Abstract
Objectives
The novel concept of subjective cognitive decline (SCD) in Parkinson's disease (PD) refers to subjective cognitive impairment without concurrent objective cognitive deficits. This study aimed to determine the prevalence and affective correlates of SCD in de novo PD patients.
Materials and Methods
A total of 139 de novo PD patients underwent comprehensive neuropsychological evaluation. PD patients with SCD (PD‐SCD) did not meet the diagnostic criteria for mild cognitive impairment in PD (PD‐MCI) based on the Movement Disorder Society Level II Criteria and were defined by a Domain‐5 Score ≥1 on the Non‐Motor Symptoms Questionnaire. Affective symptoms were measured using the Hamilton Depression Scale (HAMD) and Hamilton Anxiety Scale (HAMA).
Results
In de novo PD cohort, the prevalence of SCD was 28.1%. PD‐SCD patients performed significantly better than PD‐MCI patients on tests of five cognitive domains. The more commonly affected domains in PD‐SCD patients were memory (28.2%) and attention/working memory (25.6%). Multivariable linear regression analysis revealed that PD‐SCD was significantly associated with both HAMD (β = 4.518, 95% CI = 0.754–8.281, p = .019) and HAMA scores (β = 4.259, 95% CI = 1.054–7.464, p = .010). Furthermore, binary logistic regression analysis revealed that higher HAMD (OR = 1.128, 95% CI = 1.019–1.249, p = .020) and HAMA scores (OR = 1.176, 95% CI = 1.030–1.343, p = .017) increased the risk of PD‐SCD.
Conclusions
Our findings suggest that SCD is highly prevalent in de novo PD patients. The presence of PD‐SCD is suggestive of an underlying affective disorder.
Keywords: anxiety, de novo Parkinson's disease, depression, prevalence, subjective cognitive decline
1. INTRODUCTION
Parkinson's disease (PD) is the second most common chronic neurodegenerative disease after Alzheimer's disease (AD). 1 In addition to its defining motor symptoms, PD is characterized by a diverse spectrum of non‐motor symptoms (NMSs). Cognitive impairment is one of the most common NMSs, with PD patients at an approximately sixfold increased risk of cognitive impairment compared with healthy individuals. 2 Similar to AD, 3 the spectrum of cognitive impairment in PD is divided into three stages: an initial, high‐risk state of subjective cognitive decline (SCD), followed by mild cognitive impairment (MCI), and finally progression to dementia. 4 , 5 Both dementia and MCI can severely impact quality of life of PD patients and lead to significant economic consequences. 6 , 7 Therefore, SCD in PD patients (PD‐SCD) has received increasing attention to support earlier detection and management of cognitive decline.
According to recent reviews, 4 , 5 the novel concept of PD‐SCD refers to self‐reported cognitive decline compared with one's previous cognitive status, and normal performance on neuropsychological tests after controlling for age, gender, and education. This suggests that PD‐SCD is purely subjective cognitive impairment without concurrent objective cognitive deficits, ending the long‐standing confusion about the definition of PD‐SCD. Given that most previous studies allow for concurrent objective impairment, 8 , 9 , 10 the prevalence and affective correlates of SCD remain unclear. To date, only a few studies have estimated the prevalence of PD‐SCD, with results ranging from 15.4% to 27.2%. 11 , 12 , 13 Furthermore, although several studies have reported that PD‐SCD is associated with severe anxiety/depression symptoms, 8 , 13 , 14 , 15 this relationship is debated due to inconsistent reports. 16 , 17
To the best of our knowledge, SCD prevalence and the association between the presence of SCD and anxiety/depressive symptoms have not been previously investigated in patients with de novo PD. To address these gaps, we examined the prevalence of PD‐SCD and the bidirectional relationship between PD‐SCD and affective (anxiety and depressive) symptoms in a cohort of de novo PD patients with SCD defined according to this novel concept.
2. MATERIALS AND METHODS
2.1. Participants
A total of 139 de novo PD patients were recruited from the Affiliated Brain Hospital of Nanjing Medical University between 2018 and 2022. The inclusion criteria were as follows: newly diagnosed with PD according to the International Parkinson and Movement Disorder Society (MDS) clinical diagnostic criteria 18 ; no antiparkinsonian drug treatment; in the early‐ or middle‐stage of PD (modified Hoehn and Yahr [H‐Y] stage ≤3); and successfully completed all clinical data evaluation. Exclusion criteria were a history of neurological diseases, psychiatric disorders, or serious medical conditions such as active malignancy and end‐organ failure. All de novo PD patients enrolled were followed up for at least 1 year to confirm the diagnosis of PD based on the disease evolution and response to dopaminergic replacement therapy.
This study was approved by the Medical Ethics Committee of the Affiliated Brain Hospital of Nanjing Medical University (2015‐KY030 and 2019‐KY019‐01) and conducted in accordance with the Declaration of Helsinki. All participants provided written informed consent before participating in the study.
2.2. Clinical evaluation
Demographic and clinical data were collected for the de novo PD patients. The demographic information included age, sex, formal education, age at PD onset, and disease duration. Motor evaluation included the Unified Parkinson's Disease Rating Scale (UPDRS) part II, UPDRS part III, and modified H‐Y stage. Based on Jankovic's method, 19 PD patients were divided into tremor‐dominant (TD), indeterminate, and postural instability and gait difficulty (PIGD) subtypes. NMSs were comprehensively assessed using the Non‐Motor Symptoms Questionnaire (NMSQuest), and NMSQuest domain‐5 contains two questions to assess memory and attention. 20 , 21 Sleep problems were assessed using the Parkinson’s Disease Sleep Scale (PDSS). Affect was evaluated using the Hamilton Depression Scale (HAMD) and Hamilton Anxiety Scale (HAMA).
