Abstract
Improving maternal and child health is a global priority. Although infection with Listeria monocytogenes (LM), a small facultative anaerobic, gram-positive motile bacillus is rare, when it infects the maternal-fetoplacental unit, it can result in adverse fetal sequelae such as chorioamnionitis, preterm labour, neonatal sepsis, meningitis and neonatal death. Pregnancy-associated listeriosis may present with a plethora of diverse, non-specific symptoms such as fever, influenza-like or gastrointestinal symptoms, premature contractions and preterm labour. It has a predilection for the second and third trimester of pregnancy, occurring sporadically or as part of an outbreak, most of which have involved unpasteurised dairy products, long shelf life products, contaminated ready-to-eat food, deli meats and soft cheeses. Strains belonging to the clonal complexes 1, 4 and 6 are hypervigilant and are commonly associated with maternal-neonatal infections. Maternal listeriosis occurs as a direct consequence of LM-specific placental tropism, which is mediated by the conjugated action of internalin A and internalin B at the placental barrier. The diagnosis is established from placental culture. Penicillin, ampicillin and amoxicillin are the antimicrobials of choice. It has a high fetal morbidity of up to 30%. The authors present the case of a multiparous woman in her early 20s presenting with sepsis and preterm premature rupture of her membranes at 21 weeks gestation. A live baby was delivered spontaneously and died shortly after birth. Placental cultures and postmortem examination were consistent with the diagnosis of disseminated Listeria infection. Due to the increased susceptibility of pregnant women for LM, a high index of clinical suspicion is required to establish the diagnosis and initiate appropriate antimicrobial therapy to reduce adverse fetal outcomes.
Keywords: Infectious diseases, Obstetrics and gynaecology, Pregnancy
Background
Listeriosis is mainly a food-borne infection and typically occurs sporadically, but nosocomial clusters can also occur. During pregnancy, vertical transmission of infection from the mother to her fetus can result in devastating consequences. It is universally accepted that antenatal transplacental transmission of known ‘TORCH’ pathogens which includes Toxoplasma gondii, other infections (Listeria monocytogenes (LM), Treponema pallidum, parvovirus, HIV, varicella zoster virus and Zika virus), rubella, cytomegalovirus and herpes viruses occurs. However, fetal complications largely occur in the absence of overt illness in the woman delaying medical intervention. Despite the devastating impact of microbial infections on the developing fetus, there remains a paucity of evidence on how the pathogens breach the placental barrier to transmit vertically during pregnancy. The gastrointestinal tract is the primary site of entry for pathogenic Listeria species. It thrives and spreads intracellularly causing invasive disseminated disease to other organs including the liver, spleen, central nervous system and, in pregnant women, the placenta. To understand vertical transmission, it is necessary to understand the structure of the human placenta. By 10–12 weeks of gestation, the maternal circulation has been remodelled through the formation of spiral arteries, and the placenta becomes haemochorial (maternal blood comes into direct contact with the chorion). Given its sole interface between the maternal and fetal environments, it protects the fetus from pathogens present in maternal blood. Syncytiotrophoblasts provide protection against infection through effectors that operate in a paracrine manner. Two surface bacterial proteins, internalin A and internalin B, mediate invasion of non-phagocytic cells. Maternal listeriosis may result in preterm labour, miscarriage, stillbirth, neonatal meningitis, sepsis or neonatal death. It remains one of the infections associated with the highest fetal and neonatal morbidity, leading to fetal loss in up to 30% of cases.1
Case presentation
A multiparous woman (gravida 3 and parous 2) in her early 20s presented to the maternity assessment unit with an unbooked pregnancy at 21 weeks gestation by her last menstrual period. She received no formal antenatal care or dating ultrasound scan. She described exertional dyspnoea, severe abdominal pain and a dry cough. She was of Eastern European descent and did not speak English. Her history was taken using the translation service. She was not registered with a general practitioner, and she was unvaccinated against the COVID-19 virus. She had no comorbidities or known medication allergies. There was no history of illness such as vomiting or diarrhoea. She reported episodes of reduced fetal movements over 24 hours. Her obstetric history included two normal vaginal deliveries at term.
Respiratory examination confirmed decreased air entry bilaterally, dyspnoea and tachypnoea. Cardiovascular examination confirmed tachycardia. Abdominal and speculum examination confirmed a soft gravid uterus and a brown vaginal loss. The cervical os was closed. There was evidence of spontaneous rupture of the membranes, and thin meconium was observed. A high vaginal swab was requested.
