Abstract
Growth and functioning of Sphagnum mosses are closely linked to water level and chemistry. Sphagnum mosses occur in wet, generally acidic conditions, and when buffered, alkaline water is known to negatively impact Sphagnum. The effects of time, dose and species‐specific responses of buffered, alkaline water on Sphagnum are largely unknown.
We investigated the effects of bicarbonate and calcium on the survival, growth and physiological functioning of seven Sphagnum species occurring in contrasting environments, from raised bogs to (rich) fens. Mosses were submerged in different concentrations of bicarbonate and calcium solutions for 10 weeks under climate‐controlled circumstances.
After 2 weeks, all species exposed to the high bicarbonate treatment (2.0 mM) showed severe potassium leakage and swift discoloration. In contrast, species showed differential responses to the intermediate bicarbonate treatment (0.8 mM), some with a later onset of potassium leakage. S. squarrosum, S. teres & S. contortum generally persisted the longest, with all species dying after 6 to 10 weeks. Calcium alone, in contrast, negatively affected S. squarrosum, S. teres & S. contortum, causing discoloration and potassium leakage.
Our study shows enrichment with bicarbonate, but not calcium, is detrimental for most Sphagnum species tested. A mechanistic model was developed that is consistent with dose and duration dependence and the species specificity. Future conservation and restoration measures for Sphagnum‐dominated habitats and Sphagnum farming (cultivation, production and harvest of Sphagnum moss biomass) should limit flooding with bicarbonate‐rich waters while investigating new management options, like acidifying surface waters to lower bicarbonate levels.
Keywords: Alkalinity, Calcium tolerance, Potassium leakage, Peat moss, Peatland restoration, Sphagnum farming
Bicarbonate rather than calcium proves to be toxic (dose dependent) for seven Sphagnum species from ecological contrasting environments.

Introduction
Sphagnum mosses generally thrive in acidic, near‐continuous wet conditions that actively stimulate peat formation (Clymo 1964, 1973; Jassey et al. 2011). Successive periods of drought result in the loss of Sphagnum spp. and their functioning as ecosystem engineers (Jones et al. 1994; Potter et al. 1996; Schipperges & Rydin 1998). More knowledge about what surface water composition is suitable for Sphagnum growth is essential for peatland restoration and Sphagnum farming. Minimizing desiccation events during episodes of low precipitation may require the inlet of external surface water, especially for initial acrotelm development during restoration measures and Sphagnum farming, to sustainably grow Sphagnum on deteriorated peat (Gaudig et al. 2018; Vroom et al. 2020). Surface water composition is, however, highly variable and when inlet water is minerotrophic, the effects on Sphagnum can be detrimental (Smolders et al. 2003; Harpenslager et al. 2015; Koks et al. 2019).
Sphagnum death resulting from minerotrophic surface water is often attributed to calcium toxicity coupled with a high pH, overlooking the effect of introduced or in situ formed bicarbonate () (Clymo 1973; Vicherová et al. 2015, 2017; Plesková et al. 2016). Peatlands and wetlands characterized by a high pH (e.g. pH > 6.5) also inevitably have high concentrations, as a result of conversion from dissolved carbon dioxide (CO2) to at alkaline pH. Although some studies specifically investigated the detrimental effects of on Sphagnum, the underlying mechanisms remain elusive (Paul 1906; Skene 1915).
Sphagnum species occur along a mineral gradient differing in hydrochemical properties (Bates 1982; Rydin et al. 2006; Hájková & Hájek 2007). Rich fen species, such as S. squarrosum, S. teres & S. contortum, are frequently subjected to more buffered water (i.e. higher ) in concert with higher calcium (Ca2+) and/or magnesium (Mg2+) concentrations (Turekian & Wedepohl 1961; Vitt & Slack 1984; Vicherová & Hájek 2013; Plesková et al. 2016). Other species, such as S. fallax and S. palustre, are generally found in poor fens and ombrotrophic peatlands (Daniels & Eddy 1985; Kooijman & Kanne 1993; Limpens & Berendse 2003). In contrast, S. magellanicum and S. papillosum grow in acidic mires and ombrotrophic bogs generally lacking any systemic influence of buffered waters (Smolders et al. 2003; Hájková & Hájek 2004; Hájek et al. 2006).
Compared to ombrotrophic species, minerotrophic Sphagnum species might experience less physiological stress when subjected to buffered water (Feldmeyer‐Christe et al. 2001). Physiological stress in Sphagnum species may impair cell membrane integrity, resulting in increased electrolytes and particularly potassium (K+) loss or leakage (hereafter referred to as leakage) (Potter et al. 1996; Demidchik et al. 2014). Although the effects of combined Ca2+ and concentrations have been previously tested (Harpenslager et al. 2015; Koks et al. 2019), the differential effects of and Ca2+ have not been tested for a range of Sphagnum species. To investigate these, the following questions and hypotheses are addressed:
Is exposure to high concentrations toxic for Sphagnum? Increased concentrations were expected to eventually cause physiological damage, resulting in chlorosis and electrolyte (e.g. K+) leakage in all seven Sphagnum species.
Are the effects of species‐ and niche‐specific, and dependent on exposure time and concentration? Bog species (S. magellanicum and S. papillosum) and poor fen species (S. fallax and S. palustre) were expected to be readily damaged by high concentrations, but could have a reduced and delayed response at lower concentrations. Rich fen species (S. squarrosum, S. teres & S. contortum) were expected to survive longer at both concentrations.
Is a high Ca2+ concentration without pH increase toxic for Sphagnum? Ca2+ was not expected to affect any of the Sphagnum species.
Material and Methods
Sphagnum sample collection
In January 2018, seven different Sphagnum species were collected in the Netherlands and Germany (Table 1). All species were separately subjected to experimental treatments, except for the combination of S. teres & S. contortum. A total of 15 patches per species (group) (n = 15) were collected using a polypropylene (PP) tool (3‐cm tall; ⌀ 7.5 cm), taking the top 3 cm to minimize underlying peat influence. Before starting the experiment, 20 random Sphagnum strands per patch were weighed and each strand was marked with a small cable tie under the capitula to record length increment. The strands were put into smaller polyethylene terephthalate (PET) conical containers (11‐cm tall; ⌀ 7.5 cm) with ten drainage holes (⌀ 0.5 cm) within the larger PP containers (15‐cm tall; ⌀ 7.5 cm) (Fig. 1).
Table 1.
