Abstract
Painful post-traumatic trigeminal neuropathy (PPTTN) can result from iatrogenic injury to one or more branches of the trigeminal nerve during oral surgical procedures such as tooth extractions. Like other chronic neuropathic pain conditions, PPTTN can significantly alter the patient’s quality of life, especially when pharmacological treatment is ineffective or not tolerated. As such, new treatment options have been investigated, including local injections of botulinum toxin type A (BTX-A). A 29-year-old woman presented to our tertiary orofacial pain clinic for evaluation of chronic electric shock-like pain attacks and severe allodynia in the territory of the right inferior alveolar nerve and buccal nerve following right mandibular third molar extraction 3 years prior. Following several failed attempts at classic pharmacological management (including carbamazepine, venlafaxine, duloxetine, pregabalin, clonazepam, and amitriptyline), BTX-A injections were administered in the vicinity of the right mental nerve. This treatment provided significant improvement in the patient’s condition and overall quality of life with no significant adverse effects. Because both neuropathies were significantly improved by remote BTX-A injections, this case report provides preliminary clinical evidence supporting spinopetal transport of BTX-A, as shown in animal models, as an underlying pathophysiological mechanism of BTX-A-mediated analgesia.
Keywords: Painful post-traumatic trigeminal neuropathy, botulinum toxin type A, facial pain, inferior alveolar nerve, buccal nerve, spinopetal transport
Introduction
Painful post-traumatic trigeminal neuropathy (PPTTN) is defined as “unilateral or bilateral facial or oral pain following and caused by trauma to the trigeminal nerve(s), with other symptoms and/or clinical signs of trigeminal nerve dysfunction.” 1 PPTTN is a complication of many oral surgical procedures, including dental extractions, after which it occurs in 0.5% to 12.0% of cases. 2 Like other chronic neuropathic pain conditions, PPTTN can significantly alter the patient’s quality of life,3–5 especially when improperly diagnosed and/or managed.
The pain experienced by patients with PPTTN is typically described as a continuous burning sensation that is sometimes associated with short-lasting paroxysms, which may occur both spontaneously and following mechanical stimulation of the skin and/or mucosa in the area of innervation of the injured nerve(s). 6 Pain onset is variable, occurring either immediately following nerve injury or up to 3 to 6 months following trauma.1,6 The pain is classically associated with sensory dysfunction, either positive (hyperalgesia, allodynia, itching) or negative (hypoesthesia, anesthesia).
Current management of PPTTN commonly relies on pharmacological and/or surgical treatment.2,6 As for other neuropathic pain conditions, pharmacological treatment is based mainly on antiepileptic drugs (carbamazepine, oxcarbazepine, pregabalin, gabapentin) and/or tricyclic antidepressants (amitriptyline, nortriptyline), although no formal guidelines exist for this specific condition. Unfortunately, the analgesic effect of such drugs is often limited or hampered by significant adverse effects and/or contraindications. 2 Surgical treatment is controversial and can be offered as a second-line option in specific cases only. Indeed, many patients who have undergone peripheral surgical interventions have experienced worsening pain. 2
Considering the quite challenging management of PPTTN, new therapeutic options have emerged with the hope of obtaining an analgesic effect that is better tolerated by patients. One such therapeutic option is local injections of botulinum toxin type A (BTX-A). There is growing evidence that BTX-A may have a significant analgesic effect in both traumatic7–9 and non-traumatic painful neuropathies10–13 with few adverse effects.10,14 More specifically, there is growing evidence that BTX-A may be effective in painful post-traumatic trigeminal neuropathy,15,16 as discussed in a recent review by Moreau et al. 17 From a pathophysiological standpoint, it has been suggested that BTX-A is endocytosed in local peripheral afferents, where the protease activity of BTX-A cleaves Soluble N-ethylmaleimide-sensitive factor Attachment protein REceptor (SNARE) complexes, thus inhibiting vesicular fusion and subsequent neurotransmitter release. 17 Animal studies have shown evidence of spinopetal transport of BTX-A along the primary afferent into the dorsal root ganglia and/or the central terminal, where it can block second afferent activation.18,19 To the best of our knowledge, however, no such spinopetal transport has been evidenced in humans to date.
We herein present a case involving a young woman who presented with refractory concurrent PPTTN of the right inferior alveolar nerve and buccal nerve following extraction of her right mandibular third molar 3 years prior. The PPTTN in this case was successfully managed by BTX-A injections in the vicinity of the right mental nerve. Therapeutic and pathophysiological implications of the present case are also discussed in this report.
Case report
The reporting of this study conforms to the CARE guidelines. 20
A 29-year-old woman with no medical history presented to our tertiary orofacial pain clinic in January 2020 for chronic intractable spontaneous and evoked pain in the right labiomental and preauricular regions following bilateral mandibular third molar extractions under general anesthesia in another hospital in 2017. The patient reported significant complications following the surgery, notably pain-related speech impairment for the first 5 postoperative days, major facial and labial postoperative edema, and trismus lasting for 1 month.
