, Calderón-Larrañaga A, Marta-Moreno J, et al. Comorbidity of dementia: a cross-sectional study of primary care older patients. BMC psychiatry 2014; 14: 1–8. DOI: 10.1186/1471-244X-14-84 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 73.Volicer L, Hurley A. Comorbidity in Alzheimer's disease. Journal of Mental Health and Aging 1997; 3: 5–18. [Google Scholar]
- 74.Melis RJ, Marengoni A, Rizzuto D, et al. The influence of multimorbidity on clinical progression of dementia in a population-based cohort. PloS one 2013; 8: e84014. DOI: 10.1371/journal.pone.0084014. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 75.Mondor L, Maxwell CJ, Hogan DB, et al. Multimorbidity and healthcare utilization among home care clients with dementia in Ontario, Canada: a retrospective analysis of a population-based cohort. PLoS medicine 2017; 14: e1002249. DOI: 10.1371/journal.pmed.1002249. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 76.Cunningham NA, Cowie J, Methven K. Right at home: living with dementia and multi-morbidities. Ageing & Society 2022; 42: 632-656. DOI: 10.1017/S0144686X2000104X. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 77.Quiñones AR, Kaye J, Allore HG, et al. An Agenda for Addressing Multimorbidity and Racial and Ethnic Disparities in Alzheimer's Disease and Related Dementia. American Journal of Alzheimer's Disease & Other Dementias 2020; 35: 1–7. DOI: 10.1177/1533317520960874. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 78.National Center for Health Statistics . The International classification of diseases, 9th revision, clinical modification: ICD-9-CM. US Department of Health and Human Services, Public Health Service, Health, 1980. [Google Scholar]
- 79.World Health Organization [WHO] . International Statistical Classification of Diseases and Related Health Problems: Alphabetical index. World Health Organization, 2004. [Google Scholar]
- 80.Aldridge Z, Harrison Dening K. Dementia and the Management of Comorbidity. Practice Nurse 2019: 12–16. [Google Scholar]
- 81.National Institute for Health and Care Excellence [NICE] . Multimorbidity: clinical assessment and management. 2016. Published. [Google Scholar]
- 82.Extermann M. Measuring comorbidity in older cancer patients. European journal of cancer 2000; 36: 453–471. DOI: 10.1016/s0959-8049(99)00319-6. [DOI] [PubMed] [Google Scholar]
- 83.Yancik R, Ershler W, Satariano W, et al. Report of the national institute on aging task force on comorbidity. The Journals of Gerontology Series A: Biological Sciences and Medical Sciences 2007; 62: 275–280. DOI: 10.1093/gerona/62.3.275. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 84.Lefèvre T, d’Ivernois J-F, De Andrade V, et al. What do we mean by multimorbidity? An analysis of the literature on multimorbidity measures, associated factors, and impact on health services organization. Revue d'epidemiologie et de sante publique 2014; 62: 305–314. DOI: 10.1016/j.respe.2014.09.002. [DOI] [PubMed] [Google Scholar]
- 85.Van den Bussche H, Koller D, Kolonko T, et al. Which chronic diseases and disease combinations are specific to multimorbidity in the elderly? Results of a claims data based cross-sectional study in Germany. BMC public health 2011; 11: 1–9. DOI: 10.1186/1471-2458-11-101. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 86.Van den Akker M, Buntinx F, Metsemakers JF, et al. Multimorbidity in general practice: prevalence, incidence, and determinants of co-occurring chronic and recurrent diseases. Journal of clinical epidemiology 1998; 51: 367–375. DOI: 10.1016/s0895-4356(97)00306-5. [DOI] [PubMed] [Google Scholar]