Different selected Sphagnum species with their general microtopography and respective donor sites.
| Sphagnum species | Microtopography | Coordinates | Region; Country |
|---|---|---|---|
| S. magellanicum | Bog | 52°45'51.7"N 6°59'02.5"E | Oosterbos; the Netherlands |
| S. papillosum | Bog | 53°15'48.0"N 8°16'03.0"E | Hankhauser Moor; Germany |
| S. fallax | Poor fen | 53°15'48.0"N 8°16'03.0"E | Hankhauser Moor; Germany |
| S. palustre | Poor fen | 52°40'31.0"N 6°07'04.0"E | Weerribben‐Wieden; the Netherlands |
| S. squarrosum | Mixed fen | 52°26'43.1"N 4°56'40.3"E | Ilperveld; the Netherlands |
| S. teres | Rich fen | 52°40'53.4"N 6°07'10.6"E | Weerribben‐Wieden; the Netherlands |
| S. contortum | Rich fen | 52°40'53.4"N 6°07'10.6"E | Weerribben‐Wieden; the Netherlands |
Fig. 1.
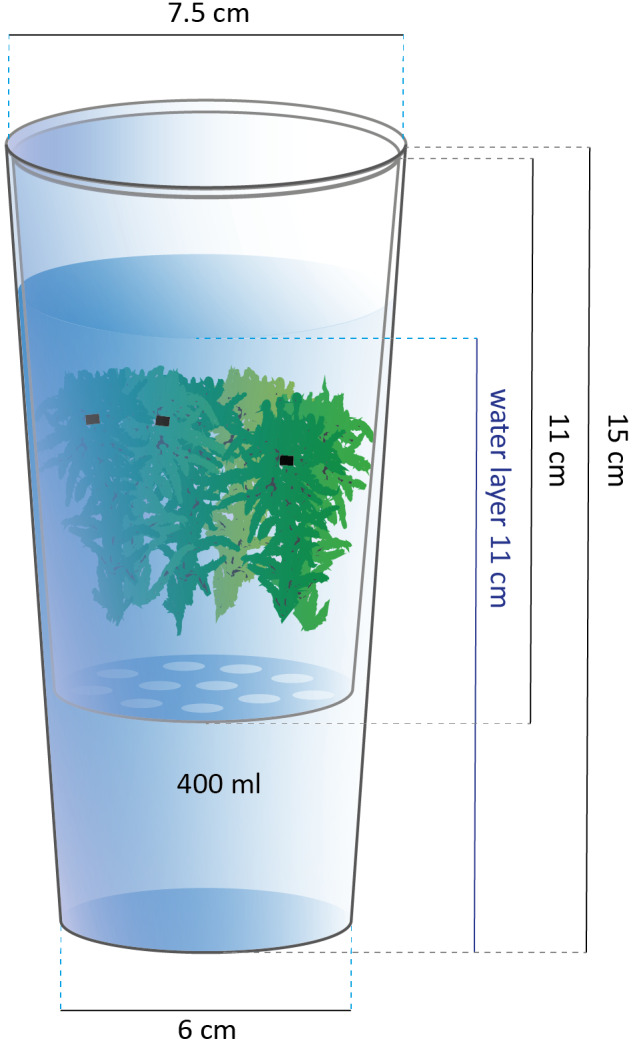
Container holding the experimental solution with the inner container holding marked Sphagnum mosses (black markers under the capitula were used to record length increment), lengths shown in cm, volume shown in ml.
Experimental conditions
The experiment was carried out in a climate‐controlled room kept at 21 °C, 55% relative humidity. All 90 containers were placed in a water bath continuously cooled to a temperature of 16 °C, resulting in 17 °C at vegetation level. Light regime consisted of a 16‐h/8‐h light/dark photoperiod providing a total of 400 µmol m−2 s−1 PAR (photosynthetically active radiation) at vegetation level (Philips Greenpower LED Toplighting Module, DR/W/FR_2MB 400V, Poland). Throughout the whole experiment, mosses were completely submerged. Before the start of the experiment, all containers were subjected to 1 week of acclimatisation in artificial rainwater solution mimicking Dutch rainwater conditions (Van Den Elzen et al. 2017).
Experimental setup
The experiment started on 27 February 2018 and lasted for 10 weeks (69 days). It included three experimental treatments and one control being rainwater solution (n = 4) in a fully randomized design (Table S1).
Two treatments had added sodium bicarbonate (NaHCO3): 0.8 mM and 2.0 mM (n = 4), and one treatment had 2.0 mM calcium chloride (CaCl2) (n = 3) (Table 2).
Table 2.
pH and dominant ions of the respective treatment solution added weekly (n = 10) (displayed are mean ± SE).
| Treatment | Cation | mmol l−1 (mM) | Anion | mmol l−1 (mM) | pH | |
|---|---|---|---|---|---|---|
| Control | / | / | / | / | 4.9 ± 0.1 | |
| 0.8 mM | Na+ | 0.8 |
|
0.8 | 6.9 ± 0.1 | |
| 2.0 mM | Na+ | 2.0 |
|
2.0 | 7.4 ± 0.1 | |
| 2.0 mM CaCl2 | Ca2+ | 2.0 | Cl− | 4.0 | 5.0 ± 0 |
During the first week, all containers received half the treatment concentration, mimicking a remnant rainwater cache mixed with external water input. Initially all containers also received a one‐time 20 µM ammonium nitrate (NH4NO3) addition. All mosses were continuously submerged in treatment water to minimize osmotic stress, gaseous discrepancies and crystallisation of salts. All containers were drained weekly and received 400 ml of freshly mixed media, to assure submergence and minimize evaporation effects.
At the end of the experiment, all containers were drained and refilled with demineralized water. This was done twice, with a 3‐h interval, removing any unbound cations from the cell walls. Mosses were stored overnight in their experimental containers to reduce excess water and making water content and fresh weight more comparable among samples.
Container water analysis
The pH in the containers was measured once at the start of the experiment then twice weekly using a portable pH meter (PHM80; Radio meter, Copenhagen, Denmark) (Appendices S1, S2). Airtight water samples were taken weekly using a 1‐ml syringe for total inorganic carbon (TIC) analysis using an infrared carbon analyser (IRGA; ABB Analytical, Frankfurt, Germany), to calculate CO2 and concentrations based on pH (Appendices S1, S2).