When the initial pain subsided, the patient developed complete right labiomental anesthesia followed several weeks later by paresthesias and finally unbearable pain in the innervation territories of the right inferior alveolar nerve and buccal nerve. The pain persisted despite numerous treatments and medical consultations in several hospitals and private practices.
At the time of the patient’s initial consultation in our clinic 3 years later, she reported continuous dull “crushing” pain accompanied by “electric shock”-like pain upon light touch to the right labiomental region. Chewing, talking, and brushing her teeth would also elicit such painful paroxysms, as did vibrations applied to the lower half of her face. Everyday activities were strongly hampered by such painful activities as talking, touching her lips, eating, kissing, sleeping on her right side, and wearing a mask (quite troublesome in the time of COVID-19-related mandatory mask-wearing). Because her professional activity (hospital nutritionist) required frequent talking, thus eliciting significant painful paroxysms, she had been placed on disability leave by her general practitioner a few weeks prior and was working part-time only at the time of her visit to our clinic.
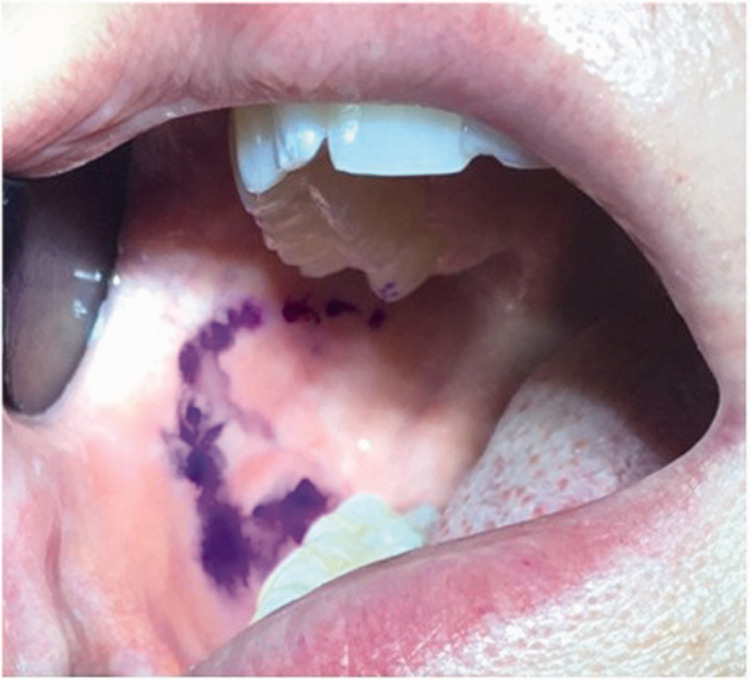
Physical examination revealed significant right labiomental mechanical (static) and thermal (cold and hot) allodynia accompanied by an avoidance response when the patient was approached. Right masseter muscle palpation was painful, but not left palpation; the pain was limited to the site of palpation, which was suggestive of reactive myospasm (i.e., local myalgia as defined in the diagnostic criteria for temporomandibular disorders 21 ). Intraoral examination revealed significant mechanical static (not dynamic) allodynia in the innervation territory of the right buccal nerve (Figure 1). No temporal summation of pain was objectified. Considering the obvious allodynia in both sites, no further sensory testing was deemed necessary. No mucosal lesion, unhealed socket, or neuroma was found along the right mandibular body and/or branch.
Figure 1.
Clinical view of the territory of intraoral buccal mucosa allodynia, mapped using a dermographic marker pen. This territory of allodynia was compatible with the innervation territory of the buccal nerve (see Reference 23 for comparison).
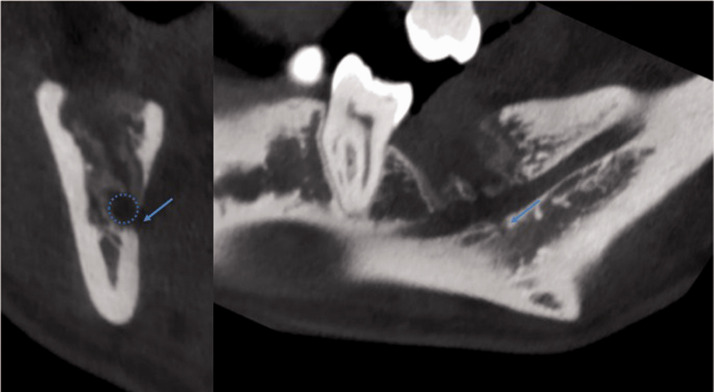
A three-dimensional cone-beam computed tomography (CBCT) imaging study had been performed 1 month after the third molar surgery (in 2017). This imaging examination showed perforation of the lingual mandibular cortex of the right lower third molar socket in the immediate vicinity of the inferior alveolar nerve bundle, the shape of which was consistent with a burr-induced iatrogenic injury (Figure 2). Brain magnetic resonance imaging had also been performed around the same period, but no significant anomalies were revealed. No further imaging studies were deemed necessary.