Investigations
Her maternity early warning score was 11 (temperature 40°C, blood pressure 95/45 mm Hg, heart rate 131 beats per minute, respiratory rate 28 breaths per minute and SpO2 98% on air). A 12-lead ECG confirmed sinus tachycardia. Her rapid 2019-nCOV PCR (Samba II) test was negative.
An anteroposterior chest radiograph showed normal lungs and pleural spaces. Urinalysis showed a trace of protein and leucocytes. A midstream urine sample was requested. Laboratory investigations confirmed an elevated white cell count (16.3×109/L), a raised C reactive protein (223 mg/L) and a raised lactate (2.0 mmol/L) (table 1). Her clotting was normal. Her blood cultures were negative. A growth scan confirmed a single intrauterine pregnancy with an estimated gestation of 21 weeks. Rubella, cytomegalovirus, parvovirus and Toxoplasma IgM were negative (table 2). An antenatal screen for hepatitis B, HIV and Treponema were negative.
Table 1.
Laboratory investigations confirmed an elevated white cell count and C reactive protein
| Full blood count | Urea and electrolytes | ||||
| White cell count | 16.3×109/L | (4–11) | Sodium | 139 mmol/L | (133–146) |
| Haemoglobin | 107 g/L | (115–165) | Potassium | 3.5 mmol/L | (3.5–5.3) |
| Mean cell volume | 90.2 fL | (80–100) | Urea | 2.2 mmol/L | (2.5–7.8) |
| Platelets | 255×109/L | (150–450) | Creatinine | 36 µmol/L | (50–100) |
| Neutrophils | 13.06×109/L | (1.7–7.50) | EGFR | >90 mL/min/1.73 m2 | (60–150) |
| Lymphocytes | 2.11×109/L | (1.00–4.00) | Liver function tests | ||
| Eosinophil count | 0.2×109/L | (0.04–0.4) | Albumin | 23 g/L | (35–50) |
| Alkaline phosphatase | 92 IU/L | (30–130) | |||
| C reactive protein | 223 mg/L | (0–7) | Alanine transaminase | 15 IU/L | (10–50) |
| Lactate | 2.0 mmol/L | (range: 0.6–2.5) | Total bilirubin | 7 µmol/L | (0–21) |
Table 2.
Serological samples were sent to the reference laboratory
| Rubella infection screen | IgG detected IgM negative |
| Cytomegalovirus screen | IgG positive IgM negative |
| Toxoplasma total antibody | Positive Toxoplasma antibody dye test 125 IU/mL Toxoplasma IgM negative |
| Parvovirus | IgM negative IgG negative |
Differential diagnosis
A diagnosis of preterm premature rupture of the membranes and maternal sepsis caused by chorioamnionitis was established. She was placed on the ‘sepsis pathway’ and managed accordingly. She was admitted to the labour ward. She was counselled by senior obstetric and neonatal consultants experienced in managing such cases. The risk of pulmonary hypoplasia, respiratory distress, limb contractures, facial deformities and neonatal mortality were discussed. The Trust’s preterm premature rupture of membranes (PPROM) pathway did not include steroids at <23 weeks gestation.
Treatment
Intravenous fluid resuscitation was initiated, and an indwelling urinary catheter was inserted. She was given treatment with intravenous antibiotics for sepsis, in this case, tazocin 4.5 g three times a day and gentamicin as per the Trust’s antimicrobial policy. Her venous thromboembolism score was 5, and she was given treatment with low molecular weight heparin, dalteparin 5000 units once daily.
