Abstract
Aims
To conduct a systematic review and network meta‐analysis to determine the comparative effectiveness of sodium‐glucose cotransporter‐2 (SGLT2) inhibitors, glucagon‐like peptide‐1 receptor agonists (GLP‐1RAs) and dipeptidyl peptidase‐4 (DPP‐4) inhibitors in patients with diabetic kidney disease (DKD).
Methods
Phase III or IV randomized, placebo‐controlled trials evaluating SGLT2 inhibitors, GLP‐1RAs or DPP‐4 inhibitors in patients with DKD were identified from the MEDLINE database. The outcomes of interest were a kidney‐specific composite outcome, kidney disease progression, major adverse cardiovascular events (MACE), hospitalization for heart failure (HHF) and cardiovascular death. A network meta‐analysis was conducted to calculate hazard ratios (HRs) and 95% confidence intervals (CIs).
Results
Sixteen trials representing a total of 46 292 patients were included. SGLT2 inhibitors significantly reduced the risk of the kidney‐specific composite outcome by 26% compared to GLP‐1RAs (HR 0.74, 95% CI 0.62‐0.88) and by 36% compared to DPP‐4 inhibitors (HR 0.64, 95% CI 0.52‐0.79). The risk of MACE was significantly reduced with SGLT2 inhibitors (by 18%; HR 0.82, 95% CI 0.72‐0.93), and with GLP‐1RAs (by 18%; HR 0.82, 95% CI 0.69‐0.96), compared to DPP‐4 inhibitors. SGLT2 inhibitors significantly reduced the risk of HHF by 28% compared to GLP‐1RAs (HR 0.72, 95% CI 0.56‐0.92) and by 41% compared to DPP‐4 inhibitors (HR 0.59, 95% CI 0.49‐0.71).
Conclusions
A clear advantage was demonstrated by SGLT2 inhibitors in reducing the risks of CV and renal events in patients with DKD, compared to GLP‐1RAs and DPP‐4 inhibitors. We recommend that SGLT2 inhibitors be considered the treatment of choice in patients with DKD.
Keywords: diabetic kidney disease, dipeptidyl peptidase‐4 inhibitors, glucagon‐like peptide‐1 receptor agonists, network meta‐analysis, sodium‐glucose cotransporter‐2 inhibitors
1. INTRODUCTION
An estimated 463 million people live with diabetes worldwide, with type 2 diabetes mellitus accounting for 90% of that total. 1 Diabetic kidney disease (DKD) is one of the major complications of type 2 diabetes mellitus, occurring in approximately 25% to 50% of adults with type 2 diabetes mellitus. 2 , 3 , 4 , 5 , 6 , 7 , 8 , 9 DKD manifests clinically as persistent microalbuminuria, reduced glomerular filtration rate (GFR), or both, eventually leading to renal impairment and end‐stage kidney disease (ESKD). 2 , 10 The incidence of kidney disease in patients with type 2 diabetes mellitus substantially increases both cardiovascular (CV) and all‐cause mortality risk; DKD is the primary cause of the excess mortality in patients with type 2 diabetes mellitus. 11
Intensive glycemic control has been shown to reduce the risk of development of microalbuminuria; however, there was little or no benefit with regard to the risk of kidney disease progression compared with standard control. 12 The renal protective effects of traditional antidiabetic drugs such as insulin, sulphonylureas and metformin have not been specifically studied in large clinical studies. In the past two decades, only renin‐angiotensin‐aldosterone system (RAAS) blockade has been shown to be effective for renoprotection in DKD. 13 , 14 Consequently, progression to ESKD and CV mortality have been two major unmet medical needs in patients with DKD. 15
There is new evidence that novel antidiabetic drug classes such as sodium‐glucose cotransporter‐2 (SGLT2) inhibitors and glucagon‐like peptide‐1 receptor agonists (GLP‐1RAs) improve CV and renal outcomes in patients with type 2 diabetes mellitus. Several large CV outcome trials (CVOTs) studying new antidiabetic drugs in patients with type 2 diabetes mellitus who were at high risk of CV disease (CVD) or who had existing CVD examined kidney effects as secondary outcomes. In the CVOTs, SGLT2 inhibitors (EMPA‐REG OUTCOME, CANVAS, DECLARE–TIMI 58) 16 , 17 , 18 and GLP‐1RAs (LEADER, SUSTAIN‐6) 19 , 20 significantly reduced the risk of new or worsening nephropathy compared with placebo. In addition, large renal outcomes trials of SGLT2 inhibitors (CREDENCE, DAPA‐CKD) 21 , 22 have demonstrated significant benefits of SGLT2 inhibitors, on top of RAAS blockade, in reducing the risk of kidney disease progression and development of ESKD. However, it is still unclear which class of drug shows the greatest effectiveness in patients with DKD. Therefore, we performed this systematic review and network meta‐analysis to determine the comparative effectiveness of SGLT2 inhibitors, GLP‐1RAs and dipeptidyl peptidase‐4 (DPP‐4) inhibitors on CV and renal outcomes in patients with DKD.
2. METHODS
This systematic review and network meta‐analysis was conducted according to a prespecified protocol (INPLASY registration number: INPLASY2021120070). The results from this network meta‐analysis are reported according to the Preferred Reporting Items for Systematic Reviews and Meta‐Analyses (PRISMA) statement.
2.1. Search strategy
We conducted a systematic search of the MEDLINE database (via PubMed) using a predefined search strategy to identify relevant randomized controlled trials (RCTs) reported in English up to July 2021. The following search algorithm was used: (“sodium‐glucose transporter 2 inhibitors” OR “dipeptidyl‐peptidase IV inhibitors” OR “glucagon‐like peptide 1”) AND (“diabetic nephropathies”[MeSH]) OR “chronic kidney disease”) AND (“diabetes mellitus, type 2”[MeSH]) AND (randomized controlled). Each search string also contained the synonyms and related keywords of the search terms mentioned above. The complete search strings are provided in Table S1.