Global cognition was measured using the Mini‐Mental State Examination (MMSE) and Montreal Cognitive Assessment Scale (MoCA). To adjust for education, a patient's MoCA score (if <30) was increased by 1 point if they had ≤12 years of education. 22 Various neuropsychological tests were administered to assess the five cognitive domains: the Digit Span Test (DST), Trail Making Test A (TMT‐A), and Stroop Color‐Word Test (SCWT) to assess attention/working memory; the Trail Making Test B (TMT‐B), Clock Drawing Test (CDT), and Animal Fluency Test (AFT) to assess executive function; the Wechsler Adult Intelligence Scale III (WAIS‐III) Similarities Test and Boston Naming Test (BNT) to assess language; the Auditory Verbal Learning Test (AVLT) and Logical Memory Test (LMT) to assess memory; and Benton's Judgment of Line Orientation Test (JLOT) and the Hooper Visual Organization Test (HVOT) to assess visuospatial function.
2.3. Cognitive classifications
PD patients were further categorized as PD with normal cognition (PD‐NC), PD‐SCD, and PD with mild cognitive impairment (PD‐MCI). PD‐MCI was defined by the MDS Level II diagnostic criteria 23 in which scores for at least two neuropsychological tests, whether in the same cognitive domain or different cognitive domains, are 1.5 standard deviations (SDs) below the average correcting for age, sex, and education. PD patients who did not meet the diagnostic criteria for PD‐MCI were categorized as PD‐SCD or PD‐NC according to NMSQuest Domain‐5. PD‐SCD was defined as an NMSQuest Domain‐5 score ≥1, similar to previous studies. 8 , 14 , 24
2.4. Statistical analysis
Demographic and clinical characteristics were reported as frequency (percent) for categorical variables and the median ± SD for continuous variables. The Shapiro–Wilk test and Kolmogorov–Smirnov test were used to assess the normality of the data. Continuous variables were compared between the PD‐NC, PD‐SCD, and PD‐MCI groups using one‐way analysis of variance (ANOVA) or the Kruskal–Wallis H‐test depending on the normality of the data, while categorical variables were compared using the Chi‐square test, followed by post hoc analysis with Bonferroni correction. Multivariable linear regression analysis and binary logistic regression analysis were performed to investigate any bidirectional relationship between the presence of PD‐SCD and affective symptoms, controlling for age, sex, and disease duration as potential confounders. Two‐tailed p‐values < .05 were considered statistically significant. All statistical analyses were performed using SPSS version 25.0 (SPSS, Inc.).
3. RESULTS
3.1. Demographic and clinical features
The demographic and clinical characteristics of the 139 de novo PD patients are summarized in Table 1. The mean age was 56.5 years and the mean disease duration was 1.9 years. The stratification resulted in 27 patients (19.4%) in the PD‐NC group, 39 patients (28.1%) in the PD‐SCD group, and 73 patients (52.5%) in the PD‐MCI group. There were no significant differences between the three groups in demographic, motor characteristics or motor subtypes, other than UPDRS part III. The post hoc analysis indicated that PD‐MCI patients had more severe UPDRS motor scores than PD‐NC patients. Furthermore, there were significant differences between the three groups in terms of NMSs. Compared with PD‐NC patients, PDSS and HAMA scores were more severe in PD‐MCI patients, while NMSQuest, HAMD, and HAMA scores were more severe in PD‐SCD patients.
TABLE 1.
Demographic and clinical characteristics of the PD‐NC, PD‐SCD, and PD‐MCI groups
| Variables | Total(n = 139, 100%) | PD‐NC(n = 27, 19.4%) | PD‐SCD(n = 39, 28.1%) | PD‐MCI(n = 73, 52.5%) | p | Post hoc |
|---|---|---|---|---|---|---|
| Age (years) | 56.5 ± 7.9 | 54.1 ± 7.5 | 55.9 ± 8.8 | 57.6 ± 7.4 | .134 | |
| Sex, Male (%) | 70 (50.4) | 17 (63.0) | 22 (56.4) | 31 (42.5) | .128 | |
| Formal education (years) | 10.5 ± 3.4 | 11.3 ± 4.0 | 11.2 ± 3.1 | 9.9 ± 3.3 | .076 | |
| Age at onset (years) | 54.6 ± 8.1 | 52.4 ± 7.3 | 53.9 ± 9.3 | 55.8 ± 7.5 | .133 | |
| Disease duration (years) | 1.9 ± 1.5 | 1.8 ± 1.3 | 2.4 ± 1.9 | 1.8 ± 1.3 | .119 | |
| UPDRS part II | 6.9 ± 3.5 | 6.0 ± 3.3 | 7.5 ± 3.6 | 6.9 ± 3.6 | .229 | |
| UPDRS part III | 19.7 ± 9.5 | 16.5 ± 9.5 | 18.3 ± 8.3 | 21.7 ± 9.8 | .028 | 0.044 a |
| H‐Y stage | 1.6 ± 0.5 | 1.5 ± 0.4 | 1.6 ± 0.5 | 1.6 ± 0.5 | .579 | |
| Motor subtype | ||||||
| TD (%) | 31 (22.3) | 4 (14.8) | 11 (28.2) | 16 (21.9) | .580 | |
| PIGD (%) | 22 (15.8) | 20 (74.1) | 23 (59.0) | 43 (58.9) | ||
| Indeterminate (%) | 86 (61.9) | 3 (11.1) | 5 (12.8) | 14 (19.2) | ||
| NMSQuest | 7.4 ± 4.3 | 5.7 ± 2.6 | 8.6 ± 4.5 | 7.5 ± 4.6 | .026 | 0.020 b |
| PDSS | 126.9 ± 21.1 | 134.3 ± 20.9 | 124.0 ± 24.4 | 125.7 ± 18.8 | .021 | 0.018 a |
| HAMD | 9.4 ± 6.8 | 6.2 ± 3.8 | 11.3 ± 9.0 | 9.5 ± 6.0 | .028 | 0.037 b |
| HAMA | 6.6 ± 5.5 | 4.0 ± 2.8 | 8.4 ± 7.8 | 6.7 ± 4.4 | .022 | 0.031 a , 0.040 b |
Note: Data are shown as the mean ± SD or n (%). p‐values were calculated using analysis of variance, Kruskal–Wallis H‐test, or Chi‐square test followed by post hoc analysis with Bonferroni adjustment. Statistically significant values (p < .05) are presented in bold.
