Abstract
Maize early endosperm development is initiated in coordination with elimination of maternal nucellar tissues. However, the underlying mechanisms are largely unknown. Here, we characterize a major quantitative trait locus for maize kernel size and weight that encodes an EXPANSIN gene, ZmEXPB15. The encoded β-expansin protein is expressed specifically in nucellus, and positively controls kernel size and weight by promoting nucellus elimination. We further show that two nucellus-enriched transcription factors (TFs), ZmNAC11 and ZmNAC29, activate ZmEXPB15 expression. Accordingly, these two TFs also promote kernel size and weight through nucellus elimination regulation, and genetic analyses support their interaction with ZmEXPB15. Importantly, hybrids derived from a ZmEXPB15 overexpression line have increased kernel weight, demonstrates its potential value in breeding. Together, we reveal a pathway modulating the cellular processes of maternal nucellus elimination and early endosperm development, and an approach to improve kernel weight.
Subject terms: Agricultural genetics, Plant molecular biology, Plant breeding, Plant development
Current studies of maize kernel development mostly focus on endosperm and embryo development, and little is known about the control of the nucellus tissue. Here, the authors report a NAC-EXPB15 pathway that regulate maize kernel size by modulating nucellus elimination and early endosperm development.
Introduction
Maize (Zea mays) is one of the most important crops with increasing global demand for food and renewable energy. Kernel size and weight are key components for crop yield-component and are also important domestication and breeding traits1. To dissect the genetic basis of maize kernel-related traits, including kernel length, width and thickness, hundred kernel weight (HKW) and unit weight, hundreds of quantitative trait loci (QTLs) have been identified (www.maizegdb.org/qtl). However, only very few were cloned and characterized. The kernel size-related QTL, qKW9, was recently found to encode a pentatricopeptide repeat (PPR) protein that affects photosynthesis and grain filling2. ZmVPS29, homologous to the Arabidopsis Vacuolar Protein Sorting29 (VPS29), is a candidate kernel morphology QTL, qKM4.08. Overexpression of this gene produces slender kernels, but increases yield per plant3. Quite some kernel-related genes have been cloned from classical mutants with defects in embryo and/or endosperm, such as defective kernel (dek), empty pericarp (emp), and small kernel (smk), and usually develop small and/or lethal kernels with limited potential for application4–8. Kernel traits are achieved through the coordinated growth of three genetically different components: embryo, endosperm, and the surrounding maternal tissues, including nucellus and pericarp9. The maternal nucellar tissue, originating from the ovule, acts as a bridge for apoplastic intercellular exchange between the mother plant and its developing offspring. It also provides an environment for the developing zygote and filial tissue10,11. However, the genetic and molecular regulation of maternal tissues and their impacts on maize kernel development are largely unknown.
Maize kernel development can be divided into three phases: early development, filling and maturation10. At an early stage, the maternal nucellar tissue degenerates by programmed cell death (PCD), and the contents of the dying cells are re-mobilized to feed the developing embryo and endosperm12,13. Interruption in nucellus PCD could lead to defective endosperm development. In Arabidopsis, the prolonged existence of nucellus due to defective degeneration in transparent testa 16 (tt16) mutant seeds leads to a loss or misplaced chalazal endosperm cyst, revealing the importance of nucellus elimination and its feedback role in timing of endosperm development11. It has thus been proposed that endosperm and nucellus undergo antagonistic development at the early stage of Arabidopsis seed development. The rice TT16 ortholog, MADS29, is expressed in the nucellus and regulates nucellus PCD by promoting the expression of PCD-related genes responsible for the cleavage of storage proteins14–16. The antagonistic development of nucellus and endosperm also occurs in rice, as suppression of MADS29 expression in nucellus impairs endosperm growth and starch accumulation15–18. However, whether and how agonistic development process of nucellar tissue can be genetically manipulated to promote the endosperm growth and ultimately increase kernel weight remains unexplored.
In this work, we identify ZmEXPB15 as a major QTL for kernel size and weight in maize, and find that it encodes an expansin protein that controls kernel size and weight through coordinating nucellus elimination and early endosperm development. The expression of ZmEXPB15 is under the direct control of two nucellus-enriched genes, ZmNAC11/ZmNAC29, which control kernel size by a similar mechanism. Further, increased kernel weight of hybrids overexpressing ZmEXPB15 demonstrates its potential application value of maize breeding.
Results
HKW9 acts maternally to control kernel size and weight
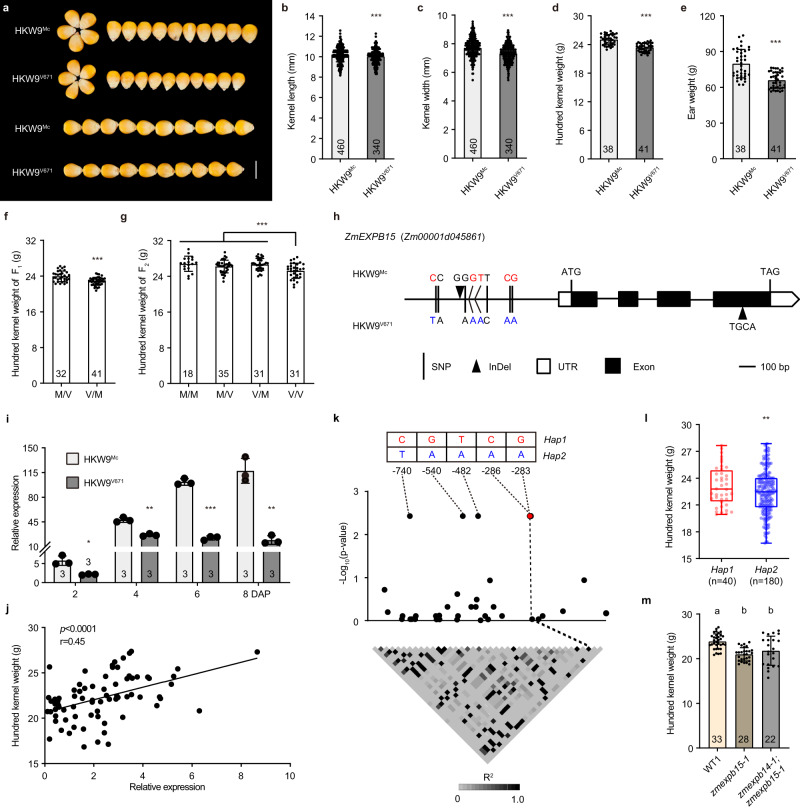
A major QTL for kernel size and weight, designated here as hundred kernel weight 9 (HKW9), was previously mapped to chromosome 9 (9.03-9.04 bin), using an F2:3 population derived from two maize elite inbred lines with different kernel size, V671 and Mc19. To precisely estimate the genetic effect of HKW9 on kernel traits, we generated near-isogenic lines (NILs), HKW9Mc and HKW9V671, harboring the HKW9 locus from Mc and V671, respectively (Fig. 1a). Over successive years of field trials, the large-kernel NIL, HKW9Mc showed a significant increase in HKW by 3.8%, in kernel length by 4.2%, in kernel width by 5.9% and in ear weight by 17.6%, compared to small-kernel NIL, HKW9V671 (Fig. 1b–e). These results demonstrated that the HKW9 QTL was responsible for multiple maize kernel related traits. Meanwhile, plant architecture, other ear traits and kernel starch accumulation were not significantly altered (Supplementary Fig. 1), suggesting a specific effect of HKW9 on kernel size.
Fig. 1. HKW9 acts maternally to control kernel size and weight.
a Comparison of mature kernels between HKW9Mc and HKW9V671. Scale bar = 1 cm. b–e Quantitative analysis of kernel length (b), kernel width (c), hundred kernel weight (d) and ear weight (e) between HKW9Mc and HKW9V671. f, g Quantitative analysis of hundred kernel weight (HKW) of F1 kernels (f) from the HKW9Mc/HKW9V671 (M/V) and HKW9V671/HKW9Mc (V/M) plants, and F2 kernels (g) from the HKW9Mc/HKW9Mc (M/M), M/V, V/M and HKW9V671/HKW9V671 (V/V) plants. h Gene structure of the candidate gene, Zm00001d045861 (ZmEXPB15), and the polymorphisms between HKW9Mc and HKW9V671. Eight SNPs (black lines) and one 1-bp InDel (black triangle) in the promoter and one 4-bp InDel (black triangle) in the last exon are shown; white boxes, untranslated regions; black boxes, coding sequences. i Expression of ZmEXPB15 is higher in large kernel NIL HKW9Mc in early developing kernels. All expression levels from three biological repeats were normalized to Actin. j Pearson correlation between ZmEXPB15 expression and the HKW among 75 inbred lines. k The association analysis of ZmEXPB15 promoter variations (−843 to −24 relative to ATG) with HKW. The white to black heat-map shows the pairwise linkage disequilibrium pattern by R2; the genotypes and positions of five significant association loci are shown on the top. l Box-and-whisker plots of HKW for the Hap1 (red box, n = 40) and Hap2 (blue box, n = 180). Each box represents the median and interquartile range, and whiskers extend to maximum and minimum values. m Quantitative analysis of the HKW in zmexpb15-1 single and zmexpb14-1;zmexpb15-1 double mutants compare to the corresponding wild-type (WT1). Number on each column is the sample size. Values in (b–g, i, l, m) are means ± s.d. (standard deviation), and the significance in (b–g, k–m) is estimated by one-way ANOVA, and the significance in (i, j) is estimated by a two-tailed Student’s t test. *P < 0.05, **P < 0.01, ***P < 0.001. The Tukey HSD test is used in (m) and statistical differences (P < 0.05) are indicated by different letters. Source data are provided as a Source Data file.
