Abstract
Background: Most of the arthroplasty surgery failure due to prosthetic joint infections (PJI) is caused by biofilm-associated Staphylococcus aureus. In a recent experimental study, savirin has been used to prevent and treat S. aureus skin infections in animal models. We explored the application of savirin in a PJI mouse model to determine its utility as an adjunct therapy to prevent PJI.
Materials and methods: The in-vitro antibacterial and antibiofilm activity of savirin, with or without antibiotics (cefazolin, rifampicin, and vancomycin), against S. aureus were investigated using broth microdilution and crystal violet staining method, respectively. The effect of savirin treatment on the expression of the key biofilm-related genes (icaA, icaD, eno, fib, ebps, and agr) in S. aureus was studied using quantitative reverse transcriptase polymerase chain reaction (qRTPCR). The in-vivo efficacy of savirin alone and with cefazolin to prevent S. aureus PJI was determined using a clinically relevant PJI mouse model. Mice were randomized into five groups (n = 8/group): 1) infected K-wire savirin treated group, 2) infected K-wire cefazolin treated group, 3) infected K-wire savirin plus cefazolin treated group, 4) infected K-wire PBS treated group, 5) sterile K-wire group. Savirin was administered subcutaneously immediately post-surgery and intravenous cefazolin was given on day seven.
Results: Savirin inhibited planktonic and biofilm in-vitro growth of S. aureus, showed enhanced inhibitory activity when combined with antibiotics, and down-regulated the expression of key S. aureus biofilm-related genes (icaA, icaD, eno, fib, ebps, and agr). Savirin significantly reduced bacterial counts on joint implants in comparison with the PBS treated control, while savirin plus cefazolin reduced bacterial counts on both implants and peri-prosthetic tissues.
Conclusion: Savirin adjuvant therapy may prevent biofilm formation and S. aureus PJI. This study gives baseline data for using savirin for the prevention as well as treatment of S. aureus PJI in future animal studies.
Keywords: S. aureus, savirin, mouse model, prosthetic joint infection, adjuvant therapy, arthroplasty
Background
Indwelling medical devices, including prosthetic joints, create a favorable environment for biofilm-related bacterial infection (Ricciardi et al., 2018). Consequently, infection-related arthroplasty failure, mainly due to S. aureus infection, is common (Peel et al., 2012). Current treatments include major surgery either to replace or debride infected prostheses, both followed by long term antibiotic use (Zimmerli et al., 2004). However, these procedures have significant drawbacks - they are costly, potentially traumatic, and have failure rates ranging from 15 to 25% (Davis, 2016; Ma et al., 2018). Biofilm, a S. aureus growth mode that contributes to prosthetic joint infection (PJI) pathogenesis, is recalcitrant to antibiotic treatment (Morris et al., 2019). Therefore, antimicrobial therapy alone is not sufficient to treat most prosthetic joint infections (Del Pozo and Patel, 2009).
Savirin (Staphylcoccus aureus virulence inhibitor), is a low molecular weight, lipophilic, synthetic molecule suitable for drug development (Sully et al., 2014). This molecule prevents AgrA attachment to promoter regions in the agr quorum sensing system (Sully et al., 2014). It inhibits activation of the agr quorum sensing system, which is responsible for controlling many important S. aureus virulence factors, resulting in increased host-mediated bacterial killing (Sully et al., 2014). Savirin has been shown to prevent as well as treat biofilm-related S. aureus infections in rodent skin and subcutaneous infection models (Sully et al., 2014). Savirin might also be active against mature biofilm, as it was able to reduce infection even when administered 24–48 h post-infection establishment (Sully et al., 2014). Savirin was not toxic in doses (5 µg and 10 µg) used subcutaneously in two animal models (Sully et al., 2014). It appears that S. aureus is less likely to develop resistance to savirin than to conventional antibiotics as multiple in-vivo or in-vitro passages did not induce resistance in S. aureus to agr inhibition by savirin, while this did induce resistance to clindamycin (Sully et al., 2014).
There are limited data that characterize the antibiofilm efficacy of savirin and no previous study has investigated its use in the prevention of prosthesis-related infection caused by S. aureus. The objective of the current study was to investigate the effect of savirin, alone and in combination with antibiotics, on S. aureus in-vitro planktonic and biofilm growth and to determine the effect of savirin treatment on the expression of the key biofilm-related genes in S. aureus. Further, this study tested savirin’s effect as an adjuvant therapy for the prevention of S. aureus PJI in a mouse model.
Materials and methods
A methicillin susceptible S. aureus clinical strain TUH_MSSA_01 isolated from a patient attending the Townsville University Hospital, Queensland, Australia was used in this study. S. aureus isolate was cultured in Luria-Bertani (LB) broth at 37°C for 48 h without shaking. Subculturing in 0.5% glucose containing LB (GLB) broth for 24 h induced ample biofilm production.
