Abstract
Background and Objectives
Status epilepticus that continues after the initial benzodiazepine and a second anticonvulsant medication is known as refractory status epilepticus (RSE). Management is highly variable because adequately powered clinical trials are missing. We aimed to determine whether propofol and midazolam were equally effective in controlling RSE in the intensive care unit, focusing on management in resource-limited settings.
Methods
Patients with RSE treated with midazolam or propofol between January 2015 and December 2018 were retrospectively identified among 9 centers across 4 continents from upper–middle-income economies in Latin America and high-income economies in North America, Europe, and Asia. Demographics, Status Epilepticus Severity Score, etiology, treatment details, and discharge modified Rankin Scale (mRS) were collected. The primary outcome measure was good functional outcome defined as a mRS score of 0–2 at hospital discharge.
Results
Three hundred eighty-seven episodes of RSE (386 patients) were included, with 162 (42%) from upper–middle-income and 225 (58%) from high-income economies. Three hundred six (79%) had acute and 79 (21%) remote etiologies. Initial RSE management included midazolam in 266 (69%) and propofol in 121 episodes (31%). Seventy episodes (26%) that were initially treated with midazolam and 42 (35%) with propofol required the addition of a second anesthetic to treat RSE. Baseline characteristics and outcomes of patients treated with midazolam or propofol were similar. Breakthrough (odds ratio [OR] 1.6, 95% CI 1.3–2.0) and withdrawal seizures (OR 2.0, 95% CI 1.7–2.5) were associated with an increased number of days requiring continuous intravenous anticonvulsant medications (cIV-ACMs). Prolonged EEG monitoring was associated with fewer days of cIV-ACMs (1–24 hours OR 0.5, 95% CI 0.2–0.9, and >24 hours OR 0.7, 95% CI 0.5–1.0; reference EEG <1 hour). This association was seen in both, high-income and upper–middle-income economies, but was particularly prominent in high-income countries. One hundred ten patients (28%) were dead, and 80 (21%) had good functional outcomes at hospital discharge.
Discussion
Outcomes of patients with RSE managed in the intensive care unit with propofol or midazolam infusions are comparable. Prolonged EEG monitoring may allow physicians to decrease the duration of anesthetic infusions safely, but this will depend on the implementation of RSE management protocols. Goal-directed management approaches including EEG targets may hold promise for patients with RSE.
Classification of Evidence
This study provides Class III data that propofol and midazolam are equivalently efficacious for RSE.
Refractory status epilepticus (RSE) is common, both in upper–middle-income and high-income economies, and associated with high morbidity and mortality.1,2 Treatment of RSE is controversial given the lack of high-quality data to guide management decisions such as randomized controlled trials or even large-scale contemporary multicenter cohort studies. Most existing studies did not use uniform data collection instruments, making comparisons between patients very challenging.3 Even less is known about practice differences between patients treated in upper–middle-income and high-income economies.4,5 The main goal of this report was to test the hypothesis that propofol and midazolam were equally effective in controlling RSE managed in the intensive care unit (ICU). We put a particular focus on studying management in resource-limited settings. We formed an international consortium called Management of RSE: an international cohort study (MORSE CODe) to compile a large contemporary international cohort study of patients with RSE and establish differences in management practice and outcomes between upper–middle-income and high-income countries. The primary research question being addressed in the study is to determine the efficacy of propofol and midazolam for the treatment of RSE.
