Abstract
Background
Few studies explored the role of hypothermic machine perfusion (HMP) in the sub‐group of non‐standard renal grafts with a biopsy‐proven advanced histological impairment. This study aimed to investigate the role of HMP in grafts with a Karpinski Score >3 in terms of the need for dialysis, creatinine reduction ratio at day‐7 (CRR7), and 3‐year graft survival.
Methods
Twenty‐three perfused grafts with Karpinski Score >3 evaluated between November 2017 and December 2018 were retrospectively analyzed and compared with a control group of 32 non‐perfused grafts transplanted between January 2014 and October 2017.
Results
After transplantation, perfused grafts had fewer cases requiring dialysis (8.7% vs. 34.4%; p = 0.051), a better reduction in serum creatinine (median at 7 days: 2.2 vs. 4.3 mg/dl; p = 0.045), and shorter length of hospital stay (median 11 vs. 15 days; p = 0.01). Three‐year death‐censored graft survival was better in the perfused cases (91.3% vs. 77.0%; p = 0.16).
In perfused grafts, initial renal resistance (RR) had the best predictive value for renal function recovery after the first week, as defined by CRR7 ≤ 70% (AUC = 0.83; p = 0.02). A cut‐off value of 0.5 mm Hg/ml/min showed a sensitivity of 82.4%, a specificity of 83.3%, and diagnostic odds ratio = 23.4. After dividing the entire population into a Low‐RR (n = 8) and a High‐RR Group (n = 15), more cases with CRR7 ≤ 70% were reported in the latter group (86.7 vs. 13.3%; p = 0.03).
Conclusion
HMP yielded promising results in kidneys with Karpinski Score >3. Initial RR should be of interest in selecting non‐standard organs for single kidney transplantation even in impaired histology.
Keywords: delayed graft function, dialysis, graft loss, Karpinski Score, perfusion, serum creatinine
Few studies explored the role of Hypothermic Machine Perfusion (HMP) in the sub‐group of non‐standard renal grafts with a biopsy‐proven advanced histological impairment used for a single kidney transplantation. Twenty‐three perfused grafts with Karpinski Score >3 evaluated between November 2017 and December 2018 were retrospectively analyzed and compared with a control group of 32 non‐perfused grafts transplanted between January 2014 and October 2017. After transplantation, perfused grafts had fewer cases requiring dialysis, a better reduction in serum creatinine, shorter length of hospital stay, and better 3‐year death‐censored graft survivals. Initial renal resistance (RR) during perfusion showed promising results in selecting non‐standard organs for single kidney transplantation even in impaired histology.

1. INTRODUCTION
Kidney transplantation (KT) represents the best therapeutic strategy for treating end‐stage renal disease. The resultant growing number of patients waiting for KT has thus pushed for an attendant increase in the number of potential donors, prompting a more liberal use of Expanded Criteria Donors (ECD). 1 However, renal transplants from such donors are often correlated with worse early and late posttransplant results. 2 , 3 , 4 Therefore, new strategies have been proposed to improve the graft selection process. For example, pretransplant histological evaluation has been primarily adopted to identify the organs usable for a single KT, a double KT, or discard. 5 , 6 , 7 Moreover, in the last decade, in‐house hypothermic machine perfusion (HMP) has provoked interest in the management of non‐standard donors due to its ability to select graft quality by evaluating the renal resistances (RR) trend 8 , 9 and to reduce the ischemic damage caused by cold storage preservation. 10
Many studies have explored the beneficial impact of HMP in the setting of KT, 11 , 12 but few have investigated the sub‐group of renal grafts with an initial severe histological impairment. 8
The primary aim of the study was to investigate the role of in‐house HMP in grafts with a histological Karpinski Score >3 used for single KT in terms of posttransplant Delayed Graft Function (DGF), graft functional recovery as evaluated by serum creatinine (sCr) decline, and 3‐year graft survival.
The secondary aim was to identify an initial RR threshold to select patients at risk for poor organ recovery.
2. MATERIALS AND METHODS
We performed a retrospective analysis of the data from 92 renal grafts from deceased‐brain donors evaluated for a single KT between November 1, 2017 and December 31, 2018. We selected this period with the intent to have a minimum of 3 years of follow‐up after transplant. Exclusion criteria were: (a) grafts directly transplanted without perfusion (n = 22); (b) perfused grafts with a Karpinski Score ≤3 (n = 41); (c) grafts discarded for inadequate quality evaluated during back‐table (n = 6). Finally, 23 grafts were enrolled in the present study. All the remaining grafts were used for single KTs.
A control group of deceased‐brain‐donor non‐perfused renal grafts with a Karpinski Score >3 was selected among the single KTs performed between January 1, 2014 and October 31, 2017. A total of 32 cases were identified. The ethics committee of Umberto I Policlinic of Rome approved the present retrospective observational study (approval number 1000/2018).
2.1. HMP protocol, management, and definitions
In our center, we adopted an HMP protocol in which all the ECD kidneys being available for transplantation after the back‐table were perfused with an in‐house HMP. An ECD was defined according to the following definition: (a) a deceased‐brain donor (DBD) aged 60 years or older; or (b) a DBD aged 50–59 years with two or more of the following comorbidities: a history of hypertension, death resulting from cerebrovascular accident, and terminal sCr ≥1.5 mg/dl. 3
During organ procurement, in vivo perfusion of the abdominal organs was performed using Celsior® (Institut Georges Lopez IGL, Lissieu, FRANCE) with a total volume of 1 L per 10 kg of donor weight. A renal biopsy was performed in all ECDs at the end of procurement. Histological graft quality was assessed by a group of expert pathologists using the Karpinski Score (Table S1). 5 In all cases, histological evaluation was available before the beginning of the KT. Grafts presenting a Karpinski Score ≤3 were judged to be usable for a single KT. For those with a score ranging from 4 to 6, the use of the graft for a single KT was decided according to the following parameters: (a) absence of diffuse gross vascular atherosclerosis in the graft; (b) absence of severe microscopically assessed pyelonephritis. In none of the cases was a double KT performed. When the Score was ≥7, the kidney was directly discarded.
LifePort® Kidney Transporter machine (Organ Recovery Systems, Brussels, Belgium) was used to perform perfusion with a pulsatile pressure. The initial pressure value used was 30 mm Hg. The perfusion solution used during the HMP was KPS‐1® solution (Organ Recovery Systems, Brussels, Belgium). Renal resistances (mm Hg/ml/min), flow (ml/min), and temperature (°C) were recorded at the beginning of perfusion, 20, 40, 60, and 120 min. All grafts were perfused for at least 120 min.