To ask whether the increased kernel size of HKW9Mc was due to a maternal effect, reciprocal crosses were performed. The HKW of F1 kernels from the large-kernel NIL, HKW9Mc, pollinated by HKW9V671, was significantly larger than that of small-kernel NIL, HKW9V671, pollinated by HKW9Mc (Fig. 1f). In addition, the HKW of HKW9Mc/HKW9V671 and HKW9V671/HKW9Mc F2 kernels was similar to that of HKW9Mc/HKW9Mc F2 kernels and larger than that of HKW9V671/HKW9V671 F2 kernels (Fig. 1g). Together, these observations indicate that the HKW9 QTL regulates kernel weight through a maternal effect.
ZmEXPB15 is the candidate gene for HKW9
The HKW9 QTL maps close to the centromere, which made fine mapping infeasible. We therefore applied transcriptome sequencing to assist in candidate gene isolation using 6 days after pollination (DAP) kernels, the stage when maternal tissues have a significant effect on kernel development. Within the 73-Mb (flanked by markers umc2370 and bnlg1209) QTL mapping region19, 27 of the annotated genes were differentially expressed between the two NILs (Supplementary Data 1), and two of them were highly and specifically expressed in developing seed based on publicly available RNA-seq data (Supplementary Fig. 2)20. These two genes, Zm00001d045792 and Zm00001d045861, both encoding β-expansin, showed high (~98%) similarity in the coding regions and were designated as ZmEXPB14 and ZmEXPB15 hereafter, respectively. By resequencing the coding regions, we found that ZmEXPB14 contained only two nonsymponous mutations in the non-conserved sites, whereas ZmEXPB15 covered a 4-bp deletion in the conserved domain of the last coding exon, leading to a frame shift of the last 60 amino acids and an addition of 24 residues in the C-terminus (Fig. 1h, Supplementary Fig. 3a, b). Thus, our main focus was first on ZmEXPB15. We further detected eight SNPs (at −740, −721, −562, −540, −482, −437, −286 and −283 bp) and one InDel (at −576 bp) within ~1 kb promoter region upstream of the ZmEXPB15 ATG. Consistently, the ZmEXPB15 transcript was found to be differentially expressed between the two NILs from an early stage (~2 DAP) of kernel development, with a higher ZmEXPB15 expression in the large-kernel NIL, HKW9Mc (Fig. 1i). Association analysis showed that ZmEXPB15 expression was positively correlated with HKW (r = 0.45, P < 0.0001, Fig. 1j) in 75 diverse inbred lines (Supplementary Data 2). To further investigate whether the promoter SNPs were responsible for the difference in HKW, we resequenced the ~1 kb promoter region in 220 diverse inbred lines (Supplementary Data 2), and found that five SNPs (at −740, −540, −482, −286 and −283 bp) in the promoter regions significantly associated with HKW (p = 3.73E-03, Fig. 1k, Supplementary Table 1). These five associated sites, which were in complete linkage disequilibrium, formed two haplotypes: one with HKW9Mc genotype and the other with HKW9V671 genotype were named Hap1 and Hap2, respectively (Fig. 1k). Inbred lines carrying Hap1 had higher ZmEXPB15 expression and larger kernels than those carrying Hap2 (Fig. 1l, Supplementary Fig. 4), consistent with the positive correlation of the ZmEXPB15 expression with the HKW.
To further verify the candidate gene for the QTL HKW9, we generated knockout mutants for both ZmEXPB14 and ZmEXPB15 genes using a CRISPR/Cas9 strategy (Supplementary Fig. 5a). Only double mutants (KO1, KO2, KO3) were obtained with two genes being edited together due to their high similarity (Supplementary Fig. 5b). We next separated the zmexpb14 and zmexpb15 mutations by screening a large (>30,000) segregating population due to their close linkage across the centromere. Single zmexpb14 mutant did not show an obvious change in HKW, whereas zmexpb15 decreased the HKW significantly (Fig. 1m, Supplementary Fig. 5c). Importantly, the double zmexpb14;zmexpb15 mutant did not show any enhancement in the HKW difference, compared to the zmexpb15 single mutant in which ZmEXPB14 expression was not altered (Fig. 1m, Supplementary Fig. 5d), indicating that ZmEXPB15 is the major player for the HKW difference. Taken together, our findings suggest that ZmEXPB15 (Zm00001d045861) is the candidate gene for the HKW9 QTL, and positively control kernel weight.
Functional validation of ZmEXPB15
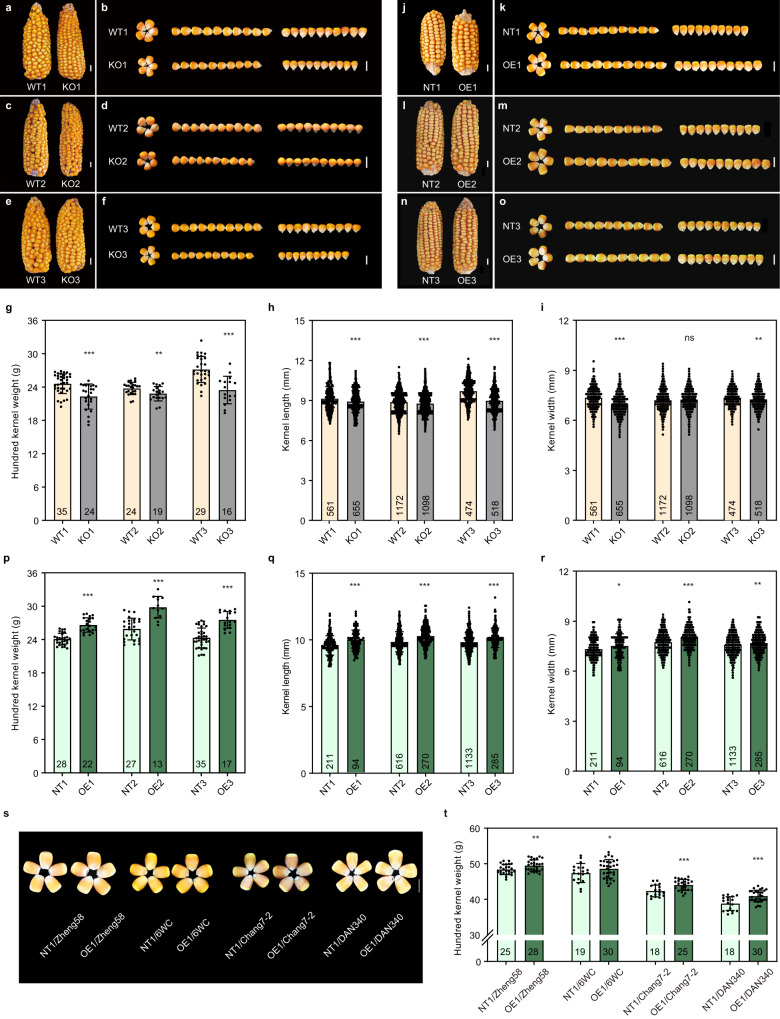
To verify the biological function of ZmEXPB15 in control of kernel size and weight, we investigated three knockout alleles (KO1, KO2, KO3), in which different truncations nearby the ATG led to early frame shifts (Supplementary Fig. 5b). All three alleles had lower HKW and smaller kernel size (Fig. 2a–i) than their corresponding wild-type (WT) plants. To further validate the effect of ZmEXPB15, we overexpressed the ZmEXPB15 large-kernel allele coding sequence using a maize ubiquitin promoter. Three independent overexpression lines (OE1, OE2, OE3) with significantly increased expression level of ZmEXPB15 (Supplementary Fig. 5e) had an increase in HKW and larger kernel length and width, compared to the corresponding non-transgenic lines (Fig. 2j–r). All these results consistently demonstrate that ZmEXPB15 positively controls kernel size and weight in maize.
Fig. 2. ZmEXPB15 positively control kernel size and weight.
a–f Three knockout lines (KO1, KO2 and KO3) with loss-of-function mutations in ZmEXPB15 had smaller kernels than the corresponding wild-type (WT1, WT2 and WT3) in a C01 inbred background. g–i Quantitative analysis of the hundred kernel weight (g), kernel length (h) and kernel width (i) in three knockout lines. j–o Three ZmEXPB15 overexpressing lines (OE1, OE2 and OE3) had larger kernels than their corresponding non-transgenic lines (NT1, NT2 and NT3) in a B104 inbred background. p–r Quantitative analysis of the hundred kernel weight (p), kernel length (q) and kernel width (r) in three overexpressing lines. s Mature kernels of four F1 hybrids derived from overexpression line OE1 and the corresponding control NT1 by crossing to four elite inbred lines (Zheng58, 6WC, Chang7-2, Dan340). t Quantitative analysis of the HKW of four F1 hybrids same as (s). Number on the bottom of each column is the sample size. Values in (g–i, p–r, t) are means ± s.d., and the significance is estimated by the one-way ANOVA. *P < 0.05, **P < 0.01, ***P < 0.001. ns, non-significance. Scale bar = 1 cm in (a–f, j–o, s). Source data are provided as a Source Data file.
Next, we evaluated the potential application of ZmEXPB15 in molecular breeding, by crossing the OE1 overexpression line (as female parent) by four different elite inbred lines. All four F1 hybrids harboring transgenic positive plants had significantly higher HKW, compared to control non-transgenic hybrids (Fig. 2s, t). The increase in HKW ranged from 2.8% to 12.4%, with 6.3% on average. The kernel length and width also increased, although the different hybrids varied in which size trait was altered (Supplementary Fig. 6a, b). In addition, most hybrids did not affect other traits, such as ear length (Supplementary Fig. 6c–e). Consistent with our finding that ZmEXPB15 acts maternally to regulate kernel weight, most reciprocal cross hybrids using OE1 as the male parent did not show a significant change in HKW (Supplementary Fig. 6f). Thus, ZmEXPB15 showed potential application in enhancing kernel weight.
