Abstract
Aims
To explore the synovial expression of mucin 1 (MUC1) and its role in rheumatoid arthritis (RA), as well as the possible downstream mechanisms.
Methods
Patients with qualified synovium samples were recruited from a RA cohort. Synovium from patients diagnosed as non-inflammatory orthopaedic arthropathies was obtained as control. The expression and localization of MUC1 in synovium and fibroblast-like synoviocytes were assessed by immunohistochemistry and immunofluorescence. Small interfering RNA and MUC1 inhibitor GO-203 were adopted for inhibition of MUC1. Lysophosphatidic acid (LPA) was used as an activator of Rho-associated pathway. Expression of inflammatory cytokines, cell migration, and invasion were evaluated using quantitative real-time polymerase chain reaction (PCR) and Transwell chamber assay.
Results
A total of 63 RA patients and ten controls were included. Expression of MUC1 was observed in both the synovial lining and sublining layer. The percentage of MUC1+ cells in the lining layer of synovium was significantly higher in RA than that in control, and positively correlated to joint destruction scores of RA. Meanwhile, MUC1+ cells in the sublining layer were positively correlated to the Krenn subscore of inflammatory infiltration. Knockdown of MUC1, rather than GO-203 treatment, ameliorated the expression of proinflammatory cytokines, cell migration, and invasion of rheumatoid synoviocytes. Knockdown of MUC1 decreased expression of RhoA, Cdc42, and Rac1. Treatment with LPA compromised the inhibition of migration and invasion, but not inflammation, of synoviocytes by MUC1 knockdown.
Conclusion
Upregulated MUC1 promotes the aggression of rheumatoid synoviocytes via Rho guanosine triphosphatases (GTPases), thereby facilitating synovitis and joint destruction during the pathological process of RA.
Cite this article: Bone Joint Res 2022;11(9):639–651.
Keywords: Rheumatoid arthritis, Fibroblast-like synoviocytes, Mucin 1, Joint destruction, Inflammation, inflammation, synovitis, fibroblast-like synoviocytes (FLSs), joint damage, joint destruction, RNAs, synoviocytes, Cdc42, cytokines
Article focus
To explore the synovial expression of mucin 1 (MUC1) and the correlation between dysregulated MUC1 and rheumatoid arthritis (RA) clinical disease parameters.
To reveal the roles and underlying mechanisms of MUC1 in inflammation, migration, and invasion of RA fibroblast-like synoviocytes (FLSs).
Key messages
Upregulated expression of MUC1 in the synovium is correlated with synovitis score as well as joint destruction score in RA.
MUC1 promotes inflammation and aggression of RA FLSs partly dependent on Rho guanosine triphosphatases (GTPases).
Strengths and limitations
Correlations between the dysregulated synovial MUC1 expression and synovitis and joint destruction in RA were revealed for the first time.
This study provides a novel mechanism for RA synovial pathology and a potential target for FLS-based therapy of RA.
Further investigations regarding the detailed molecular mechanisms are required.
Introduction
Rheumatoid arthritis (RA) is a widespread, relapsing, disabling autoimmune disease. 1 To date, a substantial proportion of RA patients fail to attain clinical remission and experience joint damage progression, even with the springing up of various therapies of monoclonal antibodies. 2 It is imperative to identify novel biomarkers and treatment targets for synovitis and structural damage in RA patients.
In recent years, mucins (MUCs) have been drawing increasing attention for their involvement in tumour biology and immunology. MUC is a family of large glycoproteins characterized by a basic structure with a highly polymorphic tandem repeat in the central region and heavy O-glycosylation modification after translation. 3 MUC1 is one of the most well-studied members of the MUC family. The mature form of MUC1 is a heterodimer composed of an extracellular N-terminal (MUC1-N) subunit and a transmembrane C-terminal (MUC1-C) subunit. MUC1 has long been recognized as an important player in respiratory diseases, digestive diseases, and cancers. 4,5 Aberrantly glycosylated MUC1 (also known as CA15-3) is upregulated in a plethora of tumours and, more importantly, is correlated with poor prognosis. Therefore, MUC1 has been widely used as a diagnostic biomarker and monitoring index for metastatic tumours, and hopefully a promising target for cancer treatment. 6,7 Several MUC1-based clinical trials have been initiated in recent years. 8-10
Fibroblast-like synoviocytes (FLSs), one of the most common groups of cells in the hypertrophic and aggressive RA synovium, actively participate in the initiation and progression of RA pathology by mediating synovial inflammation and joint destruction. 11,12 Targeting of RA FLSs has been emerging as a promising approach of RA therapy. 13 Expression of MUC3 and MUC5AC is upregulated in RA synovium compared to that of normal humans. 14 However, the expression pattern of MUC1 in the synovium, as well as the roles of MUC1 in RA pathology, remain unelucidated. In this study, we evaluated the synovial expression of MUC1 and the involvement of MUC1 in synovitis and joint destruction caused by RA FLSs.
Methods
Patient enrolment and synovium collection
RA patients were retrospectively recruited from a RA cohort from August 2010 to March 2017 at the Department of Rheumatology, as previously described by Chen et al. 15 All the RA patients were diagnosed using the 2010 American College of Rheumatology (ACR)/European League Against Rheumatism (EULAR) criteria for RA classification. 16 RA patients overlapping with serious infections, malignancies, or other autoimmune diseases were excluded. Parker-Pearson needle biopsy was conducted to obtain RA synovium. Control synovium samples were obtained from non-inflammatory orthopaedic arthropathy (Orth.A) patients who received orthopaedic surgery at the Department of Orthopedics, Sun Yat-Sen Memorial Hospital, Guangzhou, China.
Patient characteristics
A total of 63 RA patients with sufficient qualified synovium samples were enrolled in this study. Baseline demographic and clinical characteristics of those included RA patients are shown in Table I. Overall, 49 patients (77.8%) in the RA group were female. The median age of the RA patients group was 50 years (interquartile range (IQR) 44 to 60). The median disease duration was 24 months (IQR 8 to 84). As controls, ten patients who received orthopaedic surgery for meniscus injury (n = 5), plica syndrome of the knee joint (n = 1), cruciate ligament injury (n = 2), traumatic arthritis (n = 1), and knee articular cartilage damage (n = 1) were recruited. The median age of the included control patients was 33.5 years (IQR 24.25 to 45) and three (30%) of them were female.
Table I.
