Abstract
Background and Aims
Olfactory dysfunction is a recognized manifestation in patients infected with Coronavirus Disease 2019 (COVID‐19). This investigation aimed to assess the effect of mometasone furoate intranasal spray on the improvement of smell dysfunction in post‐COVID‐19 patients.
Materials and Methods
This randomized placebo‐controlled trial included 80 non‐hospitalized adult patients who had persistent anosmia or severe microsmia for more than 4 weeks due to COVID‐19 infection. The participants were randomly allocated to the intervention or placebo group to receive mometasone furoate nasal spray or sodium chloride intranasal spray during 4 weeks of follow‐up, respectively. The patients' olfactory dysfunction was assessed in terms of visual analog scale (VAS), and smell test score according to the modified version of the University of Pennsylvania smell identification test for the Iranian population.
Results
A total of 70 participants completed the follow‐up period and were analyzed in this study. By comparing the olfactory scores including smell test and VAS scores, no significant differences were found between case and control groups at baseline, 2, and 4 weeks intervals. However, the change of both olfactory scores at pre to post‐treatment intervals and 2–4 weeks was significantly higher in the mometasone group relative to the placebo group. At post‐treatment, the frequency of anosmia was 22.9% reduced in the case group compared to the control group.
Conclusion
Overall, there was no significant difference in olfactory dysfunction between the two groups during follow‐up. However, based on the significant between‐group difference in terms of olfactory scores changes, it seems that the nasal corticosteroids may be a positive effect on the recovery process of patients who received more than 2 weeks.
Level of Evidence
II Laryngoscope, 2022
Keywords: COVID‐19, intranasal corticosteroid, olfactory dysfunction, UPSIT, VAS score

Olfactory dysfunction is a recognized manifestation in patients infected with COVID‐19. This investigation is aimed to assess the effect of mometasone furoate intranasal spray on the improvement of smell dysfunction in post‐COVID‐19 patients.
INTRODUCTION
The severe acute respiratory syndrome coronavirus 2 (SARS‐CoV‐2) was firstly reported in Wuhan, China. 1 By January 2022, SARS‐CoV‐2 had infected more than 334 million people across countries/regions and killed more than 5,596,000 people 2 as a pandemic declared by the WHO. 3 Prevalent symptoms at the onset of the sickness are cough, fever, fatigue, and myalgia. 4 , 5 Anosmia and ageusia can represent the first symptoms manifested in people who have been infected with Coronavirus Disease 2019 (COVID‐19), especially for those who are asymptomatic. 6 These minor and nonspecific symptoms have been warned as strong biomarkers of COVID‐19 infection in combination with other symptoms. 7 , 8
The smell sense is the main to our link to food, avoidance behavior, and warning response to hazardous chemicals. 9 Overall, the life quality is slightly dependent on smell ability. 10 A viral upper airway infection may cause permanent anosmia. Viral infections are responsible for almost 20% of all anosmia cases. 11 As it is obvious, the neuroepithelium embedded in the nasal mucosa is damaged directly by the microorganisms or the pathogenesis involving immunological processes. 12 , 13
Although numerous studies have been conducted on COVID‐19, there is no distinct antiviral treatment approved for this condition. Steroid compounds effect on blocking both coronavirus replication and host inflammation. 14 Corticosteroids have a wide range of effects 15 and anti‐inflammatory as well as immunosuppressive features which are used in clinical contexts. Local effects include membrane stabilization, inhibition of cell migration, or alteration of mediator release. 15 , 16 These mechanisms might have a role in the rehabilitation of the olfactory with an effect on the olfactory mucosa. 15
According to the previous studies, there was a controversy concerning the effect of topical nasal corticosteroids on olfactory dysfunction of patients with anosmia‐related COVID‐19. A recent investigation demonstrated that the corticosteroid nasal spray could be useful to improve olfactory loss due to COVID‐19. 17 , 18 However, other researchers reported that intranasal corticosteroids did not affect the recovery time of anosmia due to COVID‐19. 19 , 20 , 21 Hence, some studies recommended further clinical trials will require to be undertaken. 20 , 22
The second‐generation nasal corticosteroid agents are commonly in use, such as mometasone furoate nasal spray that has pharmacokinetic characteristics minimizing their systemic bioavailability (<1%), compared to both older intranasal corticosteroid (INC) and oral agents, thereby minimizing the risk of systemic adverse events. 15 With each dose of a second‐generation INC, around 30% is deposited in the nose and attached to the glucocorticoid receptor. The remaining (70%) enter first‐pass hepatic metabolism. 23
To date, the therapy of anosmia due to COVID‐19 infection remained controversial. Hence, a more careful study of the association between COVID‐19 and olfactory loss is crucial to developing clinical treatment as well as cure rates. The present randomized clinical trial (RCT) aimed to evaluate the therapeutic effect of mometasone furoate nasal spray usage for 1 month to improve the symptoms of smell dysfunction due to COVID‐19 relative to the placebo group.
MATERIALS AND METHODS
Study Design
This double‐blind, two‐arm, parallel‐group, randomized, placebo‐controlled trial was accomplished to assess the effect of INCs on the improvement of olfactory loss in non‐hospitalized COVID‐19 patients. The study protocol was confirmed by the Ethics Committee of Mashhad University of Medical Sciences (Ethical code: IR.MUMS.REC.1399.355). Additionally, our trial study was approved in the Iranian registry of clinical trials (IRCT20200522047542N1).
