Abstract
Benzodiazepines are drugs widely used as tranquilizers and in various other indications. We treated Balb/c mice with diazepam and infected them with cowpox (CPXV) and vaccinia virus (VACV). Disease index, weight loss and the antibody response were determined. Additionally the influence of different benzodiazepines on the mitogen response of human peripheral blood lymphocytes and spleen cells was tested. Diazepam led to earlier disease onset, prolonged duration of symptoms, higher weight loss and overall disease index in VACV infected mice. CPXV infected mice developed poxviral skin lesions only after drug administration and a significant decrease in the specific antibody response was also observed. Diazepam and alprazolam also inhibited the proliferative response of human lymphocytes/spleen cells in vitro but did not show noteworthy apoptotic effects. It is surprising that even a single dose of diazepam has a profound influence on the immune system, sufficient to facilitate symptomatic infectious disease. These data provide first evidence that commonly used drugs like Valium® may augment severity of rare poxvirus infections such as CPXV or monkeypox. As VACV is still used as life vaccine against smallpox there is also a risk of enhanced side effects or possible interference with the success of vaccination.
Keywords: Poxvirus, Vaccinia, Cowpox, Benzodiazepines, Diazepam, Valium, Immunosuppression
1. Introduction
Benzodiazepines are a class of substances which function as sedative, anxiolytic, anti-convulsive and muscle relaxing drugs. They act as modulators of the gamma-amino-butyric-acid (GABA) receptor in the central nervous system. Many physiologic functions are modulated by the ionotropic GABAA receptors, ligand-gated ion channels that are permeable for chloride ions and are the most important inhibitory receptors in the central nervous system (CNS) [1]. Benzodiazepines produce allosteric changes that enhance the action of GABA by increasing the apparent affinity of the receptors and thus the frequency of opening the chloride channels. GABA receptors consist of several subunits, not all of them interacting with benzodiazepines. Classical benzodiazepine binding sites contain β-, γ2- and one of the α-subunits. Sedative effects of benzodiazepines are mediated by α1-subtypes, whereas the α2 and α3-subtypes are responsible for the anxiolytic and anticonvulsant effects [2]. Whereas most classical benzodiazepines show all of the typical effects, more recently introduced substances were developed as selective GABA receptor agonists in order to minimize side effects such as memory impairment and abuse potential [3].
In addition to the receptors in the CNS, peripheral-type benzodiazepine receptors (PBR) were discovered, named after the fact that they bind the benzodiazepine diazepam with relatively high affinity [4]. PBR are multimeric complexes, composed of 18 kDa proteins, and were first detected in kidney tissue, but are present in most peripheral organs in high density. Subcellular fractionation experiments localized PBR in the outer mitochondrial membrane and identified various porphyrins as endogenous ligands [5]. It was speculated that regulation of mitochondrial function could account for the effects of benzodiazepines on cell growth and differentiation, alteration in cardiac actions potentials as well as effects on convulsive thresholds. PBR were associated with increased steroid synthesis, with increased cell proliferation such as in cancer, gliosis, and tissue repair functions [6]. In the CNS, PBR are mainly expressed by activated microglial cells and are considered to play a role in neuroinflammatory processes in the pathogenesis of several neurodegenerative disorders [7].
Diazepam (7-chloro-1-methyl-5-phenyl-1,3-dihydro-2H-1,4-benzodiazepin-2-one), is one of the best characterized benzodiazepines and therefore on the WHO “list of essential medicines”. It has been synthesized in the late fifties and has been on the market under the trade name of Valium® since the mid-60s. It is used in the treatment of anxiety disorders, insomnia, alcohol withdrawal, as emergency medication in epileptic seizures, as premedication for surgical interventions and as part of anesthesia. Pharmacodynamic interactions are observed via metabolism routes involving the cytochrom p450 enzyme (CYP) family. Diazepam shows a high affinity to CYP 3A4 [8] and is metabolized by demethylation and hydroxylation in the human body to nordiazepam, temazepam and oxazepam, all these metabolites showing psychoactive features. The latter two undergo further phase II metabolism steps including glucuronidation.
There are only few indications that benzodiazepines might influence immune functions in humans. Use of midazolam during anesthesia has been reported to influence the interleukin response [9] and an increase of CD8 numbers has been observed upon long term use of benzodiazepines [10]. There are also very limited in vivo data on immunomodulating or immunosuppressive effects of benzodiazepines in humans. Indirect evidence arises from a cohort study in HIV positive individuals, where the use of bezodiazepines has been correlated with increased mortality. In multivariate analyses, limited to injection drug users, a positive HIV serostatus, age above 40 years, and using benzodiazepines several times daily were significantly associated with an elevated risk of death [11].
Recently we reported an unusually severe case of cowpox infection [12] in a young male with documented benzodiazepine abuse but showing no signs of immunosuppression [13]. This led us to investigate an effect of diazepam on cowpox infection in an mouse model and to investigate potential drug effects also on human lymphoid cells.