Demographic and clinical characteristics of rheumatoid arthritis patients.
| Characteristic | Data (n = 63) |
|---|---|
| Median age, yrs (IQR) | 50 (44 to 60) |
| Female, n (%) | 49 (77.8) |
| Median disease duration, mths (IQR) | 24 (8 to 84) |
| Core disease activity indicator | |
| Median ESR, mm/h (IQR) | 74 (51 to 105) |
| Median CRP, mg/l (IQR) | 38.6 (17.1 to 63.6) |
| Median RF, IU/ml (IQR) | 280 (35 to 577) |
| Median ACPA, IU/ml (IQR) | 176 (25 to 300) |
| RF positive, n (%) | 50 (79.4) |
| ACPA positive, n (%) | 50 (79.4) |
| Median DAS28-CRP (IQR) | 4.77 (4.03 to 5.41) |
| Median SDAI (IQR) | 33.74 (23.27 to 43.5) |
| Median CDAI (IQR) | 27 (20 to 40) |
| Radiological assessment | |
| Median mTSS (IQR) | 11 (3 to 34.5) |
| Median JSN subscore (IQR) | 5 (0 to 17) |
| Median JE subscore (IQR) | 6 (1 to 17) |
| RJD, n (%) | 33 (52.4) |
| Histological assessment | |
| Median Krenn synovitis score (IQR) | 5 (4 to 6) |
| Median hyperplasia of lining layer (IQR) | 2 (1 to 2) |
| Median inflammatory infiltration (IQR) | 1 (1 to 2) |
| Median activation of synovial stroma (IQR) | 2 (1 to 2) |
| Previous medication | |
| Treatment-naive*, n (%) | 23 (36.5) |
| Glucocorticoid, n (%) | 36 (57.1) |
| Methotrexate, n (%) | 13 (20.6) |
| Leflunomide, n (%) | 12 (19) |
| Sulfasalazine, n (%) | 7 (11.1) |
| Hydroxychloroquine, n (%) | 6 (9.5) |
| Biological agents, n (%) | 8 (12.7) |
Treatment-naive, without previous glucocorticoids or disease-modifying antirheumatic drug (DMARD) therapy for six months before enrolment.
ACPA, anti-cyclic citrullinated peptide antibody; CDAI, clinical disease activity index; DAS28-CRP, disease activity score in 28 joints with four variables including CRP; IQR, interquartile range; JE, joint erosion; JSN, joint space narrowing; mTSS, modified total Sharp score; RF, rheumatoid factor; RJD, radiological joint damage; SDAI, simplified disease activity index.
Clinical, radiological, and histological assessments
Demographics, disease activity, and radiological and histological assessments of RA were collected at recruitment as previously described by Chen et al. 15 Radiological assessment includes joint erosion (JE) subscore and joint space narrowing (JSN) subscore, constituting modified total Sharp score (mTSS) using Sharp/van der Heijde modified method. 17 A score of mTSS > 10 units was defined as radiological joint damage (RJD). Histological changes of synovium were graded on a scale of 0 to 9 according to the Krenn synovitis scoring system, composed of hyperplasia of lining layer subscore (0 to 3), inflammatory infiltration subscore (0 to 3), and activation of synovial stroma subscore (0 to 3). 18
Immunohistochemistry
Synovial tissue samples were fixed in 4% paraformaldehyde and embedded in paraffin. Immunohistochemistry (IHC) analysis of synovium was carried out as previously described by Chen et al. 15 Antigen retrieval was performed by boiling slides in 1 mM ethylenediaminetetraacetic acid (EDTA) buffer (PH 8.0) in a pressure cooker for 2.5 minutes. Synovium sections were incubated with rabbit anti-MUC1 antibody (1:100, ab45167, Abcam, USA) at 4°C overnight, followed by staining with a polymer detection system for IHC (PV-6000, ZSGB-Bio, China). A rabbit control immunoglobulin G (IgG) was used as a negative control. DAB substrate (ZSGB-Bio) was used for colour developing. MUC1+ cells in the lining and sublining layer in five different fields were counted under a 400× optical microscope (Leica DM2500, Leica, Germany).
Isolation and culture of FLSs
RA FLSs were isolated from fresh synovium and cultured adherently as previously described by Zou et al. 19 Freshly obtained synovial tissues were cut into small pieces and digested with 1 mg/ml collagenase I (Sigma-Aldrich, USA) for three hours at 37°C, and the isolated synoviocytes were cultured in Dulbecco’s modified Eagle’s medium (DMEM)/F12 with 10% fetal bovine serum (FBS) at 5% CO2 and 37°C. RA FLSs from passages 3 to 7 were used.
Small interfering RNA-mediated knockdown of MUC1
The small interfering RNAs (siRNAs) for MUC1 were synthesized by RiboBio (China). Three siRNAs targeting MUC1 were designed and the sequences are as follows: siMUC1-1 (CGGGATACCTACCATCCTA); siMUC1-2 (GGCTACCCAGAGAAGTTCA); and siMUC1-3 (GCCACTTTCTTGCTCAGTC). Negative control siRNA (NC) was purchased from RiboBio (siN0000001-1-5). Cells were seeded in six-well plates at 60% to 70% confluence and transiently transfected with siRNA (50 nM) using Lipofectamine RNAiMAX Reagent (Thermo Fisher Scientific, USA) as previously described by Zou et al. 19 Whole cell lysates were extracted to assess knockdown efficiency of MUC1 in RA FLSs by quantitative real-time polymerase chain reaction (qRT-PCR) and western blot (WB), and the siRNA with the best silencing efficiency was used for further experiments.
Immunofluorescence
For immunofluorescence (IF) staining of synovium, slides were incubated with anti-MUC1 antibody (1:50), anti-CD55 antibody (1:50, ab133684, Abcam), anti-CD90/Thy1 antibody (1:100, ab181469, Abcam), and anti-fibroblast activation protein (FAP) α antibody (1:100, ab207178, Abcam). Corresponding secondary antibodies conjugated with Alexa Flour 594 or Alexa Flour 488 (1:1000, Thermo Fisher Scientific) were employed for staining.
For IF staining of in vitro cultured FLSs, cells grown on glass coverslips were stained with anti-MUC1 antibody (1:50). For skeleton staining, RA FLSs were stained with Alexa Fluor 488 Phalloidin (1:1,000, Thermo Fisher Scientific).
Quantitative real-time polymerase chain reaction
Total RNA was extracted with TRIzol reagent (Thermo Fisher Scientific) according to the manufacturer’s protocol. The isolated RNA was reversely transcripted into complementary DNA (cDNA) with a PrimeScript RT kit (Takara, Japan). qRT-PCR was carried out as previously described by Zou et al. 19 Glyceraldehyde-3-phosphate dehydrogenase (GAPDH) was used as the internal reference gene. Primer sequences are shown in Supplementary Table i.
Western blot
WB was performed as previously described by Zou et al. 19 In the present research, the following primary antibodies were adopted: anti-MUC1 (1:1,000), anti-RhoA (1:1,000, 2117, Cell Signaling Technology, USA), anti-CCD42 (1:1,000, ab187643, Abcam), anti-Rac1 (1:1,000, ab155938, Abcam), and anti-GAPDH (1:1,000, 5174, Cell Signaling Technology).
Apoptosis analysis
Cells resuspended in 1× binding buffer were stained with 5 μl Annexin V-FITC (BD Biosciences, USA) for 15 minutes at room temperature and protected from light. Then, 5 μl propidium iodide (PI) (BD Biosciences) was added and then analyzed within one hour using the BD FACSCalibur flow cytometer (BD Biosciences).
Cell Counting Kit-8 assay
RA FLSs were grown in 100 μl of complete culture medium in 96-well plates. After adding 10 µl Cell Counting Kit-8 (CCK-8) (Dojindo, Japan) per well, the plates were incubated at 37°C for two hours and measured on a microplate reader to determine the absorbance at 450 nm.