Each week a 10‐ml sample per container was conserved in 0.1 ml nitric acid (65%, HNO3) for further analysis. For the elemental composition (aluminium (Al), calcium (Ca), iron (Fe), potassium (K), magnesium (Mg), manganese (Mn), sodium (Na), phosphorus (P), sulphur (S), silicon (Si) and zinc (Zn)) inductively coupled plasma emission spectrometry (ICP‐EOS) was used (ICP‐OES; iCAP 6000; Thermo‐Fischer Scientific, Bremen, Germany). This was done using a streaked optical pyrometer plasma with a power of 1150 W and a nebulizer flow of 0.6 l min−1 (sea spray nebulizer with cyclone chamber). Nitrate (), ammonium () and chloride (Cl−) concentrations of treatment solutions were analysed using the Auto Analyzer system III (SEAL Analytical, Norderstedt, Germany) (Table S1).
Sphagnum moss analysis
Weekly discoloration was measured, rating each container by visual inspection: 0 was used to indicate healthy growing mosses (no discoloration), −1, −2, −3 were mosses progressively bleaching and −4 for white bleached mosses. At −3, such a strong discoloration occurred we inferred mosses would not quickly, or ever, recover. Length increment was determined by individually measuring the marked strands, taking initial capitula size into account and averaging them per container before statistical analysis. Hereafter the mosses were weighed, oven‐dried at 70 °C until samples reached constant weight before determining dry weight. The conversion factor determined from the change in fresh/dry weight was used calculating the initial dry weight. This was then applied in calculating the relative dry weight increase in dry weight/initial dry weight (g g−1). K+ leakage was accounted for dried weight of the mosses, enabling species‐level comparisons.
Elemental plant content of Al, Ca, Fe, K, Mg, Mn, Na, P, S, Si and Zn (Table S3) were obtained by homogenizing dried plant material for 30 s per container (18,000 RPM; 0.2 mm screen) (Fritsch, Idar‐Oberstein, Germany). The homogenized material was further processed by digesting 100 mg plant matter in 4 ml nitric acid (65%) and 1 ml hydrogen peroxide (H2O2) (35%) in teflon containers. These were prepared using an Ethos D microwave (Milestone, Sorisole Lombardy, Italy). The digestate was diluted in 50 ml ultrapure mili‐Q water (Milipore, Burlington, MA, USA). Samples were analysed using ICP‐OES (see above).
Statistical analysis
The results were statistically tested using R statistical language version 4.1.2 (R Core Team 2013). Treatments and Sphagnum species were independent variables, with treatments having four and species having six categorical groups. The dependent variables at a continuous scale which consisted of Sphagnum normalized biomass development and length increase were species dependently analysed using a one‐way ANOVA (both variables having one outlier excluded, determined from boxplots being problematic for further analysis), while Na content was independently analysed using a two‐way ANOVA. Residual homogeneity and equality of variance were checked by inspecting the diagnostic plots in R (Kozak & Piepho 2017). Na content was Gaussianized using LambertW package to approach normal distribution (Goerg 2015).
The Ca, K and Mg content could not be analysed in raw or transformed format using a two‐way ANOVA because the data did not meet the requirements and were therefore analysed using a generalized linear model, fitting a gamma distribution with a Satterthwaite correction for unbalanced design. Weekly discoloration, being an ordinal scale, was analysed using an ANOVA on ranks (Boos & Brownie 1995; Carifio & Perla 2008). Significant differences were further investigated using Sidak adjustments for confidence levels and Tukey’s post‐hoc method with the Emmeans and multcomp packages (Hothorn et al. 2020; Lenth et al. 2020).
Weekly K+ leakage was additively accumulated for weekly interval analysis, while for pH the absolute values were used. Using a repeated mixed measures ANOVA from the ez package, K+ leakage was analysed with experimental weeks as within‐subject variable (Lawrence 2016). Testing for sphericity, Mauchly’s test was used with Greenhouse‐Geisser ε correction, where needed.
Significant differences resulting from the repeated mixed measures ANOVA were further investigated using pairwise comparison of least square means with Bonferroni‐adjusted significance levels for multiple comparison using the lsmeans package (Lenth 2016). Correlative analysis was done using Spearman’s rho. Error bars in line charts indicate standard error of the mean (SE), while bar charts show confidence intervals (CI95). Minimum significance was assumed at P < 0.05.
Results
Relative biomass development and length increment
Exposing Sphagnum mosses to buffered solutions reduced biomass development and length increment; however, overall treatment–species comparison was hindered with species having variable growth rates. In the 2.0 treatment, S. magellanicum (F(3,11) = 7.03, P < 0.01), S. fallax (F(3,11) = 11.98, P < 0.001) and S. squarrosum (F(3,11) = 5.23, P < 0.05) had 74–76% lower biomass development compared to the control treatment, whereas in the 0.8 treatment only S. fallax had lower biomass (P < 0.01) (Fig. 2A). The CaCl2 addition did not significantly affect biomass development.
Fig. 2.

Sphagnum species‐specific relative biomass (dry weight) increase (g g−1) (A) and length increment (cm) (B). Differences across species and interaction effects between species and treatments were not tested as relative biomass and length increment were expected to be largely species‐specific. Significant differences between treatments within a single species are indicated by different letters (displayed are mean ± CI95). White bars represent the control groups, light grey bars the 2.0 CaCl2 treatment, darker grey bars the 0.8 treatment, and darkest grey bars the 2.0 treatment.
Length increment was substantially reduced in the 2.0 treatment for S. magellanicum (F(3,11) = 6.39, P < 0.01), S. fallax (F(3,11) = 40.26, P < 0.05), S. palustre (F(3,11) = 61.26, P < 0.001), S. squarrosum (F(3,11) = 6.64, P < 0.01) and S. teres & S. contortum (F(3,11) = 7.09, P < 0.01). The 0.8 treatment negatively affected S. magellanicum (P < 0.05), S. fallax (P < 0.001), S. palustre (P < 0.001) and S. teres & S. contortum (P < 0.05). The 2.0 mM CaCl2 treatment only reduced length increment for S. fallax (P < 0.05) (Fig. 2B).
Discoloration
Increased concentrations impacted all Sphagnum species, resulting in severe discoloration that differed between species, treatment and week (from green to white/pale) (F(135,660) = 1.45, P < 0.001) (Table S4). The 2.0 CaCl2 treatment only negatively affected S. squarrosum, S. teres & S. contortum, causing discoloration (P < 0.001) (Table S4).