2. Materials and methods
2.1. Virus strains, culture
Vaccinia virus (VACV) laboratory strain WR (Western Reserve), chosen for its known virulence in mice, was obtained from Dr. B. Moss, Bethesda, MD, USA earlier. A human isolate of cowpox virus (CPXV), strain AT/Carynthia/788/07, was grown from local neck infection of a 17-year-old farm girl in Austria [14]. Both viruses were grown on RK13 rabbit kidney cells (ATCC) in roller bottles with enhanced surface (Costar) using DMEM supplemented with 5% FCS. The cells were lysed by freeze thawing, hypotonic medium and brief sonification. The virus supernates were concentrated in a superspeed centrifuge at 20,000 rpm and finally purified over sucrose gradients in a Beckman SW-28 Ultracentrifuge rotor. The resulting virus stocks were diluted in PBS and the virus titer determined by standard plaque assay on RK13 cells in 24-well plates.
2.2. Substances
The benzodiazepines diazepam, nordazepam, oxazepam, flunitrazepam, flurazepam, alprazolam, midazolam, medazepam (Pharmacy of Innsbruck Medical University Hospital, Austria) and lorazepam (Kwizda Pharma, Vienna, AT) were of analytical grade. Diazepam was dissolved in dimethylsulfoxide (DMSO) for the experiments on human PBMC or ethanol for the other experiments. The other drugs tested were dissolved in ethanol. For the experiments they were used as stock solutions of 2.5 mg/ml in solvent and added to the experiments in the indicated dilutions. Phytohemagglutinine (PHA) was purchased from DIFCO Lab., pokeweed mitogen (PWM) from GIBCO Inc., and Escherichia coli lipopolysaccharide (LPS) from Sigma–Aldrich.
2.3. Animal experiments
We used female inbred pathogen free Balb/c mice (obtained from Charles River, Germany) which were 6 weeks old and weighed about 16 g. The animals were divided into cages of groups of five per cage and allowed to adjust to the new environment for some days. They then were treated with 10 μg diazepam per gram body weight in a total volume of 100 μl injected via the intraperitoneal route (i.p.). The diazepam was dissolved in a buffer consisting of 10% ethanol and 1% Tween 20 in physiologic saline solution. The same quantity of this buffer without substance was used in the control groups. On the next day the animals were objected to short term anesthesia with ketamin/xylazin (75 and 7.5 μg/g i.p.) and infected with 10 μl of the above viral stocks via the intranasal route (i.n.). The sublethal infectious dose for i.n. inoculation had been determined in pre-experiments (not shown) for the used viral preparations and has been found to be 103 plaque forming units (p.f.u.) for VACV and 105 p.f.u. for CPXV. These viral doses were chosen for the infection at day 0.
The animals were controlled on a daily basis and the body weights were determined. The disease index (DI) was determined according to a scoring system which has been described earlier [15]. Signs of illness were scored as DI of 0 (normal), 1 (slightly ruffled skin), 2 (clearly ruffled and/or single sided conjunctivitis), 3 (hunched position and/or severe conjunctivitis of both eyes in addition to clear ruffling), 4 (score of 3 combined with difficulty moving/socializing/breathing), and 5 (death). Those animals with a DI of 4 and/or >30% loss of body weight had to be euthanized. At the end of the 14 days observation period the animals were given an anesthesia and blood drawn by heart puncture. Tails were then cut-off immediately from the CPXV infected mice and stained for 5 min with 1% trypan blue in order to visualize necrotic tissue in the poxviral skin lesions.
The work has been evaluated by the local ethics commission and has been approved by the Federal Ministery of Research section for animal experiments (permit BMWF-68.205/0246-II/10b/2008).
2.4. Determination of antibody titer against CPXV
An ELISA system was developed for detection of CPXV specific antibodies in serum. Sucrose purified virus equivalent to 106 p.f.u./well was incubated overnight at 4 °C in 50 μl of 0.1 M NaHCO4, pH 9 in 96-well plates (Nunc, Maxisorb®). After removal of the fluid the plates were air dried and exposed on a UV screen for 1 h. Unspecific binding sites were saturated with 0.5% ovalbumin in phosphate buffered saline (PBS, pH 7.4) for 1 h and twofold dilutions of the mouse sera were added in 50 μl of the same buffer containing 0.1% Tween 20 (PBST). Sera were also added to wells without antigen but treated otherwise identically as background controls. After 2 h incubation at room temperature on a shaking platform the plates were washed three times with 200 μl PBST. Bound antibody was detected with peroxidase-labeled rabbit antiserum against whole mouse immunoglobulin (from DAKO, Dk) in the same buffer and conditions as described above. Plates were again washed three times with PBST and one time with substrate buffer (0.1 M acetate, pH 5.5) and finally developed by the addition 50 μl of 5 mM ABTS (2,2-azino-di3-ethylbenzthiazolinsulfonic acid, from SIGMA) and 1 μl/ml of H2O2 in the latter buffer. After 1 h the substrate reaction was stopped by the addition of 100 μl of 0.1% sodium azide and the photometric extinction was read in a plate reader at 405 nm. The background values obtained in the wells without virus, which never exceeded 10–20% of the readout of the positive wells, were subtracted and the titer arbitrary assigned, with readout of double of the mean of the background levels used as cut-off level. The antibody titers from the five sera each were measured independently in three testings which had been performed on different days. Thus 5 × 3 antibody titers for each group were summarized and the mean value of the dilutions showing a positive result above the cut-off value calculated.