Methods
Study Design and Patients
This was a retrospective, multicenter, observational cohort study to compare the efficacy of propofol and midazolam for the treatment of RSE in the ICU. Potential sites were identified among members of the Neurointensive Care Society, and an investigator meeting was held at the Annual Neurocritical Care Society Meeting in Boca Raton, FL, on September 22, 2018. The sites were selected based on expressed interest in participation by the site principal investigator indicated at the meeting or thereafter, access to records, and availability of at least 10 patients with RSE. Inclusion and exclusion criteria and the first draft of the data collection instrument were agreed upon at this meeting. The investigators agreed to focus on consecutively admitted patients at each site treated with midazolam or propofol infusions for RSE between January 1, 2015, and December 31, 2018. Management protocols at all sites recommended treatment with either midazolam or propofol for RSE and obtaining an EEG to confirm a suspected diagnosis of psychogenic seizures and nonconvulsive status epilepticus. The STROBE guidelines were used to ensure standardized reporting of this observational study.6
We included patients (1) aged 18 years or older, (2) with RSE, (3) who were treated with continuous infusions of midazolam or propofol to manage status epilepticus, and (4) who were admitted to the ICU between January 2015 and December 2018. Episodes of RSE in the same patient were allowed to be included if separated by at least 1 month. We excluded patients who (1) were younger than 18 years, (2) did not receive either midazolam or propofol for the treatment of status epilepticus, and (3) were not admitted to the ICU. RSE was defined as the failure of status epilepticus to respond to at least a benzodiazepine and 1 additional nonanesthetic anticonvulsant medication.1,7
Standard Protocol Approvals, Registrations, and Patient Consents
Local institutional review board (IRB) approval was obtained at each site for data collection (the following ethics boards at each participating site approved this study: Human Research Protection Office, Institutional Review Board at Columbia University; Taipei Medical University Joint IRB; Hospital Luis Vernaza, Comité de Bioética; Ethics Committee of the Medical University of Innsbruck; IRB of the National Institute of Infection Evandro Chagas at Fundação Oswaldo Cruz; the Mayo Clinic Institutional Review Board; Délégation à la Recherche Clinique et al.' Innovation de l'Assistance Publique—Hôpitaux de Paris; and the Human Research Ethics Committee CEISH-USFQ, Universidad San Francisco de Quito; eTable 1, links.lww.com/WNL/C219), written informed consent was waived at all sites, and IRB approval was obtained for data sharing of deidentified patient data, as required by local regulations.
Data Collection and Outcomes
Patients were identified based on case logs maintained by each of the site PIs (S.E.H., R.S., C.T.H., S.D., P.K., N.M., R.H., T.F., and J.C.). Data were collected by research assistants and study coordinators supervised by the site PIs. Data were collected by a retrospective chart review and entered into a standardized, template database in Excel. Deidentified data were shared with the study coordinating site (Columbia University Irving Medical Center) by encrypted email. Seizure control definitions were uniformly applied and determined by each site PI based on the available data including clinical findings and EEG, as available. Cessation of clinical convulsions (generalized or focal) was the primary criterion to determine seizure control in patients with convulsive status epilepticus. EEG was not required but if available was used to determine whether nonconvulsive seizures persisted. Seizures were not classified as controlled if either clinical convulsions or EEG documented ongoing seizure activity. Patients with nonconvulsive status epilepticus were diagnosed with EEG, and seizure control required cessation of nonconvulsive seizures on EEG. Modified Rankin Scale (mRS) scores at hospital discharge were determined based on the chart review by trained research assistants and study coordinators supervised by each of the site PIs.
Demographic data (i.e., age and sex) and information on the history of present illness (i.e., etiology of status and duration of status before the initiation of treatment) were collected. Baseline predictors of outcome were captured using the Status Epilepticus Severity Score (STESS).8 EEG in this study was primarily used to monitor the management of seizures. We recorded data on the management approach of these patients including non–anesthetic anticonvulsant medications (ACMs) and anesthetic infusions (initial and secondary anesthetic infusions, starting and maintenance doses, and duration of infusions). Response to treatment was determined by quantifying the number of patients who were seizure-free within 24 hours of starting the anesthetic infusion (labeled seizure cessation). Patients with clinical or electrographic seizures occurring 6 hours after the start but during the administration of midazolam or propofol infusions were classified as having breakthrough seizures. We documented whether patients fulfilled criteria for super-RSE defined as recurrence of status epilepticus within 24 hours or more after the onset of anesthetic therapy.1 The following laboratory data on complications were collected whenever available: lactic acidosis (defined as actual base excess < −3 mmol/L), rhabdomyolysis (defined based on creatine phosphokinase elevation as diagnosed by local physicians), hyperkalemia (defined as any serum potassium level >5.5 mmol/L), and hypertriglyceridemia (defined based on any elevation above thresholds of the local laboratory). Outcome measures included placement of a tracheostomy during the admission for RSE and the modified Rankin Scale at hospital discharge.
A questionnaire was provided to each study site PI to determine details of the practice setting (number of ICU beds), EEG monitoring capability (duration of monitoring and 24/7 availability to initiate monitoring), and treatment options (types of continuous intravenous ACMs [cIV-ACMs] and confidence in medication quality; refer to eAppendix 1 and 2, links.lww.com/WNL/C219, for survey questions and survey results).