Throughout time, the discoloration (i.e. chlorosis) varied between species and treatments (Fig. 3). S. fallax was already pale at the beginning of the experiment and the initial addition of NH4NO3 in all containers resulted in S. fallax becoming greener in both the control and 2.0 CaCl2 treatment. In the 2.0 treatment, S. fallax reached the median discoloration level of ‘−3’ after 2 weeks followed by S. magellanicum, S. papillosum and S. palustre after 5 weeks. S. teres & S. contortum lasted 6 weeks, and S. squarrosum took 9 weeks to reach median level ‘−3’. In the 0.8 treatment, it took S. papillosum 6 weeks before reaching the median ‘−3’ discoloration level. S. magellanicum, S. fallax and S. teres & S. contortum took 7 weeks, S. palustre 8 weeks and S. squarrosum 9 weeks. Both S. squarrosum and S. teres & S. contortum also showed discoloration in the 2.0 CaCl2 treatment but had a later onset than in the treatments.
Fig. 3.

Discoloration during the experiment (0 are ‘healthy’ green mosses, while −4 indicate white and dead mosses) of all Sphagnum species and different treatments (displayed is the median). Blue lines represent control groups, green lines show the 2.0 CaCl2 treatment, red lines the 0.8 treatment and black lines the 2.0 treatment.
Potassium (K+) leakage and adsorption/uptake
We found evidence for K+ leakage upon exposure of Sphagnum mosses to elevated levels. K+ leakage varied between species and both treatments (F(15,66) = 8.35, P < 0.01). The extent to which species leaked varied, and S. fallax leaked in both the 0.8 and 2.0 treatments significantly more compared to S. magellanicum and S. papillosum (P < 0.05).
In contrast, in the control K+ was taken up from the treatment solution, which contained K+ ions. To a smaller extent, we observed similar K+ uptake in the 2.0 CaCl2 treatment in S. magellanicum, S. papillosum and S. palustre throughout the experiment (Fig. 4). However, S. fallax, S. squarrosum and S. teres & S. contortum eventually leaked more K+ in the 2.0 CaCl2 treatment compared to the control (P < 0.001). Nonetheless, at the end of the experiment, all species retained more K in the 2.0 CaCl2 compared to the 2.0 and 0.8 treatments (P < 0.05).
Fig. 4.

Potassium (K+) dynamics (µmol g dry weight (dw)−1) (increasing values indicate uptake, decreasing values indicate loss or leakage of K+) throughout the experiment for all Sphagnum species and experimental treatments (displayed are mean ± SE). Blue lines represent control groups, green lines the 2.0 CaCl2 treatment, red lines the 0.8 treatment and black lines the 2.0 treatment. Horizontal lines indicate thresholds when the K+ leakage is strongly reduced.
Elemental tissue content
The various treatments had differential effects on the element concentrations found in the mosses. Control values for K were species‐specific; all Sphagnum species except S. magellanicum had significantly higher K content compared to S. papillosum (F(15,66) = 9.24, P < 0.001) (Fig. 5A). K content (in cells and bound to moss tissue) differed among treatments and species (Fig. 5A). Both 0.8 and 2.0 treatments resulted in 55–88% lower K content compared to control and 2.0 CaCl2 treatment (P < 0.001) (Fig. 5A).
Fig. 5.

Tissue content (in cell and bound to moss tissue) K (A), Na (B), Ca (C) and Mg (D) (µmol g dry weight (dw)−1). Significant differences between treatments are indicated by different letters (displayed are mean ± CI95). Initial starting content per species are shown in Table S3. White bars represent control groups, light grey bars the 2.0 CaCl2 treatment, darker grey bars the 0.8 treatment and the darkest grey bars the 2.0 treatment.
Sodium (Na) was higher in all Sphagnum species in both 0.8 and 2.0 treatments compared to both control and 2.0 CaCl2 treatment (F(15,66) = 14.55, P < 0.001) (Fig. 5B; Table S3). S. teres & S. contortum had far less Na than other species, while concentrations were similar at the start of the experiment (P < 0.001; Table S3).
All Sphagnum species had 84–281% more Ca in the 2.0 CaCl2 treatment compared to the control (F(15,66) = 3.042, P < 0.001) (Fig. 5C). S. squarrosum and S. teres & S. contortum had significant higher Ca content compared to other species in the control (P < 0.05). While S. teres & S. contortum retained more Ca in both 0.8 and 2.0 treatments compared to most other species they did not differ from the control and 2.0 CaCl2 treatment. The initial Ca content (t = 0) was also higher for S. teres & S. contortum (Table S3).
The Mg content was lower in the 2.0 CaCl2 treatment compared to all other treatments for all Sphagnum species (F(15,66) = 2.002, P < 0.001) (Fig. 5D).
Temporal patterns
Both discoloration and K+ leakage revealed temporal patterns for species and treatment (F(135,594) = 6.80, P < 0.01). S. teres & S. contortum leaked more K+ after 1 week in the 2.0 treatment compared to the control (P < 0.01); S. palustre and S. squarrosum did so after 2 weeks and other species after 3 weeks in the 2.0 treatment (P < 0.001). Cumulative K+ leakage rates (Fig. 4) flattened in both treatments at similar cumulative K+ thresholds, being species‐specific and dependent on their starting K+ content (Table S3). For most species in the 2.0 treatment, K+ leakage rates levelled off after 4 to 5 weeks. In the 0.8 treatment, a similar pattern was observed, yet flattening after 9 to 10 weeks. S. teres & S. contortum started flattening after 7 weeks in the 0.8 treatment.
Throughout time, cumulative K+ loads showed an increasing correlation with the measured discoloration rates (Fig. 6). After 2 weeks, initial patterns were found (ρ = 0.48, P < 0.001), becoming stronger after 5 weeks (ρ = 0.76, P < 0.001). This differentiation in K+ leakage as discoloration of the mosses continued for both 0.8 and 2.0 treatments, with similar patterns after 10 weeks (ρ = 0.76, P < 0.001) (Fig. 6).
Fig. 6.

The correlative changes in discoloration and cumulative K+ leakage (µmol g dry weight (dw)−1) between different experimental treatments and species after 1, 2, 5 and 10 weeks, with added jitter for visibility (Displayed is Spearman's rho). Blue symbols represent control groups, green is the 2.0 CaCl2 treatment, red is the 0.8 treatment and black the 2.0 treatment. Sphagnum species are presented with different symbols (square = S. magellanicum; circle = S. papillosum; triangle = S. fallax; cross = S. palustre; x = S. squarrosum; asterisk = S. teres & S. contortum).
