Abstract
In this study, Plasmodium falciparum was detected in patients that were declared negative for malaria microscopy and rapid diagnostic test kit (mRDT), using Plasmodium 18s rRNA loop-mediated isothermal amplification (LAMP) technique. The main aim of this study was to assess the usefulness of LAMP assay for detecting pre-clinical malaria, when microscopy and mRDT were less sensitive. DNA was obtained from 100 μL of whole blood using the boil and spin method. Subsequently, the Plasmodium 18s rRNA LAMP assay was performed to amplify the specific Plasmodium 18s rRNA gene. Microscopy and mRDT negative samples [697/2223 (31.2%)] were used for this study. Compared to frequencies obtained for the other demographic variables, most of the patients were < 6 years (37.7%), females (59.0%), peri-urban dwellers (39.0%) and patients that sought outpatient department services (39.3%). Overall, the prevalence of Plasmodium 18s rRNA was 17.5%. when stratified by study variables, Plasmodium 18s rRNA LAMP positivity was higher in patients over 30 years [58/122 (54.2%)], males [69/122 (56.5%)], rural dwellers [69/122 (56.5%)] and patients that sought OPD services [68/122 (55.7%)]. The risk of being infected with Plasmodium when routine tests were negative was higher in 15–30-year group (OR = 3.03, 95% CI: 1.6–5.8, p = 0.0007), patients > 30 years (OR = 15.2, 95% CI: 8.3–27.7, p<0.001), males (OR = 2.1, 95% CI: 1.4–3.2, p = 0.0002) and rural dwellers (OR = 2.2, 95% CI:1.4–3.6, p = 0.0009). However, risk was lower in post-natal children (OR = 0.3, 95% CI: 0.18–0.51, p<0.001). Majority (81.5%) of the infected patients presented with headache, herpes labialis, diarrhea and vomiting. We demonstrated the lack of sensitivities of microscopy and mRDT for one-time diagnosis of malaria. Therefore, it is essential to utilize a sensitive technique such as Plasmodium 18s rRNA LAMP to increase the detection rate of Plasmodium infection.
Introduction
Sub-Saharan Africans are still at risk of malaria [1]. Because of that, prompt diagnosis of malaria is essential especially in children under 5 years and in pregnant women, who are at increased risk of the disease. Until malaria rapid diagnostic test (mRDT) kits were introduced in the early 1990s [2], microscopy was the mainstay. However, malaria microscopy has been found to be limited in its sensitivity to detect malaria parasites [3].
Malaria microscopy is reported to be less sensitive to parasitemia, less than 250 parasites per microliter of blood [4]. Additionally, long turnaround times, inadequate trained microscopists, lack of quality control programs, and standardized staining reagents are some of the demerits of the malaria microscopy technique [3–5]. MRDTs were manufactured to cirmumvernt the challenges of microcopy [6]. MRDTs detect various parasite antigens, namely, Plasmodium lactate dehydrogenase, Plasmodium histidine-rich proteins 2 (PfHRP2) and Plasmodium aldolase [7]. However, kits based on PfHRP2 are very common [8]. That nothswithstanding, PfHRP2 based kits are being rendered useless due to the increasing frequency of Plasmodium falciparum with mutations that make them incapable to produce the HRP 2 protein, the main marker the majority brands of mRDTs are manufactured to detect [6, 9, 10]. In Ghana [11–13] and elsewhere [11, 14, 15], various frequencies of P. falciparum with HRP2 gene deletions have been recorded, which result in false negative mRDT testing based on HRP2.
To overcome these challenges associated with malaria diagnosis, molecular testing based on nucleic acid amplification technology (NAAT) has been evaluated in several studies. Conventional polymerase chain reaction (PCR) has been the most evaluated nucleic acid amplification based technique for the detection of the P. falciparum parasite [12, 13]. In recent times, variants of PCR have been used to detect the parasite [16]. Notwithstanding the foregoing, the PCR technology is sophisticated, providing results with delay, reserved for specialized laboratories, requires skilled labor, and reactions are prone to contamination [17]. To surmount the demerits of PCR and its variants, a novel NAAT, called loop-mediated isothermal amplification (LAMP) was developed in the year 2000 by Notomi and his colleagues [18].
Since then, the LAMP assay has been used to identify several gene targets. In a literature review by Wong et al. (2018), hundreds of microorganisms have been detected by the LAMP assay [19]. LAMP assay is gaining prominence due to its rapidity, user-friendliness, high sensitivity and specificity, stability of the Bst DNA polymerase to PCR inhibitors, and amenability of the technique to outside laboratory conditions [16–20]. Additionally, reaction outcomes can be judged visually and the use of simple heating devices such as water baths and heating blocks have been employed to reduce the testing costs [21–23]. In Ghana, the LAMP assay has been widely used to detect several pathogens, namely, P. falciparum [24], Mycobacterium tuberculosis [25] and M. ulcerans [26].
In this study, P. falciparum was detected in patients that were declared negative for malaria microscopy and mRDT, using Plasmodium 18s rRNA LAMP. This study is essential because a previous study detected malaria parasites in about 12% of patients that were initially tested negative for malaria with repeated sampling [14]. Therefore, early detection of the Plasmodium parasite in patients presenting with classical signs of malaria at baseline is essential for prompt management.