Abbreviations: HAMA, Hamilton anxiety scale; HAMD, Hamilton depression scale; H‐Y, Hoehn and Yahr; NMSQuest, non‐motor symptoms questionnaire; PD, Parkinson's disease; PD‐MCI, Parkinson's disease with mild cognitive impairment; PD‐NC, Parkinson's disease with normal cognition; PD‐SCD, Parkinson's disease with subjective cognitive decline; PDSS, Parkinson’s disease sleep scale; PIGD, postural instability and gait difficulty; TD, tremor‐dominant; UPDRS, unified Parkinson's disease rating scale.
Statistically significant difference between the PD‐NC and PD‐MCI groups.
Statistically significant difference between the PD‐NC and PD‐SCD groups.
3.2. Cognitive performance
Table 2 compares the global cognition and comprehensive neuropsychological test scores between the PD‐NC, PD‐SCD, and PD‐MCI groups. Statistically significant differences between the three groups were observed for all measures except SCWT‐A‐right and SCWT‐C‐right. Post hoc analysis revealed that PD‐MCI patients performed significantly worse than PD‐NC patients on global cognition, attention/working memory (except SCWT‐right), executive function, language, memory (except AVLT‐recognition), and visuospatial function. In addition, PD‐SCD patients performed significantly better than PD‐MCI patients on global cognition (except MMSE), attention/working memory (except SCWT‐right), executive function, language, memory, and visuospatial function.
TABLE 2.
Cognitive performance among the PD‐NC, PD‐SCD, and PD‐MCI groups
| Variables | Total(n = 139, 100%) | PD‐NC(n = 27, 19.4%) | PD‐SCD(n = 39, 28.1%) | PD‐MCI(n = 73, 52.5%) | p | Post hoc |
|---|---|---|---|---|---|---|
| Global cognition | ||||||
| MMSE | 27.7 ± 2.3 | 28.2 ± 3.1 | 28.2 ± 1.4 | 27.3 ± 2.3 | .005 | 0.004 a |
| MoCA | 24.1 ± 3.4 | 25.7 ± 2.7 | 25.7 ± 3.0 | 22.8 ± 3.3 | <.001 | <0.001 a , <0.001 b |
| Attention/working memory | ||||||
| DST | 12.0 ± 2.3 | 12.7 ± 2.2 | 12.4 ± 1.9 | 11.4 ± 2.5 | .011 | 0.047 a , 0.043 b |
| TMT‐A (second) | 90.9 ± 46.7 | 68.4 ± 24.0 | 71.9 ± 24.0 | 109.3 ± 54.1 | <.001 | <0.001 a , <0.001 b |
| SCWT‐A‐time (second) | 28.1 ± 9.4 | 24.2 ± 4.6 | 25.5 ± 7.0 | 30.9 ± 10.9 | .001 | 0.006 a , 0.006 b |
| SCWT‐B‐time (second) | 40.2 ± 14.7 | 34.0 ± 9.1 | 35.1 ± 8.7 | 45.3 ± 17.0 | <.001 | 0.001 a , 0.001 b |
| SCWT‐C‐time (second) | 74.3 ± 28.1 | 65.8 ± 20.4 | 65.5 ± 18.3 | 82.2 ± 32.5 | <.001 | 0.007 a , 0.001 b |
| SCWT‐A‐right | 50.0 ± 0.9 | 50.0 ± 0.0 | 49.9 ± 0.3 | 50.0 ± 1.2 | .379 | |
| SCWT‐B‐right | 49.2 ± 2.0 | 49.8 ± 0.4 | 49.5 ± 1.3 | 48.7 ± 2.5 | .021 | |
| SCWT‐C‐right | 47.6 ± 4.1 | 48.5 ± 2.4 | 48.2 ± 3.4 | 47.0 ± 4.8 | .150 | |
| Executive function | ||||||
| TMT‐B (second) | 189.1 ± 99.8 | 137.8 ± 39.4 | 152.6 ± 30.9 | 227.5 ± 121.7 | <.001 | <0.001 a , <0.001 b |
| CDT | 8.7 ± 2.2 | 9.9 ± 0.4 | 9.6 ± 1.1 | 7.8 ± 2.6 | <.001 | <0.001 a , <0.001 b |
| AFT | 18.4 ± 5.3 | 20.7 ± 4.9 | 20.4 ± 3.8 | 16.5 ± 5.5 | <.001 | <0.001 a , <0.001 b |
| Language | ||||||
| Similarities | 16.3 ± 4.7 | 18.1 ± 4.6 | 18.1 ± 4.0 | 14.7 ± 4.5 | <.001 | 0.002 a , 0.001 b |
| BNT | 23.7 ± 3.9 | 25.5 ± 2.7 | 25.5 ± 2.4 | 22.1 ± 4.3 | <.001 | <0.001 a , <0.001 b |
| Memory | ||||||
| AVLT‐delayed recall | 5.2 ± 2.5 | 6.3 ± 2.2 | 6.4 ± 1.9 | 4.2 ± 2.4 | <.001 | <0.001 a , <0.001 b |