2.2. Study selection
Studies were included if they met the following inclusion criteria for this network meta‐analysis: (a) the trial was a Phase III or IV prospective, randomized, controlled, event‐driven CV or kidney outcome trial; (b) patients were adults with type 2 diabetes mellitus and kidney disease; (c) interventions were SGLT2 inhibitors, GLP‐1RAs or DPP‐4 inhibitors; and (d) the comparator was an active or placebo control.
2.3. Data extraction
The studies retrieved from the database search were assessed for relevance after screening of titles and abstracts. The full texts of relevant studies were then assessed for final eligibility according to the inclusion and exclusion criteria set for this network meta‐analysis. Prespecified data were extracted from each of the included studies using a standardized Excel data extraction sheet by one researcher and were independently reviewed by two researchers. The prespecified data extracted for each eligible study included study design, intervention characteristics, baseline characteristics of interest and study outcomes. Any disagreements during data extraction were resolved by consensus.
2.4. Study outcomes
The outcomes of interest for this network meta‐analysis were: a kidney‐specific composite outcome, defined as doubling of serum creatinine or a 40% or greater decline in estimated GFR (eGFR), development of ESKD, or death due to kidney disease; kidney disease progression, defined as a composite of doubling of serum creatinine or 40% or greater decline in eGFR, development of ESKD, or death due to kidney disease or CV disease; major adverse CV events (MACE), defined as a composite of CV death, nonfatal myocardial infarction (MI) or non‐fatal stroke; hospitalization for heart failure (HHF); and CV death.
2.5. Risk‐of‐bias and quality assessment
The quality of the included studies and the risk of bias were assessed using the Cochrane Risk‐of‐Bias tool. Any disagreements during assessment of risk of bias were resolved by consensus.
2.6. Statistical analysis
Hazard ratios (HRs) and 95% confidence intervals (CIs) were calculated for each study to assess the effect sizes. Statistical tests were performed using STATA 15.0, as well as the statistical packages “netmeta” for network meta‐analysis and “gemtc” for Bayesian analysis in R (version 4.1.2). All tests were two‐sided and a P value < 0.05 was considered statistically significant. The net split results were presented as forest plots. The netmeta package on R was used to evaluate the consistency between direct and indirect estimates. Potential publication bias was estimated using funnel plots and Egger tests.
A Bayesian network meta‐analysis was employed for the sensitivity analysis to analyse direct and indirect comparisons and to rank the results of the network meta‐analysis. A Markov Monte Carlo algorithm was used to derive inferences from the random‐effects Bayesian network. A total of 100 000 iterations were run for each chain, and 5000 burn‐ins were used for the outcomes. Every 10th data point for 10 000 samples per channel was extracted. Model convergence was measured using trace plots and Gelman‐Rubin plots.
3. RESULTS
3.1. Studies included in the network meta‐analysis
Out of 377 articles identified in the initial database search, a total of 20 publications from 16 studies fulfilled the eligibility criteria and were included in the final analysis (Figure S1), representing a total of 46 292 patients. All studies had placebo as the comparator. The major characteristics of the included studies are summarized in Table 1, including the criteria used to define DKD in each study. The quality evaluation of the included studies is presented in Figure S2. The network profile of the included studies indicates that the SGLT2 inhibitor‐placebo arm had the most comparisons, with seven studies, while four studies were included in the DPP‐4 inhibitor‐placebo arm and five in the GLP‐1RA‐placebo arm (Figure 1).
TABLE 1.
Study design and patient population of the included studies
| Study: first author, year | Intervention (dose) | Number of patients | Patient population (inclusion criteria) | DKD criteria (data inclusion criteria for meta‐analysis) | Median follow‐up, years |
|---|---|---|---|---|---|
| DPP‐4 inhibitors | |||||
| Udell 2015 33 |
Saxagliptin (2.5 mg OD) |
2240 | T2DM and renal impairment (eGFR 30‐50 mL/min/1.73 m2) | eGFR 30‐50 mL/min/1.73 m2 | 2 |
| Cornel 2016 34 |
Sitagliptin (100 mg OD) |
3301 | Age ≥18 years; T2DM with or without CKD |
eGFR 45‐59 mL/min/1.73 m2 (n = 2518) eGFR 30‐44 mL/min/1.73 m2 (n = 783) |
3 |
|
McGuire 2019 35 Rosenstock 2019 36 |
Linagliptin (5 mg OD) |
5147 | Age ≥18 years; T2DM with or without CKD | eGFR <60 mL/min/1.73 m2 | 2.2 |
| Ferreira 2020 37 |
Alogliptin (6.25‐25 mg OD) |
1434 | T2DM with or without CKD | eGFR <60 mL/min/1.73 m2 | 1.5 |
| GLP‐1 RAs | |||||
| Marso 2016 20 |
Semaglutide (0.5‐1 mg OW) |
939 | Age ≥50 years; T2DM with or without CKD | eGFR <60 mL/min/1.73 m2 | 2.1 |
|
Mann 2017 19 Mann 2018 38 |
Liraglutide (1.8 mg OD) |
2158 | T2DM with or without CKD | eGFR <60 mL/min/1.73 m2 | 3.8 |