The pH in the containers showed a different temporal response per treatment. In both treatments, the pH sharply increased after adding the treatment solutions (Fig. 7). These solutions had a higher pH (Table 2), nonetheless the pH continued to rise even though the treatment solution was renewed weekly. The pH in the control treatment decreased over time and the overall pH in the 2.0 CaCl2 treatment rose over time. However, within each week the pH in the 2.0 CaCl2 treatment dropped.
Fig. 7.

Weekly changes in pH throughout the experiment of all Sphagnum species and experimental treatments (displayed are mean ± SE). pH was measured twice weekly, except only once at the start (0). Blue lines represent control groups, green lines the 2.0 CaCl2 treatment, red lines the 0.8 treatment and black lines the 2.0 treatment. Week 0 is the moment just before the experimental treatments were added.
Discussion
Main findings
Our controlled laboratory experiment demonstrated that was detrimental to all seven Sphagnum species in 2.0 mM concentration, while showing more variation between different species in 0.8 mM . Nevertheless, in the 0.8 treatment all species succumbed after 6 to 10 weeks. For all species, both 0.8 and 2.0 treatments resulted in discoloration, reduced growth (biomass and length increment) and cellular K+ leakage (Figs. 2, 3, 4). Discoloration temporally correlated with K+ leakage (Fig. 6) and divalent Ca2+ accumulated over monovalent Na+ cations. Moreover, high Ca2+ tolerance was species‐specific, with S. magellanicum, S. papillosum and S. palustre not adversely affected, while S. squarrosum and S. teres & S. contortum showing reduced functioning (e.g. K+ leakage, discoloration). The current results, for the first time, show temporal patterns and dose dependency of effects for a wide range of Sphagnum species.
Increase of rather than pH seems to determine the onset of physiological stress
The die‐off of Sphagnum mosses in 2.0 mM is in accordance with similar work using S. palustre in Koks et al. (2019). To our knowledge, effects of high loads have not been further investigated for other Sphagnum mosses since initial work of Skene (1915) and Paul (1906). Skene (1915) did describe and (carbonate) affecting Sphagnum species specifically, but not the time‐dependence of this effect. With increased efforts in peatland restoration and increasing commercial interest in Sphagnum farming, more insight into the effects of loads on Sphagnum over time are of utmost importance to properly assess water quality for restoration and Sphagnum farming efforts (Wichmann et al. 2017).
Koks et al. (2019) noted that when can be suppressed, a higher pH in itself may not be toxic for Sphagnum. At pH 8 or higher, however, CO2 concentrations may become critically low, inhibiting photosynthesis. Nonetheless, CO2 concentrations were, due to the high amount of dissolved carbon, on average still higher in the 2.0 treatment than the control (Table S2). If CO2 was limiting photosynthesis, lower CO2 concentrations should have been observed compared to the control, which was not the case (Table S4). Any potential physiological stress due to high Na+ concentrations rather than is also unlikely, as previous investigations found that 2.0 mM Na+ did not harm Sphagnum (Paul 1906; Pouliot et al. 2013; Koks et al. 2019). Koks et al. (2019) also did not find any significant K+ leakage in their NaCl treatment compared to the control, while the NaCl treatment had double the Na+ cation concentrations compared to the 2.0 mM treatment in this experiment. Nonetheless former studies did not completely disentangle the potential combined negative effects of Na+ and . Our findings do warrant future studies aiming at complete disentanglement of the effects of increased pH, and increased on Sphagnum.
While we did not measure the intracellular pH, we found Sphagnum mosses subjected to lower loads in the 0.8 treatment persisted longer compared to the 2.0 treatment while having a similar or even higher pH in treatment solution (Fig. 7). But, in practice, a high pH (>6.5) in peatland waters will coincide with higher concentrations due to its dissolved carbon content (Lamers et al. 1999; Smolders et al. 2001). This further suggests the need to disentangle the effects of pH and on Sphagnum functioning.
Even though the effects of pH and have still not been fully resolved, lower loads do lead to slower responses in Sphagnum spp. compared to higher loads as in the 2.0 treatment. The 0.8 treatment showed the strongest variation in survival between species (Table S4). S. squarrosum persisted longer, remaining vital compared to poor fen and bog species in regard to discoloration (Fig. 3). However, it is not known which species‐specific traits and characteristics are the main drivers explaining these differences.
Calcium (Ca2+) and physiological stress in rich fen species
The pH of the solution was affected by increasing cation concentrations (Fig. 7). The weekly renewal of treatment solutions caused a subsequent drop in pH in the 2.0 CaCl2 treatment for all Sphagnum species (to approximately pH 4.2, whereas in the control a pH of around 5 was maintained). However, this drop in pH was reduced each week, likely as a result of a decreasing acidification capacity with all exchange sites (including the newly formed ones due to growth) becoming increasingly saturated (Clymo 1964), unlike for the control. The pH in this treatment was not artificially increased by anions like OH− or . Contrary to expectations based on their natural environment, typical bog species and poor fen species were little affected by high Ca2+ concentrations. S. magellanicum, S. papillosum, S. palustre and S. fallax submerged in low pH, but Ca2+‐rich solutions did not show any negative effects regarding discoloration (Fig. 3). This result is in accordance with findings of Koks et al. (2019) for elevated cation concentrations at similar ion loads for S. palustre. Interestingly, only minerotrophic species (S. squarrosum, S. teres & S. contortum) showed visual negative effects in this treatment.
For rich fen species the substantial lowering of the pH may have resulted in physiological stress (Fig. 4) and, consequently, a growth reduction after 4 to 5 weeks. These findings go against our assumption that increased Ca2+ concentrations, without addition, have no negative effects, especially for minerotrophic species such as S. teres & S. contortum (Clymo 1973; Vicherová et al. 2015, 2017).
Physiological limitations
The treatments with high concentrations of had severe effects on the physiological performance of all Sphagnum species tested: pigment loss, leakage of K+ and ultimately tissue and plant death. In the schematic representation of a Sphagnum cell (Fig. 8), a hypothetical interpretation of the different processes, acting in parallel, affected by high in the solution, is represented. The different elements that might together lead to the disruption of cell vitality are: 1. limited control of the cell over the apoplastic pH; 2. Influence of high external pH on the cytoplastic pH; 3. Diminished proton motive force, required to drive K+ influx and maintain ion homeostasis.
Fig. 8.

Hypothetical model showing; (A) how a proton pump releases H+ and causes an electric charge across the membrane, while uni‐ and symporters import K+ and H+ into the cell. In contrast a K+/H+ antiporter releases K+ into a rich solution while taking up H+ to maintain intracellular pH in Sphagnum cells. (B) Result of cations (Cat+) being bound to negatively charged carboxyl groups on the cell wall of Sphagnum, releasing H+ into solution.