Cell migration and invasion assay
RA FLSs were serum-starved overnight before the measurement of cell migration and invasion. For the detection of cell migration rate, RA FLSs (2.5 × 104/well) were seeded onto 6.5 mm diameter Transwell chambers (Corning, USA) with 8 μm pores and allowed to migrate toward DMEM medium supplemented with 10% FBS in the lower well for six hours. For invasion assay, a total of 4 × 104 cells were placed onto Transwell chambers precoated with Matrigel (BD Biosciences) and allowed to invade into the matrix for 24 hours. Cells moved through the membrane of the Transwell chamber were stained using crystal violet solution and quantified by counting five random 200× fields under a microscope.
Statistical analysis
SPSS (version 20.0, IBM, USA) and GraphPad Prism 8 (GraphPad Software, USA) were adopted in this research. Independent-samples t-test and Mann-Whitney U test were adopted for unpaired data. One-way analysis of variance (ANOVA) followed by Tukey’s test was used for multiple comparisons. Correlations between MUC1 expression and RA disease indicators were determined with Spearman’s rank order correlation. A two-tailed p-value < 0.05 was considered statistically significant.
Results
Synovial expression of MUC1 and the clinical significance of MUC1 in RA
Expression of MUC1 was observed in synoviocytes located in both the lining and sublining layer of the synovium (Figure 1a). The percentage of MUC1 positive (MUC1+) cells in the RA lining layer was significantly increased compared to that in the control (median 61.91% (interquartile range (IQR) 47.69% to 75.10%) vs median 37.09% (IQR 5.11% to 63.99%); p = 0.017, Mann-Whitney U test). However, the difference of MUC1 expression in the sublining layer between RA and the control group was not significant (median 85.06 (IQR 80.34 to 88.06) vs median 82.42 (IQR 17.30 to 84.03); Figure 1b). Furthermore, synovium from RA patients with RJD exhibited elevated MUC1 expression in the lining layer (median 70.24 (IQR 54.17 to 77.10) vs median 58.56 (IQR 29.65 to 68.81); p = 0.016, Mann-Whitney U test; Figure 1c).
Fig. 1.
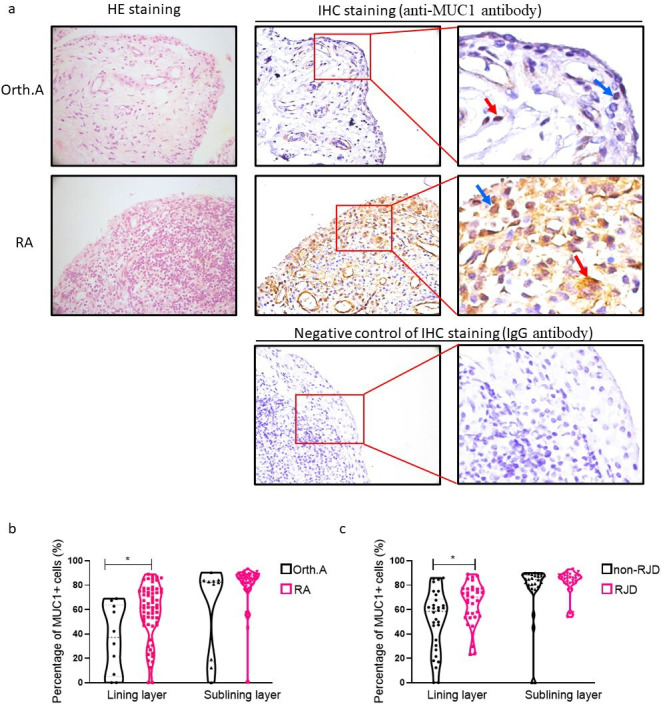
Expression of mucin 1 (MUC1) in synovium. a) Detection of MUC1 expression by immunohistochemistry (IHC) staining in synovium obtained from patients with rheumatoid arthritis (RA, n = 63) and non-inflammatory orthopaedic arthropathies (Orth.A, n = 10 ). The blue arrows and red arrows indicate MUC1-positive cells in the lining and sublining layer, respectively. Original magnification: 400×. Representative haematoxylin and eosin (H&E) staining of synovium (left column). b) and c) Percentages of MUC1-positive cells in lining and sublining layer of synovium from 63 RA patients and ten Orth.A patients were manually counted under five random 400× fields. *p < 0.05, Mann-Whitney U test. IgG, immunoglobulin G; RJD, radiological joint damage.
Positive correlations between the percentage of lining MUC1+ cells with mTSS, JSN subscore, and JE subscore were detected (r = 0.282 to 0.346; p = 0.005, p = 0.025, and p = 0.006, respectively, all Spearman’s rank order correlation; Figure 2a). Meanwhile, sublining MUC1 expression was found to be positively correlated to inflammatory infiltration subscore of the Krenn synovitis score (r = 0.267; p = 0.034, Spearman’s rank order correlation; Figure 2b). These findings imply a possible role of MUC1 in joint destruction and synovial inflammation of RA.
Fig. 2.
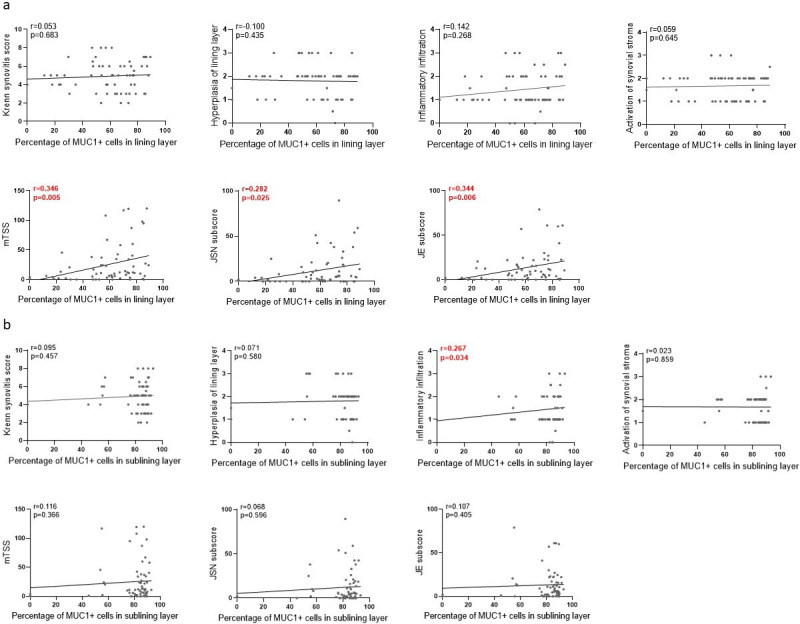
Spearman’s rank order correlation analysis between rheumatoid arthritis (RA) clinical parameters and percentages of mucin 1 (MUC1) positive cells in lining layer (a) and sublining layer (b) of RA synovium. Histological changes were assessed according to the Krenn synovitis scoring system. JE, joint erosion; JSN, joint space narrowing; mTSS, modified total Sharp score assessed by Sharp/van der Heijde modified method.
Expression of MUC1 in RA FLSs
Given that FLSs are the main resident cells in the RA synovium and are responsible for the aggressive and inflammatory phenotype of the rheumatoid pannus, the expression of MUC1 in RA FLSs was determined. MUC1 was observed to be colocalized with CD55, CD90, and FAPα in the RA synovium, implying a broad expression of MUC1 by distinct fibroblast subsets in the synovium (Figure 3a). Furthermore, expression of MUC1 was found in the nucleus, cytoplasm, as well as the plasma membrane of in vitro cultured RA FLSs (Figure 3b).