| Husain 2019 39 |
Semaglutide (14 mg OD) |
856 | Age ≥50 years; T2DM with or without CKD | eGFR <60 mL/min/1.73 m2 | 1.3 |
| Gerstein 2019 40 |
Dulaglutide (1.5 mg OW) |
2199 | Age ≥50 years; T2DM with or without CKD | eGFR <60 mL/min/1.73 m2 | 5.4 |
| Bethel 2020 41 |
Exenatide (2 mg OW) |
3177 | T2DM with or without CKD | eGFR <60 mL/min/1.73 m2 | 3.2 |
| SGLT2 inhibitors | |||||
|
Wanner 2016 42 Wanner 2018 43 |
Empagliflozin (10‐25 mg OD) | 1819 | T2DM with or without CKD | eGFR <59 mL/min/1.73 m2 | 3.1 |
| Neuen 2018 44 | Canagliflozin (100‐300 mg OD) | 2039 | T2DM with or without CKD and either age ≥30 years with established atherosclerotic vascular disease or age ≥50 years with 2 or more CV risk factors | eGFR <60 mL/min/1.73 m2 | 3.6 |
| Wiviott 2019 17 | Dapagliflozin (10 mg OD) | 1265 | Age ≥40 years; T2DM (with a glycated HbA1c of at least 6.5% but below 12.0%), and a creatinine clearance of ≥60 mL per min | eGFR <60 mL/min/1.73 m2 | 4.2 |
|
Cannon 2020 45 Cherney 2021 46 |
Ertugliflozin (5‐15 mg OD) | 1807 | Age ≥40 years; T2DM (with a HbA1c level of 7.0%‐10.5%) and established atherosclerotic CV disease | eGFR <60 mL/min/1.73 m2 | 3 |
| Perkovic 2019 22 | Canagliflozin (100 mg OD) | 4401 | Age ≥30 years; T2DM (with HbA1c level of 6.5%‐10.5%) and CKD (eGFR of 30 to <90 mL/min/1.73 m2) | eGFR 30 to <90 mL/min/1.73 m2 | 2.62 |
| Heerspink 2020 21 | Dapagliflozin (10 mg OD) | 2906 | CKD (eGFR of 25‐75 mL/min/1.73 m2 and UACR of 200‐5000 mg/g) with or without T2DM (patients with CKD + T2DM were included) | eGFR 25‐75 mL/min/1.73 m2 | 2.4 |
| Bhatt 2021 47 | Sotagliflozin (200‐400 mg OD) | 10 584 | Age ≥18 years; T2DM with or without CKD | eGFR ≥25 and ≤60 mL/min/1.73 m2 | 1.3 |
Abbreviations: CKD, chronic kidney disease; DKD, diabetic kidney disease; DPP‐4, dipeptidyl peptidase‐4; eGFR, estimated glomerular filtration rate; GLP‐1RA, glucagon‐like peptide‐1 receptor agonist; HbA1c, glycated hemoglobin; OD, once daily; OW, once weekly; SGLT2, sodium‐glucose cotransporter 2; T2DM, type 2 diabetes mellitus; UACR, urine albumin‐creatinine ratio.
FIGURE 1.
Overall network profile. This network meta‐analysis included four studies that evaluated dipeptidyl peptidase‐4 (DPP‐4) inhibitors, five that evaluated glucagon‐like peptide‐1 receptor agonists (GLP‐1Ras) and seven that evaluated sodium‐glucose cotransporter‐2 (SGLT2) inhibitors
3.2. Network meta‐analysis of treatment groups
3.2.1. Renal outcomes
Ten of the included studies reported a kidney‐specific composite outcome (six with SGLT2 inhibitors, three with GLP‐1RAs, and one with DPP‐4 inhibitors), while only four studies reported kidney disease progression (all with SGLT2 inhibitors). The within‐design heterogeneity was not significant (P = 0.6270), indicating that the network model for a kidney‐specific composite outcome was acceptable. No publication bias was found according to the funnel plot (Figure S3A) and Egger test (P = 0.1716).
The effect size estimates table and forest plots (Table 2 and Figure 2A) showed that SGLT2 inhibitors and GLP‐1RAs significantly reduced the risk of the kidney‐specific composite outcome by 36% (HR 0.64, 95% CI 0.57‐0.71) and 14% (HR 0.86, 95% CI 0.75‐0.99), respectively, compared with placebo. However, DPP‐4 inhibitors did not alter the risk of kidney‐specific outcome (HR 0.99, 95% CI 0.83‐1.18) compared with placebo.
TABLE 2.
Network meta‐analysis showing effect size (hazard ratio) and 95% confidence interval for the kidney‐specific composite outcome, major adverse cardiovascular (CV) events, CV death and hospitalization for heart failure. Comparisons between treatments should be read from left to right, and the hazard ratio is in the cell in common between the column‐defining treatment and the row‐defining treatment. Hazard ratio <1 favors the column‐defining treatment
| Kidney‐specific composite outcome | |||
|---|---|---|---|
| SGLT2 inhibitors | |||
| 0.74 (0.62,0.88) | GLP‐1RAs | ||
| 0.64 (0.52,0.79) | 0.87 (0.70,1.09) | DPP‐4 inhibitors | |
| 0.64 (0.57,0.71) | 0.86 (0.75, 0.99) | 0.99 (0.83,1.18) | Placebo |
| MACE | |||
|---|---|---|---|
| SGLT2 inhibitors | |||
| 0.95 (0.82,1.10) | GLP‐1RAs | ||
| 0.81 (0.71,0.92) | 0.85 (0.73,0.99) | DPP‐4 inhibitors | |
| 0.83 (0.76,0.91) | 0.87 (0.78, 0.98) | 1.03 (0.93,1.13) | Placebo |
| CV death | |||
|---|---|---|---|
| SGLT2 inhibitors | |||
| 0.97 (0.78,1.22) | GLP‐1RAs | ||
| 0.85 (0.71,1.02) | 0.87 (0.69, 1.10) | DPP‐4 inhibitors | |
| 0.86 (0.76,0.97) | 0.88 (0.73, 1.07) | 1.01 (0.89,1.15) | Placebo |
| HHF | |||
|---|---|---|---|
| SGLT2 inhibitors | |||
| 0.72 (0.56,0.92) | GLP‐1RAs | ||
| 0.59 (0.49,0.71) | 0.82 (0.64,1.05) | DPP‐4 inhibitors | |
| 0.64 (0.56,0.73) | 0.88 (0.72,1.09) | 1.08 (0.94,1.24) | Placebo |
Note: Shaded regions indicate the drug classes being compared, and define the columns and rows.
Abbreviations: CV, cardiovascular; DPP‐4, dipeptidyl peptidase‐4; GLP‐1RA, glucagon‐like peptide‐1 receptor agonist; HHF, hospitalization for heart failure; MACE, major adverse cardiovascular events; SGLT2, sodium‐glucose cotransporter 2.
FIGURE 2.
