Abstract
Background and Aims
Previous economic evaluations of smoking cessation interventions for pregnant women are limited to single components, which do not in isolation offer sufficient potential impact to address smoking cessation targets. To inform the development of more appropriate complex interventions, we (1) describe the development of the Economics of Smoking in Pregnancy: Household (ESIP.H) model for estimating the life‐time cost‐effectiveness of smoking cessation interventions aimed at pregnant women and (2) use a hypothetical case study to demonstrate how ESIP.H can be used to identify the characteristics of optimum smoking cessation interventions.
Methods
The hypothetical intervention was based on current evidence relating to component elements, including financial incentives, partner smoking, intensive behaviour change support, cigarettes consumption and duration of support to 12 months post‐partum. ESIP.H was developed to assess the life‐time health and cost impacts of multi‐component interventions compared with standard National Health Service (NHS) care in England. ESIP.H considers cigarette consumption, partner smoking and some health conditions (e.g. obesity) that were not included in previous models. The Markov model's parameters were estimated based on published literature, expert judgement and evidence‐based assumptions. The hypothetical intervention was evaluated from an NHS perspective.
Results
The hypothetical intervention was associated with an incremental gain in quitters (mother and partner) at 12 months postpartum of 249 [95% confidence interval (CI) = 195–304] per 1000 pregnant smokers. Over the long‐term, it had an incremental negative cost of £193 (CI = –£779 to 344) and it improved health, with a 0.50 (CI = 0.36–0.69) increase in quality‐adjusted life years (QALYs) for mothers, partners and offspring, with a 100% probability of being cost‐effective.
Conclusions
The Economics of Smoking in Pregnancy: Household model for estimating cost‐effectiveness of smoking cessation interventions aimed at pregnant women found that a hypothetical smoking cessation intervention would greatly extend reach, reduce smoking and be cost‐effective.
Keywords: Cost‐effective, cost‐utility, economic evaluation, health inequality, pregnancy, smoking cessation, tobacco
INTRODUCTION
Smoking during pregnancy (SDP) causes significant health problems, and is closely linked to health inequalities [1, 2]. In England, 11% of expectant mothers smoke at the time of delivery and the rates are higher in the most deprived regions; for example, rising to 27% in Blackpool [1]. Although approximately 44% of pregnant women express interest in cessation support, uptake of referral to stop smoking services (SSS) is approximately 12% among pregnant women, and usual care is limited and includes self‐help materials, one face‐to‐face meeting, nicotine replacement therapy (NRT) and four telephone calls after setting a quit date [3, 4, 5, 6]. Moreover, a recent UK trial found that NRT has no impact on quit rates at delivery [7]. Hence, fundamental change to the funding and delivery of NHS SSS for pregnant women will be required to deliver the current national target of 6% or fewer women to be smoking at the time of delivery by 2023 [1, 8].
The evidence suggests that long‐term conditional financial incentives combined with behavioural support and the inclusion of social supporters (e.g. partners) are the most cost‐effective interventions that help pregnant women to quit smoking and remain abstinent during the postpartum period [9, 11]. However, to date, no trial combining long‐term professional assistance and partner support with financial incentives has been published. In contrast, most interventions have been low‐intensity, short‐term and involved pregnant women only [9, 12]. As conducting pilot studies or feasibility trials to assess the impact of providing a multi‐faceted intervention that includes long‐term, intense support with a household approach would be comparatively costly, it is important to explore whether such interventions have the potential to be cost‐effective.
Decision‐analytical models are frequently used to estimate the incremental costs and benefits of an intervention beyond the available data. This allows the provision of comprehensive evidence for health‐care decision‐makers. The Medical Research Council recommends the use of economic modelling to assess the feasibility of complex interventions [13]. The existing economic models of SDP interventions have significant limitations, such as omitting the interaction between women and their partners and life‐time impacts on the offspring [14]. Jones et al. [15] developed the Economic impacts of Smoking In Pregnancy (ESIP) model, including life‐time impacts upon mothers and infants. The current study extends ESIP by adopting a household approach, considering the number of cigarettes consumed and additional health conditions [14]. This study presents the resulting new model, the Economic impacts of Smoking In Pregnancy: Household (ESIP.H), and uses the model to explore the characteristics of hypothetical smoking cessation interventions targeted at pregnant women. The study aims to contribute evidence to design better cessation services for expectant mothers, which is essential for reducing health inequalities in society.
METHODS
Description of the ESIP.H model
ESIP.H consists of five components: one decision‐tree for the within‐pregnancy component and four linked Markov chains covering the post‐pregnancy period for the mother and partner, and the offspring childhood and adulthood periods. The model allows interaction among women, partners and offspring, and runs in annual cycles. A simplified illustration of the model is provided in Figure 1.
FIGURE 1.
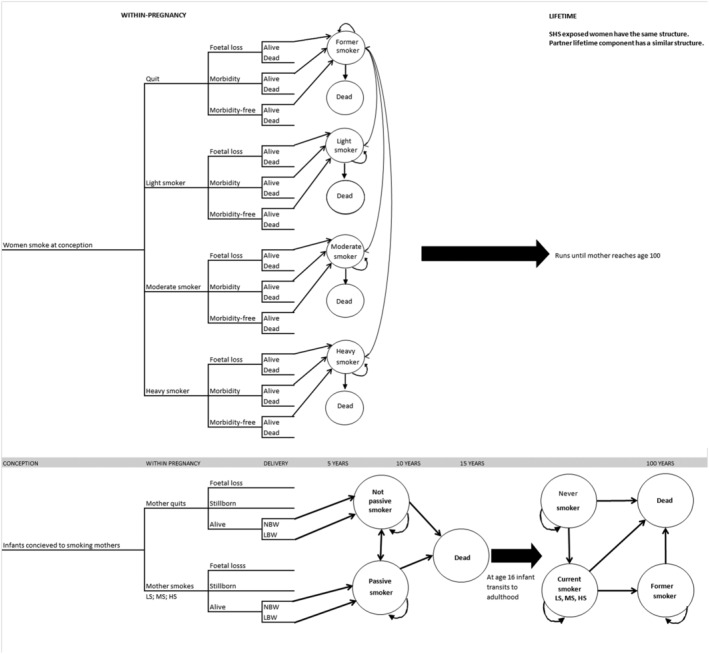
Simplified illustration of the Economics of Smoking in Pregnancy: Household (ESIP.H) model
A hypothetical cohort of 1000 singleton‐pregnancy women who smoke enter a decision tree. The year of birth of children and the average age of women are entered by the user, as the model estimates mortality based on these figures. The women are grouped according to second‐hand smoke (SHS) exposure at delivery. Next, women's smoking behaviour at the time of delivery is entered as quit, light, moderate or heavy smoker. Increased SHS exposure and severity of smoking are assumed to reduce the probability of quitting (Supporting information, Appendix A). It was also assumed that women who continue smoking during pregnancy would not reduce the number of cigarettes consumed, because no evidence was identified on the health outcomes of this [2], based on the systematic reviews which found no difference between pregnant women who quit early in pregnancy and non‐smoking women in terms of health outcomes [22, 23]. Women might die or survive during pregnancy and pregnancy might end with a live birth or stillbirth. The end of pregnancy is time zero in the life‐time models and surviving women enter the mother life‐time model, which is a Markov chain with annual cycles running up to age 100 years estimating the life‐time health and cost outcomes of smoking. The probability of quitting smoking and the probability of remaining abstinent 1 year after delivery can be specified.
The mother life‐time component is affected by the partner life‐time model, such that the partner behaviour determines the SHS exposure of the mother. Similar to the within‐pregnancy component, the quitting probability declines with increased SHS exposure and severity of smoking (Supporting information, Appendix B, Tables S1 and S2). The partner model has a similar structure to the mother life‐time model. Partners are grouped based on the smoking status of women; hence partners of smoking women are less likely to quit. The probability of starting to smoke for partners who do not smoke at delivery is also incorporated.