ZmEXPB15 is specifically expressed in nucellus of developing kernel
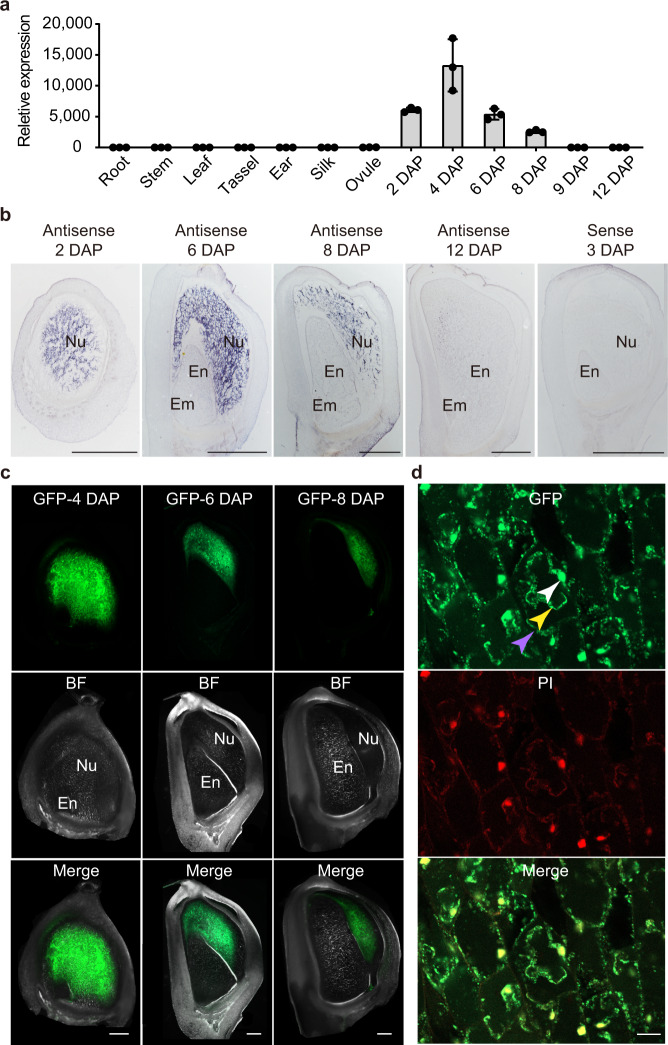
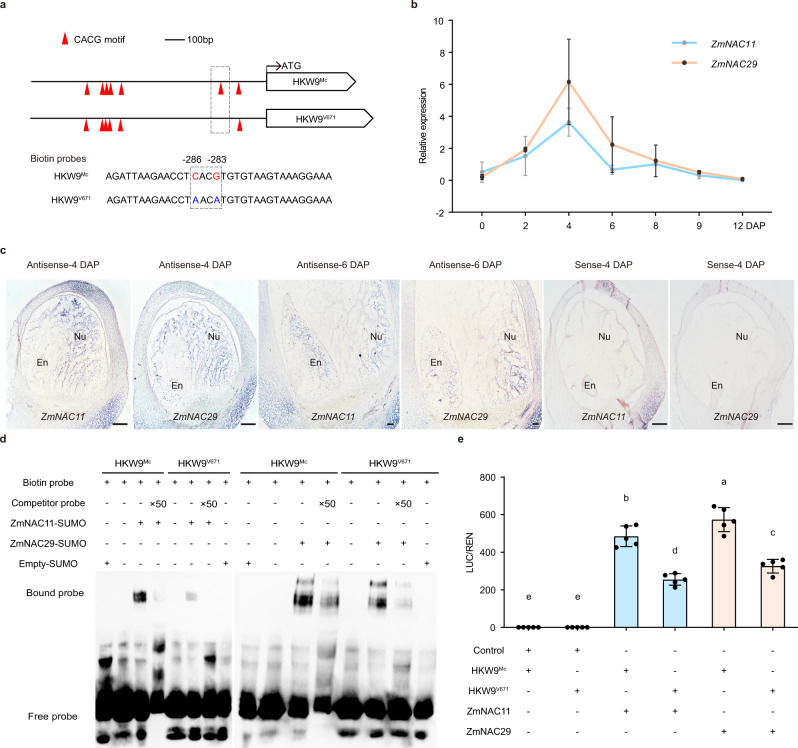
The maternal role of ZmEXPB15 in the control of kernel traits indicated that it might be expressed and function in maternal tissues during early kernel development, and was supported by its high expression in the nucellus in publicly available RNA-seq data (Supplementary Fig. 7)20. This expression profile was confirmed by our real-time quantitative reverse transcription PCR (qRT-PCR) analyses, showing that ZmEXPB15 was exclusively expressed during the early developmental stages of 2–8 DAP, with the highest level at 4 DAP, when nucellar tissue is enriched (Fig. 3a). To investigate the expression pattern more carefully, RNA in situ hybridization using an antisense probe against ZmEXPB15 was performed at 2 to 12 DAP stages, when the maternal nucellar tissue was still present. A specific expression pattern in the nucellus was observed, starting early from 2 DAP and gradually decreasing after 8 DAP, when the endosperm started to rapidly expand (Fig. 3b). We also generated a transgenic line harboring ZmEXPB15 fused with the green fluorescent protein (GFP) under its native promoter to detect its subcellular localization. A strong GFP signal was observed in the nucellus (Fig. 3c) and the signal gradually decreased, with faint florescence at 8 DAP, consistent with its dynamic transcript distribution shown by in situ hybridization. After plasmolysis, the GFP signal was clearly detected in the cell wall, cytoplasm and also nucleus (Fig. 3d). Our results demonstrate that ZmEXPB15 was specifically expressed in nucellus, and explain its maternal effect on kernel development.
Fig. 3. ZmEXPB15 accumulates specifically in nucellus of developing kernels.
a qRT-PCR analysis of ZmEXPB15 in root, stem, leaf, tassel, ear, silk and developing kernels at 0 (ovule) to 12 DAP. All expression levels from three biological repeats were normalized to Actin. Values are means ± s.d. b In situ localization of ZmEXPB15 mRNA in developing kernels at 2 to 12 DAP. Positive signals (shown in purple) are clearly restricted to the nucellus at 2 to 8 DAP (first three panels) using the ZmEXPB15 antisense probe. No signal is observed in the section of a 12-DAP kernel (fourth panel). The control was performed using 3-DAP kernel with a ZmEXPB15 sense probe (last panel). c Localization of ZmEXPB15-GFP in developing kernels at 4, 6 and 8 DAP from proZmEXPB15:ZmEXPB15-GFP transgenic plants. The ZmEXPB15-GFP signal was specifically detected in the nucellus. BF bright-field image, Merge merge of GFP and BF images. Nu nucellus, En endosperm, Em embryo. d ZmEXPB15-GFP expression was observed in the cell wall (purple arrow), cytoplasm (yellow arrow) and nucleus (white arrow) of the nucellar cells in 4-DAP kernels. PI, propidium iodide staining. Merge, merge of GFP and PI images. The experiments in (b–d) were repeated two times with a similar result. Scale bar = 1 mm in (b); 500 µm in (c); 20 µm in (d). Source data are provided as a Source Data file.
ZmEXPB15 promotes nucellus cell expansion and elimination
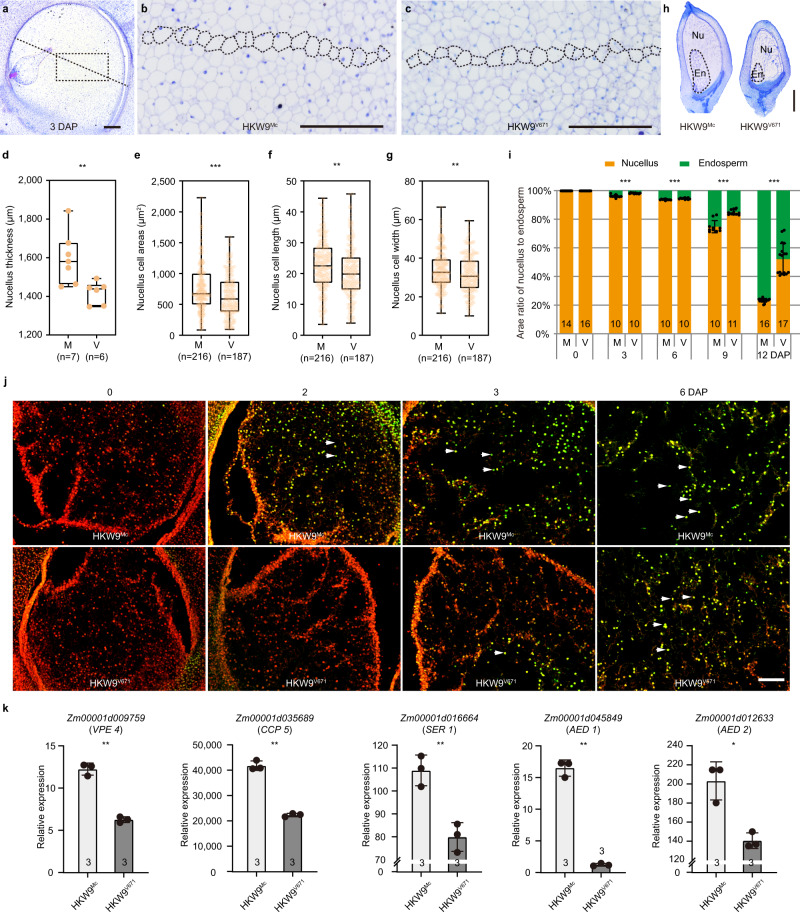
To investigate how ZmEXPB15 affects the nucellus development, we compared the nucellus size of the two NILs, and found that the nucellus in the large-kernel line HKW9Mc with higher ZmEXPB15 expression was thicker than that in HKW9V671 (Fig. 4a). The cell area, cell length and width of nucellar cells in HKW9Mc were significantly larger than those in HKW9V671, while the cell number was compatible (Fig. 4b–g; Supplementary Fig. 8a). These observations indicated that ZmEXPB15 could promote nucellus cell expansion, leading to a larger nucellus volume. During early kernel development, nucellar tissue undergoes a degenerative process recognized as programmed cell death (PCD), accompanied by the expansion of endosperm tissues21. These two antagonistic processes occur in a coordinated manner22. To investigate how ZmEXPB15 coordinates the nucellus and endosperm development, the areas of both tissues were measured in the two NILs from 0 to 12 DAP. There was no difference at 0 DAP with 100% area of nucellus, however, the ratio of nucellus to endosperm decreased differentially between the two NILs starting from 3 DAP, with a significantly lower ratio in the large-kernel HKWMc line than that in the small-kernel HKWV671 line (Fig. 4h, i). At 12 DAP, the ratio of nucellus to endosperm area in HKW9Mc was around 25%, in contrast to around 50% in HKWV671, suggesting a faster nucellus elimination and endosperm expansion present in HKW9Mc that showed a higher ZmEXPB15 expression level (Fig. 4i). Consistently, the same effect of ZmEXPB15 on the coordinated nucellus and endosperm development process was observed in the ZmEXPB15 knockout (KO1) line with a higher ratio of nucellus to endosperm area than the control, and also in the overexpression (OE1) line which showed the opposite (Supplementary Fig. 8b). These results suggested that ZmEXPB15 was involved in the nucellus elimination which coordinates endosperm development.