Materials and methods
Study design
This study was a cross-sectional study designed to detect P. falciparum in patients previously tested negative for malaria by microscopy and mRDT.
Study sites, study population, and study duration
This study was carried out in Koforidua Polyclinic, a public health facility in the New Juaben South Municipality in the Eastern Region of Ghana. The facility is the first point of contact in the municipality for primary health care needs. The facility operates an outpatient department, laboratory unit, 24-hours detention units, antenatal services, labour and recovery wards, Eye unit, Ear, Nose and Throat unit and other essential service deliver points. Patients included in this study were those tested negative for malaria by both microscopy and mRDT. Microscopy and mRDT were done by Ghana Health Service certified malaria microscopists and trained biomedical scientist certified to perform mRDT through the On-site Training and Supportive Supervision (OTSS) programs. Samples were collected for this study from February—December 2019. Individuals recruited for this study were systematically selected, with prior written consent. Further, written consent was sought from parents or guardians of the minors prior to enrolling into this study.
Inclusion and exclusion criteria
Patients included in this study were suspected of malaria but was initially tested negative for malaria parasites using microscopy and mRDT. To be included in the study, one or more of these clinical histories must be confirmed to be present by the attending clinicians, history of vomiting, presence of fever, chills, diarrhea, anemia, skin rashes, headache, nausea, mouth and lip sores, hematuria, and dizziness. Patients previously treated for malaria (inquired on direct questioning) on review visits were excluded from this study.
Clinical assessment
A pediatrician, an obstetric and gynecologist, and a physician specialist examined each patient for overt and covert signs of malaria.
Laboratory procedures
Sample collection
At least 4 mL of whole blood was collected from each patient suspected of malaria. Within 24 hours of sample collection, malaria microscopy [27] and mRDT [15] were done as previously described. Malaria negative concordant samples were further tested for Plasmodium 18s rRNA gene.
Extraction of Plasmodium DNA
Equal volume (100 μL) of whole blood was mixed with an extraction buffer (400 mM NaCl, 40 mM Tris pH 6.5, 0.4% SDS). The mixture was briefly vortexed and subsequently boiled for 5 minutes at 95°C. The haemolysate was spun at 10,000 rpm for 3 minutes. The supernatant was frozen at -30°C before the LAMP assay.
Detection of Plasmodium 18s rRNA gene by LAMP
The amplification of the Plasmodium 18s rRNA gene was done in pools of ten comprising 50μL of pooled DNA and master mix components made up of 1 μL, 2.6 μL, and 1 μL of fluorescent detector, primer mix, Bst DNA polymerase, respectively (Eiken Chemical, Japan) and 2.9 μL of nuclease free water in a total volume of 57.5 μL. Positive pools were tested individually using DNA volume of 5μL in a final volume of 12.5 μL using master mix components as stated above. This protocol has previously been published [24], using the LAMP primers presented in Table 1, which was initially used by [28].
Table 1. Oligonucleotide primers used for the 18s rRNA amplification.
| Region | Oligonucleotide primers |
|---|---|
| FIP | 5’-AGCTGGAATTACCGCGGCTGGGTTCCTAGAGAAACAATTGG-3’ |
| BIP | 5’-TGTTGCAGTTAAAACGTTCGTAGCCCAACCAGTTTAAATGAAAC-3’ |
| F3 | 5’-TGTAATTGGAATGATAGGAATTTA-3’ |
| B3 | 5’-GAAAACCTTATTTTGAACAAAGC-3’ |
| LF | 5’-GCACCAGACTTGCCCT-3’ |
| LB | 5’-TTGAATATTAAAGAA-3’ |
FIP–forward inner primer, BIP–backward inner primer, F3—forward outer primer, B3 –backward outer primer, LF—forward loop primer, LB—backward loop primer
Amplification and visualization of 18s rRNA gene
The LAMP reaction was done using Stuart Heating Block (Cole-Parmer, Vernon, United States), at a constant temperature of 65°C for 60 minutes. The enzyme was inactivated at 80°C for 5 minutes. Per the fluorescent detector used, greenish yellow end product was corresponded with a positive reaction while the brownish product was a negative test (Fig 1).
Fig 1. Quality control of LAMP assay.
EC–extraction control, RC1 –reaction control (master mix), RC2—reaction control: master mix with nuclease free water, PC 1 –positive control (24,087 parasites/μL), PC2—positive control (812 parasites/μL), PC3—positive control (6 parasites/μL), NC–negative control, NR1/2 –patient negative reactions, PR1/2 –patient positive reactions.
Internal quality controls
Six different controls were incorporated into the study, namely, positive controls (three blood samples with parasitemia, 24,087 parasites/μL, 812 parasites/μL and PBS diluted sample with estimated parasitemia 6 parasites/μL), extraction control (nuclease free water), and reaction controls (master mix). The extraction control, reaction control, and the master mix were Plasmodium LAMP negative while the positive controls were positive. The internal and patient control reactions are presented as Fig 1.