| AVLT‐recognition | 21.6 ± 2.7 | 22.0 ± 1.8 | 22.4 ± 1.6 | 21.0 ± 3.3 | .014 | 0.015 b |
| LMT‐delayed recall | 5.6 ± 2.6 | 6.8 ± 2.7 | 6.4 ± 2.4 | 4.7 ± 2.4 | <.001 | <0.001 a , 0.002 b |
| Visuospatial function | ||||||
| JLOT | 24.2 ± 3.6 | 26.1 ± 2.9 | 25.4 ± 2.3 | 22.8 ± 3.8 | <.001 | <0.001 a , <0.001 b |
| HVOT | 15.1 ± 4.8 | 18.2 ± 4.5 | 17.6 ± 3.4 | 12.6 ± 4.1 | <.001 | <0.001 a , <0.001 b |
Note: Data are shown as the mean ± SD. p‐values were calculated using analysis of variance, Kruskal–Wallis H‐test, or Chi‐square test followed by post hoc analysis with Bonferroni adjustment. Statistically significant values (p < .05) are indicated in bold.
Abbreviations: AFT, animal fluence test; AVLT, auditory verbal learning test; BNT, boston naming test; CDT, clock drawing test; DST, digit span test; HVOT, hooper visual organization test; JLOT, benton's judgment of line orientation test; LMT, logical memory test; MMSE, mini‐mental state examination; MoCA, montreal cognitive assessment; PD, Parkinson's disease; PD‐MCI, Parkinson's disease with mild cognitive impairment; PD‐NC, Parkinson's disease with normal cognition; PD‐SCD, Parkinson's disease with subjective cognitive decline; SCWT, stroop color‐word test; TMT‐A, trail making test A; TMT‐B, trail making test B;.
Statistically significant difference between the PD‐NC and PD‐MCI groups.
Statistically significant difference between the PD‐SCD and PD‐MCI groups.
Subsequently, we compared the frequency of cognitive domain deficits in the three groups (Figure 1). The cognitive domains commonly impaired in the PD‐SCD group were memory (28.2%) and attention/working memory (25.6%), whereas the domains commonly impaired in the PD‐MCI group were attention/working memory (74.0%) and executive function (72.6%). The frequencies of deficits in the five cognitive domains were significantly different among the three groups. Post hoc analysis indicated that PD‐MCI patients had a higher frequency of impairment in all domains except language compared with PD‐NC and PD‐SCD patients (p < .001).
FIGURE 1.
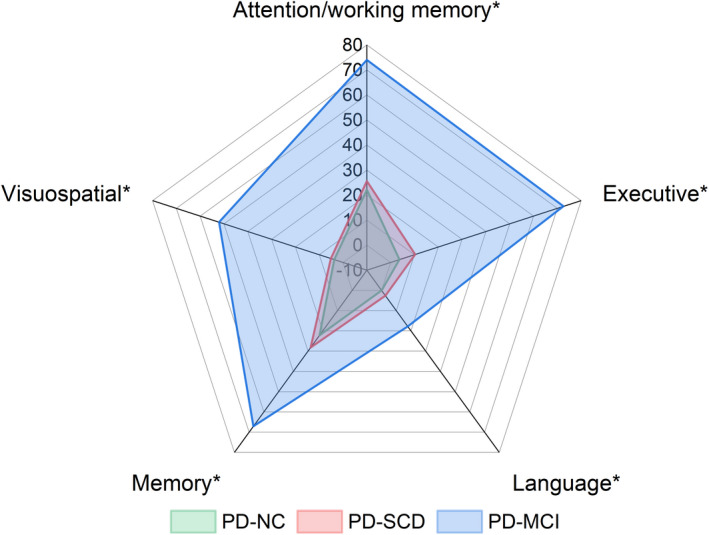
Frequency of cognitive domain deficits among the PD‐NC, PD‐SCD, and PD‐MCI groups. Group comparisons were performed using the Chi‐square test, followed by post hoc analysis with Bonferroni correction. *p < 0.05. PD, Parkinson's disease; PD‐NC, Parkinson's disease with normal cognition; PD‐SCD, Parkinson's disease with subjective cognitive decline; PD‐MCI, Parkinson's disease with mild cognitive impairment
3.3. Association between PD‐SCD presence and affective symptoms
We explored whether PD‐SCD presence was bidirectionally related to affective symptoms while controlling for age, sex, and disease duration (Table 3). The results of the multivariable linear regression analysis indicated that SCD was significantly associated with worse HAMD scores (β = 4.518, 95% CI = 0.754–8.281, p = .019) and HAMA scores (β = 4.259, 95% CI = 1.054–7.464, p = .010). According to the binary logistic regression analysis, higher HAMD scores (OR = 1.128, 95% CI = 1.019–1.249, p = .020) and HAMA scores (OR = 1.176, 95% CI = 1.030–1.343, p = .017) increased the risk of PD‐SCD.