Outcome and follow-up
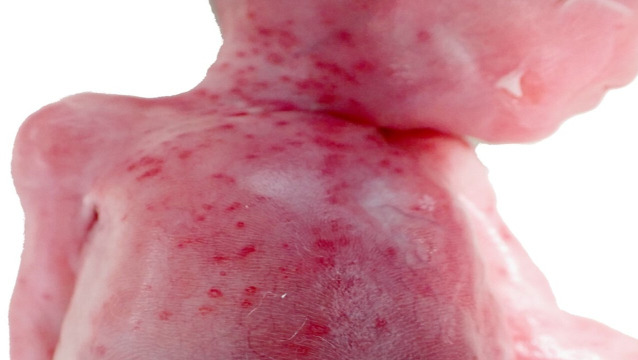
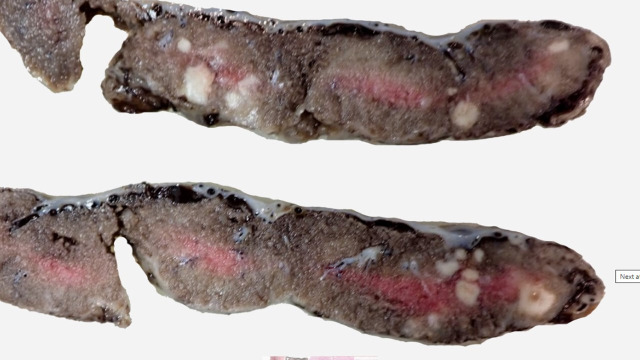
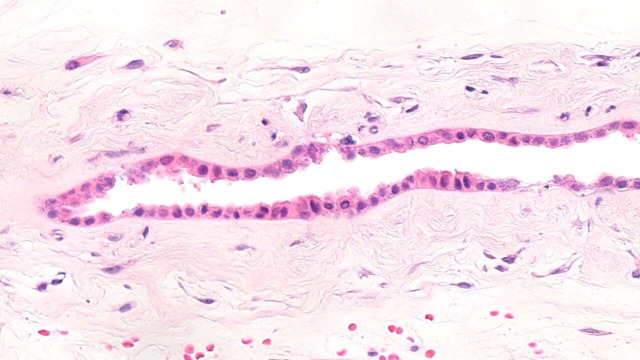
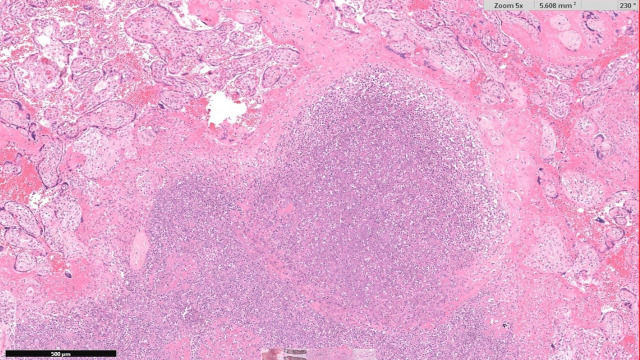
She developed acute onset of abdominal pain with perineal pressure and an urge to push. She delivered a live female baby within minutes and approximately 50 min following PPROM. The estimated blood loss was 200 mL. The perineum was intact. The neonatal team were fast bleeped. The neonatal consultant arrived within 30 s of the birth. The baby was observed gasping intermittently. The umbilical cord was clamped, and the baby was placed on the resuscitaire. Following delivery, the baby’s care was redirected as the fetal weight was 550 g, suggesting a gestation of 23 weeks. Neonatal cardiopulmonary resuscitation was performed. Chest wall movement was observed with no regular respiratory effort. Cardiovascular examination confirmed a bradycardia with a heart rate of 60 beats per minute. The skin showed multiple pustules consistent with sepsis and became macerated with minimal handling. The transwarmer was used to prevent hypothermia. The heart rate dropped to 20 beats per minute. Due to the combination of extreme prematurity, widespread diffuse petechiae and bradycardia, the decision was made not to continue with the resuscitation attempts. Ventilation breaths were continued while awaiting the translator to communicate events with the mother. Ventilation breaths were discontinued, and the baby was given to the mother for cuddles. There was no sign of respiratory effort, and the baby died after 45 min. The mother consented for an external postmortem which included a careful examination of the outside of the baby’s body, radiographs, medical photographs and examination of the placenta. She declined a complete postmortem. The external postmortem examination showed multiple macules with white centres (pustules) scattered over the trunk, thighs, neck and upper limbs (figure 1). A skeletal survey including anteroposterior and lateral ‘babygram’ views showed a femur measuring 40 mm in length and ossification consistent with a gestational age of 26 weeks (range 24–28 weeks). The weight (507.9 g) and linear measurements were consistent with a gestational age of 23 weeks. Note was made of the gas within the stomach and small bowel indicating that the baby was born alive. The placental cut surface showed white-grey abscesses, and the membranes showed evidence of severe acute chorioamnionitis (figure 2). The membrane amniocytes showed abundant intracytoplasmic rods, consistent with Listeria infection (figure 3). There was focal membrane calcification. The parenchyma showed multifocal intervillous abscess formation (figure 4). The surrounding villi showed some obliterative changes including possible expansion by mononuclear cells. There was diffuse villus oedema. The basal decidua showed focal necrosis and a focal increase in trophoblastic giant cells. Focal chorionic plate thrombosis was also noted. The appearances were consistent with disseminated congenital Listeria infection. Placental infection was the cause of spontaneous preterm labour.
Figure 1.
Multiple cutaneous pustules with erythematous bases.
Figure 2.
Placental cut surface. Multiple white-grey abscesses.
Figure 3.
Free membrane amniocytes with abundant intracytoplasmic bacteria (H&E stain, 20× objective).