Fig. 3.
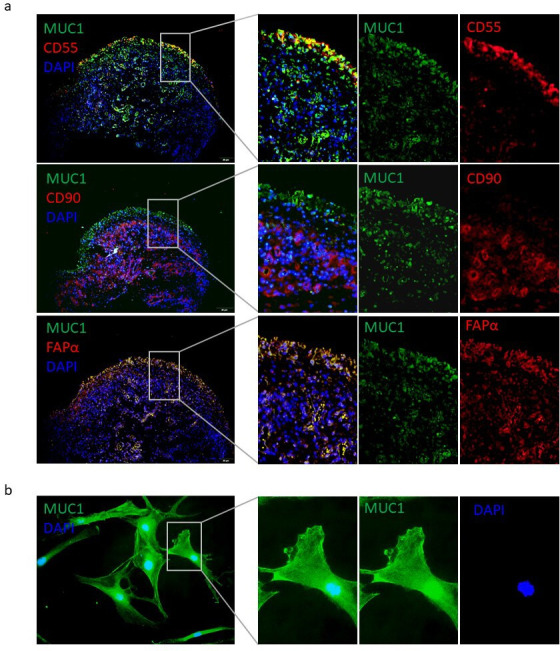
Localization of mucin 1 (MUC1) in synovium and rheumatoid arthritis (RA) fibroblast-like synoviocytes (FLSs). a) Distribution of MUC1 expression in RA synovium was detected by double immunofluorescence (IF) staining. Original magnification: 100×. b) Subcellular localization of MUC1 in RA FLSs was evaluated by indirect IF staining. Original magnification: 400×. DAPI, 4′,6-diamidino-2-phenylindole; FAPα, fibroblast activation protein α.
Effect of MUC1 on cell apoptosis and growth of RA FLSs
siRNAs targeting MUC1 were adopted for the downregulation of MUC1 and the silencing efficiency was determined (Figures 4a and 4b). Meanwhile, GO-203, a MUC1 inhibitor that blocks homodimerization and function of MUC1-C, was used for specific targeting of MUC1-C.
Fig. 4.
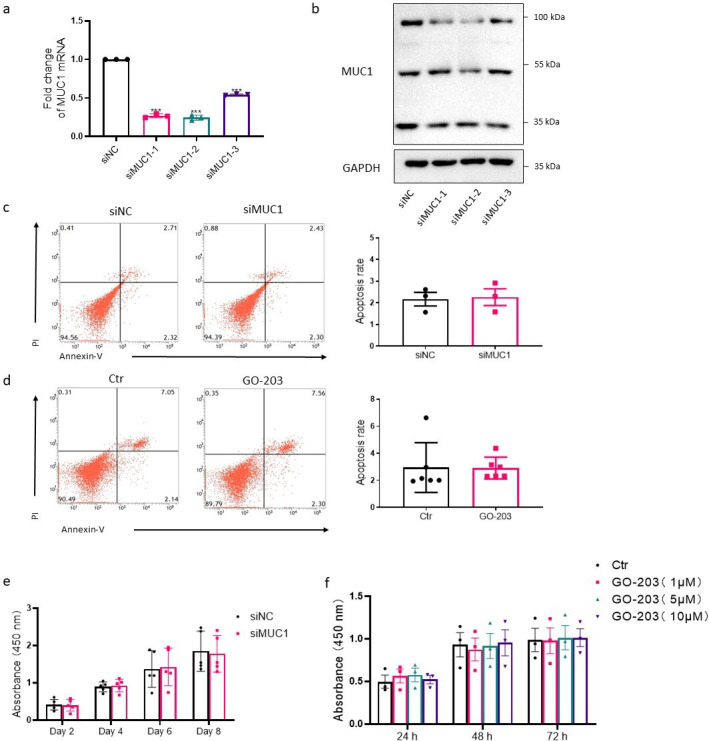
Effects of mucin 1 (MUC1) on apoptosis and proliferation in rheumatoid arthritis (RA) fibroblast-like synoviocytes (FLSs). Silencing efficiency of MUC1-targeting small interfering RNA (siRNA) was measured by a) quantitative real-time polymerase chain reaction (qRT-PCR) (n = 3) and b) western blot (WB). Three siRNAs targeting MUC1 were designed and named as siMUC1-1, siMUC1-2, and siMUC1-3, respectively. Effects of c) MUC1 silencing (n = 3) and d) MUC1 inhibitor GO-203 treatment (5 μM, 48 hours) (n = 6) on cell apoptosis of RA FLSs were measured by flow cytometry following staining with FITC-conjugated Annexin V/propidium iodide (PI). Early apoptotic cells defined as PI negative and Annexin V positive cells were quantified. e) After transfection with MUC1-targeting siRNA for the indicated time, cell viability was measured by Cell Counting Kit-8 (CCK-8) assay; n = 5 at different timepoints. f) RA FLSs were treated with different concentrations of GO-203 for the indicated time and cell viability was measured; n = 3 at different timepoints. Ctr, control; GAPDH, glyceraldehyde-3-phosphate dehydrogenase; mRNA, messenger RNA; NC, negative control.
Restrained cell apoptosis has been found in primarily cultured RA FLSs. Our results indicate that targeting MUC1 using siRNA (Figure 4c) or MUC1-C with GO-203 (Figure 4d) did not influence cell apoptosis of RA FLSs. Furthermore, the cell growth was not affected by silencing of MUC1 (Figure 4e) or GO-203 treatment (Figure 4f) in RA FLSs as measured by the CCK-8 test.
Inhibition of MUC1 significantly diminished inflammation, migration, and invasion of RA FLSs
RA FLSs are active contributors to the inflammatory microenvironment of RA synovium by producing a variety of cytokines and chemokines. Downregulation of MUC1 remarkably ameliorated the expressions of interleukin (IL)-6, IL-8, and C-C motif chemokine ligand (CCL)-2 by RA FLSs (Figure 5a). However, treatment with GO-203 did not alter the expressions of proinflammatory cytokines and chemokines in RA FLSs (Figure 5b).
Fig. 5.