Cold ischemia time (CIT) was calculated from the cross‐clamp time until the end of the HMP. The following recipient‐related data were collected in the postoperative period: daily sCr, need for dialysis, and hospital length of stay. Delayed graft function (DGF) was defined in two different ways: (a) need for dialysis within 7 days from KT, 3 and (b) creatinine reduction ratio at day 7 (CRR7) ≤70%. CRR7 was calculated using the following equation: CRR7(%) = ([sCr0–sCr7] × 100)/sCr0), where sCr0 indicates sCr levels immediately before KT and no later than 6 h after the last dialysis, and sCr7 indicates levels on day 7 post‐KT. 13
Primary non‐function (PNF) was defined as a permanent lack of graft function necessitating dialysis from the time of transplantation in a well‐perfused but never functioning graft.
Graft loss was defined as returning to dialysis or sCr clearance <15 ml/min/1.73m2 at the last evaluation. 3
2.2. Immunosuppression management
All the patients enrolled in the present analysis had a standardized immunosuppressive schedule. Induction immunosuppressive therapy consisted of basiliximab administered during KT and on the fourth postoperative day and intraoperative methylprednisolone (500 mg). Maintenance therapy was based on a triple immunosuppressive regimen composed of prednisone (20 mg daily), mycophenolate sodium (1440 mg daily), and tacrolimus (0.06 mg/kg daily). Tacrolimus was started when sCr was <3 mg/dl or on the fifth postoperative day at the latest.
2.3. Statistical analysis
Continuous variables were reported as medians and interquartile ranges (IQR). Dummy variables were reported as numbers and percentages. Mann–Whitney U test and Fisher's exact test were used to compare continuous and categorical variables, respectively.
Receiver operating characteristic (ROC) curve analysis was used for testing the performance of different variables (i.e., initial RR, initial flow, donor sCr, and Karpinski Score) in predicting DGF defined as CRR7 ≤ 70%. Different thresholds of RR were investigated in terms of sensitivity, specificity, and diagnostic odds ratios (DOR). We arbitrarily investigated the RR = 0.4, 0.5, 0.75, and 1.0 mm Hg/ml/min, approximately corresponding to the fifteenth centile, the fourth, the seventh, and the ninth decile. As for the DOR, the higher was its value, the better the test performance.
Graft survival and patient survival probabilities were estimated using the Kaplan–Meier method. The survival results were compared using the log‐rank test. In case of patient death with a functioning graft, graft survival was censored. The last censoring was performed on December 31, 2021.
Variables with a p < 0.05 were considered statistically significant. Statistical analyses were performed using the SPSS statistical package version 24.0 (SPSS Inc., Chicago, IL, USA).
3. RESULTS
3.1. Analysis of the selected cases
From November 2017 to December 2018, 92 kidneys were considered for a single KT; 29 grafts presented a Karpinski Score >3. After back‐table evaluation, six grafts were discarded due to diffuse gross atherosclerosis of the renal artery (n = 2) or severe pyelonephritis (n = 4). Four of the six discarded grafts presented a Karpinski Score of 4 (66.7%), the other two had a score of 5 (16.7%) and 6 (16.7%), respectively. Twenty‐three grafts were thus analyzed. A control group of 32 non‐perfused grafts with a Karpinski Score >3 was identified from January 2014 to October 2017.
Donor‐, recipient‐, perfusion‐, and transplant‐related characteristics of the 23 perfused versus 32 non‐perfused cases are reported in Table 1. As for the donors' characteristics, the two groups were statistically similar. The median Kidney Donor Risk Index (KDRI) and age were 1.68 versus 1.66 (p = 0.56) and 67 versus 67 years (p = 0.73) in perfused versus non‐perfused cases, respectively. Preprocurement median sCr was 1.3 versus 1.0 mg/dl (p = 0.45).
TABLE 1.
Donor‐, recipient‐, and perfusion‐related characteristics in the entire population
| Variables | Perfusion group (n = 23) | Control group (n = 32) | p‐value |
|---|---|---|---|
| Median (IQR) or n (%) | |||
| Donor‐specific variables | |||
| KDRI | 1.68 (1.36–2.17) | 1.66 (1.45–1.98) | 0.56 |
| KDPI | 92 (80–99) | 90 (84–98) | 0.87 |
| Total Karpinski Score | 4 (4–5) | 4 (4–6) | 0.18 |
| 4 | 16 (69.6) | 18 (56.3) | |
| 5 | 5 (21.7) | 5 (15.6) | 0.34 |
| 6 | 2 (8.7) | 9 (28.1) | |
| Age, years | 67 (60–74) | 67 (63–74) | 0.73 |
| Male, gender | 12 (52.2) | 19 (59.4) | 0.78 |
| Weight, kg | 80 (73–90) | 78 (65–90) | 0.55 |
| Height, cm | 170 (164–180) | 170 (160–175) | 0.56 |
| BMI | 27 (26–31) | 27 (25–32) | 0.78 |
| sCr, mg/dl | 1.3 (0.8–1.6) | 1.0 (0.7–1.6) | 0.45 |
| Sodium peak, mEq/L | 150 (145–151) | 153 (147–159) | 0.046 |
| ICU stay, days | 5 (2–5) | 6 (3–13) | 0.03 |
| VAS | 27 (0–60) | 17 (7–37) | 0.71 |
| Story of hypertension | 12 (52.2) | 17 (53.1) | 1.00 |
| Story of DM2 | 7 (30.4) | 7 (21.9) | 0.54 |
| Story of smoking | 10 (43.5) | 10 (31.3) | 0.40 |
| Story of cardiopathy | 4 (17.4) | 11 (34.4) | 0.22 |
