Abstract
Prostate cancer (PC) is one of the most common types of cancers among men, referring to the uncontrolled growth of the prostate gland. It is increasingly recognized that the interaction of the glioma-associated oncogene (GLI) pathway and androgen receptor affects PC progression. Nevertheless, the effects of resveratrol on PC progression via Hedgehog (HH) signaling remain unexplored. In this study, the castration-sensitive and castration-resistant xenograft models in mice are systematically established using two different PC cell lines (LNCaP and PC-3). Further, the Western blotting, immunohistochemistry, MTT, Transwell, and RT-qPCR analyses are performed to verify the mechanistic views of resveratrol on PC and HH signals in vitro and in vivo. Resveratrol showed epithelial–mesenchymal transition (EMT) progression, inhibiting the tumor size and expression levels of vimentin, vascular endothelial growth factor (VEGF), and matrix metalloproteinases (MMP) 7, as well as upregulating the expression profiles the E-cadherin and Annexin 2. Moreover, resveratrol inhibited the hedgehog (HH) signals and tumor necrosis factor receptor (TNFR)-associated factor 6 (TRAF6) levels exhibiting the therapeutic action on castration-sensitive and castration-resistant PC cell lines. In summary, the overexpression of TRAF6 enhanced the viability and EMT progression of cancer cells. The resveratrol could alleviate the TRAF6 effect and regulate the HH signal to affect PC progression.
Supplementary Information
The online version contains supplementary material available at 10.1007/s10616-022-00544-0.
Keywords: Resveratrol, Prostate cancer, Hedgehog, TRAF6
Introduction
Prostate cancer (PC) is the most affecting cancer in terms of the death ratio of males. Recent statistics indicated that approximately 165,000 newly diagnosed PC cases were reported in the United States in 2018. Notably, the typical PC can be treated well if diagnosed early. On the other end, PC is associated with high mortality at the later stages, requiring rapid intervention due to its highly invasive phenotype. Therefore, the development of appropriate therapeutic options against PC is of significant clinical significance, for instance, effective anti-metastasis drugs (Li et al. 2014). In general, PC is a hormone-dependent ailment, which can be treated by administering exogenous estrogen as one of the earliest treatments for this disease. Subsequently, alternative treatments have been applied to inhibit tumor growth-supporting androgens and induce cell death in the prostate glands. Androgen deprivation therapy (ADT) has gradually replaced the estrogen-based treatment. However, the utilization of ADT showed a high incidence of cardiovascular complications and a low overall survival rate (Krishna Moorthy et al. 2019). Although the PC cells initially responded to ADT, most patients with metastatic PC eventually developed undesired resistance to ADT, referred to as castration-resistant PC (CRPC). The metastatic CRPC often leads to death as the median survival of patients with CRPC is less than 2 years (Pak et al. 2019), and the mean 5-year survival rate is merely 15.4%.
Indeed, the evolutionarily conserved Hedgehog (HH) signaling pathway often contributes to cell proliferation, as well as the determination of cell fate (Kar et al. 2017; Hyuga et al. 2019). Previous reports indicated that the aberrant increase of HH signaling resulted in the abnormal dissemination of prostate epithelial cells, producing tumor cells and increasing tumor transformation, as well as epithelial–mesenchymal transition (EMT) (Montano et al. 2018). These consequences often result in tumor dissemination, promoting continuous proliferation, survival, and metastasis of PC cells (Montano et al. 2018). Resveratrol, a natural non-flavonoid polyphenol compound found in grapes, berries, and peanuts, has garnered enormous interest from researchers due to its antitumor, anti-mutagenic, anti-microbial, anti-oxidant, apoptosis induction, and regulation of estrogen properties, among others (Li et al. 2013, 2014; Dias et al. 2013). In this context, several reports demonstrated that resveratrol could substantially exhibit fundamental effects in suppressing tumor growth of PC (Li et al. 2013, 2014; Dias et al. 2013). Despite exploring various therapeutic functionalities, the precise mechanisms of action, specifically against PC, remain largely unknown.