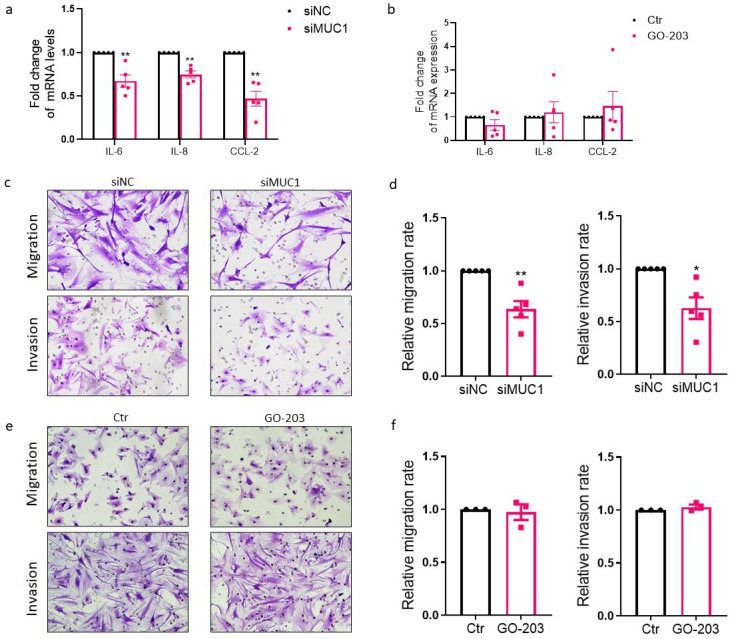
Regulation of expression of proinflammatory cytokines, migration, and invasion of rheumatoid arthritis (RA) fibroblast-like synoviocytes (FLSs) by mucin 1 (MUC1). a) RA FLSs were transfected by MUC1 small interfering RNA (siRNA), and the messenger RNA (mRNA) expression levels of proinflammatory cytokines in RA FLSs were evaluated by quantitative real-time polymerase chain reaction (qRT-PCR) (n = 5). b) RA FLSs were treated with GO-203 (5 μM) for 48 hours and expression levels of proinflammatory cytokines were measured (n = 5). Migration of RA FLSs was measured with a Transwell chamber (Corning, USA), and cells seeded onto the upper chamber were allowed to migrate towards medium with 10% fetal bovine serum (FBS) in the lower chamber for six hours. For invasion assay, Transwell chambers coated with Matrigel were used and cells were allowed to invade to the lower chamber filled with 15% FBS for 24 hours. c) to f) Inhibitory effects of MUC1 silencing by siRNA on migration and invasion (c & d; n = 5) in RA FLSs. e) to f) RA FLSs were pretreated with GO-203 (5 μM) for 48 hours and subjected to migration and invasion assay (n = 3). Data are presented as the mean and standard error of the mean (SEM). *p < 0.05, **p < 0.01, independent-samples t-test. CCL-2, C-C motif chemokine ligand-2; Ctr, control; IL, interleukin; NC, negative control; siNC, siRNA of negative control.
RA FLSs represent tumour-like behaviours such as invading adjacent cartilage and bone and migrating to distant joints. 20 Silencing of MUC1 with siRNA remarkably attenuated migration and invasion (Figures 5c and 5d) of RA FLSs. Against our expectation, treatment with GO-203 did not impair migration and invasion of RA FLSs (Figures 5e and 5f).
Regulation of Rho GTPases by MUC1 in RA FLSs
Cell motility is exquisitely regulated by dynamic assembly and reorganization of cytoskeleton. The formation of lamellipodia and filopodia at cell edges was decreased in MUC1 knockdown RA FLSs (Figure 6a). Since Rho guanosine triphosphatases (GTPases) are critical in controlling the rearrangement of cytoskeleton and other processes such as gene expression, the influences of MUC1 knockdown on RhoA, Cdc42, and Rac1 were determined. As shown in Figure 6b, no statistically significant changes of RhoA, Cdc42, and Rac1 messenger RNA (mRNA) expressions were found in MUC1 knockdown RA FLSs. Nevertheless, the protein expression levels of RhoA, Cdc42, and Rac1 were reduced by the knockdown of MUC1 (Figures 6c and 6d).
Fig. 6.
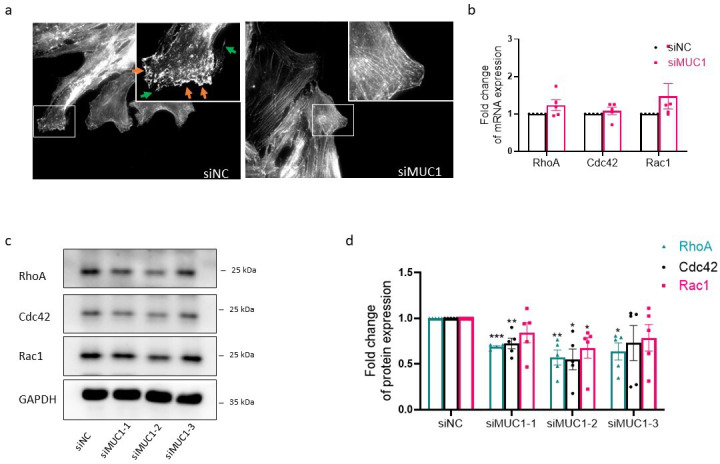
Modulation of Rho guanosine triphosphatases (GTPases) by mucin 1 (MUC1) in rheumatoid arthritis (RA) fibroblast-like synoviocytes (FLSs). a) To determine the influence of MUC1 silencing on assembly and reorganization of cytoskeleton, RA FLSs grown on glass coverslips were stained with phalloidin four hours after wounding. The green arrows indicate formation of filopodia and the orange arrows indicate lamellipodia. Original magnification 400×. b) RA FLSs were transfected with MUC1 small interfering RNA (siRNA), and quantitative real-time polymerase chain reaction (qRT-PCR) was conducted to measure the messenger RNA (mRNA) expression levels of Rho GTPases (n = 5). c) and d) Inhibitory effects of MUC1 silencing on protein expression levels of Rho GTPases in RA FLSs measured by western blot (WB) (n = 5). Data are presented as the mean and standard error of the mean (SEM). *p < 0.05, **p < 0.01, ***p < 0.001; one-way analysis of variance (ANOVA) followed by Tukey’s test was used. GAPDH, glyceraldehyde-3-phosphate dehydrogenase; NC, negative control.
Involvement of Rho GTPases in MUC1 mediated regulation of migration and invasion of RA FLSs
To determine whether Rho GTPases are required for MUC1-induced inflammation, migration, and invasion of RA FLSs, we treated RA FLSs with lysophosphatidic acid (LPA), an activator of Rho-associated pathway. The results turned out that the treatment with LPA did not change the inhibitory effects of MUC1 knockdown on expression of IL-6, IL-8, and CCL-2 (Figure 7a). Nevertheless, the inhibition of migration and invasion of RA FLSs induced by MUC1 knockdown were partly reversed (Figure 7b), implying an involvement of Rho GTPases in MUC1-mediated signalling pathway.
Fig. 7.