Figure 2.
Postoperative coronal (left) and sagittal (right) cone-beam computed tomography images of the socket of the right mandibular third molar 1 month after extraction. Note the burr-shaped lingual cortex destruction (arrows) in the immediate vicinity of the inferior alveolar nerve bundle (circle), suggestive of iatrogenic injury.
The patient’s history, physical examination findings, and CBCT results were consistent with a diagnosis of PPTTN of the right inferior alveolar nerve and buccal nerve based on the criteria established by Benoliel et al. 6 and the International Classification of Headache Disorders, 3rd edition (ICHD-3 13.1.2.3). 1 More specifically, the PPTTN affecting the inferior alveolar nerve fulfilled the five criteria described by Benoliel et al. 6 (definite neuropathic pain), whereas the PPTTN affecting the buccal nerve fulfilled only four criteria (probable neuropathic pain) because of a lack of imaging evidence. Both neuropathies could also be classified as “chronic neuropathic pain after peripheral nerve injury” according to the International Classification of Diseases, 11th revision (ICD-11). 22 From a mechanistic standpoint, the PPTTN affecting the inferior alveolar nerve was thought to be the result of iatrogenic burr-induced nerve dilaceration, whereas the buccal nerve had possibly been injured during the third molar flap incision (nerve transection) and/or reflection (nerve stretching), both frequent causes of buccal nerve injury.23,24
The patient had consulted several physicians during the few years after the extraction, and multiple pharmacological treatments were instituted either alone or in combination: carbamazepine, venlafaxine, duloxetine, clonazepam, pregabalin, and amitriptyline. All treatments were given at sufficient doses over several months (for each treatment) but with unsatisfactory results: the patient experienced either insufficient analgesic effects or too many adverse effects such as weight gain, difficulty focusing, memory loss, and fatigue. Right mental nerve block with a long-lasting anesthetic (ropivacaine at 7.5 mg/mL) suppressed the evoked labiomental pain but not the spontaneous pain. Therefore, at 3 years post-injury, the patient’s painful neuropathic condition was considered refractory according to the criteria by Smith et al., 25 and new therapeutic options were sought.
Local injections of BTX-A in the vicinity of the mental nerve were considered. The expected risks and benefits of such experimental treatment were presented to the patient, who eagerly accepted because of the significant alteration in her overall quality of life and morale. Informed verbal and written consent to undergo treatment was obtained before the first injection. At that time, the patient was still taking clonazepam at 2.5 mg/mL (6 drops three times daily) and pregabalin (150 mg daily), which provided minimal but noticeable pain relief. We decided not to modify this analgesic regimen for the time being.
Because the patient raised significant concern regarding possible speech impairment secondary to labial paralysis following the BTX-A injection, we opted for a progressive two-step injection technique.