2.5. Lymphocyte proliferation assays
2.5.1. Mitogen assays
The influence of the used substances on proliferation of human lymphocytes was investigated using a standard lymphocyte proliferation protocol involving PHA and PWM as stimulators [16]. In brief, preparations of peripheral blood mononuclear cells (PBMC) were isolated from blood donors by Ficoll density gradient centrifugation. Additionally whole spleen tissue cells were used, which have been obtained from a therapeutical spleenectomy, prepared by a cell grinder and frozen in FCS containing 10% DMSO. The cells were seeded in 96-well round-bottom plates at a concentration of 10/4 cells per well in RPMI1640 culture medium (Gibco) supplemented with 10% pooled human AB serum (Bio Whittaker). After 4 h of incubation with the indicated substances at various dilutions (1:1000–10,000) the mitogens were added in optimal concentrations ranging from 0.1 to 1 μg/ml which had been determined in pre-experiments for the used batches. The plates were incubated for 3 days at 37 °C and then labeled overnight by the addition of H3-Thymidine (Amersham) using a radioactivity of 0.5 μCi/well. The cells were finally harvested with a Canberra Packard 96-well plate harvester and counted in a microscintillation counter from the same company (Topcount®). Means of triplicate experiments were calculated and the standard error of the mean (SEM) indicated.
2.5.2. Apoptosis assay
Human spleen cells were treated with the different substances and mitogens as described above. The cells then were probed for late apoptosis by staining of the cell nuclei with propidium iodide according to published standard procedures [17] and measured in a FACScan III (Beckton Dickinson). The percentage of apoptotic cells was compared with negative controls (mitogen only and untreated cells) and positive controls consisting of cells treated with 5% ethanol.
The work with human cell material has been performed according European guidelines and EC directives for the use of human biological materials in biomedical research and has been covered by a permit of the local ethics commission to E.U. Irschick.
2.6. Statistical analysis
For analyzing the data depicted in Fig. 1, Fig. 2 a non-parametric t-test was applied assuming non-Gaussian distribution. The mean values were depicted with standard error of the mean (SEM) and the statistically significant differences were calculated as two-tailored p-value. Analyzing the ELISA data shown in Fig. 4 significance of difference of the mean antibody titers was calculated by one-way ANOVA analysis using the Dunnet's multiple comparison test. In Fig. 5 a two-way ANOVA analysis of variance was applied as a mixed model for repeated measurements. In Fig. 6 also Dunnet's multiple comparison test (one-way ANOVA analysis) was used for evaluating the differences between the different substances tested. p-Values under 0.05 were regarded as statistically significant. All statistical calculations were carried out using the Prism 5 program from GraphPad Software Inc.
Fig. 1.

Development of body weight of mice infected with VACV, CPXV and uninfected controls. The mean body weight and SEM of animals infected intranasally with 103 p.f.u. of VACV (Fig. 1a), 105 p.f.u. of CPXV (Fig. 1b) and uninfected control mice (Fig. 1c) is shown from days 0 to14 after infection. Groups of mice (n = 5) had been treated with diazepam 1 day before infection and compared to untreated control groups. Significance of difference of the readouts was calculated by t-test and the two-tailored p-value found below 0.05 in the VACV infected mice, but not significant (n.s.) in the other setups (*). One VACV infected mouse from the diazepam group had to be sacrificed on day 8 due to severe disease symptoms/weight loss (**).
Fig. 2.
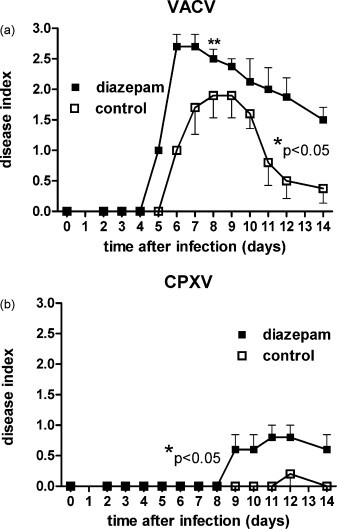
Disease index of mice infected with VACV and CPXV. The means and SEM of the recorded disease index of mice infected intranasally with 103 p.f.u. of VACV (a) and 105 p.f.u. of CPXV (b) is shown for days 0 to 14 after infection. Groups of mice (n = 5) had been treated with diazepam 1 day before infection and compared to untreated control groups. Significance of difference between the two groups was calculated by t-test and the two-tailored p-value found below 0.05 both in the VACV and CPXV infected mice (*). Uninfected mice did not show any disease symptoms. One VACV infected mouse from the diazepam group had to be sacrificed on day 8 due to severe symptoms/weight loss (**).