The decision tree identifies birth outcomes for the offspring as fetal loss, stillbirth, low birth weight (LBW), preterm and normal birth infants. Offspring then enter the childhood component depending on the birth outcome. Children have the probability of being exposed to SHS if they have smoking parents (Supporting information, Appendix A). The childhood component also predicts the smoking uptake risk based on parents’ smoking status, in response to the evidence suggesting that adolescents are more likely to start smoking if their parents are smoking (Supporting information, Appendix A, Tables S3 and S4) [24]. At age 16, they enter the adulthood component, which estimates the life‐time health and cost outcomes based on their smoking status. Age 16 was chosen because the UK national data on smoking patterns were available from 16 onwards, and there was evidence showing that children have an increased risk of smoking uptake at the age of 16 if they were exposed to smoking during pregnancy [24]. After entering the adulthood component, the offspring is assumed to be independent of their parents and hence SHS is not incorporated into the offspring adulthood component. The severity of smoking, however, is also considered in the offspring adulthood component. A summary of the model assumptions is provided in Appendix B.
Estimating mortality risks contingent on smoke exposure
The probability of maternal death during pregnancy was estimated based on the causes of death statistics [25], and maternal smoking status and SHS by partner during pregnancy were assumed to have no impact on the mortality risk due to a lack of evidence. The risks in the life‐time models (mother, partner and offspring) were determined based on the Office for National Statistics (ONS) Cohort Life Tables [26]. The impact of number of cigarettes consumed on mortality rates was incorporated. The probabilities for people who have never smoked (never smokers) were estimated by applying the following formula [27]:
The relative risks of mortality or former, light, moderate and heavy smokers were obtained from the published literature [28, 29] while the proportion of former and current smokers and the prevalence of light, moderate and heavy smokers among current smokers were obtained from national data sets [30, 31]. Only those who remained abstinent for more than 1 year were assigned the probabilities of mortality for former smokers, while those who quit smoking within the last year were assigned the risks for current smokers. In the childhood component, the mortality risks were adjusted based on birth weight.
Estimating morbidity risks contingent upon smoking status
ESIP.H included a range of morbidity risks based on the findings of a systematic review [2]. These were placental abruption, ectopic pregnancy, miscarriage, pre‐eclampsia, stillbirth, LBW, preterm birth in the decision tree, cardiovascular heart disease (CHD), chronic obstructive pulmonary disease (COPD), lung cancer (LC) and stroke in the life‐time models, and sudden infant death (SID), lower respiratory infections (LRI) and asthma in the childhood component [32]. Moreover, childhood obesity was considered in the sensitivity analysis [33, 34].
The prevalence of within pregnancy complications in the general population was estimated based on hospital episode statistics [35]. The prevalence of LBW and stillbirth were estimated based on gestation‐specific mortality data [36] and child mortality statistics [37]. English age and gender‐specific prevalence data and related relative risks identified in the literature were used for SID, LRI, asthma, obesity and the life‐time conditions [30, 38, 39, 40, 41, 42, 43, 44, 45]. The risk of stillbirth, LBW, asthma and LRI was adjusted based on gestational age [46, 47, 48, 49]. The impact of former smoking, the severity of smoking and the SHS exposure were obtained from the published literature [47, 50, 51, 52, 53, 54, 55, 56, 57, 58, 59, 60]. The estimated probabilities and an example of how smoking contingent morbidity risks were calculated appear in Supporting information, Appendix B, Tables S8–S11.
Incorporating health‐related quality of life
When incorporating the health‐related quality of life impacts, ESIP.H calculated utility loss contingent upon the severity of smoking [61] (Supporting information, Table S19). Following the same approach as Jones et al. [15], life‐years at the end of pregnancy were estimated based on the assumption that pregnancy would last 40 weeks in full term, and 10 weeks in case of ectopic pregnancy, 14 weeks for miscarriage, 33 weeks for premature birth, 38 weeks for previa and 39 weeks for pre‐eclampsia. All women who experienced a fetal loss were assigned a one‐off 0.1 utility loss [62] and those who suffered from ectopic pregnancy lost another 0.01 [63]. It was assumed that there was no utility loss for abruption, previa and pre‐eclampsia due to a lack of data. In the life‐time models, utility losses were assigned for CHD (0.27) [64], COPD (0.27) [65], LC (0.33) [66] and stroke (0.28) [67].
In the childhood component, children with asthma were assigned a utility loss of 0.1 [68]. The literature search failed to identify a UK study measuring the impact of LRI on health utilities, although some studies in other countries reported a 0.1 reduction [69, 70, 71]. Therefore, LRI was assumed to have the same impact as asthma, considering the similar symptoms of the two diseases [72].
Estimating costs for health conditions
The methods in ESIP were used to estimate the costs arising from health conditions based on recent data and by inflating the calculations from the literature to 2017/18 prices (Supporting information, Appendix A, Table S10) [15, 73]. ICD‐10 codes and Healthcare Resource Group codes were used to identify relevant NHS reference costs. The assumptions by Jones et al. [15] when estimating the within‐pregnancy costs and the use of cardiac arrest as an approximation for death were also applied in this study.
Case study: using ESIP.H to evaluate hypothetical smoking cessation interventions for pregnant women
Morgan et al. [10] identified the characteristics of the most promising smoking cessation intervention for pregnant women based on mixed methods, including a discrete choice experiment, as follows: frequent regular contacts with health‐care professionals ideally up to 12 months postpartum, vouchers up to £80 per month contingent on carbon monoxide (CO) monitoring and including social support. Based on the study by Morgan et al. [10] and several other trials [4, 16, 17], a hypothetical cessation intervention for pregnant women who smoke was designed along with a control intervention relating to usual care (Table 1):
Control intervention. Every pregnant smoker is offered one face‐to‐face contact in a maternity care setting followed by 10 weeks’ NRT, self‐help materials and four weekly telephone calls [5, 18]. No support is available for the partners.
Smoke‐free household until 12 months postpartum (SFH12m). In addition to the support for the control group (excluding the telephone calls), women and partners (or supporters) are provided with cessation support until 3 months postpartum. This includes biweekly midwife visits with £40 vouchers per negative CO specimen. Between 3 and 12 months postpartum, participants continue receiving the same support with monthly instead of biweekly midwife visits. Overall, participants receive 28 midwife visits in addition to the initial contact and are offered up to £2320 per household. If the partner smokes they need to provide a negative specimen to earn vouchers and if the partner does not smoke, they earn vouchers contingent on the woman's abstinence.
TABLE 1.
Estimated effectiveness and cost of the hypothetical SFH12m intervention
| Partner smokes | Partner does not smoke | |||
|---|---|---|---|---|
| Control | Quit rate at delivery [19, 80] | Women | 5% | 14% |
| Partners | 3% | NA | ||
| 12‐month abstinence among quitters [20, 80] | Women | 3% | 46% | |
| Partners | 3% | 46% a | ||
| Intervention cost per household | £152.18 | £152.18 | ||
| Base‐case | Quit rate at delivery [4, 7, 79] | Women | 34% | 55% |
| Partners | 15% | NA | ||
| 12‐month abstinence among quitters [4, 17, 79, 92] | Women | 27% | 84% | |
| Partners | 11% | 54% a | ||
| Intervention cost per household | £1208.98 | £2368.95 | ||
| Cautious‐case | Quit rate at delivery [16, 17] | Women | 24% | 43% |
| Partners | 9% | NA | ||
| 12‐month abstinence among quitters [17] | Women | 19% | 62% | |
| Partners | 7% | 50% a | ||
| Intervention cost per household | £937.37 | £1844.51 |
More details on how these figures were calculated are provided in Supporting information, Appendix A.
10% of these figures were applied as standard errors. NA = not applicable; SFH12m = smoke‐free household until 12 months postpartum.
These show the probability of remaining abstinent at 12 months among partners who quit smoking at delivery if they have non‐smoking partners (women) at 12 months.