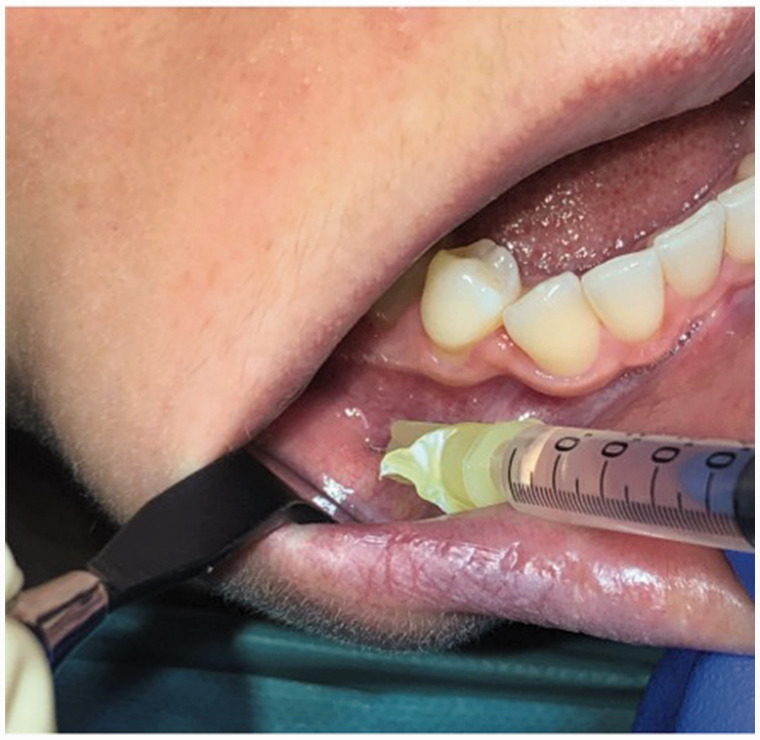
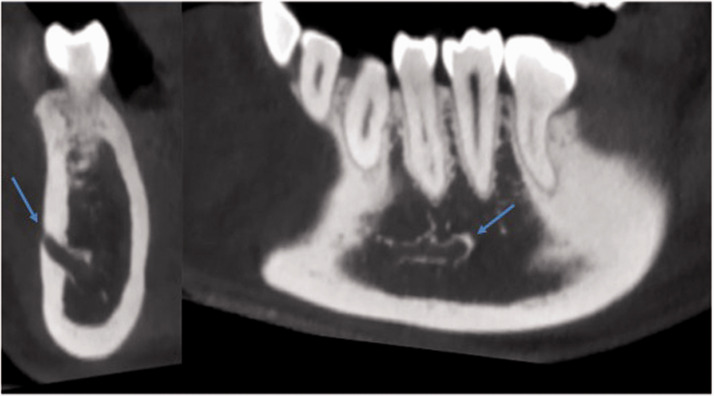
An initial dose of 25 IU of BTX-A (50 IU in 1 mL saline, Botox®; Allergan, Dublin, Ireland) was injected in the vicinity of the right mental foramen at the level of the interproximal region between the first and second premolars at a 2-cm depth from the marginal gingiva (Figure 3). This location was based on the CBCT imaging results (Figure 4). The injection was performed using a 1-mL syringe (BD Plastipak; Becton, Dickinson and Company, Franklin Lakes, NJ, USA) and a 30-G hypodermic needle (Hypodermic Needle Pro, 30G × 1/2″, 0.3 × 13 mm; Smiths Medical, Minneapolis, MN, USA). The BTX-A dose was chosen to obtain a rapid and significant analgesic effect, whereas a small injection volume (0.5 mL) was sought to limit possible diffusion into the surrounding muscles (especially those in the lower labial region). Following injection, discomfort and flushing appeared immediately but subsided rapidly. A follow-up appointment was set for day 15, and the patient was instructed to keep a detailed pain diary every day until the appointment.
Figure 3.
Clinical preoperative view of the botulinum toxin A injection in the vicinity of the right mental nerve bundle.
Figure 4.
Coronal (left) and sagittal (right) cone-beam computed tomography images (same as Figure 2) centered on the right mental foramen (arrows).
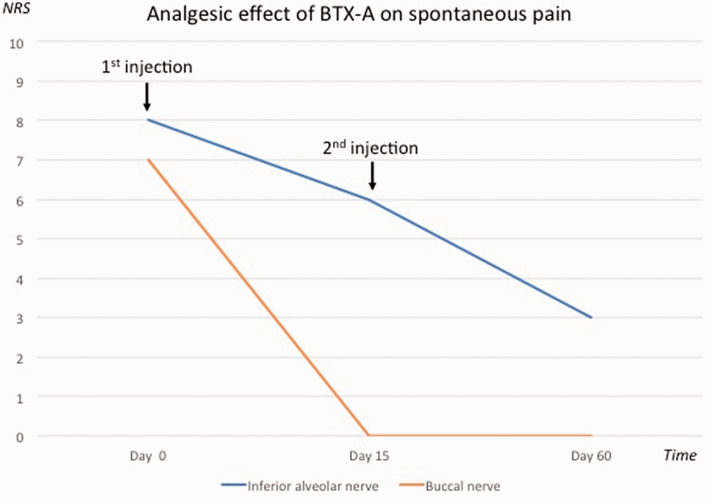
At the 15-day follow-up, the patient reported a progressive 25% improvement in spontaneous and mechanically evoked pain but persistence of thermal allodynia (Figure 5), which had appeared between days 5 and 10 following the injection. The lower lip allodynia, but not the chin allodynia, had disappeared. The intraoral buccal mucosa allodynia had also completely disappeared. No facial asymmetry was observed at rest. Labial paresis was minimal and did not impact speech or food intake (Video 1). The patient sought a second injection, duly informed of the possibility of increased labial paresis, but was clearly enthused by the preliminary analgesic results.
Figure 5.
Graphical representation of the analgesic effect of botulinum toxin A injections on spontaneous pain (self-reported by the patient on a 0 to 10 NRS), with separate evaluations for the inferior alveolar nerve territory (blue line) and buccal nerve territory (orange line).
NRS = numerical rating scale.
A second injection was performed using the same dose (25 IU) and volume (0.5 mL) but with a more distoapical injection point to ensure infiltration of the posteroinferior aspect of the mental nerve bundle (which innervates the chin region), as schematically illustrated in Figure 6. A further decrease in pain intensity was noted (63% decrease compared with baseline pain), and the chin allodynia completely disappeared (Figure 5). Spontaneous pain persisted but with a lower and tolerable intensity. The labial paresis was slightly aggravated, but still without any significant effect on speech or facial esthetics (Video 2).