Over a decade, resveratrol has been widely recognized as a multi-targeting compound. In several instances, it was reported that the abnormal expression of the tumor necrosis factor receptor (TNFR)-associated factor 6 (TRAF6) affected the EMT progression of gastric and PCs (Yang et al. 2020; Aripaka et al. 2019). In another case, miRNA-140-3p-targeted TRAF6 significantly inhibited the EMT of osteosarcoma (Guo et al. 2020). Recently, TRAF6 was considered a target factor of resveratrol, which substantially alleviated the migration and proliferation efficacies of PC and inflammation-related ailments (Khusbu et al. 2020; Chen et al. 2020; Ge et al. 2019). Previous studies indicated that SMO/TRAF6 pathway played a crucial role in the doxorubicin resistance and mediated the crosstalk between HH and NF-ƙB signals (Qu et al. 2018; Cai et al. 2019). Inspired by these facts and considerations, we propose that TRAF6/SMO signals may involve in the mechanism of resveratrol. To demonstrate these aspects, in this study, PC mouse xenograft models of PC in mice were initially constructed using different PC cell lines (LNCaP and PC-3). Further, we used these models to analyze the efficacy of resveratrol against the lipopolysaccharide (LPS)-induced EMT and changes of TRAF6, as well as HH signal. Together, we anticipate that resveratrol can be a suitable candidate for preventing the tumor metastasis of PC in mice.
Materials and methods
Cell culture
The LNCaP and PC-3 human PC cell lines were purchased from the Chinese Academy of Sciences (Shanghai institute of biology, Shanghai, China) and cultured in a basal Dulbecco’s Modified Eagle Medium (DMEM, Gibco, Carlsbad, USA) supplemented with 10% fetal bovine serum (FBS, Gibco) under 5% CO2 and 37 °C according to the instructions.
Construction of xenograft tumor model
The investigations in vivo were performed by initially establishing the tumor xenograft model in mice. The animal experiments were approved by the Research Ethics Committee of The Second Affiliated Hospital of Xi’an Jiaotong University (Xi’an, China). Male BALB/c nude mice (Four- to six-week-old, Wu Animal Center, Fuzhou, China) were housed in a controlled environment with humidity and temperature. It should be noted that the mice were allowed to acclimatize to the environment for a week before the experiment. To construct a xenograft model, PC-3 cells (5 × 106) and LNCaP cells (1 × 107) were injected subcutaneously into the mice on the back, respectively. At the determined time points, the tumor formation was monitored. Further, the developed xenograft mice (induced with PC-3 and LNCaP) were randomly distributed into four treatment groups (n = 6 each) of blank control (control), LPS (from Escherichia coli O55:B5, 5 mg/kg, L2880, Sigma Aldrich, St. Louis, MO, USA), resveratrol (Res, 50 mg/kg, cas: 501-36-0, Sigma Aldrich), and LPS + resveratrol (LPS + Res) (Dias et al. 2013). The mice were treated with the respective sample by intraperitoneal injections. At the regular intervals, the tumor volumes were monitored every week by measuring the volume based on the formula, volume = ½ (length × width2). Then, the mice were euthanized on day-29 of treatment and sacrificed to separate the tumors. The xenograft tumors were rapidly removed and weighed. Finally, a portion of the tumor was immediately placed in liquid nitrogen for subsequent biomarker analysis, while the rest of the tumor mass was sliced and fixed in 10% formalin for immunohistochemical analysis.
Western blotting (WB)
Briefly, the cells and excised tissues were initially subjected to radioimmunoprecipitation lysis buffer with protease inhibitors (Roche, Basel, Switzerland) to lyse for WB. Further, the Pierce bicinchoninic acid assay was applied for protein quantitation. Then, the quantitatively determined protein content was subjected to sodium dodecyl sulfate-polyacrylamide gel electrophoresis (SDS-PAGE, Sigma) and transferred to polyvinylidene difluoride (PVDF, Bio-Rad Hercules, CA, USA) membranes. Then, the membranes were incubated with specific primary antibodies (Abcam, Cambridge, UK) and horseradish peroxidase (HRP)-labeled secondary antibodies (Abcam), respectively. Finally, an Enhanced Chemiluminescence (ECL) solution was used for signal development, and the ImageJ software was used to semi-quantitatively analyze the blots.
Immunohistochemistry
The immunohistochemical analysis was performed on the tissue sections of xenograft mice. Initially, the slides with paraffin-embedded tissue sections were incubated at 60 °C for 1 h and then dewaxed through repeated immersion in xylene and ethanol. Subsequently, the slides were immersed in 0.1 M citrate buffer (pH 6.0) and incubated with 3% hydrogen peroxide. Then, the tissue sections were incubated with phosphate-buffered saline (PBS) containing bovine serum albumin (BSA), followed by staining with the corresponding fluorescently-labeled antibodies. Finally, the images of the tissue were captured using the fluorescent microscope.