Effectiveness of the hypothetical intervention
The expected effectiveness of the intervention was estimated based on the published literature, in the absence of specific trial data (Table 1). Quit rates at delivery in the control group were obtained from a systematic review [19] and abstinence probabilities and rates for partners from the Infant Feeding Survey [20]. As there was no evidence regarding the impact of such interventions on quit rates 1 year after delivery among women who continued smoking throughout pregnancy, it was assumed to be the same as the control group [20]. Full details of the effectiveness estimates are provided in Supporting information, Appendix A. Two different scenarios (a base‐case and a cautious‐case) were developed to consider the impacts of the assumptions.
Costs of the hypothetical intervention and control
The intervention cost was calculated based on the national reference cost for a midwife visit [21] and the data reported by two trials were used for the remaining cost components, which included the NRT, CO test and postage fees as well as the training and telephone call costs. [4, 7]. As the largest cost was the financial incentives, a systematic approach was developed to estimate the voucher cost (Supporting information, Appendix B, Tables S1–S13). The total cost of the intervention was calculated based on expected quit and reduction rates (Table 1).
Analyses and outcomes
The analysis was not pre‐registered and the results should be considered exploratory. Base‐case and cautious‐case analyses were performed for evaluating the hypothetical intervention. The novel decision analytical model, ESIP.H, was used to estimate the cost‐effectiveness. The analyses were conducted from an NHS perspective, using 2017/18 costs and future costs and benefits were discounted at 3.5% as per National Institute for Health and Care Excellence (NICE) guidelines [74].
The outcomes included number of quitters, incremental cost effectiveness ratios (ICERs) at delivery, 1 year after delivery, at the end of childhood and over the life‐time. Deterministic sensitivity analysis was performed to estimate the impact of different characteristics of the hypothetical intervention regarding partner involvement, effectiveness and cost. The analyses were also repeated by including childhood obesity, increasing the postpartum relapse rates in the control group by 6% and reducing the discount rate to 1.5%, as per NICE guidelines and the international literature [74, 75, 76, 77]. Probabilistic sensitivity analysis (PSA) was conducted to estimate the uncertainties around the model findings. The model was run until the 95% confidence interval of incremental net benefit did not include zero [78]. Cost‐effectiveness acceptability curves (CEACs) were generated to estimate the probability of cost‐effectiveness at different cost thresholds per QALY.
RESULTS
The SFH12m intervention designed for pregnant women who want to quit smoking included financial incentives up to £2,320 per household contingent of abstinence and 28 midwife visits, in addition to the standard care which included self‐hep material, NRT and telephone calls. The expected quit rate among women at delivery was estimated as 55% if the partner did not smoke and 34% if the partner smoked, with SF12m as opposed to 14 and 5%, respectively, in the control group. The cost per household was £2,369 if the partner did not smoke and £1,209 when the partner smoked, while it was £152 in the control group. These figures were entered into ESIP.H to estimate the cost‐effectiveness of SFH12m.
The outcomes generated by the ESIP.H model are provided in Table 2. SFH12m was estimated to generate 198 additional quitters 1 year after delivery per 1000 pregnant women and 39 quitters among partners. Increasing the number of quitters resulted in a significant reduction in adverse health outcomes, such as infant death (−10) and LBW (−16) (Supporting information, Appendix C, Table S15). The intervention was dominant over the comparator when the life‐time impacts upon the mothers, partners and offspring were also considered, producing greater health gains and cost‐savings (Table 2). The intervention was also cost‐effective in the cautious‐case, generating 0.30 incremental QALYs at an additional cost of £243 per household (Supporting information, Appendix C, Table S14).
TABLE 2.
ESIP.H model outputs for the hypothetical household intervention with base assumptions
| Deterministic analysis | Probabilistic sensitivity analysis | |||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Control | Experimental | Incremental | Control | Experimental | Incremental | |||||||
| Mean | Mean | Mean | Mean | 95% CI | Mean | 95% CI | Mean | 95% CI | ||||
| 12 months after delivery | ||||||||||||
| Quit rate (%) | ||||||||||||
| Mothers | 14.1 | 33.9 | 19.8 | 13.9 | 12.4 | 15.5 | 34.7 | 29.6 | 40.1 | 20.8 | 16.1 | 26.1 |
| Partners | 2.2 | 6.3 | 4.1 | 2.3 | 1.9 | 2.7 | 6.4 | 5.2 | 7.6 | 4.1 | 3.1 | 5.1 |
| Total | 16.3 | 40.0 | 23.7 | 16.2 | 14.6 | 17.8 | 41.1 | 35.2 | 47.1 | 24.9 | 19.5 | 30.4 |
| Life‐years | ||||||||||||
| Per mother | 1.67 | 1.68 | 0.006 | 1.67 | 1.67 | 1.68 | 1.68 | 1.68 | 1.68 | 0.006 | 0.004 | 0.009 |
| Per partner | 1.97 | 1.97 | 0.000 | 1.97 | 1.97 | 1.97 | 1.97 | 1.97 | 1.97 | 0.000 | 0.000 | 0.000 |
| Per offspring | 0.86 | 0.87 | 0.011 | 0.86 | 0.86 | 0.86 | 0.87 | 0.87 | 0.88 | 0.011 | 0.007 | 0.017 |
| Total | 4.50 | 4.52 | 0.017 | 4.50 | 4.50 | 4.51 | 4.52 | 4.51 | 4.53 | 0.016 | 0.010 | 0.026 |
| QALYs | ||||||||||||
| Per mother | 1.50 | 1.52 | 0.022 | 1.50 | 1.47 | 1.52 | 1.52 | 1.49 | 1.55 | 0.022 | 0.016 | 0.029 |
| Per partner | 1.83 | 1.83 | 0.003 | 1.83 | 1.80 | 1.85 | 1.83 | 1.80 | 1.86 | 0.002 | 0.001 | 0.004 |
| Per offspring | 0.84 | 0.86 | 0.014 | 0.84 | 0.82 | 0.86 | 0.86 | 0.84 | 0.87 | 0.014 | 0.009 | 0.021 |
| Total | 4.17 | 4.21 | 0.039 | 4.17 | 4.10 | 4.22 | 4.21 | 4.14 | 4.26 | 0.038 | 0.027 | 0.052 |
| Cost | ||||||||||||
| Per household (£) | £7757 | £9315 | £1558 | 7147 | 4259 | 12 913 | 8625 | 5650 | 14 477 | 1478 | 1231 | 1732 |
| Incremental cost per QALY | £40 447 | £38 894 | ||||||||||
| End of childhood (15 years after delivery) | ||||||||||||
| Life‐years | ||||||||||||
| Per mother | 12.19 | 12.19 | 0.01 | 12.19 | 12.18 | 12.19 | 12.19 | 12.19 | 12.20 | 0.01 | 0.00 | 0.01 |