Statistical Analysis
For the purposes of analysis, poor outcome was defined as a mRS score of 3–6. Patients were stratified into those recruited from hospitals within higher–middle-income (Brazil and Ecuador) and high-income (Austria, France, Taiwan, and the United States) economies based on the World Bank criteria.9 Variables were compared using the χ2 test for categorical variables and the Wilcoxon signed-rank test for continuous variables. Multivariate and univariate analyses were performed using logistic, ordinal, and negative binomial regression as appropriate. Associations between ordinal variables were tested using probability‐scale residual with or without confounders.10 All statistical tests were 2-tailed, and a p-value < 0.05 was considered statistically significant. Statistical analyses were performed using R statistical software version 4.0.2 (R Project for Statistical Computing).
Data Availability
Anonymized data not published within this article will be made available by request from any qualified investigator.
Results
Three hundred eighty-six distinct patients were enrolled from 9 centers and 6 countries recruited from higher–middle-income (e.g., Brazil and Ecuador) and high-income (e.g., Austria, France, Taiwan, and the United States) economies admitted between January 1, 2015, and December 31, 2018. Twenty-seven patients had no recorded ICU or hospital length of stay, 15 had no records of vasopressor administration, and 31 had no outcome data. One patient had 2 episodes of RSE separated by more than a month and was counted twice in the count for RSE episodes.
Clinical Characteristics
Baseline characteristics between RSE episodes treated with midazolam (N = 266) and those treated with propofol (N = 121) did not differ (Table 1), with an overall median age of 54 years (interquartile range [IQR] 33, 66), 41% being female, a median STESS of 3 (IQR 2, 4), and 30% (N = 116) having a medical history of seizures. The underlying etiology of RSE was acute in 79% (N = 306; traumatic brain injury 17%, systemic related 15%, CNS infection 13%, anoxic 11%, other 11%, epilepsy 9%, stroke 7%, subarachnoid hemorrhage 5%, intracerebral hemorrhage 5%, new-onset RSE 3%, tumors 3%, alcohol withdrawal seizure 2%, and degenerative 2%) and remote in 21% of the patients (N = 79; epilepsy 37%, brain tumor 20%, stroke 17%, traumatic brain injury 9%, intracerebral hemorrhage 5%, CNS infection 3%, systemic related 1%, alcohol withdrawal 1%, and other 8%). Focal status epilepticus was rare (N = 32, 8%) and more frequently treated with midazolam than propofol (88% vs 12%, OR 0.3 [95% CI 0.1–0.9]).
Table 1.
Baseline Characteristics and Management of Patients Included in Midazolam and Propofol Groups
Management
Half of the 387 RSE episodes were managed with continuous EEG (>24 hours; 193 [50%]), followed by prolonged EEG (1–24 hours; 98 [25%]), spot EEG (less than 1 hour; 75 [19%]), and clinical assessments without EEG (21 [5%], Table 2). The 21 patients managed without any EEG were predominantly from high-income countries (N = 21; 6 managed with midazolam and 15 with propofol, the median duration of midazolam or propofol is 1 day [IQR 1–2]). Clinical supervision was more common (OR 6.1 [95% CI 2.2–19.7]) and EEG less common (OR 0.5 [95% CI 0.3–0.9]) for patients treated with propofol. Patients with a higher predicted mortality based on the STESS score underwent a longer duration of EEG monitoring (p < 0.001, ρ = 0.28 [95% CI 0.26--0.30]). In addition to the initial benzodiazepine, patients received phenytoin/fosphenytoin (37% vs 47%), levetiracetam (27% vs 21%), valproic acid (22% vs 16%), lacosamide (14% vs 7%), and phenobarbital (1% vs 9%) before midazolam or propofol infusions, respectively.
Table 2.