Figure 4.
Intervillous abscess (H&E stain, 5× objective).
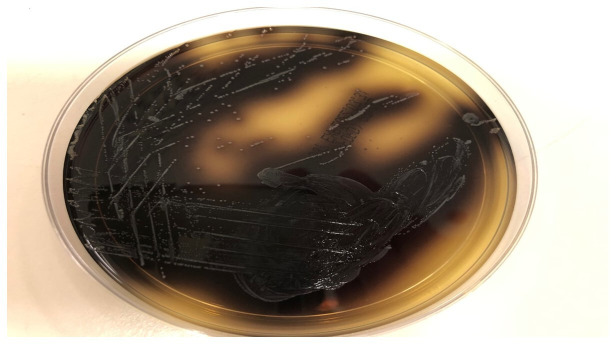
Congenital sepsis and extreme prematurity were the cause of neonatal death. Under the duty of candour, the parents were informed that a review of their care and that of their baby would be carried out. The parents did not express any concerns about their care or that of their baby. The ‘child death process pathway’ was followed, and the ‘notification of child death’ was completed. The case was also discussed with the coroner’s office, and as the baby was less than 24 weeks gestation, no further input was required. Cabergoline 1 mg was given to suppress lactation. After 24 hours incubation, the blood agar grew small grey colonies with no haemolysis (figures 5 and 6). The placenta cultures were classified as LM, sensitive to penicillin, amoxicillin and cotrimoxazole. As the patient was clinically well and already taking oral amoxicillin, the microbiologist advised that no further action was required. The source of the LM infection was soft cheese. The mother was counselled on the importance of avoiding soft cheese and unpasteurised dairy products in future pregnancies. She was discharged home, and a debrief appointment was scheduled.
Figure 5.
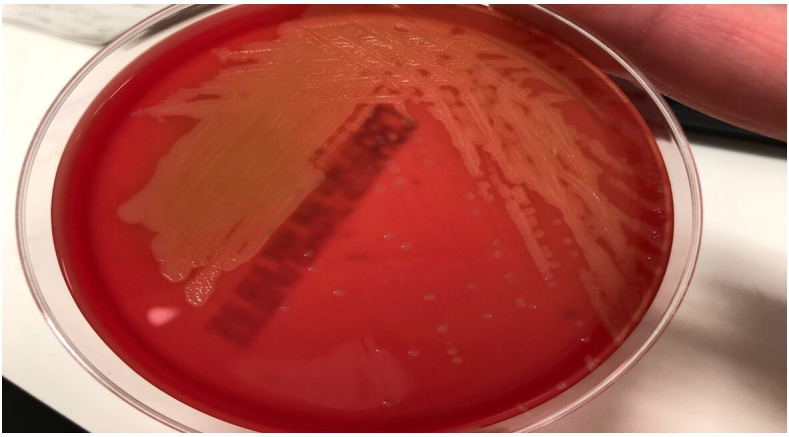
The red plate shows the isolate, Listeria monocytogenes cultured on columbia blood agar.
Figure 6.
The black colonies on the yellow agar are on the Listeria selective agar (Oxford formulation). Both media are from Thermo Fisher (Oxoid).
Discussion
LM, a small, facultatively anaerobic, gram-positive, flagellated, linear motile rod, has the ability to survive in human cells. It was first described by Hulphers in Sweden in 1911 following its discovery as a causative agent of liver abscesses, monocytosis and septicaemia in rabbits in 1926.1–3 Historically, the organism has been known as Bacillus hepatitis, Bacterium monocytogenes, Listeria hepatolytica and Bacterium monocytogenes hominis following its first reported case in humans in 1929.2–5 Its current name was finally agreed on in 1940 in honour of Lord Joseph Lister.6 It is microscopically difficult to distinguish from the commensal diphtheroids and was initially thought to be from the genus Corynebacterium.7 There are seven known species of Listeria, and only four variants affect humans, with most cases due to the following three serotypes: 1/2a, 1/2b and 4b. Farber and Perkin have provided a comprehensive review on the history of Listeria.8 Although it can survive for many months in soil, pasteurisation can eliminate it. Its identification as a major foodborne pathogen led to the implementation of a surveillance system in most countries in the Western hemisphere. In industrialised countries, Listeria has an incidence of 3–6 per million population, with a higher incidence noted among Hispanics.9 10 Interestingly, it is up to 20 times more prevalent in pregnancy than in the general population, and its crude incidence is estimated at 10–100 times higher in this group.6 11 There is enhanced susceptibility of the fetus to LM after the first trimester, suggesting that there are gestational age-specific differences in vertical transmission or that cell-mediated immunity is deficient. It rarely causes invasive disease in immunocompetent individuals and is shed symptomatically by 1% of healthy individuals.7 It has a median incubation time of 19–27.5 days, reflecting the time needed for maternal sepsis, placental and fetal infection to develop.12
Several theories on its pathogenesis in pregnancy have been hypothesised. LM crosses the epithelial barrier of the intestine, translocates to the mesenteric lymph nodes and disseminates to distant organs such as the liver and spleen, where it establishes infectious foci. In immunocompetent individuals, the infection is largely subclinical and is cleared by cell-mediated immunity. In immunocompromised individuals, including pregnancy, the primary infectious foci are inadequately resolved, and LM may be released into the bloodstream. It has been suggested that the placenta itself may become a reservoir for maternal reinfection and amplification of the bacterial load. In addition, there is a progressive increase in uterine blood flow and consequent exposure of the placenta to infection. There is also a recognised shift in the woman’s immune tolerance mechanisms, and the physiological burden of pregnancy is another known risk factor. To understand the mechanisms of vertical transmission, defined as infection of the fetus from the maternal host, it is crucial to understand the unique structure and function that exists at the maternal-fetal interface. It may cause fetal infections which can disrupt organogenesis and is associated with congenital anomalies. The mechanism of pregnancy loss in the setting of TORCH infections can be pathogen or placenta mediated or through inflammation-induced delivery. Other infections may include Escherichia coli, varicella zoster virus, Zika virus, West Nile virus and human parvovirus B19. Parasitic infections may include Plasmodium falciparum and Trypanosoma cruzi which can cause intrauterine growth restriction, preterm delivery, respiratory and cardiovascular failure. The human placenta forms the primary barrier between the maternal and fetal compartments throughout pregnancy. The maternal-fetal interface consists of fetal-derived trophoblast progenitor cells and syncytiotrophoblasts.