In-vitro antibacterial and antibiofilm activity of savirin
Broth microdilution and crystal violet staining methods were performed in triplicates using microtiter plates. The 105 cfu of S. aureus in 50 µL volume was added to eight two-fold serial dilutions of savirin (80 μg/ml to 0.62 μg/ml) in 50 µL volumes and incubated for 24 h at 37°C. This resulted in eight different savirin concentrations ranging from 40 μg/ml to 0.31 μg/ml. Antibacterial activity was determined by measuring the optical density (OD) of bacterial growth in microtiter plate wells at 600 nm. The minimum bactericidal concentration (MBC) of savirin was determined by plating the microtiter plate wells showing no bacterial growth for maximum 48 h. Microtiter plate biofilm assay procedures were adapted from a previous study (Singh et al., 2019). Planktonic bacterial culture in microtiter plate wells after 24 h of growth at 37°C in the presence of savirin was discarded and the biofilm formed was fixed with 2% sodium acetate for 10 min followed by overnight staining with 1% crystal violet. Absolute ethanol was used to reconstitute the crystal violet retained and absorbance was measured at 570 nm. S. aureus growth in the savirin diluent, DMSO (0.08%), was used as a positive control and the sterile DMSO (0.08%) was used as a negative control.
Combined inhibitory effect of savirin and antibiotics (cefazolin, rifampicin, and vancomycin) on planktonic and biofilm growth
Savirin (26.67 μg/ml to 0.42 μg/ml) in combination with cefazolin (0.5 μg/ml to 0.007 μg/ml), or vancomycin (2.5 μg/ml to 0.03 μg/ml), or rifampicin (0.015 μg/ml to 0.0002 μg/ml) was used as described above, except with a total well volume of 150 µL (50 µL each of savirin, antibiotic, and bacterial broth culture suspension). The combined effect was tested by checkerboard assay by determining fractional inhibitory concentration (FIC) index value. The interaction of savirin and antibiotics was categorized as synergy (FIC < 0.5), antagonism (FIC > 4), and additive effect (FIC = 0.5–4) (Meletiadis et al., 2010; Alhashimi et al., 2019). Inhibitory effects of combined subinhibitory concentrations of savirin and antibiotics were also compared with use of each alone.
The effect of savirin on expression of key biofilm-related genes in S. aureus
RNA extraction
RNA was extracted from log phase S. aureus culture treated with 10 μg/ml savirin (test sample) and 0.02% DMSO (control sample), using the Qiagen RNeasy mini kit. Ten µg/ml savirin was used, as this concentration reduced biofilm formation without inhibiting planktonic growth. The quality and quantity of RNA was determined using a Nanodrop 2000C spectrophotometer (Thermo Fisher Scientific, United States).
Gene expression quantification
Quantitative reverse transcriptase polymerase chain reaction (qRTPCR) was performed in triplicate for each gene by using Bio-Rad iTaq universal SYBR green one-step kit (Table 1). The reference gene used was fema because the expression of this gene was not affected when S. aureus was treated with savirin. The reaction mixture (10 µL) consisted of 5 µL of 2 × iTaq universal SYBR green reaction mix, 0.125 µL iScript reverse transcriptase, 0.8 ng RNA template in 1 µL volume, 1 nmol of primer mix in 1 µL volume, and 2.9 µL nuclease free water. The cycling conditions used on the BioRad CFX96 Touch Real-Time PCR Detection System were: reverse transcription (50°C, 10 min), polymerase activation and DNA denaturation (95°C, 1 min) followed by 40 cycles of denaturation (95°C, 10 s), and annealing/extension and plate read (60°C, 30 s). The effect of savirin treatment on the expression of key S. aureus biofilm-related genes, icaA, icaD, eno, fib, ebps, agr was determined by the comparative Ct (ΔΔCt) method using BioRad CFX Manager software (Livak and Schmittgen, 2001). The data obtained were expressed as fold changes (mean ± standard deviation) compared with control.
TABLE 1.
Primers used for qRTPCR.
| Primers | Oligonucleotide sequences (5' → 3′) | References |
|---|---|---|
| icaA (F) | CAATACTATTTCGGGTGTCTTCACTCT | Kot et al. (2018) |
| icaA (R) | CAAGAAACTGCAATATCTTCGGTAATCAT | |
| icaD (F) | TCAAGCCCAGACAGAGGGAATA | Kot et al. (2018) |
| icaD (R) | ACACGATATAGCGATAAGTGCTGTTT | |
| eno (F) | AAACTGCCGTAGGTGACGAA | Kot et al. (2018) |
| eno (R) | TGTTTCAACAGCATCTTCAGTACCTT | |
| ebps (F) | ACATTCAAATGACGCTCAAAACAAAAGT | Kot et al. (2018) |
| ebps (R) | CTTATCTTGAGACGCTTTATCCTCAGT | |
| fib (F) | GAATATGGTGCACGTCCACAATT | Kot et al. (2018) |
| fib (R) | AAGATTTTGAGCTTGAATCAATTTTTGTTCTTTTT | |
| agr (F) | AATTTGTTCACTGTGTCGATAAT | Ferreira et al. (2013) |
| agr (R) | TGGAAAATAGTTGATGAGTTGTT | |
| fema (F) | TGCCTTTACAGATAGCATGCCA | Francois et al. (2003) |
| fema R) | AGTAAGTAAGCAAGCTGCAATGACC |
Animal experiment
Ethics approval was obtained from the James Cook University Animal Ethics Committee (AEC2486). Six to 10 weeks old C57BL/6 female mice (Animal Resources Centre, Western Australia) were used. The mice were assigned to five experimental groups (8 mice/group): 1) infected K-wire savirin treated group, 2) infected K-wire cefazolin treated group, 3) infected K-wire savirin plus cefazolin treated group, 4) infected K-wire PBS treated group (positive control), and 5) sterile K-wire untreated group (negative control).