Management and Clinical Outcomes of Patients With RSE by Drug Choice and Country
The median maximum doses of midazolam and propofol were 0.5 mg/kg/hr (IQR 0.2–2.0) and 37 mcg/kg/min (IQR 23–56), respectively (propofol doses converted into mg/kg/hr for comparison 1.4 mg/kg/hr [IQR 2.2–3.4]). Of the 266 patients, 70 patients (26%) initially managed with midazolam and 42 of the 121 patients (35%) initially treated with propofol required the addition of propofol or midazolam, respectively, as a second infusion anesthetic. The risk for the second-line cIV-ACM was marginally higher for patients initially treated with propofol after accounting for STESS (OR 1.5; 95% CI 1.0–2.4), which was particularly true when restricting the analysis to patients with convulsive SE (OR 3.1; 95% CI 1.7–5.6, eTable 2, links.lww.com/WNL/C219). Additional anesthetic drips were required in both, patients initially treated with midazolam and propofol infusions: ketamine 4% and 5%, other benzodiazepines 2% and 12% of the episodes initially treated with midazolam or propofol, respectively, and barbiturates in 3% episodes initially managed with midazolam. The following ACMs were continued or added during cIV-ACM treatment: levetiracetam (40% vs 44%), valproic acid (19% vs 26%), lacosamide (20% vs 25%), phenytoin/fosphenytoin (16% vs 23%), clobazam (15% vs 11%), topiramate (6% vs 9%), lamotrigine (5% vs 5%), and phenobarbital, clonazepam, pregabaline, zonisamide, gabapentine, and carbamazepine/oxcarbamazepine each in less than 5% for episodes managed with midazolam or propofol infusions, respectively.
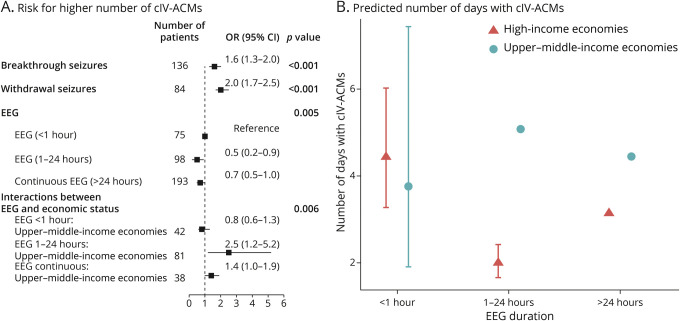
In a backward stepwise multivariate negative binomial regression model, stratified by midazolam and propofol, a higher number of cIV-ACM days were associated with breakthrough (OR 1.6, 95% CI 1.3–2.0) and withdrawal seizures (OR 2.0, 95% CI 1.7–2.5), and a lower number of days with prolonged EEG monitoring (1–24 hours OR 0.5, 95% CI 0.2–0.9, and >24 hours OR 0.7, 95% CI 0.5–1.0; reference EEG <1 hour; Figure 1). More prolonged EEG monitoring was more frequently available in high-income countries (p = 0.006), and the interaction between economic status and EEG monitoring type was associated with the number of days with cIV-ACMs (Figure 1A).
Figure 1. Predicting Days of cIV-ACMs.
Risk for a higher number of days with cIV-ACMs associated with more breakthrough and withdrawal seizures and a lower number of days with more prolonged EEG monitoring (A) most prominently seen for patients treated in high-income countries (B). cIV-ACMs = continuous intravenous anticonvulsant medications; MDZ = midazolam; PRO = propofol.
Breakthrough and withdrawal seizures were seen in 137 (41%) and 86 patients (26%), respectively. Both were observed with comparable frequency in patients managed initially with midazolam or propofol. At least 1 vasopressor was required in more than half of the patients treated with either propofol (54%) or midazolam (61%), but the number of vasopressors did not differ between the 2 treatment approaches. Adding a second anesthetic did not significantly increase the need or number of vasopressors.
Complications
Lactic acidosis, rhabdomyolysis, hyperkalemia, and hypertriglyceridemia (data available in 244 patients) were not significantly more common in either midazolam-treated or propofol-treated patients (eTable 3, links.lww.com/WNL/C219). Patients managed in upper–middle-income economies had a higher chance of developing the following complications while receiving either propofol or midazolam infusions for the treatment of RSE than those treated in high-income economies: lactic acidosis 48% vs 7% (OR 11.8, 95% CI 5.7–24.5; p < 0.0001), rhabdomyolysis (OR 13.4, 95% CI 6.1–29.4, p < 0.0001), and hyperkalemia (OR 4.2, 95% CI 1.4–12.2, p = 0.01). No difference was seen for the risk to develop hypertriglyceridemia.