Fig. 4. Nucellus cell expansion and elimination is delayed in the small-kernel HKW9V671 line.
a Longitudinal semi-thin section of 3-DAP kernels used for nucellus size measurement. The black dotted lines indicate the region where nucellus thickness was measured. b, c Enlarged image of the black boxed region in (a) of HKW9Mc (b) and HKW9V671 (c). A row of nucellar cells are framed by black-dots in (b, c) to measure the cell size. d–g Quantification of nucellus thickness (d), cell areas (e), cell length (f) and cell width (g) of HKW9Mc (M) and HKW9V671 (V). The samples in M and V are 7 and 6 in (d), and 216 and 187 in (e–g), respectively. The sample numbers in (e–g) refer to all cells counted along the black dotted lines from all independent samples. Each box represents the median and interquartile range, and whiskers extend to maximum and minimum values. h Toluidine blue stained sections of NILs kernels at 9 DAP. Nu, nucellus; En, endosperm. i Ratio of nucellus (orange box) to endosperm area (green box) in developing kernels of M and V. j TUNEL assays of the nucellus in the developing kernels from NILs. Nuclei were prestained with propidium iodide (PI) seen as red fluorescence. The green and yellow fluorescence correspond to the TUNEL-positive nuclei (partly marked white arrows). k qRT-PCR analysis of PCD-related genes in the NILs kernels at 4 DAP. Zm00001d009759 for vacuolar processing enzymes 4 (VPE4); Zm00001d035689 for cysteine protease 5 (CCP5); Zm00001d016664 for serine proteases 1 (SER1); Zm00001d045849 for aspartyl protease 1 (AED1) and Zm00001d012633 for aspartyl protease 2 (AED2). All expression levels from three biological repeats were normalized to Actin. Number on the bottom of each column is the sample size. Values are means ± s.d., two-tailed Student’s t test. *P < 0.05, **P < 0.01, ***P < 0.001. Scale bar = 200 μm in (a–c); 1 mm in (h); 500 µm in (j). Source data are provided as a Source Data file.
To confirm the speculation above, we performed a terminal deoxynucleotidyl transferase dUTP nick-end labeling (TUNEL) assay to monitor how the nucellus elimination process was regulated by ZmEXPB15. In the large-kernel line HKWMc in which ZmEXPB15 was highly expressed, the TUNEL-positive nuclei were detected in the nucellar cells of as early as 2 DAP, and increased at later stages. In contrast, the TUNEL signals in the small-kernel line HKWV671were almost undetectable at 3 DAP (Fig. 4j), although the distribution of TUNEL-positive nuclei became undistinguishable at 6 DAP between the two NILs (Fig. 4j). Further, the ZmEXPB15 knockout (KO1) line showed an obviously delayed appearance of the TUNEL-positive nuclei, and the ZmEXPB15 overexpression (OE1) line showed the opposite, compared to the controls (Supplementary Fig. 8c, d). These results indicated that ZmEXPB15 could promote nucellus PCD process. In addition, Evan’s blue staining, as another more representative marker for cell death, showed that the nucellar cells of large-kernel line HKWMc had significantly deeper staining than those in small-kernel line HKWV671 at the stage of 4 and 6 DAP (Supplementary Fig. 8e). Consistently, the ZmEXPB15-overexpressed line (OE1) also stained deeper than the control as early as 2 DAP (Supplementary Fig. 8f). These observations revealed that ZmEXPB15 promoted the PCD process and cell death of nucellus to ensure a timely endosperm development, which was likely responsible for the increased kernel size and weight.
Seed developmental PCD involves diverse classes of proteases, including cysteine proteases, serine proteases, and aspartic proteases23. We next compared the expression of some annotated protease genes between the two NILs, including vacuolar processing enzymes 4 (VPE4) homologous to barley VPE4, which has been known to execute programmed cell death in barley pericarp24,25, cysteine protease 5 (CCP5) homologous to Arabidopsis xylem cysteine proteinase 1 (XCP1), which promote catabolism in tracheary elements during xylogenesis26, serine proteases1 (SER1) homologous to Arabidopsis senescence-associated gene SAG15, which functions in protein degradation in senescing chloroplasts27, aspartyl protease1 (AED1) homologous to Arabidopsis aspartyl protease 1 (AED1), which may degrade apoplastic proteins28. All these potentially PCD-related genes were expressed at significantly lower levels in 4-DAP kernels of HKWV671 than in HKWMc (Fig. 4k). This result is in line with the delayed PCD in HKWV671 in which ZmEXPB15 was expressed at a lower level. Taken all together, we propose that ZmEXPB15 positively controls early kernel development by promoting the nucellus PCD and coordinating the antagonistic development between nucellus and endosperm.
ZmNAC11 and ZmNAC29 may activate the expression of ZmEXPB15
Our association analysis and the correlation of ZmEXPB15 expression level with kernel weight prompted us to speculate that specific cis-elements might regulate the differences in ZmEXPB15 expression between the two NILs. We thus investigated the promoter region of ZmEXPB15 and found that several CACG motifs, known binding sites for NAC transcription factors (TFs)29,30, were enriched (Fig. 5a). Two SNPs at positions of −286 bp and −283 bp upstream of the ATG resided in one CACG motif, which was also associated with HKW (Fig. 1k). To search for the potential NAC TFs binding to the promoter of ZmEXPB15, we looked at nucellus transcriptome data from 8-DAP maize kernels31, and found that ZmNAC11 and ZmNAC29, two close paralog genes, were highly expressed in nucellus (Supplementary Fig. 9). Our qRT-PCR and mRNA in situ analysis confirmed that these two NAC TFs were predominantly expressed in the nucellus and reached a peak in 4-DAP kernels, a similar expression pattern to ZmEXPB15 (Fig. 5b, c).
Fig. 5. Nucellus-expressed ZmNAC11 and ZmNAC29 bind to and activate ZmEXPB15.
a Schematic diagrams of the promoters of HKW9Mc and HKW9V671. The red triangles represent in vitro CACG motifs. The dashed box represents the positions of the biotin probes (b), qRT-PCR analysis of ZmNAC11 and ZmNAC29 expression during early kernel development of 0 to 12 DAP. All expression levels from three biological repeats were normalized to Actin. c In situ localization of ZmNAC11 and ZmNAC29 expression in the developing kenrels at 4 and 6 DAP. Positive signals were found in the nucellus (Nu) and endosperm (En) using the ZmNAC11 and ZmNAC29 antisense probes. The control was performed using 4-DAP kernel with ZmNAC11 and ZmNAC29 sense probes. Scale bars = 50 μm. d DNA binding affinities of the recombinant ZmNAC11 and ZmNAC29 proteins on the CACG motif-containing promoter regions of HKW9Mc and HKW9V671 detected by electrophoresis mobility shift assays (EMSAs). ZmNAC11 and ZmNAC29 bind more strongly to the promoter fragment (−299 to −249 upstream of the ATG) from the large-kernel line HKW9Mc than to that from the small-kernel line HKW9V671. The unlabeled intact probes were used for competition. The experiment was repeated two times with a similar result. e Transactivation activities of the ZmNAC11 and ZmNAC29 proteins on ZmEXPB15 promoters of two NILs (HKW9Mc and HKW9V671). Values are means ± s.d. (n = 5 biologically independent samples), Tukey HSD test is used and the statistical differences (P < 0.05, two-sided) are indicated by different letters. Source data are provided as a Source Data file.
To investigate the DNA binding of ZmNAC11 and ZmNAC29 to the promoter of ZmEXPB15, electrophoresis mobility shift assays (EMSAs) were performed using a probe containing the CACG motif from the promoter of ZmEXPB15, which harbors the two SNPs present in two NILs (Fig. 5a). Strong shifts of the probe were detected in the presence of the ZmNAC11 and ZmNAC29 recombinant proteins (Fig. 5d). Notably, the probe from the large-kernel NIL, HKWMc, which contains the intact CACG motif, showed a stronger shift than that from HKWV671 containing two SNPs that disrupted the motif (Fig. 5d). Next, we investigated the effects of ZmNAC11 and ZmNAC29 proteins on the transcription activity of ZmEXPB15 using the luciferase (LUC) reporter in maize leaf protoplasts. A 436-bp ZmEXPB15 promoter containing two CACG motifs was used to drive LUC (Supplementary Fig. 10a), and both ZmNAC11 and ZmNAC29 activated ZmEXPB15 expression (Fig. 5e). Consistently, the promoter from HKWMc showed a significantly stronger transcriptional activity in the presence of the NAC proteins than that from HKWV671 (Fig. 5e). Further, when a longer 1308-bp ZmEXPB15 promoter, which contains another four CACG motifs, was used, ZmEXPB15 transcription was significantly higher, suggesting a stronger binding of the two NAC proteins on the promoter (Supplementary Fig. 10b–d). When ZmNAC11 and ZmNAC29 proteins were coexpressed, the activation of ZmEXPB15 was not obviously changed (Supplementary Fig. 10e), suggesting there was no interaction between these two NAC proteins. Our findings indicate that the nucellus-enriched ZmNAC11 and ZmNAC29 TFs could bind the promoter and activate the transcription of ZmEXPB15.