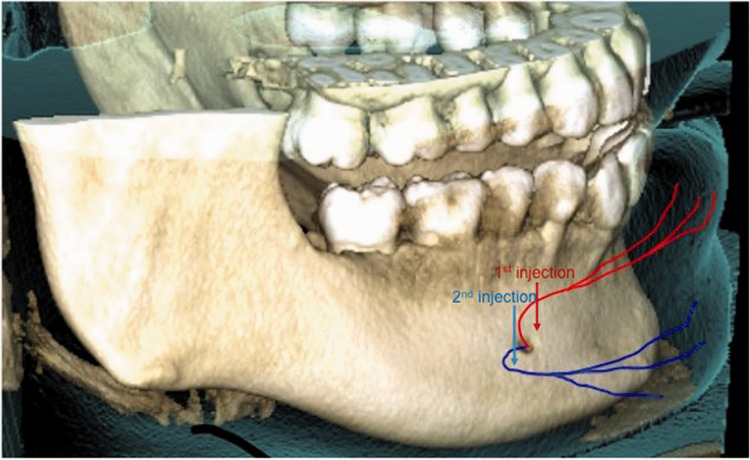
Figure 6.
Didactic illustration of the putative innervation of the patient’s labiomental region using a surface three-dimensional reconstruction of the patient’s cone-beam computed tomography volume. Putative innervation was conjectured based on the reported topography of analgesia following the first and second botulinum toxin A injections.
The overall therapeutic efficacy was evaluated based on three parameters: pain intensity (via an 11-point numerical rating scale), anxiodepressive comorbidities (via the Hospital Anxiety and Depression questionnaire), and overall impression of change (via the Patient Global Impression of Change questionnaire). The efficacy of the BTX-A injection therapy is summarized in Table 1. Overall, significant improvement was observed in all parameters based on the classic 30% improvement cutoff relevant to the pain field. 26 Significant improvement in the patient’s overall mood was also reported, as was observed by both our team and the patient’s husband.
Table 1.
Assessment of analgesic efficacy of BTX-A.
| Before BTX-A injections | After BTX-A injections | |
|---|---|---|
| Numerical rating scale score | IAN = 8/10Buccal nerve = 7/10 | IAN = 3/10Buccal nerve = 0/10 |
| Hospital Anxiety and Depression score | Anxiety = 8/21Depression = 10/21 | Anxiety = 7/21Depression = 8/21 |
| Patient Global Impression of Change score | N/A | 40% improvement |
BTX-A, botulinum toxin A; IAN, inferior alveolar nerve; N/A, not applicable.
At the 3-month follow-up, the pain had progressively returned to its initial stage (i.e., to the level reported during the first consultation in our clinic), and the patient sought another injection. A repeat injection was performed at that time with similar efficacy (decrease in the numerical rating scale score from 8 to 4). At the 6-month follow-up, upon learning of the patient’s pregnancy, the injections were stopped for safety reasons with plans to resume them after the pregnancy.
Discussion
BTX-A is one of seven types of antigenically different exotoxins produced by the gram-positive anaerobic bacteria Clostridium botulinum, which is known for its powerful paralytic effect (the main complication of botulism), and has been exploited for medical use in the fields of neuromuscular disease and cosmetology. 17 On a molecular level, BTX-A is a polypeptide composed of a 100-kDa heavy chain linked to a 50-kDa light chain by a disulfide bridge. The heavy chain binds to neurons, allowing the light chain to be translocated and exert its enzymatic protease activity. Upon endocytosis, the light chain cleaves the SNARE complexes. This prevents vesicular exocytosis of numerous neurotransmitters and neuromodulators, including acetylcholine, the main neurotransmitter responsible for muscle contraction at the neuromuscular junction level.27,28
Apart from the paralytic effect of BTX-A, recent evidence has suggested an analgesic effect resulting from blockade of the release of algogenic neuropeptides (such as substance P, neurokinin A, and calcitonin gene-related peptide), but also of glutamate, in small-diameter type C primary afferent nerve fibers. 17 This blockade prevents neurogenic inflammation and nociceptive signaling.
The analgesic effect of BTX-A is thought to take place not only in the peripheral terminal but also in the dorsal root ganglion and central terminal following spinopetal (axonal retrograde) transport as evidenced in preclinical models.18,19,29,30 Although several clinical studies have supported the analgesic effect of BTX-A in neuropathic pain and particularly in painful post-traumatic trigeminal neuropathy (as noted in a review by Moreau et al. 17 ), there is still much to learn regarding the underlying pathophysiology and efficacy of BTX-A in routine pain management.