Exposing Sphagnum mosses repeatedly to high levels of resulted in immediate K+ leakage, being significant after 3 weeks of exposure, with rates flattening after 4–5 weeks for all species in the 2.0 treatment, but whether the loss of K+ was the sole explanation for die‐off remains unknown (Demidchik et al. 2014). Nonetheless most species in the 0.8 treatment initially still took up K+, indicating correct cell functioning, but after this initial uptake, they also started to leak K+.
Leakage of K+ indicates a perturbation of K+ homeostasis, either through increased K+ efflux or inhibition of K+ influx (Demidchik et al. 2014). High concentrations may impair the cell control of apoplastic pH by scavenging H+ and thereby preventing acidification at the plasma membrane/cell wall interface (Skene 1915; Marschner & Römheld 1994) (Fig. 8). Plant cells typically maintain an electrical gradient of approximately −150 to −250 mV (negative inside) and a pH gradient (ΔpH) of 1.5 to 2 (acidic outside) across plasma membranes (Sze & Chanroj 2018). Under normal or acidic conditions, proton pumps actively pump protons out using the plasma membrane ATPase, while K+ and protons can subsequently enter the cell via K+/H+ symport (Fig. 8). Under alkaline conditions, the consumption by and the subsequent lack of protons outside the cell can easily result in an imbalance leading to an internal rise of pH, which is maintained as long as the solution is buffered (high concentration). Similarly, pH affecting K+ transport could also, for instance, be based on protonation and deprotonation of apoplastic histidine residues of the K+ uptake channel in response to the H+ pumping activity and acid metabolism, which modulates the activity of this transporter (Hoth et al. 1997).
It is not hard to imagine that Sphagnum spp. adapted to acidic conditions, are not able to adapt to sustained buffered conditions. Sze & Chanroj (2018) suggested that plants, as well as bacteria, might have K+/H+ antiporters, enabling the active release of K+ and uptake of protons under alkaline conditions maintaining the pH homeostasis (Fig. 8).
The shift in external pH from approximately pH 4 under control conditions to higher than pH 9 at high concentrations will change the proton motive force across the plasma membrane from −296 mV (−200 mV electrical potential difference across the membrane, plus −1.75 × 55 mV due to the 1.75 pH units concentration difference in H+ ions between apoplast and cytoplasm) to approximately −90 mV (assuming the membrane potential remains unaffected and the H+ concentration is now reversed to more acidic on the cytoplasmic side by 2 pH units). With the proton motive force diminished to such an extent, the normal K+ gradient across the membrane can no longer be supported through H+/K+ symporter activity, and a net efflux of K+ results.
The absence of sufficient proton motive force can additionally result in a higher vulnerability to Na+ in the medium, as H+/Na+ antiporters needed to pump out the Na+ ions that have leaked in, will not function with an insufficient force driving the H+.
In vascular plants, Ca2+ levels in the 5 to 10 mM range strongly affect the ability of plant cells to control the apoplastic pH and thereby affect the functioning of ion transporters (Martinière et al. 2018). Ca2+ toxicity in calcifuge bryophytes is assumed to be caused by insufficient control over the balance of intracellular Ca2+ uptake/efflux affecting K+ uptake, as suggested by increased survival of calcifuge Sphagnum mosses in buffered fens by increased K+ availability, and field observations that K+ facilitates Sphagnum expansion (Vicherová et al. 2015).
Yet the exact physiological mechanisms causing deterioration due to in all Sphagnum species and Ca2+ in minerotrophic species still remain a topic for further investigation.
Conclusions and recommendations
Understanding the effects of and Ca2+ on Sphagnum growth is crucial for the conservation and restoration of Sphagnum habitats and the development of Sphagnum farming aiming at cultivation, production and harvest of Sphagnum moss biomass. We show that enrichment with , but to a large extent not Ca2+, is detrimental for most Sphagnum mosses tested and also show this effect to be dose‐ and duration‐dependent and species‐specific. When restoring and/or developing Sphagnum habitats, levels should generally remain low or absent. However, regarding discoloration at elevated concentrations (0.8 mM), S. squarrosum and S. teres & S. contortum persisted longest, while potentially being more susceptible when exposed to high Ca2+ concentrations if levels were low. In natural systems, high Ca2+ levels generally coincide with /CO3 2‐ presence and both S. squarrosum and S. palustre may be best suited to the combination of these two factors (Turekian & Wedepohl 1961; Smolders et al. 2003).
For Sphagnum farming or the restoration of Sphagnum‐dominated systems, dealing with drought is crucial and may require the use of an external water source (Bragazza 2008). As the adverse effects of will depend on the species, concentration, the duration and degree of submergence, Sphagnum species may withstand alkaline water to a certain extent, depending on the condition. Bogs may be restored more effectively where sufficient peat remains (e.g. >100 cm) to buffer alkaline‐rich groundwater from upwelling beyond the moss surface. As supplying ‐rich water to combat drought can be detrimental for Sphagnum, artificial acidification of surface waters may prove to be a potential new management option in the future.
Supporting information
Table S1. Average treatment characteristics (μmol l−1) of the overall solution supplied to the containers weekly during the 10‐week experiment (n = 10) (displayed are mean ± SE).
Table S2. Average overall output water characteristics (μmol l−1) after weekly renewal, (control (n = 40), 0.8 (n = 40), 2.0 (n = 40) and 2.0 CaCl2 (n = 30), except for pH where due to two weekly measurements instead of one the n is doubled (n = 60) (displayed are mean ± SE).
Table S3. Sphagnum moss plant content (μmol g dw−1) per species and treatment (control (n = 4), 0.8 (n = 4), 2.0 HCO3 (n = 4) and 2.0 CaCl2 (n = 3)). Also, Sphagnum material used as reference and not subjected to any experiment, treatment or manipulation (n = 6) (displayed are mean ± SE).
Table S4. Changes in morphology of S. fallax, S. papillosum, S. magellanicum, S. palustre, S. squarrosum and S. teres & S. contortum for the control, 0.8 , 2.0 and 2.0 CaCl2 treatments.
