TO THE EDITOR:
Richter syndrome (RS) is the histological transformation of chronic lymphocytic leukemia (CLL) into an aggressive lymphoma, typically diffuse large B-cell lymphoma (DLBCL), with most cases clonally related to the preceding CLL.1-3 In the past 10 years, a revolution has occurred in therapeutic options for patients with CLL.4 However, little has changed for patients with RS, with only a few phase 1 and 2 clinical trials ongoing, mostly based on different drug combinations.5 Novel therapeutic perspectives for RS may come from immune-based therapies6,7 or, alternatively, from the use of antibody-drug conjugates (ADCs) targeting antigens predominantly or exclusively expressed by neoplastic cells.8
One of these antigens is CD37, a heavily glycosylated leukocyte surface antigen belonging to the tetraspanin superfamily, selectively expressed at high levels by normal mature B cells, as well by mature B-cell malignancies, including non-Hodgkin lymphoma (NHL), CLL, and DLBCL.9-11 CD37 has recently gained attention as a potential target, with consequent development of novel therapeutic agents12-16 that have shown clinical efficacy in CLL and DLBCL when used in combination with rituximab, chemotherapy, or targeted compounds.17-21
The increased interest and efficacy of anti-CD37–based therapies prompted the development of 3 different anti-CD37-ATACs (amanitin-based ADCs). These novel drugs are composed of a chimeric monoclonal IgG1 antibody specific for CD37, conjugated via either a proteolytic cleavable (anti-CD37-Ama 1 and anti-CD37-Ama 2) or a noncleavable (anti-CD37-Ama 3) linker to amanitin as the payload (supplemental Figure 1A, available on the Blood Web site).
All 3 anti-CD37 ATACs showed full-blown cytotoxicity on target-positive Raji cells (Burkitt lymphoma cell line) with half-maximal effective concentration in the low nanomolar and picomolar range (anti-CD37-Ama 1: 2.04 × 10−9 M; anti-CD37-Ama 2: 7.5 × 10−10 M; anti-CD37-Ama 3: 3.9 × 10−9 M; supplemental Figure 1B). Target specificity was demonstrated by absence of cytotoxicity up to a concentration of 10−6 M in a CD37− cell line (HEK293wt; supplemental Figure 1C). In contrast, free α-amanitin showed cytotoxicity on Raji and HEK cells (supplemental Figure 1D) with half-maximal effective concentration values in the micromolar range, which was even lower in HEK cells expressing OATP1B3 (supplemental Figure 1D), a cell surface transporter that is known to transport α-amanitin into hepatocytes.
Amanitin is an RNA polymerase II inhibitor that interrupts cellular transcription at low nanomolar concentrations.22 Consistently, anti-CD37 ATACs induced cytotoxicity by arresting RNA transcription, with the active metabolite of anti-CD37-Ama 2 displaying a similar degree of inhibition of free α-amanitin, with a half-maximal inhibitory concentration in the low nanomolar range (supplemental Figure 1E).
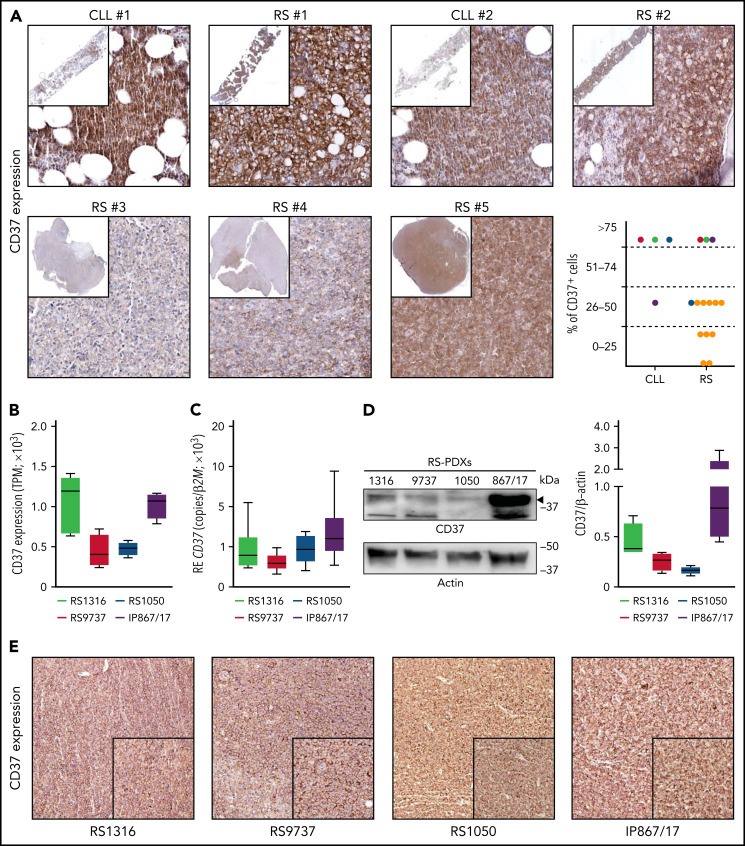