| Per partner | 12.45 | 12.45 | 0.00 | 12.45 | 12.45 | 12.46 | 12.45 | 12.45 | 12.46 | 0.00 | 0.00 | 0.00 |
| Per offspring | 10.28 | 10.40 | 0.13 | 10.27 | 10.24 | 10.30 | 10.40 | 10.35 | 10.49 | 0.13 | 0.08 | 0.20 |
| Total | 34.92 | 35.05 | 0.13 | 34.91 | 34.87 | 34.94 | 35.05 | 34.99 | 35.14 | 0.13 | 0.08 | 0.21 |
| QALYs | ||||||||||||
| Per mother | 10.84 | 10.92 | 0.08 | 10.86 | 10.64 | 11.05 | 10.94 | 10.72 | 11.12 | 0.08 | 0.05 | 0.11 |
| Per partner | 11.42 | 11.44 | 0.01 | 11.41 | 11.18 | 11.58 | 11.43 | 11.20 | 11.59 | 0.02 | 0.01 | 0.02 |
| Per offspring | 10.05 | 10.20 | 0.16 | 10.05 | 9.73 | 10.24 | 10.20 | 9.92 | 10.39 | 0.15 | 0.09 | 0.24 |
| Total | 32.31 | 32.56 | 0.25 | 32.32 | 31.78 | 32.73 | 32.57 | 32.03 | 32.95 | 0.25 | 0.17 | 0.35 |
| Cost | ||||||||||||
| Per household (£) | £30 747 | £31 161 | £414 | £29 249 | £25 719 | £34 961 | £29 728 | £26 231 | £35 467 | £480 | £36 | £915 |
| Incremental cost per QALY | £1663 | £1925 | ||||||||||
| Life‐time horizon | ||||||||||||
| Life‐years | ||||||||||||
| Per mother | 25.10 | 25.17 | 0.070 | 25.15 | 25.06 | 25.28 | 25.22 | 25.14 | 25.33 | 0.067 | 0.044 | 0.089 |
| Per partner | 24.91 | 24.95 | 0.032 | 24.94 | 24.84 | 25.08 | 24.97 | 24.87 | 25.11 | 0.032 | 0.024 | 0.041 |
| Per offspring | 24.21 | 24.51 | 0.296 | 24.18 | 24.08 | 24.25 | 24.48 | 24.34 | 24.68 | 0.297 | 0.188 | 0.470 |
| Total | 74.22 | 74.62 | 0.40 | 74.24 | 74.11 | 74.39 | 74.64 | 74.46 | 74.90 | 0.396 | 0.279 | 0.575 |
| QALYs | ||||||||||||
| Per mother | 21.59 | 21.74 | 0.154 | 21.65 | 21.24 | 21.99 | 21.81 | 21.38 | 22.16 | 0.153 | 0.104 | 0.207 |
| Per partner | 22.36 | 22.39 | 0.029 | 22.36 | 21.94 | 22.66 | 22.39 | 21.97 | 22.69 | 0.032 | 0.019 | 0.046 |
| Per offspring | 22.56 | 22.88 | 0.318 | 22.69 | 22.31 | 22.98 | 23.01 | 22.63 | 23.34 | 0.316 | 0.203 | 0.483 |
| Total | 66.51 | 67.01 | 0.501 | 66.70 | 65.97 | 67.29 | 67.20 | 66.47 | 67.77 | 0.498 | 0.363 | 0.688 |
| Cost | ||||||||||||
| Per household (£) | £70 856 | £70 669 | –£186 | £68 583 | £62 837 | £75 364 | £68 390 | £62 680 | £74 955 | –£193 | –£779 | £344 |
| Incremental cost per QALY | Dominant | Dominant | ||||||||||
ESIP.H = Economics of Smoking in Pregnancy: Household; QALY = quality‐adjusted life‐year; CI = confidence interval.
Sensitivity analyses
The deterministic sensitivity analysis showed that the inclusion of childhood obesity or the increase in the postpartum relapse rates in the control group did not change the results. The sensitivity analyses showed that the hypothetical intervention remained cost‐effective when the quit rates at delivery were reduced by one‐third and zero impact on the relapse rates were assumed (Supporting information, Appendix D, Figures S1 and S2). Similarly, the intervention remained cost‐effective when the costs per household were assumed to be 9.5‐fold and 7.5‐fold of the original figures in the base‐case and cautious‐case, respectively, given that the effects of quit and abstinence rates were constant. According to the PSA, the hypothetical intervention produced an incremental gain in quitters (mother and partner) at 12 months postpartum of 249 (95% CI,195–304) per 1000 pregnant smokers. Over the long‐term, it had an incremental negative cost of £193 (95%CI, –£779 to 344) and it improved health, with a 0.50 (95% CI, 0.36–0.69) increase in quality‐adjusted life years (QALYs) for mothers, partners and offspring. The PSA indicated some uncertainty around the incremental cost per QALY calculations (Table 2). In the base‐case household analyses most of the iterations fell into the south‐east quadrant, suggesting that SFH12m was dominant in 80% of the iterations, providing greater health gains and cost savings (Figure 2). In the cautious‐case, SFH12m was cost‐effective in all estimates. CEACs showed that the probability of cost‐effectiveness was 100% at £30 000 per QALY threshold in the long‐term analysis, although at delivery it was 0% (Figure 3).
FIGURE 2.
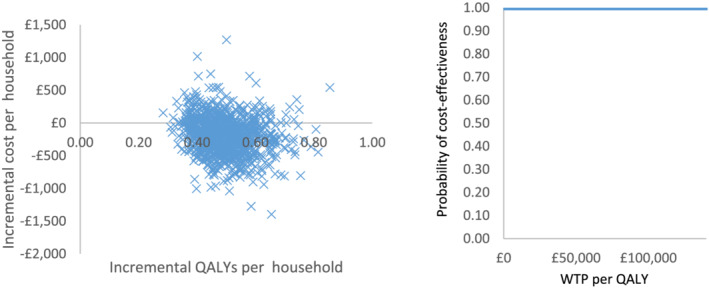
Probabilistic sensitivity analysis (PSA) and cost‐effectiveness acceptability curve (CEAC) for the smoke‐free household until 12 months postpartum (SFH12m) base‐case (life‐time horizon). Abbreviation: QALY = quality‐adjusted life‐year
FIGURE 3.
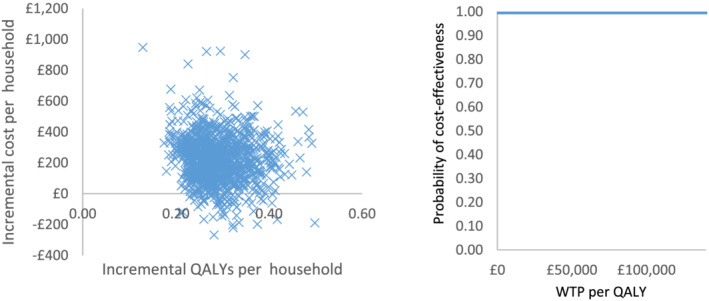
Probabilistic sensitivity analysis (PSA) and cost‐effectiveness acceptability curve (CEAC) for the smoke‐free household until 12 months postpartum (SFH12m) cautious‐case (life‐time horizon). Abbreviation: QALY ‐ quality‐adjusted life‐year
DISCUSSION
The study showed that high‐intensity, long‐term SDP interventions, including financial incentives together with a household approach has the potential to produce significant health benefits and to be cost‐effective when the impacts on the offspring and partner are also considered. Previously, offering high levels of financial incentives up to £1,800 per women were found to be cost‐effective in the United Kingdom [79]. The current study supports these findings, and suggests that SDP interventions including up to £2,320 shopping vouchers per household could be cost‐effective.
The quit rate at delivery among mothers in the hypothetical intervention (44% in the base‐case) is considerably higher than those achieved in existing interventions [9]. For instance, that figure was 23% in the CPIT study [4]. Similarly, the proportion of CO‐verified quitters 4 weeks after the admission to the NHS SSS was 29% [1]. Thus, the hypothetical cessation interventions have a potential to reduce smoking during pregnancy and the associated health problems compared with other interventions that have been investigated. Additionally, up to 15% of smoking partners could quit with the hypothetical intervention. Therefore, additional benefits could be gained by reducing SHS by partners during pregnancy.
There are also potential improvements regarding postpartum relapse rates. The biochemically verified relapse rate among women who quit at delivery was 65% 6 months after delivery and 80% 1 year after delivery in previous trials [4, 80]. With the long‐term cessation intervention lasting until 1 year postpartum, the relapse rate could decline to 34%. Hence, the hypothetical intervention has the potential to reduce the adverse health impacts of postpartum smoking considerably.