Mutations in ZmNAC11 and ZmNAC29 cause similar kernel defects to zmexpb15
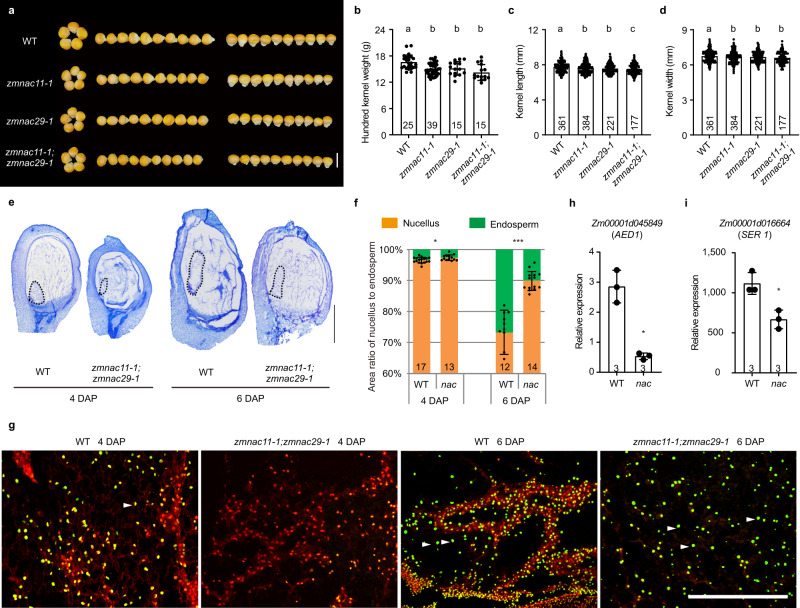
To investigate the significance of ZmEXPB15 activation by ZmNAC11 and ZmNAC29, we investigated the roles of two NAC genes on kernel traits. Knockout lines were created using CRISPR-Cas9 in the maize C01 inbred line (Supplementary Fig. 11a). Two zmnac11 knockout mutants (zmnac11-1 and zmnac11-2) and three zmnac29 knockout mutants (zmnac29-1, zmnac29-2 and zmnac29-3) all resulted in deletion or frame-shift of amino acids in the conserved NAC domain (Supplementary Fig. 11b, c). Interestingly, each single mutant produced smaller kernels with significantly decreased HKW, kernel length and width compared to the segregated wild-type sibs (Fig. 6a–d, Supplementary Fig. 11d–k). The zmnac11-1;zmnac29-1 double mutant did not show a significant enhancement compared to each single mutant (Fig. 6a–d), suggesting that these two NACs may work in one pathway. This speculation was in line with the findings that ZmNAC11 transcript was dramatically decreased in zmnac29 mutant, whereas ZmNAC29 was not affected by the loss of ZmNAC11 mutation (Supplementary Fig. 12a, b). All these results demonstrate that ZmNAC11 and ZmNAC29 both positively control kernel size and weight, with a possibility that ZmNAC29 may also work upstream of ZmNAC11.
Fig. 6. Mutations in ZmNAC11 and ZmNAC29 cause similar defects to zmexpb15 in kernel development.
a The mature kernels of zmnac11-1, zmnac29-1 and zmnac11-1;zmnac29-1 in comparison to the wild type (WT) in C01 background. b–d Quantitative analysis of hundred kernel weight (b), kernel length (c) and kernel width (d) of wild type (WT), zmnac11-1, zmnac29-1 and zmnac11-1;zmnac29-1. e Toluidine blue sections of WT and zmnac11-1;zmnac29-1 kernels at 4 and 6 DAP. f Ratio of nucellus (orange box) to endosperm area (green box) of 4-DAP and 6-DAP kernels in zmnac11-1;zmnac29-1 (nac) double mutant in comparison to the wild type (WT) control. Longitudinal median sections with maximum area of the entire endosperm were examined and quantified. g TUNEL assays of the nucellus at 4 and 6 DAP of WT and zmnac11-1;zmnac29-1 double mutant. WT kernel at 4 DAP has strong TUNEL-positive signals in nucellar cells (white arrow), whereas zmnac11-1;zmnac29-1 kernel at 4-DAP does not show obvious TUNEL signal. The TUNEL-positive signals (partly marked white arrows) are indistinguishable at 6-DAP between zmnac11-1;zmnac29-1 and WT. h, i qRT-PCR analysis shows the down-regulation of two PCD-related genes in the zmnac11-1;zmnac29-1 (nac) kernels at 4 DAP. All expression levels from three biological repeats were normalized to Actin. Scale bar = 1 cm in (a); 1 mm in (e); 500 µm in (g). Number on the bottom of each column is the sample size. Values in (b–d) are means ± s.d. and Tukey HSD test is used and statistical differences (P < 0.05, one-sided) are indicated by different letters. Values in (f, h, i) are means ± s.d., and the significance is estimated by a two-tailed Student’s t test. *P < 0.05, **P < 0.01, ***P < 0.001. Source data are provided as a Source Data file.
To explore whether the kernel defects of zmnac11 and zmnac29 were also associated with nucellus, cytological sectioning and TUNEL assay were performed using the developing kernels of zmnac11-1;zmnac29-1 double mutants. The mutants had significantly bigger nucellus and lower endosperm area at 4 and 6 DAP compared to WT sibs (Fig. 6e, f), indicating that nucellus elimination was delayed. In support of this, TUNEL-positive nuclei were barely detected in nucellar cells of 4-DAP zmnac11-1;zmnac29-1 kernels, whereas the signal was strong in wild type nucellus (Fig. 6g). However, at a later stage, the TUNEL signals in 6-DAP nucellus was indistinguishable between zmnac11-1;zmnac29-1 (nac) double mutant and wild type. Consistently, two PCD-related genes, AED1 and SER1, were also significantly down-regulated in the nac kernels (Fig. 6h, i). In addition, the HKW of F1 kernels from WT ear pollinated with the nac double mutant was significantly larger than that of nac ear pollinated with WT pollen (Supplementary Fig. 13a, b). The reciprocal cross results demonstrate that ZmNAC11 and ZmNAC29 control kernel development also maternally. Taken together, these observations suggest that ZmNAC11 and ZmNAC29 positively regulate kernel size and weight by promoting nucellus elimination, similar to ZmEXPB15.
ZmEXPB15 acts genetically with ZmNAC11 and ZmNAC29 to control kernel weight
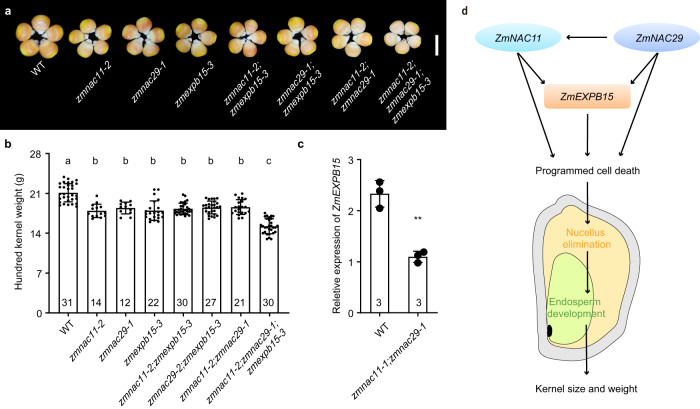
To explore the genetic interaction between zmexpb15, zmnac11 and zmnac29, the zmnac11-2;zmexpb15-3 and zmnac29-1;zmexpb15-3 double mutants and the zmnac11-2;zmnac29-1;zmexpb15-3 triple mutant were investigated. The three double mutants were similar in HKW to the segregated zmexpb15-3 single mutant, and also to zmnac11-2 and zmnac29-1 single mutants (Fig. 7a, b). Based on our findings that ZmNAC11 and ZmNAC29 could bind to ZmEXPB15 and activate its transcription, we propose that ZmEXPB15 may work in a common pathway with ZmNAC11 and ZmNAC29 in controlling kernel weight. This hypothesis is supported by the observation that ZmEXPB15 expression was significantly decreased in zmnac11 and zmnac29 mutants (Fig. 7c), whereas the expression of ZmNAC11 or ZmNAC29 was not obviously altered in zmexpb15 mutant (Supplementary Fig. 14a, b). However, zmnac11;zmnac29;zmexpb15 triple mutants had even lower HKW, compared to the single and double mutants (Fig. 7a, b), suggesting that additional players are involved in the regulation of ZmEXPB15 by ZmNAC11/ZmNAC29. Alternatively, the two NAC TFs and ZmEXPB15 might also have partially independent roles in control of kernel weight. Taken together, we propose that ZmEXPB15 may act, at least in part, in a common genetic pathway with ZmNAC11 and ZmNAC29 to control kernel weight (Fig. 7d).