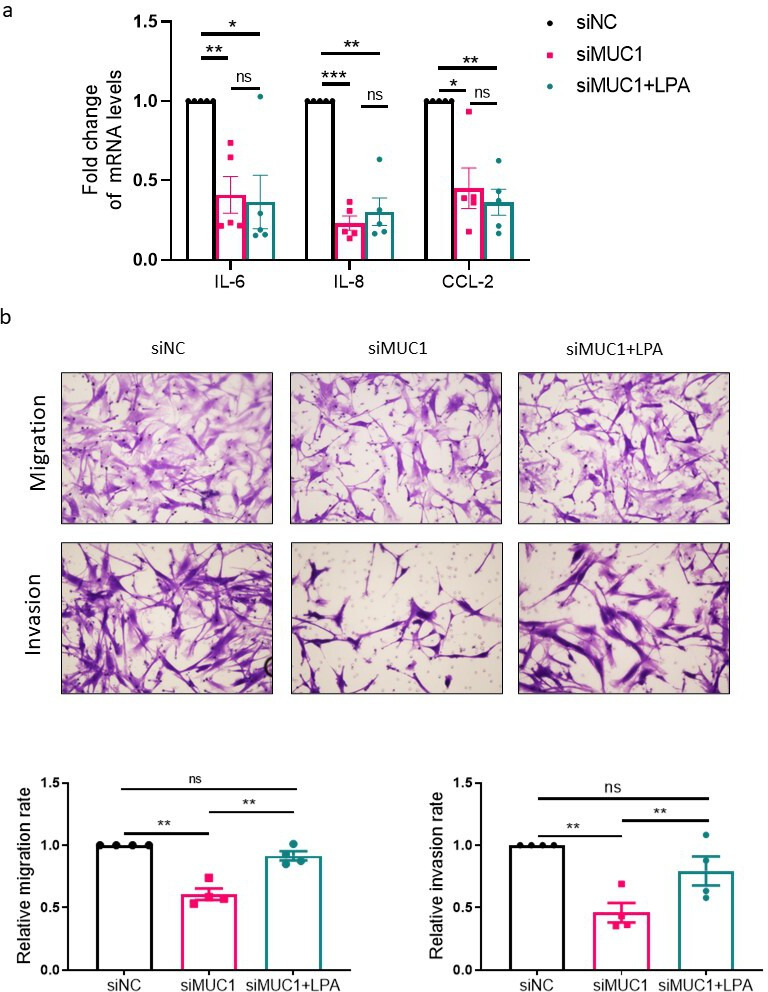
Involvement of Rho guanosine triphosphatases (GTPases) in mucin 1 (MUC1) mediated regulation of inflammation, migration, and invasion in rheumatoid arthritis (RA) fibroblast-like synoviocytes (FLSs). RA FLSs transfected with the indicated small interfering RNAs (siRNAs) were serum-starved for 24 hours and then stimulated with or without lysophosphatidic acid (LPA, 10 uM) for 24 hours. a) Quantitative real-time polymerase chain reaction (qRT-PCR) was conducted to measure the messenger RNA (mRNA) expression levels of proinflammatory cytokines (n = 5). b) Transwell chamber assay was conducted to measure migration and invasion of RA FLSs (n = 4). Data are presented as the mean and standard error of the mean (SEM). *p < 0.05, **p < 0.01, ***p < 0.001; one-way analysis of variance (ANOVA) followed by Tukey’s test was used. CCL-2, C-C motif chemokine ligand-2; IL, interleukin; NC, negative control; ns, not significant.
Discussion
This study reports the correlations between the dysregulated synovial MUC1 expression and synovitis and joint destruction in RA for the first time. Additionally, this study provides a novel mechanism based on MUC1-mediated regulation of Rho GTPases for aggression of RA FLSs, and a potential treatment target for RA.
Indeed, dysregulated MUC1 expression has been observed in various kinds of cancers and correlates to disease stages, response to therapy, and survival. 21 Apart from malignant diseases, MUC1 is also involved in some autoimmune diseases. For instance, an enhanced accumulation of MUC1 is found in the labial salivary gland from Sjögren’s syndrome patients. 22 Nevertheless, few efforts have been devoted to the expression and role of MUC1 in RA synovial pathology. In 2008, Volin et al 14 showed an enhanced accumulation of MUC3 in synovial lining cells and macrophages in RA and osteoarthritis (OA) synovium compared with that in normal synovium. MUC5AC was lowly expressed in RA and OA synovium and barely expressed in normal control synovium. However, in this study, Volin et al 14 detected negative expression of MUC1 in RA synovium, which might be explained by the insufficiency of antibody affinity and specificity. Another study in 2011, by Hamaguchi et al, 23 revealed the existence of MUC1 in synovial fluid, lining and sublining layer of RA synovium, and cultured RA FLSs. In human peripheral blood mononuclear cells (PBMCs), treatment with MUCs isolated from RA synovial fluid stimulated interleukin (IL)-6 production. 23 Our data further demonstrated the increased expression of MUC1 in RA synovium and the colocalizations of MUC1 with markers of RA FLSs including CD55, CD90, and FAPα, indicating a broad expression of MUC1 in different subsets of RA FLSs. Moreover, our study showed that synovial expression of MUC1 was correlated to joint destruction and synovitis, further supporting the pathological roles of MUC1 in RA synovial abnormality.
As a multifaceted oncoprotein, MUC1 exerts crucial regulatory influences on cell proliferation, apoptosis, metastasis, angiogenesis, and chronic inflammation. For instance, ectopic expression of MUC1 promotes the invasiveness of breast cancer cells. 24 MUC1 enhances activities of matrix metalloproteinase (MMP)-2 and MMP-9, thereby facilitating invasiveness of cancer cells. 25 In our study, we demonstrated silencing of MUC1-mitigated migration, invasion, and the production of proinflammatory mediators in RA FLSs. Considering the crucial pathological roles of RA FLSs in RA, treatments targeting RA FLSs have been emerging as a promising direction for future RA therapy. 26 Our results provide a novel potential target for RA precise therapy. Interestingly, the inhibition of MUC1-C homodimerization by GO-203 did not affect the aggressive behaviours and inflammation of RA FLSs, implying a mechanism in RA FLSs that is independent of MUC1-C subunit homodimerization. In support of these results, a study revealed that treatment with GO-203 downregulated expression of MYC1, a well-established hallmark of cancer, in cancer cells with mutant KRAS gene, but not in wild-type cancer cells, suggesting the limitation of GO-203 in blocking MUC1-mediated signal pathway. 27 Further research should be undertaken to develop new MUC1 inhibitors for a novel potential treatment of RA.
Several reports have emphasized the importance of Rho GTPases in regulating the abnormality of RA FLSs. RhoA actively participates in the regulation of invasion and adhesion of RA FLSs. 28 Rac1 promotes the invasive behaviours and proliferation of RA FLSs, possibly through c-Jun N-terminal kinase (JNK) and p21-activated kinase 1 (PAK1) activation. 29-31 Cdc42 mediates serum amyloid A (A-SAA) induced cell growth and migration of RA FLSs. 32 Apart from their roles in cell motility and invasion, Rho GTPases also act as regulators of inflammation. RhoA and its key effector Rho kinase (ROK) are engaged in the regulation of the nuclear factor kappa B (NF-κB) pathway, and consequently control proinflammatory cytokine production in RA FLSs. 33 The present research showed that inhibition of MUC1 significantly decreased protein expression of RhoA, Cdc42, and Rac1. Interestingly, RhoA, Cdc42, and Rac1 mRNA levels were not affected by MUC1 inhibition, suggesting a possible post-transcriptional regulatory mechanism of Rho GTPases by MUC1. Post-transcriptional regulatory mechanisms have been proved in the regulation of Rho GTPases. 34-36 Another study showed that long non-coding RNA (lncRNA) lowly expressed in rheumatoid fibroblast-like synoviocytes (LERFS) inhibits activation of RA FLSs by negative regulation of RhoA, Cdc42, and Rac1 through a post-transcriptional mechanism. 19 MUC1 facilitates cell-cell crosstalk, cell-matrix interaction, and intracellular signalling via distinct mechanisms, among which is post-transcriptional regulation such as modulation of protein degradation. 37 In line with our findings, MUC1 has been previously suggested to be involved in Rac1/Cdc42 mediated cell motility in breast cancer cells and 293T cells. 38 The present research provides further evidence of MUC1 mediated regulation of Rho GTPases and thus results in a novel explanation for the aggressive phenotype of RA FLSs. However, in this research, we found that the regulation of inflammation of RA FLSs by MUC1 was not dependent on RhoA, Cdc42, or Rac1. Still, we did not rule out the possibility that downstream molecules other than Rho GTPases might participate in the MUC1-related pathways. Another limitation of this research is the insufficiency in revealing the precise mechanism of MUC1-mediated modulation of RhoA, Cdc42, and Rac1 expressions. Further researches about the detailed molecular mechanisms are required for a deeper understanding of the roles and detailed mechanisms of MUC1 in regulating inflammation and aggression of RA FLSs.