Strengths and limitations
This is the first economic evaluation of cessation interventions involving long‐term and high‐intensity support for pregnant women, together with a household approach and financial incentives. In the absence of direct trial data, the study was conducted based on hypothetical scenario analyses, which required making assumptions about the effectiveness and costs based on the best available evidence. However, the number of studies was limited, and the hypothetical intervention was designed based on evidence from a range of settings including England [20], Canada [80], the United States [17] (partner smoking/abstinence, partner/significant other support), and Scotland [4] and Sweden [92] (financial incentives). We have assumed, for example, that Scottish trial results on the impact of shopping voucher incentives would be applicable in an English setting, even though smoking at the time of delivery is higher in Scotland (13%) compared to England (10%) [1, 81, 82]. It will be important to take account of new evidence as it becomes available, and the Scottish financial incentives study has led to a larger trial across the United Kingdom [83]. To reflect the range of evidence and assumptions, base‐case and cautious‐case scenarios were designed, as well as taking a conservative approach in the estimations and conducting sensitivity analyses.
ESIP.H is the first economic model with a household approach which allows estimation of the spill‐over effects and the severity of smoking. However, some limitations arising from the model assumptions should be considered when interpreting its outcomes. For example, due to the lack of data, the model assumes that having a non‐smoking partner has the same impact as not having a partner. In reality, women who continue to smoke during pregnancy are less likely to have partners than those who quit [84, 85]. However, the effect of this assumption would be limited because approximately 70% of smoking pregnant women reported having smoking partners in trials [19]. Another limitation caused by the lack of data is the assumption that the severity of smoking does not change after entering the model unless they quit and re‐start smoking. In the absence of data regarding the change in the number of cigarettes consumed daily, the implications of this assumption on model outcomes are unknown.
Another consideration is that the impact of chronic health conditions on health utilities were assumed to be constant over time, in keeping with the previous version of ESIP [15]. This impact might reduce or increase over time for specific conditions and we have not speculated on the impact of this assumption on model outcomes. Furthermore, ESIP.H does not include the impact of SHS in the offspring adulthood component. That means overestimating ICERs, considering that 26% of non‐smokers are exposed to SHS [31]. Similarly, the restriction of the model to one pregnancy per woman and singleton pregnancies should be considered, which might underestimate the benefits.
Due to a lack of evidence, ESIP.H does not incorporate e‐cigarette use, although some people use them as a cessation aid. E‐cigarettes include 95% less harmful chemicals and decreased health risks [86]. However, given the limited data on the prevalence, patterns and effects of vaping in pregnancy [87], and that NICE does not currently recommend the use of e‐cigarettes during pregnancy [88], our hypothetical intervention is focused upon support for vulnerable women to overcome their addiction to nicotine, so vaping does not represent a quit. As new evidence becomes available e‐cigarette use could be incorporated into ESIP.H.
Policy implications
The smoking cessation interventions for pregnant women published to date have limited impact on quit rates and long‐term abstinence, and they are cost‐effective mainly because the intervention costs are typically low [14]. The economic evaluation suggests that there is a case for developing interventions which require investing more resources in supporting women who smoke during pregnancy to quit, because of the comparatively high estimated quit rates and the cost‐effectiveness evidence. It would be important to pilot such an intervention to identify potential implementation challenges, such as the availability of midwives, and estimate the implementation costs before scaling‐up. It would be possible to use the ESIP.H to assess different versions of an intervention based on initial pilots. The sensitivity analyses demonstrated how ESIP.H could be used to explore the cost‐effectiveness of such interventions.
The sensitivity analysis showed that smoking during pregnancy interventions could be cost‐effective even when the impact was significantly lower (0.06 QALY gains). This is consistent with the findings of previous studies [89]. This finding indicates the need to shift the focus from the cost‐effective interventions that have little impact on quit and abstinence rates to more intensive and cost‐effective interventions.
The hypothetical intervention in this study includes longer‐term regular contact with midwives through the postpartum period rather than referral to the SSS. That is consistent with the ‘continuity of carer’ concept imposed by the ‘Better Births’ change plan for maternal care services which covers smoking during pregnancy as one of the four components of Clinical Commissioning Groups Improvement and Assessment Framework [90]. Thus, the study findings could be helpful to decision‐makers as they design their services. Achieving the national ambition of 6% or less smoking at the time of delivery requires provision of much more effective and inclusive help for expectant mothers, and the ESIP.H model has potential to help decision‐makers design optimum interventions with a household approach.
Moreover, considering the high smoking rates at the time of delivery among women from low socio‐economic backgrounds, these hypothetical interventions could help to reduce the gap in health inequalities by supporting deprived households. Similarly, because non‐smokers in the lowest income group are the most likely to be exposed to SHS [30], having a household approach could have wider impacts on reducing health inequalities.
The generalizability of health economics findings to other settings is limited, as ESIP.H used UK‐based data. Some applicability issues have been identified regarding to use of high‐income country‐based health economics evidence on smoking during pregnancy interventions in low‐ and middle‐income countries [91]. However, the characteristics of these interventions might be relevant to many other countries. Furthermore, it is possible to re‐parameterize ESIP.H for specific settings and evaluate different interventions.
CONCLUSION
Achieving the national ambition of 6% or less SDP requires provision of more inclusive and intense help for expectant mothers. The ESIP.H model has the potential to help decision‐makers to design optimum interventions with a household approach. The study demonstrates how the ESIP.H model can be used to explore the characteristics of multi‐faceted interventions that aim to tackle smoking during pregnancy. The findings indicate the importance of taking a household approach when considering the impacts of interventions to reduce SDP. The analyses suggest that long‐term interventions combining intense support and financial incentives with a household approach are likely to offer significant health benefits and to be cost‐effective, and therefore warrant further consideration by policymakers.
DECLARATION OF INTERESTS
None.
AUTHOR CONTRIBUTIONS
Tuba Avsar: Conceptualization; data curation; formal analysis; funding acquisition; investigation; methodology; project administration; resources; software; validation; visualization. Louise Jackson: Conceptualization; methodology; project administration; supervision. Pelham Barton: Conceptualization; formal analysis; methodology; project administration; software; supervision; validation. Matthew Jones: Conceptualization; methodology; software. Hugh McLeod: Conceptualization; data curation; investigation; methodology; project administration; supervision; validation.
Supporting information
Data S1: Appendix A: Technical summary of the development of ESIP.H
Appendix B: Hypothetical cessation intervention
Appendix C: Cautious‐case ESIP.H model outputs and outcomes at delivery
Appendix D: Deterministic sensitivity analysis
Appendix E: Probabilistic sensitivity analysis
Table S1. Transition probabilities for the maternal smoking behaviour change within the two years after delivery
Table S2: Transition probabilities for the smoking behaviour change in the mother and partner lifetime components
Table S3: Smoking uptake probabilities based on parental smoking
Table S4: Transition probabilities for the smoking behaviour change in the offspring adulthood component
Table S5: Probabilities of within‐pregnancy outcomes for mother and infants
Table S6: ORs (95% CI) compared to never smokers used for within pregnancy health conditions
Table S7: Prevalence of CHD, COPD, Stroke and LC amongst never smokers not exposed to SHS
Table S8: Relative risks for lifetime morbidities used in lifetime models
Table S9: Utility tariff for smokers and non‐smokers
Table S10: Unit costs used in the study
Table S11: Abstinence levels amongst mothers at different time points used for voucher cost calculations
Table S12: Abstinence levels amongst partners at different time points used for voucher cost estimations
Table S13: Estimated cost of the hypothetical intervention
Table S14. Cautious‐case ESIP.H model outputs for the hypothetical household intervention
Table S15. Outcomes at delivery for the base‐case and cautious‐case
Table S16: Distributions used in the probabilistic sensitivity analysis
Figure S1: Deterministic sensitivity analysis for SFH12m base‐case household lifetime results
Figure S2: Deterministic sensitivity analysis for SFH12m cautious‐case household lifetime results
ACKNOWLEDGEMENTS
None.
Saygın Avşar T, Jackson L, Barton P, Jones M, McLeod H. Towards optimum smoking cessation interventions during pregnancy: a household model to explore cost‐effectiveness. Addiction. 2022;117:2707–2719. 10.1111/add.15955
This research was part of the corresponding author's PhD at the University of Birmingham, which was funded by the Turkish Ministry of Education. The funder played no role in the design of the study nor in the preparation of the manuscript. TSA's time is supported by the National Institute for Health and Care Research Applied Research Collaboration (NIHR ARC) North Thames. H.McL.'s time is supported by the NIHR ARC West. The views expressed in this publication are those of the authors and not necessarily those of the NIHR ARC or the Department of Health and Social Care.