TRAF6 overexpression experiment
The expression vector of TRAF6 was purchased from Sangon Biotech Co., Ltd. (Shanghai, China). Lipofectamine™ transfection 3000 (Invitrogen, Waltham, USA) was used to transfect the recombinant overexpression plasmid of TRAF6, following the manufacturer’s instructions. In addition, opti-MEM was used to dilute the plasmids and transfection agents. Further, the mixture of plasmids and transfection agents was added to the cells (5 × 104 cells per well in the 6-well plate) and incubated at 37 °C for 5% CO2. Then, the mixture was replaced with the complete medium after 6 h of transfection. Finally, the expression was determined by RT-qPCR after incubating for 48 h.
Cell viability assay
Initially, the PC cells were seeded in the 96-well plates at a density of 1 × 104 and incubated for 24 h in the absence or presence of resveratrol at different concentrations (5, 15, 25, 35, 45, and 50 µM). Then, 50 µL of 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide (MTT, Sigma) reagent was added to the wells, and the incubation was further continued for 4 h. Further, the medium in the wells was pirated, and 150 μL of dimethyl sulfoxide (Sigma) was added to dissolve the formazan crystals and stop the reaction. The reduction of MTT was measured at 550 nm using the ELIS reader. Each experiment was repeated in triplicates. The concentration of resveratrol in vitro was selected according to the cell proliferation results. Subsequently, the cell viability after transfection was also measured using the same method.
Cell invasion assay
The cells cultured with the decarbonated serum were divided into the following four groups: control, TRAF6 overexpression (Ov-ex), Ov-ex + Resveratrol (Ov-er + Res, resveratrol, 25 µM), and Ov-er + inhibitor (Ov-er + Inh, inhibitor GANT61). The Matrigel-coated transwell chambers were used for the cell invasion study. The cells from various treatment groups were digested with trypsin, and single-cell suspensions were then prepared in a serum-free culture medium. The suspension of cells and the medium supplemented with FBS were added to the special chamber. Then, the plates were incubated at 37 °C for 24 h. Further, the culture medium was aspirated, followed by subsequently rinsing the chambers with PBS. Afterward, the chambers were fixed with methanol for 30 min, and a cotton swab was used to gently remove the non-migrated cells. Then, the chambers were stained with 0.1% crystal violet for 2 min and repeatedly washed with PBS. Finally, the migration status was observed by counting the invaded cells to evaluate the invasion index under a light microscope (Nikon, Tokyo, Japan).
qPCR
PC cells were seeded at a density of 5 × 104 cells per well in the 6-well plates for 24 h. Then, the total RNA was extracted from the cells using the TRIzol method, and the RNA concentrations were measured by NanoDrop 2000 spectrophotometer. Further, the cDNA reverse transcription kit (Life Technologies, USA) was used to synthesize cDNA from RNA. The iQ SYBR Green Supermix was used for determining the relative expressions by qPCR (ProFlex™ PCR system, ThermoFisher) through standard curve analysis. Glyceraldehyde 3-phosphate dehydrogenase (GAPDH) was used as the endogenous control gene for normalization. The primer sequences are shown in Table 1.
Table 1.
Primer sequences used for RT-qPCR measurements
| Genes | Forward | Reverse |
|---|---|---|
| Bax | TTTCTGACGGCAACTTCAACTG | CAACCACCTGGTCTTGGAT |
| Bcl-2 | CGCAGAGGGGCTACGAGT | GTTGACGCTCTCCACACACAT |
| P53 | TCCGACTATACCACTATCCACTACA | GGCACAAACACGAACCTCAAA |
| MMP7 | TGAGCTACAGTGGGAACAGG | ACCACCCCAAAGAAAATTCC |
| E-cadherin | CGCATTGCCACATACA | CGTTAGCCTCGTTCTCA |
| Vimentin | CGCTTCGCCAACTACAT | AGGGCATCCACTTCACAG |
| Annexin-2 | ACGCTGGAGTGAAGAGGAAA | AAGGCACTGAGACTCCCTCA |
| VEGF | TCTACCTCCACCATGCCAAGT | GATGATTCTGCCCTCCTCCTT |
| Gli 1 | ATCCTTACCTCCCAACCTCTGT | AACTTCTGGCTTCCTGTAGC |
| Ptch 1 | CAGGCAGCGGTAGTAGTGG | TGTAGCGGGTATTGTCGTGT |
| SMO | CTCCTACTTCCATTCGCTCAC | CAAAACAAATCCCACTCACAGA |
| GAPDH | CAATGACCCCTTCATTGACC | GACAAGCTTCCCGTTCTCAG |
Statistical analysis
All data were expressed as mean ± standard deviation (SD). Data were analyzed using SPSS 21.0 statistical software. Comparison between multiple groups was conducted using one-way ANOVA at a defined statistical significance of P < 0.05. * defines P < 0.05, ** indicates P < 0.01, and *** represents P < 0.001.
