Abstract
Introduction
Malaria is associated with high morbidity during pregnancy. Homozygous sickle cell pregnant women are even more exposed during complicated malaria. The objective of the study was to evaluate the maternal and fetal morbidity of homozygous sickle cell pregnant pregnant women with complicated malaria.
Methods
We conducted a retrospective case-control study of 982 pregnancies in sickle cell pregnant women, during which a group of sickle cell pregnant women who received antimalarial chemoprophylaxis was compared to another group without chemoprophylaxis. We analyzed the clinical evolution of pregnant women (VOCs and transfusions, pregnancy weight gain) and parasite (parasite density at the time of diagnosis of complicated malaria and during treatment for three days). We analyzed the parameters of newborns at birth (age of pregnancy at the time of delivery, birth weight, weight of the placenta and histopathological examination of the placenta.
Results
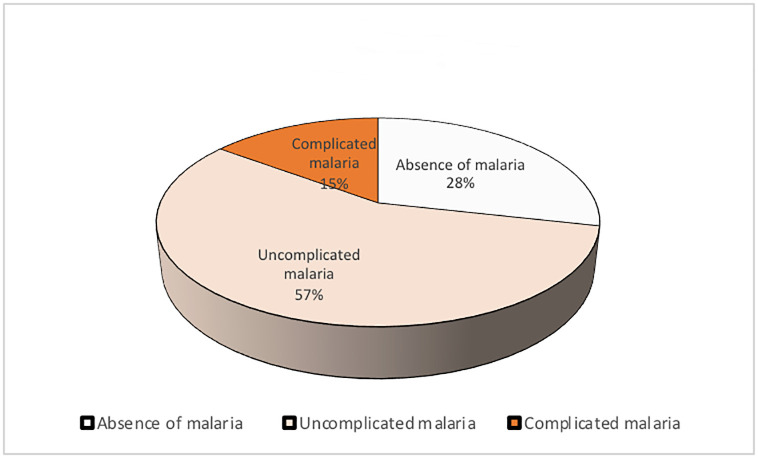
Out of 982 pregnancies, 15% of pregnant women suffered from complicated malaria, 57% suffered from uncomplicated malaria and 28% did not suffer from malaria. Pregnancy weight gain, birth weight, was better in the group of pregnant women who received chemoprophylaxis and the placenta had less histological lesions. Parasite density was low. There was a significant positive correlation between parasite density and the number of CVOs and transfusions and between parasite density and histological lesions of the placenta and low birth weight.
Conclusion
Complicated malaria is associated with high maternal and fetal morbidity in sickle cell patients. Malaria chemoprophylaxis can reduce maternal and fetal complications and parasite density during malaria infection.
Introduction
Malaria is the most widespread parasitic disease in the world in general and in the African intertropical zone in particular [1]. In Central Africa, malaria is a public health problem [2, 3].
Indeed, according to the WHO, the number of malaria cases in 2020 was estimated at 241 million, an increase of 14 million new cases compared to 2019 [4, 5]. During the year 2020, the number of deaths due to malaria was estimated at 62,700, an increase of 6,900 deaths compared to the previous year. Half of the deaths due to malaria during the year 2020 were observed in six countries including the DRC [4, 5]. Children under 5 years and pregnant women are the most vulnerable. In 2020, the mortality of children under five years was 272,000, 94% of which in Africa. Eleven percent of this mortality of children under five years old was observed in the DRC. Malaria is transmitted by the bite of an infected mosquito of the Anopheles genus and more rarely during a blood transfusion or by mother-to-child transmission during pregnancy [6, 7]. Plasmodium falciparum is responsible for complicated malaria [8]. It is responsible for half of malaria-related deaths worldwide. The other species including p. vivax, p. oval, p. malariae and p. knowlesi are not associated with complicated forms of malaria. Since October 2021, WHO recommends the use of the RTS, S/AS01 vaccine against malaria in all areas with a high endemicity of P. falciparum malaria. Malaria control strategies include vector control [9, 10]. This is why the use of impregnated mosquito nets is now recommended by the WHO in endemic areas with a high prevalence of malaria. In the DRC, impregnated mosquito nets are distributed free of charge to pregnant women during prenatal consultations. Antimalarial prophylaxis during pregnancy aims to reduce the risk of complicated malaria during pregnancy [11]. Indeed, malaria is recognized in the tropics as being one of the causes of early abortions, prematurity, fetal death in utero and low birth weight [6, 7, 11–14]. Sickle cell disease (SCD) is a constitutional hemoglobinopathy, with autosomal recessive transmission. It is characterized by the E6V mutation of the beta globin gene, in which a purine base [A] is replaced by a pyrimidine base [T]: A>T. This transversion of the purine base by a pyrimidine base results in the substitution of glutamic acid by valine at position 6 of the beta globin chain [15, 16]. The substitution of