The present report describes the successful management of two post-traumatic neuropathies affecting two branches of the same nerve (buccal nerve and inferior alveolar nerve) by perineural injections of BTX-A in the vicinity of the mental nerve, a terminal branch of the inferior alveolar nerve. This observation is consistent with the retrograde axonal transport of BTX-A shown in animal models but, to the best of our knowledge, never before evidenced in the clinical setting. The anti-allodynic and analgesic effects observed in this case may have thus resulted from the uptake of BTX-A in the mental nerve and subsequent spinopetal transport to the trigeminal ganglion and/or the trigeminal nucleus via the inferior alveolar nerve. Notably, because rare mental–buccal nerve anastomoses have been reported, 31 spinopetal transport could also have occurred in the buccal nerve, although this is less probable in the present case (considering the well-defined and limited innervation territory of the right buccal nerve as shown in Figure 1 and the resolution of the buccal allodynia following the first injection into the anterior aspect of the mental nerve, devoid of such anastomoses 31 ). To the best of our knowledge, such a perineural injection technique in the vicinity of the mental nerve has not been reported in the literature to date, especially in the treatment of an iatrogenic third molar extraction-related trigeminal nerve injury.
From a clinical standpoint, it is interesting to note that BTX-A provided significant pain relief in a patient with 3-year refractory painful neuropathy. This further supports its use in neuropathic pain management either alone or as an add-on therapy, in adherence with the most recent neuropathic pain management guidelines. 32 Nevertheless, it is possible that BTX-A does not exert an analgesic effect on all peripheral neuropathies depending on the neuropathic pain phenotype. Further studies involving specific neuropathic pain phenotype clustering could help to clarify this important issue.
From a technical standpoint, considering the remote effect of BTX-A, perineural mental nerve injections may prove to be very practical in the treatment of more proximal inferior alveolar nerve injuries resulting from commonly performed surgical interventions such as mandibular third molar extraction (as in the present case) or sagittal split mandibular osteotomy, with limited functional impairment (lip paresis) as evidenced herein (Videos 1 and 2). Nevertheless, such injections also have a limited risk of significant adverse effects related to the paralytic effect of BTX-A. Indeed, in all of the published cases in which intraoral injections of BTX-A were performed to treat trigeminal neuropathic pain, the injections were administered in the vicinity of the initial injury where the pain was felt by the patient.33,34 However, this would not have been possible in the present case because of the proximal location of the injuries and associated risks of dysphagia and/or hypoglossal nerve palsy that could have arisen from the spread of the toxin. Multiple injections of small doses and small volumes of BTX-A in the vicinity of the mental nerve seem to provide sufficient impregnation of the nerve’s branches without significant spread of the toxin that would lead to lip muscle paralysis.
Several inherent limitations apply to the present case, the findings of which should be analyzed with caution. This case report presents the successful symptomatic management of a single patient with no data regarding potential long-term treatment (painful post-traumatic trigeminal neuropathy is often chronic, if not incurable) and with a possible confounding placebo effect. However, we believe that a possible placebo effect is of minimal relevance in the present case, considering the timing of the analgesic effect (consistent with the onset of BTX-A pharmacological activity) and the severity/chronicity of the patient’s illness. Concurrent pharmacological treatments (clonazepam and pregabalin) could have also confounded the analysis of the analgesic effect of BTX-A; however, these medications were maintained at the same dosage during BTX-A treatment and had been taken at a stable dosage for many months before the first BTX-A injection. Finally, recall bias is always possible when comparing post-treatment pain with pre-treatment pain. Such bias was hopefully mitigated by the use of a pain diary, which required daily analysis of the patient’s pain levels during the onset of the pharmacological effect of BTX-A.
Conclusion
The present report describes the symptomatic management of two chronic refractory painful post-traumatic trigeminal neuropathies resulting from concurrent iatrogenic injuries to the buccal nerve and inferior alveolar nerve following mandibular third molar extraction. This symptomatic management involved perineural mental nerve BTX-A injections, bringing further support to the small volume of previously gathered scientific evidence.
A significant analgesic effect on both spontaneous and evoked pain was observed, with limited treatment-related functional impairment (minor lip paresis with no visible facial asymmetry or speech impairment). Further studies using the same injection protocol will be required to fully assess its efficacy and safety.
BTX-A injections in the vicinity of the mental nerve are a potentially effective and safe treatment option for the symptomatic management of painful post-traumatic trigeminal neuropathy, even in cases involving multiple trigeminal branches and/or chronic refractory cases. Additional patient cohorts will be required to confirm this preliminary evidence.
Footnotes
Declaration of conflicting interest: The authors declare that there is no conflict of interest.