Fig. 4.
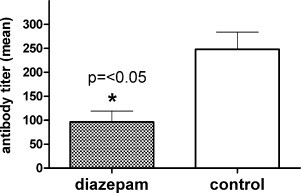
Influence of diazepam on the antibody response in CPXV infected mice. Comparison of the mean antibody titers against CPXV antigen of CPXV infected mice treated with diazepam and untreated controls is shown (n = 5 each group). SEM indicates standard error of the mean of three independent measurements of the five sera in each group. Statistical significance of the difference between the groups as calculated by one-way ANOVA analysis of variance was p < 0.05 (*).
Fig. 5.

Influence of diazepam on the proliferation of human peripheral blood mononuclear cells. PBMC were stimulated with phytohemagglutinin (PHA) (a) and bacterial lipopolysaccharide (LPS) in (b) and treated with diazepam or solvent only (DMSO) at the indicated dilutions. Incorporation of radioactive tritium (H3) by the stimulated cells was measured. Means with SEM of three independent experiments with PBMC from different donors are shown. Statistical significance of the difference between diazepam and solvent control was calculated by two-way ANOVA and found significant (p < 0.05) for the indicated dilutions (*).
Fig. 6.
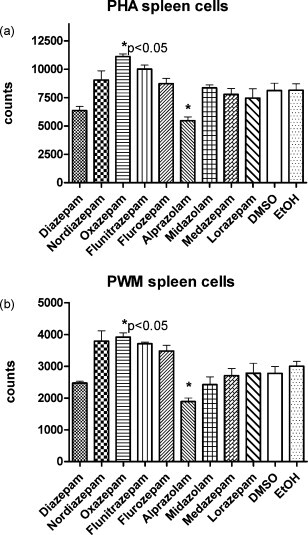
Effect of different benzodiazepines on the proliferation of whole human spleen cells. Phytohemagglutinin PHA (a) and pokeweed mitogen PWM (b) were used as mitogens. All the stock solutions of the indicated substances were used at a dilution 1:2000, solvents served as controls. Incorporation of radioactive tritium (H3) by the stimulated cells was measured. Means of triplicate experiments with SEM using spleen cells from a single human donor are shown. Statistical significance of the difference between the substances as compared to solvent controls was calculated by two-way ANOVA (Dunnet's multiple comparison test) and found significant (p < 0.05) only for oxazepam and alprazolam (*).
3. Results
3.1. Animal data
3.1.1. Development of body weight
As depicted in Fig. 1a infection with VACV led to a clear decrease of the mean body weight of the infected animals. In the diazepam group a weight loss from 16.37 g at day 0 to a minimum 11.65 g at day 8 was observed indicating a reduction of body mass due to disease of 28.8%. The mean body weight of the untreated controls decreased from 16.09 g at day 0 to 14.14 g at day 9, which represents a reduction of 12.1%, respectively. The difference in weight loss between the diazepam treated and untreated mice would have been even bigger if not one out of the five mice in the diazepam group had to be euthanized prematurely due to disease severity and extensive weight loss on day 8, which was not the case in the control group. In the CPXV infected mice (Fig. 1b) no weight loss was observed and symptoms were restricted to a stagnation of weight gain between days 2 and 5 followed by a steady increase in body weight as in the uninfected control mice (Fig. 1c).
3.1.2. Development of disease indicators
As seen in Fig. 2a diazepam led to an earlier onset of disease in the VACV infected mice on day 5 versus day 6 in the control group. Also higher disease severity was noted using the described scoring system of disease index. The untreated mice also recovered much more quickly than the diazepam group resulting in a much longer duration of severe disease in the drug treated mice and doubling of the bourdon of disease, expressed as the area under the curves. CPXV infected mice (Fig. 2b) did not suffer from apparent severe systemic disease but from day 9 on typical poxviral skin lesions developed only on the tails of the diazepam treated mice (rated as disease grade 1). This was not seen on untreated CPXV infected mice and the negative controls (i.e. mice receiving the drug but no virus). After the end of the experiment the mice were euthanized and the tails stained with trypan blue to visualize those skin lesions (Fig. 3 ).
Fig. 3.

Poxviral skin lesions on the tails of CPXV infected mice. Photographs of skin lesions caused by CPXV on day 14 on the tails of infected mice are shown. Tails (n = 5 for each group) were stained with trypan blue to visualize necrotic tissue. Diazepam treated mice (left) were compared to untreated control mice (middle). Tails from uninfected mice which had been treated with diazepam only are shown on the right-hand side.
3.1.3. Antibody response
The antibody response against CPXV was analyzed in the two groups and a significantly (p = <0.05) lower mean ELISA titer was found in the diazepam group (1:96) versus the untreated controls (1:248). Means and SEM of three independent measurements of the five sera each are shown in Fig. 4 .
3.2. Cell proliferation assays
As shown in Fig. 5 we found that diazepam dose dependently interfered with the PHA mediated stimulation of human peripheral blood mononuclear cells (PBMC) whereas the effect on LPS mediated stimulation was less pronounced. This would be an indication for a T-cell mediated mechanism rather than a B-cell mediated effect.