Acknowledgements
The authors gratefully acknowledge R. Boelens, F. Boelens, J. Siepman, R. Peters and P. van der Ven (General Instrumentation, Radboud University) for their efforts in the laboratory analysing the samples. A.H.W.K. received funding from Moosweiter. C.F. received funding by FACCE ERA‐GAS (ALW.GAS.4 Peatwise)(WET HORIZONS). This research was funded by Interreg NWE Carbon Connects Capitilisation. We also acknowledge Landschap Noord‐Holland and Staatsbosbeheer, who allowed us to collect the mosses, and particularly S. Wössner and Greifswald University who helped with fieldwork. We also thank our partners in the Moosweit‐Project (“Torfmooskultivierung zur klimaschonenden Moorentwicklung: Anbau und Ernte von kultivierten Torfmoosen”), Greifswald University, Rostock University, MoKuRa and Oldenburg University.
References
- Bates J.W. (1982) The role of exchangeable calcium in saxicolous calcicole and calcifuge mosses. New Phytologist, 90, 239–252. [Google Scholar]
- Boos D.D., Brownie C. (1995) ANOVA and rank tests when the number of treatments is large. Statistics and Probability Letters, 23, 183–191. [Google Scholar]
- Bragazza L. (2008) A climatic threshold triggers the die‐off of peat mosses during an extreme heat wave. Global Change Biology, 14, 2688–2695. [Google Scholar]
- Carifio J., Perla R. (2008) Resolving the 50‐year debate around using and misusing Likert scales. Medical Education, 42, 1150–1152. [DOI] [PubMed] [Google Scholar]
- Clymo R.S. (1964) The origin of acidity in Sphagnum bogs. The Bryologist, 67, 427–431. [Google Scholar]
- Clymo R.S. (1973) The growth of Sphagnum: some effects of environment. Journal of Ecology, 61, 849–869. [Google Scholar]
- Daniels R.E., Eddy E. (1985) Handbook of European Sphagna. Institute of Terrestrial Ecology, Huntingdon, UK. [Google Scholar]
- Demidchik V., Straltsova D., Medvedev S.S., Pozhvanov G.A., Sokolik A., Yurin V. (2014) Stress‐induced electrolyte leakage: the role of K+‐permeable channels and involvement in programmed cell death and metabolic adjustment. Journal of Experimental Botany, 65, 1259–1270. [DOI] [PubMed] [Google Scholar]
- Feldmeyer‐Christe E., Schnyder N., Bisang I. (2001) Distributions and habitats of peat mosses, Sphagnum, in Switzerland. Lindbergia, 26, 8–22. [Google Scholar]
- Gaudig G., Krebs M., Prager A., Wichmann S., Barney M., Caporn S.J.M., Emmel M., Fritz C., Graf M., Grobe A., Gutierrez P.S., Hogue‐Hugron S., Holzträger S., Irrgang S., Kämäräinen A., Karofeld E., Koch G., Koebbing J.F., Kumar S., Matchutadze I., Oberpaur C., Oestmann J., Raabe P., Rammes D., Rochefort L., Schmilewksi G., Sendžikaitė J., Smolders A., St‐Hilaire B., van de Riet B., Wright B., Wright N., Zoch L., Joosten H. (2018) Sphagnum farming from species selection to the production of growing media: a review. Mires and Peat, 20, 1–30. [Google Scholar]
- Goerg G.M. (2015) The Lambert Way to Gaussianize Heavy‐Tailed Data with the Inverse of Tukey’s h Transformation as a Special Case (T. Hu, Ed.). The Scientific World Journal 2015. [DOI] [PMC free article] [PubMed]
- Hájek M., Horsák M., Hájkova P., Dítě D. (2006) Habitat diversity of central European fens in relation to environmental gradients and an effort to standardise fen terminology in ecological studies. Perspectives in Plant Ecology, Evolution and Systematics, 8, 97–114. [Google Scholar]
- Hájková P., Hájek M. (2004) Bryophyte and vascular plant responses to base‐richness and water level gradients in Werstern Carpathian Sphagnum‐Rich Mires. Folia Geobotanica, 39, 335–351. [Google Scholar]
- Hájková P., Hájek M. (2007) Sphagnum distribution patterns along environmental gradients in Bulgaria. Journal of Bryology, 29, 18–26. [Google Scholar]
- Harpenslager S.F., Van Dijk G., Kosten S., Roelofs J.G.M., Smolders A.J.P., Lamers L.P.M. (2015) Simultaneous high C fixation and high C emissions in Sphagnum mires. Biogeosciences, 12, 4739–4749. [Google Scholar]
- Hoth S., Dreyer I., Dietrich P., Becker D., Müller‐Röber B., Hedrich R. (1997) Molecular basis of plant‐specific acid activation of K+ uptake channels. Proceedings of the National Academy of Sciences USA, 94, 4806–4810. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hothorn T., Bretz F., Westfall P., Heiberger R.M., Schuetzenmeister A., Scheibe S. (2020) multcomp: Simultaneous Inference in General Parametric Model. R package version 1.4‐12. https://cran.r‐project.org/web/packages/multcomp/ (10 February 2020).
- Jassey V.E.J., Gilbert D., Binet P., Toussaint M., Chiapusio G. (2011) Effect of a temperature gradient on Sphagnum fallax and its associated living microbial communities: a study under controlled conditions. Canadian Journal of Microbiology, 57, 226–235. [DOI] [PubMed] [Google Scholar]
- Jones C.G., Lawton J.H., Shachak M. (1994) Organisms as ecosystem engineers. Oikos, 69, 373–386. [Google Scholar]
- Koks A.H.W., Van Dijk G., Smolders A.J.P., Lamers L.P.M., Fritz C. (2019) The effects of alkalinity and cations on the vitality of Sphagnum palustre L. Mires and Peat, 24, 1–14. [Google Scholar]
- Kooijman A.M., Kanne D.M. (1993) Effects of water chemistry, nutrient supply and interspecific interactions on the replacement of Sphagnum subnitens by S. fallax in fens. Journal of Bryology, 17, 431–438. [Google Scholar]
- Kozak M., Piepho H.‐P. (2017) What’s normal anyway? Residual plots are more telling than significance tests when checking ANOVA assumptions. Journal of Agronomy and Crop Science, 00, 1–13. 10.1111/jac.12220 [DOI] [Google Scholar]
- Lamers L.P.M., Farhoush C., Van Groenendael J.M., Roelofs J.G.M. (1999) Calcareous groundwater raises bogs; the concept of ombrotrophy revisited. Journal of Ecology, 87, 639–648. [Google Scholar]
- Lawrence M. (2016) ez: Easy Analysis and Visualization of Factorial Experiments. R package version 4.4‐0. https://cran.r‐project.org/web/packages/ez/ (14 March 2019).