Results
Resveratrol inhibits tumor growth in vivo
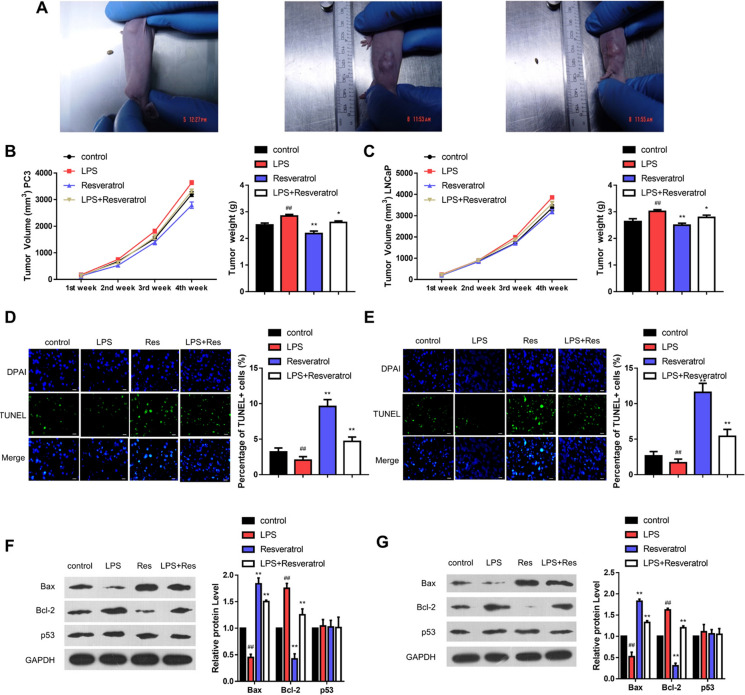
As depicted in Fig. 1A, LPS has significantly promoted the growth of PC-3- and LNCaP-derived tumors in mice. It was observed from the experimental results that the resultant volume and weight of tumors after LPS treatment were the largest of all the treatment groups. Contrarily, the resveratrol treatment group showed the smallest volume and weight of xenograft among all the treatment groups (Fig. 1B, C). At the same time, the tumor volume and weight of the LPS + resveratrol treatment group were higher than the blank treatment group (Fig. 1B, C). Further, The TUNEL experiments showed that resveratrol promoted apoptosis in the PC-3-derived (Fig. 1D) and LNCaP-derived tumors (Fig. 1E).
Fig. 1.
Resveratrol inhibits tumor growth in vivo. A Bright-field images show the formation of PC-3 and LNCaP cells-based xenografts in nude mice. B Resveratrol reduces the tumor volume induced by LPS. C Resveratrol alleviates the tumor weight induced by LPS. D The TUNEL staining shows the effect of resveratrol on the apoptosis of PC-3-induced tumors. E The TUNEL staining presents the effect of resveratrol on the apoptosis of LNCaP-induced tumors. F The WB analysis presents the effect of resveratrol on the relative expressions of Bax, Bcl-2, and p53 proteins in PC-3-induced tumors and their corresponding quantified levels. G The WB analysis indicates the effect of resveratrol on the relative expression of Bax, Bcl-2, and p53 proteins in LNCaP-induced tumors and their corresponding quantified levels (# refers to comparison with control group, # indicates P < 0.05 and ## represents P < 0.01; * refers to comparison with LPS group, * indicates P < 0.05 and ** represents P < 0.01; n.s. no significant)
Further, to explore the mechanistic views, the expression levels of various apoptotic proteins were determined using the WB analysis. The WB analysis showed that resveratrol significantly upregulated the relative expression of Bax protein level and downregulated Bcl-2 protein, indicating the promotion of apoptosis in PC-3-derived (Fig. 1F) and LNCaP-derived tumors (Fig. 1G). However, resveratrol and LPS + resveratrol treatment groups showed no significant difference in the p53 expression compared with the LPS treatment group (Fig. 1F, G). Accordingly, we have concluded that resveratrol induced apoptosis in a non-p53-dependent manner (Tover et al. 2011). Nevertheless, it should be noted that they displayed no significant difference between PC-3-derived and LNCaP-derived tumors (Fig. S1). Collectively, resveratrol could inhibit tumor growth of PC via inducing cell apoptosis.