TABLE 3.
Bidirectional association between PD‐SCD presence and affective symptoms
| β (95% CI) a | p | OR (95% CI) b | p | |
|---|---|---|---|---|
| HAMD | 4.518 (0.754–8.281) | .019 | 1.128 (1.019–1.249) | .020 |
| HAMA | 4.259 (1.054–7.464) | .010 | 1.176 (1.030–1.343) | .017 |
Abbreviations: CI, confidence interval; HAMA, Hamilton anxiety scale; HAMD, Hamilton depression scale; OR, odds ratio; PD, Parkinson's disease; PD‐NC, Parkinson's disease with normal cognition; PD‐SCD, Parkinson's disease with subjective cognitive decline.
Multivariable linear regression analysis was adjusted for age, sex, and disease duration.
Binary logistic regression analysis was adjusted for age, sex, and disease duration.
4. DISCUSSION
To our knowledge, this is the first study to explore the prevalence and affective (depression and anxiety) correlates of SCD in patients with de novo PD, utilizing a novel concept to define SCD. Notably, we found that SCD is common (28.1%) in de novo PD patients. The PD‐SCD patients generally performed better than PD‐MCI patients on tests of five cognitive domains and were frequently impaired in memory and attention/working memory domains. Importantly, SCD was bidirectionally associated with higher anxiety and depression scores. Based on these findings, the presence of SCD in de novo PD patients suggests an underlying affective disorder.
The presence of PD‐SCD in an overall intact PD patient may denote a stage of cognitive decline between PD‐NC and PD‐MCI, as has been confirmed in the ordinary population. 25 , 26 In preclinical AD patients, SCD has been shown to increase the risk of transition to dementia. 27 , 28 Similarly, the presence of PD‐SCD was highlighted as a risk factor for PD‐MCI in recent longitudinal follow‐up studies. 29 , 30 Thus, PD‐SCD is a promising concept with the potential to enable early diagnosis of impending cognitive impairment.
The prevalence of MCI in this study was 52.5%, which is higher than the reported 40% prevalence in a recent meta‐analysis. 31 This discrepancy may be primarily due to our use of a schedule of 12 cognitive evaluations spanning five cognitive domains. In addition, PD‐MCI was associated with more severe motor symptoms. 31 The PD patients enrolled in this study were not taking antiparkinsonian drugs and motor symptoms may affect cognitive assessments, especially assessment of executive function, leading to an increased prevalence of MCI.
The prevalence of SCD was 28.1% in our de novo PD cohort, which is slightly higher than previous findings. 11 , 12 , 13 This may be due to heterogeneity in assessing PD‐SCD questions. This study used a more sensitive test, namely the NMSQuest Domain‐5, to assess memory and attention function, whereas most previous studies assessed memory complaints using a single question of the NMSQuest, 24 the first item of the UPDRS, 12 or questionnaires such as the Cognitive Complaints Interview (CCI), 10 Memory Assessment Clinics Questionnaire (MAC‐Q), 13 and Parkinson's Disease Cognitive Questionnaire (PD‐CQ). 32 While some studies have used questionnaires encompassing the five cognitive domains to determine the presence of PD‐SCD, 16 , 33 , 34 these instruments require further validation. The PD‐SCD group also had higher NMSQuest scores than the PD‐NC group, which was mainly attributed to the fact that the PD‐SCD and PD‐NC groups were grouped based on NMSQuest Domain‐5.
In terms of cognitive performance, PD‐SCD patients outperformed PD‐MCI patients in the five cognitive domains. As the PD‐SCD patients in this study did not have objective cognitive impairment, it is not difficult to understand why PD‐SCD patients had better cognitive performance than PD‐MCI patients. Additionally, we found that the cognitive domains commonly affected in the PD‐SCD patients were memory and attention/working memory, which is consistent with the results of a previous study. 13 As the concept of SCD is based on research in AD patients as a generic description of early experienced cognitive deficits, 3 , 35 and memory is the main cognitive domain impaired in AD, concerns about memory impairment may have biased studies of PD‐SCD to some extent. In view of the range of cognitive deficits in non‐demented PD patients, 36 , 37 it is important to place a broad emphasis on cognitive complaints rather than memory complaints specifically in early PD. Previous studies have demonstrated that changes in the attention/working memory domain already exist in the prodromal stage of PD, 38 and that attention/working memory is the most commonly affected domain in de novo PD patients. 39 Therefore, as research on PD‐SCD continues to progress, it should become more standardized and focus on PD‐specific cognitive deficits such as attention/working memory function.