Also the effect on the mitogen proliferation of whole spleen cells from human donors seems to be rather modest as observed with different substances of the benzodiazepine class (Fig. 6a and b). In both setups with PHA and PWM a moderate decrease of proliferative activity was observed with diazepam and alprazolam, whereas oxazepam even showed an enhancing effect on proliferation as compared to the solvent controls.
3.2.1. Apoptosis
No significant increase in late apoptosis was seen in FACS analysis of human spleen cells using propidium iodide as a stain (not shown).
4. Discussion
Cowpox virus (CPXV) is known to circulate in European rodents [18] and occasionally leads to human infection. In most cases cat-transmitted such zoonotic infections are rather mild and restricted to single skin lesions [19], but severe and even lethal cases have also been observed [20]. There are no valid data on the incidence in humans, as there is no mandatory reporting and due to the self-limiting character many cases will remain undiagnosed. Although an isolate with proven virulence in humans [14], the primary isolate of CPXV used in this study seems to be rather mitigated in mice. Our Balb/c mice could handle high doses without symptoms or weight loss and the only signs of infection were a stop in weight increase for a week until the animals recovered and growth curves normalized.
Most interestingly only the diazepam treated mice developed CPXV skin lesions visible on the tails. This finding suggests that either the hematogenous spread of the virus from the respiratory tract to the skin was enhanced, or the diazepam-induced immunosuppression plays a more specific role the in development of skin lesions. In humans hematogenous spread of CPXV seems quite common, as DNA can be detected in the serum even in benign cases using highly sensitive PCR methods [21].
Vaccinia virus (VACV) has been used as a rather virulent live vaccine against smallpox virus, but the virus can still be found in Indian buffalos (“buffalopox”) and seems to persist in wild rodents at least in Brazil [22]. In our mouse model diazepam led to earlier onset of infection with VACV, increased disease severity and weight loss. Presumably also an increase in mortality, although this could not be tested, following the prescription of the committee for animal experiments.
The observation that even a single dose of diazepam before the infection substantially decreased the antibody formation in mice which are relatively immune against cowpox infection is a new finding and correlates well to our recent observations in a human case. A young drug taker with documented benzodiazepine abuse did not show the expected seroconversion after a generalized CPXV infection [13]. The antibody response seems to play a major role in the defense against orthopoxvirus infections. Especially the CD4(+) T-cell dependent anti-virus antibody production has been described to play a more important role in clearing the VACV strain WR following acute infection than CD8(+) T-cells [23].
The effects of diazepam on the antibody production have been investigated in few in vitro studies with conflicting results: both stimulatory and inhibitory effects have been demonstrated. A recent paper reported an inhibitory effect of diazepam on cytokine and antibody production in ovalbumin sensitized Balb/c mice [24]. In the contrary another group had observed stimulation by diazepam of the antibody production against sheep red blood cells (a T-cell dependent antigen) in mouse spleen cells [25]. This could be a dose dependent effect and an indication that diazepam may also stimulate certain helper T-cell functions. Evidence for such stimulatory effects may have also been observed in long term drug therapy of psychiatric patients, where benzodiazepine use has been correlated with higher CD8 T-cell numbers [10].
As mentioned earlier benzodiazepine use has been correlated with an unfavorable outcome in a HIV cohort study [11]. We also tried to assess the immune status of benzodiazepine users among HIV infected patients by comparing the mitogen control reaction of the Quantiferon Gold TB assay (Cellestis). Patients with benzodiazepine use showed lower levels of mitogen mediated immune stimulation as measured by interferon gamma, but due to low sample numbers this was not statistically significant (not shown). One possible way how benzodiazepines could negatively affect stimulated immune cells (as present in HIV patients) or prevent immunoglobulin production is via apoptosis. We did not see marked apoptotic effects on spleen cells, but novel classes of benzodiazepines have been found capable of inducing germinal centre B-cell apoptosis and have been even suggested as possible immunosuppressive agents fighting autoimmune diseases [26]. Our finding of a less pronounced diazepam effect on LPS stimulated B-cells or whole spleen cells as compared to PHA mediated stimulation of T-cells would also not fit to a predominantly B-cell mediated mechanism.
The long lasting effect of a single drug dose is surprising, but there is some rather old evidence that the immune response of rats can be altered even by administration of diazepam in the prenatal [27] or early postnatal period [28]. Effects have also been described on peritoneal macrophages [29]. Diazepam treatment decreased macrophage spreading, phagocytosis and release of H2O2 as well as serum levels of corticosteroids. Benzodiazepine receptors have been postulated to play a major role in regulating steroid production [30] but in many of the animal studies reported mere steroid effects can be excluded. Moreover as Balb/c mice have been shown to be more prone to anxious behavior than C57BL/6 [31] one would have expected a positive effect of the sedation.