Fig. 7. ZmEXPB15 may act in a common pathway with ZmNAC11 and ZmNAC29 to regulate kernel size and weight.
a Comparison of the mature kernels of wild type (WT) and different mutation combinations, zmnac11-2, zmnac29-1, zmexpb15-3 (KO3), zmnac11-2;zmexpb15-3, zmnac29-1;zmexpb15-3, zmnac11-2;zmnac29-1 and zmnac11-2;zmnac29-1;zmexpb15-3 in C01 background. Scale bar = 1 cm. b Quantitative analysis of HKW of WT, zmnac11-2, zmnac29-1, zmexpb15-3, zmnac11-2;zmexpb15-3, zmnac29-1;zmexpb15-3, zmnac11-2;zmnac29-1 and zmnac11-2;zmnac29-1;zmexpb15-3. Value is means ± s.d. Tukey HSD test is used and statistical differences (P < 0.05, one-sided) are indicated by different letters. Number on the bottom of each column is the sample size. c qRT-PCR analysis of ZmEXPB15 in WT, zmnac11-1 and zmnac29-1 kernels at 4 DAP. All expression levels from three biological repeats were normalized to Actin. Value is means ± s.d., and the significance is estimated by a two-tailed Student’s t test. ***P < 0.001. d A proposed model for the NAC-ZmEXPB15 module regulating the maternal nucellar tissue to ensure kernel size and weight in maize. ZmNAC11 and ZmNAC29 could bind and activate ZmEXPB15 to promote nucellus PCD process and further elimination, leading to an expansion and a normal growth of endosperm. Two NAC genes and ZmEXPB15 could also work independently in this process, and ZmNAC29 might work upstream of ZmNAC11. Source data are provided as a Source Data file.
Discussion
Kernel size and weight are determined by the coordinated growth of embryo, endosperm, and maternal tissues. Current studies of maize kernel development mostly focus on the endosperm and embryo growth, and little is known about the control of the nucellus, a maternal tissue. This study provides insights into the regulation of nucellus elimination and its impact on early endosperm development and consequently kernel-related traits.
Maize kernel size and weight are largely determined by the early development of the endosperm, in coordination with genetically distinct embryo and nucellus tissues during a short period around ~8 DAP22,32. Early endosperm development is accompanied by nucellus elimination though PCD33,34, however, knowledge of key genes and mechanisms coordinating these processes in maize is lacking. We identified ZmEXPB15 as the candidate gene of a hundred kernel weight QTL (HKW9), and show that it is expressed specifically in the nucellus from 2 to 8 DAP (Fig. 3a–c), coincident with the nucellus elimination period. A role of ZmEXPB15 in the regulation of nucellus elimination is supported by the delayed appearance of TUNEL signal and lower Evan’s blue staining in the small-kernel lines with lower ZmEXPB15 expression (HKWV671 and zmexpb15 mutant) (Fig. 4j and Supplementary Fig. 8). This delay is associated with downregulation of PCD-related genes (Fig. 4k). It has been proposed that nucellus and endosperm development are antagonistic in Arabidopsis11, as the nucellus degenerates in order to reallocate nutrients and space for endosperm development3. Consistent with these hypotheses, we observed that the endosperms of small-kernel lines were smaller as early as 3 DAP when the nucellus PCD was delayed, and the reduction in size became more severe at 12 DAP (Fig. 4 and Supplementary Fig. 8). These observations suggest that the reduced endosperm development in small-kernel lines is directly related to the difference in the timing of nucellus PCD, shedding light on the interaction between endosperm and maternal tissues in maize. Meanwhile, ZmEXPB15 also promotes nucellus cell expansion which is in line with its protein localization and property, leading to a larger nucellus volume. The dual role of ZmEXPB15 on nucellus cell expansion and elimination opens a window to understand how the nucellus tissue growth coordinates with endosperm development. Our findings reveal a key role of ZmEXPB15 in early endosperm development through promoting nucellus elimination and cell expansion, filling in a knowledge gap in the molecular coordination between endosperm and maternal tissues in maize.
Only a small number of genes are known to play important roles during early kernel development from fertilization to ~15 DAP in maize23. Most of these genes are mainly expressed after 6 DAP when the endosperm initiates, therefore their roles in kernel development are mostly through modulating early endosperm mitotic activity, or the differentiation and function of different endosperm cell types. To our knowledge, ZmEXPB15 is the only gene showing a nucellus-specific expression pattern in maize, and it regulates early kernel development through a distinct process, promoting nucellus PCD and elimination. At early stages of cereal seed development, the nucellus is the first maternal tissue to degenerate, and allows the remobilization of cellular contents for embryo and endosperm development13. The identification of ZmEXPB15 function highlights the importance of nucellus in early kernel development. ZmEXPB15 encodes an expansin protein, which participates in various biological processes by affecting the loosening of the cell wall35,36. We found the ZmEXPB15 protein localized in cell wall as expected, which explains its role in nucellus cell expansion (Figs. 3d, 4a–c). Our main finding on its role in nucellus PCD process opens a window on expansin protein function, which might be related to the cytoplasm and nucleus localization of the GFP-ZmEXPB15 fusion protein (Fig. 3d). Further studies are required to elucidate the underlying mechanism on how ZmEXPB15 triggers the initiation of PCD process.
The identification of two nucellus-enriched NAC TFs, ZmNAC11 and ZmNAC29 as candidate upstream regulators of ZmEXPB15, deepens our understanding on the molecular function of ZmEXPB15. Our results suggest that ZmNAC11 and ZmNAC29 may bind to the ZmEXPB15 promoter to activate expression (Fig. 5d, e), and act epistatically with ZmEXPB15 in regulating kernel size by promoting nucellus PCD (Fig. 7d). zmnac11;zmnac29;zmexpb15 triple mutant had more severe kernel defects compared to the single or double mutants (Fig. 7a, b), suggesting that the two NAC TFs regulate additional kernel related genes. This hypothesis is supported by their broader expression, not only in nucellus but also in endosperm (Fig. 5c). Our discovery of the ZmNAC11/ZmNAC29-ZmEXPB15 module that triggers nucellus PCD and promotes its elimination to accommodate endosperm development uncovers a previously unknown mechanism regulating early kernel development in maize. Recently, a NAC-expansin regulatory module, NAC25/NAC1L-EXPA2, was found to regulate endosperm cell expansion during seed germination, and to control the seed-to-seedling transition in Arabidopsis. NAC25/NAC1L directly activates EXPA2 expression specifically in endosperm, and promotes cell expansion37. In our study, the ZmNAC11/ZmNAC29-ZmEXPB15 module functions specifically in nucellus, to promote PCD process and cell elimination, and also promote its cell expansion revealing a unique function of the maize NAC-EXPANSIN module in kernel development.
Most kernel trait-related genes have been isolated from classical mutants with severe defects such as small or shrunken kernels, and are not directly applicable in breeding5. Our isolation of the maize kernel trait-related gene ZmEXPB15 in this study was based on a hundred kernel weight (HKW) QTL, HKW9. We show that ZmEXPB15 controls kernel size and weight, providing a resource to optimize maize kernel traits. To support its potential breeding value, we identified an elite haplotype (Hap1) for ZmEXPB15, which has increased ZmEXPB15 expression and HKW (Fig. 1j–l), and can be applied in molecular breeding for large kernel lines through marker-assisted selection (MAS). The large-kernel NIL, HKW9Mc, increased hundred kernel weight, kernel length and width, without obvious alteration in other agronomic traits, including starch content (Fig. 1a–e, Supplementary Fig. 1). Thus, this germplasm could be used as a large-kernel donor to improve other inbreds. In addition, ZmEXPB15 overexpression increased kernel weight and size, and this increase was consistent in hybrid combinations with different inbred lines, without an obvious negative effect in other ear traits. Therefore, our studies reveal the potential application of ZmEXPB15 in molecular breeding to generate large-kernel germplasm to improve crop yield-components.
Methods
Construction of the NIL lines
Two elite maize inbred lines, Mc and V671, that develop small and large kernels, respectively, were used for mapping hundred kernel weight (HKW) QTL. Using F2:3 families, a major QTL (HKW9) on chromosome 9 region (bin 9.03–9.04) was detected, and accounted for more than 10% of phenotypic variation19. To construct near-isogenic lines (NILs), Mc was used as the recurrent parent, and the HKW9 allele from V671 was introduced into Mc background through 4 generations of backcrossing followed by self-crossing. This resulted in two NILs, HKW9Mc and HKW9V671, harboring the HKW9 locus from Mc and V671, respectively.
Kernel trait measurement
Two near-isogenic lines (NILs) harboring the major hundred kernel weight QTL (HKW9) locus from Mc and V671, HKW9Mc and HKW9V671, were planted at different locations using a randomized block design with three replicates. For different transgenic materials (such as KO, OE, nac, nac-exp), stable generations of F3 or F4 were used for phenotypic identification. Well-pollinated ears in the middle of each row were selected to measure kernel-related traits, including the kernel length and width, and the hundred kernel weight. Microsoft Excel 2010 was used for significance test and analysis. Ear and kernel phenotype images were captured using a Nikon D7100 camera. Box plot and histogram was produce using GraphPad Prism 8 software. The kernel length and width were scored using a yield-trait scorer (YTS-MKT, GREENPHENO). One hundred kernels from the middle part of the ears were selected for measurement of hundred kernel weight. Kernel starch content was measured using the megazyme total starch assay kit (Catalog: K-TSTA-50A) according to the manufacturer’s instructions.
Cytological observation of developing kernels
For tissue sections for endosperm and nucellus area measurements, we made 1-mm median longitudinal sections of kernels with 0-12 DAP, fixed overnight in 4% (v/v) paraformaldehyde (Sigma–Aldrich), dehydrated in an ethanol gradient series (30%, 50%, 70%, 85%, 95%, and 100% ethanol), and embedded in Paraplast Plus (Sigma–Aldrich). The sample blocks were sectioned into 8-μm slices, and the longitudinal median sections were selected for measurement according to the following criteria for different developmental stage. 0 DAP selects the maximum area of the ovules, 2-3 DAP selects the maximum area of the endosperm, and for the kernels at 4 DAP and later stages, the maximum area of embryos was used for the reference, since the entire endosperm was too large to judge accurately. Finally stain with 0.5% (w/v) toluidine blue. Images were captured under the microscope (Nikon eclipse 80i).