a hydrophilic amino acid (glutamic acid) by a hydrophobic amino acid (valine), leads to the synthesis of an abnormal hemoglobin called HbS [17]. Indeed, in concentrated solution and under the influence of a drop in oxygen pressure, HbS undergoes a supramolecular process of polymerization [16, 18]. The HbS mutation is today the most common structural anomaly of hemoglobin in the world [19]. Equatorial Africa is the area with the highest prevalence of SCD and the most severe form in the world. Geographically, the distribution of HbS overlaps with that of malaria [10, 20]. Genetically, there are homozygous subjects (SS) and heterozygous subjects (AS). Homozygotes are the sick, while heterozygotes are carriers of the sickle cell trait. Heterozygous is thought to protect against complicated forms of malaria [10, 20–22]. However, homozygotes are very exposed to complicated forms of malaria [10, 20, 21]. Clinically SCD is characterized by recurrent vaso-occlusive ischemic crisis (VOC), chronic hemolysis and a high susceptibility to infections [23]. When the partial pressure of oxygen drops (hypoxia) and when associated with other unfavorable conditions such as increased temperature, acidosis, HbS crystallizes and causes deformation of the red blood cell [23]. In Africa, the DRC is the second country most affected by SCD and malaria after Nigeria.
The objective of the present study is to evaluate the fetal maternal mobility of complicated malaria in sickle cell pregnant women.
Methods
This is a retrospective case-control study that was conducted at sickle cell center of Kinshasa (CMMASS) between 2011 and 2020. The study included 982 sickle cell pregnant women. The sickle cell center of Kinshasa (CMMASS) is a center specializing in the care of sickle cell patients. It is a public center and it has the particularity of receiving many pregnant women with SCD.
Inclusion criteria
In the present study we included known sickle cell pregnant women with a confirmatory molecular diagnosis of SCD. You must have had complicated malaria during pregnancy. The diagnosis of pregnancy had to be made and confirmed by ultrasound in the first trimester of pregnancy between the 7th and 10th week of amenorrhea. The pregnant woman had to have regularly followed the prenatal consultations at the sickle cell center and had given birth in this center.
Exclusion criteria
We excluded any pregnant woman who developed pre-eclampsia during pregnancy.
Study groups
For 15 years, we have observed that many sickle cell pregnant women who receive free impregnated mosquito nets during prenatal consultations, do not accept preventive antimalarial chemoprophylaxis during pregnancy. Thus in the present study, we have two groups of pregnant women with SCD who presented the complicated malaria during pregnancy. The first group (G1) consisted of sickle cell pregnant women who had accepted preventive antimalarial chemoprophylaxis during pregnancy. The second group (G2) consisted of sickle cell pregnant women who did not accept antimalarial chemoprophylaxis during pregnancy.
Operational definitions
Uncomplicated malaria: in our study we defined uncomplicated malaria as any case of malaria with a parasite density < 100 trophozoites per microliter of blood, fever < 38°C and without any threat to vital functions.
Complicated malaria has been defined on the basis of criteria defined by the WHO, among which there is hyperparasitaemia [24, 25].
Weight gain during pregnancy (ΔP): was calculated by the difference between the weight of the pregnant woman at the time of delivery (Pf) and the weight at the start of pregnancy (Pi): (ΔP = Pf − Pi).
Low birth weight (LBW): we considered the low birth weight any newborn born at term (37 weeks) with a weight below the 10th percentile (10% of birth weight) i.e. overall a weight below 2500 g.
Preventive chemoprophylaxis is the use of drugs or drug combinations to prevent malaria infection and its consequences. It includes chemoprophylaxis, intermittent preventive treatment for pregnant women.
Preventive chemo prophylaxis
In our study we used the association Arthemeter and Lumefantrine as a molecule for preventive chemoprophylaxis. Arthemeter was dosed at 80 mg and Lumefantrine at 480 mg.