The placenta begins to form 5–6 days following fertilisation. By 12 weeks of gestation, the maternal circulation has been remodelled through the formation of spiral arteries, and the placenta becomes haemochorial.13 Floating villi are formed by an inner layer of cytotrophoblasts (CTBs) that are covered by a layer of syncytiotrophoblasts which are bathed in maternal blood flowing into the intervillous space (IVS). The IVS contains 500 mL of maternal blood exposing the villous surfaces to microbes that might be present in maternal blood. The core of the chorionic villous consists of fetal macrophages, placental fibroblasts and fetal endothelial cells lining villous capillaries. Syncytiotrophoblasts provide potent protection against both viral and non-viral pathogens. CTBs also possess innate defence mechanisms against intracellular pathogens. The decidua basalis consists of 40% immune cells, 70% decidual natural killer cells, 25% macrophages and 10% T cells.14 It also functions in immunological tolerance of the semiallogeneic fetal origin trophoblasts, which lie in direct contact with the maternal cells. Humans have a villous placenta with only one layer of syncytiotrophoblasts bathed in maternal blood. Primary syncytiotrophoblasts confer broad antiviral resistance to non-trophoblast cells through effectors that operate in a paracrine manner. It is generally accepted that LM invasion of epithelial cells requires binding of host cell E-cadherin, cell-to-cell junction protein, located in the basolateral membrane of enterocytes. It is therefore inaccessible to bacteria in the intestinal lumen. The tropism of LM for extrusion sites is not exclusive to cultured cells but also occurs in vivo. Research focusing on rabbit intestine in vivo has shown that junctions at the villus tips are the sites of invasion.15 Consistent with the aforementioned observations, other studies have shown that LM adheres to and invades exclusively at the villus tips. Research involving gerbils, mice and rabbits has established that placental infection precedes fetal infection in early onset disease.16
Maternal infection usually presents with a plethora of diverse and non-specific symptoms such as fever, reduced fetal movements and symptoms of threatened preterm labour such as abdominal pain, vaginal bleeding or PPROM. Presenting symptoms may include gastrointestinal and influenza-like symptoms. The prospective MONALISA cohort study from France reported uterine contractions, labour or an abnormal fetal heart rate in 75% of cases and fetal loss in 21% of affected women.17 Fever is not always present and has been reported in 12%–85% of cases.11 Acute hepatitis is an unusual manifestation. Early onset LM is defined as a neonate with symptoms at birth or within 48 hours of birth resulting from in utero infection and is associated with higher morbidity and mortality. The term late onset is applied when a neonate develops symptoms >48 hours after birth and is commonly caused by infection during passage through the birth canal. It is the likely cause of meningitis and central nervous system sequelae. The human lethal dose of LM for fetal/neonatal loss is 1.9×106 colony-forming units.18 Neonatal listeriosis may occur by vertical transmission, by inhalation of infected amniotic fluid, transplacentally from the maternal circulation or by ascending colonisation from the vagina. Haematogenous spread can also occur. Cutaneous eruptions such as purpura, papules and petechiae, as in our case, are infrequent symptoms.12 Neonatal listeriosis commonly presents with septicaemia, respiratory distress or meningitis. Indeed, it is the third leading cause of neonatal bacterial meningitis. It can also present with granulomatosis infantiseptica, a pathognomonic feature of LM, first reported in 1893, manifesting with widespread microabscesses, miliary granulomas, areas of necrosis and liver involvement.19 The incidence of neonatal infection is 8/100 000 live births.20
Infected infants may have pustular lesions on their skin and pharynx. They may be hypothermic, lethargic and manifest with scattered salmon-coloured truncal papules.20