Characteristics of Patients
The present trial included subjects aged 18 years or older with a definitive diagnosis of COVID‐19 infection and also persistent olfactory dysfunction between 30 to 90 days who were referred to hospital clinics of Mashhad University of Medical Sciences, from April to July 2020. The COVID‐19 patients were diagnosed using their clinical presentation, laboratory data, and a positive reverse transcription‐polymerase chain reaction. The non‐hospitalized individuals who had anosmia or microsmia due to COVID‐19 infection for 30–90 days were enrolled in this study. A complete clinical examination was performed for all patients. Participants who received any corticosteroid therapy at least 3 months before this study were excluded. Additionally, subjects who experienced smell loss before the COVID‐19 infection, patients with pregnancy, chronic rhinosinusitis, polyposis, systemic or topical corticosteroid nasal consumption, trauma history, Alzheimer's, Parkinson's, allergy, asthma, and surgery, subjects with other viral or bacterial co‐infections, and the subjects who denied to participate in follow‐up measurements, give consent, or provide data were also excluded from the study.
Sample Size
The sample size was determined based on the alpha error of 0.05 and a power of 80% (beta error = 0.2), using the two population proportion formula. The prevalence of anosmia was 79% in COVID‐19 patients, 24 and by considering the reduction of anosmia prevalence to 45% in the intervention group, the calculated sample size was 31 subjects in each group. Finally, 40 people in each group were included in the present study.
Data Collection
Individuals interested in participating in this study were evaluated for eligibility, and they were then enrolled in our study and randomly assigned to intervention and placebo groups. The demographic characteristics, medical history, symptoms, duration of olfactory dysfunction to involvement, and outcomes data were recorded in this trial.
Study Setting and Interventions
The individuals in our study were randomly allocated to the case and control groups based on block randomization. The randomized list was generated using a computer‐generated code in the block method with a block size equal to four. The patients and the physician were both completely blinded and the assignment and the randomization were performed by a third person. Furthermore, the spray bottles were made as much identical as possible to minimize biases.
The participants in the case group received one puff of 0.05% wt/vol mometasone furoate (Raha Company, Iran) intranasal spray on each side twice per day for 4 weeks. On the other hand, one puff of 0.65% wt/vol sodium chloride nasal spray on each side (Decosalin, Raha Company, Iran) was administered to the patients in the placebo group twice daily for 4 weeks. The side effects of corticosteroid therapy were recorded in the present trial.
Primary Outcomes
Different types of smell tests have been designed in various countries according to cultural adaption. The Iran Smell Identification Test (Iran‐SIT) is an altered version of the University of Pennsylvania smell identification test (UPSIT) for the Iranian residents used in this analysis. 24‐item Iran‐SIT has sufficiency to categorize adult patients in different levels as follows: subjects with normal smell function (normosmia, 19–24), ones who had mild dysfunction (mild microsmia, 14–18), ones with severely reduced smell function (severe microsmia, 10–13), and patients with the olfactory loss (anosmia, 0–9).
The patients were requested to scrape the stickers using the tip of a pen or pencil for releasing the odors. They were motivated to smell immediately the mentioned sticker as well as select one of four options. They were asked to indicate the answer nearest to their understanding if they declared that the odor they smelled was not presented in the choices. The items of a corrected answer were considered as an Iran‐SIT score.
At the same time, patients' olfactory dysfunction was evaluated based on a visual analog scale (VAS) with a score ranging from 0 as the worst to 10 as the best. Participants were followed‐up to evaluate smell function with both subjective and objective tests on the first day (baseline), 14, and 28 days after treatment. Additionally, the decrease of anosmia frequency was assessed at the end‐point of the study.
Statistical Analysis
All data were analyzed using SPSS software version 22. The qualitative variables were described using frequency (%) and compared by Chi‐square or Fisher's exact test. The quantitative variables were checked using the Kolmogorov–Smirnov test to determine the normal distribution of mentioned data. Accordingly, the variables with or without normal distribution were reported as the mean ± standard deviation, or median (percentile 25–75), respectively. The Unpaired sample t‐test or Mann–Whitney test was performed to compare quantitative variables between placebo and intervention groups. Additionally, the Wilcoxon signed‐rank test was performed to assess repeated measurements (0, 2, and 4 weeks) within the group. The p‐value <0.05 was considered significant.
RESULTS
Between April 1 and December 30, 2020, 503 subjects were assessed for eligibility, and 80 patients were included in the present study. The participants were randomly divided into the case and control groups to receive mometasone furoate or sodium chloride nasal spray for 1 month, respectively. Five subjects in each group were excluded through the period of the follow‐up, and 35 patients were analyzed in every group to evaluate the recovery of olfactory dysfunction (Fig. 1). No side effects were noted in the placebo and intervention groups of the study.
Fig. 1.