The presence of SCD in the newly diagnosed PD patients in this study was associated with higher depression and anxiety scores and showed a bidirectional relationship, which is similar to previous reports. 8 , 13 , 14 , 15 As some studies have found no relationship between SCD and mood disorders, 16 , 17 and have even proposed a link between objective cognitive impairment and affective symptoms, 15 , 40 this link remains controversial. This discrepancy may arise because PD‐SCD samples are highly heterogeneous. With an established definition of PD‐SCD, samples will become more homogeneous and the relationship between SCD and mood can be studied more thoroughly. Furthermore, although we describe a cross‐sectional relationship between SCD and mood disorders, our findings suggest that PD‐SCD is suggestive of mood disorders. As depression and anxiety are well‐established risk factors for MCI in early PD, 41 further studies are needed to determine whether PD‐SCD patients with affective symptoms have subthreshold cognitive decline and an increased risk of subsequently developing PD‐MCI.
The strength of this study lies in the use of a new definition of SCD to examine its prevalence and affective (depression and anxiety) correlates in patients with de novo PD. Nevertheless, this study is subject to some limitations. First, the NMSQuest Domain‐5 assesses memory and attention domains, not all five cognitive domains thought to be impaired in PD‐MCI. However, there is currently a lack of consensus on assessment instruments for PD‐SCD. Therefore, there is a need for a multi‐cognitive domain, standardized, and sufficiently reliable tool to assess SCD in PD patients. Second, due to the cross‐sectional study nature of this study, an association between PD‐SCD presence and affective symptoms can only be demonstrated. Longitudinal follow‐up studies are thus essential to establish causation between PD‐SCD and affective symptoms. This work is part of a longitudinal study of de novo PD patients and we will expedite follow‐up of these patients. Third, although we examined depression and anxiety symptoms associated with SCD, we did not examine other psychiatric symptoms (e.g., apathy) or possible confounders (e.g., family history of dementia) as potential factors for SCD. As studies have shown that apathy is associated with PD‐SCD, 8 future studies should investigate potential associations between these factors and PD‐SCD.
5. CONCLUSION
The findings of this study suggest that SCD is highly prevalent in de novo PD patients and that the presence of PD‐SCD is suggestive of underlying depression and anxiety disorders. Careful evaluation of affective symptoms is necessary for optimal management and treatment of PD‐SCD patients. Longitudinal studies will be essential to confirm and expand the relationship between PD‐SCD and mood symptoms.
AUTHOR CONTRIBUTIONS
JT and WL organized the project. NY drafted the preliminary manuscript and performed statistical analysis. YJ, JR, and HW collected the data. PL and HN critically revised the manuscript. All listed authors contributed to this article and approved the submitted version for publication.
CONFLICT OF INTEREST
The authors declare no conflict of interest.
PEER REVIEW
The peer review history for this article is available at https://publons.com/publon/10.1111/ane.13662.
ACKNOWLEDGEMENT
The authors greatly appreciate the active participation of the patients and the cooperation of their families during this study.
Yang, N. , Ju, Y. , Ren, J. , Wang, H. , Li, P. , Ning, H. , Tao, J. & Liu, W. (2022). Prevalence and affective correlates of subjective cognitive decline in patients with de novo Parkinson's disease. Acta Neurologica Scandinavica, 146, 276–282. doi: 10.1111/ane.13662
Funding information
National Key Research and Development Program of China, Grant/Award Number: 2017YFC1310302 and 2016YFC1306600; National Natural Science Foundation of China, Grant/Award Number: 81571348; Science and Technology Program of Jiangsu Province, Grant/Award Number: BE2019611; Science and Technology Development Project of Traditional Chinese Medicine in Jiangsu Province, Grant/Award Number: 2020ZX17; Medical Science and Technology Development Foundation of Nanjing, Grant/Award Number: YKK20094
Contributor Information
Jiaping Tao, Email: 351742801@qq.com.
Weiguo Liu, Email: wgliunbh@sina.com.
DATA AVAILABILITY STATEMENT
Raw data for this study are available from the corresponding authors upon reasonable request.