One interesting observation was that the onset of illness was faster in the drug treated mice, which argues for an early effect on the immune response. Besides suggested influences of diazepam on macrophage function [29], which could lead to interference with phagocytosis of opsonized viral particles, earlier data also suggested an effect on NK cell activity [32]. This could also contribute to a faster development of disease symptoms.
Interestingly alprazolam also showed some inhibitory effect in human lymphocyte cultures in our experiments. In an earlier report alprazolam has been reported to have stimulatory effects on mouse lymphocytes. But this has only been observed with low doses but not higher doses [33].
Midazolam and propofol have been reported to alter secretion of interleukin 8, a chemotactic agent in human neutrophils [34], and on human peripheral blood monocytes midazolam showed inhibition of the LPS-induced production of IL1-beta and TNF [9]. Midazolam was also reported to improve the outcome of septic rats most recently [35] contradicting old reports of an adverse effect of diazepam on Klebsiella infection [36]. These discrepancies highlight the difficulties in many animal studies which arise from the problem of dissecting direct drug effects on immune cells from the psychotropic effect indirectly influencing immune mechanisms. Thus positive effects of the anxiolytic benzodiazepine function on immune functions have been clearly observed also in animal studies [37].
Benzodiazepines have been used for many years, especially diazepam which has been heavily used against anxiety, stress and sleeping disorders under the trade name of Valium®. It therefore was surprising to find that it had a rather profound effect on the severity of orthopoxvirus infection in mice. It is likely that the same mechanism of immunosuppression is also present in humans as diazepam decreased the proliferative response of human lymphocytes to various mitogens in in vitro assays. A possible enhancement of severity of poxvirus infections and overall disease bourdon raises concern considering zoonotic CPXV infections which have been increasingly observed across Europe in association with infected pet rats [12], [38], [39]. Given the similarity of the disease patterns one would also expect a similar mechanism on prairie dog transmitted monkeypox virus infection which has been striking the U.S. recently [40]. Additionally our data also generate new safety issues concerning VACV, which is still used as an outdated live vaccine against smallpox. There might be safety concerns with the use of VACV in vaccinee's taking benzodiazepines and one has to consider a possibly increased vulnerability to infection associated side effects in people exposed to benzodiazepines. Finally as well a possible interference with the immune response and the success of the vaccination has to be considered in certain medical conditions.
Acknowledgements
The work has been supported by the Austrian Federal Ministry of Health as a contribution to the smallpox preparedness program (Pockenalarmplan) and the medical research fund Tyrol (MFF).
References
- 1.D’Hulst C., Atack J.R., Kooy R.F. The complexity of the GABA receptor shapes unique pharmacological profiles. Drug Discovery Today. 2009;17-18:866–875. doi: 10.1016/j.drudis.2009.06.009. [DOI] [PubMed] [Google Scholar]
- 2.McKernan R.M., Rosahl T.W., Reynolds D.S., Sur C., Wafford K.A., Atack J.R., et al. Sedative but not anxiolytic properties of benzodiazepines are mediated by the GABA(A) receptor alpha1 subtype. Nature Neuroscience. 2000;3:587–592. doi: 10.1038/75761. [DOI] [PubMed] [Google Scholar]
- 3.Iversen L. GABA pharmacology – what prospects for the future? Biochemical Pharmacology. 2004;68:1537–1540. doi: 10.1016/j.bcp.2004.06.039. [DOI] [PubMed] [Google Scholar]
- 4.Papadopoulos V. In search of the function of the peripheral-type benzodiazepine receptor. Endocrine Research. 2004;30:677–684. doi: 10.1081/erc-200043971. [DOI] [PubMed] [Google Scholar]
- 5.Snyder S.H., Verma A., Trifiletti R.R. The peripheral-type benzodiazepine receptor: a protein of mitochondrial outer membranes utilizing porphyrins as endogenous ligands. FASEB Journal. 1987;1:282–288. doi: 10.1096/fasebj.1.4.2820823. [DOI] [PubMed] [Google Scholar]
- 6.James M.L., Selleri S., Kassiou M. Development of ligands for the peripheral benzodiazepine receptor. Current Medical Chemistry. 2006;13:1991–2001. doi: 10.2174/092986706777584979. [DOI] [PubMed] [Google Scholar]