S. aureus prosthetic joint infection mouse model
Surgery was performed using previously described procedures (Bernthal et al., 2010). Mice were anesthetized with ketamine/xylazine (90 mg/kg/10 mg/kg, ip) prior to surgery. Buprenorphine (0.2 mg/kg, sc) was used as analgesic 30 min pre-surgery. Fur from the right thigh region was shaved followed by disinfection with povidone iodine. The skin was incised just above the knee and the patella was displaced to expose the tip of femoral bone. A hole was then made through the femoral intramedullary canal using a 26 G needle and a precut orthopedic-grade stainless steel Kirschner (K)-wire (diameter 0.6 mm) was inserted leaving a 1 mm protrusion into the joint space. A 2 µL S. aureus (TUH_MSSA_01) normal saline inoculum (500 cfu) was pipetted into the joint space. The kneecap was returned to its original position and the surgical site was closed with a 5–0 absorbable suture. A combination of subcutaneous (0.2 mg/kg) and oral (2.5 ml/160 ml drinking water) buprenorphine was administered for 72 h as post-surgical analgesia.
Savirin and antibiotic treatments
A single non-toxic subcutaneous dose of savirin (40 µg in 100 µL), as tested in the Vero cell line culture, was administered immediately after surgery to the infected K-wire savirin treated group. A single cefazolin (2.5 μg/g, iv) dose in 100 µL volume was administered on day 7 post-surgery to the infected K-wire cefazolin treated group. The infected K-wire savirin plus cefazolin treated group was administered a single dose of savirin (40 µg in 100 µL volume, sc) immediately after surgery followed by a single cefazolin dose (2.5 μg/g in 100 µL volume, iv) on day 7 post-surgery. Cefazolin was used in this manner not to sterilize the biofilm but to assess for any increased effect where it was used with savirin. Mice were weighed and animal well-being parameters, such as eating, drinking, mobility, interaction with other mice, and reaction to external stimuli were recorded daily. Mice were euthanized on day 14 post-surgery. The intramedullary K-wires were removed in-toto and peri-prosthetic tissues were collected aseptically for bacteriological culture.
Bacteriological analysis of K-wires and peri-prosthetic tissues
K-wires were collected in 5 ml of cold LB broth after washing three times with cold sterile PBS to remove planktonic cells. Sonication at 44 khz for 5 min using a waterbath sonicator was performed to thoroughly disrupt the attached biofilms. Similarly, tissues were collected in 800 µL of ice-cold PBS to minimize bacterial multiplication followed by homogenization using a Navy Lysis Kit (BioTools, Australia). Bacterial quantification of sonication fluids and tissue homogenates was performed by the drop dilution method whereby they were serially diluted and cultured on LB agar and mannitol salt agar (MSA) at 37°C for 48 h.
Statistical analysis
One-way ANOVA was performed using GraphPad version 8.2.0 (GraphPad Software, San Diego, California, United States) followed by Tukey post-hoc test. p-value < 0.05 indicated statistical significance.
Results
Antibacterial and antibiofilm activity of savirin
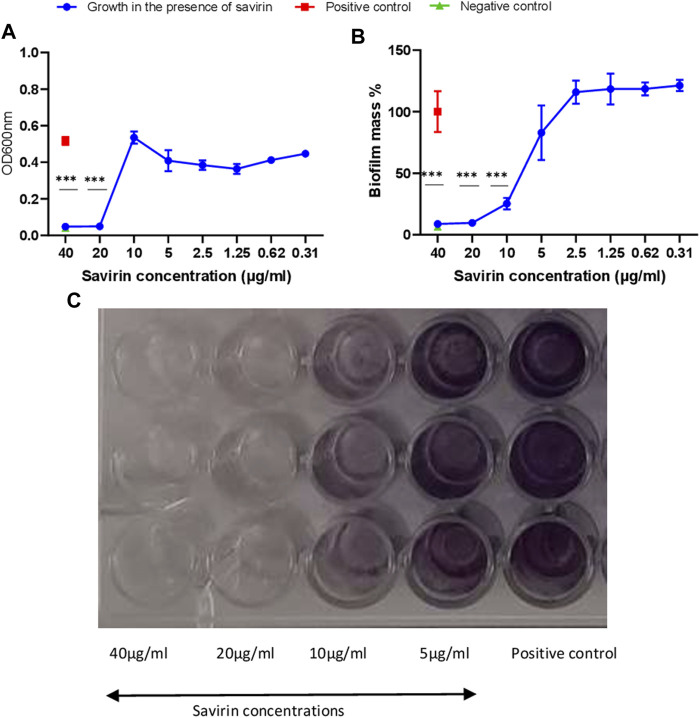
Savirin’s minimum bactericidal concentration (MBC) and minimum inhibitory concentration (MIC) were 40 μg/ml and 20 μg/ml respectively. Savirin at 40 μg/ml, 20 μg/ml and 10 μg/ml inhibited biofilm formation significantly (Figure 1).