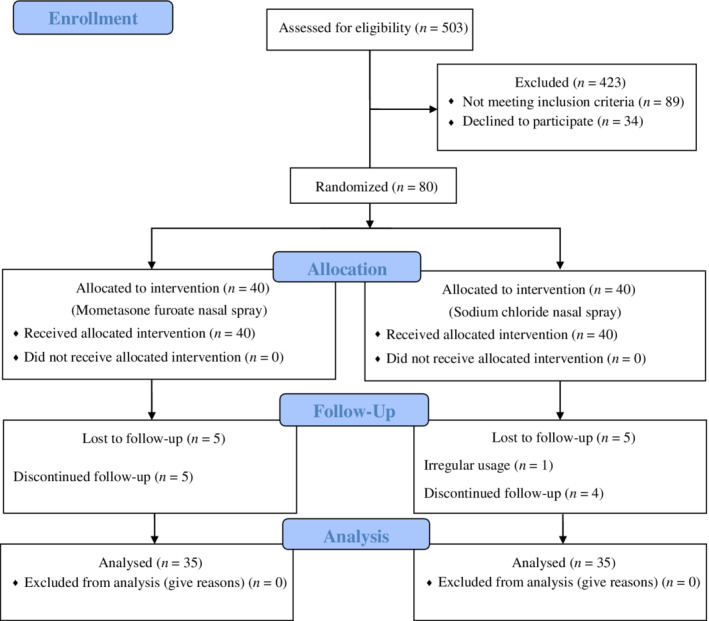
Study CONSORT diagram. [Color figure can be viewed in the online issue, which is available at www.laryngoscope.com.]
According to Table I, the mean age was 34.93 ± 12.39 and 32.23 ± 10.02 years in the placebo and intervention groups, respectively, with no significant difference between the two groups (p = 0.343). The duration of olfactory dysfunction of subjects before intervention was 56.72 ± 14.71 days in the placebo group, and 57.71 ± 17.22 days in the case group (p = 0.795). 60% and 68.5% of patients were female in the placebo and case groups, respectively (p = 0.618). No significant differences were observed in terms of smoking, and alcohol or drug addiction between groups.
TABLE I.
The Comparison of Demographics and COVID‐19‐Associated Symptoms Between Control (Placebo) and Case (Intervention) Groups.
| Variable | Placebo (n = 35) | Intervention (n = 35) | p‐Value | ||
|---|---|---|---|---|---|
| Mean ± SD | Frequency (%) | Mean ± SD | Frequency (%) | ||
| Age | 34.93 ± 12.39 | 32.23 ± 10.02 | 0.343 | ||
| Gender | |||||
| Female | 21 (60.0) | 24 (68.5) | 0.618 | ||
| Male | 14 (40.0) | 11 (31.5) | |||
| Duration of olfactory dysfunction to involvement, days | 56.72 ± 14.71 | 57.71 ± 17.22 | 0.795 | ||
| Cigarette smoking | 3 (8.5) | 5 (14.3) | 0.710 | ||
| Hookah smoking | 1 (2.8) | 5 (14.3) | 0.198 | ||
| Drug addiction | 1 (2.8) | 1 (2.8) | 1.000 | ||
| Alcohol addiction | 0 (0.0) | 4 (11.4) | 0.114 | ||
| Comorbidities | |||||
| Diabetes | 4 (11.4) | 2 (5.7) | 0.673 | ||
| Hypertension | 4 (11.4) | 1 (2.8) | 0.356 | ||
| Asthma | 0 (0.0) | 0 (0.0) | 1.000 | ||
| Autoimmune disorder | 0 (0.0) | 0 (0.0) | 1.000 | ||
| Respiratory failure | 0 (0.0) | 0 (0.0) | 1.000 | ||
| Cardiovascular disorder | 2 (5.7) | 1 (2.8) | 1.000 | ||
| Liver dysfunction | 0 (0.0) | 0 (0.0) | 1.000 | ||
| Renal dysfunction | 1 (2.8) | 1 (2.8) | 1.000 | ||
| Covid‐19 symptoms | |||||
| Headache | 19 (54.3) | 19 (54.3) | 1.000 | ||
| Fever | 27 (77.1) | 9 (25.7) | <0.001 | ||
| Cough | 24 (68.5) | 11 (31.5) | 0.004 | ||
| Hemoptysis | 4 (11.4) | 8 (22.8) | 0.342 | ||
| Sore throat | 5 (14.3) | 4 (11.4) | 1.000 | ||
| Nausea, vomiting | 2 (5.7) | 7 (20.0) | 0.151 | ||
| Dyspnea | 12 (34.3) | 9 (25.7) | 0.603 | ||
| Nasal congestion | 8 (22.8) | 9 (25.7) | 1.000 | ||
| Sneezing | 10 (28.6) | 9 (25.7) | 1.000 | ||
| Eye redness | 3 (8.5) | 1 (2.8) | 0.614 | ||
| Rhinorrhea | 5 (14.3) | 5 (14.3) | 1.000 | ||
| Parosmia | 5 (14.3) | 14 (40.0) | 0.030 | ||
| Dysgeusia | 21 (60.0) | 19 (54.3) | 0.809 | ||
COVID‐19 = coronavirus disease 2019; SD = standard deviation.
Additionally, there were no significant differences as regards diabetes (p = 0.673), hypertension (p = 0.356), asthma (p = 1.000), cardiovascular disease (p = 1.000), and renal dysfunction (p = 1.000) between both groups. None of the participants had autoimmune diseases, respiratory failure, and liver dysfunction. Based on the results of Table I, dysgeusia (57.1%), headache (54.3%), fever (51.4%), and cough (50%) were the most common symptoms during COVID‐19 infection in enrolled subjects.