Analysis of CD37 expression in cell lines and primary samples obtained from B-cell hematological malignancies revealed low levels of expression in acute lymphoblastic leukemia, Hodgkin lymphoma, and multiple myeloma, whereas CLL, mantle, and NHL cells expressed it at high levels (supplemental Figure 2A). Primary samples confirmed this pattern of expression, with CLL cells showing the highest expression, followed by follicular lymphoma and DLBCL, whereas acute lymphoblastic leukemia and multiple myeloma had low-to-undetectable expression of CD37 (supplemental Figure 2B). Importantly, RNA sequencing data performed on 14 patients with RS showed that CD37 was invariably expressed by this lymphoma (supplemental Figure 2B), as confirmed by immunohistochemical staining performed on bone marrow or lymph node biopsy specimens from patients with RS (Figure 1A). In paired CLL-RS specimens, CD37 expression, which was high in the CLL phase, was invariably maintained after RS transformation (Figure 1A).
Figure 1.
CD37 is expressed by Richter syndrome primary samples and patient-derived xenograft models. CD37 expression on primary RS samples was checked by immunohistochemistry performed on bone marrow or lymph node biopsy specimens from patients with RS (yellow dots) or paired CLL-RS samples (A; red-green-dark blue-purple dots). Representative staining of 2 paired CLL-RS and 3 RS samples. Original magnification, ×200; inset ×6. CD37 expression was checked, both at the transcript and protein levels, in our 4 RS-PDX models (RS1316, RS9737, RS1050, and IP867/17) by RNA sequencing (B; n = 4 per model; box and whiskers, minimum to maximum representation), quantitative real-time PCR (C; n = 8 per model; box and whiskers, minimum to maximum representation), western blot analysis (D; n = 4 per model; box and whiskers, minimum to maximum representation) and immunohistochemistry staining on slides of formalin-fixed and paraffin-embedded, RS-PDX–derived tumor masses (E). In western blot analyses, actin was used as a loading control. RE, relative expression; TPM, transcript per million.
We then took advantage of established PDX models, which have been shown to be useful tools to test the efficacy of novel drugs/drug combinations.8,23,24 The 4 RS-PDX models were characterized by significant levels of CD37, with 2 of them, RS1316 and IP867/17, showing slightly higher CD37 expression compared with the other 2 (RS9737 and RS1050; Figure 1B-D). Flow cytometry analyses (supplemental Figure 2C) and immunohistochemistry staining on formalin-fixed, paraffin-embedded sections of tumor masses obtained from the 4 RS-PDXs (Figure 1E) confirmed these observations.
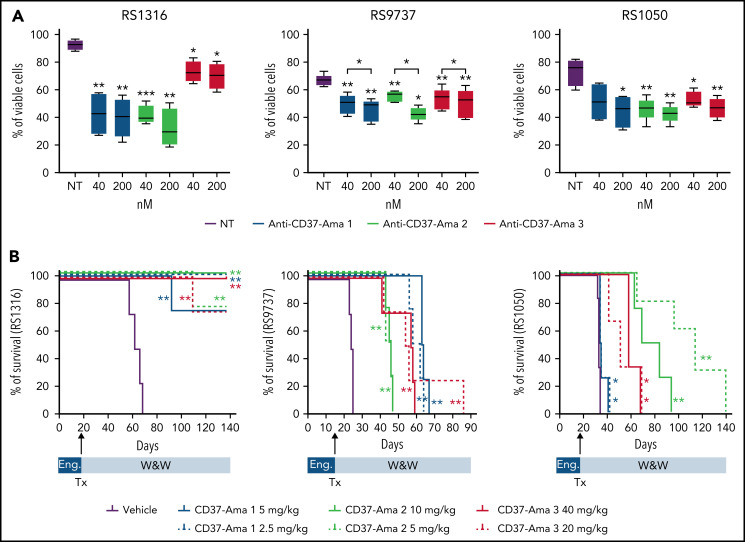
All 3 anti-CD37 ATACs (anti-CD37-Ama 1, -Ama 2, and -Ama 3) bound CD37 (supplemental Figure 2D), ruling out any interference of the linker or amatoxin with antibody-binding properties. When tested in vitro (40 and 200 nM; 72 hours) on RS-PDX–derived cells, these 3 ATACs resulted in a significant apoptotic response, with limited heterogeneity among the PDXs and compounds (supplemental Figure 3B). These results were corroborated by pronounced cleavage of caspase-3 and PARP proteins in all models (supplemental Figure 3B). All anti-CD37 ATACs induced apoptosis selectively in CD37-expressing cells. Indeed, when incubated with peripheral blood mononuclear cells from healthy donors, only CD37+ B cells underwent apoptosis, whereas no reduction in viability was measured in T lymphocytes (CD37−; supplemental Figure 4). Furthermore, no toxicity was assessed when treating these cells with nontargeted antibodies conjugated to amanitin with the same 3 linkers, underlining the specificity of anti-CD37 ATACs (supplemental Figure 4).