Data analysis
Data analysis was done by using SPSS Version 25 (Chicago, IL, USA). Univariate analysis was made to determine the proportion Plasmodium 18s rRNA positivity rate in each demographic and clinical variable. This was presented as tables and graphs. Multivariate analysis was done to identify variables at risk of Plasmodium 18s rRNA infection. P-value less than 0.05 was considered statistically significant.
Ethical approval
This study was approved by the Ghana Health Service Ethics Review Committee (GHS-REC002/03/18).
Results
Descriptive statistics of the outcome of the study samples
During the study period, 2223 patients suspected of malaria were recruited. Of this number, 579 (26%) and 947 (42.6%) patients were positive for microscopy and mRDT alone respectively wheras 568 (98.1%) samples out of the those positive for microscopy were positive for both micrscopy and mRDT. Of the total samples collected, 697 (31.4%) samples were negative for both microscopy and mRDT.
Characteristics of the study participants and their association with Plasmodium 18s rRNA status
Table 2 presents the statistical analysis of the study data. Most of the patients were children up to five years (37.7%), females (59.0%), peri-urban dwellers (39.0%), and patients that sought outpatient department services (39.3%). Overall, the prevalence of Plasmodium 18s rRNA in patients with previous negative malaria test was 17.5%. Notwithstanding the foregoing, majority of Plasmodium 18s rRNA gene was detected in patients over 30 years [58/122 (54.2%)], males [69/122 (56.5%)], rural dwellers [69/122 (56.5%)] and patients seeking OPD services [68/122 (55.7%)]. The risk of having 18S LAMP positive result was increased in patients aged 15–30 years (OR = 3.03, 95%CI: 1.6–5.8, p = 0.0007) and over 30 years (OR = 15.2, 95%CI: 8.3–27.7, p < 0.001) compared to those aged less than 6 years. Furthermore, the risk of having 18S LAMP positive result was increased in patients in males (OR = 2.1, 95% CI: 1.4–3.2, p = 0.0002), rural dwellers (OR = 2.2, 95% CI:1.4–3.6, p = 0.0009) and decreased in peri-urban dwellers (OR = 0.4, 95% CI: 0.24–0.77, p = 0.005) relative to urban dwellers. Finally, patients that sought antenatal services (OR = 0.67, 95% CI: 0.41–1.08, p = 0.1), and child welfare clinic (OR = 0.3, 95% CI: 0.18–0.51, p<0.001) had a lower risk of Plasmodium 18s rRNA positivity compared to patient that sought outpatient services.
Table 2. Demographical features of the study participants and their association with Plasmodium 18s rRNA status.
| Plasmodium 18s rRNA | |||||
|---|---|---|---|---|---|
| Variable | Total | Negative n (%) | Positive n (%) | OR (95%CI) | p-value |
| Age range | |||||
| 0–5 | 263 (37.7) | 244 (92.8) | 19 (7.2) | 1 | |
| 6–14 | 202 (29.0) | 181 (89.6) | 21 (10.4) | 1.5 (0.8–2.8) | 0.229 |
| 15–30 | 125 (17.9) | 101 (80.8) | 24 (19.2) | 3.03 (1.6–5.8) | 0.0007 |
| > 30 | 107 (15.4) | 49 (45.8) | 58 (54.2) | 15.2 (8.3–27.7) | <0.001 |
| Gender | |||||
| Male | 286 (41.0) | 217 (75.9) | 69 (24.1) | 2.1 (1.4–3.2) | 0.0002 |
| Female | 411 (59.0) | 358 (87.1) | 53 (12.9) | 1 | |
| Residential setting | |||||
| Rural | 229 (32.9) | 160 (70.0) | 69 (30.0) | 2.2 (1.4–3.6) | 0.0009 |
| Peri-urban | 271 (39.0) | 250 (92.3) | 21 (7.7) | 0.4 (0.24–0.77) | 0.005 |
| Urban | 197 (28.3) | 165 (83.8) | 32 (16.2) | 1 | |
| Service required | |||||
| Outpatient | 274 (39.3) | 206 (75.2) | 68 (24.8) | 1 | |
| Antenatal | 171 (24.5) | 140 (81.9) | 31 (18.1) | 0.67 (0.41–1.08) | 0.1 |
| Child welfare clinic | 252 (36.2) | 229 (90.9) | 23 (9.1) | 0.3 (0.18–0.51) | <0.001 |
OR–odds ratio; P–value at 95% confidence interval
Frequencies of Plasmodium 18s rRNA positivity in various rainfall patterns
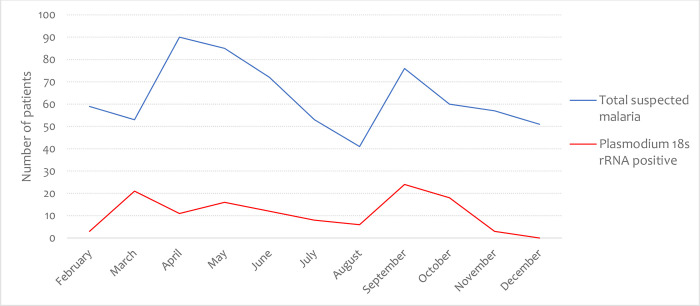
Fig 2 details the monthly prevalence of subpatent malaria during the period of the study. The Plasmodium 18s rRNA prevailed during the season of minor rains [36.5% (63/189)] compared to the major rainy season [15.6% (39/247)] and the season of no or very low [7.7% (20/261)] rains. In the major rainy season, more cases of Plasmodium were observed in May [18.8% (16/85)] compared to June and April with 16.7%, and 12.2% respectably. During the season of minor rains, each month recorded 30% or more of Plasmodium 18s rRNA positivity, however, March recorded the highest positivity rate. In the season of no or very low rains, July, and August recorded 15.9% and 14.6% Plasmodium 18s rRNA positivity rate, respectively, while December recorded no positive Plasmodium 18r rRNA.