Management pregnant women during pregnancy
During the pregnancy, we had given two doses of chemoprophylaxis to group 1 at the 14th week and at the 28th week. All pregnant women in both groups received regular supplementation with omega 3, magnesium pidolat and folic acid. Any case of complicated malaria during pregnancy was treated with the artesunat administered parenterally. The dose administered was 2.4 mg/Kg on arrival, then successively at the 12th hour, 24th hour, 48th hour and finally at the 72nd hour. After 72 hours, the treatment was relayed orally with the association Arthemeter dosed at 80 mg and lumefantrine dosed at 480 mg for 3 days.
Laboratory analyzes
Hemoglobin assay and thick smear analysis
The determination of hemoglobin and the analysis of the thick film were carried out at each appointment of the prenatal consultation.
The diagnosis of malaria
The diagnosis of malaria was made on the basis of two tests. The rapid test (RDT) using the immunochromatography technique and the microscopic diagnostic test for malaria. For the microscopic diagnosis of malaria, we had carried out the thin smear and the thick drop. The thin smear was intended to make the diagnosis of the plasmodium species, and the thick smear allowed us to determine the density of the parasites per microliter of blood according to WHO recommendations [24, 25].
Histopathological analysis of the placenta
The placentas of newborns of mothers who had complicated malaria during pregnancy were analyzed in the anatomopathological department of the University Hospital of Kinshasa. Indeed, after delivery, the placenta was analyzed macroscopically in search of any morphological abnormality.
The umbilical cord was cut at the base. Then we weighed the placenta after cutting the membranes of the placenta. We had taken three samples of the placenta from three different sites: at the base of the cord, from the middle zone of the placenta and from the marginal zone. The biopsy specimens were fixed in 10% buffered formalin and then sent to the pathological anatomy laboratory of the University Hospital of Kinshasa. According to the types of lesions observed, we have classified the placentas into five types as defined below.
Placenta type 1: represents placentas that had no lesions after histopathological analysis.
Placenta type 2: these are placentas that had multiple areas of calcification, infarction and fibrin deposition.
Placenta type 3: these are placentas had lesions of thrombosis of chorio allantoic vessels and chorioangiosis.
Type 4 placenta: these are placentas that had type 3 lesions with ischemic necrosis lesions with deposition of fibrinoid substances.
Placenta type 5: these are placentas that had type 4 lesions with chronic villitis and the presence of parasites: trophozoites.
Variables of interest
In the present study, the variables of interest were: the age of the pregnant woman, the parity, the hemoglobin level, the gravidic weight gain, the weight of the newborn, the age of pregnancy at delivery, placental weight and histopathological lesions of the placenta.
Ethical approval
The study was approved by the ethical committee of the school of public health of the University of Kinshasa (Approval reference: ESP/CE/079/2016), DRC. Informed consent was obtained from all patients before their inclusion in the study. The informed consent obtained was written and our study did not include minors.
Statistical analyzes
Our data was processed with SPSS software. We calculated the frequencies and the means and standard deviations. Our averages were compared using the Student t test. The value of p<0.05 was considered as the level of significance.
Results
Our study included 982 mothers with SCD, among whom 556 (57%) had presented uncomplicated malaria, 146 (15%) had presented complicated malaria and 280 (28%) had not presented malaria during pregnancy. Fig 1 below shows the distribution of malaria in our study population.
Fig 1. Distribution of malaria during the pregnancy in study population.
Table 1 above presents the characteristics of our pregnant women. We observe that there was no difference in age and parity between the mothers of the two groups. However, the difference between the mean Hb level during pregnancy and the gravidic weight gain between the groups was highly significant (p<0.001) in favor of group 1.
Table 1. Periods and therapeutic doses of artemether during complicated malaria.
| Characteristics | Group 1 (n = 62) | Group 2 (n = 85) | p |
|---|---|---|---|
| Age (years) | 23.60 ± 4.00 | 24.03 ± 3.75 | 0.498 |
| Parity (n) | 1.40 ± 0.52 | 1.30 ± 0.46 | 0.199 |
| Hb (g/dl) | 7.43 ± 1.77 | 5.95 ± 0.98 | <0.001 |
| Gravidic weight gain (Kg) | 5.48 ± 1.11 | 3.19 ± 0.52 | <0.001 |
Group 1: is the study group i.e. pregnant women who had agreed to receive chemoprophylaxis during pregnancy.