FIGURE 1.
Planktonic (A) and biofilm (B,C) growth of S. aureus in the presence of different savirin concentrations. Triplicate wells were used for each treatment (N = 3) and experiments were repeated twice. Data are presented as mean ± standard deviation (SD) and the error bars indicate SD (*** = p < 0.001).
Combined antibacterial and antibiofilm activity of savirin and antibiotics (cefazolin, rifampicin, and vancomycin)
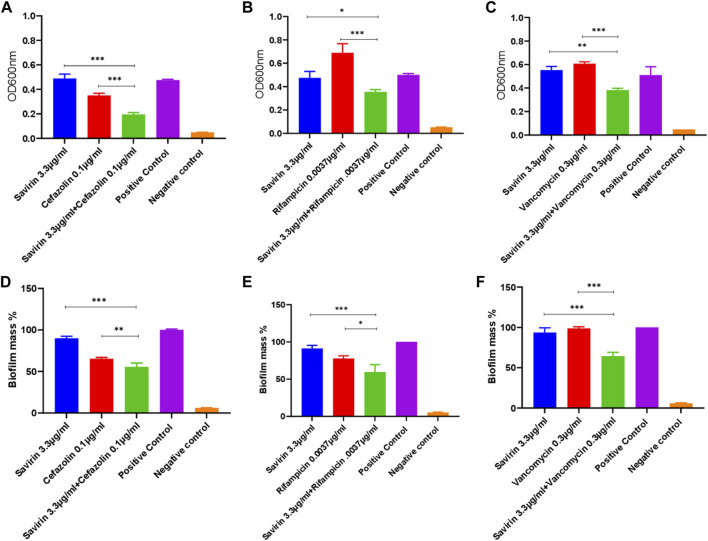
Multiple savirin and antibiotic concentration combinations were tested. The MICs of cefazolin, rifampicin, and vancomycin for the S. aureus strain used were 0.5 μg/ml, 0.015 μg/ml, and 2.5 μg/ml respectively. Combined sub-inhibitory concentrations of savirin and antibiotics showed significant enhanced antibacterial and antibiofilm activity compared with that of each alone (Figure 2). The fractional inhibitory concentration (FIC) index values for all three savirin and antibiotics combination ranged from 0.75 to 2 indicating an additive effect. The sub-inhibitory concentrations of savirin and antibiotics were chosen because they have no/minimal inhibitory activity when used alone such that enhanced combined inhibitory activity could be observed.
FIGURE 2.
Inhibition of planktonic (A,B,C) and biofilm (D,E,F) growth by combined savirin and antibiotics (cefazolin, rifampicin, and vancomycin) sub-inhibitory concentrations compared with savirin and antibiotics alone (*** = p < 0.001, ** = p < 0.01, * = p < 0.05). Savirin concentration used in this experiment was sub-inhibitory. Savirin in combination with antibiotics showed enhanced antibacterial and antibiofilm activity against S. aureus compared with savirin alone. Experiments were performed in triplicates (N = 3) and data are presented as mean ± standard deviation (SD) with error bars indicating SD.
Effect of savirin on the expression of S. aureus biofilm-related genes
In the TUH_MSSA_01 strain, savirin downregulated all the biofilm-related genes tested significantly (> 2-fold) in comparison with the untreated positive control (Figure 3).
FIGURE 3.
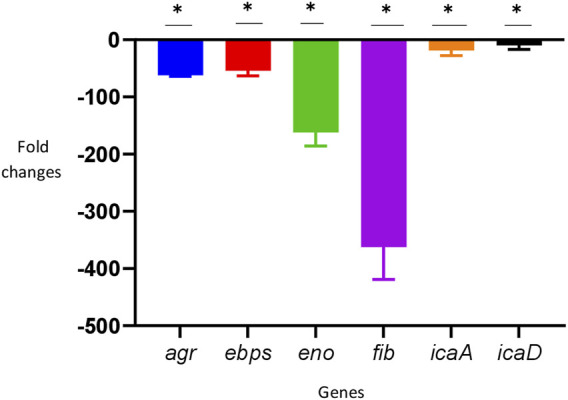
Downregulation of biofilm-related genes by savirin after 8 h of culture (* = downregulated > 2-fold). The reference gene used was fema and the experiment was performed in triplicate (N = 3). To determine the effect of savirin (10 ug/ml) treatment on the S. aureus biofilm-related genes, comparative Ct (ΔΔCt) method was used and the results were expressed as mean fold changes ± standard deviation (SD) in comparison with savirin diluent (0.02% Dimethyl Sulphoxide) treated control. The error bars indicate SD.