Laboratory investigations including a full blood count may reveal a leucocytosis. A serological test to detect the presence of antilisteriolysin O antibodies in blood can also be performed. Real-time PCR has a high sensitivity for early (<24 hours) detection of LM. Multiplex molecular panel also provides a rapid and sensitive tool for diagnosis. A ‘tumbling’ motion may be evident on gram stain or wet mount microscopy. It can also be confirmed by positive catalase and hippurate tests, as well as the presence of a narrow zone of beta-haemolysis on blood agar plates. LM grows well from blood or cerebrospinal fluid without the need for enrichment or selective media. The gold standard for diagnosis of maternal-fetal listeriosis is via placental cultures. Abscesses or necrosis in small blood vessels may occur in placental tissue. LM can be particularly challenging to isolate by culture, and blood cultures have a low diagnostic sensitivity of 55%.17 Maternal vaginal samples are usually negative, reflecting the haematogenous seeding of the placenta.17 Presently, stool culture is not recommended.
There are no uniform management guidelines, and recommendations are based primarily on small case series, in vitro susceptibility testing, animal-model experimentation or expert opinions. Penicillin, ampicillin and amoxicillin are the antimicrobial of choice as they can penetrate into the host cell, maintain a high cellular concentration and bind to the penicillin-binding protein-3. The current recommendation for first line therapy is intravenous amoxicillin >6 g/day for 2 weeks, except if the maternal central nervous system is involved where 4 weeks are recommended.18 A macrolide antibiotic is recommended for those women with a penicillin allergy. Although gentamicin has a synergistic effect with amoxicillin, it has no antilisteria intracellular efficacy in macrophages or placental tissue.21 22
Cephalosporin also has no effect on LM. In cases of a penicillin allergy, trimethoprim with sulfamethoxazole is a secondary line treatment. Caution is advised with the use of trimethoprim as it could damage the fetal heart and nervous system during early pregnancy due to folate antagonism. Trimethoprim may also cause haemolytic anaemia in male infants due to glucose-6-phosphate deficiency. Sulfonamides also pose the risk of a sulfur allergy or kernicterus. Erythromycin results in subtherapeutic concentrations in the amniotic fluid, and the dose should be increased. In cases of maternal sepsis with multiorgan involvement, termination of the pregnancy may be required to save the mother’s life. Furthermore, a new nanovaccine formulation, GNP (gold glyconanoparticle)-GAPDH (glyceraldehyde-3-phosphate dehydrogenase)1–22, that reverses all effects of listeriosis and confers LM-specific immunity has been proposed.23
In 2011, the Centre for Disease Control and Prevention published a recommended schedule for treating individuals facing an elevated risk of infection.1 Patient education on risk prevention should be incorporated into antenatal clinical practice. The gestational age at the time of infection has a fundamental effect on fetal prognosis. It has a case fatality rate of 20%–30% if the infection occurs during the second or third trimester of pregnancy.1 Only 5% of pregnancies with maternal LM have a favourable outcome.17 The MONALISA National Prospective Cohort Study showed that 82% faced adverse outcomes such as fetal loss or premature birth <32 weeks gestation.17
Although LM has been proven as an invaluable model micro-organism using mouse models, guinea pigs (Cavia porcellus), the Mongolian gerbil (Meriones unguiculatus) and non-human primates such as the rhesus macaques (Macaca mulatta) and cynomolgus macaques (Macaca fascilurais) to decipher the events associated with placental invasion by a pathogen, finding models that replicate the human-placental interface remains a challenge. In addition, it has enabled key discoveries in basic immunology and cell biology to be made. Although the use of mouse models has provided some insights, nevertheless, further research is required to delineate the virulence factors of LM replication in the placenta and its dissemination into fetal tissues. Although the placentae of primates and guinea pigs have a similar architecture to that of humans, their lack of genetic tractability limits some studies. Recently, the development of stem cell-derived organoid models of both maternal and fetal types at the maternal-fetal interface has opened up exciting new avenues to model this interface.24 25 Further studies underpinning the strategies used by pathogens to bypass these trophoblast-intrinsic defences are required to fully understand how and why LM is able to reach and invade the placenta so silently inducing adverse fetal outcomes. The advent of novel molecular assays may also help with the diagnostic challenge. Improved maternal surveillance and targeted health education during pregnancy are required to increase awareness of the risk factors for LM.