Outcomes
Rates of tracheostomy (143 [39%]), and length of hospital (28 days [13–52 days]) or ICU stay (13 days [5–29 days]), did not differ between those treated with midazolam and propofol (Table 2). Prolonged EEG monitoring was associated with longer ICU stays (p = 0.004, ρ = 0.17 [95% CI 0.15–0.20]), but rates of tracheostomy or hospital length of stay did not differ. One hundred ten patients (28%) were dead at hospital discharge, and 44% (N = 48) of these patients underwent withdrawal of life-sustaining therapy (WLST). WLST was less common in patients treated with propofol than those with midazolam (7% vs 15%, OR 0.4 [95% CI 0.1–0.8]), even after taking STESSs into account, but overall mortality did not differ between patients treated with midazolam or propofol (midazolam 28% vs propofol 29%). Favorable functional outcomes (mRS 0–2) during hospital discharge were seen in 80 patients (21%) with comparable rates for patients treated with midazolam (30%) and propofol (39%; Table 2).
Economic Status
EEG in general and more prolonged monitoring in particular were less frequently available in upper–middle-income economies when compared with high-income countries (eTables 1–3, links.lww.com/WNL/C219). Although most medications used for the management of patients with RSE were available at all sites, two-thirds of practitioners in upper–middle-income countries voiced concern about the quality of medications that they had available to treat patients with RSE, whereas none of the practitioners from high-income countries were concerned about this. Although treatment of patients with status epilepticus started before hospital arrival in 83% of the sites from high-income countries, this is not practiced at any of the sites from upper–middle-income countries.
One hundred sixty-two patients (42%) with RSE were included from upper–middle-income and 225 (58%) from high-income economies. STESSs of patients from upper–middle-income and high-income countries did not differ. Patients from upper–middle-income countries were younger, less frequently had a seizure history, and more commonly had convulsive SE and less commonly nonconvulsive SE (Table 3). Patients from upper–middle-income countries underwent shorter duration of EEG monitoring, received less often multiple vasopressors, and had more often breakthrough and withdrawal seizures (Table 2). A second anesthetic infusion was more frequently required in patients managed in upper–middle-income compared with high-income countries (second infusion required 40% vs 21%, OR = 2.4 [95% CI 1.5–3.8]), which was particularly true when restricting the analysis to patients with convulsive SE only (eTable 2, links.lww.com/WNL/C219). Higher rates of death were seen for patients from upper–middle-income countries when compared with those from high-income countries after accounting for individual patient STESSs (OR 1.8 [95% CI 1.1–2.8]). Rates of WLST and functional outcomes among survivors did not differ between upper–middle-income and high-income countries (Table 2).
Table 3.
Basic Characteristics and Management of Patients Between High-Income and Upper-Middle-Income Economic Status
Classification of Evidence
This study provides Class III data that propofol and midazolam are equivalently efficacious for RSE.
Discussion
In this large, international, multicenter cohort study of RSE patients with predominantly acute underlying etiologies treated with propofol or midazolam infusions in the ICU, we observed comparable outcomes including efficacy and complications between the 2 anesthetic agents, with mortality rates that were higher in upper–middle-income and if anything lower in high-income economies than most prior series from high-income countries.11 Patients initially managed with propofol infusions were more likely to require the addition of a second anesthetic infusion (i.e., adding or switching to midazolam infusions) than those initially started on midazolam (i.e., adding or switching to propofol infusions). Despite minor differences in management including the use of continuous EEG monitoring and duration of anesthetic infusions, we did not find differences in seizure control or functional outcomes between the 2 medications, although it is important to recognize that drug effects cannot directly be compared because of lack of randomization. Recent nonrandomized case series have shown comparable rates of seizure control with midazolam or propofol.1,12,13 Outcomes are difficult to compare between practice settings because of differences in baseline characteristics of patients, etiology of status epilepticus, hospital management approaches, and postacute care support.3,4 Functional outcomes at discharge and mortality were worse for patients from upper–middle-income countries even after adjusting for STESS. Access to affordable monitoring techniques may be impactful in all practice settings.
Seizures in the critical care context are often electrographic only, particularly when used to monitor the treatment of RSE. Earlier observational cohort studies focused on the detection of seizures in unresponsive patients suggested that routine EEGs would miss a large portion of electrographic seizures14 particularly in comatose patients with acute brain injury.15 Screening the initial 30 minutes for epileptiform activity16 or preset serial short-duration studies17 may increase the pretest probability of detecting seizures. The cohort studied here offers a worldwide comparison of treatment practices for RSE patients with major differences in EEG utilization to monitor the management of RSE. The optimal duration of EEG monitoring is controversial to this date. In this study, half of the cohort underwent continuous EEG for more than 24 hours (50%), followed by prolonged EEG (1–24 hours; 25%), spot EEG (less than 1 hour; 19%), and clinical assessments without EEG (5%). Although sicker patients (as per the STESS) underwent more prolonged monitoring, shorter administration of midazolam and propofol was seen for those with longer monitoring. This observation was made particularly in high-income countries, supporting the notion of a more targeted RSE management approach.