- 7.Venneti S., Lopresti B.J., Wiley C.A. The peripheral benzodiazepine receptor (translocator protein 18 kDa) in microglia: from pathology to imaging. Progress in Neurobiology. 2006;80:308–322. doi: 10.1016/j.pneurobio.2006.10.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.He Y.A., Roussel F., Halpert J.R. Analysis of homotropic and heterotropic cooperativity of diazepam oxidation by CYP3A4 using site-directed mutagenesis and kinetic modeling. Archives of Biochemistry and Biophysics. 2003;409:92–101. doi: 10.1016/s0003-9861(02)00484-8. [DOI] [PubMed] [Google Scholar]
- 9.Taupin V., Jayais P., Descamps-Latscha B., Cazalaa J.B., Barrier G., Bach J.F., et al. Benzodiazepine anesthesia in humans modulates the interleukin-1 beta, tumor necrosis factor-alpha and interleukin-6 responses of blood monocytes. Journal of Neuroimmunology. 1991;35:13–19. doi: 10.1016/0165-5728(91)90157-3. [DOI] [PubMed] [Google Scholar]
- 10.Cosentino M., Fietta A., Caldiroli E., Marino F., Rispoli L., Comelli M., et al. Assessment of lymphocyte subsets and neutrophil leukocyte function in chronic psychiatric patients on long-term drug therapy. Progress in Neuropsychopharmacology and Biological Psychiatry. 1996;20:1117–1129. doi: 10.1016/s0278-5846(96)00100-5. [DOI] [PubMed] [Google Scholar]
- 11.van Haastrecht H.J., van Ameijden E.J., van den Hoek J.A., Mientjes G.H., Bax J.S., Coutinho R.A. Predictors of mortality in the Amsterdam cohort of human immunodeficiency virus (HIV)-positive and HIV-negative drug users. American Journal of Epidemiology. 1996;143:380–391. doi: 10.1093/oxfordjournals.aje.a008752. [DOI] [PubMed] [Google Scholar]
- 12.Hönlinger B., Huemer H.P., Romani N., Czerny C.P., Eisendle K., Höpfl R. Generalized cowpox infection probably transmitted from a rat. British Journal of Dermatology. 2005;153:451–453. doi: 10.1111/j.1365-2133.2005.06731.x. [DOI] [PubMed] [Google Scholar]
- 13.Huemer H.P., Himmelreich A., Hönlinger B., Pavlic M., Eisendle K., Höpfl R., et al. “Recreational” drug abuse associated with failure to mount a proper antibody response after a generalised orthopoxvirus infection. Infection. 2007;35:469–473. doi: 10.1007/s15010-007-6194-9. [DOI] [PubMed] [Google Scholar]
- 14.Richter S., Huemer H.P., Revilla-Fernández S., Zenz W., Strenger V., Müller M., et al. Diagnosis of Cowpox virus in Austria, 2007. Infection. 2008;36(Suppl. I):59. [Google Scholar]
- 15.Berhanu A., Wilson R.L., Kirkwood-Watts D.L., King D.S., Warren T.K., Lund S.A., et al. Vaccination of Balb/c mice with Escherichia coli-expressed vaccinia virus proteins A27L, B5R, and D8L protects mice from lethal vaccinia virus challenge. Journal of Virology. 2008;82:3517–3529. doi: 10.1128/JVI.01854-07. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Froebel K.S., Pakker N.G., Aiuti F., Bofill M., Choremi-Papadopoulou H., Economidou J., et al. Standardization and quality assurance of lymphocyte proliferation assays for use in the assessment of immune function. Journal of Immunological Methods. 1999;227:85–97. doi: 10.1016/s0022-1759(99)00082-4. [DOI] [PubMed] [Google Scholar]
- 17.Riccardi C., Nicoletti I. Analysis of apoptosis by propidium iodide staining and flow cytometry. Nature Protocols. 2006;1:1458–1461. doi: 10.1038/nprot.2006.238. [DOI] [PubMed] [Google Scholar]
- 18.Essbauer S., Pfeffer M., Meyer H. Zoonotic poxviruses. Veterinary Microbiology. 2010;140:229–236. doi: 10.1016/j.vetmic.2009.08.026. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Hawranek T., Tritscher M., Muss W.H., Jecel J., Nowotny N., Kolodziejek J., et al. Feline orthopoxvirus infection transmitted from cat to human. Journal of the American Academy of Dermatology. 2003;49(3):513–518. doi: 10.1067/s0190-9622(03)00762-x. [DOI] [PubMed] [Google Scholar]
- 20.Czerny C.P., Zeller-Lue C., Eis-Hübinger A.M., Kaaden O.R., Meyer H. Characterization of a cowpox-like orthopox virus which had caused a lethal infection in man. Archives of Virology. 1997;Suppl. 13:13–24. doi: 10.1007/978-3-7091-6534-8_2. [DOI] [PubMed] [Google Scholar]
- 21.Nitsche A., Kurth A., Pauli G. Viremia in human Cowpox virus infection. Journal of Clinical Virology. 2007;40:160–162. doi: 10.1016/j.jcv.2007.07.014. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Trindade G.S., Emerson G.L., Carroll D.S., Kroon E.G., Damon I.K. Brazilian vaccinia viruses and their origins. Emerging Infectious Diseases. 2007;13:965–972. doi: 10.3201/eid1307.061404. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Xu R., Johnson A.J., Liggitt D., Bevan M.J. Cellular and humoral immunity against vaccinia virus infection of mice. Journal of Immunology. 2004;172:6265–6271. doi: 10.4049/jimmunol.172.10.6265. [DOI] [PubMed] [Google Scholar]