Group 2: is the group of cases i.e. pregnant women who did not accept chemoprophylaxis during pregnancy.
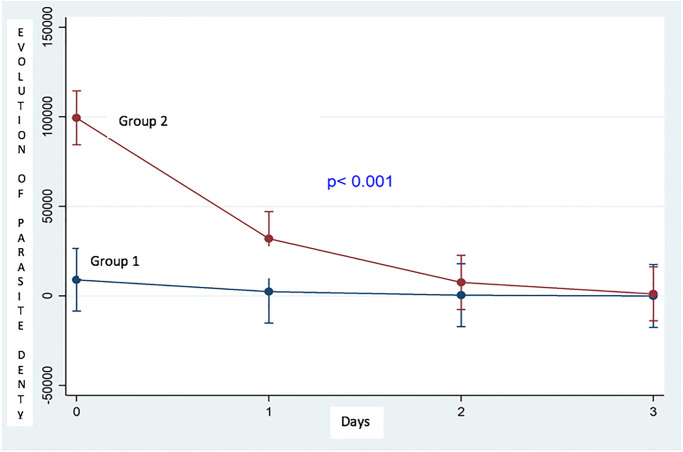
Fig 2 above shows the evolution of the parasite density during the treatment for 3 days. It appears that from the first day to the third the parasite densities of the pregnant women of group 2 were very high compared to the pregnant women of group 1. The statistical differences were significant (p<0.001).
Fig 2. Evolution of parasite density during treatment in the study population.
Table 2 above shows the characteristics of the newborns and the average weight of the placentas. We observe that the mean age at childbirth was similar in the two groups. However, fetal weight, placental weight and frequency of low birth weight were different between the two groups and the differences were statistically significant (p<0.001).
Table 2. Characteristics of newborns and placentas.
| characteristics of newborns and placentas | Group 1 (n = 62) | Group 2 (n = 85) | p |
|---|---|---|---|
| Age at childbirth (weeks) | 37.41 ± 0.50 | 37.56 ± 0.52 | 0.042 |
| Fetal weight (gr) | 2721.55± 202.51 | 2503.69 ± 175.66 | <0.001 |
| Placenta weight (gr) | 612.09 ± 148.02 | 438.33 ± 145.27 | <0.001 |
| LBW (%) | 12.90±1.09 | 44.05±1.99 | <0.001 |
LBW: low birth weight
Table 3 shows the frequencies of the types of histopathological lesions observed in the placentas. It appears that the frequency of type 1 placenta (placenta without histological lesion) was higher in group 1 than in group 2. The statistical difference was significant (p<0.001). However, the frequency of placenta type 5 (placenta with ischemic necrosis lesions with deposition of fibrinoid substances, with chronic villitis and presence of trophozoites) was higher in group 2, the statistical difference was also significant (p<0.001).
Table 3. Frequencies of placental lesions.
| type of histopathological lesions of the placenta | Group 1 (n = 62) | Group 2 (n = 85) | p |
|---|---|---|---|
| Type 1 (%) | 59.68 | 23.81 | <0.001 |
| Type 2 (%) | 24.19 | 19.05 | 0.452 |
| Type 3 (%) | 3.23 | 14.29 | 0.024 |
| Type 4 (%) | 3.23 | 3.57 | 0.909 |
| Type 5 (%) | 9.68 | 39.29 | <0.001 |
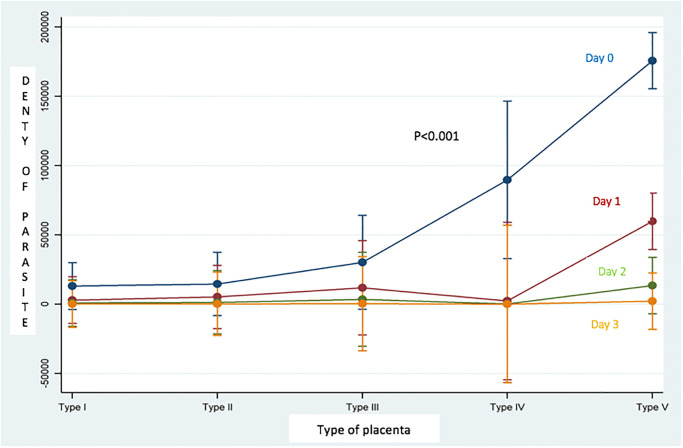
Fig 3 shows the correlation between parasite density and the risk of histological damage to the placenta in our cohort. It appears that there is a positive correlation between the high parasite density and the type 5 placenta (placenta with ischemic necrosis lesions with deposition of fibrinoid substances, with chronic villitis and presence of trophozoites). Conversely, we observe a positive correlation between the low parasite densities with the type 1 placenta (without histological lesion).