Table II is depicted the olfactory scores according to the smell test and VAS in the follow‐up period. By comparing the median scores of smell test based on Iran‐SIT, no significant differences were determined between the case and placebo groups at the beginning of the study (p = 0.953), 2 weeks (p = 0.827), and 4 weeks after treatment (p = 0.120). Additionally, there were no statistically significant differences in median VAS scores between groups at the mentioned times (p = 0.353, 0.280, and 0.076, respectively). However, statistically significant differences were observed in terms of smell test and VAS scores within the placebo or intervention group relative to the baseline (p ≤ 0.001).
TABLE II.
Comparison of Olfactory Score Based on Smell Test (Iran‐SIT) and VAS Score Between and Within the Case and Control Groups
| Variable | Placebo | Intervention | p‐Value* | p‐Value † | p‐Value ‡ |
|---|---|---|---|---|---|
| Median (25th–75th) | Median (25th–75th) | ||||
| Baseline smell test score | 4 (1–14) | 4 (1–12) | 0.953 | ‐ | ‐ |
| Smell test score after 2 weeks | 8 (4–17) | 8 (4–18) | 0.827 | <0.001 | <0.001 |
| Smell test score after 4 weeks | 10 (6–23) | 19 (10–22) | 0.120 | <0.001 | <0.001 |
| Baseline VAS score | 1 (0–5) | 1 (0–5) | 0.353 | ‐ | ‐ |
| VAS score after 2 weeks | 3 (2–8) | 3 (2–6) | 0.280 | 0.001 | <0.001 |
| VAS score after 4 weeks | 4 (3–9) | 7 (5–9) | 0.076 | <0.001 | <0.001 |
Iran‐SIT = Iran Smell Identification Test; VAS = Visual Analog Scale.
Between groups.
Within the placebo group.
Within the intervention group.
The median (25th–75th) scores of the smell test were determined 4 (1–14) and 10 (6–23) in the control group at the baseline and endpoint of the study versus 4 (1–12) and 19 (10–22) in the case group, respectively (Fig. 2A and Table II). Furthermore, the median VAS scores were 1 (0–5) and 1 (0–5) in the control and case groups at the pre–treatment compared to 4 (3–9) and 7 (5–9) at the post‐treatment (Fig. 2B and Table II).
Fig. 2.
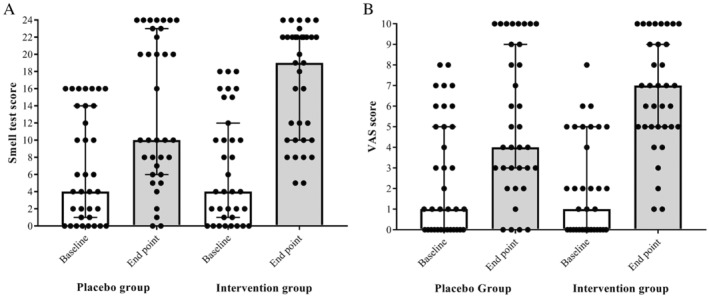
Comparison of olfactory scores between placebo and case groups at the pre and post‐intervention. Smell test score based on UPSIT (A) and VAS score (B). UPSIT = University of Pennsylvania smell identification test; VAS = visual analog scale.
As shown in Table III, the changes observed in olfactory scores based on the smell test and VAS were significant between groups at 0–4 weeks and 2–4 weeks intervals (p ≤ 0.001). Our results revealed that the mean change of smell identification test and VAS scores were 6.57 ± 3.62 and 2.66 ± 2.26 in the placebo group after 4 weeks, versus 10.08 ± 4.22, and 4.66 ± 2.36 in the case group, respectively. According to the olfactory dysfunction types of participants, anosmia and normosmia were reported at 40% and 42.8% in the placebo group after 4 weeks versus 17.1% and 54.3% in the case group, respectively (Table IV, and Fig. 3). At the endpoint of the study, the frequency of anosmia was 22.9% reduced in the case group relative to the placebo group. However, no significant differences were observed between groups regarding the severity of smell dysfunction during the follow‐up period.
TABLE III.
Assessment of the Changes in Smell Test (Iran‐SIT) and VAS Scores Between Two Groups During Follow‐Up.
| Variable | Placebo | Intervention | p‐Value |
|---|---|---|---|
| Smell test score initial and after 2 weeks | 2.88 ± 2.81 | 3.31 ± 2.53 | 0.505 |
| Smell test score initial and after 4 weeks | 6.57 ± 3.62 | 10.08 ± 4.22 | <0.001 |
| Smell test score after 2 and 4 weeks | 3.68 ± 2.68 | 6.77 ± 3.64 | <0.001 |
| VAS score initial and after 2 weeks | 2.05 ± 2.50 | 1.73 ± 1.55 | 0.619 |
| VAS score initial and after 4 weeks | 2.66 ± 2.26 | 4.66 ± 2.36 | 0.001 |
| VAS score after 2 and 4 weeks | 0.48 ± 0.75 | 2.27 ± 2.09 | 0.001 |
Variables were expressed as the mean ± standard deviation (SD).
Iran‐SIT = Iran Smell Identification Test; VAS = Visual Analog Scale.
TABLE IV.