| Story of hepatopathy | 2 (8.7) | 2 (6.3) | 1.00 |
| Story of dyslipidemia | 4 (17.4) | 2 (6.3) | 0.22 |
| Cause of death | |||
| Anoxia | 1 (4.3) | 1 (3.1) | |
| Blunt trauma | 5 (21.7) | 4 (12.5) | 0.63 |
| CVA | 17 (73.9) | 27 (84.4) | |
| Episode(s) of hypotension | 6 (26.1) | 5 (15.6) | 0.50 |
| Episode(s) of cardiac arrest | 3 (13.0) | 2 (6.3) | 0.64 |
| Recipient‐specific variables | |||
| Age, years | 61 (53–68) | 62 (58–68) | 0.44 |
| Male, gender | 13 (56.5) | 22 (68.8) | 0.40 |
| Weight, kg | 71 (70–76) | 73 (64–82) | 0.73 |
| Height, cm | 165 (160–165) | 168 (165–176) | 0.04 |
| BMI | 24 (23–27) | 24 (23–27) | 0.92 |
| Renal pathology | |||
| ADPKD | 4 (17.4) | 3 (9.4) | |
| Angiosclerosis | 2 (8.7) | 6 (18.8) | |
| DM2 | 2 (8.7) | 4 (12.5) | |
| GN | 3 (13.0) | 6 (18.8) | 0.46 |
| Lupus | 2 (8.7) | 0 (–) | |
| Re‐KT | 2 (8.7) | 4 (12.5) | |
| Other | 3 (13.0) | 6 (18.8) | |
| Unknown | 5 (21.7) | 3 (9.4) | |
| Time of dialysis, years | 5 (4–5) | 3 (3–4) | 0.02 |
| Arterial hypertension | 18 (78.3) | 26 (81.3) | 1.00 |
| CIT min | 623 (484–745) | 695 (604–900) | 0.046 |
| CIT min (no perfusion time) | 380 (298–419) | 695 (604–900) | <0.0001 |
| Perfusion‐specific variables | |||
| Perfusion, min | 247 (125–330) | – | – |
| Flow, ml/min | |||
| 0 min | 46 (30–56) | ||
| 20 min | 78 (51–98) | ||
| 40 min | 81 (60–100) | – | – |
| 60 min | 85 (60–105) | ||
| 120 min | 87 (65–110) | ||
| Delta flow, % | +124 (+71 to +200) | ||
| RR, mm Hg/ml/min | |||
| 0 min | 0.57 (0.46–0.87) | ||
| 20 min | 0.34 (0.26–0.53) | ||
| 40 min | 0.28 (0.22–0.47) | – | – |
| 60 min | 0.28 (0.22–0.41) | ||
| 120 min | 0.28 (0.23–0.36) | ||
| Delta RR, % | −60 (−67 to −48) | ||
| sCr, mg/dl | |||
| At KT | 6.1 (4.6–7.9) | 7.1 (5.9–8.2) | 0.22 |
| 7 days after KT | 2.2 (1.5–5.4) | 4.3 (2.7–8.1) | 0.03 |
| CRR7 | 57.9 (31.6–72.7) | 36.2 (3.0–56.2) | 0.045 |
| ≤70% | 16 (69.6) | 28 (87.5) | 0.17 |
| eGFR (ml/min/1.73 mq) a | |||
| At KT | 12 (8–15) | 10 (9–13) | 0.27 |
| 7 days after KT | 26 (14–49) | 19 (10–32) | 0.051 |
| Delta value | 15 (4–34) | 7 (0–13) | 0.051 |
| Need for dialysis first week | 2 (8.7) | 11 (34.4) | 0.051 |
| LOS days | 11 (9–13) | 15 (11–24) | 0.01 |
Abbreviations: ADPKD, autosomal dominant polycystic disease; BMI, body mass index; CIT, cold ischemia time; CRR7, creatinine reduction ratio at day 7; CVA, cerebrovascular accident; DM2, diabetes mellitus type 2; eGFR, estimated glomerular filtration rate; GN, glomerulonephritis; ICU, intensive care unit; KDPI, kidney donor profile index; KDRI, kidney donor risk index; KT, kidney transplant; LOS, length of stay; RR, resistance; sCr, serum creatinine; VAS, vasoactive Score.
eGFR estimated using the EPI‐CKD formula.
Out of the 23 perfused grafts, 16 (69.6%), 5 (21.7%), and 2 (8.7%) presented a Karpinski Score of 4, 5, and 6, respectively. Out of the 32 non‐perfused grafts, 18 (56.3%), 5 (15.6%), and 9 (28.1%) presented a Karpinski Score of 4, 5, and 6, respectively.
As for the recipients' characteristics, median age and cold ischemia time were 61 versus 62 years (p = 0.44) and 623 versus 695 min (p = 0.046) in the perfused versus non perfused cases, respectively.
In the 23 perfused grafts, the median time between donor cross‐clamp and the start of HMP was 193 min (IQR = 157–285). As for the in‐house HMP, the median duration was 247 min (IQR = 125–330), and all kidneys were perfused for at least 120 min. At the start of perfusion, the median flow and RR were 46 ml/min (IQR = 30–56) and 0.57 mm Hg/ml/min (0.46–0.87), respectively. After 2 h, the same parameters were 87 ml/min (IQR = 65–110) and 0.28 mm Hg/ml/min (IQR = 0.23–0.36). All grafts were used for a single KT.
During the first week after KT, two (8.7%) versus 11 (34.4%) patients needed dialysis (p = 0.051), with 16 (69.6%) versus 28 (87.5%) patients having an impaired sCr reduction defined as a CRR7 ≤ 70% (p = 0.17) in the perfused versus non‐perfused cases. The median sCr at day 7 (2.2 vs. 4.3 mg/dl; p = 0.03) and CRR7 values (57.9% vs. 36.2%; p = 0.045) were observed in the perfused versus non‐perfused cases.
The median length of hospital stay was shorter in the perfused group (median: 11 vs. 15 days; p = 0.01).
After a median follow‐up of 43 months (IQR = 37–60), two (8.7%) versus nine (28.1%) grafts were lost in the perfused versus non‐perfused group. We reported one (4.3%) versus seven (21.9%) cases of patient death in the perfused versus non‐perfused group. Three‐year death‐censored graft survival was 91.3% versus 77.0% in the perfused versus non‐perfused group (p = 0.16). Three‐year patient survival was 95.7% versus 87.5% in the perfused versus non‐perfused group (p = 0.19).
3.1.1. RR thresholds for the prediction of DGF
We investigated five different donor‐, graft‐, and perfusion‐related aspects to identify the best predictors for DGF defined as CRR7 ≤ 70% (Table 2). Unfortunately, we could not perform the same analysis for DGF defined as the need for dialysis within the first week post‐KT due to the small number of reported cases (n = 2).
TABLE 2.