Fig 3. Correlation between parasite density and type of placental lesion.
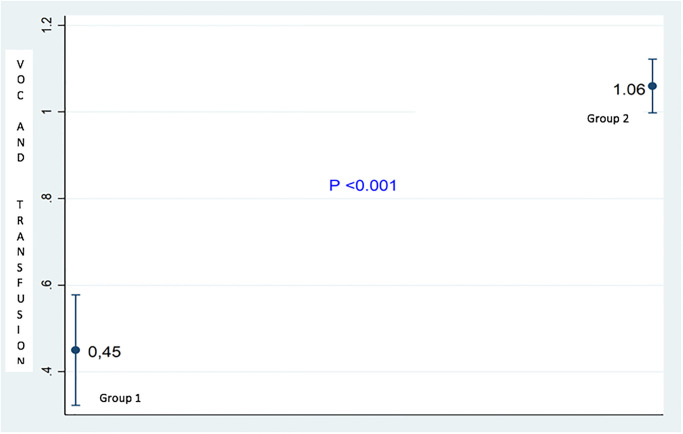
Fig 4 shows the frequency of VOCs and transfusions during the malaria attack in the two groups. It appears that during the crisis of complicated malaria, the number of VOCs and transfusions were higher in group 2 than in group 1. The statistical difference was highly significant (p<0.001).
Fig 4. Frequencies of VOCs and transfusions during the crisis of complicated malaria.
Discussion
Malaria is a parasitic disease that is associated with great morbidity during pregnancy and in children under five years of age [9, 10, 26–28]. In homozygous sickle cell pregnant women, malaria is responsible for complicated VOCs [29–31] and frequent haemolysis [29–32] which compromises the vital prognosis of the patients [30–32]. During pregnancy, complicated malaria is associated with a high risk of LBW [29–33]. The objective of antimalarial chemoprophylaxis is to prevent malaria infection and its consequences. Our study aimed to evaluate the maternal and fetal morbidity of complicated malaria in sickle cell pregnant women. Our study showed that complicated malaria is frequent during pregnancy. Indeed, 15% of pregnant women in our series suffered from complicated malaria during pregnancy. We have observed that chemoprophylaxis reduces parasite density in complicated malaria. This observation proves the protection of antimalarial chemoprophylaxis during pregnancy. In general, pregnant women with SCD have low weight gain during pregnancy and their newborns have LBW [29–33]. Our study showed that complicated malaria increases the risk of LBW. Indeed, in our series, pregnancy weight gain and birth weight were lower in pregnant women who had not received preventive chemoprophylaxis against malaria. LBW has an impact on the weight of the placenta. Our results showed that low weight placentas with high parasite densities were associated with greater histopathological lesions. The histological lesions encountered in our study (ischemic necrosis and vessel thrombosis) could be directly associated with poor quality exchanges between the mother and the fetus. The disruption of exchanges between the mother and the fetus explains the LBW. Indeed, we observed a positive correlation between the types of placental lesions, parasite density and low birth weight. The etiologies of the histological lesions of the placenta can be the frequency and the severity of the VOCs which increase during the complicated malaria such as disassemble in our study, but also the vasoocclusion of the vessels of the placenta can involve the lesions of ischemic necrosis and thrombosis. vessels. Conversely, the inflammatory phenomenon secondary to vasoocclusion explains inflammatory lesions of the placenta such as chronic villitis. Our study showed that complicated malaria is associated with frequent VOCs and a risk of worsening anemia leading to multiple transfusions during pregnancy.
Conclusion
Complicated malaria is associated with high maternal and fetal morbidity in sickle cell patients. Malaria chemoprophylaxis can reduce both the parasite density during malaria infection but also reduces the risk of maternal and fetal complications. The risk of low birth weight is associated with damage to the placenta.
Supporting information
(XLSX)
Acknowledgments
The authors would like to thank the biologist Jonas KAPAY and his team for the quality of the laboratory analyses. thanks also to the midwife Mamie BAYA and her team for the quality of follow-up of pregnant women during this study. Finally, thanks to all the pregnant women who agreed to participate in this study.