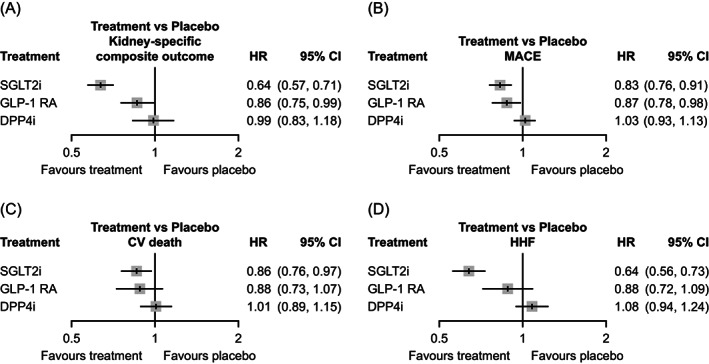
Risk of renal and cardiovascular (CV) outcomes with different antidiabetic drug classes. Forest plots comparing the risks of (A) kidney‐specific outcome, (B) major adverse cardiovascular events (MACE), (C) cardiovascular (CV) death and (D) hospitalization for heart failure (HHF) with different antidiabetic drug classes compared to placebo. DPP‐4, dipeptidyl peptidase‐4; GLP‐1RA, glucagon‐like peptide‐1 receptor agonist; SGLT2, sodium‐glucose cotransporter‐2
There was strong consistency between direct and indirect evidence for the kidney‐specific composite outcome (Figure 3A). SGLT2 inhibitors significantly reduced the risk of the kidney‐specific composite outcome by 26% compared to GLP‐1RAs (HR 0.74, 95% CI 0.62‐0.88) and by 36% compared to DPP‐4 inhibitors (HR 0.64, 95% CI 0.52‐0.79). GLP‐1RAs did not significantly alter the risk of the kidney‐specific composite outcome compared to DPP‐4 inhibitors (HR 0.87, 95% CI 0.70‐1.09; Table 2).
FIGURE 3.
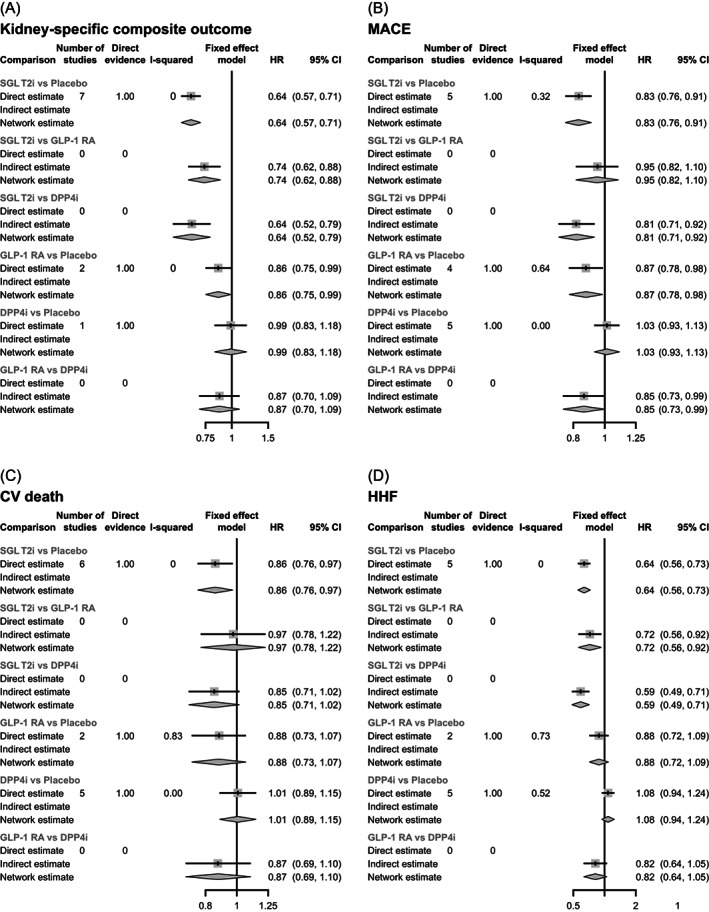
Comparison between treatments for (A) kidney‐specific composite outcome, (B) major adverse cardiovascular events (MACE), (C) cardiovascular (CV) death and (D) hospitalization for heart failure (HHF). DPP‐4, dipeptidyl peptidase‐4; GLP‐1RA, glucagon‐like peptide‐1 receptor agonist; SGLT2, sodium‐glucose cotransporter‐2 inhibitor
The rankogram (Figure S4A) showed that SGLT2 inhibitors had the highest probability of reducing the risk of the kidney‐specific composite outcome (100% probability of being the best treatment), followed by GLP‐1RAs (88.5% probability of being the second‐best treatment) and DPP‐4 inhibitors (10.9% probability of being the second‐best treatment, 44.7% probability of being the third‐best treatment).
As all four studies that reported kidney disease progression assessed SGLT2 inhibitors versus placebo, a traditional meta‐analysis was performed. No publication bias was found according to the funnel plot (Figure S3B) and Harbord test (Egger test, P = 0.971). SGLT2 inhibitors reduced the risk of kidney disease progression by 30% (HR 0.70, 95% CI 0.63‐0.77) compared with placebo (Figure S5).
3.2.2. CV outcomes
Fifteen of the included studies reported outcomes for MACE (six with SGLT2 inhibitors, four with GLP‐1RAs and five with DPP‐4 inhibitors); 13 studies reported outcomes for CV death (six with SGLT2 inhibitors, two with GLP‐1RAs, and five with DPP‐4 inhibitors); and 13 studies reported outcomes for HHF (six with SGLT2 inhibitors, two with GLP‐1RAs and five with DPP‐4 inhibitors). Although the DAPA‐CKD study reported outcomes for MACE and HHF, data for the DKD subgroup were unavailable. 21 Therefore, the MACE and HHF data from the DAPA‐CKD study were excluded from the network meta‐analysis, resulting in the inclusion of 14 studies for MACE and 12 studies for HHF. The within‐design heterogeneity was not significant for MACE (P = 0.0950), CV death (P = 0.5741) or HHF (P = 0.1680). No publication bias was found for any of the three outcomes according to the funnel plots (Figure S3C‐E) and Egger tests (P = 0.7147 for MACE, P = 0.5957 for CV death and P = 0.5198 for HHF).