Effect of savirin and/or cefazolin treatment on bacterial concentrations on K-wire implants and peri-prosthetic joint tissues
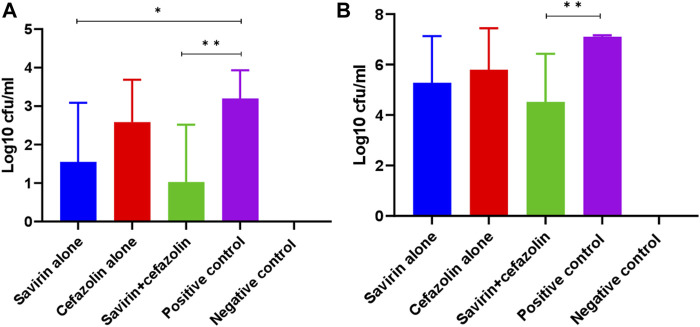
In this study, cefazolin showed better in-vitro activity when combined with savirin compared with other antibiotics tested. Additionally, this is the most common prophylactic antibiotic used in arthroplasty surgery. Therefore, cefazolin was chosen to use in the animal experiment over other antibiotics tested in-vitro. On animal well-being parameters testing, we did not report any adverse effects with the savirin concentration used in this study. Savirin significantly reduced bacterial counts on K-wires removed from the femur of mice with experimentally-induced prosthesis-associated septic arthritis in comparison with the PBS treated control (log10 cfu/ml, 3.2 versus 1.6) (p < 0.05). Similarly, savirin plus cefazolin reduced bacterial counts on both implants (log10 cfu/ml, 3.2 versus 1) and peri-prosthetic tissues (log10 cfu/ml, 7.1 versus 4.5) in comparison with the PBS treated control (p < 0.01) (Figure 4). The absence of an effect of cefazolin alone given on day 7 is keeping with persistence of S. aureus infection due to biofilm, indicating that this antibiotic failed to cure established biofilm.
FIGURE 4.
Bacterial counts on (N = 8) implant (A) and periprosthetic tissue (B) of different mice groups on day 14 post-surgery (** = p < 0.01, * = p < 0.05). Savirin alone reduced bacterial concentration on K-wire, while savirin plus cefazolin reduced bacterial concentration on both K-wire and periprosthetic tissue. The data are presented as mean log10 cfu/ml ± standard deviation (SD). The error bars indicate SD.
Discussion
Savirin showed the in-vitro antibacterial and antibiofilm activity, potentiated the in-vitro activity of selected antibiotics (cefazolin, rifampicin, and vancomycin) against S. aureus, and downregulated all the biofilm-related genes tested. In the PJI mouse model, this molecule prevented S. aureus infection.
This study reported the MBC and MIC of savirin for S. aureus to be 40 μg/ml and 20 μg/ml respectively. An earlier study also reported the direct in-vitro antibacterial activity of savirin (MIC = 36.8 μg/ml) (Mahdally et al., 2021). However, the mechanism of the action is unknown.
The inhibitory roles of savirin (5 μg/ml) against the agr quorum sensing system and a few other AgrA regulated genes, hla, psm alpha, pvl (lukS), have been reported previously (Sully et al., 2014). However, the same study showed no effect of savirin (5 μg/ml) treatment in the expression of other biofilm-related genes by microarray analysis (Sully et al., 2014). Consequently, inhibition of agr, a gene responsible for biofilm dispersal in S. aureus, by savirin (5 μg/ml) would have been expected to enhance biofilm formation (Vuong et al., 2000). However, while 5 μg/ml savirin had no effect, 10 μg/ml showed significant antibiofilm activity in this study. Downregulation of other biofilm-related genes might have negated the effect of agr disruption in S. aureus biofilm. Additionally, agr has strain-specific roles in staphylococcal biofilm formation and dispersal, and agr disruption might have increased, decreased, or no effect in biofilm formation in different strains (O'Neill et al., 2007; Yarwood and Schlievert, 2003). The discrepancy between the results of the previous and this study might be related to the higher savirin (10 μg/ml) concentration used in this study, and the different S. aureus strains and growth conditions used. The ebps, eno, and fib genes encode cell surface associated proteins and promote S. aureus adherence and colonization (Hartford et al., 2001; Downer et al., 2002; Tristan et al., 2003; Carneiro et al., 2004), while icaA and icaD induce bacterial slime production (Arciola et al., 2006). Therefore these genes are essential for the initial attachment of planktonic S. aureus cells to biofilm maturation and their downregulation may inhibit biofilm formation. In this study, savirin downregulated the ebps, eno, and fib genes more significantly compared with the icaA and icaD genes. Savirin perhaps prevents biofilm formation mainly by inhibiting S. aureus initial adherence and colonization then followed by prevention of extracellular matrix production. However, to determine the extent to which each genes' downregulation affected the biofilm formation, further studies using individual gene mutants strains are needed. In the prosthetic joint infection mouse model, savirin significantly reduced bacterial counts on K-wires and savirin plus cefazolin reduced bacterial counts on both implants and peri-prosthetic tissues in comparison with the PBS treated control. This indicates that savirin alone has in-vivo antibiofilm activity probably due to the prevention of S. aureus adherence to the K-wire prosthesis but no antibacterial activity. Savirin, instead, disarmed the bacteria by inhibiting biofilm, which were then cleared partially by cefazolin used on day 7. Savirin prevented the adherence of S. aureus to k-wire and monolayer formation by inhibiting the ebps, eno, and fib genes followed by further prevention of microcolony formation and biofilm maturation by inhibiting ica locus. There was no significant reduction in the bacterial counts in both the implants and tissues due to savirin plus cefazolin treatment compared with savirin alone treatment. This is inconsistent with the in-vitro results, where savirin plus cefazolin had significantly enhanced activity compared with savirin alone. It may be that the in-vivo diminished effect of savirin in our study relates to its rapid elimination before administering cefazolin to mice, leading to S. aureus biofilm formation to which cefazolin has limited activity. None of the treatments used in mice sterilized the implant or tissue infection even though the drugs showed complete in-vitro growth inhibition. This was probably due to the sub-inhibitory concentrations of savirin and cefazolin, to which S. aureus was being exposed in-vivo. However, more animal studies to determine the concentrations of savirin and cefazolin in the blood or knee joint tissues of mice, at different time points, are needed to reach definitive conclusions in this regard. This could also help to determine the optimal dose to sterilize infections in the mouse model.