Prediction of CRR7 ≤ 70% and RR threshold values investigation
| Variables | AUC | SE | 95%CI | p | |
|---|---|---|---|---|---|
| Lower | Upper | ||||
| RR initial time | 0.83 | 0.09 | 0.67 | 1.00 | 0.02 |
| RR 120 min | 0.81 | 0.12 | 0.58 | 1.00 | 0.03 |
| Flow initial time | 0.26 | 0.11 | 0.05 | 0.47 | 0.09 |
| Karpinski Score | 0.71 | 0.11 | 0.49 | 0.92 | 0.14 |
| Donor sCr | 0.62 | 0.15 | 0.34 | 0.91 | 0.38 |
| Initial RR | Sensitivity | Specificity | DOR | ||
| 0.4 mm Hg/ml/min | 87.5 | 14.3 | 1.2 | ||
| 0.5 mm Hg/ml/min | 82.4 | 83.3 | 23.4 | ||
| 0.75 mm Hg/ml/min | 52.9 | 100.0 | ∞ | ||
| 1.0 mm Hg/ml/min | 29.4 | 100.0 | ∞ | ||
Abbreviations: AUC, area under the curve; CI, confidence intervals; DOR, diagnostic odds ratios; RR, renal resistances; sCr, serum creatinine; SE, standard error.
The initial RR (renal resistance at the beginning of HMP) showed the best diagnostic ability, with an AUC = 0.83 (p = 0.02). The diagnostic ability of RR substantially remained constant after 2 h of perfusion, with an AUC = 0.81 (p = 0.03). Flow during perfusion, donor age, and Karpinski Score all failed to predict post‐KT DGF.
Four thresholds for initial RR were investigated, respectively 0.4, 0.5, 0.75, and 1.0 mm Hg/ml/min, approximately corresponding to the fifteenth centile, the fourth, the seventh, and the ninth decile. The cut‐off value of 0.5 mm Hg/ml/min showed the highest sensitivity and specificity (82.4 and 83.3, respectively), with an excellent DOR of 23.4.
We thus divided our population into two groups: Low‐RR Group (initial resistances <0.5 mm Hg/ml/min; n = 8) and High‐RR Group (initial resistances ≥0.5 mm Hg/ml/min; n = 15).
Table 3 reports the donor‐, recipient‐, and perfusion‐specific characteristics of the 23 transplanted cases stratified into the two groups.
TABLE 3.
Donor‐, recipient‐, and perfusion‐related characteristics in the two groups of low and high RR
| Variables | Low‐RR (n = 8) | High‐RR (n = 15) | p‐value |
|---|---|---|---|
| Median (IQR) or n (%) | |||
| Donor‐specific variables | |||
| KDRI | 1.47 (1.30–2.00) | 1.68 (1.38–2.22) | 0.43 |
| KDPI | 85 (76–97) | 92 (81–99) | 0.51 |
| Total Karpinski Score | 4 (4–4) | 4 (4–5) | 0.27 |
| 4 | 7 (87.5) | 9 (60.0) | 0.35 |
| 5 | 1 (12.5) | 4 (26.7) | 0.62 |
| 6 | 0 (–) | 2 (13.3) | 0.53 |
| Age, years | 60 (56–78) | 67 (61–74) | 0.21 |
| Male, gender | 6 (75.0) | 6 (40.0) | 0.19 |
| Weight, kg | 83 (76–90) | 80 (73–90) | 0.47 |
| Height, cm | 179 (165–180) | 168 (164–177) | 0.21 |
| BMI | 28 (26–31) | 27 (26–31) | 1.00 |
| sCr, mg/dl | 1.2 (0.9–1.8) | 1.4 (0.8–1.4) | 0.68 |
| Sodium peak, mEq/L | 152 (150–161) | 150 (144–150) | 0.02 |
| ICU stay, days | 4 (2–7) | 5 (2–5) | 0.83 |
| VAS | 30 (11–61) | 5 (0–50) | 0.33 |
| Story of hypertension | 4 (50.0) | 8 (53.3) | 1.00 |
| Story of DM2 | 4 (50.0) | 3 (20.0) | 0.18 |
| Story of smoking | 4 (50.0) | 6 (40.0) | 0.69 |
| Story of cardiopathy | 1 (12.5) | 3 (20.0) | 1.00 |
| Story of hepatopathy | 2 (25.0) | 0 (–) | 0.11 |
| Story of dyslipidemia | 1 (12.5) | 3 (20.0) | 1.00 |
| Cause of death | |||
| Anoxia | 0 (–) | 1 (6.7) | |
| Blunt trauma | 1 (12.5) | 4 (26.7) | 0.52 |
| CVA | 7 (87.5) | 10 (66.7) | |
| Episode(s) of hypotension | 4 (50.0) | 2 (13.3) | 0.13 |
| Episode(s) of cardiac arrest | 2 (25.0) | 1 (6.7) | 0.27 |
| Recipient‐specific variables | |||
| Age, years | 58 (53–64) | 63 (57–69) | 0.21 |
| Male, gender | 5 (62.5) | 8 (53.3) | 1.00 |
| Weight, kg | 71 (56–77) | 71 (70–76) | 0.89 |
| Height, cm | 165 (161–165) | 165 (160–165) | 0.86 |
| BMI | 24 (20–28) | 24 (24–27) | 0.39 |
| Renal pathology | 0.13 | ||
| ADPKD | 2 (25.0) | 2 (13.3) | |
| Angiosclerosis | 0 (–) | 2 (13.3) | |
| DM2 | 1 (12.5) | 1 (6.7) | |
| GN | 2 (25.0) | 1 (6.7) | |
| Lupus | 2 (25.0) | 0 (–) | |
| Re‐KT | 1 (12.5) | 1 (6.7) | |
| Other | 0 (–) | 3 (20.0) | |
| Unknown | 0 (–) | 5 (33.3) | |
| Time of dialysis, years | 5 (3–8) | 5 (4–5) | 0.74 |
| Arterial hypertension | 7 (87.5) | 11 (73.3) | 0.62 |
| CIT, min | 375 (269–479) | 380 (300–405) | 1.00 |
| Perfusion‐specific variables | |||
| Perfusion, min | 183 (126–311) | 267 (156–365) | 0.20 |