Slightly less than half (42%) of the patients were recruited from upper–middle-income countries, while the remainder (58%) originated from high-income countries. STESSs were originally developed on cohorts recruited from high-income countries8 but have since been validated in the context of upper–middle-income countries.18,19 These patients also more frequently developed complications during infusion of midazolam or propofol when compared with those from high-income economies including lactic acidosis, rhabdomyolysis, and hyperkalemia. These were seen equally frequent in patients treated with midazolam or propofol and are likely a reflection of the overall population illness and critical care management rather than specific to the choice of RSE treatments. Although STESSs did not differ between the 2 groups, higher mortality and worse functional outcomes were seen for patients from upper–middle-income countries suggesting that treatment practices may provide additional opportunities to improve outcomes. Potential avenues include starting status epilepticus treatment before hospital arrival, shortening the duration of cIV-ACMs possibly supported by more prolonged EEG monitoring more rapidly, and assuring more certainty about the quality of medications used for the management of patients with RSE. However, it is important to stress that patients from upper–middle-income countries were younger, less frequently had a seizure history, and more commonly had convulsive SE and less commonly nonconvulsive SE. Care of patients with RSE is expensive and represents a major burden for healthcare systems worldwide.1,2 Costs associated with higher medication utilization vs availability of monitoring technology need to be further investigated in future cost-effectiveness studies. To determine efficacy, future studies may explore if deeper but shorter duration of therapeutic coma compared with more prolonged infusions would be associated with less complications and shorter length of stay.12 The World Bank classification scheme is based on the total amount of money earned by a nation's people and is also known as the gross national income (GNI). In the presented study, we chose this classification scheme because GNI despite known limitations20 is widely seen as a proxy measure for a country's ability to provide financial means for health services to its population.21,22
Findings reported here are only applicable for RSE episodes treated in the ICU. Several limitations are worth mentioning, most importantly that these data are not the result of a randomized controlled clinical trial and comparisons between treatments or management approaches will therefore be limited. The limitations of the retrospective review do also limit accurate assessments of complications. In the absence of adequately powered randomized controlled studies,23 a large case series such as this one may provide some guidance for planning of future trials and reflect current practice. Second, interpreting findings based on data lumping high-income with upper–middle-income countries has to be made with reservation. We noted a lack of prehospital treatment in upper–middle-income countries suggesting a potentially impactful approach to improve care of patients with status epilepticus.24 Adequately controlling for severity of illness is challenging, and the STESS that was developed as a short-term predictor of mortality does not comprehensively control for etiologies and for severity of the etiology.8 Many additional differences including types and severity of status epilepticus etiologies and baseline illness, access to rehabilitation services, and other unmeasured socioeconomic differences need to be considered. Third, we did not detect any patients who met clinical criteria for propofol infusion syndrome, but this being a clinical diagnosis, limitations of the retrospective data collection need to be acknowledged. In general, our data collection does have limited information on side effects and complications. Fourth, we were not able to get higher-level long-term outcome measures because of the study design. Fifth, prolonged EEG monitoring for more than 24 hours was most frequently seen in patients from high-income countries, while clinical observation and spot EEGs were more commonly the monitoring practice in patients from upper–middle-income countries. This needs to be taken into account when interpreting the results because RSE is mostly nonconvulsive and may go unnoticed by clinical observation alone. However, it is unclear why the monitoring was associated with improved outcomes because this may represent a surrogate marker for unmeasured practice differences between those centers that had and those that did not have prolonged monitoring readily available. Sixth, all included sites from higher–middle-income countries were located in South America, and it cannot be excluded that detected signals are unique to these countries or the South American continent. Countries were stratified based on GNI because this measure has been linked to investments into healthcare systems, but it is important that alternate classification schemes based on the development status such as those provided by the United Nations may equally apply. Seventh, we are unable to provide information on time to initiating treatment of RSE. Finally, drug doses need to be interpreted with caution because medication impurities have been reported,25 particularly for drugs marketed in upper–middle-income countries. In this context, EEG end points rather than drug dosing may be particularly useful to guide RSE management.