Evaluation of the Olfactory Dysfunction Types Such as Anosmia, and Severe or Mild Microsmia Based on Smell Test Between Two Groups in the Follow‐Up Period.
| Variable | Placebo | Intervention | p‐Value |
|---|---|---|---|
| Frequency (%) | Frequency (%) | ||
| Baseline | 1.000 | ||
| Anosmia | 21 (60.0) | 22 (62.8) | |
| Severe | 4 (11.4) | 4 (11.4) | |
| Mild | 10 (28.6) | 9 (25.7) | |
| After 2 weeks | 0.986 | ||
| Anosmia | 19 (54.3) | 19 (54.3) | |
| Severe | 2 (5.7) | 2 (5.7) | |
| Mild | 7 (20.0) | 8 (22.8) | |
| Normosmia | 7 (20.0) | 6 (17.1) | |
| After 4 weeks | 0.172 | ||
| Anosmia | 14 (40.0) | 6 (17.1) | |
| Severe | 5 (14.3) | 7 (20.0) | |
| Mild | 1 (2.8) | 3 (8.6) | |
| Normosmia | 15 (42.8) | 19 (54.3) |
Fig. 3.
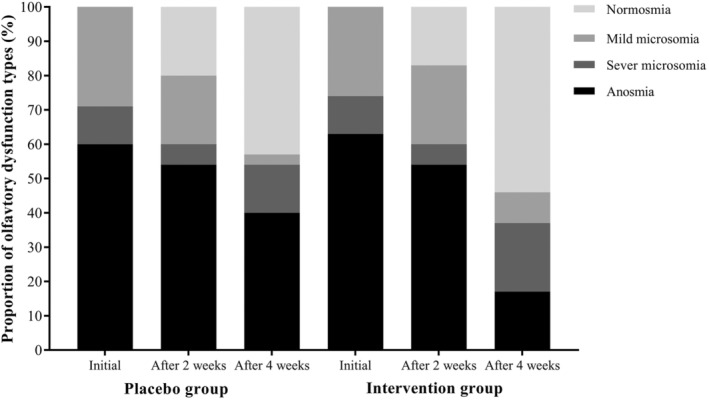
The percentage of olfactory dysfunction types in the case and control groups during the follow‐up period.
DISCUSSION
In the current study, we assessed the improvement of patients who suffered from olfactory dysfunction after COVID‐19 infection. Although the olfactory scores according to the VAS and smell test were not significant in the case group relative to the placebo group, significant differences were found between the two groups in terms of score changes based on VAS and UPSIT at pre and post‐treatment. Additionally, there were significant differences in terms of mentioned scores within the placebo or intervention groups during the follow‐up period.
Olfactory dysfunction is prevalent in the mild severity of COVID‐19 patients. Even though acute smell loss was self‐reported by over a third of the infected patients, 25 it is suggested that it could be an early symptom associated with COVID‐19. 26 However, the accurate mechanism of olfactory disorder is yet to be identified and, there is no acceptable anosmia treatment. Nevertheless, the majority of patients who present with anosmia appear to improve entirely after a few weeks, and a slight proportion appears to exhibit persistent smell dysfunction with problematic clinical manifestations including phantosmia or parosmia. 27
Recent evidence suggested the medical management for olfactory dysfunction such as systemic and topical steroids, traditional Japanese medicine, zinc, vitamin A, alpha‐lipoic acid, as well as theophylline, minocycline, and acupuncture. 28 , 29 Therefore, the present RCT study was evaluated to define the efficiency of mometasone furoate on the recovery of smell dysfunction in post‐COVID‐19 patients. Several kinds of research have demonstrated that the pathogenesis of olfactory dysfunction is related to atrophy of the olfactory mucosa, inflammatory neuropathy, olfactory nerve damage, and epithelial dysfunction in cases with long‐term anosmia. 30 , 31
A clear young female predominance was observed in our participants, which is also supported by previously conducted studies. The mean age of patients in our study was 32.23 ± 10.02 and 34.93 ± 12.39 in the case and placebo groups, respectively. Additionally, no statistical differences were determined in terms of gender and age between the mentioned groups. Giacomelli et al. examined 59 hospitalized cases with COVID‐19 who reported smell or taste disturbance (34%). Furthermore, females (52.6%) illustrated olfactory and taste disorders more frequently than males (25%) (p = 0.03). 32
Similarly, in a meta‐analytic study, notable gender differences in terms of odor identification were shown in younger adults (aged 18–50 years) not older participants (>50 years). 33 In a cohort of 42 participants with mild COVID‐19, it was observed that subjects younger than 40 years improved olfaction more quickly compared to those who were older than 40 years. 25 In a study carried out in Iran, the significance of smell dysfunction measured with UPSIT was equal in males and females. 8 They demonstrated that the specificity and sensitivity of smell tests for COVID‐19 patients below the age of 65 would appear to be strong because age‐related differences regarding olfactory function mainly occur over the age of 65. 8
Given that the median time from the onset of symptoms varies from 8 to 12 days in the studies, oral steroids should not be prescribed in the first 2 weeks of suspected COVID‐19 post‐viral anosmia. It should be rather considered only in cases with anosmia that persists beyond at least 14 days after careful consideration of possible risks and benefits. 34 In the current study, the treatment was initiated later than 1 month after the onset of the olfactory dysfunction. It was shown that dysgeusia and anosmia were short‐term and resolved within 7–14 days in most COVID‐19 patients, whereas the improvement time has typically lasted some weeks to months for other post‐viral conditions. 27 , 35 , 36
According to the findings, some degree of smell loss is present in almost all COVID‐19 patients at the end of their intense recovery period. 8 Other studies demonstrated that dyspnea (45.6%, 95% CI 10.9%–80.4%) was one of the most common clinical signs. 37 , 38 The incidence rate of anosmia in patients with COVID‐19 infection ranged between 22% and 68%. The description of taste dysfunction was more heterogeneous with dysgeusia, ageusia, or distorted taste, which were present in 33%, 20%, and 21% of COVID‐19 cases, respectively. 7 , 24 , 25 It has been explained that corticosteroids recover smell function directly by an effect on olfactory receptor neuron function in extra to their anti‐inflammatory activities. 39 Furthermore, Kim et al. reported a comparison between pre and post‐treatment olfactory test results in terms of subjective improvements in smell. Based on their report, the threshold test demonstrated a better correlation in comparison with the identification test. 39
Our results revealed that there was no significant difference in smell dysfunction between the mometasone and placebo groups at the end of the intervention, whereas the changes in Iran‐SIT and VAS scores between mentioned groups were significant after 4 weeks of treatment. Based on olfactory score changes it seems that the nasal corticosteroids may be a helpful effect on the recovery process of patients who received 4 weeks. The median changes of UPSIT and VAS were 6 and 2 in the placebo group versus 10, and 5 in the case group respectively, during the pre and post‐treatment. As the olfactory dysfunction types, 40% and 17% anosmia were observed at the post‐treatment in control and case groups, respectively. At the endpoint of intervention, the decrease of anosmia frequency was 22.9% in the case group compared to the placebo group.