For semi-thin sections, the kernels were cut longitudinally, leaving 1/5 of the middle part about 2 mm thick. The samples were immediately fixed in 50% FAA solution, resin embedding was performed with the kit Technovit 7100 (Kulzer, Germany), stained with 0.5% (w/v) toluidine blue solution after section, and photographed with Nikon camera (Nikon eclipse 80i). Finally, the cell size was measured and calculated by Image J 1.50i.
For scanning electron microscopy observation, endosperms at 16 DAP were cut into 2-mm3 pieces. Fresh tissues were fixed overnight in 2.5% (w/v) glutaraldehyde in sodium phosphate buffer (0.1 M, pH 7.2) at 4 °C for 12 h. Samples were dehydrated in a graded series of ethanol, and then dried to the critical point, sputter-coated with gold in an E-100 ion sputtering process, and observed with a scanning electron microscope (6390LV; JEOL, Tokyo, Japan).
Phylogenetic analysis
The ZmEXPB15 sequences from HKW9Mc and HKW9V671 were aligned using ClustalX software with the default parameters. The phylogenetic tree for multiple NAC transcription factors of maize was constructed using the MEGA program version 7 (www.megasoftware.net) and the neighbor-joining method, whose parameters are Poisson correction, pairwise deletion, and boot-strap (500 replicates; random seed).
Genetic transformation
To create knockout mutants for ZmEXPB15, ZmNAC11 and ZmNAC29, the guide RNA sequences (Supplementary Figs. 5c, 11b) against target genes were designed at the website of CRISPR-P 2.0 (http://crispr.hzau.edu.cn/CRISPR2/)38, then were cloned into a CRISPR/Cas9 plant expression vector pCPB-ZmUbi-hspCas939, followed by the agrobacterium-mediated transformation into the maize inbred line C01. The primers used for the transgenic genotype detection were listed in Supplementary Data 3.
To overexpress ZmEXPB15, the full CDS of the ZmEXPB15 copy from the large-kernel NIL, HKW9Mc, driven by the ubiquitin promoter was ligated to the binary vector pCAMBIA3301 and then transformed into the maize inbred line B104. To construct proZmEXPB15:ZmEXPB15-GFP, the genomic fragment including 1.5 kb promoter, full CDS and eGFP-fused gene was amplified and inserted into the pCAMBIA3301 entry vector, and then transformed into the maize inbred line B104. The Basta (bar) gene was amplified to distinguish the transgenic and non-transgenic plants. The primers used for the transgenic genotype detection were listed in Supplementary Data 3.
RT-qPCR assay
Immature (0~12 DAP) kernels from three independent ears were dissected and quickly frozen in liquid nitrogen, and were ground into a fine powder. Total RNA was extracted with TRIzol reagent (Thermo Fisher Scientific) according to the manufacturer’s instruction, and was treated with DNaseI digestion. For the reverse transcription, 1 μg of total RNA was used in each 20 μL reaction and the first cDNA strand was synthesized using SuperScriptII (Invitrogen). The qRT-PCR analysis was carried out with three independent RNA samples using a Bio-Rad CFX96 Touch Real-Time PCR detection system using Actin as an internal control, and the relative expression of the target transcripts was calculated using the 2−ΔΔCT method40. The primers used for the amplicons were listed in Supplementary Data 3.
RNA in situ hybridization
Immature C01 kernels at 0~12 DAP were collected and fixed in FAA solution containing 5% formalin (v/v), 50% ethanol (v/v), and 5% acetic acid (v/v) for 16 h at 4 °C, and then replaced with 70% ethanol twice and dehydrated with an ethanol series, followed by the substitution with xylene and embedding in Paraplast Plus (Sigma–Aldrich). For making sense and antisense RNA probes, gene-specific primers (listed in Supplementary Data 3) were used to amplify ZmEXPB15, ZmNAC11 and ZmNAC29. The amplified products were cloned into pSPT18 (Roche) and linearized with Hind III or EcoR I. The sense and antisense probes were separately synthesized by RNA polymerase using SP6 or T7 primer with digoxigenin-labelled UTP. RNA hybridization and immunologic detection of the hybridized probes were performed according to Greb41, with the addition of 8% polyvinyl alcohol to the detection buffer to minimize diffusion of the reaction products. Slides were exposed for 12–15 h before mounting and imaging, and were visualized under a microscope (Nikon eclipse 80i).
Confocal microscopy observation
Kernels at 2~8 DAP from proZmEXPB15:ZmEXPB15-GFP plant were cut longitudinally with a thickness of about 1 mm and observed under a confocal microscope (Olympus FV1000) with an excitation wavelength at 488 nm.
Terminal deoxynucleotidyl transferase dUTP nick-end labeling assay
The paraplast fixed developing kernels at 0~6 DAP were from above used for the toluidine blue staining. The paraplastp was removed by the axylol treatment, and the sections were hydrated with an ethanol series and treated with proteinase K in PBS. The endogenous peroxidase activity was quenched by the treatment with H2O2. The sections were incubated at 37 °C for 60 min in the presence of TdT using the TUNEL apoptosis detection kit (DeadEndTM fluorometric TUNEL system; Promega). The TUNEL-positive signal was detected using DeadEndTM Fluorometric TUNEL System (Promega) following the manufacturer’s instructions.
Evan’s blue staining
The nucellus tissue from different days after pollination was dyed in Evan’s blue solution and shaken for 20 min to ensure that all tissues were exposed to Evan’s blue dye solution. Rinse thoroughly with deionized water until the unbound dye washes away from the surface, then photograph. Quantitative Evan’s blue staining was performed by extracting Evan’s blue dye from stained nucellus tissue, in which 100 mg of tissue was thoroughly ground with 1 mL 1% sodium dodecyl sulfate (SDS) cell lysis buffer and sieved. Directly transfer 250 µL of supernatant to a 96-well microtiter plate. Measure the optical density at 600 nm spectrophotometrically using a plate reader by taking 1% SDS as blank. The concentration of Evan’s blue can be estimated using a reference standard curve42.
RNA-seq analysis
Total RNA of HKW9Mc and HKW9V671 were extracted from 6-DAP kernels using TRIzol reagent, followed by cleanup and DNase I treatment using an RNeasy Mini Kit (Qiagen). Three independent biological replicates from different ears were performed. cDNA libraries were constructed following the standard Illumina protocol and sequenced on the Illumina NovaSeq platform by Novogene. The sequence reads were trimmed using the tool Trimmomatic (v.0.33; http://www.usadellab.org/cms/?page5trimmomatic) and were mapped to the B73 RefGen_v4.34 reference genome using the programHISAT2 (v.2.1.0; http://ccb.jhu.edu/software/hisat2/faq.shtml). The program StringTie (v.1.3.3b) was employed to reconstruct the transcripts and to estimate gene expression levels43. The program HTSeq (v.0.6.1; https://pypi.org/project/HTSeq/) was used to count the number of reads per gene. The differentially expressed genes (DEGs) were identified by DEseq2 (http://www.bioconductor.org/packages/devel/bioc/html/DESeq2.html) by a p-value of <0.001, absolute value of log2[fold change] > 0.5. DEGs are listed in Supplementary Data 1.
Transient expression assays in maize protoplast
For promoter activation assay, the full coding sequences of ZmNAC11 and ZmNAC29 driven by the 35 S promoter were cloned into the effector vector pGreenII 62-SK, and different truncated promoter of the ZmEXPB15 from HKW9Mc and HKW9V671 were cloned into the reporter vector pGreenII 0800-LUC. The empty pGreenII 62-SK vector was used as control. The transient dual-luciferase assays were performed in the maize protoplasts collected from the leaves of 2-week-old etiolated seedling of inbred line B73. The luciferase signal was detected using dual-luciferase assay reagents (Promega) following the manufacturer’s instructions. The ratio of LUC/REN activity was measured using multi-mode microplate reader (SpectraMax i3x). Primers are listed in Supplementary Data 3.
Recombinant protein expression and purification
The full coding sequences of ZmNAC11 and ZmNAC29 were cloned into the recombinant protein expression vectors pET-28a-SUMO vector (Novagen), and were introduced into E.coli BL21 (DE3) strain. The recombinant proteins, His-SUMO-ZmNAC11 and His-SUMO-ZmNAC29, were induced with 0.2 mM isopropyl-β-Ɗ-thiogalactoside (IPTG) for 16 h at 16 °C, and were purified with columns equipped with Ni2+ affinity resin (GE Healthcare). Primers are listed in Supplementary Data 3.
Electrophoretic mobility shift assay
Oligonucleotide probes from −299 to −249 upstream of the ZmEXPB15 promoters from HKW9Mc and HKW9V671 were synthesized and labeled with the Pierce Biotin 3′ End DNA Labeling Kit. The purified recombinant His-SUMO-ZmNAC11 and His-SUMO-ZmNAC29 proteins were used to detect the binding affinity with the biotin-labeled probes by the LightShiftTM Chemiluminescent EMSA kit (Thermo Fisher Scientific) according to the standard manufacturer’s manual. Primers are listed in Supplementary Data 3.
Reporting summary
Further information on research design is available in the Nature Research Reporting Summary linked to this article.
Supplementary information
Description of Additional Supplementary Files
Source data
Acknowledgements
This work was supported by grant from the National Key Research and Development Program of China (2021YFF1000500, F.Q.) and the National Natural Science Foundation of China (31971951, F.Q. and 31771796, F.Q.). The computations in this paper were run on the bioinformatics computing platform of the National Key Laboratory of Crop Genetic Improvement, Huazhong Agricultural University.
Author contributions
F.Q. and F.Y. conceived and designed the project. Q.S., Y.L., A.H., W.Z., Q.N., Z.T., K.L. and L.M. performed the experiments. D.G. and W.Z. designed the CRISPR sgRNAs and prepared the constructs for maize transformation. H.Z. and Z.T. analysed the RNA-seq data. Q.S., F.Y., Z.Z. and D.J. analysed the data and wrote the manuscript.