| Flow, ml/min | |||
| 0 min | 53 (50–78) | 32 (17–53) | 0.008 |
| 20 min | 96 (81–124) | 53 (35–82) | 0.009 |
| 40 min | 95 (90–126) | 65 (50–97) | 0.03 |
| 60 min | 95 (89–127) | 71 (54–102) | 0.03 |
| 120 min | 98 (89–128) | 80 (60–92) | 0.03 |
| Delta flow (from 0 to 120), % | +61 (+14 to +142) | +140 (+96 to +231) | 0.02 |
| RR, mm Hg/ml/min | |||
| 0 min | 0.43 (0.31–0.48) | 0.76 (0.57–1.35) | <0.001 |
| 20 min | 0.27 (0.31–0.48) | 0.50 (0.32–0.73) | 0.005 |
| 40 min | 0.23 (0.20–0.26) | 0.43 (0.26–0.57) | 0.007 |
| 60 min | 0.23 (0.20–0.27) | 0.35 (0.25–0.43) | 0.01 |
| 120 min | 0.23 (0.19–0.28) | 0.33 (0.26–0.40) | 0.04 |
| Delta RR (from 0 to 120), % | −41 (−57 to −6) | −63 (−70 to −54) | 0.001 |
| sCr, mg/dl | |||
| At KT | 6.2 (5.5–7.6) | 6.1 (4.1–8.9) | 0.83 |
| 7 days after KT | 1.7 (1.5–3.4) | 3.4 (1.5–5.4) | 0.29 |
| CRR7 | 72 (52–78) | 46 (29–63) | 0.07 |
| ≤70% | 3 (13.3) | 13 (86.7) | 0.03 |
| eGFR (ml/min/1.73 mq) a | |||
| At KT | 13 (9–14) | 11 (7–18) | 0.83 |
| 7 days after KT | 45 (12–52) | 22 (14–39) | 0.68 |
| Delta value | 32 (5–40) | 11 (4–25) | 0.29 |
| Need for dialysis 1st week | 0 (–) | 2 (13.3) | 0.53 |
| LOS days | 11 (9–12) | 12 (9–15) | 0.27 |
Abbreviations: ADPKD, autosomal dominant polycystic disease; BMI, body mass index; CIT, cold ischemia time; CRR7, creatinine reduction ratio at day 7; CVA, cerebrovascular accident; DM2, diabetes mellitus type 2; eGFR, estimated glomerular filtration rate; GN, glomerulonephritis; ICU, intensive care unit; KDPI, kidney donor profile index; KDRI, kidney donor risk index; KT, kidney transplant; LOS, length of stay; RR, resistance; sCr, serum creatinine; VAS, vasoactive Score.
eGFR estimated using the EPI‐CKD formula.
No substantial differences in donor characteristics were reported between the two groups. The median CIT was slightly inferior in the Low‐RR Group (375 vs. 380 min; P = 1.00), while perfusion duration was markedly inferior, although not significant (183 vs. 267; p = 0.20). As previously reported, all grafts were perfused for at least 2 h. The longest perfusion time was 7.4 h.
All the variables related to HMP are reported in Table 3 and Figure 1. Figure 2 showed the RR trend of all the grafts undergoing HMP.
FIGURE 1.
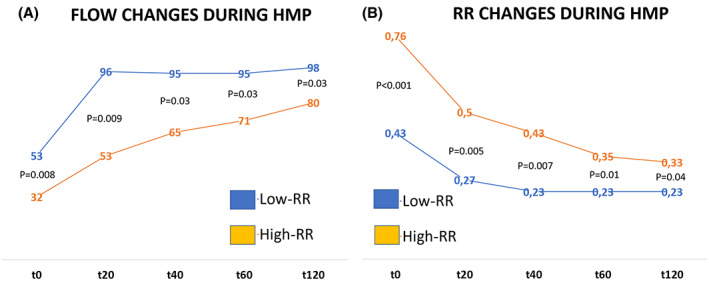
Median flow (A) and RR (B) changes during the HMP at different time points in the low‐RR and high‐RR groups. Y‐axis reports the median values of flow expressed in ml/min (A), and the median values of RR expressed in mm Hg/ml/min (B).
FIGURE 2.
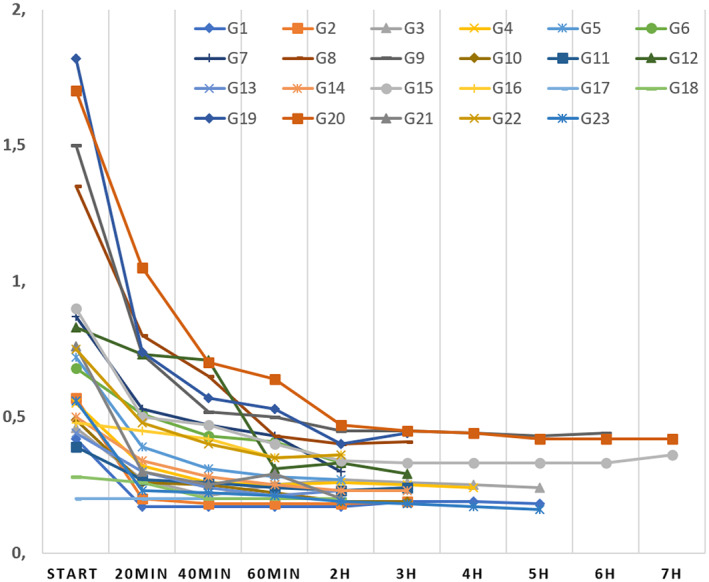
RR changes at different time points observed in the 23 patients transplanted. Y‐axis reports the values of RR expressed in mm Hg/ml/min.
Significant differences between the two groups were observed in terms of renal flows and RRs. By definition, at the start of HMP, grafts in the High‐RR group showed higher median RRs (0.76 vs. 0.43 mm Hg/ml/min; p < 0.001) and consequently lower flows (32 vs. 53 ml/min; p = 0.008). While both groups showed a decrease of RR with time and a corresponding increase in flow, significant differences between the two groups could still be observed after 2 h of perfusion, with High‐RR cases still presenting higher RRs (0.33 vs. 0.23 mm Hg/ml/min; p = 0.04) and lower flows (80 vs. 98 ml/min; p = 0.03).
After perfusion, all grafts were successfully transplanted. Concerning early recovery of renal function, although median sCr values 7 days after KT were markedly lower in the Low‐RR Group (1.7 vs. 3.4 mg/dl), this difference was not statistically significant (p = 0.29). According to the classical definition of DGF, namely the need for dialysis during the first week after transplant, no differences were observed between the groups, with 2/15 (13.3%) versus zero cases found in the High‐ versus Low‐RR Group, respectively (p = 0.53). However, when DGF was defined as a CRR7 ≤ 70%, the number of DGF cases in the High‐RR Group was statistically significantly superior (86.7 vs. 13.3%; p = 0.03).
Although not statistically significant, the median length of stay was shorter in the Low‐RR Group (11 vs. 12 days; p = 0.27).