Resveratrol affects the EMT pathway
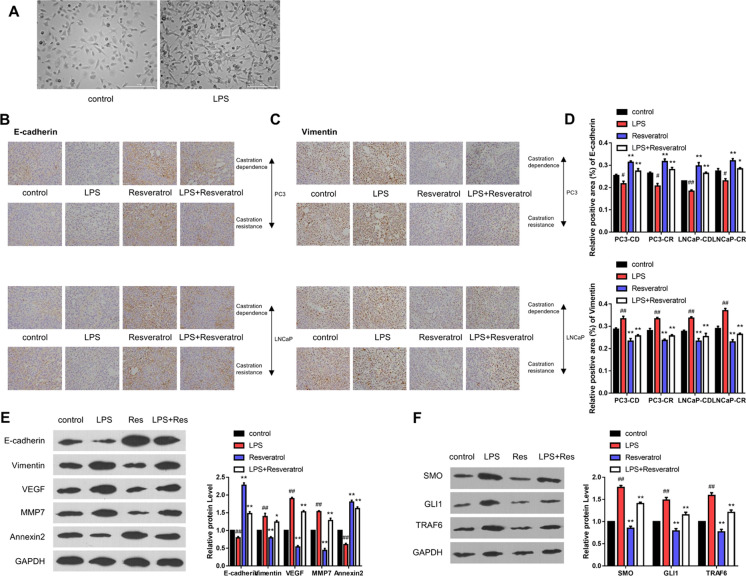
It was observed from the experimental results that the LPS exposure has significantly induced the morphological EMT changes of PC-3 cells in vitro (Fig. 2A). Further, in vivo experiments also showed that expression of the mesenchymal marker vimentin was increased, while the relative expression of E-cadherin was decreased in the LPS treatment group (P < 0.01; Fig. 2B, C, D). Interestingly, the resveratrol treatment has significantly inhibited these effects of LPS, in which the relative expression of E-cadherin was increased, and vimentin level was decreased. In addition, resveratrol treatment downregulated the expression of various genes associated with metastasis, including matrix metalloproteinase-7 (MMP-7) and vascular endothelial growth factor (VEGF). In contrast, the annexin expression was significantly upregulated (Fig. 2E). Furthermore, resveratrol significantly inhibited PC metastasis and reduced tumor angiogenesis. Together, these results showed that the anti-invasion effects of resveratrol were exerted by inhibiting HH signal and EMT pathways. Further, the LPS-mediated EMT activated the HH signal and increased the expression of glioma-associated oncogene homolog (GLI1), TRAF6, and SMO. On the other hand, resveratrol inhibited the HH pathway and reduced the expression of GLI1, TRAF6, and SMO (P < 0.05; Fig. 2F).
Fig. 2.
Resveratrol inhibits EMT progression and HH signal in vitro. A Representative microscopic images of PC-3 cells show the EMT morphological changes induced by LPS treatment. B Resveratrol enhances the expression of E-cadherin. C Resveratrol decreases the vimentin level. D The immunohistochemical staining presents the relative expressions of E-cadherin and vimentin. E The WB analysis shows the relative expressions of the various proteins of EMT, i.e., E-cadherin, Vimentin, VEGF, MMP-7, and Annexin 2, compared to GAPDH. F Resveratrol inhibits the HH signal in vivo (# refers to comparison with control group, # indicates P < 0.05, ## represents P < 0.01; * refers to comparison with LPS group, * indicates P < 0.05, ** represents P < 0.01; n.s. means no significant)
Resveratrol acts the role through HH signal
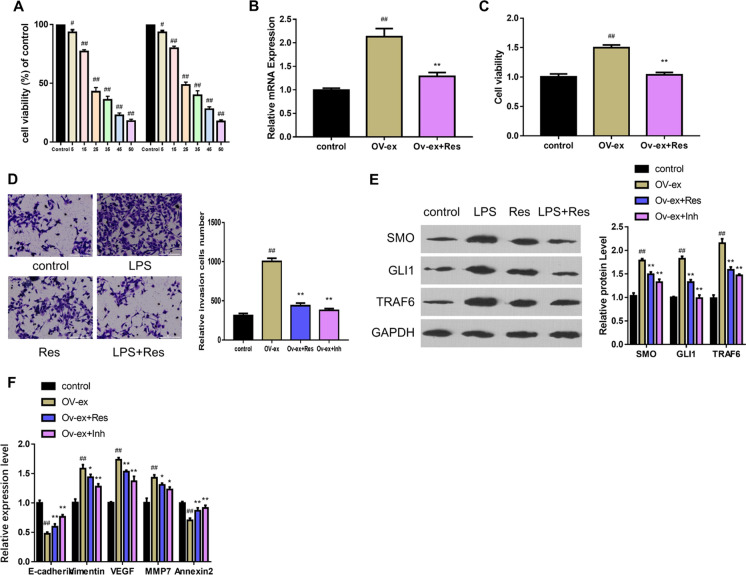
From the in vitro investigations, it was observed that resveratrol showed significant inhibitory effects on the viabilities of PC cell lines. The proliferation rate of these cells gradually decreased (P < 0.05; Fig. 3A) with an increase in the concentration of resveratrol. To investigate the mechanism of resveratrol, the HH signal inhibitors of GANT61 and TRAF6 overexpression vector were used. As depicted in Fig. 3B and C, the viabilities of the cell were enhanced after transfection, in which the level of TRAF6 was positively correlated to the cell activation. As resveratrol inhibited the proliferation of PC cells, it could be correlated that resveratrol had a similar potential with HH signal inhibitor. In the presence of an HH inhibitor, the invasion ability by TRAF6 overexpression was slightly reduced (Fig. 3D). Moreover, the relative expressions of various proteins, i.e., SMO, GLI1, and TRFA6, were downregulated by resveratrol and inhibitor. However, GANT61 treatment was more effective than resveratrol (Fig. 3E). Predictably, the relative expression levels of E-cadherin and Annexin were upregulated by resveratrol and inhibitor, while Vimentin, VEGF, and MMP7 expression levels were decreased compared with TRAF6 overexpression (Fig. 3F).