The effect size estimates table and forest plots for each comparison (Table 2, Figure 2B‐D) showed that SGLT2 inhibitors significantly reduced the risk of MACE by 17% (HR 0.83, 95% CI 0.76‐0.91), the risk of CV death by 14% (HR 0.86, 95% CI 0.76‐0.97) and the risk of HHF by 36% (HR 0.64, 95% CI 0.56‐0.73) compared with placebo. While GLP‐1RAs significantly reduced the risk of MACE by 13% (HR 0.87, 95% CI 0.78‐0.98), the risk of CV death (HR 0.88, 95% CI 0.73‐1.07) and HHF (HR 0.88, 95% CI 0.72‐1.09) was not significantly different compared with placebo. In contrast, the risk of MACE (HR 1.03, 95% CI 0.93‐1.13), CV death (HR 1.01, 95% CI 0.89‐1.15) and HHF (HR 1.08, 95% 0.94‐1.24) was comparable between DPP‐4 inhibitors and placebo groups.
For all outcomes, there was strong consistency between direct and indirect evidence (Figure 3B‐D). As shown in Table 2, the risk of MACE was significantly reduced with SGLT2 inhibitors by 19% (HR 0.81, 95% CI 0.71‐0.92) and with GLP‐1RAs (by 15%; HR 0.85, 95% CI 0.73‐0.99) compared to DPP‐4 inhibitors. However, the risk of MACE was comparable between SGLT2 inhibitors and GLP‐1RAs (HR 0.95, 95% CI 0.82‐1.10). The risk of CV death was not significantly different among all three treatments (HR 0.97, 95% CI 0.78‐1.22 between SGLT2 inhibitors and GLP‐1RAs; HR 0.85, 95% CI 0.71‐1.02 between SGLT2 inhibitors and DPP‐4 inhibitors; and HR 0.87, 95% CI 0.69‐1.10 between GLP‐1RAs and DPP‐4 inhibitors). SGLT2 inhibitors significantly reduced the risk of HHF by 28% compared to GLP‐1RAs (HR 0.72, 95% CI 0.56‐0.92) and by 41% compared to DPP‐4 inhibitors (HR 0.59, 95% CI 0.49‐0.71). The risk of HHF was comparable between GLP‐1RAs and DPP‐4 inhibitors (HR 0.82, 95% CI 0.64‐1.05).
The rankograms (Figure S4B‐D) showed that SGLT2 inhibitors and GLP‐1RAs had a similar probability of being the best treatment for reducing the risk of MACE (54.5% and 45.1%, respectively), with DPP‐4 inhibitors have a 30.3% probability of being the third‐best treatment. Similarly, SGLT2 inhibitors and GLP‐1RAs had a 58.2% and 40.4% probability of being the best treatment to reduce risks of CV death, respectively, while there was a 35.0% probability that DPP‐4 inhibitors were the third‐best treatment. In contrast, SGLT2 inhibitors had the highest probability of being the best treatment for HHF (99.5%), followed by GLP‐1RAs (86.1% probability of being the second‐best treatment) and placebo (77.3% probability of being the third‐best treatment).
Analyses of MACE and HHF outcomes including data from the DAPA‐CKD study are shown in Figures S6 to S8. The results were consistent with the main analysis.
3.3. Sensitivity analyses
The results of the Bayesian sensitivity analyses were consistent with the frequentist analysis. The network plots corresponded with the number of studies included for each outcome (Figure S9A‐D). The rankograms showed that SGLT2 inhibitors were the most likely to reduce the risks of the kidney‐specific outcome and HHF, followed by GLP‐1RAs, while SGLT2 inhibitors and GLP‐1RAs had comparable probabilities of reducing the risks of MACE and CV death (Figure S10A‐D).
4. DISCUSSION
This network meta‐analysis of 16 RCTs compared SGLT2 inhibitors, GLP‐1RAs and DPP‐4 inhibitors to identify the drug class that lowers CV and renal risk by the greatest extent in patients with DKD. The inclusion of the latest results from 16 high‐quality CV and renal outcome trials encompassing 46 292 patients allowed for better statistical power to compare multiple outcomes and drug classes. The main strength of this study is that the analysis was focused specifically on patients with DKD. This allowed, for the first time, comparison of the efficacy of novel antidiabetic drug classes on CV and renal outcomes in patients with DKD.
The results showed that, in comparison with other treatments, intervention with SGLT2 inhibitors led to the most favourable outcomes. SGLT2 inhibitors significantly lowered the risks of MACE, CV death, HHF and the kidney‐specific composite outcome by 17%, 14%, 36% and 36%, respectively, compared with placebo. While GLP‐1RAs lowered the risks of MACE and the kidney‐specific composite outcome by 13% and 14%, respectively, there was no benefit with regard to the risk of CV death and HHF compared with placebo. In contrast, DPP‐4 inhibitors did not significantly alter the risk of kidney‐specific outcome and CV outcomes as compared to placebo. These data on the comparative efficacy of antidiabetic therapies based on the totality of available data will help clinicians make informed treatment decisions for patients with DKD.
4.1. Advantages of SGLT2 inhibitors and GLP‐1RAs
In a comparative analysis, SGLT2 inhibitor therapy was associated with a significantly greater reduction in the risk of the kidney‐specific composite outcome and HHF compared with GLP‐1RAs in patients with DKD. The results of this network meta‐analysis concur with previous meta‐analyses that showed the superiority of SGLT2 inhibitors in reducing the risk of renal outcomes compared to GLP‐1RAs in patients with type 2 diabetes mellitus. 23 , 24 In a previous network meta‐analysis of CVOTs in patients with type 2 diabetes, use of SGLT2 inhibitors was associated with a 21% greater reduction in the risk of HHF and a 31% greater reduction in the risk of renal composite outcome compared with GLP‐1RAs. 23 Furthermore, in another network meta‐analysis in patients with type 2 diabetes and chronic kidney disease (CKD), GLP‐1RAs did not reduce the risk of either a CV (MACE) or a renal composite outcome. 24 Of note, no trial involving GLP‐1RAs has assessed renal outcomes as the primary outcome in patients with type 2 diabetes and CKD. Data pertaining to GLP‐1RAs and renal function in patients with CKD have come from CVOTs in which renal outcomes were assessed as secondary or exploratory outcomes. The FLOW trial evaluating the efficacy and safety of semaglutide by injection on primary renal endpoints is currently ongoing and the results are expected in 2024. In the present analysis, we analysed the treatment effects specifically in patients with type 2 diabetes mellitus and established CKD (DKD). The p‐scores and probability rankings confirmed the results from the network meta‐analysis. Based on the totality of available evidence, SGLT2 inhibitors had the highest p‐rank scores for all outcomes, indicating that they have a greater probability than the other treatments of reducing risks of both CV and renal events.