Learning points.
Listeria monocytogenes (LM) is a small gram-positive bacterium that is transmitted through contaminated food. Pregnant women are 18 times more likely to become infected after eating contaminated products. This is partly due to reduced T cell immunity and insufficient decidual T cells. Its incubation period is estimated at 3 weeks. LM exhibits a specific placental tropism that can also invade both extravillous trophoblast cells and syncytiotrophoblasts.
LM has a diverse and non-specific clinical presentation. Women may present with influenza-like symptoms, headache, myalgia, gastrointestinal upset or other digestive-related symptoms. Fever is present in 25% of cases. Mothers may report reduced fetal movements or symptoms of threatened preterm labour such as abdominal pain, vaginal bleeding or premature rupture of membranes. Maternal infection can lead to chorioamnionitis or meconium-stained amniotic fluid during delivery.
Infection due to LM is rare in neonates, and thus its clinical presentation and outcomes are not commonly reported. Granulomatosis infantiseptica is pathognomonic pustular skin lesions that are characteristic of severe neonatal infection. The major determinant of neonatal outcome is the gestational age at birth.
Early initiation of antibiotics such as penicillin, amoxicillin and ampicillin is recommended. A macrolide antibiotic is recommended for those women with a penicillin allergy. Erythromycin results in subtherapeutic doses in the amniotic fluid, and the dose should therefore be increased.
Acknowledgments
Mr Bruce MacMaster, Senior Biomedical Scientist, Department of Microbiology and Pathology. University Hospitals of North Midlands NHS Trust.
Footnotes
Contributors: LD: wrote the case report. MP: literature search. WS: pathology report and figures. GF: final editing and approval of the case report.
Funding: The authors have not declared a specific grant for this research from any funding agency in the public, commercial or not-for-profit sectors.
Case reports provide a valuable learning resource for the scientific community and can indicate areas of interest for future research. They should not be used in isolation to guide treatment choices or public health policy.
Competing interests: Yes.
Provenance and peer review: Not commissioned; externally peer reviewed.
Ethics statements
Patient consent for publication
Consent obtained directly from patient(s).
References
- 1.Gray ML, Killinger AH. Listeria monocytogenes and listeric infections. Bacteriol Rev 1966;30:309–82. 10.1128/br.30.2.309-382.1966 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Murray EGD, Webb RA, Swann MBR. A disease of rabbits characterised by a large mononuclear leucocytosis, caused by a hitherto undescribed bacillusBacterium monocytogenes (n.sp.). J Pathol Bacteriol 1926;29:407–39. 10.1002/path.1700290409 [DOI] [Google Scholar]
- 3.Hulphers G. Lefvernekos HOS kaninorsakad a fen EJ forut beskrifven bakteria. Svenska Vet Tidskr 1911;2:265–8. 10.1515/JPM.2011.035 [DOI] [Google Scholar]
- 4.Pirie JHH. A new disease of wild rodents, ‘tiger river disease’. Publications of the South African Institute for Medical Research 1927;3:163–86. [Google Scholar]
- 5.Nyfeldt A. Etiologie de la mononucleose infectieuse. Compt rend Soc Biol 1929;101:590–6. 10.1001/archpedi.1977.02120170017001 [DOI] [Google Scholar]
- 6.Gellin BG, Broome CV. Listeriosis. JAMA 1989;261:1313–20. 10.1001/jama.1989.03420090077035 [DOI] [PubMed] [Google Scholar]
- 7.Portnoy DA, Auerbuch V, Glomski IJ. The cell biology of listeria monocytogenes infection: the intersection of bacterial pathogenesis and cell-mediated immunity. J Cell Biol 2002;158:409–14. 10.1083/jcb.200205009 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Farber JM, Losos JZ. Listeria monocytogenes: a foodborne pathogen. CMAJ 1988;138:413–8. [PMC free article] [PubMed] [Google Scholar]