Both grafts lost after transplant showed an initial RR >0.5 mm Hg/ml/min. Three‐year graft survival rates were 100.0 versus 86.7% in Low‐RR versus High‐RR Groups, respectively (p = 0.31) (Figure 3).
FIGURE 3.
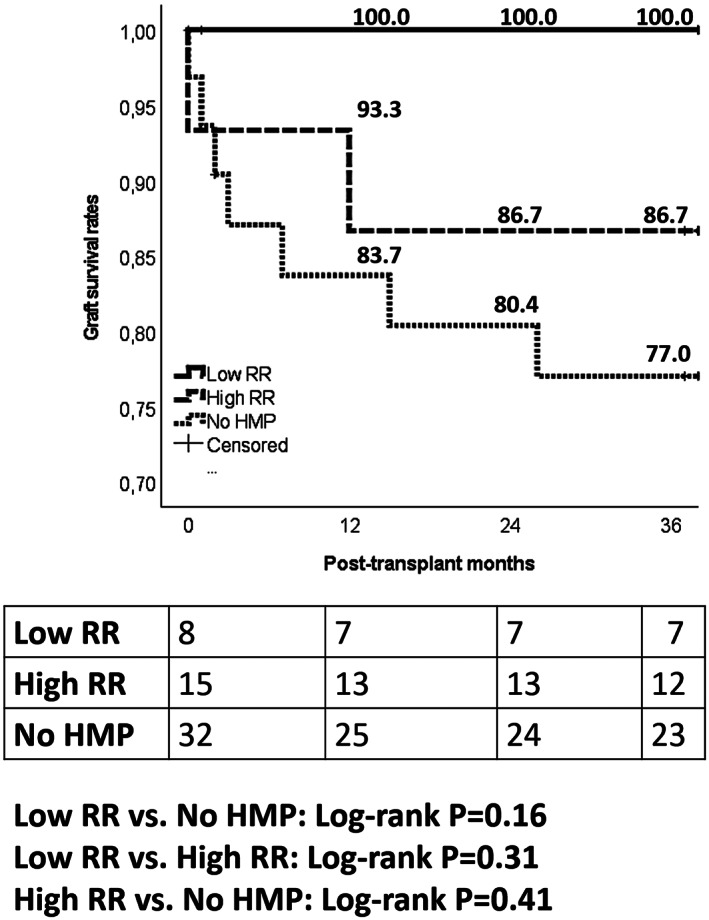
Three‐year death‐censored graft survival rates observed in the low‐RR perfused, high‐RR perfused, and non‐perfused groups.
4. DISCUSSION
In the present study, we observed that the use of in‐house HMP resulted in low rates of DGF and 3‐year graft loss (2/23; 8.7%), even when used in grafts with an impaired histological status (Karpinski >3). Compared with a group of non‐perfused grafts with Karpinski >3, the perfused grafts consented to obtain a better decline of sCr, a reduction in the number of cases needing dialysis, and shorter LOS.
Several previously published preclinical and clinical studies investigated the mechanisms underlying the positive effect of HMP. For example, a link between HMP and preservation of the endothelial lining was found. 14 Chatauret et al. demonstrated that non‐oxygenated machine perfusion preservation increased the phosphorylation of eNOS with an AMPK‐dependent pathway in the renal cortex at the end of the procedure and in the renal artery after re‐oxygenation. 15 HMP also showed effectiveness in reducing the ischemia/reperfusion injury by decreasing pro‐inflammatory cytokines expression and adhesion molecules such as ICAM‐1. 16 From the bench to the clinics, a recent Cochrane systematic review based on 16 studies (N = 2266) comparing HMP with cold storage showed an overall reduction of 22.0% for the risk of DGF in DBD donors (p = 0.006). 10
In our series, no statistical relevance was reported in 3‐year death‐censored graft survival, although better survivals were reported in the perfused group (91.3% vs. 77.0%). These results are probably connected with the small sample size of the investigated population.
Moreover, we highlighted the potential for using the additional information gathered during perfusion (such as RR) as a useful selection tool. As for the role of RR in the selection of the perfused grafts, evidence in the existing literature is weaker. This result may be mainly ascribed to potential selection biases in studies investigating this aspect, such as grafts being systematically discarded based on arbitrarily defined RR thresholds. 17 However, we should emphasize that, in our series, no organ was refused due to high RR values; organ discard before perfusion was based on the presence of microscopic or macroscopic adverse conditions (i.e., pyelonephritis, diffuse atherosclerosis).
A study from China based on 76 grafts from deceased‐cardiac donors showed that terminal RRs were an independent predictor of DGF (odds ratio [OR] = 3.12; p = 0.01) and graft survival (hazard ratio [HR] = 2.06; p = 0.03). 18 The same group even proposed a scoring model able to identify the risk of DGF based on the combination of HMP duration (OR = 1.17; p = 0.043), RR (OR = 2.19; p < 0.001), and flow rate (OR = 0.93; p = 0.01). 19
Sandal et al. identified two different terminal RR thresholds able to predict the risk for death‐censored graft failure, namely 0.2 (HR = 2.42; p = 0.04) and 0.4 (HR = 2.67; p = 0.07) mm Hg/ml/min. 12
An Italian study based on 35 KT performed using ECD showed that recipients of kidneys with RR ≤1.0 within 1 h of HMP had a lower PNF/DGF rate (11 vs. 44%; p = 0.03) and faster sCr decrease (post‐KT day 10: 1.79 mg/dl vs. 4.33 mg/dl; p = 0.02). 8
Likewise, our results showed a correlation between RR and DGF. Interestingly, while our study found RR at the beginning of perfusion to be most predictive of DGF, many previous studies reported terminal RR as most predictive. 8 , 12 , 18 , 19 In our series, initial RR showed an excellent AUC = 0.83 for the diagnosis of DGF, with the cut‐off = 0.5 mm Hg/ml/min presenting excellent sensitivity = 82.4 and specificity = 83.3.
More specifically, investigating this threshold, we observed that cases starting with a higher initial RR (>0.5 mm Hg/ml/min) maintained higher RR and lower flows even after 2 h of perfusion, although all of them showed a significant improvement in these latter values. These data suggest that the perfusion starting‐point status could be valuable additional information for determining the global graft quality. We can postulate that diffuse chronic histological alterations (such as interstitial fibrosis or atherosclerosis) that are poorly investigable in their entity through biopsy alone could be better accounted for using the initial value of RR. That continuous perfusion cannot minimize their negative effect in the posttransplant clinical course.