There is only one other study that reported the influence of savirin on the prevention and treatment of S. aureus skin and subcutaneous infections in mouse models (Sully et al., 2014). In the previous study, savirin reduced infection even when administered 24–48 h post infection establishment implying its effectiveness against established S. aureus mature biofilms (Sully et al., 2014). Savirin doses of 5 µg and 10 µg were used to prevent and treat skin and subcutaneous tissue infections induced with S. aureus infectious doses 2×107 to 4 × 107 cfu (Sully et al., 2014). In this study, a higher savirin dose (40 µg) but a lower S. aureus infective dose (500 cfu) than the previous study was used and confirmed that savirin prevented infection. The infective dose used in the PJI model was chosen to establish a chronic septic arthritis, while the savirin dose was chosen because of theoretical concerns of reduced penetration into joints or bones.
Since the detailed animal toxicity profile, pharmacodynamics, and pharmacokinetics of savirin are not known, this study was unable to use higher savirin doses through different routes that might have sterilized the infection. Dose and toxicity finding studies including pharmacodynamics and pharmacokinetics studies are needed to allow for further animal model experiments. Testing of savirin in large animal prosthetic joint infection models that use materials and techniques used in a modern arthroplasty surgery is also recommended. These large animal models can better represent the real human infection pathogenesis compared with the simple mouse model used in this study.
S. aureus did not develop resistance against low concentration of savirin (5 μg/ml) in comparison with antibiotics (Sully et al., 2014). At 5 μg/ml, savirin does not directly inhibit bacteria but acts as a quorum sensing (QS) inhibitor and disarms bacteria exerting low selection pressure (Pan and Ren, 2009). However, the possibility of quorum sensing inhibitor resistance development among Gram-negative bacteria has been postulated (Defoirdt et al., 2010). Additionally, induction of dysfunctional agr has been reported, therefore the development of savirin (5 μg/ml) resistance through selection of agr dysfunctional mutants or stimulation of drug efflux requiring higher savirin concentration is possible (Somerville et al., 2002; Sully et al., 2014). Savirin’s binding site to AgrA includes a known mutation in agrA in human isolates mainly in strains colonizing the nose before the initiation of infection (Smyth et al., 2012). These S. aureus agrA mutant strains have been shown not to be efficiently transmitted between patients (Shopsin et al., 2010). With this information in mind it may be that agrA mutant S. aureus strains would not be a serious problem particularly in relation to PJI. However, there may be the possibility of resistance development of S. aureus against the direct inhibitory higher concentration of savirin and this needs to be investigated.
This study also explored the in-vitro antibacterial and antibiofilm activity of savirin against S. epidermidis (MIC = 40 μg/ml) and MRSA (MBC = 40 μg/ml, MIC = 20 μg/ml). Savirin was not active against P. aeruginosa, vancomycin resistant Enterococcus, and Klebsiella pneumoniae (detailed data not shown). These data indicate that savirin is, more broadly, an anti-staphylococcal agent with activity against both planktonic and biofilm growth forms. However, in the previous study no effect of savirin has been reported against S. epidermidis (Sully et al., 2014). This difference between results of the previous and this study might again be attributed to the higher concentration of savirin used in this study, and the different bacterial strains and growth conditions used in the two studies.
From our results, it can be concluded that savirin should be considered for the development of an adjuvant therapy for the prevention of S. aureus PJI. This study lays a foundation for studying this molecule for the prevention and treatment of S. aureus PJI.
Acknowledgments
We presented these data and the abstract has been published as conference proceeding at the Australian Society For Microbiology National Meeting 2021, ASM MIM conference 2021, and ASMR QLD Symposium 2021.
Data availability statement
The original contributions presented in the study are included in the article/supplementary material, further inquiries can be directed to the corresponding author.
Ethics statement
The animal study was reviewed and approved by James Cook University Animal Ethics Committee.