Funding information Turkish Ministry of Education, Grant/Award Number: PhD Funding
REFERENCES
- 1. Health and Social Care Information Centre (HSCI) . Statistics on Women's Smoking Status at Time of Delivery Leeds, UK, HSCI; 2019. [Google Scholar]
- 2. Avşar TS, McLeod H, Jackson L. Health outcomes of smoking during pregnancy and the postpartum period: an umbrella review. BMC Pregnancy Childbirth. 2021;21:254–353. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3. Chamberlain C, O'Mara‐Eves A, Porter J, Coleman T, Perlen SM, Thomas J, et al. Psychosocial interventions for supporting women to stop smoking in pregnancy. Cochrane Database Syst Rev. 2013;2:1–293. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4. Tappin D, Bauld L, Purves D, Boyd K, Sinclair L, MacAskill S, et al. Financial incentives for smoking cessation in pregnancy: randomised controlled trial. BMJ. 2015;350:h134. [DOI] [PubMed] [Google Scholar]
- 5. Fahy SJ, Cooper S, Coleman T, Naughton F, Bauld L. Provision of smoking cessation support for pregnant women in England: results from an online survey of NHS stop smoking services for pregnant women. BMC Health Serv Res. 2014;14:1–11. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6. Naughton F, Vaz LR, Coleman T, Orton S, Bowker K, Leonardi‐Bee J et al. Interest in and use of smoking cessation support across pregnancy and postpartum. Nicotine Tob Res, 2020; 22: 1178–1186. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7. Cooper S, Lewis S, Thornton JG, Marlow N, Watts K, Britton J, et al. The SNAP trial: a randomised placebo‐controlled trial of nicotine replacement therapy in pregnancy—clinical effectiveness and safety until 2 years after delivery, with economic evaluation. Health Technol Asses. 2014;18: 1–128. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8. Department of Health (DoH) . Towards a Smoke‐free Generation: A Tobacco Plan for England. London, UK: Health Behaviours, DoH ; 2017. [Google Scholar]
- 9. Chamberlain C, O'Mara‐Eves A, Porter J, Coleman T, Perlen SM, Thomas J, et al. Psychosocial interventions for supporting women to stop smoking in pregnancy. Cochrane Database Syst Rev. 2017;2:CD001055. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10. Morgan H, Hoddinott P, Thomson G, Crossland N, Farrar S, Yi D, et al. Psychosocial interventions for supporting women to stop smoking in pregnancy. Health Technol Assess. 2015;19(30):1–564. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11. World Health Organization (WHO) . Recommendations for the Prevention and Management of Tobacco Use and Second‐Hand Smoke Exposure in Pregnancy Geneva; Switzerland: WHO; 2013. [PubMed] [Google Scholar]
- 12. Coleman T, Chamberlain C, Cooper S, Leonardi‐Bee J. Efficacy and safety of nicotine replacement therapy for smoking cessation in pregnancy: systematic review and meta‐analysis. Addiction. 2011;106:52–61. [DOI] [PubMed] [Google Scholar]
- 13. Skivington K, Matthews L, Simpson SA, Craig P, Baird J, Blazeby JM, et al. A new framework for developing and evaluating complex interventions: update of Medical Research Council guidance. BMJ. 2021;374:n2061. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14. Jones M, Lewis S, Parrott S, Coleman T. Systematic critical review of previous economic evaluations of smoking cessation during pregnancy. BMJ Open. 2015;5:e008998. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15. Jones M, Smith M, Lewis S, Parrott S, Coleman T. A dynamic, modifiable model for estimating cost‐effectiveness of smoking cessation interventions in pregnancy: application to an RCT of self‐help delivered by text message. Addiction. 2018;114:353–65. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16. Donatelle RJ, Prows SL, Champeau D, Hudson D. Randomised controlled trial using social support and financial incentives for high risk pregnant smokers: significant other supporter (SOS) program. Tob Control. 2000;9:67–9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17. McBride CM, Baucom DH, Peterson BL, Pollak KI, Palmer C, Westman E, et al. Prenatal and postpartum smoking abstinence a partner‐assisted approach. Am J Prev Med. 2004;27:232–8. [DOI] [PubMed] [Google Scholar]
- 18. Cooper S, Orton S, Campbell KA, Ussher M, Coleman‐Haynes N, Whitemore R, et al. Attitudes to e‐cigarettes and cessation support for pregnant women from English stop smoking services: a mixed methods study. Int J Environ Res Public Health. 2019;16:110–117. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19. Jones M. The development of the Economic impacts of Smoking In Pregnancy (ESIP) model for measuring the impacts of smoking and smoking cessation during pregnancy Nottingham, UK: University of Nottingham; 2015. [Google Scholar]
- 20. Health and Social Care Information Centre (HSCI) . Infant Feeding Survey Leeds, UK: HSCI; 2010. [Google Scholar]
- 21. Department of Health (DoH) . NHS Reference Costs: Financial Year 2017–18 London, UK: DoH; 2018. [Google Scholar]
- 22. Agutu CA. The Timing if Maternal Smoking Cessation in Pregnancy and the Risk of Low Birth Weight: a Systematic Review and Meta‐Analysis. Nottingham, UK: Division of Epidemiology and Public Health, University of Nottingham; 2012.
- 23. Nicoletti D, Appel LD, Siedersberger Neto P, Guimarães GW, Zhang L. Maternal smoking during pregnancy and birth defects in children: a systematic review with meta‐analysis. Cad Saude Publica. 2014;30:2491–529. [DOI] [PubMed] [Google Scholar]
- 24. Leonardi‐Bee J, Jere ML, Britton J. Exposure to parental and sibling smoking and the risk of smoking uptake in childhood and adolescence: a systematic review and meta‐analysis. Thorax. 2011;66:847–55. [DOI] [PubMed] [Google Scholar]
- 25. Office for National Statistics (ONS) . Mortality Statistics: Death Registered in England and Wales 2018. Available at: https://www.ons.gov.uk/peoplepopulationandcommunity/birthsdeathsandmarriages/deaths/datasets/deathsregisteredinenglandandwalesseriesdrreferencetables. Accessed 5 May 2019.
- 26. Office for National Statistics (ONS) . Historic and Projected Mortality Data from the Period and Cohort Life Tables, 2016‐based London, UK: ONS; 2018. [Google Scholar]
- 27. Flack S, Taylor M, Trueman P. Cost‐Effectiveness of Interventions for Smoking Cessation York, UK: York Health Economics Consortium; 2007. [Google Scholar]
- 28. Doll R, Peto R, Wheatley K, Gray R, Sutherland I. Mortality in relation to smoking: 40 years’ observations on male British doctors. BMJ Clin Res. 1994;309:901–11. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29. Doll SR, Peto R, Boreham J, Sutherland I. Mortality in relation to smoking: 50 years’ observations on male British doctors. BMJ. 2004;328:1519–28. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30. Office for National Statistics (ONS) . Health Survey England 2017: Adult Health Related Behaviours. London, UK: ONS; 2018. Available at: https://digital.nhs.uk/data-and-information/publications/statistical/health-survey-for-england/2017. Accessed 5 May 2019. [Google Scholar]
- 31. Office for National Statistics (ONS) . Health Survey England 2017: Adult Health Related Behaviours Compendium: Adult Smoking Habits in Great Britain. 2018. Available at: https://www.ons.gov.uk/peoplepopulationandcommunity/healthandsocialcare/drugusealcoholandsmoking/datasets/adultsmokinghabitsingreatbritain. Accessed 5 May 2019.