Abdelalim et al. suggested that using mometasone furoate spray as a topical corticosteroid had no significant effect on the treatment of post‐COVID‐19 anosmia in comparison with the olfactory training alone during 3 weeks of follow‐up. 19 The VAS olfactory score could significantly improve at the end‐point of treatment within both groups. The smell score was assessed only based on the VAS, and no placebo group was designed in this study, which leads to limitations in their results. The authors recommended that the intervention of mentioned corticosteroid therapy had no superiority over the olfactory training in the smell scores, recovery rate, and treatment of post‐COVID‐19 anosmia.
Kasiri et al. evaluated the effect of mometasone spray on the improvement of post‐COVID‐19 anosmia or severe microsmia relative to the placebo group within 4 weeks of intervention. 17 According to their results, no notable differences were seen in terms of olfactory score and amount of score changes based on IRAN‐SIT between the two groups. The severity of olfactory dysfunction was significantly different at the end‐point of the study. Although the VAS scores were reported significant between and within groups during the follow‐up period, there were no significant changes between case and control groups. Additionally, no significant differences were found in the results of Iranian UPSIT between the two groups. They noted that the corticosteroid nasal spray with olfactory training could accelerate the recovery of smell loss symptoms due to COVID‐19 than the olfactory training.
A recent study by Rashid et al. has shown that the use of nasal drops of betamethasone 3 times daily for until 1 month had no notable effect on the duration of anosmia relative to placebo. 20 In another investigation, 40 local corticosteroid injection was administered to the nasal mucosa close to the olfactory cleft, and the recovery rate in the smell loss was obtained at 49.6% instantly after treatment in the patients diagnosed with upper respiratory tract infection. Another study revealed that patients who were diagnosed with the post‐viral olfactory loss after treatment using corticosteroids had higher odor identification rates. 41
The researchers showed that olfactory function in the cases of idiopathic hyposmia did not improve significantly after the administration of topical corticosteroid (mometasone). Regardless, oral corticosteroids had a positive outcome in this regard. 15 Veer Singh et al, exhibited that after the use of triamcinolone paste in the mouth and fluticasone spray in the nose there was a statistically notable improvement in identifying all the odors and tastes on day 5 relative to day 1. 18 In a study conducted by Gregorio et al., 42 13 patients have prescribed topical corticosteroid alone (38.4%) with different responses. The rate of recovery was greater in chronic rhinosinusitis cases; however, it was lower in idiopathic hyposmia cases. 43
LIMITATIONS OF THE STUDY AND SUGGESTIONS
Although this investigation supplies an explanation for factors that impact olfactory dysfunction duration, treatment outcomes, and the association between subjective and objective smell tests, it has some limitations. The treatment will not be successful until a more complete understanding of the pathomechanisms of inflammation is achieved in chronic sinusitis. The use of systemic corticosteroids may be accompanied by a better result compared to nasal corticosteroids; however, it was not possible due to its inverse effects on the immunity of the patients during the COVID‐19 pandemic. Therefore, multicentric studies with a larger sample size of participants and also a longer follow‐up period of more than 1 month are required to comprehend whether or not mometasone is a reasonable therapy for anosmia and hyposmia due to COVID‐19 infection. Additionally, further clinical trials based on other types of topical corticosteroid treatment or combination therapy will be recommended.
CONCLUSION
In the present study, the assessment of olfactory dysfunction based on smell test and VAS scores demonstrated no significant differences between case and placebo groups, whereas statistically significant differences were observed within both groups. Although at the end of the intervention, anosmia and normosmia were reported 40% and 42.8% in the placebo group versus 17.1% and 54.3% in the case group, respectively, no significant differences were found between groups. Hence, the frequency of anosmia was 22.9% reduced in the case group relative to the placebo group. Overall, no significant differences were found in olfactory dysfunction between the two groups at the end of the intervention. On the other hand, the change of smell test and VAS scores were significantly higher in the case group relative to the control group. Hence, it seems that the nasal corticosteroids may be a positive effect on the recovery process of patients who received more than 2 weeks. Accordingly, we recommended further randomized clinical trials to assess the effect of topical corticosteroids on anosmic patients due to the COVID‐19 infection.