Fig 2. Monthly trends in suspected and Plasmodium 18s rRNA positive cases.
Major rainfall–April–June, minor rainfall–March, September and October, no or very low rains—July, August, November–February.
Clinical history of patients infected with P. falciparum
Table 3 indicates the clinical history of the patients with LAMP positive reactions. The 122 patients found to be positive for 18S rRNA presented with a total of 190 symptoms. Majority of the patients (155/190; 81.5%) presented with headache (n = 50, 26.3%), herpes labialis (n = 39, 20.5%), diarrhea (n = 35, 18.4%), and vomiting (n = 31, 16.3).
Table 3. The proportions of the clinical features of the LAMP positive patients.
| Clinical history | LAMP positive | % |
|---|---|---|
| Headache | 50 | 26.3 |
| Herpes labialis | 39 | 20.5 |
| Diarrhea | 35 | 18.4 |
| Vomiting | 31 | 16.3 |
| Fever | 9 | 4.7 |
| Dizziness | 7 | 3.7 |
| Chills | 5 | 2.6 |
| Nausea | 5 | 2.6 |
| Skin rashes | 4 | 2.1 |
| Anemia | 3 | 1.6 |
| Hematuria | 2 | 1.1 |
Discussion
This study reported that microscopy and mRDT underestimated the detection of Plasmodium in suspected patients by 17.5%. This could be due to the reported high sensitivity of LAMP for detecting malaria parasites, even in asymptomatic situations. In a previous study, the limit of detection for LAMP for detecting malaria parasites was 0.25 parasites/μL of blood [24]. In this study, the final product fluorescence observed for a sample dilution containing 6 parasites/μL of blood was similar to a sample containing 24,087 parasites/μL. this underscores the sensitivity of the LAMP assay, hence, able to detect P. falciparum is blood samples that previously was deemed negative by routine methods.
Children under 6 years and pregnant women were also found to be positive for 18s rRNA LAMP. These cohorts were mostly at risk of malaria, therefore, prompt treatment following early detection is essential to prevent deleterious sequelae associated with Plasmodium infection [22, 29]. The risk of submicroscopic malaria was significantly higher in patients 15 years or more compared to patients less than 6 years. The main reason could be due to competent Plasmodium immunity which increases with age [30]. This easily result in either asymptomatic or submicroscopic malara, therefore, children (especially under 6 years of age) easily developed clinical malaria compared to older patients due to incompetent antimalarial immunity [31]. It was also observed that the positivity rate was less in patients aged 6–29 years compared to patients over 30 years. It could possibly be that they were exposed to the parasite due to prolonged nocturnal and diurnal working life which is common among these age group [32].
Aninagyei (2020) reported that microscopy underestimated the detection rate of Plasmodium falciparum by 12% on single testing. When repeated testing was done at 6 hours and 12 hours after baseline testing, the positivity rates were 35% and 42%, respectively. This was because parasitemia at hour 6 was submicroscopic but at the 12th hour, continuous and unchecked multiplication of the parasites yielded enough parasitemia to be detected microscopically [14]. During the same period, the positivity rate of mRDT increased from 42.9% at baseline testing to 47.5% 12 hours after baseline testing [14]. It is obvious that a significant proportion of patients presenting with clinical signs of malaria may test positive for malaria either with microscopy or mRDT within 12 hours or more. However, repeated sampling for 12 hours or more may not be feasible since those that temporarily responded to tepid sponging or other interventions may be discharged and may return to the hospital with a severe form of the disease. In the previous study the proportion malaria was underreported by 12% while in this study, LAMP was positive in only 7.2% that were previously tested negative by routine methods. This observation was quite surprising since a similar or higher prevalence was expected. The reason that could be ascribed to this observation was probably due to the differences in the prevalence of malaria in these study sites. The prevalence of malaria in the Ga West Municipality, a contiguous district to the study site for the Aninagyei (2020) study, was reported to be 36.3% [33], which was significantly higher than the prevalence of malaria in Kwahu-South (11.9%) [34], a contiguous district to the New Juaben South Municipality, the study district for this study. Secondly, the study site for the previous study was peri-urban while that of this study was urban. These factors could account for the differences of prevalence observed in this study.
The risk of P. falciparum positivity was reduced in children between 0–5 years and pregnant women. This could be as a result of the free distribution and use of long-lasting insecticide treated nets to children and pregnant women in Ghana [35] and the mandatory use of sulfadoxine/pyrimethamine prophylaxis by pregnant women which is directly administred by the health care providers in Ghana [36]. Additionally, P. falciparum cytoadherence especially in placenta [37] could account for the reduced sensitivity of the LAMP assay due possibly to very few presence of peripheral parasitemia.