4.2. Risks associated with the use of DPP‐4 inhibitors
The results presented in this study strengthen the data from previous meta‐analyses which showed that DPP‐4 inhibitors did not significantly alter CV and renal risks compared to placebo. 23 , 25 A comparative cohort study that used data from clinical practice in 13 countries also concluded that SGLT2 inhibitors were associated with improved CV benefits compared to DPP‐4 inhibitors. 26 Overall, it appears that DPP‐4 inhibitors confer limited benefits and may not increase the risk of cardiorenal outcomes in patients with DKD.
4.3. Mechanism of action of SGLT2 inhibitors
Overall, this study proves the superiority of SGLT2 inhibitors in reducing the risk of CV and renal events, not only compared to placebo, but also compared to GLP‐1RAs and DPP‐4 inhibitors. Several possible mechanisms have been proposed to explain the benefits of SGLT2 inhibitors. SGLT2 inhibitors block SGLT2 cotransporters in the renal proximal tubule, resulting in increased glycosuria and a mild natriuretic and diuretic effect. 27 This leads to metabolic benefits including improved glycemic control, weight loss and blood pressure improvements that confer CV and renal protection. 28 It has been proposed that SGLT2 inhibitors improve ventricular loading conditions (due to natriuretic and diuretic effects), optimize cardiac metabolism, thus improving cardiac efficiency and output and inhibit the Na+/H+ exchanger (NHE 1) in the myocardium, thus providing favourable effects on the risk of heart failure. 29 , 30 The net result of these processes is a reduced risk of CV and renal outcomes with SGLT2 inhibition, as seen in major CVOTs 16 , 17 , 18 and renal outcome trials 19 , 20 and confirmed in this meta‐analysis. SGLT2 inhibitors also lower the reabsorption of sodium in the renal proximal tubule, thus restoring glomerular feedback and reducing intraglomerular pressure. 31 While this may result in an acute decrease in eGFR levels, eGFR stability is seen in the long run, leading to a nephroprotective effect. 31
4.4. Latest updates in guideline recommendations for the management of DKD
The latest Standards of Medical Care in Diabetes guidelines from the American Diabetes Association (ADA) recommend the use of SGLT2 inhibitors in patients with stage 3 CKD or higher and type 2 diabetes mellitus regardless of glycemic control, to slow the progression of CKD and to reduce CV risks. 32 The results of this meta‐analysis lend further support to these recommendations. The ADA suggests the use of GLP‐1RAs with proven CV benefit if SGLT2 inhibitors are not tolerated or contradicted. GLP‐1RAs are suggested “for CV risk reduction if such risk is a predominant problem, as they reduce risks of CV events and appear to possibly slow CKD progression.” The results of this meta‐analysis do not confirm the CV risk reduction benefits of GLP‐1RAs in patients with DKD; although GLP‐1RAs slightly reduced CV and renal risks compared to placebo, the decrease was not statistically significant for most outcomes.
4.5. Study limitations
To the best of our knowledge, this is the first analysis to evaluate SGLT2 inhibitors, GLP‐1RAs and DPP‐4 inhibitors specifically in patients with DKD. This meta‐analysis also includes the latest high‐quality data from recent CVOTs and renal outcome trials. While results from this network meta‐analysis are fairly comprehensive, the study does have some limitations. Firstly, as expected, there may be certain differences across RCTs in the patient population, stage of CKD (although data were specifically extracted for patients with eGFR <90 mL/min/1.73 m2), types of drugs within each drug class, drug dose and duration of treatment. These factors may cause heterogeneity and thus potentially impact results of the meta‐analysis. Secondly, as we used data from subgroup analyses of CVOTs, there is a concern that the patients with DKD may not have been fully randomized. Thirdly, the criteria used to define DKD were different in each study included in the analysis.
5. CONCLUSIONS
SGLT2 inhibitors show a clear advantage in reducing the risks of both CV and renal events in patients with DKD, compared to GLP‐1RAs and DPP‐4 inhibitors. GLP‐1RAs also show CV and renal outcome benefits, compared to placebo, but the benefit was not better than SGLT2 inhibitors or DPP‐4 inhibitors for most outcomes. In contrast, DPP‐4 inhibitors did not improve either CV or renal outcomes. Based on the results of this meta‐analysis, we recommend that SGLT2 inhibitors should be considered the treatment of choice in patients with type 2 diabetes mellitus and kidney disease.
AUTHOR CONTRIBUTIONS
All authors contributed to the meta‐analysis design, data analysis and to the drafting, review and final approval of the manuscript.
CONFLICT OF INTEREST
All authors have no conflict of interest to declare.
PEER REVIEW
The peer review history for this article is available at https://publons.com/publon/10.1111/dom.14702.
Supporting information
Appendix S1 Supporting information.
ACKNOWLEDGMENTS
Editorial assistance was provided by Syed Abdul Haseeb (MS, CMPP) and Akshaya Srinivasan (PhD) of MediTech Media, Asia Pacific.
Cao H, Liu T, Wang L, Ji Q. Comparative efficacy of novel antidiabetic drugs on cardiovascular and renal outcomes in patients with diabetic kidney disease: A systematic review and network meta‐analysis. Diabetes Obes Metab. 2022;24(8):1448‐1457. doi: 10.1111/dom.14702
Hongwei Cao and Tao Liu contributed equally to this work.
Funding information Funding was provided by AstraZeneca China in accordance with Good Publication Practice (GPP3) guidelines.
DATA AVAILABILITY STATEMENT
Data will be made available upon reasonable request.