ACKNOWLEDGMENTS
The authors are particularly grateful to the patients and their family members who volunteered to participate in this study. The authors would like to appreciate the Clinical Research Development Unit, Ghaem Hospital and Faculty of Medicine, Mashhad University of Medical Sciences for assisting with the data analysis.
Editor's Note: This Manuscript was accepted for publication on August 2, 2022.
The authors have no other funding, financial relationships, or conflicts of interest to disclose.
m.h. and m.k. contributed equally to this work.
BIBLIOGRAPHY
- 1. Zhang J‐j, Dong X, Cao Y‐Y, et al. Clinical characteristics of 140 patients infected with SARS‐CoV‐2 in Wuhan, China. Allergy. 2020;75:1730‐1741. [DOI] [PubMed] [Google Scholar]
- 2. WHO . Coronavirus disease 2019 (COVID‐19): situation report, 191. 2020.
- 3. Cucinotta D, Vanelli M. WHO declares COVID‐19 a pandemic. Acta Bio‐Medica: Atenei Parmensis. 2020;91(1):157‐160. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4. Huang C, Wang Y, Li X, et al. Clinical features of patients infected with 2019 novel coronavirus in Wuhan, China. Lancet. 2020;395(10223):497‐506. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5. Qiu H, Wu J, Hong L, Luo Y, Song Q, Chen D. Clinical and epidemiological features of 36 children with coronavirus disease 2019 (COVID‐19) in Zhejiang, China: an observational cohort study. Lancet Infect Dis. 2020;20:689‐696. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6. Vaira LA, Salzano G, Deiana G, De Riu G. Anosmia and ageusia: common findings in COVID‐19 patients. Laryngoscope. 2020;130(7):1787‐1793. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7. Yan CH, Faraji F, Prajapati DP, Boone CE, DeConde AS. Association of chemosensory dysfunction and Covid‐19 in patients presenting with influenza‐like symptoms. Int Forum Allergy Rhinol. 2020;10(7):806‐813. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8. Moein ST, Hashemian SM, Mansourafshar B, Khorram‐Tousi A, Tabarsi P, Doty RL. Smell dysfunction: a biomarker for COVID‐19. Int Forum Allergy Rhinol. 2020;10(8):944‐950. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9. Rouby C, Thomas‐Danguin T, Vigouroux M, et al. The Lyon clinical olfactory test: validation and measurement of hyposmia and anosmia in healthy and diseased populations. Int J Otolaryngol. 2011;2011:203805. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10. Nordin S, Brämerson A. Complaints of olfactory disorders: epidemiology, assessment and clinical implications. Curr Opin Allergy Clin Immunol. 2008;8(1):10‐15. [DOI] [PubMed] [Google Scholar]
- 11. Ovesen T. COVID‐19 anosmia. Tidsskrift Den Norske Legeforening. 2020;7:5‐6. [DOI] [PubMed] [Google Scholar]
- 12. Jafek BW, Murrow B, Michaels R, Restrepo D, Linschoten M. Biopsies of human olfactory epithelium. Chem Senses. 2002;27(7):623‐628. [DOI] [PubMed] [Google Scholar]
- 13. Chen M, Reed RR, Lane AP. Chronic inflammation directs an olfactory stem cell functional switch from neuroregeneration to immune defense. Cell Stem Cell. 2019;25(4):501‐513.e5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14. Matsuyama S, Kawase M, Nao N, et al. The inhaled corticosteroid ciclesonide blocks coronavirus RNA replication by targeting viral NSP15. bioRxiv. 2020. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15. Heilmann S, Huettenbrink K‐B, Hummel T. Local and systemic administration of corticosteroids in the treatment of olfactory loss. Am J Rhinol. 2004;18(1):29‐33. [PubMed] [Google Scholar]
- 16. Holm A, Fokkens W. Topical corticosteroids in allergic rhinitis; effects on nasal inflammatory cells and nasal mucosa. Clin Exp Allergy. 2001;31(4):529‐535. [DOI] [PubMed] [Google Scholar]
- 17. Kasiri H, Rouhani N, Salehifar E, Ghazaeian M, Fallah S. Mometasone furoate nasal spray in the treatment of patients with COVID‐19 olfactory dysfunction: a randomized, double blind clinical trial. Int Immunopharmacol. 2021;98:107871. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18. Singh CV, Jain S, Parveen S. The outcome of fluticasone nasal spray on anosmia and triamcinolone oral paste in dysgeusia in COVID‐19 patients. Am J Otolaryngol. 2021;42(3):102892. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19. Abdelalim AA, Mohamady AA, Elsayed RA, Elawady MA, Ghallab AF. Corticosteroid nasal spray for recovery of smell sensation in COVID‐19 patients: a randomized controlled trial. Am J Otolaryngol. 2021;42(2):102884. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20. Rashid RA, Zgair A, Al‐Ani RM. Effect of nasal corticosteroid in the treatment of anosmia due to COVID‐19: a randomised double‐blind placebo‐controlled study. Am J Otolaryngol. 2021;42(5):103033. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21. Clemency BM, Varughese R, Gonzalez‐Rojas Y, et al. Efficacy of inhaled ciclesonide for outpatient treatment of adolescents and adults with symptomatic COVID‐19: a randomized clinical trial. JAMA Intern Med. 2022;182(1):42‐49. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22. Saussez S, Vaira LA, Chiesa‐Estomba CM, et al. Short‐term efficacy and safety of oral and nasal corticosteroids in Covid‐19 patients with olfactory dysfunction: a European multicenter study. Pathogens. 2021;10(6):698. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23. Lipworth BJ, Jackson CM. Safety of inhaled and intranasal corticosteroids. Drug Saf. 2000;23(1):11‐33. [DOI] [PubMed] [Google Scholar]