Finally, these ATACs were tested in vivo in 3 of 4 RS-PDX models (RS1316, RS9737, and RS1050), which can grow in a systemic way, mimicking the human disease. After IV injection, RS cells are engrafted and localized in different tissues, including spleen, bone marrow, peripheral blood, and even the brain, effectively establishing a progressive lymphoma.25 Anti-CD37 ATACs were administered IV at 2 different doses as a single treatment (anti-CD37-Ama 1: 2.5 and 5 mg/kg; anti-CD37-Ama 2: 5 and 10 mg/kg; and anti-CD37-Ama 3: 20 and 40 mg/kg). The mice were then compared with a vehicle control group for survival and disease distribution in different organs (spleen, liver, peripheral blood, bone marrow, kidney, brain, and lung), as assessed by flow cytometry using tumor-specific human markers. Consistent with the in vitro data, RS1316 showed the best responses in terms of improvement of survival after anti-CD37 ATAC administration, with complete disease regression in 21 of 24 treated mice (P = .0067; Figure 2A, left panel) and no or very limited tumor cells in the different organs (supplemental Figure 5A). At variance with RS1316, all RS9737, and RS1050 mice died of disease (supplemental Figure 5B), but anti-CD37 ATAC-treated mice displayed a marked increase in survival (Figure 2B). Specifically, mice treated with anti-CD37 ATACs displayed heterogenous responses among the 3 different compounds, with no significant differences between the 2 doses (vehicle: 24 days vs anti-CD37-Ama 2: 45 days vs anti-CD37-Ama 1: 61 days; P = .0091). Anti-CD37-Ama 3 showed a higher degree of heterogeneity of in vivo response (41-59 days with the higher dose vs 42-86 days with the lower one; P = .0091). Likewise, the RS1050 model showed a significant increase in survival when mice were treated with anti-CD37 ATACs, although with some differences among the compounds, and with the only exception of the anti-CD37-Ama 1 that resulted only in a mild in vivo effect (Figure 2B; right panel). Vehicle-treated mice were euthanized on days 32 to 34 after injection of RS cells, with neoplastic cells localizing in several tissues (supplemental Figure 5C). Anti-CD37-Ama 3 resulted in a statistically significant increased survival (41-69 days after injection), with comparable results when considering the 2 doses of drug (P = .0107 for the 40-mg/kg dose and P = .0193 for the lower dose). Anti-CD37-Ama 2 treatment further improved the mice’s survival (63-84 days for the higher dose and 65-120 days for the 5-mg/kg dose).
Figure 2.
Anti-CD37 ATACs induce apoptosis in vitro and prolong survival of RS-PDX mice. (A) The apoptotic response of RS cells exposed for 72 hours to different doses (40 and 200 nM) of anti-CD37-Ama 1, anti-CD37-Ama 2, and anti-CD37-Ama 3 or left untreated was analyzed with conventional cytofluorimetric annexin V/PI staining (n = 5 per model; box and whiskers, minimum to maximum representation). (B) RS cells were injected IV into NOD/SCID/γ chain−/− mice and left to engraft (Eng) for 2 weeks. The mice were then randomly assigned to different treatment groups (4 mice per group). After anti-CD37 ATAC administration (Tx), mice were monitored (W&W, watch and wait) and survival analyses performed, generating Kaplan-Mayer curves. (A) Asterisks on the box plots refer to statistical significance vs the untreated condition; P values with bars refer to the indicated conditions. Statistical analyses were performed with the paired t test. *P < .05; **P < .01; ***P < .001.