- 9.Pagliano P, Arslan F, Ascione T. Epidemiology and treatment of the commonest form of listeriosis: meningitis and bacteraemia. Infez Med 2017;25:210–6. [PubMed] [Google Scholar]
- 10.Silk BJ, Date KA, Jackson KA, et al. Invasive listeriosis in the foodborne diseases active surveillance network. food net. Further targeted prevention needed for higher-risk groups. Clin Infect Dis 2012;5:396–404. 10.1093/cid/cis268 [DOI] [PubMed] [Google Scholar]
- 11.Elinav H, Hershko-Klement A, Solt I, et al. Pregnancy-associated listeriosis: many beliefs, few facts. Lancet Infect Dis 2015;15:1128–30. 10.1016/S1473-3099(15)00302-3 [DOI] [PubMed] [Google Scholar]
- 12.Kelly CS, Gibson JL. Listeriosis as a cause of fetal wastage. Obstetrics & Gynecology 1972;40:91–7. 10.1097/00006250-197207000-00016 [DOI] [PubMed] [Google Scholar]
- 13.Arora N, Sadovsky Y, Dermody TS, et al. Microbial vertical transmission during human pregnancy. Cell Host Microbe 2017;21:561–7. 10.1016/j.chom.2017.04.007 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Manaster I, Mandelboim O, Mandelsohn O. The unique properties of uterine NK cells. Am J Reprod Immunol 2010;63:434–44. 10.1111/j.1600-0897.2009.00794.x [DOI] [PubMed] [Google Scholar]
- 15.Pentecost M, Otto G, Theriot JA, et al. Listeria monocytogenes invades the epithelial junctions at sites of cell extrusion. PLoS Pathog 2006;2:e3. 10.1371/journal.ppat.0020003 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Kaur S, Malik SVS, Vaidya VM, et al. Listeria monocytogenes in spontaneous abortions in humans and its detection by multiplex PCR. J Appl Microbiol 2007;103:1889–96. 10.1111/j.1365-2672.2007.03414.x [DOI] [PubMed] [Google Scholar]
- 17.Charlier C, Perrodeau Élodie, Leclercq A, et al. Clinical features and prognostic factors of listeriosis: the MONALISA national prospective cohort study. Lancet Infect Dis 2017;17:510–9. 10.1016/S1473-3099(16)30521-7 [DOI] [PubMed] [Google Scholar]
- 18.Roulo RM, Fishburn JD, Amosu M, et al. Dose response of Listeria monocytogenes invasion, fetal morbidity, and fetal mortality after oral challenge in pregnant and nonpregnant Mongolian gerbils. Infect Immun 2014;82:4834–41. 10.1128/IAI.01514-14 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Lee PJ, Krilov LR. Listeria. Pediatr Rev 2018;39:153–5. 10.1542/pir.2017-0062 [DOI] [PubMed] [Google Scholar]
- 20.Temple ME, Nahata MC. Treatment of listeriosis. Ann Pharmacother 2000;34:656–61. 10.1345/aph.19315 [DOI] [PubMed] [Google Scholar]
- 21.Carryn S, Van Bambeke F, Mingeot-Leclercq MP. Activity of beta-lactams (ampicillin, meropenem), gentamicin, azithromycin and moxifloxacin against intracellular listeria monocytogenes in a 24 H THP-1 human macrophage model. J Antimicrob Chemother 2003;51:1051–2. 10.1093/jac/dkg189 [DOI] [PubMed] [Google Scholar]
- 22.Bakardjiev AI, Stacy BA, Portnoy DA. Growth of listeria monocytogenes in the guinea pig placenta and role of cell-to-cell spread in fetal infection. J Infect Dis 2005;191:1889–97. 10.1086/430090 [DOI] [PubMed] [Google Scholar]
- 23.Calderon-Gonzalez R, Frande-Cabanes E, Teran-Navarro H, et al. GNP-GAPDH1-22 nanovaccines prevent neonatal listeriosis by blocking microglial apoptosis and bacterial dissemination. Oncotarget 2017;8:53916–34. 10.18632/oncotarget.19405 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Imanishi M, Routh JA, Klaber M, et al. Estimating the attack rate of pregnancy-associated listeriosis during a large outbreak. Infect Dis Obstet Gynecol 2015;2015:1–5. 10.1155/2015/201479 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Turco MY, Gardner L, Kay RG, et al. Trophoblast organoids as a model for maternal–fetal interactions during human placentation. Nature 2018;564:263–7. 10.1038/s41586-018-0753-3 [DOI] [PMC free article] [PubMed] [Google Scholar]