Fig. 3.
Resveratrol alleviates EMT by targeting TRAF6 and regulates HH signal. A MTT assay results show the cell viability at the different concentrations of resveratrol. B RT-PCR analysis shows the TRAF6 expression after transfection; C TRAF6 overexpression enhances the cell viability. D Resveratrol downregulates the increase of cell invasion induced by TRAF6 overexpression. E The WB analysis determines relative levels of factors in the HH signal pathway. F RT-PCR determines the relative expressions of various EMT factors (# refers to comparison with control group, # indicates P < 0.05 and ## represents P < 0.01; * refers to comparison with Ov-ex group, * indicates P < 0.05 and ** represents P < 0.01; n.s. means no significant)
Discussion
CRPC has become a significant challenge for PC therapy (Sun et al. 2019). In this context, various androgen receptor (AR)-dependent mechanisms are involved in some crucial pathways for developing CRPC, including but not limited to amplification and mutations of AR, aberrant expression of AR splice variants, increased androgen production, as well as altered expression of AR co-activation and co-inhibition factors. However, several reports demonstrated that CRPC tumors in castrated mice required selective pressure, which might not be associated with androgens and involved no CRPC cells induced by overexpression of AR.
To this end, a crosstalk between the HH signaling and AR pathways, as well as their effects on the tumor microenvironment, may undoubtedly aid in the formation of a castration-resistant phenotype. Notably, several reports demonstrated the aberrant HH signaling pathway might promote tumorigenesis, survival, and invasion by regulating the cell cycle, and cell apoptosis (Kar et al. 2017; Pak et al. 2019; Yang et al. 2019). Owing to these facts, it could be anticipated that there exists a relationship between HH protein and pT stage and pathological G. It should be noted that the expression of GLI2 was positively correlated with pathology (Gonnissen et al. 2017). In this context, several reports mainly focused on blocking HH pathway-mediated paracrine signals to delay the CRPC-assisted tumor progression (Yang et al. 2019; Cortes et al. 2019). However, the current anti-PC therapies show some undesired shortcomings. Compared to various classical therapies available, natural products and dietary xenoestrogens from plants often show improved inhibitory actions against PC development, which can be explored to become anticancer drugs (Massah et al. 2021). Among various naturally available products, resveratrol has garnered enormous interest due to various pharmacological actions (Li et al. 2013, 2014; Dias et al. 2013).
Interestingly, a stilbene synthase-triggered formation resulted in the formation of cis- and trans-isomers of resveratrol. These altered isomeric structures could offer unique properties and target multiple receptor domains (Ashrafizadeh et al. 2021). In addition, several reports demonstrated that resveratrol could synergistically improve the inhibitory actions of doxorubicin on the formation of angiogenesis in tumors (Uvez et al. 2020). In several instances, resveratrol showed its therapeutic effects on chemotherapy-sensitive and tumor cell apoptosis, as well as targeting microRNA, and ERK1/2, among others. Previous studies indicated that resveratrol could exert fundamental effects in suppressing tumor growth of PC. However, the specific mechanisms are yet to be explored comprehensively (Li et al. 2013, 2014; Dias et al. 2013). This study explored that resveratrol could significantly inhibit the changes induced by EMT and upregulate the relative expression levels of E-cadherin and Annexin 2.