To exclude any toxicity related to amanitin administration, ad hoc experiments were performed on NSG mice to analyze the effects of this toxin on the liver, administering the 3 ADCs at the higher dose with the same schedule adopted for RS models. Hepatic enzymes were measured, and histology of the liver was performed at days 7 and 21 after administration of ADC. Elevation of aspartate aminotransferase and alanine aminotransferase was noted 7 days after treatment, with levels diminishing after 21 days. However, regarding hepatic tissue and morphology, no signs of hepatic toxicity related to the administration of amanitin was note at the histological level in treated mice, suggesting a transient elevation of the enzymes, but no permanent liver damage (supplemental Figure 6 and supplemental Table 2).
Taken together, the data indicate that CD37 may represent a good candidate for targeting RS cells with highly selective amanitin-based ADCs.
Supplementary Material
The online version of this article contains a data supplement.
Acknowledgments
This work was supported by the Italian Association for Cancer Research (AIRC) (My First AIRC Grant MFAG-23107 (T.V.); investigator grant IG-23095 (S.D.); the Italian Ministry of Health (GR-2016-02364298 (T.V.); the Ministry of Education, University and Research-MIUR “Progetto strategico di Eccellenza Dipartimentale” grant D15D18000410001 (S.D.) as part of the Department of Medical Sciences, University of Turin; Fondazione CRT Erogazioni Ordinarie 2021 I tornata (T.V.); and Heidelberg Pharma research funds (S.D.).
Footnotes
Original data are available by e-mail request to the corresponding author (silvia.deaglio@unito.it or tiziana.vaisitti@unito.it).
Authorship
Contribution: T.V., C.O., M.K., A.P., and S.D. designed the research; M.G.P., O.J., and A.D. provided material; T.V., N.V., M.M., L.B., A.I., G.L., J.C.C., and A.D. performed the experiments; T.V., C.O., M.K., A.P., and S.D. analyzed the results, produced the figures, and wrote the manuscript; and all authors approved the final version of the manuscript.
Conflict-of-interest disclosure: T.V. has received research funds from AstraZeneca. N.V., C.O., M.K., and A.P. are employed by Heidelberg Pharma. S.D. has received research funds from AstraZeneca, VelosBio Inc, and Heidelberg Pharma. The remaining authors declare no competing financial interests.
Correspondence: Silvia Deaglio, Department of Medical Sciences, University of Turin, Via Nizza 52, 10126 Torino, Turin, Italy; e-mail: silvia.deaglio@unito.it; and Tiziana Vaisitti, Department of Medical Sciences, University of Turin, Via Nizza 52, 10126 Torino, Turin, Italy; e-mail: tiziana.vaisitti@unito.it.
REFERENCES
- 1.Chigrinova E, Rinaldi A, Kwee I, et al. Two main genetic pathways lead to the transformation of chronic lymphocytic leukemia to Richter syndrome. Blood. 2013;122(15):2673-2682. [DOI] [PubMed] [Google Scholar]
- 2.Rossi D, Gaidano G. Richter syndrome: pathogenesis and management. Semin Oncol. 2016;43(2):311-319. [DOI] [PubMed] [Google Scholar]
- 3.Rossi D, Spina V, Deambrogi C, et al. The genetics of Richter syndrome reveals disease heterogeneity and predicts survival after transformation. Blood. 2011;117(12):3391-3401. [DOI] [PubMed] [Google Scholar]
- 4.Hallek M. On the architecture of translational research designed to control chronic lymphocytic leukemia. Hematology Am Soc Hematol Educ Program. 2018;2018:1-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Iannello A, Deaglio S, Vaisitti T. Novel approaches for the treatment of patients with Richter’s syndrome. Curr Treat Options Oncol. 2022;23(4):526-542. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Ding W, LaPlant BR, Call TG, et al. Pembrolizumab in patients with CLL and Richter transformation or with relapsed CLL. Blood. 2017;129(26):3419-3427. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Rogers KA, Huang Y, Dotson E, et al. Use of PD-1 (PDCD1) inhibitors for the treatment of Richter syndrome: experience at a single academic centre. Br J Haematol. 2019;185(2):363-366. [DOI] [PubMed] [Google Scholar]