Even though females have been found to be more likely to have malaria than males [38, 39], our study reported otherwise. The risk of 18s rRNA positivity was found to be more than doubled for male gender, Those studies reported on clinical malaria while in our study, subpatent malaria was the case. It was reported in another Ghanaian study [40] that being male increased the odds of asymptomatic malaria by 18%. The higher prevalence of asymptomatic malaria infections among males than females have been reported as a result of gender roles with regards to division of labour, hormonal or host genetic factors [41, 42].
On the other hand, rural dwellers have been reported in several studies to be associated with malaria transmission. This is because the vector for transmission of Plasmodium has been reported to survive well in forested areas where there is stagnation of clear rain water in crevices, humid atmosphere, and tree holes which favor oviposition sites. Additionally, the presence of temporary pools and gullies are common in rural areas [31, 32, 34, 35]. Furthermore, rural dwellers have the habit of outdoor activities, especially at night, when mosquito biting rates are high [43].
The study also found that Plasmodium infection was higher in the months of March, September, and October, compared to the other months. In these months, irregular rains were frequent. Irregular rainfall patterns offer enough time for the mosquito eggs to hatch into larvae and subsequently to the pupal stage and finally, to the adult stage. During these periods, room temperature rises due to frequent switches between sunny and short rains. Increased room temperature is associated with infrequent insecticide treated net usage [44]. Taking together, vector inoculation rates increase, which increase the carrier rate of the Plasmodium parasites. Unfortunately, there is no reported evidence of the actual state of entomological inoculation rates in the study municipality. There is the need to establish this, in a future study, to give credence to this observations.
Conclusion
We demonstrate the lack of sensitivity of microscopy and mRDT for one-time diagnosis of malaria. These routine techniques have been shown to underestimate Plasmodium positivity rate by 17.5%, with a significant number of children (9.1%) and pregnant women (18.1%) declared negative for malaria. Therefore, it is essential to utilize a sensitive technique such as Plasmodium 18s rRNA LAMP to increase the detection rate of Plasmodium detection. In conclusion, LAMP technology is highly recommended for detection of subpatent parasitemia because it has been reported to be of high sensitivity, simple to use, cost-effective, and user-friendly [24].
Acknowledgments
We are grateful to the management and staff of Koforidua Polyclinic in the New Juaben South Municipality in the Eastern Region of Ghana for supporting this study.
List of abbreviations
- HRP
histidine-rich protein
- LAMP
loop-mediated isothermal amplification
- mRDT
malaria rapid diagnostic test kit
- NAAT
nucleic acid amplification technologies
- PCR
polymerase chain reaction
Data Availability
All data and related metadata underlying the findings are provided in the paper.
Funding Statement
The authors received no specific funding for this work.
References
- 1.Badmos AO, Alaran AJ, Adebisi YA, Bouaddi O, Onibon Z, Dada A, et al. What sub-Saharan African countries can learn from malaria elimination in China. Trop Med Health [Internet]. 2021. Dec 24;49(1):86. Available from: https://tropmedhealth.biomedcentral.com/articles/10.1186/s41182-021-00379-z [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Cunningham J, Jones S, Gatton ML, Barnwell JW, Cheng Q, Chiodini PL, et al. A review of the WHO malaria rapid diagnostic test product testing programme (2008–2018): performance, procurement and policy. Malar J [Internet]. 2019. Dec 2;18(1):387. Available from: https://malariajournal.biomedcentral.com/articles/10.1186/s12936-019-3028-z [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Wangai L, Karau M, Njiruh P, Sabah O, Kimani F, Magoma G, et al. Sensitivity of microscopy compared to molecular diagnosis of P. falciparum: Implications on malaria treatment in epidemic areas in Kenya. African J Infect Dis [Internet]. 2011. May 24;5(1). Available from: http://www.ajol.info/index.php/ajid/article/view/66504 doi: 10.4314/ajid.v5i1.66504 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Moody A. Rapid Diagnostic Tests for Malaria Parasites. Clin Microbiol Rev [Internet]. 2002. Jan;15(1):66–78. Available from: https://journals.asm.org/doi/10.1128/CMR.15.1.66-78.2002 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Payne D. Use and limitations of light microscopy for diagnosing malaria at the primary health care level. Bull World Health Organ [Internet]. 1988;66(5):621–6. Available from: http://www.ncbi.nlm.nih.gov/pubmed/2463112 [PMC free article] [PubMed] [Google Scholar]