REFERENCES
- 1. Bloem BR, Okun MS, Klein C. Parkinson's disease. Lancet. 2021;397(10291):2284‐2303. doi: 10.1016/s0140-6736(21)00218-x [DOI] [PubMed] [Google Scholar]
- 2. Aarsland D, Andersen K, Larsen JP, Lolk A, Nielsen H, Kragh‐Sørensen P. Risk of dementia in Parkinson's disease: a community‐based, prospective study. Neurology. 2001;56(6):730‐736. doi: 10.1212/wnl.56.6.730 [DOI] [PubMed] [Google Scholar]
- 3. Jessen F, Amariglio RE, van Boxtel M, et al. A conceptual framework for research on subjective cognitive decline in preclinical Alzheimer's disease. Alzheimers Dement. 2014;10(6):844‐852. doi: 10.1016/j.jalz.2014.01.001 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4. Aarsland D, Batzu L, Halliday GM, et al. Parkinson disease‐associated cognitive impairment. Nat Rev Dis Primers. 2021;7(1):47. doi: 10.1038/s41572-021-00280-3 [DOI] [PubMed] [Google Scholar]
- 5. Oedekoven C, Egeri L, Jessen F, Wagner M, Dodel R. Subjective cognitive decline in idiopathic Parkinson's disease: a systematic review. Ageing Res Rev. 2022;74:101508. doi: 10.1016/j.arr.2021.101508 [DOI] [PubMed] [Google Scholar]
- 6. Chandler JM, Nair R, Biglan K, et al. Characteristics of Parkinson's disease in patients with and without cognitive impairment. J Parkinsons Dis. 2021;11(3):1381‐1392. doi: 10.3233/jpd-202190 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7. Vossius C, Larsen JP, Janvin C, Aarsland D. The economic impact of cognitive impairment in Parkinson's disease. Mov Disord. 2011;26(8):1541‐1544. doi: 10.1002/mds.23661 [DOI] [PubMed] [Google Scholar]
- 8. Chua CY, Koh MRE, Chia NS, et al. Subjective cognitive Complaints in early Parkinson's disease patients with normal cognition are associated with affective symptoms. Parkinsonism Relat Disord. 2021;82:24‐28. doi: 10.1016/j.parkreldis.2020.11.013 [DOI] [PubMed] [Google Scholar]
- 9. Pan C, Ren J, Hua P, et al. Subjective cognitive complaints in newly‐diagnosed parkinson's disease with and without mild cognitive impairment. Front Neurosci. 2021;15:761817. doi: 10.3389/fnins.2021.761817 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10. Dujardin K, Duhamel A, Delliaux M, Thomas‐Antérion C, Destée A, Defebvre L. Cognitive complaints in Parkinson's disease: its relationship with objective cognitive decline. J Neurol. 2010;257(1):79‐84. doi: 10.1007/s00415-009-5268-2 [DOI] [PubMed] [Google Scholar]
- 11. Han LL, Wang L, Xu ZH, et al. Disease progression in Parkinson's disease patients with subjective cognitive complaint. Ann Clin Transl Neurol. 2021;8(10):2096‐2104. doi: 10.1002/acn3.51461 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12. Hogue O, Fernandez HH, Floden DP. Predicting early cognitive decline in newly‐diagnosed Parkinson's patients: A practical model. Parkinsonism Relat Disord. 2018;56:70‐75. doi: 10.1016/j.parkreldis.2018.06.031 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13. Baschi R, Nicoletti A, Restivo V, Recca D, Zappia M, Monastero R. Frequency and correlates of subjective memory complaints in Parkinson's disease with and without mild cognitive impairment: data from the parkinson's disease cognitive impairment study. J Alzheimers Dis. 2018;63(3):1015‐1024. doi: 10.3233/jad-171172 [DOI] [PubMed] [Google Scholar]
- 14. Barbosa RP, Mendonça MD, Caetano AP, Lampreia TM, Miguel R, Bugalho PM. Cognitive complaints in Parkinson's disease patients: from subjective cognitive complaints to dementia and affective disorders. J Neural Transm (Vienna). 2019;126(10):1329‐1335. doi: 10.1007/s00702-019-02042-8 [DOI] [PubMed] [Google Scholar]
- 15. Lehrner J, Moser D, Klug S, et al. Subjective memory complaints, depressive symptoms and cognition in Parkinson's disease patients. Eur J Neurol. 2014;21(10):1276‐1284, e77. doi: 10.1111/ene.12470 [DOI] [PubMed] [Google Scholar]
- 16. Dupouy J, Ory‐Magne F, Mekies C, et al. Cognitive complaint in early Parkinson's disease: A pilot study. Acta Neurol Scand. 2018;137(1):59‐66. doi: 10.1111/ane.12808 [DOI] [PubMed] [Google Scholar]
- 17. Hong JY, Lee JE, Sohn YH, Lee PH. Neurocognitive and atrophic patterns in Parkinson's disease based on subjective memory complaints. J Neurol. 2012;259(8):1706‐1712. doi: 10.1007/s00415-011-6404-3 [DOI] [PubMed] [Google Scholar]
- 18. Postuma RB, Berg D, Stern M, et al. MDS clinical diagnostic criteria for Parkinson's disease. Mov Disord. 2015;30(12):1591‐1601. doi: 10.1002/mds.26424 [DOI] [PubMed] [Google Scholar]
- 19. Jankovic J, McDermott M, Carter J, et al. Variable expression of Parkinson's disease: a base‐line analysis of the DATATOP cohort. The Parkinson Study Group. Neurology Oct. 1990;40(10):1529‐1534. doi: 10.1212/wnl.40.10.1529 [DOI] [PubMed] [Google Scholar]
- 20. Chaudhuri KR, Martinez‐Martin P, Schapira AH, et al. International multicenter pilot study of the first comprehensive self‐completed nonmotor symptoms questionnaire for Parkinson's disease: the NMSQuest study. Mov Disord. 2006;21(7):916‐923. doi: 10.1002/mds.20844 [DOI] [PubMed] [Google Scholar]
- 21. Chaudhuri KR, Martinez‐Martin P, Brown RG, et al. The metric properties of a novel non‐motor symptoms scale for Parkinson's disease: Results from an international pilot study. Mov Disord. 2007;22(13):1901‐1911. doi: 10.1002/mds.21596 [DOI] [PubMed] [Google Scholar]