As for the postoperative course, initial RR was correlated with recovery of early graft function as defined by CRR7. Better results were observed in the Low‐RR Group (CRR7 ≤ 70%: 13.3 vs. 86.7%; p = 0.03). Although we also reported a trend for initial RR associated with a lower need for dialysis and shorter length of stay, a similar significant predictive value could not be demonstrated, possibly due to the limited number of patients in our study.
Our study's peculiarity is that we investigated the potential role of RR during in‐house HMP as a tool to select which grafts could be usable for a single KT in histologically impaired (Karpinski>3) DBD grafts.
The decision to use these organs for performing a single transplant overrules the Remuzzi Score recommendation of limiting the use of kidneys for a single KT to a Karpinski Score ≤3. 20 However, growing evidence suggests that these recommendations could be slightly modified under specific conditions. For example, an Italian study proposed modifying the decision protocol, considering the scores 4–5 as usable for a single KT if donor glomerular filtration rate was ≥60 ml/min 21
Another study comparing the results after a single KT selected on clinical grounds alone versus clinical‐histological protocol did not show any difference in graft survival and long‐term sCr values 22
In a study performed by Bissolati et al., in which the histological features were investigated in the context of reconditioned organs, Karpinski Score did not correlate with PNF/DGF rate (p = 0.87) and postoperative sCr trend (p = 0.80). 8 These data are in line with our findings, in which the postperfusion results were excellent, although non‐standard organs were used for a single KT
All these aspects allow for some speculation on the observed results. For example, initial RR may be used as a non‐invasive method for organ viability assessment, offering a valuable source of additional information compared to biopsy score alone. RR May have the potential to improve the evaluation of the quality of the entire cortex, whereas a biopsy is unfortunately only able to offer information on a small number of nephrons, tubules, and vessels. This limitation is well recognized in some studies, in which the risk of unnecessarily discarding organs due to renal biopsy results is underlined 22 , 23
Furthermore, HMP helps minimize the deterioration process in the perfused organ seen in cold storage, mainly by limiting ischemia–reperfusion injury, a well‐recognized risk factor for poor outcomes and increased immunological accidents. The use of HMP may thus expand the pool of transplantable kidneys with non‐standard organs, although without risk of jeopardizing the outcome. In addition, data on pretransplant HMP RR could be helpful to refine the posttransplant surveillance and tailoring the most appropriate immunosuppressive regimen. In support of this claim, a study from Poland showed that patients with initially low RR (<0.19 mm Hg/ml/min) presented two‐fold lower rates of acute rejection compared to patients transplanted with grafts with RR≥0.19 mm Hg/ml/min 24
All these speculations require further studies and more extensive series, with the intent to construct mathematical models aimed at “weighing” the valuable role of hypothermic reconditioning and the selective role of RR in combination with the other histological features
The present study has some limitations. As in many previous publications, this is a preliminary experience based on a small number of patients. Although we intended to identify a potential RR cut‐off with the discriminative ability for DGF, the population we studied to obtain our RR threshold of >0.5 was too small to warrant its clinical application. Indeed, the positive predictive value of the threshold was relatively low. Only two of 15 cases with a high initial RR developed DGF. We can only propose to use the RR values as part of a broader assessment of donor quality. Another limit is that this was a monocentric study. Therefore, the outcome could potentially be influenced by local prerogatives. Multicenter studies are needed with the intent to overcome the present limitations
In our series, we used basiliximab instead of thymoglobulin for induction therapy. This strategy derived from the major experience of our center in managing the anti‐CD25 monoclonal antibody in a setting in which we explored the efficacy of HMP in marginal grafts. In consideration of the potential benefits of thymoglobulin in this type of patient should appear (marginal kidneys with increased susceptibility to CNI toxicity), we are confident to implement the use of this drug in the future
Lastly, the retrospective nature of the analysis impaired our ability to obtain relevant pieces of information obtainable from the biopsies, like the degree of acute tubular necrosis. Prospective collection of data in well‐designed prospective studies is required for improving our knowledge on the correlation between graft histological damages and posttransplant course
5. CONCLUSIONS
Using in‐house HMP for preconditioning kidneys with a Karpinski score >3 resulted in reduced DGF and 3‐year graft loss rates compared with non‐perfused grafts. However, these results are not statistically relevant due to the small sample size reported, and a larger dataset is required for confirming the positive impact of HMP. Initial RR represents a helpful parameter for selecting non‐standard organs to be safely used for single KT, even in impaired histological findings. Initial RR has also been shown to hold a substantial predictive value for early recovery of renal function
AUTHOR CONTRIBUTIONS
Franco Ruberto, Mario Piazzolla, and Quirino Lai were responsible for the conception, design, analysis, and writing of the study; Franco Ruberto, Mario Piazzolla, Quirino Lai, Pietro Santopietro, Veronica Zullino, and Massimo Rossi were involved with the collection and interpretation of data; Renzo Pretagostini, Francesco Giovanardi, Francesco Nudo, Manuela Garofalo, Luca Poli, Francesco Pugliese, and Pasquale B. Berloco participated in data management, reviewing, and editing of the manuscript.
CONFLICT OF INTEREST
The authors have no conflicts of interest to declare about the present study.
Supporting information
Appendix S1
ACKNOWLEDGMENT
Open access funding enabled by Universita degli Studi di Roma La Sapienza.
Ruberto F, Lai Q, Piazzolla M, Brisciani M, Pretagostini R, Garofalo M, et al. The role of hypothermic machine perfusion in selecting renal grafts with advanced histological score. Artif. Organs. 2022;46:1771–1782. 10.1111/aor.14308
Correction added on 2st July 2022, after First online publication open access funding statement has been added.