Moreover, the abnormal activation of HH and levels of SMO, GLI1, and TRAF6 were significantly inhibited by resveratrol. As the expression of TRAF6 is significantly associated with cell viability, the effects of resveratrol on TRAF6 and HH signals were verified in vitro. It was observed from the cell proliferation results that resveratrol alleviated viability and invasion abilities of cells through targeting TRAF6. In addition, resveratrol showed a similar potential ability to the GANT61 inhibitor. Further, resveratrol downregulated the level of TRAF6, and the stable interactions of SMO-TRAF6 were interrupted by regulating the HH signal. Together, our findings indicated that the TRAF6 was overexpressed in EMT, and resveratrol played a crucial role in PC-EMT by targeting TRAF6 and inhibiting the HH pathway activation.
Conclusion
Resveratrol could inhibit the LPS-induced EMT both in vivo and in vitro, and this process was shown to be mediated by HH signaling. Our results have provided new insights into the treatment of PC.
Supplementary Information
Below is the link to the electronic supplementary material.
Figure S1 Differences between PC-3-derived and LNCaP-derived tumors for 4 weeks. Graphical representations illustrate the tumor volume of PC-3 and LNCaP cells in vivo in the (A) the control group, (B) the LPS group, (C) the resveratrol group, (D) the LPS+resveratrol group. (*refers to PC-3 comparison with LNCaP group, * indicates P < 0.05; n.s. means no significant) (JPG 383 kb)
Acknowledgements
We sincerely acknowledge the support of the Second Affiliated Hospital of Xi’an Jiaotong University.
Author contributions
Conceptualization and original draft preparation: JL; data curation: ZW; formal analysis: HL; investigation: JC; methodology: NN and YL; software: XZ and TC; Review and editing: JL.
Declarations
Conflict of interest
The authors declare that they have no conflicts of competing interests.
Consent to participate
Not applicable.
Footnotes
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
References
- Aripaka K, et al. TRAF6 function as a novel co-regulator of Wnt3a target genes in prostate cancer. EBioMedicine. 2019;45:192–207. doi: 10.1016/j.ebiom.2019.06.046. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ashrafizadeh M, et al. Antitumor activity of resveratrol against gastric cancer: a review of recent advances with an emphasis on molecular pathways. Cancer Cell Int. 2021;21(1):66. doi: 10.1186/s12935-021-01773-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cai K, et al. Targeting the crosstalk between the hedgehog and NF-κB signaling pathways in multiple myeloma. Leuk Lymphoma. 2019;60(3):772–781. doi: 10.1080/10428194.2018.1493727. [DOI] [PubMed] [Google Scholar]
- Chen Q, et al. Resveratrol attenuates neuroinflammation after deep hypothermia with circulatory arrest in rats. Brain Res Bull. 2020;155:145–154. doi: 10.1016/j.brainresbull.2019.12.008. [DOI] [PubMed] [Google Scholar]
- Cortes JE, et al. Hedgehog signaling inhibitors in solid and hematological cancers. Cancer Treat Rev. 2019;76:41–50. doi: 10.1016/j.ctrv.2019.04.005. [DOI] [PubMed] [Google Scholar]
- Dias S, et al. Trimethoxy-resveratrol and piceatannol administered orally suppress and inhibit tumor formation and growth in prostate cancer xenografts. Prostate. 2013;73(11):1135–1146. doi: 10.1002/pros.22657. [DOI] [PubMed] [Google Scholar]
- Ge Y, et al. Resveratrol protects BV2 mouse microglial cells against LPS-induced inflammatory injury by altering the miR-146a-5p/TRAF6/NF-κB axis. Immunopharmacol Immunotoxicol. 2019;41(5):549–557. doi: 10.1080/08923973.2019.1666406. [DOI] [PubMed] [Google Scholar]
- Gonnissen A, et al. Tissue microarray analysis indicates hedgehog signaling as a potential prognostic factor in intermediate-risk prostate cancer. BMC Cancer. 2017;17(1):634. doi: 10.1186/s12885-017-3619-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Guo Q, et al. By targeting TRAF6, miR-140-3p inhibits TGF-β1-induced human osteosarcoma epithelial-to-mesenchymal transition, migration, and invasion. Biotech Lett. 2020;42(11):2123–2133. doi: 10.1007/s10529-020-02943-9. [DOI] [PubMed] [Google Scholar]