- 87.Suls J, Green PA, Boyd CM. Multimorbidity: Implications and directions for health psychology and behavioral medicine. Health psychology 2019; 38: 772. 782, DOI: 10.1037/hea0000762. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 88.Hodgson N, Gitlin LN, Winter L, et al. Undiagnosed illness and neuropsychiatric behaviors in community-residing older adults with dementia. Alzheimer disease and associated disorders 2011; 25: 109. 115, DOI: 10.1097/WAD.0b013e3181f8520a. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 89.Alzheimer's Society . Urinary tract infections (UTIs) and dementia: Factsheet 528LP. 2019. [Google Scholar]
- 90.Fick DM, Agostini JV, Inouye SK. Delirium superimposed on dementia: a systematic review. Journal of the American Geriatrics Society 2002; 50: 1723–1732. DOI: 10.1046/j.1532-5415.2002.50468.x. [DOI] [PubMed] [Google Scholar]
- 91.Manni B, Federzoni L, Zucchi P, et al. Prevalence and management of delirium in community dwelling older people with dementia referred to a memory clinic. Aging Clinical and Experimental Research 2021; 33: 2243–2250. DOI: 10.1007/s40520-020-01753-3. [DOI] [PubMed] [Google Scholar]
- 92.Mitchell SL, Teno JM, Kiely DK, et al. The clinical course of advanced dementia. New England Journal of Medicine 2009; 361: 1529–1538. DOI: 10.1056/NEJMoa0902234. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 93.Alzheimer's Society . Worst hit: dementia during coronavirus. 2020. [Google Scholar]
- 94.Wang Q, Davis PB, Gurney ME, et al. COVID-19 and dementia: Analyses of risk, disparity, and outcomes from electronic health records in the US. Alzheimer's & Dementia 2021; 17: 1297–1306. 10.1002/alz.12296 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 95.Tuijt R, Frost R, Wilcock J, et al. Life under lockdown and social restrictions-the experiences of people living with dementia and their carers during the COVID-19 pandemic in England. BMC geriatrics 2021; 21: 1–12. 10.1186/s12877-021-02257-z [DOI] [PMC free article] [PubMed] [Google Scholar]
- 96.Porter T, Ong BN, Sanders T. Living with multimorbidity? The lived experience of multiple chronic conditions in later life. Health 2020; 24: 701–718. DOI: 10.1177/1363459319834997. [DOI] [PubMed] [Google Scholar]
- 97.Kuring J, Mathias J, Ward L. Prevalence of depression, anxiety and PTSD in people with dementia: a systematic review and meta-analysis. Neuropsychology review 2018; 28: 393–416. DOI: 10.1007/s11065-018-9396-2. [DOI] [PubMed] [Google Scholar]
- 98.Korczyn AD, Halperin I. Depression and dementia. Journal of the neurological sciences 2009; 283: 139–142. DOI: 10.1016/j.jns.2009.02.346. [DOI] [PubMed] [Google Scholar]
- 99.Ng TP, Feng L, Nyunt MSZ, et al. Metabolic syndrome and the risk of mild cognitive impairment and progression to dementia: follow-up of the Singapore longitudinal ageing study cohort. JAMA neurology 2016; 73: 456–463. DOI: 10.1001/jamaneurol.2015.4899. [DOI] [PubMed] [Google Scholar]
- 100.Leder D. The absent body. University of Chicago Press, 1990. [Google Scholar]
- 101.Paterson BL. The shifting perspectives model of chronic illness. Journal of nursing scholarship 2001; 33: 21–26. DOI: 10.1111/j.1547-5069.2001.00021.x. [DOI] [PubMed] [Google Scholar]
- 102.Engel GL. The need for a new medical model: a challenge for biomedicine. Science 1977; 196: 129–136. DOI: 10.1126/science.847460. [DOI] [PubMed] [Google Scholar]
- 103.Lai AX, Kaup AR, Yaffe K, et al. High occurrence of psychiatric disorders and suicidal behavior across dementia subtypes. The American Journal of Geriatric Psychiatry 2018; 26: 1191–1201. DOI: 10.1016/j.jagp.2018.08.012. [DOI] [PubMed] [Google Scholar]
- 104.Sörensen S, Duberstein P, Gill D, et al. Dementia care: mental health effects, intervention strategies, and clinical implications. The Lancet Neurology 2006; 5: 961–973. [DOI] [PubMed] [Google Scholar]
- 105.Stall NM, Kim SJ, Hardacre KA, et al. Association of informal caregiver distress with health outcomes of community-dwelling dementia care recipients: a systematic review. Journal of the American Geriatrics Society 2019; 67: 609–617. DOI: 10.1111/jgs.15690. [DOI] [PubMed] [Google Scholar]