Funding: This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
ORCID iD: Nathan Moreau https://orcid.org/0000-0003-2725-5292
Ethics statement
No review board approval was requested for the publication of the present case report (according to French bylaw, no ethics board approval is required to publish a single case report as long as patient consent is obtained, as occurred in the present case). All identifying information was removed from the case presentation. The patient provided written consent for treatment and publication of this case report.
Data availability
Data pertaining to the present case report can be obtained, following anonymization, upon reasonable request to the corresponding author.
Supplemental material
Supplemental material for this article is available online.
References
- 1.Headache classification committee of the International Headache Society (IHS). The International Classification of Headache Disorders, 3rd edition. Cephalalgia 2018; 38: 1–211. doi: 10.1177/0333102417738202. [DOI] [PubMed]
- 2.Benoliel R, Kahn J, Eliav E. Peripheral painful traumatic trigeminal neuropathies. Oral Dis 2012; 18: 317–332. doi: 10.1111/j.1601-0825.2011.01883. [DOI] [PubMed] [Google Scholar]
- 3.O’Connor AB. Neuropathic pain: Quality-of-life impact, costs and cost effectiveness of therapy. Pharmacoeconomics 2009; 27: 95–112. doi: 10.2165/00019053-200927020-00002. [DOI] [PubMed] [Google Scholar]
- 4.Renton T, Yilmaz Z. Profiling of patients presenting with posttraumatic neuropathy of the trigeminal nerve. J Orofac Pain 2011; 25: 333–344. [PubMed] [Google Scholar]
- 5.Finnerup NB, Attal N, Haroutounian S, et al. Pharmacotherapy for neuropathic pain in adults: A systematic review and meta-analysis. Lancet Neurol 2015; 14: 162–173. doi: 10.1016/S1474-4422(14)70251-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Benoliel R, Zadik Y, Eliav E, et al. Peripheral painful traumatic trigeminal neuropathy: Clinical features in 91 cases and proposal of novel diagnostic criteria. J Orofac Pain 2012; 26: 49–58. [PubMed] [Google Scholar]
- 7.Wittekindt C, Liu WC, Preuss SF, et al. Botulinum toxin A for neuropathic pain after neck dissection: A dose-finding study. Laryngoscope 2006; 116: 1168–1171. doi: 10.1097/01.mlg.0000217797.05523.75. [DOI] [PubMed] [Google Scholar]
- 8.Ranoux D, Attal N, Morain F, et al. Botulinum toxin type A induces direct analgesic effects in chronic neuropathic pain. Ann Neurol 2008; 64: 274–283. doi: 10.1002/ana.21427. [DOI] [PubMed] [Google Scholar]
- 9.Han ZA, Song DH, Oh HM, et al. Botulinum toxin type A for neuropathic pain in patients with spinal cord injury. Ann Neurol 2016; 79: 569–578. doi: 10.1002/ana.24605. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Tsai CP, Liu CY, Lin KP, et al. Efficacy of botulinum toxin type a in the relief of carpal tunnel syndrome: A preliminary experience. Clin Drug Investig 2006; 26: 511–515. doi: 10.2165/00044011-200626090-00004. [DOI] [PubMed] [Google Scholar]
- 11.Yuan RY, Sheu JJ, Yu JM, et al. Botulinum toxin for diabetic neuropathic pain: A randomized double-blind crossover trial. Neurology 2009; 72: 1473–1478. [DOI] [PubMed] [Google Scholar]
- 12.Xiao L, Mackey S, Hui H, et al. Subcutaneous injection of botulinum toxin a is beneficial in postherpetic neuralgia. Pain Med 2010; 11: 1827–1833. doi: 10.1111/j.1526-4637.2010.01003.x. [DOI] [PubMed] [Google Scholar]
- 13.Apalla Z, Sotiriou E, Lallas A, et al. Botulinum toxin A in postherpetic neuralgia: A parallel, randomized, double-blind, single-dose, placebo-controlled trial. Clin J Pain 2013; 29: 857–864. doi: 10.1097/AJP.0b013e31827a72d2. [DOI] [PubMed] [Google Scholar]
- 14.Layeeque R, Hochberg J, Siegel E, et al. Botulinum toxin infiltration for pain control after mastectomy and expander reconstruction. Ann Surg 2004; 240: 608–613. doi: 10.1097/01.sla.0000141156.56314.1f. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Yoon SH, Merrill RL, Choi JH, et al. Use of botulinum toxin type A injection for neuropathic pain after trigeminal nerve injury. Pain Med 2010; 11: 630–632. doi: 10.1111/j.1526-4637.2010.00801.x. [DOI] [PubMed] [Google Scholar]
- 16.Cuadrado ML, García-Moreno H, Arias JA, et al. Botulinum neurotoxin type-A for the treatment of atypical odontalgia. Pain Med 2016; 17: 1717–1721. doi: 10.1093/pm/pnw040. [DOI] [PubMed] [Google Scholar]