- Hyuga T, et al. Hedgehog signaling for urogenital organogenesis and prostate cancer: an implication for the epithelial–mesenchyme interaction (EMI) Int J Mol Sci. 2019;21(1):58. doi: 10.3390/ijms21010058. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kar S, et al. SOX2 function and Hedgehog signaling pathway are co-conspirators in promoting androgen independent prostate cancer. Biochim Biophys Acta. 2017;1863(1):253–265. doi: 10.1016/j.bbadis.2016.11.001. [DOI] [PubMed] [Google Scholar]
- Khusbu F, et al. Resveratrol induces depletion of TRAF6 and suppresses prostate cancer cell proliferation and migration. Int J Biochem Cell Biol. 2020;118:105644. doi: 10.1016/j.biocel.2019.105644. [DOI] [PubMed] [Google Scholar]
- Krishna Moorthy H, Laxman Prabhu GG, Venugopal P. The resurgence of estrogens in the treatment of castration-resistant prostate cancer. Indian J Urol. 2019;35(3):189–196. doi: 10.4103/iju.IJU_56_19. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Li G, et al. Dietary resveratrol prevents development of high-grade prostatic intraepithelial neoplastic lesions: involvement of SIRT1/S6K axis. Cancer Prev Res (phila) 2013;6(1):27–39. doi: 10.1158/1940-6207.CAPR-12-0349. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Li J, et al. A novel anticancer effect of resveratrol: reversal of epithelial–mesenchymal transition in prostate cancer cells. Mol Med Rep. 2014;10(4):1717–1724. doi: 10.3892/mmr.2014.2417. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Li J, et al. A novel anti-cancer effect of resveratrol: reversal of epithelial–mesenchymal transition in prostate cancer cells. Mol Med Rep. 2014;10(4):1717–1724. doi: 10.3892/mmr.2014.2417. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Massah S, et al. GLI activation by the estrogen receptor in breast cancer cells: regulation of cancer cell growth by Gli3. Mol Cell Endocrinol. 2021;522:111136. doi: 10.1016/j.mce.2020.111136. [DOI] [PubMed] [Google Scholar]
- Montano M, et al. Dual regulation of decorin by androgen and Hedgehog signaling during prostate morphogenesis. Dev Dyn. 2018;247(5):679–685. doi: 10.1002/dvdy.24619. [DOI] [PubMed] [Google Scholar]
- Pak S, et al. The small molecule WNT/β-catenin inhibitor CWP232291 blocks the growth of castration-resistant prostate cancer by activating the endoplasmic reticulum stress pathway. J Exp Clin Cancer Res. 2019;38(1):342. doi: 10.1186/s13046-019-1342-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pietrobono S, Gagliardi S, Stecca B. Non-canonical Hedgehog signaling pathway in cancer: activation of GLI transcription factors beyond smoothened. Front Genet. 2019;10:556. doi: 10.3389/fgene.2019.00556. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Qu C, et al. Smoothened stabilizes and protects TRAF6 from degradation: a novel non-canonical role of smoothened with implications in lymphoma biology. Cancer Lett. 2018;436:149–158. doi: 10.1016/j.canlet.2018.08.020. [DOI] [PubMed] [Google Scholar]
- Sun W, et al. Combination of phospholipase Cε knockdown with GANT61 sensitizes castration-resistant prostate cancer cells to enzalutamide by suppressing the androgen receptor signaling pathway. Oncol Rep. 2019;41(5):2689–2702. doi: 10.3892/or.2019.7054. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tovar C, et al. MDM2 antagonists boost antitumor effect of androgen withdrawal: implications for therapy of prostate cancer. Mol Cancer. 2011;10:49. doi: 10.1186/1476-4598-10-49. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Uvez A, et al. Synergistic interactions between resveratrol and doxorubicin inhibit angiogenesis both in vitro and in vivo. Pol J Vet Sci. 2020;23(4):571–580. doi: 10.24425/pjvs.2020.135803. [DOI] [PubMed] [Google Scholar]
- Yang R, et al. Polymeric micellar delivery of novel microtubule destabilizer and Hedgehog signaling inhibitor for treating chemoresistant prostate cancer. J Pharmacol Exp Ther. 2019;370(3):864–875. doi: 10.1124/jpet.119.256628. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yang M, et al. TRAF6 promotes gastric cancer cell self-renewal, proliferation, and migration. Stem Cells International. 2020;2020:3296192. doi: 10.1155/2020/3296192. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Figure S1 Differences between PC-3-derived and LNCaP-derived tumors for 4 weeks. Graphical representations illustrate the tumor volume of PC-3 and LNCaP cells in vivo in the (A) the control group, (B) the LPS group, (C) the resveratrol group, (D) the LPS+resveratrol group. (*refers to PC-3 comparison with LNCaP group, * indicates P < 0.05; n.s. means no significant) (JPG 383 kb)