In summary, our study indicates that upregulated MUC1 aggregates the inflammatory and aggressive phenotype of RA FLSs, partly via regulation of the Rho GTPases pathway. Our results revealed a novel role of MUC1 in synovitis and joint destruction caused by RA FLSs and a potential treatment strategy by targeting MUC1.
Author contributions
Y. Zou: Investigation, Methodology, Formal analysis, Writing – original draft, Writing – review & editing.
X. Zhang: Investigation, Formal analysis, Writing – original draft.
J. Liang: Data curation, Formal analysis, Writing – original draft.
L. Peng: Investigation, Writing – original draft.
J. Qin: Investigation, Writing – original draft.
F. Zhou: Investigation, Writing – original draft.
T. Liu: Investigation, Writing – original draft.
L. Dai: Supervision, Formal analysis, Writing – review & editing.
Funding statement
The authors disclose receipt of the following financial or material support for the research, authorship, and/or publication of this article: financial support from the National Natural Science Foundation of China (No. 81801605 and 81971527), Guangdong Natural Science Foundation (No. 2018A030313690), Guangdong Basic and Applied Basic Research Foundation (No. 2019A1515011928), and Science and Technology Program of Guangzhou (No. 201904010088).
ICMJE COI statement
The authors have no conflicts of interest to declare.
Data sharing
Data from this study are available from the corresponding author on reasonable request.
Acknowledgements
The authors thank Dr Jianda Ma for valuable discussion.
Ethical review statement
This study was performed in compliance with the Declaration of Helsinki and was approved by the Medical Ethics Committee of Sun Yat-Sen Memorial Hospital (SYSEC-2009-06 and SYSEC-KY-KS-012). All participants gave written informed consent.
Open access funding
The authors report that the open access funding for their manuscript was self-funded.
Supplementary material
Table showing primers used for quantitative real-time polymerase chain reaction in this research, and figure showing the activation of Rho guanosine triphosphatase by lysophosphatidic acid stimulation.
© 2022 Author(s) et al. This is an open-access article distributed under the terms of the Creative Commons Attribution Non-Commercial No Derivatives (CC BY-NC-ND 4.0) licence, which permits the copying and redistribution of the work only, and provided the original author and source are credited. See https://creativecommons.org/licenses/by-nc-nd/4.0/
Contributor Information
Yaoyao Zou, Email: zouyy29@mail.sysu.edu.cn.
Xuepei Zhang, Email: zhangxp27@mail2.sysu.edu.cn.
Jinjian Liang, Email: liangjj38@mail.sysu.edu.cn.
Liqin Peng, Email: penglq6@mail.sysu.edu.cn.
Jiale Qin, Email: qinjle@mail2.sysu.edu.cn.
Feng Zhou, Email: zhouf58@mail2.sysu.edu.cn.
Ting Liu, Email: liut98@mail2.sysu.edu.cn.
Lie Dai, Email: dailie@mail.sysu.edu.cn.
References
- 1. Miossec P. Rheumatoid arthritis: still a chronic disease. Lancet. 2013;381(9870):884–886. 10.1016/S0140-6736(12)62192-8 [DOI] [PubMed] [Google Scholar]
- 2. Lillegraven S, Prince FHM, Shadick NA, et al. . Remission and radiographic outcome in rheumatoid arthritis: application of the 2011 ACR/EULAR remission criteria in an observational cohort. Ann Rheum Dis. 2012;71(5):681–686. 10.1136/ard.2011.154625 [DOI] [PubMed] [Google Scholar]
- 3. Hollingsworth MA, Swanson BJ. Mucins in cancer: protection and control of the cell surface. Nat Rev Cancer. 2004;4(1):45–60. 10.1038/nrc1251 [DOI] [PubMed] [Google Scholar]
- 4. Nath S, Mukherjee P. MUC1: a multifaceted oncoprotein with a key role in cancer progression. Trends Mol Med. 2014;20(6):332–342. 10.1016/j.molmed.2014.02.007 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5. Kato K, Lillehoj EP, Lu W, Kim KC. MUC1: The first respiratory mucin with an anti-inflammatory function. J Clin Med. 2017;6(12):E110. 10.3390/jcm6120110 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6. Kufe DW. MUC1-C oncoprotein as a target in breast cancer: activation of signaling pathways and therapeutic approaches. Oncogene. 2013;32(9):1073–1081. 10.1038/onc.2012.158 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7. Wu X, Yin Z, McKay C, et al. . Protective epitope discovery and design of MUC1-based vaccine for effective tumor protections in immunotolerant mice. J Am Chem Soc. 2018;140(48):16596–16609. 10.1021/jacs.8b08473 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8. Lin Y, Chen S, Zhong S, An H, Yin H, McGowan E. Phase I clinical trial of PD-1 knockout anti-MUC1 CAR-T cells in the treatment of patients with non-small cell lung cancer. Ann Oncol. 2019;30:xi12. 10.1093/annonc/mdz448 [DOI] [Google Scholar]
- 9. O’Cearbhaill RE, Deng W, Chen L-M, et al. . A phase II randomized, double-blind trial of a polyvalent Vaccine-KLH conjugate (NSC 748933 IND# 14384) + OPT-821 versus OPT-821 in patients with epithelial ovarian, fallopian tube, or peritoneal cancer who are in second or third complete remission: An NRG Oncology/GOG study. Gynecol Oncol. 2019;155(3):393–399. 10.1016/j.ygyno.2019.09.015 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10. Patel JD, Lee J-W, Carbone DP, et al. . Phase II study of immunotherapy with tecemotide and bevacizumab after chemoradiation in patients with unresectable stage III non-squamous non-small-cell lung cancer (NS-NSCLC): a trial of the ECOG-ACRIN cancer research group (E6508). Clin Lung Cancer. 2020;21(6):520–526. 10.1016/j.cllc.2020.06.007 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11. Chu CQ. Fibroblasts in rheumatoid arthritis. N Engl J Med. 2020;383(17):1679–1681. 10.1056/NEJMcibr2024718 [DOI] [PubMed] [Google Scholar]
- 12. Bottini N, Firestein GS. Duality of fibroblast-like synoviocytes in RA: passive responders and imprinted aggressors. Nat Rev Rheumatol. 2013;9(1):24–33. 10.1038/nrrheum.2012.190 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13. Ji M, Ryu HJ, Hong JH. Signalling and putative therapeutic molecules on the regulation of synoviocyte signalling in rheumatoid arthritis. Bone Joint Res. 2021;10(4):285–297. 10.1302/2046-3758.104.BJR-2020-0331.R1 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14. Volin MV, Shahrara S, Haines GK, Woods JM, Koch AE. Expression of mucin 3 and mucin 5AC in arthritic synovial tissue. Arthritis Rheum. 2008;58(1):46–52. 10.1002/art.23174 [DOI] [PubMed] [Google Scholar]