REFERENCES
- 1. Rao PS, Schaubel DE, Guidinger MK, Andreoni KA, Wolfe RA, Merion RM, et al. A comprehensive risk quantification score for deceased donor kidneys: the kidney donor risk index. Transplantation. 2009;88:231–6. [DOI] [PubMed] [Google Scholar]
- 2. Pretagostini R, Lai Q, Poli L, Levi Sandri GB, Travaglia D, Rossi M, et al. Predictive characteristics of delayed graft function after expanded and standard criteria donor kidney transplantations. Transplant Proc. 2009;41:1149–51. [DOI] [PubMed] [Google Scholar]
- 3. Lai Q, Nudo F, Morabito V, et al. Donor quality scoring systems and early renal function measurements in kidney transplantation. In: Ortiz J, Andre J editors. Understanding the complexities of kidney transplantation. Intech Open; 2011. 10.5772/17845 [DOI] [Google Scholar]
- 4. Querard AH, Foucher Y, Combescure C, Dantan E, Larmet D, Lorent M, et al. Comparison of survival outcomes between expanded criteria donor and standard criteria donor kidney transplant recipients: a systematic review and meta‐analysis. Transpl Int. 2016;29:403–15. [DOI] [PubMed] [Google Scholar]
- 5. Karpinski J, Lajoie G, Cattran D, Fenton S, Zaltzman J, Cardella C, et al. Outcome of kidney transplantation from high‐risk donors is determined by both structure and function. Transplantation. 1999;67:1162–7. [DOI] [PubMed] [Google Scholar]
- 6. Remuzzi G, Cravedi P, Perna A, Dimitrov BD, Turturro M, Locatelli G, et al. Long‐term outcome of renal transplantation from older donors. N Engl J Med. 2006;354:343–52. [DOI] [PubMed] [Google Scholar]
- 7. Anglicheau D, Loupy A, Lefaucheur C, Pessione F, Létourneau I, Côté I, et al. A simple clinico‐histopathological composite scoring system is highly predictive of graft outcomes in marginal donors. Am J Transplant. 2008;8:2325–34. [DOI] [PubMed] [Google Scholar]
- 8. Bissolati M, Gazzetta PG, Caldara R, Guarneri G, Adamenko O, Giannone F, et al. Renal resistance trend during hypothermic machine perfusion is more predictive of postoperative outcome than biopsy score: preliminary experience in 35 consecutive kidney transplantations. Artif Organs. 2018;42:714–22. [DOI] [PubMed] [Google Scholar]
- 9. Jochmans I, Moers C, Smits JM, Leuvenink HGD, Treckmann J, Paul A, et al. The prognostic value of renal resistance during hypothermic machine perfusion of deceased donor kidneys. Am J Transplant. 2011;11:2214–20. [DOI] [PubMed] [Google Scholar]
- 10. Tingle SJ, Figueiredo RS, Moir JA, Goodfellow M, Talbot D, Wilson CH. Machine perfusion preservation versus static cold storage for deceased donor kidney transplantation. Cochrane Database Syst Rev. 2019;3:CD011671. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11. Patel K, Nath J, Hodson J, Inston N, Ready A. Outcomes of donation after circulatory death kidneys undergoing hypothermic machine perfusion following static cold storage: a UKpopulation‐based cohort study. Am J Transplant. 2018;18:1408–14. [DOI] [PubMed] [Google Scholar]
- 12. Sandal S, Paraskevas S, Cantarovich M, Baran D, Chaudhury P, Tchervenkov JI, et al. Renal resistance thresholds during hypothermic machine perfusion and transplantation outcomes ‐ a retrospective cohort study. Transpl Int. 2018;31:658–69. [DOI] [PubMed] [Google Scholar]
- 13. Johnston O, Okelly P, Spencer S, Donohoe J, Walshe JJ, Little DM, et al. Reduced graft function (with or without dialysis) vs immediate graft function—a comparison of long‐term renal allograft survival. Nephrol Dial Transplant. 2006;21:2270–4. [DOI] [PubMed] [Google Scholar]
- 14. De Deken J, Kocabayoglu P, Moers C. Hypothermic machine perfusion in kidney transplantation. Curr Opin Organ Transplant. 2016;21:294–300. [DOI] [PubMed] [Google Scholar]
- 15. Chatauret N, Coudroy R, Delpech PO, Vandebrouck C, Hosni S, Scepi M, et al. Mechanistic analysis of non‐oxygenated hypothermic machine perfusion's protection on warm ischemic kidney uncovers greater eNOS phosphorylation and vasodilation. Am J Transplant. 2014;14:2500–14. [DOI] [PubMed] [Google Scholar]
- 16. Tozzi M, Franchin M, Soldini G, Ietto G, Chiappa C, Maritan E, et al. Impact of static cold storage VS hypothermic machine preservation on ischemic kidney graft: inflammatory cytokines and adhesion molecules as markers of ischemia/reperfusion tissue damage. Our preliminary results. Int J Surg. 2013;11:S110–4. [DOI] [PubMed] [Google Scholar]
- 17. Jochmans I, O'Callaghan JM, Pirenne J, Ploeg RJ. Hypothermic machine perfusion of kidneys retrieved from standard and high‐risk donors. Transpl Int. 2015;28:665–76. [DOI] [PubMed] [Google Scholar]
- 18. Ding CG, Tian PX, Ding XM, Xiang HL, Li Y, Tian XH, et al. Beneficial effect of moderately increasing hypothermic machine perfusion pressure on donor after cardiac death renal transplantation. Chin Med J (Engl). 2018;131:2676–82. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19. Ding CG, Li Y, Tian XH, Hu XJ, Tian PX, Ding XM, et al. Predictive score model for delayed graft function based on hypothermic machine perfusion variables in kidney transplantation. Chin Med J (Engl). 2018;131:2651–7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20. Remuzzi G, GrinyòJ RP, et al. Early experience with dual kidney transplantation in adults using expanded donor criteria. Double Kidney Transplant Group (DKG). J Am Soc Nephrol. 1999;10:2591–8. [DOI] [PubMed] [Google Scholar]
- 21. Carta P, Zanazzi M, Caroti L, Buti E, Mjeshtri A, di Maria L, et al. Impact of the pre‐transplant histological score on 3‐year graft outcomes of kidneys from marginal donors: a single‐Centre study. Nephrol Dial Transplant. 2013;28:2637–44. [DOI] [PubMed] [Google Scholar]
- 22. Casati C, Colombo VG, Perrino M, Rossetti OM, Querques M, Giacomoni A, et al. Renal transplants from older deceased donors: use of preimplantation biopsy and differential allocation to dual or single kidney transplant according to histological score has no advantages over allocation to single kidney transplant by simple clinical indication. J Transplant. 2018;2018:4141756. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23. Kasiske BL, Stewart DE, Bista BR, Salkowski N, Snyder JJ, Israni AK, et al. The role of procurement biopsies in acceptance decisions for kidneys retrieved for transplant. Clin J Am Soc Nephrol. 2014;9:562–71. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24. Wszolaa M, Domagalab P, Serwanska‐Swieteka M, et al. Should immunosuppression after kidney transplant be adjusted based on renal resistance during pretransplant hypothermic machine perfusion? Transpl Proc. 2019;51:2676–82. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Appendix S1