- 106.Dröes RM, Chattat R, Diaz A, et al. Social health and dementia: a European consensus on the operationalization of the concept and directions for research and practice. Aging & Mental Health 2017; 21: 4–17. DOI: 10.1080/13607863.2016.1254596. [DOI] [PubMed] [Google Scholar]
- 107.Regan M. The interface between dementia and mental health: an evidence review. 2016. [Google Scholar]
- 108.World Health Organization [WHO] . Global action plan on the public health response to dementia 2017–2025. Report no. 9241513489, 2017. [Google Scholar]
- 109.Sachdev PS. Social health, social reserve and dementia. Current Opinion in Psychiatry 2022; 35: 111–117. DOI: 10.1097/YCO.0000000000000779. [DOI] [PubMed] [Google Scholar]
- 110.Guthrie B, Payne K, Alderson P, et al. Adapting clinical guidelines to take account of multimorbidity. Bmj 2012; 345: e6341. [DOI] [PubMed] [Google Scholar]
- 111.Together in Dementia Everyday [TIDE] & the Dementia Engagement and Empowerment Project [DEEP] . End of Life Care and Post Bereavement Support - Shifting the Conversation from Difficult to Important. 2018. [Google Scholar]
- 112.Wilkinson H. The perspectives of people with dementia: Research methods and motivations. Jessica Kingsley Publishers, 2002. [DOI] [PubMed] [Google Scholar]
- 113.Reid DI. Doing Relationship-centred Dementia Care: Learning from Each Other for Better Dementia Support. Jessica Kingsley Publishers, 2021. [Google Scholar]
- 114.Nguyen H, Manolova G, Daskalopoulou C, et al. Prevalence of multimorbidity in community settings: a systematic review and meta-analysis of observational studies. Journal of comorbidity 2019; 9: 2235042X19870934. DOI: 10.1177/2235042X19870934. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 115.Calderón-Larrañaga A, Vetrano DL, Onder G, et al. Assessing and Measuring Chronic Multimorbidity in the Older Population: A Proposal for Its Operationalization. The Journals of Gerontology Series A: Biological Sciences and Medical Sciences 2016: 72, glw233. 1423, DOI: 10.1093/gerona/glw233. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 116.Ipsos MORI. Framing Multiple Long-Term Conditions. 2018. [Google Scholar]
- 117.Singer M. Introduction to syndemics: A critical systems approach to public and community health. John Wiley & Sons, 2009. [Google Scholar]
- 118.Singer M, Bulled N, Ostrach B, et al. Syndemics and the biosocial conception of health. Lancet 2017; 389: 941-950. journal article. DOI: 10.1016/S0140-6736(17)30003-X. [DOI] [PubMed] [Google Scholar]
- 119.Iliffe S, Manthorpe J. Dementia: beyond multi-morbidity. Journal of Public Mental Health 201716, 172, 179. DOI: 10.1108/JPMH-05-2017-0019 [DOI] [Google Scholar]
- 120.Iliffe S, Manthorpe J. Dementia: is the biopsychosocial model vindicated? : British Journal of General Practice, 2017, p. 67, 344–345. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 121.Lee ES, Koh HL, Ho EQ-Y, et al. Systematic review on the instruments used for measuring the association of the level of multimorbidity and clinically important outcomes. BMJ Open 2021; 11: e041219. DOI: 10.1136/bmjopen-2020-041219. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Supplemental Material for Conceptualising comorbidity and multimorbidity in dementia: A scoping review and syndemic framework by Rosie Dunn, Eleanor Clayton, Emma Wolverson and Andrea Hilton in Journal of Multimorbidity and Comorbidity
Supplemental Material for Conceptualising comorbidity and multimorbidity in dementia: A scoping review and syndemic framework by Rosie Dunn, Eleanor Clayton, Emma Wolverson and Andrea Hilton in Journal of Multimorbidity and Comorbidity
Supplemental Material for Conceptualising comorbidity and multimorbidity in dementia: A scoping review and syndemic framework by Rosie Dunn, Eleanor Clayton, Emma Wolverson and Andrea Hilton in Journal of Multimorbidity and Comorbidity