- 24. Lechien JR, Chiesa‐Estomba CM, De Siati DR, et al. Olfactory and gustatory dysfunctions as a clinical presentation of mild‐to‐moderate forms of the coronavirus disease (COVID‐19): a multicenter European study. Eur Arch Otorhinolaryngol. 2020;277(8):2251‐2261. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25. Levinson R, Elbaz M, Ben‐Ami R, et al. Anosmia and dysgeusia in patients with mild SARS‐CoV‐2 infection. medRxiv. 2020;52(8):600‐602. [DOI] [PubMed] [Google Scholar]
- 26. Soler ZM, Patel ZM, Turner JH, Holbrook EH. A primer on viral‐associated olfactory loss in the era of COVID‐19. Int Forum Allergy Rhinol. 2020;10(7):814‐820. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27. Bakhshaee M, Barzegar‐Amini M, Motedayen Z, et al. Olfactory dysfunction in patients infected with 2019 novel coronavirus. Iran J Otorhinolaryngol. 2021;33(3):163‐171. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28. Swain SK. Management of olfactory dysfunction in COVID‐19 patients: a review. MGM J Med Sci. 2021;8(3):297. [Google Scholar]
- 29. Casale TB, Dykewicz MS. Clinical implications of the allergic rhinitis‐asthma link. Am J Med Sci. 2004;327(3):127‐138. [DOI] [PubMed] [Google Scholar]
- 30. Maiese A, Manetti AC, Bosetti C, et al. SARS‐CoV‐2 and the brain: a review of the current knowledge on neuropathology in COVID‐19. Brain Pathol. 2021;31(6):e13013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31. Vaira L, Hopkins C, Sandison A, et al. Olfactory epithelium histopathological findings in long‐term coronavirus disease 2019 related anosmia. J Laryngol Otol. 2020;134(12):1123‐1127. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32. Giacomelli A, Pezzati L, Conti F, et al. Self‐reported olfactory and taste disorders in patients with severe acute respiratory coronavirus 2 infection: a cross‐sectional study. Clin Infect Dis. 2020;71:889‐890. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33. Wang X, Zhang C, Xia X, Yang Y, Zhou C. Effect of gender on odor identification at different life stages: a meta‐analysis. Rhinology. 2019;57(5):322‐330. [DOI] [PubMed] [Google Scholar]
- 34. Walker A, Hopkins C, Surda P. The use of Google trends to investigate the loss of smell related searches during COVID‐19 outbreak. Int Forum Allergy Rhinol. 2020;10(7):839‐847. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35. Lee DY, Lee WH, Wee JH, Kim J‐W. Prognosis of postviral olfactory loss: follow‐up study for longer than one year. Am J Rhinol Allergy. 2014;28(5):419‐422. [DOI] [PubMed] [Google Scholar]
- 36. Mori TA, Sugiura M, Matsumoto K, et al. Clinical study of olfactory disturbance. Acta Otolaryngol. 1998;118(538):197‐201. [PubMed] [Google Scholar]
- 37. Rodriguez‐Morales AJ, Cardona‐Ospina JA, Gutiérrez‐Ocampo E, et al. Clinical, laboratory and imaging features of COVID‐19: a systematic review and meta‐analysis. Travel Med Infect Dis. 2020;34:101623. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38. Yang J, Zheng Y, Gou X, et al. Prevalence of comorbidities and its effects in patients infected with SARS‐CoV‐2: a systematic review and meta‐analysis. Int J Infect Dis. 2020;94:91‐95. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39. Kim DH, Kim SW, Hwang SH, et al. Prognosis of olfactory dysfunction according to etiology and timing of treatment. Otolaryngology–Head and Neck Surgery. 2017;156(2):371‐377. [DOI] [PubMed] [Google Scholar]
- 40. Fukazawa K. A local steroid injection method for olfactory loss due to upper respiratory infection. Chem Senses. 2005;30(suppl_1):i212‐i213. [DOI] [PubMed] [Google Scholar]
- 41. Seo BS, Lee HJ, Mo J‐H, Lee CH, Rhee C‐S, Kim J‐W. Treatment of postviral olfactory loss with glucocorticoids, Ginkgo biloba, and mometasone nasal spray. Arch Otolaryngol Head Neck Surg. 2009;135(10):1000‐1004. [DOI] [PubMed] [Google Scholar]
- 42. Gregorio LL, Caparroz F, Nunes LMA, Neves LR, Macoto EK. Olfaction disorders: retrospective study. Braz J Otorhinolaryngol. 2014;80(1):11‐17. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43. Shu CH, Lee PL, Shiao AS, Chen KT, Lan MY. Topical corticosteroids applied with a squirt system are more effective than a nasal spray for steroid‐dependent olfactory impairment. Laryngoscope. 2012;122(4):747‐750. [DOI] [PubMed] [Google Scholar]