- 6.Michael OS, Orimadegun AE, Falade CO. PERSISTENCE OF PLASMODIUM FALCIPARUM HRP2 ANTIGEN AFTER EFFECTIVE ANTIMALARIAL THERAPY. Ann Ibadan Postgrad Med [Internet]. 2021. Jun;19(1):15–21. Available from: http://www.ncbi.nlm.nih.gov/pubmed/35330886 [PMC free article] [PubMed] [Google Scholar]
- 7.Nyataya J, Waitumbi J, Mobegi VA, Noreddin A, El Zowalaty ME. Plasmodium falciparum Histidine-Rich Protein 2 and 3 Gene Deletions and Their Implications in Malaria Control. Diseases [Internet]. 2020. May 20;8(2):15. Available from: https://www.mdpi.com/2079-9721/8/2/15 doi: 10.3390/diseases8020015 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Bakari C, Jones S, Subramaniam G, Mandara CI, Chiduo MG, Rumisha S, et al. Community-based surveys for Plasmodium falciparum pfhrp2 and pfhrp3 gene deletions in selected regions of mainland Tanzania. Malar J [Internet]. 2020. Dec 4;19(1):391. Available from: https://malariajournal.biomedcentral.com/articles/10.1186/s12936-020-03459-3 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Hänscheid T. Current strategies to avoid misdiagnosis of malaria. Clin Microbiol Infect [Internet]. 2003. Jun;9(6):497–504. Available from: https://linkinghub.elsevier.com/retrieve/pii/S1198743X14639999 doi: 10.1046/j.1469-0691.2003.00640.x [DOI] [PubMed] [Google Scholar]
- 10.Ohrt C, Purnomo, Sutamihardja MA, Tang D, Kain KC. Impact of Microscopy Error on Estimates of Protective Efficacy in Malaria‐Prevention Trials. J Infect Dis [Internet]. 2002. Aug 15;186(4):540–6. Available from: https://academic.oup.com/jid/article-lookup/doi/10.1086/341938 [DOI] [PubMed] [Google Scholar]
- 11.Amoah LE, Abuaku B, Bukari AH, Dickson D, Amoako EO, Asumah G, et al. Contribution of P. falciparum parasites with Pfhrp 2 gene deletions to false negative PfHRP 2 based malaria RDT results in Ghana: A nationwide study of symptomatic malaria patients. Carvalho LH, editor. PLoS One [Internet]. 2020. Sep 4;15(9):e0238749. Available from: https://dx.plos.org/10.1371/journal.pone.0238749 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Thomson R, Beshir KB, Cunningham J, Baiden F, Bharmal J, Bruxvoort KJ, et al. pfhrp2 and pfhrp3 Gene Deletions That Affect Malaria Rapid Diagnostic Tests for Plasmodium falciparum: Analysis of Archived Blood Samples From 3 African Countries. J Infect Dis [Internet]. 2019. Sep 26;220(9):1444–52. Available from: https://academic.oup.com/jid/article/220/9/1444/5524476 doi: 10.1093/infdis/jiz335 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Gendrot M, Fawaz R, Dormoi J, Madamet M, Pradines B. Genetic diversity and deletion of Plasmodium falciparum histidine-rich protein 2 and 3: a threat to diagnosis of P. falciparum malaria. Clin Microbiol Infect [Internet]. 2019. May;25(5):580–5. Available from: https://linkinghub.elsevier.com/retrieve/pii/S1198743X18306311 doi: 10.1016/j.cmi.2018.09.009 [DOI] [PubMed] [Google Scholar]
- 14.Aninagyei E. Repeated sampling improved the sensitivity of malaria microscopy in children under six years. BMC Res Notes [Internet]. 2020;13(1):1–5. Available from: 10.1186/s13104-020-05359-w [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Aninagyei E, Doku ET, Adu P, Egyir-Yawson A, Acheampong DO. Storage related haematological and biochemical changes in Plasmodium falciparum infected and sickle cell trait donor blood. BMC Hematol. 2018;18(1):1–10. doi: 10.1186/s12878-018-0128-x [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Leski TA, Taitt CR, Swaray AG, Bangura U, Reynolds ND, Holtz A, et al. Use of real-time multiplex PCR, malaria rapid diagnostic test and microscopy to investigate the prevalence of Plasmodium species among febrile hospital patients in Sierra Leone. Malar J [Internet]. 2020. Dec 21;19(1):84. Available from: https://malariajournal.biomedcentral.com/articles/10.1186/s12936-020-03163-2 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Liu HY, Hopping GC, Vaidyanathan U, Ronquillo YC, Hoopes PC, Moshirfar M. Polymerase Chain Reaction and Its Application in the Diagnosis of Infectious Keratitis. Med hypothesis, Discov Innov Ophthalmol J [Internet]. 2019;8(3):152–5. Available from: http://www.ncbi.nlm.nih.gov/pubmed/31598517 [PMC free article] [PubMed] [Google Scholar]