REFERENCES
- 1. Saeedi P, Petersohn I, Salpea P, et al. Global and regional diabetes prevalence estimates for 2019 and projections for 2030 and 2045: results from the International Diabetes Federation Diabetes Atlas, 9th edition. Diabetes Res Clin Pract. 2019;157:107843. [DOI] [PubMed] [Google Scholar]
- 2. Afkarian M, Zelnick LR, Hall YN, et al. Clinical manifestations of kidney disease among US adults with diabetes, 1988‐2014. JAMA. 2016;316(6):602‐610. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3. Jitraknatee J, Ruengorn C, Nochaiwong S. Prevalence and risk factors of chronic kidney disease among type 2 diabetes patients: a cross‐sectional study in primary care practice. Sci Rep. 2020;10(1):6205. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4. Duan JY, Duan GC, Wang CJ, et al. Prevalence and risk factors of chronic kidney disease and diabetic kidney disease in a central Chinese urban population: a cross‐sectional survey. BMC Nephrol. 2020;21(1):115. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5. Hussain S, Habib A, Najmi AK. Limited knowledge of chronic kidney disease among type 2 diabetes mellitus patients in India. Int J Environ Res Public Health. 2019;16(8):1443. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6. Wu B, Bell K, Stanford A, et al. Understanding CKD among patients with T2DM: prevalence, temporal trends, and treatment patterns‐NHANES 2007‐2012. BMJ Open Diabetes Res Care. 2016;4(1):e000154. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7. Kung K, Chow KM, Hui EM, et al. Prevalence of complications among Chinese diabetic patients in urban primary care clinics: a cross‐sectional study. BMC Fam Pract. 2014;15:8. [DOI] [PMC free article] [PubMed] [Google Scholar] [Retracted]
- 8. Assogba GF, Couchoud C, Roudier C, et al. Prevalence, screening and treatment of chronic kidney disease in people with type 2 diabetes in France: the ENTRED surveys (2001 and 2007). Diabetes Metab. 2012;38(6):558‐566. [DOI] [PubMed] [Google Scholar]
- 9. Retnakaran R, Cull CA, Thorne KI, Adler AI, Holman RR, Group US . Risk factors for renal dysfunction in type 2 diabetes: U.K. Prospective Diabetes Study 74. Diabetes. 2006;55(6):1832‐1839. [DOI] [PubMed] [Google Scholar]
- 10. Thomas MC, Brownlee M, Susztak K, et al. Diabetic kidney disease. Nat Rev Dis Primers. 2015;1(1):15018. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11. Afkarian M, Sachs MC, Kestenbaum B, et al. Kidney disease and increased mortality risk in type 2 diabetes. J Am Soc Nephrol. 2013;24(2):302. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12. Ruospo M, Saglimbene VM, Palmer SC, et al. Glucose targets for preventing diabetic kidney disease and its progression. Cochrane Database Syst Rev. 2017;6:CD010137. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13. Brenner BM, Cooper ME, de Zeeuw D, et al. Effects of losartan on renal and cardiovascular outcomes in patients with type 2 diabetes and nephropathy. N Engl J Med. 2001;345(12):861‐869. [DOI] [PubMed] [Google Scholar]
- 14. Lewis EJ, Hunsicker LG, Clarke WR, et al. Renoprotective effect of the angiotensin‐receptor antagonist irbesartan in patients with nephropathy due to type 2 diabetes. N Engl J Med. 2001;345(12):851‐860. [DOI] [PubMed] [Google Scholar]
- 15. Anders H‐J, Huber TB, Isermann B, Schiffer M. CKD in diabetes: diabetic kidney disease versus nondiabetic kidney disease. Nat Rev Nephrol. 2018;14(6):361‐377. [DOI] [PubMed] [Google Scholar]
- 16. Neal B, Perkovic V, Mahaffey KW, et al. Canagliflozin and cardiovascular and renal events in type 2 diabetes. N Engl J Med. 2017;377(7):644‐657. [DOI] [PubMed] [Google Scholar]
- 17. Wiviott SD, Raz I, Bonaca MP, et al. Dapagliflozin and cardiovascular outcomes in type 2 diabetes. N Engl J Med. 2019;380(4):347‐357. [DOI] [PubMed] [Google Scholar]
- 18. Zinman B, Wanner C, Lachin JM, et al. Empagliflozin, cardiovascular outcomes, and mortality in type 2 diabetes. N Engl J Med. 2015;373(22):2117‐2128. [DOI] [PubMed] [Google Scholar]
- 19. Mann JFE, Ørsted DD, Brown‐Frandsen K, et al. Liraglutide and renal outcomes in type 2 diabetes. N Engl J Med. 2017;377(9):839‐848. [DOI] [PubMed] [Google Scholar]
- 20. Marso SP, Bain SC, Consoli A, et al. Semaglutide and cardiovascular outcomes in patients with type 2 diabetes. N Engl J Med. 2016;375(19):1834‐1844. [DOI] [PubMed] [Google Scholar]
- 21. Heerspink HJL, Stefánsson BV, Correa‐Rotter R, et al. Dapagliflozin in patients with chronic kidney disease. N Engl J Med. 2020;383(15):1436‐1446. [DOI] [PubMed] [Google Scholar]
- 22. Perkovic V, Jardine MJ, Neal B, et al. Canagliflozin and renal outcomes in type 2 diabetes and nephropathy. N Engl J Med. 2019;380(24):2295‐2306. [DOI] [PubMed] [Google Scholar]
- 23. Fei Y, Tsoi M‐F, Cheung BMY. Cardiovascular outcomes in trials of new antidiabetic drug classes: a network meta‐analysis. Cardiovasc Diabetol. 2019;18(1):112. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24. Yamada T, Wakabayashi M, Bhalla A, et al. Cardiovascular and renal outcomes with SGLT‐2 inhibitors versus GLP‐1 receptor agonists in patients with type 2 diabetes mellitus and chronic kidney disease: a systematic review and network meta‐analysis. Cardiovasc Diabetol. 2021;20(1):14. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25. Bae JH, Park E‐G, Kim S, Kim SG, Hahn S, Kim NH. Comparative renal effects of dipeptidyl peptidase‐4 inhibitors and sodium‐glucose cotransporter 2 inhibitors on individual outcomes in patients with type 2 diabetes: a systematic review and network meta‐analysis. Endocrinol Metab (Seoul). 2021;36(2):388‐400. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26. Kohsaka S, Lam CSP, Kim DJ, et al. Risk of cardiovascular events and death associated with initiation of SGLT2 inhibitors compared with DPP‐4 inhibitors: an analysis from the CVD‐REAL 2 multinational cohort study. Lancet Diabetes Endocrinol. 2020;8(7):606‐615. [DOI] [PubMed] [Google Scholar]