- 32. Saygın Avşar T, McLeod H, Jackson L. Health outcomes of maternal smoking during pregnancy and postpartum period for the mother and infant: protocol for an umbrella review. Syst Rev. 2018;7:235–9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33. Koshy G, Delpisheh A, Brabin BJ. Dose response association of pregnancy cigarette smoke exposure, childhood stature, overweight and obesity. Eur J Public Health. 2011;21:286–91. [DOI] [PubMed] [Google Scholar]
- 34. Messiah SE, Arheart KL, Lipshultz SE, Bandstra ES, Miller TL. Perinatal factors associated with cardiovascular disease risk among preschool‐age children in the United States: an analysis of 1999‐2008 NHANES data. Int J Pediatr. 2012;2012:157237. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35. Health and Social Care Information Centre (HSCI) . Hospital Episode Statistics. NHS Maternity Statistics—England. Leeds, UK: HSCI; 2018. [Google Scholar]
- 36. Office for National Statistics (ONS) . Gestation‐specific infant mortality in England and Wales: Gestation mortality ref. tables. 2014. Available at: https://data.gov.uk/dataset/bb825554-d3d6-4a9f-9b2d-210b4d014b13/gestation-specific-infant-mortality-in-england-and-wales. Accessed 21 March 2018.
- 37. Office for National Statistics (ONS) . Child and infant mortality in England and Wales: 2017. 2019. Available at: https://www.ons.gov.uk/peoplepopulationandcommunity/birthsdeathsandmarriages/deaths/bulletins/childhoodinfantandperinatalmortalityinenglandandwales/2017. Accessed 10 March 2020.
- 38. Flowers H. Chronic Disease Prevalence by Age, Sex, SHA and UK country in 2008 London, UK: Eastern Region Public Health Observatory, Department of Health; 2010. [Google Scholar]
- 39. European Respiratory Society . The Economic Burden of Lung Disease. 2014. Available at: https://www.erswhitebook.org/chapters/the-economic-burden-of-lung-disease/. Accessed 10 March 2020.
- 40. Office for National Statistics (ONS) . Mid‐year population statistics for England and Wales. 2019. Available at: https://www.ons.gov.uk/peoplepopulationandcommunity/populationandmigration/populationestimates. Accessed 10 March 2020.
- 41. Health and Social Care Information Centre (HSCI) . Hospital Episode Statistics for England 2017 Leeds, UK, HSCI; 2018. [Google Scholar]
- 42. Burke H, Leonardi‐Bee J, Hashim A, Pine‐Abata H, Chen Y, Cook DG et al. Prenatal and passive smoke exposure and incidence of asthma and wheeze: systematic review and meta‐analysis. Pediatrics 2012; 129: 735–744. [DOI] [PubMed] [Google Scholar]
- 43. Office for National Statistics (ONS) . Unexplained deaths in infancy, England and Wales: 2016. 2018. Available at: https://www.ons.gov.uk/peoplepopulationandcommunity/birthsdeathsandmarriages/deaths/bulletins/unexplaineddeathsininfancyenglandandwales/2016. Accessed 5 May 2019.
- 44. Health and Social Care Information Centre (HSCI) . Health Survey for England 2016: Adult Health Trends Leeds, UK, 2017: HSCI. [Google Scholar]
- 45. Public Health England (PHE . Cancer Prevalence in England: 21 year prevalence by demographic and geographic measures London, UK: PHE; 2016. [Google Scholar]
- 46. Dietz PM, England LJ, Shapiro‐Mendoza CK, Tong VT, Farr SL, Callaghan WM. Infant morbidity and mortality attributable to prenatal smoking in the U.S. Am J Prev Med. 2010;39:45–52. [DOI] [PubMed] [Google Scholar]
- 47. Pineles BL, Hsu S, Park E, Samet JM. Systematic review and meta‐analyses of perinatal death and maternal exposure to tobacco smoke during pregnancy. Am J Epidemiol. 2016;184:87–97. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48. Mu M, Ye S, Bai MJ, Liu GL, Tong Y, Wang SF, et al. Birth weight and subsequent risk of asthma: a systematic review and meta‐analysis. Heart Lung Circ. 2014;23:511–9. [DOI] [PubMed] [Google Scholar]
- 49. Singleton RJ, Wirsing EA, Haberling DL, Christensen KY, Paddock CD, Hilinski JA, et al. Risk factors for lower respiratory tract infection death among infants in the United States. 1999–2004. Pediatrics. 2009;124:e768–76. [DOI] [PubMed] [Google Scholar]
- 50. Aliyu MH, Lynch ON, Wilson RE, Alio AP, Kristensen S, Marty PJ, et al. Association between tobacco use in pregnancy and placenta‐associated syndromes: a population‐based study. Arch Gynecol Obstet. 2011;283:729–34. [DOI] [PubMed] [Google Scholar]
- 51. Bouyer J, Coste J, Shojaei T, Pouly JL, Fernandez H, Gerbaud L, et al. Risk factors for ectopic pregnancy: a comprehensive analysis based on a large case‐control, population‐based study in France. Am J Epidemiol. 2003;157:185–94. [DOI] [PubMed] [Google Scholar]
- 52. Pineles BL, Park E, Samet JM. Systematic review and meta‐analysis of miscarriage and maternal exposure to tobacco smoke during pregnancy. Am J Epidemiol. 2014;179:807–23. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53. Tikkanen M, Nuutila M, Hiilesmaa V, Paavonen J, Ylikorkala O. Prepregnancy risk factors for placental abruption. Acta Obstet Gynecol Scand. 2006;85:40–4. [DOI] [PubMed] [Google Scholar]
- 54. Cui H, Gong TT, Liu C‐X, Wu Q‐J. Associations between passive maternal smoking during pregnancy and preterm birth: evidence from a meta‐analysis of observational studies. PLOS ONE. 2016;11:e0147848. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55. Salmasi G, Grady R, Jones J, McDonald SD. Environmental tobacco smoke exposure and perinatal outcomes: a systematic review and meta‐analyses. Acta Obstet Gynecol Scand. 2010;89:423–41. [DOI] [PubMed] [Google Scholar]
- 56. Shah NR, Bracken MB. A systematic review and meta‐analysis of prospective studies on the association between maternal cigarette smoking and preterm delivery. Am J Obstet Gynecol. 2000;182:465–72. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57. Lee PN, Forey BA, Coombs KJ, Hamling JS, Thornton AJ. Epidemiological evidence relating environmental smoke to COPD in lifelong non‐smokers: a systematic review. F1000Res. 2018;7:146–79. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58. Oono IP, Mackay DF, Pell JP. Meta‐analysis of the association between secondhand smoke exposure and stroke. J Public Health. 2011;33:496–502. [DOI] [PubMed] [Google Scholar]
- 59. Lee P, Fry JS, Forey B, Hamling JS, Thornton AJ. Environmental tobacco smoke exposure and lung cancer: a systematic review. World J MEta‐Anal. 2016;4(2):10–43. [Google Scholar]
- 60. He J, Vupputuri S, Allen K, Prerost MR, Hughes J, Whelton PK. Passive smoking and the risk of coronary heart disease—a meta‐analysis of epidemiologic studies. N Engl J Med. 1999;340:920–6. [DOI] [PubMed] [Google Scholar]
- 61. Maheswaran H, Petrou S, Rees K, Stranges S. Estimating EQ‐5D utility values for major health behavioural risk factors in England. J Epidemiol Community Health. 2013;67:172–80. [DOI] [PubMed] [Google Scholar]
- 62. Gillespie P, O'Neill C, Adams E, Turner K, O'Donovan D, Brugha R et al. The cost and cost‐effectiveness of opportunistic screening for chlamydia trachomatis in Ireland. Sex Transm Infect 2012;88:222–8. [DOI] [PubMed] [Google Scholar]
- 63. Partridge JC, Sendowski MD, Martinez AM, Caughey AB. Resuscitation of likely nonviable infants: a cost–utility analysis after the born‐alive infant protection act. Am J Obstet Gynecol. 2012;206:49–59. PMID: e1–49.e10. [DOI] [PubMed] [Google Scholar]
- 64. Community Pharmacy Medicines Management Project Evaluation Team . The MEDMAN study: a randomized controlled trial of community pharmacy‐led medicines management for patients with coronary heart disease. Fam Pract. 2007;24:189–200. [DOI] [PubMed] [Google Scholar]