- 17.Moreau N, Dieb W, Descroix V, et al. Topical review: Potential use of botulinum toxin in the management of painful posttraumatic trigeminal neuropathy. J Oral Facial Pain Headache 2017; 31: 7–18. doi: 10.11607/ofph.1753 [DOI] [PubMed] [Google Scholar]
- 18.Antonucci F, Rossi C, Gianfranceschi L, et al. Long-distance retrograde effects of botulinum neurotoxin A. J Neurosci 2008; 28: 3689–3696. doi: 10.1523/JNEUROSCI.0375-08.2008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Matak I, Riederer P, Lackovic Z. Botulinum toxin’s axonal transport from periphery to the spinal cord. Neurochem Int 2012; 61: 236–239. doi: 10.1016/j.neuint.2012.05.001. [DOI] [PubMed] [Google Scholar]
- 20.Gagnier JJ, Kienle G, Altman DG, et al. ; CARE Group. The CARE guidelines: Consensus-based clinical case reporting guideline development. Headache 2013; 53: 1541–1547. [DOI] [PubMed] [Google Scholar]
- 21.Schiffman E, Ohrbach R, Truelove E, et al. Diagnostic criteria for temporomandibular disorders (DC/TMD) for clinical and research applications: Recommendations of the international RDC/TMD consortium network and orofacial special interest group. J Oral Facial Pain Headache 2014; 28: 6–27. doi: 10.11607/jop.1151. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Scholz J, Finnerup NB, Attal N, et al. The IASP classification of chronic pain for ICD-11: Chronic neuropathic pain. Pain 2019; 160: 53–59. doi: 10.1097/j.pain.0000000000001365. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Hendy CW, Smith KG, Robinson PP. Surgical anatomy of the buccal nerve. Br J Oral Maxillofac Surg 1996; 34: 457–460. doi: 10.1016/s0266-4356(96)90108-4. [DOI] [PubMed] [Google Scholar]
- 24.Takezawa K, Ghabriel M, Townsend G. The course and distribution of the buccal nerve: Clinical relevance in dentistry. Aust Dent J 2018; 63: 66–71. doi: 10.1111/adj.12543. [DOI] [PubMed] [Google Scholar]
- 25.Smith BH, Torrance N, Ferguson JA, et al. Towards a definition of refractory neuropathic pain for epidemiological research. An international delphi survey of experts. BMC Neurol 2012; 12: 29. doi: 10.1186/1471-2377-12-29. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Farrar JT, Young JP, Jr, LaMoreaux L, et al. Clinical importance of changes in chronic pain intensity measured on an 11-point numerical pain rating scale. Pain 2001; 4: 149–158. doi: 10.1016/S0304-3959(01)00349-9. [DOI] [PubMed] [Google Scholar]
- 27.Kim DW, Lee SK, Ahnn J. Botulinum toxin as a pain killer: Players and actions in antinociception. Toxins (Basel) 2015; 7: 2435–2453. doi: 10.3390/toxins7072435 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Pellett S, Yaksh TL, Ramachandran R. Current status and future directions of botulinum neurotoxins for targeting pain processing. Toxins (Basel) 2015; 7: 4519–4563. doi: 10.3390/toxins7114519. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Restani L, Antonucci F, Gianfranceschi L, et al. Evidence for anterograde transport and transcytosis of botulinum neurotoxin A (BoNT/A). J Neurosci 2011; 31: 15650–15659. doi: 10.1523/JNEUROSCI.2618-11.2011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Koizumi H, Goto S, Okita S, et al. Spinal central effects of peripherally applied botulinum neurotoxin A in comparison between its subtypes A1 and A2. Front Neurol 2014; 5: 98. doi: 10.3389/fneur.2014.00098. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Won SY, Yang HM, Woo HS, et al. Neuroanastomosis and the innervation territory of the mental nerve. Clin Anat 2014; 27: 598–602. doi: 10.1002/ca.22310. [DOI] [PubMed] [Google Scholar]
- 32.Attal N. Pharmacological treatments of neuropathic pain: The latest recommendations. Rev Neurol (Paris) 2019; 75: 46–50. doi: 10.1016/j.neurol.2018.08.005. [DOI] [PubMed] [Google Scholar]
- 33.Herrero Babiloni A, Kapos FP, Nixdorf DR. Intraoral administration of botulinum toxin for trigeminal neuropathic pain. Oral Surg Oral Med Oral Pathol Oral Radiol 2016; 121: e148–e153. [DOI] [PubMed] [Google Scholar]
- 34.Moreno-Hay I, Mishra P, Okeson JP. Intraoral administration of botulinum toxin for continuous dentoalveolar neuropathic pain: A case series. J Oral Facial Pain Headache 2019; 33: 160–164. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
Data pertaining to the present case report can be obtained, following anonymization, upon reasonable request to the corresponding author.