Author contributions
NP conceptualized the study, performed the lab work, analyzed the data, and prepared the first draft of this manuscript. SM-H helped in the lab work, revised the manuscript, and gave critical comments to improve the manuscript. CR and JW supervised the study, revised the manuscript, and gave critical comments to improve the manuscript. DE conceptualized and supervised the study, substantially revised the manuscript, and gave critical comments to improve the manuscript.
Funding
This study was supported by Townsville University Hospital Study Education, and Research Trust Account (SERTA) funding.
Conflict of interest
NP is a recipient of a James Cook University Postgraduate Research Scholarship that covers tuition fee and living expenses.
The remaining authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Publisher’s note
All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article, or claim that may be made by its manufacturer, is not guaranteed or endorsed by the publisher.
References
- Alhashimi M., Mayhoub A., Seleem M. N. (2019). Repurposing salicylamide for combating multidrug resistant Neisseria gonorrhoeae . Antimicrob. Agents Chemother. 63, e01225–19. 10.1128/AAC.01225-19 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Arciola C. R., Campoccia D., Baldassarri L., Donati M. E., Pirini V., Gamberini S., et al. (2006). Detection of biofilm formation in Staphylococcus epidermidis from implant infections. Comparison of a PCR method that recognizes the presence of ica genes with two classic phenotypic methods. J. Biomed. Mat. Res. A 76, 425–430. 10.1002/jbm.a.30552 [DOI] [PubMed] [Google Scholar]
- Bernthal N. M., Stavrakis A. I., Billi F., Cho J. S., Kremen T. J., Simon S. I., et al. (2010). A mouse model of post-arthroplasty Staphylococcus aureus joint infection to evaluate in vivo the efficacy of antimicrobial implant coatings. PLoS One 5, e12580. 10.1371/journal.pone.0012580 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Carneiro C. R., Postol E., Nomizo R., Reis L. F., Brentani R. R. (2004). Identification of enolase as a laminin binding protein on the surface of Staphylococcus aureus . Microbes Infect. 6, 604–608. 10.1016/j.micinf.2004.02.003 [DOI] [PubMed] [Google Scholar]
- Davis J. (2016). Prosthetic joint infections in Australia and New Zealand: The first 275 patients from the PIANO (prosthetic joint infection in Australia and New Zealand observational) study. Orthop. Proc. 98-B, 58. The British editorial society of bone & joint surgery. 10.1302/1358-992X.98BSUPP_23.EBJIS2016-058 [DOI] [Google Scholar]
- Defoirdt T., Boon N., Bossier P. (2010). Can bacteria evolve resistance to quorum sensing disruption? PLoS Pathog. 6, e1000989. 10.1371/journal.ppat.1000989 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Del Pozo J. L., Patel R. (2009). Clinical practice. Infection associated with prosthetic joints. N. Engl. J. Med. 361, 787–794. 10.1056/NEJMcp0905029 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Downer R., Roche F., Park P. W., Mecham R. P., Foster T. J. (2002). The elastin binding protein of Staphylococcus aureus (EbpS) is expressed at the cell surface as an integral membrane protein and not as a cell wall-associated protein. J. Biol. Chem. 277, 243–250. 10.1074/jbc.M107621200 [DOI] [PubMed] [Google Scholar]
- Ferreira F. A., Souza R. R., de Sousa Moraes B., de Amorim Ferreira A. M., Américo M. A., Fracalanzza S. E., et al. (2013). Impact of agr dysfunction on virulence profiles and infections associated with a novel methicillin resistant Staphylococcus aureus (MRSA) variant of the lineage ST1-SCCmec IV. BMC Microbiol. 13, 93. 10.1186/1471-2180-13-93 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Francois P., Pittet D., Bento M., Pepey B., Vaudaux P., Lew D., et al. (2003). Rapid detection of methicillin resistant Staphylococcus aureus directly from sterile or nonsterile clinical samples by a new molecular assay. J. Clin. Microbiol. 41, 254–260. 10.1128/jcm.41.1.254-260.2003 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hartford O. M., Wann E. R., Höök M., Foster T. J. (2001). Identification of residues in the Staphylococcus aureus fibrinogen-binding MSCRAMM clumping factor A (ClfA) that are important for ligand binding. J. Biol. Chem. 276, 2466–2473. 10.1074/jbc.M007979200 [DOI] [PubMed] [Google Scholar]
- Kot B., Sytykiewicz H., Sprawka I. (2018). Expression of the biofilm-associated genes in methicillin resistant Staphylococcus aureus in biofilm and planktonic conditions. Int. J. Mol. Sci. 19, 3487. 10.3390/ijms19113487 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Livak K. J., Schmittgen T. D. (2001). Analysis of relative gene expression data using real time quantitative PCR and the 2(-Delta Delta C(T)) Method. Methods 25, 402–408. 10.1006/meth.2001.1262 [DOI] [PubMed] [Google Scholar]
- Ma C. Y., Lu Y. D., Bell K. L., Wang J. W., Ko J. Y., Wang C. J., et al. (2018). Predictors of treatment failure after 2-stage reimplantation for infected total knee arthroplasty: A 2- to 10-year follow-up. J. Arthroplasty 33, 2234–2239. 10.1016/j.arth.2018.02.007 [DOI] [PubMed] [Google Scholar]