- 18.Notomi T. Okayama Hiroto, Masubuchi Harumi, Yonekawa Toshihiro, Watanabe Keiko, Nobuyuki Amino, et al. Loop-mediated isothermal amplification of DNA. Nucleic Acids Res [Internet]. 2000. Jun 15;28(12):63e – 63. Available from: https://academic.oup.com/nar/article-lookup/doi/10.1093/nar/28.12.e63 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Wong Y-P, Othman S, Lau Y-L, Radu S, Chee H-Y. Loop-mediated isothermal amplification (LAMP): a versatile technique for detection of micro-organisms. J Appl Microbiol [Internet]. 2018. Mar;124(3):626–43. Available from: https://onlinelibrary.wiley.com/doi/10.1111/jam.13647 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Postigo M, Mendoza-León A, Pérez HA. Malaria diagnosis by the polymerase chain reaction: a field study in southeastern Venezuela. Trans R Soc Trop Med Hyg [Internet]. 1998. Sep;92(5):509–11. Available from: doi: 10.1016/s0035-9203(98)90893-8 [DOI] [PubMed] [Google Scholar]
- 21.Sriworarat C, Phumee A, Mungthin M, Leelayoova S, Siriyasatien P. Development of loop-mediated isothermal amplification (LAMP) for simple detection of Leishmania infection. Parasit Vectors [Internet]. 2015. Dec 14;8(1):591. Available from: http://www.parasitesandvectors.com/content/8/1/591 doi: 10.1186/s13071-015-1202-x [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Odari EO, Maiyo A, Lwembe R, Gurtler L, Eberle J, Nitschko H. Establishment and evaluation of a loop-mediated isothermal amplification (LAMP) assay for the semi-quantitative detection of HIV-1 group M virus. J Virol Methods [Internet]. 2015. Feb;212:30–8. Available from: https://linkinghub.elsevier.com/retrieve/pii/S0166093414004029 doi: 10.1016/j.jviromet.2014.10.012 [DOI] [PubMed] [Google Scholar]
- 23.Nagai Y, Iwade Y, Nakano M, Akachi S, Kobayashi T, Nishinaka T. Rapid and simple identification of Beijing genotype strain of Mycobacterium tuberculosis using a loop-mediated isothermal amplification assay. Microbiol Immunol [Internet]. 2016. Jul;60(7):459–67. Available from: https://onlinelibrary.wiley.com/doi/10.1111/1348-0421.12389 [DOI] [PubMed] [Google Scholar]
- 24.Aninagyei E, Smith-Graham S, Boye A, Egyir-Yawson A, Acheampong DO. Evaluating 18s-rRNA LAMP and selective whole genome amplification (sWGA) assay in detecting asymptomatic Plasmodium falciparum infections in blood donors. Malar J [Internet]. 2019;18(1):1–11. Available from: 10.1186/s12936-019-2850-7 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Otchere ID, Afum T, Morgan P, Musah AB. Evaluation of a loop-mediated amplification test for rapid diagnosis of tuberculosis in Ghana. 2022;(July). [Google Scholar]
- 26.Ablordey A, Ahotor E, Narh CA, King SA, Cruz I, Ndung’u JM, et al. Evaluation of different DNA extraction methods and loop-mediated isothermal amplification primers for the detection of Mycobacterium ulcerans in clinical specimens. BMC Infect Dis [Internet]. 2021. Dec 23;21(1):598. Available from: https://bmcinfectdis.biomedcentral.com/articles/10.1186/s12879-021-06308-z [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Aninagyei E, Tetteh CD, Oppong M, Boye A, Acheampong DO. Efficacy of Artemether-Lumefantrine on various Plasmodium falciparum Kelch 13 and Pfmdr1 genes isolated in Ghana. Parasite Epidemiol Control [Internet]. 2020. Nov;11:e00190. Available from: https://linkinghub.elsevier.com/retrieve/pii/S2405673120300593 doi: 10.1016/j.parepi.2020.e00190 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Poon LL, Wong BW, Ma EH, Chan KH, Chow LM, Abeyewickreme W, et al. Sensitive and Inexpensive Molecular Test for Falciparum Malaria: Detecting Plasmodium falciparum DNA Directly from Heat-Treated Blood by Loop-Mediated Isothermal Amplification,. Clin Chem [Internet]. 2006. Feb 1;52(2):303–6. Available from: https://academic.oup.com/clinchem/article/52/2/303/5626787 doi: 10.1373/clinchem.2005.057901 [DOI] [PubMed] [Google Scholar]
- 29.Schumacher R-F, Spinelli E. MALARIA IN CHILDREN. Mediterr J Hematol Infect Dis [Internet]. 2012. Nov 7;4(1):e2012073. Available from: http://www.mjhid.org/index.php/mjhid/article/view/2012.073 doi: 10.4084/MJHID.2012.073 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Laurens MB. The Immunologic Complexity of Growing Up with Malaria—Is Scientific Understanding Coming of Age? Pasetti MF, editor. Clin Vaccine Immunol [Internet]. 2016. Feb;23(2):80–3. Available from: https://journals.asm.org/doi/10.1128/CVI.00697-15 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Addy JWG, Bediako Y, Ndungu FM, Valetta JJ, Reid AJ, Mwacharo J, et al. 10-year longitudinal study of malaria in children: Insights into acquisition and maintenance of naturally acquired immunity. Wellcome Open Res [Internet]. 2022. Feb 4;6:79. Available from: https://wellcomeopenresearch.org/articles/6-79/v3 doi: 10.12688/wellcomeopenres.16562.3 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Mawili-Mboumba DP, Akotet MKB, Kendjo E, Nzamba J, Medang MO, Mbina J-RM, et al. Increase in malaria prevalence and age of at risk population in different areas of Gabon. Malar J [Internet]. 