- 8.Vaisitti T, Arruga F, Vitale N, et al. ROR1 targeting with the antibody-drug conjugate VLS-101 is effective in Richter syndrome patient-derived xenograft mouse models. Blood. 2021;137(24):3365-3377. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Barrena S, Almeida J, Yunta M, et al. Aberrant expression of tetraspanin molecules in B-cell chronic lymphoproliferative disorders and its correlation with normal B-cell maturation. Leukemia. 2005;19(8):1376-1383. [DOI] [PubMed] [Google Scholar]
- 10.Beckwith KA, Byrd JC, Muthusamy N. Tetraspanins as therapeutic targets in hematological malignancy: a concise review. Front Physiol. 2015;6:91. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Xu-Monette ZY, Li L, Byrd JC, et al. Assessment of CD37 B-cell antigen and cell of origin significantly improves risk prediction in diffuse large B-cell lymphoma. Blood. 2016;128(26):3083-3100. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Deckert J, Park PU, Chicklas S, et al. A novel anti-CD37 antibody-drug conjugate with multiple anti-tumor mechanisms for the treatment of B-cell malignancies. Blood. 2013;122(20):3500-3510. [DOI] [PubMed] [Google Scholar]
- 13.Pagel JM, Spurgeon SE, Byrd JC, et al. Otlertuzumab (TRU-016), an anti-CD37 monospecific ADAPTIR(™) therapeutic protein, for relapsed or refractory NHL patients. Br J Haematol. 2015;168(1):38-45. [DOI] [PubMed] [Google Scholar]
- 14.Pereira DS, Guevara CI, Jin L, et al. AGS67E, an anti-CD37 monomethyl auristatin E antibody-drug conjugate as a potential therapeutic for B/T-cell malignancies and AML: a new role for CD37 in AML. Mol Cancer Ther. 2015;14(7):1650-1660. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Repetto-Llamazares AH, Larsen RH, Patzke S, et al. Targeted cancer therapy with a novel anti-CD37 beta-particle emitting radioimmunoconjugate for treatment of non-Hodgkin lymphoma. PLoS One. 2015;10(6):e0128816. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Maaland AF, Heyerdahl H, O’Shea A, et al. Targeting B-cell malignancies with the beta-emitting anti-CD37 radioimmunoconjugate 177Lu-NNV003. Eur J Nucl Med Mol Imaging. 2019;46(11):2311-2321. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Balzarotti M, Magagnoli M, Canales MA, et al. A phase Ib, open-label, dose-escalation trial of the anti-CD37 monoclonal antibody, BI 836826, in combination with gemcitabine and oxaliplatin in patients with relapsed/refractory diffuse large B-cell lymphoma. Invest New Drugs. 2021;39(4):1028-1035. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Danilov AV, Spurgeon SE, Siddiqi T, et al. A phase Ib, open label, dose escalation trial of the anti-CD37 monoclonal antibody, BI 836826, in combination with ibrutinib in patients with relapsed/refractory chronic lymphocytic leukemia. Invest New Drugs. 2021;39(4):1099-1105. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Kolstad A, Illidge T, Bolstad N, et al. Phase 1/2a study of 177Lu-lilotomab satetraxetan in relapsed/refractory indolent non-Hodgkin lymphoma. Blood Adv. 2020;4(17):4091-4101. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Robak T, Hellmann A, Kloczko J, et al. Randomized phase 2 study of otlertuzumab and bendamustine versus bendamustine in patients with relapsed chronic lymphocytic leukaemia. Br J Haematol. 2017;176(4):618-628. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Stilgenbauer S, Aurran Schleinitz T, Eichhorst B, et al. Phase 1 first-in-human trial of the anti-CD37 antibody BI 836826 in relapsed/refractory chronic lymphocytic leukemia. Leukemia. 2019;33(10):2531-2535. [DOI] [PubMed] [Google Scholar]
- 22.Kaplan CD, Larsson KM, Kornberg RD. The RNA polymerase II trigger loop functions in substrate selection and is directly targeted by alpha-amanitin. Mol Cell. 2008;30(5):547-556. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Vaisitti T, Gaudino F, Ouk S, et al. Targeting metabolism and survival in chronic lymphocytic leukemia and Richter syndrome cells by a novel NF-κB inhibitor. Haematologica. 2017;102(11):1878-1889. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Iannello A, Vitale N, Coma S, et al. Synergistic efficacy of the dual PI3K-δ/γ inhibitor duvelisib with the Bcl-2 inhibitor venetoclax in Richter syndrome PDX models. Blood. 2021;137(24):3378-3389. [DOI] [PubMed] [Google Scholar]
- 25.Vaisitti T, Braggio E, Allan JN, et al. Novel Richter syndrome xenograft models to study genetic architecture, biology, and therapy responses. Cancer Res. 2018;78(13):3413-3420. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.