The results of this large, international, multicenter cohort study suggest that optimizing prehospital treatment and goal-directed management protocols including prolonged EEG monitoring may offer opportunities for patients with RSE. Comparable outcomes were observed for RSE patients with predominantly acute underlying etiologies treated with propofol or midazolam infusions.
Glossary
- cIV-ACMs
continuous intravenous anticonvulsant medications
- GNI
gross national income
- ICU
intensive care unit
- IRB
institutional review board
- mRs
modified Rankin Scale
- RSE
refractory status epilepticus
- STESS
Status Epilepticus Severity Score
- WLST
withdrawal of life-sustaining therapy
Appendix. Authors

Footnotes
Class of Evidence: NPub.org/coe
Study Funding
No targeted funding reported.
Disclosure
S. Demeret thanks the iCRIN program funded by “Investissements d'Avenir” ANR-10-IAIHU-06 and the Paris Brain Institute (ICM) that provided local research assistant support for this work. J. Claassen reports grants during the conduct of the study from the James S. McDonnell Foundation, reports grants from the National Institute of Neurological Disorders and Stroke, and is a minority shareholder of iCE Neurosystems. All these do not directly apply to the submitted work. The other authors declare no relevant disclosures. Go to Neurology.org/N for full disclosures.
References
- 1.Shorvon S, Ferlisi M. The treatment of super-refractory status epilepticus: a critical review of available therapies and a clinical treatment protocol. Brain. 2011;134(pt 10):2802-2818. pubmed.ncbi.nlm.nih.gov/21914716/. [DOI] [PubMed] [Google Scholar]
- 2.Narayanan JT, Murthy JMK. Nonconvulsive status epilepticus in a neurological intensive care unit: profile in a developing country. Epilepsia. 2007;48(5):900-906. pubmed.ncbi.nlm.nih.gov/17433050/. [DOI] [PubMed] [Google Scholar]
- 3.Ferlisi M, Hocker S, Trinka E, Shorvon S, International Steering Committee of the StEp Audit. The anesthetic drug treatment of refractory and super-refractory status epilepticus around the world: results from a global audit. Epilepsy Behav. 2019;101(pt B):106449. pubmed.ncbi.nlm.nih.gov/31420291/. [DOI] [PubMed] [Google Scholar]
- 4.Silva GS, Maldonado NJ, Mejia-Mantilla JH, et al. , Galapagos Neurocritical Care Summit Investigators. Neuroemergencies in South America: how to fill in the gaps? Neurocrit Care. 2019;31(3):573-582. pubmed.ncbi.nlm.nih.gov/31342447/. [DOI] [PubMed] [Google Scholar]
- 5.Venkatasubba Rao CP, Suarez JI, Martin RH, et al. , PRINCE Study Investigators. Global survey of outcomes of neurocritical care patients: analysis of the PRINCE study part 2. Neurocrit Care. 2020;32(1):88-103. pubmed.ncbi.nlm.nih.gov/31486027/. [DOI] [PubMed] [Google Scholar]
- 6.von Elm E, Altman DG, Egger M, Pocock SJ, Gøtzsche PC, Vandenbroucke JP, STROBE Initiative. The strengthening the reporting of observational studies in epidemiology (STROBE) statement: guidelines for reporting observational studies. Int J Surg. 2014;12(12):1495-1499. pubmed.ncbi.nlm.nih.gov/25046131/. [DOI] [PubMed] [Google Scholar]
- 7.Rossetti AO, Lowenstein DH. Management of refractory status epilepticus in adults: still more questions than answers. Lancet Neurol. 2011;10(10):922-930. pubmed.ncbi.nlm.nih.gov/21939901/. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Rossetti AO, Logroscino G, Milligan TA, Michaelides C, Ruffieux C, Bromfield EB. Status Epilepticus Severity Score (STESS): a tool to orient early treatment strategy. J Neurol. 2008;255(10):1561-1566. pubmed.ncbi.nlm.nih.gov/18769858/. [DOI] [PubMed] [Google Scholar]
- 9.World Bank Country and Lending Groups–World Bank Data Help Desk. Accessed March 13, 2021. datahelpdesk.worldbank.org/knowledgebase/articles/906519. [Google Scholar]