- 15. Chen Y-L, Jing J, Mo Y-Q, et al. . Presence of hepatitis B virus in synovium and its clinical significance in rheumatoid arthritis. Arthritis Res Ther. 2018;20(1):130. 10.1186/s13075-018-1623-y [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16. Aletaha D, Neogi T, Silman AJ, et al. . 2010 Rheumatoid arthritis classification criteria: an American College of Rheumatology/European League Against Rheumatism collaborative initiative. Arthritis Rheum. 2010;62(9):2569–2581. 10.1002/art.27584 [DOI] [PubMed] [Google Scholar]
- 17. Lin J-Z, Liang J-J, Ma J-D, et al. . Myopenia is associated with joint damage in rheumatoid arthritis: a cross-sectional study. J Cachexia Sarcopenia Muscle. 2019;10(2):355–367. 10.1002/jcsm.12381 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18. Krenn V, Perino G, Rüther W, et al. . 15 years of the histopathological synovitis score, further development and review: A diagnostic score for rheumatology and orthopaedics. Pathol Res Pract. 2017;213(8):874–881. 10.1016/j.prp.2017.05.005 [DOI] [PubMed] [Google Scholar]
- 19. Zou Y, Xu S, Xiao Y, et al. . Long noncoding RNA LERFS negatively regulates rheumatoid synovial aggression and proliferation. J Clin Invest. 2018;128(10):4510–4524. 10.1172/JCI97965 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20. Mousavi MJ, Karami J, Aslani S, et al. . Transformation of fibroblast-like synoviocytes in rheumatoid arthritis; from a friend to foe. Auto Immun Highlights. 2021;12(1):3. 10.1186/s13317-020-00145-x [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21. Horm TM, Schroeder JA. MUC1 and metastatic cancer. Cell Adh Migr. 2013;7(2):187–198. 10.4161/cam.23131 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22. Castro I, Albornoz N, Aguilera S, et al. . Aberrant MUC1 accumulation in salivary glands of Sjögren’s syndrome patients is reversed by TUDCA in vitro. Rheumatology (Oxford). 2020;59(4):742–753. 10.1093/rheumatology/kez316 [DOI] [PubMed] [Google Scholar]
- 23. Hamaguchi M, Kawahito Y, Ishino H, et al. . Mucin from rheumatoid arthritis synovial fluid enhances interleukin-6 production by human peripheral blood mononuclear cells. Hum Immunol. 2011;72(3):241–248. 10.1016/j.humimm.2010.12.013 [DOI] [PubMed] [Google Scholar]
- 24. Sachdeva M, Mo Y-Y. MicroRNA-145 suppresses cell invasion and metastasis by directly targeting mucin 1. Cancer Res. 2010;70(1):378–387. 10.1158/0008-5472.CAN-09-2021 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25. Mori Y, Akita K, Tanida S, et al. . MUC1 protein induces urokinase-type plasminogen activator (uPA) by forming a complex with NF-κB p65 transcription factor and binding to the uPA promoter, leading to enhanced invasiveness of cancer cells. J Biol Chem. 2014;289(51):35193–35204. 10.1074/jbc.M114.586461 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26. Nygaard G, Firestein GS. Restoring synovial homeostasis in rheumatoid arthritis by targeting fibroblast-like synoviocytes. Nat Rev Rheumatol. 2020;16(6):316–333. 10.1038/s41584-020-0413-5 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27. Bouillez A, Rajabi H, Pitroda S, et al. . Inhibition of MUC1-C suppresses MYC expression and attenuates malignant growth in KRAS mutant lung adenocarcinomas. Cancer Res. 2016;76(6):1538–1548. 10.1158/0008-5472.CAN-15-1804 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28. Xiao Y, Liang L, Pan Y, et al. . Inhibitory effects of simvastatin on migration and invasion of rheumatoid fibroblast-like synoviocytes by preventing geranylgeranylation of RhoA. Rheumatol Int. 2013;33(2):389–399. 10.1007/s00296-012-2383-7 [DOI] [PubMed] [Google Scholar]
- 29. Chan A, Akhtar M, Brenner M, Zheng Y, Gulko PS, Symons M. The GTPase Rac regulates the proliferation and invasion of fibroblast-like synoviocytes from rheumatoid arthritis patients. Mol Med. 2007;13(5–6):297–304. 10.2119/2007-00025.Chan [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30. Lao M, Shi M, Zou Y, et al. . Protein inhibitor of activated STAT3 regulates migration, invasion, and activation of fibroblast-like synoviocytes in rheumatoid arthritis. J Immunol. 2016;196(2):596–606. 10.4049/jimmunol.1403254 [DOI] [PubMed] [Google Scholar]
- 31. Moran EM, Connolly M, Gao W, McCormick J, Fearon U, Veale DJ. Interleukin-17A induction of angiogenesis, cell migration, and cytoskeletal rearrangement. Arthritis Rheum. 2011;63(11):3263–3273. 10.1002/art.30582 [DOI] [PubMed] [Google Scholar]
- 32. Connolly M, Veale DJ, Fearon U. Acute serum amyloid A regulates cytoskeletal rearrangement, cell matrix interactions and promotes cell migration in rheumatoid arthritis. Ann Rheum Dis. 2011;70(7):1296–1303. 10.1136/ard.2010.142240 [DOI] [PubMed] [Google Scholar]
- 33. He Y, Xu H, Liang L, et al. . Antiinflammatory effect of Rho kinase blockade via inhibition of NF-kappaB activation in rheumatoid arthritis. Arthritis Rheum. 2008;58(11):3366–3376. 10.1002/art.23986 [DOI] [PubMed] [Google Scholar]
- 34. Nomikou E, Livitsanou M, Stournaras C, Kardassis D. Transcriptional and post-transcriptional regulation of the genes encoding the small GTPases RhoA, RhoB, and RhoC: implications for the pathogenesis of human diseases. Cell Mol Life Sci. 2018;75(12):2111–2124. 10.1007/s00018-018-2787-y [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35. Xing L, Yao X, Williams KR, Bassell GJ. Negative regulation of RhoA translation and signaling by hnRNP-Q1 affects cellular morphogenesis. Mol Biol Cell. 2012;23(8):1500–1509. 10.1091/mbc.E11-10-0867 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36. Chen H-H, Yu H-I, Chiang W-C, Lin Y-D, Shia B-C, Tarn W-Y. hnRNP Q regulates Cdc42-mediated neuronal morphogenesis. Mol Cell Biol. 2012;32(12):2224–2238. 10.1128/MCB.06550-11 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37. Pochampalli MR, el Bejjani RM, Schroeder JA. MUC1 is a novel regulator of ErbB1 receptor trafficking. Oncogene. 2007;26(12):1693–1701. 10.1038/sj.onc.1209976 [DOI] [PubMed] [Google Scholar]
- 38. Shen Q, Rahn JJ, Zhang J, et al. . MUC1 initiates Src-CrkL-Rac1/Cdc42-mediated actin cytoskeletal protrusive motility after ligating intercellular adhesion molecule-1. Mol Cancer Res. 2008;6(4):555–567. 10.1158/1541-7786.MCR-07-2033 [DOI] [PubMed] [Google Scholar]