- 22. Nasreddine ZS, Phillips NA, Bédirian V, et al. The montreal cognitive assessment, MoCA: a brief screening tool for mild cognitive impairment. J Am Geriatr Soc. 2005;53(4):695‐699. doi: 10.1111/j.1532-5415.2005.53221.x [DOI] [PubMed] [Google Scholar]
- 23. Litvan I, Goldman JG, Tröster AI, et al. Diagnostic criteria for mild cognitive impairment in Parkinson's disease: movement disorder society task force guidelines. Mov Disord. 2012;27(3):349‐356. doi: 10.1002/mds.24893 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24. Erro R, Santangelo G, Barone P, et al. Do subjective memory complaints herald the onset of mild cognitive impairment in Parkinson disease? J Geriatr Psychiatry Neurol. 2014;27(4):276‐281. doi: 10.1177/0891988714532015 [DOI] [PubMed] [Google Scholar]
- 25. Reisberg B, Gauthier S. Current evidence for Subjective Cognitive Impairment (SCI) as the pre‐Mild Cognitive Impairment (MCI) stage of subsequently manifest Alzheimer's disease. Int Psychogeriatr. 2008;20(1):1‐16. doi: 10.1017/s1041610207006412 [DOI] [PubMed] [Google Scholar]
- 26. Reid LM, Maclullich AM. Subjective memory complaints and cognitive impairment in older people. Dement Geriatr Cogn Disord. 2006;22(5–6):471‐485. doi: 10.1159/000096295 [DOI] [PubMed] [Google Scholar]
- 27. Mitchell AJ, Beaumont H, Ferguson D, Yadegarfar M, Stubbs B. Risk of dementia and mild cognitive impairment in older people with subjective memory complaints: meta‐analysis. Acta Psychiatr Scand. 2014;130(6):439‐451. doi: 10.1111/acps.12336 [DOI] [PubMed] [Google Scholar]
- 28. Burmester B, Leathem J, Merrick P. Subjective cognitive complaints and objective cognitive function in aging: a systematic review and meta‐analysis of recent cross‐sectional findings. Neuropsychol Rev. 2016;26(4):376‐393. doi: 10.1007/s11065-016-9332-2 [DOI] [PubMed] [Google Scholar]
- 29. Purri R, Brennan L, Rick J, et al. Subjective cognitive complaint in parkinson's disease patients with normal cognition: canary in the coal mine? Mov Disord. 2020;35(9):1618‐1625. doi: 10.1002/mds.28115 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30. Galtier I, Nieto A, Lorenzo JN, Barroso J. Subjective cognitive decline and progression to dementia in Parkinson's disease: a long‐term follow‐up study. J Neurol. 2019;266(3):745‐754. doi: 10.1007/s00415-019-09197-0 [DOI] [PubMed] [Google Scholar]
- 31. Baiano C, Barone P, Trojano L, Santangelo G. Prevalence and clinical aspects of mild cognitive impairment in Parkinson's disease: A meta‐analysis. Mov Disord. 2020;35(1):45‐54. doi: 10.1002/mds.27902 [DOI] [PubMed] [Google Scholar]
- 32. Santangelo G, Vitale C, Trojano L, et al. Subthreshold depression and subjective cognitive complaints in Parkinson's disease. Eur J Neurol. 2014;21(3):541‐544. doi: 10.1111/ene.12219 [DOI] [PubMed] [Google Scholar]
- 33. Koster DP, Higginson CI, MacDougall EE, Wheelock VL, Sigvardt KA. Subjective cognitive complaints in parkinson disease without dementia: a preliminary study. Appl Neuropsychol Adult. 2015;22(4):287‐292. doi: 10.1080/23279095.2014.925902 [DOI] [PubMed] [Google Scholar]
- 34. Copeland JN, Lieberman A, Oravivattanakul S, Tröster AI. Accuracy of patient and care partner identification of cognitive impairments in parkinson's disease‐mild cognitive impairment. Mov Disord. 2016;31(5):693‐698. doi: 10.1002/mds.26619 [DOI] [PubMed] [Google Scholar]
- 35. Jessen F, Amariglio RE, Buckley RF, et al. The characterisation of subjective cognitive decline. Lancet Neurol. 2020;19(3):271‐278. doi: 10.1016/s1474-4422(19)30368-0 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36. Aarsland D, Bronnick K, Williams‐Gray C, et al. Mild cognitive impairment in Parkinson disease: a multicenter pooled analysis. Neurology. 2010;75(12):1062‐1069. doi: 10.1212/WNL.0b013e3181f39d0e [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37. Brennan L, Devlin KM, Xie SX, et al. Neuropsychological subgroups in non‐demented parkinson's disease: a latent class analysis. J Parkinsons Dis. 2017;7(2):385‐395. doi: 10.3233/jpd-171081 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38. Weintraub D, Chahine LM, Hawkins KA, et al. Cognition and the course of prodromal Parkinson's disease. Mov Disord. 2017;32(11):1640‐1645. doi: 10.1002/mds.27189 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39. Monastero R, Cicero CE, Baschi R, et al. Mild cognitive impairment in Parkinson's disease: the Parkinson's Disease Cognitive Study (PACOS). J Neurol. 2018;265(5):1050‐1058. doi: 10.1007/s00415-018-8800-4 [DOI] [PubMed] [Google Scholar]
- 40. Hong JY, Lee Y, Sunwoo MK, Sohn YH, Lee PH. Subjective cognitive complaints and objective cognitive impairment in parkinson's disease. J Clin Neurol. 2018;14(1):16‐21. doi: 10.3988/jcn.2018.14.1.16 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41. Hu MT, Szewczyk‐Królikowski K, Tomlinson P, et al. Predictors of cognitive impairment in an early stage Parkinson's disease cohort. Mov Disord. 2014;29(3):351‐359. doi: 10.1002/mds.25748 [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
Raw data for this study are available from the corresponding authors upon reasonable request.