Peer review
Peer review information
Nature Communications thanks Nicolas Doll, Nese Sreenivasulu, Haiyang Wang and the other, anonymous, reviewer(s) for their contribution to the peer review of this work. Peer reviewer reports are available.
Data availability
The plant materials generated in this study are available from the corresponding authors upon request. The RNA-seq datasets are available from the NCBI Sequence Read Archive under the accession numbers PRJNA756197 and SRR15525330-SRR15525335. Source data are provided with this paper.
Competing interests
The authors declare no competing interests.
Footnotes
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Contributor Information
Fang Yang, Email: fyang@mail.hzau.edu.cn.
Fazhan Qiu, Email: qiufazhan@mail.hzau.edu.cn.
Supplementary information
The online version contains supplementary material available at 10.1038/s41467-022-33513-4.
References
- 1.Kesavan, M., Song, J. T. & Seo, H. S. Seed size: A priority trait in cereal crops. Physiol. Plant.147, 113–120 (2013). [DOI] [PubMed] [Google Scholar]
- 2.Huang, J. et al. The kernel size-related quantitative trait locus qKW9 encodes a pentatricopeptide repeat protein that aaffects photosynthesis and grain filling. Plant Physiol.183, 1696–1709 (2020). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Chen, L. et al. The retromer protein ZmVPS29 regulates maize kernel morphology likely through an auxin-dependent process(es). Plant Biotechnol. J.18, 1004–1014 (2020). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Cai, M. et al. Emp10 encodes a mitochondrial PPR protein that affects the cis-splicing of nad2 intron1 and seed development in maize. Plant J.91, 132–144 (2017). [DOI] [PubMed] [Google Scholar]
- 5.Dai, D., Ma, Z. & Song, R. Maize kernel development. Mol. Breeding41 (2021). [DOI] [PMC free article] [PubMed]
- 6.Pan, Z. et al. ZmSMK9, a pentatricopeptide repeat protein, is involved in the cis-splicing of nad5, kernel development and plant architecture in maize. Plant Sci.288, 110205 (2019). [DOI] [PubMed] [Google Scholar]
- 7.Ren, X. et al. EMPTY PERICARP11 serves as a factor for splicing of mitochondrial nad1 intron and is required to ensure proper seed development in maize. J. Exp. Bot.68, 4571–4581 (2017). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Zhao, H. et al. Loss of function of an RNA polymerase III subunit leads to impaired maize kernel development. Plant Physiol.184, 359–373 (2020). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Sreenivasulu, N. & Wobus, U. Seed-development programs: a systems biology-based comparison between dicots and monocots. Annu. Rev. Plant Biol.64, 189–217 (2013). [DOI] [PubMed] [Google Scholar]
- 10.Doll, N. M. et al. Signaling in early maize kernel development. Mol. Plant. 10, 375-388 (2017). [DOI] [PubMed] [Google Scholar]
- 11.Xu, W. et al. Endosperm and nucellus develop antagonistically in Arabidopsis seeds. Plant Cell.28, 1343–1360 (2016). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Wu, H. M. & Cheung, A. Y. Programmed cell death in plant reproduction. Plant Mol. Biol.44, 267–281 (2000). [DOI] [PubMed] [Google Scholar]
- 13.Dominguez, F. & Cejudo, F. J. Programmed Cell Death (PCD): An essential process of cereal seed development and germination. Front. Plant Sci.5, 366 (2014). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Dominguez, F. & Cejudo, F. J. Identification of a nuclear-localized nuclease from wheat cells undergoing programmed cell death that is able to trigger DNA fragmentation and apoptotic morphology on nuclei from human cells. Biochem. J.397, 529–536 (2006). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Yang, X. et al. Live and Let Die - The Bsister MADS-box gene OsMADS29 controls the degeneration of cells in maternal tissues during seed development of rice (Oryza sativa L.). PLoS One7, e51435 (2012). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Yin, L. & Xue, H. The MADS29 transcription factor regulates the degradation of the nucellus and the nucellar projection during rice seed development. Plant Cell24, 1049–1065 (2012). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Lu, J. & Magnani, E. Seed tissue and nutrient partitioning, a case for the nucellus. Plant Reprod.31, 309–317 (2018). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Nayar, S., Sharma, R., Tyagi, A. K. & Kapoor, S. Functional delineation of rice MADS29 reveals its role in embryo and endosperm development by affecting hormone homeostasis. J. Exp. Bot.64, 4239–4253 (2013). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Liu, Y. et al. Genetic analysis and major QTL detection for maize kernel size and weight in multi-environments. Theor. Appl. Genet.127, 1019–1037 (2014). [DOI] [PubMed] [Google Scholar]
- 20.Yi, F. et al. High temporal-resolution transcriptome landscape of early maize seed development. Plant Cell31, 974–992 (2019). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Krishnan, S. & Dayanandan, P. Structural and histochemical studies on grain-filling in the caryopsis of rice (Oryza sativa L.). J. Biosci.28, 455–469 (2003). [DOI] [PubMed] [Google Scholar]
- 22.Doll, N. M., Depege-Fargeix, N., Rogowsky, P. M. & Widiez, T. Signaling in early maize kernel development. Mol. Plant10, 375–388 (2017). [DOI] [PubMed] [Google Scholar]
- 23.Lopez-Fernandez, M. P. & Maldonado, S. Programmed cell death in seeds of angiosperms. J. Integr. Plant Biol.57, 996–1002 (2015). [DOI] [PubMed] [Google Scholar]
- 24.Radchuk, V. et al. Vacuolar processing enzyme 4 contributes to maternal control of grain size in barley by executing programmed cell death in the pericarp. N. Phytol.218, 1127–1142 (2018). [DOI] [PubMed] [Google Scholar]
- 25.Radchuk, V. et al. Grain filling in barley relies on developmentally controlled programmed cell death. Commun. Biol.4, 428 (2021). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Avci, U. et al. Cysteine proteases XCP1 and XCP2 aid micro-autolysis within the intact central vacuole during xylogenesis in Arabidopsis roots. Plant J.56, 303–315 (2008). [DOI] [PubMed] [Google Scholar]
- 27.Nakabayashi, K. et al. Identification of clp genes expressed in senescing Arabidopsis leaves. Plant Cell Physiol.40, 504–514 (1999). [DOI] [PubMed] [Google Scholar]
- 28.Till, C. J. et al. The Arabidopsis thaliana N-recognin E3 ligase PROTEOLYSIS1 influences the immune response. Plant Direct3, e00194 (2019). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.He, X. et al. The nitrate-inducible NAC transcription factor TaNAC2-5A controls nitrate response and increases wheat yield. Plant Physiol.169, 1991–2005 (2015). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Tang, W. et al. Genome-wide associated study identifies NAC42-activated nitrate transporter conferring high nitrogen use efficiency in rice. Nat. Commun.10, 5279 (2019). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Zhan, J. et al. RNA sequencing of laser-capture microdissected compartments of the maize kernel identifies regulatory modules associated with endosperm cell differentiation. Plant Cell27, 513–531 (2015). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Sabelli, P. A. & Larkins, B. A. The contribution of cell cycle regulation to endosperm development. Sex. Plant Reprod.22, 207–219 (2009). [DOI] [PubMed] [Google Scholar]
- 33.Berger, F. Endosperm: The crossroad of seed development. Curr. Opin. Plant Biol.6, 42–50 (2003). [DOI] [PubMed] [Google Scholar]
- 34.Leroux, B. M. et al. Maize early endosperm growth and development: From fertilization through cell type differentiation. Am. J. Bot.101, 1259–1274 (2014). [DOI] [PubMed] [Google Scholar]
- 35.Cosgrove, D. J. Plant expansins: diversity and interactions with plant cell walls. Curr. Opin. Plant Biol.25, 162–172 (2015). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Marowa, P., Ding, A. & Kong, Y. Expansins: roles in plant growth and potential applications in crop improvement. Plant Cell Rep.35, 949–965 (2016). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Sanchez-Montesino, R. et al. A regulatory module controlling GA-mediated endosperm cell expansion is critical for seed germination in arabidopsis. Mol. Plant12, 71–85 (2019). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Liu, H. et al. CRISPR-P 2.0: An improved CRISPR-Cas9 tool for genome editing in plants. Mol. Plant10, 530–532 (2017). [DOI] [PubMed] [Google Scholar]
- 39.Li, C. et al. RNA-guided Cas9 as an in vivo desired-target mutator in maize. Plant Biotechnol. J.15, 1566–1576 (2017). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Livak, K. J. & Schmittgen, T. D. Analysis of relative gene expression data using real-time quantitative PCR and the 2(-Delta Delta C(T)) Method. Methods25, 402–408 (2001). [DOI] [PubMed] [Google Scholar]
- 41.Greb, T. et al. Molecular analysis of the LATERAL SUPPRESSOR gene in Arabidopsis reveals a conserved control mechanism for axillary meristem formation. Genes Dev.17, 1175–1187 (2003). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Vijayaraghavareddy, P., Adhinarayanreddy, V., Vemanna, R. S., Sreeman, S. & Makarla, U. Quantification of membrane damage/cell death using evan’s blue staining technique. Bio-Protoc.7, e2519 (2017). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Pertea, M., Kim, D., Pertea, G. M., Leek, J. T. & Salzberg, S. L. Transcript-level expression analysis of RNA-seq experiments with HISAT, StringTie and Ballgown. Nat. Protoc.11, 1650–1667 (2016). [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Description of Additional Supplementary Files
Data Availability Statement
The plant materials generated in this study are available from the corresponding authors upon request. The RNA-seq datasets are available from the NCBI Sequence Read Archive under the accession numbers PRJNA756197 and SRR15525330-SRR15525335. Source data are provided with this paper.