- 65. Starkie HJ, Briggs AH, Chambers MG, Jones P. Predicting EQ‐5D values using the SGRQ. Value Health. 2011;14:354–60. [DOI] [PubMed] [Google Scholar]
- 66. Pickard AS, Neary MP, Cella D. Estimation of minimally important differences in EQ‐5D utility and VAS scores in cancer. Health Qual Life Outcomes. 2007;5:70–8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 67. Haacke C, Althaus A, Spottke A, Siebert U, Back T, Dodel R. Long‐term outcome after stroke: evaluating health‐related quality of life using utility measurements. Stroke. 2006;37:193–8. [DOI] [PubMed] [Google Scholar]
- 68. Carroll KN, Gebretsadik T, Griffin MR, Dupont WD, Mitchel EF, Wu P, et al. Maternal asthma and maternal smoking are associated with increased risk of bronchiolitis during infancy. Pediatrics. 2007;119:1104–12. [DOI] [PubMed] [Google Scholar]
- 69. Jiang X, Sun L, Wang B, Yang X, Shang L, Zhang Y. Health‐related quality of life among children with recurrent respiratory tract infections in Xi'an, China. PLOS ONE. 2013;8:e56945. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 70. Rolfsjord LB, Skjerven HO, Bakkeheim E, Carlsen KH, Hunderi JO, Kvenshagen BK, et al. Children hospitalised with bronchiolitis in the first year of life have a lower quality of life nine months later. Acta Paediatr. 2015;104:53–8. [DOI] [PubMed] [Google Scholar]
- 71. Sarria EE, Mundstock E, Machado DG, Mocelin HT, Fischer GB, Furlan SP, et al. Health‐related quality of life in patients with bronchiolitis obliterans. J Pediatr. 2018;94:374–9. [DOI] [PubMed] [Google Scholar]
- 72. Athanazio R. Airway disease: similarities and differences between asthma, COPD and bronchiectasis. Clinics. 2012;67:1335–43. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 73. Curtis L, Burns A. Unit Costs of Health and Social Care 2018 Canterbury, Kent: Personal Social Services Research Unit, University of Kent; 2018. [Google Scholar]
- 74. National Institute for Care and Excellence (NICE) . Methods for the Development of NICE Public Health Guidance London, UK: NICE; 2013. [PubMed] [Google Scholar]
- 75. Claxton K, Paulden M, Gravelle H, Brouwer W, Culyer AJ. Discounting and decision making in the economic evaluation of health‐care technologies. Health Econ. 2011;20:2–15. [DOI] [PubMed] [Google Scholar]
- 76. Attema AE, Brouwer WB, Claxton K. Discounting in economic evaluations. Pharmacoeconomics. 2018;36:745–58. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 77. National Institute for Health and Care Excellence (NICE) . Guide to the Methods of Technology Appraisal London, UK: NICE; 2013. [PubMed] [Google Scholar]
- 78. Hatswell AJ, Bullement A, Briggs A, Paulden M, Stevenson MD. Probabilistic sensitivity analysis in cost‐effectiveness models: determining model convergence in cohort models. Pharmacoeconomics. 2018;36:1421–6. [DOI] [PubMed] [Google Scholar]
- 79. Boyd KA, Briggs AH, Bauld L, Sinclair L, Tappin D. Are financial incentives cost‐effective to support smoking cessation during pregnancy? Addiction. 2016;111:360–70. [DOI] [PubMed] [Google Scholar]
- 80. Ratner PA, Johnson JL, Bottorff JL. Smoking relapse and early weaning among postpartum women: is there an association? Birth. 1999;26:76–82. [DOI] [PubMed] [Google Scholar]
- 81. Public Health Scotland (PHS) . PHS's Scotland's SMR02 data scheme (Table 3 maternal smoking status). Scotland UK: PHS; 2021.
- 82. Centers for Disease Control and Prevention (CDC . ). Cigarette Smoking During Pregnancy: United States 2016. NCHS Data Brief no. 305. Atlanta, GA: CDC; 2018. [PubMed]
- 83. Sinclair L, McFadden M, Tilbrook H, Mitchell A, Keding A, Watson J et al. The smoking cessation in pregnancy incentives trial (CPIT): study protocol for a phase III randomised controlled trial. Trials 2020; 21: 1–12. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 84. Kia F, Tosun N, Carlson S, Allen S. Examining characteristics associated with quitting smoking during pregnancy and relapse postpartum. Addict Behav. 2018;78:114–9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 85. Prady SL, Kiernan K, Bloor K, Pickett KE. Do risk factors for post‐partum smoking relapse vary according to marital status? Matern Child Health J 2012; 16: 1364–1373. [DOI] [PubMed] [Google Scholar]
- 86. McNeill A, Brose LS, Calder R, Bauld L, Robson D. Evidence Review of e‐Cigarettes and Heated Tobacco Products London, UK: Public Health England; 2018. [Google Scholar]
- 87. Calder R, Gant E, Bauld L, McNeill A, Robson D, Brose LS. Vaping in pregnancy: a systematic review. Nicotine Tob Res 2021; 23: 1451–1458. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 88. National Institute for Care and Excellence (NICE) . Evidence Reviews for Nicotine Replacement Therapies and E‐Cigarettes in Pregnancy: Update. NICE guideline NG209 London, UK: NICE; 2021. [PubMed] [Google Scholar]
- 89. Taylor M. Economic Analysis of Interventions for Smoking Cessation in Pregnancy London, UK: National Institute for Care and Excellence; 2009. [Google Scholar]
- 90. NHS England . Better Births: Improving Maternity Services in England. Leeds, UK: NHS England; 2016. [Google Scholar]
- 91. Saygın Avşar T, Jackson L, McLeod H. Potential for health economics to influence policies on tobacco use during pregnancy in low‐income and middle‐income countries: a qualitative case study. BMJ Open. 2021;11:e045624. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 92. Nohlert E, Tegelberg Å, Tillgren P, Johansson P, Rosenblad A, Helgason ÁR. Comparison of a high and a low intensity smoking cessation intervention in a dentistry setting in Sweden—a randomized trial. BMC Public Health. 2009;9:1–10. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Data S1: Appendix A: Technical summary of the development of ESIP.H
Appendix B: Hypothetical cessation intervention
Appendix C: Cautious‐case ESIP.H model outputs and outcomes at delivery
Appendix D: Deterministic sensitivity analysis
Appendix E: Probabilistic sensitivity analysis
Table S1. Transition probabilities for the maternal smoking behaviour change within the two years after delivery
Table S2: Transition probabilities for the smoking behaviour change in the mother and partner lifetime components
Table S3: Smoking uptake probabilities based on parental smoking
Table S4: Transition probabilities for the smoking behaviour change in the offspring adulthood component
Table S5: Probabilities of within‐pregnancy outcomes for mother and infants
Table S6: ORs (95% CI) compared to never smokers used for within pregnancy health conditions
Table S7: Prevalence of CHD, COPD, Stroke and LC amongst never smokers not exposed to SHS
Table S8: Relative risks for lifetime morbidities used in lifetime models
Table S9: Utility tariff for smokers and non‐smokers
Table S10: Unit costs used in the study
Table S11: Abstinence levels amongst mothers at different time points used for voucher cost calculations
Table S12: Abstinence levels amongst partners at different time points used for voucher cost estimations
Table S13: Estimated cost of the hypothetical intervention
Table S14. Cautious‐case ESIP.H model outputs for the hypothetical household intervention
Table S15. Outcomes at delivery for the base‐case and cautious‐case
Table S16: Distributions used in the probabilistic sensitivity analysis
Figure S1: Deterministic sensitivity analysis for SFH12m base‐case household lifetime results
Figure S2: Deterministic sensitivity analysis for SFH12m cautious‐case household lifetime results