2013. Dec 2;12(1):3. Available from: https://malariajournal.biomedcentral.com/articles/10.1186/1475-2875-12-3 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Rufai T, Aninagyei E, Akuffo KO, Ayin CT-M, Nortey P, Quansah R, et al. Malaria and typhoid fever coinfection among patients presenting with febrile illnesses in Ga West Municipality, Ghana. medRxiv [Internet]. 2022;2022.04.12.22273780. Available from: http://medrxiv.org/content/early/2022/04/16/2022.04.12.22273780.abstract [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Owusu EDA, Buabeng V, Dadzie S, Brown CA, Grobusch MP, Mens P. Characteristics of asymptomatic Plasmodium spp. parasitaemia in Kwahu-Mpraeso, a malaria endemic mountainous district in Ghana, West Africa. Malar J [Internet]. 2016. Dec 22;15(1):38. Available from: http://www.malariajournal.com/content/15/1/38 doi: 10.1186/s12936-015-1066-8 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Darko E, Tetteh J, Ayanore MA, Damoah-Aferi I. Socio-demographic determinants associated with ownership and use of long lasting insecticide treated nets among pregnant women in the Wa Municipality of Ghana. Pan Afr Med J [Internet]. 2019;33. Available from: http://www.panafrican-med-journal.com/content/article/33/81/full/ doi: 10.11604/pamj.2019.33.81.16245 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Yeboah DF, Afoakwah R, Nwaefuna EK, Verner O, Boampong JN. Quality of Sulfadoxine-Pyrimethamine Given as Antimalarial Prophylaxis in Pregnant Women in Selected Health Facilities in Central Region of Ghana. J Parasitol Res [Internet]. 2016;2016:1–6. Available from: http://www.hindawi.com/journals/jpr/2016/9231946/ doi: 10.1155/2016/9231946 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Valiyaveettil M, Achur RN, Alkhalil A, Ockenhouse CF, Gowda DC. Plasmodium falciparum Cytoadherence to Human Placenta: Evaluation of Hyaluronic Acid and Chondroitin 4-Sulfate for Binding of Infected Erythrocytes. Exp Parasitol [Internet]. 2001. Oct;99(2):57–65. Available from: https://linkinghub.elsevier.com/retrieve/pii/S0014489401946429 doi: 10.1006/expr.2001.4642 [DOI] [PubMed] [Google Scholar]
- 38.Quaresima V, Agbenyega T, Oppong B, Awunyo JADA, Adu Adomah P, Enty E, et al. Are Malaria Risk Factors Based on Gender? A Mixed-Methods Survey in an Urban Setting in Ghana. Trop Med Infect Dis [Internet]. 2021. Sep 2;6(3). Available from: http://www.ncbi.nlm.nih.gov/pubmed/34564545 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Deku JG, Lokpo SY, Kye-Amoah KK, Orish VN, Ussher FA, Esson J, et al. Malaria Burden and Trend Among Clients Seeking Healthcare in the Western Region: A 4-Year Retrospective Study at the Sefwi-Wiawso Municipal Hospital, Ghana. Open Microbiol J [Internet]. 2018. Dec 31;12(1):404–11. Available from: https://openmicrobiologyjournal.com/VOLUME/12/PAGE/404/ [Google Scholar]
- 40.Mensah BA, Myers-Hansen JL, Obeng Amoako E, Opoku M, Abuaku BK, Ghansah A. Prevalence and risk factors associated with asymptomatic malaria among school children: repeated cross-sectional surveys of school children in two ecological zones in Ghana. BMC Public Health [Internet]. 2021. Dec 17;21(1):1697. Available from: https://bmcpublichealth.biomedcentral.com/articles/10.1186/s12889-021-11714-8 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Pathak S, Rege M, Gogtay NJ, Aigal U, Sharma SK, Valecha N, et al. Age-Dependent Sex Bias in Clinical Malarial Disease in Hypoendemic Regions. Snounou G, editor. PLoS One [Internet]. 2012. Apr 25;7(4):e35592. Available from: https://dx.plos.org/10.1371/journal.pone.0035592 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.van Eijk AM, Sutton PL, Ramanathapuram L, Sullivan SA, Kanagaraj D, Priya GSL, et al. The burden of submicroscopic and asymptomatic malaria in India revealed from epidemiology studies at three varied transmission sites in India. Sci Rep [Internet]. 2019. Dec 19;9(1):17095. Available from: http://www.nature.com/articles/s41598-019-53386-w doi: 10.1038/s41598-019-53386-w [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Monroe A, Asamoah O, Lam Y, Koenker H, Psychas P, Lynch M, et al. Outdoor-sleeping and other night-time activities in northern Ghana: implications for residual transmission and malaria prevention. Malar J [Internet]. 2015. Dec 28;14(1):35. Available from: https://malariajournal.biomedcentral.com/articles/10.1186/s12936-015-0543-4 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Ahorlu CS, Adongo P, Koenker H, Zigirumugabe S, Sika-Bright S, Koka E, et al. Understanding the gap between access and use: a qualitative study on barriers and facilitators to insecticide-treated net use in Ghana. Malar J [Internet]. 2019. Dec 12;18(1):417. Available from: https://malariajournal.biomedcentral.com/articles/10.1186/s12936-019-3051-0 [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
All data and related metadata underlying the findings are provided in the paper.