Data Availability
All relevant data are within the paper and its Supporting information files.
Funding Statement
The authors received no specific funding for this work.
References
- 1.Newton PN, Fernández FM, Plançon A, Mildenhall DC, Green MD, Ziyong L, et al. A collaborative epidemiological investigation into the criminal fake artesunate trade in South East Asia. PLoS Med. 2008;5(2):e32. doi: 10.1371/journal.pmed.0050032 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.World health Organization. Severe Falciparum malaria. Trans R Soc Trop Med Hyg. 2000;94 (suppl 1):1–90. PubMed | Google Scholar. [PubMed] [Google Scholar]
- 3.Snow RW, Sartorius B, Kyalo D, Maina J, Amratia P, Mundia CW, et al. The prevalence of Plasmodium falciparum in sub-Saharan Africa since 1900 Nature. 2017. Oct 26;550(7677):515–518. Epub 2017 Oct 11. doi: 10.1038/nature24059 . [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.WHO. World malaria report 2020: 20 years of global progress and challenges. Geneva: World Health Organization; 2020. Licence: CC BY-NC-SA 3.0 IGO; 2020.
- 5.World Malaria Report 2021, Key Messages, WHO/UCN/GMP/2021.08.
- 6.McClure EM, Goldenberg RL, Dent AE, Meshnick SR. A systematic review of the impact of malaria prevention in pregnancy on low birth weight and maternal anemia. Int J Gynaecol Obstet. 2013;121:103–9. doi: 10.1016/j.ijgo.2012.12.014 [DOI] [PubMed] [Google Scholar]
- 7.Blencowe H, Krasevec J, de Onis M, Black RE, An X, Stevens GA, et al. National, regional, and worldwide estimates of low birthweight in 2015, with trends from 2000: a systematic analysis. Lancet Glob Health. 2019;7:e849–60. doi: 10.1016/S2214-109X(18)30565-5 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Rowe JA, Opi DH, Williams TN. Blood groups and malaria: fresh insights into pathogenesis and identification of targets for intervention. Curr Opin Hematol. 2009;16(6):480–7. doi: 10.1097/MOH.0b013e3283313de0 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Bakken Line and Iversen Per Ole: The impact of malaria during pregnancy on low birth weight in East-Africa: a topical review. Bakken and Iversen Malar J (2021) 20:348 doi: 10.1186/s12936-021-03883-z [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Fried Michal and Patrick E. Duffy: malaria during pregnancy. Cold Spring Harb Perspect Med 2017;7:a025551. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.WHO. Intermittent preventive treatment in pregnancy (IPTp). Geneva: World Health Organization. https://www.who.int/malaria/areas/preve ntive_therapies/pregnancy/en/. Accessed 29 May 2021.
- 12.McClure EM, Goldenberg RL, Dent AE, Meshnick SR. A systematic review of the impact of malaria prevention in pregnancy on low birth weight and maternal anemia. Int J Gynaecol Obstet. 2013;121:103–9. doi: 10.1016/j.ijgo.2012.12.014 [DOI] [PubMed] [Google Scholar]
- 13.Blencowe H, Krasevec J, de Onis M, Black RE, An X, Stevens GA, et al. National, regional, and worldwide estimates of low birthweight in 2015, with trends from 2000: a systematic analysis. Lancet Glob Health. 2019;7:e849–60. doi: 10.1016/S2214-109X(18)30565-5 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Eisele TP, Larsen DA, Anglewicz PA, Keating J, Yukich J, Bennett A, et al. Malaria prevention in pregnancy, birthweight, and neonatal mortality: a meta-analysis of 32 national cross-sectional datasets in Africa. Lancet Infect Dis. 2012;12:942–9. doi: 10.1016/S1473-3099(12)70222-0 [DOI] [PubMed] [Google Scholar]
- 15.Ingram VM. 1956. A specific chemical difference between the globins of normal human and sicklecell anaemia haemoglobin. Nature 178:792–94 [doi: 10.1038/178792a0 ]. [DOI] [PubMed] [Google Scholar]