- 10.Li C, Shepherd BE. Test of association between two ordinal variables while adjusting for covariates. J Am Stat Assoc. 2010;105(490):612-620. /pmc/articles/PMC2946253/. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Delaj L, Novy J, Ryvlin P, Marchi NA, Rossetti AO. Refractory and super-refractory status epilepticus in adults: a 9-year cohort study. Acta Neurol Scand. 2017;135(1):92-99. onlinelibrary.wiley.com/doi/full/10.1111/ane.12605. [DOI] [PubMed] [Google Scholar]
- 12.Muhlhofer WG, Layfield S, Lowenstein D, et al. Duration of therapeutic coma and outcome of refractory status epilepticus. Epilepsia. 2019;60(5):921-934. pubmed.ncbi.nlm.nih.gov/30957219/. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Claassen J, Hirsch LJ, Emerson RG, Bates JE, Thompson TB, Mayer SA. Continuous EEG monitoring and midazolam infusion for refractory nonconvulsive status epilepticus. Neurology. 2001;57(6):1036-1042. ncbi.nlm.nih.gov/pubmed/11571331. [DOI] [PubMed] [Google Scholar]
- 14.Pandian JD, Cascino GD, So EL, Manno E, Fulgham JR. Digital video-electroencephalographic monitoring in the neurological-neurosurgical intensive care unit: clinical features and outcome. Arch Neurol. 2004;61(7):1090-1094. pubmed.ncbi.nlm.nih.gov/15262740/. [DOI] [PubMed] [Google Scholar]
- 15.Claassen J, Mayer SA, Kowalski RG, Emerson RG, Hirsch LJ. Detection of electrographic seizures with continuous EEG monitoring in critically ill patients. Neurology. 2004;62(10):1743-1748. ncbi.nlm.nih.gov/pubmed/15159471. [DOI] [PubMed] [Google Scholar]
- 16.Shafi MM, Westover MB, Cole AJ, Kilbride RD, Hoch DB, Cash SS. Absence of early epileptiform abnormalities predicts lack of seizures on continuous EEG. Neurology. 2012;79(17):1796-1801. pubmed.ncbi.nlm.nih.gov/23054233/. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Alvarez V, Sierra-Marcos A, Oddo M, Rossetti AO. Yield of intermittent versus continuous EEG in comatose survivors of cardiac arrest treated with hypothermia. Crit Care. 2013;17(5):R190. pubmed.ncbi.nlm.nih.gov/24007625/. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.El-Tamawy MS, Amer H, Kishk NA, et al. External validation of STESS and EMSE as outcome prediction scores in an Egyptian cohort with status epilepticus. Epilepsy Behav. 2020;102:106686. pubmed.ncbi.nlm.nih.gov/31760201/. [DOI] [PubMed] [Google Scholar]
- 19.Zhang Y, Chen D, Xu D, Tan G, Liu L. Clinical utility of EMSE and STESS in predicting hospital mortality for status epilepticus. Seizure. 2018;60:23-28. pubmed.ncbi.nlm.nih.gov/29906706/. [DOI] [PubMed] [Google Scholar]
- 20.Stiglitz J, Sen AFJ. Mismeasuring Our Lives: Why GDP Do’sn't Add up. A Report by the Commission on the Measurement of Economic Performance and Social Progress: The New Press; 2010. [Google Scholar]
- 21.Preston SH. Dynamic equilibrium of economic and demographic transition, economic development and cultural change. Am Econ Rev. 1961;76:203-232. [Google Scholar]
- 22.Pritchett LSL. Wealthier is healthier. J Hum Resour. 1996;31:841-868. [Google Scholar]
- 23.Rossetti AO, Milligan TA, Vulliémoz S, Michaelides C, Bertschi M, Lee JW. A randomized trial for the treatment of refractory status epilepticus. Neurocrit Care. 2011;14(1):4-10. pubmed.ncbi.nlm.nih.gov/20878265/. [DOI] [PubMed] [Google Scholar]
- 24.Alldredge BK, Gelb AM, Isaacs SM, et al. A comparison of lorazepam, diazepam, and placebo for the treatment of out-of-hospital status epilepticus. N Engl J Med. 2001;345(9):631-637. pubmed.ncbi.nlm.nih.gov/11547716/. [DOI] [PubMed] [Google Scholar]
- 25.Janvier S, De Spiegeleer B, Vanhee C, Deconinck E. Falsification of biotechnology drugs: current dangers and/or future disasters? J Pharm Biomed Anal. 2018;161:175-191. pubmed.ncbi.nlm.nih.gov/30165334/. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
Anonymized data not published within this article will be made available by request from any qualified investigator.