- 27. Imprialos KP, Sarafidis PA, Karagiannis AI. Sodium‐glucose cotransporter‐2 inhibitors and blood pressure decrease: a valuable effect of a novel antidiabetic class? J Hypertens. 2015;33(11):2185‐2197. [DOI] [PubMed] [Google Scholar]
- 28. Storgaard H, Gluud LL, Bennett C, et al. Benefits and harms of sodium‐glucose co‐transporter 2 inhibitors in patients with type 2 diabetes: a systematic review and meta‐analysis. PLoS One. 2016;11(11):e0166125. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29. Sattar N, McLaren J, Kristensen SL, Preiss D, McMurray JJ. SGLT2 inhibition and cardiovascular events: why did EMPA‐REG outcomes surprise and what were the likely mechanisms? Diabetologia. 2016;59(7):1333‐1339. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30. Packer M, Anker SD, Butler J, Filippatos G, Zannad F. Effects of sodium‐glucose cotransporter 2 inhibitors for the treatment of patients with heart failure: proposal of a novel mechanism of action. JAMA Cardiol. 2017;2(9):1025‐1029. [DOI] [PubMed] [Google Scholar]
- 31. Heerspink HJ, Desai M, Jardine M, Balis D, Meininger G, Perkovic V. Canagliflozin slows progression of renal function decline independently of glycemic effects. J Am Soc Nephrol. 2017;28(1):368‐375. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32. American Diabetes Association Professional Practice Committee . 11. Chronic kidney disease and risk management: standards of medical Care in Diabetes—2022. Diabetes Care. 2021;45(supplement_1):S175‐S184. [DOI] [PubMed] [Google Scholar]
- 33. Udell JA, Bhatt DL, Braunwald E, et al. Saxagliptin and cardiovascular outcomes in patients with type 2 diabetes and moderate or severe renal impairment: observations from the SAVOR‐TIMI 53 trial. Diabetes Care. 2015;38(4):696‐705. [DOI] [PubMed] [Google Scholar]
- 34. Cornel JH, Bakris GL, Stevens SR, et al. Effect of sitagliptin on kidney function and respective cardiovascular outcomes in type 2 diabetes: outcomes from TECOS. Diabetes Care. 2016;39(12):2304‐2310. [DOI] [PubMed] [Google Scholar]
- 35. McGuire DK, Alexander JH, Johansen OE, et al. Linagliptin effects on heart failure and related outcomes in individuals with type 2 diabetes mellitus at high cardiovascular and renal risk in CARMELINA. Circulation. 2019;139(3):351‐361. [DOI] [PubMed] [Google Scholar]
- 36. Rosenstock J, Perkovic V, Johansen OE, et al. Effect of linagliptin vs placebo on major cardiovascular events in adults with type 2 diabetes and high cardiovascular and renal risk: the CARMELINA randomized clinical trial. JAMA. 2019;321(1):69‐79. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37. Ferreira JP, Mehta C, Sharma A, Nissen SE, Rossignol P, Zannad F. Alogliptin after acute coronary syndrome in patients with type 2 diabetes: a renal function stratified analysis of the EXAMINE trial. BMC Med. 2020;18(1):165. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38. Mann JFE, Fonseca V, Mosenzon O, et al. Effects of liraglutide versus placebo on cardiovascular events in patients with type 2 diabetes mellitus and chronic kidney disease. Circulation. 2018;138(25):2908‐2918. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39. Husain M, Birkenfeld AL, Donsmark M, et al. Oral semaglutide and cardiovascular outcomes in patients with type 2 diabetes. N Engl J Med. 2019;381(9):841‐851. [DOI] [PubMed] [Google Scholar]
- 40. Gerstein HC, Colhoun HM, Dagenais GR, et al. Dulaglutide and renal outcomes in type 2 diabetes: an exploratory analysis of the REWIND randomised, placebo‐controlled trial. Lancet. 2019;394(10193):131‐138. [DOI] [PubMed] [Google Scholar]
- 41. Bethel MA, Mentz RJ, Merrill P, et al. Microvascular and cardiovascular outcomes according to renal function in patients treated with once‐weekly exenatide: insights from the EXSCEL trial. Diabetes Care. 2020;43(2):446‐452. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42. Wanner C, Inzucchi SE, Lachin JM, et al. Empagliflozin and progression of kidney disease in type 2 diabetes. N Engl J Med. 2016;375(4):323‐334. [DOI] [PubMed] [Google Scholar]
- 43. Wanner C, Lachin JM, Inzucchi SE, et al. Empagliflozin and clinical outcomes in patients with type 2 diabetes mellitus, established cardiovascular disease, and chronic kidney disease. Circulation. 2018;137(2):119‐129. [DOI] [PubMed] [Google Scholar]
- 44. Neuen BL, Ohkuma T, Neal B, et al. Cardiovascular and renal outcomes with canagliflozin according to baseline kidney function. Circulation. 2018;138(15):1537‐1550. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45. Cannon CP, Pratley R, Dagogo‐Jack S, et al. Cardiovascular outcomes with ertugliflozin in type 2 diabetes. N Engl J Med. 2020;383(15):1425‐1435. [DOI] [PubMed] [Google Scholar]
- 46. Cherney DZI, Charbonnel B, Cosentino F, et al. Effects of ertugliflozin on kidney composite outcomes, renal function and albuminuria in patients with type 2 diabetes mellitus: an analysis from the randomised VERTIS CV trial. Diabetologia. 2021;64(6):1256‐1267. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47. Bhatt DL, Szarek M, Pitt B, et al. Sotagliflozin in patients with diabetes and chronic kidney disease. N Engl J Med. 2021;384(2):129‐139. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Appendix S1 Supporting information.
Data Availability Statement
Data will be made available upon reasonable request.