- 16.Ingram VM. 1957. Gene mutations in human haemoglobin: the chemical difference between normal and sickle cell haemoglobin. Nature 180:326–28 [doi: 10.1038/180326a0 ]. [DOI] [PubMed] [Google Scholar]
- 17.Serjeant GR, Vichinsky E. Variability of homozygous sickle cell disease: the role of alpha and beta globin chain variation and other factors. Blood Cells Mol Dis. 2018;70:66–77. doi: 10.1016/j.bcmd.2017.06.004 [DOI] [PubMed] [Google Scholar]
- 18.Perutz MF, Mitchison JN. State of hemoglobin in sickle cell anemia. Nature, 1950; 166: 677–679. doi: 10.1038/166677a0 [DOI] [PubMed] [Google Scholar]
- 19.Rees DC, Williams TN, Gladwin MT. Sickle-cell disease. Lancet 2010;376:2018–31. doi: 10.1016/S0140-6736(10)61029-X [DOI] [PubMed] [Google Scholar]
- 20.Piel FB, Patil AP, Howes RE, Nyangiri OA, Gething PW, Williams TN, et al. Global distribution of the sickle cell gene and geographical confirmation of the malaria hypothesis. Nat Commun. 2010;1(1):104. doi: 10.1038/ncomms1104 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Aluoch JR. The presence of sickle cells in the peripheral blood film. Specificity and sensitivity of diagnosis of homozygous sickle cell disease in Kenya. Trop Geogr Med. 1995;47(2):89–91. [PubMed] [Google Scholar]
- 22.Kosiyo Paul, Otieno Walter, Gitaka Jesse, Munde Elly O. and Collins Ouma: Haematological abnormalities in children with sickle cell disease and non-severe malaria infection in western Kenya. Infectious Diseases (2021) 21:329 doi: 10.1186/s12879-021-06025-7 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Sundd Prithu, Gladwin Mark T., Novelli Enrico M.: Pathophysiology of Sickle Cell Disease, Annu Rev Pathol. 2019. January 24; 14: 263–292. doi: 10.1146/annurev-pathmechdis-012418-012838 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.WHO Basic malaria microscopy– 2nd edition. Part 1: Learner’s guide. ISBN 978 92 4 154782 6.
- 25.World health Organization. Severe Falciparum malaria. Trans R Soc Trop Med Hyg. 2000;94 (suppl 1):1–90. PubMed | Google Scholar. [PubMed] [Google Scholar]
- 26.WHO. World malaria report 2020. Geneva: World Health Organiza- tion; 2020. https://www.who.int/publications/i/item/9789240015791. Accessed 29 May 2021.
- 27.Wastnedge E, Patel S, Goh MY, Rudan I. The global burden of sickle cell disease in children under ve years of age: a systematic review and meta- analysis. J Glob Health. 2018;8:021103. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Piel FB, Steinberg MH, Rees DC. Sickle cell disease. N Engl J Med. 2017;76:1561–73. [DOI] [PubMed] [Google Scholar]
- 29.Jain D, Atmapoojya P, Colah R, Lodha P. Sickle cell desaese and pregnany. Mediterr J Hematol Infect Dis 2019. Jul 1;11(1):e2019040. eCollection 2019. doi: 10.4084/MJHID.2019.040 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Boga C, Ozdogu H. Pregnancy and sickle cell disease: A review of the current literature. Crit Rev Oncol Hematol 2016. Feb;98:364–74. doi: 10.1016/j.critrevonc.2015.11.018 [DOI] [PubMed] [Google Scholar]
- 31.Howard J, Oteng-Ntim E. The obstetric management of sickle cell disease. Best Pract Res Clin Obstet Gynaecol 2012. Feb;26(1):25–36. Epub 2011 Nov 22. doi: 10.1016/j.bpobgyn.2011.10.001 [DOI] [PubMed] [Google Scholar]
- 32.Rogers TD, Molokie R. Sickle cell disease and pregnancy. Obstet Gynecol Clin North Am 2010. Jun;37(2):223–37. doi: 10.1016/j.ogc.2010.02.015 [DOI] [PubMed] [Google Scholar]
- 33.Ngô C, Kayem G, Habibi A, Benachi A, Goffinet F, Galactéros F, et al. Pregnancy in sickle cell disease: maternal and fetal outcomes in a population receiving prophylactic partial exchange transfusions. Eur J Obstet Gynecol Reprod Biol. 2010. Oct;152(2):138–42. Epub 2010 Sep 16. doi: 10.1016/j.ejogrb.2010.05.022 . [DOI] [PubMed] [Google Scholar]