- 24.Wey S.P., Wu H.Y., Chang F.C., Jan T.R. Methamphetamine and diazepam suppress antigen-specific cytokine expression and antibody production in ovalbumin-sensitized Balb/c mice. Toxicology Letters. 2008;181:157–162. doi: 10.1016/j.toxlet.2008.07.015. [DOI] [PubMed] [Google Scholar]
- 25.Okimura T., Nagata I. Effect of benzodiazepine derivatives: I. Augmentation of T cell-dependent antibody response by diazepam in mouse spleen cells. Journal of Immunopharmacology. 1986;8:327–346. doi: 10.3109/08923978609026493. [DOI] [PubMed] [Google Scholar]
- 26.Bednarski J.J., Lyssiotis C.A., Roush R., Boitano A.E., Glick G.D., Opipari A.W., Jr. A novel benzodiazepine increases the sensitivity of B cells to receptor stimulation with synergistic effects on calcium signalling and apoptosis. Journal of Biological Chemistry. 2004;279:29615–29621. doi: 10.1074/jbc.M403507200. [DOI] [PubMed] [Google Scholar]
- 27.Schlumpf M., Parmar R., Ramseier H.R., Lichtensteger W. Prenatal benzodiazepine immunosuppression: possible involvement of peripheral benzodiazepine site. Developmental Pharmacology and Therapeutics. 1990;15:178–185. doi: 10.1159/000457643. [DOI] [PubMed] [Google Scholar]
- 28.Dostál M., Benesová O., Tejkalová H., Soukupová D. Immune response of adult rats is altered by administration of diazepam in the first postnatal week. Reproductive Toxicology. 1995;9:115–121. doi: 10.1016/0890-6238(94)00061-1. [DOI] [PubMed] [Google Scholar]
- 29.Massoco C.O., Palermo-Neto J. Diazepam effects on peritoneal macrophage activity and corticosterone serum levels in Balb/c mice. Life Sciences. 1999;65:2157–2165. doi: 10.1016/s0024-3205(99)00481-6. [DOI] [PubMed] [Google Scholar]
- 30.Ferrarese C., Appollonio I., Bianchi G., Frigo M., Marzorati C., Pecora N., et al. Benzodiazepine receptors and diazepam binding inhibitor: a possible link between stress, anxiety and the immune system. Psychoneuroendocrinology. 1993;18:3–22. doi: 10.1016/0306-4530(93)90051-l. [DOI] [PubMed] [Google Scholar]
- 31.Depino A.M., Gross C. Simultaneous assessment of autonomic function and anxiety-related behavior in Balb/c and C57BL/6 mice. Behavioural Brain Research. 2007;177:254–260. doi: 10.1016/j.bbr.2006.11.025. [DOI] [PubMed] [Google Scholar]
- 32.Stepień H., Agro A., Padol I., Stanisz A. Inhibitory effect of diazepam on human natural killer activity in vitro. Cytobios. 1994;77:131–136. [PubMed] [Google Scholar]
- 33.Fride E., Skolnick P., Arora P.K. Immunoenhancing effects of alprazolam in mice. Life Sciences. 1990;47:2409–2420. doi: 10.1016/0024-3205(90)90485-a. [DOI] [PubMed] [Google Scholar]
- 34.Galley H.F., Dubbels A.M., Webster N.R. The effect of midazolam and propofol on interleukin-8 from human polymorphonuclear leukocytes. Anesthesia and Analgesia. 1998;86:1289–1293. doi: 10.1097/00000539-199806000-00030. [DOI] [PubMed] [Google Scholar]
- 35.Qiao H., Sanders R.D., Ma D., Wu X., Maze M. Sedation improves early outcome in severely septic Sprague–Dawley rats. Critical Care. 2009;13:R136. doi: 10.1186/cc8012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Laschi A., Descotes J., Tachon P., Evreux J.C. Adverse influence of diazepam upon resistance to Klebsiella pneumoniae infection in mice. Toxicology Letters. 1983;16:281–284. doi: 10.1016/0378-4274(83)90188-1. [DOI] [PubMed] [Google Scholar]
- 37.Kaledin V.I., Llnitskaya S.I., Nikolin V.P., Popova N.A., Smagin D.A., Kudryavtseva N.N. Limiting effect of diazepam on Lewis Lung carcinoma metastasis in anxious male mice. Experimental Oncology. 2009;31:62–64. [PubMed] [Google Scholar]
- 38.Becker C., Kurth A., Hessler F., Kramp H., Gokel M., Hoffmann R., et al. Cowpox virus infection in pet rat owners: not always immediately recognized. Deutsches Ärzteblatt International. 2009;106:329–334. doi: 10.3238/arztebl.2009.0329. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Ninove L., Domart Y., Vervel C., Voinot C., Salez N., Raoult D., et al. Cowpox virus transmission from pet rats to humans, France. Emerging Infectious Diseases. 2009;15:781–784. doi: 10.3201/eid1505.090235. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Centers for Disease Control and Prevention (CDC). Update: multistate outbreak of monkeypox – Illinois, Indiana, Kansas, Missouri, Ohio, and Wisconsin, 2003. MMWR Morbidity Mortality Weekly Reports, vol. 52(27); 2003. p. 642–6. [PubMed]