- Lenth R.V. (2016) Least‐squares means: the R package lsmeans. Journal of Statistical Software, 69, 1–33. [Google Scholar]
- Lenth R., Singmann H., Love J., Buerkner P., Herve M. (2020) em‐means: Estimated marginal means, aka least‐squares means. R package version 1.4.4. https://cran.r‐project.org/web/packages/emmeans (10 February 2020).
- Limpens J., Berendse F. (2003) How litter quality affects mass loss and N loss from decomposing Sphagnum . Oikos, 103, 537–547. [Google Scholar]
- Marschner H., Römheld V. (1994) Strategies of plants for acquisition of iron. Plant and Soil, 165, 261–274. [Google Scholar]
- Martinière A., Gibrat R., Sentenac H., Dumont X., Gaillard I., Paris N. (2018) Uncovering pH at both sides of the root plasma membrane interface using noninvasive imaging. Proceedings of the National Academy of Sciences, 115, 4–9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Paul H. (1906) Zur Kalkfeindlichkeitsfrage der Torfmoose. Berichte Deutschen Botanischen Gesellschaft, 24, 148–154. [Google Scholar]
- Plesková Z., Jiroušek M., Peterka T., Hájek T., Dítě D., Hájková P., Navrátilová J., Šímová A., Syrovátka V., Hájek M. (2016) Testing inter‐regional variation in pH niches of fen mosses. Journal of Vegetation Science, 27, 352–364. [Google Scholar]
- Potter L., Foot J.P., Caporn S.J.M., Lee J.A. (1996) Responses of four Sphagnum species to acute ozone fumigation. Journal of Bryology, 19, 19–32. [Google Scholar]
- Pouliot R., Rochefort L., Graf M.D. (2013) Fen mosses can tolerate some saline conditions found in oil sands process water. Environmental and Experimental Botany, 89, 44–50. [Google Scholar]
- R Core Team . (2013) R: a language and environment for statistical computing. R Foundation for Statistical Computing, Vienna, Austria. [Google Scholar]
- Rydin H., Gunnarsson U., Sundberg S. (2006) The role of Sphagnum in peatland development and persistence. In: Wieder R.K., Vitt D.H. (Eds), Boreal peatland ecosystems. Springer, Berlin, Germany, pp 47–65. [Google Scholar]
- Schipperges B., Rydin H. (1998) Response of photosynthesis of Sphagnum species from contrasting microhabitats to tissue water content and repeated desiccation. New Phytologist, 140, 677–684. [DOI] [PubMed] [Google Scholar]
- Skene M. (1915) The acidity of Sphagnum and its relation to chalk and mineral salts. Annals of Botany, 29, 65–87. [Google Scholar]
- Smolders A.J.P., Tomassen H.B.M., Pijnappel H.W., Lamers L.P.M., Roelofs J.G.M. (2001) Substrate‐derived CO2 is important in the development of Sphagnum spp. New Phytologist, 152, 325–332. [Google Scholar]
- Smolders A.J.P., Tomassen H.B.M., Van Mullekom M., Lamers L.P.M., Roelofs J.G.M. (2003) Mechanisms involved in the re‐establishment of Sphagnum‐dominated vegetation in rewetted bog remnants. Wetlands Ecology and Management, 11, 403–418. [Google Scholar]
- Sze H., Chanroj S. (2018) Plant endomembrane dynamics: studies of K+/H+ antiporters provide insights on the effects of pH and ion homeostasis. Plant Physiology, 177, 875–895. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Turekian K.K., Wedepohl K.H. (1961) Distribution of the elements in some major units of the earth’s crust. Geological Society of America Bulletin, 72, 175–192. [Google Scholar]
- Van Den Elzen E., Kox M.A.R., Harpenslager S.F., Hensgens G., Fritz C., Jetten M.S.M., Ettwig K.F., Lamers L.P.M. (2017) Symbiosis revisited: phosphorus and acid buffering stimulate N2 fixation but not Sphagnum growth. Biogeosciences, 14, 1111–1122. [Google Scholar]
- Vicherová E., Hájek M., Hájek T. (2015) Calcium intolerance of fen mosses: physiological evidence, effects of nutrient availability and successional drivers. Perspectives in Plant Ecology, Evolution and Systematics, 17, 347–359. [Google Scholar]
- Vicherová E., Hájek M., Šmilauer P., Hájek T. (2017) Sphagnum establishment in alkaline fens: importance of weather and water chemistry. Science of the Total Environment, 580, 1429–1438. [DOI] [PubMed] [Google Scholar]
- Vicherová E., Hájek T. (2013) Desiccation tolerance of Sphagnum revisited: a puzzle resolved. Plant Biology, 16, 1–9. [DOI] [PubMed] [Google Scholar]
- Vitt D.H., Slack N.G. (1984) Niche diversification of Sphagnum relative to environmental factors in Northern Minnesota USA peatlands. Canadian Journal of Botany, 62, 1409–1430. [Google Scholar]
- Vroom R.J.E., Temmink R.J.M., van Dijk G., Joosten H., Lamers L.P.M., Smolders A.J.P., Krebs M., Gaudig G., Fritz C. (2020) Nutrient dynamics of Sphagnum farming on rewetted bog grassland in NW Germany. Science of the Total Environment, 726, 1–11. [DOI] [PubMed] [Google Scholar]
- Wichmann S., Prager A., Gaudig G. (2017) Establishing Sphagnum cultures on bog grassland, cut‐over bogs, and floating mats: procedures, costs and area potential in Germany. Mires and Peat, 20, 1–19. [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Table S1. Average treatment characteristics (μmol l−1) of the overall solution supplied to the containers weekly during the 10‐week experiment (n = 10) (displayed are mean ± SE).
Table S2. Average overall output water characteristics (μmol l−1) after weekly renewal, (control (n = 40), 0.8 (n = 40), 2.0 (n = 40) and 2.0 CaCl2 (n = 30), except for pH where due to two weekly measurements instead of one the n is doubled (n = 60) (displayed are mean ± SE).
Table S3. Sphagnum moss plant content (μmol g dw−1) per species and treatment (control (n = 4), 0.8 (n = 4), 2.0 HCO3 (n = 4) and 2.0 CaCl2 (n = 3)). Also, Sphagnum material used as reference and not subjected to any experiment, treatment or manipulation (n = 6) (displayed are mean ± SE).
Table S4. Changes in morphology of S. fallax, S. papillosum, S. magellanicum, S. palustre, S. squarrosum and S. teres & S. contortum for the control, 0.8 , 2.0 and 2.0 CaCl2 treatments.