- Mahdally N. H., George R. F., Kashef M. T., Al-Ghobashy M., Murad F. E., Attia A. S. (2021). Staquorsin: A novel Staphylococcus aureus agr-mediated quorum sensing inhibitor impairing virulence in vivo without notable resistance development. Front. Microbiol. 12, 700494. 10.3389/fmicb.2021.700494 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Meletiadis J., Pournaras S., Roilides E., Walsh T. J. (2010). Defining fractional inhibitory concentration index cut offs for additive interactions based on self-drug additive combinations, Monte Carlo simulation analysis, and in vitro-in vivo correlation data for antifungal drug combinations against Aspergillus fumigatus . Antimicrob. Agents Chemother. 54, 602–609. 10.1128/AAC.00999-09 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Morris J. L., Letson H. L., Elliott L., Grant A. L., Wilkinson M., Hazratwala K., et al. (2019). Evaluation of bacteriophage as an adjunct therapy for treatment of peri-prosthetic joint infection caused by Staphylococcus aureus . PloS one 14 (12), e0226574. 10.1371/journal.pone.0226574 [DOI] [PMC free article] [PubMed] [Google Scholar]
- O'Neill E., Pozzi C., Houston P., Smyth D., Humphreys H., Robinson D. A., et al. (2007). Association between methicillin susceptibility and biofilm regulation in Staphylococcus aureus isolates from device-related infections. J. Clin. Microbiol. 45, 1379–1388. 10.1128/JCM.02280-06 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pan J., Ren D. (2009). Quorum sensing inhibitors: A patent overview. Expert Opin. Ther. Pat. 19, 1581–1601. 10.1517/13543770903222293 [DOI] [PubMed] [Google Scholar]
- Peel T. N., Cheng A. C., Buising K. L., Choong P. F. (2012). Microbiological aetiology, epidemiology, and clinical profile of prosthetic joint infections: Are current antibiotic prophylaxis guidelines effective? Antimicrob. Agents Chemother. 56, 2386–2391. 10.1128/AAC.06246-11 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ricciardi B. F., Muthukrishnan G., Masters E., Ninomiya M., Lee C. C., Schwarz E. M. (2018). Staphylococcus aureus evasion of host immunity in the setting of prosthetic joint infection: Biofilm and beyond. Curr. Rev. Musculoskelet. Med. 11 (3), 389–400. 10.1007/s12178-018-9501-4 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Shopsin B., Eaton C., Wasserman G. A., Mathema B., Adhikari R. P., Agolory S., et al. (2010). Mutations in agr do not persist in natural populations of methicillin resistant Staphylococcus aureus . J. Infect. Dis. 202, 1593–1599. 10.1086/656915 [DOI] [PubMed] [Google Scholar]
- Singh A. K., Yadav S., Chauhan B. S., Nandy N., Singh R., Neogi K., et al. (2019). Classification of clinical isolates of Klebsiella pneumoniae based on their in vitro biofilm forming capabilities and elucidation of the biofilm matrix chemistry with special reference to the protein content. Front. Microbiol. 10, 669. 10.3389/fmicb.2019.00669 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Smyth D. S., Kafer J. M., Wasserman G. A., Velickovic L., Mathema B., Holzman R. S., et al. (2012). Nasal carriage as a source of agr-defective Staphylococcus aureus bacteremia. J. Infect. Dis. 206, 1168–1177. 10.1093/infdis/jis483 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Somerville G. A., Beres S. B., Fitzgerald J. R., DeLeo F. R., Cole R. L., Hoff J. S., et al. (2002). In vitro serial passage of Staphylococcus aureus: Changes in physiology, virulence factor production, and agr nucleotide sequence. J. Bacteriol. 184, 1430–1437. 10.1128/jb.184.5.1430-1437.2002 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sully E. K., Malachowa N., Elmore B. O., Alexander S. M., Femling J. K., Gray B. M., et al. (2014). Selective chemical inhibition of agr quorum sensing in Staphylococcus aureus promotes host defense with minimal impact on resistance. PLoS Pathog. 10, e1004174. 10.1371/journal.ppat.1004174 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tristan A., Ying L., Bes M., Etienne J., Vandenesch F., Lina G. (2003). Use of multiplex PCR to identify Staphylococcus aureus adhesins involved in human hematogenous infections. J. Clin. Microbiol. 41, 4465–4467. 10.1128/jcm.41.9.4465-4467.2003 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Vuong C., Saenz H. L., Gotz F., Otto M. (2000). Impact of the agr quorum sensing system on adherence to polystyrene in Staphylococcus aureus . J. Infect. Dis. 182, 1688–1693. 10.1086/317606 [DOI] [PubMed] [Google Scholar]
- Yarwood J. M., Schlievert P. M. (2003). Quorum sensing in Staphylococcus infections. J. Clin. Invest. 112, 1620–1625. 10.1172/JCI20442 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zimmerli W., Trampuz A., Ochsner P. E. (2004). Prosthetic joint infections. N. Engl. J. Med. 351, 1645–1654. 10.1056/NEJMra040181 [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
The original contributions presented in the study are included in the article/supplementary material, further inquiries can be directed to the corresponding author.